Abstract
Vaccines have had a profound impact on the management and prevention of infectious disease. In addition, development of vaccines against chronic diseases has attracted considerable interest as an approach to prevent, rather that treat, conditions such as cancer, Alzheimer’s disease, and others. Subunit vaccines consist of non-genetic components of the infectious agent or disease-related epitope. In this review, we discuss peptide-based vaccines and their potential in three therapeutic areas: infectious disease, Alzheimer’s disease, and cancer. We discuss factors that contribute to vaccine efficacy and how these parameters may potentially be modulated by design. We examine both clinically tested vaccines as well as nascent approaches and explore current challenges and potential remedies. While peptide vaccines hold substantial promise in the prevention of human disease, many obstacles remain that have hampered their clinical use; thus, continued research efforts to address these challenges is warranted.
Graphical Abstract
1. INTRODUCTION
Vaccines are arguably the most successful biomedical advance in preventing disease. Each year, over 100 million children globally receive vaccinations to prevent diseases that were once widespread and linked to serious medical conditions or even death. Globally distributed childhood vaccines include those for measles, mumps, rubella, seasonal influenza virus, tetanus, polio, hepatitis B, cervical cancer, diptheria, pertussis, and others. Additionally, vaccines for diseases that are endemic to certain regions, such as Yellow fever virus whose mosquito vectors circulate in tropical and subtropical regions year-round, are administered to the general population. Altogether, it is estimated that vaccination prevents between 2–3 million deaths annually (WHO).1
Despite these successes, there are many diseases for which development of a safe and effective vaccine remains elusive. At present, all widely utilized vaccines prevent infectious disease. Microbial pathogens that have exceptionally broad sequence diversity among their constituent family members (e.g., HIV-1), or pathogens such as influenza virus that undergo significant annual antigenic drift have been especially difficult to approach from a vaccine perspective.2–4 Malaria has also been a challenging vaccine target due to the many stages of the parasite life cycle.5 Dengue virus is the most globally distributed arbovirus with ~390 million infections worldwide each year, but development of a Dengue vaccine has been challenging due to a complex immunopathology in which induction of sub-neutralizing antibody levels contributes to an enhanced form of the disease.6
Infectious disease vaccines aim to induce a protective immune response in a naïve host by exposing the immune system to epitopes contained on the pathogen prior to exposure to the infectious agent itself. The major challenges that confront infectious disease vaccines stem from the nature of the epitopes against which the immune response is directed; in some cases, immunodominant epitopes arising from natural infection may not be those that are most desirable (e.g., susceptible to neutralization and/or highly conserved). In contrast, vaccines targeting diseases that involve “self” antigens (e.g., cancer or neurodegenerative disease) provide an additional complication in that the immune system suppresses responses to “self” antigens. In fact, immunological dysregulation of self-responses is suspected to be causative for many autoimmune disorders such as rheumatoid arthritis, lupus, and Graves’ disease. Nonetheless, the potential to develop vaccines against chronic diseases remains appealing. In the cases of both cancer and Alzheimer’s disease (on which we focus here), therapeutic promise via passive immunization provides the underlying rationale that vaccines could be developed to invoke similar protective responses but without the continual need for administration of a therapeutic agent. In immune-oncology in particular, it has become clear that activation of antigen-specific T cell responses will become a critical factor for development of successful immunotherapies against solid tumors.
In this review, we discuss the development of peptide-based vaccine approaches in three specific contexts: infectious disease, Alzheimer’s disease, and cancer. We focus on these areas because each has an instructive mix of clinical successes and remaining challenges. In addition, we focus attention on cases that have either advanced to clinical stage or on approaches that utilize structure-based design as a key aspect. While this discussion is by no means exhaustive of all peptide vaccines that have been or are currently under development, our goal is to provide the reader with chemical and structural insights into vaccine design using peptides. We begin the review with a general discussion of factors to consider in peptide vaccine design.
1.1. Stimulation of immune responses by peptides.
The vast majority of vaccines against infectious diseases, the largest class of vaccines, consist of inactivated or live attenuated pathogens. For example, the small pox vaccine was first derived by Edward Jenner in 1796 from a related but non-pathogenic strain that only infects cattle (cowpox). The seasonal influenza vaccine is composed of mixtures of viral strains grown in eggs and then heat inactivated. In general, inactive or attenuated pathogens can stimulate a robust immune response because they contain both B- and T- cell epitopes presented in a conformation that is relevant to the pathogen. Subunit vaccines that consist primarily of peptides or proteins, in contrast, can face limitations with respect to immunogenicity and thus may require multiple immunizations to achieve similar levels of immune response. Nonetheless, a variety of approaches to enhance subunit vaccine responses, including presentation of epitopes in multimeric format (e.g., virus-like particles, VLPs, or nanoparticles) or use of immunostimulatory adjuvants, have been utilized. Here, we discuss considerations when trying to elicit peptide-specific B- or T-cell responses.
B-cell responses.
The elicitation of epitope-specific antibodies is a primary mechanism of protection for many vaccines. For infectious diseases, often the targeted epitope, which is bound by the antigen-binding fragment (Fab) region of the IgG, is a site of susceptibility for “neutralization” by antibodies. Neutralizing antibodies can inhibit infection by blocking host cell attachment or entry by pathogens, or by inducing pathogen-antibody immune complexes that are cleared systemically (e.g., agglutination/opsonization). In addition, both neutralizing and non-neutralizing pathogen-specific antibodies may induce a number of immune mechanisms via the antibody Fc region that result ultimately in the destruction and/or clearance of the pathogen or pathogen-infected cells (Fig. 1A). For the most part, protective antibodies target epitopes that lie on the surface of the pathogen (e.g., the viral glycoprotein or bacterial capsid).
Figure 1.
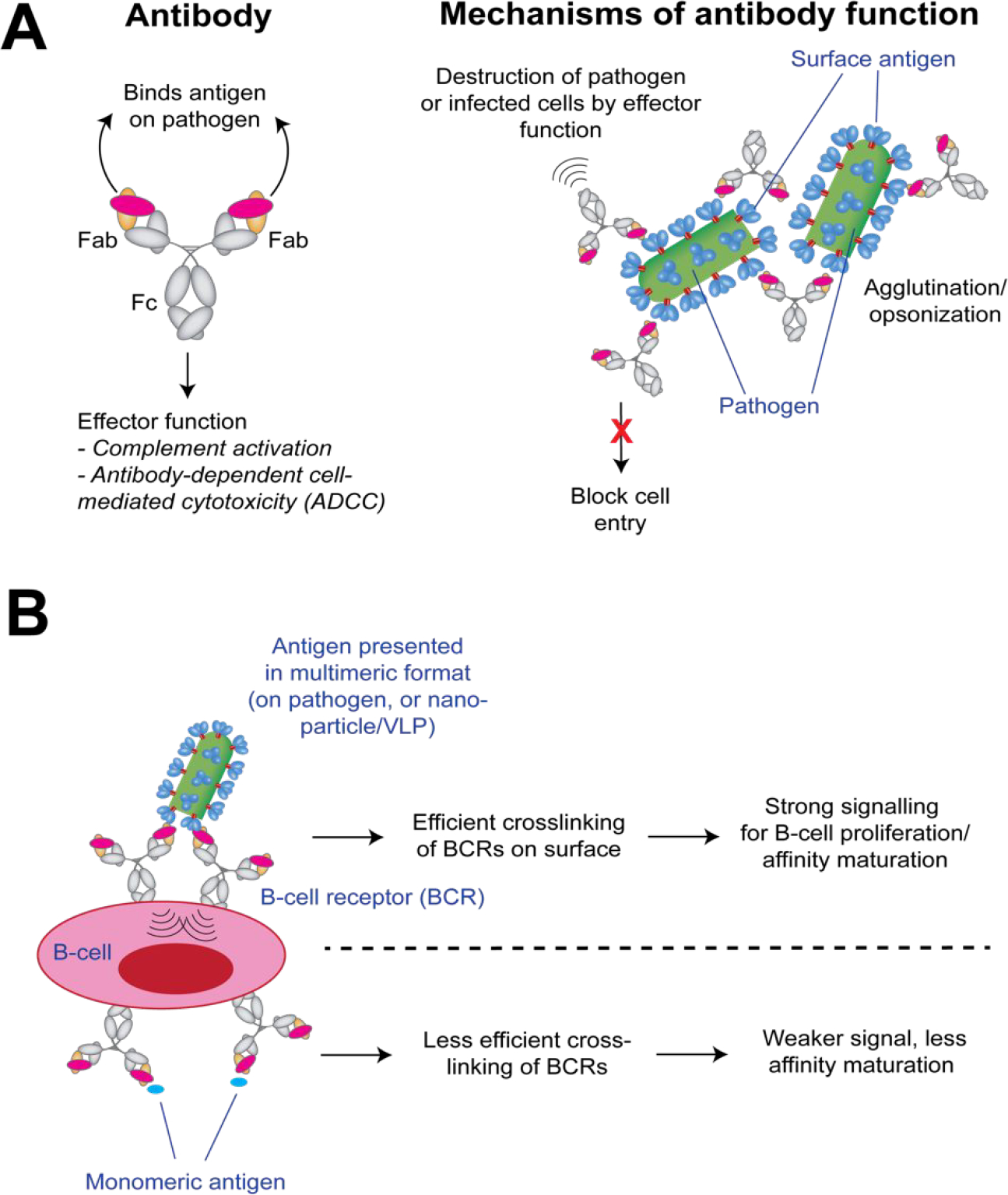
Antibody function and affinity maturation. (A) Mechanisms by which antibodies can protect against microbial pathogens. For the overall antibody architecture, the Fab region binds the antigen or pathogen, and the Fc region is responsible for effector function. (B) Affinity maturation requires cross-linking of B-cell receptors on the surface to signal survival and expansion of that clone. This cross-linking is more efficiently stimulated when antigens are presented in a multimeric format (e.g., on the pathogen, or on a nanoparticle or VLP).
Generally, the elicitation of protective antibodies requires affinity maturation from the germline, a process that is stimulated by crosslinking B-cell receptors (BCRs) on a specific B-cell (Fig. 1B). To this end, monomeric peptides are often poorly immunogenic relative to those corresponding sequences on viral, bacterial, or parasitic external proteins because, when presented in those contexts, multiple copies of the epitope on the pathogen surface permit efficiently cross-link BCRs and thus stimulate antibody affinity maturation. One strategy to improve immunogenicity is to link the desired peptide epitope to a VLP or nanoparticle to allow ordered, multivalent epitope presentation that can more efficiently crosslink BCRs.
Another mechanism by which antibodies can afford protection is by binding secreted or shedded factors that are linked to a microorganism’s pathogenesis. For example, antibodies against bacterial toxins such as tetanus toxoid, anthrax toxin, or Staphylococcus aureus enterotoxin B are protective in animal models7–9. These toxins are produced by the pathogen and contribute to expanded infection by inducing effects such as hemorrhaging or inflammation, thus providing the pathogen an opportunity to infect further damaged tissue. Thus, fragments or inactive variants of these toxins can themselves be candidates for vaccines.
Antibodies can target their epitopes in conformation-dependent or -independent manners10. The “structural epitope” (i.e., those residues on the antigen whose side chains make direct contacts with the antibody combining site) can include positions that are close or distant in primary protein sequence. Larger structural epitopes are generally conformation-dependent and include residues from multiple secondary structural elements, and thus antibody recognition is dependent on globular fold, at least in the region of the epitope. These larger epitopes have been mimicked by structural protein/peptide engineering,11–12 or by “mimetope” selection whereby a naïve library of peptides are selected for their ability to bind the antibody by phage display or other display methods.13 Epitopes that are conformation-independent are generally linear stretches of residues; while the stretch of amino acids need not be in a specific conformation to be recognized by the antibody, they typically are induced to adopt some local secondary structure upon antibody binding. Linear epitopes are generally found in protein loops and are prime candidates for peptide vaccine design. As with most peptide-targeting approaches, however, there is an advantage to rigidifying peptide epitope conformations so that they most closely match the epitope structure when bound to the antibody.
T-cell responses.
Stimulation of epitope-specific T-cells is another mechanism by which vaccines can induce protective immunity. In the context of infectious diseases, recruitment of T-cells can result in the rapid destruction and clearing of the pathogen itself or of infected host cells thereby stemming the spread of the infection. In the context of immuno-oncology, a major mechanism by which tumors evade immune surveillance is by local downregulation of cancer-specific T-cells. Immunotherapies that globally upregulate T-cells, such as anti-PD1 monoclonal antibodies (mAbs), have shown great promise against leukemias (“blood cancers”),14 but a current challenge is how to stimulate T-cells that are embedded within solid tumors, which systemically administered mAbs cannot access.
Epitope specificity for T-cells is mediated by the T-cell receptor (TCR), which binds peptides presented in the “peptide binding groove” of class I or class II major histocompatibility complexes (MHCs, also known as human leukocyte antigen, HLA, for humans) on antigen presenting cells (APCs) (Fig. 2A). Whole antigens are internalized and proteolyzed by APCs and then short peptides (8–11 residues in length for class I, and 11–30 residues in length for class II) are loaded into MHCs (or HLAs) and presented on the APC surface. TCRs that are specific for the peptide epitope then bind those peptide-MHC complexes (pMHCs or pHLA) and a variety of proteins at the T-cell/APC interface orchestrate expansion of that T-cell clone. The T-cell synapse proteins can be co-stimulatory or inhibitory; PD1 (an inhibitory synapse protein) is overexpressed by many cancer cells to reduce T-cell responses and thus allow the cancer cell to evade destruction by T-cells.
Figure 2.
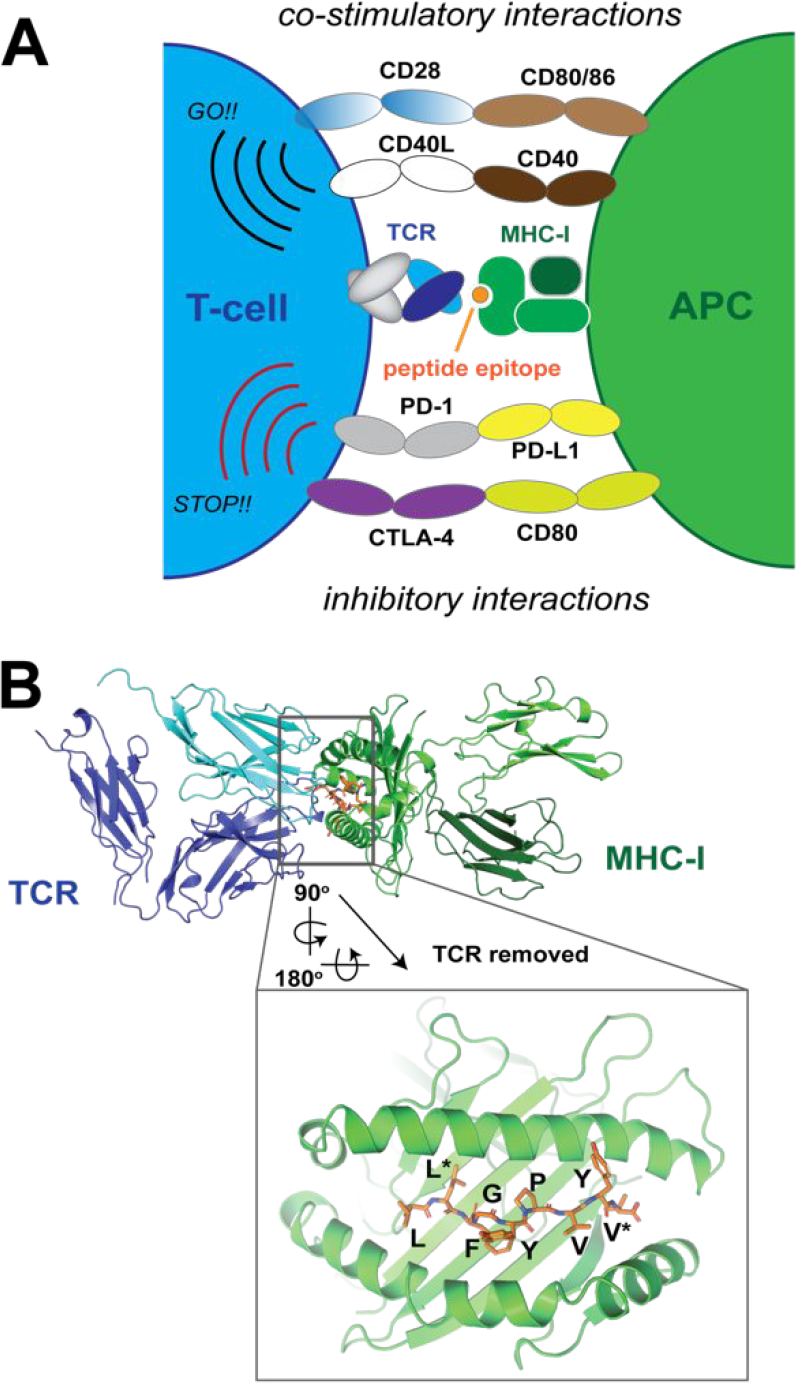
Stimulation of T-cells. (A) Interactions between pMHC and TCR at the T-cell/APC interface. Some co-stimulatory and inhibitory interactions are also shown. (B) Binding of a peptide epitope into a class I MHC. A model HLA-A2-restricted epitope for HTLV-1 is shown (PDB 1A07). Anchor positions of the peptide are indicated in the inset with asterisks.
Peptides presented in class I MHCs are typically short; class I MHC peptides follow a sequence pattern of X-(L/I)- X(6–7)-(V/L) where L/I and V/L represents residues whose side chains anchor the peptide to the pMHC and thus are oriented toward the interior of the peptide binding groove and away from the TCR (Fig. 2B).15 The other positions point toward the TCR and interactions with these residues mediate the epitope specificity. The sequences of class II MHC peptides are more varied but also contain anchor positions. The epitope peptide backbone binds snugly in the peptide binding groove with an extended backbone conformation, although bulging is accommodated for longer peptides in both class I and II MHCs. Furthermore, recognition of peptides requires a free N-terminal amine group.
Peptides that are loaded into MHCs or HLAs must conform to the above sequence requirements, but this does not guarantee that a particular epitope will be immunogenic. Nonetheless, the presentation of known immunogenic sequences can be accomplished by simply loading peptide repeats onto APCs such as dendritic cells.16 For immuno-oncology, this can be one method to expand tumor-infiltrating lymphocytes (TILs) that can then be reinfused into patients for adoptive transfer cell therapy.16 In other circumstances, systemic delivery of the peptides themselves or DNA encoding the epitopes is sufficient to stimulate T-cell expansion in vivo.17
Interactions between proteins at the T-cell interface are generally clustered, and thus individual protein-protein interactions, including those between pMHC and the TCR, or PD-1 and its primary ligand, PD-L1, are low affinity (KD ~ μM range) when measured using soluble forms of each component. Interactions between the peptide-binding platform of the MHC and TCR are central to the T-cell/APC interface, and thus TCRs cannot recognize their peptide epitopes without epitope presentation in this format. Furthermore, the antigen specificity of the T-cell is dependent on the TCR-pMHC interaction, and thus the structural features of the epitope-MHC-TCR ternary complex can be an important consideration for T-cell targeted vaccines. Recognition of particular TCRs on cells using soluble peptide-loaded MHC (pMHC) protein requires presentation of the pMHC in a multivalent fashion. This is most commonly achieved by biotinylation of the pMHC and subsequent complexing with streptavidin, which provides 3–4 pMHCs per streptavidin molecule. Folding of MHCs is dependent on the peptide, thus exogenous expression of pMHCs typically involves fusion of the peptide epitope to the MHC using a polypeptide linker. A number of in vitro and chemical methods have also been devised to allow exchange of the bound peptide with exogenously added peptides.18–20
1.2. Considerations for peptide vaccines.
Immunodominance.
For both B- and T-cell epitopes, not all regions of a protein antigen are equally immunogenic. While antibodies that arise in response to infection typically target a number of epitopes on the pathogen, higher numbers of antibodies mature toward some epitopes versus others. For T-cell responses as well, some regions of an antigen may result in more efficient expansion of T-cells than others. Issues of immunodominance are an important consideration for any vaccine design strategy but particularly for peptide vaccines that focus on only a single or a few critical epitopes. A common strategy is to utilize naturally-occurring antibodies or TCRs as a template for vaccine design, following the logic that if a particular epitope has already elicited a B- or T-cell response during natural disease, then it is sufficiently immunogenic to allow induction of similar responses by administration of a vaccine. In other cases, epitopes that elicit an immune response most favorable for mitigating the disease may not be the most immunodominant, and thus vaccination with critical epitopes may skew the immune response to yield protective responses. A good example of this is in HIV-1, where the vast majority of antibodies that arise during natural infection target non-conserved or non-neutralizing epitopes.4 A number of HIV-1 vaccine programs seek to focus the immune response on the most conserved epitopes, and those that represent sites of susceptibility for virus neutralization. Similar challenges confront development of broad vaccines for other viral pathogens such as influenza and dengue virus.2, 12 In such contexts, peptide-based vaccines may confer some advantage over vaccines consisting of larger protein sequences or whole inactivated virus as they are smaller and may elicit a more focused immune response toward critical neutralizing epitopes.21
In typical peptide vaccination protocols, the epitope of interest is conjugated to a carrier protein or presented in a multimeric format (VLP or nanoparticle). Such strategies can boost immune responses by increasing the half-life of the epitope by decreasing renal clearance and susceptibility to proteolytic degradation. Linkage to carrier proteins is typically achieved by chemical conjugation. The carriers are generally known to have immunogenic properties, and thus the simple covalent linking of epitopes to immunogenic species can often be sufficient to enhance the immune response. Related to this, the immunogenicity of peptide or protein sequences can be augmented through linkage to short sequences that are known to stimulate an immune response. An example of this is PADRE, a universal helper T-cell epitope that can be fused to peptide or protein sequences to stimulate antibody responses.
Epitope structure.
As discussed above, T-cell epitope backbone conformations are limited by the steric restriction of binding into the MHC peptide binding groove, but antibody epitopes can be much more heterogenous in conformation. Antibodies that are specific for linear peptide sequences typically contain a groove at the combining site, whereas those that bind protein surfaces that span multiple secondary structural elements are generally flatter. Peptide epitopes can bind antibodies in α-helical, β-strand/extended, or loop conformations. The precise conformation that the peptide epitope adopts in the antigen-antibody complex can sometimes be important for the activity of the antibody. In these cases where structure is thought to be an important aspect, the presentation of peptide vaccines in a conformationally-relevant manner then becomes a key factor for vaccine design. Conformational dependence of the epitope may be important because it allows recognition of the epitope by the antibody within the larger context of the globular antigen fold. Alternatively, the function of the epitope may be important for disease, and function is structure-dependent. Thus, binding and blocking the functionally relevant conformation is critical to the biological activity of the induced antibodies.
In cases where epitope conformation is important, a variety of approaches have been implemented to constrain peptide epitopes. These include covalent side chain-side chain crosslinking by inclusion of disulfide bonds or other covalent constraints, or integration of the epitope into a larger scaffold that contains elements that induce the relevant peptide conformation. An elegant example of the latter, is the case where scaffolds to present a critical epitope for protective antibody (motavizumab) targeting the F protein of respiratory syncytial virus (RSV) were developed by computational methods (Fig. 3).22 A designed immunogen (FFL_01) was used to vaccinate non-human primates, which induced antibodies (e.g., 17-HD9) that bound the RSV epitope in a manner that mimicked motavizumab (the template for design) but with a different angle of approach to the antigen.
Figure 3.
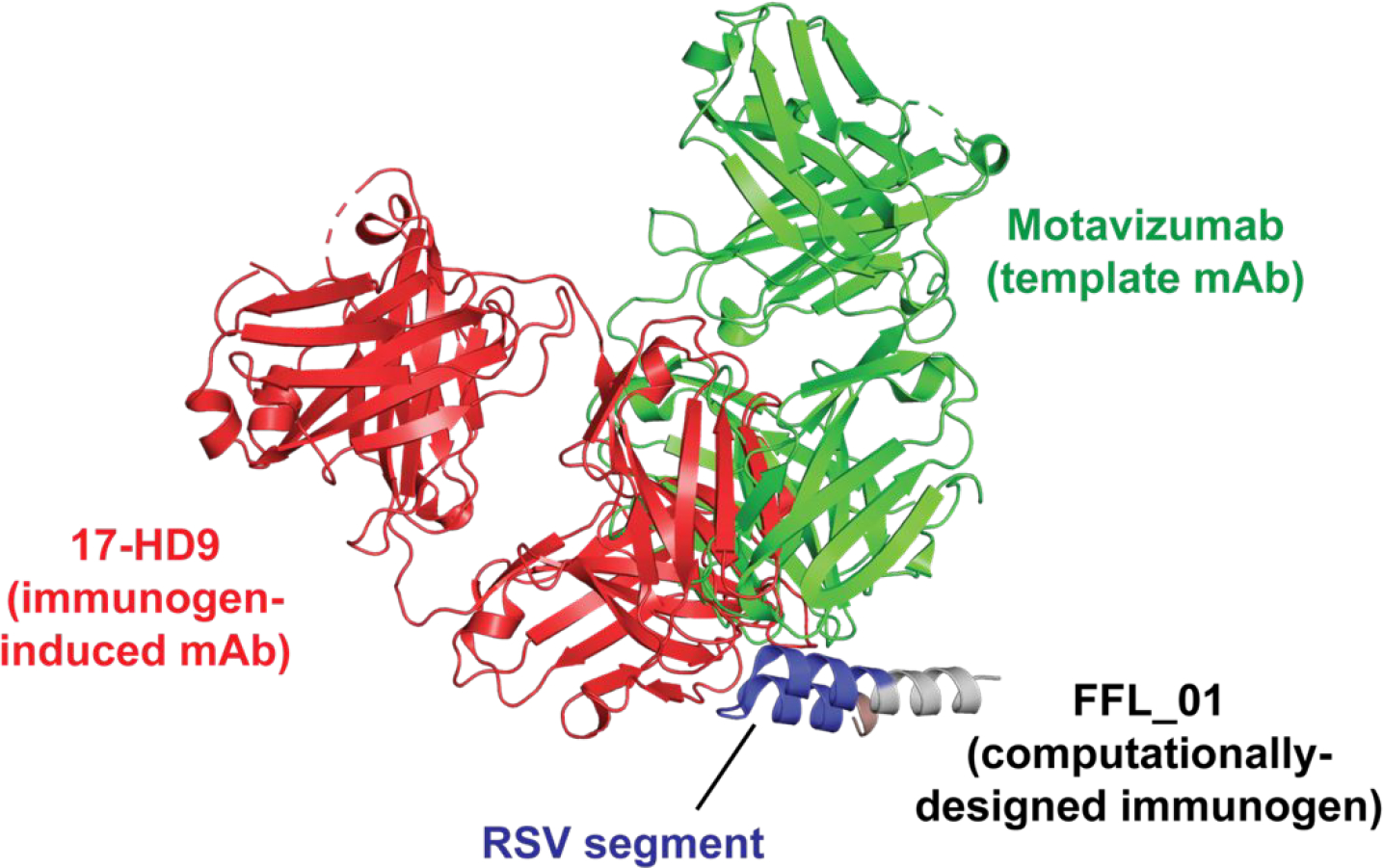
Computational design of an immunogen (FFL_001) for RSV. The scaffolded epitope from RSV F protein, shown in blue, was templated on the RSV antibody motavizumab. FFL_001 elicited antibodies in non-human primates (e.g., 17-HD9) that bound the epitope in a conformationally identical manner but not with the same angle of approach.
Adjuvants and formulations.
Most vaccines are injected with an adjuvant to stimulate an immune response. The nature of adjuvants can vary extensively and are an important consideration for peptide vaccination studies. For example, conformationally-designed epitopes may require adjuvants that do not denature or emulsify the antigens. An additional consideration is that some adjuvants that are utilized in rodents are not approved for use in larger animals (e.g., non-human primates) or humans. It is difficult to predict a priori which adjuvants may yield the best immune response, and often an adjuvant screen can be informative.
2. VACCINES FOR INFECTIOUS DISEASES
As discussed above, vaccines targeting microbial pathogens are the largest class of currently employed vaccines. Consequently, there is significant interest in developing novel peptide-based infectious disease vaccines for many pathogens. Here, we focus specifically on just a few examples (malaria, Hepatitis C virus, influenza virus, and HIV-1) where candidates are in advanced clinical development, or where structure-based design allows a unique approach to next-generation immunogen development.
2.1. Malaria parasite.
Malaria is an Anopheles mosquito-borne disease which remains a significant public health threat. Five species of Plasmodium parasites caused an estimated 219 million cases and 435,000 related deaths in 2017.23 Most severe disease and death cases are due to P. falciparum, although P. vivax can provoke severe disease and relapses as well.24 Currently, there is no licensed vaccine against P. falciparum and P. vivax parasites mostly due to a complicated multi-stage parasitic life cycle. During the Plasmodium full life cycle, the parasite resides in two hosts (mosquito and human) and undergoes 10 morphological transitions.25 During a blood meal, the mosquito ingests plasmodium gametocytes that will give rise to an ookinete after fertilization. The ookinete is transferred to the mid-gut for maturation and then becomes an oocyst. Mature oocysts called sporozoites will enter the mosquito salivary glands and be transmitted to humans during a blood meal.5 Sporozoites in the human blood stream will enter hepatocytes and undergo maturation into merozoites and multiply heavily.26 After rupture of the hepatocyte cells, the merozoites will then invade the red blood cells (RBCs) and start the asexual blood cycle, which is composed of 4 morphological stages (ring, trophozoite, schizont and merozoite).27 The rupture of RBCs by large amount of merozoites is the cause of malaria fever symptoms. In parallel to the asexual blood cycle, some parasites do not undergo the 4 stages of maturation but instead will produce female and male gametocytes inside the RBC.28 Those particular RBCs will be ingested by a mosquito to complete the full plasmodium life cycle. During this complex life cycle, the parasite morphology will vary significantly, which makes antigen identification for vaccine development challenging.
Vaccine strategies.
Three main strategies exist for malaria vaccine development, which target distinct stages of the parasite life cycle: (1) Prevention of sporozoite invasion of the liver (pre-erythocyte vaccine); (2) inhibition of erythrocyte entry (blood stage vaccine); and (3) blockage of oocyst formation in mosquito (transmission blocking vaccine).29 Most malaria vaccine approaches focus on subunit vaccines that contain one or more antigenic proteins, although some approaches use live-attenuated whole parasites.30 Among subunit vaccines, two specific antigens are of interest: the circumsporozoite protein (CSP) and the apical membrane antigen 1 (AMA-1) found respectively in sporozoites and merozoites.
The CSP, localized at the surface of the sporozoite, is composed of 412 amino acids and is critical for sporozoite establishment and development in the liver.31 A 37 tetrapeptide repeat Asn-Pro-Asn-Ala (NPNA) and a thrombospondin conserved domain are two CSP key elements that have been identified as immunogenic epitopes.32 The most advanced phase III vaccine trial (RTS,S) uses a ~188 amino acids truncated CSP where the two key domains are fused to each other.33 Other efforts to develop a shorter antigenic CSP fragment, which is easier to produce on a large scale, are under way.34 Development of a 20 amino acid peptide mimetic called UK39, which includes 5 NPNA repeats, showed similar structural and antigenic properties as the native CSP NPNA repeat region (Fig. 4A).35 UK39 contains a designed covalent amide linkage to stabilize the loop conformation between glutamate and 4-aminoproline residues, in addition to an N-terminal phosphatidylethanolamine for coupling to the surface of immune-potentiating influenza virosomes (IRIV).36–37 IRIV is an established antigen-delivery platform for multi-subunit vaccine eliciting CD4-T cell and antibody responses when the antigens are display on the virosome surface.38 Immunization of mice and rabbits with UK39 led to production of sporozoite cross-reactive IgGs that inhibited migration and invasion of hepatocytes by sporozoites.39
Figure 4.
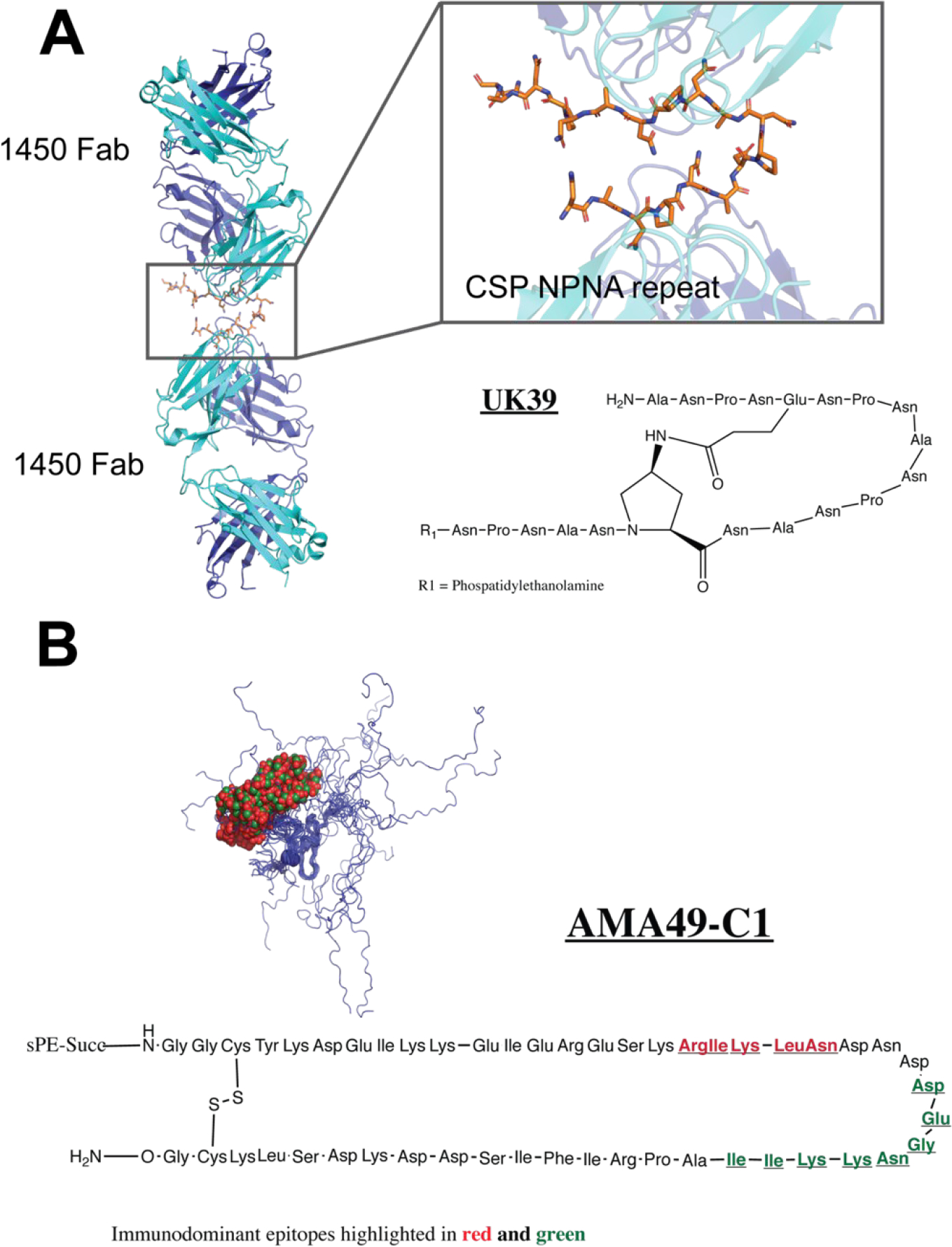
Peptide vaccines for malaria parasite. (A) Structure of peptide mimetic UK39, which was designed based on the X-ray structure of 1450 Fab bound to the NPNA repeat of CSP (PDB 6D11). (B) Structure of cyclic peptide AMA49-C1, a mimetic of AMA1. The two immunodominant regions (459–464, red, and 467–475, green) are highlighted on the chemical structure. An NMR structure of the P. falciparum AMA1 residues 436–545 shows that these segments, shown as red and green Cα spheres, respectively fall within the disordered region (PDB 1HN6).
The second antigen of interest, the apical membrane antigen 1 (AMA-1), is a type I integral membrane protein localized at the surface of the merozoite.40–41 After release of the merozoites from the liver, AMA-1 is believed to play an important role in the invasion of erythrocytes and during parasite blood stage development.42–43 The AMA-1 ectodomain is comprised of three subdomains named I, II and III, and the overall protein structure is stabilized by eight intramolecular disulfide.40 An epitope mapping study of the AMA-1 semi-conserved loop I of domain III showed that a cyclic synthetic peptide including residues 446–490, denominated AMA49-C1, was capable of eliciting blood stage parasite cross-reacting antibodies in mice and rabbits (Fig. 4B).44 Based on encouraging animal study results with CSP and AMA-1 synthetic peptide antigens, human clinical trials were started in early 2006. Similar to UK39, AMA49-C1 was conjugated to PE and presented on IRIVs. Clinical trial phase Ia and Ib demonstrated safety and immunogenicity of individual or combination of virosome-formulated UK39 and AMA49-C1 peptides, opening the door for multi-component malaria vaccine targeting different stages of parasite development.36–37
2.2. Hepatitis C virus.
Despite recent advances in treatments, hepatitis C virus (HCV) remains a global health concern that is a leading cause of liver disease and liver cancer.45–46 Chronic HCV infection can lead to cirrhosis, liver failure, and hepatocellular carcinoma. High treatment costs as well as a high rate of asymptomatic and untreated patients make a vaccine to prevent HCV of substantial interest. Currently, no approved vaccines exist, but candidates are under investigation in preclinical and clinical studies.
One hurdle in the design of an effective HCV vaccine has been the high diversity of the virus, arising from error-prone replication that allows the virus to escape immune surveillance.47–48 Clearance of HCV infection therefore requires a robust and cross-reactive CD4 and CD8 T-cell response as well as neutralizing antibodies.49–50 Identification and characterization of cytotoxic T lymphocytes (CTL) epitopes as well as broadly neutralizing antibodies that target conserved epitopes of the E1 and E2 glycoproteins on the viral surface has prompted the exploration of peptide-based vaccine strategies.51
IC41 is a vaccine candidate that consists of five synthetic peptides (IPEP 83, 84, 87, 89,1426) from core, NS3, and NS4 proteins harboring HCV CD4 and CD8 T-cell epitopes along with the synthetic adjuvant poly-L-arginine.52 The vaccine targets HLA A2-restricted epitopes that are conserved among the different HCV genotypes: HCV genotypes 1a (100%, 100%, 83%, 100%, 100% for the respective five peptides), 1b (98%, 90%, 15%, 94%, 88%) and 2 (91%, 96%, 13%, 91%, 87%). Immunization in healthy volunteers was generally well tolerated and elicited an HCV peptide-specific Th1/Tc1 response.52 In trials of therapeutic vaccination in chronic HCV patients, IFN-γ secreting T-cells were induced and the peptide vaccine caused no adverse effects. However, T-cell responses were too weak to induce significant changes in HCV RNA in the majority of patients, suggesting that further optimization is required.53 Increased dosing as well as intradermal injection of IC41 demonstrated enhanced response rates.54 Modest reduction in viral load was observed in HCV genotype 1 infected patients after IC41 vaccination, suggesting that investigating combination treatments with antivirals may hold therapeutic promise.55
More recent efforts have employed a number of different strategies in the design of HCV peptide vaccines. One approach utilized the structure of the broadly neutralizing antibody, human mAb HCV1, in complex with a conserved linear epitope (epitope I; residues 412–423) of HCV E2 to design novel immunogens.56,57 These included two cyclic peptides, C1 and C2, that used the β-hairpin structure of θ-defensin as a scaffold to present the HCV epitope (Fig. 5). The X-ray structure of the designed C1 immunogen bound to HCV1 closely resembled the mAb complexed with its native linear epitope. Additionally, a bivalent E2-based antigen was designed, in which epitope I was engineered at another site of E2 (residues 625 to 644). Mice vaccinated with the designed immunogens produced a robust antibody response against epitope I that demonstrated neutralization against HCV. Another strategy involving cyclic peptides to mimic HCV-envelope E2 was recently employed58 that demonstrated that cyclic epitope mimics of epitope II of the HCV E2 protein, and not their linear counterparts, demonstrated specificity for neutralizing mAb HC84.1.59–60
Figure 5.
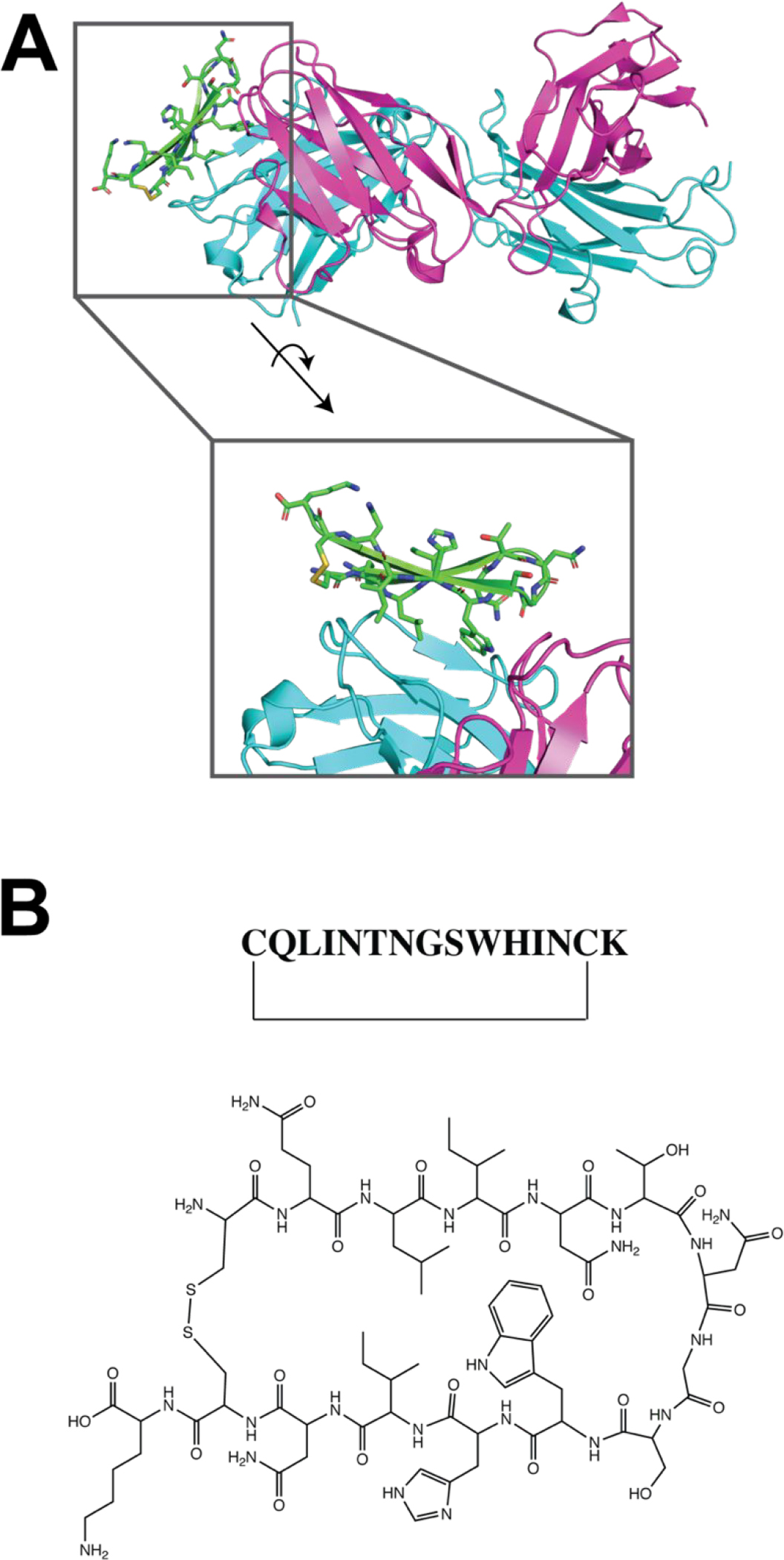
HCV peptide vaccine. (A) X-ray structure of C1 immunogen bound to Fab HCV1 (PDB 5KZP). (B) Chemical structure of HCV peptide C1, modeled after the epitope I structure.
A different approach aimed to broaden the T-cell response to HCV by immunization with a mixture of peptides spanning non-structural protein 3 (NS3) in cationic liposomes.61 The peptide vaccine was composed of a panel of 62 20-residue peptides that spanned the entire NS3 protein. Vaccination studies in mice induced a broader and more robust CD4+ and CD8+ T cell response than recombinant NS3 protein. Furthermore, the T cell response targeted both immunodominant as well as other epitopes, which may be important in combating T-cell exhaustion and chronic HCV infection.
2.3. Influenza virus and HIV-1.
Two of the most challenging viruses for vaccine development have been influenza virus and human immunodeficiency virus (type 1), HIV-1, both of which carry an extraordinary breadth of sequence diversity.2–4, 62 In both viruses, clade- or strain-specific vaccine antigens have been developed and are protective but are of limited clinical use, since they provide protection against only a small fraction of circulating viruses. For HIV-1 in particular, given the chronic nature of the infection and the continual battle between host and virus for immune clearance/evasion, there can be significant viral genetic diversity within a single individual infection. Consequently, peptide vaccine strategies for both viruses have focused on highly conserved regions and epitopes. While neutralization of a particular epitope by an antibody is likely not an absolute requirement for protection for epitope design, most efforts have concentrated on those regions of the viral envelope glycoproteins that are targeted by broadly neutralizing antibodies (bNAbs) that have been derived from patients.
Among the most potent influenza bNAbs are those that target the highly conserved “stem” region of hemagglutinin HA2.11, 63–64 The stem region and HA2 in general are critical for the viral membrane fusion pathway that is required for viral entry into the host cell. Vaccination of rodents or primates with designed protein immunogens that display this region in a conformationally-relevant manner have recently been shown to elicit protective responses.11 Similarly, synthetic peptide vaccines containing these segments are protective, albeit with lower overall titer.65–66
The most advanced influenza peptide vaccine is Multimeric-001, which contains both B- and T-cell linear epitopes from HA but also matrix 1 (M1) and nucleoprotein (NP) combined into a single recombinantly expressed polypeptide.67–69 Multimeric-001 has been shown to induce a protective response in mice and elicited humoral and cellular responses toward a limited subset of influenza strains in healthy volunteers in phase I trials.
For HIV-1, the V3 loop of gp120 was found to be a target of neutralizing antibodies arising from the Rv144 clinical trial, and consequently has attracted a great deal of interest as a target for peptide-based vaccine design.70–77 A complicating aspect of targeting this region is that it contains two glycosylation sites, one of which is required for binding and recognition by model bNAb PGT128.73 Both mono- and di-glycosylated forms of the V3 peptide have been synthesized using chemical or chemical/chemoenzymatic approaches.72, 78 In addition, more recently, the development of multicomponent and multivalent V3 glycopeptides have been examined.79 Based on binding, V3 glycopeptides containing designed structural constraints to induce reverse turn were the most likely to be recognized by PGT128 and other V3 antibodies and induced the most cross-reactive sera in mice or non-human primates (Fig. 6).78 However, as of yet the induction of neutralizing sera has not been reported with any such immunogen.
Figure 6.
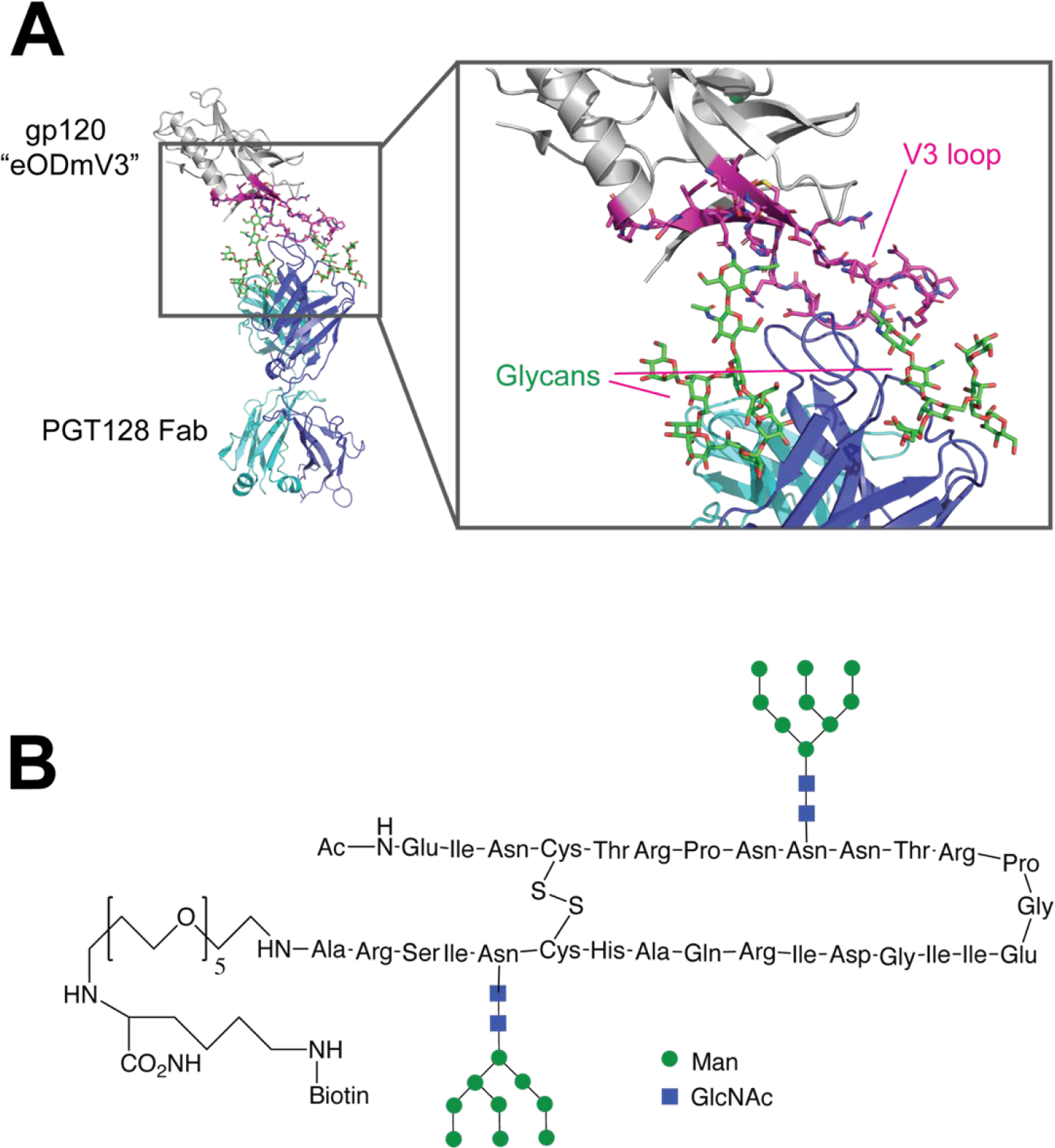
Synthetic HIV-1 gp120 V3 glycopeptide vaccine. (A) X-ray structure of a modified HIV-1 gp120 outer domain containing the V3 region (“eODmV3”) in complex with PGT129 Fab (PDB 3TYG). The V3 region that serves as the basis for glycopeptide vaccine design is colored magenta, and glycans are shown in green. (B) Cyclic V3 glycopeptide immunogen design.
Recently, the fusion peptide region of gp41 has been shown to be a target of human bNAb VRC34.80 The fusion peptide plays a critical role during viral infection, as it anchors to the target cell membrane in a series of glycoprotein conformational changes that ultimately result in fusion of the host and viral cell membranes.81–82 Viral membrane fusion is a critical event for delivery of the viral genome during infection, and thus interference with this pathway inhibits viral entry in vitro and in vivo. Fusion peptides conjugated to keyhole limpet hemocyanin (KLH) induce broadly neutralizing responses, albeit less broad and potent than VRC34.83–84 The difference in breadth and potency may have structural origins in that the conformation of the fusion peptide is different when bound to VRC34 and one of the more potent vaccine-induced antibodies (2712-vFP16.02). The breadth of neutralizing antibodies could be improved in guinea pigs upon priming with FP-KLH conjugate followed by extensive boosting with intact trimer.
3. VACCINES FOR ALZHEIMER’S DISEASE
Alzheimer’s Disease (AD) is a chronic, progressive neurodegenerative disorder afflicting over 5 million adults in the United States and nearly 50 million worldwide.85,86 The financial public health burden of AD is substantial; over 200 billion US dollars are spent on direct care of AD patients annually. AD is the fifth leading cause of death in the US for adults over 65 years of age. Presentation of the disease is characterized by cognitive decline, including short-term memory loss, language impairment and executive dysfunction. Disturbances in mood and behavior as well as functional impairment are features of later stages of the disease, which ultimately leads to death. Currently, no effective treatments exist that reverse disease progression. The most significant efforts in therapeutic development have focused on targeting pathologic species of β-amyloid (Aβ) and Tau proteins.
There is extensive evidence that the abberant aggregation of two proteins, Aβ and Tau, plays an important role in the pathological neurodegeneration that is the hallmark of AD. Consequently, there is much interest in the possibility that immunization with epitopes from these proteins could result in preventative clearance of neurotoxic forms of these proteins or avoid formation of aggregates altogether. An added challenge for this approach is how to penetrate the blood-brain barrier, which is generally inaccessible to antibodies. Nonetheless, a number of peptide vaccine candidates have progressed to clinical studies, suggesting that certain types of immune responses may clear or prevent aggregates from accumulating in the brain.
3.1. Pathological roles of Aβ & Tau
While the cause of AD remains unknown, an imbalance between production and clearance of β-amyloid is thought to be central to disease progression87. Amyloid precursor protein (APP) is an integral membrane protein that is expressed in many tissues, and particularly concentrated in neuronal synapses. APP undergoes extensive post-translational modification, including proteolytic processing by α-, β-, and γ- secretases (Fig. 7A). Digestion of APP by α-secretase occurs within the amyloidogenic region, and thus products arising from this processing (AICD, P3, and C83) are non-amyloidogenic. However, proteolysis by β- then γ-secretase results in the production of amyloidogenic APP fragments Aβ(1–40) and Aβ(1–42). These segments form a diverse array of soluble oligomers as well as fibrillar amyloid plaques, whose improper accumulation is a defining feature of the disease.
Figure 7.
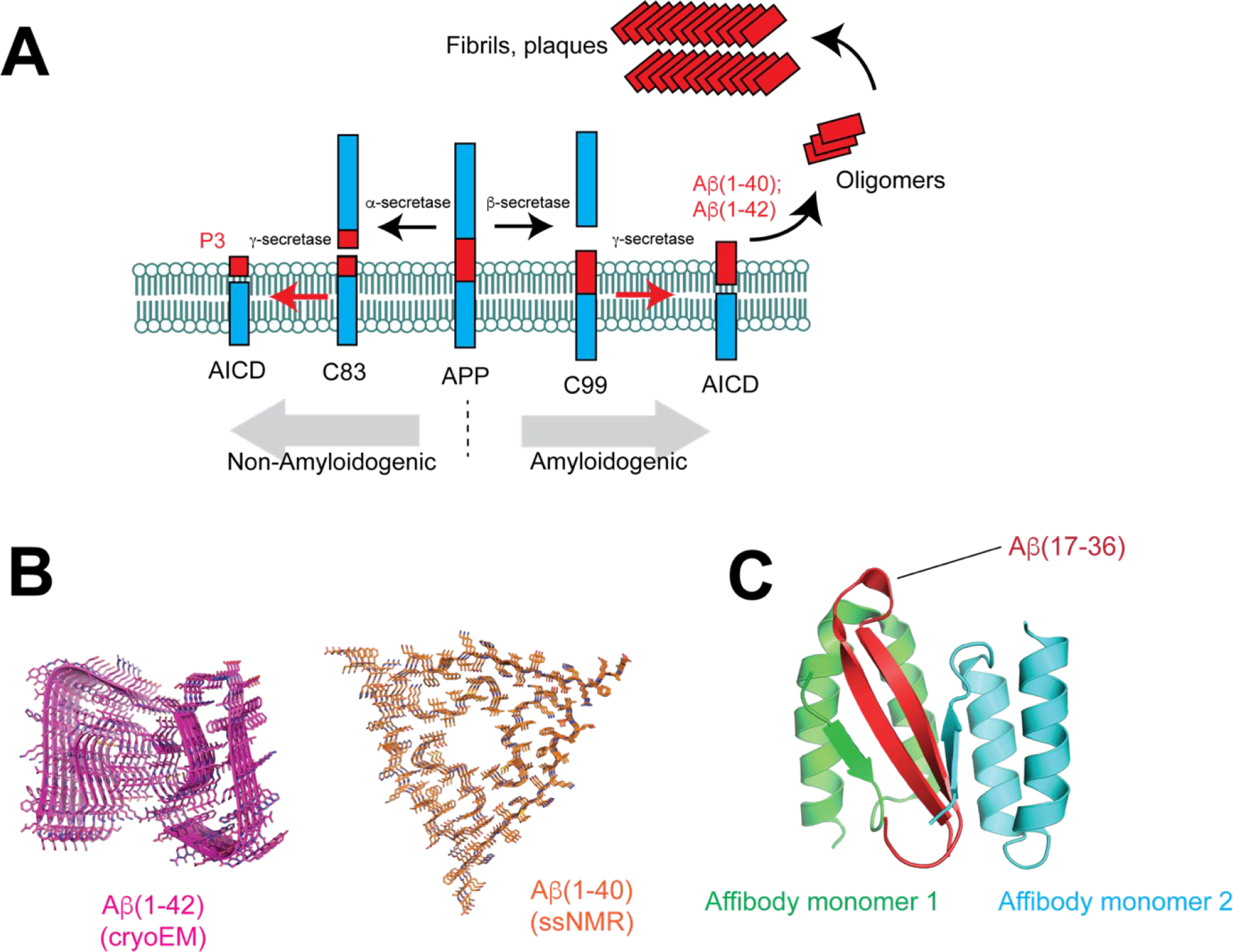
Aβ and its role in Alzheimer’s Disease. (A) Processing of Amyloid precursor protein (APP) by α-, β-, and γ-secretases. Cleavage by α-secretase leads to non-amyloidenic species C83, P3, and AICD. However, cleavage by β- and γ-secretases results in production of Aβ(1–40) and Aβ(1–42) fragments that can form oligomers and ultimately fibrils and plaques. (B) Cross β-structure of Aβ(1–42) (PDB 5OQV) and Aβ(1–40) (PDB 2M4J). (C) Solution NMR structure of Aβ(1–40) in complex with an affibody (PDB 2OTK.
During the pathogenesis of AD, Aβ exists in a number of structurally distinct states, ultimately progressing to form mature, insoluble fibrils that constitute plaques. The structure of Aβ fibrils has been solved by solid-state NMR (Aβ(1–40)) 88 and more recently, cryo-electron microscopy (Aβ(1–42))89. The fibril contains two protofilaments each composed of a parallel cross-β structure (Fig. 7B). Emerging evidence suggests that soluble prefibrillar species are the most neurotoxic, but the intrinsic heterogeneity and metastability of these oligomers has impeded structural studies. The structure of a monomeric Aβ fragment in complex with an affibody protein90, determined by NMR, revealed a β-hairpin comprising residues 17–36 (Fig. 7C). How different Aβ species contribute to the neurotoxicity observed in AD remains largely unknown. It has been shown that Aβ oligomers can form β-barrel pores in a membrane environment, suggesting that disruption of the plasma membrane may be one mechanism by which Aβ oligomers cause toxicity91. Large non-fibril assemblies formed by Aβ-like peptides designed to adopt well-defined oligomers have also been visualized by X-ray crystallography and contain pore-like features.92–93 The diversity of Aβ species has emerged as an important challenge in the development of Aβ-targeted therapeutics.94
Another central pathological mechanism of AD involves Tau protein, which forms neurofibrillary tangles (NFTs) in the brain.95 Tau protein is a microtubule associated protein that is expressed mainly in neurons. Six isoforms exist in the brain, all of which are formed by alternative splicing of the microtubule-associated protein tau (MAPT) gene. Tau is post-translationally modified, including ~80 possible phosphorylation sites that are targets of a diverse array of kinases and phosphatases. Hyperphosphorylation as well as truncation of Tau are thought to contribute to the misfolding and subsequent fibril formation observed in AD. Structurally, tau filaments have been studied by cryo-electron microscopy.96 The core tau filament is composed of residues 306–378 of tau, forming a combined cross-β/β-helix structure of two protofilaments (paired helical, and straight filaments, Fig. 8). Tau lesions are closely correlated with the degree of neurodegeneration in AD,97 which supports its potential as a therapeutic target. Moreover, the distribution of NFTs defines clinical subtypes of AD and NFTs precede Aβ plaque formation. The diversity of physiologic and pathogenic tau isoforms and modification has been a longstanding challenge in the design of tau-targeting agents as potential AD therapies. Nonetheless, different strategies to specifically target pathological tau species are under investigation.
Figure 8.
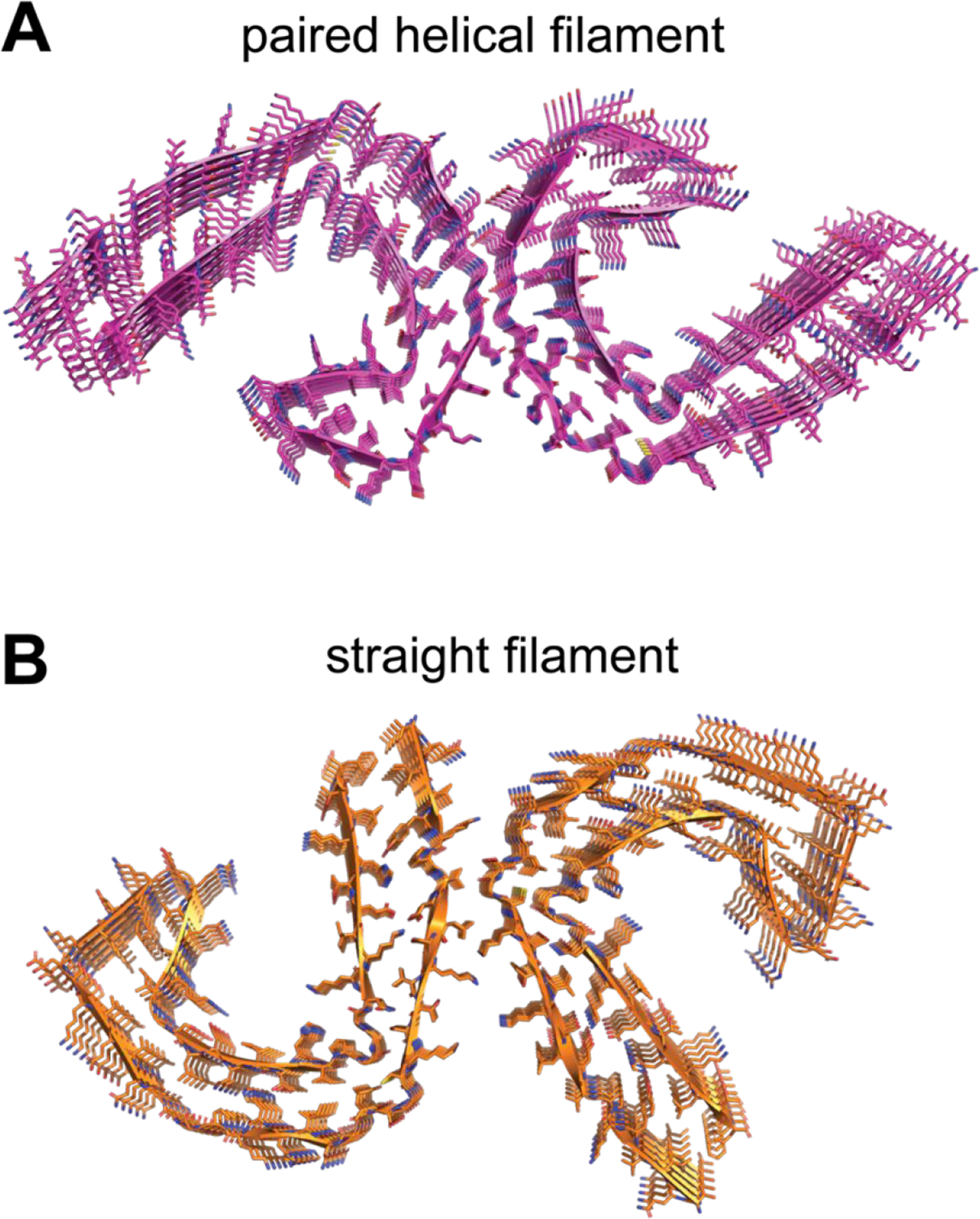
Cross-β/β-helix structure of Tau paired helical (A, PDB 5O3L) or straight (B, PDB 5O3T) filaments.
3.2. Active Immunotherapeutic Strategies.
Early studies supporting the notion of a vaccine for AD involved immunization with synthetic human Aβ(1–42). Preliminary studies demonstrated efficacy in reducing plaque burden in animal models, but clinical trials in humans showed adverse reactions that resulted in the termination of further investigation. These trials that involved immunization with Aβ(1–42) caused meningoencephalitis in 6% of treated patients. Post-mortem analysis indicated a T-cell mediated autoimmune response in these patients. Still, efforts to identify vaccine candidates that elicit an immune response specific to pathologic forms of Aβ, and more recently, Tau, are ongoing. Table 1 lists current candidates under investigation in clinical trials.
Table 1 –
Alzheimer’s Disease Peptide Vaccines in Clinical Development
| Vaccine | Description | Clinical Phase | References |
|---|---|---|---|
| CAD106 | Aβ(1–6) coupled to Qβ VLP | Phase II/III | 98–101 |
| UB311 | Aβ(1–14) fused to helper T-cell epitope | Phase II | 102–103 |
| Lu AF20513 | Aβ(1–12) fused to tetanus toxin epitopes | Phase I | 104 |
| ABvac40 | Multiple repeats of Aβ(33–40) conjugated to KLH | Phase II | 105 |
| ACI-35 | Phosphorylated tau(393–408) with palmitic acid for liposome assembly | Phase I | 106–107 |
| AADvac-1 | Tau(294–305) conjugated to KLH | Phase II | 108–109 |
Mechanistically, active immunization may result in clearance of pathologic Aβ or Tau conformers by eliciting activation of Fc receptor-dependent phagocytosis by microglia. It is also possible that antibodies elicited in the periphery may act as a “peripheral sink” and sequester neurotoxic species to the periphery from the CNS.110 Evidence for both of these mechanisms in animal models exists, but future studies are needed to provide further mechanistic insight into these processes.
3.3. Current Aβ peptide vaccine candidates.
Several vaccine candidates comprised of different N-terminal fragments of Aβ are being explored. Targeting of the N-terminus is due in part to the immunogenic profile of the Aβ peptide; the N-terminus harbors B-cell epitopes whereas the C-terminus is thought to comprise T-cell epitopes. Thus, there is considerable interest in designing an Aβ vaccine that generates a robust anti-Aβ B-cell response while avoiding activation of Aβ-specific T-cells.
CAD106 combines multiple copies of the Aβ(1–6) N-terminal peptide fragment coupled to a Qβ VLP (Fig. 9A). The Aβ(1–6) peptide (DAEFRH) was extended by a GGC spacer and covalently conjugated to the E. coli RNA phage Qβ VLP, such that each particle contains ~350–550 Aβ peptide fragments.98 This VLP carrier was selected to provide an ordered, multivalent scaffold for antigen presentation. In addition to lacking the C-terminal T-cell epitope, the peptide antigen is shorter than typical T-cell epitopes and was computationally determined to be unreactive towards MHC class I and II molecules. In both APP transgenic mice and primates, immunization led to Aβ antibody titers of all IgG subclasses, with Aβ(3–6) as the minimal epitope. Notably, amyloid accumulation in two APP transgenic mouse lines was reduced as observed by plaque number and area and no increased microhemorrhage or adverse inflammatory reactions were observed. In humans, phase I and II clinical trials have demonstrated that repeated CAD106 administration is generally well tolerated and strong serological responses are induced.99–101 Preliminary 18F-florbetapir PET studies suggest that change in PET SUVR correlated inversely with anti-Aβ titers, but further studies with larger patient cohorts are required to evaluate clinical efficacy.
Figure 9.
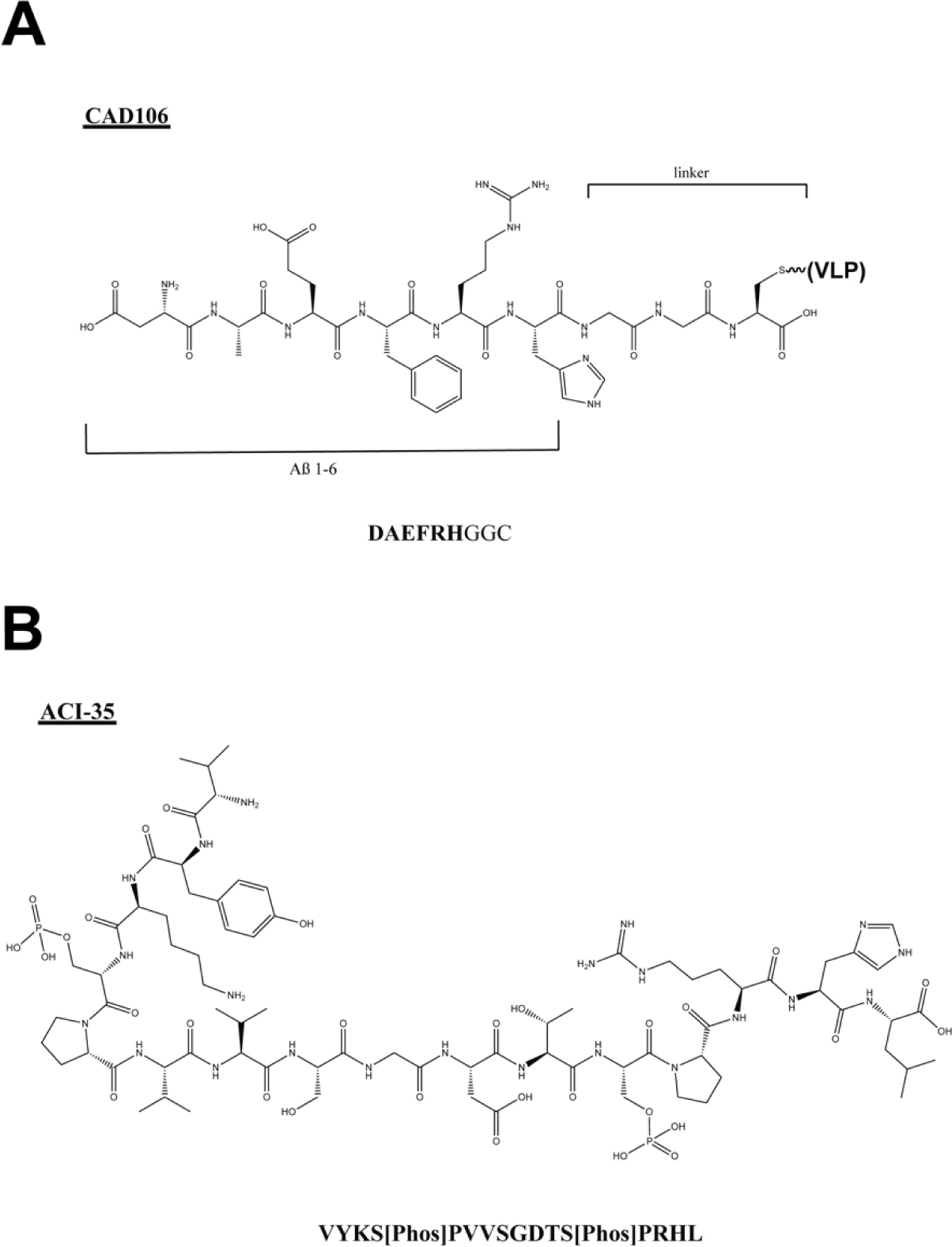
Chemical structures of peptide components of (A) CAD106 and (B) ACI-35.
UB-311 is composed of two synthetic peptides, each consisting of Aβ(1–14) fused to different helper T-cell epitopes (UBITh ®), formulated in a Th2-biased delivery system.102 It is another example of a “next-generation” Aβ vaccine that seeks to eliminate adverse inflammatory responses while maintaining N-terminal anti-Aβ antibodies. The T-helper cell peptide epitopes used are derived from the highly antigenic measles virus fusion protein (MVF 288–302) and hepatitis B virus surface antigen (HBsAg19–33).111 Sites within these epitopes were optimized by combinatorial mutagenesis and selected for broad responsiveness in genetically diverse backgrounds. The peptides are mixed in an equimolar ratio with polyanionic CpG oligodeoxynucleotides to form stable micron-sized particulates mediated by electrostatic interaction. This design strategy biases Th2 type regulatory T-cell responses over Th1 pro-inflammatory T-cell response. UB-311 showed a favorable immunogenic profile in APP transgenic mice, baboons, and macaques. In AD patients, a 100% responder rate was achieved and high levels of anti-Aβ response that bind Aβ monomers, oligomers and fibrils was observed.103
Lu AF20513 includes three copies of Aβ(1–12) interspersed with P30 and P2 Th epitopes from the tetanus toxoid vaccine.104 The goal of this strategy is to eliminate anti-Aβ or anti-APP-specific T-cell responses by activating CD4+ T-lymphocytes specific to foreign tetanus toxoid antigen that exist in previously immunized individuals. Immunization resulted in anti-Aβ antibodies that reduced AD pathology in Tg2576 mice. A strong humoral response was also found in guinea pigs and monkeys, and clinical trials are currently underway to determine safety and tolerability in humans.
While most Aβ vaccine design strategies have focused on the N-terminal epitopes, other approaches are also in development. ABvac40 is a vaccine candidate containing multiple repeats of Aβ(33–40) C-terminal fragment of Aβ(1–40) conjugated to KLH. Aβ(1–40) is the predominant variant of secreted Aβ, and though less toxic and prone to aggregation than Aβ(1–42), studies have demonstrated that high levels of Aβ(1–40) in the brain correlate with AD severity.112–113 Additionally, certain anti-C terminal Aβ mAbs do not bind parental APP as the epitope is concealed within the transmembrane portion, unlike N-terminal directed antibodies. Phase I trials demonstrated ABvac40 is well tolerated in humans and elicits specific anti-Aβ(1–40) antibodies.105
3.4. Current Tau peptide vaccine candidates.
More recently, pathologic conformations of Tau have been targeted in the development of AD vaccines. The major challenge has been the identification of pathologic epitopes that would elicit a selective antibody response that does not engage the many physiologic species of Tau. Several examples of Tau peptide-based vaccines are currently in the pipeline (Table 1).
ACI-35 is a liposome-based vaccine candidate that contains 16 copies of a synthetic Tau fragment (Tau393–408) with phosphorylation of residues S396 and S404 (Fig. 9B).106–107 These Tau phospho-peptides are modified to include two palmitic acid chains at each terminus to allow for assembly into liposomes. CD spectra of the liposome vaccine demonstrates an ordered, β-sheet configuration, which mimics aggregated Tau. In Tau.P301L mice, ACI-35 induced robust antibody titers that markedly reduced Tau lesions in the brain. The vaccine is currently being investigated for safety and efficacy in humans.
AADvac-1 is a synthetic peptide derived from Tau294–305 sequence coupled to KLH. This Tau sequence was determined based on immunization of mice with disordered Tau protein 151–391 followed by mAb isolation and screening for disruption of Tau-Tau interaction in vitro.114 One mAb, DC8E8 was found to reduce Tau oligomerization as measured by thioflavin T fluorescence and also reduced insoluble tau oligomers in transgenic mouse brains. Epitope mapping studies using deletions of full-length Tau, competition studies, as well as structural analysis by X-ray crystallography revealed that the DC8E8 epitope is HXPGGG, which is present 4 times on full length tau. These studies informed the design of AADvac-1, which comprises the Tau 294–305 epitope (KDNIKHVPGGGS) and demonstrated 95% reduction of tau hyperphosphorylation in a rat model of AD following immunization.108 Preliminary studies in humans show that AADvac-1 is well tolerated and further study is warranted in larger trials.109
4. CANCER VACCINES
The boom in immuno-oncology over the past decade has shown that manipulation of the immune response in order to counter the immunosuppressive evasion mechanisms that cancer cells utilize is a powerful approach to treating cancer. For the most part, efforts focus on inducing T-cell responses, since it is believed that T-cells generally have the capability of clearing tumors in the absence of immunosuppressive mechanisms. Most cancer cells can be differentiated from healthy cells by either upregulation/overexpression of certain endogenous proteins, or mutation of those proteins. Thus, any gene product that is expressed differentially or in a mutated form in cancer cells relative to healthy cells is a potential vaccine target. Here, we describe efforts against two targets (Folate receptor, and HER2), and then discuss general “next generation” strategies to use peptides as cancer vaccines or to stimulate T-cells for adoptive cell therapy.
4.1. Folate receptor.
Folate (reduced form) or folic acid (oxidized form) is part of the vitamin B family. Folate is required for proper cell function since it is a necessary cofactor for purine and pyrimidine biosynthesis.115 Folate also plays a key role in protein and phospholipid methylation.116 Folate, which is overall a lipophilic molecule, is transported into the cell by three distinct proteins: 1) the reduced folate carrier (RFC), 2) the proton-coupled folate transporter (PCFT) and 3) the folate receptor (FR).117 Multiple isoforms of FR have been identified, FR-α, FR-β, and FR- γ, which each have a specific tissue distribution and share 70–80% of sequence identity.118–119 The membrane associated form of FR (α and β) can transport folate into the cell. Paradoxically, the major FR-α isoform, with the exception of placental, is mostly expressed at the apical (luminal) surface of epithelial cells, which is not in direct contact with circulating folate.120 Under normal cellular conditions, FR-α expression level is low and is restricted to various epithelial cells, including those in the kidney proximal tubule, placenta, breast, choroid plexus, lung, salivary glands and female reproductive tract.121–123 The role of folate in cancer is not well understood and appears to have different effects depending on the circumstances. For example, in ovarian cancer it was shown that downregulation of the RFC is associated with disease-free survival but upregulation of the FR-α is correlated with tumor progression.124
The key to successful development of an ovarian epithelial or breast cancer vaccine is the identification of tumor-associated antigens that induce CTLs. Based on the observation that normal ovarian epithelium expresses basal levels of FR, but in cancerous ovarian tissue FR expression is >20 fold higher than normal tissue,124–126 several groups have identified circulating FR-α-antigen reactive lymphocytes in ovarian cancer patients and subsequently FR-α-derived immunogenic peptides.127 Pre-existent immunity indicates that FR-α naturally contains immunogenic peptides, making FR-α an ideal candidate for a therapeutic peptide vaccine for ovarian cancer.
Despite advances in surgery, immunological and adjuvant systemic therapies, ovarian cancer causes the highest number of death in the U.S. of gynecologic cancers.128 Regarding breast cancer, an estimated 40,000 deaths occur annually in the US.129 These numbers highlight the need for new therapeutic strategies. Two independent groups have identified several FR-α immunogenic peptides from two distinct HLA-restricted groups. E39 (FR-α 191–199) and E41 (FR-α 245–253) are both HLA-A2-restricted MHC class I FR-α peptides and are efficiently presented to CD8+ T-cells (Table 2). A 2008 phase I clinical trial for advanced stage ovarian cancer used a multi-peptide vaccine approach including E39 with four other MHC class I and one MHC class II peptides along with immunoadjuvant.17 This trial showed good overall safety but moderate functional T-cell response established by enzyme-linked immunospot (ELISpot). Using predictive algorithms, Knutson et al have identified 14 potential MHC class II FR-α peptides and have screened breast and ovarian cancer patients to confirm that 70% of patients demonstrated immunity against at least one peptide and that more than 25% of patients recognized 5 peptides by ELISpot (Table 2).130 Based on those results, a phase 1 clinical trial using five FR-α peptide (FR30, FR56, FR76, FR113 and FR238) admixed with GM-CSF, called TIPV200, was tested on ovarian and breast cancer patients.131 Vaccination was well tolerated and more than 90% of the patients slowly developed an immunity over a 5 month period that persisted at least a year. Currently, Tapimmune is running three distinct phase II clinical trials with TPIV200: 1) TPIV200 in combination with cancer immunotherapy durvalumab for ovarian cancer that progressed after receiving platinium-based chemotherapy; 2) TPIV200 alone as a maintenance therapy for ovarian cancer; and 3) TPIV200 as a treatment for triple negative breast cancer.
Table 2 –
Peptide Vaccines Based on the Folate Receptor
| Vaccine | Sequence | Position |
|---|---|---|
| FR30 | RTELLNVCMNAKHHKEK | 30–46 |
| FR56 | QCRPWRKNACCSTNT | 56–70 |
| FR76 | KDVSYLYRFNWNHCGEMA | 76–93 |
| FR113 | LGPWIQQVDQSWRKERV | 113–129 |
| E39 | EIWTHSTKV | 191–199 |
| FR238 | PWAAWPFLLSLALMLLWL | 238–255 |
| E41 | LLSLALMLL | 245–253 |
4.2. HER2
HER2/neu (also called erB-2, CD340) is a member of the human epidermal growth factor receptor family and one of the most studied oncogenes in cancer. The HER2 signaling pathway promotes cell growth and division.132–134 The HER2 receptor is embedded in the cell membrane by a transmembrane domain and also contains an extracellular ligand binding domain as well as an intracellular tyrosine kinase domain. When HER2 is activated by extracellular ligands, it dimerizes and undergoes transphosphorylation to mediate intracellular signaling and stimulate proliferation. Gene amplification and HER2 protein overexpression is linked to tumor cell proliferation and anti-apoptotic signaling and is found in 15–30% of human breast cancers.135–137 Aberrant HER2 expression is also known to occur in ovarian, uterine, stomach, and other cancers.138 HER2 is the target of the breast cancer drug trastuzumab (Herceptin), which is a mAb that induces an immune-mediated response leading to internalization and down regulation of HER2.139–140 Another drug, pertuzumab blocks a distinct site of HER2 and has been shown to improve survival in HER2-positive breast cancer.141–142 The success of these passive immunotherapeutic approaches targeting HER2 has led to interest in the development of active immunization strategies, which have the potential to elicit a broader anti-tumor immune response with minimal toxicity.
NeuVax (Nelipepimut-S or E75) is a 9-amino acid peptide derived from the extracellular domain of HER2 (369–377; KIFGSLAFL) combined with GM-CSF. It is an immunodominant MHC class I, HLA-A2 and HLA-A3 restricted epitope. Early studies found that the peptide binds HLA-A2/A3 and promotes T-cells to lyse HER2-positive cancer cell lines.143–144 In mouse models, T-cells stimulated with this peptide efficiently lysed HER2 expressing colon and renal cell carcinoma cells.145 NeuVax stimulates specific CD8+ CTLs that recognize and destroy HER2 expressing cancer cells. Human trials demonstrated that NeuVax is well tolerated in humans.146–148 A phase III clinical trial determined that NeuVax monotherapy did not impact breast cancer recurrence compared to placebo.149 Still, NeuVax may hold promise in combination therapies. Two phase II clinical trials investigating NeuVax treatment in HER2 positive breast cancer combined with trastuzumab (NCT02297898 & NCT01570038) are ongoing.
GP2 is a 9-amino acid, MHC class I peptide derived from the transmembrane domain of HER2 (654–662; IISAVVGIL).150–151 This peptide was found to be expressed in HER2 positive ovarian and breast tumors and capable of inducing a CTL response in vitro.152 Clinical testing demonstrated that the vaccine was well tolerated and that patients demonstrated increased HER2-specific CTLs.153 A subsequent phase II study of HLA-A2+, clinically disease-free, high risk breast cancer patients with HER2-positive tumors was conducted.154 Overall recurrence was not reduced in vaccinated patients, but the results suggested possible clinical activity in select HER2-positive cancer patients treated with trastuzumab.
IMU-131 (HerVaxx) is a fusion peptide made of three peptides derived from the extracellular domain of the HER2 conjugated to the carrier protein diphtheria toxin. The three peptides P4, P6, and P7, are B-cell epitopes of the HER2 extracellular domain.155 Immunization studies in c-neu transgenic mice demonstrated delayed tumor onset and reduced growth.156 Phase I trials in women with metastatic breast cancer indicated a robust immune response and that immunization was generally well tolerated.157 Immunogenicity was further optimized by conjugation with CRM197 along with the adjuvant montanide.158
B-Vaxx is another combination of HER2 peptides under investigation in clinical trials. Previous work identified the first generation of HER2 B-cell epitopes (628–647 and 316–339) through a combination of computer algorithms, preclinical testing in vitro and in mice, as well as phase I clinical trials, which indicated safety and effectiveness in eliciting antibody responses in the majority of patients.159–161 B-vaxx peptides were engineered to mimic conformational epitopes based on those defined by the HER2/pertuzumab and HER2/trastuzumab complexes (Fig. 10A). Pertuzumab binds the dimerization loop of subdomain II of the extracellular domain, thereby impeding dimerization and subsequent HER2-mediated signal transduction.162 Three peptides that span the dimerization loop epitope, comprising of residues 266–296, 298–333, and 315–333, were evaluated for their potential to act as vaccine candidates.163 Cyclic, conformational peptides of these sequences were engineered with different disulfide pairings. Vaccination studies in both mice and rabbits demonstrated immunogenicity and one epitope (266–296) reduced the tumor burden in transgenic BALB-neuT mice (Fig. 10B). This peptide, in combination with a similarly engineered peptide that contains the HER2 residues 597–626, which comprises the trastuzumab epitope,164 constitute B-Vaxx (Figure 10B) and were evaluated in phase I trials (Fig. 10B).165 These peptides also incorporated a promiscuous T-cell epitope. The vaccine was well tolerated and generated a sustained humoral response in the majority of patients. Further studies are underway to determine the therapeutic potential of vaccination with HER2-derived peptides in cancer treatment.
Figure 10.
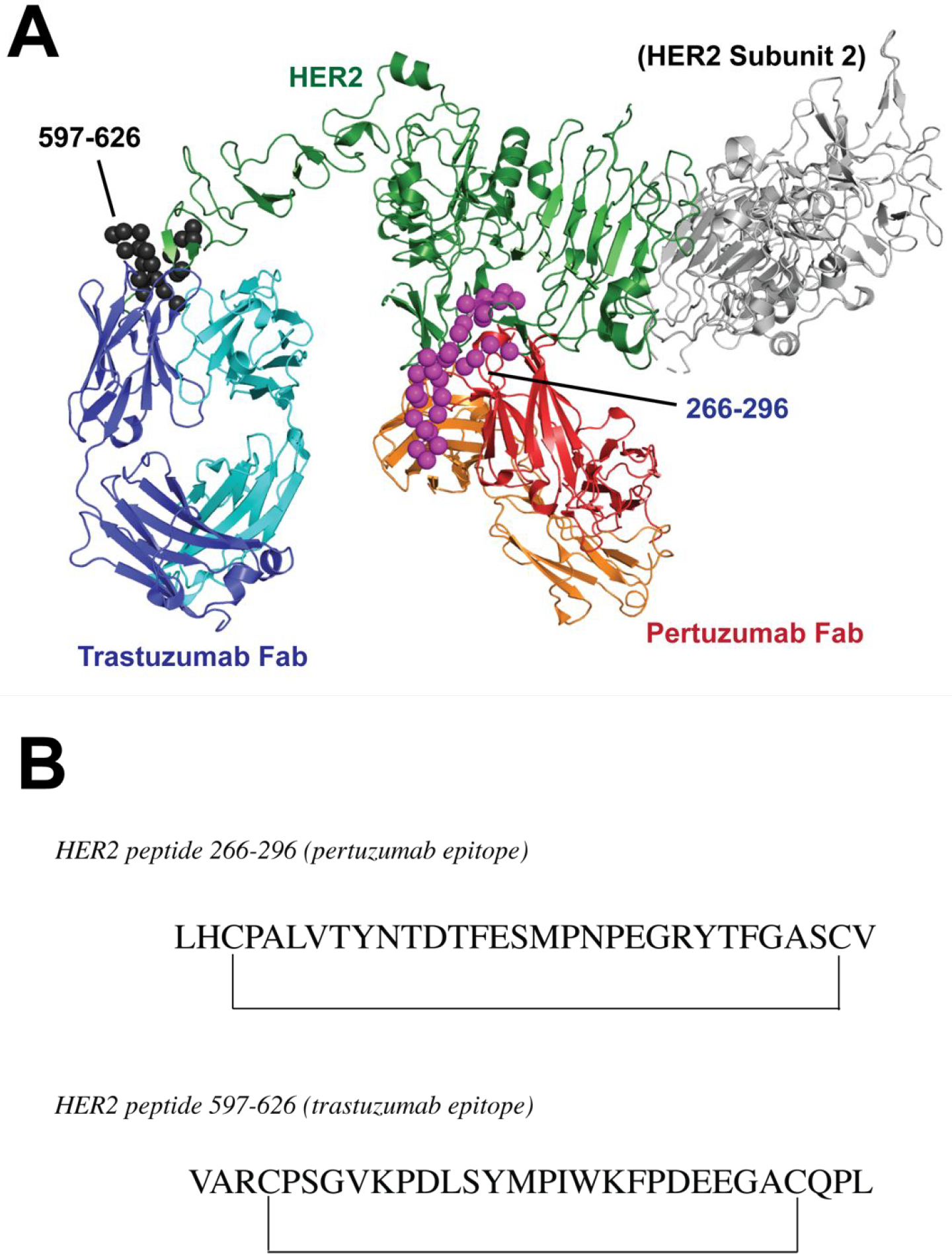
(A) Overlay of X-ray structures of HER2 bound to Pertuzumab Fab (PDB 1S78) and Trastuzumab Fab (PDB ID 1N8Z). HER2 is a dimer, but one of the subunits is colored gray and shown uncomplexed with Fab for clarity. B-Vaxx components are modeled after regions 266–296 (magenta spheres) and 597–626 (black spheres). (B) B-Vaxx peptide epitopes.
4.3. Emerging approaches for identification of and vaccination with cancer epitopes.
Immunotherapy is an exciting new frontier for the treatment of cancer. The past decade has seen major clinical advances for both antibody/protein and cellular therapies. One continual challenge, however, is treatment of solid tumors, which can be difficult to penetrate with macromolecules. The first clinically deployed chimeric antigen receptor T-cell therapy (CAR-T) was for chronic lymphocytic leukemia (CLL), a blood cancer, utilizing CD19 (generic B-cell marker) as the targeting moiety.166 Checkpoint inhibitor antibodies (anti-CTLA4 and anti-PD1) are not effective as monotherapies against most solid tumors.14, 167 Many solid tumors are susceptible to destruction by T-cells, but generally those T-cells within the tumor are actively suppressed by the cancer cells themselves and/or exhausted due to long-term exposure to the tumor-associated antigen. Thus, a significant current effort is to develop new strategies for identification of novel peptide epitopes that are specific to tumor cells.168 In theory, the knowledge gained from such studies could be utilized in active immunization strategies to turn “cold” tumors into “hot” tumors that are susceptible to destruction by general T-cell activation by combination treatment with checkpoint inhibitor therapies.169 Another possibility is the use of novel epitopes to stimulate tumor infiltrating lymphocytes (TILs) ex vivo for adoptive cellular therapy.16
One interesting genomic strategy involves mining whole exome sequencing data from tumors to identify mutations that may constitute novel tumor-specific epitopes (“neoepitopes”) that may recognize TILs. In a seminal 2013 study, Robbins et al. identified 55 putative mutations that fell within predicted HLA-A class I epitopes.170 The corresponding nonamer and decamer peptides were synthesized and tested for their ability to stimulate tumor-derived TILs when presented on HLA-A class I expressing cells. From this screen, two novel peptide epitopes (from non-obvious antigens casein kinase 1, α 1 protein and growth arrest specific 7 gene) were shown to stimulate both patient-derived TILs as well as patient peripheral blood mononuclear cells. Subsequently, a similar strategy was utilized to treat a patient with metastatic cholangiocarcinoma that was refractory to chemotherapy.171 A class II HLA neoepitope in ERBB2 interacting protein was identified and used to expand a TIL culture, which was subsequently infused (along with other therapeutics) which led to a near complete regression.
In addition to identifying natural peptide epitopes for TILs, there is a desire to optimize or probe de novo the reactivity profiles of TCRs from TILs. Here, peptide libraries, synthetic or yeast-displayed, have proven useful. Synthetic one-bead-one-compound methods have been used in a variety of cases to either probe the requirements for natural epitope recognition by alanine scanning, or to enhance reactivity of naturally isolated tumor-associated peptide epitopes.172–175 Recently, a yeast-display approach, in which an optimized HLA-A class I protein was used to present a library of naïve peptides for selection against orphan TCRs from TILs of patients with colorectal adenocarcinoma.176 Peptide epitopes, both mutated and unmutated, were discovered and then shown to activate T-cells that had been retrovirally transduced with the patient-derived TILs. These strategies open an exciting avenue to identification and development of next-generation vaccine candidates.
Recent work has demonstrated that peptide vaccination can enhance the proliferation and activity of CAR-Ts, which may improve their efficacy against solid tumors. One strategy involves using CAR-Ts prepared from lymphocytes that harbor specificity against a particular virus through either the endogenous TCR or a second antigen receptor177–178. Subsequent therapeutic vaccination with peptide antigen was shown to stimulate the anti-tumor response of CAR-Ts. Another method recently described involves peptides conjugated to an amphiphilic lipid that directs the target epitope to lymph nodes179–180. These so-called “amph-ligands” contain a bifunctional distearoyl phosphoethanolamine which binds albumin and can also insert into cell membranes181 as well as either a peptide or small molecule antigen attached by a PEG linker. The amph-ligands accumulated in the lymph nodes and readily inserted into the membrane of dendritic cells. This synthetic antigen presentation stimulated a robust CAR-T response that improved therapeutic activity of the CAR-Ts in multiple solid tumor models in mice. Further investigation is needed to evaluate the potential of this strategy in humans.
5. CONCLUDING REMARKS
The examples discussed herein show how peptides can be harnessed to manipulate the immune system for prophylactic or therapeutic benefit. Like all vaccines, a continual challenge with peptide vaccines stems from the fact that immune responses are still very difficult to predict. Thus, development of optimal immunogens often requires a laborious trial-and-error process of animal immunizations followed by characterization of resulting immune responses. Even with this, there can often be species differences, and thus what works in a mouse may not work in a primate. Still, recent efforts in both structure-based immunogen design as well as de novo analysis of TCR specificities have provided a few shining examples of success. There is increasing evidence that the structure of peptide B-cell epitopes can be important for eliciting antibodies of desired function (e.g., neutralizing activity), and thus methods aimed at stabilizing or presenting peptide epitopes in conformationally relevant contexts are likely to improve the success of vaccines that aim to elicit antibody responses. For infectious disease, this may be especially important in the context of neutralizing epitopes where function of the epitope is often tied to its three-dimensional structure. For Alzheimer’s Disease, targeting the appropriate pathologic oligomeric state of Aβ or Tau may prove to be an important factor for next-generation immunotherapies or vaccines.
For cancer vaccines, much of the current effort is focused on devising new methods to identify new T-cell epitopes that are specific to the tumor. In this case, many of the peptide epitopes can be assessed in vitro utilizing peptide-MHC complexes or using HLA-presenting cells and T-cells. An exciting prospect is that this strategy could be scaled up into a personalized therapeutic approach whereby each patient’s tumor or TILs are sequenced, and then patient-specific peptide vaccines or peptide-stimulated adoptive cell therapies are generated then utilized. Potentially, this strategy could provide greater efficacy than a general approach, such as global upregulation of T-cells (e.g., checkpoint inhibitors), as there is the potential to activate endogenous antitumor responses.
Finally, a likely continued major challenge for peptide vaccines will be the weaker overall immune response that subunit vaccines tend to elicit in comparison to vaccines that contain inactivated or attenuated pathogen. While this issue is of particular relevance in infectious diseases, it may also pertain to other disease areas as well, since stronger immune responses can often be associated with greater protection and durability. There are a number of exciting technologies with potential to overcome this challenge, such as the development of peptide-presenting nanoparticles. However, additional research into this area is warranted. One approach that we have not discussed here, but that has been successful for larger subunit vaccines, is delivery not of the protein itself, but of the genetic material that encodes the protein via liposome-delivered mRNA, in vivo electroporation, or adeno-associated virus (AAV). In this case, the immunogen is produced by the host (typically muscle cells), and thus the immune response can be greater because of the sustained level of immunogen. Whether or not this paradigm could be adapted to peptide-based immunogens remains to be determined but is an exciting proposition.
In summary, the development of peptide vaccines to combat human disease holds great promise but also will face continued challenges. Vaccines have been highly beneficial for reducing mortality and illness due to infectious disease and have the potential to have similar impact in chronic diseases.
FUNDING STATEMENT
J. R. L. gratefully acknowledges funding from the NIH (R01-AI125462, R01-AI132256, and R42-AI122403) as well as the Irma T. Hirschl Trust. R. J. M. was supported in part by the Einstein Medical Scientist Training Program (T32-GM007288).
ABBREVIATIONS
- AAV
Adeno-associated virus
- AMA-1
Apical membrane antigen 1
- AD
Alzheimer’s disease
- APC
Antigen-presenting cell
- APP
Amyloid precursor protein
- BCR
B-cell receptor
- bNAb
Broadly neutralizing antibody
- CNS
Central nervous system
- CSP
Circumsporozoite protein
- CTL
Cytotoxic T-lymphocyte
- FR
Folate receptor
- HA
Hemagluttinin
- HCV
Hepatitis C virus
- HIV-1
Human immunodeficiency virus type 1
- HLA
Human leukocyte antigen
- KLH
Keyhole limpet hemocyanin
- IRIV
Immuno-potentiating influenza virosome
- mAb
Monoclonal antibody
- MAPT
Microtubule-associated protein tau
- MHC
Major histocompatibility complex
- NFT
Neurofibrillary tangles
- PET
Positron emission tomography
- pHLA
Peptide/HLA complex
- pMHC
Peptide/MHC complex
- RBC
Red blood cell
- RSV
Respiratory syncytial virus
- TCR
T-cell receptor
- TIL
Tumor-infiltrating lymphocyte
- VLP
Virus-like particle
Biographies
Ryan Malonis received his B.S. in Biochemistry from Boston College in 2013. He worked as a research assistant at the New York University School of Medicine before entering the Medical Scientist Training Program at the Albert Einstein College of Medicine as an M.D.-Ph.D. candidate in 2015. He is currently performing his thesis research in the laboratory of Professor Jonathan Lai in the Department of Biochemistry. His research is focused on monoclonal antibody isolation and characterization, to study the humoral immune response to emerging viral pathogens. His scientific interests include protein and antibody engineering, rational vaccine design, B-cell immunology as well as viral pathogenesis and host-pathogen interactions.
Jonathan Lai received his B. Sc. (Hons.) in Biochemistry from Queen’s University (Kingston, Ontario, Canada) in 1999; and his Ph. D. in 2004 in Biophysics and Chemistry from the University of Wisconsin-Madison where he trained with Professor Samuel H. Gellman as a Natural Sciences and Engineering Research Council (NSERC) of Canada PGS B scholar. He was Helen Hay Whitney post-doctoral fellow from 2004–2007 in Biological Chemistry at Harvard Medical School where he worked in the groups of Professor Christopher T. Walsh and Stephen C. Harrison. He began his independent faculty position at the Albert Einstein College of Medicine in 2007, where he is currently Professor of Biochemistry. He is the recipient of the Arnold and Mabel Young Investigator Award, and the Irma T. Hirschl/Monique Weill-Caulier Career Scientist Award. Professor Lai’s group is broadly interested in protein engineering and antibody isolation strategies for the development of novel immunotherapies and vaccines.
Olivia Vergnolle received a B.S. and M.S. in Cellular and Molecular Biology from University Pierre and Marie Curie, France in 2005. She obtained her Biochemistry Ph.D. at Cambridge University as a Marie Curie early stage training fellow under the guidance of Professor Peter Leadlay, where she investigated how polyketide synthase enzyme control stereochemistry during natural product biosynthesis. After moving to New York City as a postdoctoral researcher, she focused her research on bacterial enzymes involve in Mycobacterium tuberculosis virulence in the laboratories of Professor Luis Quadri and Professor John Blanchard. She is currently a Research Assistant Professor at the Albert Einstein College of Medicine in Professor Jonathan Lai’s group. Her main research interest is centered on the development of immunogen- or antibody-based antiviral treatment to respond to current and emerging viral threats.
Footnotes
CONFLICT OF INTEREST STATEMENT
The Albert Einstein College of Medicine has filed a US patent application on the vaccine candidates discussed in reference 12 entitled “Dengue virus glycoprotein E DIII variants and uses thereof” (PCT/US2017/017637) with J. R. L. as co-inventor.
REFERENCES
- 1.Global Vaccine Action Plan 2011–2020. World Health Organization: https://www.who.int/immunization/global_vaccine_action_plan/GVAP_doc_2011_2020/en/. Accessed on 11/26/19 [Google Scholar]
- 2.Kwong PD; Wilson IA HIV-1 and Influenza Antibodies: Seeing Antigens in New Ways. Nature Immunol. 2009, 10, 573–578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Burton DR; Hangartner L Broadly Neutralizing Antibodies to HIV and Their Role in Vaccine Design. Ann. Rev. Immunol. 2016, 34, 635–659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Burton DR; Poignard P; Stanfield RL; Wilson IA Broadly Neutralizing Antibodies Present New Prospects to Counter Highly Antigenically Diverse Viruses. Science 2012, 337, 183–186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Arama C; Troye-Blomberg M The Path of Malaria Vaccine Development: Challenges and Perspectives. J. Intern. Med. 2014, 275, 456–66. [DOI] [PubMed] [Google Scholar]
- 6.Halstead SB Safety Issues from a Phase 3 Clinical Trial of a Live-Attenuated Chimeric Yellow Fever Tetravalent Dengue Vaccine. Hum. Vacc. Immunother. 2018, 14, 2158–2162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Verreault D; Ennis J; Whaley K; Killeen SZ; Karauzum H; Aman MJ; Holtsberg R; Doyle-Meyers L; Didier PJ; Zeitlin L; Roy CJ Effective Treatment of Staphylococcal Enterotoxin B Aerosol Intoxication in Rhesus Macaques by Using Two Parenterally Administered High-Affinity Monoclonal Antibodies. Antimicrob. Agents Chemother. 2019, 63, pii: e02049–18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pohl MA; Rivera J; Nakouzi A; Chow SK; Casadevall A Combinations of Monoclonal Antibodies to Anthrax Toxin Manifest New Properties in Neutralization Assays. Infect. Immun. 2013, 81, 1880–1888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Arunachalam B; Ghosh S; Talwar GP; Raghupathy R A Single Human Monoclonal Antibody that Confers Total Protection from Tetanus. Hybridoma 1992, 11, 165–179. [DOI] [PubMed] [Google Scholar]
- 10.Koellhoffer JF; Higgins CD; Lai JR Protein Engineering Strategies for the Development of Viral Vaccines and Immunotherapeutics. FEBS Lette. 2014, 588, 298–307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Impagliazzo A; Milder F; Kuipers H; Wagner MV; Zhu X; Hoffman RM; van Meersbergen R; Huizingh J; Wanningen P; Verspuij J; de Man M; Ding Z; Apetri A; Kukrer B; Sneekes-Vriese E; Tomkiewicz D; Laursen NS; Lee PS; Zakrzewska A; Dekking L; Tolboom J; Tettero L; van Meerten S; Yu W; Koudstaal W; Goudsmit J; Ward AB; Meijberg W; Wilson IA; Radosevic K A Stable Trimeric Influenza Hemagglutinin Stem as a Broadly Protective Immunogen. Science 2015, 349, 1301–1306. [DOI] [PubMed] [Google Scholar]
- 12.Frei JC; Wirchnianski AS; Govero J; Vergnolle O; Dowd KA; Pierson TC; Kielian M; Girvin ME; Diamond MS; Lai JR Engineered Dengue Virus Domain III Proteins Elicit Cross-Neutralizing Antibody Responses in Mice. J. Virol. 2018, 92, pii: e01023–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Saphire EO; Montero M; Menendez A; van Houten NE; Irving MB; Pantophlet R; Zwick MB; Parren PW; Burton DR; Scott JK; Wilson IA Structure of a High-Affinity “Mimotope” Peptide Bound to HIV-1-Neutralizing Antibody b12 Explains its Inability io Elicit gp120 Cross-Reactive Antibodies. J. Mol. Biol. 2007, 369, 696–709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sharma P; Allison JP Immune Checkpoint Targeting in Cancer Therapy: Toward Combination Strategies with Curative Potential. Cell 2015, 161, 205–214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Garboczi DN; Ghosh P; Utz U; Fan QR; Biddison WE; Wiley DC Structure of the Complex Between Human T-Cell Receptor, Viral Peptide and HLA-A2. Nature 1996, 384, 134–141. [DOI] [PubMed] [Google Scholar]
- 16.Yang JC; Rosenberg SA Adoptive T-Cell Therapy for Cancer. Adv. Immunol. 2016, 130, 279–294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chianese-Bullock KA; Irvin WP Jr.; Petroni GR; Murphy C; Smolkin M; Olson WC; Coleman E; Boerner SA; Nail CJ; Neese PY; Yuan A; Hogan KT; Slingluff CL Jr. A Multipeptide Vaccine Is Safe and Elicits T-Cell Responses in Participants with Advanced Stage Ovarian Cancer. J. Immunother. 2008, 31, 420–430. [DOI] [PubMed] [Google Scholar]
- 18.McShan AC; Natarajan K; Kumirov VK; Flores-Solis D; Jiang J; Badstubner M; Toor JS; Bagshaw CR; Kovrigin EL; Margulies DH; Sgourakis NG Peptide Exchange on MHC-I by TAPBPR is Driven by a Negative Allostery Release Cycle. Nat. Chem. Biol. 2018, 14, 811–820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Luimstra JJ; Garstka MA; Roex MCJ; Redeker A; Janssen GMC; van Veelen PA; Arens R; Falkenburg JHF; Neefjes J; Ovaa H A Flexible MHC Class I Multimer Loading System for Large-Scale Detection of Antigen-Specific T Cells. J. Exp. Med. 2018, 215, 1493–1504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rodenko B; Toebes M; Hadrup SR; van Esch WJ; Molenaar AM; Schumacher TN; Ovaa H Generation of Peptide-MHC Class I Complexes through UV-Mediated Ligand Exchange. Nat. Protoc. 2006, 1, 1120–1132. [DOI] [PubMed] [Google Scholar]
- 21.Ghaffari-Nazari H; Tavakkol-Afshari J; Jaafari MR; Tahaghoghi-Hajghorbani S; Masoumi E; Jalali SA Improving Multi-Epitope Long Peptide Vaccine Potency by Using a Strategy that Enhances CD4+ T Help in BALB/c Mice. PloS One 2015, 10, e0142563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Correia BE; Bates JT; Loomis RJ; Baneyx G; Carrico C; Jardine JG; Rupert P; Correnti C; Kalyuzhniy O; Vittal V; Connell MJ; Stevens E; Schroeter A; Chen M; Macpherson S; Serra AM; Adachi Y; Holmes MA; Li Y; Klevit RE; Graham BS; Wyatt RT; Baker D; Strong RK; Crowe JE Jr.; Johnson PR; Schief WR Proof of Principle for Epitope-Focused Vaccine Design. Nature 2014, 507, 201–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.World malaria report 2018. World Health Organization; 2018: https://www.who.int/malaria/publications/world-malaria-report-2018/en/. Accessed on 11/26/19. [Google Scholar]
- 24.White MT; Shirreff G; Karl S; Ghani AC; Mueller I Variation in Relapse Frequency and the Transmission Potential of Plasmodium Vivax Malaria. Proc. Biol. Sci. 2016, 283, 20160048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mackinnon MJ; Marsh K, The selection landscape of malaria parasites. Science 2010, 328 (5980), 866–71. [DOI] [PubMed] [Google Scholar]
- 26.Hafalla JC; Silvie O; Matuschewski K Cell biology and immunology of malaria. Immunol Rev 2011, 240 (1), 297–316. [DOI] [PubMed] [Google Scholar]
- 27.Collins WE; Jeffery GM A Retrospective Examination of Sporozoite- and Trophozoite-Induced Infections with Plasmodium Falciparum: Development of Parasitologic and Clinical Immunity During Primary Infection. Am. J. Trop. Med. Hyg. 1999, 61, 4–19. [DOI] [PubMed] [Google Scholar]
- 28.Drakeley C; Sutherland C; Bousema JT; Sauerwein RW; Targett GA The Epidemiology of Plasmodium Falciparum Gametocytes: Weapons of Mass Dispersion. Trends Parasitol. 2006, 22, 424–430. [DOI] [PubMed] [Google Scholar]
- 29.Ouattara A; Laurens MB Vaccines Against Malaria. Clin. Infect. Dis. 2015, 60, 930–936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Draper SJ; Sack BK; King CR; Nielsen CM; Rayner JC; Higgins MK; Long CA; Seder RA Malaria Vaccines: Recent Advances and New Horizons. Cell Host Microbe 2018, 24, 43–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Dame JB; Williams JL; McCutchan TF; Weber JL; Wirtz RA; Hockmeyer WT; Maloy WL; Haynes JD; Schneider I; Roberts D; Sanders GS; Reddy EP; Diggs CL; Miller LH Structure of the Gene Encoding the Immunodominant Surface Antigen on the Sporozoite of the Human Malaria Parasite Plasmodium Falciparum. Science, 1984, 225, 593–599. [DOI] [PubMed] [Google Scholar]
- 32.Kappe SH; Buscaglia CA; Nussenzweig V Plasmodium Sporozoite Molecular Cell Biology. Annu. Rev. Cell Dev. Biol. 2004, 20, 29–59. [DOI] [PubMed] [Google Scholar]
- 33.Sedegah M; Kim Y; Ganeshan H; Huang J; Belmonte M; Abot E; Banania JG; Farooq F; McGrath S; Peters B; Sette A; Soisson L; Diggs C; Doolan DL; Tamminga C; Villasante E; Hollingdale MR; Richie TL Identification of Minimal Human MHC-Restricted CD8+ T-Cell Epitopes within the Plasmodium Falciparum Circumsporozoite Protein (CSP). Malar. J. 2013, 12, 185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zavala F; Tam JP; Masuda A, Synthetic Peptides as Antigens for the Detection of Humoral Immunity to Plasmodium Falciparum Sporozoites. J. Immunol. Methods 1986, 93, 55–61. [DOI] [PubMed] [Google Scholar]
- 35.Ghasparian A; Moehle K; Linden A; Robinson JA Crystal Structure of an NPNA-Repeat Motif from the Circumsporozoite Protein of the Malaria Parasite Plasmodium Falciparum. Chem. Commun. 2006, 2, 174–176. [DOI] [PubMed] [Google Scholar]
- 36.Cech PG; Aebi T; Abdallah MS; Mpina M; Machunda EB; Westerfeld N; Stoffel SA; Zurbriggen R; Pluschke G; Tanner M; Daubenberger C; Genton B; Abdulla S, Virosome-Formulated Plasmodium Falciparum AMA-1 & CSP Derived Peptides as Malaria Vaccine: Randomized Phase 1b Trial in Semi-Immune Adults & Children. PLoS One 2011, 6, e22273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Genton B; Pluschke G; Degen L; Kammer AR; Westerfeld N; Okitsu SL; Schroller S; Vounatsou P; Mueller MM; Tanner M; Zurbriggen R A Randomized Placebo-Controlled Phase Ia Malaria Vaccine Trial of Two Virosome-Formulated Synthetic Peptides in Healthy Adult Volunteers. PLoS One 2007, 2, e1018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zurbriggen R, Immunostimulating Reconstituted Influenza Virosomes. Vaccine 2003, 21, 921–924. [DOI] [PubMed] [Google Scholar]
- 39.Okitsu SL; Kienzl U; Moehle K; Silvie O; Peduzzi E; Mueller MS; Sauerwein RW; Matile H; Zurbriggen R; Mazier D; Robinson JA; Pluschke G, Structure-Activity-Based Design of a Synthetic Malaria Peptide Eliciting Sporozoite Inhibitory Antibodies in a Virosomal Formulation. Chem. Biol. 2007, 14, 577–587. [DOI] [PubMed] [Google Scholar]
- 40.Hodder AN; Crewther PE; Matthew ML; Reid GE; Moritz RL; Simpson RJ; Anders RF, The Disulfide Bond Structure of Plasmodium Apical Membrane Antigen-1. J. Biol. Chem. 1996, 271, 29446–29452. [DOI] [PubMed] [Google Scholar]
- 41.Marshall VM; Zhang L; Anders RF; Coppel RL Diversity of the Vaccine Candidate AMA-1 of Plasmodium Falciparum. Mol. Biochem. Parasitol. 1996, 77, 109–113. [DOI] [PubMed] [Google Scholar]
- 42.Triglia T; Healer J; Caruana SR; Hodder AN; Anders RF; Crabb BS; Cowman AF Apical Membrane Antigen 1 Plays a Central Role in Erythrocyte Invasion by Plasmodium Species. Mol. Microbiol. 2000, 38, 706–718. [DOI] [PubMed] [Google Scholar]
- 43.Urquiza M; Suarez JE; Cardenas C; Lopez R; Puentes A; Chavez F; Calvo JC; Patarroyo ME Plasmodium Falciparum AMA-1 Erythrocyte Binding Peptides Implicate AMA-1 as Erythrocyte Binding Protein. Vaccine 2000, 19, 508–13. [DOI] [PubMed] [Google Scholar]
- 44.Mueller MS; Renard A; Boato F; Vogel D; Naegeli M; Zurbriggen R; Robinson JA; Pluschke G Induction of Parasite Growth-Inhibitory Antibodies by a Virosomal Formulation of a Peptidomimetic of Loop I From Domain III of Plasmodium Falciparum Apical Membrane Antigen 1. Infect. Immun. 2003, 71, 4749–4758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Alter MJ Epidemiology of Hepatitis C Virus Infection. World J. Gastroenterol. 2007, 13, 2436–2441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hajarizadeh B; Grebely J; Dore GJ Epidemiology and Natural History of HCV Infection. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 553–562. [DOI] [PubMed] [Google Scholar]
- 47.Luxenburger H; Neumann-Haefelin C; Thimme R; Boettler T HCV-Specific T Cell Responses During and After Chronic HCV Infection. Viruses 2018, 10, pii: E645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Park S-H; Rehermann B Immune Responses to HCV and Other Hepatitis Viruses. Immunity 2014, 40, 13–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Keck Z-Y; Pierce BG; Lau P; Lu J; Wang Y; Underwood A; Bull RA; Prentoe J; Velázquez-Moctezuma R; Walker MR; Luciani F; Guest JD; Fauvelle C; Baumert TF; Bukh J; Lloyd AR; Foung SKH Broadly Neutralizing Antibodies From an Individual that Naturally Cleared Multiple Hepatitis C Virus Infections Uncover Molecular Determinants for E2 Targeting and Vaccine Design. PLoS Pathog. 2019, 15, e1007772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.de Jong YP; Dorner M; Mommersteeg MC; Xiao JW; Balazs AB; Robbins JB; Winer BY; Gerges S; Vega K; Labitt RN; Donovan BM; Giang E; Krishnan A; Chiriboga L; Charlton MR; Burton DR; Baltimore D; Law M; Rice CM; Ploss A Broadly Neutralizing Antibodies Abrogate Established Hepatitis C Virus Infection. Sci. Transl. Med. 2014, 6, 254ra129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Cashman SB; Marsden BD; Dustin LB, The Humoral Immune Response to HCV: Understanding is Key to Vaccine Development. Front. Immunol. 2014, 5, 550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Firbas C; Jilma B; Tauber E; Buerger V; Jelovcan S; Lingnau K; Buschle M; Frisch J; Klade CS, Immunogenicity And Safety of a Novel Therapeutic Hepatitis C Virus (HCV) Peptide Vaccine: A Randomized, Placebo Controlled Trial for Dose Optimization in 128 Healthy Subjects. Vaccine 2006, 24, 4343–4353. [DOI] [PubMed] [Google Scholar]
- 53.Klade CS; Wedemeyer H; Berg T; Hinrichsen H; Cholewinska G; Zeuzem S; Blum H; Buschle M; Jelovcan S; Buerger V; Tauber E; Frisch J; Manns MP Therapeutic Vaccination of Chronic Hepatitis C Nonresponder Patients with the Peptide Vaccine IC41. Gastroenterology 2008, 134, 1385–1395. [DOI] [PubMed] [Google Scholar]
- 54.Firbas C; Boehm T; Buerger V; Schuller E; Sabarth N; Jilma B; Klade CS, Immunogenicity and Safety of Different Injection Routes and Schedules of IC41, A Hepatitis C Virus (HCV) Peptide Vaccine. Vaccine 2010, 28, 2397–2407. [DOI] [PubMed] [Google Scholar]
- 55.Klade CS; Schuller E; Boehm T; von Gabain A; Manns MP Sustained Viral Load Reduction in Treatment-Naive HCV Genotype 1 Infected Patients after Therapeutic Peptide Vaccination. Vaccine 2012, 30, 2943–2950. [DOI] [PubMed] [Google Scholar]
- 56.Pierce BG; Boucher EN; Piepenbrink KH; Ejemel M; Rapp CA; Thomas WD; Sundberg EJ; Weng Z; Wang Y Structure-Based Design of Hepatitis C Virus Vaccines that Elicit Neutralizing Antibody Responses To A Conserved Epitope. J. Virol. 2017, 91, e01032–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kong L; Lee DE; Kadam RU; Liu T; Giang E; Nieusma T; Garces F; Tzarum N; Woods VL; Ward AB Structural Flexibility at a Major Conserved Antibody Target on Hepatitis C Virus E2 Antigen. Proc. Natl. Acad. Sci. USA 2016, 113, 12768–12773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Meuleman TJ; Dunlop JI; Owsianka AM; van de Langemheen H; Patel AH; Liskamp RMJ, Immobilization by Surface Conjugation of Cyclic Peptides for Effective Mimicry of the HCV-Envelope E2 Protein as a Strategy toward Synthetic Vaccines. Bioconjug. Chem. 2018, 29, 1091–1101. [DOI] [PubMed] [Google Scholar]
- 59.Keck Z.-y.; Xia J; Wang Y; Wang W; Krey T; Prentoe J; Carlsen T; Li AY-J; Patel AH; Lemon SM; Bukh J; Rey FA; Foung SKH. Human Monoclonal Antibodies to a Novel Cluster of Conformational Epitopes on HCV E2 with Resistance to Neutralization Escape in a Genotype 2a Isolate. PLoS Pathog. 2012, 8, e1002653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Krey T; Meola A; Keck Z.-y.; Damier-Piolle L; Foung SKH; Rey FA, Structural Basis of HCV Neutralization by Human Monoclonal Antibodies Resistant to Viral Neutralization Escape. PLoS Pathog. 2013, 9, e1003364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Filskov J; Mikkelsen M; Hansen PR; Christensen JP; Thomsen AR; Andersen P; Bukh J; Agger EM Broadening CD4+ And CD8+ T Cell Responses Against Hepatitis C Virus by Vaccination with NS3 Overlapping Peptide Panels in Cross-Priming Liposomes. J. Virol. 2017, 91, e00130–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Julien JP; Lee PS; Wilson IA, Structural Insights into Key Sites of Vulnerability on HIV-1 Env And Influenza HA. Immnol. Rev. 2012, 250, 180–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Lang S; Xie J; Zhu X; Wu NC; Lerner RA; Wilson IA, Antibody 27F3 Broadly Targets Influenza A Group 1 and 2 Hemagglutinins through a Further Variation in VH1–69 Antibody Orientation on the HA Stem. Cell Rep. 2017, 20, 2935–2943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ekiert DC; Bhabha G; Elsliger MA; Friesen RH; Jongeneelen M; Throsby M; Goudsmit J; Wilson IA, Antibody Recognition of a Highly Conserved Influenza Virus Epitope. Science 2009, 324, 246–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Wang TT; Tan GS; Hai R; Pica N; Ngai L; Ekiert DC; Wilson IA; Garcia-Sastre A; Moran TM; Palese P Vaccination with a Synthetic Peptide from the Influenza Virus Hemagglutinin Provides Protection Against Distinct Viral Subtypes. Proc. Natl. Acad. Sci. USA 2010, 107, 18979–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Jiang Z; Gera L; Mant CT; Hirsch B; Yan Z; Shortt JA; Pollock DD; Qian Z; Holmes KV; Hodges RS Platform Technology to Generate Broadly Cross-Reactive Antibodies to Alpha-Helical Epitopes in Hemagglutinin Proteins from Influenza A Viruses. Biopolymers 2016, 106, 144–159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Atsmon J; Caraco Y; Ziv-Sefer S; Shaikevich D; Abramov E; Volokhov I; Bruzil S; Haima KY; Gottlieb T; Ben-Yedidia T Priming by a Novel Universal Influenza Vaccine (Multimeric-001)-A Gateway For Improving Immune Response in the Elderly Population. Vaccine 2014, 32, 5816–5823. [DOI] [PubMed] [Google Scholar]
- 68.Atsmon J; Kate-Ilovitz E; Shaikevich D; Singer Y; Volokhov I; Haim KY; Ben-Yedidia T, Safety And Immunogenicity of Multimeric-001--A Novel Universal Influenza Vaccine. J. Clin. Immunol. 2012, 32, 595–603. [DOI] [PubMed] [Google Scholar]
- 69.van Doorn E; Liu H; Ben-Yedidia T; Hassin S; Visontai I; Norley S; Frijlink HW; Hak E, Evaluating the Immunogenicity and Safety of a Biondvax-Developed Universal Influenza Vaccine (Multimeric-001) Either as a Standalone Vaccine or as a Primer to H5N1 Influenza Vaccine: Phase IIb Study Protocol. Medicine 2017, 96, e6339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Akapirat S; Karnasuta C; Vasan S; Rerks-Ngarm S; Pitisuttithum P; Madnote S; Savadsuk H; Rittiroongrad S; Puangkaew J; Phogat S; Tartaglia J; Sinangil F; de Souza MS; Excler JL; Kim JH; Robb ML; Michael NL; Ngauy V; O’Connell RJ; Karasavvas N Characterization of HIV-1 gp120 Antibody Specificities Induced in Anogenital Secretions of RV144 Vaccine Recipients After Late Boost Immunizations. PLoS One 2018, 13, e0196397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Barouch DH; Whitney JB; Moldt B; Klein F; Oliveira TY; Liu J; Stephenson KE; Chang H-W; Shekhar K; Gupta S; Nkolola JP; Seaman MS; Smith KM; Borducchi EN; Cabral C; Smith JY; Blackmore S; Sanisetty S; Perry JR; Beck M; Lewis MG; Rinaldi W; Chakraborty AK; Poignard P; Nussenzweig MC; Burton DR Therapeutic Efficacy of Potent Neutralizing HIV-1-Specific Monoclonal Antibodies in SHIV-Infected Rhesus Monkeys. Nature 2013, 503, 224–228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Cai H; Orwenyo J; Giddens JP; Yang Q; Zhang R; LaBranche CC; Montefiori DC; Wang LX Synthetic Three-Component HIV-1 V3 Glycopeptide Immunogens Induce Glycan-Dependent Antibody Responses. Cell Chem. Biol. 2017, 24, 1513–1522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Kong L; Lee JH; Doores KJ; Murin CD; Julien JP; McBride R; Liu Y; Marozsan A; Cupo A; Klasse PJ; Hoffenberg S; Caulfield M; King CR; Hua Y; Le KM; Khayat R; Deller MC; Clayton T; Tien H; Feizi T; Sanders RW; Paulson JC; Moore JP; Stanfield RL; Burton DR; Ward AB; Wilson IA, Supersite of Immune Vulnerability on the Glycosylated Face of HIV-1 Envelope Glycoprotein gp120. Nat. Struct. Mol. Biol. 2013, 20, 796–803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.McLellan JS; Pancera M; Carrico C; Gorman J; Julien JP; Khayat R; Louder R; Pejchal R; Sastry M; Dai K; O’Dell S; Patel N; Shahzad-ul-Hussan S; Yang Y; Zhang B; Zhou T; Zhu J; Boyington JC; Chuang GY; Diwanji D; Georgiev I; Kwon YD; Lee D; Louder MK; Moquin S; Schmidt SD; Yang ZY; Bonsignori M; Crump JA; Kapiga SH; Sam NE; Haynes BF; Burton DR; Koff WC; Walker LM; Phogat S; Wyatt R; Orwenyo J; Wang LX; Arthos J; Bewley CA; Mascola JR; Nabel GJ; Schief WR; Ward AB; Wilson IA; Kwong PD Structure of HIV-1 gp120 V1/V2 Domain with Broadly Neutralizing Antibody PG9. Nature 2011, 480, 336–343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Mouquet H; Scharf L; Euler Z; Liu Y; Eden C; Scheid JF; Halper-Stromberg A; Gnanapragasam PN; Spencer DI; Seaman MS; Schuitemaker H; Feizi T; Nussenzweig MC; Bjorkman PJ Complex-type N-glycan Recognition by Potent Broadly Neutralizing HIV Antibodies. Proc. Natl. Acad. USA 2012, 109, E3268–3277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Orwenyo J; Cai H; Giddens J; Amin MN; Toonstra C; Wang LX Systematic Synthesis and Binding Study of HIV V3 Glycopeptides Reveal the Fine Epitopes of Several Broadly Neutralizing Antibodies. ACS Chem. Biol. 2017, 12, 1566–1575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Pejchal R; Doores KJ; Walker LM; Khayat R; Huang PS; Wang SK; Stanfield RL; Julien JP; Ramos A; Crispin M; Depetris R; Katpally U; Marozsan A; Cupo A; Maloveste S; Liu Y; McBride R; Ito Y; Sanders RW; Ogohara C; Paulson JC; Feizi T; Scanlan CN; Wong CH; Moore JP; Olson WC; Ward AB; Poignard P; Schief WR; Burton DR; Wilson IA, A Potent and Broad Neutralizing Antibody Recognizes and Penetrates the HIV Glycan Shield. Science 2011, 334, 1097–1103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Alam SM; Aussedat B; Vohra Y; Meyerhoff RR; Cale EM; Walkowicz WE; Radakovich NA; Anasti K; Armand L; Parks R; Sutherland L; Scearce R; Joyce MG; Pancera M; Druz A; Georgiev IS; Von Holle T; Eaton A; Fox C; Reed SG; Louder M; Bailer RT; Morris L; Abdool-Karim SS; Cohen M; Liao HX; Montefiori DC; Park PK; Fernandez-Tejada A; Wiehe K; Santra S; Kepler TB; Saunders KO; Sodroski J; Kwong PD; Mascola JR; Bonsignori M; Moody MA; Danishefsky S; Haynes BF Mimicry of an HIV Broadly Neutralizing Antibody Epitope with a Synthetic Glycopeptide. Sci. Transl. Med. 2017, 9, pii: eaai7521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Cai H; Zhang R; Orwenyo J; Giddens J; Yang Q; LaBranche CC; Montefiori DC; Wang LX Multivalent Antigen Presentation Enhances the Immunogenicity of a Synthetic Three-Component HIV-1 V3 Glycopeptide Vaccine. ACS Cent. Sci. 2018, 4, 582–589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Kong R; Xu K; Zhou T; Acharya P; Lemmin T; Liu K; Ozorowski G; Soto C; Taft JD; Bailer RT; Cale EM; Chen L; Choi CW; Chuang GY; Doria-Rose NA; Druz A; Georgiev IS; Gorman J; Huang J; Joyce MG; Louder MK; Ma X; McKee K; O’Dell S; Pancera M; Yang Y; Blanchard SC; Mothes W; Burton DR; Koff WC; Connors M; Ward AB; Kwong PD; Mascola JR Fusion Peptide of HIV-1 as a Site of vulnerability to Neutralizing Antibody. Science, 2016, 352, 828–833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Eckert DM; Kim PS Mechanisms of Viral Membrane Fusion and its Inhibition. Annu. Rev. Biochem. 2001, 70, 777–810. [DOI] [PubMed] [Google Scholar]
- 82.Dai Z; Tao Y; Liu N; Brenowitz MD; Girvin ME; Lai JR, Conditional Trimerization and Lytic Activity of HIV-1 gp41 Variants Containing the Membrane-Associated Segments. Biochemistry 2015, 54, 1589–1599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Cheng C; Xu K; Kong R; Chuang GY; Corrigan AR; Geng H; Hill KR; Jafari AJ; O’Dell S; Ou L; Rawi R; Rowshan AP; Sarfo EK; Sastry M; Saunders KO; Schmidt SD; Wang S; Wu W; Zhang B; Doria-Rose NA; Haynes BF; Scorpio DG; Shapiro L; Mascola JR; Kwong PD, Consistent Elicitation of Cross-clade HIV-neutralizing Responses Achieved in Guinea Pigs after Fusion Peptide Priming By Repetitive Envelope Trimer Boosting. PLoS One 2019, 14, e0215163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Xu K; Acharya P; Kong R; Cheng C; Chuang GY; Liu K; Louder MK; O’Dell S; Rawi R; Sastry M; Shen CH; Zhang B; Zhou T; Asokan M; Bailer RT; Chambers M; Chen X; Choi CW; Dandey VP; Doria-Rose NA; Druz A; Eng ET; Farney SK; Foulds KE; Geng H; Georgiev IS; Gorman J; Hill KR; Jafari AJ; Kwon YD; Lai YT; Lemmin T; McKee K; Ohr TY; Ou L; Peng D; Rowshan AP; Sheng Z; Todd JP; Tsybovsky Y; Viox EG; Wang Y; Wei H; Yang Y; Zhou AF; Chen R; Yang L; Scorpio DG; McDermott AB; Shapiro L; Carragher B; Potter CS; Mascola JR; Kwong PD Epitope-based Vaccine Design Yields Fusion Peptide-Directed Antibodies that Neutralize Diverse Strains of HIV-1. Nat. Med. 2018, 24, 857–867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Taylor CA; Greenlund SF; McGuire LC; Lu H; Croft JB Deaths from Alzheimer’s Disease—United States, 1999–2014. MMWR Morb. Mort. Wkly. Rep. 2017, 66, 521–526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Prince M; Comas-Herrera A; Knapp M; Guerchet M; Karagiannidou M World Alzheimer Report 2016: Improving Healthcare for People Living with Dementia: Coverage, Quality and Costs Now and in the Future. 2016. Alzheimer’s Disease International: https://www.alz.co.uk/research/world-report-2016. Accessed on 11/26/19. [Google Scholar]
- 87.Selkoe DJ; Hardy J The Amyloid Hypothesis of Alzheimer’s Disease at 25 Years. EMBO Mol. Med. 2016, 8, 595–608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Lu JX; Qiang W; Yau WM; Schwieters CD; Meredith SC; Tycko R Molecular Structure of Beta-Amyloid Fibrils in Alzheimer’s Disease Brain Tissue. Cell 2013, 154, 1257–1268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Gremer L; Scholzel D; Schenk C; Reinartz E; Labahn J; Ravelli RBG; Tusche M; Lopez-Iglesias C; Hoyer W; Heise H; Willbold D; Schroder GF Fibril Structure of Amyloid-Beta(1–42) by Cryo-electron Microscopy. Science 2017, 358, 116–119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Hoyer W; Grönwall C; Jonsson A; Ståhl S; Härd T Stabilization of a β-hairpin in Monomeric Alzheimer’s Amyloid-β Peptide Inhibits Amyloid Formation. Proc. Natl. Acad. Sci. USA 2008, 105, 5099–5104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Serra-Batiste M; Ninot-Pedrosa M; Bayoumi M; Gairí M; Maglia G; Carulla N, Aβ42 Assembles into Specific β-barrel Pore-Forming Oligomers In Membrane-Mimicking Environments. Proc. Natl. Acad. Sci. USA 2016, 113, 10866–10871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Kreutzer AG; Hamza IL; Spencer RK; Nowick JS \ X-ray Crystallographic Structures of a Trimer, Dodecamer, and Annular Pore Formed by an Abeta17–36 Beta-hairpin. J. Am. Chem. Soc. 2016, 138, 4634–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Kreutzer AG; Yoo S; Spencer RK; Nowick JS Stabilization, Assembly, and Toxicity of Trimers Derived from Abeta. J. Am. Chem. Soc. 2017, 139, 966–975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Wisniewski T; Goñi F Immunotherapeutic Approaches for Alzheimer’s Disease. Neuron 2015, 85, 1162–1176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Novak P; Kontsekova E; Zilka N; Novak M Ten Years of Tau-Targeted Immunotherapy: The Path Walked and the Roads Ahead. Front. Neurosci. 2018, 12, 798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Fitzpatrick AW; Falcon B; He S; Murzin AG; Murshudov G; Garringer HJ; Crowther RA; Ghetti B; Goedert M; Scheres SH Cryo-EM Structures of Tau Filaments from Alzheimer’s Disease. Nature 2017, 547, 185–190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Nelson PT; Alafuzoff I; Bigio EH; Bouras C; Braak H; Cairns NJ; Castellani RJ; Crain BJ; Davies P; Del Tredici K; Duyckaerts C; Frosch MP; Haroutunian V; Hof PR; Hulette CM; Hyman BT; Iwatsubo T; Jellinger KA; Jicha GA; Kovari E; Kukull WA; Leverenz JB; Love S; Mackenzie IR; Mann DM; Masliah E; McKee AC; Montine TJ; Morris JC; Schneider JA; Sonnen JA; Thal DR; Trojanowski JQ; Troncoso JC; Wisniewski T; Woltjer RL; Beach TG Correlation of Alzheimer disease neuropathologic changes with Cognitive Status: A Review of the Literature. J. Neuropath. Exp. Neurol. 2012, 71, 362–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Wiessner C; Wiederhold K-H; Tissot AC; Frey P; Danner S; Jacobson LH; Jennings GT; Lüönd R; Ortmann R; Reichwald J; Zurini M; Mir A; Bachmann MF; Staufenbiel M The Second-Generation Active Aβ Immunotherapy CAD106 Reduces Amyloid Accumulation in APP Transgenic Mice While Minimizing Potential Side Effects. J. Neurosci. 2011, 31, 9323–9331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Winblad B; Andreasen N; Minthon L; Floesser A; Imbert G; Dumortier T; Maguire RP; Blennow K; Lundmark J; Staufenbiel M; Orgogozo J-M; Graf A Safety, tolerability, and Antibody Response of Active Aβ Immunotherapy with CAD106 in Patients with Alzheimer’s Disease: Randomised, Double-blind, Placebo-controlled, First-in-human study. Lancet Neurol. 2012, 11, 597–604. [DOI] [PubMed] [Google Scholar]
- 100.Farlow MR; Andreasen N; Riviere M-E; Vostiar I; Vitaliti A; Sovago J; Caputo A; Winblad B; Graf A Long-term Treatment with Active Aβ Immunotherapy with CAD106 in Mild Alzheimer’s Disease. Alzheimers Res. Ther 2015, 7, 23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Vandenberghe R; Riviere M-E; Caputo A; Sovago J; Maguire RP; Farlow M; Marotta G; Sanchez-Valle R; Scheltens P; Ryan JM; Graf A, Active Aβ immunotherapy CAD106 in Alzheimer’s disease: A phase 2b study. Alzheimer’s & dementia (New York, N. Y.) 2016, 3 (1), 10–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Wang CY; Finstad CL; Walfield AM; Sia C; Sokoll KK; Chang T-Y; Fang XD; Hung CH; Hutter-Paier B; Windisch M Site-specific UBITh® Amyloid-β Vaccine for Immunotherapy of Alzheimer’s Disease. Vaccine 2007, 25, 3041–3052. [DOI] [PubMed] [Google Scholar]
- 103.Wang CY; Wang P-N; Chiu M-J; Finstad CL; Lin F; Lynn S; Tai Y-H; De Fang X; Zhao K; Hung C-H; Tseng Y; Peng W-J; Wang J; Yu C-C; Kuo B-S; Frohna PA UB-311, A Novel UBITh(®) Amyloid β Peptide Vaccine for Mild Alzheimer’s Disease. Alzheimers Dement. 2017, 3, 262–272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Davtyan H; Ghochikyan A; Petrushina I; Hovakimyan A; Davtyan A; Poghosyan A; Marleau AM; Movsesyan N; Kiyatkin A; Rasool S; Larsen AK; Madsen PJ; Wegener KM; Ditlevsen DK; Cribbs DH; Pedersen LO; Agadjanyan MG Immunogenicity, Efficacy, Safety, and Mechanism of Action of Epitope Vaccine (Lu AF20513) for Alzheimer’s Disease: Prelude to a Clinical Trial. J. Neurosci. 2013, 33, 4923–4934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Lacosta A-M; Pascual-Lucas M; Pesini P; Casabona D; Pérez-Grijalba V; Marcos-Campos I; Sarasa L; Canudas J; Badi H; Monleón I; San-José I; Munuera J; Rodríguez-Gómez O; Abdelnour C; Lafuente A; Buendía M; Boada M; Tárraga L; Ruiz A; Sarasa M Safety, Tolerability and Immunogenicity of an Active Anti-Aβ40 Vaccine (ABvac40) in Patients with Alzheimer’s Disease: A Randomised, Double-Blind, Placebo-controlled, Phase I Trial. Alzheimers Res. Ther 2018, 10, 12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Hickman DT; López-Deber MP; Ndao DM; Silva AB; Nand D; Pihlgren M; Giriens V; Madani R; St-Pierre A; Karastaneva H; Nagel-Steger L; Willbold D; Riesner D; Nicolau C; Baldus M; Pfeifer A; Muhs A, Sequence-independent Control of Peptide Conformation in Liposomal Vaccines for Targeting Protein Misfolding Diseases. J. Biol. Chem. 2011, 286, 13966–13976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Theunis C; Crespo-Biel N; Gafner V; Pihlgren M; López-Deber MP; Reis P; Hickman DT; Adolfsson O; Chuard N; Ndao DM; Borghgraef P; Devijver H; Van Leuven F; Pfeifer A; Muhs A, Efficacy and Safety of A Liposome-Based Vaccine against Protein Tau, Assessed in Tau.P301L Mice That Model Tauopathy. PLoS One 2013, 8, e72301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Kontsekova E; Zilka N; Kovacech B; Novak P; Novak M First-in-man Tau Vaccine Targeting Structural Determinants Essential for Pathological Tau–Tau Interaction Reduces tau Oligomerisation and Neurofibrillary Degeneration in an Alzheimer’s Disease Model. Alzheimers Res. Ther. 2014, 6, 44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Novak P; Schmidt R; Kontsekova E; Kovacech B; Smolek T; Katina S; Fialova L; Prcina M; Parrak V; Dal-Bianco P; Brunner M; Staffen W; Rainer M; Ondrus M; Ropele S; Smisek M; Sivak R; Zilka N; Winblad B; Novak M FUNDAMANT: An Interventional 72-week Phase 1 Follow-up Study of AADvac1, An Active Immunotherapy against Tau Protein Pathology in Alzheimer’s Disease. Alzheimers Res. Ther. 2018, 10, 108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Liu Y-H; Wang Y-R; Xiang Y; Zhou H-D; Giunta B; Mañucat-Tan NB; Tan J; Zhou X-F; Wang Y-J Clearance of Amyloid-Beta in Alzheimer’s Disease: Shifting the Action Site from Center to Periphery. Mol. Neurobiol. 2015, 51, 1–7. [DOI] [PubMed] [Google Scholar]
- 111.Wang CY; Walfield AM, Site-specific Peptide Vaccines for Immunotherapy and Immunization against Chronic Diseases, Cancer, Infectious Diseases, and for Veterinary Applications. Vaccine 2005, 23, 2049–56. [DOI] [PubMed] [Google Scholar]
- 112.Naslund J; Schierhorn A; Hellman U; Lannfelt L; Roses AD; Tjernberg LO; Silberring J; Gandy SE; Winblad B; Greengard P; Nordstedt C; Terenius L Relative abundance of Alzheimer Abeta Amyloid Peptide Variants in Alzheimer Disease and Normal Aging. Proc. Natl. Acad. USA 1994, 91, 8378–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Lacosta AM; Insua D; Badi H; Pesini P; Sarasa M Neurofibrillary Tangles of Abetax-40 in Alzheimer’s Disease Brains. J. Alzheimers Dis. 2017, 58, 661–667. [DOI] [PubMed] [Google Scholar]
- 114.Kontsekova E; Zilka N; Kovacech B; Skrabana R; Novak M Identification of Structural Determinants on Tau Protein Essential for its Pathological Function: Novel Therapeutic Target for Tau Immunotherapy in Alzheimer’s Disease. Alzheimers Res. Ther. 2014, 6, 45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Stover PJ Physiology of Folate and Vitamin B12 in Health and Disease. Nutr. Rev. 2004, 62, S3–12. [DOI] [PubMed] [Google Scholar]
- 116.Wani NA; Hamid A; Kaur J Folate Status in Various Pathophysiological Conditions. IUBMB Life 2008, 60, 834–842. [DOI] [PubMed] [Google Scholar]
- 117.Vergote IB; Marth C; Coleman RL Role of the Folate Receptor in Ovarian Cancer Treatment: Evidence, Mechanism, and Clinical Implications. Cancer Metastasis Rev 2015, 34, 41–52. [DOI] [PubMed] [Google Scholar]
- 118.Elwood PC Molecular Cloning and Characterization of the Human Folate-binding Protein cDNA from Placenta and Malignant Tissue Culture (KB) Cells. J. Biol. Chem. 1989, 264, 14893–14901. [PubMed] [Google Scholar]
- 119.Shen F; Wu M; Ross JF; Miller D; Ratnam M Folate Receptor Type Gamma is Primarily a Secretory Protein Due to Lack of an Efficient Signal for Glycosylphosphatidylinositol Modification: Protein Characterization and Cell Type Specificity. Biochemistry 1995, 34, 5660–5665. [DOI] [PubMed] [Google Scholar]
- 120.Chancy CD; Kekuda R; Huang W; Prasad PD; Kuhnel JM; Sirotnak FM; Roon P; Ganapathy V; Smith SB Expression And Differential Polarization of the Reduced-Folate Transporter-1 and the Folate Receptor Alpha in Mammalian Retinal Pigment Epithelium. J. Biol. Chem. 2000, 275, 20676–20684. [DOI] [PubMed] [Google Scholar]
- 121.Parker N; Turk MJ; Westrick E; Lewis JD; Low PS; Leamon CP Folate Receptor Expression in Carcinomas and Normal Tissues Determined by a Quantitative Radioligand Binding Assay. Anal. Biochem. 2005, 338, 284–93. [DOI] [PubMed] [Google Scholar]
- 122.Smith SB; Kekuda R; Gu X; Chancy C; Conway SJ; Ganapathy V Expression of Folate Receptor Alpha in the Mammalian Retinol Pigmented Epithelium and Retina. Invest. Ophthalmol. Vis. Sci. 1999, 40, 840–848. [PubMed] [Google Scholar]
- 123.Solanky N; Requena Jimenez A; D’Souza SW; Sibley CP; Glazier JD Expression of Folate Transporters in Human Placenta and Implications for Homocysteine Metabolism. Placenta 2010, 31, 134–43. [DOI] [PubMed] [Google Scholar]
- 124.Siu MK; Kong DS; Chan HY; Wong ES; Ip PP; Jiang L; Ngan HY; Le XF; Cheung AN Paradoxical Impact of Two Folate Receptors, FRalpha and RFC, in Ovarian Cancer: Effect on Cell Proliferation, Invasion and Clinical Outcome. PLoS One 2012, 7, e47201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Peoples GE; Anderson BW; Lee TV; Murray JL; Kudelka AP; Wharton JT; Ioannides CG Vaccine Implications of Folate Binding Protein, A Novel Cytotoxic T Lymphocyte-recognized Antigen System in Epithelial Cancers. Clin. Cancer Res. 1999, 5, 4214–23. [PubMed] [Google Scholar]
- 126.Bagnoli M; Canevari S; Figini M; Mezzanzanica D; Raspagliesi F; Tomassetti A; Miotti S A Step Further in Understanding the Biology of the Folate Receptor in Ovarian Carcinoma. Gynecol. Oncol. 2003, 88, S140–144. [DOI] [PubMed] [Google Scholar]
- 127.Peoples GE; Anderson BW; Fisk B; Kudelka AP; Wharton JT; Ioannides CG, Ovarian Cancer-associated Lymphocyte Recognition of Folate Binding Protein Peptides. Ann. Surg. Oncol. 1998, 5, 743–750. [DOI] [PubMed] [Google Scholar]
- 128.Siegel RL; Miller KD; Jemal A Cancer Statistics, 2018. CA Cancer J. Clin 2018, 68, 7–30. [DOI] [PubMed] [Google Scholar]
- 129.Siegel RL; Miller KD; Jemal A Cancer statistics, 2019. CA Cancer J. Clin 2019, 69, 7–34. [DOI] [PubMed] [Google Scholar]
- 130.Knutson KL; Krco CJ; Erskine CL; Goodman K; Kelemen LE; Wettstein PJ; Low PS; Hartmann LC; Kalli KR T-cell Immunity to The Folate Receptor Alpha is Prevalent in Women with Breast or Ovarian Cancer. J. Clin. Oncol. 2006, 24, 4254–4261. [DOI] [PubMed] [Google Scholar]
- 131.Kalli KR; Block MS; Kasi PM; Erskine CL; Hobday TJ; Dietz A; Padley D; Gustafson MP; Shreeder B; Puglisi-Knutson D; Visscher DW; Mangskau TK; Wilson G; Knutson KL Folate Receptor Alpha Peptide Vaccine Generates Immunity in Breast and Ovarian Cancer Patients. Clin. Cancer Res. 2018, 24, 3014–3025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Yarden Y; Sliwkowski MX Untangling the ErbB Signalling Network. Nat. Rev. Mol. Cell Biol. 2001, 2, 127–137. [DOI] [PubMed] [Google Scholar]
- 133.Landgraf R HER2 therapy. HER2 (ERBB2): Functional Diversity from Structurally Conserved Building Blocks. Breast Cancer Res. 2007, 9, 202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Brennan PJ; Kumogai T; Berezov A; Murali R; Greene MI HER2/neu: Mechanisms of Dimerization/Oligomerization. Oncogene 2000, 19, 6093–6101. [DOI] [PubMed] [Google Scholar]
- 135.Zhou BP; Hung M-C Dysregulation of Cellular Signaling by HER2/neu in Breast Cancer. Semin. Oncol. 2003, 30, 38–48. [DOI] [PubMed] [Google Scholar]
- 136.Moasser MM The Oncogene HER2: Its Signaling and Transforming Functions and its Role in Human Cancer Pathogenesis. Oncogene 2007, 26, 6469–6487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Gutierrez C; Schiff R HER2: Biology, Detection, and Clinical Implications. Arch. Pathol. Lab. Med. 2011, 135, 55–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Yan M; Schwaederle M; Arguello D; Millis SZ; Gatalica Z; Kurzrock R HER2 Expression Status in Diverse Cancers: Review of Results from 37,992 Patients. Cancer Metastasis Rev. 2015, 34, 157–164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Nahta R; Esteva FJ, Trastuzumab: Triumphs and Tribulations. Oncogene 2007, 26, 3637–3643. [DOI] [PubMed] [Google Scholar]
- 140.Cho H-S; Mason K; Ramyar KX; Stanley AM; Gabelli SB; Denney DW; Leahy DJ Structure of the Extracellular Region of HER2 Alone and In Complex with the Herceptin Fab. Nature 2003, 421, 756–760. [DOI] [PubMed] [Google Scholar]
- 141.Rimawi MF; Schiff R; Osborne CK Targeting HER2 for the Treatment of Breast Cancer. Annu. Rev. Med. 2015, 66, 111–28. [DOI] [PubMed] [Google Scholar]
- 142.Swain SM; Baselga J; Kim S-B; Ro J; Semiglazov V; Campone M; Ciruelos E; Ferrero J-M; Schneeweiss A; Heeson S; Clark E; Ross G; Benyunes MC; Cortés J Pertuzumab, Trastuzumab, and Docetaxel in HER2-Positive Metastatic Breast Cancer. New Engl. J. Med. 2015, 372, 724–734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Fisk B; Blevins TL; Wharton JT; Ioannides CG Identification of an Immunodominant Peptide of HER-2/neu Protooncogene Recognized by Ovarian Tumor-Specific Cytotoxic T Lymphocyte Lines. J. Exp. Med. 1995, 181, 2109–2117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Kono K; Takahashi A; Sugai H; Fujii H; Choudhury AR; Kiessling R; Matsumoto Y Dendritic Cells Pulsed with HER-2/neu-derived Peptides Can Induce Specific T-Cell Responses in Patients with Gastric Cancer. Clin. Cancer Res. 2002, 8, 3394–400. [PubMed] [Google Scholar]
- 145.Brossart P; Stuhler G; Flad T; Stevanovic S; Rammensee H-G; Kanz L; Brugger W Her-2/neu-derived Peptides Are Tumor-Associated Antigens Expressed by Human Renal Cell and Colon Carcinoma Lines and Are Recognized by In Vitro Induced Specific Cytotoxic T Lymphocytes. Cancer Res. 1998, 58, 732–736. [PubMed] [Google Scholar]
- 146.Holmes JP; Gates JD; Benavides LC; Hueman MT; Carmichael MG; Patil R; Craig D; Mittendorf EA; Stojadinovic A; Ponniah S; Peoples GE Optimal Dose and Schedule of an HER-2/neu (E75) Peptide Vaccine to Prevent Breast Cancer Recurrence. Cancer 2008, 113, 1666–1675. [DOI] [PubMed] [Google Scholar]
- 147.Mittendorf EA; Clifton GT; Holmes JP; Clive KS; Patil R; Benavides LC; Gates JD; Sears AK; Stojadinovic A; Ponniah S; Peoples GE Clinical Trial Results of the HER-2/neu (E75) Vaccine to Prevent Breast Cancer Recurrence in High-Risk Patients: From US Military Cancer Institute Clinical Trials Group Study I-01 and I-02. Cancer 2012, 118, 2594–2602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Holmes JP; Clifton GT; Patil R; Benavides LC; Gates JD; Stojadinovic A; Mittendorf EA; Ponniah S; Peoples GE Use of Booster Inoculations to Sustain the Clinical Effect of an Adjuvant Breast Cancer Vaccine. Cancer 2011, 117, 463–471. [DOI] [PubMed] [Google Scholar]
- 149.Mittendorf EA; Lu B; Melisko M; Price Hiller J; Bondarenko I; Brunt AM; Sergii G; Petrakova K; Peoples GE Efficacy and Safety Analysis of Nelipepimut-S Vaccine to Prevent Breast Cancer Recurrence: A Randomized, Multicenter, Phase III Clinical Trial. Clin. Cancer Res. 2019, 25, 4248–4254. [DOI] [PubMed] [Google Scholar]
- 150.Krasniqi E; Barchiesi G; Pizzuti L; Mazzotta M; Venuti A; Maugeri-Saccà M; Sanguineti G; Massimiani G; Sergi D; Carpano S; Marchetti P; Tomao S; Gamucci T; De Maria R; Tomao F; Natoli C; Tinari N; Ciliberto G; Barba M; Vici P Immunotherapy in HER2-Positive Breast Cancer: State of the Art and Future Perspectives. J. Hematol. Oncol. 2019, 12, 111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Clifton GT; Mittendorf EA; Peoples GE Adjuvant HER2/neu Peptide Cancer Vaccines in Breast Cancer. Immunotherapy 2015, 7, 1159–1168. [DOI] [PubMed] [Google Scholar]
- 152.Peoples GE; Goedegebuure PS; Smith R; Linehan DC; Yoshino I; Eberlein TJ Breast and Ovarian Cancer-Specific Cytotoxic T Lymphocytes Recognize the Same HER2/neu-derived Peptide. Proc. Natl. Acad. Sci. USA 1995, 92, 432–436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Mittendorf EA; Storrer CE; Foley RJ; Harris K; Jama Y; Shriver CD; Ponniah S; Peoples GE Evaluation of the HER2/neu-derived Peptide GP2 for Use in a Peptide-based Breast Cancer Vaccine Trial. Cancer 2006, 106, 2309–2317. [DOI] [PubMed] [Google Scholar]
- 154.Mittendorf EA; Ardavanis A; Litton JK; Shumway NM; Hale DF; Murray JL; Perez SA; Ponniah S; Baxevanis CN; Papamichail M; Peoples GE Primary Analysis of a Prospective, Randomized, Single-Blinded Phase II Trial Evaluating the HER2 peptide GP2 vaccine in Breast Cancer Patients to Prevent Recurrence. Oncotarget 2016, 7, 66192–66201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Jasinska J; Wagner S; Radauer C; Sedivy R; Brodowicz T; Wiltschke C; Breiteneder H; Pehamberger H; Scheiner O; Wiedermann U; Zielinski CC Inhibition of Tumor Cell Growth by Antibodies Induced After Vaccination with Peptides Derived From the Extracellular Domain of Her-2/neu. Int. J. Cancer 2003, 107, 976–83. [DOI] [PubMed] [Google Scholar]
- 156.Wagner S; Jasinska J; Breiteneder H; Kundi M; Pehamberger H; Scheiner O; Zielinski CC; Wiedermann U Delayed Tumor Onset and Reduced Tumor Growth Progression after Immunization with a Her-2/neu Multi-peptide Vaccine and IL-12 in c-neu Transgenic Mice. Breast Cancer Res. Treat. 2007, 106, 29–38. [DOI] [PubMed] [Google Scholar]
- 157.Wiedermann U; Wiltschke C; Jasinska J; Kundi M; Zurbriggen R; Garner-Spitzer E; Bartsch R; Steger G; Pehamberger H; Scheiner O; Zielinski CC A Virosomal Formulated Her-2/neu Multi-peptide Vaccine Induces Her-2/neu-specific Immune Responses in patients with Metastatic Breast Cancer: A Phase I Study. Breast Cancer Res. Treat. 2010, 119, 673–683. [DOI] [PubMed] [Google Scholar]
- 158.Tobias J; Jasinska J; Baier K; Kundi M; Ede N; Zielinski C; Wiedermann U Enhanced and Long Term Immunogenicity of a Her-2/neu Multi-epitope Vaccine Conjugated to the carrier CRM197 in Conjunction with the adjuvant Montanide. BMC Cancer 2017, 17, 118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Kaumaya PT; Foy KC; Garrett J; Rawale SV; Vicari D; Thurmond JM; Lamb T; Mani A; Kane Y; Balint CR; Chalupa D; Otterson GA; Shapiro CL; Fowler JM; Grever MR; Bekaii-Saab TS; Carson WE 3rd Phase I Active Immunotherapy with combination of Two Chimeric, Human Epidermal Growth Factor Receptor 2, B-cell Epitopes Fused to a Promiscuous T-cell Epitope in Patients with Metastatic and/or Recurrent Solid Tumors. J. Clin. Oncol. 2009, 27, 5270–5277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Dakappagari NK; Pyles J; Parihar R; Carson WE; Young DC; Kaumaya PT A Chimeric Multi-human Epidermal Growth Factor Receptor-2 B Cell Epitope Peptide Vaccine Mediates Superior Antitumor Responses. J. Immunol. 2003, 170, 4242–4253. [DOI] [PubMed] [Google Scholar]
- 161.Dakappagari NK; Douglas DB; Triozzi PL; Stevens VC; Kaumaya PT Prevention of Mammary Tumors with a Chimeric HER-2 B-cell Epitope Peptide Vaccine. Cancer Res. 2000, 60 (14), 3782–9. [PubMed] [Google Scholar]
- 162.Franklin MC; Carey KD; Vajdos FF; Leahy DJ; de Vos AM; Sliwkowski MX Insights into ErbB Signaling From the Structure of the ErbB2-pertuzumab Complex. Cancer Cell 2004, 5, 317–328. [DOI] [PubMed] [Google Scholar]
- 163.Allen SD; Garrett JT; Rawale SV; Jones AL; Phillips G; Forni G; Morris JC; Oshima RG; Kaumaya PT, Peptide Vaccines of the HER-2/neu Dimerization Loop are Effective in Inhibiting Mammary Tumor Growth In Vivo. J. Immunol. 2007, 179, 472–482. [DOI] [PubMed] [Google Scholar]
- 164.Garrett JT; Rawale S; Allen SD; Phillips G; Forni G; Morris JC; Kaumaya PT Novel Engineered Trastuzumab Conformational Epitopes Demonstrate In Vitro and In Vivo Antitumor Properties against HER-2/neu. J. Immunol. 2007, 178, 7120–7131. [DOI] [PubMed] [Google Scholar]
- 165.Bekaii-Saab T; Wesolowski R; Ahn DH; Wu C; Mortazavi A; Lustberg M; Ramaswamy B; Fowler J; Wei L; Overholser J; Kaumaya PTP Phase I Immunotherapy Trial with Two Chimeric HER-2 B-Cell Peptide Vaccines Emulsified in Montanide ISA 720VG and Nor-MDP Adjuvant in Patients with Advanced Solid Tumors. Clin. Cancer Res. 2019, 25, 3495–3507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Bhoj VG; Arhontoulis D; Wertheim G; Capobianchi J; Callahan CA; Ellebrecht CT; Obstfeld AE; Lacey SF; Melenhorst JJ; Nazimuddin F; Hwang WT; Maude SL; Wasik MA; Bagg A; Schuster S; Feldman MD; Porter DL; Grupp SA; June CH; Milone MC Persistence of Long-lived Plasma Cells and Humoral Immunity in Individuals Responding to CD19-directed CAR T-cell Therapy. Blood 2016, 128, 360–370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Marshall HT; Djamgoz MBA Immuno-Oncology: Emerging Targets and Combination Therapies. Front. Oncol. 2018, 8, 315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Ilyas S; Yang JC Landscape of Tumor Antigens in T Cell Immunotherapy. J. Immunol. 2015, 195, 5117–5122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Duan F; Duitama J; Al Seesi S; Ayres CM; Corcelli SA; Pawashe AP; Blanchard T; McMahon D; Sidney J; Sette A; Baker BM; Mandoiu II; Srivastava PK Genomic and Bioinformatic Profiling of Mutational Neoepitopes Reveals New Rules to Predict Anticancer Immunogenicity. J. Exp. Med. 2014, 211, 2231–2248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Robbins PF; Lu YC; El-Gamil M; Li YF; Gross C; Gartner J; Lin JC; Teer JK; Cliften P; Tycksen E; Samuels Y; Rosenberg SA Mining exomic sequencing data to Identify Mutated Antigens Recognized by Adoptively Transferred Tumor-Reactive T Cells. Nature Med. 2013, 19, 747–752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Tran E; Turcotte S; Gros A; Robbins PF; Lu YC; Dudley ME; Wunderlich JR; Somerville RP; Hogan K; Hinrichs CS; Parkhurst MR; Yang JC; Rosenberg SA Cancer Immunotherapy Based on Mutation-specific CD4+ T Cells in a Patient with Epithelial Cancer. Science 2014, 344, 641–655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Pinilla C; Rubio-Godoy V; Dutoit V; Guillaume P; Simon R; Zhao Y; Houghten RA; Cerottini JC; Romero P; Valmori D Combinatorial Peptide Libraries as an Alternative Approach to the Identification of Ligands for Tumor-reactive Cytolytic T Lymphocytes. Cancer Res. 2001, 61, 5153–5160. [PubMed] [Google Scholar]
- 173.Rubio-Godoy V; Ayyoub M; Dutoit V; Servis C; Schink A; Rimoldi D; Romero P; Cerottini JC; Simon R; Zhao Y; Houghten RA; Pinilla C; Valmori D Combinatorial Peptide Library-based Identification of Peptide Ligands for Tumor-reactive Cytolytic T Lymphocytes of Unknown Specificity. Eur. J. Immunol. 2002, 32, 2292–2299. [DOI] [PubMed] [Google Scholar]
- 174.Rubio-Godoy V; Dutoit V; Zhao Y; Simon R; Guillaume P; Houghten R; Romero P; Cerottini JC; Pinilla C; Valmori D Positional Scanning-synthetic Peptide Library-based Analysis of Self- and Pathogen-derived Peptide Cross-reactivity with Tumor-reactive Melan-A-specific CTL. J. Immunol. 2002, 169, 5696–5707. [DOI] [PubMed] [Google Scholar]
- 175.Cole DK; Edwards ES; Wynn KK; Clement M; Miles JJ; Ladell K; Ekeruche J; Gostick E; Adams KJ; Skowera A; Peakman M; Wooldridge L; Price DA; Sewell AK Modification of MHC Anchor Residues Generates Heteroclitic Peptides that Alter TCR Binding and T Cell Recognition. J. Immunol. 2010, 185, 2600–2610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Gee MH; Han A; Lofgren SM; Beausang JF; Mendoza JL; Birnbaum ME; Bethune MT; Fischer S; Yang X; Gomez-Eerland R; Bingham DB; Sibener LV; Fernandes RA; Velasco A; Baltimore D; Schumacher TN; Khatri P; Quake SR; Davis MM; Garcia KC Antigen Identification for Orphan T Cell Receptors Expressed on Tumor-Infiltrating Lymphocytes. Cell 2018, 172, 549–563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Tanaka M; Tashiro H; Omer B; Lapteva N; Ando J; Ngo M; Mehta B; Dotti G; Kinchington PR; Leen AM; Rossig C; Rooney CM Vaccination Targeting Native Receptors to Enhance the Function and Proliferation of Chimeric Antigen Receptor (CAR)-Modified T Cells. Clin Cancer Res. 2017, 23, 3499–3509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Wang X; Wong CW; Urak R; Mardiros A; Budde LE; Chang WC; Thomas SH; Brown CE; La Rosa C; Diamond DJ; Jensen MC; Nakamura R; Zaia JA; Forman SJ CMVpp65 Vaccine Enhances the Antitumor Efficacy of Adoptively Transferred CD19-Redirected CMV-Specific T Cells. Clin. Cancer Res. 2015, 21, 2993–3002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Ma L; Dichwalkar T; Chang JYH; Cossette B; Garafola D; Zhang AQ; Fichter M; Wang C; Liang S; Silva M; Kumari S; Mehta NK; Abraham W; Thai N; Li N; Wittrup KD; Irvine DJ, Enhanced CAR-T Cell Activity Against Solid Tumors by Vaccine Boosting through the Chimeric Receptor. Science 2019, 365, 162–168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Liu H; Moynihan KD; Zheng Y; Szeto GL; Li AV; Huang B; Van Egeren DS; Park C; Irvine DJ Structure-based Programming of Lymph-node Targeting in Molecular Vaccines. Nature 2014, 507, 519–522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Liu H; Kwong B; Irvine DJ Membrane Anchored Immunostimulatory Oligonucleotides for In Vivo Cell Modification and Localized Immunotherapy. Angew. Chem. Int. Ed. 2011, 50, 7052–7055. [DOI] [PMC free article] [PubMed] [Google Scholar]


