Abstract
Purpose
To provide a practical overview of the management of the potential organ donor in the intensive care unit.
Methods
Seven areas of donor management were considered for this review: hemodynamic management; fluids and electrolytes; respiratory management; endocrine management; temperature management; anaemia and coagulation; infection management. For each subchapter, a narrative review was conducted.
Results and conclusions
Most elements in the current recommendations and guidelines are based on pathophysiological reasoning, epidemiological observations, or extrapolations from general ICU management strategies, and not on evidence from randomized controlled trials. The cardiorespiratory management of brain-dead donors is very similar to the management of critically ill patients, and the same applies to the management of anaemia and coagulation. Central diabetes insipidus is of particular concern, and should be diagnosed based on clinical criteria. Depending on the degree of vasopressor dependency, it can be treated with intermittent desmopressin or continuous vasopressin, intravenously. Temperature management of the donor is an area of uncertainty, but it appears reasonable to strive for a core temperature of > 35 °C. The indications and controversies regarding endocrine therapies, in particular thyroid hormone replacement therapy, and corticosteroid therapy, are discussed. The potential donor should be assessed clinically for infections, and screening tests for specific infections are an essential part of donor management. Although the rate of infection transmission from donor to receptor is low, certain infections are still a formal contraindication to organ donation. However, new antiviral drugs and strategies now allow organ donation from certain infected donors to be done safely.
Keywords: Organ transplantation, Brain death, Organ donor, Tissue and Organ procurement
Take-home message
| A concise and critical overview of the essential elements in the management of the brain-dead donor. |
Introduction
Mortality on the waiting list for transplantation remains high [1]. Efforts to increase organ donation rates in the population, such as opting-out laws or campaigns to promote donation consent, are part of the solution to this challenge. Other important elements involve the early identification of potential donors, and include organ donation as an option in the end-of-life care of every intensive care unit (ICU) patient [1]. On the other hand, organs from deceased potential donors are often refused because of suboptimal quality [2]. Adequate donor management to maximize the number of organs that can be offered for donation is essential to expand the donation pool. Such management may ensure an optimal future function in the recipient, and can be viewed as an ultimate form of respect towards the donor and his family, because it involves meticulous care for the bodily functions of the deceased.
Protection and optimization of organ functions is at the heart of intensive care medicine. The timeframe between brain death diagnosis and referral to the operating room for procurement provides a window of opportunity to apply interventions that can preserve and even improve organ function. The role of intensivists in this process is key. The purpose of the present review is to provide a practical and yet critical overview of general interventions and organ-specific measures to maximize the chance of successful transplantation.
Hemodynamic management
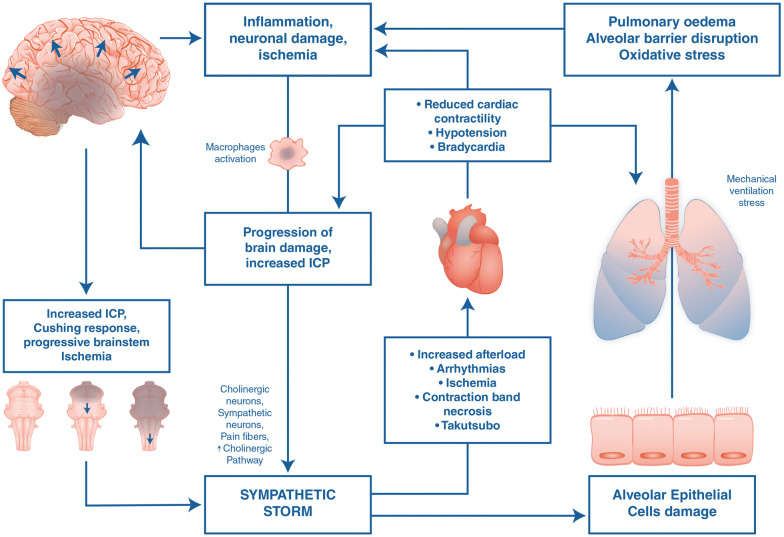
The hemodynamic response after brain death is well-described [3, 4], and hemodynamic instability is probably the number one challenge, or at least the most obvious one, in the management of the brain-dead donor. A primary devastating injury to the brain and/or brain stem results in a massive immediate activation of the sympathetic nervous system, leading to an increase in arterial blood pressure and cardiac afterload, followed by left atrial pressure elevation, increased pressure in the pulmonary capillary bed, pulmonary vasoconstriction, and endothelial damage [3] (Fig. 1). This sympathetic storm may lead to cardiac muscle damage [5], and contribute to respiratory problems, described below. In a small retrospective cohort study, Esmolol was used to mitigate this response [6], but needs stronger evidence before it can be broadly recommended. Thereafter, vasoplegia becomes the most dominant hemodynamic feature, requiring fluids and/or vasopressors. Brain stem death includes the vagal nuclei, and, therefore, only the sympathetic nervous system influences the heart rate. An intravenous injection of atropine will fail to increase the heart rate after brain stem herniation [7]. Endocrine changes might contribute to fluid depletion or vasoplegia. The specific fluid and endocrine management of the brain-dead donor will be described in the sections below.
Fig. 1.
Overview of the sympathetic storm and the pro-inflammatory cascade caused by devastating brain injury with brain stem ischemia. As brain damage progresses and intracranial pressure rises to the point where cerebral perfusion is impaired, progressive ischemic damage through the entire brain and brain stem will cause hypothalamic activation of the autonomic system (so-called autonomic storm), characterized by a systemic stress response with increase in circulating catecholamines. Hemodynamic repercussions: Activation of alpha-1-adrenergic receptors causes vasoconstriction and increased arterial blood pressure (first phase of the Cushing reflex). The circulating high levels of catecholamines can lead to increased oxygen consumption, arrhythmias and cardiac injury. As a result of activation of baroreceptors in the aortic arch and damage to brain stem vasomotor nuclei with loss of peripheral vascular tone, parasympathetic activation leads to hypotension, reduced cardiac contractility, and bradycardia (second phase of the Cushing reflex). Respiratory repercussions: The acute increase in intracranial pressure and the consequent catecholamine release and generalized inflammatory response produces a transient increase in systemic intravascular pressure that damages the alveolar epithelial cells and increases pulmonary capillary permeability to protein. The respiratory system is then vulnerable to further inflammatory insults (the so-called “second hit”) caused by mechanical stress induced by mechanical ventilation activating a vicious circle where the respiratory function may worsen damage of the central nervous system
As with every hemodynamic problem, identification of the cause of the instability (i.e. fluid depletion, reduced cardiac output, vasoplegia, …) is crucial to determine the right therapy. There are no major studies dictating which types of monitoring should be applied in brain-dead donors [8], but it is common sense that hemodynamic shock states should be evaluated using invasive arterial blood pressure measurements, lactate levels, (mixed) venous oxygen saturations, and echocardiography to evaluate myocardial contractility and exclude other cardiac pathology. Serial echocardiography can be used to assess potential recovery in neurogenic stunned myocardium, and guide continued support in potential donors [9], which may increase the number and quality of donor hearts. In some cases, specific invasive monitoring with pulmonary artery catheter (PAC) [8] or arterial waveform analysis-based techniques [10] can be useful to steer management. It is difficult to set universal hemodynamic treatment goals for all donors, and the recommended mean blood pressure targets of 60–70 mmHg [2, 8] are not based on evidence from randomized controlled trials (RCTs). In the Monitoring Organ Donors to Improve Transplantation Results (MOnIToR) RCT, stopped prematurely due to lack of resources, management based on blood pressure, pulse pressure variation, and cardiac index targets was not superior to conventional therapy in increasing the number of organs transplanted per donor [11].
Guidelines propose dopamine as the catecholamine of choice, and to use norepinephrine sparingly because of theoretical concerns that it may increase pulmonary capillary permeability, induce excessive mesenteric or coronary vasoconstriction, or increase the afterload of the left heart [8]. In a small prospective trial where brain-dead cardiac donors were weaned of norepinephrine and switched to dopamine and/or vasopressin, norepinephrine exposure was associated with reduced end-systolic elastance, which was in turn associated with worse cardiac graft function and worse survival at 1 year in heart transplant recipients [12]. An RCT of low-dose dopamine (4 µg/kg/min) in 264 brain-dead kidney donors resulted in a significantly reduced need for dialysis in the recipient of the dopamine-treated grafts [13]. Patients on norepinephrine > 0.4 µg/kg/min or other adrenergic agents were excluded from this study, and 78.4% and 85.8% of donors received concomitant norepinephrine in the dopamine and control groups respectively. A recent retrospective study of 135 kidney donors found a similar rate of catecholamine use of 85.8% (dopamine 60%, noradrenaline 49%, dobutamine 11%, adrenaline 3%) [14], while only noradrenaline, and not dopamine, was associated with a decreased incidence of graft loss. Apparently, several combinations of vasopressors and catecholamines are being used in donors, and in the absence of RCTs comparing their effect on graft outcomes, the superiority of one regimen over the other is not established.
Fluids and electrolytes
Hypovolemia is a frequent finding in the potential organ donor, because of profound peripheral vasodilatation and central diabetes insipidus (CDI), consequent on brain death-related central sympatholysis and hormone dysregulation, respectively. Donor management guidelines emphasize the importance of the prevention or immediate correction of hypovolemia to maintain perfusion in potentially transplantable organs [8, 15]. Brain death-related pathophysiological changes make the clinical assessment of volume status in organ donors even more challenging, compared to general ICU patients [16]. Appropriate monitoring is essential to guide fluid replacement, but optimal monitoring modalities remain undefined [1, 7, 13]. The negative results of the multicentre MOnIToR trial make it difficult to make specific recommendations with regard to fluid resuscitation targets [11]. Euvolemia, even while this is an ill-defined concept, is the primary therapeutic goal, and isotonic crystalloid solutions are the preferred choice for volume replacement in the organ donor. In the absence of evidence of superiority of one over the other, 0.9% saline or lactated Ringer solution are both recommended [8]. Starch-based synthetic colloids should be avoided because of their known adverse effects in critically ill patients generally [17, 18], and because hydroxyethyl starch has been associated with a 41% increase in the risk of delayed graft failure after renal transplantation [19]. A perceived challenge in volume management is the potential for a conflict between treatment goals for kidney and lung donation. Traditionally, aggressive fluid resuscitation was believed to result in improved kidney retrieval rates, while a conservative fluid replacement strategy benefited lung procurement. However, in a recent cohort study, a lung-targeted management strategy, including a restrictive fluid balance, had no adverse effect on kidney graft survival compared against historical controls [20].
CDI is an early sign of brain death-related endocrinopathy, and is reported in 46–86% of brain-dead organ donors [16]. It is a consequence of failure of posterior pituitary function and depletion of anti-diuretic hormone (ADH), and is characterized by polyuria, hyperosmolality, and hypernatremia. Other causes of high urine output and hypernatremia, such as osmotic diuresis secondary to hyperglycaemia or prior administration of mannitol, should be excluded, and the diagnosis of CDI confirmed using established clinical criteria (Table 1), prior to commencing treatment. CDI should be treated with desmopressin or vasopressin depending on the patient’s clinical status [21]. Desmopressin (1-deamino-8-d-arginine vasopressin) is a vasopressin analogue with greater affinity for the V2 receptor. It has a primary antidiuretic action and is the preferred choice for CDI in the absence of hypotension. Dosing is largely empirical. Small doses minimize over-prolonged action and risk of hypervolemia, and can be repeated to obtain the desired clinical effect [22]. In practice, an initial IV dose of desmopressin of 1–4 μg is usual, repeated as necessary until urine volume and serum sodium concentration come under control. If further correction of hypernatremia is required once volume status is stabilized, hypotonic fluids such as 5% dextrose can be considered while being mindful to avoid hyperglycaemia. Vasopressin infusion is indicated when CDI occurs in association with hypotension refractory to fluid resuscitation; it acts equally at all three vasopressin receptors, so has pressor in addition to antidiuretic actions. Vasopressin use is associated with increased organ retrieval rates, although it is not known whether this effect is related to its reversal of hypotension, treatment of CDI, or both [23]. Maintaining serum sodium < 155 mEq/L during the management of CDI is recommended because some studies report worse liver graft survival with higher concentrations [8].
Table 1.
Diagnostic criteria for central diabetes insipidus
| Clinical feature | Diagnostic finding |
|---|---|
| Increased urine volume | Urine output > 3–4 L/day or > 2.5–3.0 mL/kg/h |
| Hypernatremia | Serum sodium concentration > 145 mmol/L |
| Normal or increased serum osmolality | Serum osmolality > 305 mmol/kg |
| Inappropriately dilute urine | Urine osmolality < 200 mmol/kg or specific gravity < 1.005a |
aWhilst waiting for laboratory tests, a simple, but not 100% reliable, bedside test of urine specific gravity (SG) may be useful. In the presence of high urine output and high serum sodium, urine SG < 1.005 is suggestive of diabetes insipidus
Respiratory management
The combination of raised hydrostatic pressures and capillary damage in the pulmonary vascular bed unbalances Starling forces across the endothelium, causing leakage of plasma into interstitium and alveolar space, resulting in neurogenic pulmonary oedema (NPE) [24] (Fig. 1). Massive brain injury increases the vulnerability of the lungs [25, 26] to mechanical or ischemia–reperfusion injury, through increased expression of inflammatory mediators, neutrophil infiltration and activated macrophages in the alveolar space, membrane lipid peroxidation, and alveolar haemorrhage. This ‘double hit’, hemodynamic and inflammatory, causes a clinical picture similar to the acute respiratory distress syndrome (ARDS). This could not only impede the potential for lung donation, but also endanger the homeostasis of other organs. The management of the potential donor is thus aimed at maintaining gas exchange to protect other organs, while taking care to preserve the lung [8].
Respiratory targets include a physiologic pH (7.35–7.45), with the use of the minimal fraction of inspired oxygen (FiO2) necessary to achieve a partial pressure of oxygen (PaO2) above 100 mmHg, an oxygen saturation (SpO2) above 95%, and a partial pressure of carbon dioxide (PaCO2) of 35–40 mmHg. Previous guidelines recommended the use of liberal tidal volumes [10–15 mL/kg of predicted body weight (PBW)] [27] with positive end expiratory pressure (PEEP) of at least 5 cmH2O to treat NPE. However, ventilator-induced lung injury is common in organ donors [28], and recent experimental evidence shows that protective ventilation attenuates the severity of lung injury [29]. A protective ventilator strategy bundle (using 6–8 mL/kg of PBW, PEEP equal to 8–10 cmH2O, a closed circuit for tracheal suction, alveolar recruitment manoeuvres after any disconnection, and the use of continuous positive airway pressure during apnoea test), compared to the conventional strategy in a multicentre RCT [30], increased the number of eligible and transplanted lungs, while the number of other transplanted organs was not influenced. Current guidelines recommend ‘low stretch protocols’, using lower tidal volumes of 6 mL/kg PBW, plateau pressures < 30 cmH2O, and measures to recruit atelectatic lung [8], an approach similar to the management of acute lung injury patients [31, 32].
In so-called extended-donor-criteria lung donors, a good evaluation of the lung combined with appropriate donor management might still lead to acceptable receptor outcomes [33]. An observational survey conducted in 13 Italian centres [34] revealed that nearly half of potential lung donors had a PaO2/FiO2 ratio of < 300, making them ineligible for lung donation. In these patients, ventilator settings remained unchanged after the diagnosis of brain death, and no strategies to prevent lung decruitment were performed, suggesting that respiratory management may have been suboptimal.
Endocrine management
Thyroid hormone replacement
Alterations in the thyroid axis are common after brain death, with usually low levels of the biologically active triiodothyronine (T3). Low T3 levels were historically ascribed to hypothalamic–pituitary dysfunction following brain death. However, several studies of brain-dead organ donors demonstrated residual pituitary function in the majority of patients, due to internal carotids supply, with normal to elevated thyroid-stimulating hormone levels [35, 36]. Thyroxine (T4) levels usually remain in the normal range and levels of inactive reverse-T3 are normal or elevated. This constellation points to non-thyroidal illness rather than central hypothyroidism, with increased peripheral inactivation of thyroid hormone in the presence of a functioning thyroid gland, as also occurs in general ICU patients [35, 36]. Since prolonged and/or severe hypothyroidism may lead to myocardial dysfunction, low T3 levels have been hypothesized to provoke hemodynamic instability in the potential donor. It remains unclear, however, whether non-thyroidal illness occurring after brain death should be treated. A large observational study including data from 63,593 brain-dead organ donors independently associated thyroid hormone substitution with an increased number of procured organs [37]. However, due to the observational design, a causal relationship cannot be inferred. Moreover, the apparent benefit of thyroid hormone replacement was not confirmed by an RCT, which could not find any impact on donor hemodynamics or number of procured organs [38]. However, the relatively low number of patients with hemodynamic stability included in RCTs may preclude a conclusion in this patient subgroup. Hence, consensus guidelines have recommended to consider thyroid hormone replacement in hemodynamically unstable donors [8]. For this purpose, both T4 and T3 substitution have been used, although T4 is increasingly degraded to inactive reverse-T3. Commonly used doses are reported in Table 2.
Table 2.
Commonly used drug regimens for endocrine management of the brain-dead organ donor
| Hormone/drug | Dose | Remark | Evidence |
|---|---|---|---|
| Thyroid hormone | |||
| Thyroxine (T4) | 20 µg IV bolus | Precursor of T3 | More organs procured in observational studies [37] |
| 10 µg/h IV maintenance | Low intrinsic biological activity | No proof in benefit in RCTs [38] | |
| Increased conversion into inactive rT3 | |||
| Slow onset | |||
| Triiodothyronine (T3) | 4 µg IV bolus | Active hormone | More organs procured in observational studies [37] |
| 3 µg/h IV maintenance | Rapid onset | No proof in benefit in RCTs [38] | |
| May trigger arrhythmias | |||
| Corticosteroid | |||
| Methylprednisolone | 1000 mg IV od | Monitor blood glucose levels | Improved donor hemodynamics, increased organ procurement. and improved graft and recipient survival in some observational studies [37, 40] |
| OR | Only after blood sampling for tissue typing | Mixed results from RCTs, largely neutral [40] | |
| 15 mg/kg IV od | |||
| OR | |||
| 250 mg IV bolus | |||
| 100 mg/h IV maintenance | |||
| Hydrocortisone | 50 mg IV bolus | Monitor blood glucose levels | Lower vasopressor need in observational study, without difference in organ procurement rate [41] |
| 10 mg/h IV maintenance | Only after blood sampling for tissue typing | No RCT evidence | |
| OR | |||
| 300 mg/d IV | |||
| Insulin | Continuous IV infusion | Frequently measure blood glucose | Supported by observational studies [8, 42] |
| Adjust dose to preset target blood glucose | At least avoid severe hyperglycaemia and large blood glucose fluctuations | No RCT evidence in this population | |
IV intravenous, od once daily, RCT randomized controlled trial, rT3 reverse-T3
Corticosteroid treatment
Corticosteroid treatment of brain-dead organ donors has been advocated for two reasons. The first reason is to treat presumed hypothalamic–pituitary–adrenal (HPA) axis failure, which could potentially mediate hemodynamic instability. However, as with the thyroid axis, the HPA axis is usually not deficient after brain death [35, 36]. Moreover, in observational studies, donor hemodynamic instability was not associated with hypocortisolaemia or absent corticotropin responsiveness of the adrenals [35, 39]. Nevertheless, corticosteroids may improve hemodynamics through their vasopressor effects [40, 41]. The second potential reason to administer corticosteroids is to reduce inflammation, which may negatively impact graft function [8]. Observational studies have suggested increased organ procurement rates and improved graft and recipient survival by administering corticosteroids [37, 40]. High-quality RCT evidence is lacking, however. A systematic review revealed only one RCT showing clinical benefit, i.e. reduced hepatic ischemia–reperfusion injury and acute rejection, which was not confirmed by a subsequent RCT [40]. The large heterogeneity in study design and concurrent therapies, and the poor quality of most RCTs, preclude a strong conclusion [40]. In most studies, high doses of methylprednisolone were administered (Table 2). Theoretically, corticosteroid-induced or corticosteroid-aggravated hyperglycaemia could have outweighed any potential benefit. More recently, lower doses of hydrocortisone have been studied [40, 41]. A relatively small observational study found improved blood glucose control by such strategy, without a benefit on patient-centred outcomes [40]. In summary, the indications for corticosteroid treatment in brain-dead organ donors remains unclear, but could be considered in case of hemodynamic instability. Importantly, they should only be administered after sampling for tissue typing, since they may reduce human leukocyte antigen expression [8].
Hyperglycaemia
As in critically ill patients, brain-dead patients usually have hyperglycaemia due to insulin resistance and unsuppressed gluconeogenesis. Hyperglycaemia has been associated with reduced donor renal function and with pancreas allograft loss [8, 42]. Besides that, severe hyperglycaemia may induce osmotic diuresis leading to fluid depletion and electrolyte abnormalities, and is associated with an increased risk of infections in critically ill patients [42]. The ideal blood glucose target for potential donors remains unclear, due to the lack of RCTs in this population. Consensus guidelines recommend to treat at least severe hyperglycaemia (> 180 mg/dL) [8].
Nutrition
The optimal feeding strategy for brain-dead organ donors remains unclear, since clinical studies are lacking. Consensus guidelines recommend to continue nutritional support as if brain death would not have occurred [8]. In view of the potentially deleterious effects of early parenteral nutrition in critically ill patients—increasing risk of infections and prolonging organ failure—it seems prudent not to initiate early parenteral nutrition in potential donors [43].
Temperature management
Temperature dysregulation following death by neurologic criteria is inevitable [4, 44], manifesting as a progressive reduction of the internal temperature, unless temperature is actively corrected. This phenomenon is secondary to a combination of loss of hypothalamic control, fall in metabolic rate, absence of muscular activity, and increased heat loss because of profound vasoplegia. Hypothermia may have negative consequences because it can activate intravascular coagulation and produce organ damage [45]. Myocardial contractility reduces, and the heart becomes more susceptible to arrhythmias. Independent from this early coagulopathy, hypotension, and injury severity, admission hypothermia has been associated with reduced organ donation in trauma patients [46]. In addition, a body temperature above 35 °C is a necessary criterion to be able to pronounce brain death in most national legislations about organ donation [47]. Initial measures for reaching the temperature target includes thermal blankets reducing passive heat loss, hot air devices, warmed fluid infusions, and, in extreme conditions, intravascular devices.
In a recent RCT in 370 organ donors, including predominantly recipients of kidneys from extended criteria donors [48], mild hypothermia in the organ donor (34–35 °C) significantly reduced the requirement for early dialysis after kidney transplantation compared with normothermia (36.5–37.5 °C). A more recent retrospective cohort study [49] showed that donor’s lower temperature was associated with lower serum creatinine levels before procurement, but failed to validate a graft survival advantage. Therefore, the current target to keep the body temperature above 35 °C seems reasonable [1], although a higher target may be warranted in the presence of severe cardiovascular instability requiring high doses of vasopressors or inotropes.
Anaemia and coagulation
Anaemia
RCTs in paediatric as well as in adult critically ill patients have demonstrated that restrictive red blood cell (RBC) transfusion strategies are equal or non-inferior to liberal strategies with regard to organ function and outcomes [50]. Although certain subpopulations might benefit from a higher haemoglobin levels, an RBC transfusion target of 7 g/dL is recommended in hemodynamically stable patients [51]. Whether the same target can be extended to brain-dead donors is unclear [8] since no RCTs have addressed this issue, or have examined the effect of transfusion on oxygen delivery (DO2) to tissues or organs. Brain-dead donors typically have a decreased oxygen consumption (VO2). At least in some centres, haemoglobin thresholds for RBC transfusions appear to be higher than 7 g/dL. In a retrospective review at the Vanderbilt University Medical Center, the haemoglobin trough level was above 7 g/dL in 86.5% of donors who received RBC transfusions; in 15.8% it was even above 10 g/dL [52]. Kidneys transplanted from brain-dead donors who had received one or more RBC transfusion had a lower probability of delayed graft function [53]. Currently, universal recommendations on RBC transfusion targets in brain-dead donors cannot be made. Clinicians should integrate their decision to transfuse in a global therapeutic strategy targeting the cardiovascular system, and consider RBC transfusions in cases of hemodynamic instability with signs of tissue hypoxia [10]. Maybe computerized decision support systems such as the ‘Digital Intern’ could support clinicians for individualized transfusion dosage in brain-dead donors [54].
Coagulation
Brain death is associated with a pronounced systemic inflammatory response, together with an activation of coagulation. In particular, an increased fibrin formation, hypofibrinolysis, as well as a higher platelet activation paired with a profound dysregulation in the von Willebrand factor production (which promotes platelet attachment to damaged vasculature), are observed [55]. This prothrombotic state may contribute to formation of microthrombi in transplantable organs, and potentially to a deterioration of their function. However, no specific strategies to avoid these processes have been validated in clinical trials. In brain-dead donors due to traumatic brain injury (TBI), the incidence of disseminated intravascular coagulation (DIC) can be as high as 15–25% [56], especially if the donor had a general bleeding and received multiple units of blood products. Still, DIC in the donor is not a contraindication for transplantation, as it does not affect long-term or even early graft function in heart, lung [56], and kidney [57] transplantation. Severe thrombopenia could complicate the surgical procedures of organ procurement, but there is uncertainty with regard to the minimum safe platelet level for surgery. In addition, a low platelet level in donors suffering from immune idiopathic thrombopenia was associated with a lower unadjusted 5-year patient and graft survival in liver transplant recipients, but no effects on other transplanted organs was observed [58]. While good clinical evidence is lacking, an international normalized ratio (INR) of < 1.5 and a platelet count of > 50,000/mm3 have been proposed as therapeutic goals [10], including the transfusion of clotting factors and/or platelets to achieve these goals before surgery. There are no conclusive evidence-based data on thromboprophylaxis in the brain-dead organ donor. It might be reasonable in patients with normal coagulation and platelets to prescribe low-molecular weight heparin (LMWH) because of the pro-coagulative status.
Infection management
Infections in the donor might complicate organ donation. The actual rate of unexpected infection transmission from donor to receptor is low, occurring in less than 1% of solid organ transplant recipients [59, 60]. Nevertheless, the consequences may be devastating, and sometimes even fatal. Although the effectiveness of pre-transplant donor infectious screening programs is not known, they are an essential element in the pre-transplant evaluation of potential donors [61]. The assessment of the donor should involve a review of medical and social history (including previous infections, travels, contact with animals, other environmental exposures, sexual history, and intravenous drug abuse) [8, 59]. The infection screening protocols and its different elements varies across centres and geographical regions [59, 61, 62]. A summary of common screening tests can be found in Table 3. In the ICU, where the potential donor is usually located, the incidence of infection (mainly pneumonia or catheter-related bloodstream infections) can reach up to 40%, especially when the length of stay is prolonged [63]. Bacteraemia or sepsis are not contraindications to donation, provided appropriate antibiotics have been administered for at least 48 h prior to procurement [8], and in the absence of shock, multi-organ failure or poor response to antibiotic treatment. Some infections, such as fungal infections or bacteraemia, are only diagnosed after transplantation, and in those cases, it is important to communicate well with the coordinating transplant organization to allow for treatment of the receptor [59]. Bacterial meningitis in the donor is not a contraindication either, after adequate treatment for at least 24–48 h aimed at the known or presumed pathogen, and treatment of the recipient with the same antibiotics for 5–10 days [8]. However, meningo-encephalitis due to rare pathogens (including Mycobacterium, lymphocytic chorio-meningitis virus, West Nile virus, rabies, Cryptococcus, Coccidioides, Aspergillus, and Balamuthia) may be a challenge, and specific guidance can be found on the website of the Organ Procurement and Transplantation Network [64]. When a donor has febrile illness with unclear etiology, signs of meningitis or encephalitis, or focal or global neurological deficits of an unknown origin, the organs should not be offered for procurement [8]. In endemic areas, Zika virus transmission from donor to receptor has been documented [65], and maybe donors who have recently travelled to these areas should be screened [60]. Organs from HIV-positive donors have been transplanted successfully to HIV-positive receptors [66], and this is now an acceptable practice. Because new antiviral agents against hepatitis C have become available, successful use of organs from hepatitis C-positive donors has been reported with good outcomes, provided the receptor was treated based on viral load monitoring [67], or pre-emptively [68].
Table 3.
Routine infection screening for potential organ donors
| Clinical assessment |
| Physical examination |
| Medical and social history |
| Travel |
| Animals |
| Environmental |
| Sexual |
| Intravenous drug abuse |
| Chest radiograph |
| Routine laboratory |
| White blood cell count |
| CRP |
| Cultures: bacterial and fungi |
| Blood cultures |
| Urine cultures for kidney donors |
| Broncho-alveolar lavage for lung donors |
| Viral serology |
| CMV, HIV, HSV antibody |
| HBV surface antigen, core antibody, surface antibody |
| HCV antibody |
| VZV antibody |
| Other serology |
| Toxoplasma antibody |
| Syphilis screening: RPR |
| Tuberculosis screening |
| Purified protein or interferon-gamma |
| Strong suspicion based on medical/social history |
| NAT for HIV, HBV, HCV |
| Strong suspicion based on stay in endemic areas or areas with epidemiological exposure |
| Strongyloides serology |
| Coccidioides serology |
| Trypanosoma serology |
| Blastomyces serology |
| HEV serology |
| Cryptococcus antigen |
| WNV antibody |
| Zika NAT |
This table was build based on references [8, 59, 61, 62]
CMV cytomegalovirus, HBV hepatitis B virus, HCV hepatitis C virus, HEV hepatitis E virus, HIV human immunodeficiency virus, HSV herpes simplex virus, WNV West Nile virus, VZV varicella zoster virus, CRP C-reactive protein, NAT nucleic acid amplification test, RPR rapid plasma reagin
Conclusions
Brain death induces a marked physiological response in many organ systems, especially the cardiorespiratory and endocrine systems. This poses a particular challenge in the management of these patients, and may lead to loss of potential organs for donation if left untreated. In essence, the hemodynamic and respiratory management of brain-dead donors does not differ that much from the management of the critically ill patient, with a focus on substrate supply and consumption matching, and avoiding therapy-induced harm such as ventilator-induced injury or excessive vasoconstriction. Specific endocrine therapies, such as the use of vasopressin or analogues, steroid therapy, and thyroid hormone replacement, are often indicated. Infections in the donor are a special challenge, in particular because of the risk of transmission. The most important management strategies are summarized in Table 4.
Table 4.
The most important (potential) organ-protective therapies in the brain-dead donor, at a glance
| Organ-protective therapy | Evidence of an effect on the numbers of organs retrieved, or improved organ function in the donor? |
|---|---|
| Hemodynamic management | |
| Low-dose dopamine (4 µg/kg/min) in kidney donors | One small RCT, reduced need for dialysis in kidney recipients [13] |
| Other inotropes or vasopressors | Observational data |
| Fluid and electrolyte management | |
| Goal-directed fluid management | One RCT, prematurely stopped; not superior to conventional therapy [11] |
| Crystalloid solutions: NaCl 0.9% or Ringer’s lactate | Observational data |
| Avoid the use of starches | One prospective cohort study, propensity score-corrected; independent predictor of DGF [19] |
| Diabetes insipidus treatment: desmopressin or vasopressin | Extrapolation from evidence in non-brain-dead critically ill patients |
| Respiratory management | |
| Lung-protective ventilation and recruitment manoeuvres | One RCT, prematurely stopped; increased number of eligible and harvested lungs [30] |
| Endocrine management | |
| Thyroid hormone in hemodynamically unstable donors | Observational data; higher number of procured organs [37] |
| Four double blind placebo-controlled RCTs; no evidence of benefit [38] | |
| Corticosteroid treatment | 14 observational trials; most suggest some form of benefit [40] |
| 11 RCTs, poor quality, many confounders; all but one are neutral [40] | |
| Temperature management | |
| Maintain body T° 34°–35° | One RCT; lower incidence of DGF in kidney recipients, compared to 36, 5°–37, 5° [48] |
| Maintain body T° > 35° | Legal requirement in many countries |
| Blood transfusion and coagulation management | |
| RBC transfusion trigger unknown | Observational data |
| Keep INR < 1.5 and platelet count > 50,000/mm3 | Observational data, single-centre practice [10] |
| LMWH prophylaxis | Extrapolation from evidence in non-brain-dead critically ill patients |
| Infection management | |
| Screen the donor for infections | International guidelines, based on epidemiological data [62, 64] |
| HIV-positive donor may donate to HIV positive receptor | One prospective non-randomized cohort study; similar outcomes compared to non-HIV [66] |
| Donation from HCV positive donor | One prospective observational study (HTx); DAAs reduce viral load [67] |
| One prospective observational study (KTx); prophylactic DAAs reduce viral load [68] | |
RCT randomized controlled trial, DGF delayed graft function, RBC red blood cell, INR international normalized ratio, HIV human immunodeficiency virus, HCV hepatitis C virus, HTx heart transplant, KTx kidney transplant, DAA direct anti-viral agent
Unfortunately, for many of the management strategies in brain-dead donors, we lack evidence from RCTs that demonstrate an effect on receptor outcomes. Hence, some of the elements in brain-dead donor management protocols are based on pathophysiological reasoning, epidemiological observations, or extrapolations from general ICU management strategies.
Acknowledgements
Geert Meyfroidt is funded by the Fonds Wetenschappelijk Onderzoek as senior clinical investigator (grant no. 1846118N).
Compliance with ethical standards
Conflicts of interest
MS is Editor in Chief of the Journal of Neurosurgical Anesthesiology. GC is Editor in Chief of Intensive Care Medicine. GM and IML are Associate Editors of Intensive Care Medicine. CR is Junior Editor of Intensive Care Medicine.
Ethical approval
An approval by an ethics committee was not applicable.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Citerio G, Cypel M, Dobb GJ, et al. Organ donation in adults: a critical care perspective. Intensive Care Med. 2016;42:305–315. doi: 10.1007/s00134-015-4191-5. [DOI] [PubMed] [Google Scholar]
- 2.Tullius SG, Rabb H. Improving the supply and quality of deceased-donor organs for transplantation. N Engl J Med. 2018;378:1920–1929. doi: 10.1056/NEJMra1507080. [DOI] [PubMed] [Google Scholar]
- 3.Van Loon J, Shivalkar B, Plets C, et al. Catecholamine response to a gradual increase of intracranial pressure. J Neurosurg. 1993;79:705–709. doi: 10.3171/jns.1993.79.5.0705. [DOI] [PubMed] [Google Scholar]
- 4.Smith M. Physiologic changes during brain stem death—lessons for management of the organ donor. J Heart Lung Transplant. 2004;23:S217–S222. doi: 10.1016/j.healun.2004.06.017. [DOI] [PubMed] [Google Scholar]
- 5.Novitzky D, Rhodin J, Cooper DKC, et al. Ultrastructure changes associated with brain death in the human donor heart. Transpl Int. 1997;10:24–32. doi: 10.1007/BF02044338. [DOI] [PubMed] [Google Scholar]
- 6.Audibert G, Charpentier C, Seguin-Devaux C, et al. Improvement of donor myocardial function after treatment of autonomic storm during brain death. Transplantation. 2006;82:1031–1036. doi: 10.1097/01.tp.0000235825.97538.d5. [DOI] [PubMed] [Google Scholar]
- 7.Drory Y, Ouaknine G, Kosary IZ, Kellermann JJ. Electrocardiographic findings in brain death; description and presumed mechanism. Chest. 1975;67:425–432. doi: 10.1378/chest.67.4.425. [DOI] [PubMed] [Google Scholar]
- 8.Kotloff RM, Blosser S, Fulda GJ, et al. Management of the potential organ donor in the ICU: society of critical care medicine/American college of chest physicians/association of organ procurement organizations consensus statement. Crit Care Med. 2015;43:1291–1325. doi: 10.1097/CCM.0000000000000958. [DOI] [PubMed] [Google Scholar]
- 9.Lazzeri C, Guetti C, Migliaccio ML, et al. The utility of serial echocardiograms for organ procurement in brain death. Clin Transplant. 2017;31:e13094. doi: 10.1111/ctr.13094. [DOI] [PubMed] [Google Scholar]
- 10.Dictus C, Vienenkoetter B, Esmaeilzadeh M, et al. Critical care management of potential organ donors: our current standard. Clin Transplant. 2009;23:2–9. doi: 10.1111/j.1399-0012.2009.01102.x. [DOI] [PubMed] [Google Scholar]
- 11.Al-Khafaji A, Elder M, Lebovitz DJ, et al. Protocolized fluid therapy in brain-dead donors: the multicenter randomized MOnIToR trial. Intensive Care Med. 2015;41:418–426. doi: 10.1007/s00134-014-3621-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Stoica SC, Satchithananda DK, White PA, et al. Noradrenaline use in the human donor and relationship with load-independent right ventricular contractility. Transplantation. 2004;78:1193–1197. doi: 10.1097/01.tp.0000137792.74940.4f. [DOI] [PubMed] [Google Scholar]
- 13.Schnuelle P, Gottmann U, Hoeger S, et al. Effects of donor pretreatment with dopamine on graft function after kidney transplantation. JAMA. 2009;302:1067–1075. doi: 10.1001/jama.2009.1310. [DOI] [PubMed] [Google Scholar]
- 14.Birtan D, Arslantas MK, Altun GT, et al. Effect of vasoactive therapy used for brain-dead donors on graft survival after kidney transplantation. Transplant Proc. 2018;50:1289–1291. doi: 10.1016/j.transproceed.2018.02.058. [DOI] [PubMed] [Google Scholar]
- 15.McKeown DW, Bonser RS, Kellum JA. Management of the heartbeating brain-dead organ donor. Br J Anaesth. 2012;108:i96–i107. doi: 10.1093/bja/aer351. [DOI] [PubMed] [Google Scholar]
- 16.Maciel CB, Greer DM. ICU management of the potential organ donor: state of the art. Curr Neurol Neurosci Rep. 2016;16:86. doi: 10.1007/s11910-016-0682-1. [DOI] [PubMed] [Google Scholar]
- 17.Perner A, Haase N, Guttormsen AB, et al. Hydroxyethyl starch 130/0.42 versus Ringer’s acetate in severe sepsis. N Engl J Med. 2012;367:124–134. doi: 10.1056/NEJMoa1204242. [DOI] [PubMed] [Google Scholar]
- 18.Myburgh JA, Finfer S, Bellomo R, et al. Hydroxyethyl starch or saline for fluid resuscitation in intensive care. N Engl J Med. 2012;367:1901–1911. doi: 10.1056/NEJMoa1209759. [DOI] [PubMed] [Google Scholar]
- 19.Patel MS, Niemann CU, Sally MB, et al. The impact of hydroxyethyl starch use in deceased organ donors on the development of delayed graft function in kidney transplant recipients: a propensity-adjusted analysis. Am J Transplant. 2015;15:2152–2158. doi: 10.1111/ajt.13263. [DOI] [PubMed] [Google Scholar]
- 20.Miñambres E, Ballesteros MA, Rodrigo E, et al. Aggressive lung donor management increases graft procurement without increasing renal graft loss after transplantation. Clin Transplant. 2013;27:52–59. doi: 10.1111/j.1399-0012.2012.01690.x. [DOI] [PubMed] [Google Scholar]
- 21.Youn TS, Greer DM. Brain death and management of a potential organ donor in the intensive care unit. Crit Care Clin. 2014;30:813–831. doi: 10.1016/j.ccc.2014.06.010. [DOI] [PubMed] [Google Scholar]
- 22.Tisdall M, Crocker M, Watkiss J, Smith M. Disturbances of sodium in critically ill adult neurologic patients. J Neurosurg Anesthesiol. 2006;18:57–63. doi: 10.1097/01.ana.0000191280.05170.0f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Plurad DS, Bricker S, Neville A, et al. Arginine vasopressin significantly increases the rate of successful organ procurement in potential donors. Am J Surg. 2012;204:856–861. doi: 10.1016/j.amjsurg.2012.05.011. [DOI] [PubMed] [Google Scholar]
- 24.Busl KM, Bleck TP. Neurogenic pulmonary edema. Crit Care Med. 2015;43:1710–1715. doi: 10.1097/CCM.0000000000001101. [DOI] [PubMed] [Google Scholar]
- 25.López-Aguilar J, Villagrá A, Bernabé F, et al. Massive brain injury enhances lung damage in an isolated lung model of ventilator-induced lung injury*. Crit Care Med. 2005;33:1077–1083. doi: 10.1097/01.ccm.0000162913.72479.f7. [DOI] [PubMed] [Google Scholar]
- 26.Mascia L, Sakr Y, Pasero D, et al. Extracranial complications in patients with acute brain injury: a post-hoc analysis of the SOAP study. Intensive Care Med. 2008;34:720–727. doi: 10.1007/s00134-007-0974-7. [DOI] [PubMed] [Google Scholar]
- 27.MacLean A, Dunning J. The retrieval of thoracic organs: donor assessment and management. Br Med Bull. 1997;53:829–843. doi: 10.1093/oxfordjournals.bmb.a011651. [DOI] [PubMed] [Google Scholar]
- 28.Wheeler AP, Bernard GR. Acute lung injury and the acute respiratory distress syndrome: a clinical review. Lancet. 2007;369:1553–1564. doi: 10.1016/S0140-6736(07)60604-7. [DOI] [PubMed] [Google Scholar]
- 29.Krebs J, Tsagogiorgas C, Pelosi P, et al. Open lung approach with low tidal volume mechanical ventilation attenuates lung injury in rats with massive brain damage. Crit Care. 2014;18:R59. doi: 10.1186/cc13813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mascia L, Pasero D, Slutsky AS, et al. Effect of a lung protective strategy for organ donors on eligibility and availability of lungs for transplantation. JAMA. 2010;304:2620. doi: 10.1001/jama.2010.1796. [DOI] [PubMed] [Google Scholar]
- 31.ARDSNet Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med. 2000;342:1301. doi: 10.1056/NEJM200005043421801. [DOI] [PubMed] [Google Scholar]
- 32.Slutsky AS, Ranieri VM. Ventilator-induced lung injury. N Engl J Med. 2013;369:2126–2136. doi: 10.1056/NEJMra1208707. [DOI] [PubMed] [Google Scholar]
- 33.Botha P. Extended donor criteria in lung transplantation. Curr Opin Organ Transplant. 2009;14:206–210. doi: 10.1097/mot.0b013e328326c834. [DOI] [PubMed] [Google Scholar]
- 34.Mascia L, Bosma K, Pasero D, et al. Ventilatory and hemodynamic management of potential organ donors: an observational survey*. Crit Care Med. 2006;34(2):321–327. doi: 10.1097/01.ccm.0000196828.87358.6e. [DOI] [PubMed] [Google Scholar]
- 35.Howlett TA, Keogh AM, Perry L, et al. Anterior and posterior pituitary function in brain-stem-dead donors. A possible role for hormonal replacement therapy. Transplantation. 1989;47:828–834. doi: 10.1097/00007890-198905000-00016. [DOI] [PubMed] [Google Scholar]
- 36.Gramm HJ, Meinhold H, Bickel U, et al. Acute endocrine failure after brain death? Transplantation. 1992;54:851–857. doi: 10.1097/00007890-199211000-00016. [DOI] [PubMed] [Google Scholar]
- 37.Novitzky D, Mi Z, Sun Q, et al. Thyroid hormone therapy in the management of 63,593 brain-dead organ donors. Transplantation. 2014;98:1119–1127. doi: 10.1097/TP.0000000000000187. [DOI] [PubMed] [Google Scholar]
- 38.MacDonald PS, Aneman A, Bhonagiri D, et al. A systematic review and meta-analysis of clinical trials of thyroid hormone administration to brain dead potential organ donors. Crit Care Med. 2012;40:1635–1644. doi: 10.1097/CCM.0b013e3182416ee7. [DOI] [PubMed] [Google Scholar]
- 39.Dimopoulou I, Tsagarakis S, Anthi A, Milou E, Ilias I, Stavrakaki K, Charalambidis C, Tzanela M, Orfanos S, Mandragos K, Nikolaos Thalassinos CR. High prevalence of decreased cortisol reserve in brain-dead potential organ donors. Crit Care Med. 2003;31:1113–1117. doi: 10.1097/01.CCM.0000059644.54819.67. [DOI] [PubMed] [Google Scholar]
- 40.Dupuis S, Amiel J-A, Desgroseilliers M, et al. Corticosteroids in the management of brain-dead potential organ donors: a systematic review. Br J Anaesth. 2014;113:346–359. doi: 10.1093/bja/aeu154. [DOI] [PubMed] [Google Scholar]
- 41.Pinsard M, Ragot S, Mertes P, et al. Interest of low-dose hydrocortisone therapy during brain-dead organ donor resuscitation: the CORTICOME study. Crit Care. 2014;18:R158. doi: 10.1186/cc13997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Marvin MR, Morton V. Glycemic control and organ transplantation. J Diabetes Sci Technol. 2009;3:1365–1372. doi: 10.1177/193229680900300616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Casaer MP, Van den Berghe G. Nutrition in the acute phase of critical illness. N Engl J Med. 2014;370:1227–1236. doi: 10.1056/NEJMra1304623. [DOI] [PubMed] [Google Scholar]
- 44.Hahnenkamp K, Böhler K, Wolters H, et al. Organ-protective intensive care in organ donors. Dtsch Aerzteblatt Online. Dtsch Arztebl Int. 2016;113(33–34):552–558. doi: 10.3238/arztebl.2016.0552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Weiss S, Kotsch K, Francuski M, et al. Brain death activates donor organs and is associated with a worse I/R injury after liver transplantation. Am J Transplant. 2007;7:1584–1593. doi: 10.1111/j.1600-6143.2007.01799.x. [DOI] [PubMed] [Google Scholar]
- 46.Joseph B, Khalil M, Pandit V, et al. Hypothermia in organ donation: a friend or foe? J Trauma Acute Care Surg. 2014;77(4):559–563. doi: 10.1097/TA.0000000000000413. [DOI] [PubMed] [Google Scholar]
- 47.Citerio G, Crippa IA, Bronco A, et al. Variability in brain death determination in Europe: looking for a solution. Neurocrit Care. 2014;21:376–382. doi: 10.1007/s12028-014-9983-x. [DOI] [PubMed] [Google Scholar]
- 48.Niemann CU, Feiner J, Swain S, et al. Therapeutic hypothermia in deceased organ donors and kidney-graft function. N Eng J Med. 2015;373:405–414. doi: 10.1056/NEJMoa1501969. [DOI] [PubMed] [Google Scholar]
- 49.Schnuelle P, Mundt HM, Drüschler F, et al. Impact of spontaneous donor hypothermia on graft outcomes after kidney transplantation. Am J Transplant. 2018;18:704–714. doi: 10.1111/ajt.14541. [DOI] [PubMed] [Google Scholar]
- 50.Hébert PC, Carson JL. Transfusion threshold of 7 g per deciliter—the new normal. N Engl J Med. 2014;371:1459–1461. doi: 10.1056/NEJMe1408976. [DOI] [PubMed] [Google Scholar]
- 51.Docherty AB, Turgeon AF, Walsh TS. Best practice in critical care: anaemia in acute and critical illness. Transfus Med. 2018;28:181–189. doi: 10.1111/tme.12505. [DOI] [PubMed] [Google Scholar]
- 52.Coberly EA, Booth GS. Ten-year retrospective review of transfusion practices in beating-heart organ donors. Transfusion. 2016;56:339–343. doi: 10.1111/trf.13340. [DOI] [PubMed] [Google Scholar]
- 53.De La Cruz JS, Sally MB, Zatarain JR, et al. The impact of blood transfusions in deceased organ donors on the outcomes of 1,884 renal grafts from United Network for Organ Sharing Region. J Trauma Acute Care Surg. 2015;79(4 Suppl 2):S164–S170. doi: 10.1097/TA.0000000000000670. [DOI] [PubMed] [Google Scholar]
- 54.Connor JP, Raife T, Medow JE. Outcomes of red blood cell transfusions prescribed in organ donors by the Digital Intern, an electronic decision support algorithm. Transfusion. 2018;58:366–371. doi: 10.1111/trf.14424. [DOI] [PubMed] [Google Scholar]
- 55.Lisman T, Leuvenink HGD, Porte RJ, Ploeg RJ. Activation of hemostasis in brain dead organ donors: an observational study. J Thromb Haemost. 2011;9:1959–1965. doi: 10.1111/j.1538-7836.2011.04442.x. [DOI] [PubMed] [Google Scholar]
- 56.Valdivia M, Chamorro C, Romera MA, et al. Effect of posttraumatic donor’s disseminated intravascular coagulation in intrathoracic organ donation and transplantation. Transplant Proc. 2007;39:2427–2428. doi: 10.1016/j.transproceed.2007.07.052. [DOI] [PubMed] [Google Scholar]
- 57.Garrouste C, Baudenon J, Gatault P, et al. No impact of disseminated intravascular coagulation in kidney donors on long-term kidney transplantation outcome: A multicenter propensity-matched study. Am J Transplant. 2018;19(2):448–456. doi: 10.1111/ajt.15008. [DOI] [PubMed] [Google Scholar]
- 58.Trotter PB, Robb M, Summers D, et al. Donors with immune thrombocytopenia: do they pose a risk to transplant recipients? Am J Transplant. 2017;17:796–802. doi: 10.1111/ajt.14105. [DOI] [PubMed] [Google Scholar]
- 59.Fishman JA, Greenwald MA, Grossi PA. Transmission of infection with human allografts: essential considerations in donor screening. Clin Infect Dis. 2012;55:720–727. doi: 10.1093/cid/cis519. [DOI] [PubMed] [Google Scholar]
- 60.Aguilar C, Husain S, Lortholary O. Recent advances in understanding and managing infectious diseases in solid organ transplant recipients. F1000Research. 2018;7:661. doi: 10.12688/f1000research.14262.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Fischer SA. Is this organ donor safe?: Donor-derived infections in solid organ transplantation. Surg Clin N Am. 2019;99:117–128. doi: 10.1016/j.suc.2018.09.009. [DOI] [PubMed] [Google Scholar]
- 62.Len O, Garzoni C, Lumbreras C, et al. Recommendations for screening of donor and recipient prior to solid organ transplantation and to minimize transmission of donor–derived infections. Clin Microbiol Infect. 2014;20:10–18. doi: 10.1111/1469-0691.12557. [DOI] [PubMed] [Google Scholar]
- 63.Vincent J-L, Marshall JC, Namendys-Silva SA, et al. Assessment of the worldwide burden of critical illness: the intensive care over nations (ICON) audit. Lancet Respir Med. 2014;2:380–386. doi: 10.1016/S2213-2600(14)70061-X. [DOI] [PubMed] [Google Scholar]
- 64.Organ Procurement and Transplantation Network Guidance for Recognizing Central Nervous System Infections in Potential Deceased Organ Donors. https://optn.transplant.hrsa.gov/resources/guidance/guidance-for-recognizing-central-nervous-system-infections-in-potential-deceased-organ-donors/. Accessed 10 Dec 2018
- 65.Nogueira ML, Estofolete CF, Terzian ACB, et al. Zika virus infection and solid organ transplantation: a new challenge. Am J Transplant. 2017;17:791–795. doi: 10.1111/ajt.14047. [DOI] [PubMed] [Google Scholar]
- 66.Muller E, Barday Z, Mendelson M, Kahn D. HIV-positive–to–HIV-positive kidney transplantation—results at 3 to 5 years. N Engl J Med. 2015;372:613–620. doi: 10.1056/NEJMoa1408896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Schlendorf KH, Zalawadiya S, Shah AS, et al. Early outcomes using hepatitis C-positive donors for cardiac transplantation in the era of effective direct-acting anti-viral therapies. J Hear Lung Transplant. 2018;37:763–769. doi: 10.1016/j.healun.2018.01.1293. [DOI] [PubMed] [Google Scholar]
- 68.Durand CM, Bowring MG, Brown DM, et al. Direct-acting antiviral prophylaxis in kidney transplantation from hepatitis C virus-infected donors to noninfected recipients. Ann Intern Med. 2018;168:533. doi: 10.7326/M17-2871. [DOI] [PMC free article] [PubMed] [Google Scholar]