Abstract
Purpose
Severe bronchiolitis is the leading cause of admission to the pediatric intensive care unit (PICU). Nasal continuous positive airway pressure (nCPAP) has become the primary respiratory support, replacing invasive mechanical ventilation (MV). Our objective was to evaluate the economic and clinical consequences following implementation of this respiratory strategy in our unit.
Methods
This was a retrospective cohort analysis of 525 infants with bronchiolitis requiring respiratory support and successively treated during two distinct periods with invasive MV between 1996 and 2000, P1 (n = 193) and nCPAP between 2006 and 2010, P2 (n = 332). Costs were estimated using the hospital cost billing reports.
Results
Patients’ baseline characteristics were similar between the two periods. P2 is associated with a significant decrease in the length of ventilation (LOV) (4.1 ± 3.5 versus 6.9 ± 4.6 days, p < 0.001), PICU length of stay (LOS) (6.2 ± 4.6 versus 9.7 ± 5.5 days, p < 0.001) and hospital LOS. nCPAP was independently associated with a shorter duration of ventilatory support than MV (hazard ratio 1.8, 95 % CI 1.5–2.2, p < 0.001). nCPAP was also associated with a significant decrease in ventilation-associated complications, and less invasive management. The mean cost of acute viral bronchiolitis-related PICU hospitalizations was significantly decreased, from 17,451 to 11,205 € (p < 0.001). Implementation of nCPAP led to a reduction of the total annual cost of acute viral bronchiolitis hospitalizations of 715,000 €.
Conclusion
nCPAP in severe bronchiolitis is associated with a significant improvement in patient management as shown by the reduction in invasive care, LOV, PICU LOS, hospital LOS, and economic burden.
Keywords: Bronchiolitis, Nasal CPAP, Mechanical ventilation, Noninvasive ventilation, Cost-effectiveness, Economic burden
Introduction
Acute viral bronchiolitis is the most common cause of seasonal acute respiratory failure in infancy throughout the world and is responsible for approximately 600,000 deaths worldwide each year [1, 2]. In recent years, the hospitalization rate among infants with acute bronchiolitis has significantly increased in developed countries [3, 4]. Up to 8 % of infants hospitalized with acute viral bronchiolitis required admission to a pediatric intensive care unit (PICU) owing to recurrent apnea or acute respiratory failure needing ventilatory support [5]. Currently, most PICU teams provide non-invasive nasal continuous positive airway pressure (nCPAP) ventilation as the first-line therapy when required.
Treatment of acute viral bronchiolitis-related hospitalization represents a significant seasonal healthcare burden in all developed countries. Even if representing a limited proportion of all acute viral bronchiolitis, the cost-effectiveness of nCPAP in this setting is unknown.
The main objective of our retrospective study was to compare the clinical evolution and economic impact of two different types of respiratory support: invasive mechanical ventilation and nCPAP—the new standard of care in our PICU.
Materials and methods
Study design and population
We performed a large retrospective analysis of infants admitted to the PICU at the Kremlin-Bicetre tertiary care center for severe acute bronchiolitis needing any type of ventilatory support. Medical records from all infants admitted for bronchiolitis between 1 January 1996 and 28 February 2000, and between 1 January 2006 and 28 February 2010, were obtained. During the first period (1996–2000, P1), the only ventilatory support available was endotracheal mechanical ventilation. During the second period (2006–2010, P2) the primary ventilatory support was non-invasive nCPAP. Criteria for inclusion in the cohorts were admission in the PICU for severe acute bronchiolitis and acute respiratory failure or respiratory syncytial virus (RSV)-related apnea events requiring ventilatory support, either in invasive mechanical ventilation (MV) or nCPAP mode. Exclusion criteria included acute bronchiolitis stabilized with nasal oxygen supplementation only and patients with missing data. The study was approved by Ethical Review Board of the French Speaking Society of Intensive Care, which waived the need for informed consent based on the retrospective design of the study (n° 12–390).
Data collection
For each patient, the demographic data collected included age, gender, weight, gestational age, and presence of congenital heart disease (CHD), bronchopulmonary dysplasia (BPD), airway obstruction, or any other underlying disease. BPD was defined in accordance with recognized criteria [6]. Because of the retrospective design, pre-PICU admission data illustrating disease severity prior to the initiation of ventilatory support was composed of objective measures such as respiratory rate (RR), capillary partial pressure of carbon dioxide (PCO2), pulse oximetry (SpO2), and inspired fraction of inspired O2 (FiO2). The PCR viral status, use of antibiotics, and pulmonary radiologic abnormalities on chest X-ray were also included. The Pediatric Risk of Mortality (PRISM) score was systematically calculated at both hospital and PICU admission, and was also collected. PICU stay data included measurements of maximum PCO2 and minimum SpO2/FiO2 ratio on the first day of admission. All invasive studies and access were collected, such as number of labs and chest X-rays during the PICU stay, and the need of central venous lines. Healthcare-associated pneumonia were based on Centers for Disease Control and Prevention (CDC) criteria and defined by new onset of fever (temperature >38.5 °C), new infiltrate(s) on chest X-ray, and positive tracheal aspirate culture when available. Total length of ventilatory support (invasive alone, invasive + nCPAP, or nCPAP alone) and PICU length of stay (LOS), post-PICU general ward LOS, and PICU readmission were all collected.
Costs assessment
Total costs were assessed using the institutional billing database. Cost information was obtained from the hospital accounting reports. Costs are given per day of hospitalization in the unit. This global data includes all the associated costs, staff salaries, equipment, procedures, and amortization of premises. This value is calculated for each hospital unit on the basis of actual expenses paid the previous year.
The cost burden for both periods was estimated using the median LOS in PICU and general ward for each period. We used the cost accounting from 2009, the most recent year available to estimate total cost using the following formula: total cost (Euros) = [mean PICU LOS (day) × PICU cost accounting] + [mean general ward LOS (day) × general ward cost accounting].
Routine management during P1 (1996–2000)
All patients were admitted to the PICU within the first 24 h following general pediatric ward admission. When ventilatory support was mandated, endotracheal intubation was performed using either a cuffed or uncuffed endotracheal tube (Mallinckrodt™, Covidien, Dublin, Ireland). All infants were ventilated in a time-cycled, pressure-limited ventilation mode (Babylog 8000, Draeger Medical, Lübeck, Germany) in order to achieve a normal range of tidal volume (6–8 ml/kg) with an infant dual-heated ventilator circuit (Fisher and Paykel Healthcare, Auckland, New Zealand). Sedation and neuromuscular blockade were used at the discretion of the attending physician.
Routine management during P2 (2006–2010)
All patients were admitted to the PICU within 24 h of initial hospitalization. nCPAP was applied in the case of acute respiratory failure defined by tachypnea (>2 SD for age) associated with a transcutaneous carbon dioxide pressure (PtcCO2) of at least 50 mmHg and marked chest wall retraction, irrespective of SaO2 level and/or inspiratory oxygen fraction and for RSV-related apnea events. However, clinical conditions leading to emergency endotracheal intubation were considered if any of the following were present: cardiac arrest or hemodynamic instability, major respiratory acidosis (defined by a pH < 7.2), refractory apnea to nCPAP and caffeine and major altered consciousness.
nCPAP was delivered through nasal prongs (Fisher and Paykel Healthcare, Auckland, New Zealand) using an ICU ventilator (Babylog 8000 or Evita 2 Dura Neoflow; Draeger Medical, Lübeck, Germany) and a standard heated humidifier chamber (MR 290 model, Fisher and Paykel Healthcare, Auckland, New Zealand). In all patients, initial PEEP level of nCPAP was set at 7 cmH2O, previously identified as the optimal PEEP in the setting of acute viral bronchiolitis [7]. nCPAP was first performed continuously, then switched to alternate spontaneous breathing and nCPAP support at the discretion of the attending nurse. Duration of ventilatory support encompassed time on both continuous and intermittent nCPAP. Routine sedation was prohibited; however, if necessary, oral hydroxyzine (1 mg/kg × 3 per day) or codeine (0.5 mg/kg × 3 per day) was used.
In both period P1 and P2, all infants underwent routinely chest physiotherapy including increased exhalation technique and were fed enterally using a nasoduodenal tube. The decision to start antimicrobial drugs was left at the discretion of the attending physician. Corticosteroids were not used routinely during both periods. Bronchodilators or adrenaline were used mainly before PICU admission, more often during P1.
Statistical analysis
Baseline demographic data were expressed as number and percentage for binary or ordinary data, and means (SD) for continuous data unless skewed, in which case median and interquartile range (IQR) were reported. Continuous data were compared using the Wilcoxon test and dichotomic data using the Fischer exact test. Time failure data (length of ventilatory support and length of stay) were summarized as medians and 95 % confidence interval (95 % CI).
Survival curves for length of ventilatory support and length of stay were estimated on the basis of the whole cohort using the Kaplan–Meier method, then compared across period groups by using the log-rank test. We additionally adjusted survival analyses for prognostic baseline covariates (age, gestational age, PRISM score, RSV infection, antibiotic use), using a Cox model. Measures of the period effect for survival data were hazard ratios (HR).
For secondary outcomes, we compared the number of chest X-rays, blood check exams, antimicrobial therapy, nutritional support route (enteral vs parenteral), healthcare-associated pneumonia, ARDS, pneumothorax events, and mortality rate using the Fisher test.
All tests were two-tailed, with p values of 0.05 or less considered as statistically significant.
Statistical analysis was carried out with Stata 12 (StataCorp. 2007, Stata Statistical Software: Release 12, College Station, TX: StataCorp. LP).
Results
Patients
During the early period P1, there were 193 cases of severe acute viral bronchiolitis in 186 infants, and 332 bronchiolitis episodes in 325 infants during P2. Seventeen patients were excluded because of missing data during P1 and five during P2. The percentage of invasive ventilation (IV) dramatically decreases from 2003 with a concomitant increase of the use of nCPAP. The percentage of infants without any ventilatory support staying in spontaneous breath during all the PICU LOS remains stable over the whole period (Fig. 1).
Fig. 1.
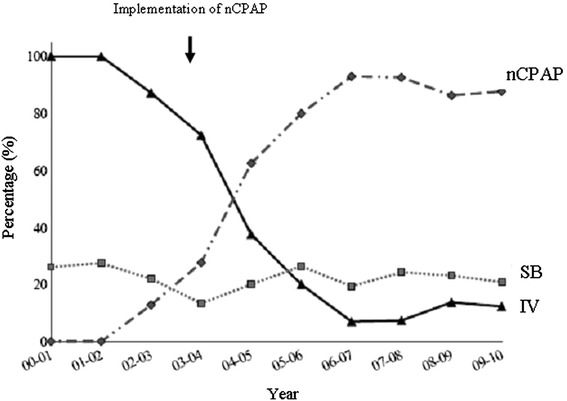
Distribution of ventilatory support in our PICU since 2000. The percentage of invasive ventilation (IV) among all ventilatory support (solid line) dramatically decreases from 2003 with a concomitant increase of the nasal continuous positive pressure (nCPAP) (dashed line). The dotted line represents the percentage of infants admitted to the unit without any ventilatory support, remaining in spontaneous breathing (SB) during their PICU stay. This percentage remains stable over the whole period
Baseline characteristics were similar between the two periods, as summarized in Table 1. Prematurity was the most frequent underlying disease, with 70 (37.6 ± 3.4 %) affected infants during P1 and 111 (34.1 ± 3.4 %) infants during P2. The number of infants with a history of any CHD was higher during P1 (12 versus 4).
Table 1.
Demographic and clinical characteristics of patients
| P1 (1996–2000) n = 193 | P2 (2006–2010) n = 332 | p values | |
|---|---|---|---|
| Age (days) | 59 ± 71 | 53 ± 63 | 0.479 |
| Weight (kg) | 3.93 ± 1.3 | 4.01 ± 1.1 | 0.193 |
| Gestational age (weeks) | 36.8 ± 3.4 | 36.9 ± 3.5 | 0.711 |
| Birth weight (kg) | 2.7 ± 0.8 | 2.8 ± 0.9 | 0.05 |
| Prematurity (%) | 36.3 | 33.4 | 0.59 |
| RSV (%) | 83.1 | 74.7 | 0.03 |
| Atelectasis (%) | 38.3 | 37 | 0.805 |
Data are expressed in mean ± standard deviation
RSV respiratory syncytial virus
Respiratory management
Most infants were ventilated before PICU admission: during P1, 81 % were intubated before PICU admission and during P2, 67 % had ventilatory support with either nCPAP (55 %) or mechanical ventilation (12 %). All respiratory parameters are summarized in Table 2. During P2, there was a significant decrease in the length of ventilatory support relative to P1: the median (95 % CI) length of ventilator support was respectively 6.0 (5.3–6.7) days for P1 and 3.0 (2.7–3.3) days for P2. This reduction was strongly associated with the period and nCPAP with an HR of 1.83 (1.53–2.19), p < 0.001 (Fig. 2a). This effect remains significant after adjustment for prognostic baseline covariates: PRISM score, age, gestational age, RSV infection, and antibiotics [HR (95 % CI) = 1.76 (1.45–2.14), p < 0.001] (Fig. 2b).
Table 2.
Respiratory management of patients before PICU admission and during PICU stay
| P1 (1996–2000) n = 193 | P2 (2006–2010) n = 332 | p values | |
|---|---|---|---|
| Respiratory pattern before PICU admission and ventilatory support | |||
| Invasive ventilation, n (%) | 156 (81) | 40 (12) | |
| nCPAP (%) | 0.5 | 55 | |
| Nasal canula oxygen (%) | 18 | 32 | |
| PCO2 (kPa) | 7.4 ± 2.1 | 8.06 ± 2.1 | 0.0004 |
| SaO2 (%) | 96 ± 5 | 96 ± 6 | 0.475 |
| FiO2 (%) | 39 ± 15 | 36 ± 16 | 0.006 |
| Respiratory evolution during PICU and gas exchange between 2 h and 6 h of admission | |||
| nCPAP failure (%) | 6 | ||
| Length of invasive ventilation (days) | 6.9 ± 4.6 | 5.1 ± 4.5 | 0.0002 |
| Length of ventilation (days) | 6.9 ± 4.6 | 4.1 ± 3.5 | <0.0001 |
| PCO2 (kPa) | 5.4 ± 1.4 | 6.2 ± 1.1 | <0.0001 |
| SaO2 (%) | 96 ± 4 | 97 ± 3 | 0.314 |
| Maximal FiO2 (%) | 52 ± 23 | 43 ± 18 | <0.0001 |
nCPAP nasal continuous positive airway pressure, PCO 2 capillary partial pressure of carbon dioxide, SaO 2 pulse oximetry, FiO 2 inspired fraction of inspired O2
Fig. 2.
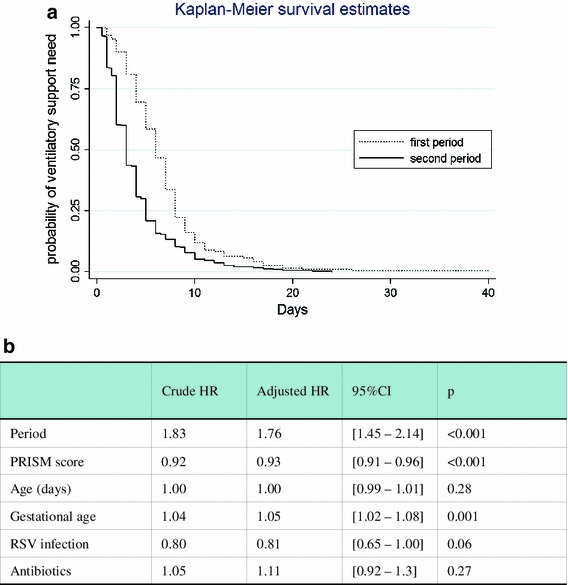
a Probability curves of being on ventilatory support were established on the basis of the whole cohort using the Kaplan–Meier method, then compared across period groups by using the log-rank test. During the second period, there was a significant decrease of length of ventilatory support relative to the first period with an HR (95 % CI) of 1.83 (1.53–2.19). b HRs and 95 % CIs for length of ventilator support in the second period as compared with the first period. This effect remains significant after adjustment for prognostic baseline covariates: PRISM score, age, gestational age, RSV infection, and antibiotics
Furthermore, nCPAP was associated with a low rate of treatment failure between 15 and 3 % in the most recent year, illustrating the success of nCPAP implementation in clinical practice. Even in the case of nCPAP failure during P2, the length of invasive ventilatory support was shorter compared to P1 (Table 2).
Complications and associated therapy during PICU stay
A significant reduction in PICU LOS of 3 days was observed between P1 and P2. Similarly, during P2, there was a significant decrease in adverse ventilation-associated events such as healthcare-associated pneumonia (24 versus 10 %, p < 0.01), and postextubation stridor (39.4 versus 3.3 %, p < 0.01). As shown in Table 3, P2 was associated with significantly less invasive procedures (labs, central venous catheters), chest X-rays, blood transfusions, antibiotic therapy, and parenteral nutrition.
Table 3.
Management in the PICU, therapies and complications
| P1 (n = 193) | P2 (n = 332) | p | |
|---|---|---|---|
| PICU LOS (days), median (IQR) | 9 (8–10) | 5 (4.5–5.5) | <0.0001 |
| Chest X-ray, mean ± SD | 7.7 ± 5.9 | 2 ± 4.1 | <0.0001 |
| Blood labs, mean ± SD | 6.2 ± 7.7 | 2.9 ± 5 | <0.0001 |
| Corticosteroids, n (%) | 35 (17.9) | 49 (14.7) | 0.1 |
| Bronchodilators, n (%) | 97 (50.3) | 42 (12.7) | <0.0001 |
| Antibiotic therapy, n (%) | 111 (57.5) | 98 (29.5) | <0.0001 |
| Central venous catheter, n (%) | 30 (15.5) | 19 (5.7) | <0.0001 |
| Blood transfusion, n (%) | 23 (11.9) | 23 (6.9) | 0.06 |
| Parenteral nutrition, n (%) | 24 (12.4) | 11 (3.3) | <0.001 |
| Nosocomial pneumonia, n (%) | 46 (23.8) | 33 (9.9) | <0.0001 |
| ARDS, n (%) | 18 (9.3) | 15 (4.5) | 0.04 |
| Pneumothorax, n (%) | 2 (1) | 3 (0.9) | 0.98 |
| Death, n (%) | 2 (1) | 3 (0.9) | 0.98 |
PICU LOS pediatric intensive care unit length of stay, ARDS acute respiratory distress syndrome
Five infants died during the study periods, with similar mortality rate between P1 and P2 including two cases of Down syndrome, and one case of CHD, and facial malformation respectively. The main cause of death was hospital-acquired pneumonia with sepsis alteration.
Post PICU outcome
Hospital stay after PICU discharge was significantly reduced during the second period, from 7.4 ± 5.7 to 5 ± 3.9 days, respectively. During P2, we observed a significant decrease in the total hospital LOS of 5.9 days. Moreover, the reduction in PICU LOS was not associated with an increased number of readmissions to the PICU as demonstrated by similar rates of readmission between P1 and P2, (3.1 and 4 % respectively).
Cost assessment
The billing costs used in this study, obtained from the hospital cost accounting reports, were an average of 1,806 € ($2,408) per day of PICU care and 1,198 € ($1,597) per day of general ward care. We did not measure the individual costs. With the reduction in length of both PICU and general pediatric ward stay, we calculated a total cost reduction during P2 of 715,000 € ($950,000) per year and of 3,575,000 € ($4,750,000) for the whole 4-year period (Table 4).
Table 4.
The billing costs obtained from the hospital cost accounting reports
| P1 | P2 | p | |
|---|---|---|---|
| Mean PICU cost (€) | 17,451 ± 9,978 | 11,205 ± 8,296 | <0.001 |
| Mean PICU cost ($) | 23,209 | 14,902 | <0.001 |
| Mean hospital cost (€) | 27,572 ± 12,166 | 16,801 ± 9,642 | <0.001 |
| Mean hospital cost ($) | 36,670 | 22,345 | <0.001 |
For each PICU stay, the billing cost was assessed using the hospital cost accounting reports given per day of stay
Discussion
This study evaluated changes in the management of severe acute bronchiolitis in more than 500 infants over two distinct periods. There is a significant change in the global management of these infants and the major change in practice is the modification of ventilatory support with the use of a pre-emptive nCPAP strategy in severe bronchiolitis. This new management is associated with a significant improvement in supportive care as reflected by a dramatic reduction of the use of invasive mechanical ventilation, length of any respiratory support, PICU and hospital LOS, and economic burden.
Severe acute bronchiolitis is a potentially life-threatening respiratory condition that affects infants in predictable seasonal epidemics. Hospital admission rates in the USA and Europe are reported to be between 20 and 30 per 1,000 for children under 1 year [8–10]. PICU admission rates range from 4 to 15 % for healthy infants and this percentage increases accordingly with the presence of comorbidities [11]. The treatment of severe bronchiolitis is mainly supportive [12]. Invasive ventilation is still frequently used to support these infants, but it is associated with significant airway and infectious adverse events [13]. In observational studies, early use of nCPAP was associated with a significant decrease in both RR and PCO2, suggesting that nCPAP might be an effective ventilation strategy in moderate to severe bronchiolitis with a low rate of failure [14–20]. In addition, nCPAP failure does not seem to be associated with a worse evolution. Moreover, recent physiological studies demonstrated that severe bronchiolitis is associated with increased work of breathing, as reflected by high levels of diaphragmatic and esophageal pressure–time products, and significantly improved with application of a continuous PEEP [7, 21, 22]. To our knowledge, this is the first study which clearly demonstrates the clinical and economic benefit of nCPAP in severe acute bronchiolitis. This large observational cohort study, with more than 300 cases of nCPAP, found that nCPAP is independently associated with a shorter duration of ventilatory support, PICU LOS, and hospital LOS. These findings confirm previous observational studies which showed a similar reduction in time of ventilatory support of 2–3 days [17, 18, 23].
The second period is associated with significant changes in the overall clinical management of infants with acute bronchiolitis. One of the most relevant changes was the significant decrease in the number of chest X-rays performed between the two periods. Although current guidelines do not recommend its use for the diagnosis and follow-up of bronchiolitis, most emergency departments and PICUs are still performing systematic chest X-rays, particularly in the most critically ill patients. Children are four times more sensitive than adults to the ionizing radiation with an accordingly elevated risk of delayed cancer, given their more active cell proliferation. Shu et al. [24] reported that children with three or more postnatal X-rays had a 1.5–2 times higher risk of cancer compared to those who were never exposed.
Apart from affecting PICU and hospital LOS, we noted that several associated therapies such antibiotics, chest X-rays, number of labs drawn, use of central venous catheters, blood transfusions, and parenteral nutrition were significantly reduced during the second period. Bronchiolitis with fever has been associated with a lower risk of bacteremia (0.2 %) compared to children with a fever of unknown origin (2–7 %) [25]. Although antimicrobial therapy is no longer recommended for bronchiolitis, it is commonly used in hospitalized children, with a rate varying widely between 34 and 99 % [26, 27]. In addition, infants with bronchiolitis requiring invasive mechanical ventilation have high rates of bacterial co-infection varying from 21 to 26 % [28, 29] and Kneyber et al. [29] reported antibiotic usage reaching 95 % in these setting. In our series, the decrease in antibiotics use from 58 to 30 %, between the two periods, is partly due to increased physician awareness of the potential harmful effects of antibiotic selection pressure. Nevertheless, we could not exclude a relationship between increasing use of nCPAP as a standard of care and the decrease in healthcare-associated pneumonia from 24 to 10 % [26]. Similarly, in adults, non-invasive ventilation (NIV) was shown to decrease significantly the rate of nosocomial bacterial pneumonia from 20 to 8 % [30]. This study highlights the fact that although there has been a change in the approach to ventilator support over time, there has also been a significant change in many aspects of the overall management of PICU patients between those two decades that can moderate the conclusion.
Bronchiolitis is known to be the leading cause of hospitalization for infants less than 1 year old, but data on the economic burden of bronchiolitis hospitalizations are limited. In 2002, in the USA, bronchiolitis hospitalizations accounted for 21.1 % of children less than 1 year old with an annual direct medical cost of approximately $543 million. Younger age, male gender, mechanical ventilation, and co-diagnosis of pneumonia were all markers predictive of higher hospitalization cost and longer hospital LOS [31]. The use of NIV in adults with acute exacerbation of chronic obstructive disease significantly increased between 1998 and 2008 and was associated with clinical improvement and decrease in LOS [32]. To our knowledge, our study is the first to evaluate the economic impact of nCPAP in children. ICU admissions for severe bronchiolitis comprise only a small proportion of all bronchiolitis cases, but account for a larger proportion of all bronchiolitis-related hospitalization charges. In the USA, some authors demonstrate that bronchiolitis managed with invasive mechanical ventilation is associated with costs 6–9 times higher than when managed without any ventilatory support [33–35]. In this study, we further demonstrate that nCPAP is a cost-effective strategy with a trend to reduction of the total economic burden, mostly owing to a significant decrease in PICU and hospital LOS as actual individual costs could not be measured.
We are aware that the before–after study design limits the ability to assess whether nCPAP as the first ventilatory support therapy is superior to invasive ventilation and it was not the objective. Thus, this large descriptive study, even with some methodological limitations, highlights that nCPAP used as a pre-emptive treatment strategy is associated with significant reduction of the percentage of invasive ventilation requirement and treatment invasiveness. The percentage of patients without any ventilatory support remains stable over the whole period, before the use of nCPAP and after the implementation of nCPAP as a pre-emptive ventilatory strategy in our unit (Fig. 1). This suggests that the nCPAP was not used for infants who in fact did not require any ventilatory support and allowed us to suggest that nCPAP is associated with a reduction of the total economic burden of severe bronchiolitis. We chose to analyze this effect in two different periods because, as shown in Fig. 1, invasive ventilation was exclusive before 2000 and nCPAP was predominant after 2006, whereas between 2000 and 2006 none of these support was predominant.
Conclusion
This is the first large study describing the cost-effectiveness of nCPAP in the management of severe acute bronchiolitis. Thus, it appears that the new global management with the early use of nCPAP is associated with a clear reduction in treatment invasiveness and complications, as well as a significant reduction in the economic burden of severe bronchiolitis. This descriptive study is a first step and highlights the need for further prospective controlled studies.
Acknowledgment
This project was done with no specific support.
Conflicts of interest
The authors have no conflicts of interest to disclose.
Footnotes
Take-home message: nCPAP as the primary ventilatory support in severe bronchiolitis is associated with a significant decrease of treatment invasiveness, PICU and hospital length of stays, and economic burden.
References
- 1.Simoes EA. Respiratory syncytial virus infection. Lancet. 1999;354(9181):847–852. doi: 10.1016/S0140-6736(99)80040-3. [DOI] [PubMed] [Google Scholar]
- 2.Simoes EA. Respiratory syncytial virus and subsequent lower respiratory tract infections in developing countries: a new twist to an old virus. J Pediatr. 1999;135(6):657–661. doi: 10.1016/S0022-3476(99)70079-X. [DOI] [PubMed] [Google Scholar]
- 3.Greenough A. Role of ventilation in RSV disease: CPAP, ventilation, HFO, ECMO. Paediatr Respir Rev. 2009;10(Suppl 1):26–28. doi: 10.1016/S1526-0542(09)70012-0. [DOI] [PubMed] [Google Scholar]
- 4.Shay DK, et al. Bronchiolitis-associated hospitalizations among US children, 1980–1996. JAMA. 1999;282(15):1440–1446. doi: 10.1001/jama.282.15.1440. [DOI] [PubMed] [Google Scholar]
- 5.Leader S, Kohlhase K. Recent trends in severe respiratory syncytial virus (RSV) among US infants, 1997 to 2000. J Pediatr. 2003;143(5 Suppl):S127–S132. doi: 10.1067/S0022-3476(03)00510-9. [DOI] [PubMed] [Google Scholar]
- 6.Jobe AH, Bancalari E. Bronchopulmonary dysplasia. Am J Respir Crit Care Med. 2001;163(7):1723–1729. doi: 10.1164/ajrccm.163.7.2011060. [DOI] [PubMed] [Google Scholar]
- 7.Essouri S, et al. Optimal level of nasal continuous positive airway pressure in severe viral bronchiolitis. Intensive Care Med. 2011;37(12):2002–2007. doi: 10.1007/s00134-011-2372-4. [DOI] [PubMed] [Google Scholar]
- 8.Levine DA, et al. Risk of serious bacterial infection in young febrile infants with respiratory syncytial virus infections. Pediatrics. 2004;113(6):1728–1734. doi: 10.1542/peds.113.6.1728. [DOI] [PubMed] [Google Scholar]
- 9.Simoes EA, Carbonell-Estrany X. Impact of severe disease caused by respiratory syncytial virus in children living in developed countries. Pediatr Infect Dis J. 2003;22(2 Suppl):S13–S18. doi: 10.1097/01.inf.0000053881.47279.d9. [DOI] [PubMed] [Google Scholar]
- 10.Deshpande SA, Northern V. The clinical and health economic burden of respiratory syncytial virus disease among children under 2 years of age in a defined geographical area. Arch Dis Child. 2003;88(12):1065–1069. doi: 10.1136/adc.88.12.1065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lebel MH, et al. Respiratory failure and mechanical ventilation in severe bronchiolitis. Arch Dis Child. 1989;64(10):1431–1437. doi: 10.1136/adc.64.10.1431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Smyth RL, Openshaw PJ. Bronchiolitis. Lancet. 2006;368(9532):312–322. doi: 10.1016/S0140-6736(06)69077-6. [DOI] [PubMed] [Google Scholar]
- 13.Jorgensen J, et al. Incidence of and risk factors for airway complications following endotracheal intubation for bronchiolitis. Otolaryngol Head Neck Surg. 2007;137(3):394–399. doi: 10.1016/j.otohns.2007.03.041. [DOI] [PubMed] [Google Scholar]
- 14.Beasley JM, Jones SE. Continuous positive airway pressure in bronchiolitis. Br Med J (Clin Res Ed) 1981;283(6305):1506–1508. doi: 10.1136/bmj.283.6305.1506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Soong WJ, Hwang B, Tang RB. Continuous positive airway pressure by nasal prongs in bronchiolitis. Pediatr Pulmonol. 1993;16(3):163–166. doi: 10.1002/ppul.1950160305. [DOI] [PubMed] [Google Scholar]
- 16.Thia LP, et al. Randomised controlled trial of nasal continuous positive airways pressure (CPAP) in bronchiolitis. Arch Dis Child. 2008;93(1):45–47. doi: 10.1136/adc.2005.091231. [DOI] [PubMed] [Google Scholar]
- 17.Campion A, et al. Non-invasive ventilation in infants with severe infection presumably due to respiratory syncytial virus: feasibility and failure criteria. Arch Pediatr. 2006;13(11):1404–1409. doi: 10.1016/j.arcped.2006.08.003. [DOI] [PubMed] [Google Scholar]
- 18.Larrar S, et al. Effects of nasal continuous positive airway pressure ventilation in infants with severe acute bronchiolitis. Arch Pediatr. 2006;13(11):1397–1403. doi: 10.1016/j.arcped.2006.07.005. [DOI] [PubMed] [Google Scholar]
- 19.Ganu SS, et al. Increase in use of non-invasive ventilation for infants with severe bronchiolitis is associated with decline in intubation rates over a decade. Intensive Care Med. 2012;38(7):1177–1183. doi: 10.1007/s00134-012-2566-4. [DOI] [PubMed] [Google Scholar]
- 20.Cavari Y, et al. Non invasive positive pressure ventilation in infants with respiratory failure. Pediatr Pulmonol. 2012;47(10):1019–1025. doi: 10.1002/ppul.22561. [DOI] [PubMed] [Google Scholar]
- 21.Cambonie G, et al. Nasal continuous positive airway pressure decreases respiratory muscles overload in young infants with severe acute viral bronchiolitis. Intensive Care Med. 2008;34(10):1865–1872. doi: 10.1007/s00134-008-1201-x. [DOI] [PubMed] [Google Scholar]
- 22.Milesi C, et al. Is treatment with a high flow nasal cannula effective in acute viral bronchiolitis? A physiologic study. Intensive Care Med. 2013;39(6):1088–1094. doi: 10.1007/s00134-013-2879-y. [DOI] [PubMed] [Google Scholar]
- 23.Javouhey E, et al. Non-invasive ventilation as primary ventilatory support for infants with severe bronchiolitis. Intensive Care Med. 2008;34(9):1608–1614. doi: 10.1007/s00134-008-1150-4. [DOI] [PubMed] [Google Scholar]
- 24.Shu XO, et al. Association of paternal diagnostic X-ray exposure with risk of infant leukemia. Investigators of the Childrens Cancer Group. Cancer Epidemiol Biomarkers Prev. 1994;3(8):645–653. [PubMed] [Google Scholar]
- 25.Greenes DS, Harper MB. Low risk of bacteremia in febrile children with recognizable viral syndromes. Pediatr Infect Dis J. 1999;18(3):258–261. doi: 10.1097/00006454-199903000-00010. [DOI] [PubMed] [Google Scholar]
- 26.Vogel AM, et al. Variations in bronchiolitis management between five New Zealand hospitals: can we do better? J Paediatr Child Health. 2003;39(1):40–45. doi: 10.1046/j.1440-1754.2003.00069.x. [DOI] [PubMed] [Google Scholar]
- 27.Kabir ML, et al. Evaluation of hospitalized infants and young children with bronchiolitis—a multi centre study. Mymensingh Med J. 2003;12(2):128–133. [PubMed] [Google Scholar]
- 28.Thorburn K, et al. High incidence of pulmonary bacterial co-infection in children with severe respiratory syncytial virus (RSV) bronchiolitis. Thorax. 2006;61(7):611–615. doi: 10.1136/thx.2005.048397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kneyber MC, et al. Concurrent bacterial infection and prolonged mechanical ventilation in infants with respiratory syncytial virus lower respiratory tract disease. Intensive Care Med. 2005;31(5):680–685. doi: 10.1007/s00134-005-2614-4. [DOI] [PubMed] [Google Scholar]
- 30.Girou E, et al. Secular trends in nosocomial infections and mortality associated with noninvasive ventilation in patients with exacerbation of COPD and pulmonary edema. JAMA. 2003;290(22):2985–2991. doi: 10.1001/jama.290.22.2985. [DOI] [PubMed] [Google Scholar]
- 31.Pelletier AJ, Mansbach JM, Camargo CA., Jr Direct medical costs of bronchiolitis hospitalizations in the United States. Pediatrics. 2006;118(6):2418–2423. doi: 10.1542/peds.2006-1193. [DOI] [PubMed] [Google Scholar]
- 32.Chandra D, et al. Outcomes of noninvasive ventilation for acute exacerbations of chronic obstructive pulmonary disease in the United States, 1998–2008. Am J Respir Crit Care Med. 2012;185(2):152–159. doi: 10.1164/rccm.201106-1094OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Willson DF, Landrigan CP, Horn SD, Smout RJ. Complications in infants hospitalized for bronchiolitis or respiratory syncytial virus pneumonia. J Pediatr. 2003;143(Suppl 5):S142–S149. doi: 10.1067/S0022-3476(03)00514-6. [DOI] [PubMed] [Google Scholar]
- 34.Howard TS, Hoffman LH, Stang PE, Simoes EA. Respiratory syncytial virus pneumonia in the hospital setting: length of stay, charges, and mortality. J Pediatr. 2000;137(2):227–232. doi: 10.1067/mpd.2000.107525. [DOI] [PubMed] [Google Scholar]
- 35.Katz B, Lo J, Sorrentino M. Costs of respiratory syncytial virus infection at a tertiary care children’s hospital. Pharm Ther. 2003;28(5):343–345. [Google Scholar]


