Abstract
Infectious diseases and their pandemics periodically attract public interests due to difficulty in treating the patients and the consequent high mortality. Sepsis caused by an imbalanced systemic inflammatory response to infection often leads to organ failure and death. The current therapeutic intervention mainly includes “the sepsis bundles,” antibiotics (antibacterial, antiviral, and antifungal), intravenous fluids for resuscitation, and surgery, which have significantly improved the clinical outcomes in past decades; however, the patients with fulminant sepsis are still in desperate need of alternative therapeutic approaches. One of the potential supportive therapies, extracorporeal blood treatment, has emerged and been developed for improving the current therapeutic efficacy. Here, I overview how the treatment of infectious diseases has been assisted with the extracorporeal adjuvant therapy and the potential utility of various nanobiotechnology and microfluidic approaches for developing new auxiliary therapeutic methods.
Keywords: Microfluidics, Nanobiotechnology, Extracorporeal devices, Infectious diseases, Blood treatment, Sepsis
Introduction
Sepsis, a systemic inflammatory disease, is caused by an imbalanced overwhelming systemic inflammatory response to infection, which often leads to organ failure and death. Because it progresses rapidly while the determination of a cause of infection takes hours to days, the current state of the art treatment of sepsis in hospitals requires careful assessment of patients’ conditions, followed by proper treatment based on the “sepsis bundle”1. It was not until the 20th century that Hugo Schottmüller2 and Roger Bone3 proposed the modern clinical definition of sepsis. Before this era, sepsis was regarded as severe biological decay that may accompany fevers4 or a syndrome that might have resulted from invisible living creatures5,6.
This new foundation for sepsis has shifted the direction of the sepsis research from a source of infection to the host inflammatory response, which has led considerable efforts in drug development for anti- inflammatory therapies. Xigris, for example, a recombinant form of human activated protein C that has anticoagulative and cytoprotective effects was developed7. After extensive clinical research that reported no efficacy of Xigris for treating septic patients8, Eli Lilly and Co. withdrew Xigris from the market9. Despite the lack of proper drugs that effectively mitigate sepsis, the mortality of sepsis has gradually reduced thanks to consolidated efforts of clinicians, such as the Surviving Sepsis Campaign Guidelines, which was recently updated in 20181. However, we are still in desperate need of developing a new strategy to fight against infectious outbreaks worldwide whenever infectious diseases, such as severe acute respiratory syndrome (SARS)10, Ebola11, middle east respiratory syndrome (MERS)12, surfaced and threatened the world. In the past five years, we experienced multiple severe outbreaks of Ebola (2014-2016 in West Africa), MERS (2015 in the Republic of Korea), and the recent coronavirus outbreak (COVID-19 in Wuhan, China). However, we mainly rely on taking preventative measures against epidemics due to the lack of proper therapeutic strategy in handling severe septic conditions.
Because sepsis is a complicated and dynamic syndrome that involves enormous numbers of parameters that regulate the inflammatory network, it was challenging to inhibit a particular pathway because the disease can still progress by detouring the inhibited pathways as we have already witnessed the case of Xigris. Thus, some of the researchers and clinicians have paid attention to removing a source of infection that persistently triggered a catastrophic inflammatory response for developing an adjuvant therapy. This idea was evidenced by several clinical studies supporting that viral and bacterial loads in the blood are a major contributor to the severity and mortality of sepsis13,14. Due to the rapid progress of sepsis and a high frequency (~70%) of negative results of blood culture in septic human blood15, identification of a source of infection in time is often elusive even in state-of-the-art clinical settings. For this, an auxiliary therapeutic approach could be a preferable treatment option for clinicians while the conventional “sepsisbundle” procedures are mitigating symptoms of severe septic patients. This has led to developing several medical devices that have been available in the market for the past decades. This review will mainly scrutinize extracorporeal blood treatment approaches for treating sepsis and how the researchers in the interdisciplinary fields of nanobiotechnology and biochips could make potential contributions to improving the efficacy of the biomedical devices addressing the unmet needs in sepsis treatment.
Pathogen-capturing Reagents Immobilized on Fiber Surface
Efforts first dedicated to removing infectious reagents in whole blood of septic patients were developed by modifying the conventional extracorporeal blood treatment devices, such as hemofiltration devices. The inner surface of the fiber bundles of the device was functionalized with synthetic proteins16 or antibiotics17 that were known to capture a range of Gram-negative pathogens or lipopolysaccharides (LPS) (Figure 1A). The fibers of Toraymyxin® and oXiris®, for example, are functionalized with polymyxin B (antibiotics) and polyethyleneimine (PEI) (positively charged molecule), respectively, which are known to bind Gram-negative bacteria and LPS (negatively charged moiety). This approach has been extensively attempted in clinical trials as well as in large animal models because it can be simply integrated with the existing blood filtration devices that had already been approved for treating patients with organ failures. However, the devices based on this principle were more effective in depleting free LPS or pathogen debris probably caused by antibiotic treatment18 than removing intact bacteria that were planktonic in whole blood. This is most likely due to the low binding efficiency of relatively large bacterial cells (~ 1–2 µm in diameter) in comparison to submicrometer-sized LPS molecules. If we consider only stochastic movements of pathogenic materials and capturing matrix, the binding efficiency of planktonic particles in a solution to the capturing matrix is determined by the thermal fluctuation (kd) and collisions caused by shear (kshear)19. Because the collision rate constant of diffusion, kd, is given by
| 1 |
where rc is the radius of a pathogen (~ 0.5 µm), and rb is the radius of a capturing particle, kb is the Boltzmann constant, T is the temperature (K), and η is the fluid viscosity (N s m−2), the binding efficiency of free bacteria to a solid wall surface via thermal fluctuation where rb approaches an infinite radius becomes negligible. Thus, physical contact of bacterial cells to the surface is an essential prerequisite to achieve the complete removal of pathogens from blood in this principle.
Figure 1.
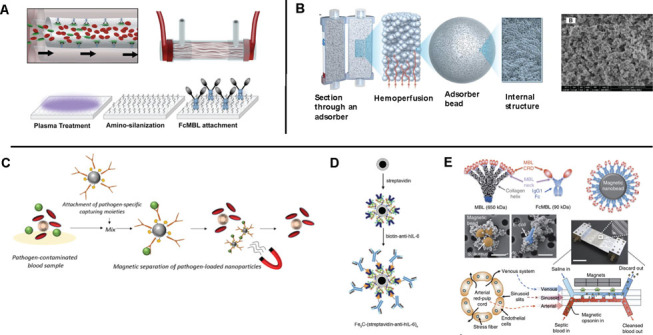
(A) A schematic drawing of surface adsorption-based extracorporeal devices. The inner surface of fibers bundled in the device is functionalized with proteins or synthetic polymers, which are known to capture a wide range of pathogens and toxins in blood18. (B) An illustration showing a cross-sectional view of porous beads and their internal structures of the CytoSorb® filter, which was developed for lowering cytokine levels in blood of septic patients20. (C, D, E) Magnetic particle-assisted blood treatment approaches. (C) Magnetic particles are functionalized with pathogen-capturing moieties21. (D) Magnetic particles coated with anti-IL6 was developed for removing a proinflammatory cytokine in blood22. (E) An engineered human FcMBL molecule conjugated on magnetic nanoparticles where the FcMBL molecules were designed to be oriented with carbohydrate recognition domains facing outward23.
Moreover, the efficiency of capturing LPS in blood should be significantly higher than whole bacteria because the frequency of LPS encountering the surface is greater than individual intact bacterial cells due to their enormous numbers after being scattered by bactericidal antibiotics. A report published by researchers at Harvard University also demonstrated that the use of antibiotics significantly increases the depletion efficiency of pathogen-associated molecular patterns (PAMPs) in a septic rodent animal model18. Thus, it is recommended to use the LPS adsorbing devices, particularly along with antibiotic treatment. However, in the case of infection caused by multidrug-resistant (MDR) or pan-drug resistant (PDR) bacteria where the use of antibiotics is often ineffective, this approach may not be a preferable strategy.
Porous Polymer Beads Absorbing Cytokines
The severity of sepsis is often driven by overwhelming amounts of cytokines produced throughout the body, the cytokine storm. Cytokines are a broad category of small proteins responsible for cell to cell signaling (< 40 kDa), and their productions and surges in the host immune system are asynchronous. Because a variety of cytokines are responsible for the rapid progress of sepsis, an idea of reducing levels of all small molecules in septic blood was set out to develop for treating septic patients. Hemofiltration (HF) was initially applied to lowering the concentration of cytokines; however, due to the relatively inefficient rate in depleting cytokines, the clinical trial did not find a significantly improved outcome for treating septic patients24. After a septic animal study in rats showed that physical adsorption of cytokines (IL-6) to HF filter surfaces was attributed to reducing cytokine levels in blood25, the many researchers started to investigate how to maximize physical adsorption of cytokines in an extracorporeal circuit while demonstating their prototype devices in a small animal model26 and developing mathematical models predicting the cytokine removal efficiency27. The most well-known product based on this principle is Cyto-Sorb® in which polymer beads with large amounts of the porous cavity in a range of 250 Angstroms to about 2000 Angstroms28 hold up small molecules, such as cytokines, while whole septic blood flows through the device (Figure 1B). However, because pro-inflammatory (IL-1, IL-12, IL-18, TNF-α, IFN-γ) and anti-inflammatory (IL-4, IL-6, IL-10, IL-10, IL-13) cytokines finely orchestrate complicated molecular cascades of responses to infection, therapeutic strategies aiming to reduce cytokine levels in blood are still disputable. One of the challenges in this approach is to find the right time to treat patients with adsorbing filters because levels of various cytokines in patients are dynamic and often unpredictable29.
Magnetic Nanoparticle-assisted Blood Cleansing Devices
Magnetic separation is a classical isolation method for harvesting a particular fraction of cells, molecules, or impurity from heterogeneous mixtures, such as whole blood30, various biological liquid samples31–33, and nanotube solutions34. This principle has been applied to reducing pathogen levels in whole blood circulating in an extracorporeal circuit in vitro and in vivo animal models (Figure 1C-E). Advantages of the magnetic particles-based blood treatment methods over other competitors described above include that it offers the considerably higher collision rate constant of diffusion (kd, Equation (1)) and the larger active surface areas (Acap) capturing pathogens (kd = 10−17~ 10−18 m3/s, Acap = ~10~102 m2/g)19,35 in comparison to the surface-adhesion-based bacterial capture method (kd→0, Acap = 4~8 m2)36. It can also continuously feed fresh capturing reagents into the bloodstream at every moment instead of repassing blood through the capturing surface matrix multiple times, which may gradually compromise the capturing ability. There are several parameters in magnetic blood treatment that determine the pathogen depletion efficiency in septic blood; 1) opsonin molecules that capture a broad range of pathogens without having to carry out prior identification of pathogens, 2) sizes and concentrations of magnetic particles, 3) magnetic forces exerted on the magnetic particle-bacteria complexes, and 4) a design and dimensions of microfluidic channels. Because there is a high degree of variety of carbohydrate chains or proteins expressed on bacteria, viruses, and fungi, it has been challenging to develop a generic opsonin molecule that targets a wide range of pathogens. Researchers initially employed antibody molecules or metal chelators conjugated on magnetic particles to deplete a particular microorganism37,38, metal ions (Pb2+), drugs, and cytokines (IL-6)22 in blood (Figure 1E). These approaches would be practical when we could have beforehand identified a source of infection or intoxication; however, in most infection cases, instantaneous identification of causative pathogens is a daunting task. To address this, Lee, J.-J. et al. developed synthetic ligand zinc-coordinated bis(dipicolylamine) (bis-Zn-DPA)-coated magnetic nanoparticles, which was supposed to form bonds with anionic phospholipids present on the outer surface of both Gram-positive and Gram-negative bacteria (Figure 2A). They also demonstrated its utility using Gram-negative bacteria, E. coli, bound with bis-Zn-DPA-coated magnetic nanoparticles39. The engineered human opsonin molecule (Fc-mannose binding lectin, FcMBL) was also demonstrated to deplete a broad range of pathogens in whole blood (Figure 2A)23. The second technical issue is how to optimize the size and concentrations of magnetic nanoparticles to maximize the efficiency of the blood cleansing system. Kang, J.H. et al. developed a theoretical model combining an orthokinetic collision model and a magnetophoretic model to predict the optimal size and concentrations of the magnetic nanoparticles that yield the most efficient depletion of pathogens in a buffer solution, plasma, and whole blood, respectively (Figure 2B).19 Besides, the magnetic force exerted on the magnetic particles and their complexes bound with pathogens is a determinant of magnetic separation, which is proportional to magnetic flux density gradients along the direction of magnetic pulling. Although an electromagnetic system to pull out magnetic particles offers simplicity for switching the magnetic forces on and off, it appears unfavorable for this particular application because of the limited magnetic flux density gradients due to electromagnetic heating40. The permanent magnets assembled with alternating polarization generate the highly enhanced magnetic flux density gradients at a gap adjacent to two assembled magnets23. Recently, the Halbach magnetic array was experimentally demonstrated to manipulate magnetic nanoparticles at distances up to 18 mm and wash magnetic particles used in a multi-step-immunoassay process in a deep well plate41 (Figure 2C, i). The ferromagnetic microstructures also have been employed to augment the magnetic flux density gradients to manipulate superparamagnetic nanoparticles that exhibit relatively weak magnetic moments (Figure 2C, ii–iii)34,42. Finally, the demonstration of a new design principle for microfluidic channels to remove magnetic particles more effectively in viscous fluids was successfully implemented where secondary spiral flows combined with magnetophoresis significantly enhanced the magnetic separation efficiency even in undiluted whole blood in a continuous manner (Figure 2D) 43. Given the recent progress and persistent efforts dedicated to magnetically removing pathogens from whole blood, the magnetic separation principle could be one day incorporated in a biomedical device from which many septic patients in dire need benefit in the near future.
Figure 2.
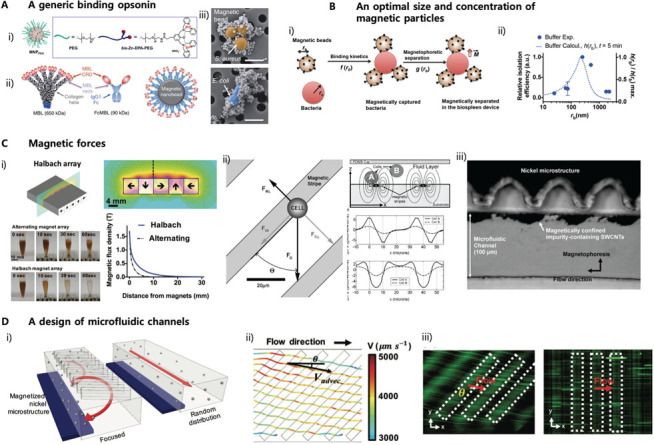
Experimental parameters that determine the magnetic depletion efficiency of pathogens in blood. (A) An opsonin molecule that captures a wide range of pathogens, including Gram-positive, Gram-negative, fungi, and viruses. i) zinc-coordinated bis(dipicolylamine) (bis-Zn-DPA) coated on magnetic particles forms coordination bonds with anionic phospholipids present on pathogen surfaces39. ii) An engineered human C-type lectin, mannose-binding lectin (MBL), fused to human IgG1 Fc (FcMBL) 23. iii) Pseudo-colored SEM images of S. aureus (upper) and E. coli (bottom) captured by magnetic nanoparticles coated with Fc-MBL23. (B) i) An optimal size and concentration of magnetic particles was predicted by a theoretical model combining an orthokinetic collision and a magnetophoretic model19. ii) The theoretical prediction (dotted line) was corroborated by the experimental results (dark circles) 19. (C) The magnetic forces acting on magnetic particles can be improved by assembling permanent magnets with polarization angles varying clockwise by 90°. (Halbach arrays)41 or ii) integrating ferromagnetic microstructures to enhance the magnetic flux density gradients42. iii) The nickel microstructures allows carbon nanotubes containing superparamagnetic impurity to be selectively separated in the microfluidic channel when exposed to external magnetic fields34. (D) i) Slanted obstacle arrays patterned in a microfluidic channels induce asymmetric pressure gradients across the channel44,45, which results in secondary lateral flows dragging the magnetic particles in spiral directions43. ii) COMSOL Multiphysics® simulation predicted the deflected paths of the magnetic particles, which correspond to the experimental results (iii, left panel) 43.
Commercial Products Available in the Market and Tested in Clinical Trials
The extracorporeal biomedical devices that have acquired the approval worldwide for treating septic patients predominantly base on the surface-adsorption principles. (Table 1) Due to their variable and inconclusive therapeutic efficacy, the cost covered by insurance supported either by government or private sectors has regional differences; for instance, in the Republic of Korea, use of the ToraymyxinTM filters is not currently subsidized by general health insurance carriers, while in Japan, it can be reimbursed by the Japanese national health insurance since 199417. However, despite insufficient promising results of adjunctive extracorporeal treatment obtained by randomized controlled trials (RCTs), sporadic clinical studies showing positive efficacy of treating septic patients in several cases of the clinical studies46,47 keep the devices being used in an intensive care unit. One of the difficulties in pursuing RCTs of extracorporeal devices treating sepsis is the lack of patients who can be involved in the clinical trials, but this could be resolved when the therapeutic efficacy becomes notably outstanding without controversy. Besides the products shown in Table 1, several startup companies48,49 have been striving to bring their technologies to the bedside. (Table 1)
Table 1.
A summary of extracorporeal devices for treating sepsis.
| Product Name | Principle | Company | Country | Targets of removal |
|---|---|---|---|---|
| oXiris® | Adsorption | Baxter Int. Inc. | USA | Endotoxins, cytokines, uremic toxin |
| Toraymyxin™ | Toray Industries | Japan | Gram-negative bacteria, endotoxin | |
| Alteco® LPS Adsorber | Alteco Medical AB | Sweden | Endotoxin | |
| Seraph® | ExThera Medical | USA | Bacteria (MRSA, E.coli, ESBL-K. pneumoniae, VRE, etc.) | |
| Cytosorb® | CytoSorbents | USA | Cytokines | |
| Under development | BOA Biomedical Inc.* | USA | A broad range of pathogens | |
| Under development | Magnetic particle separation | hemotune* | Switzerland | Endotoxin, cytokines, drugs, heavy metals |
* Startup company
Technical and Experimental Challenges
The most crucial role of the extracorporeal adjuvant therapy is to lower PAMPs levels in whole blood. However, despite the use of the endotoxin activity assay (EAATM) since 200450, there is no such a tool approved worldwide for measuring PAMPs levels in human whole blood yet at this moment. Moreover, recent studies reporting the sequencing results of pathogens in septic patient blood reveal that genome sequencing of pathogens in septic blood retains a considerable frequency of false outcomes, particularly when their concentrations are low51. Because depletion of PAMPs in blood in extracorporeal therapy varies with the effectiveness of antibiotics18, the development of rapid diagnostic tools for antibiotic susceptibility test is also required52. Thus, the diagnostic techniques and their capability should be preeminently revolutionized for clinicians to achieve successful sepsis therapy. As for the magnetic particles-based blood-cleansing approach, the biocompatibility of the particles is the priority to address the safety issue in a phase I clinical trial. As the size of magnetic particles approaches to a nanoscale regime, the magnetic removal efficiency of nanoparticles turns out problematic, and their cytotoxicity becomes even more significant. To this end, Hermann et al. demonstrated that carbon-coated magnetic particles might not induce toxicity in a long-term exposure study when the particles were delivered intravenously53. Similar approaches where magnetic particles are decorated with various functional materials are encouraged to pursue to address the unmet needs in this particular application in the future. Rapid assessment of residual magnetic nanoparticles remaining in whole blood after blood-cleansing treatment is also critical to ensure that a minimal amount of the nanoparticles return to the patients. However, the currently available tools to quantitate the magnetic nanoparticles or measure the magnetic susceptibility of whole blood require dedicated equipment, such as a superconducting quantum interference device (SQUID) or a vibrating sample magnetometer (VSM), which are often unavailable in a standard research laboratory. Jang, B.H. et al. developed a microfluidic device where they can rapidly assess magnetic particle concentrations in a range of 0.01 mg/mL to 5 mg/mL by measuring the diamagnetic repelling velocity of polystyrene microspheres.54 Also, Bougas, L. et al. reported a method of detecting sub-picomolar magnetic nanoparticles in flowing complex fluids55, which would be useful when integrated with the extracorporeal device to monitor low amounts of the magnetic particles returning to the patients.
While most extracorporeal blood treatment methods have targeted lowering endotoxin or cytokine levels in blood of the septic patients, extracorporeal removal of viral particles has rarely been explored56. Although the previously reported opsonin molecule, FcMBL23, is known to bind several virus particles, no data clearly demonstrating the depletion of viruses in human blood are available in public yet. Thus, additional efforts for proving the utility of extracorporeal devices decreasing viral particle concentrations in blood are also required to be established.
Most in vivo animal studies have been demonstrated in rodent sepsis models, including cecal ligation and puncture (CLP) and intravenous or intraperitoneal infection. Before bringing the developed technology to clinical trials, it is essential to test the efficacy in a large animal model, such as swine57, rabbit, or baboon58. However, the current animal models provide poor predictability of human clinical outcomes58. Although the current sepsis animal models do not represent pathophysiological outcomes of human septic patients, fortunately, we could still utilize the animal models for developing the extracorporeal blood treatment devices. This is because we only assess a degree of pathogen levels depleted in the blood of septic animals, which may not much differ from the depletion efficiency in human blood, rather than studying pathophysiological or pharmacokinetic studies.
Summary and Outlook
The research resources and efforts that have been made for studying cancer, cardiovascular diseases, and other diseases have always outweighed those dedicated to infectious diseases. Moreover, the development processes of conventional extracorporeal devices did not actively involve interdisciplinary efforts in the past. As we overviewed above, there are many technical challenges that researchers in interdisciplinary fields ranging from medicine and biology to nanotechnology, polymer sciences, and complex fluid dynamics, could work together to revolutionize the current extracorporeal therapeutic strategies. Besides removing pathogenic materials from blood, elimination of activated leukocytes may have positive impacts on therapeutic efficacy59; however, more fundamental and clinical evaluations are required to follow because the detailed primary mechanisms and their consequences governing sepsis progression remain largely unknown16. It is also important to note that once proving their therapeutic efficiency in RCTs, it would be worth exploring whether the extracorporeal therapeutic scenario could not only help decrease the mortality of septic patients but also improve the post-therapeutic life quality because about half of the survivors of sepsis suffer from the post-sepsis syndrome60.
Acknowledgements
This work was supported by a grant funded by Samsung Research Funding and Incubation Center for Future Technology (SRFC-IT16 02-02), and the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (Grant No. NRF-2019R1C1C1006124).
Conflict of Interests The authors declare no competing financial interests.
References
- 1.Levy MM, Evans LE, Rhodes A. The surviving sepsis campaign bundle: 2018 update. Intensive Care Med. 2018;44:925–928. doi: 10.1007/s00134-018-5085-0. [DOI] [PubMed] [Google Scholar]
- 2.Gary T, Mingle D, Yenamandra A. The evolving definition of sepsis. Int. Clin. Pathol. J. 2016;2:154–159. [Google Scholar]
- 3.Bone RC, Balk RA, Cerra FB, Dellinger RP, Fein AM, Knaus WA, Schein R, Sibbald W J. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. CHEST Journal. 1992;101:1644–1655. doi: 10.1378/chest.101.6.1644. [DOI] [PubMed] [Google Scholar]
- 4.Rittirsch D, Flierl MA, Ward PA. Harmful molecular mechanisms in sepsis. Nat. Rev. Immunol. 2008;8:776–787. doi: 10.1038/nri2402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Baron RM, Baron MJ, Perrella MA. Pathobiology of Sepsis: Are We Still Asking the Same Questions? Am. J. Respir. Cell Mol. Biol. 2006;34:129–134. doi: 10.1165/rcmb.F308. [DOI] [PubMed] [Google Scholar]
- 6.Hernandez Botero Johan Sebastian, Florian Perez Maria Cristina. Sepsis - An Ongoing and Significant Challenge. 2012. The History of Sepsis from Ancient Egypt to the XIX Century. [Google Scholar]
- 7.Abraham E, Laterre P-F, Garg R, Levy H, Talwar D, Trzaskoma BL, François B, Guy JS, Brückmann M, Rea-Neto Á, Rossaint R, Perrotin D, Sablotzki A, Arkins N, Utterback BG, Macias WL. Drotrecogin alfa (activated) for adults with severe sepsis and a low risk of death. N. Engl. J. Med. 2005;353:1332–1341. doi: 10.1056/NEJMoa050935. [DOI] [PubMed] [Google Scholar]
- 8.Ranieri VM, Thompson BT, Barie PS, Dhainaut JF, Douglas IS, Finfer S, Gårdlund B, Marshall JC, Rhodes A, Artigas A. Drotrecogin alfa (activated) in adults with septic shock. N. Engl. J. Med. 2012;366:2055–2064. doi: 10.1056/NEJMoa1202290. [DOI] [PubMed] [Google Scholar]
- 9.FDA Drug Safety Communication: Voluntary market withdrawal of Xigris [drotrecogin alfa (activated)] due to failure to show a survival benefit (https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-voluntary-market-withdrawal-xigris-drotrecogin-alfa-activated-due).
- 10.Peiris JSM, Yuen KY, Osterhaus ADME, Stöhr K. The Severe Acute Respiratory Syndrome. N. Engl. J. Med. 2003;349:2431–2441. doi: 10.1056/NEJMra032498. [DOI] [PubMed] [Google Scholar]
- 11.Ilunga Kalenga O, Moeti M, Sparrow A, Nguyen V-K, Lucey D, Ghebreyesus TA. The ongoing Ebola epidemic in the Democratic Republic of Congo, 2018–2019. N. Engl. J. Med. 2019;381:373–383. doi: 10.1056/NEJMsr1904253. [DOI] [PubMed] [Google Scholar]
- 12.Arabi YM, Balkhy HH, Hayden FG, Bouchama A, Luke T, Baillie JK, Al-Omari A, Hajeer AH, Senga M, Denison MR, Nguyen-Van-Tam JS, Shindo N, Bermingham A, Chappell JD, van Kerkhove MD, Fowler RA. Middle East respiratory syndrome. N. Engl. J. Med. 2017;376:584–594. doi: 10.1056/NEJMsr1408795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chuang Y-C, Chang S-C, Wang W-K. High and increasing Oxa-51 DNA load predict mortality in Acinetobacter baumannii bacteremia: implication for pathogenesis and evaluation of therapy. PLoS ONE. 2010;5:e14133. doi: 10.1371/journal.pone.0014133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Towner JS, Rollin PE, Bausch DG, Sanchez A, Crary SM, Vincent M, Lee WF, Spiropoulou CF, Ksiazek TG, Lukwiya M, Kaducu F, Downing R, Nichol ST. Rapid diagnosis of Ebola hemorrhagic fever by reverse transcription-PCR in an outbreak setting and assessment of patient viral load as a predictor of outcome. J. Virol. 2004;78:4330–4341. doi: 10.1128/JVI.78.8.4330-4341.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chung H, Lee JH, Jo YH, Hwang JE, Kim J. Circulating monocyte counts and its impact on outcomes in patients with severe sepsis including septic shock. SHOCK. 2019;51:423–429. doi: 10.1097/SHK.0000000000001193. [DOI] [PubMed] [Google Scholar]
- 16.Monard C, Rimmelé T, Ronco C. Extracorporeal blood purification therapies for sepsis. Blood Purif. 2019;47:1–14. doi: 10.1159/000499520. [DOI] [PubMed] [Google Scholar]
- 17.Shoji H. Extracorporeal endotoxin removal for the treatment of sepsis: endotoxin adsorption cartridge (Toraymyxin) Ther. Apheresis Dial. 2003;7:108–114. doi: 10.1046/j.1526-0968.2003.00005.x. [DOI] [PubMed] [Google Scholar]
- 18.Didar TF, Cartwright MJ, Rottman M, Graveline AR, Gamini N, Watters AL, Leslie DC, Mammoto T, Rodas MJ, Kang JH, Waterhouse A, Seiler BT, Lombardo P, Qendro EI, Super M, Ingber DE. Improved treatment of systemic blood infections using antibiotics with extracorporeal opsonin hemoadsorption. Biomaterials. 2015;67:382–392. doi: 10.1016/j.biomaterials.2015.07.046. [DOI] [PubMed] [Google Scholar]
- 19.Kang JH, Um E, Diaz A, Driscoll H, Rodas M J, Domansky K, Watters AL, Super M, Stone HA, Ingber DE. Optimization of pathogen capture in flowing fluids with magnetic nanoparticles. Small. 2015;11:5657–5666. doi: 10.1002/smll.201501820. [DOI] [PubMed] [Google Scholar]
- 20.Cytosorbents-How it works, https://www.mddionline.com/file/cytosorbents-how-it-worksjpg.
- 21.Herrmann IK, Schlegel AA, Graf R, Stark WJ, Beck-Schimmer B. Magnetic separation-based blood purification: a promising new approach for the removal of disease-causing compounds? J. Nano-biotechnol. 2015;13:49. doi: 10.1186/s12951-015-0110-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Herrmann IK, Urner M, Koehler FM, Hasler M, Roth-Z’Graggen B, Grass RN, Ziegler U, Beck-Schimmer B, Stark WJ. Blood purification using functionalized core/shell nanomagnets. Small. 2010;6:1388–1392. doi: 10.1002/smll.201000438. [DOI] [PubMed] [Google Scholar]
- 23.Kang JH, Super M, Yung CW, Cooper RM, Domansky K, Graveline AR, Mammoto T, Berthet JB, Tobin H, Cartwright MJ, Watters A L, Rottman M, Waterhouse A, Mammoto A, Gamini N, Rodas MJ, Kole A, Jiang A, Valentin TM, Diaz A, Takahashi K, Ingber D E. An extracorporeal blood-cleansing device for sepsis therapy. Nat. Med. 2014;20:1211–1216. doi: 10.1038/nm.3640. [DOI] [PubMed] [Google Scholar]
- 24.Cole L, Bellomo R, Hart G, Journois D, Davenport P, Tipping P, Ronco C. A phase II randomized, controlled trial of continuous hemofiltration in sepsis. Crit. Care Med. 2002;30:100–106. doi: 10.1097/00003246-200201000-00016. [DOI] [PubMed] [Google Scholar]
- 25.Kellum JA, Dishart MK. Effect of hemofiltration filter adsorption on circulating IL-6 levels in septic rats. Crit. Care. 2002;6:429–433. doi: 10.1186/cc1528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Song M, Winchester J, Albright RL, Capponi VJ, Choquette MD, Kellum JA. Cytokine removal with a novel adsorbent polymer. Blood Purif. 2004;22:428–434. doi: 10.1159/000080235. [DOI] [PubMed] [Google Scholar]
- 27.DiLeo MV, Kellum JA, Federspiel WJ. A simple mathematical model of cytokine capture using a hemoadsorption device. Ann. Biomed. Eng. 2009;37:222–229. doi: 10.1007/s10439-008-9587-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Young, W.-T., Albright, R.L., Golobish, T.D., Capponi, V. & Chan, P. Size- selective polymer system, US 8211310 B2.
- 29.Shukla AM. Extracorporeal therapy in sepsis: are we there yet? Kidney Int. 2012;81:336–338. doi: 10.1038/ki.2011.375. [DOI] [PubMed] [Google Scholar]
- 30.Oh S, Jung H, Seo H, Min M-K, Kim B, Hahn YK, Kang JH, Choi S. Magnetic activated cell sorting (MACS) pipette tip for immunomagnetic bacteria separation. Sens. Actuators, B. 2018;272:324–330. [Google Scholar]
- 31.Wilson S, Lane A, Rosedale R, Stanley C. Concentration of Mycobacterium tuberculosis from sputum using ligand-coated magnetic beads. Int. J. Tuberc. Lung. Dis. 2010;14:1164–1168. [PubMed] [Google Scholar]
- 32.Kang JH, Krause S, Tobin H, Mammoto A, Kanapathipillai M, Ingber DE. A combined micromagnetic-microfluidic device for rapid capture and culture of rare circulating tumor cells. Lab Chip. 2012;12:2175–2181. doi: 10.1039/c2lc40072c. [DOI] [PubMed] [Google Scholar]
- 33.Song S-H, Lee JH, Yoon J, Park W. Functional microparticle R&D for IVD and cell therapeutic technology: Large-scale commercialized products. BioChip J. 2019;13:95–104. [Google Scholar]
- 34.Kang JH, Park J-K. Magnetophoretic continuous purification of single-walled carbon nanotubes from catalytic impurities in a microfluidic device. Small. 2007;3:1784–1791. doi: 10.1002/smll.200700334. [DOI] [PubMed] [Google Scholar]
- 35.Sari AY, Eko AS, Candra K, Hasibuan DP, Ginting M, Sebayang P, Simamora P. Synthesis, properties and application of glucose coated Fe3O4 nanoparticles prepared by co-precipitation method. IOP Conf. Ser.: Mater. Sci. Eng. 2017;214:012021. [Google Scholar]
- 36.Scott K. Handbook of Industrial Membranes. 1995. INTRODUCTION TO MEMBRANE SEPARATIONS; pp. 3–185. [Google Scholar]
- 37.Xia N, Hunt T, Mayers B, Alsberg E, Whitesides G, Westervelt R, Ingber D. Combined microfluidic-micromagnetic separation of living cells in continuous flow. Biomed. Microdevices. 2006;8:299–308. doi: 10.1007/s10544-006-0033-0. [DOI] [PubMed] [Google Scholar]
- 38.Yung CW, Fiering J, Mueller AJ, Ingber DE. Micromagnetic-microfluidic blood cleansing device. Lab Chip. 2009;9:1171–1177. doi: 10.1039/b816986a. [DOI] [PubMed] [Google Scholar]
- 39.Lee Jung-Jae, Jeong Kyung Jae, Hashimoto Michinao, Kwon Albert H., Rwei Alina, Shankarappa Sahadev A., Tsui Jonathan H., Kohane Daniel S. Synthetic Ligand-Coated Magnetic Nanoparticles for Microfluidic Bacterial Separation from Blood. Nano Letters. 2013;14(1):1–5. doi: 10.1021/nl3047305. [DOI] [PubMed] [Google Scholar]
- 40.Microfluidic magnetic particle sorting and separation: a short review. https://www.elveflow.com/microfluidic-tutorials/microfluidic-reviews-and-tutorials/magnetic-particle-separation-a-short-review/.
- 41.Kang JH, Driscoll H, Super M, Ingber DE. Application of a Halbach magnetic array for longrange cell and particle separations in biological samples. Appl. Phys. Lett. 2016;108:213702. [Google Scholar]
- 42.Inglis DW, Riehn R, Austin RH, Sturm JC. Continuous microfluidic immunomagnetic cell separation. Appl. Phys. Lett. 2004;85:5093–5095. [Google Scholar]
- 43.Jung SH, Hahn YK, Oh S, Kwon S, Um E, Choi S, Kang JH. Advection flows-enhanced magnetic separation for high-throughput bacteria separation from undiluted whole blood. Small. 2018;14:e1801731. doi: 10.1002/smll.201801731. [DOI] [PubMed] [Google Scholar]
- 44.Chung AJ. A minireview on inertial microfluidics fundamentals: Inertial particle focusing and secondary Ffow. BioChip J. 2019;13:53–63. [Google Scholar]
- 45.Choi S, Park J-K. Continuous hydrophoretic separation and sizing of microparticles using slanted obstacles in a microchannel. Lab Chip. 2007;7:890–897. doi: 10.1039/b701227f. [DOI] [PubMed] [Google Scholar]
- 46.Chang T, Tu Y-K, Lee C-T, Chao A, Huang C-H, Wang M-J, Yeh Y-C. Effects of polymyxin B hemoperfusion on mortality in patients with severe sepsis and septic shock: A systemic review, meta-analysis update, and disease severity subgroup meta-analysis. Crit. Care Med. 2017;45:e858–e864. doi: 10.1097/CCM.0000000000002362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Klein DJ, Foster D, Walker PM, Bagshaw SM, Mekonnen H, Antonelli M. Polymyxin B hemoperfusion in endotoxemic septic shock patients without extreme endotoxemia: a post hoc analysis of the EUPHRATES trial. Intensive Care Med. 2018;44:2205–2212. doi: 10.1007/s00134-018-5463-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hemotune. https://www.hemotune.ch/.
- 49.BOA Biomedical Inc. https://otd.harvard.edu/news/new-startup-boa-biomedical-launches-to-reduce-sepsis-deaths.
- 50.Romaschin AD, Klein DJ, Marshall JC. Bench-to-bedside review: Clinical experience with the endotoxin activity assay. Crit. Care. 2012;16:248. doi: 10.1186/cc11495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Salter SJ, Cox MJ, Turek EM, Calus ST, Cookson WO, Moffatt MF, Turner P, Parkhill J, Loman NJ, Walker AW. Reagent and laboratory contamination can critically impact sequence-based microbiome analyses. BMC Biol. 2014;12:87. doi: 10.1186/s12915-014-0087-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kim S, Masum F, Jeon JS. Recent developments of chip-based phenotypic antibiotic susceptibility testing. BioChip J. 2019;13:43–52. [Google Scholar]
- 53.Herrmann IK, Beck-Schimmer B, Schumacher CM, Gschwind S, Kaech A, Ziegler U, Clavien P-A, Günther D, Stark WJ, Graf R, Schlegel AA. In vivo risk evaluation of carbon-coated iron carbide nanoparticles based on short- and long-term exposure scenarios. Nanomedicine. 2016;11:783–796. doi: 10.2217/nnm.16.22. [DOI] [PubMed] [Google Scholar]
- 54.Jang BH, Kwon S, Kang JH. Measurement of the magnetic susceptibility of subtle paramagnetic solutions using the diamagnetic repulsion of polymer microparticles. Lab Chip. 2019;19:2356–2361. doi: 10.1039/c9lc00245f. [DOI] [PubMed] [Google Scholar]
- 55.Bougas L, Langenegger LD, Mora CA, Zeltner M, Stark WJ, Wickenbrock A, Blanchard JW, Budker D. Nondestructive in-line sub-picomolar detection of magnetic nanoparticles in flowing complex fluids. Sci. Rep. 2018;8:3491. doi: 10.1038/s41598-018-21802-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Shahidi Bonjar, A.H. Antiviral therapy: a perspective. Drug Des., Dev. Ther. DOI 10.2147/DDDT.S83037 (2016). [DOI] [PMC free article] [PubMed]
- 57.Park I, Lee JH, Jang D-H, Kim D, Chang H, Kwon H, Kim S, Kim TS, Jo YH. Characterization of fecal peritonitis-induced sepsis in a porcine model. J. Surg. Res. 2019;244:492–501. doi: 10.1016/j.jss.2019.06.094. [DOI] [PubMed] [Google Scholar]
- 58.Fink MP. Animal models of sepsis. Virulence. 2014;5:143–153. doi: 10.4161/viru.26083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Pino CJ, Yevzlin AS, Tumlin J, Humes HD. Cell-based strategies for the treatment of kidney dysfunction: a review. Blood Purif. 2012;34:117–123. doi: 10.1159/000341649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Huang CY, Daniels R, Lembo A, Hartog C, O’Brien J, Heymann T, Reinhart K, Nguyen H B, Sepsis Survivors Engagement Project SSEP Life after sepsis: an international survey of survivors to understand the post-sepsis syndrome. Int. J. Qual. Health Care. 2019;31:191–198. doi: 10.1093/intqhc/mzy137. [DOI] [PubMed] [Google Scholar]


