Abstract
Lactoferrin is a milk protein that exhibits broad-spectrum antimicrobial properties. Previous studies indicated that supplemental lactoferrin may alter the microbial populations in the gut of nonruminants and increase preweaning weight gains in calves. In the present study, 40 Holstein calves were used to examine the effects of supplemental lactoferrin (0, 1, 2, or 3 g/d) on health, growth, and feed intake from 3 d of age to 2 wk postweaning. Lactoferrin was mixed and fed with a nonmedicated milk replacer. Calves were housed in individual pens and offered a textured, nonmedicated starter and water for ad libitum consumption. Body weight and heart girth were measured weekly. Intakes of milk replacer and starter were determined daily. Fecal consistency was monitored three times per week. Calves were weaned when they met certain criteria based on body weight gain and starter intake. Preweaning fecal score responded quadratically, with the group fed 1 g/d of lactoferrin having the lowest score. Overall and preweaning number of days medicated responded in the same manner as fecal score. Preweaning average daily gain and gain-to-feed ratio increased linearly with lactoferrin supplementation, whereas postweaning gain-to-feed ratio decreased linearly with lactoferrin. Overall average daily heart girth gain increased linearly with lactoferrin. Body weight, weaning age, and dry matter intake were not different among treatments. Based on the observed improved gain-to-feed ratios, increased average daily gains, improved fecal scores, and reduced morbidity in preweaned calves, it appears that lactoferrin may be a beneficial supplement in the diets of neonatal calves prior to weaning.
Key words: calves, health, lactoferrin, weaning
Abbreviation key: ADG, average daily gain; LF, lactoferrin
Introduction
It is well accepted that colostrum is an essential part of the newborn calf's diet. The primary reason for the benefits of colostrum is the passive transfer of maternal immunoglobulins to the newborn. In addition to immunoglobulins, colostrum and milk contain many other bioactive substances that are important for the development of the immune system and for fighting infections in the newborn (Shah, 2000). Lactoferrin (LF) is among these bioactive compounds. Lactoferrin is an iron-binding glycoprotein present in the colostrum and milk of most mammals (Masson and Heremans, 1971) and in many body secretions including bronchial mucus (Fahy et al., 1993) and tears (Kijlstra et al., 1983). Compared to other species, bovine milk and colostrum have relatively low concentrations of LF. Bovine colostrum contains approximately 2 mg of LF per milliliter (Tsuji et al., 1990), and mature milk contains 20 to 200 μg/ml (Masson and Heremans, 1971). Lactoferrin is also present at high concentrations (up to 100 mg/ml) in the mammary secretions of the nonlactating cow (Rejman et al., 1989). As an antimicrobial protein, LF produced by the mammary gland may serve a dual role protecting both the mammary gland and the neonatal intestine from infection.
Diarrhea is the most common ailment in young calves causing more than 52% of preweaning deaths (NAHMS, 1993). Diarrhea can be caused by a variety of bacteria or viruses, but the most common pathogens are enterotoxigenic Escherichia coli, rotavirus, coronavirus, Cryptosporidium, and Salmonellae (Tromp, 1990). Studies have shown that LF has activity against at least two of these pathogens, E. coli (Teraguchi et al., 1994) and rotavirus (Superti et al., 1997). These data suggest that LF may prevent infection by these organisms in the calf. Evidence has shown that LF has bacteriostatic activity in vivo. Orally administered bovine LF suppresses the proliferation of intestinal E. coli in milk-fed mice (Teraguchi et al., 1994). If similar activity is observed in the gastrointestinal tract of young calves, there is potential for using LF as a preventative supplement to reduce the occurrence of disease or as a treatment for neonatal diarrhea.
Because bovine colostrum and milk contain a low LF concentration, supplementing the diets of preweaned calves with LF could improve their health and overall performance. Results from a recent study conducted in our laboratory demonstrated that calves fed 1 and 10 g/d of LF during the preweaning period weighed more, had increased ADG, tended to consume more dry feed, and tended to have increased feed efficiency (gain/DMI; Joslin et al., 2002). Calves fed 1 g/d of LF had a greater preweaning ADG than calves fed 10 g/d LF.
The objective of the present study was to further examine the effects of supplemental LF on calf health, growth, feed intake, and feed efficiency.
Materials and Methods
Calves, Diets, and Treatments
This experiment was reviewed and approved by the University of New Hampshire Institutional Animal Care and Use Committee (Approval #010201).
At birth, 40 Holstein calves (36 heifers and four bulls) were randomly assigned by blocks of four, to one of four treatments: 0 (control), 1, 2, or 3 g/d of LF. The iron saturation of the LF (Agri-Cell, Methuen, MA) was 13.2 mg/100 g. All calves received 2 L of good quality colostrum, tested by a colostrometer, within 3 h after birth, and another 2 L of good quality colostrum 8 to 12 h later. Calves were removed from their dam before the first colostrum feeding and placed in a naturally ventilated, enclosed calf room, in individual pens. Pens were bedded with kiln-dried sawdust. The calves remained in their pens for the duration of the study. On the day of birth, an initial BW was obtained before the second feeding of colostrum. On d 2, calves were fed a nonmedicated, all-milk protein milk replacer (Dairy Maid, Blue Seal Feeds, Londonderry, NH) in two feedings at 0700 and 1500 h. Beginning on d 2, every day each calf received 1.2% of its initial BW, in milk replacer powder. The milk replacer powder was divided into two equal portions, and each portion was reconstituted in 2 L of warm water immediately before feeding. Starting on d 3, and continuing until 14 d postweaning, calves had unlimited access to a nonmedicated starter (Calf Starter, Blue Seal Feeds) and fresh water. Starting on d 3, LF treatment was mixed with milk replacer and divided equally among the two feedings. Fresh starter and milk replacer were given to calves twice daily. Starter orts were collected and weighed daily. The starter consisted of steam-flaked corn, crimped oats, molasses, and pellets. Calves were weaned abruptly when they met the following four criteria: 1) minimum of 21 d old, 2) daily starter DMI was ≥1% of initial BW, for three consecutive days, 3) cumulative starter DMI was ≥9% of initial BW, and 4) weight gain was ≥12% of initial BW (Greenwood et al., 1997). Calves were removed from the study at 14 d postweaning.
Feed Intake and Feed Analysis
Feed intake was calculated on a daily basis. The DM of the orts was determined daily, and the DM of the starter and milk replacer powder was determined for each 22.7-kg bag. The DM of the milk replacer powder, starter, and orts was determined by drying samples in a forced-air convection oven (VWR Scientific Products Corp., Boston, MA) at 60°C for 24 h. Samples of starter and milk replacer powder were saved from each bag of feed and stored at −20°C. When the study was completed, samples were composited by shipment (20 bags/shipment of starter, and 10 bags/shipment of milk replacer) for nutrient analysis. Composited starter samples were dried in the forced-air convection oven at 60°C for 6 h and ground through a 1-mm screen using a Wiley mill (Thomas Scientific, Swedesboro, NJ). Milk replacer and starter were analyzed for CP (AOAC, 1979), fat, or fatty acid content (AOAC, 1995), and Ca, P, Mg, and Fe (AOAC, 1990). Starter was also analyzed for NDF (Goering and Van Soest, 1970). Water was tested for iron levels by atomic absorption (Clesceri et al., 1989). Milk replacer was reconstituted to 12.2% DM for determination of LF levels using ELISA (Bethyl Laboratories, Inc., Montgomery, TX).
Measurements
Calves were weighed and measured for heart girth once per week. Calves born on Friday through Monday were weighed and measured every Monday, and calves born on Tuesday through Thursday were weighed and measured every Thursday. Calves were also weighed at birth, weaning, and 14 d postweaning. Evaluation of fecal consistency was performed by three independent scorers every Monday, Wednesday, and Friday. Two of the three scorers were blind to treatment. Feces were scored on a scale of 1 to 4, with 1 = firm and 4 = watery diarrhea. Sick days were recorded as any day that a calf received medication. Medication was given to calves that had a fecal score greater than or equal to 3 (antibiotic and electrolyte treatment), or calves that had a rectal temperature greater than 39°C.
Statistical Analysis
A randomized, complete block design was used, and calves were randomly assigned to treatments in blocks of four. Analysis of variance was conducted using the MIXED procedure of SAS (SAS release 8.2, 2001). The mixed effects model used was:
where
- Yijk
is the dependent variable,
- μ
is the overall mean,
- bi
is the random effect of block i (i = 1, …10),
- Lj
is the fixed effect of the jth lactoferrin level (j = 0, …3),
- K
is the regression coefficient of the covariate C,
- Cijk
is the value of the covariate variable for the kth calf, of the ith block, of the jth lactoferrin level (k = 1, …4), and
- eijk
is the random error associated with the calf k, in block i, that received lactoferrin level j.
Initial BW was used as a covariate for all measures. Significance was determined at a probability value ≤0.05. Data were tested for linear, quadratic, and cubic responses to level of LF supplementation. The data from one calf, on the 2-g treatment, were removed from the statistical analysis, due to abnormal eating habits and severe illness.
Results
Chemical analyses of milk replacer and starter are shown in Table 1 . Iron content of the water used to mix the LF and milk replacer was less than the detection limit of the assay (0.10 mg/kg). Lactoferrin content of the reconstituted milk replacer was 0.014 ± 0.002 mg/ml.
Table 1.
Chemical analysis of milk replacer and starter.
| Item | Milk replacer | Starter |
|---|---|---|
| DM, % | 97.58 | 89.60 |
| CP, % DM | 21.91 | 23.16 |
| Fatty acids, % DM | 21.04 | … |
| Fat, % DM | … | 4.15 |
| NDF, % DM | … | 23.89 |
| Ca, % DM | 1.38 | 0.93 |
| P, % DM | 0.95 | 0.81 |
| Mg, % DM | 0.20 | 0.36 |
| Fe, mg/kg | 88.50 | 162.20 |
Preweaning fecal scores followed a quadratic pattern, with the group fed the 1-g treatment having the lowest score and the control group having the highest score (Figure 1 ). During the second week, calves fed LF had lower fecal scores than did controls (Table 2 ). Preweaning (Figure 2 ), wk 3, and overall number of days medicated responded in a quadratic fashion (Table 2). The group fed the 1-g treatment had the lowest number of days medicated, and the control group had the highest. The number of days medicated in wk 6 decreased linearly with increasing amounts of LF.
Figure 1.
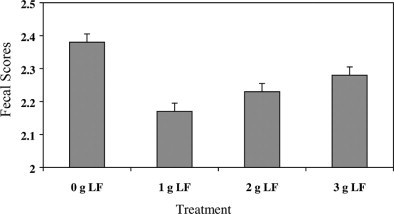
Least square means of preweaning fecal scores of calves fed different amounts of lactoferrin during the preweaning period (fecal score: 1 = firm to 4 = watery diarrhea). LF = Lactoferrin. Standard error = 0.05. Quadratic response P = 0.01.
Table 2.
Least square means of overall, preweaning, and postweaning fecal score and number of days medicated of calves fed different amounts of lactoferrin during the preweaning period.
| Lactoferrin1 (g/d) |
Contrast2,3 (P =) |
||||||
|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | SE | L | Q | |
| n | 10 | 10 | 9 | 10 | … | … | … |
| Fecal scores4 | |||||||
| wk 1 | 2.28 | 2.13 | 2.13 | 2.15 | 0.09 | NS | NS |
| wk 2 | 2.61 | 2.15 | 2.28 | 2.18 | 0.10 | 0.01 | 0.06 |
| wk 3 | 2.27 | 2.23 | 2.33 | 2.38 | 0.11 | NS | NS |
| wk 4 | 2.24 | 2.24 | 2.19 | 2.41 | 0.09 | NS | NS |
| wk 5 | 2.45 | 2.38 | 2.52 | 2.49 | 0.11 | NS | NS |
| wk 6 | 2.45 | 2.53 | 2.59 | 2.48 | 0.13 | NS | NS |
| wk 7 | 2.28 | 2.37 | 2.19 | 2.05 | 0.30 | NS | NS |
| Preweaning | 2.38 | 2.17 | 2.23 | 2.28 | 0.05 | NS | 0.01 |
| Postweaning | 2.39 | 2.43 | 2.46 | 2.48 | 0.09 | NS | NS |
| Overall | 2.38 | 2.29 | 2.33 | 2.35 | 0.05 | NS | NS |
| Days medicated | |||||||
| wk 1 | 0.70 | 0.00 | 0.12 | 0.00 | 0.29 | NS | NS |
| wk 2 | 1.41 | 0.23 | 1.11 | 1.20 | 0.45 | NS | NS |
| wk 3 | 2.20 | 0.20 | 0.55 | 1.40 | 0.54 | NS | 0.01 |
| wk 4 | 0.67 | 0.45 | 0.29 | 1.31 | 0.39 | NS | NS |
| wk 5 | 0.67 | 0.65 | 1.20 | 1.21 | 0.55 | NS | NS |
| wk 6 | 0.71 | 0.82 | 0.65 | 0.14 | 0.33 | 0.04 | NS |
| wk 7 | 0.60 | 0.00 | 0.05 | 0.44 | 0.54 | NS | NS |
| Preweaning | 4.70 | 1.59 | 2.01 | 3.40 | 0.89 | NS | 0.02 |
| Postweaning | 1.98 | 0.86 | 1.83 | 2.01 | 0.82 | NS | NS |
| Overall | 6.67 | 2.45 | 3.84 | 5.41 | 1.30 | NS | 0.03 |
Calves were supplemented with either 0, 1, 2, or 3 g/d of lactoferrin added to milk replacer during the preweaning period.
Contrasts: L = linear and Q = quadratic; NS = nonsignificant (P > 0.10).
A cubic effect was observed for wk 2 fecal score (P = 0.05), all other cubic effects were NS.
Fecal score: 1 = firm through 4 = watery diarrhea.
Figure 2.
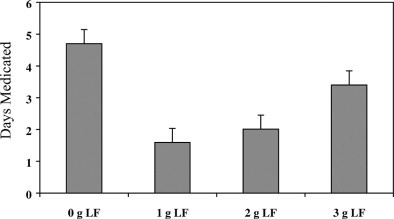
Least square means of preweaning number of days medicated of calves fed different amounts of lactoferrin during the preweaning period. LF = Lactoferrin. Standard error = 0.89. Quadratic response P = 0.02.
Calves consumed all colostrum and milk replacer. Body weight and weaning age were not different among treatments (Table 3 ). No differences in starter DMI were observed (Table 3). However, at wk 5 there was a linear trend (P = 0.10) for increasing starter DMI with increasing amounts of LF. As mentioned previously, the average weaning age for these calves was around 28 d, and while there was not a significant response seen in weaning age, the calves fed 0 and 1 g of LF tended to have later weaning dates than the calves fed 2 and 3 g LF. While only 1 to 2 d separate the weaning ages, this could make a difference in the average daily starter DMI seen during the week after weaning (wk 5).
Table 3.
Least square means of BW, weaning age, weekly, overall, preweaning, and postweaning, starter DMI, ADG, and gain/DMI for calves fed different amounts of lactoferrin during the preweaning period.
| Lactoferrin1 (g/d) |
Contrast2,3 (P =) |
||||||
|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | SE | L | Q | |
| n | 10 | 10 | 9 | 10 | … | … | … |
| Body weight, kg | |||||||
| Initial | 43.2 | 43.4 | 42.9 | 43.0 | 0.2 | NS | NS |
| Weaning | 50.3 | 51.3 | 51.3 | 51.5 | 0.7 | NS | NS |
| Final | 60.5 | 60.6 | 60.6 | 59.9 | 1.4 | NS | NS |
| Weaning age, d | 28.0 | 29.0 | 26.5 | 26.6 | 1.3 | NS | NS |
| Starter DMI, kg/d | |||||||
| wk 1 | 0.04 | 0.05 | 0.03 | 0.02 | 0.01 | NS | NS |
| wk 2 | 0.13 | 0.13 | 0.11 | 0.10 | 0.02 | NS | NS |
| wk 3 | 0.27 | 0.27 | 0.30 | 0.29 | 0.05 | NS | NS |
| wk 4 | 0.64 | 0.63 | 0.68 | 0.72 | 0.10 | NS | NS |
| wk 5 | 1.19 | 1.11 | 1.30 | 1.38 | 0.11 | 0.10 | NS |
| wk 6 | 1.69 | 1.52 | 1.73 | 1.65 | 0.12 | NS | NS |
| wk 7 | 1.78 | 1.55 | 1.86 | 1.94 | 0.19 | NS | NS |
| Preweaning | 0.24 | 0.23 | 0.23 | 0.23 | 0.02 | NS | NS |
| Postweaning | 1.41 | 1.38 | 1.39 | 1.44 | 0.06 | NS | NS |
| Overall | 0.74 | 0.72 | 0.75 | 0.75 | 0.05 | NS | NS |
| ADG, kg/d | |||||||
| wk 2 | 0.04 | 0.26 | 0.18 | 0.25 | 0.09 | NS | NS |
| wk 3 | 0.25 | 0.26 | 0.36 | 0.35 | 0.11 | NS | NS |
| wk 4 | 0.44 | 0.34 | 0.45 | 0.35 | 0.09 | NS | NS |
| wk 5 | 0.60 | 0.44 | 0.48 | 0.68 | 0.09 | NS | 0.04 |
| wk 6 | 0.57 | 0.52 | 0.70 | 0.65 | 0.13 | NS | NS |
| wk 7 | 1.08 | 0.87 | 0.90 | 0.78 | 0.32 | NS | NS |
| Preweaning | 0.28 | 0.30 | 0.35 | 0.36 | 0.03 | 0.02 | NS |
| Postweaning | 0.74 | 0.66 | 0.66 | 0.60 | 0.08 | NS | NS |
| Overall | 0.44 | 0.42 | 0.46 | 0.45 | 0.03 | NS | NS |
| Gain/DMI, kg/kg | |||||||
| wk 2 | 0.08 | 0.40 | 0.27 | 0.42 | 0.14 | NS | NS |
| wk 3 | 0.30 | 0.39 | 0.45 | 0.37 | 0.13 | NS | NS |
| wk 4 | 0.43 | 0.34 | 0.45 | 0.34 | 0.09 | NS | NS |
| wk 5 | 0.48 | 0.37 | 0.36 | 0.49 | 0.06 | 0.07 | NS |
| wk 6 | 0.33 | 0.30 | 0.37 | 0.34 | 0.07 | NS | NS |
| wk 7 | 0.61 | 0.55 | 0.36 | 0.27 | 0.18 | NS | NS |
| Preweaning | 0.38 | 0.41 | 0.48 | 0.49 | 0.04 | 0.01 | NS |
| Postweaning | 0.52 | 0.46 | 0.45 | 0.42 | 0.04 | 0.04 | NS |
| Overall | 0.43 | 0.42 | 0.44 | 0.43 | 0.02 | NS | NS |
Calves were supplemented with either 0, 1, 2, or 3 g/d of lactoferrin added to milk replacer during the preweaning period.
Contrasts: L = linear and Q = quadratic; NS = non-significant (P > 0.10).
There were no significant cubic effects.
Preweaning average daily gain (ADG) increased linearly with LF supplementation (Table 3). A quadratic response occurred in wk 5; the calves receiving the 0- and 3-g treatments had higher ADG than the calves receiving the 1- and 2-g treatments (Table 3).
Preweaning feed efficiency (gain/DMI) increased linearly with increasing LF supplementation, whereas postweaning feed efficiency decreased linearly (Table 3). Similar to ADG, wk-5 feed efficiency tended (P = 0.07) to respond quadratically, with the 0-g and 3-g treatments having higher feed efficiency than the 1-g and 2-g treatments. Overall, average daily heart girth gain increased linearly (P = 0.02) with LF level (0.25, 0.24, 0.29, 0.31 cm/d; SE = 0.02) for treatments 0, 1, 2, and 3 g/d.
Discussion
The results of the present study may be explained by three of the functions of LF: antibacterial activity (Teraguchi et al., 1994), growth factor activity (Zhang et al., 2001), and ability to stimulate glucose absorption (Teraguchi et al., 1998). Improved intestinal health, as demonstrated by reduced fecal scores, and reduced numbers of days medicated is probably a function of the antibacterial activity and immunomodulatory properties of LF. Increased growth responses are probably related to the ability of LF to increase intestinal growth and nutrient absorption, and therefore, feed efficiency.
Diarrhea is a major problem in young calves, especially during the preweaning period. In the present study, preweaning fecal scores were the lowest for the calves fed 1 g/d LF and the highest for the control calves. Preweaning and total number of days medicated followed a similar response pattern. It has been demonstrated that LF has bacteriostatic and bactericidal properties that are effective against pathogenic bacteria such as E. coli (Dionysius et al., 1993; Teraguchi et al., 1994). More specifically, calves treated with a preparation containing LF and the lactoperoxidase system were able to survive a lethal intestinal infection of enterotoxigenic E. coli (Still et al., 1990). With E. coli being the principle pathogen responsible for neonatal calf diarrhea, supplemental LF may have reduced the establishment of E. coli in the gut of the calf, leading to reduced bouts of diarrhea. Similarly, LF has been shown to be effective against the diarrhea-causing pathogen, rotavirus (Superti et al., 1997). If calves in this study were exposed to rotavirus, then LF may have played a role in reducing the occurrence of infection by this virus.
From the results of this study, it appears that of the doses given, 1 g/d LF was the optimal dose for reducing disease. This finding is consistent with previous results from our laboratory, where calves fed 1 g/d of LF had lower numbers of days medicated than calves fed 0 or 10 g/d of LF (Joslin et al. 2002). However, in the previous study, this result was not significant, probably due to a small sample size. The results of these two studies make it apparent that the higher doses of LF are not as effective at reducing disease. Supplementing the milk replacer with 1 g/d of LF brought the total concentration of LF in the milk replacer to a concentration similar to that found in average bovine whole milk (250 mg/ml). The intestinal system of the newborn calf may be best adapted to utilizing this low level of LF to optimize health. Adding LF to milk replacer may be a way to make it more similar to milk, which may benefit the health of calves. It is clear from the results of this study that 1 g/d of supplemental LF improves health during the preweaning period. It should be noted here that the calves in this study were well-managed and generally healthy. The fact that LF improved the health of calves in a high-quality environment suggests that where conditions are less desirable, LF may be even more effective at improving health.
While LF supplementation resulted in health responses, it also resulted in growth and feed efficiency responses. During the preweaning period, when the calves were receiving treatments, ADG and gain/DMI increased linearly as the level of supplemental LF increased. Intake of DM was not affected by LF supplementation. These results are similar to those of our previous study except that they found that calves fed the 1-g treatment had higher ADG and gain/DMI than calves fed the 10-g treatment (Joslin et al., 2002). This suggests that the linear effect of LF supplementation on ADG and gain/DMI does not continue up to 10 g/d.
It cannot be determined from this study whether the growth and feed efficiency responses to LF supplementation are indirect effects of improved health or direct effects on increased nutrient absorption. Lactoferrin is known to act as a growth factor by stimulating mucosal growth of the small intestine in vivo. Zhang et al. (2001) observed that the small intestine weight of rat pups consuming milk containing 12 mg/ml of human LF increased 27%, and intestinal length increased by 6.5% compared with rat pups consuming milk containing no human LF. In the present study, it is possible that the supplemental LF was acting as a growth factor in the small intestine of the calf. A more developed intestinal mucosa will have greater absorptive ability, allowing the calf to absorb more nutrients. Therefore, calves that received supplemental LF may have been able to absorb more nutrients from their feed and may have been more efficient at converting feed into BW gain.
During the postweaning period, after calves had stopped receiving supplemental LF, gain/DMI decreased linearly with increased feeding of LF during the preweaning period (Table 3). Our previous study did not show this postweaning effect. In spite of the decreased feed efficiency, postweaning ADG was not different among treatments. More research is needed to determine how LF is affecting feed efficiency, both preweaning and postweaning.
Over the entire experimental period, average daily heart girth gain increased linearly with increased LF supplementation. Increased hearth girth gains were probably due to increased skeletal growth. Perhaps the increased feed efficiency during the preweaning period allowed the LF-supplemented calves to grow more than the control calves.
It seems that supplemental LF improves the growth rates and health of preweaned calves. More research should be performed to further understand the mode of action of LF and its improvement in growth rates of calves. Due to the reduced occurrences of diarrhea, providing young calves with supplemental LF could be economically beneficial to the dairy producer, because of reduced costs for medication and labor. The reduced occurrence of diarrhea, seen in the preweaned calf, supplemented with 1 g/d of LF, is supported by the differences seen in wk 2 for fecal scores and wk 2 and 3 for number of days medicated. Based on these results, it may be beneficial for dairy producers to feed 1 g/d of LF to calves for the first 21 d of life.
Using LF as a preventative supplement or as a treatment for diarrhea could reduce the use of traditional antibiotics. Due to the ever-increasing prevalence of antibiotic-resistant strains of bacteria, LF may play an important role in future medicine. Several recent studies have shown that LF is effective against antibiotic resistant bacteria and that it is able to reduce the resistance of some bacteria to antibiotics (Aguila et al., 2001; Leitch and Willcox, 2001). Lactoferrin could be used as a replacement for traditional antibiotics, or it could be used adjunctively with antibiotics to treat antibiotic-resistant bacteria.
Conclusions
Lactoferrin supplementation at 1 g/d reduced fecal scores and the number of days medicated. Average daily gain and feed efficiency were increased by LF supplementation of preweaned dairy calves. However, feed efficiency responded in the opposite manner postweaning. Research with calves should evaluate the efficacy of LF doses that are less than 1 g/d and the efficacy of supplementing LF in the diet after weaning. It should be determined if LF would be most useful as a preventative supplement or as a treatment for diarrhea.
Acknowledgments
The authors thank the University of New Hampshire Dairy Teaching and Research Center staff for their assistance in conducting this experiment. Authors also express their gratitude to Blue Seal Feeds, Inc. for providing the milk replacer and calf starter, to ImmuCell Corporation for providing the LF used in this experiment, and to Celeste Dietterle for typing the manuscript.
References
- Aguila A., Herrera A.G., Morrison D., Cosgove B., Perojo A., Montesinos I., Perez J., Seirra G., Gemmell C.G., Brock J.H. Bacteriostatic activity of human lactoferrin against Staphylococcus aureus is a function of its iron-binding properties and is not influenced by antibiotic resistance. FEMS Immunol. Med. Microbiol. 2001;31:145–152. doi: 10.1111/j.1574-695X.2001.tb00511.x. [DOI] [PubMed] [Google Scholar]
- Association of Official Analytical Chemists Protein (crude) in animal feed semiautomated method 976.06. J. AOAC. 1979;62:290. [Google Scholar]
- Association of Official Analytical Chemists . 15th ed. AOAC; Washington, DC: 1990. Official Methods of Analysis. [Google Scholar]
- Association of Official Analytical Chemists . 16th ed. AOAC; Washington, DC: 1995. Official Methods of Analysis. [Google Scholar]
- Clesceri L.S., Greenberg A.E., Trussell R.R. 17th ed. Am. Publ. Health Assoc., Inc.; Washington, DC: 1989. Standard Methods for the Examination of Water and Wastewater. [Google Scholar]
- Dionysius D.A., Grieve P.A., Milne J.M. Forms of lactoferrin: their antibacterial effect on enterotoxigenic Escherichia coli. J. Dairy Sci. 1993;76:2597–2606. doi: 10.3168/jds.S0022-0302(93)77594-3. [DOI] [PubMed] [Google Scholar]
- Fahy J.V., Steiger D.J., Liu J., Basbaum C.B., Finkbeiner W.E., Boushey H.A. Markers of mucus secretion and DNA levels in induced sputum from asthmatic and from healthy subjects. Am. Rev. Respir. Dis. 1993;147:1132–1137. doi: 10.1164/ajrccm/147.5.1132. [DOI] [PubMed] [Google Scholar]
- Goering H.K., Van Soest P.J. Agric. Handbook No. 379. ARS-USDA; Washington, DC: 1970. Forage Fiber Analysis (Apparatus, Reagents, Procedures, and Some Applications) [Google Scholar]
- Greenwood R.H., Morrill J.L., Titgemeyer E.C. Using dry feed intake as a percentage of initial body weight as a weaning criterion. J. Dairy Sci. 1997;80:2542–2546. doi: 10.3168/jds.S0022-0302(97)76208-8. [DOI] [PubMed] [Google Scholar]
- Joslin R.S., Erickson P.S., Santoro H.M., Whitehouse N.L., Schwab C.G., Rejman J.J. Lactoferrin supplementation to dairy calves. J. Dairy Sci. 2002;85:1237–1242. doi: 10.3168/jds.S0022-0302(02)74187-8. [DOI] [PubMed] [Google Scholar]
- Kijlstra A., Jeurissen S.H., Koning K.M. Lactoferrin levels in normal human tears. Br. J. Opthalmol. 1983;67:199–202. doi: 10.1136/bjo.67.3.199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leitch E.C., Willcox M.D. Lactoferrin-induced reduction of vanB vancomycin resistance in enterococci. Int. J. Antimicrob. Agents. 2001;18:399–402. doi: 10.1016/s0924-8579(01)00415-0. [DOI] [PubMed] [Google Scholar]
- Masson P.L., Heremans J.F. Lactoferrin in milk from different species. Comp. Biochem. Physiol. 1971;39B:119–129. doi: 10.1016/0305-0491(71)90258-6. [DOI] [PubMed] [Google Scholar]
- National Animal Health Monitoring System . USDA, Animal and Plant Health Inspection Service, Veterinary Services, Fort; Collins, CO: 1993. Dairy herd management practices focusing on preweaned heifers. [Google Scholar]
- Rejman J.J., Hurley W.L., Bahr J.M. Enzyme-linked immunosorbent assays of bovine lactoferrin and a 39-kilodalton protein found in mammary secretions during involution. J. Dairy Sci. 1989;72:555–560. doi: 10.3168/jds.S0022-0302(89)79141-4. [DOI] [PubMed] [Google Scholar]
- SAS/STAT User's Guide . SAS Inst., Inc.; Cary, NC: 2001. Version 8.2 Edition. [Google Scholar]
- Shah N.P. Effects of milk-derived bioactives: an overview. Br. J. Nutr. 2000;84(Suppl. 1):S3–S10. doi: 10.1017/s000711450000218x. [DOI] [PubMed] [Google Scholar]
- Still J., Delahaut P., Coppe P., Kaeckenbeeck A., Perraudin J.P. Treatment of induced enterotoxigenic colibacilosis (scours) in calves by the lactoperoxidase system and lactoferrin. Ann. Rech. Vet. 1990;21:143–152. [PubMed] [Google Scholar]
- Superti F., Ammendolia M.G., Valenti P., Seganti L. Antirotaviral activity of milk proteins: lactoferrin prevents rotavirus infection in the enterocyte-like cell line HT-29. Med. Microbiol. Immunol. (Berl.). 1997;186:83–91. doi: 10.1007/s004300050049. [DOI] [PubMed] [Google Scholar]
- Teraguchi S., Ozawa K., Yasuda S., Shin K., Fukuwatari Y., Shimamura S. The bacteriostatic effects of orally administered bovine lactoferrin on intestinal Enterobacteriaceae of SPF mice fed bovine milk. Biosci. Biotech. Biochem. 1994;58:482–487. doi: 10.1271/bbb.57.360. [DOI] [PubMed] [Google Scholar]
- Teraguchi S., Ogata T., Shin K., Kingaku M., Fukuwatari Y., Kawase K., Hayasawa H., Tomita M. The mechanism of in vivo bacteriostasis of bovine lactoferrin. Adv. Exp. Med. Biol. 1998;443:239–246. doi: 10.1007/978-1-4757-9068-9_28. [DOI] [PubMed] [Google Scholar]
- Tomita M., Takase M., Wakabayashi H., Bellamy W. Antimicrobial peptides of lactoferrin. In: Hutchens T.W., editor. Lactoferrin: Structure and Function. Plenum Press; New York: 1994. pp. 209–218. [Google Scholar]
- Tromp A.M. A practitioner's views on fluid therapy in calves. Vet. Clin. North Am. Food Anim. Pract. 1990;6:103–110. doi: 10.1016/s0749-0720(15)30897-5. [DOI] [PubMed] [Google Scholar]
- Tsuji S., Hirata Y., Mukai F. Comparison of lactoferrin content in colostrums between different cattle breeds. J. Dairy Sci. 1990;73:125–128. doi: 10.3168/jds.S0022-0302(90)78654-7. [DOI] [PubMed] [Google Scholar]
- Wharton B.A., Balmer S.E., Scott P.H. Fecal flora in the newborn. Effect of lactoferrin and related nutrients. Adv. Exp. Med. Biol. 1994;357:91–98. [PubMed] [Google Scholar]
- Zhang P., Sawicki V., Lewis A., Hanson L., Nuijens J.H., Neville M.C. Human lactoferrin in the milk of transgenic mice increases intestinal growth in 10-day-old suckling neonates. Adv. Exp. Biol. 2001;501:107–113. doi: 10.1007/978-1-4615-1371-1_13. [DOI] [PubMed] [Google Scholar]


