Abstract
Objective
To describe the daily practice of mechanical ventilation (MV), and secondarily, its outcome in pediatric intensive care units (PICUs).
Design
Prospective cohort of infants and children who received MV for at least 12 h.
Setting
Thirty-six medical surgical PICUs.
Patients
All consecutive patients admitted to the PICUs during 2-month period.
Measurements and main results
Of the 1893 patients admitted, 659 (35%) received MV for a median time of 4 days (25th percentile, 75%: 2, 6). Median of age was 13 months (25th percentile, 75%: 5, 48). Common indications for MV were acute respiratory failure (ARF) in 72% of the patients, altered mental status in 14% of the patients, and ARF on chronic pulmonary disease in 10% of the patients. Median length of stay in the PICUs was 8 days (25th percentile, 75%: 5, 13). Overall mortality rate in the PICUs was 15% (confidence interval 95%: 13–18) for the entire population, 50% (95% CI: 25–74) in patients who received MV because of acute respiratory distress syndrome, 24% (95% CI: 16–35) in patients who received MV for altered mental status and 16% (95% CI: 9–29) in patients who received MV for ARF on chronic pulmonary disease.
Conclusion
One in every 3 patients admitted to the PICUs requires ventilatory support.. The ARF was the most common reason for MV, and survival of unselected infants and children receiving MV for more than 12 h was 85%.
Electronic Supplementary Material
Supplementary material is available in the online version of this article at http://dx.doi.org/10.1007/s00134-004-2225-5
Keywords: Mechanical ventilation, Children, Infants, Survey, Weaning, Outcomes
Introduction
Mechanical ventilation (MV) with positive pressure is a technique that has been employed in the PICUs with increasing frequency. The percentage of mechanically ventilated infants and children varies from 30 to 64% [1, 2, 3, 4]. Since its introduction into the modern ICUs, MV has undergone continuous evolution. There has been an explosion of new ventilator modes, many of which have been incorporated into routine clinical practice without evidence of their efficacy o their superiority over other modes of ventilation. Indeed, in most cases physicians must rely only on short studies performed on small numbers of patients to help them decide which mode of ventilatory support they should use for their patients with acute respiratory failure.
The introduction of new modes of ventilator support requires an understanding of the epidemiology and outcome on the daily practice of the MV. There are remarkably few epidemiological studies concerning of infants and children mechanically ventilated. Harel and coworkers reported the results of a survey conducted by mail in the United States [5]. Of the pediatric critical care physicians that responded, most of them indicated that synchronized intermittent mandatory ventilation (SIMV) was their mode of first choice; however, the main limitation of mail survey is that they represented self-reported practices rather than the actual practices.
As a high-cost technology, MV will likely be increasingly scrutinized due the increased focus on improving cost-efficiency and documenting patient outcomes [6, 7, 8]. It is possible that examining the daily practice in “average” PICUs will enhance the information available on use of ventilatory modes, setting, and outcomes of mechanically ventilated infants and children.
Methods
We conducted a prospective cohort study between 1 April 1999 and 31 May 1999 in 36 volunteer PICUs located in seven countries (Argentina, n=10; Costa Rica, n=1; UK, n=2; Greece, n=2; Panama, n=1; Spain, n=18; and USA, n=2). Before the collection of data, the study protocol was reviewed and approved by Institutional Review Committees of each hospital. For a PICU to be included in the study, it had to possess six or more beds and at least 60% of the staff had to have undergone PICU training and/or more than 5 years of experience in a PICU. Neonatal ICUs, postoperative recovery rooms, and postoperative cardiovascular ICUs were excluded. Patients were enrolled if they met the following conditions: age between 1 months and 15 years; and a requirement of either invasive or noninvasive MV for more than 12 h. Patients receiving MV at the study PICUs for less than 12 h or patients in whom MV was started at another hospital and its duration was longer than 24 h were excluded.
To minimize a change in behavior as a result of being observed, only the investigators and research coordinators of a given PICU were aware of the study. Each investigator and research coordinator was provided with a comprehensive manual describing data collection requirements and definitions. To assure the quality of the data before the initiation of the study, forms were filled in at least 10 patients during 1 month in three different PICUs. Based on difficulties detected during initial evaluation, forms were modified before the initiation of the study. Each questionnaire was checked by the main authors (J.A.F., F.F., A.E.) to identify omissions and inconsistent data were corrected.
Data sheets were used to collect information on each hospital, each PICU study, and each patient receiving ventilatory support. The information on the PICU and the hospital included the number of beds in the hospital, number of beds in the PICU, number of patients in the PICU at the beginning of the study, and number of patients receiving MV during that time. The following information was collected in each enrolled patient:
Demographic data. The sex, age, and weight of each patient, date of admission to the PICUs, PRISM score at the time of admission to the PICUs [9], chronic functional status, date of initiating MV, and mode of access to the patient’s airway: orotracheal intubation, nasotracheal intubation, facial mask, or tracheostomy were recorded. If a patient had undergone a tracheostomy during the study, the date of surgery was recorded.
Reason for MV. The indication for the initiation of MV on admission was selected from the following predefined list of categories: (a) acute exacerbation of chronic pulmonary disease which described patients with bronchopulmonary dysplasia, cystic fibrosis, pulmonary or thoracic malformations, requiring MV due to infection, bronchospasm, or another acute episode; (b) altered mental status which described patients requiring MV due to loss of consciousness secondary to organic condition, infection, or electrolyte disturbance, such as cerebral hemorrhage, meningitis, and hypernatremia, respectively; (c) neuromuscular disease, which described patients whose respiratory failure was due to diseases and disorders of the peripheral nerves, myoneural junction, or muscle; (d) acute respiratory failure (ARF), which described patients without a pre-existing obstructive or restrictive lung disease requiring MV because of respiratory failure.
Whenever a patient had more than one cause of indication for MV, the data collector recorded the reason judged dominant.
The patients who fell in the category of ARF were separated into the following subgroups: (a) pneumonia, defined by the development of a new alveolar infiltrate or worsening of previous alveolar infiltrates, accompanied by fever/hypothermia, and leukocytosis/leukopenia; (b) bronchiolitis defined by pre-established criteria [10]: tachypnea; cough; chest retractions; prolonged expiratory time; pulmonary rales; and hyperinflation of the lungs on chest radiographs; and the patient requiring ventilatory support due to any of the following: refractory hypoxemia; excessive work of breathing; respiratory pauses or apnea, or both; respiratory acidosis (pH<7.25, PaCO2>60 mmHg); (c) aspiration, defined by visualization of gastric contents in the airway or in a tracheal aspirate; (d) postoperative state, consisting of patients who required the continuation of MV following surgery because of a serious underlying medical problem or the high risk of the operative procedure; (e) sepsis/septic shock, defined by pre-established criteria [11]; (f) upper airway obstruction due to infection, e.g., epiglottitis, laryngotracheobronchitis; etc. (g) ARDS, defined according to the criteria of ATS/ERS consensus conference [12] and the patients met criteria for ARDS within 48 h from PICU or hospital admission; (h) trauma, MV was initiated due to chest, abdominal, or cranial trauma; (i) heart failure, consisting of patients with dyspnea, bilateral alveolar infiltrates, hypoxemia, and evidence of cardiogenic shock due to heart rate abnormalities, cardiomyopathies, or congenital heart disease; (g) any other etiology of ARF not mentioned above.
-
3.
Ventilator data. The first arterial blood gas measurements at 8:00 a.m. and corresponding settings were recorded daily while patients were mechanically ventilated for a maximum of 28 days. The ventilator modes were defined as in a previous study [5]. The duration of MV was defined as the time elapsed from the initiation of ventilatory support to the onset of weaning. The onset of weaning was the time the physicians in charge considered the patient was able to be discontinued from MV and either the level of pressure support or the mandatory respiratory rate were decreased or a trial of spontaneous breathing (SBT) was performed. The duration of weaning was defined as the time elapsed from the onset of it to the extubation. Accidental removal of endotracheal tubes (ETT) was defined as the presence of any of the following events: patient related if ETT was removed deliberately by the patients, either with their hands or by making voluntary movements that led directly to the removal; care provider related if ETT was removed as a consequence of inadequate handling by medical personnel. The rate of accidental removal of ETT was calculated, as follows: (a) accidental removal to the number of ETT placed/number of ETT placed; or (b) accidental removal according to the time at risk–number of ETT accidentally removed ×100/sum of the time that ETT remained placed. The need for reintubation within 48 h after extubation and the time of reintubation were recorded. All patients were followed-up until discharge from the hospital or death.
Statistical analysis
Data are shown as mean and standard deviation, median with the interquartile range and proportions as appropriate. All categorical variables were analyzed with the chi-square test, except where small size required the use of Fisher’s exact test. Comparison of continuous variables was made with Student’s t test for variables with normal distribution or the Mann-Whitney U test for variables with abnormal distribution and the Kruskal-Wallis test was used to compare continuous variables among more than two groups.
Results
During the 2-month period study, 1893 patients were admitted to the PICUs and 659 (35%; 95% CI: 36–37) of them received MV for more than 12 h. The median number of patients admitted in each PICU was 32 (25, 75th percentile, 28–73) and the median number of patients mechanically ventilated in each PICU was 14 (25th percentile, 75% 12–27). Demographic characteristics and the indication for MV are listed in Table 1.
Table 1.
Characteristics of the studied patients on admission to PICU
| Age: months, median (percentile 25, 75) | 13 (5, 48) |
| Females: n (%, 95% CI) | 262 (40, 36–44) |
| Weight: kg, median (percentile 25, 75) | 9 (5, 16) |
| PRISM score: points, median (percentile 25, 75) | 13 (11, 16) |
| Prior functional status | |
| Normal: n (%, 95% CI) | 541 (82, 79–81) |
| Chronic condition: n (%, 95% CI) | 105 (16, 13–19) |
| Problem | |
| Medical: n (%, 95% CI) | 429 (65, 61–69) |
| Surgical: n (%, 95% CI) | 230 (35, 31–38) |
| Reason for the initiation of mechanical ventilation | |
| Acute respiratory failure: n (%, 95% CI)a | 472 (72, 68–75) |
| Acute pulmonary disease | 172 (25, 22–29) |
| Pneumonia: n (%, 95% CI) | 100 (15, 13–18) |
| Bronchiolitis, n (%, 95% CI) | 35 (5, 4–7) |
| ARDS, n (%, 95% CI) | 16 (2, 1–4) |
| Aspiration, n (%, 95% CI) | 15 (2, 1–4) |
| Upper airway obstruction, n (%, 95% CI) | 5 (1, 0–2) |
| Postoperative, n (%, 95% CI) | 161 (24, 21–28) |
| Sepsis, n (%, 95% CI) | 70 (11, 8–13) |
| Trauma, n (%, 95% CI) | 31 (5, 3–7) |
| Congestive heart failure, n (%, 95% CI) | 22 (3, 2–5) |
| Other: n (%, 95% CI) | 71 (11, 9–13) |
| Acute or chronic respiratory failure | |
| Chronic obstructive disease: n (%, 95% CI) | 51 (8, 6–10) |
| Asthma: n (%, 95% CI) | 10 (1, 1–3) |
| Altered mental status: n (%, 95% CI) | 87 (14, 11–17) |
| Neuromuscular disease: n (%, 95% CI) | 6 (1, 0–2) |
aMore than one cause of acute respiratory failure per patient was permitted
All the PICUs included in the study were medical–surgical. The median number of hospital beds was 272 (25th, 75th percentiles: 146–482) and the number of the beds in the PICUs was a mean of 11±4. Eighty-five percent of the participants in PICUs were located at pregraduate teaching hospital and 97% in postgraduate teaching hospitals.
The access to the airway for delivery of MV was through an ETT in 635 (96%; 95% CI: 94–97) of patients, a tracheostomy in 11 (2%; 95% CI: 1–3), and facial mask in 10 (1.5%; 95% CI: 1–3). Of the ETT, 398 (63%) were passed through the mouth and 237 (37%) through the nose. In those patients who underwent a tracheostomy during the study period, it was performed after a median of 12 days from the date of tracheal intubation (25th and 75th percentiles: 6, 18).
The modes of MV most commonly used during the whole study period are shown in the Fig. 1. Two hundred fifty-seven patients (39%; 95% CI: 35–43) received SIMV alone or in combination with pressure support ventilation (PSV). One hundred sixty-six patients (25%; 95% CI: 22–29) received pressure control ventilation (PCV) and an almost equal number of patients, 165 (23%; 95% CI: 20–26), received volume limited ventilation-assisted/control (VLV–A/C). Through the period of study, 53% of the patients received more than one mode of ventilation and there were not differences in the ventilatory modes used among the categories of respiratory failure.
Fig. 1.
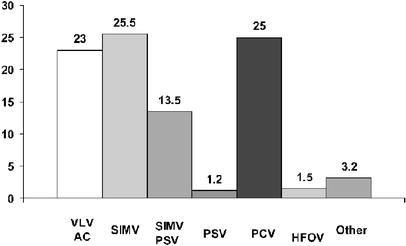
Modes of mechanical ventilation used during study period. VLV-A/C volume limited ventilation—assist/control, SIMV synchronized intermittent mandatory ventilation, PCV pressure control ventilation, PSV pressure support ventilation, SIMV+PSV synchronized intermittent mandatory plus pressure support ventilation, HFOV high-frequency oscillatory ventilation
The settings of tidal volume (TV), mandatory respiratory rate, positive end-expiratory pressure (PEEP), and peak inspiratory pressure (PIP), at different days of MV, are shown in the Table 2. In patients with ARDS, PIP was significantly higher than in patients with acute on chronic pulmonary diseases (p<0.001). Similarly, a significantly higher level of PEEP was employed in patients with ARDS when compared with patients with acute on chronic pulmonary disease (p<0.001).
Table 2.
Ventilatory parameters and blood arterial gases on days 1, 4, and 6 of mechanical ventilation. Data are expressed as median (interquartile range). ARDS acute respiratory distress syndrome, PEEP positive end-expiratory pressure
| Day 1 | Day 4 | Day 6 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Overall | Acute on CPDc | ARDS | Overall | Acute on CPD | ARDS | Overall | Acute on CPD | ARDS | |
| Ventilatory parameters | |||||||||
| Tidal volume (ml/kg) | 11 (9, 13) | 11 (10, 14) | 11 (10, 14) | 10 (9, 13) | 14 (9, 19) | 10 (8, 14) | 11 (9, 14) | 17 (14, 20) | 11 (7, 14) |
| Respiratory rate (bpm) | 25 (20, 30) | 25 (20, 32) | 24 (22, 30) | 26 (20, 34) | 30 (25, 35) | 24 (21, 28) | 26 (20, 35) | 26 (18, 30) | 24 (21, 28) |
| Patients with PEEP (n, %) | 549 (83%) | 46 (71%) | 15 (95%) | 255 (75%) | 25 (69%) | 12 (80%) | 153 (80%) | 14 (67%) | 12 (91%) |
| Applied PEEP (cm H2O)a | 4 (2, 5) | 4 (3, 5) | 8 (5, 10) | 4 (3, 5) | 4 (3, 5) | 6 (6, 10) | 4 (3, 5) | 3 (2, 5) | 5 (4, 9) |
| Peak pressure (cm H2O) | 24 (20, 28) | 28 (23, 30) | 36 (23, 40) | 25 (20, 30) | 26 (21, 33) | 37 (28, 40) | 25 (20, 30) | 39 (23, 35) | 33 (24, 40) |
| Blood arterial gases | |||||||||
| pH | 7.39 (7.34, 7.45) | 7.39 (7.32, 7.45) | 7.40 (7.36, 7.45) | 7.41 (7.36, 7.46) | 7.36 (7.43, 7.50) | 7.41 (7.28, 7.47) | 7,41 (7.35, 7.46) | 7.40 (7.36, 7.43) | 7.43 (7.35, 7.46) |
| PaCO2 (mm Hg) | 39 (32, 46) | 48 (38, 60) | 37 (32, 52) | 41 (36, 50) | 47 (38, 62) | 45 (41, 50) | 45 (39, 54) | 57 (44, 69) | 46 (40,50) |
| Ratio PaO2 to FiO2 | 221 (125, 364) | 126 (92, 204) | 88 (65, 181) | 191 (124, 278) | 135 (111, 185) | 124 (111, 139) | 167 (106, 257) | 125 (90, 184) | 105 (72,213) |
aOnly includes patients with PEEP
b p<0.001 (Kruskal-Wallis test)
cAcute on CPD denotes acute on chronic pulmonary disease and is constituted by patients with chronic obstructive disease and asthma
The weaning was initiated after 5±6 days of MV, and an additional 2.6±3.6 days elapsed before the removal of ventilatory support; thus, 46% of the total time of MV was devoted to liberation from ventilatory support. The methods of weaning used during the whole period of the study are shown in the Fig. 2. A total of 590 attempts of weaning were performed in 563 patients (85%). The SBT was used in 175 (31%; 95% CI: 27–35) of the attempts and the reduction gradual of either pressure support or mandatory respiratory frequency was used in 372 (66%; 95% CI 62–70) of the attempts.
Fig. 2.
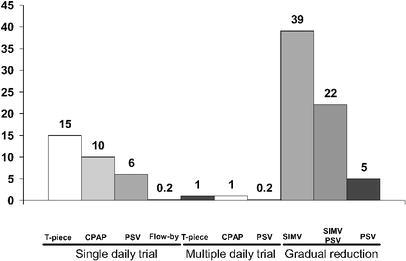
Modes of weaning used in 590 attempts. SIMV synchronized intermittent mandatory ventilation, PSV pressure support ventilation, SIMV+PSV synchronized intermittent mandatory plus pressure support ventilation, CPAP continuous positive airway pressure
During the period of the study the patients had 635 ETT in place during a total time 4204 days. The rate of accidental extubation was 4% (0.61 per 100 days of intubation). The extubation outcome is shown in Table 3. The mortality was significantly higher among patients who required reintubation when compared with those successfully extubated after planned extubation (19 vs 5%; p<0.05).
Table 3.
Extubation outcomes
| Mortality: n (%, 95% CI) | ||
|---|---|---|
| Extubation: n (%, 95% CI) | ||
| Overall | 550 (85, 82–87) | 26 (5, 3–7) |
| Scheduled | 524 (81, 78–84) | 24 (5, 3–7) |
| Accidental | 26 (4, 3–6) | 2 (8, 1–27) |
| Reintubation: n (%, 95% CI) | ||
| Overall | 56 (10, 8–13) | 11 (20, 11–33) |
| After scheduled extubation | 48 (9, 7–12) | 9 (19, 9–33) |
| After accidental extubation | 8 (31, 15–52) | 2 (25, 4–64) |
The outcomes according to the reason for the initiation of MV are shown in Table 4. Both, the in-unit mortality and the hospital mortality, were significantly higher among patients with ARDS when compared with those patients with acute on chronic pulmonary disease or ARF non-ARDS (p<0.05 and p<0.01), respectively. Similarly, the mortality among patients with coma was higher than the mortality of patients with ARF non-ARDS (p<0.05).
Table 4.
Outcome of the mechanically ventilated children
| Overall (n=659) | Acute or chronic pulmonary disease (n=61) | Acute respiratory distress syndrome (n=16) | Acute respiratory failure, no ARDS (n=456) | Coma (n=87) | p valuec | |
|---|---|---|---|---|---|---|
| Days of mechanical ventilation, median (percentile: 25, 75%) | ||||||
| Survivors | 3 (2, 6) | 4 (2, 8) | 3 (2, 4) | 3 (2, 6) | 3 (2, 4) | 0.02 |
| Non-survivors | 4 (3, 7) | 4 (2, 8.5) | 4 (3, 6) | 4 (3, 7) | 4 (3, 6) | 0.03 |
| Days of weaning, median (percentile: 25, 75%) | ||||||
| Survivors | 2 (1, 2) | 1 (2, 3) | 2 (1, 3) | 2 (1, 2) | 2 (1, 3) | 0.80 |
| Non-survivors | 2 (1, 5) | 1 (1, 5) | – | 2 (1, 6) | 1.5 (1, 4) | 0.40 |
| Days of stay in the PICU, median (percentile: 25, 75%) | ||||||
| Survivors | 8 (5, 13) | 9 (5, 15) | 17 (6, 27) | 8 (5, 13) | 6 (4, 9) | 0.005 |
| Non-survivors | 7 (4, 13) | 7 (5, 22) | 5 (3, 10.5) | 7 (4, 12) | 5 (3, 10) | 0.17 |
| Days of stay in the hospital, median (percentile: 25, 75%) | ||||||
| Survivors | 17 (10, 32) | 20 (10, 31.5) | 31 (11, 47) | 17.5 (11, 34) | 13 (10, 24) | 0.14 |
| Non-survivors | 11.5 (5, 27.5) | 20 (5.5, 41) | 5 (3, 18.5) | 12 (5, 23) | 5 (3, 18) | 0.09 |
| Mortality | ||||||
| PICU: n [%, (95% CI)] | 101 [15 (13–18)] | 10 [16 (9–29)]a | 8 [50 (25–74)] | 60 [13 (10–17)]a,b | 21 [24 (16–35)] | |
| Hospital: n [% (95% CI)] | 116 [18 (15–21)] | 13 [21 (12–34 )]a | 8 [50 (25–74)] | 70 [15 (12–19)]a,b | 23 [26 (18–37)] | |
a p<0.05 for the comparison between acute or chronic pulmonary disease and ARDS and for the comparison between ARF non-ARDS and coma
b p<0.01 for the comparison between ARF non-ARDS and ARDS
cKruskal-Wallis test
Discussion
Our study represented the largest cohort of infants and children mechanically ventilated for more than 12 h from whom data were collected prospectively; however, most of the PICUs participating were from Argentina or Spain, so it is difficult to know to what extent our data contribute to the characterization of the daily practice of MV in PICUs around the world.
We found that 35% of the infants and children admitted to the PICUs received MV for more than 12 h. The percentage of pediatric patients mechanically ventilated has been reported to occur at rates of 30–64% [1, 2, 3, 4]. Martinot et al. [2] reported the results of a multicentric French study, with over 720 admitted patients, in which 64% of them were mechanically ventilated. The same percentage of use of MV was found in 1061 patients from six Latin American PICUs [3]. Lopez-Herce et al. [4] reported the results of a questionnaire survey of PICUs from Spain in which, of over 9585 patients admitted during a year, 32% received MV. We found that the percentage of mechanically ventilated children compares more closely with this later study. The wide variation in the percentage of ventilated patients could be related to several factors such as regionalization of PICUs, political admission and discharges of PICUs, severity of illness, and seasonal variation. The time period of this study missed respiratory syncytial virus season for most of the participating PICUs; hence, the percentage of patients receiving MV would have been higher.
Acute respiratory failure is the most common underlying diagnosis indicating the need for MV, being the case in 75% of our ventilated patients. Among the subgroups of ARF, the higher proportions were the acute pulmonary disease and postoperative state. Acute pulmonary disease was seen to be the main reason for the initiation of MV in developing countries [13, 14], whereas postoperative state seems to be the main indication for institute ventilatory support in developed countries [15]. To date, there has been significant progress in our understanding of pathophysiology in patients with ARDS, and innovative therapies for ARDS in children are available. Accordingly, several attempts have been made to obtain precise information on the incidence of ARDS. In both, adult and children ventilated patients, the reported incidence of ARDS ranged from 2 to 4% [16, 17, 18, 19, 20, 21]. Now, we have found that ARDS accounted for only 2% of patients who required MV at admission to the PICUs.
The ventilatory mode most frequently used was SIMV alone or in combination with PSV in 39% of patients. Harel et al. [5], in a questionnaire survey carried out in physicians in caring for patients in PICUs during 1995, reported that the preferred mode for initiating the MV in infants and children was SIMV with a percentage ranging from 65 to 82%, followed by PCV (2–17%); however, our results show a greater proportion of use of PCV and VLV–A/C than the one preferred by the physicians in the aforementioned study. Different reasons may be attributed for the lack of uniformity in the selection of ventilatory modes in the pediatric population. One reason is the lack of published studies assessing the most beneficial ventilatory mode in terms of mortality or in reducing the time that patient remains on ventilatory support
Weaning from MV accounted for 46% of the total time spent in receiving ventilatory support, and the most commonly used weaning method was SIMV, which was gradually reduced in 39% of the patients. In Harel et al.’s study [5] this was also the preferred mode for liberation from MV, with percentages ranging between 34 and 38%.
Weaning is an aspect of MV that has been the subject of several randomized and controlled trials. In adult patients it has been demonstrated that SIMV is the worst method for weaning when it was compared with SBT or PSV [22, 23]. Indeed, two large recent studies in children showed that not all the patients need gradual reduction of MV [13, 24]. The use of SBT appears to have changed over the past few years. For example, this method was not reported in Harel et al.’s [5] study, but we found that 30% of mechanically ventilated patients used this method to gain liberation from MV. The SBT has been shown to be a useful approach in identifying those patients who are ready for extubation [13, 14, 25]. Although, its implementation in infants and children mechanically ventilated could simplify the approach to weaning and shorten the duration of MV, it is an aspect that requires further investigation.
The selection of ventilatory settings in pediatric patients with ARF at different days of ventilatory support has been infrequently reported; thus, comparing our results with other series is difficult. Nevertheless, the selection of ventilatory settings in our patients with ARDS reflects a more cautious use by PICU physicians than in the beginning of the past decade. Before the recommendation of American College of Chest Physicians [6], three studies reported a maximum level of PIP utilization ranging from 63±17 to 73±18 cm H2O [18, 19, 20]. We have found that the median peak inspiratory pressure used on these patients with ARDS from days 1–6 was never higher than 37 cm of water. Similarly, the set tidal volume values that we found in those patients with ARDS between days 1 and 6 of MV ranged between 13 and 10 ml/kg, respectively. These tidal volume values are also remarkably lower than previously reported by a study in the beginning of 1990 [20].
The incidence of unplanned endotracheal extubation that we found is very similar to that reported by others, which has ranged from 3 to 13% [26, 27, 28, 29, 30]. Furthermore, the reintubation rate after unplanned endotracheal extubation and later re-institution of MV was 30%. These data suggest that many infants and children are kept intubated longer than necessary. More frequent assessment for the eligibility to wean and a protocol-driven weaning from MV should help to identify those patients who remain unnecessarily on the ventilator. Likewise, a re-intubation rate of 10–20% or so is consistently found among pediatric studies [13, 14, 15, 24, 25, 31] and is confirmed in the present study. Like us, several studies on pediatric and adult populations have reported that patients requiring reintubation have a significantly higher mortality than patients who are successfully extubated [13, 30, 32, 33]; however, in this study the mortality rate of reintubated patients was not different for the entire group who received MV. Again, it is possible for reintubated patients to begin a new course of MV where the probability of mortality is very similar to that of those patients who received MV for more than 12 h [17].
Several investigators have evaluated the survival of mechanically ventilated pediatric patients due to acute hypoxemic respiratory failure and ARDS [18, 19, 20, 34]. These descriptive studies performed in unselected and heterogeneous population have showed that the mortality rate ranges from 50 to 75%. The mortality rate in this study in infants and children with ARDS was approximately 50%. The mortality rates in adult and pediatric patients in the controlled clinical studies on ARDS and acute lung injury ranged from 30 to 40% [35, 36]. The higher mortality in observational studies as compared with clinical trials might be explained, at least in part, by restrictive inclusion criteria for the enrollment of patients with poor prognosis in the controlled clinical trials [17, 37].
There are few published epidemiological data on mortality in infants and children mechanically ventilated. We have found that the overall mortality for the entire population is approximately 15%. Among adults ventilated, the reported overall mortality is higher than 30% [17]. The survival of mechanically ventilated patients not only may be influenced by personnel and physical characteristics of the PICU, but also those factors present at the start of MV such as immunocompromise, multiple congenital anomalies, malignancies disease, and mainly, development of complications in the ventilators and the patients’ management in the ICU [17]. Epidemiological studies focusing on these aspects could cast light on the factors related to survival of infants and children submitted to MV.
Conclusion
In conclusion, our study offers a large amount of information concerning use of setting, modes and outcomes of MV, as well as the primary indication to institute the ventilatory support. We believe that this descriptive study could come closer to the daily practice of MV in pediatric ICU.
Electronic Supplementary Material
Acknowledgement
We are indebted to A. Fernandez, F. Ibelli, G. Fernández, and J.L. Do Pico, for artwork. List of participants: 1. Argentina: P. Neira, G. Sheehan, (Hospital de Niños R Gutiérrez, Buenos Aires); R. Álvarez, G. Debaisis (Hospital de Niños P. Elizalde, Buenos Aires); N. Demo, G. Pujales (Hospital Materno Infantil, San Luis); P. Minces, E. Schniltzer (Hospital Italiano, Buenos Aires); S. Sáenz, M. Centeno (Hospital de Niños de la Santísima Trinidad, Córdoba); M.E. Ratto, C. Peltzer (Hospital de Niños Sor María Ludovica, La Plata, Buenos Aires); D. Allende, O. Maliarchuk (Hospital Provincial Castro Rendón, Neuquén); M. Cabezas, I. Oviedo (Hospital de Niños J.J. Vilela, Rosario, Santa Fe); R. Jabornisky, F. Avellaneda (Hospital de Niños Juan Pablo II, Corrientes); M.G. Rodríguez, G. Domínguez (Clínica Bazterrica, Buenos Aires). Costa Rica: A. Baltodano Agüero (Hospital Nacional de Niños “Dr. Carlos Sáenz H”. Costa Rica). Greece: D. Paraschou (Children’s Hospital Pesenti); M. Tamiolaki (Children’s Hospital Santa Sophia, Athens). Panama: M. Johnson, A. Vargas (Hospital de Niños de Panamá). Spain: P. Azcón (Hospital Universitario Virgen de las Nieves, Granada); J. Balcells (Hospital Vall d’Hebrón, Barcelona); J.C. de Carlos (Hospital Son Dureta, Palma de Mallorca); J. Espinosa (Hospital Materno-Infantil, Badajoz); M. Frías (Hospital Universitario Reina Sofía, Córdoba); E. García-Sánchez (Hospital Universitario Gregorio Marañón, Madrid); A. Hernández (Hospital Puerta del Mar, Cádiz); A. Medina (Hospital Central de Asturias, Oviedo); I. Martos (Hospital Ramón y Cajal, Madrid); P. Oliva (Hospital La Paz, Madrid); J. Pilar (Hospital de Cruces, Baracaldo); M. Pons (Hospital Sant Joan de Deu, Esplugues); F. Rodríguez Amuedo (Hospital Materno-Infantil, Málaga); J.I. Sánchez Díaz (Hospital 12 de Octubre, Madrid); J.M. Sánchez López (Complejo Hospitalario Materno-Infantil, Las Palmas de Gran Canaria); J.P. Santidrián (Hospital Universitario Marqués de Valdecilla, Santander); A. Serrano (Hospital Universitario del Niño Jesús, Madrid); C. Valdovinos (Hospital Miguel Servet, Zaragoza). UK: J. Fraser (Bristol Royal Hospital for Sick Children, Bristol) D. Macrae, A. Petros (Great Ormond Street Hospital for Children, London). USA: G. Reyes (PICU Hope children’s Hospital, Illinois); Nemours.org (PICU Alfred I. duPont Hospital for Children, Wilmington, Del.). This work was supported in part by Karelsie Foundation.
References
- 1.Salyer Resp Care Clin North Am. 1996;2:471. [PubMed] [Google Scholar]
- 2.Martinot Arch Pediatr. 1997;4:730. doi: 10.1016/S0929-693X(97)83410-0. [DOI] [PubMed] [Google Scholar]
- 3.Earle Crit Care Med. 1997;25:1462. doi: 10.1097/00003246-199709000-00011. [DOI] [PubMed] [Google Scholar]
- 4.Lopez-Herce Intensive Care Med. 2000;26:62. doi: 10.1007/s001340050013. [DOI] [PubMed] [Google Scholar]
- 5.Harel Heart Lung. 1998;27:238. doi: 10.1016/s0147-9563(98)90035-8. [DOI] [PubMed] [Google Scholar]
- 6.American Chest. 1993;104:1833. doi: 10.1378/chest.104.6.1833. [DOI] [PubMed] [Google Scholar]
- 7.Task Crit Care Med. 1991;19:275. [Google Scholar]
- 8.Esteban Am J Respir Crit Care Med. 2000;161:1450. doi: 10.1164/ajrccm.161.5.9902018. [DOI] [PubMed] [Google Scholar]
- 9.Pollack Crit Care Med. 1988;16:1110. doi: 10.1097/00003246-198811000-00006. [DOI] [PubMed] [Google Scholar]
- 10.Lebel Arch Dis Child. 1989;64:1431. doi: 10.1136/adc.64.10.1431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Proulx Chest. 1996;109:1033. [Google Scholar]
- 12.Bernard Am J Respir Crit Care Med. 1994;149:818. doi: 10.1164/ajrccm.149.3.7509706. [DOI] [PubMed] [Google Scholar]
- 13.Farias Intensive Care Med. 2001;27:1649. doi: 10.1007/s001340101035. [DOI] [PubMed] [Google Scholar]
- 14.Farias Intensive Care Med. 2002;28:752. doi: 10.1007/s00134-002-1306-6. [DOI] [PubMed] [Google Scholar]
- 15.Thiagarajan Am J Respir Crit Care Med. 1999;160:1562. doi: 10.1164/ajrccm.160.5.9810036. [DOI] [PubMed] [Google Scholar]
- 16.Knaus Am J Respir Crit Care Med. 1994;150:311. doi: 10.1164/ajrccm.150.2.8049808. [DOI] [PubMed] [Google Scholar]
- 17.Esteban J Am Med Assoc. 2002;287:345. doi: 10.1001/jama.287.3.345. [DOI] [Google Scholar]
- 18.Timmons J Pediatr. 1991;119:896. [Google Scholar]
- 19.De Crit Care Med. 1992;20:1223. [Google Scholar]
- 20.Davis J Pediatr. 1993;123:35. [Google Scholar]
- 21.Lodha Indian Pediatr. 2001;38:1154. [PubMed] [Google Scholar]
- 22.Brochard Am J Respir Crit Care Med. 1994;150:896. doi: 10.1164/ajrccm.150.4.7921460. [DOI] [PubMed] [Google Scholar]
- 23.Esteban N Engl J Med. 1995;332:345. [Google Scholar]
- 24.Randolph J Am Med Assoc. 2002;288:2561. doi: 10.1001/jama.288.20.2561. [DOI] [Google Scholar]
- 25.Farias Intensive Care Med. 1998;24:1070. doi: 10.1007/s001340050718. [DOI] [PubMed] [Google Scholar]
- 26.Orlowski Crit Care Med. 1980;8:324. doi: 10.1097/00003246-198006000-00002. [DOI] [PubMed] [Google Scholar]
- 27.Scott PH, Eigen H, Moye LA, Georgitis J, Laughlin J. Predictability and consequences of spontaneous extubation in a pediatric ICU. Crit Care Med. 1985;13:228–232. doi: 10.1097/00003246-198504000-00004. [DOI] [PubMed] [Google Scholar]
- 28.Little Crit Care Med. 1990;18:163. doi: 10.1097/00003246-199002000-00007. [DOI] [PubMed] [Google Scholar]
- 29.Betbese Crit Care Med. 1998;26:1180. doi: 10.1097/00003246-199807000-00016. [DOI] [PubMed] [Google Scholar]
- 30.Epstein Am J Respir Crit Care Med. 2000;161:1912. doi: 10.1164/ajrccm.161.6.9908068. [DOI] [PubMed] [Google Scholar]
- 31.Venkataraman Crit Care Med. 2000;28:2991. doi: 10.1097/00003246-200008000-00051. [DOI] [PubMed] [Google Scholar]
- 32.Epstein Chest. 1997;112:186. doi: 10.1378/chest.112.1.186. [DOI] [PubMed] [Google Scholar]
- 33.Esteban Am J Respir Crit Care Med. 1997;156:459. doi: 10.1164/ajrccm.156.2.9610109. [DOI] [PubMed] [Google Scholar]
- 34.Timmons Chest. 1995;108:789. doi: 10.1378/chest.108.3.789. [DOI] [PubMed] [Google Scholar]
- 35.Arnold Crit Care Med. 1994;22:1530. [PubMed] [Google Scholar]
- 36.The N Engl J Med. 2000;342:1301. [Google Scholar]
- 37.Estenssoro Crit Care Med. 2002;30:2450. doi: 10.1097/00003246-200211000-00008. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


