Abstract
Purpose
To analyze the impact on patient outcome of ventilator-associated events (VAEs) as defined by the Centers for Disease Control and Prevention (CDC) in 2008, 2013, and the correlation with ventilator-associated pneumonia (VAP) or tracheobronchitis (VAT).
Methods
This was a prospective, observational, multicenter, international study conducted at 13 intensive care units (ICUs); thirty consecutive adults mechanically ventilated for ≥ 48 h per site were eligible, with daily follow-up being recorded in a collaborative web database; VAEs were assessed using the 2013 CDC classification and its 2015 update.
Results
A total of 2856 ventilator days in 244 patients were analyzed, identifying 33 VAP and 51 VAT episodes; 30-day ICU mortality was significantly higher (42.8 vs. 19.6%, p < 0.007) in patients with VAP than in those with VAT. According to the 2013 CDC definitions, 117 VAEs were identified: 113 (96%) were infection-related ventilator-associated complication-plus (IVAC-plus), while possible ventilator-associated pneumonia (PVAP) was found in 64 (56.6%) of them. VAE increased the number of ventilator days and prolonged ICU and hospital LOS (by 5, 11, and 12 days, respectively), with a trend towards increased 30-day mortality (43 vs 28%, p = 0.06). Most episodes (26, 55%) classified as IVAC-plus without PVAP criteria were due to atelectasis. PVAP significantly increased (p < 0.05) ventilator days as well as ICU and hospital LOS (by 10.5, 14, and 13 days, respectively). Only 24 (72.7%) of VAP and 15 (29.4%) of VAT episodes met IVAC-plus criteria.
Conclusions
Respiratory infections (mainly VAT) were the most common complication. VAE algorithms only identified events with surrogates of severe oxygenation deterioration. As a consequence, IVAC definitions missed one fourth of the episodes of VAP and three fourths of the episodes of VAT. Identifying VAT (often missed by IVAC-plus criteria) is important, as VAP and VAT have different impacts on mortality.
Electronic supplementary material
The online version of this article (10.1007/s00134-018-5269-7) contains supplementary material, which is available to authorized users.
Keywords: Ventilator-associated pneumonia, Ventilator-associated tracheobronchitis, Ventilator-associated events, Surveillance, Hypoxemia
Introduction
Despite progress in the treatment of infectious diseases, ventilator-associated pneumonia (VAP) remains a major infection among adult critically ill patients with overall attributable mortality of 13% [1, 2]. Infections of the lower respiratory tract are the main reason for antibiotic prescription in the intensive care unit (ICU), and they are associated with increased length of stay (LOS) and costs [1, 3, 4].
A global consensus on VAP diagnosis has been difficult to achieve, largely due to the high variability in the interpretation of current definitions and the use of different tests for microbiological confirmation. Moreover, many complications in adult patients receiving mechanical ventilation, such as acute respiratory distress syndrome (ARDS), pulmonary edema, atelectasis, and pulmonary embolisms, may be misinterpreted as respiratory infections due to the lack of specificity of certain diagnostic criteria such as chest X-ray [5]. Previous reports have highlighted the poor correlation between clinical diagnosis of VAP and the histopathology findings [6, 7]. All these limitations have implications for clinical practice, surveillance initiatives, and the design of preventive strategies [8].
In 2013 the Centers for Disease Control and Prevention (CDC) proposed a new diagnostic algorithm for ventilator-associated events (VAEs) based on more objective criteria and easily measurable clinical features. It divided the causes of respiratory worsening in critically ill patients into four tiers: ventilator associated complication (VAC), iInfection-related ventilator associated complication (IVAC), probable ventilator-associated pneumonia, and possible ventilator associated pneumonia. In the 2015 update of this algorithm, probable and possible ventilator-associated pneumonia were amalgamated as possible ventilator-associated pneumonia (PVAP) and a new category, infection-related ventilator-associated complication-plus (IVAC-plus) was created.
The EU-VAE project was developed with the aim of analyzing the different definitions, incidences, risk factors, outcomes and impact of VAEs in non-USA ICUs, according to the 2013 CDC definition and its 2015 update [3, 9]. Preliminary data were reported at ECCMID 2016 and 2017 [10, 11].
Patients and methods
A prospective, observational, international, multicenter study was conducted in 13 ICUs from eight countries (Australia, France, Greece, Iran, Italy, Slovenia, Spain, and Turkey) with the capacity for treating adults with medical or surgical conditions or trauma. Nine (69%) of these ICUs were located in university hospitals.
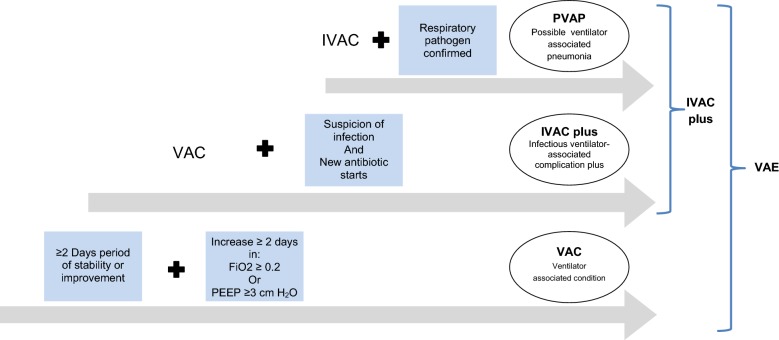
Persons over 18 years of age who had been on mechanical ventilation for more than 48 h were eligible; the first 30 subjects from each participating center were followed up daily for 30 days. The exclusion criteria were mechanical ventilation for less than 48 h, age below 18, and presence of respiratory viral infection. Only the first episode of VAE in each patient was included in the study. Subjects with incomplete follow-up data were eliminated. Extubation, ICU discharge, or death in the ICU were recorded as outcome end-points. The following characteristics were recorded: age, sex, weight, APACHE II score [12], immunosuppressive therapy, reason for and site of intubation, hospital and ICU LOS, implementation of preventive measures for ventilator-associated respiratory infections (VARI), clinical and laboratory parameters of inflammatory response [13], microbiological data, use of antibiotics, sedation and medical paralysis, fluid balance, and need for tracheostomy during the ICU stay. Daily follow-up data were recorded in a collaborative web database (http://compartint.net/euvae/). The current adult definitions for VAEs implemented in the CDC 2013 National Healthcare Safety Network and its 2015 update [3, 14] were assessed (Table 1). There are three definition tiers within the VAE algorithm: (1) VAE, (2) IVAC, and (3) PVAP (Fig. 1). Cases of VAP or ventilator-associated tracheobronchitis (VAT) were recorded according to the 2008 CDC criteria [15]. In brief, VAP was defined as the presence of a new or progressive and persistent pulmonary infiltrate, consolidation, or cavitation plus at least three of the following in a patient under mechanical ventilation for > 48 h: (1) temperature ≥ 38 °C or ≤ 36 °C with no other recognized cause; (2) leukocyte count ≥ 12,000/mm3 or < 4000/mm3; (3) new onset of purulent tracheal secretions or change in character of sputum, or increased respiratory secretions, or increased suctioning requirements; (4) new-onset or worsening dyspnea or tachypnea; (5) wheezing, rales, or bronchial breath sounds; (6) worsening gas exchange, increased oxygen requirement, or increased ventilator demand; (7) altered mental status in a patient > 70 years old. The definition of VAT [16, 17] was based on the absence of clinical and radiographic evidence of pneumonia and presence of the following criteria: positive culture obtained by deep tracheal aspirate plus at least two of these signs in a patient under mechanical ventilation for > 48 h: fever > 38 °C, new or increased purulent tracheal secretions, rhonchi, or wheezing (Table 1). Quantitative definitions of significant growth in cultures and purulent respiratory secretions are detailed in the electronic supplementary material (ESM) appendixes 2 and 3. Atelectasis was defined as collapse of a part of the lung due to a decrease in the amount of air in the alveoli, resulting in volume loss and increased density. Pulmonary edema was diagnosed based on air space opacification in a classic batwing distribution, possibly accompanied by air bronchograms, with peribronchial cuffing and perihilar haze, septal lines (Kerley lines), and thickening of interlobar fissures. The final diagnosis was established by the investigator based on the radiologist’s interpretation. In unclear cases, lung ultrasound was performed to differentiate opacities [18]. An episode of mechanical ventilation was defined as the period between tracheal intubation (day 1) and 24 h after successful extubation (or disconnection from the ventilator in the case of tracheotomized patients).
Table 1.
| PVAP (CDC 2013 definition) | VAP (CDC 2008 definition) | VAT | |
|---|---|---|---|
| Clinical criteria |
Increase in FiO2 ≥ 0.20 or in PEEP ≥ 3 cmH2O with a previous period of stability/improvement ≥ 2 days And Suspicion of infectionc And Beginning of a new antibiotic |
One of the following: 1. Worsening gas Exchangea 2. Tachypnea or dyspnea 3. Change in sputum characteristicsb 4. Rales or bronchial breath sounds And at least one: 1. Suspicion of infectionc 2. Altered mental status in adults ≥70 years oldg |
Suspicion of infectionc |
| Radiological criteria | Not included | New or progressive infiltrate, consolidation or cavitation | Absence of radiologic criteria for pneumonia |
| Microbiological criteria |
1.Significative growth of a pathogen in respiratory samplesd 2. Insufficient growth of a pathogenic microorganism plus purulent sputumf 3. Pathogenic microorganism in pleural fluid cultures 4. Histopathologic evidence of lung infectione 5. Positive test for pathogenic virus in respiratory samples 6. Positive test for Legionella species |
1. Significative growth of a pathogen in respiratory samplesd 2. > 5% Cells with intracellular bacteria in bronchoalveolar lavage 3. Pathogenic microorganism in pleural fluid cultures 4. Histopathologic evidence of lung infectione 5. Positive growth in blood cultureg |
Positive endotracheal aspirate culture And Purulent sputumf |
PVAP possible ventilator associated pneumonia, VAP ventilator associated pneumonia, VAT ventilator associated tracheobronchitis
aWorsening gas exchange: increased oxygen requirements or in ventilator demand
bChange in sputum characteristics: new onset of purulent respiratory secretions or increase in its production or in suctioning requirements
cSuspicion of infection: leukocytosis (≥ 12,000 cells/mL) or leukopenia (≤ 4000 cells/mL) or fever (≥ 38 °C) or hypothermia (≤ 36 °C)
dSignificative growth in respiratory samples: endotracheal aspirate: ≥ 105 CFU/mL, bronchoalveolar lavage: ≥ 104 CFU/mL, lung tissue: ≥ 104 CFU/g, protected specimen brush: ≥ 103 CFU/mL
eHistopathologic evidence of lung infection: abscess formation or foci of consolidation with intense polymorphonuclear accumulation or positive quantitative culture of parenchyma or evidence of parenchyma invasion by fungus or virus
fPurulent sputum: 325 neutrophils and ≤ 10 squamous epithelial cells per low power field
gWithout other recognized focus
Fig. 1.
Ventilator associated events, CDC 2013 classification and 2015 update [3, 10, 34]
Continuous data were reported as medians and interquartile ranges and categorical data as numbers and percentages; the T test or Mann–Whitney test was used for comparison of continuous variables, while the Chi squared test or Fisher’s exact test was used to compare categorical variables. The clustered Wilcoxon rank sum test was used for clustered data following the Datta–Satten method. Relative risks and 95% confidence intervals were calculated; a p value ≤ 0.05 was considered statistically significant and a p value between 0.05 and ≤ 0.10 was considered as showing a trend toward statistical significance. All analyses were performed in R, version 3.4.1 (R Core Team 19).
The study protocol was approved by the institutional review board on human research of each participating center and at Vall d´Hebron University hospital as the coordinating center [PR(AG)28/2014]. Patients, or the relatives of unconscious patients, were asked to provide written consent prior to participating in the study.
Results
The study cohort comprised 244 adults (of a total of 1078 eligible patients) who between them had 2856 ventilator days with full follow-up. A flow-chart showing the patients enrolled and the exclusion criteria can be found in the ESM (appendix 4). The median APACHE II score at admission of the patients included was 20.6 (25–75% IQR 14–27). The ICU LOS was 14 days (25–75% IQR 8–26.2), the hospital LOS was 25 days (25–75% IQR 11.7–45), and 30-day mortality (for the entire cohort) was 21.7%. Almost all patients were intubated within 24 h of hospitalization; sites of and reasons for intubation are detailed in the ESM (appendix 5). The patients’ baseline characteristics are shown in Table 2.
Table 2.
| Variable | All (n 244) | 2013 CDC criteria (VAE vs. No VAE) | 2013 CDC criteria (PVAP vs. No PVAP) | ||||
|---|---|---|---|---|---|---|---|
| VAE (n 117) | No VAE (n 127) | P | PVAP (n 64) | No PVAP (n 178) | p | ||
| Male n (%) | 154 (63.1) | 76 (65) | 78 (61) | 0.56 | 50 (32.5) | 104 (58.4) | 0.01 |
| Age median (95% CI) | 56 (49–69) | 60 (50–73) | 56 (45–67) | 0.27 | 61 (44–74) | 56 (49–67) | 0.18 |
| Weight kg median (95% CI) | 80 (70–90) | 80 (70–92) | 80 (70–87) | 0.94 | 86 (67–93.7) | 75 (70–85) | 0.009 |
| APACHE II at admission median (95% CI) | 20 (14–27) | 19 (9–21) | 22.5 (16–29) | 0.06 | 19.5 (13–24) | 21 (14–28) | 0.49 |
| Preexisting condition n (%) | |||||||
| Diabetes | 65 (28) | 38 (33) | 27 (22) | 0.07 | 13 (20) | 52 (30) | 0.12 |
| Kidney disease | 33 (14) | 15 (13) | 18 (15) | 0.64 | 5 (7) | 28 (16) | 0.08 |
| Cancer | 20 (8) | 8 (7) | 12 (10) | 0.41 | 3 (5) | 17 (10) | 0.20 |
| Heart disease | 65 (27) | 36 (31) | 29 (24) | 0.20 | 20 (31) | 45 (26) | 0.43 |
| Liver disease | 6 (3) | 1 (1) | 5 (4) | 0.10 | – | 6 (3) | 0.12 |
| Lung disease | 48 (20) | 25 (21) | 23 (19) | 0.60 | 9 (14) | 39 (22) | 0.14 |
| Immunosuppression | 23 (10) | 9 (8) | 14 (12) | 0.31 | 7 (11) | 16 (10) | 0.74 |
| Type of patient n (%) | 0.07 | 0.31 | |||||
| Medical | 141(58) | 62 (53) | 79 (62) | 34 (51) | 107 (61) | ||
| Surgical | 61 (25) | 37 (32) | 24 (19) | 21(32) | 40 (22) | ||
| Traumatic | 42 (17) | 18 (15) | 24 (19) | 11 (16) | 31 (17) | ||
| Tracheostomy n (%) | 44 (29) | 36 (47) | 8 (11) | < 0.001 | 27 (50) | 17 (17) | < 0.001 |
| MV days median (95% CI) | 9 (5–15) | 12 (9–21) | 7 (5–10) | < 0.001 | 17.5 (9–28) | 7 (5–11) | < 0.001 |
| ICU LOS median (95% CI) | 14 (8–26) | 23 (10–36) | 12 (7–20) | 0.001 | 25(12–38) | 11 (7–21) | < 0.001 |
| Hospital LOS median (95% CI) | 25 (11–45) | 31 (17–54) | 19 (10–38) | 0.04 | 32 (10–55) | 19 (10–39) | 0.02 |
| Mortality n (%) | 53 (22) | 31 (43) | 22(29) | 0.06 | 22(40) | 31(33) | 0.38 |
ICU intensive care unit, MV mechanical ventilation, LOS length of stay, VAE ventilator associated event, PVAP possible ventilator associated pneumonia
Eighty-four VARI were recorded (29.3 per 1000 ventilator days). Using the 2008 CDC criteria, 33 (39.3%) of these incidents were VAP (12.2 per 1000 ventilator days) and 51 (60.7%) were VAT (17.8 per 1000 ventilator days) (Fig. 2). In addition, 33 (39%) adults with VARI did not meet the criteria of VAE because they did not require sustained positive end-expiratory pressure (PEEP) or Fi02 increase. Patients with VARI had a significantly longer ventilation period than non-VARI patients (excess: 6 days). Details of hospital LOS and mortality can be found in the ESM (appendix 6). Patients classified as having VAP presented higher mortality than VAT patients (42.8 vs 19.6%, p = 0.007). However, no statistically significant differences were observed in number of ventilator days or in ICU or hospital LOS (ESM appendix 7).
Fig. 2.
Previous and new 2013 CDC classification [3, 10, 34]. jVAE ventilator-associated event, aVARI ventilator-associated respiratory infection, bLOS length of stay, cMV Days invasive mechanical ventilation days, dVAP ventilator associated pneumonia, eVAT ventilator associated tracheobrochitis, fVAC ventilator associated condition, gIVAC infectious ventilator-associated complication, hPVAP possible ventilator associated pneumonia, iARDS acute respiratory distress syndrome, kSuspicion of infection: leukocytosis (≥ 12,000 cells/mL) or leukopenia (≤ 4000 cells/mL) or fever (≥ 38 °C) or hypothermia (≤ 36 °C)
Using the 2013 CDC definition (Table 1), 117 episodes of VAE were recorded (40.8 per 1000 ventilator days). VAE were present after a median of 4 days’ ventilation (25–75% IQR 3–7); 59 VAEs (50.9%) developed within the first 4 days of mechanical ventilation (ESM appendixes 10, 11). The main cause of VAE was IVAC-plus (39.6 per 1000 ventilatory days). IVAC-plus accounted for 113 (96%) episodes, 64 (56.6%) of which met the CDC criteria for PVAP (22.3 per 1000 ventilator days). The rest of the events were classified as VAC (5.58 per 1.000 ventilator days). In patients classified as IVAC-plus without PVAP criteria, most episodes were due to atelectasis (26, 55%); a minority (six, 12.7%) were ARDS, and only two (4.2%) were pulmonary edema (Fig. 2). Both VAE and IVAC-plus definitions had a negative predictive value of 98% but predicted VAP in only one out of three adults (ESM appendix 8).
Patients in the VAE group had significant increases in ventilator days (5 days) and in ICU and hospital LOS (11 and 12 days, respectively) compared with the non-VAE group (Table 2). A trend towards an increase in 30-day mortality was observed in the VAE group (43 vs 28%, p 0.065) (Table 2).
PVAP patients had increases (p < 0.05) of 10.5 days of ventilation, 14 days of ICU stay and 13 days of hospital stay compared to patients without PVAP. Compared to VARI, the PVAP group had 4.5 day more days of ventilation (p < 0.027), 6.5 more days in the ICU and 7 days more in hospital (p < 0.05) (ESM appendix 9). Non-significant differences were detected when non-infected patients were compared using either the 2013 or 2015 classifications (non-VARI vs. non-VAE) (appendix 6 ESM). Correlations between the 2008 and 2013 CDC classifications are shown in Fig. 3.
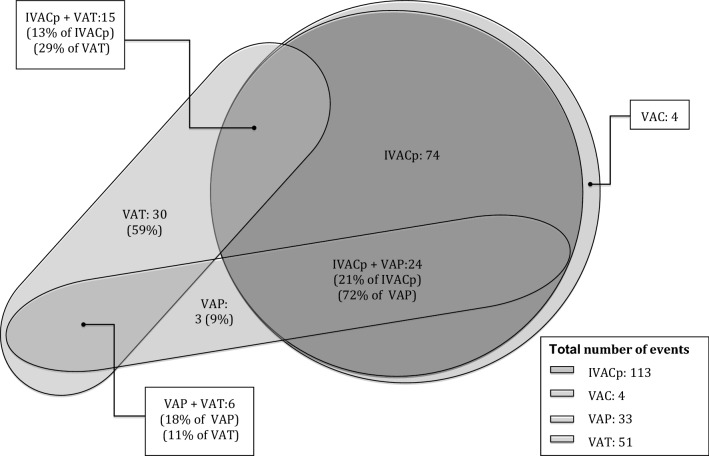
Fig. 3.
Relationship between 2008 and 2013 CDC classifications [10, 15, 36]. 2013 classification is represented with circles and 2008 classification with ovals. VAC ventilator associated condition, IVACp infectious ventilator-associated complication plus, VAP ventilator associated pneumonia, VAT ventilator associated tracheobronchitis
Discussion
To the best of our knowledge, this is the first prospective, international, multicenter study to assess the incidence and outcomes of VAEs in adult ventilated subjects in ICUs. This study identified both VAP and VAT (often missed in earlier studies). Moreover, our study systematically implemented the 2013 CDC classification of VAE and its 2015 update and demonstrated the frequent occurrence of these events in mechanically ventilated adults. Our findings suggest that respiratory infection was the main cause of VAEs, but both the 2013 and 2008 definitions missed a significant proportion of respiratory infections (three out of every four VAT episodes did not meet the criteria of IVAC).
Our findings are consistent with the results of a recent meta-analysis by Fan and colleagues [20] in demonstrating that VAE were associated with increased mortality, more days of mechanical ventilation, and greater hospital LOS than traditional VAP criteria. Worsening in FiO2 and in PEEP are commonly used in clinical practice as surrogates of poor oxygenation [21–23], and both are employed by the VAE definition to detect deterioration in oxygenation. Several studies have reported notable reductions in the time taken to detect VAE when automatic surveillance was used [24, 25]. Not surprisingly, the association between poor oxygenation and outcomes in patients with VAP has previously been reported [21, 22]. In 2012, Klompas et al [23] evaluated the feasibility of 32 possible surveillance definitions for VAP, reporting that only those including quantitative evidence of respiratory deterioration were associated with increased morbidity and mortality. In the same way, the VAE paradigm broadens the spectrum of complications associated with mechanical ventilation, identifying conditions severe enough to cause respiratory worsening, including respiratory infection-related events such as VAP as well as tracheobronchitis and non-infective and extra-pulmonary events. PaO2/FiO2 ratio, the presence of infection, and the level of PEEP [23, 26–28] enhanced the power of VAE surveillance. In our cohort, PVAP was the most frequent complication but non-negligible incidences of atelectasis, ARDS, and pulmonary edema were also documented. Similar results have been reported elsewhere [20, 29, 30], and there is growing evidence that many of these events are preventable [14, 31, 32]. In our study, only 59% of patients with suspicion of pulmonary infection had PVAP and almost 39% of cases were due to a noninfectious event (atelectasis, ARDS, pulmonary edema). In the pediatric population, the presence of VAE was also associated with worse outcomes, including mortality; however, IVAC was less prevalent than in adults. Indeed, the incidence of ARDS and pulmonary edema was lower, while atelectasis was the most frequent event [33, 34]. This confirms that the newer VAE classification moves the focus beyond respiratory infections but selects only the most severe cases.
Our study suggests that identifying VAT (often missed by IVAC-plus criteria) is important, because VAP and VAT have different impacts on mortality. Low agreement has previously been reported between the novel CDC definition and VAP in series in the USA and elsewhere [20, 35, 36]. In the present cohort, almost 30% of VAP cases were not diagnosed as IVAC-plus and only 29% of VAT episodes met the IVAC-plus criteria (Fig. 3). These findings suggest that VAE selects only the most severe cases and discards those without sufficient deterioration in oxygenation to increase the ventilation demands to a level required to reach diagnosis thresholds.
IVAC-plus was the most prevalent event in VAE surveillance and is defined by the suspicion of infection and the beginning of a new antibiotic treatment. The 2015 VAE update [3] amalgamated overall events meeting or exceeding the definition of IVAC into the category IVAC-plus, which includes patients with a confirmed respiratory infection (PVAP) and those with hypoxemia in a non-confirmed pulmonary infectious-related context (IVAC). In our cohort, 43% of IVAC-plus episodes were classified as IVAC, the main causes being atelectasis, ARDS, and pulmonary edema, while the remaining 57% were classified as PVAP and were associated with worse outcomes (Fig. 2). Our study suggests that VAT produced a sustained increase ≥ 2 days in FiO2 ≥ 0.2 or PEEP ≥ 3 cm H2O and fulfilled VAE criteria in around 25% of episodes.
The strengths of our study include the use of a large database with prospective data collection, the inclusion of medical, trauma, and surgical patients, its prospective, multicenter international design and its use of predefined outcomes rather than simply reported rates. This study also has some potential limitations: Its initial design limits the blinding process, which is a potential source of bias. Selection bias is a potential important concern, and due to the heterogeneity of the ICUs’ case mix it might affect generalization. Owing to the high proportion of university hospitals, some of the results might not be globally representative, while the exclusion of children, chronically ill patients, and patients ventilated outside the ICU may limit the extrapolation of the results to other settings. Our cohort presented a lower VAC rate than those reported in previous studies [35, 37], possibly due to differences in institutional antibiotic policies and practices between hospitals. Klompas [38] reported high interobserver variability in diagnosis of respiratory infections, and several studies reported differences in surveillance practice across hospitals [15, 39]; however, these differences could be used to identify variables as independent factors. Finally, given the low incidence of some of the events, the power to demonstrate statistical differences is sometimes limited by small cohort size.
Our study also has important implications that drive new research questions for future developments. There is a need to investigate the effects of new management algorithms for ventilated adults using more subtle definitions of VAE, as has been done in children [33]. Further studies should identify better breakpoints for ventilatory settings and should not omit VAT.
Conclusions
Respiratory infections (mainly VAT) were the most common complications in mechanically ventilated adults. All tiers were associated with significantly worse outcomes. Our findings suggest that the VAE algorithms identified only those events with surrogates of severe oxygenation deterioration. As a consequence, IVAC definitions missed one fourth of the episodes of VAP and three fourths of the episodes of VAT. Identifying VAT (often missed by IVAC-plus criteria) is important, with VAP and VAT having different impacts on patient outcome.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
This research was carried out as part of a PhD program in Health Science at the Universitat Autónoma de Barcelona, Spain and was supported by the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) Study Group for Infections in Critically Ill Patients (ESGCIP), Basel, Switzerland and by Centro de Investigación Biomédica en Red—CIBERES CB06/06/036, Madrid, Spain. We thank Dr. Vandana KE, India, for English editing of the manuscript and critical comments.
Aliye Bastug, Ankara Numune Training and Research Hospital, Turkey; Amir Vahedian-Azimi, Baqiyatallah University of Medical Sciences, Vanak Square, Tehran, Iran; AsumanInan, Haydarpasa Numune Hospital, Istanbul, Turkey; Benito Almirante Gragera, Vall d´Hebron Barcelona Hospital Campus, Barcelona, Spain; Despoina Koulenti, University of Queensland, Brisbane, Australia and Attikon University Hospital, Athens, Greece; Garyphallia Poulakou, Attikon University Hospital, Athens, Greece; George Dimopoulos, Attikon University Hospital, Athens, Greece; Ilkay Bozkurt, Ondokuz Mayis University, Samsun, Turkey; Igor Muzlovic, University Medical Centre, Ljubljana, Slovenia; Izarne Totorika Hospital Universitario de Donostia, Donostia, Spain; Jordi Rello, Vall d'Hebron Barcelona Hospital Campus, Barcelona, Spain; Kostoula Arvaniti, Papageorgiou General Hospital, Thessaloniki, Greece; Leonel Lagunes, Vall d’Hebron Barcelona Hospital Campus, Barcelona, Spain; Lilla Bouadma, Hôpital Bichat-Claude-Bernard, Paris, France; Loreto Vidaur, Hospital Universitario de Donostia, Donostia, Spain; Marina Oikonomou, Papageorgiou General Hospital, Thessaloniki, Greece; MatteoBassetti, Infectious Diseases Clinic, Santa Maria Misericordia Hospital, University of Udine, Udine, Italy; Nefise Oztoprak, Antalya Education and Research Hospital, Turkey; Saad Nseir, Hospital Universitaire Lille, Lille, France; Sergio Ramírez-Estrada, Vall d'Hebron Barcelona Hospital Campus, Barcelona, Spain; Yolanda Peña-López, Vall d’Hebron Barcelona Hospital Campus, Barcelona, Spain
Abbreviations
- ARDS
Acute respiratory distress syndrome
- CDC
Centers for disease control and prevention
- CFU
Colony-forming unit
- ECCMID
European Congress of Clinical Microbiology and Infectious Diseases
- ESM
Electronic supplementary material
- ICU
Intensive care unit
- IVAC
Infection-related ventilator-associated complication
- LOS
Length of stay
- PVAP
Possible ventilator-associated pneumonia
- VAC
Ventilator-associated condition
- VAE
Ventilator-associated event
- VAP
Ventilator-associated pneumonia
- VARI
Ventilator-associated respiratory infection
- VAT
Ventilator-associated tracheobronchitis
Footnotes
The members of the EU-VAE study Investigators Group are listed in the Acknowledgements and in the electronic supplementary material.
Contributor Information
Jordi Rello, Phone: 34 932746209, Email: jrello@crips.es.
the EU-VAE Study Investigators Group:
Aliye Bastug, Amir Vahedian-Azimi, AsumanInan, Benito Almirante Gragera, Despoina Koulenti, Garyphallia Poulakou, George Dimopoulos, Ilkay Bozkurt, Igor Muzlovic, Izarne Totorika, Jordi Rello, Kostoula Arvaniti, Leonel Lagunes, Lilla Bouadma, Loreto Vidaur, Marina Oikonomou, Matteo Bassetti, Nefise Oztoprak, Saad Nseir, Sergio Ramírez-Estrada, and Yolanda Peña-López
References
- 1.Rello J, Diaz E, Mañez R, Sole-Violan J, Valles J, Vidaur L, et al. Improved survival among ICU-hospitalized patients with community-acquired pneumonia by unidentified organisms: a multicenter case–control study. Eur J Clin Microbiol Infect Dis. 2017;36:123–130. doi: 10.1007/s10096-016-2779-5. [DOI] [PubMed] [Google Scholar]
- 2.Melsen WG, Rovers MM, Groenwold RHH, Bergmans DCJJ, Camus C, Bauer TT, et al. Attributable mortality of ventilator-associated pneumonia: a meta-analysis of individual patient data from randomised prevention studies. Lancet Infect Dis. 2017;13:665–671. doi: 10.1016/S1473-3099(13)70081-1. [DOI] [PubMed] [Google Scholar]
- 3.Center for Disease Control and Prevention. Ventilator-associated event. http://www.cdc.gov/nhsn/pdfs/pscmanual/10-vae_final.pdf. Accessed 18 Jun 2017
- 4.Vincent J-L, Rello J, Marshall J, Silva E, Anzueto A, Martin CD, et al. International study of the prevalence and outcomes of infection in intensive care units. JAMA. 2009;302:2323–2329. doi: 10.1001/jama.2009.1754. [DOI] [PubMed] [Google Scholar]
- 5.Klompas M. Does this patient have ventilator-associated pneumonia? JAMA. 2007;297:1583–1593. doi: 10.1001/jama.297.14.1583. [DOI] [PubMed] [Google Scholar]
- 6.Tejerina E, Esteban A, Fernández-Segoviano P, Frutos-Vivar F, Aramburu J, Ballesteros DR-BJ. Accuracy of clinical definitions of ventilator-associated pneumonia: comparison with autopsy findings. J Crit Care. 2010;25:62–68. doi: 10.1016/j.jcrc.2009.05.008. [DOI] [PubMed] [Google Scholar]
- 7.Kerlin MP, Trick WE, Anderson DJ, Babcock HM, Lautenbach E, Gueret R, et al. Interrater reliability of surveillance for ventilator-associated events and pneumonia. Infect Control Hosp Epidemiol. 2017;38:172–178. doi: 10.1017/ice.2016.262. [DOI] [PubMed] [Google Scholar]
- 8.Muscedere JG, Day A, Heyland DK. Mortality, attributable mortality, and clinical events as end points for clinical trials of ventilator-associated pneumonia and hospital-acquired pneumonia. Clin Infect Dis. 2010;51:120–125. doi: 10.1086/653060. [DOI] [PubMed] [Google Scholar]
- 9.Magill SS, Klompas M, Balk R, Burns SM, Deutschman CS, Diekema D, et al. Developing a new national approach to surveillance for ventilator-associated events: executive summary. Am J Infect Control. 2013;41:1096–1099. doi: 10.1016/j.ajic.2013.07.001. [DOI] [PubMed] [Google Scholar]
- 10.Ramírez-Estrada S. Ventilator-associated events in European intensive care units. The EU-VAE project. Abstract in: 26th European Congress of Clinical Microbiology and Infectious Diseases-ECMID; 2016 April 9–12, Amsterdam, Netherlands. Abstract EV089642
- 11.Ramírez-Estrada S. Looking inside the ventilator-associated events in European intensive care units—the EU-VAE project. Oral presentation in: 27th European Congress of Clinical Microbiology and Infectious Diseases-ECMID; 2017 April 22–25, Vienna, Austria. Oral presentation: OS010643
- 12.Knaus WA, Draper EA, Wagner DPZJ. APACHE II: a severity of disease classification system. Crit Care Med. 1985;8:18–29. [PubMed] [Google Scholar]
- 13.Bone RC, Balk RA, Cerra FB, Dellinger RP, Fein AM, Knaus WA, et al. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Chest. 1992;101(1644):55. doi: 10.1378/chest.101.6.1644. [DOI] [PubMed] [Google Scholar]
- 14.Magill SS, Li Q, Gross C, Dudeck M, Allen-Bridson K, Edwards JR. Incidence and characteristics of ventilator-associated events reported to the national healthcare safety network in 2014. Crit Care Med. 2016;44:2154–2162. doi: 10.1097/CCM.0000000000001871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pearson D, Hayashi Y, Richards B, Lipman J (2014) VAP, VAC, IVAC, and ventilator-associated events: the need for objectivity for surveillance. ICU Management & Practice. https://healthmanagement.org/c/icu/issuearticle/vap-vac-ivac-and-ventilator-associated-events-the-need-for-objectivity-for-surveillance. Accessed 15 Dec 2017
- 16.Nseir S, Favory R, Jozefowicz E, Decamps F, Dewavrin F, Brunin G, et al. Antimicrobial treatment for ventilator-associated tracheobronchitis: a randomized controlled multicenter study. Crit Care Med. 2008;12:62. doi: 10.1186/cc6890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Martin-Loeches I, Povoa P, Rodríguez A, et al. TAVeM study. Incidence and prognosis of ventilator-associated tracheobronchitis (TAVeM): a multicentre, prospective, observational study. Lancet Respir Med. 2015;3:859–868. doi: 10.1016/S2213-2600(15)00326-4. [DOI] [PubMed] [Google Scholar]
- 18.Lichtenstein DA. Lung ultrasound in the critically ill. Ann Intensive Care. 2014;9(4):1. doi: 10.1186/2110-5820-4-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Core Team R. R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing; 2017. [Google Scholar]
- 20.Fan Y, Gao F, Wu Y, Zhang J, Zhu M, Xiong L. Does ventilator-associated event surveillance detect ventilator-associated pneumonia in intensive care units? A systematic review and meta-analysis. Crit Care. 2016;20:338. doi: 10.1186/s13054-016-1506-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Vidaur L, Planas K, Sierra R, Dimopoulos G, Ramirez A, Lisboa T, et al. Ventilator-associated pneumonia: impact of organisms on clinical resolution and medical resources utilization. Chest. 2008;133:625–632. doi: 10.1378/chest.07-2020. [DOI] [PubMed] [Google Scholar]
- 22.Luna CM, Blanzaco D, Niederman MS, Matarucco W, Baredes NC, Desmery P, et al. Resolution of ventilator-associated pneumonia: prospective evaluation of the clinical pulmonary infection score as an early clinical predictor of outcome. Crit Care Med. 2003;3:676–682. doi: 10.1097/01.CCM.0000055380.86458.1E. [DOI] [PubMed] [Google Scholar]
- 23.Klompas M, Magill S, Robicsek A, et al. Objective surveillance definitions for ventilator-associated pneumonia. Crit Care Med. 2012;12:3154–3161. doi: 10.1097/CCM.0b013e318260c6d9. [DOI] [PubMed] [Google Scholar]
- 24.Mann T, Ellsworth J, Huda N, Neelakanta A, Chevalier T, Sims KL, et al. Building and validating a computerized algorithm for surveillance of ventilator-associated events. Infect Control Hosp Epidemiol. 2015;36:999–1003. doi: 10.1017/ice.2015.127. [DOI] [PubMed] [Google Scholar]
- 25.Nuckchady D, Heckman MG, Diehl NN, Creech T, Carey D, Domnick R, et al. Assessment of an automated surveillance system for detection of initial ventilator-associated events. Am J Infect Control. 2015;43:1119–1121. doi: 10.1016/j.ajic.2015.05.040. [DOI] [PubMed] [Google Scholar]
- 26.Laffey JG, Bellani G, Pham T, Fan E, Madotto F, Bajwa EK, et al. Potentially modifiable factors contributing to outcome from acute respiratory distress syndrome: the LUNG SAFE study. Intensive Care Med. 2016;42(1865–1876):38. doi: 10.1007/s00134-016-4571-5. [DOI] [PubMed] [Google Scholar]
- 27.Villar J, Pérez-Méndez L, Blanco J, Añón JM, Blanch L, Belda J, et al. A universal definition of ARDS: the PaO2/FiO2 ratio under a standard ventilatory setting a prospective, multicenter validation study. Intensive Care Med. 2013;39:583–592. doi: 10.1007/s00134-012-2803-x. [DOI] [PubMed] [Google Scholar]
- 28.Bein T, Grasso S, Moerer O, Quintel M, Guerin C, Deja M, et al. The standard of care of patients with ARDS: ventilatory settings and rescue therapies for refractory hypoxemia. Intensive Care Med. 2016;42:699–711. doi: 10.1007/s00134-016-4325-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Boyer AF, Schoenberg N, Babcock H, McMullen KM, Micek ST, Kollef MH. A prospective evaluation of ventilator-associated conditions and infection-related ventilator-associated conditions. Chest. 2015;147:68–81. doi: 10.1378/chest.14-0544. [DOI] [PubMed] [Google Scholar]
- 30.Klompas M, Khan Y, Kleinman K, Evans RS, Lloyd JF, Stevenson K, et al. Multicenter evaluation of a novel surveillance paradigm for complications of mechanicalventilation. PLoS One. 2011;6:e18062. doi: 10.1371/journal.pone.0018062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nair GB, Niederman MS. Ventilator-associated pneumonia: present understanding and ongoing debates. Intensive Care Med. 2015;41:34–48. doi: 10.1007/s00134-014-3564-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lewis SC, Li L, Murphy MV, Klompas M, Prevention Epicenters CDC. Risk factors for ventilator-associated events: a case-control multivariable analysis. Crit Care Med. 2014;42:1839–1848. doi: 10.1097/CCM.0000000000000338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Peña-López Y, et al. Assessing prediction accuracy for outcomes of ventilator-associated events and infctions in critically ill children: a prospective cohort study. Clin Microbiol Infect. 2018;24(7):732–737. doi: 10.1016/j.cmi.2017.10.004. [DOI] [PubMed] [Google Scholar]
- 34.Gionfriddo A, et al. Retrospective application of new pediatric ventilator-associated pneumonia criteria identifies a high-risk population. Pediatr Crit Care Med. 2018;19(6):507–512. doi: 10.1097/PCC.0000000000001522. [DOI] [PubMed] [Google Scholar]
- 35.Muscedere J, Sinuff T, Heyland DK, Dodek PM, Keenan SP, Wood G, et al. The clinical impact and preventability of ventilator-associated conditions in critically ill patients who are mechanically ventilated. Chest. 2013;144:1453–1460. doi: 10.1378/chest.13-0853. [DOI] [PubMed] [Google Scholar]
- 36.Klouwenberg PMCK, Van Mourik MSM, Ong DSY, Horn J, Schultz MJ, Cremer OL, et al. Electronic implementation of a novel surveillance paradigm for ventilator-associated events feasibility and validation. Am J Respir Crit Care Med. 2014;189:947–955. doi: 10.1164/rccm.201307-1376OC. [DOI] [PubMed] [Google Scholar]
- 37.Bouadma L, Sonneville R, Garrouste-Orgeas M, Darmon M, Souweine B, Voiriot G, et al. Ventilator-associated events: prevalence, outcome, and relationship with ventilator-associated pneumonia. Crit Care Med. 2015;43:1798–1806. doi: 10.1097/CCM.0000000000001091. [DOI] [PubMed] [Google Scholar]
- 38.Klompas M. Interobserver variability in ventilator-associated pneumonia surveillance. Am J Infect Control. 2010;38:237–239. doi: 10.1016/j.ajic.2009.10.003. [DOI] [PubMed] [Google Scholar]
- 39.Horan TC, Andrus M, Dudeck MA. CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control. 2008;36:309–332. doi: 10.1016/j.ajic.2008.03.002. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.