Respiratory syncytial virus (RSV) is the most common cause of clinically significant respiratory infections in infants and children. It is estimated that there are over 75,000 annual hospitalizations in the United States for pneumonia and/or bronchiolitis because of RSV infections (1). Although certain populations are at increased risk of hospitalization because of RSV infection (e.g., <6 wk of age, immunocompromised, heart disease, lung disease), it is unclear why these and other individuals develop a more severe clinical course that leads to acute lung injury (ALI) or acute respiratory distress syndrome (ARDS) (2,3).
Although the young and very old are some of the most susceptible to RSV, it is evident that no one can escape RSV infection. There have been multiple speculations on the roles that the different arms of the immune system may play in the prevention of RSV infections. The importance of the humoral system and specific antibody production continues to be debated. Despite the ability of RSV hyperimmunoglobulin to decrease the rate of hospitalizations in infants with bronchopulmonary dysplasia, the role of humoral immunity in nonpremature infants (with maternal passive antibody protection to RSV) and the elderly (with a long history of repeated infections) remains unclear as to why these cohorts continue to be at risk (3–5). Interestingly, an inactivated RSV vaccine trial in the 1960s demonstrated that recipients of the vaccine developed a more severe clinical disease when subsequently challenged with a wild-type strain of virus, suggesting that preformed RSV-specific antibodies may play a role in the pathogenesis of severe disease (6–8). It has also been shown that secondary infections occur just as often with homologous virus subgroups as with alternate viral subgroups, again suggesting a lesser role for humoral immunity in RSV protection. Not surprisingly, even less is understood about the role of cell-mediated mechanisms for the prevention of RSV infections (4,9). Early studies by Graham et al. in mice have shown that although cytotoxic lymphocytes (CLs) are important for viral clearance, depletion of CLs resulted in significantly less disease (10). In addition, Cannon et al. similarly showed that transfer of CLs into RSV-infected mice also resulted in worse lung pathology (11). These results reflect our lack of understanding of the pathophysiology of viral immune responses, and leave it unclear as to the precise role(s) that the different arms of the immune system play in preventing RSV infections.
The recognition that RSV results in characteristic necrosis and sloughing of the bronchiolar epithelium is longstanding, and has been largely attributed to direct cellular toxicity of the virus itself. Although it is clear that the virus is capable of many of these direct effects, it has been suggested that immune-mediated mechanisms may actually be responsible for the pathogenesis of the disease, as suggested by the histologic findings of marked lymphocytic peribronchial and interstitial tissue infiltration (3,4). A role for dysregulated immune responses to pathogens, which subsequently lead to the development of secondary organ damage and disease, is not a new concept, and has been postulated to be involved in various autoimmune and immune dysregulation disorders. In fact, an “over zealous” or uncontrolled immune response to infections, especially viruses, is sine qua non to the development of a relatively rare pediatric immune dysregulation syndrome, hemophagocytic lymphohistiocytosis (HLH) (12). In this disorder, half of the affected infants and children harbor genetic mutations in genes of the perforin/granzyme pathway, a pathway that has been shown to be critical in the clearance of tumor cells and certain viruses by cytotoxic lymphocytes (e.g., natural killer cells and CD8+ T cells) (12,13). A critical understanding of this disorder has come from murine knockout models, which have demonstrated that the pathogenesis of this disorder does not appear to be due to lack of viral clearance, but rather the uncontrolled immune response to the viral infection (14–16). These results have suggested that the same pathway involved in cellular cytotoxicity and viral clearance (i.e., perforin/granzyme pathway), may also be involved in immune regulation. Although the genetic etiologies of the remainder of the “less severe” HLH subjects are unclear, the observation that many of these subjects have underlying autoimmune disorders (e.g., systemic-onset juvenile rheumatoid arthritis, Crohn's disease, lupus) suggests a role for immune dysregulation in the pathophysiology of this disorder. Interestingly, some of the most effective medical treatments for these subjects include the blockade of certain cytokine pathways such as IL-1 (e.g., Anakinra) in systemic-onset juvenile rheumatoid arthritis, and TNF (e.g., Remicaide) in rheumatoid arthritis. Whether or not these or other biologic modifiers are capable of treating other severe immune dysregulation syndromes such as HLH, or severe infections such as RSV, has not been fully evaluated (12).
In the article by Bem et al. in this issue of Pediatric Research (17), the authors set out to determine whether or not a cell-mediated immune response could be detected in children intubated from severe RSV infection. They compared their findings with results obtained from children intubated for other clinical reasons, and without RSV infection. They evaluated tracheal aspirates for granzymes A and B, which are predominantly expressed in CLs, as a marker of a cell-mediated response (13). They found significantly elevated levels and activity of both granzyme molecules, which also corresponded to elevated leukocyte counts and IL-8 cytokine levels in the airways of RSV-infected subjects compared to controls. In addition, they found that granzyme B expression was limited to the lymphoid compartment, and was not observed in myeloid or neutrophil cells, a point of contention that continues to be debated as to the cellular subsets that naturally express granzyme B (18,19). Interestingly, they also found elevated granzyme B expression in CD4+ T cells in bronchoalveolar lavage fluid from RSV-infected subjects. The role for this cytotoxic enzyme in a classically defined “noncytotoxic” T cell compartment such as CD4+ T cells is still unclear; however, prior reports have described expression of granzymes in both human and mouse CD4+ T regulatory cell compartments, suggesting their role in immune regulation (20–22). Still, the role of CD4+ T cell granzyme expression in immunity to pathogens and/or immune regulation remains largely unstudied.
The observations by Bem et al. extend our understanding of the cell-mediated responses that occur in vivo to respiratory infections such as RSV (17,23,24). Their results support prior suggestions that the immune response, in particular cytotoxic lymphocytes, may actually play a detrimental role in the development of severe RSV infections. These findings also suggest that one reason for the discrepancy of RSV more adversely affecting the young and elderly may be in their immune system's lack of proper regulation. Further understanding of how the immune system responds (appropriately and inappropriately) to various infections will be critical to our development of more effective clinical interventions (Fig. 1). Although more research is clearly needed to establish how different immune cell types contribute to the actual clinical pathology observed during infections, the work by Bem et al. helps to pave the way to our understanding of the importance of a balanced immune response.
Figure 1.
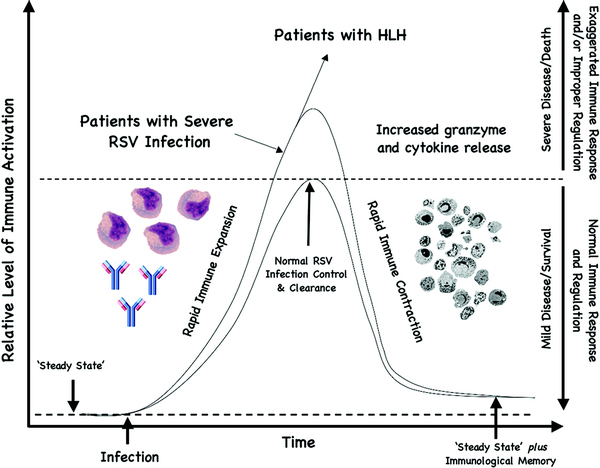
Illustration of a hypothetical balanced vs. imbalanced immune response to an infection. From a “steady state,” normal individuals are able to make a rapid immune response, and just as rapidly are able to undergo immune contraction back to the steady state with appropriate “immunologic memory” to that particular infectious agent after proper control and elimination (demonstrated by the small curve). Subjects with exaggerated immune responses and/or improper regulation develop more severe clinical presentations (demonstrated by the larger curve). Hemophagocytic lymphohistiocytosis (HLH) subjects fail to undergo normal immune contraction and require immunosuppression/chemotherapy to prevent progressive end-organ damage and/or death. Patients who develop severe RSV infections are proposed to undergo exaggerated cell-mediated immune response (e.g., increased granzyme B and cytokine release) and/or improper immune regulation, as suggested by data from Bem et al. and demonstrated by the large curve.
References
- 1.Shay DK, Holman RC, Newman RD, Liu LL, Stout JW, Anderson LJ. Bronchiolitis-associated hospitalizations among U.S. children, 1980–1996. JAMA. 1999;282:1440–1446. doi: 10.1001/jama.282.15.1440. [DOI] [PubMed] [Google Scholar]
- 2.Openshaw PJ. Immunopathological mechanisms in respiratory syncytial virus disease. Springer Semin Immunopathol. 1995;17:187–201. doi: 10.1007/BF00196165. [DOI] [PubMed] [Google Scholar]
- 3.Heilman CA, From the National Institute of Allergy and Infectious Diseases and the World Health Organization Respiratory syncytial and parainfluenza viruses. J Infect Dis. 1990;161:402–406. doi: 10.1093/infdis/161.3.402. [DOI] [PubMed] [Google Scholar]
- 4.La Via WV, Marks MI, Stutman HR. Respiratory syncytial virus puzzle: clinical features, pathophysiology, treatment, and prevention. J Pediatr. 1992;121:503–510. doi: 10.1016/S0022-3476(05)81135-7. [DOI] [PubMed] [Google Scholar]
- 5.Welliver RC. Review of epidemiology and clinical risk factors for severe respiratory syncytial virus (RSV) infection. J Pediatr. 2003;143:S112–S117. doi: 10.1067/S0022-3476(03)00508-0. [DOI] [PubMed] [Google Scholar]
- 6.Chin J, Magoffin R, Shearer L, Schieble J, Lennette E. Field evaluation of a respiratory syncytial virus vaccine and a trivalent parainfluenza virus vaccine in a pediatric population. Am J Epidemiol. 1969;89:449–463. doi: 10.1093/oxfordjournals.aje.a120957. [DOI] [PubMed] [Google Scholar]
- 7.Kapikian AZ, Mitchell RH, Chanock RM, Shvedoff RA, Stewart CE. An epidemiologic study of altered clinical reactivity to respiratory syncytial (RS) virus infection in children previously vaccinated with an inactivated RS virus vaccine. Am J Epidemiol. 1969;89:405–421. doi: 10.1093/oxfordjournals.aje.a120954. [DOI] [PubMed] [Google Scholar]
- 8.Kim HW, Canchola JG, Brandt CD, Pyles G, Chanock RM, Jensen K, Parrott RH. Respiratory syncytial virus disease in infants despite prior administration of antigenic inactivated vaccine. Am J Epidemiol. 1969;89:422–434. doi: 10.1093/oxfordjournals.aje.a120955. [DOI] [PubMed] [Google Scholar]
- 9.Isaacs D, Bangham C, McMichael A. Cell-mediated cytotoxic response to respiratory syncytial virus in infancts with bronchiolitis. Lancet. 1987;2:769–771. doi: 10.1016/S0140-6736(87)92502-5. [DOI] [PubMed] [Google Scholar]
- 10.Graham BS, Bunton LA, Wright PF, Karzon DT. Role of T lymphocytes subsets in the pathogenesis of primary infection and rechallenge with respiratory syncytial virus in mice. J Clin Invest. 1991;88:1026–1033. doi: 10.1172/JCI115362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cannon MJ, Openshaw PJ, Askonas BA. Cytotoxic T cells clear virus but augment lung pathology in mice infected with respiratory syncytial virus. J Exp Med. 1988;168:1163–1168. doi: 10.1084/jem.168.3.1163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Verbsky JW, Grossman WJ. Hemophagocytic lymphohistiocytosis: diagnosis, pathophysiology, treatment, and future perspectives. Ann Med. 2006;38:20–31. doi: 10.1080/07853890500465189. [DOI] [PubMed] [Google Scholar]
- 13.Russell JH, Ley TJ. Lymphocyte-mediated cytotoxicity. Annu Rev Immunol. 2002;20:323–370. doi: 10.1146/annurev.immunol.20.100201.131730. [DOI] [PubMed] [Google Scholar]
- 14.Badovinac VP, Hamilton SE, Harty JT. Viral infection results in massive CD8+ T cell expansion and mortality in vaccinated perforin-deficient mice. Immunity. 2003;18:463–474. doi: 10.1016/S1074-7613(03)00079-7. [DOI] [PubMed] [Google Scholar]
- 15.Badovinac VP, Tvinnereim AR, Harty JT. Regulation of antigen-specific CD8+ T cell homeostasis by perforin and interferon-gamma. [comment] Science. 2000;290:1354–1358. doi: 10.1126/science.290.5495.1354. [DOI] [PubMed] [Google Scholar]
- 16.Harty JT, Badovinac VP. Influence of effector molecules on the CD8(+) T cell response to infection. Curr Opin Immunol. 2002;14:360–365. doi: 10.1016/S0952-7915(02)00333-3. [DOI] [PubMed] [Google Scholar]
- 17.Bem RA, Bos AP, Bots M, Wolbink AM, van Ham SM, Medema JP, Lutter R, van Woensel JBM. Activation of the granzyme pathway in children with severe respiratory syncytial virus infection. Pediatr Res. 2008;63:650–655. doi: 10.1203/PDR.0b013e31816fdc32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Grossman WJ, Ley TJ. Granzymes A and B are not expressed in human neutrophils. [comment] Blood. 2004;104:906–907. doi: 10.1182/blood-2004-03-0858. [DOI] [PubMed] [Google Scholar]
- 19.Wagner C, Iking-Koner C, Denefleh B, Stegmaier S, Hug F, Hansch G. Granzyme B and perforin: constitutive expression in human polymorphonuclear neutrophils. Blood. 2004;103:1099–1104. doi: 10.1182/blood-2003-04-1069. [DOI] [PubMed] [Google Scholar]
- 20.Grossman WJ, Verbsky JW, Barchet W, Colonna M, Atkinson JP, Ley TJ. Human T regulatory cells can use the perforin pathway to cause autologous cell death. Immunity. 2004;21:589–601. doi: 10.1016/j.immuni.2004.09.002. [DOI] [PubMed] [Google Scholar]
- 21.Grossman WJ, Verbsky JW, Tollefson BL, Kemper C, Atkinson JP, Ley TJ. Differential expression of granzymes A and B in human cytotoxic lymphocyte subsets and T regulatory cells. Blood. 2004;104:2840–2848. doi: 10.1182/blood-2004-03-0859. [DOI] [PubMed] [Google Scholar]
- 22.Gondek DC, Lu LF, Quezada SA, Sakaguchi S, Noelle RJ. Contact-mediated supppressioin by CD4+CD25+ regulatory cells involves a granzyme B-dependent, perforin-independent mechanism. J Immunol. 2005;174:1783–1786. doi: 10.4049/jimmunol.174.4.1783. [DOI] [PubMed] [Google Scholar]
- 23.Hashimoto S, Kobayashi A, Kooguchi K, Kitamura Y, Onodera H, Nakajima H. Upregulation of two death pathways of perforin/granzyme and FasL/Fas in septic acute respiratory distress syndrome. Am J Respir Crit Care Med. 2000;161:237–243. doi: 10.1164/ajrccm.161.1.9810007. [DOI] [PubMed] [Google Scholar]
- 24.Miyazaki H, Kuwano K, Yoshida K, Maeyama T, Yoshimi M, Fujita M, Hagimoto N, Yoshida R, Nakanishi Y. The perforin mediated apoptotic pathway in lung injury and fibrosis. J Clin Pathol. 2004;57:1292–1298. doi: 10.1136/jcp.2003.015495. [DOI] [PMC free article] [PubMed] [Google Scholar]


