Abstract
During the last decade, the propagation of immunological knowledge describing the critical role of dendritic cells (DC) in the induction of efficacious immune responses has promoted research and development of vaccines systematically targeting DC. Based on the promise for the rational design of vaccine platforms, the current review will provide an update on particle-based vaccines of both viral and synthetic origin, giving examples of recombinant virus carriers such as adenoviruses and biodegradable particulate carriers. The viral carriers carry pathogen-associated molecular patterns (PAMP), used by the original virus for targeting DC, and are particularly efficient and versatile gene delivery vectors. Efforts in the field of synthetic vaccine carriers are focussing on decorating the particle surface with ligands for DC receptors such as heparan sulphate glycosaminoglycan structures, integrins, Siglecs, galectins, C-type lectins and toll-like receptors. The emphasis of this review will be placed on targeting the porcine immune system, but reference will be made to advances with murine and human vaccine delivery systems where information on DC targeting is available.
Keywords: Vaccine targeting, Dendritic cells, Particulate vaccines receptor ligands
1. “Targeting” for efficient vaccination
The ideal scenario for efficacious vaccination requires initiation of a potent immune response by a vaccine formulated to resemble the situation of the natural infection [1], [2]. In reality, vaccines are manufactured to resemble as close as possible the pathogen in question, but lacking a pathogenic nature; elaboration of this approach has sought to dissect the vaccine into the most pertinent immunogenic component(s) for promotion of protective immune defences. With virus vaccines based on the whole virus, approaches have included the application of attenuated variants as well as inactivated antigen. Moreover, a study of the evolution in vaccine design to combat viral infections shows how manipulations of the vaccine composition have increased the success rate for generating efficacious vaccines. An appreciation of these approaches, and their evolution, shows three main elements essential for successful vaccination. (i) The antigenic characteristics of the vaccine itself. (ii) The mode of vaccine delivery; until the last decade often ignored or relegated to the position of an “also-ran”. This has a particular significance in that efficient vaccine delivery relates directly to vaccine proficiency at targeting the host immune system. (iii) Adjuvant application, a major tool and often critically important component for an efficacious vaccine. This is a major provider of the “danger” signals necessary for activating the innate responses essential for promoting efficacious specific immune defences. Although viruses and bacterial components can themselves provide danger signals, these may require efficient replication of the pathogen, which is not possible with inactivated or subunit vaccines. Alternatively, only a week danger signal may be provided due to a low-level interaction with the appropriate receptors, or the presence of inhibitory signals.
Overall, the vaccine must be efficiently delivered to the appropriate compartments of the immune system, leading to the correct signalling of the immune system to process the vaccine, and promotion of efficacious vaccination. Accordingly, an important issue for vaccine design is consideration of the ligands carried by a number of pathogens, which interact with receptors on cells of the immune system. Adjuvants also contain components interacting with immune cells, particularly of the innate immune defences. In both respects, the involvement of dendritic cells (DC) is crucial, due to their role as central players in the immune defence development (Supplementary Fig. 1). DC ensure both appropriate recognition of danger signals and correct processing of the vaccine antigen, promoting efficacious development of innate and acquired immune defences [3].
2. Immune defence induction via vaccination: the critical role for DC
In order to appreciate how vaccines can be targeted to the immune system, for enhancing their efficacy, it is necessary to appreciate the central role played by the DC therein, because these are critical targets for vaccine delivery (Supplementary Fig. 1). Most inactivated, protein-based vaccines induce acquired (specific) immune responses involving MHC Class II-restricted T-helper (Th) and B lymphocytes: Antibody-dependent immune defences. An additional defence important for removing infected or “altered self” host cells is that involving MHC Class I-restricted cytotoxic T lymphocyte (CTL) responses, although these are less evident following vaccination with inactivated vaccines. Overall, the central and key player for inducing efficacious acquired immunity is the DC [4]. These areas are covered in detail elsewhere [3]. Following endocytosis of the vaccine, DC process the antigen towards presentation of derived peptides to T lymphocytes (in association with either MHC Class II or Class I molecules). Advances in murine and rat immunology have shown that DC may “deliver” antigen in a more intact form to B lymphocytes, releasing the antigen to stimulate the B lymphocytes [5]. Furthermore, DC release cytokines of major importance for B lymphocyte activity: type I interferon (IFN), IL-6, IL-12 and BAFF [6].
Efficacious vaccination is dependent on the efficiency with which the vaccine interacts with the DC subsets. This raises the question of vaccine delivery, which will influence how both conventional DC (cDC) and plasmacytoid DC (pDC) – as well as monocytes and macrophages – become involved. DC possess a broad spectrum of cell surface receptors involved in the initiation, promotion and execution of immune responses [7]; importantly, the expression patterns for these receptors vary between cDC and pDC. Among these are the TLR, scavenger receptors, and C-type lectin receptors (Fig. 1 ). All are important pattern recognition receptors (PRR), recognizing PAMP on pathogens – and indeed antigens, adjuvants and vaccines – essential for the development of innate immune responses and defences [8], [9]. With PAMP binding to PRR on the DC surface, they also offer potential as targets for vaccine delivery. Ligation of particular PRR evokes rapid activation of the cell, inducing pro-inflammatory cytokine release and upregulation of co-stimulatory molecules on leukocytes, as seen with DC and macrophages [9]. With other PRR, such as certain C-type lectin receptors, their ligation leads to enhanced endocytosis. For DC acting as antigen-presenting cells (APC), the ligation can also lead to induction of DC maturation—an essential step licensing these cells to induce potent effector as well as memory immune responses [10], [11]. A good example of PAMP ligating PRR to activate cDC is seen with bacterial lipopeptides and lipoproteins recognized by heterodimeric TLR2-TLR1 or TLR2-TLR6. Lipopeptides deliver a strong activation signal to DC, which is the basis of their potent activity as immunoadjuvants in vitro and in vivo, at least in rodents and primates [12], [13], [14]. C-type lectin PRR such as the multilectin DEC-205 receptor and the mannose receptor are more involved in enhanced endocytosis. Ligation of these receptors has also shown potential for vaccine targeting and activation of DC [15]. In the murine system, ligation of DEC-205 enhances receptor-dependent endocytic processes leading to efficient recycling of the antigen through late endosomes—such processing is likely to enhance antigen presentation to CD4+ helper T lymphocytes. In contrast, ligation of the mannose receptor tends more to promote recycling through the peripheral endosomal compartment.
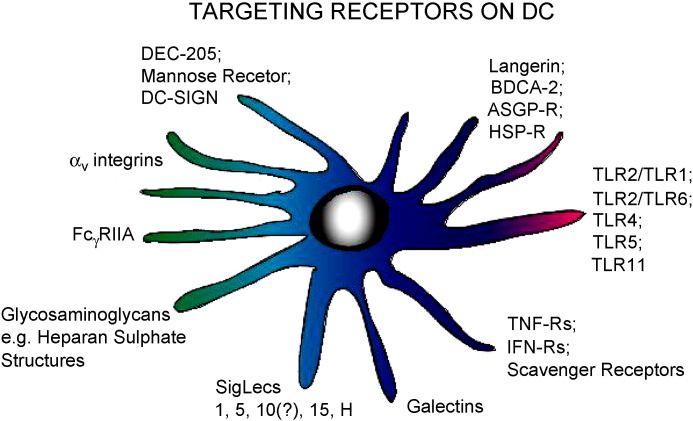
Fig. 1.
Examples of known receptors on DC, which may have potential for targeting vaccines, based on work performed with vaccine targeting to DC, or from analyses of the ligands with which the receptors interact.
These few examples of how targeting specific receptors on DC can result in a different form of signalling to the DC is an important consideration for targeting vaccines to the immune system. Dependent on the receptor being targeted, the antigen may be “delivered” to different compartments of the DC, with important consequences for the type of immune response induced. Consequently the following sections will use selected examples of vaccine developments to demonstrate how vaccine may be targeting the immune system. When possible, examples will be given for the porcine field. This will be elaborated using more general advances in immunology characterizing the interaction of DC with vaccine carriers promoting interaction with surface receptors on the DC.
3. Recombinant vector vaccines with potential for targeting the porcine immune system
An important element in the concept of recombinant vector vaccines is selecting immunogenic structures of the pathogen to replace the pathogen itself, ensuring its efficient delivery to the immune system as in the form of a biologically active carrier. This approach helps reduce the risks associated with handling the live pathogen, and the problems of incomplete inactivation or reversion of attenuation. The approach must ensure that the vaccine retains a high level of immunogenicity, which is more difficult to achieve with subunit or protein/peptide vaccines.
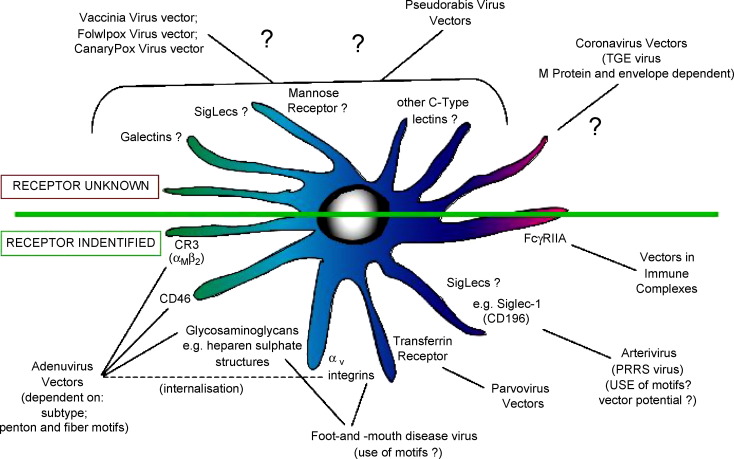
Viruses possess natural ligands for binding to receptors of the cells they infect; receptors have been identified for some viruses employed for vaccine design, but not for all (Fig. 2 ). Although many viruses infect epithelial cells, a large number can interact directly with DC, in particular monocytotropic viruses. Also a restricted infection of epithelial cells can be immunogenic after antigen “transfer” to the DC, following exocytosis from the producing cell.
Fig. 2.
Examples of viruses employed as vectors for vaccine delivery, as well as vaccines which can provide information in terms of potential receptors for vaccine targeting. The figure also shows the receptors targeted by certain of the viruses, when this is known, as well as receptors which may be employed by virus vectors for targeting DC.
A disadvantage of viral vectors is the immune response against the vector itself, which restricts its application in terms of multiple use for different vaccines. However, the formation of antibody complexes with the vaccine can have certain advantages in terms of the immune reaction induced. For example, FcγRI-targeting of adenovirus vector can enhance vector-mediated gene transfer into human DC, leading to an increased immune response against the vector-encoded antigen [16].
3.1. Vaccinia virus and other poxvirus recombinant vaccines
3.1.1. Past and current use as vaccine vectors
Vaccinia virus was one of the earliest viruses employed as a vector for the generation of recombinant vaccines, generally for all species [17]. In 1986, a recombinant vaccinia virus expressing influenza virus haemagglutinin was tested in cattle, sheep and poultry [18]. On the porcine front, a vaccinia virus was employed to generate a recombinant vaccine expressing pseudorabies virus (PRV) glycoprotein gp50 [19]. An overview of reports on vaccinia virus recombinants employed as vectors for porcine vaccines is given in Table 1 , including the recent application of the recombinant modified Ankara vaccinia virus [20] (Table 1a).
Table 1.
Examples of porcine vaccines based on poxvirus vectors.
| (a) Porcine vaccines based on a vaccinia virus vector | ||
|---|---|---|
| Target pathogen | Target antigen | Reference |
| Japanese encephalitis virus | prM and E proteins | [174] |
| Japanese encephalitis virus | prM, E, and NS1 proteins | [175] |
| PRSSV | GP5 and M proteins | [20] |
| PRV | gp50 glycoprotein | [19] |
| TGEV | Spike glycoprotein, membrane and nucleoprotein | [176] |
| (b) Porcine vaccines based on other poxvirus vectors | |||
|---|---|---|---|
| Vector | Target pathogen | Target antigen | Reference |
| Fowlpox virus | FMDV | P1, 2A, 3C | [177] |
| Fowlpox virus | PRRSV | GP5/GP3 | [178] |
| ORF virus (Parapoxvirus ovis) | CSFV | E2 glycoprotein | [179] |
| ORF virus | PRV | gC or gD glycoprotein | [180] |
| Parapoxvirus and vaccinia virus | Vector alone | IFN induction | [23], [181] |
| Swinepox virus | General review | General review | [21] |
| Swinepox virus | Feline leukemia virus | Gag and Env | [22] |
Related to the application of vaccinia vectors, swine poxvirus has also been proposed as the basis for a porcine recombinant vaccine [21] (Table 1b). Although it has not been tested as a porcine vaccine vector, swine poxvirus has clear potential, for example from the work with feline leukaemia virus antigens and infection of porcine cells [22] (Table 1b). Both fowlpox virus and parapoxvirus (ORF virus) have been employed as vectors for potential porcine vaccines (Table 1b). Interestingly, the ORF virus vector also displays adjuvant potential for inducing IFN responses [23] (Table 1b).
3.1.2. Potential for targeting DC
Whether vaccinia virus can enter porcine DC leading to viral DNA replication or transcription is uncertain. From studies using mouse and human cells, it appears that the virus does interact with DC [24], [25], [26], [27], [28]; to date, the receptor involved has not been defined, although it is possible to propose potential candidates (Fig. 2). Certainly, vaccinia virus is less efficient at binding to human DC compared with a human epithelial cell line [24]. Jenne et al. [25] also demonstrated difficulties for vaccinia virus to replicate in human DC – immature as well as mature – leading to an abortive infection in which proteins under control of the late promoters were lacking. Nevertheless, a certain level of protein translation does occur in DC, which could promote antigen-specific lymphocyte proliferation.
As mentioned above, DC processing of the antigen alone is inadequate for efficient promotion of immune responses; induction of DC maturation is also required, typically through “danger” signalling. With respect to vaccine vectors, there is a reported absence of vaccinia virus-induced maturation in the DC [24]. Moreover, a reduced expression of certain costimulatory molecules – CD80 and CD83, but not CD86 or MHC – on the virus-infected DC, together with a reduced capacity for stimulating T-cell proliferation was observed [25]. The modified vaccinia virus Ankara was even more problematic for human DC in terms of reduced cellular protein synthesis leading to increased apoptosis in the virus-infected DC [27]. While these latter events would be advantageous to the development of cytotoxic immunity, through cross-presentation of the apoptotic cells, the results overall query the value of direct interaction of vaccinia virus vectors with DC. Yates and Alexander-Miller [28] showed that when vaccinia virus infected immature DC, there was no induction of maturation, yielding DC incapable of initiating T-cell activation. It was only when mature DC were infected with vaccinia virus that functional antigen-presenting cells were generated, capable of activating vaccinia virus-specific CD8+ lymphocytes. Moreover, Nagorsen et al. [26] noted that the efficiency of the vaccine virus vector-delivered vaccine was dependent on the HLA haplotype and the epitopes carried by the vaccine.
3.2. Pseudorabies virus recombinant vaccines
Non-transmissible pseudorabies virus vectors have been created by deletion of the essential gD required for PRV penetration [29]. Considering that this virus can still spread from cell-to-cell, a biologically safer quadruple glycoprotein-deleted PRV lacking gG, gI and gE was generated [30]. With such PRV vectors, vaccines have been generated encoding proteins from a number of viruses Table 2 .
Table 2.
Examples of porcine vaccines based on a pseudorabies virus vector.
| Target pathogen | Target antigen | Reference |
|---|---|---|
| CSFV | E2 envelope glycoprotein | [29] |
| FMDV | Capsid precursor protein (P1) | [182] |
| FMDV | VP1 | [183] |
| FMDV; porcine parvovirus | P1-2A; VP2 | [184] |
| Japanese encephalitis virus | NS1 protein | [185] |
| PCV2 | Capsid (ORF2) protein | [186] |
| PCV2 | ORF1-ORF2 fusion protein | [187] |
| PRRSV | GP5 and M proteins | [188] |
| PRRSV | Modified GP5 membrane protein | [189] |
| PRRSV | GP5 | [190] |
| PRRSV | GP5 membrane protein | [191] |
| Pseudorabies virus | All except gD, gE, TK | [192] |
| Swine influenza virus (H3N2) | HA | [193] |
| TGEV | S protein | [194] |
Although the potential of PRV vectors for targeting DC has not been investigated, the reported capacity of PRV to infect and replicate efficiently in macrophages would indicate a tropism for monocytic cells including DC [31] (Fig. 2). Unfortunately, it has been reported that PRV infection of macrophages is associated with immunological malfunction [32], [33]. In contrast, PRV was seen to be a potent activator of porcine pDC [34], which would be beneficial for its use as vector. Such results show that the interaction of PRV with cDC and pDC needs further characterization before one can propose this as a vaccine delivery vehicle targeting DC. Indeed, the above results on both poxviruses and PRV demonstrate the need to understand the type and status of the DC being targeted by a vaccine carrier, to ensure the appropriate processing leading to activation of effector immune defences.
3.3. Adenovirus recombinant vaccines
3.3.1. Past and current use as vaccine vectors
Adenovirus has been receiving much attention as a vector for delivery of vaccines [35], [36], [37]. Recombinant replication-defective adenoviruses have been generated for a number of porcine pathogens (Table 3 ). These efforts have employed primarily human adenovirus serotype 5 or porcine adenovirus serotype 3, although canine adenovirus type 2 has also been tested. Adenovirus-vectored antigens have also been produced in cell culture and goat milk, for application as recombinant vaccines rather than vectored vaccines [37], [38].
Table 3.
Examples of porcine vaccines based on adenovirus vectors.
| Target pathogen | Target antigen | Reference |
|---|---|---|
| General overview | General overview | Porcine adenovirus type 3: [35] |
| Vector only | Vector development | Porcine adenovirus type 3: [195] |
| CSFV | gp55 (E2) glycoprotein | Porcine adenovirus type 3: [196] |
| CSFV | gp55 (E2) glycoprotein | Porcine adenovirus type 3: [197] |
| FMDV | VP1 | Canine adenovirus type 2: [198] |
| FMDV | P1 and protease | Human adenovirus type 5: [36], [199] |
| FMDV | P1 and protease/IFN-α | Human adenovirus type 5: [40] |
| Mycoplasma hyopneumoniae | P97 adhesin | Human adenovirus type 5: [200] |
| PCV2 | Capsid (ORF2) protein | Human adenovirus type 5: [201] |
| PCV2 | Capsid (ORF2) protein | Human adenovirus type 5: [202] |
| PRRSV | GP3 envelope protein | Human adenovirus type 5: [203] |
| PRRSV | GP5 and M proteins | Human adenovirus type 5: [204] |
| Porcine respiratory corona virus | Spike glycoprotein | Human adenovirus type 5: [205] |
| PRV | gD glycoprotein | Porcine adenovirus type 3: [206] |
| PRV | gD glycoprotein | Human adenovirus type 5: [207] |
| Swine influenza virus | Haemagglutinin | Human adenovirus type 5: [208] |
| Swine influenza virus | Haemagglutinin | Human adenovirus type 5: [209] |
| Swine influenza virus | Haemagglutinin | Human adenovirus type 5: [210] |
| TGEV | Spike glycoprotein | Human adenovirus type 5: [39] |
Although porcine adenovirus might appear to be the more obvious candidate of choice, application of human adenovirus does have high potential. Torres et al. [39] analysed the efficiency of this vector for targeting cells and organs of vaccinated pigs. They found adenovirus antigen associated with epithelial cells of bronchioles, macrophages, type II pneumocytes, and follicular dendritic cells. However, it was unclear whether these adenovirus-positive cells were accommodating replicating virus – therefore with the potential for producing the heterologous antigen in a vectored vaccine – or endocytosed antigen produced elsewhere.
Human adenovirus have also been engineered to express immunostimulatory cytokines, for promoting innate resistance against infection. A successful example of this is vaccination against foot-and-mouth disease virus (FMDV); a recombinant human adenovirus expressing FMDV P1 capsid precursor protein and 3C protease was co-administered with an adenovirus expressing porcine IFN-α [40]. This is particularly intriguing considering the importance of type I IFN to the maturation of cDC and in the development of innate responses following vaccination [41].
3.3.2. Adenovirus interaction with DC
The mechanism by which adenoviruses may interact with DC can be inferred from studies employing human monocytes/macrophages, human cell lines and murine bone marrow-derived DC (Fig. 2). Moreover, the immunomodulatory capacity of adenoviruses has been demonstrated using murine bone marrow-derived DC, in terms of maturation of the DC [42], [43], [44], [45], [46], phenotypic modification [42], [47], [48], and cytokine induction [42], [45], [46], [47], [48], [49]. Related to this, Miller et al. [43] noted that the induced maturation of DC depended on virus entry rather than transcription of the viral genome, while Molinier-Frenkel et al. [44] identified the penton capsomer and the fibre protein (and its knob domain) as major inducers of DC maturation.
Bontkes et al. [50] found that adenovirus alone was poor at infecting human pDC. In contrast, Basner-Tschakarjan et al. [46] were able to transduce murine bone marrow-derived (with Flt3-L) cDC and pDC with adenovirus, leading to cell maturation and production of IFN-α. This effect was observed in the pDC only when TLR9-dependent signalling was engaged. Such discriminatory results from adenovirus interaction with DC subsets may relate to the receptors required by different adenovirus serotypes, or differences between rodents and humans (see the comment in the next paragraph concerning CD46 expression). Interestingly, the targeting of adenovirus to human DC was enhanced when the virus was engineered to express CD40L on its capsid [51]. Bontkes et al. [50] also used a CD40 targeting approach to improve virus interaction with the DC; although this did not improve transduction efficiency, it did increase the maturation of the pDC.
3.3.3. Potential for targeting adenovirus to DC
The most straightforward approach for targeting vaccines to DC would be to employ virus vectors known to interact with a particular receptor on the DC. In this context, adenoviruses have a clear application. Receptors have been identified on human DC, as well as human or murine monocytes and macrophages, with which particular adenoviruses can interact. However, an initial problem was realised when the major cell receptor for virus binding was characterized as the coxsackie-adenovirus receptor (CAR) [52]. Despite being a widely expressed cell adhesion molecule, this receptor has not been found on DC or monocytic cell populations [53], [54]. In contrast, another receptor – CD46 – is expressed on DC [55], [56]. However, CD46 is employed by subgroup B adenoviruses such as adenovirus 35. Subgroup C adenoviruses such as the widely applied vaccine vector adenovirus 5, as well as canine adenovirus type 2 vaccine vector, employ CAR [54], [55], [56]. Nevertheless, the work with CD46 has shown its importance as a receptor for vaccine targeting, being present on many subtypes of human DC. Interestingly, it is not expressed on murine DC [57].
This variation among adenovirus subtypes with respect to receptor targeting would create uncertainty about the application of vectors such as the commonly employed adenovirus 5. Yet, such vaccines have proven successful in pigs (see Table 3). One explanation may be that the adenovirus vector targeted cells other than DC, such as epithelial cells expressing CAR. Alternatively, the vector may have used receptors other than CAR and CD46 on DC. Integrins such as the complement receptor CR3 (CD11b/CD18, αMβ2) [58], and glycosaminoglycans such as heparan sulphate structures on the cell surface [59] may also function as adenovirus receptors. In addition, certain adenoviruses can employ αv integrins for interacting with cells, at least cell lines [60], [61] and human monocytes and macrophages [59].
3.3.4. Appreciation of adenovirus targeting mechanisms
When considering adenovirus targeting of cells, it is important to bear in mind that the virus interaction with cell surface receptors is not a single event. Adenovirus can bind to cells via its fibre protein, after which the viral penton base interacts with cellular integrins such as αvβ3 and αvβ5 to promote virus entry. This role of the penton base appears to be dependent on the RGD motifs in the five identical subunits, at least with cells lines [60], [61]. Involvement of the αv integrins may be more important for virus internalisation than for attachment to the cell [60]. Yet, modifying the viral fibre knob of adenovirus type 5 to carry an RGD motif results in enhanced infectivity for murine DC [62]. While the interaction of the virus fibre protein is clearly important for binding to cell lines, penton base interaction with αMβ2 integrin is important for binding to human monocytes and macrophages [58]. Akin to attachment via the fibre knob, interaction with αMβ2 integrin still required the additional step involving αv integrins for virus internalisation.
In addition to the integrins as targets for adenovirus, interaction with cell surface glycosaminoglycan structures can occur. This may be particularly pertinent when considering that αv integrins are more efficiently expressed following activation of monocytes, macrophage or DC. Wickham et al. [59] demonstrated that adenovirus type 5 modified to carry a heparan sulphate-binding heptalysine motif were more efficient at transducing human monocytes and macrophages. A candidate heparin-binding KKTK motif was also identified in the third pseudorepeat of the fibre shaft from adenoviruses 2 and 5 [63]. Moreover, many viruses can bind to heparan sulphate structures on cells, particularly following cell culture passage of the virus as is often the case when producing vaccine viruses. However, heparan sulphate structures are rather ubiquitously expressed, and may not be ideal for enhancing DC targeting over interaction with other cell types. On the contrary, CR3 is more restricted to cells of the innate immune system, namely DC, macrophages and monocytes. Indeed, in contrast to adenoviruses type 2 and 5 (subgroup C adenoviruses), adenovirus type 3 (subgroup B) did not require heparan sulphate glycosaminoglycans for binding to and infection of cell lines [63].
3.3.5. Modifying cell targeting by adenoviruses
Modulation of vaccine targeting can be achieved by introducing a ligand on to the vaccine delivery vehicle, to enhance targeting of the desired receptor. Again, adenoviruses provide some interesting examples of the value in this approach. A chimeric adenovirus was created by modifying the subgroup C adenovirus type 5 to carry the subgroup B adenovirus 35 fibre antigen [55]. This increased interaction with human DC up to 100-fold. A similar approach may be achieved by applying short peptide motifs known for their capacity to interact with integrins expressed on DC, as described above in Section 3.3.3 [62]. One disadvantage with this approach is that certain motifs, such as RGD, have the potential to bind with different integrins. Nevertheless, the presence of the RGD motif in chimeric adenovirus penton bases apparently did not influence their interaction with integrins [54], suggesting an involvement of other structures; perhaps fibre antigens carrying the KKTK motif of the adenovirus 35 would be important [55]. Indeed, Perreau et al. [54] proposed that chimeric viruses could employ the non-integrin-binding capacity of the canine adenovirus 2 knob in combination with the capacity of the human adenovirus 5 to interact with integrins.
How different targeting events would influence DC handling of a vaccine is dependent on the cell receptor being targeted. Indeed, human adenovirus 5 and canine adenovirus 2 appear to be endocytosed into distinct intracellular compartments [54]. The roles of different receptors leading to distinct compartmentalisation within the cell, as well as the influence of multiple receptor involvement, are important considerations when analysing the potential for targeting vaccines to DC. While there is information concerning the binding of particular vaccines to DC, there is less information on the endocytic route taken thereafter. This is unfortunate, because the endocytic process will determine if an antigen is released (for interaction with B lymphocytes), processed through MHC Class II (for Th lymphocytes), or processed via the immunoproteasome for presentation in association with MHC Class I (for Tc lymphocytes). The endocytic pathway employed will also determine if genetic information carried by a viral vector vaccine is translated into protein. These are important issues, which will be raised again later during discussion of C-type lectin receptor targeting.
3.4. Other virus receptors on DC with potential for vaccine targeting
3.4.1. Small icosahedral DNA virus receptors
Canine parvovirus is reported to interact with the transferrin receptor (TfR) [64] (Fig. 2). This is referred to as an asymmetric binding where only a few receptor molecules (5 ± 2 TfR dimmers) can bind per capsid. The authors suggested that the asymmetric binding of TfR might be due to the virus possessing only one unique site with the conformation for binding to the TfR. Alternatively, the binding of the virus to the TfR might induce the asymmetry. Whatever the mechanism, this binding differs from that associated with picornavirus infection, in which up to 60 sites on the icosahedron can be involved [65].
3.4.2. Small icosahedral RNA virus receptors
Numerous non-enveloped viruses, including FMDV, are known for their potential to interact with heparan sulphate structures on cell surfaces [66], [67], [68], [69]. Heparan sulphate structures on DC are important for binding cell-culture passaged FMDV, relating to vaccine viruses [70] (Fig. 2). This virus also carries ligands for integrin receptors [71]: αvβ1 [72], αvβ3 [73], [74]) αvβ6 [71], and αvβ8 [72]. However, it appears that the preferred integrins for virus binding are either not expressed or are poorly expressed on the DC [70]. Whether ligation of heparan sulphate structures is the best target remains to be elucidated.
3.4.3. The potential of coronaviruses for vaccine targeting
Transmissible gastroenteritis virus (TGEV) has also generated interest with respect to DC targeting. In addition, TGEV has been employed as a vaccine vector; reviewed by Enjuanes et al. [75]. TGEV efficiently induces IFN production by pDC [41], [76], [77], [78], an ability which is independent of virus replication. Viral pseudoparticles expressing M and E proteins, and resembling intact virions, retained the interferogenic activity of the virus [79]; neither the virus genome nor the spike (S) protein necessary for infection of cells were essential for IFN induction. The induction of IFN did require virus interaction with the cell surface, but not all virion structures were important. Relating to the above report from Baudoux et al. [79], Riffault et al. [80] had shown that antibody against the TGEV receptor aminopeptidase N did not prevent the IFN induction; nor was antibody against surface M protein effective. Moreover, reconstructing detergent solubilised virus into virosomes did not reconstitute IFN induction capacity. The authors concluded that the induction of IFN required a complex native envelope protein structure. As such, it has been difficult to identify the receptors involved in this IFN induction, although it does seem certain that the induction is being signalled from interaction with receptors on the pDC surface. Insofar as being applicable for targeting vaccine to DC, one may employ the pseudoparticles expressing M and E proteins [79].
3.4.4. Sialic acids, sialylated structures on DC with potential for vaccine targeting
Although influenza virus per se has not been applied as a viral vector, derived virosomes (based on influenza virus envelope plus surface glycoproteins) have high potential. In this context, the virus biology is interesting in terms of DC targeting. Sialic residues on cells, particularly α-2,3 and α-2,6-linked sialic acids, are well established as receptors for influenza viruses. The efficiency of the virosomal vaccine carrier (for example: [81], [82], [83]) would suggest that α-2,6-linked sialic acids on DC (at least murine and human DC) were the target for such vaccines. Indeed, Angel et al. [84] demonstrated that virosomes interacting with pDC are reliant on sialic acid structures on the cell surface; endocytosis of the virosomes by pDC was inhibited by neuraminidase treatment. However, one also has to consider the targeting of the C-type lectin mannose receptors on DC; Reading et al. [85] demonstrated the importance of the mannose receptor for influenza virus infection of murine macrophages. Nevertheless, sialic acid residues on DC would offer the potential for vaccine targeting, particularly when the vaccine construct carries the haemagglutinin glycoprotein.
Another important consideration is the varying phenotype of DC, with respect to DC subsets and particularly DC differentiation and maturation. Sialylated structures on DC are modulated during differentiation or interaction with B cells resulting in an increase in expression of α-2-3-sialylated O-glycans, as well as α-2-6- and α-2-3-sialylated N-glycans [86], [87]. Interestingly, such sialylated glycans are important for DC endocytic activity. Maturation of the DC resulted in a reduced expression of the glycans, relating to the reduced endocytic activity of mature DC.
3.5. Particulate vaccines based on non-replicating viral particles
An alternative approach to the use of live, non-transmissible, replicating vaccine vectors is the use of virus-like particles (VLP). These lack a viral genome, physically carrying the vaccine antigen of interest within their virion structure. With the foreign (“vaccine”) antigen being within a chimeric structure, replication is no longer necessary. In fact, chimeric vectors can be regarded as “delivering” the vaccine in a manner similar to that for an inactivated vaccine. Chimeric viruses based on non-enveloped VLPs can also be defined in terms of structures or sequences interacting with DC receptors. Indeed, it can be seen that the non-enveloped viruses mentioned in Section 3.4 would be candidates for chimeric VLP-based vaccines.
There is quite a variety of vectors employed for bearing the antigen of interest (Table 4 ). These range from chimeric peptide constructs to viruses and bacteria [88], [89], [90], [91]. One value of employing non-enveloped virus particles for chimeric constructs is the potential of displaying heterologous antigens, allowing for targeting to the immune system via homologous or heterologous viral ligands for receptors on the DC. Good examples of this approach are the reported applications of porcine parvovirus and porcine circovirus type 2 (PCV2; Table 4), which have been employed for both homologous vaccines [92] and for delivering heterologous epitopes [93], [94], [95].
Table 4.
Examples of porcine vaccines based on antigen display using chimeric constructs.
| Vector | Target pathogen | Target antigen | Reference |
|---|---|---|---|
| Bacteriophage T4** | FMDV** | Capsid protomer** | [91] |
| Bacteriophage M13 | Taenia solium | KETc1, KETc12, GK1; KETc7 | [89] |
| Bamboo mosaic virus | FMDV | VP1 capsid protein | [211] |
| Porcine parvovirus (VLP) | PCV2 | Capsid (ORF2) protein residues 165–200 | [95] |
| Porcine parvovirus (VLP)* | Parvovirus* | All* | [92] |
| PCV1 (non-pathogenic) | PCV2 | Capsid (ORF2) protein | [94] |
| PRRSV (vaccine strain) | PRRSV (virulent) | All | [212] |
Tested in mice.
Tested primarily in mice.
4. Particle-based vaccine delivery
4.1. The advent of biodegradable vaccine carriers
Biodegradable polymers such as poly(lacticco-glycolic acid) (PLGA) have been applied for protein-based vaccine delivery for over a decade [96]. In addition, polyethyleneimine (PEI) loaded PLGA carriers as well as histidine-rich polymers and lipids have been used for nucleic acid delivery [97]. These polymers offer a main advantage over the vaccine vectors described above in Section 3, as well as those based on other viruses, bacteria or their products. Biodegradable polymers do not induce a strong or durable immune response against themselves. Moreover, they are efficiently degraded into non-toxic metabolites, primarily via the Krebs cycle. Particulate carriers are also flexible in their design, composition and structure, which has led to the demonstration of their high potential as vaccine carriers [98], [99], [100], [101], [102], [103], [104], [105], [106]. The design of these particles has employed a number of components, the main ones being PLGA, PEG-PLGA, PLA, PEG-PLA, hydrophobised cholesterol and polycationic formulations. A list of reviews on this topic is found in Table 5 .
Table 5.
Reviews on application of nanoparticle vaccine delivery systems.
| Title | Reference |
|---|---|
| Nanoparticles and microparticles as vaccine-delivery systems | [213] |
| Implication of nanoparticles/microparticles in mucosal vaccine delivery | [214] |
| The use of soluble polymers and polymer microparticles to provide improved vaccine responses after parenteral and mucosal delivery | [215] |
| Vaccine delivery—current trends and future | [216] |
| Polylactide-co-glycolide microparticles with surface adsorbed antigens as vaccine delivery systems | [217] |
| Nanoparticles as carriers for nasal vaccine delivery | [110] |
| Biodegradable poly(lactic-co-glycolic acid) microparticles for injectable delivery of vaccine antigens | [218] |
| DNA-loaded biodegradable microparticles as vaccine delivery systems and their interaction with dendritic cells | [130] |
| Immunological aspects of polymer microsphere vaccine delivery systems | [219] |
| Microparticles as vaccine adjuvants and delivery systems | [168] |
| Recent developments in vaccine delivery systems | [166] |
| Recent advances in vaccine adjuvants | [167] |
| Microparticle vaccine approaches to stimulate mucosal immunisation | [220] |
| Chitosan and its derivatives in mucosal drug and vaccine delivery | [221] |
| Nanoparticles and microparticles for drug and vaccine delivery | [96] |
Particulate formulations offer a wide applicability for in vivo delivery routes. In addition to their use for parenteral delivery, translocation of nano- and microparticles into systemic immune tissues has been observed after intranasal administration in mice, concomitant with development of immune responses [107], [108], [109]. An important consideration in this respect is the particle size, which appears to be a determinant for the performance of biodegradable particle vaccine delivery [110]. Particulate vaccine delivery has also been successful via the oral route (for example, reviewed by O’Hagan et al. [111]). Moreover, oral and nasal delivery of chitosan microparticles has been compared with respect to diphtheria vaccine efficacy in mice [109].
Despite these advances and high application value, there is little information on the biological characteristics of DC interaction with and processing of these particle-based vaccines. Moreover, further analyses are required on how such vaccine constructs can activate the “danger” recognition by DC necessary for promoting DC maturation and the development of active immunity.
4.2. Biodegradable vaccine carriers employed for porcine vaccinology
The recombinant viruses employed for porcine vaccine delivery (Table 1, Table 2, Table 3) can be regarded as nanoparticle-based vaccines. Indeed, the adenovirus vectors are effectively nanoparticles delivering a DNA vaccine. In that context, the capacity of the adenovirus to target particular receptors on the DC offers the potential for defined vaccine targeting to the immune system, which can be taken into the field of the biodegradable particle-based vaccine delivery vehicle (see Section 6 below). Similarly, VLP (Table 4) are nanoparticles carrying protein vaccine cargoes, albeit forming an integral part of the viral structure. Indeed, the bacteriophages T4 expressing the FMDV P1 protein (together with the T4 expressing FMDV 3C) have been referred to as a nanoparticle surface gene–protein display system [91].
Insofar as the biodegradable vaccine carriers are concerned, there is much less information on their application for porcine vaccines. This is somewhat surprising considering the extent of their application for human vaccines, and vaccines tested in murine models (see the reviews listed in Table 5). Nevertheless, there are a number of articles demonstrating the potential of biodegradable carriers for delivery of porcine vaccines (Table 6 ). These also include chitosan-based nanoparticles to entrap plasmids carrying immunomodulatory CpG-ODN motifs [112], [113]. Elaboration of this approach is seen with the application of chitosan-based nanoparticles carrying plasmids encoding porcine IL-2 [90] or IL-6 [112]. For these purposes, the authors employed the net positive charge of chitosan nanoparticles to entrap the DNA in the chitosan polymer matrix by ionic cross-linking.
Table 6.
Potential of nanoparticle/microparticle-based vaccine and adjuvant delivery systems for application in the porcine field.
| Delivery vehicle | Vaccine antigen | Vaccine adjuvant | Reference |
|---|---|---|---|
| Chitosan-nanoparticles | Pasteurella multocida and CSFV* | Porcine IL-2 gene + CpG-ODN; (DNA)* | [112] |
| Chitosan-nanoparticles encapsulating IL-2 gene | Porcine paratyphoid vaccine* | Porcine IL-2 gene (DNA)* | [90] |
| Chitosan-nanoparticles decorated with CpG-ODN | Porcine paratyphoid vaccine* | CpG-ODN (DNA)* | [113] |
| PLGA microspheres | IgY (model protein for oral vaccine delivery) | None (oral vaccination) | [222] |
| PLGA (200 nm) plus dimethyl-dioctadecyl-ammonium bromide cationic surfactant. | DNA (anionic): surface coated on to particles | None (in reconstituted gastric mucus) | [223] |
| Negative-charged fluorescent particles (50–500 nm) | None (testing transcutaneous delivery) | None (applied to pig skin in diffusion chambers) | [116] |
| Enteric-coated polymers (AQ6) | Actinobacillus pleuropneumonia | None (oral) | |
| Alum (sub-cutaneous) | [224] | ||
| Starch microparticles | Ovalbumin (surface coated on to particles) | None (applied to porcine nasal mucosa in vitro) | [225] |
| Microspheres from enteric-coating material (aqueous acrylic polymer) plus talc and glycerol | Mycoplasma hypopneumoniae | None (oral vaccination) | [226] |
| Nanoparticles and microbeads compared | Trichinella spiralis L1 muscular larvae. | Various adjuvants compared | [227] |
| Bioadhesive intranasal delivery system (esterified hyaluronic microspheres) | Influenza virus vaccine H1N1. | Mucosal adjuvants LTK63 and LTR72 (detoxified heat-labile enterotoxin) | [228] |
| Saccharomyces cerevisiae | None (testing particle uptake) | None (testing transcytosis by M cells) | [229]. |
| Gold particles | Influenza virus (H1N1) DNA vaccine | None (gene gun-based DNA vaccination) | [114] |
| Microspheres | Mycoplasma hypopneumoniae | Priming with adjuvanted, boosting with encapsulated antigen | [230] |
Immunised mice with a porcine vaccine and/or porcine cytokine-based adjuvant.
Zhao et al. [112] tested their delivery system in mice, co-administered with a bivalent vaccine against Pasteurella multocida and classical swine fever virus (CSFV). Their results were encouraging in the context of how the nanoparticle-delivered adjuvant enhanced the development of specific immunity and resistance to challenge infection. It will be interesting to see how this application of nanoparticle-delivered adjuvant functions in the porcine host, particularly considering the differences between murine and porcine immune system. As far as delivery of vaccine antigen is concerned, early results using ovalbumin as a model are showing considerable promise both in vitro with porcine DC (Supplementary Fig. 2) and in vivo (Harwood et al., unpublished results).
4.3. Particulate vaccine carriers and nucleic acid vaccine delivery
4.3.1. General considerations
Application of nucleic acid vaccines demonstrates the factors to be considered in the context of particle-delivered vaccines. An important influence is the particle charge. Positively charged particles facilitate association with nucleic acid, and also protein antigens with the appropriate negative charge; via ionic cross-linking with the particles. A major consideration – for both anionic and cationic particles – is that sufficient charge be available for binding the cargo. The particle charge is also important concerning mode and efficiency of delivery in vivo. For example, Macklin et al. [114] successfully employed gold particles for epidermal delivery of a DNA vaccine to immunise pigs against H1N1 influenza virus, using a device for particle-mediated gene transfer to the epidermis [115]. Negatively charged nanoparticle constructs also show high potential for delivery through pig skin [116]. Efficient permeation of pig skin was obtained with the negatively charged particles ranging in size from 50 to 500 nm, leading the authors to propose such nanoparticles as potential delivery vectors for transdermal vaccination.
Considering that the nucleic acid must be delivered to the target cell in which it will most efficiently transcribe, strong candidate targets are epithelial, fibroblastic or muscle cells; cells with dividing nuclei would increase the chance of DNA entry into the cell nucleus for transcription. The low rate of nuclear division in DC may restrict their capacity to handle DNA vaccines [117]. Nevertheless, targeting DNA vaccines to DC can be successful when applied via viral and bacterial vectors. For example, adenoviruses targeting DC do promote transcription of their DNA (see Section 3.3); the Salmonella typhimurium vector interacts efficiently with murine DC in vitro, and promotes expression of the encoded antigen both in vitro and in vivo [118], [119].
DNA vaccine processing by DC is less promising when the DNA is applied directly to the DC. Ceppi et al. [117] showed that porcine DC directly transfected with DNA do not translocate the DNA into the nucleus nor permit transcription. Franco et al. [120], showed uptake of a mycobacterial DNA vaccine by human macrophages and DC, but no apparent nuclear translocation or protein production, though mRNA was detectable in monocytes. Transfected cells could induce PBMC proliferation, but this was obtained with PBMC from patients positive or negative for reactivity against mycobacterial antigens.
Following direct intradermal inoculation of mice with DNA vaccine encoding influenza virus nucleoprotein (NP), Bot et al. [121] found both MHC Class II+ and MHC Class II− cells carrying the NP antigen. However, MHC Class II is insufficient for defining DC or Langerhans cells. Fibrocytes, which are important for activating cytotoxic T-cells, are also MHC Class II positive; it was interesting that adoptive transfer of the NP+ MHC Class II+ cells did induce cytotoxic T-cells. Although the authors argued against it, the results do not rule out translocation of endocytosed material, especially considering the capacity of DC to endocytose free antigen and also antigen-producing cells for cross-priming. Indeed, Watkins et al. [122] concluded that EGFP synthesised following DNA vaccination of sheep was leaching out of the skin for associating with DC in the afferent lymph. This was based on observations that both plasmid and protein were found in lymph cells and plasma, but only the protein was detected in lymph DC.
4.3.2. Increasing the efficiency of the DNA vaccine for activating DC
A number of groups have sought to improve the consequence of DNA vaccine interaction with DC, mostly in murine models. One approach was to encode DC-activating or DC-recruiting cytokines (reviewed by Kutzler and Weiner [123]). In these cases, the DC are not necessarily the targets for the vaccine, but become involved by enhanced attraction to the site of antigen production, for endocytosis of the antigens or antigen-producing cells. DC can also be directly activated by DNA vaccines, through CpG motifs in the DNA interacting with TLR9, although Spies et al. [124] showed that such activation can be TLR9-independent.
Another approach was to increase the targeting of the encoded antigen rather than the DNA. Indeed, You et al. [125] stated that DC showed a limited capacity for uptake of DNA vaccines. Their solution was to encode an antigen with IgG Fc in a fusion protein for vaccination of mice, promoting interaction with the FcR of the DC. Nchinda et al. [126] and Gu et al. [127] employed similar approaches with mice, using DNA encoding a fusion protein with a single chain Fv antibody specific for the DC receptor DEC 205 [126], or a fusion protein with heat shock protein as the targeting moiety [127].
4.3.3. Application of gold particles for DNA delivery
The above processes do not consider the targeting of the vaccine by delivery vehicles. In the context of DNA vaccines, much work has been performed using gold particle-based delivery, particularly in mice. Although many of these studies mentioned that DC were the key players in promoting immune response development, the majority did not directly quantify vaccine interaction with the DC. This is a major point of criticism raised by Lauterbach et al. [128]. They sought to elaborate on this question by using DNA constructs for either ubiquitous expression (CMV promoter) or specific expression in DC (CD11c promoter). Following biolistic vaccination of mice with DNA-coated gold particles, both forms of construct efficiently induced antibody responses. However, the DNA encoding for DC-specific expression was insufficient at inducing optimum T lymphocyte responses. Porgador et al. [129] also noted that lymph node cells directly expressing DNA-encoded antigen following vaccination of mice were rare. They did find DC carrying the encoded peptide, leading to the proposal that the DC were involved in cross-priming of material produced by transfected myeloblasts.
4.3.4. Application of biodegradable particle for DNA delivery
Gold particles for vaccine delivery can be problematic, due to their non-biodegradability and risk of persistence and toxicity. This is not a problem for biodegradable micro/nanoparticle delivery, which has also been promoted for “targeting” DNA vaccines to DC. However, many of these articles use the observation that an immune response was generated against the antigens encoded by the DNA as evidence for targeting of the DC (for example, see the review by Jilek et al. [130]). This is rather speculative, and cannot be considered as proof. On the other hand, the biodegradable particles themselves do interact efficiently with DC (reviewed by Jilek et al. [130]). Insofar as DNA vaccine delivery is concerned, the most convincing work comes from in vitro studies using cell lines. Jilek et al. [131] successfully transfected a murine DC line with PLGA microparticles carrying DNA encoding green fluorescent protein (GFP). However, the number of cells expressing GFP was low, which the authors related to the requirement for DNA to escape the phagosome wherein the PLGA particles would be degraded. This is a critical point considering the cytosolic localisation of phagosomes contrasting with the cellular localisation of DNA transcription machinery. Indeed, Walter and Merkle [132] showed that particle transfection efficiency is low in phagocytic cells compared with non-phagocytic cells.
Biodegradable particles can target DC in vivo, as observed with DC and macrophage endocytosis of PLGA microparticles in the spleen [133] and draining lymph nodes [134] of vaccinated mice. However, these authors did not determine if the encoded antigen was expressed in DC or non-immune cells; the particles were found in both lymphoid and non-lymphoid organs. Moreover, Walter and Merkle [132] proposed that application of biodegradable particle-based DNA delivery in vivo is very likely to lead to transfection of non-phagocytic cells such as fibroblasts.
4.3.5. Future prospectives for nucleic acid vaccination
Although much of the work to date has been in mice, DNA vaccination has been reported in pigs. Dincer et al. [135] analysed epidermal delivery of DNA-gold particles in pigs and mini-pigs. Antigen expression was detectable as early as 4 h post-vaccination, but it is uncertain if this reflected interaction of the DNA with non-immune cells. Nevertheless, one can conclude that particle-based DNA vaccination can be successful in pigs. Application of DNA-loaded PLGA microparticles may have a future for porcine vaccination considering the promise shown from studies in mice, guinea pigs, non-human primates and humans (reviewed by Jilek et al. [130]). While these particles can be targeted to DC, and certain DC may well be capable of allowing DNA transcription, particle-based delivery of DNA vaccines would be improved by targeting non-immune cells. Both Walter and Merkle [132] and Porgador et al. [129] suggested that the cells directly targeted by the particles for DNA transcription may be non-phagocytic cells.
One may also consider the reported differences seen with processing of DNA and RNA by DC [117]. This work would suggest that RNA vaccines have a higher potential for biodegradable particle-based delivery to DC. Certainly, DC will accommodate RNA translation and promote processing of the derived protein for induction of specific immunity [70]. It will therefore be interesting to see how the work advances on biodegradable nanoparticles enhancing the uptake of RNA by porcine DC (Supplementary Fig. 3).
5. Targeting vaccines to DC: potential for biodegradable particle targeting
5.1. General considerations
There have been a number of reports on vaccine carriers targeting DC, but primarily in the murine and human field. Gamvrellis et al. [136] reviewed a number of vaccine constructs which could facilitate uptake by DC. In order to pursue this topic with respect to targeting vaccines to porcine DC, it is necessary to appreciate the advances made in the murine and human fields. Current work is seeking to characterize how DC interact with vaccine carriers, particularly the biodegradable nanoparticle carriers. These have a high potential for application across a number of species and for different vaccines. This is due to their uptake relying on relatively “simple” components such as ionic charge, carbohydrate moieties in the particle structure, or particle size, rather than peptide-based (or lipoprotein- or glycoprotein-based) entities as is the case with many viral vectors. Moreover, biodegradable particles lend themselves more readily to modification, in both design and “decoration” to carry specific ligands for targeting DC receptors (Fig. 1).
Biodegradable particle structure has been modified and analysed in terms of how this influences DC endocytosis and intracytoplasmic delivery of the vaccine cargos. Not only can they be employed as carriers of protein and nucleic acid-based vaccines (Table 5, Table 6), but also as carriers of the adjuvant (Table 6). When the two approaches can be combined, an efficient mutual targeting of vaccine and adjuvant may be achievable. When targeting a vaccine to DC, it is necessary to concomitantly deliver a “danger” signal to promote activation and maturation of the DC system. Dependent on the “danger” signal, it may be desirable to co-deliver this with the vaccine in the same particle, or to use two different particles—one for the vaccine (targeting for example cDC) and one for the adjuvant (targeting for example pDC).
5.2. Choice of vaccine cargo
When attempting concomitant delivery of vaccine and adjuvant to the DC, it is likely that DNA vaccines would not be the appropriate vaccine choice—protein or RNA vaccines may have greater potential. RNA vaccines also need to target cells, but unlike DNA do not require a dividing nucleus for translation of their encoded genes into protein. DC can certainly accommodate RNA translation, as witnessed when DC are transfected with RNA encoding the complete genomic sequence of classical swine fever virus (CSFV) [137] or FMDV [70]). Unfortunately, RNA does not efficiently enter DC without assistance [137]. Electroporation or lipofection [70], [117] do allow “delivery” of the RNA, but these are not applicable of techniques for in vivo application, except if one relates lipofection to the application of liposomes for vaccine delivery. In this respect, nanoparticles offer a high potential for RNA delivery to DC, as they do for DNA delivery to epithelial or muscle cells.
The element which must be considered when applying nanoparticles for targeting protein or RNA vaccines to DC is the influence of targeting on the cellular site of vaccine delivery. Protein is ideally delivered into an acidifying endosomal system, to promote its processing for presentation in association with MHC Class II to Th lymphocytes. For processing in association with MHC Class I, leading to activation of Tc lymphocytes, a different intracellular localisation is required. In the case of RNA, a cytosolic delivery is required, to ensure that the delivered RNA is not degraded by cellular RNases. PLGA particles are reported to escape from endosomes, delivering their payloads into the cytoplasm [138], [139]. Nevertheless, the processes of cellular uptake, retention and processing are not well characterized for particulate vaccine delivery vehicles interacting with DC. This is certainly the case for particles decorated to target and/or activate PRR on the DC. How this would influence delivery to intracellular compartments is of pertinence when choosing the targeting ligand. A good example is the comparison of material delivered through ligation of the DEC205 receptor compared with ligation of the mannose receptor. Targeting DEC205 can lead to antigen recycling through late endosomes, whereas ligation of the mannose receptor may promote more of a recycling process through the peripheral endosomal compartment [15].
5.3. Potential for DC targeting
5.3.1. Potential for DC targeting using antibody against cell receptors
A simple approach for DC targeting is the application of antibodies against defined DC receptors. While this has clear potential as a targeting ligand, the overall applicability of such an approach is less certain, whether that be for human or porcine vaccines. Heterologous antibody risks inducing an immune response from the host, while isologous antibody can pose problems when in immune complexes with the vaccine. For the latter, one has to consider removing the Fc portion of the antibody, or preventing its interaction with FcR on cells of the innate immune defences. Nevertheless, antibody interacting with the vaccine carrier would have a role to play, as is the case during the natural humoral responses against an antigen: Immune complex formation would push the equilibrium of the immune response towards memory development.
An example of antibody-based DC-targeting is seen with the Siglec (sialic acid-binding immunoglobulin superfamily lectin) receptors, primarily expressed on cells of the immune system [140]. Siglecs bind specific glycan structures containing sialic acid [141], [142]. When murine pDC were targeted with ovalbumin conjugated to anti-Siglec-H antibody [143], an efficient endocytosis of the ovalbumin was observed, leading to stimulation of CD8+ T lymphocytes. The Siglec-5 receptor can also be targeted with specific antibody for enhanced endocytosis. Anti-Siglec-5 (Fab)2 fragments mediated rapid uptake into early endosomes of human monocytes [144].
One should note that the Siglec family is distributed throughout the immune system, but many are actually inhibitory receptors, carrying immunoreceptor tyrosine-based inhibitory motifs (ITIM). Siglec-10 is one such receptor on human DC [145], as is Siglec-7 on human DC and monocytes [146]. In contrast, Siglec-H actually lacks tyrosine-based signaling motifs in its cytoplasmic tail, functioning as an efficient endocytic receptor on murine pDC and macrophages, as observed with anti-Siglec-H antibody targeting [143]. Siglec-15 is another receptor of this family which does not carry an ITIM, being expressed on human macrophages and DC [147]. Lysine residues in its transmembrane domain interact with DNAX activation proteins DAP12 and DAP10. Interestingly, Siglec-15 expression appears to be conserved among species [147].
Another important characteristic of the Siglec expression on DC is the modulation associated with maturing cells, a modulation also noted with another glycan-binding DC receptor family, the β-galactoside-binding galectins [148]. Maturation-dependent modulation is also seen with the expression of C-type lectin receptors on DC [149]. In contrast to many of the galectin and Siglec reactions [148], the expression of C-type lection receptors is downregulated as the DC mature, a phenomenon related to reduced antigen uptake by maturing DC [149]. C-type lectin receptors such as DC-SIGN have also been targeted using specific antibodies, in murine and primate models for vaccine delivery (for example [150], [151]).
5.3.2. Potential for DC targeting using ligands for cell receptors
In addition to using antibody against DC receptors for vaccine targeting, a number of ligands for particular receptors have been identified, which offer potential for vaccine targeting. Siglec-15 preferentially recognizes Neu5Acα2-6GalNAcα [147]. Galectins bind glycoconjugates containing β-galactose structures [148], [152]. However, it is uncertain how vaccine particles interacting with different Siglecs would signal the DC, and how this would influence the outcome of vaccine handling by the cells. Interaction of a vaccine with a Siglec carrying an ITIM would not necessarily be detrimental for vaccine targeting, because it is not yet certain how DC would respond to vaccines using Siglec ligands for targeting purposes. For example, Siglec-10, which carries an ITIM, is reported to bind human red blood cells and soluble sialoglycoconjugates [145]. The Siglec-H, which lacks tyrosine-based signaling motifs, is endocytosed leading to the processing and presentation of captured antigens [153]. Yet, this Siglec signals intracellularly via the DAP12 adaptor with which it is associated, leading to reduced pDC responses against TLR ligands. Siglec-15 also signals via DAP12 and DAP10, but it has been suggested that this signaling implies delivery of activating signals to the cell [147]. Another receptor, Siglec-1 (CD169), is reported to be the receptor for porcine reproductive and respiratory syndrome virus (PRRSV) binding to and internalisation by porcine macrophages [154]. Considering that virus interaction with this receptor would lead to internalisation, targeting Siglec-1 may prove a useful vaccine targeting strategy. Siglec-1 is also expressed by DC, being upregulated on mature cells, along with Siglec-2 and Siglec-7 as well as two of the galectins—galectin-3 and galectin-8 [148].
There are also a number of ligands defined for C-type lectin receptors – such as DC-SIGN – which would provide useful targeting moieties. Examples of these are the GlcNac(β1-3)-Gal(β1-4)-Glc-R oligosaccharide forming the outer core of Neisseria meningitidis lipopolysaccharide [155], and Gal(β1-4)(Fuc(α1-3))GlcNAc and Fuc(α1-3)Gal(β1-4)(Fuc(α1-3))GlcNAc determinants relating to the glycosphingolipids of Schistosoma mansoni [156]. With respect to the studies on Neisseria meningitidis lipopolysaccharide interaction with DC-SIGN [155], mannan and N-acetylglucosamine (GlcNac) were found to interfere with bacterial binding. Mannan, including oxidized and reduced derivatives, enhanced antigen targeting to and endocytosis by DC. This resulted in an activation of the DC and immune response induction in vivo, at least in the mouse [157]. Moreover, the differential application of the oxidized and reduced forms of mannan can favour a more Th1 or Th2 type response, respectively [158].
Certainly, mannan and other mannosylated structures, which can bind to the mannose receptor CD206 as well as DC-SIGN, have been employed for targeting mannose-binding C-type lectins on DC (Table 7 ). A number of vaccine antigens also offer this targeting facility, through mannose-containing moieties inherent in their structures. Either these or synthetic structures can be conjugated to the vaccine delivery vehicles, an approach which has found favour with the mannosylation of biodegradable nanoparticle vaccine carriers.
Table 7.
Mannosylated nanoparticle/microparticle-based vaccine enhancing targeting of the immune system.
| Delivery vehicle | Vaccine antigen | Reference |
|---|---|---|
| Mannosylated anionic poly(varepsilon-caprolactone)-poly(ethyleneglycol)-poly(varepsilon-caprolactone) | Human basic fibroblast growth factor | [231] |
| Mannosylated cationic liposomes: N-[1-(2,3-dioleyloxy)propyl]-N,N,N-trimethylammonium chloride (DOTMA)/cholesten-5-yloxy-N-(4-((1-imino-2-d-thiomannosyl-ethyl)amino)butyl)formamide (Man-C4-Chol)/Chol | DNA vaccine | [232] |
| Mannosylated cationic liposomes: cholesten-5-yloxy-N-{4-[(1-imino-2-d-thiomannosylethyl)amino]butyl-formamide (Man-C4-Chol) with cationic lipid | Ovalbumin | [233] |
| Mannosylated cationic nanoparticles: emulsifying wax plus CTAB cationic surfactant; mannosylation of nanoparticles with or without entrapped endosomolytic agents, dioleoyl phosphatidylethanolamine (DOPE) and cholesterol | DNA vaccine | [234] |
5.3.3. TLR receptors on DC with potential for vaccine targeting
Related to the approach with ligands for C-type lectin and Siglec receptors, it is also possible to target other DC receptors, including the TLRs (Fig. 1) important for adjuvant-dependent activation of the cells. Certainly, TLR ligands such as chemically defined lipopeptides have potential as adjuvants [159]. Although lipopeptides have not been reported in terms of vaccine delivery for pigs, lipopeptides and lipoproteins have been tested for their adjuvant potential. These have been employed either as an integral antigenic component of the vaccine [160], [161], [162], or admixed with the vaccine antigen in a more classical manner [163]. The disadvantage of lipoproteins is that they themselves can be antigenic. In contrast, lipopeptides such as the synthetic compounds based on tri-palmitoyl-S-glyceryl cysteine (Pam3Cys) and di-palmitoyl-S-glyceryl cysteine (Pam2Cys) tend to serve primarily as PAMPs for TLR2 dimers, thus allowing for their application in a more generic sense [164].
Related to this area is the application of lipid-based vaccine carriers, liposomes [165]. Liposomal vaccine delivery and application as an adjuvant have been widely reported for murine and human studies [166], [167], [168], [169], [170], [171]. VLPs can also be constructed to bear lipopeptide surface structures. By inference to the capacity of TLR2 homo- and heterodimers to bind such structures, this allows for potential vaccine targeting and activation of DC [169], [172], [173].
6. Overall conclusions
This overview on particulate vaccine targeting to the immune system has presented the current knowledge concerning the relationship between the surface structure of vaccine delivery vehicles and the manner by which DC interact with the particle cargo. Although there is some evidence that vaccine targeting to DC has high potential for porcine vaccines, most of the information comes from inference to work with murine and human DC. Therein, a major effort is being made in the development of both viral and synthetic vaccine carriers. A critically important consideration therein is how receptor ligation influences the characteristics of vaccine interaction with the DC. Whether protein or RNA delivery is being employed, the antigen ultimately present in the DC – delivered by the carrier or translated from the delivered RNA – has to be processed via endosomal recycling and processing. Accordingly, current research on vaccine targeting to DC is seeking to determine the influence of the vaccine delivery vehicle surface decoration on modulation of DC activity. This is linking to the cytosolic localisation of the delivered vaccine cargo, which will be different for a protein cargo compared with an RNA cargo. The key element is to define the manner of targeting to the DC which will promote the appropriate delivery, leading to efficacious immune defence development (Supplementary Fig. 4).
Footnotes
Supplementary data associated with this article can be found, in the online version, at doi:10.1016/j.dci.2008.07.015.
Appendix A. Supplementary data
References
- 1.Janeway C. Immunogenicity signals 1,2,3… and 0. Immunol Today. 1989;10:283–286. doi: 10.1016/0167-5699(89)90081-9. [DOI] [PubMed] [Google Scholar]
- 2.Fearon D. Happy coupling: recruiting both antigen and effector function. Nat Biotechnol. 1997;15:618–619. doi: 10.1038/nbt0797-618. [DOI] [PubMed] [Google Scholar]
- 3.Summerfield A, McCullough KC, editors. The porcine dendritic cell, Developmental and Comparative Immunology 2009;33:299–309. [DOI] [PMC free article] [PubMed]
- 4.Banchereau J., Steinman R.M. Dendritic cells and the control of immunity. Nature. 1998;392:245–252. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- 5.MacPherson G., Kushnir N., Wykes M. Dendritic cells, B cells and the regulation of antibody synthesis. Immunol Rev. 1999;172:325–334. doi: 10.1111/j.1600-065x.1999.tb01376.x. [DOI] [PubMed] [Google Scholar]
- 6.Jego G., Pascual V., Palucka A.K., Banchereau J. Dendritic cells control B cell growth and differentiation. Curr Dir Autoimmun. 2005;8:124–139. doi: 10.1159/000082101. [DOI] [PubMed] [Google Scholar]
- 7.Gordon S. Pattern recognition receptors: doubling up for the innate immune response. Cell. 2002;111:927–930. doi: 10.1016/s0092-8674(02)01201-1. [DOI] [PubMed] [Google Scholar]
- 8.Uenishi H, Shinkai H, editors. Porcine TLR: a front line of pathogen monitoring and possible implication in disease resistance. Developmental and Comparative Immunology 2009;33:353–61. [DOI] [PubMed]
- 9.Pulendran B. Modulating vaccine responses with dendritic cells and Toll-like receptors. Immunol Rev. 2004;199:227–250. doi: 10.1111/j.0105-2896.2004.00144.x. [DOI] [PubMed] [Google Scholar]
- 10.Lanzavecchia A., Sallusto F. Dynamics of T lymphocyte responses: intermediates, effectors, and memory cells. Science. 2000;290:92–97. doi: 10.1126/science.290.5489.92. [DOI] [PubMed] [Google Scholar]
- 11.McCullough K.C., Summerfield A. Basic concepts of immune response and defense development. ILAR J. 2005;46:230–240. doi: 10.1093/ilar.46.3.230. [DOI] [PubMed] [Google Scholar]
- 12.Wiedemann F., Link R., Pumpe K., Jacobshagen U., Schaefer H.E., Wiesmuller K.H. Histopathological studies on the local reactions induced by complete Freund's adjuvant (CFA), bacterial lipopolysaccharide (LPS), and synthetic lipopeptide (P3C) conjugates. J Pathol. 1991;164:265–271. doi: 10.1002/path.1711640313. [DOI] [PubMed] [Google Scholar]
- 13.BenMohamed L., Gras-Masse H., Tartar A., Daubersies P., Brahimi K., Bossus M. Lipopeptide immunization without adjuvant induces potent and long-lasting B, T helper, and cytotoxic T lymphocyte responses against a malaria liver stage antigen in mice and chimpanzees. Eur J Immunol. 1997;27:1242–1253. doi: 10.1002/eji.1830270528. [DOI] [PubMed] [Google Scholar]
- 14.Seth A., Yasutomi Y., Jacoby H., Callery J.C., Kaminsky S.M., Koff W.C. Evaluation of a lipopeptide immunogen as a therapeutic in HIV type 1-seropositive individuals. AIDS Res Hum Retroviruses. 2000;16:337–343. doi: 10.1089/088922200309214. [DOI] [PubMed] [Google Scholar]
- 15.Mahnke K., Guo M., Lee S., Sepulveda H., Swain S.L., Nussenzweig M. The dendritic cell receptor for endocytosis, DEC-205, can recycle and enhance antigen presentation via major histocompatibility complex class II-positive lysosomal compartments. J Cell Biol. 2000;151:673–684. doi: 10.1083/jcb.151.3.673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sapinoro R., Maguire C.A., Burgess A., Dewhurst S. Enhanced transduction of dendritic cells by FcgammaRI-targeted adenovirus vectors. J Gene Med. 2007;9:1033–1045. doi: 10.1002/jgm.1112. [DOI] [PubMed] [Google Scholar]
- 17.Ulaeto D., Hruby D.E. Uses of vaccinia virus in vaccine delivery. Curr Opin Biotechnol. 1994;5:501–504. doi: 10.1016/0958-1669(94)90064-7. [DOI] [PubMed] [Google Scholar]
- 18.Boyle D.B., Coupar B.E., Parsonson I.M., Bagust T.J., Both G.W. Responses of cattle, sheep and poultry to a recombinant vaccinia virus expressing a swine influenza haemagglutinin. Res Vet Sci. 1986;41:40–44. [PubMed] [Google Scholar]
- 19.Marchioli C.C., Yancey R.J., Jr., Petrovskis E.A., Timmins J.G., Post L.E. Evaluation of pseudorabies virus glycoprotein gp50 as a vaccine for Aujeszky's disease in mice and swine: expression by vaccinia virus and Chinese hamster ovary cells. J Virol. 1987;61:3977–3982. doi: 10.1128/jvi.61.12.3977-3982.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zheng Q., Chen D., Chen D., Bi Z., Cao R., Zhou B. Co-expressing GP5 and M proteins under different promoters in recombinant modified vaccinia virus Ankara (rMVA)-based vaccine vector enhanced the humoral and cellular immune responses of porcine reproductive and respiratory syndrome virus (PRRSV) Virus Genes. 2007;35:585–595. doi: 10.1007/s11262-007-0161-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tripathy D.N. Swinepox virus as a vaccine vector for swine pathogens. Adv Vet Med. 1999;41:463–480. doi: 10.1016/S0065-3519(99)80035-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Winslow B.J., Cochran M.D., Holzenburg A., Sun J., Junker D.E., Collisson E.W. Replication and expression of a swinepox virus vector delivering feline leukemia virus Gag and Env to cell lines of swine and feline origin. Virus Res. 2003;98:1–15. doi: 10.1016/j.virusres.2003.08.005. [DOI] [PubMed] [Google Scholar]
- 23.Fachinger V., Schlapp T., Strube W., Schmeer N., Saalmüller A. Poxvirus-induced immunostimulating effects on porcine leukocytes. J Virol. 2000;74:7943–7951. doi: 10.1128/jvi.74.17.7943-7951.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Drillien R., Spehner D., Bohbot A., Hanau D. Vaccinia virus-related events and phenotypic changes after infection of dendritic cells derived from human monocytes. Virology. 2000;268:471–481. doi: 10.1006/viro.2000.0203. [DOI] [PubMed] [Google Scholar]
- 25.Jenne L., Hauser C., Arrighi J.F., Saurat J.H., Hügin A.W. Poxvirus as a vector to transduce human dendritic cells for immunotherapy: abortive infection but reduced APC function. Gene Ther. 2000;7:1575–1583. doi: 10.1038/sj.gt.3301287. [DOI] [PubMed] [Google Scholar]
- 26.Nagorsen D., Panelli M., Dudley M.E., Finkelstein S.E., Rosenberg S.A., Marincola F.M. Biased epitope selection by recombinant vaccinia-virus (rVV)-infected mature or immature dendritic cells. Gene Ther. 2003;10:1754–1765. doi: 10.1038/sj.gt.3302066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chahroudi A., Garber D.A., Reeves P., Liu L., Kalman D., Feinberg M.B. Differences and similarities in viral life cycle progression and host cell physiology after infection of human dendritic cells with modified vaccinia virus Ankara and vaccinia virus. J Virol. 2006;80:8469–8481. doi: 10.1128/JVI.02749-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Yates N.L., Alexander-Miller M.A. Vaccinia virus infection of mature dendritic cells results in activation of virus-specific naive CD8+ T cells: a potential mechanism for direct presentation. Virology. 2007;359:349–361. doi: 10.1016/j.virol.2006.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Peeters B., Bienkowska-Szewczyk K., Hulst M., Gielkens A., Kimman T. Biologically safe, non-transmissible pseudorabies virus vector vaccine protects pigs against both Aujeszky's disease and classical swine fever. J Gen Virol. 1997;78:3311–3315. doi: 10.1099/0022-1317-78-12-3311. [DOI] [PubMed] [Google Scholar]
- 30.Mettenleiter T.C., Klupp B.G., Weiland F., Visser N. Characterization of a quadruple glycoprotein-deleted pseudorabies virus mutant for use as a biologically safe live virus vaccine. J Gen Virol. 1994;75:1723–1733. doi: 10.1099/0022-1317-75-7-1723. [DOI] [PubMed] [Google Scholar]
- 31.Iglesias G., Pijoan C., Molitor T. Interactions of Pseudorabies virus with swine alveolar macrophages I: virus replication. Arch Virol. 1989;104:107–115. doi: 10.1007/BF01313812. [DOI] [PubMed] [Google Scholar]
- 32.Iglesias G., Pijoan C., Molitor T. Interactions of pseudorabies virus with swine alveolar macrophages: effects of virus infection on cell functions. J Leukoc Biol. 1989;45:410–415. doi: 10.1002/jlb.45.5.410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shibata I., Urono K., Samegai Y., Okada M., Inaba Y. Replication of virulent and attenuated strains of Aujeszky's disease virus in swine alveolar macrophages. J Vet Med Sci. 1994;56:465–468. doi: 10.1292/jvms.56.465. [DOI] [PubMed] [Google Scholar]
- 34.Vincent I.E., Balmelli C., Meehan B., Allan G., Summerfield A., McCullough K.C. Silencing of natural interferon producing cell activation by porcine circovirus type 2 DNA. Immunology. 2007;120:47–56. doi: 10.1111/j.1365-2567.2006.02476.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hammond J.M., Johnson M.A. Porcine adenovirus as a delivery system for swine vaccines and immunotherapeutics. Vet J. 2005;169:17–27. doi: 10.1016/j.tvjl.2003.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Grubman M.J. Development of novel strategies to control foot-and-mouth disease: marker vaccines and antivirals. Biologicals. 2005;33:227–234. doi: 10.1016/j.biologicals.2005.08.009. [DOI] [PubMed] [Google Scholar]
- 37.Sanchez O., Barrera M., Rodriguez M.P., Frias M.T., Figueroa N.E., Naranjo P. Classical swine fever virus E2 glycoprotein antigen produced in adenovirally transduced PK-15 cells confers complete protection in pigs upon viral challenge. Vaccine. 2008;26:988–997. doi: 10.1016/j.vaccine.2007.11.014. [DOI] [PubMed] [Google Scholar]
- 38.Toledo J.R., Sanchez O., Montesino R., Farnos O., Rodriguez M.P., Alfonso P. Highly protective E2-CSFV vaccine candidate produced in the mammary gland of adenoviral transduced goats. J Biotechnol. 2008;133:370–376. doi: 10.1016/j.jbiotec.2007.09.014. [DOI] [PubMed] [Google Scholar]
- 39.Torres J.M., Alonso C., Ortega A., Mittal S., Graham F., Enjuanes L. Tropism of human adenovirus type 5-based vectors in swine and their ability to protect against transmissible gastroenteritis coronavirus. J Virol. 1996;70:3770–3780. doi: 10.1128/jvi.70.6.3770-3780.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Moraes M.P., Chinsangaram J., Brum M.C., Grubman M.J. Immediate protection of swine from foot-and-mouth disease: a combination of adenoviruses expressing interferon alpha and a foot-and-mouth disease virus subunit vaccine. Vaccine. 2003;22:268–279. doi: 10.1016/s0264-410x(03)00560-7. [DOI] [PubMed] [Google Scholar]
- 41.Guzylack-Piriou L., Piersma S., McCullough K.C., Summerfield A. Role of natural interferon-producing cells and T lymphocytes in porcine monocyte-derived dendritic cell maturation. Immunology. 2006;118:78–87. doi: 10.1111/j.1365-2567.2006.02343.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Korst R.J., Mahtabifard A., Yamada R., Crystal R.G. Effect of adenovirus gene transfer vectors on the immunologic functions of mouse dendritic cells. Mol Ther. 2002;5:307–315. doi: 10.1006/mthe.2002.0538. [DOI] [PubMed] [Google Scholar]
- 43.Miller G., Lahrs S., Shah A.B., DeMatteo R.P. Optimization of dendritic cell maturation and gene transfer by recombinant adenovirus. Cancer Immunol Immunother. 2003;52:347–358. doi: 10.1007/s00262-003-0379-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Molinier-Frenkel V., Prévost-Blondel A., Hong S.S., Lengagne R., Boudaly S., Magnusson M.K. The maturation of murine dendritic cells induced by human adenovirus is mediated by the fiber knob domain. J Biol Chem. 2003;278:37175–37182. doi: 10.1074/jbc.M303496200. [DOI] [PubMed] [Google Scholar]
- 45.Schumacher L., Ribas A., Dissette V.B., McBride W.H., Mukherji B., Economou J.S. Human dendritic cell maturation by adenovirus transduction enhances tumor antigen-specific T-cell responses. J Immunother. 2004;27:191–200. doi: 10.1097/00002371-200405000-00003. [DOI] [PubMed] [Google Scholar]
- 46.Basner-Tschakarjan E., Gaffal E., O’Keeffe M., Tormo D., Limmer A., Wagner H. Adenovirus efficiently transduces plasmacytoid dendritic cells resulting in TLR9-dependent maturation and IFN-alpha production. J Gene Med. 2006;8:1300–1306. doi: 10.1002/jgm.964. [DOI] [PubMed] [Google Scholar]
- 47.Morelli A.E., Larregina A.T., Ganster R.W., Zahorchak A.F., Plowey J.M., Takayama T. Recombinant adenovirus induces maturation of dendritic cells via an NF-kappaB-dependent pathway. J Virol. 2000;74:9617–9628. doi: 10.1128/jvi.74.20.9617-9628.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Salucci V., Lena A.M., Ciliberto G., Scarselli E., La Monica N. Adenovirus transduction and culture conditions affect the immunogenicity of murine dendritic cells. Scand J Immunol. 2005;62:206–217. doi: 10.1111/j.1365-3083.2005.01658.x. [DOI] [PubMed] [Google Scholar]
- 49.Hirschowitz E.A., Weaver J.D., Hidalgo G.E., Doherty D.E. Murine dendritic cells infected with adenovirus vectors show signs of activation. Gene Ther. 2000;7:1112–1120. doi: 10.1038/sj.gt.3301210. [DOI] [PubMed] [Google Scholar]
- 50.Bontkes H.J., Ruizendaal J.J., Schreurs M.W., Kramer D., Meijer C.J., Hooijberg E. Antigen gene transfer to human plasmacytoid dendritic cells using recombinant adenovirus and vaccinia virus vectors. Cell Oncol. 2005;27:175–182. doi: 10.1155/2005/753549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Korokhov N., Noureddini S.C., Curiel D.T., Santegoets S.J., Scheper R.J., de Gruijl T.D. A single-component CD40-targeted adenovirus vector displays highly efficient transduction and activation of dendritic cells in a human skin substrate system. Mol Pharm. 2005;2:218–223. doi: 10.1021/mp050002w. [DOI] [PubMed] [Google Scholar]
- 52.Zhang Y., Bergelson J.M. Adenovirus receptors. J Virol. 2005;79:12125–12131. doi: 10.1128/JVI.79.19.12125-12131.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Rea D., Schagen F.H., Hoeben R.C., Mehtali M., Havenga M.J., Toes R.E. Adenoviruses activate human dendritic cells without polarization toward a T-helper type 1-inducing subset. J Virol. 1999;73:10245–10253. doi: 10.1128/jvi.73.12.10245-10253.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Perreau M., Mennechet F., Serratrice N., Glasgow J.N., Curiel D.T., Wodrich H. Contrasting effects of human, canine, and hybrid adenovirus vectors on the phenotypical and functional maturation of human dendritic cells: implications for clinical efficacy. J Virol. 2007;81:3272–3284. doi: 10.1128/JVI.01530-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Rea D., Havenga M.J., van Den Assem M., Sutmuller R.P., Lemckert A., Hoeben R.C. Highly efficient transduction of human monocyte-derived dendritic cells with subgroup B fiber-modified adenovirus vectors enhances transgene-encoded antigen presentation to cytotoxic T cells. J Immunol. 2001;166:5236–5244. doi: 10.4049/jimmunol.166.8.5236. [DOI] [PubMed] [Google Scholar]
- 56.Loré K., Adams W.C., Havenga M.J., Precopio M.L., Holterman L., Goudsmit J. Myeloid and plasmacytoid dendritic cells are susceptible to recombinant adenovirus vectors and stimulate polyfunctional memory T cell responses. J Immunol. 2007;179:1721–1729. doi: 10.4049/jimmunol.179.3.1721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Hsu C., Boysen M., Gritton L.D., Frosst P.D., Nemerow G.R., Von Seggern D.J. In vitro dendritic cell infection by pseudotyped adenoviral vectors does not correlate with their in vivo immunogenicity. Virology. 2005;332:1–7. doi: 10.1016/j.virol.2004.11.014. [DOI] [PubMed] [Google Scholar]
- 58.Huang S., Kamata T., Takada Y., Ruggeri Z.M., Nemerow G.R. Adenovirus interaction with distinct integrins mediates separate events in cell entry and gene delivery to hematopoietic cells. J Virol. 1996;70:4502–4508. doi: 10.1128/jvi.70.7.4502-4508.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Wickham T.J., Tzeng E., Shears L.L., 2nd, Roelvink P.W., Li Y., Lee G.M. Increased in vitro and in vivo gene transfer by adenovirus vectors containing chimeric fiber proteins. J Virol. 1997;71:8221–8229. doi: 10.1128/jvi.71.11.8221-8229.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wickham T.J., Mathias P., Cheresh D.A., Nemerow G.R. Integrins alpha v beta 3 and alpha v beta 5 promote adenovirus internalization but not virus attachment. Cell. 1993;73:309–319. doi: 10.1016/0092-8674(93)90231-e. [DOI] [PubMed] [Google Scholar]
- 61.Mathias P., Wickham T., Moore M., Nemerow G. Multiple adenovirus serotypes use alpha v integrins for infection. J Virol. 1994;68:6811–6814. doi: 10.1128/jvi.68.10.6811-6814.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Worgall S., Busch A., Rivara M., Bonnyay D., Leopold P.L., Merritt R. Modification to the capsid of the adenovirus vector that enhances dendritic cell infection and transgene-specific cellular immune responses. J Virol. 2004;78:2572–2580. doi: 10.1128/JVI.78.5.2572-2580.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Dechecchi M.C., Tamanini A., Bonizzato A., Cabrini G. Heparan sulfate glycosaminoglycans are involved in adenovirus type 5 and 2-host cell interactions. Virology. 2000;268:382–390. doi: 10.1006/viro.1999.0171. [DOI] [PubMed] [Google Scholar]
- 64.Hafenstein S., Palermo L.M., Kostyuchenko V.A., Xiao C., Morais M.C., Nelson C.D. Asymmetric binding of transferrin receptor to parvovirus capsids. Proc Natl Acad Sci USA. 2007;104:6585–6589. doi: 10.1073/pnas.0701574104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Rossmann M.G., He Y., Kuhn R.J. Picornavirus-receptor interactions. Trends Microbiol. 2002;10:324–331. doi: 10.1016/s0966-842x(02)02383-1. [DOI] [PubMed] [Google Scholar]
- 66.Jackson T., Ellard F.M., Ghazaleh R.A., Brookes S.M., Blakemore W.E., Corteyn A.H. Efficient infection of cells in culture by type O foot-and-mouth disease virus requires binding to cell surface heparan sulfate. J Virol. 1996;70:5282–5287. doi: 10.1128/jvi.70.8.5282-5287.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Sa-Carvalho D., Rieder E., Baxt B., Rodarte R., Tanuri A., Mason P.W. Tissue culture adaptation of foot-and-mouth disease virus selects viruses that bind to heparin and are attenuated in cattle. J Virol. 1997;71:5115–5123. doi: 10.1128/jvi.71.7.5115-5123.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Baranowski E., Ruiz-Jarabo C.M., Sevilla N., Andreu D., Beck E., Domingo E. Cell recognition by foot-and-mouth disease virus that lacks the RGD integrin-binding motif: flexibility in aphthovirus receptor usage. J Virol. 2000;74:1641–1647. doi: 10.1128/jvi.74.4.1641-1647.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Rigden R.C., Carrasco C.P., Summerfield A., McCullough K.C. Macrophage phagocytosis of foot-and-mouth disease virus may create infectious carriers. Immunology. 2002;106:537–548. doi: 10.1046/j.1365-2567.2002.01460.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Harwood L.J., Gerber H., Sobrino F., Summerfield A., McCullough K.C. Dendritic cell internalisation of foot-and-mouth disease virus: influence of heparan sulphate binding on virus uptake and immune response induction. J Virol. 2008 doi: 10.1128/JVI.00021-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Jackson T., Sheppard D., Denyer M., Blakemore W., King A.M. The epithelial integrin alphavbeta6 is a receptor for foot-and-mouth disease virus. J Virol. 2000;74:4949–4956. doi: 10.1128/jvi.74.11.4949-4956.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Jackson T., Clark S., Berryman S., Burman A., Cambier S., Mu D. Integrin alphavbeta8 functions as a receptor for foot-and-mouth disease virus: role of the beta-chain cytodomain in integrin-mediated infection. J Virol. 2004;78:4533–4540. doi: 10.1128/JVI.78.9.4533-4540.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Berinstein A., Roivainen M., Hovi T., Mason P.W., Baxt B. Antibodies to the vitronectin receptor (integrin alpha V beta 3) inhibit binding and infection of foot-and-mouth disease virus to cultured cells. J Virol. 1995;69:2664–2666. doi: 10.1128/jvi.69.4.2664-2666.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Neff S., Sa-Carvalho D., Rieder E., Mason P.W., Blystone S.D., Brown E.J. Foot-and-mouth disease virus virulent for cattle utilizes the integrin alpha(v)beta3 as its receptor. J Virol. 1998;72:3587–3594. doi: 10.1128/jvi.72.5.3587-3594.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Enjuanes L.S.I., Alonso S., Escors D., Zúñiga S. Coronavirus reverse genetics and development of vectors for gene expression. Curr Top Microbiol Immunol. 2005;287:161–197. doi: 10.1007/3-540-26765-4_6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Riffault S., Carrat C., Besnardeau L., La Bonnardière C., Charley B. In vivo induction of interferon-alpha in pig by non-infectious coronavirus: tissue localization and in situ phenotypic characterization of interferon-alpha-producing cells. J Gen Virol. 1997;78:2483–2487. doi: 10.1099/0022-1317-78-10-2483. [DOI] [PubMed] [Google Scholar]
- 77.Riffault S., Carrat C., van Reeth K., Pensaert M., Charley B. Interferon-alpha-producing cells are localized in gut-associated lymphoid tissues in transmissible gastroenteritis virus (TGEV) infected piglets. Vet Res. 2001;32:71–79. doi: 10.1051/vetres:2001111. [DOI] [PubMed] [Google Scholar]
- 78.Charley B., Riffault S., Van Reeth K. Porcine innate and adaptative immune responses to influenza and coronavirus infections. Ann N Y Acad Sci. 2006;1081:130–136. doi: 10.1196/annals.1373.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Baudoux P., Carrat C., Besnardeau L., Charley B., Laude H. Coronavirus pseudoparticles formed with recombinant M and E proteins induce alpha interferon synthesis by leukocytes. J Virol. 1998;72:8636–8643. doi: 10.1128/jvi.72.11.8636-8643.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Riffault S., Grosclaude J., Vayssier M., Laude H., Charley B. Reconstituted coronavirus TGEV virosomes lose the virus ability to induce porcine interferon-alpha production. Vet Res. 1997;28:105–114. [PubMed] [Google Scholar]
- 81.Glück R., Mischler R., Brantschen S., Just M., Althaus B., Cryz S.J., Jr. Immunopotentiating reconstituted influenza virus virosome vaccine delivery system for immunization against hepatitis A. J Clin Invest. 1992;90:2491–2495. doi: 10.1172/JCI116141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Lambkin R., Oxford J.S., Bossuyt S., Mann A., Metcalfe I.C., Herzog C. Strong local and systemic protective immunity induced in the ferret model by an intranasal virosome-formulated influenza subunit vaccine. Vaccine. 2004;22:4390–4396. doi: 10.1016/j.vaccine.2003.10.054. [DOI] [PubMed] [Google Scholar]
- 83.Felnerova D., Viret J.F., Glück R., Moser C. Liposomes and virosomes as delivery systems for antigens, nucleic acids and drugs. Curr Opin Biotechnol. 2004;15:518–529. doi: 10.1016/j.copbio.2004.10.005. [DOI] [PubMed] [Google Scholar]
- 84.Angel J., Chaperot L., Molens J.P., Mezin P., Amacker M., Cabral M.G. Virosome-mediated delivery of tumor antigen to plasmacytoid dendritic cells. Vaccine. 2007;25:3913–3921. doi: 10.1016/j.vaccine.2007.01.101. [DOI] [PubMed] [Google Scholar]
- 85.Reading P.C., Miller J.L., Anders E.M. Involvement of the mannose receptor in infection of macrophages by influenza virus. J Virol. 2000;74:5190–5197. doi: 10.1128/jvi.74.11.5190-5197.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Santos L., Draves K.E., Boton M., Grewal P.K., Marth J.D., Clark E.A. Dendritic cell-dependent inhibition of B cell proliferation requires CD22. J Immunol. 2008;180:4561–4569. doi: 10.4049/jimmunol.180.7.4561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Videira P.A., Amado I.F., Crespo H.J., Alguero M.C., Dall’olio F., Cabral M.G. Surface alpha2-3- and alpha2-6-sialylation of human monocytes and derived dendritic cells and its influence on endocytosis. Glycoconj J. 2008;25:259–268. doi: 10.1007/s10719-007-9092-6. [DOI] [PubMed] [Google Scholar]
- 88.Aoki H., Sakoda Y., Jukuroki K., Takada A., Kida H., Fukusho O. Induction of antibodies in mice by a recombinant baculovirus expressing pseudorabies virus glycoprotein B in mammalian cells. Vet Microbiol. 1999;68:197–207. doi: 10.1016/s0378-1135(99)00110-8. [DOI] [PubMed] [Google Scholar]
- 89.Manoutcharian K., Diaz-Orea A., Gevorkian G., Fragoso G., Acero G., Gonzalez E. Recombinant bacteriophage-based multiepitope vaccine against Taenia solium pig cysticercosis. Vet Immunol Immunopathol. 2004;99:11–24. doi: 10.1016/j.vetimm.2003.12.009. [DOI] [PubMed] [Google Scholar]
- 90.Yang Y., Chen J., Li H., Wang Y., Xie Z., Wu M. Porcine interleukin-2 gene encapsulated in chitosan nanoparticles enhances immune response of mice to piglet paratyphoid vaccine. Comp Immunol Microbiol Infect Dis. 2007;30:19–32. doi: 10.1016/j.cimid.2006.09.006. [DOI] [PubMed] [Google Scholar]
- 91.Ren Z.J., Tian C.J., Zhu Q.S., Zhao M.Y., Xin A.G., Nie W.X. Orally delivered foot-and-mouth disease virus capsid protomer vaccine displayed on T4 bacteriophage surface: 100% protection from potency challenge in mice. Vaccine. 2008;26:1471–1481. doi: 10.1016/j.vaccine.2007.12.053. [DOI] [PubMed] [Google Scholar]
- 92.Lo-Man R., Rueda P., Sedlik C., Deriaud E., Casal I., Leclerc C. A recombinant virus-like particle system derived from parvovirus as an efficient antigen carrier to elicit a polarized Th1 immune response without adjuvant. Eur J Immunol. 1998;28:1401–1407. doi: 10.1002/(SICI)1521-4141(199804)28:04<1401::AID-IMMU1401>3.0.CO;2-M. [DOI] [PubMed] [Google Scholar]
- 93.Fenaux M., Opriessnig T., Halbur P.G., Meng X.J. Immunogenicity and pathogenicity of chimeric infectious DNA clones of pathogenic porcine circovirus type 2 (PCV2) and nonpathogenic PCV1 in weanling pigs. J Virol. 2003;77:11232–11243. doi: 10.1128/JVI.77.20.11232-11243.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Fenaux M., Opriessnig T., Halbur P.G., Elvinger F., Meng X.J. A chimeric porcine circovirus (PCV) with the immunogenic capsid gene of the pathogenic PCV type 2 (PCV2) cloned into the genomic backbone of the nonpathogenic PCV1 induces protective immunity against PCV2 infection in pigs. J Virol. 2004;78:6297–6303. doi: 10.1128/JVI.78.12.6297-6303.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Pan Q., He K., Huang K. Development of recombinant porcine parvovirus-like particles as an antigen carrier formed by the hybrid VP2 protein carrying immunoreactive epitope of porcine circovirus type 2. Vaccine. 2008 doi: 10.1016/j.vaccine.2008.02.037. [DOI] [PubMed] [Google Scholar]
- 96.Kreuter J. Nanoparticles and microparticles for drug and vaccine delivery. J Anat. 1996;189:503–505. [PMC free article] [PubMed] [Google Scholar]
- 97.Ogris M., Brunner S., Schuller S., Kircheis R., Wagner E. PEGylated DNA/transferrin-PEI complexes: reduced interaction with blood components, extended circulation in blood and potential for systemic gene delivery. Gene Ther. 1999;6:595–605. doi: 10.1038/sj.gt.3300900. [DOI] [PubMed] [Google Scholar]
- 98.Gu X.G., Schmitt M., Hiasa A., Nagata Y., Ikeda H., Sasaki Y. A novel hydrophobized polysaccharide/oncoprotein complex vaccine induces in vitro and in vivo cellular and humoral immune responses against HER2-expressing murine sarcomas. Cancer Res. 1998;58:3385–3390. [PubMed] [Google Scholar]
- 99.Johansen P., Estevez F., Zurbriggen R., Merkle H.P., Gluck R., Corradin G. Towards clinical testing of a single-administration tetanus vaccine based on PLA/PLGA microspheres. Vaccine. 2000;19:1047–1054. doi: 10.1016/s0264-410x(00)00343-1. [DOI] [PubMed] [Google Scholar]
- 100.Vajdy M., O’Hagan D.T. Microparticles for intranasal immunization. Adv Drug Deliv Rev. 2001;51:127–141. doi: 10.1016/s0169-409x(01)00167-3. [DOI] [PubMed] [Google Scholar]
- 101.Cui Z., Mumper R.J. The effect of co-administration of adjuvants with a nanoparticle-based genetic vaccine delivery system on the resulting immune responses. Eur J Pharm Biopharm. 2003;55:11–18. doi: 10.1016/s0939-6411(02)00129-7. [DOI] [PubMed] [Google Scholar]
- 102.Katare Y.K., Panda A.K., Lalwani K., Haque I.U., Ali M.M. Potentiation of immune response from polymer-entrapped antigen: toward development of single dose tetanus toxoid vaccine. Drug Deliv. 2003;10:231–238. doi: 10.1080/drd_10_4_231. [DOI] [PubMed] [Google Scholar]
- 103.Locher C.P., Putnam D., Langer R., Witt S.A., Ashlock B.M., Levy J.A. Enhancement of a human immunodeficiency virus env DNA vaccine using a novel polycationic nanoparticle formulation. Immunol Lett. 2003;90:67–70. doi: 10.1016/j.imlet.2003.02.001. [DOI] [PubMed] [Google Scholar]
- 104.Zhou S., Liao X., Li X., Deng X., Li H. Poly-d,l-lactide-co-poly(ethylene glycol) microspheres as potential vaccine delivery systems. J Control Release. 2003;86:195–205. doi: 10.1016/s0168-3659(02)00423-6. [DOI] [PubMed] [Google Scholar]
- 105.Farokhzad O.C., Jon S., Khademhosseini A., Tran T.N., Lavan D.A., Langer R. Nanoparticle-aptamer bioconjugates: a new approach for targeting prostate cancer cells. Cancer Res. 2004;64:7668–7672. doi: 10.1158/0008-5472.CAN-04-2550. [DOI] [PubMed] [Google Scholar]
- 106.Chong C.S., Cao M., Wong W.W., Fischer K.P., Addison W.R., Kwon G.S. Enhancement of T helper type 1 immune responses against hepatitis B virus core antigen by PLGA nanoparticle vaccine delivery. J Control Release. 2005;102:85–99. doi: 10.1016/j.jconrel.2004.09.014. [DOI] [PubMed] [Google Scholar]
- 107.Tobio M., Gref R., Sanchez A., Langer R., Alonso M.J. Stealth PLA-PEG nanoparticles as protein carriers for nasal administration. Pharm Res. 1998;15:270–275. doi: 10.1023/a:1011922819926. [DOI] [PubMed] [Google Scholar]
- 108.Eyles J.E., Bramwell V.W., Williamson E.D., Alpar H.O. Microsphere translocation and immunopotentiation in systemic tissues following intranasal administration. Vaccine. 2001;19:4732–4742. doi: 10.1016/s0264-410x(01)00220-1. [DOI] [PubMed] [Google Scholar]
- 109.van der Lubben I.M., Kersten G., Fretz M.M., Beuvery C., Coos Verhoef J., Junginger H.E. Chitosan microparticles for mucosal vaccination against diphtheria: oral and nasal efficacy studies in mice. Vaccine. 2003;21:1400–1408. doi: 10.1016/s0264-410x(02)00686-2. [DOI] [PubMed] [Google Scholar]
- 110.Koping-Hoggard M., Sanchez A., Alonso M.J. Nanoparticles as carriers for nasal vaccine delivery. Expert Rev Vaccines. 2005;4:185–196. doi: 10.1586/14760584.4.2.185. [DOI] [PubMed] [Google Scholar]
- 111.O’Hagan D.T., Singh M., Ulmer J.B. Microparticle-based technologies for vaccines. Methods. 2006;40:10–19. doi: 10.1016/j.ymeth.2006.05.017. [DOI] [PubMed] [Google Scholar]
- 112.Zhao ZZ, Zhang HB, Chen Q, Su D, Xie Z, Wang YY, et al. Promotion of immunity of mice to Pasteurella multocida and hog cholera vaccine by pig interleukin-6 gene and CpG motifs. Comp Immunol Microbiol Infect Dis 2008, in press [available online 14 February 2008]. [DOI] [PubMed]
- 113.Fu M.L., Ying S.C., Wu M., Li H., Wu K.Y., Yang Y. Regulating effects of novel CpG chitosan-nanoparticles on immune responses of mice to porcine paratyphoid vaccines. Biomed Environ Sci. 2006;19:315–322. [PubMed] [Google Scholar]
- 114.Macklin M.D., McCabe D., McGregor M.W., Neumann V., Meyer T., Callan R. Immunization of pigs with a particle-mediated DNA vaccine to influenza A virus protects against challenge with homologous virus. J Virol. 1998;72:1491–1496. doi: 10.1128/jvi.72.2.1491-1496.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Swain W.F., Macklin M.D., Neumann G., McCabe D.E., Drape R., Fuller J.T. Manipulation of immune responses via particle-mediated polynucleotide vaccines. Behring Inst Mitt. 1997;98:73–78. [PubMed] [Google Scholar]
- 116.Kohli A.K., Alpar H.O. Potential use of nanoparticles for transcutaneous vaccine delivery: effect of particle size and charge. Int J Pharm. 2004;275:13–17. doi: 10.1016/j.ijpharm.2003.10.038. [DOI] [PubMed] [Google Scholar]
- 117.Ceppi M., Ruggli N., Tache V., Gerber H., McCullough K.C., Summerfield A. Double-stranded secondary structures on mRNA induce type I interferon (IFN alpha/beta) production and maturation of mRNA-transfected monocyte-derived dendritic cells. J Gene Med. 2005;7:452–465. doi: 10.1002/jgm.685. [DOI] [PubMed] [Google Scholar]
- 118.Paglia P., Medina E., Arioli I., Guzman C.A., Colombo M.P. Gene transfer in dendritic cells, induced by oral DNA vaccination with Salmonella typhimurium, results in protective immunity against a murine fibrosarcoma. Blood. 1998;92:3172–3176. [PubMed] [Google Scholar]
- 119.Cochlovius B., Stassar M.J., Schreurs M.W., Benner A., Adema G.J. Oral DNA vaccination: antigen uptake and presentation by dendritic cells elicits protective immunity. Immunol Lett. 2002;80:89–96. doi: 10.1016/s0165-2478(01)00313-3. [DOI] [PubMed] [Google Scholar]
- 120.Franco L.H., Wowk P.F., Silva C.L., Trombone A.P., Coelho-Castelo A.A., Oliver C. A DNA vaccine against tuberculosis based on the 65 kDa heat-shock protein differentially activates human macrophages and dendritic cells. Genet Vaccines Ther. 2008;6:3–13. doi: 10.1186/1479-0556-6-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Bot A., Stan A.C., Inaba K., Steinman R., Bona C. Dendritic cells at a DNA vaccination site express the encoded influenza nucleoprotein and prime MHC class I-restricted cytolytic lymphocytes upon adoptive transfer. Int Immunol. 2000;12:825–832. doi: 10.1093/intimm/12.6.825. [DOI] [PubMed] [Google Scholar]
- 122.Watkins C., Hopkins J., Harkiss G. Reporter gene expression in dendritic cells after gene gun administration of plasmid DNA. Vaccine. 2005;23:4247–4256. doi: 10.1016/j.vaccine.2005.03.023. [DOI] [PubMed] [Google Scholar]
- 123.Kutzler M.A., Weiner D.B. Developing DNA vaccines that call to dendritic cells. J Clin Invest. 2004;114:1241–1244. doi: 10.1172/JCI23467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Spies B., Hochrein H., Vabulas M., Huster K., Busch D.H., Schmitz F. Vaccination with plasmid DNA activates dendritic cells via Toll-like receptor 9 (TLR9) but functions in TLR9-deficient mice. J Immunol. 2003;171:5908–5912. doi: 10.4049/jimmunol.171.11.5908. [DOI] [PubMed] [Google Scholar]
- 125.You Z., Huang X., Hester J., Toh H.C., Chen S.Y. Targeting dendritic cells to enhance DNA vaccine potency. Cancer Res. 2001;61:3704–3711. [PubMed] [Google Scholar]
- 126.Nchinda G., Kuroiwa J., Oks M., Trumpfheller C., Park C.G., Huang Y. The efficacy of DNA vaccination is enhanced in mice by targeting the encoded protein to dendritic cells. J Clin Invest. 2008;118:1427–1436. doi: 10.1172/JCI34224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Gu Q.L., Huang X., Ren W.H., Shen L., Liu B.Y., Chen S.Y. Targeting hepatitis B virus antigens to dendritic cells by heat shock protein to improve DNA vaccine potency. World J Gastroenterol. 2007;13:5911–5917. doi: 10.3748/wjg.v13.i44.5911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Lauterbach H., Gruber A., Ried C., Cheminay C., Brocker T. Insufficient APC capacities of dendritic cells in gene gun-mediated DNA vaccination. J Immunol. 2006;176:4600–4607. doi: 10.4049/jimmunol.176.8.4600. [DOI] [PubMed] [Google Scholar]
- 129.Porgador A., Irvine K.R., Iwasaki A., Barber B.H., Restifo N.P., Germain R.N. Predominant role for directly transfected dendritic cells in antigen presentation to CD8+ T cells after gene gun immunization. J Exp Med. 1998;188:1075–1082. doi: 10.1084/jem.188.6.1075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Jilek S., Merkle H.P., Walter E. DNA-loaded biodegradable microparticles as vaccine delivery systems and their interaction with dendritic cells. Adv Drug Deliv Rev. 2005;57:377–390. doi: 10.1016/j.addr.2004.09.010. [DOI] [PubMed] [Google Scholar]
- 131.Jilek S., Zurkaulen H., Pavlovic J., Merkle H.P., Walter E. Transfection of a mouse dendritic cell line by plasmid DNA-loaded PLGA microparticles in vitro. Eur J Pharm Biopharm. 2004;58:491–499. doi: 10.1016/j.ejpb.2004.03.038. [DOI] [PubMed] [Google Scholar]
- 132.Walter E., Merkle H.P. Microparticle-mediated transfection of non-phagocytic cells in vitro. J Drug Target. 2002;10:11–21. doi: 10.1080/10611860290007478. [DOI] [PubMed] [Google Scholar]
- 133.Lunsford L., McKeever U., Eckstein V., Hedley M.L. Tissue distribution and persistence in mice of plasmid DNA encapsulated in a PLGA-based microsphere delivery vehicle. J Drug Target. 2000;8:39–50. doi: 10.3109/10611860009009208. [DOI] [PubMed] [Google Scholar]
- 134.Trombone A.P., Silva C.L., Almeida L.P., Rosada R.S., Lima K.M., Oliver C. Tissue distribution of DNA-Hsp65/TDM-loaded PLGA microspheres and uptake by phagocytic cells. Genet Vaccines Ther. 2007;5:9–16. doi: 10.1186/1479-0556-5-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Dincer Z., Jones S., Haworth R. Preclinical safety assessment of a DNA vaccine using particle-mediated epidermal delivery in domestic pig, minipig and mouse. Exp Toxicol Pathol. 2006;57:351–357. doi: 10.1016/j.etp.2006.03.014. [DOI] [PubMed] [Google Scholar]
- 136.Gamvrellis A., Leong D., Hanley J.C., Xiang S.D., Mottram P., Plebanski M. Vaccines that facilitate antigen entry into dendritic cells. Immunol Cell Biol. 2004;82:506–516. doi: 10.1111/j.0818-9641.2004.01271.x. [DOI] [PubMed] [Google Scholar]
- 137.Ceppi M., de Bruin M.G., Seuberlich T., Balmelli C., Pascolo S., Ruggli N. Identification of classical swine fever virus protein E2 as a target for cytotoxic T cells by using mRNA-transfected antigen-presenting cells. J Gen Virol. 2005;86:2525–2534. doi: 10.1099/vir.0.80907-0. [DOI] [PubMed] [Google Scholar]
- 138.Prabha S., Zhou W.Z., Panyam J., Labhasetwar V. Size-dependency of nanoparticle-mediated gene transfection: studies with fractionated nanoparticles. Int J Pharm. 2002;244:105–115. doi: 10.1016/s0378-5173(02)00315-0. [DOI] [PubMed] [Google Scholar]
- 139.Panyam J., Labhasetwar V. Biodegradable nanoparticles for drug and gene delivery to cells and tissue. Adv Drug Deliv Rev. 2003;55:329–347. doi: 10.1016/s0169-409x(02)00228-4. [DOI] [PubMed] [Google Scholar]
- 140.Crocker P.R. Siglecs in innate immunity. Curr Opin Pharmacol. 2005;5:431–437. doi: 10.1016/j.coph.2005.03.003. [DOI] [PubMed] [Google Scholar]
- 141.Crocker P.R., Varki A. Siglecs in the immune system. Immunology. 2001;103:137–145. doi: 10.1046/j.0019-2805.2001.01241.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Varki A., Angata T. Siglecs—the major subfamily of I-type lectins. Glycobiology. 2006;16:1R–27R. doi: 10.1093/glycob/cwj008. [DOI] [PubMed] [Google Scholar]
- 143.Zhang J., Raper A., Sugita N., Hingorani R., Salio M., Palmowski M.J. Characterization of Siglec-H as a novel endocytic receptor expressed on murine plasmacytoid dendritic cell precursors. Blood. 2006;107:3600–3608. doi: 10.1182/blood-2005-09-3842. [DOI] [PubMed] [Google Scholar]
- 144.Lock K., Zhang J., Lu J., Lee S.H., Crocker P.R. Expression of CD33-related siglecs on human mononuclear phagocytes, monocyte-derived dendritic cells and plasmacytoid dendritic cells. Immunobiology. 2004;209:199–207. doi: 10.1016/j.imbio.2004.04.007. [DOI] [PubMed] [Google Scholar]
- 145.Li N., Zhang W., Wan T., Zhang J., Chen T., Yu Y. Cloning and characterization of Siglec-10, a novel sialic acid binding member of the Ig superfamily, from human dendritic cells. J Biol Chem. 2001;276:28106–28112. doi: 10.1074/jbc.M100467200. [DOI] [PubMed] [Google Scholar]
- 146.Nicoll G., Ni J., Liu D., Klenerman P., Munday J., Dubock S. Identification and characterization of a novel siglec, siglec-7, expressed by human natural killer cells and monocytes. J Biol Chem. 1999;274:34089–34095. doi: 10.1074/jbc.274.48.34089. [DOI] [PubMed] [Google Scholar]
- 147.Angata T., Tabuchi Y., Nakamura K., Nakamura M. Siglec-15: an immune system Siglec conserved throughout vertebrate evolution. Glycobiology. 2007;17:838–846. doi: 10.1093/glycob/cwm049. [DOI] [PubMed] [Google Scholar]
- 148.Bax M., Garcia-Vallejo J.J., Jang-Lee J., North S.J., Gilmartin T.J., Hernandez G. Dendritic cell maturation results in pronounced changes in glycan expression affecting recognition by siglecs and galectins. J Immunol. 2007;179:8216–8224. doi: 10.4049/jimmunol.179.12.8216. [DOI] [PubMed] [Google Scholar]
- 149.Pyz E., Marshall A.S., Gordon S., Brown G.D. C-type lectin-like receptors on myeloid cells. Ann Med. 2006;38:242–251. doi: 10.1080/07853890600608985. [DOI] [PubMed] [Google Scholar]
- 150.Kretz-Rommel A., Qin F., Dakappagari N., Torensma R., Faas S., Wu D. In vivo targeting of antigens to human dendritic cells through DC-SIGN elicits stimulatory immune responses and inhibits tumor growth in grafted mouse models. J Immunother. 2007;30:715–726. doi: 10.1097/CJI.0b013e318135472c. [DOI] [PubMed] [Google Scholar]
- 151.Pereira C.F., Torensma R., Hebeda K., Kretz-Rommel A., Faas S., Figdor C.G. In vivo targeting of DC-SIGN-positive antigen-presenting cells in a nonhuman primate model. J Immunother. 2007;30:705–714. doi: 10.1097/CJI.0b013e31812e6256. [DOI] [PubMed] [Google Scholar]
- 152.Barondes S.H., Castronovo V., Cooper D.N., Cummings R.D., Drickamer K., Feizi T. Galectins: a family of animal beta-galactoside-binding lectins. Cell. 1994;76:597–598. doi: 10.1016/0092-8674(94)90498-7. [DOI] [PubMed] [Google Scholar]
- 153.Blasius A.L., Colonna M. Sampling and signaling in plasmacytoid dendritic cells: the potential roles of Siglec-H. Trends Immunol. 2006;27:255–260. doi: 10.1016/j.it.2006.04.005. [DOI] [PubMed] [Google Scholar]
- 154.Vanderheijden N., Delputte P.L., Favoreel H.W., Vandekerckhove J., Van Damme J., van Woensel P.A. Involvement of sialoadhesin in entry of porcine reproductive and respiratory syndrome virus into porcine alveolar macrophages. J Virol. 2003;77:8207–8215. doi: 10.1128/JVI.77.15.8207-8215.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Steeghs L., van Vliet S.J., Uronen-Hansson H., van Mourik A., Engering A., Sanchez-Hernandez M. Neisseria meningitidis expressing lgtB lipopolysaccharide targets DC-SIGN and modulates dendritic cell function. Cell Microbiol. 2006;8:316–325. doi: 10.1111/j.1462-5822.2005.00623.x. [DOI] [PubMed] [Google Scholar]
- 156.Meyer S., van Liempt E., Imberty A., van Kooyk Y., Geyer H., Geyer R. DC-SIGN mediates binding of dendritic cells to authentic pseudo-LewisY glycolipids of Schistosoma mansoni cercariae, the first parasite-specific ligand of DC-SIGN. J Biol Chem. 2005;280:37349–37359. doi: 10.1074/jbc.M507100200. [DOI] [PubMed] [Google Scholar]
- 157.Sheng K.C., Pouniotis D.S., Wright M.D., Tang C.K., Lazoura E., Pietersz G.A. Mannan derivatives induce phenotypic and functional maturation of mouse dendritic cells. Immunology. 2006;118:372–383. doi: 10.1111/j.1365-2567.2006.02384.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Apostolopoulos V., Pietersz G.A., Loveland B.E., Sandrin M.S., McKenzie I.F. Oxidative/reductive conjugation of mannan to antigen selects for T1 or T2 immune responses. Proc Natl Acad Sci USA. 1995;92:10128–10132. doi: 10.1073/pnas.92.22.10128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Bessler W.G., Jung G. Synthetic lipopeptides as novel adjuvants. Res Immunol. 1992;143:548–553. doi: 10.1016/0923-2494(92)80067-u. [DOI] [PubMed] [Google Scholar]
- 160.Goethe R., Gonzales O.F., Lindner T., Gerlach G.F. A novel strategy for protective Actinobacillus pleuropneumoniae subunit vaccines: detergent extraction of cultures induced by iron restriction. Vaccine. 2000;19:966–975. doi: 10.1016/s0264-410x(00)00212-7. [DOI] [PubMed] [Google Scholar]
- 161.Meens J., Selke M., Gerlach G.F. Identification and immunological characterization of conserved Mycoplasma hyopneumoniae lipoproteins Mhp378 and Mhp651. Vet Microbiol. 2006;116:85–95. doi: 10.1016/j.vetmic.2006.03.011. [DOI] [PubMed] [Google Scholar]
- 162.Aranda J., Garrido M.E., Cortés P., Llagostera M., Barbé J. Analysis of the protective capacity of three Streptococcus suis proteins induced under divalent-cation-limited conditions. Infect Immun. 2008;76:1590–1598. doi: 10.1128/IAI.00987-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Rau H., Revets H., Cornelis P., Titzmann A., Ruggli N., McCullough K.C. Efficacy and functionality of lipoprotein OprI from Pseudomonas aeruginosa as adjuvant for a subunit vaccine against classical swine fever. Vaccine. 2006;24:4757–4768. doi: 10.1016/j.vaccine.2006.03.028. [DOI] [PubMed] [Google Scholar]
- 164.Moyle P.M., Toth I. Self-adjuvanting lipopeptide vaccines. Curr Med Chem. 2008;15:506–516. doi: 10.2174/092986708783503249. [DOI] [PubMed] [Google Scholar]
- 165.Cheng G., Zhao X., Yan W., Wang W., Zuo X., Huang K. Alpha interferon is a powerful adjuvant for a recombinant protein vaccine against foot-and-mouth disease virus in swine, and an effective stimulus of in vivo immune response. Vaccine. 2007;25:5199–5208. doi: 10.1016/j.vaccine.2007.04.089. [DOI] [PubMed] [Google Scholar]
- 166.O’Hagan D.T. Recent developments in vaccine delivery systems. Curr Drug Targets Infect Disord. 2001;1:273–286. doi: 10.2174/1568005014606008. [DOI] [PubMed] [Google Scholar]
- 167.Singh M. Recent advances in vaccine adjuvants. Pharm Res. 2002;19:715–728. doi: 10.1023/a:1016104910582. [DOI] [PubMed] [Google Scholar]
- 168.O’Hagan D.T., Singh M. Microparticles as vaccine adjuvants and delivery systems. Expert Rev Vaccines. 2003;2:269–283. doi: 10.1586/14760584.2.2.269. [DOI] [PubMed] [Google Scholar]
- 169.Moyle P.M., McGeary R.P., Blanchfield J.T., Toth I. Mucosal immunisation: adjuvants and delivery systems. Curr Drug Deliv. 2004;1:385–396. doi: 10.2174/1567201043334588. [DOI] [PubMed] [Google Scholar]
- 170.Patel G.B., Chen W. Archaeosome immunostimulatory vaccine delivery system. Curr Drug Deliv. 2005;2(4):407–421. doi: 10.2174/156720105774370285. [DOI] [PubMed] [Google Scholar]
- 171.Christensen D., Korsholm K.S., Rosenkrands I., Lindenström T., Andersen P., Agger E.M. Cationic liposomes as vaccine adjuvants. Expert Rev Vaccines. 2007;6:785–796. doi: 10.1586/14760584.6.5.785. [DOI] [PubMed] [Google Scholar]
- 172.Allsopp C.E., Plebanski M., Gilbert S., Sinden R.E., Harris S., Frankel G. Comparison of numerous delivery systems for the induction of cytotoxic T lymphocytes by immunization. Eur J Immunol. 1996;26:1951–1959. doi: 10.1002/eji.1830260841. [DOI] [PubMed] [Google Scholar]
- 173.Boato F., Thomas R.M., Ghasparian A., Freund-Renard A., Moehle K., Robinson J.A. Synthetic virus-like particles from self-assembling coiled-coil lipopeptides and their use in antigen display to the immune system. Angew Chem Int Ed Engl. 2007;46:9015–9018. doi: 10.1002/anie.200702805. [DOI] [PubMed] [Google Scholar]
- 174.Nam J.H., Cha S.L., Cho H.W. Immunogenicity of a recombinant MVA and a DNA vaccine for Japanese encephalitis virus in swine. Microbiol Immunol. 2002;46:23–28. doi: 10.1111/j.1348-0421.2002.tb02672.x. [DOI] [PubMed] [Google Scholar]
- 175.Konishi E., Pincus S., Paoletti E., Laegreid W.W., Shope R.E., Mason P.W. A highly attenuated host range-restricted vaccinia virus strain, NYVAC, encoding the prM, E, and NS1 genes of Japanese encephalitis virus prevents JEV viremia in swine. Virology. 1992;190:454–458. doi: 10.1016/0042-6822(92)91233-k. [DOI] [PubMed] [Google Scholar]
- 176.Murata H., Britton P., Kaeffer B., Aynaud J.M., Chevaleyre C., Salmon H. Specific cytotoxic lymphocyte response in swine against structural proteins of transmissible gastro-enteritis virus: a study using lymphoblastoid cell line and recombinant vaccinia virus. Vet Res. 1998;29:467–474. [PubMed] [Google Scholar]
- 177.Ma M., Jin N., Shen G., Zhu G., Liu H.J., Zheng M. Immune responses of swine inoculated with a recombinant fowlpox virus co-expressing P12A and 3C of FMDV and swine IL-18. Vet Immunol Immunopathol. 2008;121:1–7. doi: 10.1016/j.vetimm.2007.06.029. [DOI] [PubMed] [Google Scholar]
- 178.Shen G., Jin N., Ma M., Jin K., Zheng M., Zhuang T. Immune responses of pigs inoculated with a recombinant fowlpox virus coexpressing GP5/GP3 of porcine reproductive and respiratory syndrome virus and swine IL-18. Vaccine. 2007;25:4193–4202. doi: 10.1016/j.vaccine.2007.03.010. [DOI] [PubMed] [Google Scholar]
- 179.Voigt H., Merant C., Wienhold D., Braun A., Hutet E., Le Potier M.F. Efficient priming against classical swine fever with a safe glycoprotein E2 expressing Orf virus recombinant (ORFV VrV-E2) Vaccine. 2007;25:5915–5926. doi: 10.1016/j.vaccine.2007.05.035. [DOI] [PubMed] [Google Scholar]
- 180.Fischer T., Planz O., Stitz L., Rziha H.J. Novel recombinant parapoxvirus vectors induce protective humoral and cellular immunity against lethal herpesvirus challenge infection in mice. J Virol. 2003;77:9312–9323. doi: 10.1128/JVI.77.17.9312-9323.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Büttner M., Czerny C.P., Lehner K.H., Wertz K. Interferon induction in peripheral blood mononuclear leukocytes of man and farm animals by poxvirus vector candidates and some poxvirus constructs. Vet Immunol Immunopathol. 1995;46:237–250. doi: 10.1016/0165-2427(94)05357-x. [DOI] [PubMed] [Google Scholar]
- 182.He Y., Qian P., Zhang K., Yao Q., Wang D., Xu Z. Construction and immune response characterization of a recombinant pseudorabies virus co-expressing capsid precursor protein (P1) and a multiepitope peptide of foot-and-mouth disease virus in swine. Virus Genes. 2008;36:393–400. doi: 10.1007/s11262-008-0204-6. [DOI] [PubMed] [Google Scholar]
- 183.Qian P., Li X.M., Jin M.L., Peng G.Q., Chen H.C. An approach to a FMD vaccine based on genetic engineered attenuated pseudorabies virus: one experiment using VP1 gene alone generates an antibody responds on FMD and pseudorabies in swine. Vaccine. 2004;22:2129–2136. doi: 10.1016/j.vaccine.2003.12.005. [DOI] [PubMed] [Google Scholar]
- 184.Hong Q., Qian P., Li X.M., Yu X.L., Chen H.C. A recombinant pseudorabies virus co-expressing capsid proteins precursor P1-2A of FMDV and VP2 protein of porcine parvovirus: a trivalent vaccine candidate. Biotechnol Lett. 2007;29:1677–1683. doi: 10.1007/s10529-007-9459-6. [DOI] [PubMed] [Google Scholar]
- 185.Xu G., Xu X., Li Z., He Q., Wu B., Sun S. Construction of recombinant pseudorabies virus expressing NS1 protein of Japanese encephalitis (SA14-14-2) virus and its safety and immunogenicity. Vaccine. 2004;22:1846–1853. doi: 10.1016/j.vaccine.2003.09.015. [DOI] [PubMed] [Google Scholar]
- 186.Song Y., Jin M., Zhang S., Xu X., Xiao S., Cao S. Generation and immunogenicity of a recombinant pseudorabies virus expressing cap protein of porcine circovirus type 2. Vet Microbiol. 2007;119:97–104. doi: 10.1016/j.vetmic.2006.08.026. [DOI] [PubMed] [Google Scholar]
- 187.Ju C., Fan H., Tan Y., Liu Z., Xi X., Cao S. Immunogenicity of a recombinant pseudorabies virus expressing ORF1-ORF2 fusion protein of porcine circovirus type 2. Vet Microbiol. 2005;109:179–190. doi: 10.1016/j.vetmic.2005.06.001. [DOI] [PubMed] [Google Scholar]
- 188.Jiang Y., Fang L., Xiao S., Zhang H., Pan Y., Luo R. Immunogenicity and protective efficacy of recombinant pseudorabies virus expressing the two major membrane-associated proteins of porcine reproductive and respiratory syndrome virus. Vaccine. 2007;25:547–560. doi: 10.1016/j.vaccine.2006.07.032. [DOI] [PubMed] [Google Scholar]
- 189.Jiang Y.B., Fang L.R., Xiao S.B., Zhang H., Chen H.C. Construction and immunogenicity of recombinant pseudorabies virus expressing the modified GP5m protein of porcine reproduction and respiratory syndrome virus. Sheng Wu Gong Cheng Xue Bao. 2005;21:858–864. [PubMed] [Google Scholar]
- 190.Qiu H.J., Tian Z.J., Tong G.Z., Zhou Y.J., Ni J.Q., Luo Y.Z. Protective immunity induced by a recombinant pseudorabies virus expressing the GP5 of porcine reproductive and respiratory syndrome virus in piglets. Vet Immunol Immunopathol. 2005;106:309–319. doi: 10.1016/j.vetimm.2005.03.008. [DOI] [PubMed] [Google Scholar]
- 191.Tian Z.J., Qiu H.J., Ni J.Q., Zhou Y.J., Cai X.H., Zhou G.H. Construction and characterization of a recombinant pseudorabies virus expressing porcine reproductive and respiratory syndrome virus GP5. Yi Chuan Xue Bao. 2005;32:1248–1255. [PubMed] [Google Scholar]
- 192.Shiau A.L., Liu C.W., Wang S.Y., Tsai C.Y., Wu C.L. A simple selection system for construction of recombinant gD-negative pseudorabies virus as a vaccine vector. Vaccine. 2002;20:1186–1195. doi: 10.1016/s0264-410x(01)00426-1. [DOI] [PubMed] [Google Scholar]
- 193.Tian Z.J., Zhou G.H., Zheng B.L., Qiu H.J., Ni J.Q., Yang H.L. A recombinant pseudorabies virus encoding the HA gene from H3N2 subtype swine influenza virus protects mice from virulent challenge. Vet Immunol Immunopathol. 2006;111:211–218. doi: 10.1016/j.vetimm.2006.01.015. [DOI] [PubMed] [Google Scholar]
- 194.Yin J., Ren X., Tian Z., Li Y. Assembly of pseudorabies virus genome-based transfer vehicle carrying major antigen sites of S gene of transmissible gastroenteritis virus: potential perspective for developing live vector vaccines. Biologicals. 2007;35:55–61. doi: 10.1016/j.biologicals.2006.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Reddy P.S., Idamakanti N., Hyun B.H., Tikoo S.K., Babiuk L.A. Development of porcine adenovirus-3 as an expression vector. J Gen Virol. 1999;80:563–570. doi: 10.1099/0022-1317-80-3-563. [DOI] [PubMed] [Google Scholar]
- 196.Hammond J.M., Jansen E.S., Morrissy C.J., Hodgson A.L., Johnson M.A. Protection of pigs against ‘in contact’ challenge with classical swine fever following oral or subcutaneous vaccination with a recombinant porcine adenovirus. Virus Res. 2003;97:151–157. doi: 10.1016/j.virusres.2003.08.003. [DOI] [PubMed] [Google Scholar]
- 197.Hammond J.M., Jansen E.S., Morrissy C.J., Williamson M.M., Hodgson A.L., Johnson M.A. Oral and sub-cutaneous vaccination of commercial pigs with a recombinant porcine adenovirus expressing the classical swine fever virus gp55 gene. Arch Virol. 2001;146:1787–1793. doi: 10.1007/s007050170064. [DOI] [PubMed] [Google Scholar]
- 198.Liu Y., Hu R., Zhang S., Zhang F., Li Z., Wei X. Expression of the foot-and-mouth disease virus VP1 protein using a replication-competent recombinant canine adenovirus type 2 elicits a humoral antibody response in a porcine model. Viral Immunol. 2006;19:202–209. doi: 10.1089/vim.2006.19.202. [DOI] [PubMed] [Google Scholar]
- 199.Mayr G.A., Chinsangaram J., Grubman M.J. Development of replication-defective adenovirus serotype 5 containing the capsid and 3C protease coding regions of foot-and-mouth disease virus as a vaccine candidate. Virology. 1999;263:496–506. doi: 10.1006/viro.1999.9940. [DOI] [PubMed] [Google Scholar]
- 200.Okamba F.R., Moreau E., Cheikh Saad Bouh K., Gagnon C.A., Massie B., Arella M. Immune responses induced by replication-defective adenovirus expressing the C-terminal portion of the Mycoplasma hyopneumoniae P97 adhesin. Clin Vaccine Immunol. 2007;14:767–774. doi: 10.1128/CVI.00415-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Wang X., Jiang P., Li Y., Jiang W., Dong X. Protection of pigs against post-weaning multisystemic wasting syndrome by a recombinant adenovirus expressing the capsid protein of porcine circovirus type 2. Vet Microbiol. 2007;121:215–224. doi: 10.1016/j.vetmic.2006.11.027. [DOI] [PubMed] [Google Scholar]
- 202.Wang X., Jiang W., Jiang P., Li Y., Feng Z., Xu J. Construction and immunogenicity of recombinant adenovirus expressing the capsid protein of porcine circovirus 2 (PCV2) in mice. Vaccine. 2006;24:3374–3380. doi: 10.1016/j.vaccine.2005.12.068. [DOI] [PubMed] [Google Scholar]
- 203.Jiang W., Jiang P., Li Y., Wang X., Du Y. Analysis of immunogenicity of minor envelope protein GP3 of porcine reproductive and respiratory syndrome virus in mice. Virus Genes. 2007;35:695–704. doi: 10.1007/s11262-007-0143-7. [DOI] [PubMed] [Google Scholar]
- 204.Jiang W., Jiang P., Li Y., Tang J., Wang X., Ma S. Recombinant adenovirus expressing GP5 and M fusion proteins of porcine reproductive and respiratory syndrome virus induce both humoral and cell-mediated immune responses in mice. Vet Immunol Immunopathol. 2006;113:169–180. doi: 10.1016/j.vetimm.2006.05.001. [DOI] [PubMed] [Google Scholar]
- 205.Callebaut P., Enjuanes L., Pensaert M. An adenovirus recombinant expressing the spike glycoprotein of porcine respiratory coronavirus is immunogenic in swine. J Gen Virol. 1996;77:309–313. doi: 10.1099/0022-1317-77-2-309. [DOI] [PubMed] [Google Scholar]
- 206.Hammond J.M., Jansen E.S., Morrissy C.J., van der Heide B., Goff W.V., Williamson M.M. Vaccination of pigs with a recombinant porcine adenovirus expressing the gD gene from pseudorabies virus. Vaccine. 2001;19:3752–3758. doi: 10.1016/s0264-410x(01)00084-6. [DOI] [PubMed] [Google Scholar]
- 207.Adam M., Lepottier M.F., Eloit M. Vaccination of pigs with replication-defective adenovirus vectored vaccines: the example of pseudorabies. Vet Microbiol. 1994;42:205–215. doi: 10.1016/0378-1135(94)90019-1. [DOI] [PubMed] [Google Scholar]
- 208.Wesley R.D. Overcoming maternal antibody interference by vaccination with human adenovirus 5 recombinant viruses expressing the hemagglutinin and the nucleoprotein of swine influenza virus. Vet Microbiol. 2006;118:67–75. doi: 10.1016/j.vetmic.2006.07.014. [DOI] [PubMed] [Google Scholar]
- 209.Wesley R.D., Lager K.M. Evaluation of a recombinant human adenovirus-5 vaccine administered via needle-free device and intramuscular injection for vaccination of pigs against swine influenza virus. Am J Vet Res. 2005;66:1943–1947. doi: 10.2460/ajvr.2005.66.1943. [DOI] [PubMed] [Google Scholar]
- 210.Tang M., Harp J.A., Wesley R.D. Recombinant adenovirus encoding the HA gene from swine H3N2 influenza virus partially protects mice from challenge with heterologous virus: A/HK/1/68 (H3N2) Arch Virol. 2002;147:2125–2141. doi: 10.1007/s00705-002-0870-y. [DOI] [PubMed] [Google Scholar]
- 211.Yang C.D., Liao J.T., Lai C.Y., Jong M.H., Liang C.M., Lin Y.L. Induction of protective immunity in swine by recombinant bamboo mosaic virus expressing foot-and-mouth disease virus epitopes. BMC Biotechnol. 2007;7:62. doi: 10.1186/1472-6750-7-62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Wang Y., Liang Y., Han J., Burkhart K.M., Vaughn E.M., Roof M.B. Attenuation of porcine reproductive and respiratory syndrome virus strain MN184 using chimeric construction with vaccine sequence. Virology. 2008;371:418–429. doi: 10.1016/j.virol.2007.09.032. [DOI] [PubMed] [Google Scholar]
- 213.Singh M., Chakrapani A., O’Hagan D.T. Nanoparticles and microparticles as vaccine-delivery systems. Expert Rev Vaccines. 2007;6:797–808. doi: 10.1586/14760584.6.5.797. [DOI] [PubMed] [Google Scholar]
- 214.Vyas S.P., Gupta P.N. Implication of nanoparticles/microparticles in mucosal vaccine delivery. Expert Rev Vaccines. 2007;6:401–418. doi: 10.1586/14760584.6.3.401. [DOI] [PubMed] [Google Scholar]
- 215.Davis S.S. The use of soluble polymers and polymer microparticles to provide improved vaccine responses after parenteral and mucosal delivery. Vaccine. 2006;24:7–10. doi: 10.1016/j.vaccine.2005.01.102. [DOI] [PubMed] [Google Scholar]
- 216.Azad N., Rojanasakul Y. Vaccine delivery—current trends and future. Curr Drug Deliv. 2006;3:137–146. doi: 10.2174/156720106776359249. [DOI] [PubMed] [Google Scholar]
- 217.Singh M., Kazzaz J., Ugozzoli M., Malyala P., Chesko J., O’Hagan D.T. Polylactide-co-glycolide microparticles with surface adsorbed antigens as vaccine delivery systems. Curr Drug Deliv. 2006;3:115–120. doi: 10.2174/156720106775197565. [DOI] [PubMed] [Google Scholar]
- 218.Jiang W., Gupta R.K., Deshpande M.C., Schwendeman S.P. Biodegradable poly(lactic-co-glycolic acid) microparticles for injectable delivery of vaccine antigens. Adv Drug Deliv Rev. 2005;57:391–410. doi: 10.1016/j.addr.2004.09.003. [DOI] [PubMed] [Google Scholar]
- 219.Eyles J.E., Carpenter Z.C., Alpar H.O., Williamson E.D. Immunological aspects of polymer microsphere vaccine delivery systems. J Drug Target. 2003;11:509–514. doi: 10.1080/10611860410001670017. [DOI] [PubMed] [Google Scholar]
- 220.Brayden D.J., Baird A.W. Microparticle vaccine approaches to stimulate mucosal immunisation. Microbes Infect. 2001;3:867–876. doi: 10.1016/s1286-4579(01)01445-9. [DOI] [PubMed] [Google Scholar]
- 221.van der Lubben I.M., Verhoef J.C., Borchard G., Junginger H.E. Chitosan and its derivatives in mucosal drug and vaccine delivery. Eur J Pharm Sci. 2001;14:201–207. doi: 10.1016/s0928-0987(01)00172-5. [DOI] [PubMed] [Google Scholar]
- 222.Torché A.M., Le Dimna M., Le Corre P., Mesplède A., Le Gal S., Cariolet R. Immune responses after local administration of IgY loaded-PLGA microspheres in gut-associated lymphoid tissue in pigs. Vet Immunol Immunopathol. 2006;109:209–217. doi: 10.1016/j.vetimm.2005.08.016. [DOI] [PubMed] [Google Scholar]
- 223.Dawson M., Krauland E., Wirtz D., Hanes J. Transport of polymeric nanoparticle gene carriers in gastric mucus. Biotechnol Prog. 2004;20:851–857. doi: 10.1021/bp0342553. [DOI] [PubMed] [Google Scholar]
- 224.Liao C.W., Chiou H.Y., Yeh K.S., Chen J.R., Weng C.N. Oral immunization using formalin-inactivated Actinobacillus pleuropneumoniae antigens entrapped in microspheres with aqueous dispersion polymers prepared using a co-spray drying process. Prev Vet Med. 2003;61:1–15. doi: 10.1016/s0167-5877(02)00195-2. [DOI] [PubMed] [Google Scholar]
- 225.Osth K., Strindelius L., Larhed A., Ahlander A., Roomans G.M., Sjöholm I. Uptake of ovalbumin-conjugated starch microparticles by pig respiratory nasal mucosa in vitro. J Drug Target. 2003;11:75–82. doi: 10.1080/1061186031000093254. [DOI] [PubMed] [Google Scholar]
- 226.Lin J.H., Weng C.N., Liao C.W., Yeh K.S., Pan M.J. Protective effects of oral microencapsulated Mycoplasma hyopneumoniae vaccine prepared by co-spray drying method. J Vet Med Sci. 2003;65:69–74. doi: 10.1292/jvms.65.69. [DOI] [PubMed] [Google Scholar]
- 227.Aucouturier J., Deville S., Perret C., Vallée I., Boireau P. Assessment of efficacy and safety of various adjuvant formulations with a total soluble extract of Trichinella spiralis. Parasite. 2001;8:126–132. doi: 10.1051/parasite/200108s2126. [DOI] [PubMed] [Google Scholar]
- 228.Singh M., Briones M., O’Hagan D.T. A novel bioadhesive intranasal delivery system for inactivated influenza vaccines. J Control Release. 2001;70:267–276. doi: 10.1016/s0168-3659(00)00330-8. [DOI] [PubMed] [Google Scholar]
- 229.Beier R., Gebert A. Kinetics of particle uptake in the domes of Peyer's patches. Am J Physiol. 1998;275:G130–G137. doi: 10.1152/ajpgi.1998.275.1.G130. [DOI] [PubMed] [Google Scholar]
- 230.Weng C.N., Tzan Y.L., Liu S.D., Lin S.Y., Lee C.J. Protective effects of an oral microencapsulated Mycoplasma hyopneumoniae vaccine against experimental infection in pigs. Res Vet Sci. 1992;3(1):42–46. doi: 10.1016/0034-5288(92)90082-d. [DOI] [PubMed] [Google Scholar]
- 231.Gou M., Dai M., Li X., Yang L., Huang M., Wang Y. Preparation of mannan modified anionic PCL-PEG-PCL nanoparticles at one-step for bFGF antigen delivery to improve humoral immunity. Colloids Surf B Biointerfaces. 2008 doi: 10.1016/j.colsurfb.2007.12.014. [DOI] [PubMed] [Google Scholar]
- 232.Lu Y., Kawakami S., Yamashita F., Hashida M. Development of an antigen-presenting cell-targeted DNA vaccine against melanoma by mannosylated liposomes. Biomaterials. 2007;28:3255–3262. doi: 10.1016/j.biomaterials.2007.03.028. [DOI] [PubMed] [Google Scholar]
- 233.Hattori Y., Kawakami S., Lu Y., Nakamura K., Yamashita F., Hashida M. Enhanced DNA vaccine potency by mannosylated lipoplex after intraperitoneal administration. J Gene Med. 2006;8:824–834. doi: 10.1002/jgm.910. [DOI] [PubMed] [Google Scholar]
- 234.Cui Z., Mumper R.J. Topical immunization using nanoengineered genetic vaccines. J Control Release. 2002;81:173–184. doi: 10.1016/s0168-3659(02)00051-2. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.