Highlights
► IFN therapies have been plagued by severe side effects. ► Discovery of PRRs rejuvenated interest for immunomodulatory therapies. ► Panviral therapeutics will target key regulators of innate immune responses. ► Future targeted immunomodulatory therapies will reduce side effects. ► Panviral therapeutics in combination with DAAs to achieve viral eradication.
Abstract
Interferons (IFNs) have long been used as an immunomodulatory therapy for a large array of acute and chronic viral infections. However, IFN therapies have been plagued by severe side effects. The discovery of pathogen recognition receptors (PRR) rejuvenated the interest for immunomodulatory therapies. The successes obtained with Toll-like receptor (TLR) agonists in activating immune cells and as adjuvant for prophylactic vaccines against different viruses paved the way to targeted immunomodulatory therapy. Better characterization of pathogen-induced immune disorders and newly discovered regulators of innate immunity have now the potential to specifically withdraw prevailing subversion mechanisms and to transform antiviral treatments by introducing panviral therapeutics with less adverse effects than IFN therapies.
Current Opinion in Virology 2012, 2:622–628
This review comes from a themed issue on Antivirals and resistance
Edited by Daniel Lamarre and Mark A Wainberg
For a complete overview see the Issue and the Editorial
Available online 25th September 2012
1879-6257/$ – see front matter, © 2012 Elsevier B.V. All rights reserved.
Introduction
The innate immune system is the first line of defense for organisms that possess an adaptive immune system. It relies on the presence of specific receptors able to recognize recurring pattern in molecules associated with pathogens but not with host cells, allowing discrimination between self and non-self. These receptors are named pattern recognition receptors (PRR) and recognized pathogen-associated molecular patterns (PAMP) to induce the expression of cytokines and chemokines that restrict dissemination, eliminate pathogens and instruct pathogen-specific adaptive immune responses. In the recent years, tremendous advances in the characterization of PRR families, nucleic acid sensing, downstream signaling pathways and effector responses have revealed essential role of novel proteins and dynamic protein interactions network in the triggering of immune responses to intracellular pathogen such as viruses. In the near future, targeting specific regulators of PRR-mediated innate response to withdraw viral subversion mechanisms, and access to novel surrogate measurable effector markers, hold the promise of new panviral therapeutics that will minimize adverse effects associated with type I IFN therapy. This review briefly summarizes strategies and challenges of present and future targeted immunomodulatory therapies according to our increasing knowledge in regulation of innate immunity and of virus-induced immune host dysfunction.
Toward a better understanding of the innate immune response to viral infection
Signaling PPRs include the major families of Toll-like receptors (TLRs), retinoic acid-inducible gene I (RIG-I)-like receptors (RLRs) and nucleotide-binding oligomerization domain (NOD)-like receptors (NLRs). Pathogen sensing takes place in all nucleated cells to generate cell-intrinsic innate immunity and in professional antigen presenting cells (APCs) to promote specific adaptive immune responses. While TLRs sense PAMPs in the extracellular space and endosomes, RLRs and NLRs function as pathogen sensors in intracellular compartments [1]. Interestingly, only a few of the known 13 TLRs have the ability to recognize viral molecules: TLR3 for viral dsRNA, TLR7/8 for viral ssRNA and TLR9 for viral unmethylated CpG DNA. Three cytosolic sensors of viral RNA have been characterized thus far: RIG-I for the sensing of 5′ triphosphate structure and blunt-end base paring, MDA5 for the sensing of long dsRNA and LGP2 a CARDless regulator of its counterparts [2]. Following their activation, the CARD domain of RIG-I and MDA5 interacts with the CARD domain of the signaling adaptor MAVS (mitochondrial antiviral signaling protein) [3••]. Both TLR and RLR viral sensing pathways converge to activate IFN regulatory factor IRF3-mediated and IRF7-mediated type I IFN (α/β) antiviral response and NF-κB-mediated inflammatory pathway [4] (Figure 1 ). Recent studies aims at better defining innate immune responses have identified several novel signaling and regulatory molecules [5]. Global proteomic analysis has further revealed signaling modules with high interconnectivity and adaptor proteins regulating signalosome assembly upon antiviral response and type I IFN production [6••].
Figure 1.
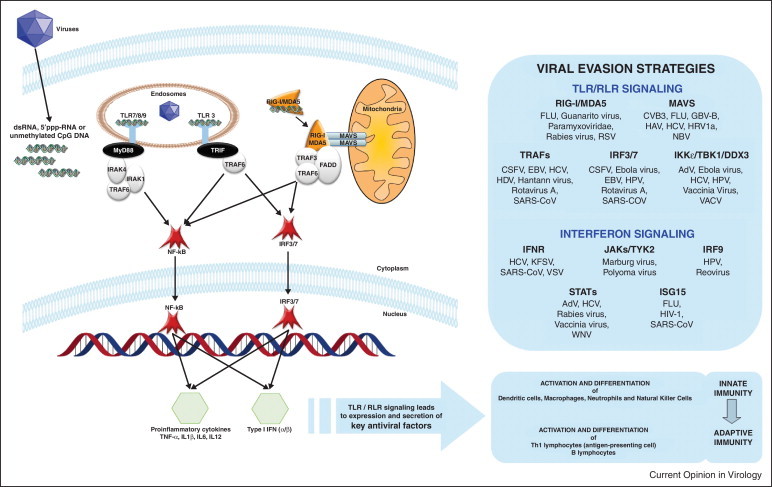
TLR and RLR signaling. Viral nucleic acids are recognized by endosomal and cytoplasmic PRRs. Activation of MYD88-dependant TLR7/8/9 signaling, TRIF-dependant TLR3 signaling and RIG-I/MDA5 signaling results in nuclear translocation of IRF3/7 and NF-κB transcriptional factors, leading to type I IFN and proinflammatory cytokines production. Effectors of innate immune response allow mounting of an optimal adaptive immune response. Viral evasion strategies are also identified that interfere with TLR/RLR and IFN signaling pathways. Abbreviations: AdV: Adenovirus, CSFV: Classical Swine Fever Virus, CVB3: Coxsackievirus B3, EBV: Epstein-Barr Virus, GBV-B: GB virus B, HAV/HBV/HCV/HDV: Hepatitis A/B/C/D virus, HIV-1: Human Immunodeficiency Virus type 1, HPV: Human Papillomavirus, HRV1a: Human Rhinovirus 1a, KFSV: Kaposi's Sarcoma Virus, RSV: Respiratory Syncytial Virus, SARS-CoV: SARS coronavirus, VACV: Vaccinia Virus, VSV: Vesicular Stomatitis Virus, WNV: West Nile Virus.
PRR signaling in initiation of specific adaptive immune response
TLR-mediated and RLR-mediated antiviral responses take place at the site of infection in nonimmune cells and resting immune cells, where secreted pro-inflammatory cytokines and type I IFNs increase expression of MHC class II antigens, CD40 and CD86 on APCs [7]. Cytokines produced at sites of infection play a key role in the activation and differentiation of dendritic cells (DC), macrophages, neutrophils and NK cells, all major players of the innate immune response [8] (Figure 1). When mature DCs detect virus derived antigens, they migrate to the lymph nodes to present antigens to CD4+ and CD8+ T cells and B cells, inducing their activation [9]. Thus, modulation of PRR-mediated antiviral responses can have important ripple effects on both qualitative and quantitative aspects of the specific adaptive immune responses to maximize the therapeutic potential of immunomodulatory drugs [10].
Negative regulation of innate immune response and pathological consequences
Antiviral innate response must be tightly regulated in order to prevent uncontrolled production of cytokines that might have deleterious effects on the host. Type I IFN signature induced by PRR activation has been observed in diverse autoimmune disorders including diabetes, and is believed to play a role in the induction of chronic inflammatory disorders such as asthma and rheumatoid arthritis. In the recent years, a better picture has emerged in the biology of regulators illustrating the existence of numerous negative regulators that often play a nonredundant role and target the same positive regulator [5]. Many negative regulators have been characterized that are either involved in direct interaction with PRRs, dissociation of adaptors complexes, degradation of signal proteins or transcriptional regulation [12]. Post-translational modifications (phosphorylation and ubiquitination) have emerged as key mechanisms to regulate innate immune responses. Degradation of signal proteins mediated by the ubiquitin-proteasome and autophagy systems plays crucial roles in negative regulation of TLR signaling, and unlike disruption of adaptors contributes to termination of signaling as these degradations are irreversible [11]. Examples include proteins SOCS and PIN1 that promote polyubiquitination and proteasomal degradation of Mal adaptor and IRF3/7 respectively, to suppress type I IFN and antiviral responses. Recently, miRNAs have also emerged as fine tuners of innate immune responses, which target mRNAs encoding TLRs, intracellular signaling proteins and cytokines. Examples include miR-146 that targets IRAK1 and TRAF6, and miR-155 that targets MYD88, TAB2 and IKKɛ [12]. Thus, targeting specific negative regulator of the innate immune response may offer a new immunotherapeutic strategy to treat a range of infectious and inflammatory diseases [13].
Viral subversion mechanisms
Cellular defence has evolutionarily challenged viruses that in turn have developed strategies to counteract innate immune response. Indeed TLR and RLR sensing pathways are fundamental targets for virus-encoded immune suppression. These viral subversion mechanisms include recruitment of ubiquitin proteasome system, mimicry of the host cell components and sequestration and cleavage of key components of the immune system. One notable example is MAVS adaptor that is targeted by numerous viruses through proteolytic cleavage by hepatitis C virus (HCV), hepatitis A virus (HAV), Coxsackievirus B3 (CVB3), human rhinovirus 1a (HRV1a) and GB virus B (GBV-B), through decrease of the mitochondrial membrane potential by influenza A virus (FLU) or through inhibition of its interaction with RIG-I by hepatitis B virus (HBV). Processes of viral evasion are varied and are beyond the scope of this review, but are recapitulated in Figure 1 (reviewed in [14]). Importantly, host proteins targeted by multiples viruses highlight key players of innate immunity, which represent potential therapeutic targets to restore antiviral response and eventually cure cells from viruses. However, these specific viral evasion strategies must also be taken into account when developing immunomodulatory therapeutics to provide the greatest clinical benefits.
IFNs: pioneer of panviral therapies
Type I IFNs were rapidly used as a therapeutic agent against HBV and HCV, and demonstrated antiviral activity against infection with SARS-CoV [15], FLU [16], West Nile virus (WNV) [17], yellow fever virus (YFV) [18] and Ebola virus [19]. Refinement of therapies was explored with the development of improved IFN molecules like consensus interferon (CIFN: a completely synthetic interferon) [20], albinterferon (a fusion protein between IFNα2a and human albumin) [21] and Y shape interferon [22]. Recently, virus-induced type III IFNs (IFN-λ1-3: IL-29, IL28A, IL28B) have gained a lot of interest to treat viral infections since naturally occurring variants of the IL28B gene have been a major prediction factor in spontaneous and treatment-induced clearance of HCV [23, 24]. Early clinical trials of recombinant pegylated-IFN-λ1 in HCV-infected patients showed reduced adverse effects compared to IFN-α, likely linked to minimal expression of IFN-λ receptors in hematopoietic cells [25, 26].
TLR targeted therapies (Table 1)
Table 1.
Development status of TLR-targeting molecules for treatment of viral infections.
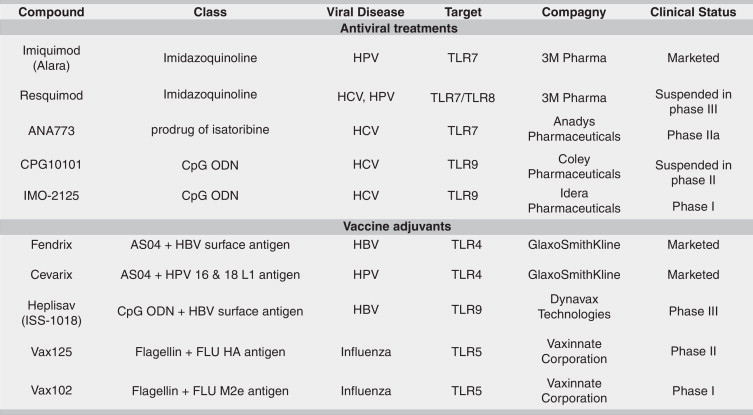
The discovery of TLRs heralded the rebirth of interest in innate immunity. Their specificity in recognizing most classes of pathogens, as well as their role in the pathogenesis of multiple diseases represent the strongest evidences that TLRs are valuable therapeutic targets. TLR targeted drugs have been approved and small-molecule compounds are being investigated in the treatment of viral infections as stand-alone treatment or adjunct to direct acting antivirals (DAAs).
Imidazoquinolines
The most advanced examples of TLR agonists are Imiquimod (Aldara, 3M) and Resiquimod (R-848, 3M), which are members of the imidazoquinolinamines [27•]. Imiquimod is the only approved TLR7 agonist and is used for topical treatment of external genital and perianal warts resulting from human papillomavirus (HPV) infection [28]. Resiquimod is a mixed TLR7/8 agonist that reached phase III trial for the treatment of genital herpes before being suspended due to a lack of efficacy [29].
Isatoribine
ANA-773 (Anadys Pharmaceuticals) is a second generation of orally bioavailable prodrug of isatoribine that signals through TLR7, which is expressed in B cells and DCs [30]. In HCV infected patients, ANA-773 was generally well tolerated and resulted in a significant −1.26 log10 decrease in HCV RNA levels following 10 days of treatments [31]. ANA-773 is now assessed in phase IIa, and its efficacy will be evaluated in combination with ribavirin and DAAs as an IFN replacement.
Immunomodulatory oligonucleotides
Synthetic cytosine-phosphate-guanine containing oligodeoxynucleotides (CpG-ODNs) are potent TLR9 agonists, which interact directly with DCs to stimulate cytokine release and induce adaptive immune responses [32]. In Phase I clinical trials, subcutaneously administration of IMO-2125 (Idera Pharmaceuticals) as monotherapy resulted in a more than −1 log10 decrease in HCV RNA levels in prior nonresponders to PEG-IFN/ribavirin after 4 weeks [33], and in combination with ribavirin to a −2.4 log10 decrease in HCV RNA in treatment-naïve patients at day 29 [34•, 35]. On the basis of its efficacy, IMO-2125 could provide an alternative to IFNs for HCV therapy. However, Idera Pharmaceuticals delayed a phase II study after the observation of atypical lymphocytic proliferation in preclinical toxicology study.
Vaccine adjuvants using TLR agonists
TLR agonists have been an extensively explored area in the development of vaccine adjuvants for prophylactic and therapeutic applications by linking innate and adaptive immune systems. The proof-of-concept of this approach was made with the AS04 adjuvant system that combines monophosphoryl lipid A (MPLA), an agonist of the TLR4 receptor and aluminium salt [36, 37, 38]. AS04 has been approved in prophylactic vaccine against HBV (Fendrix, GlaxoSmithKline) [39] and HPV 16 and 18 (Cervarix, GlaxoSmithKline) [40]. The mechanism of action of AS04 is mediated by a transient and local activation of NF-kB activity and cytokine production, thus providing an innate immune signal for optimal activation of APCs [41]. Other notable examples of adjuvants in clinical development are Heplisav and VaxInnate. Heplisav is a HBV vaccine comprised of an immunostimulatory sequence (ISS-1018, Dynavax Technologies) that targets TLR9 receptor and HBV surface antigen. In phase III clinical trials, Heplisav demonstrated earlier and higher protection with fewer doses than currently licensed vaccines [42•]. VaxInnate Corporation is developing vaccines using highly conserved influenza immunogens fused to TLR5 agonist Salmonella typhimurium flagellin type 2 as an adjuvant to potentially protect against all strains of seasonal and pandemic FLU strains (VAX102, VAX125, VAX128 and VAX168) [43, 44, 45].
Future immunomodulatory targeted therapy and panviral approaches (Table 2)
Table 2.
Current and future development of immunomodulatory targeted therapy [49••, 50, 51, 52, 53, 54, 55, 56].

In the past decade, many newly emerging or re-emerging virus infections and fear of future pandemics have accentuated the need for novel antiviral therapy. Panviral therapeutics with a targeted therapy approach would be an ideal treatment for acute and chronic viral infections, either as a standalone treatment or in combination with DAAs. The major challenge in developing future immunomodulatory therapy will be to minimize adverse effects. The aggravation of psoriatic plaques in HPV-infected patients treated with Imiquimod illustrates that triggering innate immune responses can lead to uncontrolled activation of the inflammatory response. Furthermore, immunomodulatory molecules, such as peptidoglycans, that bind to multiple PRRs (TLR2, NOD proteins and peptidoglycan recognition proteins) increase the risk of undesired side effects. Development of therapeutics will require more extensive structural information of receptor–ligand interaction to maximize the specificity and avoid undesired interactions.
The selection of specific targets will require a comprehensive knowledge of innate immunity signaling pathways and regulators that are induced by and common to numerous viral infections. The mapping of an innate immune protein interaction network regulating IFNB1 has revealed signaling modules with high interconnectivity including MAVS, TBK1 and IRAK [6••]. Each module interacts with many signaling proteins of the pathway offering multiple drug targets with specific immune effector function. Using a genome-wide RNAi screen assessing virus-induced IFNB1 transcription in human cells, we identified novel proteins and pathways capable of negatively and positively regulating innate immune responses (unpublished data). Comprehensive epistasis analysis of the various regulators acting at different steps of the antiviral responses from virus sensing, signal propagation/amplification up to feedback regulation, offers valuable information for selection of drug targets. In principle, strategies of targeted therapy could include small molecule-mediated activation of positive regulators or inhibition of negative regulators. An example of targeting a negative regulator could be the immuno-miRNA miR-155, which is induced by virus infection and downregulate MYD88, IRAK3, TAB2 and IKKɛ gene expression to suppress TLR signaling [12]. Silencing miR-155 function using antagomirs or locked nucleic acid (LNA) in infected cells could potentially restore TLR signaling.
A better knowledge of surrogate end point measurable makers of immune effector function (correlating with pan antiviral efficacy) in relevant infected biological material will undoubtedly enhance selection process and therapeutic value of drug targets. Indeed, microarray analysis of infected primary cells can be used to identify early and late response innate immune genes, as well as virus-mediated inhibition of these genes [46, 47, 48]. Finally, the knowledge of virus-induced immune host dysfunction and of immune proteins targeted by multiples viruses will validate key viral host interfaces, leading to hypothesis-driven selection of therapeutic targets intended to restore innate immune responses.
Conclusions
TLRs agonists reflect substantial promise as therapeutic targets and demonstrate the huge potential of targeting innate immunity in fighting viral infections. In the future, integration of structural, proteomics and functional genomics data will pave the way to the identification of key regulators of innate immunity. Targeting immune regulators that promote PRR signaling to maintain transient activation of innate immune responses upon viral infection should pioneer the discovery of panviral therapeutics. Such targeted immunomodulatory therapy approach could change the way we treat infectious diseases by allowing a single treatment to be effective against numerous viruses, with minimal viral breakthrough. In the near future, the increasing availability and potency of new targeted immunomodulatory panviral therapeutics could allow the re-thinking of temporal aspects of treatments that, in combination with available DAAs, could achieve viral eradication. The ultimate goal is to shape TLR-dependent and RLR-dependent innate immune responses to restore antiviral effects and to generate an optimal global immune response, while controlling inflammation.
References and recommended reading
Papers of particular interest, published within the period of review, have been highlighted as:
• of special interest
•• of outstanding interest
Acknowledgements
This work was supported by grants from the Canadian Institutes for Health Research (CIHR-CI6-103135) and by the Novartis/Canadian Liver Foundation Hepatology Research Chair to DL. NT is a recipient of a Ph.D. scholarship from the Fonds de Recherche en Santé du Québec (FRSQ).
References
- 1.Takeuchi O., Akira S. Pattern recognition receptors and inflammation. Cell. 2010;140:805–820. doi: 10.1016/j.cell.2010.01.022. [DOI] [PubMed] [Google Scholar]
- 2.Loo Y.M., Gale M., Jr. Immune signaling by RIG-I-like receptors. Immunity. 2011;34:680–692. doi: 10.1016/j.immuni.2011.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3••.Hou F., Sun L., Zheng H., Skaug B., Jiang Q.X., Chen Z.J. MAVS forms functional prion-like aggregates to activate and propagate antiviral innate immune response. Cell. 2011;146:448–461. doi: 10.1016/j.cell.2011.06.041. [DOI] [PMC free article] [PubMed] [Google Scholar]; Describes how viral infection induces the formation of prion-like aggregates of MAVS on mitochondria that potently activate IRF3.
- 4.Kawai T., Akira S. Toll-like receptors and their crosstalk with other innate receptors in infection and immunity. Immunity. 2011;34:637–650. doi: 10.1016/j.immuni.2011.05.006. [DOI] [PubMed] [Google Scholar]
- 5.Eisenacher K., Krug A. Regulation of RLR-mediated innate immune signaling — it is all about keeping the balance. Eur J Cell Biol. 2012;91:36–47. doi: 10.1016/j.ejcb.2011.01.011. [DOI] [PubMed] [Google Scholar]
- 6••.Li S., Wang L., Berman M., Kong Y.Y., Dorf M.E. Mapping a dynamic innate immunity protein interaction network regulating type I interferon production. Immunity. 2011;35:426–440. doi: 10.1016/j.immuni.2011.06.014. [DOI] [PMC free article] [PubMed] [Google Scholar]; Proteomic mapping of ligand-dependent interactions reveals dynamic remodeling of the human innate immunity interactome.
- 7.Hervas-Stubbs S., Perez-Gracia J.L., Rouzaut A., Sanmamed M.F., Le Bon A., Melero I. Direct effects of type I interferons on cells of the immune system. Clin Cancer Res. 2011;17:2619–2627. doi: 10.1158/1078-0432.CCR-10-1114. [DOI] [PubMed] [Google Scholar]
- 8.Wang B.X., Fish E.N. The yin and yang of viruses and interferons. Trends Immunol. 2012;33:190–197. doi: 10.1016/j.it.2012.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gao Y., Majchrzak-Kita B., Fish E.N., Gommerman J.L. Dynamic accumulation of plasmacytoid dendritic cells in lymph nodes is regulated by interferon-beta. Blood. 2009;114:2623–2631. doi: 10.1182/blood-2008-10-183301. [DOI] [PubMed] [Google Scholar]
- 10.Desmet C.J., Ishii K.J. Nucleic acid sensing at the interface between innate and adaptive immunity in vaccination. Nat Rev Immunol. 2012;12:479–491. doi: 10.1038/nri3247. [DOI] [PubMed] [Google Scholar]
- 11.Kato H., Takahasi K., Fujita T. RIG-I-like receptors: cytoplasmic sensors for non-self RNA. Immunol Rev. 2011;243:91–98. doi: 10.1111/j.1600-065X.2011.01052.x. [DOI] [PubMed] [Google Scholar]
- 12.O’Neill L.A., Sheedy F.J., McCoy C.E. MicroRNAs: the fine-tuners of Toll-like receptor signalling. Nat Rev Immunol. 2011;11:163–175. doi: 10.1038/nri2957. [DOI] [PubMed] [Google Scholar]
- 13.Kondo T., Kawai T., Akira S. Dissecting negative regulation of Toll-like receptor signaling. Trends Immunol. 2012 doi: 10.1016/j.it.2012.05.002. [DOI] [PubMed] [Google Scholar]
- 14.Ramos H.J., Gale M., Jr. RIG-I like receptors and their signaling crosstalk in the regulation of antiviral immunity. Curr Opin Virol. 2011;1:167–176. doi: 10.1016/j.coviro.2011.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kumaki Y., Day C.W., Wandersee M.K., Schow B.P., Madsen J.S., Grant D., Roth J.P., Smee D.F., Blatt L.M., Barnard D.L. Interferon alfacon 1 inhibits SARS-CoV infection in human bronchial epithelial Calu-3 cells. Biochem Biophys Res Commun. 2008;371:110–113. doi: 10.1016/j.bbrc.2008.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gao L., Yu S., Chen Q., Duan Z., Zhou J., Mao C., Yu D., Zhu W., Nie J., Hou Y. A randomized controlled trial of low-dose recombinant human interferons alpha-2b nasal spray to prevent acute viral respiratory infections in military recruits. Vaccine. 2010;28:4445–4451. doi: 10.1016/j.vaccine.2010.03.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kalil A.C., Devetten M.P., Singh S., Lesiak B., Poage D.P., Bargenquast K., Fayad P., Freifeld A.G. Use of interferon-alpha in patients with West Nile encephalitis: report of 2 cases. Clin Infect Dis. 2005;40:764–766. doi: 10.1086/427945. [DOI] [PubMed] [Google Scholar]
- 18.Julander J.G., Ennis J., Turner J., Morrey J.D. Treatment of yellow fever virus with an adenovirus-vectored interferon DEF201, in a hamster model. Antimicrob Agents Chemother. 2011;55:2067–2073. doi: 10.1128/AAC.01635-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Jahrling P.B., Geisbert T.W., Geisbert J.B., Swearengen J.R., Bray M., Jaax N.K., Huggins J.W., LeDuc J.W., Peters C.J. Evaluation of immune globulin and recombinant interferon-alpha2b for treatment of experimental Ebola virus infections. J Infect Dis. 1999;179(Suppl. 1):S224–S234. doi: 10.1086/514310. [DOI] [PubMed] [Google Scholar]
- 20.Alavian S.M., Behnava B., Tabatabaei S.V. Comparative efficacy and overall safety of different doses of consensus interferon for treatment of chronic HCV infection: a systematic review and meta-analysis. Eur J Clin Pharmacol. 2010;66:1071–1079. doi: 10.1007/s00228-010-0881-7. [DOI] [PubMed] [Google Scholar]
- 21.Flisiak R., Flisiak I. Albinterferon-alpha 2b: a new treatment option for hepatitis C. Expert Opin Biol Ther. 2010;10:1509–1515. doi: 10.1517/14712598.2010.521494. [DOI] [PubMed] [Google Scholar]
- 22.Ashour MKF: Clinical trial of the efficacy, dosing, safety and tolerability of Y-shaped pegylated interferon (YPEG-IFNα-2a) plus ribavirin in egyptian patients with untreated chronic hepatitis C. In American Association for the study of liver diseases. San Francisco, CA; 2011.
- 23.Ge D., Fellay J., Thompson A.J., Simon J.S., Shianna K.V., Urban T.J., Heinzen E.L., Qiu P., Bertelsen A.H., Muir A.J. Genetic variation in IL28B predicts hepatitis C treatment-induced viral clearance. Nature. 2009;461:399–401. doi: 10.1038/nature08309. [DOI] [PubMed] [Google Scholar]
- 24.Thomas D.L., Thio C.L., Martin M.P., Qi Y., Ge D., O’Huigin C., Kidd J., Kidd K., Khakoo S.I., Alexander G. Genetic variation in IL28B and spontaneous clearance of hepatitis C virus. Nature. 2009;461:798–801. doi: 10.1038/nature08463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Muir A.J., Shiffman M.L., Zaman A., Yoffe B., de la Torre A., Flamm S., Gordon S.C., Marotta P., Vierling J.M., Lopez-Talavera J.C. Phase 1b study of pegylated interferon lambda 1 with or without ribavirin in patients with chronic genotype 1 hepatitis C virus infection. Hepatology. 2010;52:822–832. doi: 10.1002/hep.23743. [DOI] [PubMed] [Google Scholar]
- 26.Donnelly R.P., Kotenko S.V. Interferon-lambda: a new addition to an old family. J Interferon Cytokine Res. 2010;30:555–564. doi: 10.1089/jir.2010.0078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27•.Hedayat M., Netea M.G., Rezaei N. Targeting of Toll-like receptors: a decade of progress in combating infectious diseases. Lancet Infect Dis. 2011;11:702–712. doi: 10.1016/S1473-3099(11)70099-8. [DOI] [PubMed] [Google Scholar]; Comprehensive review on the therapeutic targeting of TLRs in infectious diseases either as antimicrobial treatment or vaccine adjuvants.
- 28.Wagstaff A.J., Perry C.M. Topical imiquimod: a review of its use in the management of anogenital warts, actinic keratoses, basal cell carcinoma and other skin lesions. Drugs. 2007;67:2187–2210. doi: 10.2165/00003495-200767150-00006. [DOI] [PubMed] [Google Scholar]
- 29.Pockros P.J., Guyader D., Patton H., Tong M.J., Wright T., McHutchison J.G., Meng T.C. Oral resiquimod in chronic HCV infection: safety and efficacy in 2 placebo-controlled, double-blind phase IIa studies. J Hepatol. 2007;47:174–182. doi: 10.1016/j.jhep.2007.02.025. [DOI] [PubMed] [Google Scholar]
- 30.Kronenberger B., Zeuzem S. Current and future treatment options for HCV. Ann Hepatol. 2009;8:103–112. [PubMed] [Google Scholar]
- 31.Bergmann J.F., de Bruijne J., Hotho D.M., de Knegt R.J., Boonstra A., Weegink C.J., van Vliet A.A., van de Wetering J., Fletcher S.P., Bauman L.A. Randomised clinical trial: anti-viral activity of ANA773, an oral inducer of endogenous interferons acting via TLR7, in chronic HCV. Aliment Pharmacol Ther. 2011;34:443–453. doi: 10.1111/j.1365-2036.2011.04745.x. [DOI] [PubMed] [Google Scholar]
- 32.Vicari A.P., Schmalbach T., Lekstrom-Himes J., Morris M.L., Al-Adhami M.J., Laframboise C., Leese P., Krieg A.M., Efler S.M., Davis H.L. Safety, pharmacokinetics and immune effects in normal volunteers of CPG 10101 (ACTILON), an investigational synthetic toll-like receptor 9 agonist. Antivir Ther. 2007;12:741–751. [PubMed] [Google Scholar]
- 33.Muir A., Ghalib R., Lawitz E., Patel K., Rodriguez-torres M., Sheikh A., Sapp S., Taylor R., Bexon A., Sullivan T. A phase 1, multi-center, randomized, placebo-controlled, dose-escalation study of IMO-2125, a TLR9 agonist, in hepatitis C nonresponders. EASL 45th Annual Meeting April; Vienna, Austria; 2010. [Google Scholar]
- 34•.Horscroft N.J., Pryde D.C., Bright H. Antiviral applications of Toll-like receptor agonists. J Antimicrob Chemother. 2012;67:789–801. doi: 10.1093/jac/dkr588. [DOI] [PubMed] [Google Scholar]; Update on the development of antiviral TLR agonists including description of the chemistry applied in the develpment of the therapeutic agents.
- 35.Guyader D., Bogomolov P., Kobalava Z., Moiseev V., Szlavik J., Astruc B., Varkonyi I., Sullivan T., Horgan K., Bexon A. IMO-2125 plus ribavirin gives substantial first-dose viral load reductions, cumulative antiviral effect, is well tolerated in naïve genotype 1 HCV patients: a phase 1 trial. EASL 46th Annual Meeting March; Berlin, Germany; 2011. [Google Scholar]
- 36.Mata-Haro V., Cekic C., Martin M., Chilton P.M., Casella C.R., Mitchell T.C. The vaccine adjuvant monophosphoryl lipid A as a TRIF-biased agonist of TLR4. Science. 2007;316:1628–1632. doi: 10.1126/science.1138963. [DOI] [PubMed] [Google Scholar]
- 37.Gavin A.L., Hoebe K., Duong B., Ota T., Martin C., Beutler B., Nemazee D. Adjuvant-enhanced antibody responses in the absence of toll-like receptor signaling. Science. 2006;314:1936–1938. doi: 10.1126/science.1135299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.van Duin D., Medzhitov R., Shaw A.C. Triggering TLR signaling in vaccination. Trends Immunol. 2006;27:49–55. doi: 10.1016/j.it.2005.11.005. [DOI] [PubMed] [Google Scholar]
- 39.Kundi M. New hepatitis B vaccine formulated with an improved adjuvant system. Expert Rev Vaccines. 2007;6:133–140. doi: 10.1586/14760584.6.2.133. [DOI] [PubMed] [Google Scholar]
- 40.Schwarz T.F. Clinical update of the AS04-adjuvanted human papillomavirus-16/18 cervical cancer vaccine, Cervarix. Adv Ther. 2009;26:983–998. doi: 10.1007/s12325-009-0079-5. [DOI] [PubMed] [Google Scholar]
- 41.Didierlaurent A.M., Morel S., Lockman L., Giannini S.L., Bisteau M., Carlsen H., Kielland A., Vosters O., Vanderheyde N., Schiavetti F. AS04, an aluminum salt- and TLR4 agonist-based adjuvant system, induces a transient localized innate immune response leading to enhanced adaptive immunity. J Immunol. 2009;183:6186–6197. doi: 10.4049/jimmunol.0901474. [DOI] [PubMed] [Google Scholar]
- 42•.Harandi A.M., Davies G., Olesen O.F. Vaccine adjuvants: scientific challenges and strategic initiatives. Expert Rev Vaccines. 2009;8:293–298. doi: 10.1586/14760584.8.3.293. [DOI] [PubMed] [Google Scholar]; Highlights the promise of TLR agonists in the development of novel vaccine adjuvants.
- 43.Talbot H.K., Rock M.T., Johnson C., Tussey L., Kavita U., Shanker A., Shaw A.R., Taylor D.N. Immunopotentiation of trivalent influenza vaccine when given with VAX102, a recombinant influenza M2e vaccine fused to the TLR5 ligand flagellin. PLoS ONE. 2010;5:e14442. doi: 10.1371/journal.pone.0014442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Taylor D.N., Treanor J.J., Sheldon E.A., Johnson C., Umlauf S., Song L., Kavita U., Liu G., Tussey L., Ozer K. Development of VAX128, a recombinant hemagglutinin (HA) influenza-flagellin fusion vaccine with improved safety and immune response. Vaccine. 2012 doi: 10.1016/j.vaccine.2012.06.086. [DOI] [PubMed] [Google Scholar]
- 45.Treanor J.J., Taylor D.N., Tussey L., Hay C., Nolan C., Fitzgerald T., Liu G., Kavita U., Song L., Dark I. Safety and immunogenicity of a recombinant hemagglutinin influenza-flagellin fusion vaccine (VAX125) in healthy young adults. Vaccine. 2010;28:8268–8274. doi: 10.1016/j.vaccine.2010.10.009. [DOI] [PubMed] [Google Scholar]
- 46.Jouan L., Chatel-Chaix L., Melancon P., Rodrigue-Gervais I.G., Raymond V.A., Selliah S., Bilodeau M., Grandvaux N., Lamarre D. Targeted impairment of innate antiviral responses in the liver of chronic hepatitis C patients. J Hepatol. 2011 doi: 10.1016/j.jhep.2011.07.017. [DOI] [PubMed] [Google Scholar]
- 47.Jouan L., Melancon P., Rodrigue-Gervais I.G., Raymond V.A., Selliah S., Boucher G., Bilodeau M., Grandvaux N., Lamarre D. Distinct antiviral signaling pathways in primary human hepatocytes and their differential disruption by HCV NS3 protease. J Hepatol. 2010;52:167–175. doi: 10.1016/j.jhep.2009.11.011. [DOI] [PubMed] [Google Scholar]
- 48.Rodrigue-Gervais I.G., Jouan L., Beaule G., Sauve D., Bruneau J., Willems B., Sekaly R.P., Lamarre D. Poly(I:C) and lipopolysaccharide innate sensing functions of circulating human myeloid dendritic cells are affected in vivo in hepatitis C virus-infected patients. J Virol. 2007;81:5537–5546. doi: 10.1128/JVI.01741-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49••.Botos I., Segal D.M., Davies D.R. The structural biology of Toll-like receptors. Structure. 2011;19:447–459. doi: 10.1016/j.str.2011.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]; Highlights the variation in the nature of the interactions of TLR paralog extracellular domains following ligand-induced dimerization.
- 50.Ireton R.C., Gale M., Jr. RIG-I like receptors in antiviral immunity and therapeutic applications. Viruses. 2011;3:906–919. doi: 10.3390/v3060906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Jiang F., Ramanathan A., Miller M.T., Tang G.Q., Gale M., Jr., Patel S.S., Marcotrigiano J. Structural basis of RNA recognition and activation by innate immune receptor RIG-I. Nature. 2011;479:423–427. doi: 10.1038/nature10537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Luo D., Ding S.C., Vela A., Kohlway A., Lindenbach B.D., Pyle A.M. Structural insights into RNA recognition by RIG-I. Cell. 2011;147:409–422. doi: 10.1016/j.cell.2011.09.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Chakravarthy K.V., Bonoiu A.C., Davis W.G., Ranjan P., Ding H., Hu R., Bowzard J.B., Bergey E.J., Katz J.M., Knight P.R. Gold nanorod delivery of an ssRNA immune activator inhibits pandemic H1N1 influenza viral replication. Proc Natl Acad Sci U S A. 2010;107:10172–10177. doi: 10.1073/pnas.0914561107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Luke J.M., Simon G.G., Soderholm J., Errett J.S., August J.T., Gale M., Jr., Hodgson C.P., Williams J.A. Coexpressed RIG-I agonist enhances humoral immune response to influenza virus DNA vaccine. J Virol. 2011;85:1370–1383. doi: 10.1128/JVI.01250-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kobiyama K., Takeshita F., Ishii K.J., Koyama S., Aoshi T., Akira S., Sakaue-Sawano A., Miyawaki A., Yamanaka Y., Hirano H. A signaling polypeptide derived from an innate immune adaptor molecule can be harnessed as a new class of vaccine adjuvant. J Immunol. 2009;182:1593–1601. doi: 10.4049/jimmunol.182.3.1593. [DOI] [PubMed] [Google Scholar]
- 56.Gottwein E., Cullen B.R. Viral and cellular microRNAs as determinants of viral pathogenesis and immunity. Cell Host Microbe. 2008;3:375–387. doi: 10.1016/j.chom.2008.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]


