Abstract
Background
Little is known about pain and opiate use at home directly after total knee replacement (TKR). Due to adverse effects, low opiate use is desired. An electronic health app (PainCoach) was developed to guide patients in pain control and opiate use.
Objective
The aim of this paper was to investigate the effects of the PainCoach app on pain control and opiate use in patients who underwent TKR during the first 2 weeks at home after surgery.
Methods
In an unblinded randomized controlled trial, patients scheduled for TKR were offline recruited and randomized to a PainCoach group or control group. In the PainCoach group, the PainCoach app was downloaded on each patient’s smartphone or tablet. In response to the patient’s input of the pain experienced, the PainCoach app gave advice on pain medication use, exercises/rest, and when to call the clinic. This advice was the same as that received during usual care. The control group received usual care. The primary outcomes were opiate use and visual analog scale (VAS) pain scores at rest, during activity, and at night during the first 2 weeks at home after surgery, which were collected daily from day 1 until 14 postoperatively by online questionnaires. The actual amount of app use was recorded, and active use was defined as ≥12 total app uses.
Results
The pain scores did not differ between the groups. The PainCoach group (n=38) used 23.2% less opiates (95% CI −38.3 to −4.4; P=.02) and 14.6% more acetaminophen (95% CI 8.2-21.3; P<.001) when compared with the findings in the control group (n=33). The PainCoach app was used 12 (IQR 4.5-22.0) times per patient. In the active PainCoach subgroup (n=19), the following were noted when compared with the findings in the control group: 4.1 times faster reduction of the VAS pain score during activity (95% CI −7.5 to −0.8; P=.02), 6.3 times faster reduction of the VAS pain score at night (95% CI −10.1 to −2.6; P=.001), 44.3% less opiate use (95% CI −59.4 to −23.5; P<.001), 76.3% less gabapentin use (95% CI −86.0 to −59.8; P<.001), and 21.0% more acetaminophen use (95% CI 12.6-30.0; P<.001).
Conclusions
The use of the PainCoach app contributes to reduced opiate use in the initial period at home after TKR. Active use of this app leads to a further reduction in opiate use and improved pain control.
Trial Registration
ClinicalTrials.gov NCT03961152; https://clinicaltrials.gov/ct2/show/NCT03961152
Keywords: opiate use, pain, total knee replacement, app, eHealth
Introduction
Total knee replacement (TKR) is a successful treatment option for patients with end-stage knee osteoarthritis (OA) [1]. Moderate-to-severe pain after TKR can be expected [2,3]. Local infiltration anesthesia (LIA) techniques and so-called fast-track recovery programs have resulted in reduced pain and early mobilization, subsequently reducing the length of stay in hospital and increasing patient satisfaction [4-7]. Previous research established several factors associated with increased pain after TKR [8-19] (Table 1). Postoperative pain inhibits recovery, increases morbidity, and may result in chronic pain, ultimately limiting the effectiveness of TKR [6,20]. Therefore, pain should be controlled optimally both in the hospital and at home.
Table 1.
Factors associated with increased pain after total knee replacement.
| Factor | Association with increased pain after TKRa |
| Gender | Being female [8-12] |
| Age | Older age [8,10,13] |
| BMIb | Higher BMIb [8,10] |
| ASAc score | Higher ASAc score [10] |
| Pain catastrophization | Higher pain catastrophization score [12,14-17] |
| Comorbidity | Presence of comorbidities [8,10,13,18] |
| Previous knee surgery | Having a history of knee surgery [10] |
| Preoperative pain | Higher preoperative pain severity [8,12,18,19] |
| Social support | Poor social support [13] |
| Preoperative mental health | Poor preoperative mental health [8,10,13,18] |
aTKR: total knee replacement.
bBMI: body mass index.
cASA: American Society of Anesthesiologists.
Although pain is usually under control during hospital stay, less is known about pain control in the initial period at home after TKR. Current pain management strategies include a combination of nonsteroidal anti-inflammatory drugs (NSAIDs), nonnarcotic medication, opiates, and exercise [4]. Although opiates are very effective for reducing pain, serious adverse effects, such as nausea, itching, reduced gut mobility, and urinary retention, often occur [21]. Addiction to opiates is an ever increasing problem and may ultimately lead to an increased risk of death [22]. The amount of opiate use should therefore be kept to a minimum. Orthopedic surgery, however, accounts for an estimated 8.8% of prolonged prescription opiate use [23]. Therefore, alternative pain management strategies are needed. Electronic health (eHealth) apps can be used to guide patients in improving their pain management strategies at home. An important benefit of these apps is that patients can access the information provided directly and anywhere whenever necessary [24-29]. The number of older adults with internet access and acceptance of internet-based interventions is increasing, and patients tend to remember up to 80% of the information acquired from interactive education [30,31].
With this in mind, to manage pain better and potentially decrease opiate use, an eHealth app named PainCoach was developed. This app aims to help patients control their pain better in the initial period at home after TKR, including optimal use of the available pain medication. This study aimed to determine the effects of PainCoach on pain control and opiate use in TKR patients in the first 2 weeks at home after surgery. The hypothesis was that the use of this app would decrease pain and opiate use.
Methods
Study Design
An unblinded, randomized, controlled, single-center trial was performed at Kliniek ViaSana (Mill, The Netherlands). Patients with an American Society of Anesthesiologists (ASA) score of I-II, a body mass index (BMI) of ≤35, and a plan to undergo primary TKR between February and June 2016 were enrolled. Four experienced high-volume knee surgeons performed all surgeries, and three experienced anesthesiologists administered spinal anesthesia. The same type of TKR implant was used in all patients (NexGen LPS, ZimmerBiomet, Warsaw, Indiana). All surgeries were performed using a tourniquet. The pain management protocol consisted of preoperatively administered medication, LIA injections during surgery directly before cementing the implant, and a step-wise postoperative pain management protocol (Multimedia Appendix 1). Patients were excluded if they did not possess a smartphone or tablet, had a contraindication to any of the medications used in the study, did not have an email address, did not have internet at home, did not have a thorough command of the Dutch language, had memory disorders, or had surgery under general anesthesia. Patients were recruited over the phone by the research staff after being scheduled for primary TKR under spinal anesthesia, and contraindication to any of the medications used in the study and presence of memory disorders were checked by the anesthesiologists. Patients were asked over the phone if they possessed a smartphone or tablet, had an email address, had internet at home, and had a thorough command of the Dutch language. Patient information and informed consent were sent by postal service if a patient met the criteria and was interested to participate. Patients were considered lost to follow-up if they completed less than two postoperative questionnaires during the first 2 weeks at home. Power analysis (significance level: .05, power: 90%) showed that 35 patients would be needed in each group to detect a difference of 10 points on a visual analog scale (VAS) for pain (VAS pain, 0-100). Written informed consent was obtained from all participants. The study was approved by the medical ethics committee of St. Anna Hospital (Geldrop, The Netherlands, Study ID: 5.12) and was registered at Clinicaltrials.gov retrospectively (ID: NCT03961152).
Randomization
Included unblinded patients were randomly assigned to the PainCoach or control group using lots presented in sealed opaque envelopes during admission. All lots were created and sealed by a researcher in the ratio of 1:1. A blinded nurse presented the envelops to a patient, and the patient selected one to complete randomization. All patients received the usual pain management care including pre-, peri-, and postoperative pain medication (Multimedia Appendix 1), participated in group information meetings, received an information booklet, and could contact the clinic at any time (24 hours a day/7 days a week) in case of any remaining questions. In the PainCoach group, in addition to receiving the aforementioned usual care, the PainCoach app (Interactive Studios, Rosmalen, The Netherlands) was downloaded on each patient’s smartphone or tablet, using a unique download code. In this way, the PainCoach app was not available to the control group. An unblinded nurse provided the code and assisted the patient by completing the download process of the app during admission. The app gave the same advice as that during usual care. After only entering the date of surgery as patient data, the app allowed patients to input their pain level (no pain, bearable pain, unbearable pain, or untenable pain) whenever they wanted until day 14 after surgery. Based on the patient’s input and taking into account the number of days after surgery, the app provided advice on pain medication use, physiotherapy exercises including videos, use of ice or heat packs, rest, immobilization of the operated leg, and when to call the clinic (Multimedia Appendix 2). Patients in the PainCoach group were not subjected to any treatment that was different from that in the control group (ie, advice on pain management was delivered in an extra and different way, but the pain medication itself was exactly the same for both groups). During the study, no major changes or revisions were made to the PainCoach app.
Outcomes and Measurements
Beside the actual amount of app use, all the outcome measurements were assessed using a digital, online, automated collection system (OnlinePROMs, Interactive Studios, Rosmalen, The Netherlands), which automatically sent an invitation by email to complete an online questionnaire preoperatively, daily from day 1 to 14, and at 1 month postoperatively. In case of nonresponse to the preoperative or 1-month questionnaire, an automatic reminder was sent after 3 days. The invitation to complete the daily questionnaire was sent at 5 pm, and patients had access to the questionnaire until midnight.
The primary outcomes were opiate use and pain score of the operated knee at rest, during activity, and at night in the first 2 weeks at home after TKR. The pain score was measured on a VAS for pain, which ranged from 0 (no pain) to 100 (worst imaginable pain), preoperatively, daily from day 1 to 14, and at 1 month postoperatively [32-35]. Severe pain was defined as a VAS pain score from 70 to 100. Opiate (oxycodon; 5 mg per tablet; different manufacturers) use was recorded in quantities per 24 hours from day 1 to 14.
The secondary outcomes in the first 2 weeks at home and 1 month after TKR included other pain medication use (ie, NSAIDs [diclofenac], acetaminophen, or gabapentin; different manufacturers), which was also recorded in quantities per 24 hours from day 1 to 14. Additionally, pain acceptance at rest, during activity, and at night was assessed with a happy smiley (acceptable pain) and a sad smiley (unacceptable pain) preoperatively, daily from day 1 to 14, and at 1 month postoperatively. Experiences with the executed recommended physiotherapy exercises were recorded daily from day 1 to 14 on a 3-item scale (did too much, exactly enough, or could have done more exercises). Moreover, function and quality of life were measured preoperatively and 1 month postoperatively. Knee function was assessed using the Knee Injury and Osteoarthritis Outcome Score–Physical Function Short-form (KOOS-PS) on a scale from 0 (no difficulty) to 100 (extreme difficulty) [36]. The Oxford Knee Score was used to measure combined function and pain on a scale from 0 (most severe symptoms) to 48 (least severe symptoms) [37]. Quality of life was measured using the EuroQol-5 Dimensions (EQ-5D) 3-level version (EQ-5D-3L) questionnaire consisting of the following two scores: EQ VAS score, which is assessed on a scale from 0 (worst imaginable health state) to 100 (best imaginable health state), and EQ-5D descriptive system [38]. The PainCoach app’s perceived effectiveness (usability, added value, and likelihood of being recommended to others) was recorded on a 5-item scale ranging from totally agree to totally disagree at day 14 after surgery. Each downloaded app had its own app code that was used to record the actual amount of app use. As the admission period was generally 1 or 2 days, outcomes were measured until day 14 after surgery, and outcomes at home were investigated, the outcome active PainCoach app use was defined as using the app at least 12 times in total.
Preoperative opiate and other pain medication use, age, gender, ASA score, BMI, preoperative comorbidities, history of knee surgery on the same side, Charnley score, date of surgery, date of discharge, and complication data were collected from the electronic patient records. Pain coping, anxiety, education level, and marital status were determined preoperatively using an online questionnaire. Pain coping was measured using the pain coping and cognition list scored from 1 (totally disagree) to 6 (totally agree), and it had the following four categories: catastrophizing, pain coping, internal pain management, and external pain management [39].
Statistical Analysis
Analysis was performed using SPSS version 25.0 (IBM Corp, Armonk, New York). All measured outcomes from day 1 until day 14 after surgery were recoded into measured outcomes for days at home by subtraction of the admission period. Patient characteristics were analyzed using descriptive statistics, and data were checked for normal distribution. Differences in mean, median, or percentage were tested using the independent two-sample t-test, Mann-Whitney U test, likelihood analysis, Fisher’s test, or Pearson’s chi-squared test, depending on the type of data. Mixed linear models were used to analyze the overall rate of decrease or increase for continuous data, and generalized linear models were used to analyze the percentage decrease or increase for count and nominal data. Additional analysis was performed to compare the active PainCoach subgroup with the control group, with correction for differences in preoperative data. Statistical significance was set at P<.05, and trends were defined as .05<P<.10.
Results
Patient Characteristics
A total of 97 patients were included, and of these, 76 patients were randomized. Because of loss to follow-up, the final analysis was performed with 71 patients (PainCoach group, n=38; control group, n=33) (Figure 1). The response rates for the daily questionnaires at home were 91% in the PainCoach group and 89% in the control group.
Figure 1.
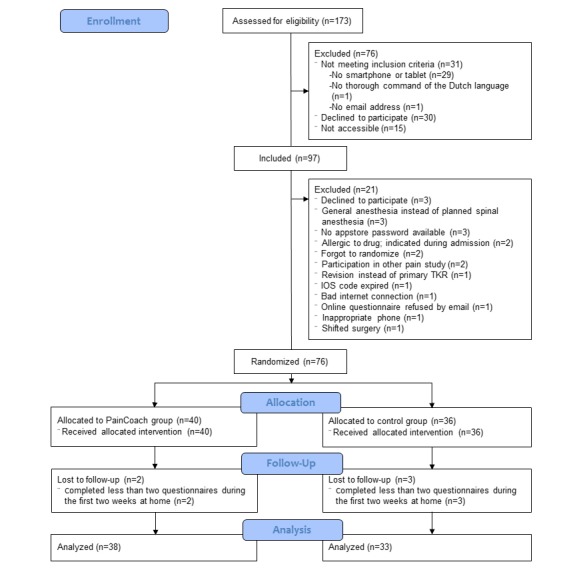
Study flowchart. IOS: iPhone operating system; TKR: total knee replacement.
No statistically significant differences in patient characteristics were found between the PainCoach group and control group. The preoperative VAS pain score at night was significantly lower in the active PainCoach subgroup (n=19) than in the control group (P=.02) (Table 2).
Table 2.
Characteristics of patients in the PainCoach group, active PainCoach subgroup, and control group.
| Characteristic | 1. PainCoach (n=38) |
2. Active PainCoach (n=19) |
3. Control (n=33) |
P value (1 vs 3) |
P value (2 vs 3) |
||
| Gender (male), n (%) | 23 (61) | 13 (68) | 19 (58) | .80 | .44 | ||
| Age (years), mean (SD) | 62.6 (7.0) | 62.8 (6.1) | 64.6 (7.5) | .24 | .38 | ||
| BMIa, mean (SD) | 27.6 (3.5) | 26.7 (3.4) | 27.8 (3.0) | .83 | .24 | ||
| ASAb (I), n (%) | 18 (47) | 11 (58) | 12 (36) | .35 | .13 | ||
| Preoperative comorbidities, n (%) | 14 (37) | 8 (42) | 17 (52) | .21 | .51 | ||
| Preoperative prescription, n (%) |
|
|
|
|
|
||
|
|
NSAIDsc | 5 (13) | 3 (16) | 6 (18) | .20 | .48 | |
|
|
Acetaminophen | 1 (3) | 0 (0) | 1 (3) | .92 | >.99 | |
|
|
Opiate | 3 (8) | 0 (0) | 0 (0) | .24 | >.99 | |
|
|
Gabapentin | 0 (0) | 0 (0) | 0 (0) | >.99 | >.99 | |
| Preoperative anxiety, n (%) |
|
|
|
.59 | >.99 | ||
|
|
No anxiety | 33 (87) | 18 (95) | 30 (91) |
|
|
|
|
|
Some anxiety | 5 (13) | 1 (5) | 3 (9) |
|
|
|
|
|
Much anxiety | 0 (0) | 0 (0) | 0 (0) |
|
|
|
| History of knee surgery on the same side, n (%) | 27 (71) | 15 (79) | 21 (64) | .51 | .25 | ||
| Charnley score, n (%) |
|
|
|
.64 | .98 | ||
|
|
One knee affected with OAd | 22 (58) | 12 (63) | 19 (58) |
|
|
|
|
|
Both knees affected with OA | 7 (18) | 3 (16) | 6 (18) |
|
|
|
|
|
Contralateral TKRe | 5 (13) | 1 (5) | 2 (6) |
|
|
|
|
|
Multiple joints affected with OA | 4 (11) | 3 (16) | 6 (18) |
|
|
|
| Education level, n (%) |
|
|
|
.33 | .30 | ||
|
|
Primary school | 3 (8) | 1 (5) | 1 (3) |
|
|
|
|
|
Secondary school | 14 (37) | 7 (37) | 10 (31) |
|
|
|
|
|
Tertiary school | 21 (55) | 11 (58) | 21 (66) |
|
|
|
| Marital status, n (%) |
|
|
|
.19 | .09 | ||
|
|
Married | 29 (76) | 18 (95) | 22 (67) |
|
|
|
|
|
Otherf | 9 (24) | 1 (5) | 11 (33) |
|
|
|
| Pain coping, mean (SD) |
|
|
|
|
|
||
|
|
Catastrophization | 2.5 (0.7) | 2.5 (0.8) | 2.3 (0.6) | .15 | .32 | |
|
|
Pain coping | 3.6 (1.0) | 3.8 (1.0) | 3.7 (0.8) | .68 | .66 | |
|
|
Internal pain management | 4.1 (0.8) | 4.2 (0.9) | 3.9 (0.8) | .24 | .29 | |
|
|
External pain management | 2.7 (0.8) | 2.7 (0.7) | 2.5 (0.8) | .31 | .36 | |
| Preoperative VASg pain, median (IQRh) |
|
|
|
|
|
||
|
|
Knee at rest | 33.0 (20.8-52.8) | 33.0 (13.0-43.0) | 32.0 (17.8-49.0) | .65 | .82 | |
|
|
Knee during activity | 60.5 (36.5-77.3) | 57.0 (30.0-75.0) | 60.0 (43.3-73.8) | .82 | .69 | |
|
|
Knee at night | 20.5 (4.8-42.5) | 15.0 (1.0-30.0) | 35.5 (15.0-58.5) | .11 | .02i | |
| Preoperative acceptable pain, n (%) |
|
|
|
|
|
||
|
|
Knee at rest | 29 (76) | 16 (84) | 27 (82) | .40 | >.99 | |
|
|
Knee during activity | 16 (42) | 10 (53) | 13 (39) | .90 | .41 | |
|
|
Knee at night | 28 (74) | 16 (84) | 27 (82) | .28 | >.99 | |
| Preoperative KOOS-PSj, median (IQR) | 47.3 (41.6-55.3) | 46.1 (40.3-54.4) | 48.5 (40.3-57.9) | .76 | .96 | ||
| Preoperative OKSk, mean (SD) | 25.3 (7.2) | 27.0 (7.2) | 24.8 (5.6) | .75 | .23 | ||
| Preoperative EQ-5Dl descriptive system, median (IQR) | 0.775 (0.471-0.783) | 0.775 (0.516-0.807) | 0.775 (0.651-0.807) | .27 | .81 | ||
| Preoperative EQ VASm, median (IQR) | 86.0 (73.6-94.3) | 87.0 (79.0-93.0) | 86.0 (74.0-95.5) | .89 | .72 | ||
| Complications, n (%) | 3 (8) | 2 (11) | 1 (3) | .62 | .55 | ||
aBMI: body mass index.
bASA: American Society of Anesthesiologists.
cNSAIDs: nonsteroidal anti-inflammatory drugs.
dOA: osteoarthritis.
eTKR: total knee replacement.
fOther marital status: single, living together, divorced, widow(er), living apart together relationship, different.
gVAS: visual analog scale.
hIQR: interquartile range.
iSignificant difference (P<.05).
jKOOS-PS: Knee Injury and Osteoarthritis Outcome Score–Physical Function Short-form.
kOKS: Oxford Knee Score.
lEQ-5D: EuroQol-5 Dimensions.
mEQ VAS: EuroQol visual analog scale.
Visual Analog Scale Pain Scores and Opiate Use
During the first 2 weeks at home, the PainCoach group had VAS pain scores of 17.0 (IQR 5.0-30.0) at rest, 20.0 (IQR 7.0-35.0) during activity, and 17.0 (IQR 4.0-37.0) at night. The control group had VAS pain scores of 20.0 (IQR 7.0-33.0) at rest, 21.0 (IQR 10.0-38.0) during activity, and 20.5 (IQR 8.0-40.0) at night. Pain was classified as severe on one or more days in 21% (8/38) of patients from the PainCoach group and 30% (10/33) of patients from the control group. No statistically significant differences were found between the two groups in terms of the VAS pain scores at rest, during activity, and at night (Figure 2A-C, Table 3). Regarding opiate use, the PainCoach group used a mean of 0.4 (SD 0.7) tablets a day and the control group used a mean of 0.5 (SD 0.8) tablets a day. Opiate use was significantly reduced by 23.2% in the PainCoach group when compared with the finding in the control group (95% CI −38.3 to −4.4; P=.02) (Figure 2A-C, Table 3). One month after surgery, no statistically significant differences in the VAS pain scores were found between the PainCoach group and control group (Table 4).
Figure 2.
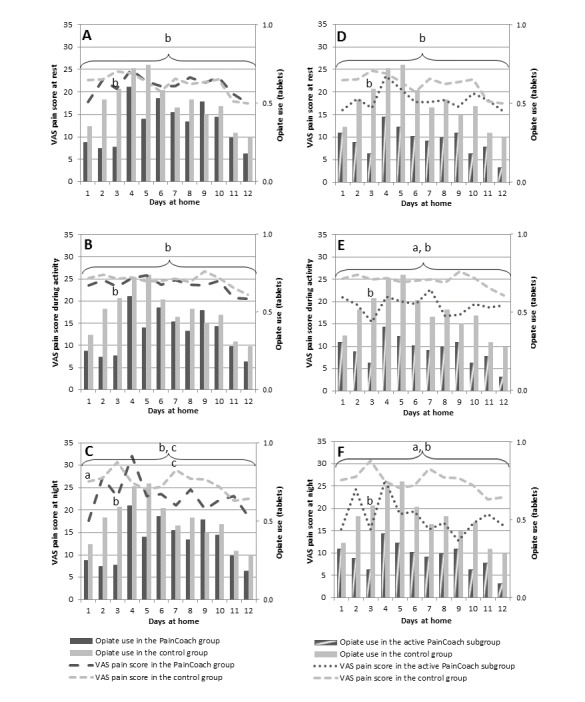
VAS pain scores and opiate use in the PainCoach group and control group at rest (A), during activity (B), and at night (C) and in the active PainCoach subgroup and control group at rest (D), during activity (E), and at night (F) on separate days and in the overall first period at home. a: significant difference in VAS pain (P<.05); b: significant difference in opiate use (P<.05); c: trend in VAS pain (.05<P<.10); VAS: visual analog scale.
Table 3.
Findings in the PainCoach group, active PainCoach subgroup, and control group during the first 2 weeks at home.
| Variable | 1. PainCoach versus control | 2. Active PainCoach versus control | P value (1) | P value (2) | |
| VASa pain, decrease or increase (rate) |
|
|
|
|
|
|
|
Knee at rest | ↓0.3 | ↓1.9 | .86 | .27 |
|
|
Knee during activity | ↓1.0 | ↓4.1 | .48 | .02b |
|
|
Knee at night | ↓3.0 | ↓6.3 | .06c | <.001b |
| Medication use, decrease or increase (%) |
|
|
|
|
|
|
|
Opiate | ↓23.2 | ↓44.3 | .02b | <.001b |
|
|
NSAIDsd | ↓9.2 | ↓12.8 | .08c | .06c |
|
|
Acetaminophen | ↑14.6 | ↑21.0 | <.001b | <.001b |
|
|
Gabapentin | ↑4.6 | ↓76.3 | .71 | <.001b |
| Acceptable pain, decrease or increase (%) |
|
|
|
|
|
|
|
Knee at rest | ↓31.3 | ↓20.3 | .11 | .25 |
|
|
Knee during activity | ↓17.2 | ↑31.1 | .40 | .38 |
|
|
Knee at night | ↓21.1 | ↑36.4 | .21 | .25 |
| Experience with the executed recommended exercises—exactly enough, decrease or increase (%) | ↓33.1 | ↓8.7 | .02b | .67 | |
aVAS: visual analog scale.
bSignificant difference (P<.05).
cTrend (.05<P<.10).
dNSAIDs: nonsteroidal anti-inflammatory drugs.
Table 4.
Findings in the PainCoach group, active PainCoach subgroup, and control group 1 month after surgery.
|
|
1. PainCoach (n=38) |
2. Active PainCoach (n=19) |
3. Control (n=33) |
P value (1 vs 3) |
P value (2 vs 3) |
|
| VASa pain, median (IQRb) |
|
|
|
|
|
|
|
|
Knee at rest | 11.5 (5.0-20.8) | 11.5 (4.3-18.8) | 10.0 (5.0-25.0) | .77 | .53 |
|
|
Knee during activity | 14.0 (7.0-28.8) | 12.5 (9.3-26.3) | 15.0 (8.0-35.0) | .49 | .59 |
|
|
Knee at night | 15.0 (7.0-33.0) | 15.0 (5.0-33.0) | 15.0 (7.0-27.8) | .79 | .89 |
| Acceptable pain, n (%) |
|
|
|
|
|
|
|
|
Knee at rest | 31 (96.9) | 16 (100.0) | 28 (96.6) | >.99 | >.99 |
|
|
Knee during activity | 30 (93.8) | 15 (93.8) | 25 (86.2) | .41 | .64 |
|
|
Knee at night | 26 (81.3) | 14 (87.5) | 26 (89.7) | .48 | >.99 |
| KOOS-PSc, mean (SD) | 36.5 (10.5) | 33.5 (8.4) | 39.6 (9.8) | .24 | .04d | |
| OKSe, mean (SD) | 28.4 (8.4) | 29.9 (9.1) | 26.8 (6.2) | .42 | .18 | |
| EQ-5Df descriptive system, median (IQR) | 0.775 (0.693-0.843) | 0.811 (0.775-0.857) | 0.775 (0.651-0.811) | .34 | .11 | |
| EQ VASg, median (IQR) | 80.0 (70.0-90.0) | 83.5 (70.0-90.0) | 80.0 (65.5-89.5) | .56 | .32 | |
aVAS: visual analog scale.
bIQR: interquartile range.
cKOOS-PS: Knee Injury and Osteoarthritis Outcome Score–Physical Function Short-form.
dSignificant difference (P<.05).
eOKS: Oxford Knee Score.
fEQ-5D: EuroQol-5 Dimensions.
gEQ VAS: EuroQol visual analog scale.
Adjusted analyses showed that the active PainCoach subgroup had VAS pain scores of 10.0 (IQR 4.0-26.3) at rest, 12.0 (IQR 5.0-25.0) during activity, and 10.0 (IQR 2.8-28.0) at night during the first 2 weeks at home. Pain was reported as severe on one or more days in 16% (3/19) of patients from the active PainCoach subgroup. The VAS pain score during activity significantly decreased 4.1 times faster in the active PainCoach subgroup when compared with the finding in the control group (95% CI −7.5 to −0.8; P=.02) (Figure 2E, Table 3). The VAS pain score at night significantly decreased 6.3 times faster in the active PainCoach subgroup when compared with the finding in the control group (95% CI −10.1 to −2.6; P=.001) (Figure 2F, Table 3). The mean opiate use was 0.3 (SD 0.5) tablets a day in the active PainCoach subgroup. Opiate use was significantly reduced by 44.3% in the active PainCoach subgroup when compared with the finding in the control group (95% CI −59.4 to −23.5; P<.001) (Figure 2D-F, Table 3). One month after surgery, no statistically significant differences in VAS pain scores were found between the active PainCoach subgroup and control group (Table 4).
Other Pain Medication Use, Pain Acceptance, and Experience With Executed Recommended Exercises
In the PainCoach group, there was a statistically significant 14.6% increase in acetaminophen use (95% CI 8.2-21.3; P<.001) and no statistically significant differences in NSAID use and gabapentin use when compared with the findings in the control group during the first 2 weeks at home (Table 3). Overall pain medication use was below the advised maximum in both groups. Pain acceptance was 86.5% at rest, 86.5% during activity, and 79.4% at night in the PainCoach group and was 90.4% at rest, 88.6% during activity, and 83.0% at night in the control group, without statistically significant differences between the two groups. Regarding experience with executing recommended exercises, the PainCoach group had statistically significant 33.1% reduced experience with executing exactly enough exercises when compared with the findings in the control group (69.7% vs. 77.5%; 95% CI −52.0 to −6.7; P=.02) (Table 3). At 1 month after surgery, no statistically significant differences were found when comparing both groups (Table 4).
Adjusted analyses comparing the active PainCoach subgroup with the control group showed statistically significant 21.0% increased acetaminophen use in the active PainCoach subgroup (95% CI 12.6-30.0; P<.001) during the first 2 weeks at home. Additionally, the active PainCoach subgroup had statistically significant 76.3% decreased gabapentin use when compared with the findings in the control group (mean 0.1 [SD 0.3] tablets a day vs. 0.4 [SD 1.0] tablets a day; 95% CI −86.0 to −59.8; P<.001) (Table 3). In the active PainCoach subgroup, pain acceptance was 88.4% at rest, 90.9% during activity, and 87.4% at night. Regarding pain acceptance and experience with executing recommended exercises, no statistically significant differences were found between the active PainCoach subgroup and control group (Table 3). One month after surgery, the mean KOOS-PS was significantly lower in the active PainCoach subgroup (33.5 [SD 8.4]) than in the control group (39.6 [SD 9.8]) (P=.048) (Table 4).
PainCoach App Use
Among 28 patients who provided appropriate responses, 25 (89%) reported ease of app use, 22 (79%) found that the app added value, and 22 (79%) would recommend the app to friends and family. The PainCoach app was used 12 (IQR 4.5-22.0) times per patient on 7 (IQR 4.0-9.0) days at home. The number of patients with at least one entry in the PainCoach app ranged from 11 (30%) to 26 (70%) per day at home (Multimedia Appendix 2). The app was most frequently used between 9 and 10 am and mostly for advice on bearable pain.
Discussion
Principal Findings
This study aimed to determine the effects of an eHealth app, the PainCoach app, on pain control and opiate use in patients who underwent TKR during the first 2 weeks at home after surgery. The hypothesis was that the app would decrease pain and opiate use. As indicated by the main findings, there was no statistically significant difference in pain scores between the two groups and opiate use was significantly reduced by 23.2% in the PainCoach group when compared with the finding in the control group. In the active PainCoach subgroup, however, pain during activity and at night significantly decreased 4.1 and 6.3 times faster, respectively, and opiate use significantly reduced by 44.3% when compared with the findings in the control group.
Overall, low pain scores and high levels of pain acceptance were found in this study. Only 21% (8/38) of patients in the PainCoach group and 30% (10/33) in the control group classified their pain as severe during one or more days at home. Other studies have stated that the most painful period after TKR surgery was the initial period at home, with 23%-30% of patients rating their average pain as severe [40,41]. Aside from the use of modern LIA techniques and a step-wise pain management protocol postoperatively, a possible explanation for the reported low pain and high acceptance scores in this study could be the guidance program that was provided to all patients who underwent TKR in Kliniek ViaSana. As less anxiety is associated with lower pain scores [14,19], the guidance provided might have resulted in less anxiety and therefore lower pain scores. The reported overall low pain scores also probably explain why no difference in pain scores was found between the PainCoach group and control group. Although overall pain scores were low, active use of the PainCoach app resulted in even lower pain scores during activity and at night when compared with the findings in the control group. These findings are in line with the results of a previous study showing that pain decreased by 0.7 points on a scale from 0 to 10 in patients with OA after online “pain coping skills” training [29]. Others have stated that 80% of interactive information is remembered compared with 20% of auditory information and 40% of read information [30,42,43]. As the PainCoach app is an interactive tool, it is logical that active use will result in better use of the pain management strategies provided and subsequently lower pain scores.
Opiate addiction caused 74 deaths in the Netherlands in 2016, and this number is increasing each year [44]. Using the PainCoach app, opiate use reduced by 23.2%, and active PainCoach app use resulted in a further reduction (44.3%). Because of a lack of standardized opiate prescribing protocols in orthopedic surgery, it is difficult to compare the reported amount of opiate use in this study with that in other studies. In one available study, a daily average morphine dose at discharge of 155 (SD 63) mg was prescribed to patients who underwent TKR, which would be the equivalent of 11 tablets per day of the opiate used in this study (oxycodon, 5 mg per tablet) and is far above the average use of 0.4 opiate tablets per day in this study [45]. The low preoperative opiate use of patients in this study might have contributed to the low opiate use after surgery, as preoperative opiate use is a strong predictor for prolonged opiate use after TKR [42,46,47]. With lower opiate use, acetaminophen use was higher, with a 14.6% increase in the PainCoach group and 21.0% increase in the active PainCoach subgroup. It can be concluded that because of the advice provided by the PainCoach app, opiate use was substituted by acetaminophen use. Opiate use was only advised in the presence of severe enough reported pain in the app. Therefore, it is concluded that the app helps to reduce the risk of the adverse effects of opiate use [48,49].
A shorter hospital stay is associated with a higher burden among patients, who need to take responsibility for aftercare shortly after surgery. Recent studies have shown that patients feel uncertain and left alone after discharge, which could increase anxiety and affect their pain coping and subsequent management [50,51]. Patients might need more individualized guidance, and the PainCoach app was developed to satisfy this need. The app scored high on usability, likelihood of being recommended to others, and added value. The results of this study show that the PainCoach app is a successful pain management tool, and its active use is recommended for the best effects on pain and opiate use.
To our knowledge, this is the first randomized controlled trial to examine the effects of eHealth with regard to controlling pain and reducing opiate use after TKR. The strengths of this study are that the actual amount of app use was measured and because of the unique download codes adopted, it was not possible for the control group to use the PainCoach app. The shortcomings are that the additional analysis was underpowered and the cost-effectiveness of the PainCoach app was not investigated. Furthermore, as there is no short validated questionnaire in Dutch for measuring pain acceptance, an expert group decided to assess pain acceptance using happy and sad smileys as the best alternative. In the population of this study, opiate use was already low. The app might have a much stronger effect in patient populations where preoperative opiate use is much higher. It is questionable if the PainCoach app is effective in the overall TKR population, as this study investigated the effects in patients having ASA I-II and BMI ≤35, which represent around 80% of the total TKR population [52,53]. Future research should focus on a larger sample size of the total TKR population, determination of the cost-effectiveness of the app, and use of the app in populations that have much higher preoperative opiate use.
Conclusions
The use of the PainCoach app contributes to reduced opiate use in the initial period at home after TKR. Active use of this app leads to further reduction in opiate use and improved pain control.
Acknowledgments
We thank the nurses for their help with executing this study, Klaartje Pijnappels for her assistance with data collection, and all patients who participated in this study.
Abbreviations
- ASA
American Society of Anesthesiologists
- BMI
body mass index
- eHealth
electronic health
- EQ
EuroQol
- EQ-5D
EuroQol-5 Dimensions
- EQ-5D-3L
EuroQol-5 Dimensions 3-level version
- EQ VAS
EuroQol visual analog scale
- IQR
interquartile range
- KOOS-PS
Knee Injury and Osteoarthritis Outcome Score–Physical Function Short-form
- LIA
local infiltration anesthesia
- NSAIDs
nonsteroidal anti-inflammatory drugs
- TKR
total knee replacement
- VAS
visual analog scale
Appendix
Pain management protocol.
Content and use of the PainCoach app.
CONSORT‐EHEALTH checklist (V 1.6.1).
Footnotes
Authors' Contributions: YP was responsible for designing the study, data collection, and study coordination; MP performed the statistical analysis supervised by YP; and YP, MP, AS, and MB were involved in drafting and revising the manuscript. All authors approved the final version of the manuscript.
Conflicts of Interest: None declared.
References
- 1.Jüni P, Reichenbach S, Dieppe P. Osteoarthritis: rational approach to treating the individual. Best Pract Res Clin Rheumatol. 2006 Aug;20(4):721–40. doi: 10.1016/j.berh.2006.05.002. [DOI] [PubMed] [Google Scholar]
- 2.de Beer JD, Winemaker MJ, Donnelly GA, Miceli PC, Reiz JL, Harsanyi Z, Payne LW, Darke AC. Efficacy and safety of controlled-release oxycodone and standard therapies for postoperative pain after knee or hip replacement. Can J Surg. 2005 Aug;48(4):277–83. http://www.canjsurg.ca/vol48-issue4/48-4-277/ [PMC free article] [PubMed] [Google Scholar]
- 3.Lin P. An evaluation of the effectiveness of relaxation therapy for patients receiving joint replacement surgery. J Clin Nurs. 2012 Mar;21(5-6):601–8. doi: 10.1111/j.1365-2702.2010.03406.x. [DOI] [PubMed] [Google Scholar]
- 4.Dalury DF. A state-of-the-art pain protocol for total knee replacement. Arthroplast Today. 2016 Mar;2(1):23–25. doi: 10.1016/j.artd.2016.01.004. https://linkinghub.elsevier.com/retrieve/pii/S2352-3441(16)00006-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kehlet H, Wilmore DW. Evidence-based surgical care and the evolution of fast-track surgery. Ann Surg. 2008 Aug;248(2):189–98. doi: 10.1097/SLA.0b013e31817f2c1a. [DOI] [PubMed] [Google Scholar]
- 6.Wu CL, Rowlingson AJ, Partin AW, Kalish MA, Courpas GE, Walsh PC, Fleisher LA. Correlation of postoperative pain to quality of recovery in the immediate postoperative period. Reg Anesth Pain Med. 2005;30(6):516–22. doi: 10.1016/j.rapm.2005.07.190. http://rapm.bmj.com/cgi/pmidlookup?view=long&pmid=16326335. [DOI] [PubMed] [Google Scholar]
- 7.Trasolini NA, McKnight BM, Dorr LD. The Opioid Crisis and the Orthopedic Surgeon. J Arthroplasty. 2018 Nov;33(11):3379–3382.e1. doi: 10.1016/j.arth.2018.07.002. [DOI] [PubMed] [Google Scholar]
- 8.Dowsey MM, Nikpour M, Choong PF. Outcomes following large joint arthroplasty: does socio-economic status matter? BMC Musculoskelet Disord. 2014 May 06;15:148. doi: 10.1186/1471-2474-15-148. https://bmcmusculoskeletdisord.biomedcentral.com/articles/10.1186/1471-2474-15-148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fillingim RB, King CD, Ribeiro-Dasilva MC, Rahim-Williams B, Riley JL. Sex, gender, and pain: a review of recent clinical and experimental findings. J Pain. 2009 May;10(5):447–85. doi: 10.1016/j.jpain.2008.12.001. http://europepmc.org/abstract/MED/19411059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jiang Y, Sanchez-Santos MT, Judge AD, Murray DW, Arden NK. Predictors of Patient-Reported Pain and Functional Outcomes Over 10 Years After Primary Total Knee Arthroplasty: A Prospective Cohort Study. J Arthroplasty. 2017 Jan;32(1):92–100.e2. doi: 10.1016/j.arth.2016.06.009. https://linkinghub.elsevier.com/retrieve/pii/S0883-5403(16)30274-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Racine M, Tousignant-Laflamme Y, Kloda LA, Dion D, Dupuis G, Choinière M. A systematic literature review of 10 years of research on sex/gender and experimental pain perception - part 1: are there really differences between women and men? Pain. 2012 Mar;153(3):602–18. doi: 10.1016/j.pain.2011.11.025. [DOI] [PubMed] [Google Scholar]
- 12.Sullivan M, Tanzer M, Stanish W, Fallaha M, Keefe FJ, Simmonds M, Dunbar M. Psychological determinants of problematic outcomes following Total Knee Arthroplasty. Pain. 2009 May;143(1-2):123–9. doi: 10.1016/j.pain.2009.02.011. [DOI] [PubMed] [Google Scholar]
- 13.Escobar A, Quintana JM, Bilbao A, Azkárate J, Güenaga JI, Arenaza JC, Gutierrez LF. Effect of patient characteristics on reported outcomes after total knee replacement. Rheumatology (Oxford) 2007 Jan;46(1):112–9. doi: 10.1093/rheumatology/kel184. [DOI] [PubMed] [Google Scholar]
- 14.Blackburn J, Qureshi A, Amirfeyz R, Bannister G. Does preoperative anxiety and depression predict satisfaction after total knee replacement? Knee. 2012 Oct;19(5):522–4. doi: 10.1016/j.knee.2011.07.008. [DOI] [PubMed] [Google Scholar]
- 15.Edwards RR, Haythornthwaite JA, Smith MT, Klick B, Katz JN. Catastrophizing and depressive symptoms as prospective predictors of outcomes following total knee replacement. Pain Res Manag. 2009;14(4):307–11. doi: 10.1155/2009/273783. doi: 10.1155/2009/273783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Riddle DL, Wade JB, Jiranek WA, Kong X. Preoperative pain catastrophizing predicts pain outcome after knee arthroplasty. Clin Orthop Relat Res. 2010 Mar;468(3):798–806. doi: 10.1007/s11999-009-0963-y. http://europepmc.org/abstract/MED/19585177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Vissers MM, Bussmann JB, Verhaar JA, Busschbach JJ, Bierma-Zeinstra SM, Reijman M. Psychological factors affecting the outcome of total hip and knee arthroplasty: a systematic review. Semin Arthritis Rheum. 2012 Feb;41(4):576–88. doi: 10.1016/j.semarthrit.2011.07.003. [DOI] [PubMed] [Google Scholar]
- 18.Lingard EA, Riddle DL. Impact of psychological distress on pain and function following knee arthroplasty. J Bone Joint Surg Am. 2007 Jun;89(6):1161–9. doi: 10.2106/JBJS.F.00914. [DOI] [PubMed] [Google Scholar]
- 19.Brander VA, Stulberg SD, Adams AD, Harden RN, Bruehl S, Stanos SP, Houle T. Predicting total knee replacement pain: a prospective, observational study. Clin Orthop Relat Res. 2003 Nov;(416):27–36. doi: 10.1097/01.blo.0000092983.12414.e9. [DOI] [PubMed] [Google Scholar]
- 20.De Luca ML, Ciccarello M, Martorana M, Infantino D, Letizia Mauro G, Bonarelli S, Benedetti MG. Pain monitoring and management in a rehabilitation setting after total joint replacement. Medicine (Baltimore) 2018 Oct;97(40):e12484. doi: 10.1097/MD.0000000000012484. http://Insights.ovid.com/pubmed?pmid=30290604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Niemeläinen M, Kalliovalkama J, Aho AJ, Moilanen T, Eskelinen A. Single periarticular local infiltration analgesia reduces opiate consumption until 48 hours after total knee arthroplasty. A randomized placebo-controlled trial involving 56 patients. Acta Orthop. 2014 Dec;85(6):614–9. doi: 10.3109/17453674.2014.961399. http://europepmc.org/abstract/MED/25238439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Reece AS, Hulse GK. Impact of lifetime opioid exposure on arterial stiffness and vascular age: cross-sectional and longitudinal studies in men and women. BMJ Open. 2014 Jun 02;4(6):e004521. doi: 10.1136/bmjopen-2013-004521. http://bmjopen.bmj.com/cgi/pmidlookup?view=long&pmid=24889849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Schoenfeld AJ, Jiang W, Chaudhary MA, Scully RE, Koehlmoos T, Haider AH. Sustained Prescription Opioid Use Among Previously Opioid-Naive Patients Insured Through TRICARE (2006-2014) JAMA Surg. 2017 Dec 01;152(12):1175–1176. doi: 10.1001/jamasurg.2017.2628. http://europepmc.org/abstract/MED/28813584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Currie M, Philip LJ, Roberts A. Attitudes towards the use and acceptance of eHealth technologies: a case study of older adults living with chronic pain and implications for rural healthcare. BMC Health Serv Res. 2015 Apr 16;15:162. doi: 10.1186/s12913-015-0825-0. https://bmchealthservres.biomedcentral.com/articles/10.1186/s12913-015-0825-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hochstenbach LM, Zwakhalen SM, Courtens AM, van Kleef M, de Witte LP. Feasibility of a mobile and web-based intervention to support self-management in outpatients with cancer pain. Eur J Oncol Nurs. 2016 Aug;23:97–105. doi: 10.1016/j.ejon.2016.03.009. https://linkinghub.elsevier.com/retrieve/pii/S1462-3889(16)30027-8. [DOI] [PubMed] [Google Scholar]
- 26.Jibb LA, Stevens BJ, Nathan PC, Seto E, Cafazzo JA, Johnston DL, Hum V, Stinson JN. Implementation and preliminary effectiveness of a real-time pain management smartphone app for adolescents with cancer: A multicenter pilot clinical study. Pediatr Blood Cancer. 2017 Oct;64(10) doi: 10.1002/pbc.26554. [DOI] [PubMed] [Google Scholar]
- 27.Machado GC, Pinheiro MB, Lee H, Ahmed OH, Hendrick P, Williams C, Kamper SJ. Smartphone apps for the self-management of low back pain: A systematic review. Best Pract Res Clin Rheumatol. 2016 Dec;30(6):1098–1109. doi: 10.1016/j.berh.2017.04.002. [DOI] [PubMed] [Google Scholar]
- 28.Naylor MR, Naud S, Keefe FJ, Helzer JE. Therapeutic Interactive Voice Response (TIVR) to reduce analgesic medication use for chronic pain management. J Pain. 2010 Dec;11(12):1410–9. doi: 10.1016/j.jpain.2010.03.019. http://europepmc.org/abstract/MED/20620119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rini C, Porter LS, Somers TJ, McKee DC, DeVellis RF, Smith M, Winkel G, Ahern DK, Goldman R, Stiller JL, Mariani C, Patterson C, Jordan JM, Caldwell DS, Keefe FJ. Automated Internet-based pain coping skills training to manage osteoarthritis pain: a randomized controlled trial. Pain. 2015 May;156(5):837–48. doi: 10.1097/j.pain.0000000000000121. http://europepmc.org/abstract/MED/25734997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sandberg EH, Sharma R, Sandberg WS. Deficits in retention for verbally presented medical information. Anesthesiology. 2012 Oct;117(4):772–9. doi: 10.1097/ALN.0b013e31826a4b02. [DOI] [PubMed] [Google Scholar]
- 31.Zickuhr K, Madden M. Pew Research Center's Internet & American Life Project. 2012. [2018-02-08]. Older adults and internet use https://www.sainetz.at/dokumente/studien/Older_adults_and_internet_use_2012.pdf.
- 32.Price DD, McGrath PA, Rafii A, Buckingham B. The validation of visual analogue scales as ratio scale measures for chronic and experimental pain. Pain. 1983 Sep;17(1):45–56. doi: 10.1016/0304-3959(83)90126-4. [DOI] [PubMed] [Google Scholar]
- 33.Delgado DA, Lambert BS, Boutris N, McCulloch PC, Robbins AB, Moreno MR, Harris JD. Validation of Digital Visual Analog Scale Pain Scoring With a Traditional Paper-based Visual Analog Scale in Adults. J Am Acad Orthop Surg Glob Res Rev. 2018 Mar;2(3):e088. doi: 10.5435/JAAOSGlobal-D-17-00088. http://europepmc.org/abstract/MED/30211382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chiu LY, Sun T, Ree R, Dunsmuir D, Dotto A, Ansermino JM, Yarnold C. The evaluation of smartphone versions of the visual analogue scale and numeric rating scale as postoperative pain assessment tools: a prospective randomized trial. Can J Anaesth. 2019 Jun;66(6):706–715. doi: 10.1007/s12630-019-01324-9. [DOI] [PubMed] [Google Scholar]
- 35.Jamison RN, Gracely RH, Raymond SA, Levine JG, Marino B, Herrmann TJ, Daly M, Fram D, Katz NP. Comparative study of electronic vs. paper VAS ratings: a randomized, crossover trial using healthy volunteers. Pain. 2002;99(1):341–347. doi: 10.1016/s0304-3959(02)00178-1. [DOI] [PubMed] [Google Scholar]
- 36.Perruccio AV, Stefan LL, Canizares M, Tennant A, Hawker GA, Conaghan PG, Roos EM, Jordan JM, Maillefert J, Dougados M, Davis AM. The development of a short measure of physical function for knee OA KOOS-Physical Function Shortform (KOOS-PS) - an OARSI/OMERACT initiative. Osteoarthritis Cartilage. 2008 May;16(5):542–50. doi: 10.1016/j.joca.2007.12.014. http://linkinghub.elsevier.com/retrieve/pii/S1063-4584(07)00405-0. [DOI] [PubMed] [Google Scholar]
- 37.Haverkamp D, Breugem SJ, Sierevelt IN, Blankevoort L, van Dijk CN. Translation and validation of the Dutch version of the Oxford 12-item knee questionnaire for knee arthroplasty. Acta Orthop. 2005 Jun;76(3):347–52. [PubMed] [Google Scholar]
- 38.EuroQol Group EuroQol--a new facility for the measurement of health-related quality of life. Health Policy. 1990 Dec;16(3):199–208. doi: 10.1016/0168-8510(90)90421-9. [DOI] [PubMed] [Google Scholar]
- 39.Stomp-van den Berg SG, Vlaeyen JW, Ter Kuile MM, Spinhoven P, van Breukelen G, Kole-Snijders AM. Pijn Coping en Cognitie Lijst (PCCL) [2018-02-08]. Meetinstrumenten chronische pijn deel 2 https://docplayer.nl/13110836-Meetinstrumenten-chronische-pijn-deel-2-pijn-coping-en-cognitie-lijst-pccl.html.
- 40.Chan EY, Blyth FM, Nairn L, Fransen M. Acute postoperative pain following hospital discharge after total knee arthroplasty. Osteoarthritis Cartilage. 2013 Sep;21(9):1257–63. doi: 10.1016/j.joca.2013.06.011. http://linkinghub.elsevier.com/retrieve/pii/S1063-4584(13)00847-9. [DOI] [PubMed] [Google Scholar]
- 41.Aasvang EK, Luna IE, Kehlet H. Challenges in postdischarge function and recovery: the case of fast-track hip and knee arthroplasty. Br J Anaesth. 2015 Dec;115(6):861–6. doi: 10.1093/bja/aev257. https://linkinghub.elsevier.com/retrieve/pii/S0007-0912(17)31433-2. [DOI] [PubMed] [Google Scholar]
- 42.Bol N, Smets EM, Rutgers MM, Burgers JA, de Haes HC, Loos EF, van Weert JC. Do videos improve website satisfaction and recall of online cancer-related information in older lung cancer patients? Patient Educ Couns. 2013 Sep;92(3):404–12. doi: 10.1016/j.pec.2013.06.004. [DOI] [PubMed] [Google Scholar]
- 43.Selinger CP, Carbery I, Warren V, Rehman AF, Williams CJ, Mumtaz S, Bholah H, Sood R, Gracie DJ, Hamlin PJ, Ford AC. The relationship between different information sources and disease-related patient knowledge and anxiety in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2017 Dec;45(1):63–74. doi: 10.1111/apt.13831. [DOI] [PubMed] [Google Scholar]
- 44.van Laar MW, van Gestel B, Cruts AA, van der Pol PM, Ketelaars AP, Beenakkers EM, Meijer RF, Croes EA, Brunt TM. Nationale Drug Monitor - Jaarbericht 2017. Utrecht: Trimbos instituut; 2018. [Google Scholar]
- 45.Hernandez NM, Parry JA, Taunton MJ. Patients at Risk: Large Opioid Prescriptions After Total Knee Arthroplasty. J Arthroplasty. 2017 Aug;32(8):2395–2398. doi: 10.1016/j.arth.2017.02.060. [DOI] [PubMed] [Google Scholar]
- 46.Bedard NA, DeMik DE, Dowdle SB, Callaghan JJ. Trends and risk factors for prolonged opioid use after unicompartmental knee arthroplasty. Bone Joint J. 2018 Jan;100-B(1 Supple A):62–67. doi: 10.1302/0301-620X.100B1.BJJ-2017-0547.R1. http://europepmc.org/abstract/MED/29292342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Franklin PD, Karbassi JA, Li W, Yang W, Ayers DC. Reduction in narcotic use after primary total knee arthroplasty and association with patient pain relief and satisfaction. J Arthroplasty. 2010 Sep;25(6 Suppl):12–6. doi: 10.1016/j.arth.2010.05.003. [DOI] [PubMed] [Google Scholar]
- 48.Sinatra RS, Jahr JS, Reynolds LW, Viscusi ER, Groudine SB, Payen-Champenois C. Efficacy and safety of single and repeated administration of 1 gram intravenous acetaminophen injection (paracetamol) for pain management after major orthopedic surgery. Anesthesiology. 2005 Apr;102(4):822–31. doi: 10.1097/00000542-200504000-00019. [DOI] [PubMed] [Google Scholar]
- 49.Yeh Y, Reddy P. Clinical and economic evidence for intravenous acetaminophen. Pharmacotherapy. 2012 Jun;32(6):559–79. doi: 10.1002/j.1875-9114.2011.01085.x. [DOI] [PubMed] [Google Scholar]
- 50.Specht K, Agerskov H, Kjaersgaard-Andersen P, Jester R, Pedersen BD. Patients' experiences during the first 12 weeks after discharge in fast-track hip and knee arthroplasty - a qualitative study. Int J Orthop Trauma Nurs. 2018 Nov;31:13–19. doi: 10.1016/j.ijotn.2018.08.002. [DOI] [PubMed] [Google Scholar]
- 51.Sjøveian AK, Leegaard M. Hip and knee arthroplasty - patient's experiences of pain and rehabilitation after discharge from hospital. Int J Orthop Trauma Nurs. 2017 Nov;27:28–35. doi: 10.1016/j.ijotn.2017.07.001. [DOI] [PubMed] [Google Scholar]
- 52.LROI LROI Rapportage 2017. [2019-12-09]. Distribution of ASA score of patients who underwent a primary knee arthroplasty per hospital in The Netherlands in 2016 http://www.lroi-rapportage.nl/knee-primary-knee-arthroplasty-demographics-practice-variation-asa-score.
- 53.LROI LROI Rapportage 2017. [2019-12-09]. Distribution of BMI of patients who underwent a primary knee arthroplasty per hospital in The Netherlands in 2016 http://www.lroi-rapportage.nl/knee-primary-knee-arthroplasty-demographics-practice-variation-body-mass-index.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Pain management protocol.
Content and use of the PainCoach app.
CONSORT‐EHEALTH checklist (V 1.6.1).


