Abstract
Aim:
The association of transporters gene polymorphisms with chloroquine/primaquine malaria treatment response was investigated in a Brazilian population.
Patients & methods:
Totally, 164 Plasmodium vivax malaria infected patients were included. Generalized estimating equations were performed to determine gene influences on parasitemia and/or gametocytemia clearance over treatment time.
Results:
Significant interaction between SLCO2B1 genotypes and treatment over time for parasitemia clearance rate on day 2 were observed (p FDR = 0.002). SLCO1A2 and SLCO1B1 gene treatment over time interactions were associated with gametocytemia clearance rate (p FDR = 0.018 and p FDR = 0.024). ABCB1, ABCC4 and SLCO1B3 were not associated with treatment response.
Conclusion:
The present work presents the first pharmacogenetic report of an association between chloroquine/primaquine responses with OATP transporters.
Keywords: : chloroquine and primaquine, malaria, transporter genes
First draft submitted: 2 May 2017; Accepted for publication: 12 July 2017; Published online: 4 October 2017
Plasmodium vivax is the major cause of malaria outside Africa and represents a real challenge for malaria eradication in Asia and America continents, because of its intrinsic characteristic to develop dormant hypnozoite forms in the liver that cause subsequent infections in the blood [1–3].
In Brazil, chloroquine (CQ) and primaquine (PQ) combined therapy is the first choice treatment protocol for uncomplicated P. vivax malaria [4,5]. This treatment protocol with quinine derivatives combined targets asexual schizonts in blood and tissues, sexual gametocytes in blood and also hypnozoites in the liver [6–8]. Interindividual variability in CQ and PQ concentrations were reported in different populations [9–16]. Recently, the association of CYP450 gene variants with CQ/PQ treatment and relapses were reported [17,18]; however, the effect of genetic variants in membrane transporters were not tested so far.
The potential role of drug transporters in antimalarial treatment became clear after the observation that malaria parasite expresses transporter genes in its digestive vacuole as defense mechanism against, for example, CQ. It is likely that there is a large overlap in substrate specificity between drug transporters in Plasmodium spp. and humans, therefore, genetic variants of drug transporters in humans might foster the development of drug resistance by, for example, lowering drug concentrations in red blood cells [19].
Transporters are integral membrane proteins that mediate the translocation of chemicals into and out of cells using active and passive mechanisms. ATP-binding cassette (ABC) and solute carrier (SLC) transporter families are formed by influx and efflux transporters expressed on membranes of polarized cells and have been shown to significantly affect concentrations of drugs in plasma and peripheral tissues, thus affecting drug efficacy and toxicity [20–22]. Membrane transporters such as MDR1 and multidrug proteins (MRP) are members of the ABC family and use ATP to move substrates across membranes [21,22]. Instead, the organic anion-transporting polypeptides (OATPs) move substrates against a concentration gradient without ATP expenditure [20], and together with ABC transporters are responsible for transport and availability of several endogenous and exogenous compounds. CQ seems to be an ATP-binding transporters inhibitor; however, some evidences indicate that this drug is a potential substrate for these transporters. The MRP transport system is responsible for CQ cellular direct efflux in multidrug-resistant tumor cells [23,24], and this drug could also act as substrate or inhibitor of human MDR1 transporter [25,26]. PQ also inhibits MDR1 and MRP1 drug transport without being a substrate [26]. Recently, CQ was described as an important inhibitor of OATP1A2 and OATP1B1 functions, representing a possible role in drug–drug interactions and malaria treatment [27,28].
To improve the understanding of how genetic variants influence CQ/PQ malaria treatment, the present study aims to investigate if ABCB1, ABCC4, SLCO1A2, SLCO1B1, SLCO1B3 and SLCO2B1 polymorphisms are associated with P. vivax malaria treatment response in a Brazilian population.
Patients & methods
Study population
The study population was composed of 164 P. vivax malaria patients. All subjects were born in Para state. This big Brazilian Amazonian state presents different risk of infection and transmission among different regions and cities [29]. Some patients were infected in their home cities whereas others while traveling to endemic regions. All subjects were diagnosed and treated in Belém at the Evandro Chagas Institute between 2007 and 2009. Sample and collection procedures were previously described [17]. Briefly, patients were clinically examined and received the standard treatment of 1500 mg of CQ associated with 210 mg of PQ in short regimen during a week (first day CQ 600 mg and PQ 30 mg, second and third days CQ 450 mg and PQ 30 mg, and last 4 days PQ 30 mg) as recommended by the Brazilian health authorities [30]. During the week treatment, patient response was daily accompanied by clinical examinations. Parasitemia and gametocytemia were daily estimated (density per microliter of blood by counting the number of parasites per 100 fields and double-checked blindly by two expert microscopists). Parasite counts were obtained before treatment and in the next 7 days of treatment. All subjects provided their written informed consent to participate in this study. Participants younger than 18 years (n = 17) had the informed consents signed by parents to participate in the study. The Ethics Committees of the Evandro Chagas Institute and Federal University of Pará approved the study protocol (CEP/IEC-0035 and CEP-CCS/UFPA 061/07).
Genotyping
Genomic DNA was extracted from peripheral blood leukocytes using proteinase K digestion and standard phenol–chloroform procedures [31]. SNPs in ABCB1, ABCC4, SLCO1A2, SLCO1B1, SLCO1B3 and SLCO2B1 genes were determined by allelic discrimination with Taqman 5′-nuclease assays (Supplementary Table 1; real-time PCR, Applied Biosystems, CA, USA) according to the manufacturer's recommended protocol.
Statistical analysis
Allele and genotype frequencies were estimated by gene counting. Deviation from Hardy–Weinberg equilibrium was verified by χ2 with Bonferroni correction. Haplotypes and linkage disequilibrium were estimated with PHASE 2.1.1 [32]. The individual proportions of European, African and Amerindian genetic ancestry from the study population were estimated as previously described [33]. Generalized estimating equation (GEE) is a repeated measure analysis focused on average changes in response over time and the impact of covariates on these changes. This method models the mean response as a linear function of covariates of interest via transformation or link function and can be used in studies in which data are asymmetric or the data distribution is difficult to verify due to small sample size [34]. This analysis was performed to determine the genetic influence in parasitemia or gametocytemia clearance by treatment over time considering a Gaussian distribution with an identity link function and an exchangeable correlation matrix structure. Parasitemia and gametocytemia levels were log-transformed before analysis due to their skewed distribution, but back-transformed values are presented in the results as geometric means. Age, gender, co-medication, parasitemia baseline level, gametocytemia baseline level and African and Amerindian genetic ancestry entered in models as covariates based on conceptual analyses of the literature and/or by means of a statistical definition (association with the study factor and with the outcome at p ≤ 0.15). Based on previous investigations with the same sample, CYP2C8 genotypes were also used as a covariate in the gametocytemia analyses [17]. GEE analysis was performed with the SPSS18.0 (IBM Company, NY, USA) statistical package for Windows®. Benjamini–Hochberg procedure (false discovery rate [FDR]) for multiple comparisons was performed to control for multiple testing and corrected p-values were presented. Statistical significance was defined as a two-tailed p-value <0.05. To determine the effect sizes, Cohen's d test was calculated based on standardized differences between means [35].
Results
Major demographic and clinical features from the study population are summarized in Table 1. Malaria patients were aged between 12 and 88 years (36.0 ± 15.6 years), and 29 patients (17.6%) used other medications in combination with CQ/PQ treatment to manage malaria symptoms or pre-existing diseases. All patients completed the 7 days treatment and adverse drug reactions were not reported. After complete treatment all patients presented negative results for parasites and gametocytes in blood and were monthly followed for 6 months. Twenty-seven patients (16.5%) presented relapses after at least 1 month of treatment and repeated the therapeutic regimen with no more relapses until the end of the follow-up period.
Table 1. . Study group main characteristics.
| Characteristics | Malaria patients |
|---|---|
| n | 164 |
| Age | 36.0 (15.6) |
| Gender (male %) | 68.5 |
| Baseline parasitemia (parasites/μl) | 8554.35 (50–75,000) |
| Baseline gametocytemia (gametocytes/μl) | 110.93 (0–4500) |
| Genetic ancestry | |
| African | 0.243 (0.09) |
| European | 0.415 (0.11) |
| Native American | 0.340 (0.12) |
| Co-medication (%) | |
| Antiemetic | 4.2 |
| Antipyretic/analgesic | 12.2 |
| Antacid | 2.4 |
| Antibiotic | 1.2 |
| Anthelmintic | 0.6 |
| Anticonvulsant | 0.6 |
| ACE inhibitor | 0.6 |
Values for age and genetic ancestry are expressed as mean (SD).
Values for parasitemia and gametocytemia are expressed as median (range).
SD: Standard deviation.
ABCB1, ABCC4, SLCO1A2, SLCO1B1, SLCO1B3 and SLCO2B1 allele and genotype frequencies are shown in Supplementary Table 2. Haplotype frequencies are shown in Supplementary Table 3. Genotype distributions did not deviate significantly from Hardy–Weinberg equilibrium in the study population. ABCB1, SLCO1B1 and SLCO1B3 allelic, genotype and haplotype frequencies showed no statistical differences from frequencies observed in a general population sample from Belém (data not shown, but available upon request) [36].
Influence of ABCB1 & SLCO2B1 in parasitemia clearance
ABCB1 and SLCO2B1 genes were associated with the clearance rate over treatment time in the model adjusting for age, gender, co-medication, parasitemia baseline level and genetic ancestry. After FDR procedure, ABCB1 gene was no longer associated with parasitemia clearance rate over treatment time. No main gene effect was observed for both genes (Table 2).
Table 2. . Mean parasitemia over treatment time according to ABCB1 and SLCO2B1 genes.
| Gene | Treatment | Mean parasitemia (parasites/μl) | p | pFDR | d† | ||||
|---|---|---|---|---|---|---|---|---|---|
| Gene | Gene*time | Gene | Gene*time | ||||||
| ABCB‡ | CGC/CGC (n = 43) | TnonGT (n = 83) | Others (n = 31) | ||||||
| Day 0 | 3894.85 | 3574.30 | 3680.04 | ||||||
| (3211.5–4723.6) | (2728.8–4681.8) | (2729–4962.5) | |||||||
| Day 1 | 360.53 | 244.93 | 279.32 | 0.568 | 0.044 | NS | 0.132 | 0.41 | |
| (186.23–697.95) | (141.20–424.86) | (153.34–508.80) | |||||||
| Day 2 | 0.44 | 0.78 | 1.98 | ||||||
| (0.12–1.67) | (0.29–2.10) | (0.55–7.12) | |||||||
| Day 3 | 0.01 | 0.02 | 0.01 | ||||||
| (0.005–0.014) | (0.012–0.048) | (0.004–0.025) | |||||||
| SLCO2B1§ | AA (n = 26) | AG (n = 79) | GG (n = 50) | ||||||
| Day 0 | 3626.79 | 3722.19 | 3758.23 | ||||||
| (2734.5–4810.2) | (2879.6–4811.3) | (2809.1–5028.0) | |||||||
| Day 1 | 321.52 | 339.01 | 201.12 | 0.073 | <0.0002 | NS | 0.002 | 0.89 | |
| (163.85–630.90) | (225.43–509.82) | (82.73–488.94) | |||||||
| Day 2 | 0.10 | 0.86 | 3.23 | ||||||
| (0.02–0.48) | (0.33–2.28) | (0.99–10.54) | |||||||
| Day 3 | 0.01 | 0.02 | 0.01 | ||||||
| (0.004–0.018) | (0.011–0.036) | (0.005–0.030) | |||||||
Mean parasitemia adjusted for age, gender, genetic ancestry, co-medication and parasitemia baseline level.
Parasitemia is expressed as geometric mean (95% CI).
†Effect size Cohen's d.
‡Haplotypes based on SNPs 3435C>T, 2677G>A/T and 1236C>T.
§Genotypes from SNP 935G>A.
NS: Not significant.
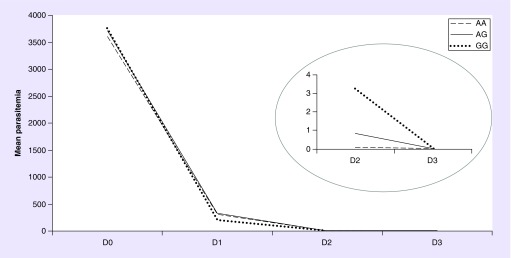
The model including treatment over time with SLCO2B1 genotypes is represented in Figure 1. Although no main genotype effect was observed (pFDR = 0.100), a significant interaction effect between genotypes and treatment over time for parasitemia clearance rate during treatment (pFDR = 0.002) was observed. On day 2 SLCO2B1 AA carriers showed an interaction effect between genotypes and parasitemia clearance rate over treatment time when compared with GG genotype carriers (AA vs AG: p = 0.076; AA vs GG: p = 0.002; and AG vs GG: p = 0.262). Considering Cohen's classification, this interaction presents a large effect size (d = 0.89) [37].
Figure 1. . Mean parasitemia clearance during chloroquine/primaquine regimen according to SLCO2B1 genotypes.
Generalized estimating equations method with age, gender, co-medication, parasitemia baseline level and genetic ancestry as covariates; gene*time pFDR = 0.002 and d = 0.89. In the right circle, it was highlighted the mean parasitemia clearance in the days 2 and 3. On day 2, AA genotype showed a higher parasitemia clearance rate over treatment time than GG genotypes. Mean parasitemia means are presented as parasites/ul (for details, see ‘Patients & methods’ section).
ABCC4, SLCO1A2, SLCO1B1 and SLCO1B3 variants were not associated with parasitemia clearance rate.
Influence of SLCO1A2 & SLCO1B1 in gametocytemia clearance
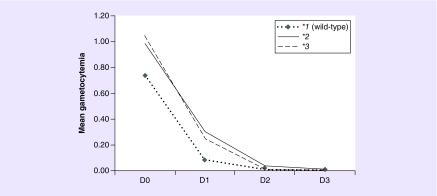
SLCO1A2 and SLCO1B1 genes were associated with the gametocytemia clearance rate over treatment time in GEE analyses adjusted for age, gender, co-medication, parasitemia baseline level, CYP2C8 genotypes and genetic ancestry. SLCO1A2 was associated with gametocytemia (p = 0.003); SLCO1A2*2 carriers presented higher mean gametocytemia SLCO1A2*1 homozygotes (p = 0.002). The model including treatment over time in the presence of SLCO1A2 genotypes is represented in Figure 2; a significant interaction effect between genotypes and treatment over time for gametocytemia clearance rate during treatment was observed (pFDR = 0.018). The model showed that SLCO1A2*2 and SLCO1A2*3 carriers have clearance of gametocytes at a lower rate as compared with SLCO1A2*1 homozygotes during treatment. Following effect size Cohen's interpretation scale, SLCO1A2 model showed medium effect size (d = 0.59).
Figure 2. . Mean gametocytemia clearance during chloroquine/primaquine regimen according to SLCO1A2 haplotypes.
Generalized estimating equations method with age, gender, co-medication, gametocytemia baseline level, CYP2C8 genotypes and genetic ancestry as covariates. On day 2 SLCO2B1 AA carriers showed an interaction effect between genotypes and parasitemia clearance rate over treatment time when compared with GG genotype carriers (AA vs AG: p = 0.076; AA vs GG: p = 0.002; and AG vs GG: p = 0.262). Gene*time pFDR = 0.018 and d = 0.59. Gametocytemia means are presented as gametocytes/ul (for details, see ‘Patients & methods’ section).
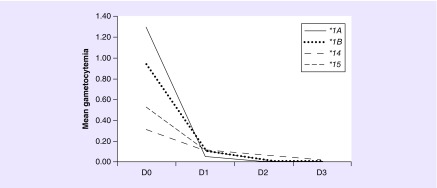
The main effect of SLCO1B1 genotype was not associated with mean gametocytemia in the analysis (p = 0.086), nevertheless, a significant interaction effect between genotype and gametocytemia clearance rate over time (pFDR = 0.024) was observed. The model represented in Figure 3 showed lower gametocytemia clearance rate over time by SLCO1B1*14 carriers compared with SLCO1B1*1a and SLCO1*1b allele carriers (on second day: *1a vs *14; p = 0.001 and *1b vs *14; p = 0.002; Figure 3). This model showed large effect sizes (d = 1.20) based on Cohen's interpretation scale [37].
Figure 3. . Mean gametocytemia clearance during chloroquine/primaquine regimen according to SLCO1B1 haplotypes.
Generalized estimating equations method with age, gender, co-medication, gametocytemia baseline level, CYP2C8 genotypes and genetic ancestry as covariates; gene*time pFDR = 0.024 and d = 1.20. A lower gametocytemia clearance rate over time by SLCO1B1*14 carriers compared with SLCO1B1*1a and SLCO1*1b allele carriers is represented (on second day: *1a vs *14; p = 0.001 and *1b vs *14; p = 0.002). Gametocytemia means are presented as gametocytes/ul (for details, see ‘Patients & methods’ section).
Models including ABCB1, ABCC4, SLCO1B2 and SLCO1B3 were not associated with gametocytemia clearance rate.
Discussion
The main finding of the present study was that SLC transporters influence P. vivax malaria treatment in a Brazilian Amazonian population. CQ and PQ have a synergistic effect as schizonticide, gametocide and hipnozontocide, and could be either substrates or inhibitors for these transporters [15,19,20].
Several studies showed fundamental differences in the pharmacodynamics and pharmacokinetic action of CQ between Plasmodium vivax and P. falciparum [38,39]. Moreover, CQ resistance grades in P. vivax are not fully understood as it is for P. falciparum. The patterns observed in in vitro tests for resistance did not show relapses or recrudescence before 28 days [8,40]. Current P. vivax in vivo studies are unable to distinguish a recrudescence of CQ-resistant parasites from a relapse or a new infection [41].
Knowledge about the mechanisms and molecular markers of CQ resistance in P. vivax remains limited. Due to intrinsic biological dissimilarities, extrapolation from P. falciparum may not reflect the true drug resistance mechanisms of P. vivax, thus limiting the number of parasite markers definitively identified in P. vivax [42]. Although how the interactions among CQ and PQ with OATP1A2, OATP1B1 and OATP2B1 occur is not fully understood, the results of the present study suggest that polymorphisms in these transporters should alter the availability of both drugs in different tissues interfering with their action in a specific manner affecting the different sexual forms of P. vivax. Thus, it is an important variability factor that should be considered together with the metabolism variability of CQ and PQ on treatment response.
SLCs’ organic anion-transporting polypeptides (OATPs) are plasma membrane proteins that mediate the active cellular influx of a variety of amphipathic compounds. These proteins were expressed in apical and basolateral membranes of polarized cells in tissues such as liver and kidneys, as well as in the intestinal wall and the blood–brain barrier, and may affect pharmacokinetics and effects of their substrates [43,44].
The nonsynonymous 935G>A SNP in SLCO2B1 was associated with parasitemia clearance rate in malaria treatment in this study population. Homozygous patients for the 935A allele showed a significant higher parasitemia clearance rate over treatment time when compared with GG homozygotes. OATP2B1 also is expressed in the sinusoidal membrane of hepatocytes in the luminal membrane of small intestinal enterocytes, suggesting that it participate in drugs uptake from blood and absorption [45,46]. Recently, the expression of this transporter on red blood cell membrane was described, but the action of antimalarial drugs such as CQ as inhibitor or substrate of OATP2B1 was not observed in vitro [28]. Despite this, 935G>A SNP showed an important effect in CQ/PQ treatment variability in the present study and pharmacokinetic mechanisms should be further investigated.
OATP1B1 is mainly expressed in the sinusoidal membrane of human hepatocytes diffusely distributed in the liver lobulus [44]. This protein mediates uptake of its substrates from blood into the liver [45] and is encoded by SLCO1B1 gene. Polymorphisms in SLCO1B1 were well characterized and associated with statins pharmacogenetics [47–52]. In the present study, the SLCO1B1*14 allele carriers have lower gametocytemia clearance rate over treatment time in malaria. This allele is formed by 388G and 463A SNP variants, which were associated with reduced transport activity in vitro [53]. Recently, it has been shown that CQ increases OATP1B1 levels in different models in vitro and interacts with this transporter as an inhibitor decreasing transport activity [27].
OATP1A2 is highly expressed in the brain, but it is also expressed in, intestine, kidneys, lung and testes in different amounts [54–56]. OATP1A2 is encoded by SLCO1A2 gene, and its polymorphisms were mainly associated with cancer treatment pharmacokinetics [57,58]. The results of the present work showed that SLCO1A2*2 and SLCO1A2*3 presented lower gametocytes clearance rates over treatment time. These alleles are associated with changes in the protein sequence (I13T and E172D), and are related to an increased and decreased uptake of substrates, respectively [59]. Recently, the expression of this transporter on red blood cell membrane was described as well as the quinine transport activity inside these cells [28]. CQ is an important OATP1A2 inhibitor [28], therefore, the influence of OATP1A2 in CQ and PQ treatment should be better investigated.
The present study also showed the association of ABCB1 gene haplotypes on parasitemia clearance rate over treatment time. MDR1 (or P-glycoprotein) is responsible for the active efflux of many drugs, by biliary and renal excretion [20,21,60,61]. The present results showed that ABCB1 T/nonG/T haplotype carriers (3435C>T, 2677G>A/T and 1236C>T) showed a lower parasitemia clearance rate over treatment time when compared with wild-type haplotype CGC homozygotes; however, after the FDR procedure, this difference was no longer statistically significant. CQ seems to be a broad inhibitor of ABC transporters and a potential substrate of some ABCs. The significance loss in the analysis after multiple comparisons correction could be related to the polymorphism small effect size. The role of ABCB1 variants on CQ pharmacokinetics in malaria treatment should be better investigated in future studies.
The observational-naturalistic design of our study, moderate sample size and the absence of CQ/PQ plasma levels information in our patients are limitations of the present study. However, this design might be valuable to better appreciate the role of genetic factors in routine clinical practice beyond the realm of controlled clinical trials, but some caveats of this kind of studies should be considered. First, we had no internal control to correct for any effect of time (e.g., regression to the mean) or expectancy bias because we did not have a placebo arm in this trial. Second, we did not control for parasite resistance. Nevertheless, CQ-resistant P. vivax has been estimated to vary from 4.4 to 10% in the Amazonian region [62] and all patients presented negative results after treatment. The role of transporter gene variability is associated with the rate of clearance and not with efficacy per se. Although it is not possible to exclude that the effects we observed were due to lack of adherence to treatment, there is no reason to expect a preferential compliance to CQ/PQ treatment according to transporter gene genotypes. This study design also did not allow to evaluate PQ effect in liver hypnozoites, or to evaluate the effect of transporter genes in relapses occurrence.
Conclusion
The present study reports important effects of transporters gene variants in CQ/PQ pharmacokinetics, which could represent an important factor to permit P. vivax resistance to this treatment. The study was the first to describe these pharmacogenetic influences in P. vivax malaria treatment and the results found should be replicated in larger and independent samples.
Future perspective
Despite all the efforts to develop a multiple drug therapy that has a good response in the malaria treatment, this disease still is an important morbidity and mortality factor in several world regions, among them the Amazonian region. For now, pharmacogenetic studies in this kind of disease are scarce, mainly because of their complexity. However, the present study reports an important contribution to the development of the personalized treatment in the malaria disease. More studies analyzing the role of the genetic background in the drug response could help in better drug therapy prescription, minimizing their adverse effects and improving their effectiveness.
Summary points.
To improve the understanding of how genetic variants influence chloroquine/primaquine malaria treatment, the present study investigated if ABCB1, ABCC4, SLCO1A2, SLCO1B1, SLCO1B3 and SLCO2B1 polymorphisms are associated with Plasmodium vivax malaria treatment response.
Analyses of the effect of different genotypes on treatment efficacy were performed using generalized estimating equations to determine the genetic influence on parasitemia or gametocytemia clearance over time.
ABCB1 and SLCO2B1 genes were associated with the parasitemia rate over treatment time in the model adjusting for age, gender, co-medication, parasitemia baseline level and genetic ancestry. After FDR procedure, ABCB1 gene was no longer associated with parasitemia rates.
A significant interaction effect between SLCO2B1 genotypes and treatment over time for parasitemia elimination rates during treatment (pFDR = 0.002) was observed. On day two SLCO2B1 AA carriers showed higher parasitemia elimination rates than GG genotype carriers (AA vs AG: p = 0.076; AA vs GG: p = 0.002; and AG vs GG: p = 0.262). Considering Cohen's classification, this interaction presents a large effect size (d = 0.89).
SLCO1A2 and SLCO1B1 genes were associated with gametocytemia rate over treatment time in generalized estimating equation analyses adjusted for age, gender, co-medication, parasitemia baseline level, CYP2C8 genotypes and genetic ancestry. SLCO1A2 was associated with gametocytemia (p = 0.003).
A significant interaction effect between genotypes and treatment over time for gametocytemia elimination rates during treatment was observed (pFDR = 0.018). The model showed that SLCO1A2*2 and SLCO1A2*3 carriers eliminates gametocytes in a lower rate as compared with SLCO1A2*1 homozygotes during treatment.
The main effect of SLCO1B1 genotype was not associated with mean gametocytemia in the analysis (p = 0.086), nevertheless, a significant interaction effect between genotype and treatment over time (pFDR = 0.024) was observed.
SLCO1B1*14 carriers showed lower gametocytemia elimination rates compared with SLCO1B1*1a and SLCO1*1b allele carriers (on second day: *1a vs *14; p = 0.001 and *1b vs *14; p = 0.002). This model showed large effect sizes (d = 1.20) based on Cohen's interpretation scale.
Supplementary Material
Footnotes
Financial & competing interest disclosure
Financial support was provided by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Brazil). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
Ethical conduct of research
The authors state that they have obtained appropriate institutional review board approval or have followed the principles outlined in the Declaration of Helsinki for all human or animal experimental investigations. In addition, for investigations involving human subjects, informed consent has been obtained from the participants involved.
References
Papers of special note have been highlighted as: • of interest; •• of considerable interest
- 1.WHO. www.who.int/malaria/publications/world-malaria-report-2016/report/en/ World Malaria Report 2016.
- 2.Howes RE, Battle KE, Mendis KN, et al. Global epidemiology of Plasmodium vivax . Am. J. Trop. Med. Hyg. 2016;95(Suppl. 6):5–34. doi: 10.4269/ajtmh.16-0141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mueller I, Galinski MR, Baird JK, et al. Key gaps in the knowledge of Plasmodium vivax, a neglected human malaria parasite. Lancet Infect. Dis. 2009;9(9):555–566. doi: 10.1016/S1473-3099(09)70177-X. [DOI] [PubMed] [Google Scholar]
- 4.Ferreira MU, Castro MC. Challenges for malaria elimination in Brazil. Malar. J. 2016;15(1):284. doi: 10.1186/s12936-016-1335-1. [DOI] [PMC free article] [PubMed] [Google Scholar]; • Reviews major lessons learned from past and current malaria control policies in Brazil.
- 5.WHO. Guidelines for the Treatment of Malaria (3rd Edition) www.who.int/malaria/publications/atoz/9789241549127/en/ [Google Scholar]
- 6.Thomé R, Lopes SC, Costa FT, Verinaud L. Chloroquine: modes of action of an undervalued drug. Immunol. Lett. 2013;153(1–2):50–57. doi: 10.1016/j.imlet.2013.07.004. [DOI] [PubMed] [Google Scholar]
- 7.Vale N, Moreira R, Gomes P. Primaquine revisited six decades after its discovery. Eur. J. Med. Chem. 2009;44(3):937–953. doi: 10.1016/j.ejmech.2008.08.011. [DOI] [PubMed] [Google Scholar]
- 8.Baird JK. Resistance to therapies for infection by Plasmodium vivax . Clin. Microbiol. Rev. 2009;22(3):508–534. doi: 10.1128/CMR.00008-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hellgren U, Alván G, Jerling M. On the question of interindividual variations in chloroquine concentrations. Eur. J. Clin. Pharmacol. 1993;45(4):383–385. doi: 10.1007/BF00265960. [DOI] [PubMed] [Google Scholar]
- 10.Walker O, Dawodu AH, Adeyokunnu AA, Salako LA, Alvan G. Plasma chloroquine and desethylchloroquine concentrations in children during and after chloroquine treatment for malaria. Br. J. Clin. Pharmacol. 1983;16(6):701–705. doi: 10.1111/j.1365-2125.1983.tb02244.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dua VK, Gupta NC, Kar PK, et al. Chloroquine and desethylchloroquine concentrations in blood cells and plasma from Indian patients infected with sensitive or resistant Plasmodium falciparum . Ann. Trop. Med. Parasitol. 2000;94(6):565–570. doi: 10.1080/00034983.2000.11813579. [DOI] [PubMed] [Google Scholar]
- 12.Kim YR, Kuh HJ, Kim MY, et al. Pharmacokinetics of primaquine and carboxyprimaquine in Korean patients with vivax malaria. Arch. Pharm. Res. 2004;27(5):576–580. doi: 10.1007/BF02980134. [DOI] [PubMed] [Google Scholar]
- 13.Fletcher KA, Evans DA, Gilles HM, Greaves J, Bunnag D, Harinasuta T. Studies on the pharmacokinetics of primaquine. Bull. World Health Organ. 1981;59(3):407–412. [PMC free article] [PubMed] [Google Scholar]
- 14.Goller JL, Jolley D, Ringwald P, Biggs BA. Regional differences in the response of Plasmodium vivax malaria to primaquine as anti-relapse therapy. Am. J. Trop. Med. Hyg. 2007;76(2):203–207. [PubMed] [Google Scholar]
- 15.Pukrittayakamee S, Tarning J, Jittamala P, et al. Pharmacokinetic interactions between primaquine and chloroquine. Antimicrob. Agents Chemother. 2014;58(6):3354–3359. doi: 10.1128/AAC.02794-13. [DOI] [PMC free article] [PubMed] [Google Scholar]; • Overview of pharmacokinetic interactions between primaquine and chloroquine.
- 16.Höglund R, Moussavi Y, Ruengweerayut R, Cheomung A, Äbelö A, Na-Bangchang K. Population pharmacokinetics of a three-day chloroquine treatment in patients with Plasmodium vivax infection on the Thai-Myanmar border. Malar. J. 2016;29(15):129. doi: 10.1186/s12936-016-1181-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sortica VA, Lindenau JD, Cunha MG, et al. The effect of SNPs in CYP450 in chloroquine/primaquine Plasmodium vivax malaria treatment. Pharmacogenomics. 2016;17(17):1903–1911. doi: 10.2217/pgs-2016-0131. [DOI] [PMC free article] [PubMed] [Google Scholar]; •• Showing the role of CYP450 in malaria treatment.
- 18.Silvino AC, Costa GL, Araújo FC, et al. Variation in human cytochrome P-450 drug-metabolism genes: a gateway to the understanding of Plasmodium vivax relapses. PLoS ONE. 2016;11(7):e0160172. doi: 10.1371/journal.pone.0160172. [DOI] [PMC free article] [PubMed] [Google Scholar]; •• Showing the role of CYP450 in malaria relapses.
- 19.Kerb R, Fux R, Mörike K, et al. Pharmacogenetics of antimalarial drugs: effect on metabolism and transport. Lancet Infect. Dis. 2009;9(12):760–774. doi: 10.1016/S1473-3099(09)70320-2. [DOI] [PubMed] [Google Scholar]
- 20.Chen Z, Shi T, Zhang L, et al. Mammalian drug efflux transporters of the ATP binding cassette (ABC) family in multidrug resistance: a review of the past decade. Cancer Lett. 2016;370(1):153–164. doi: 10.1016/j.canlet.2015.10.010. [DOI] [PubMed] [Google Scholar]
- 21.Hong M. Biochemical studies on the structure-function relationship of major drug transporters in the ATP-binding cassette family and solute carrier family. Adv. Drug Deliv. Rev. 2016 doi: 10.1016/j.addr.2016.06.003. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 22.Zhou F, Zhu L, Wang K, Murray M. Recent advance in the pharmacogenomics of human solute carrier transporters (SLCs) in drug disposition. Adv. Drug Deliv. Rev. 2016 doi: 10.1016/j.addr.2016.06.004. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 23.Vezmar M, Georges E. Direct binding of chloroquine to the multidrug resistance protein (MRP): possible role for MRP in chloroquine drug transport and resistance in tumor cells. Biochem. Pharmacol. 1998;56(6):733–742. doi: 10.1016/s0006-2952(98)00217-2. [DOI] [PubMed] [Google Scholar]
- 24.Oerlemans R, van der Heijden J, Vink J, et al. Acquired resistance to chloroquine in human CEM T cells is mediated by multidrug resistance-associated protein 1 and provokes high levels of crossresistance to glucocorticoids. Arthritis Rheum. 2006;54(2):557–568. doi: 10.1002/art.21569. [DOI] [PubMed] [Google Scholar]
- 25.Crowe A, Ilett KF, Karunajeewa HA, Batty KT, Davis TME. Role of P glycoprotein in absorption of novel antimalarial drugs. Antimicrob. Agents Chemother. 2006;50(10):3504–3506. doi: 10.1128/AAC.00708-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hayeshi R, Masimirembwa C, Mukanganyama S, Ungell AL. The potential inhibitory effect of antiparasitic drugs and natural products on P-glycoprotein mediated efflux. Eur. J. Pharm. Sci. 2006;29(1):70–81. doi: 10.1016/j.ejps.2006.05.009. [DOI] [PubMed] [Google Scholar]
- 27.Alam K, Pahwa S, Wang X, et al. Downregulation of organic anion transporting polypeptide (OATP) 1b1 transport function by lysosomotropic drug chloroquine: implication in OATP-mediated drug–drug interactions. Mol. Pharm. 2016;13(3):839–851. doi: 10.1021/acs.molpharmaceut.5b00763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hubeny A, Keiser M, Oswald S, et al. Expression of organic anion transporting polypeptide 1A2 in red blood cells and its potential impact on antimalarial therapy. Drug Metab. Dispos. 2016;44(10):1562–1568. doi: 10.1124/dmd.116.069807. [DOI] [PubMed] [Google Scholar]
- 29.Brazilian Ministry of Health. Epidemiological bulletin n43. 2015. http://portalsaude.saude.gov.br/
- 30.Brazilian Ministry of Health. Manual of Malaria laboratory diagnosis. 2009. http://bvsms.saude.gov.br/bvs/publicacoes/manual_diagnostico_laboratorial_malaria_2ed.pdf
- 31.Sambrook J, Fritsch EF, Maniatis T. Molecular Cloning: A Laboratory Manual (2nd Edition) Cold Spring Harbor Laboratory Press; NY, USA: 1989. [Google Scholar]
- 32.Stephens M, Donnelly P. A comparison of Bayesian methods for haplotype reconstruction from population genotype data. Am. J. Hum. Genet. 2003;73(5):1162–1169. doi: 10.1086/379378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sortica VA, Cunha MG, Ohnishi MD, et al. IL1B, IL4R, IL12RB1 and TNF gene polymorphisms are associated with Plasmodium vivax malaria in Brazil. Malar. J. 2012;11:409. doi: 10.1186/1475-2875-11-409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Liang K-Y, Zeger SL. Longitudinal data analysis using generalized linear models. Biometrika. 1986;73(1):13–22. [Google Scholar]
- 35.Fritz CO, Morris PE, Richler JJ. Effect size estimates: current use, calculations, and interpretation. J. Exp. Psychol. Gen. 2012;141(1):2–18. doi: 10.1037/a0024338. [DOI] [PubMed] [Google Scholar]
- 36.Sortica Vde A, Ojopi EB, Genro JP, et al. Influence of genomic ancestry on the distribution of SLCO1B1, SLCO1B3 and ABCB1 gene polymorphisms among Brazilians. Basic Clin. Pharmacol. Toxicol. 2012;110(5):460–468. doi: 10.1111/j.1742-7843.2011.00838.x. [DOI] [PubMed] [Google Scholar]
- 37.Cohen J, Cohen P, West SG, Aiken LS. Applied Multiple Regression/Correlation Analysis for the Behavioral Science. Erlbaum; NJ, USA: 2002. [Google Scholar]
- 38.Wirjanata G, Sebayang BF, Chalfein F, et al. Contrasting ex vivo efficacies of “reversed chloroquine” compounds in chloroquine-resistant Plasmodium falciparum and P. vivax isolates. J. Antimicrob. Agents Chemother. 2015;59(9):5721–5726. doi: 10.1128/AAC.01048-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sharrock WW, Suwanarusk R, Lek-Uthai U, et al. Plasmodium vivax trophozoites insensitive to chloroquine. Malar. J. 2008;7:94. doi: 10.1186/1475-2875-7-94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Siqueira AM, Alencar AC, Melo GC, et al. Fixed-dose artesunate–amodiaquine combination vs chloroquine for treatment of uncomplicated blood stage P. vivax infection in the Brazilian Amazon: an open-label randomized, controlled trial. Clin. Infect. Dis. 2017;64(2):166–174. doi: 10.1093/cid/ciw706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Price RN, Auburn S, Marfurt J, Cheng Q. Phenotypic and genotypic characterization of drug-resistant Plasmodium vivax . Trends Parasitol. 2012;28:522–529. doi: 10.1016/j.pt.2012.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Gonçalves LA, Cravo P, Ferreira MU. Emerging Plasmodium vivax resistance to chloroquine in South America: an overview. Mem. Inst. Oswaldo Cruz. 2014;109(5):534–539. doi: 10.1590/0074-0276130579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hagenbuch B, Meier PJ. Organic anion transporting polypeptides of the OATP/SLC21 family: phylogenetic classification asOATP/SLCO superfamily, new nomenclature and molecular/functional properties. Pflugers Arch. 2004;447(5):653–665. doi: 10.1007/s00424-003-1168-y. [DOI] [PubMed] [Google Scholar]
- 44.Kalliokoski A, Niemi M. Impact of OATP transporters on pharmacokinetics. Br. J. Pharmacol. 2009;158(3):693–705. doi: 10.1111/j.1476-5381.2009.00430.x. [DOI] [PMC free article] [PubMed] [Google Scholar]; •• Compiles the knowledge about the expression and function of human OATP transporters, their substrate and inhibitor specificities, as well as pharmacogenetics.
- 45.Kullak-Ublick GA, Ismair MG, Stieger B, et al. Organic anion-transporting polypeptide B (OATP-B) and its functional comparison with three other OATPs of human liver. Gastroenterology. 2001;120(2):525–533. doi: 10.1053/gast.2001.21176. [DOI] [PubMed] [Google Scholar]
- 46.Kobayashi D, Nozawa T, Imai K, Nezu J, Tsuji A, Tamai I. Involvement of human organic anion transporting polypeptide OATP-B (SLC21A9) in pH-dependent transport across intestinal apical membrane. J. Pharmacol. Exp. Ther. 2003;306(2):703–708. doi: 10.1124/jpet.103.051300. [DOI] [PubMed] [Google Scholar]
- 47.Ho RH, Tirona RG, Leake BF, et al. Drug and bile acid transporters in rosuvastatin hepatic uptake: function, expression, and pharmacogenetics. Gastroenterology. 2006;130(6):1793–1806. doi: 10.1053/j.gastro.2006.02.034. [DOI] [PubMed] [Google Scholar]
- 48.Niemi M. Role of OATP transporters in the disposition of drugs. Pharmacogenomics. 2007;8(7):787–802. doi: 10.2217/14622416.8.7.787. [DOI] [PubMed] [Google Scholar]; • Provides an up-to-date review of human OATPs and their substrates, and a current compilation of their DNA sequence variations.
- 49.Pasanen MK, Neuvonen M, Neuvonen PJ, Niemi M. SLCO1B1 polymorphism markedly affects the pharmacokinetics of simvastatin acid. Pharmacogenet. Genomics. 2006;16(12):873–979. doi: 10.1097/01.fpc.0000230416.82349.90. [DOI] [PubMed] [Google Scholar]
- 50.Couvert P, Giral P, Dejager S, et al. Association between a frequent allele of the gene encoding OATP1B1 and enhanced LDL-lowering response to fluvastatin therapy. Pharmacogenomics. 2008;9(9):1217–1227. doi: 10.2217/14622416.9.9.1217. [DOI] [PubMed] [Google Scholar]
- 51.Romaine SP, Bailey KM, Hall AS, Balmforth AJ. The influence of SLCO1B1 (OATP1B1) gene polymorphisms on response to statin therapy. Pharmacogenomics J. 2010;10(1):1–11. doi: 10.1038/tpj.2009.54. [DOI] [PubMed] [Google Scholar]
- 52.Sortica VA, Fiegenbaum M, Lima LO, et al. SLCO1B1 gene variability influences lipid-lowering efficacy on simvastatin therapy in Southern Brazilians. Clin. Chem. Lab. Med. 2012;50(3):441–448. doi: 10.1515/cclm.2011.804. [DOI] [PubMed] [Google Scholar]
- 53.Michalski C, Cui Y, Nies AT, et al. A naturally occurring mutation in the SLC21A6 gene causing impaired membrane localisation of the hepatocyte uptake transporter. J. Biol. Chem. 2002;277(45):43058–43063. doi: 10.1074/jbc.M207735200. [DOI] [PubMed] [Google Scholar]
- 54.Kullak-Ublick GA, Fisch T, Oswald M, et al. Dehydroepiandrosterone sulfate (DHEAS): identification of a carrier protein in human liver and brain. FEBS Lett. 1998;424(3):173–176. doi: 10.1016/s0014-5793(98)00168-9. [DOI] [PubMed] [Google Scholar]
- 55.Lee W, Glaeser H, Smith LH, et al. Polymorphisms in human organic aniontransporting polypeptide 1A2 (OATP1A2): implications for altered drug disposition and central nervous system drug entry. J. Biol. Chem. 2005;280(10):9610–9617. doi: 10.1074/jbc.M411092200. [DOI] [PubMed] [Google Scholar]
- 56.Franke RM, Scherkenbach LA, Sparreboom A. Pharmacogenetics of the organic anion transporting polypeptide 1A2. Pharmacogenomics. 2009;10(3):339–344. doi: 10.2217/14622416.10.3.339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Gong IY, Kim RB. Impact of genetic variation in OATP transporters to drug disposition and response. Drug Metab. Pharmacokinet. 2013;28(1):4–18. doi: 10.2133/dmpk.dmpk-12-rv-099. [DOI] [PubMed] [Google Scholar]; • Addresses the present evidence of relevance of genetic variability in OATP1B1, OATP1B3, OATP2B1 and OATP1A2 in terms of drug response, efficacy and optimal therapeutics.
- 58.Zhou Y1, Yuan J, Li Z, et al. Genetic polymorphisms and function of the organic anion-transporting polypeptide 1A2 and its clinical relevance in drug disposition. Pharmacology. 2015;95(3–4):201–208. doi: 10.1159/000381313. [DOI] [PubMed] [Google Scholar]
- 59.Badagnani I, Castro RA, Taylor TR, et al. Interaction of methotrexate with organic-anion transporting polypeptide 1A2 and its genetic variants. J. Pharmacol. Exp. Ther. 2006;318(2):521–529. doi: 10.1124/jpet.106.104364. [DOI] [PubMed] [Google Scholar]
- 60.Deeley RG, Westlake C, Cole SP. Transmembrane transport of endo- and xenobiotics by mammalian ATP-binding cassette multidrug resistance proteins. Physiol. Rev. 2006;86(3):849–899. doi: 10.1152/physrev.00035.2005. [DOI] [PubMed] [Google Scholar]
- 61.Fung KL, Gottesman MM. A synonymous polymorphism in a common MDR1 (ABCB1) haplotype shapes protein function. Biochim. Biophys. Acta. 2009;1794(5):860–871. doi: 10.1016/j.bbapap.2009.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.de Santana Filho FS, Arcanjo AR, Chehuan YM, et al. Chloroquine-resistant Plasmodium vivax, Brazilian Amazon. Emerg. Infect. Dis. 2007;13(7):1125–1126. doi: 10.3201/eid1307.061386. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.