Abstract
Severe acute respiratory syndrome (SARS) was caused by a previously unrecognized animal coronavirus that exploited opportunities provided by 'wet markets' in southern China to adapt to become a virus readily transmissible between humans. Hospitals and international travel proved to be 'amplifiers' that permitted a local outbreak to achieve global dimensions. In this review we will discuss the substantial scientific progress that has been made towards understanding the virus—SARS coronavirus (SARS-CoV)—and the disease. We will also highlight the progress that has been made towards developing vaccines and therapies The concerted and coordinated response that contained SARS is a triumph for global public health and provides a new paradigm for the detection and control of future emerging infectious disease threats.
Main
The past 150 years saw the emergence of three pandemics from southern China: plague during the late nineteenth century and two influenza pandemics (Asian flu of 1957 and Hong Kong flu of 1968)1,2. In November 2002, a new 'plague' was emerging in Guangdong Province, China. On 21 February 2003 a physician from Guangdong spent a single day in hotel 'M' in Hong Kong, during which time he transmitted an infection to 16 other guests. These, in turn, seeded outbreaks of the disease in Hong Kong, Toronto, Singapore and Vietnam3. Within weeks, SARS had spread to affect more than 8,000 people in 25 countries across 5 continents (Fig. 1; World Health Organization, http://www.who.int/csr/sars/country/table2004_04_21/ en_21/en/print.html). By the end of the global outbreak (5 July 2003), it had killed 774 people—a small number in comparison with the fatalities during the previous pandemics of plague and influenza. But the rapidity of spread by air travel, immediate media coverage and today's globalization of economic activity all contributed to the far more pronounced impact of SARS.
Figure 1. The global spread of SARS.
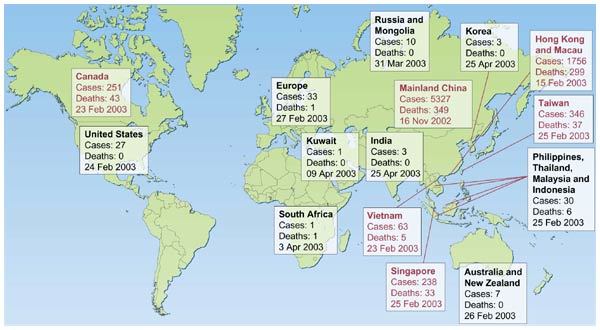
The number of probable cases of SARS and the date of onset of the first case in each country (or group of countries) is denoted. The countries denoted in red are those where substantial local transmission occurred. The data are based on World Health Organization, http://www.who.int/csr/sars/country/ table2004_04_21/en_21/en/print.html and the figure is adapted from ref. 15.
The speed of the scientific response in understanding this new viral disease was unparalleled. The clinical syndrome was described4,5,6, the etiological agent identified7,8,9, diagnostic tests devised9,10 and the genome completely sequenced11,12 within weeks of the virus's emergence from mainland China. Just 1.5 years later, the first phase 1 vaccine trials are underway, and several other vaccine candidates are under evaluation in animal models13. Previous reviews have addressed aspects of the clinical presentation14,15,16, etiology17, virology18,19,20, laboratory diagnosis21, epidemiology (ref. 22 and World Health Organization, http://www.who.int/csr/sars/en/whoconsensus.pdf), infection control, clinical management and public health23,24,25. Here we emphasize aspects of pathogenesis and their correlation to clinical outcome, and discuss the progress that has been made towards antiviral treatment and vaccine development.
The virus, its origins and evolution
SARS probably first emerged in Guangdong around November 2002 (refs. 26,27). Many of the affected individuals in November and December 2002 had contact with the live-game trade27. The disease was described as an “infectious atypical pneumonia” because of its propensity to cause clusters of disease in families and healthcare workers28. The etiological agent of SARS was identified as a new coronavirus not previously endemic in humans7,8,9. The lack of serological evidence of previous infection in healthy humans suggested that SARS-CoV had recently emerged in the human population and that animal-to-human interspecies transmission seemed the most probable explanation for its emergence. Specimens collected from apparently healthy animals (e.g., Himalayan palm civets (Paguma larvata) and raccoon dogs (Nyctereutes procyonoides)) found in live wild-game animal markets in Guangdong yielded a SARS-CoV-like virus with more than 99% nucleotide homology to the human SARS-CoV29. But the wild-animal reservoir in nature still has not been identified conclusively. Many workers who handled animals in these wet markets had antibody to the related animal SARS-CoV-like virus although they had no history of a SARS-like disease29,30. Taken together with the observation that a number of the SARS-affected individuals in November and December 2002 had epidemiological links to the wild-game animal trade27, it is likely that these wet markets in Guangdong (Fig. 2) provided the interface for transmission to humans. The early interspecies transmissions to humans were probably inefficient, causing little human disease or transmission between humans. Eventually, the animal precursor SARS-CoV-like virus probably adapted to more efficient human-to-human transmission, and SARS emerged. As two authors aptly stated, this was “one small step to man, one giant leap to mankind”31.
Figure 2. Wet markets in Guangdong: 'Wet markets' selling live poultry, fish, reptiles and other mammals are commonplace across southeast Asia and southern China to service the cultural demand for freshly killed meat and fish produce.

In some regions (e.g., Guangdong province, China), increasing affluence has led to the proliferation of markets housing a range of live 'wild' animal species, such as civet cats, pictured, linked to the restaurant trade servicing the demand for these exotic foods.
(AP Photo/Xinhua, Liu Dawei)
The new coronavirus associated with SARS (SARS-CoV) is phylogenetically distinct from all previously known human and animal coronaviruses11,12,32,33. There is also evidence that SARS-CoV evolved towards greater 'fitness' in the human host during the course of the outbreak. Compared with animal SARS-like viruses and early human SARS-CoV strains, human viruses isolated later during the outbreak had acquired a 29- (in some, a 415-) nucleotide deletion in open reading frame (ORF)8 (refs. 29,34). The biological significance of these deletions, however, is not clear. Similarly, SARS-CoV in individuals before February 2003 was genetically more diverse than the later isolates26,34,35. The spike protein (the viral surface glycoprotein which mediates viral attachment and entry into the cell; Fig. 3) of early isolates contained higher rates of nonsynonymous mutations, probably reflecting the ongoing adaptation to the new host. The relative genetic homogeneity of SARS-CoV isolates from later in the outbreak34,35,36,37 may reflect a virus better adapted to the new host. The fact that much of the global spread arose from one index case in Hotel M in Hong Kong3,35 may also contribute to this genetic homogeneity.
Figure 3. Schematic diagram of the SARS coronavirus structure (reproduced from ref. 20).
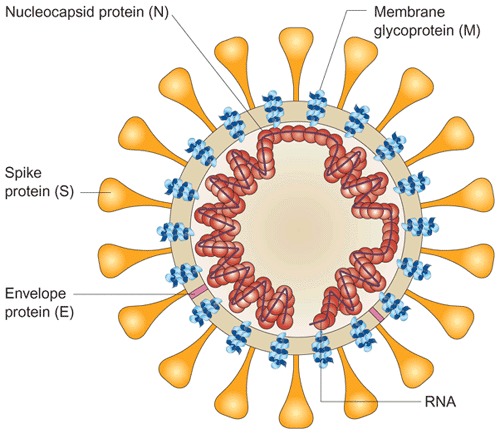
The viral surface proteins (spike, envelope and membrane) are embedded in a lipid bilayer envelope derived from the host cell. Unlike group 2 coronaviruses, SARS-CoV does not possess a hemagglutinin esterase glycoprotein. The single-stranded positive-sense viral RNA is associated with the nucleocapsid protein.
A ban on the sale of wildlife in wet markets in Guangdong imposed during the later period of the SARS outbreak was lifted in September 2003. Between 16 December and 30 January 2004, there were four new cases of SARS, the first nonlaboratory-associated cases diagnosed in humans since the end of the SARS outbreak in July 2003. Epidemiological linkage and phylogenetic data suggest that the associated viruses were new introductions from animals (Y. Guan, unpublished observations)34,38,39. These human cases were relatively mild and did not lead to secondary transmission, reflecting that the animal precursor virus is probably not well adapted to efficient human-to-human transmission. This is probably a recapitulation of events in late 2002 in the run-up to the SARS outbreak in 2003. This time, the findings led to the reintroduction of the ban on wild-game animal markets and there have been no further naturally acquired human cases since.
It is likely that the precursor of SARS-CoV has repeatedly crossed the species barrier but only occasionally has it succeeded in adapting to human–human transmission. This adaptation clearly occurred in late 2002 and it may happen again in the future. But given the present understanding and awareness about SARS, we expect that such re-emergence is unlikely to lead to a global outbreak on the scale of 2003.
Transmission between humans
The major routes of transmission of SARS are droplet infection, aerosolization and fomites (refs. 40,41 and World Health Organization, http://www.who.int/csr/sars/en/whoconsensus.pdf). Deposition of infected droplets or aerosols on the respiratory mucosal epithelium probably initiates viral infection. Whether infection can occur through the oral or conjunctival routes is unknown, but SARS-CoV has been detected in tears42. Although exposure to the animal precursor of SARS-CoV seems to have resulted in asymptomatic infection29,30, once the virus had adapted to human-to-human transmission in the later part of the outbreak, asymptomatic infection seemed to be rare43. Other peculiarities about SARS-CoV transmission were also evident. Transmission was infrequent during the first five days of illness44 and, unlike transmission of influenza, was relatively inefficient in the household setting45. Despite SARS's fearsome reputation and global spread, the average number of secondary infectious cases generated by one case (R0) was low (2.2–3.7); in contrast, the R0 of influenza ranges from 5 to 25 (ref. 22). Although not unique to SARS, 'superspreading events' (in which a few affected individuals disproportionately contribute to transmission) were characteristic of the outbreak22,46. The factors underlying the superspreading phenomenon of SARS are poorly understood but may include coinfection with other viruses and host factors, as well as behavioral and environmental factors.
Clinical symptoms
The clinical symptoms of SARS-CoV infection are those of lower respiratory tract disease4,5,6,7,14. Besides fever, malaise and lymphopenia, affected individuals have slightly decreased platelet counts, prolonged coagulation profiles and mildly elevated serum hepatic enzymes. Chest radiography reveals infiltrates with subpleural consolidation or 'ground glass' changes compatible with viral pneumonitis. But although the main clinical symptoms are those of severe respiratory illness, SARS-CoV actually causes infection of other organs: some affected individuals have watery diarrhea, and virus can be cultured from the feces and urine, as well as the respiratory tract47,48,49. In addition, RT–PCR has identified the virus in the serum, plasma and peripheral blood leucocytes50,51. Individuals with SARS also have a pronounced peripheral T-cell lymphocytopenia: numbers of CD4+ and CD8+ cells are both reduced, and more than one-third of individuals have a CD4+ T-cell count of less than 200 cells/mm3 (refs. 52,53), suggesting increased susceptibility to secondary infections. The mechanisms underlying the T-cell lymphopenia remain to be elucidated.
Around 20–30% of individuals with SARS require management in intensive care units14 and the overall fatality rate is ∼15% (World Health Organization, http://www.who.int/csr/sars/en/whoconsensus.pdf). The age dependence of disease severity and mortality is notable; during the outbreak, mortality rates of affected individuals in Hong Kong who were 0–24, 25–44, 45–64 and >65-year old were 0%, 6%, 15% and 52%, respectively (World Health Organization, http://www.who.int/csr/sars/en/WHOconsensus.pdf). None of the 1–12-year-olds infected with SARS-CoV in Hong Kong had disease severe enough to require intensive care or mechanical ventilation54,55. This progressive age dependence in mortality is not totally explained by comorbid factors and the underlying biological basis remains unclear.
Virus tropism and pathogenesis
Quantitative studies of viral load have provided insights into the pathogenesis of SARS. Viral load is higher in the lower respiratory tract than in the upper airways56,57. Viral load in the upper respiratory tract47 and feces57 is low during the first 4 days and peaks at around day 10 of illness. In marked contrast, viral load in influenza peaks soon after onset of clinical symptoms58. This unusual feature of SARS-CoV infection explains its low transmissibility early in the illness. It also explains the poor diagnostic sensitivity of the first-generation RT–PCR diagnostic tests on upper respiratory tract and fecal specimens collected early in the illness (reviewed in ref. 21).
Affected individuals with high serum viral loads have a poor prognosis59. Between days 10–15 of illness, high viral load in nasopharyngeal aspirates, feces and serum, as well as detection of virus in multiple anatomic sites, are independently predictive of adverse clinical outcome60. Serial studies of viral load throughout illness also reflect clinical outcome61. Taken together, these findings suggest that poor clinical outcome is associated with continued uncontrolled viral replication. SARS-CoV RNA can be invariably detected in the lungs of individuals dying of SARS, but viral load is higher in those dying earlier in the course of the illness (<21 days)62.
The respiratory tracts of affected individuals who die during the first ten days of illness show diffuse alveolar damage with a mixed alveolar infiltrate, lung edema and hyaline membrane formation. Macrophages are a prominent component of the cellular exudates in the alveoli and lung interstitium63,64. Multinucleate syncytia of macrophage or epithelial cell origin are sometimes seen later in the disease. Immunohistochemistry, in situ hybridization and electron microscopy on autopsy or tissue biopsy have unequivocally demonstrated SARS-CoV replication in pneumocytes in the lung and enterocytes in the intestine65,66,67,68. Individual reports of virus detection by in situ hybridization or immunohistochemistry in other tissues69 await confirmation by electron microscopy70.
In the large and small intestines, the virus replicates in enterocytes71. Viral particles primarily are seen on the apical surface of enterocytes and rarely in the glandular epithelial cells. But there is no villous atrophy or cellular infiltrate in the intestinal epithelium and the pathogenic mechanisms responsible for watery diarrhea in individuals with SARS is unclear. Some human intestinal epithelial cell lines support productive replication of SARS-CoV72 and gene expression arrays have shown that virus replication is associated with the expression of an antiapoptotic host cellular response, perhaps explaining the lack of enterocyte destruction in vivo73.
The virus receptor and entry into cells
Studies using pseudotyped lentiviruses, carrying the spike, membrane and envelope surface glycoproteins of SARS-CoV (Fig. 3) separately and in combination demonstrated that the spike protein is both necessary and sufficient for virus attachment on susceptible cells74,75,76,77. The SARS-CoV spike protein uses a mechanism similar to that of class 1 fusion proteins in mediating membrane fusion78,79. There is no consensus as to whether the virus entry occurs through a pH-dependent receptor-mediated endocytosis or through direct membrane fusion at the cell surface74,77,80. The receptor for SARS-CoV was identified as the metallopeptidase ACE-2 (refs. 81,82). The soluble ACE-2 ectodomain blocks SARS-CoV infection76, and amino acids 270–510 of the spike protein are required for interaction with ACE-2 (ref. 83). Other coronaviruses use different cell receptors and enter cells either by means of fusion at the plasma membrane or through receptor-mediated endocytosis84.
Immunostaining techniques have identified ACE-2 on the surface of type 1 and 2 pneumocytes, the enterocytes of all parts of the small intestine and the proximal tubular cells of the kidney. This localization explains the documented tissue tropism of SARS-CoV for the lung and gastrointestinal tract and its isolation from the urine. But it is notable that colonic enterocytes lack ACE-2 protein expression although SARS-CoV replication does occur in colonic epithelium71,85. In contrast, whereas ACE-2 is strongly expressed on the endothelial cells of small and large arteries and veins of all tissues studied and the smooth muscle cells of the intestinal tract, there is no evidence of virus infection at any of these sites. This lack of virus infection in tissues that express the putative receptor prompts the question of whether a coreceptor is required for successful virus infection70. Vasculitis is known to occur in individuals with SARS but its relation to infection of endothelial cells is unknown. Because only the basal layer of the nonkeratinized squamous epithelium of the upper respiratory tract expresses ACE-2 (ref. 85), undamaged epithelium of the nasopharynx is unlikely to support SARS-CoV replication. Other receptors for virus entry that are independent of ACE-2 expression may exist.
Pseudotyped virus containing the spike protein has also been shown to bind to dendritic cell-specific intercellular adhesion molecule 3-grabbing nonintegrin (DC-SIGN)74. DC-SIGN is a type-II transmembrane adhesion molecule found on dendritic cells consisting of a C-type lectin domain that recognizes carbohydrate residues on a variety of pathogens. Unlike the ACE-2 receptor on pneumocytes and enterocytes, DC-SIGN does not permit SARS-CoV infection of the dendritic cells. Instead, binding of SARS-CoV to DC-SIGN allows dendritic cells to transfer infectious SARS-CoV to susceptible target cells74. A similar mechanism has been described for dengue virus, human immune deficiency virus (HIV) and cytomegalovirus, and may be relevant in SARS pathogenesis.
Many details of SARS-CoV pathogenesis remain to be elucidated, but the development of a full-length infectious cDNA clone of SARS-CoV should permit precise manipulation of the virus genome and will help our understanding of the viral determinants of pathogenesis86.
The host response
Several inflammatory cytokines (IL-1β, IL-6 and IL-12) and chemokines chemotactic for monocytes (MCP-1) and neutrophils (IP-10) are elevated in adults and children with SARS87,88,89,90. The increased levels of monocyte-tropic chemokines may contribute to the prominently monocytic macrophagic infiltrate observed in the lung63. But increases of these same chemokines occur in other viral diseases (e.g., influenza)91 and are not a unique feature of SARS. In addition, ELISPOT assays of peripheral blood leukocytes have revealed prolonged immunological dysregulation in individuals with SARS92. It is difficult to evaluate the overall pathogenic significance of these findings because immunological markers in the peripheral blood do not always reflect the local microenvironment of the lung93.
Genetic factors associated with susceptibility to, or severity of, SARS are under investigation. HLA-B*4601 has been associated with severe SARS disease in Taiwan94 but not Hong Kong95. HLA-B*0703 has also been associated with disease susceptibility and HLA-DRB1*0301 with resistance to SARS. The coinheritance of B*0703 and B60 was significantly higher in individuals with SARS than in the general population95. The mechanisms underlying these disease associations remain to be elucidated.
Animal models
Key to the development of effective antiviral drugs and vaccines against SARS-CoV was the development of animal models of SARS (Table 1). SARS-CoV seems to cause infection in cynomolgous macaques following intratracheal inoculation96,97,98. But whereas some researchers find evidence of disease pathology reminiscent of that seen in individuals dying of SARS and can show SARS-CoV antigen and viral particles in the pneumocytes of infected macaques96,97, others only find evidence of a mild upper-airway disease and low levels of virus by RT-PCR98. These differences in outcome may reflect differences in the viral strain, pre-exposure history and age of the animals, route of inoculation, stage of infection at which necropsy was performed or other factors.
Table 1.
Animal models for SARS coronavirus infection and disease
| Animal model | Virus strain, dose and route of challenge | Viral replication detected in | Disease and pathology | Transmission from animal to animal | Reference (ref no) |
|---|---|---|---|---|---|
| Cynomolgus macaque | HKU-39849 strain. 106 TCID50 intratracheal,intranasal and conjunctival. | Sputum day 2–6, nasal swab day 2–6, pharyngeal swab day 2–6. | Lethargy, respiratory distress, skin rash. | Not done | Fouchier, R.A.M. et al. 2003 (96) |
| Day 4–6: multifocal lung pathology similar to human disease. | Kuiken, T. et al. 2003 (97) | ||||
| Necropsy at day 6: Lung 104–5 TCID50/g tissue. | Viral antigen in pneumocytes. | ||||
| Cynomolgus macaque (Rhesus macaques were also studied but results were less notable and are not summarized here) | Tor2 strain. 107 PFU intratracheal or intravenous. | Oral and nasal swabs: culture negative, but RT-PCR evidence of low levels of virus. | Mild cough and decreased activity on days 2–3 after challenge. Sneezing at day 8–10. Necropsy at day 12–14: Lung: small foci of consolidation in 1 of 2 macaques with intratracheal inoculation. Virus antigen negative. Viral RNA detected in lung (2 of 4 lungs), mediastinal lymph node (3 of 4 nodes). Viral RNA also detected in both in intravenously injected animals. | Not done | Rowe, T. et al. 2004 (98) |
| Ferret | HKU-39849 strain. 106 TCID50 intratracheal. | Nasopharynx: day 2–14; peak viral titers 105 TCID50/ml. Lungs: peak viral titers 106 TCID50/ml. | Lethargy (3 of 6), Death (1 of 6)Multifocal lung pathology. Milder than in macaques. | Yes | Martina, B.E.E. et al. 2003 (99) |
| Domestic cat | HKU-39849 strain. 106 TCID50 intratracheal. | Nasopharynx: day 2–10; Peak viral titers 105/ml. Lungs: peak viral titers 106 TCID50/ml. | No clinical disease. | Yes | Martina, B.E.E. et al. 2003 (99) |
| Mice (BALB/c) (4–6 weeks old) | Urbani strain. 103–105 TCID50 intranasal inoculation. | Lungs and nasal turbinates peak viral titers at days 1–3 after infection. Minimal pathology ordisease. Virus is cleared by day 7. Titers in lung > titers in nasal turbinates. | Viral antigen in bronchiolar epithelial cells. | Not done | Subbarao, K. et al, 2004 (101) |
| African green monkey (adult) | Urbani strain. 106.3 TCID50 intranasal and intratracheal combined inoculation. | Nasal swab: for 5–7 days, peak titers 101.5–3 TCID50 Tracheal lavage: 3 days, peak titres 102.5–3 TCID50. | No clinical disease. | Not done | Bukreyev, A. et al. 2004 (100) |
| Golden Syrian hamster | Urbani strain. 103 TCID50 intranasal inoculation. | Nasal turbinates: peak titers 106.2 TCID50. | Lung pathology but no clinical disease. | Not done | Buchholz, U.J. et al. 2004 (102) |
| Lungs: peak titers 105.6 TCID50. | Roberts, A. et al. (in the press) (103) |
Other animal models include ferrets, cats, Golden Syrian hamsters, mice and African green monkeys (Table 1)99,100,101,102,103. These animal models support viral replication in the upper and lower respiratory tracts96,97,98,99,100,101,102,103. Ferrets and hamsters also develop notable lung pathology. Infected cats and ferrets transmit SARS-CoV to noninfected animals held in the same cage99. Natural asymptomatic infection in cats was documented during the community outbreak at Amoy Gardens, Hong Kong (World Health Organization, http://www.who.int/csr/sars/en/whoconsensus.pdf).
These animal models of SARS differ from natural human disease in that the period between infection and peak disease pathology or peak viral load is shorter than is found in human disease and because the disease pathology, when present, is self-limited and rarely progresses to a fatal outcome as occurs with SARS. They also do not accurately reproduce the intestinal component of the human disease. But these models provide the only options presently available for addressing questions relevant to therapeutics and vaccine development. They can provide useful information providing their limitations are recognized.
Antiviral therapy
Several potential antiviral agents have been evaluated in vitro, and a few have been tested in animal models. Screening of currently available antiviral drugs and chemical libraries reveals that interferons, glycyrrhizin, baicalin, reserpine, niclosamide, luteolin, tetra-O-galloyl-β-D-glucose and the protease inhibitors have in vitro activity against SARS-CoV104,105,106,107,108. Differences in in vitro susceptibility of SARS-CoV to interferon (IFN)-β1b, IFN-α2 and ribavirin106,109,110,111 probably relate to differences in the testing methods used. Overall, IFN-αn1/n3, leukocytic IFN-α, IFN-β and the HIV protease inhibitors (especially nelfinavir) are consistently active in vitro and should be considered for animal studies and randomized placebo-controlled clinical trials. Type 1 interferons render uninfected cells refractory to SARS-CoV replication through a MxA-independent mechanism112, whereas the HIV protease inhibitors may block the activity of the main SARS-CoV proteinase113. So far, only interferons have been tested in animal models: in cynomolgous macaques, pegylated IFN-αn2 provided prophylaxis but was only marginally effective for early treatment114. No randomized placebo-controlled trials have been performed for any of these antiviral drugs, although treatment studies using historical controls have suggested clinical benefit from IFN-α (infacon-1)115 and the combination of a protease inhibitor with ribavirin61.
The rapidity with which the SARS-CoV genome was sequenced, the determination of the structure of potential drug targets116 and the prediction of functional properties of SARS-CoV proteins based on prior knowledge of homologs from other coronaviruses117 have allowed identification of potential new drug targets. Peptides derived from the heptad-repeat-2 region of the spike protein have been shown to block virus infection, albeit at much higher molar concentrations than similar inhibitors needed to prevent HIV entry78,79. Short interfering RNAs also seems to be effective in decreasing viral replication in cell lines118,119,120, but this remains an experimental strategy rather than one immediately amenable to clinical application. Screening of combinatorial chemical libraries has identified inhibitors of SARS protease, helicase and spike-protein–mediated cell entry121.
For successful treatment of influenza, antiviral drugs must be administered within 48 hours of disease onset to obtain substantial clinical effect. But because the SARS-CoV load increases until day 10 of illness47, and in light of the correlation of high viral load in the second week of illness with adverse outcome60, the window of opportunity for antiviral therapy may be wider.
Active and passive immunization
Much scientific effort has been focused on developing a vaccine to protect against future outbreaks of SARS-CoV. The commercial viability of developing a vaccine for SARS-CoV will ultimately depend on whether the virus re-emerges in the near future. As discussed above, it is unlikely that future outbreaks will reach global proportions, but nevertheless, vaccines or passive immunization would be relevant in the context of protecting high-risk individuals such as laboratory and health-care workers. A vaccine could also be considered in the setting of the farmed-game-animal trade, if farming of civets for human consumption continues. In the short time since the virus was identified, substantial progress has been made toward developing a vaccine.
Immunodominant B- and T-cell epitopes of SARS-CoV are being defined122,123,124. Natural human infection with SARS-CoV leads to a long-lived neutralizing antibody response and immune sera crossneutralize diverse human SARS-CoV125, suggesting that active immunization against SARS may be a feasible proposition. But so far there has been no known instance of human re-exposure to SARS-CoV to confirm that the naturally acquired immune response confers protection from reinfection. When SARS-CoV spike, envelope, membrane and nucleocapsid proteins were individually expressed in an attenuated parainfluenza type 3 vector, only the recombinants expressing the spike protein induced neutralizing antibody and protected from challenge in hamsters102 (Table 2). Mucosal immunization of African green monkeys with this parainfluenza–spike protein chimeric virus led to neutralizing antibody and protection from viral replication in the upper and lower respiratory tracts after challenge with live SARS-CoV100, and spike protein–encoding DNA vaccines stimulated neutralizing antibody production and protection from live-virus challenge in mice126. These studies confirm the assumption that the spike protein is the dominant protective antigen for SARS. Experiments using adoptive transfer and T-cell depletion showed that humoral immunity alone can confer protection126. Other vaccine strategies have included the use of naked DNA127,128,129, adenoviral vectors130 or modified vaccinia (Ankara)131 and inactivated whole virus132,133. Many investigators have optimized the codon usage of the gene target to improve expression. In summary, all vaccines based on the spike protein seem to induce neutralizing antibody responses, and those carrying nucleoprotein can induce nucleoprotein-specific cell-mediated immunity. But thus far only four studies have used live SARS-CoV to challenge immunized animals (Table 2). An inactivated vaccine with alum adjuvant, which induces neutralizing antibody in mice, is entering phase 1 human clinical trials in China13.
Table 2.
Studies on active immunization in SARS
| Type of vaccine | Gene target or antigen | Animal and dosage | Immune response | Protection from SARS CoV challenge? | Reference (ref. no.) |
|---|---|---|---|---|---|
| Inactivated vaccine. Infected Vero E6 cells treated with β-propiolactone and chromatography. | Whole virus | BALB/c mice. 0.1–0.3 μ g intramuscularly 2 doses 3 weeks apart. | Neutralizing antibodies. | Not done | Tang, L. et al. DNA Cell Biol. 2004 (133) |
| UV inactivated whole virion. | Whole virus | BALB/c mice, 10ug with or without alum adjuvant; subcutaneous. 2 doses 7 weeks apart. | Neutralizing antibody and T-cell responses. | Not done | Takasuka, N. et al. Int. Immunol. 2004 (132) |
| Recombinant antigen. Specifically constructed S2 fragments (Leu803 to Ala828 in F3 and Pro1061 to Ser1093 in F9). | Spike protein | Rabbits 0.5–1 mg, BALB/c mice 50–100 μg. Boosted every 2 weeks. | Neutralizing antibodies (in vitro pseudotyped virus assay). | Not done | Zhang, H. et al. J. Virol. 2004 (123) |
| DNA vaccine. Codon-optimized spike protein. | Spike protein | BALB/c mice. 25 μg intramuscularly 3 doses 3 weeks apart. | Specific neutralizing antibodies; T-cell responses (CD4+ and CD8+ T cells against spike protein). | >106 decrease in lung titer; ~102 decrease in nasal titer. Protection mediated by humoral rather than T-cell mechanisms. | Yang, Z.Y. et al. Nature 2004 (126) |
| DNA vaccine. N-linked calreticulin. | Nucleocapsid protein | C57BL/6 mice. DNA-coated gold beads 3 doses 1 week apart. | Antibody and CD8+ T-cell responses against nucleocapsid protein. Protects mice against vaccinia expressing SARS-CoV nucleocapsid. | Not done | Kim, T.W. et al. J. Virol. 2004 (127) |
| DNA vaccine with spike protein. | Spike protein | Balb/c mice. 150 μg intramuscularly 3 doses 3 weeks apart. | Neutralizing antibodies TH1 switch. | Not done | Zeng, F. et al. Biochem. Biophys Res. Commun. 2004 (128) |
| DNA vaccine with nucleocapsid protein. | Nucleocapsid protein | C3H/He mice. 0.05 μg intramuscularly 3 doses 2 weeks apart. | Cytotoxic T cells Nucleocapsid-specific antibodies. | Not done | Zhu, M.S. et al. Immunol. Lett. 2004 (129) |
| Adenoviral vector. Codon-optimized spike, membrane and nucleocapsid proteins. | Separate constructs for spike, membrane, and nucleocapsid proteins | Rhesus macaques. 1011 PFU of all three vectors (spike, membrane and nucleocapsid proteins) intramuscularly 2 doses 4 weeks apart. | Neutralizing antibody response. T-cell responses against nucleocapsid. | Not done | Gao, W. et al. Lancet 2003 (130) |
| Modified vaccinia Ankara (MVA) with spike protein. | Spike protein | BALB/c mice. 107 PFU intramuscularly or intranasally 2 doses 4 weeks apart. | Neutralizing antibodies. | Decreased lung/nasal titer; passive transfer of immune serum confers protection. | Bisht, H. et al. Proc. Natl. Acad Sci. USA 2004 (131) |
| Recombinant attenuated parainfluenza type 3 vector with spike protein. | Spike protein | African green monkeys. One dose of 106 TCID50 intranasally and intratracheally. | Neutralizing antibodies. | Prevents viral shedding. | Bukreyev, A. et al. Lancet 2004 (100) |
| Recombinant parinfluenza virus type 3 vector. | Spike, membrane, envelope and nucleocapsid proteins | Golden Syrian hamsters. 106 TCID50 Single dose, intranasally. | Only spike protein construct induced neutralizing antibody. | Only spike protein vaccine construct induces protection against challenge. | Buchholtz, U.J. et al. Proc. Natl. Acad. Sci. USA 2004 (102) |
Experience with coronavirus vaccines for animals is relevant for SARS vaccine development134. One problem facing animal coronavirus vaccines has been strain variation among field isolates, leading to variable vaccine efficacy. A further concern is the experience with feline infectious peritonitis coronavirus, in which prior immunization led to enhanced disease rather than protection135. In the case of SARS-CoV, neither vaccination nor passive transfer of antibody has yet been reported to lead to disease enhancement, but challenge with live SARS-CoV has occurred soon after immunization. Whether waning immunity or low titers of antibody lead to SARS disease enhancement remains unclear; the recent suggestion that immunized ferrets became more ill after challenge clearly needs to be confirmed or refuted13.
Passive transfer of immune serum protects naive mice from SARS-CoV infection101, and hyperimmune globulin with sufficient neutralizing activity for use in humans could be prepared from pooled convalescent human plasma or from horses immunized with inactivated vaccine. Alternatively, monoclonal antibodies with sufficient neutralizing antibody activity have been developed by screening phage-display antibody libraries and by immortalizing B-cell repertoires of convalescent SARS individuals with Epstein-Barr virus (Table 3)136,137,138. One of these (80R) blocks the virus–ACE-2 receptor interaction through binding to the spike protein S1 domain136. Passive immunization of ferrets and mice was effective in suppressing viral replication in lungs, but less so in the nasopharynx137,138. No randomized placebo control trial evaluated antibody therapy for pre- or post-exposure prophylaxis in at-risk groups during the SARS outbreak. Retrospective analysis of outcome in a limited human study using human SARS convalescent plasma suggested that passive immunization had no obvious adverse effects139.
Table 3.
Studies on passive immunization in SARS with neutralizing monoclonal antibodies
| Type of antibody and method of generation | Antibody target | Animal, route and dose of passive immunization and virus challenge | Response | Reference (ref. no.) |
|---|---|---|---|---|
| Human monoclonal antibody (IgG1). Epstein-Barr virus immortalization of memory B cell repertoire from SARS patients augmented by CpG2006. | Spike protein | BALB/c mice. Monoclonal antibody administered intraperitoneally 2 days prior to intranasal challenge with 104 TCID50 SARS-CoV. | 6-log10 decrease in viral titer in lungs (less decrease in nasal swabs). | Traggiai, E. Nat. Med. 2004 (138) |
| Human monoclonal antibody (IgG1). Screen naive antibody phage display library IgG1. | Spike protein | Ferret. 10 mg/kg of monoclonal antibody administered 24 hours before intratracheal challenge with 104 TCID50 of virus. | 3.3-log10 decrease in viral titer in lung; decreased shedding from throat Protects from lung pathology. | ter Meulen, J. Lancet 2004 (137) |
The antigenic diversity of SARS-CoV-like precursor viruses in the wild-animal reservoir is undefined. In the event of a new interspecies transmission event prompting another SARS outbreak, the crossprotection afforded by current vaccine constructs based on the human SARS-CoV of 2003 is unknown and is likely to influence the efficacy of both passive and active immunization strategies.
Lessons learned
SARS provided a painful reminder of the global impact of emerging infectious diseases. It illustrated how microbes, with their evolutionary drive to preserve and propagate their genes, exploit new opportunities and niches created by modern society140. Interspecies transmission of viruses to humans clearly has occurred throughout human history. But recent developments allowed SARS-CoV increased opportunity to adapt to human-to-human transmission and, subsequently, to spread globally. In particular, large centralized wet markets and hospitals proved to be venues for amplification of transmission to humans, and the burgeoning increase of international travel (currently ∼700 million travelers annually) exploded the local outbreak of an emerging infection into a potential pandemic.
Because most recent emerging infectious disease threats have a zoonotic origin, we need to better understand the microbial ecology of livestock and wildlife. In the context of increased attention and research funding directed at preparedness to combat bioterrorism threats, it is relevant to note that nature remains the greatest 'bioterrorist.' Although microbes that cause commercially important diseases in livestock are well studied, organisms that pose threats to human health are not necessarily ones known to cause disease in livestock, or for that matter, in wildlife. Nipah virus, Hendra virus and SARS-CoV all have a wildlife reservoir. Furthermore, at present there is concern over the possible role played by wild birds and ducks in the maintenance and spread of avian influenza A (H5N1) in Asia141. Greater understanding of the viral ecology of apparently healthy domestic animals and wildlife is therefore important. For example, the attention on ecological studies arising from the Nipah virus and SARS outbreaks have already led to the identification of a number of new viruses, including Tioman, Menangle, Australian bat lyssavirus and a novel group 1 coronavirus142,143. Some of these are now known to be associated with human or livestock disease. But prioritizing such research efforts and assessing the public health relevance—if any—of such findings, poses challenges.
Three incidents of laboratory-acquired SARS have arisen from biohazard level 3 and 4 laboratories, with community transmission arising from one (World Health Organization, http://www.wpro.who.int/sars/docs/update/update_07022004.asp). These incidents were associated with lapses in biohazard level 3 and 4 practices. SARS-CoV can be safely handled in biohazard level 3 laboratories provided that biohazard level 3 practices are rigorously complied with. But as hospital health-care workers learned to their cost, SARS-CoV is an unforgiving virus; one lapse may be one too many, and it is irrelevant whether the lapse occurs in a biohazard level 3 or 4 laboratory.
Despite the impressive speed of scientific understanding of the disease, the global success in containing SARS owed much to traditional public health methods of clinical case identification, contact investigation, infection control at healthcare facilities, patient isolation and community containment (that is, quarantine)25. But the application of such measures in modern society during the control of SARS highlighted several ethical and medical dilemmas, many of which arose from the need to balance individual freedoms against the common good144,145.
SARS signaled a paradigm shift in international public health. It highlighted the need for rapid information exchange regarding unusual infectious disease outbreaks and the possibility of146, and the need for147, a coordinated global response to emerging infectious disease threats. During the early stages of the outbreak, the WHO acted independently, issuing travel alerts and geographically specific travel advisories, without the express consent of the countries affected. The need for such measures was acknowledged post hoc by member states at the World Health Assembly meeting in May 2003 where the WHO was formally empowered to take such actions, as necessary, in the future. Although future emerging pandemics (e.g., influenza because of its transmissibility during early illness) may not be quelled through similar measures, the success of containing SARS remains a triumph for global public health.
Acknowledgements
We thank K.V. Holmes, L.L.M. Poon and J.M. Nicholls for critical comment on the manuscript; A. Frazier for scientific editing; and F. Wong for secretarial assistance. We acknowledge research funding from the United States National Institutes of Health (grant AI95357), the Wellcome Trust (grant 067072/D/02/Z) and the Research Fund for the Control of Infectious Diseases from the Government of Hong Kong Special Administrative Region.
Competing interests
The authors declare no competing financial interests.
References
- 1.Smith MD. Manson's Tropical Diseases. 2003. Plague; pp. 1125–1131. [Google Scholar]
- 2.Shortridge KF, Stuart-Harris CH. An influenza epicentre? Lancet. 1982;2:812–813. doi: 10.1016/S0140-6736(82)92693-9. [DOI] [PubMed] [Google Scholar]
- 3.Centers for Disease Control and Prevention. Update: outbreak of severe acute respiratory syndrome—worldwide, 2003. Morbid. Mortal. Wkly. Rpt.52, 241–248 (2003). [PubMed]
- 4.Tsang KW, et al. A cluster of cases of severe acute respiratory syndrome in Hong Kong. N. Engl. J. Med. 2003;348:1977–1985. doi: 10.1056/NEJMoa030666. [DOI] [PubMed] [Google Scholar]
- 5.Lee N, et al. A major outbreak of severe acute respiratory syndrome in Hong Kong. N. Engl. J. Med. 2003;348:1986–1994. doi: 10.1056/NEJMoa030685. [DOI] [PubMed] [Google Scholar]
- 6.Poutanen SM, et al. Identification of severe acute respiratory syndrome in Canada. N. Engl. J. Med. 2003;348:1995–2005. doi: 10.1056/NEJMoa030634. [DOI] [PubMed] [Google Scholar]
- 7.Peiris JS, et al. Coronavirus as a possible cause of severe acute respiratory syndrome. Lancet. 2003;361:1319–1325. doi: 10.1016/S0140-6736(03)13077-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ksiazek TG, et al. A novel coronavirus associated with severe acute respiratory syndrome. N. Engl. J. Med. 2003;348:1953–1966. doi: 10.1056/NEJMoa030781. [DOI] [PubMed] [Google Scholar]
- 9.Drosten C, et al. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N. Engl. J. Med. 2003;348:1967–1976. doi: 10.1056/NEJMoa030747. [DOI] [PubMed] [Google Scholar]
- 10.Poon LL, et al. Rapid diagnosis of a coronavirus associated with severe acute respiratory syndrome (SARS) Clin. Chem. 2003;49:953–955. doi: 10.1373/49.6.953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Marra MA, et al. The genome sequence of the SARS associated coronavirus. Science. 2003;300:1399–1404. doi: 10.1126/science.1085953. [DOI] [PubMed] [Google Scholar]
- 12.Rota PA, et al. Characterization of a novel coronavirus associated with severe acute respiratory syndrome. Science. 2003;300:1394–1399. doi: 10.1126/science.1085952. [DOI] [PubMed] [Google Scholar]
- 13.Enserink M. One year after outbreak. SARS virus yields some secrets. Science. 2004;304:1097. doi: 10.1126/science.304.5674.1097. [DOI] [PubMed] [Google Scholar]
- 14.Peiris JSM, Yuen KY, Osterhaus ADME, Stohr K. The severe acute respiratory syndrome. N. Engl. J. Med. 2003;349:2431–2441. doi: 10.1056/NEJMra032498. [DOI] [PubMed] [Google Scholar]
- 15.Christian MD, Poutanen SM, Loufty MR, Muller MP, Low DE. Severe acute respiratory syndrome. Clin. Infect. Dis. 2004;38:1420–1427. doi: 10.1086/420743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rainer TH. Severe acute respiratory syndrome: clinical features, diagnosis and management. Curr. Opin. Pulm. Med. 2004;10:159–165. doi: 10.1097/00063198-200405000-00003. [DOI] [PubMed] [Google Scholar]
- 17.Drosten C, Preiser W, Gunther S, Schmitz H, Doerr HW. Severe acute respiratory syndrome: identification of the aetiologic agent. Trends Mol. Med. 2003;9:325–327. doi: 10.1016/S1471-4914(03)00133-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Holmes KV. SARS coronavirus: a new challenge for prevention and therapy. J. Clin. Invest. 2003;111:1605–1609. doi: 10.1172/JCI18819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Davidson A, Siddell S. Potential for antiviral treatment of severe acute respiratory syndrome. Curr. Opin. Infect. Dis. 2003;26:565–571. doi: 10.1097/00001432-200312000-00009. [DOI] [PubMed] [Google Scholar]
- 20.Stadler K, et al. SARS—beginning to understand a new virus. Nat. Rev. Microbiol. 2003;1:209–218. doi: 10.1038/nrmicro775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Poon LLM, Guan Y, Nicholls NJ, Yuen KY, Peiris JSM. The aetiology, origins and diagnosis of SARS. Lancet Infect. Dis. 2004;4:663–671. doi: 10.1016/S1473-3099(04)01172-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Anderson RM, et al. Epidemiology, transmission dynamics, and control of SARS. The 2002–2003 epidemic. Phil. Trans. R. Soc. Lond. B. 2004;359:1091–1105. doi: 10.1098/rstb.2004.1490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Poutanen SM, Low DE. Severe acute respiratory syndrome: an update. Curr. Opin. Infect. Dis. 2004;17:287–294. doi: 10.1097/01.qco.0000136924.45049.7e. [DOI] [PubMed] [Google Scholar]
- 24.Poutanen SM, McGeer AJ. Transmission and control of severe acute respiratory syndrome. Curr. Infect. Dis. Rep. 2004;6:220–227. doi: 10.1007/s11908-004-0012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Weinstein RA. Planning for epidemics—the lessons of SARS. N. Engl. J. Med. 2004;350:2332–2334. doi: 10.1056/NEJMp048082. [DOI] [PubMed] [Google Scholar]
- 26.Zhong NS, et al. Epidemiology and cause of severe acute respiratory syndrome (SARS) in Guangdong, People's Republic of China, in February 2003. Lancet. 2003;362:1355–1358. doi: 10.1016/S0140-6736(03)14630-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Xu RH, et al. Epidemiologic clues to SARS origin in China. Emerg. Infect. Dis. 2004;10:1030–1037. doi: 10.3201/eid1006.030852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhong NS, Zeng GQ. Our strategies for fighting severe acute respiratory syndrome (SARS) Am. J. Respir. Crit. Care Med. 2003;168:7–9. doi: 10.1164/rccm.200305-707OE. [DOI] [PubMed] [Google Scholar]
- 29.Guan Y, et al. Isolation and characterization of viruses related to SARS coronavirus from animals in southern China. Science. 2003;302:276–278. doi: 10.1126/science.1087139. [DOI] [PubMed] [Google Scholar]
- 30.Centers for Disease Control and Prevention. Prevalence of IgG antibody to SARS-associated coronavirus in animal traders—Guangdong Province, China, 2003. Morb. Mortal. Wkly. Rep.52, 986–987 (2003). [PubMed]
- 31.Klempner MS, Shapiro DS. Crossing the species barrier—one small step to man, one giant leap to mankind. N. Engl. J. Med. 2004;350:1171–1172. doi: 10.1056/NEJMp048039. [DOI] [PubMed] [Google Scholar]
- 32.Snijder EJ, et al. Unique and conserved features of genomes and proteome of SARS-coronavirus, an early split-off from the coronavirus group 2 lineage. J. Mol. Biol. 2003;33:991–1004. doi: 10.1016/S0022-2836(03)00865-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gorbalenya AE, Snijder AE, Spaan WJ. Severe acute respiratory syndrome coronavirus phylogeny: toward consensus. J. Virol. 2004;78:7863–7866. doi: 10.1128/JVI.78.15.7863-7866.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.The Chinese SARS Molecular Epidemiology Consortium. Molecular evolution of the SARS coronavirus during the course of the SARS epidemic in China. Science303, 1666–1669 (2004). [DOI] [PubMed]
- 35.Guan Y, et al. Molecular epidemiology of SARS coronavirus in Hong Kong. Lancet. 2004;363:99–104. doi: 10.1016/S0140-6736(03)15259-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ruan Y, et al. Comparative full-length genome sequence analysis of 14 SARS coronavirus isolates and common mutations associated with putative origins of infection. Lancet. 2003;361:1779–1785. doi: 10.1016/S0140-6736(03)13414-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yeh SH, et al. Characterization of severe acute respiratory syndrome coronavirus genomes in Taiwan: molecular epidemiology and genome evolution. Proc. Natl. Acad. Sci. USA. 2004;101:2542–2547. doi: 10.1073/pnas.0307904100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zhong N. Management and prevention of SARS in China. Phil. Trans. R. Soc. Lond. B. 2004;359:1115–1116. doi: 10.1098/rstb.2004.1491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Liang G, et al. Laboratory diagnosis of four recent sporadic cases of community-acquired SARS, Guangdong Province, China. Emerg. Infect. Dis. 2004;10:1774–1781. doi: 10.3201/eid1010.040445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wong TW, et al. Cluster of SARS among medical students exposed to single patient, Hong Kong. Emerg. Infect. Dis. 2004;10:269–276. doi: 10.3201/eid1002.030452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Seto WH, et al. Effectiveness of precautions against droplets and contact in prevention of nosocomial transmission of severe acute respiratory syndrome (SARS) Lancet. 2003;361:1519–1520. doi: 10.1016/S0140-6736(03)13168-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Loon SC, et al. The severe acute respiratory syndrome coronavirus in tears. Br. J. Ophthalmol. 2004;88:861–863. doi: 10.1136/bjo.2003.035931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Leung GM, et al. SARS CoV antibody prevalence in all Hong Kong patient contacts. Emerg. Infect. Dis. 2004;10:1653–1656. doi: 10.3201/eid1009.040155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lipsitch M, et al. Transmission dynamics and control of severe acute respiratory syndrome. Science. 2003;300:1966–1970. doi: 10.1126/science.1086616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Goh DLM, et al. Secondary household transmission of SARS, Singapore. Emerg. Infect. Dis. 2004;10:232–234. doi: 10.3201/eid1002.030676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Shen Z, et al. Super-spreading SARS events in Beijing, 2003. Emerg. Infect. Dis. 2004;10:256–260. doi: 10.3201/eid1002.030732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Peiris JS, et al. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: a prospective study. Lancet. 2003;361:1767–1772. doi: 10.1016/S0140-6736(03)13412-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chan KH, et al. Detection of SARS coronavirus (SCoV) by RT-PCR, culture, and serology in patients with acute respiratory syndrome (SARS) Emerg. Infect. Dis. 2004;10:294–299. doi: 10.3201/eid1002.030610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Chan PK, et al. Laboratory diagnosis of SARS. Emerg. Infect. Dis. 2004;10:825–830. doi: 10.3201/eid1005.030682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Li L, et al. SARS-coronavirus replicates in mononuclear cells of peripheral blood (PBMCs) from SARS patients. J. Clin. Virol. 2003;28:239–244. doi: 10.1016/S1386-6532(03)00195-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ng EK, et al. Serial analysis of the plasma concentration of SARS coronavirus RNA in pediatric patients with severe acute respiratory syndrome. Clin. Chem. 2003;49:2085–2088. doi: 10.1373/clinchem.2003.024588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Wong RSM, et al. Haematological manifestations in patients with severe acute respiratory syndrome: retrospective analysis. BMJ. 2003;326:1358–1362. doi: 10.1136/bmj.326.7403.1358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Li T, et al. Significant changes of peripheral T lymphocyte subsets in patients with severe acute respiratory syndrome. J. Infect. Dis. 2004;189:648–651. doi: 10.1086/381535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Leung CW, et al. Severe acute respiratory syndrome in children. Pediatrics. 2004;113:e535–e543. doi: 10.1542/peds.113.6.e535. [DOI] [PubMed] [Google Scholar]
- 55.Hon KL, et al. Clinical presentation and outcome of severe acute respiratory syndrome in children. Lancet. 2003;361:1701–1703. doi: 10.1016/S0140-6736(03)13364-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Drosten C, et al. Evaluation of advanced reverse transcription PCR assays and an alternative PCR target region for detection of severe acute respiratory syndrome-associated coronavirus. J. Clin. Microbiol. 2004;42:2043–2047. doi: 10.1128/JCM.42.5.2043-2047.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Cheng PKC, et al. Viral shedding patterns of coronavirus in patients with probable severe acute respiratory syndrome. Lancet. 2004;363:1699–1700. doi: 10.1016/S0140-6736(04)16255-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kaiser L, Briones MS, Hayden FG. Performance of virus isolation and Directigen Flu A to detect influenza A virus in experimental human infection. J. Clin. Virol. 1999;14:191–197. doi: 10.1016/S1386-6532(99)00058-X. [DOI] [PubMed] [Google Scholar]
- 59.Ng EK, et al. Quantitation analysis and prognostic implication of SARS coronavirus RNA in the plasma and serum of patients with severe acute respiratory syndrome. Clin. Chem. 2003;49:1976–1980. doi: 10.1373/clinchem.2003.024125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Hung IF, et al. Viral loads in clinical specimens and SARS manifestations. Emerg. Infect. Dis. 2004;10:1550–1557. doi: 10.3201/eid1009.040058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Chu CM, et al. The role of lopinavir/ritonavir in treatment of SARS: initial virological and clinical findings. Thorax. 2004;59:252–256. doi: 10.1136/thorax.2003.012658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Mazzulli T, et al. Severe acute respiratory syndrome–associated coronavirus in lung tissue. Emerg. Infect. Dis. 2003;10:20–24. doi: 10.3201/eid1001.030404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Nicholls JM, et al. Lung pathology of fatal severe acute respiratory syndrome. Lancet. 2003;361:1773–1778. doi: 10.1016/S0140-6736(03)13413-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Franks TJ, et al. Lung pathology of severe acute respiratory syndrome (SARS): a study of 8 autopsy cases from Singapore. Hum. Pathol. 2003;34:743–748. doi: 10.1016/S0046-8177(03)00367-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Chow KC, et al. Detection of severe acute respiratory syndrome-associated coronavirus in pneumocytes of the lung. Am. J. Clin. Pathol. 2004;121:574–580. doi: 10.1309/C0EDU0RAQBTXBHCE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.To KF, et al. Tissue and cellular tropism of the coronavirus associated with severe acute respiratory syndrome: an in situ hybridization study of fatal cases. J. Pathol. 2004;202:157–163. doi: 10.1002/path.1510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Nakajima N, et al. SARS Coronavirus-infected cells in lung detected by new in situ hybridization technique. Jpn. J. Infect. Dis. 2003;56:139–141. [PubMed] [Google Scholar]
- 68.Chong PY, et al. Analysis of deaths during the severe acute respiratory syndrome (SARS) epidemic in Singapore. Arch. Pathol. Lab. Med. 2004;128:195–204. doi: 10.5858/2004-128-195-AODDTS. [DOI] [PubMed] [Google Scholar]
- 69.Ding Y, et al. Organ distribution of severe acute respiratory syndrome (SARS) associated coronavirus (SARS CoV) in SARS patients: implications for pathogenesis and virus transmission pathways. J. Pathol. 2004;203:622–630. doi: 10.1002/path.1560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.To KF, Lo AWI. Exploring the pathogenesis of severe acute respiratory syndrome (SARS): the tissue distribution of the coronavirus (SARS CoV) and its putative receptor, angiotensin-converting-enzyme 2 (ACE-2) J. Pathol. 2004;203:740–743. doi: 10.1002/path.1597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Leung WK, et al. Enteric involvement of severe acute respiratory syndrome-associated coronavirus infection. Gastroenterology. 2003;125:1011–1017. doi: 10.1016/j.gastro.2003.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Chan PKS, et al. Persistent infection of SARS coronavirus in colonic cells in vitro. J. Med. Virol. 2004;74:1–7. doi: 10.1002/jmv.20138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Cinatl J, et al. Infection of cultured intestinal epithelial cells with severe acute respiratory syndrome coronavirus. Cell Mol. Life Sci. 2004;61:2100–2112. doi: 10.1007/s00018-004-4222-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Yang ZY, et al. pH-dependent entry of severe acute respiratory syndrome coronavirus is mediated by the spike glycoprotein and enhanced by dendritic cell transfer through DC SIGN. J. Virol. 2004;78:5642–5650. doi: 10.1128/JVI.78.11.5642-5650.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Simmons G, et al. Characterization of severe acute respiratory syndrome-associated coronavirus (SARS CoV) spike glycoprotein-mediated viral entry. Proc. Natl. Acad. Sci. USA. 2004;101:4240–4245. doi: 10.1073/pnas.0306446101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Hoffmann H, et al. Susceptibility to SARS coronavirus S protein-derived infection correlates with expression of angiotensin converting enzyme 2 and infection can be blocked by soluble receptor. Biochem. Biophys. Res. Comm. 2004;319:1216–1221. doi: 10.1016/j.bbrc.2004.05.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Han DP, Kim HG, Kim YB, Poon LLM, Cho MW. Development of a safe neutralization assay for SARS CoV and characterization of S-glycoprotein. Virology. 2004;326:140–149. doi: 10.1016/j.virol.2004.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Bosch BJ, et al. Severe acute respiratory syndrome associated coronavirus (SARS CoV) infection inhibition using spike protein heptad repeat-derived peptides. Proc. Natl. Acad. Sci. USA. 2004;101:8455–8460. doi: 10.1073/pnas.0400576101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Liu S, et al. Interaction between heptad repeat 1 and 2 regions in spike protein of SARS-associated coronavirus: implications for virus fusogenic mechnism and identification of fusion inhibitors. Lancet. 2004;363:938–947. doi: 10.1016/S0140-6736(04)15788-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Qinfen Z, et al. The life cycle of SARS coronavirus in Vero E6 cells. J. Med. Virol. 2004;73:332–337. doi: 10.1002/jmv.20095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Li W, et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. 2003;426:450–454. doi: 10.1038/nature02145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Wang P, et al. Expression cloning of functional receptor used by SARS coronavirus. Biochem. Biophys. Res. Commun. 2004;315:439–444. doi: 10.1016/j.bbrc.2004.01.076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Babcock GJ, Esshaki DJ, Thomas WD, Ambrosino DM. Amino acids 270 to 510 of the severe acute respiratory syndrome spike protein are required for interaction with receptor. J. Virol. 2004;78:4552–4560. doi: 10.1128/JVI.78.9.4552-4560.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Lai MMC, Holmes KV. Fields' Virology. 2001. Coronaviridae and their replication; pp. 1163–1185. [Google Scholar]
- 85.Hammling I, et al. Tissue distribution of ACE-2 protein, the functional receptor for SARS coronavirus. J. Pathol. 2004;203:631–637. doi: 10.1002/path.1570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Yount B, et al. Reverse genetics with a full-length infectious cDNA of severe acute respiratory syndrome coronavirus. Proc. Natl. Acad. Sci. USA. 2003;100:12995–13000. doi: 10.1073/pnas.1735582100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Wong CK, et al. Plasma inflammatory cytokines and chemokines in severe acute respiratory syndrome. Clin. Exp. Immunol. 2004;136:95–103. doi: 10.1111/j.1365-2249.2004.02415.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zhang Y, et al. Analysis of serum cytokines in patients with severe acute respiratory syndrome. Infect. Immun. 2004;72:4410–4415. doi: 10.1128/IAI.72.8.4410-4415.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Ng PC, et al. Inflammatory cytokine profile in children with severe acute respiratory syndrome. Pediatrics. 2004;113:e7–e14. doi: 10.1542/peds.113.1.e7. [DOI] [PubMed] [Google Scholar]
- 90.Lee CH, et al. Altered p38 mitogen-activated protein kinase expression in different leukocytes with increment of immunosuppressive mediators in patients with severe acute respiratory syndrome. J. Immunol. 2004;172:7841–7847. doi: 10.4049/jimmunol.172.12.7841. [DOI] [PubMed] [Google Scholar]
- 91.Peiris JSM, et al. Re-emergence of fatal human influenza A subtype H5N1 disease. Lancet. 2004;363:617–619. doi: 10.1016/S0140-6736(04)15595-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Jones BM, et al. Prolonged disturbance of in vitro cytokine production inpatients with severe acute respiratory syndrome (SARS) treated with ribavirin and steroids. Clin. Exp. Immunol. 2004;135:467–473. doi: 10.1111/j.1365-2249.2003.02391.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Openshaw PJM. What does the peripheral blood tell you in SARS? Clin. Exp. Immunol. 2004;136:11–12. doi: 10.1111/j.1365-2249.2004.02448.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Lin M, et al. Association of HLA class 1 with severe acute respiratory syndrome coronavirus infection. BMC Med. Genet. 2003;4:9. doi: 10.1186/1471-2350-4-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Ng MHL, et al. Association of human-leukocyte-antigen class 1 (B*0703) and class II (DRB1*0301) genotypes with susceptibility and resistance to the development of severe acute respiratory syndrome. J. Infect. Dis. 2004;190:5151–5158. doi: 10.1086/421523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Fouchier RAM, et al. Koch's postulates fulfilled for SARS virus. Nature. 2003;423:240. doi: 10.1038/423240a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Kuiken T, et al. Newly discovered coronavirus as the primary cause of severe acute respiratory syndrome. Lancet. 2003;362:263–270. doi: 10.1016/S0140-6736(03)13967-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Rowe T, et al. Macaque model for severe acute respiratory syndrome. J. Virol. 2004;78:11410–11414. doi: 10.1128/JVI.78.20.11401-11404.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Martina BE, et al. Virology: SARS virus infection of cats and ferrets. Nature. 2003;425:915. doi: 10.1038/425915a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Bukreyev A, et al. Mucosal immunisation of African green monkeys (Cercopithicus aethiops) with an attenuated parainfluenza virus expressing the SARS coronavirus spike protein for the prevention of SARS. Lancet. 2004;363:2122–2127. doi: 10.1016/S0140-6736(04)16501-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Subbarao K, et al. Prior infection and passive transfer of neutralizing antibody prevent replication of severe acute respiratory syndrome coronavirus in the respiratory tract of mice. J. Virol. 2004;78:3572–3577. doi: 10.1128/JVI.78.7.3572-3577.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Buchholtz UJ, et al. Contributions of the structural proteins of severe acute respiratory syndrome coronavirus to protective immunity. Proc. Natl. Acad. Sci. USA. 2004;101:9804–9809. doi: 10.1073/pnas.0403492101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Roberts, A. et al. SARS coronavirus infection in Golden Syrian hamsters. J. Virol. (in the press). [DOI] [PMC free article] [PubMed]
- 104.Wu CY, et al. Small molecules targeting severe acute respiratory syndrome human coronavirus. Proc. Natl. Acad. Sci. USA. 2004;101:10012–10017. doi: 10.1073/pnas.0403596101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Wu CJ, et al. Inhibition of severe acute respiratory syndrome coronavirus replication by niclosamide. Antimicrob. Agents Chemother. 2004;48:2693–2696. doi: 10.1128/AAC.48.7.2693-2696.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Chen F, et al. In vitro susceptibility of 10 clinical isolates of SARS coronavirus to selected antiviral compounds. J. Clin. Virol. 2004;31:69–75. doi: 10.1016/j.jcv.2004.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Yi Y, et al. Small molecules blocking the entry of severe acute respiratory syndrome coronavirus into host cells. J. Virol. 2004;78:11334–11339. doi: 10.1128/JVI.78.20.11334-11339.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Yamamoto N, et al. HIV protease inhibitor nelfinavir inhibits replication of SARS-associated coronavirus. Biochem. Biophys. Res. Commun. 2004;318:719–725. doi: 10.1016/j.bbrc.2004.04.083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Tan EL, et al. Inhibition of SARS coronavirus infection in vitro with clinically approved antiviral drugs. Emerg. Infect. Dis. 2004;10:581–586. doi: 10.3201/eid1004.030458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Hensley LE, et al. Interferon-β 1a and SARS coronavirus replication. Emerg. Infect. Dis. 2004;10:317–319. doi: 10.3201/eid1002.030482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Stroher U, et al. Severe acute respiratory syndrome-related coronavirus is inhibited by interferon-α. J. Infect. Dis. 2004;189:1164–1167. doi: 10.1086/382597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Spiegel M, Pichlmair A, Muhlberger E, Haller O, Weber F. The antiviral effect of interferon-β against SARS coronavirus is not mediated by MxA protein. J. Clin. Virol. 2004;30:211–213. doi: 10.1016/j.jcv.2003.11.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Zhang XW, Yap YL. Old drugs as lead compounds for a new disease? Binding analysis of SARS coronavirus main proteinase with HIV, psychotic, and parasitic drugs. Bioorg. Med. Chem. 2004;12:2517–2521. doi: 10.1016/j.bmc.2004.03.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Haagmans BL, et al. Pegylated interferon-α protects type 1 pneumocytes against SARS coronavirus infection in macaques. Nat. Med. 2004;10:290–293. doi: 10.1038/nm1001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Loutfy MR, et al. Interferon alfacon-1 plus corticosteroids in severe acute respiratory syndrome: a preliminary study. J. Am. Med. Assoc. 2003;290:3222–3228. doi: 10.1001/jama.290.24.3222. [DOI] [PubMed] [Google Scholar]
- 116.Anand K, et al. Coronavirus main protease (3Clpro) structure: basis for design of antiviral drugs. Science. 2003;300:1763–1767. doi: 10.1126/science.1085658. [DOI] [PubMed] [Google Scholar]
- 117.Ivanov KA, et al. Multiple enzymatic activities associated with severe acute respiratory syndrome coronavirus helicase. J. Virol. 2004;78:5619–5632. doi: 10.1128/JVI.78.11.5619-5632.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.He ML, et al. Inhibition of SARS-associated coronavirus infection and replication by RNA interference. JAMA. 2003;290:2665–2666. doi: 10.1001/jama.290.20.2665. [DOI] [PubMed] [Google Scholar]
- 119.Wang Z, et al. Inhibition of severe acute respiratory syndrome virus replication by small interfering RNA in mammalian cells. J. Virol. 2004;78:7523–7527. doi: 10.1128/JVI.78.14.7523-7527.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Zhang Y, et al. Silencing SARS CoV spike protein expression in cultured cells by RNA interference. FEBS Lett. 2004;560:141–146. doi: 10.1016/S0014-5793(04)00087-0. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 121.Kao RY, et al. Identification of novel small molecule inhibitors of severe acute respiratory syndrome associated coronavirus by chemical genetics. Chem. Biol. 2004;11:1293–1299. doi: 10.1016/j.chembiol.2004.07.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Lu L, et al. Immunological characterization of the spike protein of the severe acute respiratory syndrome coronavirus. J. Clin. Microbiol. 2004;42:1570–1576. doi: 10.1128/JCM.42.4.1570-1576.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Zhang H, et al. Identification of an antigenic determinant on the S2 domain of the severe acute respiratory syndrome coronavirus spike protein capable of inducing neutralizing antibodies. J. Virol. 2004;78:6938–6945. doi: 10.1128/JVI.78.13.6938-6945.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Wang YD, et al. T cell epitopes in severe acute respiratory syndrome (SARS) coronavirus spike protein elicit a specific T-cell immune response in patients who recover from SARS. J. Virol. 2004;78:5612–5618. doi: 10.1128/JVI.78.11.5612-5618.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Nie Y, et al. Neutralizing antibodies in patients with severe acute respiratory syndrome-associated coronavirus infection. J. Infect. Dis. 2004;190:1119–1126. doi: 10.1086/423286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Yang ZY, et al. A DNA vaccine induces SARS coronavirus neutralization and protective immunity in mice. Nature. 2004;428:561–564. doi: 10.1038/nature02463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Kim TW, et al. Generation and characterization of DNA vaccines targeting the nucleocapsid protein of severe acute respiratory syndrome coronavirus. J. Virol. 2004;78:4638–4645. doi: 10.1128/JVI.78.9.4638-4645.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Zeng F, et al. Characterization of humoral responses in mice immunized with plasmid DNAs encoding SARS-CoV spike gene fragments. Biochem. Biophys. Res. Commun. 2004;315:1134–1139. doi: 10.1016/j.bbrc.2004.01.166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Zhu MS, et al. Induction of SARS-nucleoprotein-specific immune response by use of DNA vaccine. Immunol Lett. 2004;92:237–243. doi: 10.1016/j.imlet.2004.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Gao W, et al. Effects of a SARS-associated coronavirus vaccine in monkeys. Lancet. 2003;362:1895–1896. doi: 10.1016/S0140-6736(03)14962-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Bisht H, et al. Severe acute respiratory syndrome coronavirus spike protein expressed by attenuated vaccinia virus protectively immunizes mice. Proc. Natl. Acad. Sci. USA. 2004;101:6641–6646. doi: 10.1073/pnas.0401939101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Takasuka N, et al. A subcutaneously injected UV-inactivated SARS coronavirus vaccine elicits systemic humoral immunity in mice. Int. Immunol. 2004;16:1423–1430. doi: 10.1093/intimm/dxh143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Tang L, et al. Inactivated SARS-CoV vaccine prepared from whole virus induces a high level of neutralizing antibodies in BALB/c mice. DNA Cell Biol. 2004;23:391–394. doi: 10.1089/104454904323145272. [DOI] [PubMed] [Google Scholar]
- 134.Cavanagh D. Severe acute respiratory syndrome vaccine development: experiences of vaccination against avian infectious bronchitis coronavirus. Avian Pathol. 2003;32:567–582. doi: 10.1080/03079450310001621198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Olsen CW. A review of feline infectious peritonitis virus: molecular biology, immunopathogenesis, clinical aspects, and vaccination. Vet. Microbiol. 1993;36:1–37. doi: 10.1016/0378-1135(93)90126-R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Sui J, et al. Potent neutralization of severe acute respiratory syndrome (SARS) coronavirus by a human mAB to S1 protein that blocks receptor association. Proc. Natl. Acad. Sci. USA. 2004;101:2536–2641. doi: 10.1073/pnas.0307140101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Ter Muelen J, et al. Human monoclonal antibody as prophylaxis for SARS coronavirus infection in ferrets. Lancet. 2004;363:2139–2141. doi: 10.1016/S0140-6736(04)16506-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Traggian E, et al. An efficient method to make human monoclonal antibodies from memory B cells: potent neutralization of SARS coronavirus. Nat. Med. 2004;10:871–875. doi: 10.1038/nm1080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Soo YOY, et al. Retrospective comparison of convalescent plasma with continuing high dose methyl prednisolone treatment in SARS patients. Clin. Microbiol. Infect. 2004;10:657–658. doi: 10.1111/j.1469-0691.2004.00956.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Smolinski MS, Hamburg MA, Lederberg J. Microbial Threats to Health: Emergence, Detection and Response. 2003. [PubMed] [Google Scholar]
- 141.Li KS, et al. Genesis of a highly pathogenic and potentially pandemic H5N1 influenza virus in eastern Asia. Nature. 2004;430:209–213. doi: 10.1038/nature02746. [DOI] [PubMed] [Google Scholar]
- 142.Mackenzie JS, Field HF, Guyatt KJ. Managing emerging diseases borne by fruit bats (flying foxes), with particular reference to henipaviruses and Australian bat lyssavirus. J. Appl. Microbiol. 2003;94(Suppl):59S–69S. doi: 10.1046/j.1365-2672.94.s1.7.x. [DOI] [PubMed] [Google Scholar]
- 143.Poon, L.L.M. et al. Identification of a novel coronavirus in bats. J. Virol. (in the press). [DOI] [PMC free article] [PubMed]
- 144.Singer PA, et al. Ethics and SARS: Lessons from Toronto. BMJ. 2003;327:1342–1344. doi: 10.1136/bmj.327.7427.1342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.O'Neill O. Informed consent and public health. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2004;359:1133–1136. doi: 10.1098/rstb.2004.1486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.World Health Organization. A multicentre collaboration to investigate the cause of severe acute respiratory syndrome. Lancet361, 1730–1733 (2003). [DOI] [PMC free article] [PubMed]
- 147.Fidler DP. Germs, governance, and global public health in the wake of SARS. J. Clin. Invest. 2004;113:799–804. doi: 10.1172/JCI200421328. [DOI] [PMC free article] [PubMed] [Google Scholar]


