Abstract
Alcohol impairs resolution of respiratory viral infections. Numerous immune response pathways are altered in response to alcohol misuse, including alcohol-induced ciliary dysfunction in the lung. We hypothesized that mucociliary clearance-mediated innate immunity to respiratory syncytial virus (RSV) would be compromised by alcohol exposure. Cilia were assayed using Sisson-Ammons Video Analysis by quantitating the average number of motile points in multiple whole field measurements of mouse tracheal epithelial cells grown on air-liquid interface. Pretreatment with ethanol alone (100 mM for 24 hr) had no effect on the number of motile cilia. A single dose (TCID50 1 × 105) of RSV resulted in a significant (p<0.05) decrease in motile cilia after 2 days. Ethanol pretreatment significantly (p<0.05) potentiated RSV-induced cilia loss by 2 days. Combined RSV and ethanol treatment led to a sustained activation-induced auto-downregulation of PKC epsilon (PKCε). Ethanol-induced enhancement of ciliated cell detachment was confirmed by dynein ELISA and LDH activity from the supernates. RSV-induced cilia loss was evident until 7 days, when RSV-only infected cells demonstrated no significant cilia loss vs. control cells. However, cells pretreated with ethanol showed significant cilia loss until 10 days post-RSV infection. To address the functional significance of ethanol-enhanced cilia detachment, mice fed alcohol ad libitum (20% for 12 wk) were infected once with RSV and clearance measured by plaque forming assay from lung homogenates for up to 7 days. After 3 days, RSV plaque formation was no longer detected from lung of control mice, while significant (p<0.01) RSV plaque forming units were detected at 7 days in alcohol-fed mice. Alcohol-fed mice demonstrated enhanced cilia loss and delayed cilia recovery from tracheal measurements in wild type C57BL/6 mice, but not PKCε KO mice. These data suggest that alcohol worsens RSV-mediated injury to ciliated epithelium in a PKCε-dependent manner.
Keywords: RSV Respiratory Syncytial Virus, CBF Ciliary Beat Frequency, MTEC Mouse Tracheal Epithelial Cell, PKC Protein Kinase C
Introduction
Alcohol misuse has long been an established contributor to pneumonia deaths (Schmidt and De Lint, 1972). Adults average 2–4 viral upper respiratory infections (Monto et al., 2001; Turner, 1998) while children average 3–8 viral upper respiratory infections (Rosenstein et al., 1998) each year. Viral upper respiratory infections are associated with 20 million days of work missed, 22 million school absences, and 45 million days bedridden (Adams, 1999). Approximately 25 million office visits to primary care providers for upper respiratory infections take place each year (Gonzales et al., 2001) with an associated cost in excess of ~$25 billion annually (Fendrick et al., 2001).
Viral-induced lung injury is a significant public health problem as respiratory viral infections cause ~60% of chronic lung disease exacerbations. Respiratory viral infections directly infect the bronchial airways leading to activation of host innate immunity. Typically recognized as life threatening in newborns, respiratory syncytial virus (RSV) is now recognized as a serious pathogen in the aging population (65+ years old) (Falsey et al., 2005). Worldwide, RSV leads to more than 64 million infections and 160,000 deaths each year with no effective vaccines or anti-viral therapies currently available.
The pathogenesis of RSV lung infection of the lungs specifically impacts the airways and is characterized by ciliated cell infection and detachment, airway lumen debris accumulation, and multi-nucleated syncytia formation (Johnson et al., 2007). The primary cellular target of RSV infection is the ciliated airway epithelial cell (Pickles, 2013). RSV infection profoundly induced airway inflammation, resulting in airway hyperesponsiveness, mucus hypersecretion, plasma leakage, and inflammatory cell recruitment (Gern and Busse, 2000).
Mucociliary clearance is the mechanical component of innate defense to inhaled pathogens orchestrated by the airway epithelium. Mucus is secreted to trap inhaled particles, pathogens and toxins while cilia move in a “whip-like” manner through the airway surface liquid to propel trapped particles and pathogens out of the lung. Importantly, cilia beating can speed up or slow down in response to their environment. RSV infection leads to a rapid decrease in CBF (Sisson et al., 1994; Tristram et al., 1998b). Infection subsequently results in the loss of cilia and selective detachment of ciliated cells (Fishaut et al., 1978). This detachment is likely related to the fact that ciliated epithelial cells attach to basal cells via desmosomes (Evans et al., 1989).
Agents that slow cilia beating (organic dusts, aldehydes, bisulfites, neuropeptides, and cigarette smoke) share a common theme: protein kinase C epsilon (PKCε) activation (Wyatt et al., 2012). RSV has also been shown to activate PKC and PKCε (Monick et al., 2001). Sustained PKCε activation eventually results in the auto-downregulation of the kinase (Chen, 1993). Down-regulation of PKC activity is functionally equivalent to PKC inhibition. We have previously shown that PKCε activation is a prerequisite to ciliary slowing and that inhibition of PKCε results in ciliated cell detachment in vitro (Wyatt et al., 2012). This PKCε-mediated slowing and subsequent detachment response was observed when airway epithelial cells were co-exposed to cigarette smoke and alcohol. Alcohol alters cilia beating and lung host defense (Sapkota and Wyatt, 2015). Chronic alcohol exposure results in the uncoupling of cilio-stimulatory pathways (Wyatt and Sisson, 2001). Post-viral pneumonia occurs with a greater frequency in patients with alcoholism (Ruben and Cate, 1987). Based on these observations, we hypothesized that alcohol co-exposure enhances RSV infection injury by compromising cilia-mediated viral clearance.
Materials and methods
Mouse tracheal epithelial cells grown at air-liquid interface
C57BL/6J female mice (6–8 weeks of age) were euthanized using CO2 inhalation, the tracheas were removed, opened lengthwise and placed in a 1.5 mg/ml solution of pronase (Sigma-Aldrich, St. Louis, MO) for 18–24 hr. The pronase was neutralized with 10% fetal bovine serum (FBS). The tracheas were rinsed in 10 ml of Ham’s F-12 medium containing 10% FBS. The tracheas were removed and the solutions centrifuged to collect the epithelial cells. Differential adherence was performed by plating cells onto an uncoated 60 mm polystyrene plastic culture dish in MTEC basic media with 10% FBS. The MTEC basic media consists of a 1:1 solution of Ham’s F-12 and DMEM supplemented with Pen/Strep (10,000 U/ml), Amphotericin B (250 μg/ml), Gentamicin (40 mg/ml), and glutamine (4 mM). The cells were incubated for 4 hr to remove adhered fibroblasts, the suspended cells washed and counted. Cells were plated on Type I collagen (Becton Dickinson, Bedford, MA) coated membrane inserts, and grown submerged in MTEC supplementary media which consists of basic media supplemented with insulin, transferrin, EGF, BPE, cholera toxin, retinoic acid and 5% FBS (Navarette et al., 2012). By the 6th day, the cells were confluent and cultured at air-liquid interface (ALI). At this point, cells were fed basally only with MTEC basic media supplemented with 10% NuSerum (Fisher, Waltham, MA) and retinoic acid until fully ciliated. Ciliated MTEC were pretreated basally with 100 mM ethanol for 24 hr or apically with 1 × 105 TCID50 RSV, as per experimental condition.
Mice and chronic alcohol feeding
Female BALB/c mice, between 6–8 wk of age, were purchased from Jackson Laboratory (Bar Harbor, ME). Mice were acclimated for a minimum of 1 wk prior to starting the experimental protocol. Animals were randomly assigned to ethanol feeding or water feeding as control. We utilized the Cook model of ethanol feeding (Cook et al., 2004; Cook et al., 2007), whereby mice acclimated to ethanol feeding by gradually increasing the concentration of ethanol in drinking water over a 1 wk period (10% ethanol [w/v] in sterile water for 2–3 days, followed by 15% [w/v] in sterile water for two additional days, followed by 18% ethanol [w/v] for the remainder of the experiment) (Elliott et al., 2007). No significant weight loss was observed due to alcohol feeding or RSV infection. For in vitro MTEC assays, transgenic PKCε knockout mice (B6;129S4-Prkcetm1Msg/J and B6;129X1-Prkcdtm1Msg/J on C57Bl/6J background) were sourced from Jackson, bred, and genotyped at UNMC. All animal experiments were conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. In addition, animal experiments were completed with the approval and oversight of the University of Nebraska Medical Center’s Institutional Animal Care and Use Committee.
RSV infection
Mice from ethanol-feeding and control-feeding groups were randomly chosen to receive an RSV inoculation. Mice were lightly anesthetized with isoflurane and intranasally instilled with RSV (Advanced Biotechnologies, Eldersburg, MD) diluted in 50 μL of sterile saline, at 3,250 TCID50 U per mouse. Mock-infected mice were similarly anesthetized and instilled with saline as vehicle control. Body weight was monitored to assure that no more than 20% of initial starting body weight was lost during the infection period. Subsets of mice were euthanized at days 1, 2, 3, and 7 post-inoculation.
TCID50/mL method – viral infectivity assay
Murine lungs were weighed and homogenized in Earl’s Minimum Essential Medium (EMEM) (ATCC, Manassas, VA) for the viral infectivity assay. Briefly, human epithelial type 2 (HEp-2) cells (ATCC) were sub-cultured into 96-well plates (Corning, Pittsburgh, PA) at a rate of 5 × 105 cells per well in EMEM supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin (Life Technologies, Grand Island, NY). Upon reaching 85–90% confluence, cell layers were washed with phosphate-buffered saline (PBS), and then inoculated with 10-fold dilutions of mouse lung homogenates. After a 2-h incubation at 37 °C in a 5% CO2 incubator, the lung homogenates were removed by aspiration. Serum-free EMEM was added to all the wells, and plates were incubated for up to 7 days at 37 °C, 5% CO2. Each well was examined for viral infection, and quantitation of virus was determined using the Reed-Munich equation (Warren et al., 2015).
Cilia beat frequency- Whole field analysis
Ciliary beat frequency (CBF) analysis and measurement of the total number of motile points during whole field analysis was accomplished using the Sisson Ammons Video Analysis method as previously validated and characterized (Sisson et al., 2003).
Protein kinase C activity assay
Determination of PKC isoform catalytic activity was accomplished in fractionates from axonemes, cells, and tissues by direct isoform-specific substrate peptide phosphorylation assays as previously described (Wyatt et al., 2010).
Dynein ELISA
Detached ciliated cells and shed cilia released into the media supernatant from exposure conditions were quantitated by dynein ELISA using an antibody to ciliary 13S dynein protein as previously characterized (Sisson et al., 1994).
Cell viability assay
Cell and tissue viability was determined by the lactate dehydrogenase (LDH) activity assay (Sigma, St. Louis, MO). No significant LDH release was detected in cells under any treatment condition.
Statistical methods
Data are representative of three separate experiments and presented as mean ± SEM. Statistical analysis was conducted using GraphPad Prism Version 6 (San Diego, CA). Statistical significance was determined by Student’s t test for studies determining differences between RSV and RSV+EtOH alone where measured outcome was not detected in the non-infected (CTRL) or ethanol-treated (EtOH). For all other analyses, one-way ANOVAs were used to determine differences among treatment groups and a Bonferroni post hoc analysis was used to account for multiple comparisons if the p value was <0.05. In all analyses, p values less than 0.05 were considered statistically significant.
Results
The combination of alcohol and RSV potentiates cilia slowing, coincident with increased PKC epsilon activity in mouse tracheal epithelial cell cultures.
Because RSV slows cilia beating (Sisson et al., 1994) and activates PKCε (Monick et al., 2001) in airway epithelial cells, we investigated the effect of alcohol exposure on RSV-infected ciliated epithelial cells in culture with regard to these responses. Wild-type mouse tracheal epithelial cells (MTEC) were isolated and cultured on air-liquid interface to produce confluent monolayers with a ciliated phenotype. Cells were then treated with or without 1 × 105 TCID50 of RSV in culture media for 1–6 hr. CBF was measured, media supernates collected, and cell monolayers harvested for PKCε activity assay. As expected, RSV alone significantly (ap<0.05 vs media at matched time point) slowed CBF by 3 hr exposure and decreased cilia beating was observed relative to media control up to 6 hr (Figure 1A). However, 24 hr pre-treatment with 100 mM alcohol resulted in both an earlier (1 hr) RSV-induced CBF decrease compared to media control (cp<0.01 vs media at matched time point) as well as a significantly greater decrease in the magnitude of cilia slowing compared to RSV only-treated cells by 3–6 hr (bp<0.05 vs RSV only at matched time point). Complete ciliostasis occurred at 6 hr whereby the time course was halted. As previously reported (Wyatt and Sisson, 2001), alcohol alone (1 hr) increased CBF, but rapidly returned to baseline levels with no CBF slowing observed relative to media control.
Fig. 1. The combination of alcohol and RSV potentiates cilia slowing coincident with PKC epsilon action in mouse tracheal epithelial cell cultures.

Ciliated mouse tracheal epithelial cells (MTEC) cultured at air-liquid interface and pretreated 24 hr with or without 100 mM alcohol (EtOH) were infected with 1 × 105 TCID50 respiratory syncytial virus (RSV) for 1–6 hr. Control cells received no treatment (Media). A) Ciliary beat frequency (CBF) decreased in RSV-treated MTEC (ap<0.05 vs Media at matched time points of 3–6 hr) while EtOH+RSV further enhanced cilia slowing due to RSV only (bp<0.05 vs RSV only at matched time points of 3–6 hr). EtOH+RSV potentiated the time of RSV-induced CBF decrease compared to media control (cp<0.01 vs Media at 1 hr). B) Protein kinase C epsilon (PKCε) activity was increased by RSV (ap<0.05 vs Media at 1 hr), enhanced by EtOH+RSV (bp<0.01 vs Media at matched time points of 1–2 hr), and significantly decreased with EtOH+RSV (cp<0.05 vs Media or RSV only at matched time points of 5–6 hr). Experiments were performed independently three times each using triplicate data measurements (n=9).
In contrast, RSV infection increased PKCε activity in media-treated cells by 1 hr (ap<0.05 vs media; Figure 1B). Pre-treatment with alcohol resulted in an enhancement of magnitude and duration of RSV-stimulated PKCε activity (bp<0.01 vs media at matched time points). PKCε, however, quickly declined after 2 hr, and a significant decrease from even baseline kinase activity was observed by 5 hr (cp<0.05 vs media at matched time points). No change in baseline PKCε activity was observed under media control or alcohol-only treatment conditions.
The combination of alcohol and RSV results in enhanced cilia loss, ciliated cell detachment, and cell death in mouse tracheal epithelial cell cultures.
RSV infection of ciliated epithelium causes cilia shedding, ciliated cell detachment, and cell death (Villenave et al., 2013). To determine the effect of alcohol treatment on these known responses to RSV, we measured the amount of cilia and LDH released into the culture supernates after exposure to RSV in cells pre-treated with or without alcohol. Quantitative ELISA for cilia axoneme dynein revealed that a significant (ap<0.05 vs media) amount of mouse tracheal epithelial cilia were shed into the culture supernate in cells treated with 1 × 105 TCID50 RSV for 6 hr as compared to media control-treated cells (Figure 2A). Although alcohol treatment alone for 24 hr resulted in no increase in supernate dynein, supernates from cells pretreated with alcohol followed by RSV demonstrated a significant (bp<0.001 vs RSV only) increase in the amount of cilia-specific protein detected. Similarly, RSV infection resulted in a significant (ap<0.005 vs media) increase in LDH release into the supernatant media and this release was significantly increased (bp<0.02 vs RSV only) in those cells pre-treated with alcohol (Figure 2B). Importantly, no cell death or LDH release was observed in 100 mM alcohol-treated cells at any time point.
Fig. 2. The combination of alcohol and RSV results in worsened cilia loss and cell death in mouse tracheal epithelial cell cultures.
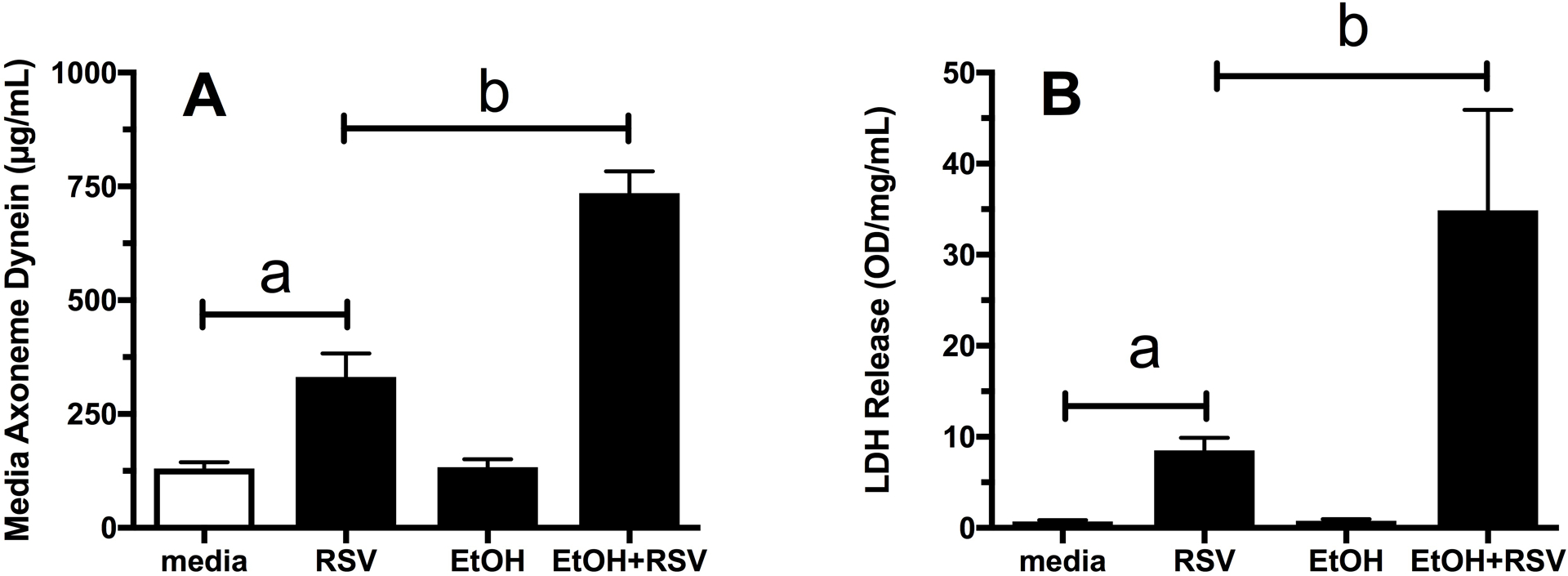
RSV-induced damage to ciliated cells was quantitated by dynein ELISA and LDH release. A) RSV infection resulted in increased axonemal dynein in the culture media by 6 hr (ap<0.05 vs Media) while 24 hr pretreatment with EtOH increased RSV-induced cilia loss (bp<0.001 vs RSV only). B) RSV infection increased LDH release (ap<0.005 vs Media) and this release was enhanced in EtOH pre-treated MTEC (bp<0.02 vs RSV only). Experiments were performed independently three times each using triplicate data measurements (n=9).
Alcohol feeding in mice potentiates RSV-induced cilia slowing and cilia loss and delays recovery of the ciliated epithelium.
To determine the in vivo relevancy of the alcohol potentiation effect on cilia slowing and detachment, alcohol-fed mice were infected with 3,250 TCID50 RSV and sacrificed at 1, 2, 3, and 7 days whereupon tracheal cilia were examined. In mice that consumed water or alcohol ad libitum for 8–10 wk (weeks), no changes were observed in baseline CBF of tracheal ring cilia (Figure 3A). Compared to control, non-infected mice, RSV-infected mice demonstrated a significant (ap<0.05 vs Control mice) decrease in CBF by 24 hr, which recovered to baseline levels by day 2. Importantly, the RSV-mediated decrease in CBF failed to show any recovery in the alcohol-fed mice, even at 1 wk. By wk 2, a return to baseline was observed (data not shown). In addition, alcohol had a profound effect upon cilia loss in vivo. By 48 hr post-RSV infection, a significant loss (bp<0.05 vs Control mice) of motile cilia was observed (Figure 3B) in both RSV and RSV+EtOH trachea. Recovery of motile cilia was detected 1 wk after RSV infection. However, the significant (ap<0.05 vs Control mice) loss of motile cilia was detected 24 hr earlier in alcohol-treated RSV-infected mice. In addition, no recovery in motile points was detected in the alcohol-fed infected mice.
Fig. 3. Alcohol feeding in mice potentiates and sustains RSV-induced cilia slowing and cilia loss and delays recovery of the ciliated epithelium.

Female BALB/c mice were ethanol-fed or water-fed for 8 wks prior to RSV infection (3,250 TCID50 U/mouse) and sacrificed 1–7 days post infection. A) CBF decreased in tracheal epithelium of RSV-infected mice (ap<0.05 vs Control mice) by 24 hr and recovered to baseline levels by day 2. In alcohol- (EtOH) fed mice, the RSV-mediated CBF decrease persisted through 1 wk (ap<0.05 vs Control and RSV only mice @1wk). B) RSV caused a loss of motile cilia (bp<0.05 vs Control mice at 48 hr) which recovered by 1 wk after infection. In alcohol-fed RSV-infected mice, cilia loss was detected 24 hr earlier and sustained longer than RSV only (ap<0.05 vs RSV only at 24 hr and 1 wk and Control mice at all time points). n=6–10 mice per group from replicated experiments.
Alcohol feeding in mice potentiates and sustains RSV-induced PKC epsilon auto-downregulation.
In these same mouse tracheal epithelial samples, PKCε activity was assayed. RSV infection resulted in significantly (bp<0.05 vs Control mice) decreased PKCε activity after 24 hr, but gradually returned to baseline by 3–7 d (Figure 4). The significant (ap<0.01 vs Control mice) decrease in PKCε activity below baseline levels after RSV infection remained in place by 1 wk in the alcohol-fed mice. Alcohol feeding alone had no effect on PKCε activity.
Fig. 4. Alcohol feeding in mice potentiates and sustains RSV-induced PKC epsilon auto-downregulation.

PKCε activity was assayed in the same mouse tracheal epithelial samples from in vivo-exposed mice shown in Fig.3. RSV infection decreased PKCε activity (bp<0.05 vs Control mice at 24–48 hr), but returned to baseline by day 3–7. Decreased PKCε activity below baseline levels (autodownregulation) after RSV infection persisted in EtOH-fed mice (ap<0.01 vs RSV only at 72 hr -1 wk and Control mice at all times). n=6–10 mice per group from replicated experiments.
Alcohol feeding in mice prevents normal clearance of RSV.
To determine the functional consequences of the alcohol and RSV-associated slowing of CBF and cilia loss observed in vivo, RSV titer was assayed in whole lung homogenates after infection in control- and alcohol-fed mice. RSV was detected from lung homogenates in similar amounts after 24 hr post-infection in control-fed and alcohol-fed mice (Figure 5). However, while RSV was cleared from the lungs of control-fed mice by 48 hr, a significant amount (ap<0.05 vs RSV-only mice at 2–7 d) of RSV was detected in the lungs of alcohol-fed mice for up to 1 wk. Virus was not detected in the lungs of alcohol-fed mice from 10–14 days after infection, and no virus was detected in the uninfected control animals (data not shown).
Fig. 5. Alcohol feeding in mice prevents normal clearance of RSV.

RSV titers were determined in lung homogenates (100 μg/mL) from 1–7 days post-infection in control- and alcohol-fed mice. RSV was equally detected in lung homogenates of control- and alcohol-fed mice at 24 hr post infection. RSV continued to be detected in the lungs of alcohol-fed mice out to 1 wk, compared to control-fed mice (ap<0.05 vs RSV only mice at 2–7 d). n=6–10 mice per group from replicated experiments.
The combination of alcohol feeding and RSV infection does not slow or detach cilia in MTECs from PKC epsilon knockout mice (PKCε-KO).
To demonstrate a role for PKCε in the enhancement of cilia slowing and cilia loss due to RSV and alcohol co-exposure, we examined mouse tracheal ciliated cells (MTEC) from PKCε knockout (PKCε-KO) mice. MTEC from wild type and PKCε-KO mice were cultured under air-liquid interface conditions and treated with 1 × 105 TCID50 RSV in the presence of 100 mM alcohol for 3–8 hrs, optimal experimental time points as determined above. A significant difference (ap<0.01 vs wild type mice) in cilia slowing and number of motile points was observed between wild type MTEC and MTEC from PKCε-KO by 3 hr (Figure 6). As a novel PKC isoform class control, no difference in cilia slowing was observed between MTEC made from wild type and PKC delta KO mice. Similarly, the reduction in cilia motile points after combined alcohol and RSV treatment observed in wild type and PKC delta KO cultures at 8 hr was absent in the PKCε KO mouse cells.
Fig. 6. The combination of alcohol feeding and RSV infection does not slow or detach cilia in MTECs from PKCε knockout mice.

MTEC from wild type and PKCε KO mice were cultured under air-liquid interface conditions and treated with 1 × 105 TCID50 RSV in the presence of 100 mM EtOH. Baseline CBF and the number of motile points were preserved in PKCε KO MTEC (ap<0.01 vs wild type or PKCδ KO mice) for both A) cilia slowing at 3 hr and B) number of motile points at 8 hr.
Discussion
RSV preferentially infects the airway epithelium, resulting in the formation of large, multi-nucleated syncytial cells and damage to the ciliated cells resulting in cilia slowing, the shedding of cilia, the replication of virus, and the detachment of ciliated cells from the basal epithelium (Villenave et al., 2013). In normal, healthy humans, repair and resolution from the infection takes place by 48 hr post-infection (Tristram et al., 1998a). Lack of a timely resolution to viral infection can result in a secondary bacterial infection due to weakened innate lung defense (Prasso and Deng, 2017). Because alcohol misuse has been associated with increased pneumonia for over two centuries (Green and Kass, 1965), we hypothesized that alcohol enhances the severity and duration of RSV infection.
In the present study using in vitro mouse tracheal epithelial cell cultures, we demonstrate that RSV initially causes a significant decrease in ciliary beating through the activation of PKCε. Alcohol enhances the magnitude of both cilia slowing and PKCε activation. This activation eventually results in the auto-downregulation of PKCε and subsequent increased ciliated cell detachment in the alcohol and RSV co-exposed cells. In mice fed alcohol in their drinking water then inoculated with RSV, cilia beating is significantly slowed, PKCε auto-downregulation is pronounced, ciliated cell loss is sustained, and RSV clearance is impaired versus those animals that are fed water only (Figure 7). The resultant functional consequence to this alcohol and RSV co-exposure is a prolonged period of time before complete clearance of virus from the mouse lungs. This enhanced RSV-mediated injury requires the presence of PKCε activation, and subsequent auto-inhibition response, as PKCε-KO mice appear to be protected against the enhanced injury due to alcohol. Importantly, it has already been suggested that the auto-inhibition of PKC can lead to desmosome disruption and cellular detachment (McHarg et al., 2014).
Fig. 7.

Model diagram for temporal regulation of cilia slowing by PKCε activation leading to subsequent ciliated cell detachment by PKCε auto-downregulation.
The auto-downregulation of PKC is functionally equivalent to inhibiting the kinase as shown by many earlier studies (Parker et al., 1995). Previously, we have reported that inhibition of PKCε using chemical inhibitors of the catalytic region of the enzyme, or inhibition through siRNA can result in the selective detachment of the ciliated epithelial cells (Slager et al., 2006). Furthermore, the activation of PKCε is associated with the slowing of the cilia (Salathe et al., 1993; Wong et al., 1998). Earlier reports by others examining RSV-induced cilia slowing also reported increased activation of PKCε (Monick et al., 2001). Because our previous alcohol and cigarette smoke co-exposure studies defined a unique role for PKCε activation leading to enhanced lung injury not observed under alcohol-only conditions (Wyatt et al., 2012), it is logical to ask whether alcohol could worsen RSV-mediated injury through enhanced PKCε signaling.
The impact of alcohol on host defense against RSV is clearly multifaceted and complicated. Human susceptibility to RSV infection is increased in those individuals who misuse alcohol (Simet and Sisson, 2015) and alcohol consumption during pregnancy is associated with increased severity of RSV infection in newborns (Libster et al., 2015). It has been suggested that alcohol-induced reductions in lung surfactant may play a role in increased susceptibility to RSV (Lazic et al., 2007). RSV infection is an increasingly recognized problem in older people. While RSV may be a self-limited upper respiratory infection in younger adults, in older people, it is more likely to progress into a pneumonia (Branche and Falsey, 2015). Older people with underlying heart or lung disease or immunocompromise such as heavy alcohol use are at highest risk of developing pneumonia and death (Falsey and Walsh, 2005). It is possible that PKCε plays a role in the pathogenicity of RSV with aging. PKCε is elevated in the airway epithelium with aging, which slows cilia beat frequency at baseline (Bailey et al., 2014). The elderly are known to have impaired mucociliary clearance (Ho et al., 2001; Incalzi et al., 1989; Proenca de Oliveira-Maul et al., 2013). This impairment could weaken the first line of defense against RSV infection. It is not known how alcohol misuse affects the risk of RSV infection in the elderly. However, alcohol use disorders in older people are continuing to increase (Han et al., 2017).
Alcohol-mediated alterations in interferon response and other cytokines are observed after RSV infection (Jerrells et al., 2007). In addition, we have also demonstrated that both early and late adaptive immunity to RSV infection are altered by chronic alcohol consumption in our mouse model (Warren et al., 2016). Furthermore, alcohol exposure impairs adaptive immune responses, including decreased numbers of virus-specific pulmonary CD8 T cells, a decreased size and frequency of pulmonary B cell foci, and reduced production of influenza-specific antibodies after viral infection (McGill et al., 2009). Indeed, a limitation of our study is translating our observations to the complex human immune system when using a murine model where strain, age, or sex differences may apply. Our findings in the present study add to the complexity of the altered response to RSV in the alcohol lung through the identification of early injury to the cilio-mechanical nature of innate lung defense.
Alcohol-mediated injury to lung defense may be of relevance to society in a manner other than human public health. Bovine RSV (bRSV) infection is a significant problem in cattle production on concentrated animal feeding operations (CAFOs). Similar to humans, cattle who contract bRSV can progress to a secondary bacterial lung infection leading to significant morbidity and mortality, thus negatively impacting the costs of meat production (Sacco et al., 2014). In recent years, it has become widespread practice to feed cattle wet, alcohol-containing corn byproducts discarded after the production of bioethanol (Weiss et al., 2017). Cattle are particularly susceptible to the effects of alcohol on their lung epithelium (Wyatt and Sisson, 2001). Chronic consumption of alcohol by cattle on feedlots could be one factor leading to the significant increase in bovine respiratory disease (i.e. “shipping fever”) observed during this time period. Further epidemiologic and laboratory research is required, however, to address this concern.
In conclusion, the consumption of alcohol results in a significant impairment of clearance of RSV from the lung. This is due, in part, by a significant alcohol-mediated slowing of the cilia in terms of both magnitude and duration as well as an alcohol-enhanced detachment of the ciliated cells and loss of cilia in response to RSV. These temporal processes are dependent upon the activation and auto-downregulation of PKCε as a mechanism of this injury. Future research should focus upon whether manipulation of PKCεin RSV-infected mice can produce a protective response or decreased magnitude of lung injury. Development of therapeutic approaches will be much more challenging as the PKC family of enzymes plays a pleiotropic role in cellular physiology.
Acknowledgments
Funding sources for this research are as follows: VA BX000728 (T.A.W.), NIAAA AA008769 (J.H.S.), NIAAA AA019859 (S.M.S.) and NIA AG0535553 (K.L.B.). TAW is the recipient of a Research Career Scientist Award (IK6 BX003781) from the Department of Veterans Affairs. The authors wish to acknowledge the editorial assistance from Ms. Lisa Chudomelka and expert assistance on culturing RSV provided by Dr. Steven Varga at the University of Iowa.
References
- Adams P (1999). Costs associated with VRIs in 1996. Vital Health Stat 10, 200. [Google Scholar]
- Bailey KL, Bonasera SJ, Wilderdyke M, Hanisch BW, Pavlik JA, DeVasure J, Robinson JE, Sisson JH, and Wyatt TA (2014). Aging causes a slowing in ciliary beat frequency, mediated by PKCepsilon. Am. J. Physiol. Lung Cell. Mol. Physiol 306, L584–589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Branche AR, and Falsey AR (2015). Respiratory syncytial virus infection in older adults: an under-recognized problem. Drugs Aging 32, 261–269. [DOI] [PubMed] [Google Scholar]
- Chen CC (1993). Protein kinase C alpha, delta, epsilon and zeta in C6 glioma cells. TPA induces translocation and down-regulation of conventional and new PKC isoforms but not atypical PKC zeta. FEBS Lett 332, 169–173. [DOI] [PubMed] [Google Scholar]
- Cook RT, Schlueter AJ, Coleman RA, Tygrett L, Ballas ZK, Jerrells TR, Nashelsky MB, Ray NB, Haugen TH, and Waldschmidt TJ (2007). Thymocytes, Pre‐B Cells, and Organ Changes in a Mouse Model of Chronic Ethanol Ingestion—Absence of Subset‐Specific Glucocorticoid‐Induced Immune Cell Loss. Alcoholism: Clinical and Experimental Research 31, 1746–1758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cook RT, Zhu X, Coleman RA, Ballas ZK, Waldschmidt TJ, Ray NB, LaBrecque DR, and Cook BL (2004). T-cell activation after chronic ethanol ingestion in mice. Alcohol 33, 175–181. [DOI] [PubMed] [Google Scholar]
- Elliott MK, Sisson JH, and Wyatt TA (2007). Effects of cigarette smoke and alcohol on ciliated tracheal epithelium and inflammatory cell recruitment. Am J Respir Cell Mol Biol 36, 452–459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans MJ, Cox RA, Shami SG, Wilson B, and Plopper CG (1989). The role of basal cells in attachment of columnar cells to the basal lamina of the trachea. Am. J. Respir. Cell Mol. Biol 1, 463–469. [DOI] [PubMed] [Google Scholar]
- Falsey AR, Hennessey PA, Formica MA, Cox C, and Walsh EE (2005). Respiratory syncytial virus infection in elderly and high-risk adults. N Engl J Med 352, 1749–1759. [DOI] [PubMed] [Google Scholar]
- Falsey AR, and Walsh EE (2005). Respiratory syncytial virus infection in elderly adults. Drugs Aging 22, 577–587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fendrick AM, Sarnes MW, Nightengale B, and Monto AS (2001). IN2: The economic burden of viral respiratory infection in the United States. Value in Health 4, 412. [DOI] [PubMed] [Google Scholar]
- Fishaut M, Schwartzman JD, McIntosh K, and Mostow SR (1978). Behavior of respiratory syncytial virus in piglet tracheal organ culture. J Infect Dis 138, 644–649. [DOI] [PubMed] [Google Scholar]
- Gern JE, and Busse WW (2000). The role of viral infections in the natural history of asthma. J Allergy Clin Immunol 106, 201–212. [DOI] [PubMed] [Google Scholar]
- Gonzales R, Malone DC, Maselli JH, and Sande MA (2001). Excessive antibiotic use for acute respiratory infections in the United States. Clin Infect Dis 33, 757–762. [DOI] [PubMed] [Google Scholar]
- Green GM, and Kass EH (1965). The influence of bacterial species on pulmonary resistance to infection in mice subjected to hypoxia, cold stress, and ethanolic intoxication. Br. J. Exp. Pathol 46, 360–366. [PMC free article] [PubMed] [Google Scholar]
- Han BH, Moore AA, Sherman S, Keyes KM, and Palamar JJ (2017). Demographic trends of binge alcohol use and alcohol use disorders among older adults in the United States, 2005–2014. Drug Alcohol Depend 170, 198–207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho JC, Chan KN, Hu WH, Lam WK, Zheng L, Tipoe GL, Sun J, Leung R, and Tsang KW (2001). The effect of aging on nasal mucociliary clearance, beat frequency, and ultrastructure of respiratory cilia. American Journal of Respiratory and Critical Care Medicine 163, 983–988. [DOI] [PubMed] [Google Scholar]
- Incalzi RA, Maini CL, Fuso L, Giordano A, Carbonin PU, and Galli G (1989). Effects of aging on mucociliary clearance. Compr. Gerontol. A 3 Suppl, 65–68. [PubMed] [Google Scholar]
- Jerrells TR, Pavlik JA, Devasure J, Vidlak D, Costello A, Strachota JM, and Wyatt TA (2007). Association of chronic alcohol consumption and increased susceptibility to and pathogenic effects of pulmonary infection with respiratory syncytial virus in mice. Alcohol 41, 357–369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson JE, Gonzales RA, Olson SJ, Wright PF, and Graham BS (2007). The histopathology of fatal untreated human respiratory syncytial virus infection. Modern Pathology 20, 108. [DOI] [PubMed] [Google Scholar]
- Lazic T, Wyatt TA, Matic M, Meyerholz DK, Grubor B, Gallup JM, Kersting KW, Imerman PM, Almeida-De-Macedo M, and Ackermann MR (2007). Maternal alcohol ingestion reduces surfactant protein A expression by preterm fetal lung epithelia. Alcohol 41, 347–355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Libster R, M Ferolla F, Hijano DR, Acosta PL, Erviti A, and Polack FP (2015). Alcohol during pregnancy worsens acute respiratory infections in children. Acta Paediatrica 104, e494–499. [DOI] [PubMed] [Google Scholar]
- McGill J, Meyerholz DK, Edsen-Moore M, Young B, Coleman RA, Schluetter AJ, Waldschmidt TJ, Cook RT, and Legge KL (2009). Fetal exposure to ethanol has long-term effects on the severity of influenza virus infections. J of Immunology 182, 7803–7808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McHarg S, Hopkins G, Lim L, and Garrod D (2014). Down-regulation of desmosomes in cultured cells: the roles of PKC, microtubules and lysosomal/proteasomal degradation. PloS One 9, e108570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monick M, Staber J, Thomas K, and Hunninghake G (2001). Respiratory syncytial virus infection results in activation of multiple protein kinase C isoforms leading to activation of mitogen-activated protein kinase. J Immunol 166, 2681–7. [DOI] [PubMed] [Google Scholar]
- Monto AS, Fendrick AM, and Sarnes MW (2001). Respiratory illness caused by picornavirus infection: a review of clinical outcomes. Clin. Ther 23, 1615–1627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Navarette CR, Sisson JH, Nance E, Allen-Gipson DS, Hanes J, and Wyatt TA (2012). Particulate Matter in Cigarette Smoke Increases Ciliary Axoneme Beating through Mechanical Stimulation. J Aerosol Med & Pulm Drug Deliv 25, 159–168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker PJ, Bosca L, Dekker L, Goode NT, Hajibagheri N, and Hansra G (1995). Protein kinase C (PKC)-induced PKC degradation: a model for down-regulation. Biochem. Soc. Trans 23, 153–155. [DOI] [PubMed] [Google Scholar]
- Pickles RJ (2013). Human airway epithelial cell cultures for modeling respiratory syncytial virus infection In Challenges and Opportunities for Respiratory Syncytial Virus Vaccines, Springer; ) pp. 371–387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prasso JE, and Deng JC (2017). Postviral Complications: Bacterial Pneumonia. Clin. Chest Med 38, 127–138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Proenca de Oliveira-Maul J, Barbosa de Carvalho H, Goto DM, Maia RM, Flo C, Barnabe V, Franco DR, Benabou S, Perracini MR, Jacob-Filho W, et al. (2013). Aging, diabetes, and hypertension are associated with decreased nasal mucociliary clearance. Chest 143, 1091–1097. [DOI] [PubMed] [Google Scholar]
- Rosenstein N, Phillips WR, Gerber MA, Marcy SM, Schwartz B, and Dowell SF (1998). The common cold—principles of judicious use of antimicrobial agents. Pediatrics 101, 181–184. [Google Scholar]
- Ruben FL, and Cate TR (1987). Influenza pneumonia. Semin Respir Infect 2, 122–129. [PubMed] [Google Scholar]
- Sacco RE, McGill J, Pillatzki AE, Palmer M, and Ackermann MR (2014). Respiratory syncytial virus infection in cattle. Vet. Pathol 51, 427–436. [DOI] [PubMed] [Google Scholar]
- Salathe M, Pratt MM, and Wanner A (1993). Protein kinase C-dependent phosphorylation of a ciliary membrane protein and inhibition of ciliary beating. J Cell Sci 106, 1211–1220. [DOI] [PubMed] [Google Scholar]
- Sapkota M, and Wyatt TA (2015). Alcohol, aldehydes, adducts and airways. Biomolecules 5, 2987–3008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt W, and De Lint J (1972). Causes of death of alcoholics. Q. J. Stud. Alcohol 33, 171–185. [PubMed] [Google Scholar]
- Simet SM, and Sisson JH (2015). Alcohol’s Effects on Lung Health and Immunity. Alcohol. Res 37, 199–208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sisson JH, Papi A, Beckmann JD, Leise KL, Wisecarver J, Brodersen BW, Kelling CL, Spurzem JR, and Rennard SI (1994). Smoke and viral infection cause cilia loss detectable by bronchoalveolar lavage cytology and dynein ELISA. Am J Respir Crit Care Med 149, 205–213. [DOI] [PubMed] [Google Scholar]
- Sisson JH, Stoner JA, Ammons BA, and Wyatt TA (2003). All-digital image capture and whole-field analysis of ciliary beat frequency. J Microsc 211, 103–111. [DOI] [PubMed] [Google Scholar]
- Slager RE, Sisson JH, Pavlik JA, Johnson JK, Nicolarsen JR, Jerrells TR, and Wyatt TA (2006). Inhibition of protein kinase C epsilon causes ciliated bovine bronchial cell detachment. Exp Lung Res 32, 349–362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tristram DA, Hicks W, and Hard R (1998a). Respiratory syncytial virus and human bronchial epithelium. Archives of Otolaryngology–Head & Neck Surgery 124, 777–783. [DOI] [PubMed] [Google Scholar]
- Tristram DA, H. W Jr, and Hard R (1998b). Respiratory syncytial virus and human bronchial epithelium. Arch Otolaryngol Head Neck Surg 124, 777–783. [DOI] [PubMed] [Google Scholar]
- Turner RB (1998). The common cold. Pediatr Ann 27, 790–795. [DOI] [PubMed] [Google Scholar]
- Villenave R, Shields MD, and Power UF (2013). Respiratory syncytial virus interaction with human airway epithelium. Trends Microbiol 21, 238–244. [DOI] [PubMed] [Google Scholar]
- Warren KJ, Pavlik JA, DeVasure JM, Dickinson JD, Sisson JH, and Wyatt TA (2016). RSV-specific anti-viral immunity is disrupted by chronic ethanol consumption. Alcohol 55, 35–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warren KJ, Olson MM, Thompson NJ, Cahill ML, Wyatt TA, Yoon KJ, Loiacono CM, and Kohut ML (2015). Exercise Improves Host Response to Influenza Viral Infection in Obese and Non-Obese Mice through Different Mechanisms. PLoS One 10, e0129713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss CP, Gentry WW, Cole NA, McCollum FT, and Jennings JS (2017). Effects of feeding condensed distiller’s solubles and crude glycerin alone or in combination on finishing beef cattle performance, carcass characteristics, and in vitro fermentation. J. Anim. Sci 95, 922–929. [DOI] [PubMed] [Google Scholar]
- Wong LB, Park CL, and Yeates DB (1998). Neuropeptide Y inhibits ciliary beat frequency in human ciliated cells via nPKC, independently of PKA. Am J Physiol 275, C440–448. [DOI] [PubMed] [Google Scholar]
- Wyatt T, and Sisson J (2001). Chronic ethanol downregulates PKA activation and ciliary beating in bovine bronchial epithelial cells. American Journal of Physiology-Lung Cellular and Molecular Physiology 281, L575–L581. [DOI] [PubMed] [Google Scholar]
- Wyatt TA, Sisson JH, Allen-Gipson DS, McCaskill MK, Boten JA, DeVasure JM, Bailey KL, and Poole JA (2012). Co-Exposure to Cigarette Smoke and Alcohol Decreases Airway Epithelial Cell Cilia Beating in a Protein Kinase C epsilon-Dependent Manner. Am J Pathol 181, 431–440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wyatt TA, Slager RE, Heires AJ, Devasure JM, Vonessen SG, Poole JA, and Romberger DJ (2010). Sequential activation of protein kinase C isoforms by organic dust is mediated by tumor necrosis factor. Am. J. Respir. Cell Mol. Biol 42, 706–715. [DOI] [PMC free article] [PubMed] [Google Scholar]


