Abstract
Chilo iridescent virus (CIV) is the type species for the family Iridoviridae, which are large, isometric, cytoplasmic dsDNA viruses. We examined the mechanism of apoptosis induction by CIV. High CIV doses (CIVXS; 400 μg/ml), UV-irradiated virus (CIVUV; 10 μg/ml) and CVPE (CIV protein extract; 10 μg/ml) induced apoptosis in 60% of treated Choristoneura fumiferana (IPRI-CF-124T) cells. Normal doses of infectious CIV (10 μg/ml) induced apoptosis in only 10% of C. fumiferana (CF) cells. Apoptosis was inhibited by Z-IETD-FMK, an apical caspase inhibitor, indicating that CIV-induced apoptosis requires caspase activity. The putative caspase in CF cells was designated Cf-caspase-i. CIVUV or CVPE enhanced Cf-caspase-i activity by 80% at 24 h relative to mock-treated cells. Since the MAP kinase pathway induces or inhibits apoptosis depending on the context, we used JNK inhibitor SP600125 and demonstrated drastic suppression of CVPE-induced apoptosis. Thus, the JNK signaling pathway is significant for apoptosis in this system. Virus interaction with the cell surface was not sufficient for apoptosis since CIVUV particles bound to polysterene beads failed to induce apoptosis. Endocytosis inhibitors (bafilomycin or ammonium chloride) negated apoptosis induction by CIVUV, CIVXS or CVPE indicating that entry through this mode is required. Given the weak apoptotic response to infectious CIV, we postulated that viral gene expression inhibited apoptosis. CIV infection of cells pretreated with cycloheximide induced apoptosis in 69% of the cells compared to 10% in normal infections. Furthermore, blocking viral DNA replication with aphidicolin or phosphonoacetic acid suppressed apoptosis and Cf-caspase-i activity, indicating that early viral expression is necessary for inhibition of apoptosis, and de novo synthesis of viral proteins is not required for induction. We show for the first time that, in a member of the family Iridoviridae, apoptosis: (i) requires entry and endocytosis of virions or virion proteins, (ii) is inhibited under conditions permitting early viral expression, and (iii) requires the JNK signaling pathway. This is the first report of JNK signal requirement during apoptosis induction by an insect virus.
Keywords: Chilo iridescent virus, Iridovirus, Apoptosis, Inhibition, Endocytosis, Viral early expression, JNK, Apical caspase, Insect, Lepidopteran cells
Introduction
Chilo iridescent virus (CIV) is the type species for the family Iridoviridae (Iyer et al., 2006). The Iridoviridae are large, cytoplasmic DNA viruses with an icosahedral outer capsid, an internal lipid membrane, and an electron-dense core containing a circularly permuted double-stranded DNA genome (209 kbp). CIV infects a wide range of insects (Williams et al., 2005), replicates productively in the cotton boll weevil Anthonomus grandis (Henderson et al., 2001), and induces metamorphic arrest and mortality in this host (McLaughlin et al., 1972). Earlier work showed that UV-treated CIV or viral “soluble” protein extract inhibited host protein synthesis in mosquito cells (Cerutti and Devauchelle, 1980). Recently, our group showed that a CIV virion protein extract (CVPE) induces mortality in neonate boll weevil larvae (Bilimoria, 2001) and apoptosis in spruce budworm Choristoneura fumiferana and boll weevil cell cultures (Paul et al., 2007).
Apoptosis is generally characterized by several criteria. These include caspase activation, cytoplasmic shrinkage, phosphatidylserine exposure, chromatin condensation, DNA fragmentation, and cell blebbing (Hay and Kannourakis, 2002). Apoptosis proceeds through signal transduction pathways, usually involving activation of cysteine-aspartate-specific proteases, i.e., caspases. Various signals activate initiator (apical) caspases, which then trigger effector (executioner) caspases. In Drosophila, one mechanism includes interaction of Eiger (invertebrate tumor necrosis factor ligand) with the wengen membrane receptor. This leads to activation of JNK, which then triggers the central initiator caspase DRONC, instigating a cascade triggering the effector caspase Drice and ultimately apoptosis (Igaki et al., 2002). The Eiger-wengen mediated pathway appears to be the invertebrate equivalent of the “extrinsic” pathway found in mammalian systems. Alternate induction mechanisms in Drosophila include activation of DRONC through ER stress or through bcl-2-type stimulation and could represent invertebrate equivalents of “intrinsic” pathways. Induction of apoptosis in uninfected Spodoptera frugiperda (Lepidoptera: Noctuidae) cells appears to involve the equivalents of mammalian apical caspase and caspase-3 (Manji and Friesen, 2001) as initiator and effector caspases, respectively.
Recently, three iridoviruses from the genera Ranavirus and Lymphocystivirus were shown to induce apoptosis in cell culture (Essbauer and Ahne, 2002, Chinchar et al., 2003, Hu et al., 2004, Imajoh et al., 2004). Apoptosis induction by frog virus-3 required caspases, and induction of apoptosis by red sea bream iridovirus involved effector caspases-3 and 6 (Chinchar et al., 2003, Imajoh et al., 2004). However, the role of viral entry and endocytosis in the induction of apoptosis by the Iridoviridae is not established, and the significance of viral gene expression in apoptosis modulation has not been studied.
In this report, we show for the first time that in a member of the family Iridoviridae: (i) entry and endocytosis of virions or virion proteins are required for apoptosis induction, (ii) apoptosis is inhibited under conditions permitting early viral expression, (iii) apoptosis requires the JNK signaling pathway. This is the first report of JNK signal requirement during apoptosis induction by an insect virus.
Results
Normal and UV-treated Chilo iridescent virus induce apoptotic blebbing in spruce budworm cells
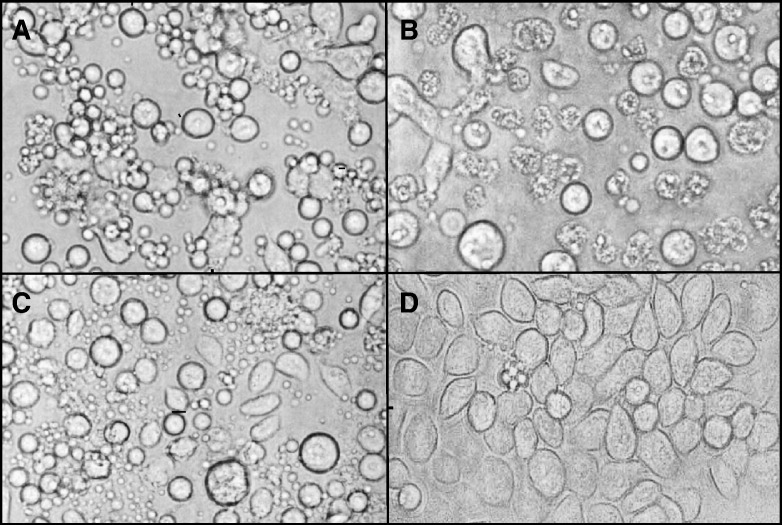
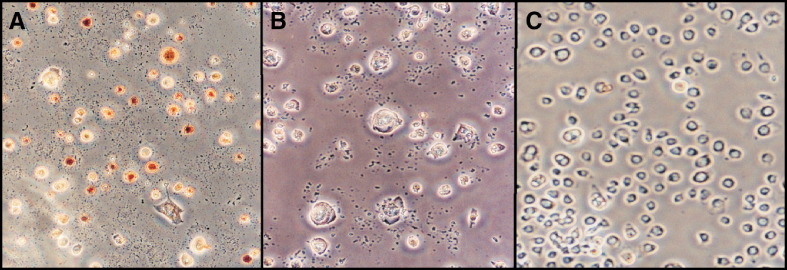
To determine if CIV replication was necessary for induction of apoptosis in C. fumiferana (CF) cells, we treated these cells with normal and UV-inactivated CIV (CIVUV) as described in Materials and methods. Normal, active CIV was used at two concentrations: high dosage (CIVXS; 400 μg/ml) and standard dosage (CIV; 10 μg/ml). Infected cells were incubated at 28 °C as well as 21 °C for 24 h and observed for cell blebbing. Fig. 1 shows that CIVXS induced 70% blebbing (A), whereas CIV induced 10% blebbing and 55% lysis in these cells (B). Lysis was indicated by the presence of cell debris. CIVUV (10 μg/ml) induced 62% blebbing and 5% lysis in CF cells (C). Mock-treated cells incubated at 21 °C or 28 °C did not undergo significant blebbing or lysis (D). The above results are consistent with apoptosis induction using CIV virion protein extract (CVPE) (Bilimoria, 2001, Paul et al., 2007).
Fig. 1.
Induction of blebbing in Choristoneura fumiferana (CF124T) cells treated with Chilo iridescent virus and UV-treated CIV. (A) Cells treated with very large doses of CIV (400 μg/ml). (B) Cells infected with normal doses of CIV (10 μg/ml). (C) Cells treated with UV-irradiated CIV (CIVUV; 10 μg/ml). Incubation for panel A through panel C was at 21 °C. (D) Mock-treated cells were incubated at 21 °C or 28 °C. Observations were made by phase-contrast microscopy. Cells were photographed 24 h after infection or treatment. Magnification: 200×.
Apoptosis induced by high-dosage CIV, CVPE and UV-irradiated CIV is negated by inhibitors of caspase activity
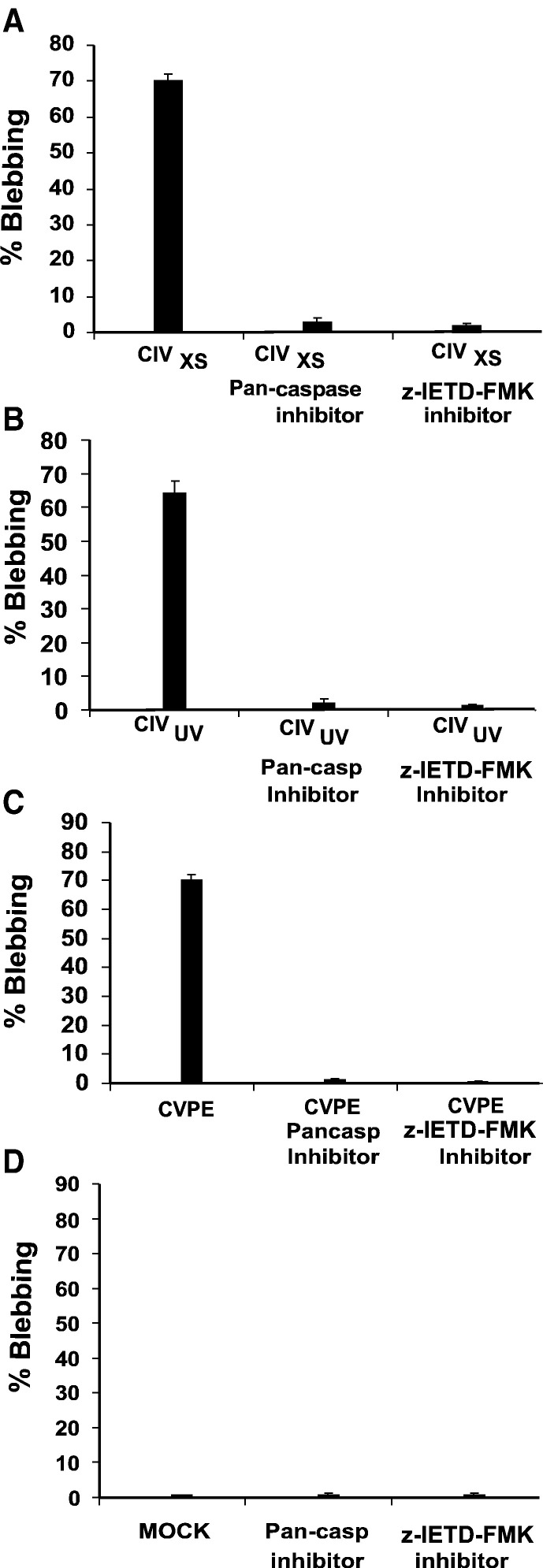
To determine whether apoptosis induced by high-dosage CIV, CVPE, or UV-irradiated CIV was caspase-dependent, CF cells were pretreated with pancaspase inhibitor (Z-VAD-FMK, Calbiochem, CA) and challenged with the above inducers. Pancaspase inhibitor reduced blebbing as follows: CIVXS-induced by 93% (Fig. 2A), CIVUV-induced by 97% (Fig. 2B), CVPE-induced by 99% (Fig. 2C). To establish whether the apoptotic pathway in this system requires an apical caspase, CF cells were pretreated with Z-IETD-FMK (a mammalian caspase-8 inhibitor shown to inhibit lepidopteran, Sf-caspase-X) (Manji and Friesen, 2001) prior to challenge with CIVXS, CIVUV or CVPE. The results show that Z-IETD-FMK blocked apoptotic blebbing by at least 97% for CIVXS (Fig. 2A); 96% for CIVUV (Fig. 2B); 96% for CIVUV; and 98% for CVPE (Fig. 2C). Mock treatment with RBSS yielded less than 2% blebbing (Fig 2D). These results suggest that apoptosis induction in CF cells by high-dosage CIV, CVPE, and UV-treated CIV is dependent on Sf-caspase-X-like activity, which we have designated C. fumiferana initiator caspase (Cf-caspase-i).
Fig. 2.
Effect of pancaspase and apical caspase inhibitors on apoptosis in CF124T cells. Cells were treated with pancaspase inhibitor (Z-VAD-FMK; 50 μM), apical caspase inhibitor (Z-IETD-FMK; 50 μM), or mock-treated and then incubated at 21 °C for 24 h. Inhibitor concentrations and incubation conditions were maintained at these levels for the following. (A) Cells were pretreated with caspase inhibitors for 1 h and then treated with high doses of CIV (400 μg/ml; CIVXS). (B) Cells were pretreated with caspase inhibitors for 1 h and then treated with CIVUV (10 μg/ml). (C) Cells were pretreated with caspase inhibitors for 1 h and then challenged with CVPE (10 μg/ml). (D) Control cells were mock-treated or treated with caspase inhibitors. Percent of the cell population showing blebbing was determined for each treatment set. Results presented are means of 4 separate experiments in which 100 cells from 5 random fields were counted. Error bars represent standard deviations of means for four experiments.
CVPE and UV-irradiated CIV induce apical caspase activity
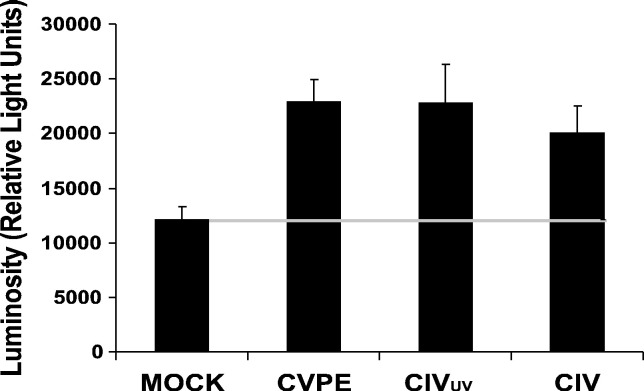
To confirm whether an apical enzyme is indeed activated by CVPE, UV-irradiated CIV, and CIV as suggested by the inhibitor data (Fig. 2), we tested for apical caspase activity using Caspase-Glo Assay kit (Promega, WI) as described in Materials and methods. In this assay the LETD sequence, which is specific for apical caspases, was used as substrate (Lien et al., 2004). Fig. 3 shows that CVPE increased apical caspase activity by 89% as compared to mock treatment by 24 h post-treatment. UV-irradiated CIV increased apical caspase activity by 88%, and enzyme activity in CIV-infected cells increased by 65% compared to mock treatments. These results are consistent with the caspase inhibitor data (Fig. 2) and further support a major role for an apical caspase during apoptosis induction in the CIV system.
Fig. 3.
Activation of apical caspase in CF-124T cells treated with CVPE, CIVUV, or CIV. CF cells were treated with CVPE (10 μg/ml), CIVUV (10 μg/ml), or CIV (10 μg/ml) and incubated at 21 °C. Cells were assayed after 24 h post-treatment for activation of apical caspase using Caspase-Glo Caspase-8 Assay Kit (Promega, WI) as per manufacturer's protocol. Activity is represented as % luminosity. Mock treatment was with RBSS only (bar and horizontal gray line).
JNK inhibitor negates apoptosis induction by CVPE
JNK plays an important role in regulation of Drosophila apoptosis through several pathways (Mizutani et al., 2003, Kanda and Miura, 2004). JNK signaling can augment or inhibit apoptosis depending on the context. Therefore, we examined the effect of JNK inhibitor in the CIV-CF cell system. Fig. 4 shows that pretreatment with JNK inhibitor SP600125 (25 nM) reduced CVPE-induced apoptosis by 87%, suggesting a major role for JNK signaling in the regulation of CIV-induced apoptosis. Treatment with JNK inhibitor alone or mock treatment with Tris-NaCl buffer induced negligible blebbing.
Fig. 4.
Effect of JNK inhibitor on CVPE-induced apoptosis. CF124T cells were pretreated with JNK inhibitor SP600125 (25 nM in Tris–NaCl, pH 7.4) for 1 h and then treated with CVPE (10 μg/ml). Cells were incubated at 21 °C for 24 h. Control cultures were mock-treated with Tris–NaCl buffer (pH 7.4) or with JNK inhibitor in Tris–NaCl buffer (pH 7.4). Bars represent percent blebbing based on counts of 200 cells per field. Error bars indicate standard deviations for four independent experiments.
Inhibition of endocytosis negates apoptosis induction by CVPE, UV-irradiated CIV and high-dose CIV
It was important to determine whether interaction of viral proteins with the cell surface in the CIV-CF cell system is sufficient for inducing apoptosis or whether there is a requirement for cellular entry. The use of ammonium chloride to inhibit acidification of endosomes in insect cells is well documented (Hacker and Hardy, 1997, IJkel et al., 2000). Fig. 5A shows that 10 mM ammonium chloride reduced apoptosis induction by CVPE (83%), CIVUV (77%), and high-dose CIV (69%). Fig. 5B shows that bafilomyin A1 (1 μM), an inhibitor of endocytosis (Mizutani et al., 2003, Long et al., 2006), also reduced apoptosis induction by CVPE (93%), CIVUV (93%), and high-dose CIV (91%) in CF cells. These results suggest that induction of apoptosis by CVPE, UV-irradiated CIV, or high-dose CIV is endocytosis-dependent.
Fig. 5.
Effect of endocytosis inhibition on induction of apoptosis by CVPE, CIVUV, or CIV. (A) Cells were pretreated with ammonium chloride (AC; 10 mM) for 30 min prior to addition of CVPE (10 μg/ml), CIVUV (10 μg/ml), or CIVXS (400 μg/ml) and incubated at 21 °C for 24 h. (B) Cells were pretreated with bafilomycin A1 (Bf; 1 μM) for 30 min prior to addition of CVPE (10μg/ml), CIVUV (10μg/ml), or CIV (400 μg/ml) and incubated at 21 °C for 24 h. (C) CIVUV (10μg/ml) was attached to polysterene beads as described in Materials and methods. Cells were challenged with this complex, incubated at 21 °C for 24 h, and stained with safranin to differentiate between blebs and beads. Bars in plates A, B, and C represent percent blebbing based on counting 200 cells per field. Error bars indicate standard deviations for four independent experiments per set.
Virus–cell surface interaction is not sufficient for induction of apoptosis
To confirm that induction of apoptosis requires viral entry rather than surface interaction of virions with cells, we attached CIVUV to polysterene beads as described in Materials and methods (Paran et al., 2001). This would allow interaction of virions with the cell surface but preclude viral entry.
UV-treated CIV (100 μg/ml) was attached to 12 mg polysterene beads (average diameter 10 μm) per manufacturer's protocol (Polysciences, Inc., PA). Beads were extensively washed after conjugation with virus, and attachment of virus to beads was confirmed using a virion-associated kinase assay. Kinase activity has been demonstrated in CIV particles (Monnier and Devauchelle, 1980), and our group recently showed kinase activity in CVPE (Paul et al., 2007). Therefore, interaction of bead-bound CIV with cells was confirmed with kinase assays on whole cells. CIVUV–bead conjugate and gamma 32P-ATP were added to cells and incubated for 1 h at 28 °C. Staurosporine and genistein were added to stop all kinase activity after incubation. The cell lysate was analyzed on SDS–PAGE and visualized by phosphorimaging. Interaction of CIVUV–bead conjugate with the cell surface was confirmed by the presence of at least one phosphorylated cellular protein. CIVUV–beads in supplemented media devoid of cells did not phosphorylate any polypeptides in the media (data not shown). Similarly, addition of gamma 32P-ATP to cells did not show extracellular kinase activity or blebbing. CIVUV and CIVUV–bead conjugate were added to CF cells, incubated at 21 °C for 24 h, and assayed for cell blebbing by differentially staining the cells but not beads with Gram's safranin. Fig. 5C shows that polysterene-bound CIVUV induced only 2% cell blebbing compared to 65% for unbound, UV-treated virus. Polysterene beads alone or conjugated with BSA induced less than 1% blebbing (Fig. 5C).
Low-pH-dependent endocytosis is required for CIV infection
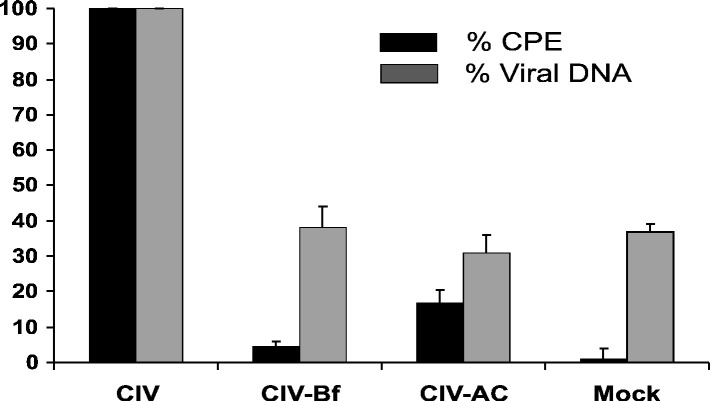
Since CVPE and CIVUV required low-pH-dependent endocytosis for induction of apoptosis, we hypothesized that CIV infection should also require endocytosis. Therefore, we tested for virus replication in the presence and absence of ammonium chloride (10 mM) and bafilomyin A1 (1 μM) by observing for viral cytopathic effect and DNA replication. Fig. 6 shows that CIV-induced cytopathic effect (consisting of hypertrophy, syncytia formation, granularity, or lysis) was drastically reduced in the presence of AC (85% reduction) and bafilomycin A1 (95% decrease). Fig. 6 also shows that viral DNA levels (determined by dot blot analysis; see Materials and methods) in CIV infections containing AC or Bf were significantly lower (more than 95% after correction for mock infections) than those in normal CIV infections and barely above those in mock-infected cells. These results indicate that low-pH-dependent endocytosis is required for CIV infection.
Fig. 6.
Requirement for low-pH-dependent endocytosis in CIV infection. Parallel cultures of cells were pretreated with ammonium chloride (AC; 10 mM) and bafilomycin A (Bf; 1 μM) for 30 min prior to CIV (10 μg/ml) infection, infected with CIV (10 μg/ml), or mock-treated with RBSS. Cytopathic effect (CPE) monitored was hypertrophy, syncytia formation, granularity, or lysis. Cells from parallel cultures were harvested at 48 hpi and blotted on a nitrocellulose membrane (Hybond). Viral DNA replication was detected as described in Materials and methods. Dots were quantified using ImageJ software (NIH). Mock treatments consisted of RBSS.
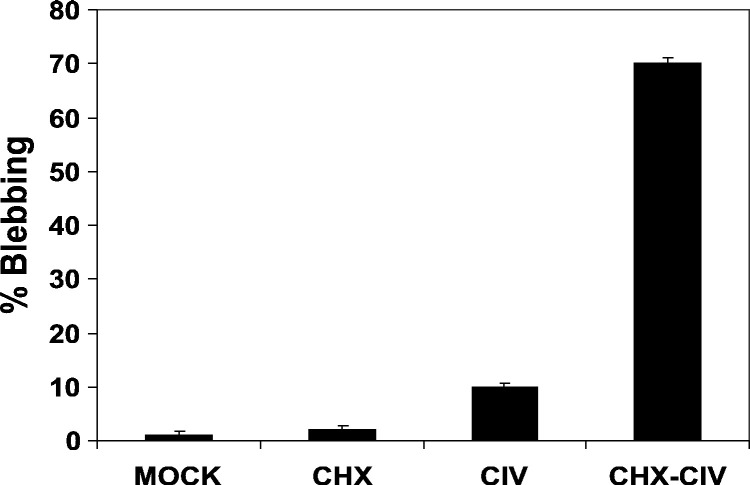
Pretreatment with cycloheximide augments apoptosis by CIV and CVPE
In order to determine whether de novo protein synthesis is required for induction of apoptosis after CIV infection, we added cycloheximide (200 μg/ml) to CF cells 1 h prior to infection. In cells with cycloheximide, CIV infection resulted in 70% apoptotic cells as determined by blebbing assay (Fig. 7 ) and 60% apoptosis as determined by TUNEL assay (Fig. 8 ). Similar levels of blebbing (75%) were observed when CF cells were exposed to CIVUV after prior treatment with CHX (data not shown). CIV infections without inhibitor resulted in less than 10% apoptosis (Fig. 7, Fig. 8B). Cycloheximide alone or mock infections resulted in less than 2% blebbing. These results indicate that de novo protein synthesis is not required for apoptosis induction upon infection of CF cells with CIV.
Fig. 7.
Effect of cycloheximide on apoptosis induction in CIV-infected CF124T cells. Cells were treated and incubated at 21 °C for 24 h: MOCK (Tris–NaCl, pH 7.4); CHX (cycloheximide, 200 μg/ml); CIV (10 μg/ml). CF cells were pretreated with CHX for 1 h and then infected with CIV (CHX-CIV). Controls included CIV infection in the absence of CHX. Percent blebbing was determined as described in the legend for Fig. 2.
Fig. 8.
Use of the TUNEL assay to determine the requirement for viral gene expression in the reduction of CIV-induced apoptosis. (A) CF124T cells treated with cycloheximide (CHX; 200 μg/ml) 1 h prior to infection with CIV (10 μg/ml); (B) CIV-infected cells without CHX treatment; (C) uninfected CHX-treated cells. Cells were incubated at 21 °C, observed 24 h after treatment, and processed for TUNEL assay as described in Materials and methods.
Viral early expression inhibits CIV-induced apoptosis
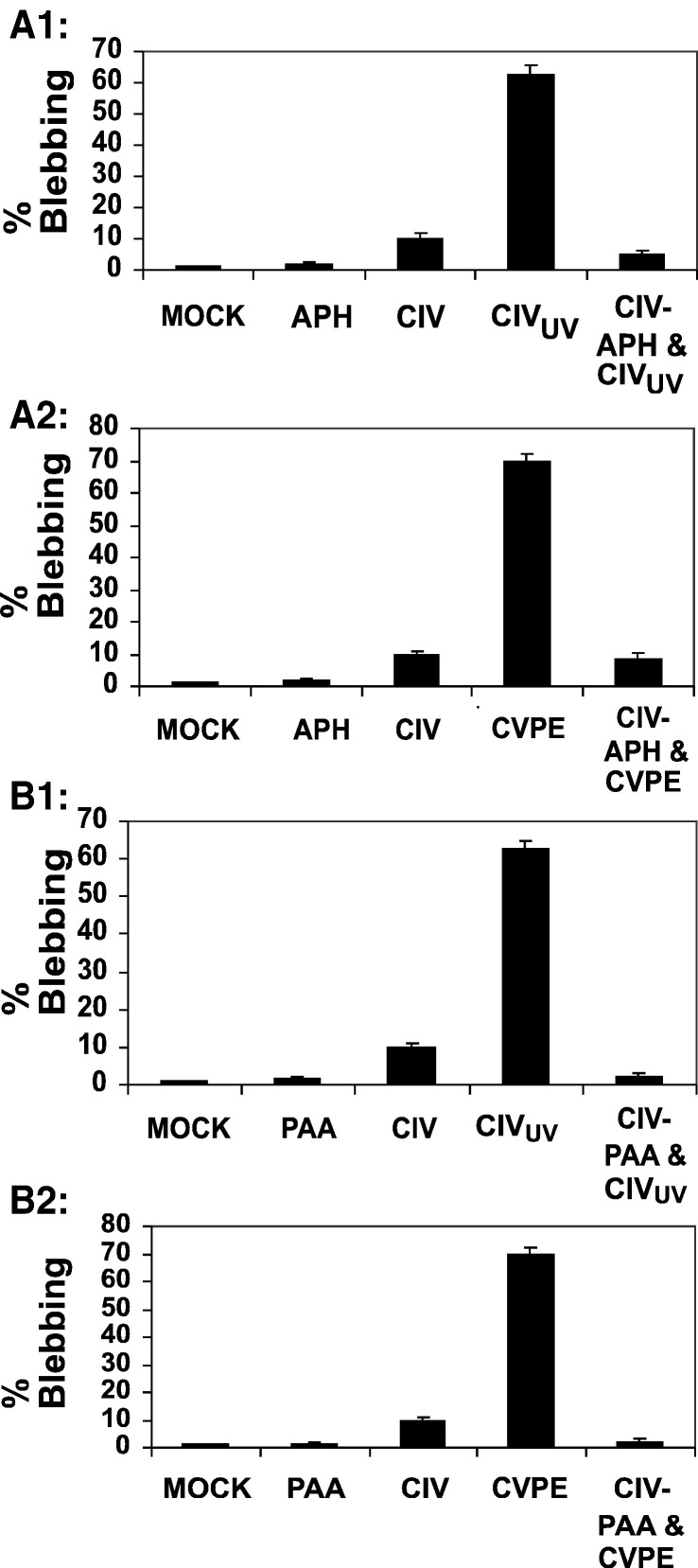
CIV infection induces a much lower level of apoptosis in CF cells than CIVXS, CVPE, or CIVUV (Fig. 1, Fig. 2, Fig. 7, Fig. 8). Moreover, inhibition of viral protein synthesis enhances CIV-induced apoptosis, suggesting that CIV gene expression is required for suppression of apoptosis. To determine if viral early gene expression blocks apoptosis induction upon CIV infection, we pretreated CF cells with aphidicolin (5 μg/ml) (D'Costa et al., 2001) or with phosphonoacetic acid (PAA; 200 μg/ml) prior to infection. Fig. 9 shows that CIV infection induced only 2% blebbing in the presence of aphidicolin and 1% blebbing with phosphonoacetic acid pretreatment compared to 10% during CIV infections of untreated cells. Thus, blebbing levels are significantly lower under conditions permitting only early gene expression compared to those allowing both early and late gene expression. Late gene expression down regulates many early genes in CIV (D'Costa et al., 2004), which probably leads to the observed 10% enhancement of blebbing. Fig. 9 also shows that inhibition of viral DNA replication in CIV infections where CIVUV or CVPE had been added as apoptosis inducers resulted in similarly drastic reductions in apoptotic blebbing. Cells challenged with CIV and CIVUV together (data not shown) manifested blebbing in 65% of the cell population, probably due to an overwhelming dose of viral proteins. Reciprocal experiments involving CVPE treatment for 1 h followed by CIV and PAA resulted in 40% blebbing and 35% cell lysis, probably due to excessive dose of viral proteins (data not shown). These results suggest that one or more viral early gene products inhibited apoptosis induction by CIV, CVPE, or CIVUV.
Fig. 9.
Effect of early viral gene expression on apoptosis induction by CIVUV and CVPE in CF124T cells. Late gene expression in CIV-infected CF124T cells was blocked by inhibiting viral DNA replication with 5 μg/ml aphidicolin (APH) or 200 μg/ml phosphonoacetic acid (PAA). Viral early gene expression was permitted for 6 h after infection with CIV (10 μg/ml), and cells were challenged with CIVUV (10 μg/ml) or with CVPE (10 μg/ml). Cells were then incubated at 21 °C and observed for apoptotic blebbing 24 h after initial treatment or infection. MOCK: untreated cells; APH/PAA: cells treated with aphidicolin or phosphonoacetic acid, respectively; CIVUV/CVPE: cells treated with CIVUV only (A1, B1) or with CVPE only (A2, B2); CIV-APH/PAA and CIVUV/CVPE: cells pretreated with APH or PAA, infected with CIV, then challenged with CIVUV (A1, B1) or CVPE (A2, B2) at 6 hpi. Results presented are means of 3 separate experiments in which 100 cells from 5 random fields were counted. Error bars represent standard deviations.
CIV early gene expression inhibits apical caspase activity
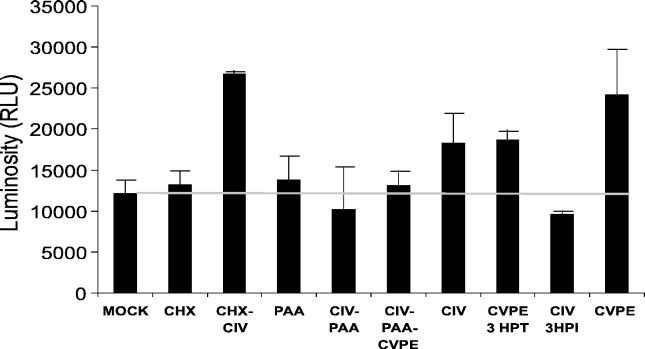
Since CIV early gene expression blocked apoptosis, it was important to determine the effect of this temporal phase on apical caspase activity (putative Cf-caspase-i) vital to apoptosis induction in this system (Fig. 2, Fig. 3). Therefore, we assayed Cf-caspase-i activity in CF cells at 24 h post-infection (or treatment) under conditions allowing early but not late viral expression. Fig. 10 shows that significant levels of apical caspase activity were detected in CIV infections with or without inhibition of early gene expression with cycloheximide (CHX-CIV; CIV) and in cells treated with CIV virion protein extract (CVPE). On the other hand, when phosphonoacetic acid restricted viral expression to early genes, CIV infection suppressed Cf-caspase-i activity to mock infection levels (Fig. 10: CIV-PAA). Challenge of CIV-PAA with CVPE did not induce Cf-caspase-i activity (Fig. 10: CIV-PAA-CVPE), indicating strong inhibition of this pathway by viral early gene expression. Cf-caspase-i activity was very low at 3 h post-CIV infection (Fig. 10: CIV-3HPI), which is within the immediate-early phase of the CIV replication cycle (D'Costa et al., 2004). On the other hand, Cf-caspase-i activity 3 h after CVPE treatment was high (Fig 10: CVPE-3HPT). Cf-caspase-i levels at initiation of infection or treatment were at mock infection levels. These data strongly suggest that one or more factors expressed during the early temporal phase of the CIV gene expression program inhibit Cf-caspase-i activity.
Fig. 10.
Effect of CIV early gene expression on activation of apical caspase. CF124T cells were assayed for caspase-8 activity after treatment with CVPE. Incubation was at 21 °C for 24 h except where noted. Reagents and inhibitors were in RBSS at the following concentrations: CIV (10 μg/ml), phosphonoacetic acid (PAA; 200 μg/ml), cycloheximide (CHX; 200 μg/ml), and CVPE (10 μg/ml). CHX: treatment with cycloheximide alone. CHX-CIV: cells pretreated with CHX and infected with CIV compared to treatment with CHX only. PAA: phosphonoacetic acid-treated cells. CIV-PAA: cells pretreated with PAA and infected with CIV. CIV-PAA-CVPE: cells pretreated with PAA, infected with CIV, and then challenged with CVPE after 6 hpi. CIV: cells infected with CIV (10 μg/ml) at 24 hpi. CVPE 3HPT: cells treated with CVPE and assayed at 3 h post treatment. CIV 3HPI: apical caspase assay carried out 3 h after infection with CIV compared to CVPE 3HPT. CVPE: cells treated with CVPE alone. Caspase-8 assay was carried out as described in Materials and methods. Values indicated are relative luminosity units. MOCK treatment was with RBSS only (bar and horizontal gray line). Values for all treatments at t = 0 were not significantly different from mock treatments. Error bars represent standard deviations for 3 independent experiments.
Discussion
We previously showed that Chilo iridescent virus capsid protein extract (CVPE) induced apoptosis and inhibition of host protein synthesis in C. fumiferana (IPRI-CF-124T) and A. grandis (BRL-AG-3A) cells (Bilimoria, 2001, Paul et al., 2007). In the present report, we demonstrate that apoptosis induction upon infection with CIV or treatment with virion components requires endocytosis, is strongly suppressed by viral early expression, does not require de novo viral protein synthesis, and is dependent on apical caspase and JNK-like factors. These are the first such findings for a member of the family Iridoviridae.
Induction of profuse apoptotic blebbing with very high doses of CIV (400μg/ml) probably results from virion components initiating rapid apoptosis, which preempts viral gene expression and consequent viral replication. These results are consistent with our observation that UV-irradiated CIV is capable of inducing apoptosis in CF cells. The ability of UV-irradiated cytoplasmic DNA viruses to induce apoptosis varies with virus and cell type (Hay and Kannourakis, 2002).
Low-pH-dependent endocytosis of CVPE and of UV-irradiated CIV was necessary for induction of apoptosis, implicating a requirement for cellular entry. This was further supported by preclusion of cellular entry when CIVUV was attached to polysterene beads and concurrent abolition of apoptotic activity (Fig. 5). Binding of CVPE and CIVUV to polysterene beads did not present steric hindrance problems as determined by the ability of virion-associated kinase activity to phosphorylate at least one cellular polypeptide. The requirement for cellular entry in induction of apoptosis has been shown for other cytoplasmic DNA viruses such as African swine fever virus (ASFV) (Carrascosa et al., 2002) and vaccinia virus (Ramsey-Ewing and Moss, 1998). Several viruses outside this group also require cellular entry as a prerequisite for apoptosis induction. These include Autographa californica nucleopolyhedrovirus (AcMNPV) (LaCount and Friesen, 1997), Sindbis virus (Jan and Griffin, 1999), human reovirus (Connolly and Dermody, 2002), and murine coronavirus (Liu et al., 2003).
The JNK pathway plays a critical role in insect immunity. JNK (coded by basket in Drosophila) (Riesgo-Escovar et al., 1996, Sluss et al., 1996) is activated upon infection of lepidopteran (Wojda et al., 2004) and Drosophila species (Kanda and Miura, 2004). In Drosophila, Eiger (invertebrate tumor necrosis factor; TNF) functions as a cell death ligand and binds to the TNF receptor-like protein, wengen. Eiger requires the JNK pathway and the mammalian caspase-9 homologue DRONC, which it activates (Igaki et al., 2002). JNK-mediated proapoptotic action does not always require gene expression (Lei and Davis, 2003).
Our data show that JNK and apical caspase inhibitors each block CIV-induced apoptosis in C. fumiferana cells, and this is consistent with the general scheme outlined for apoptosis in Drosophila. Therefore, as summarized in Fig. 11 , we postulate that Z-IETD-FMK inhibits a DRONC-like caspase (Cf-caspase-i) to suppress apoptosis (Fig. 2, Fig. 3) in CF cells. The JNK inhibitor, SP600125, blocks JNK activity and impedes DRONC-mediated apoptosis in CF cells (Fig. 4). Preliminary data (not shown) indicate that JNK inhibitor also inhibits caspase activity, suggesting that JNK is upstream of apical caspase activation. Thus, the suppression of apoptosis by inhibitors of apical caspase and JNK underscores the importance of the JNK signaling pathway in iridovirus-induced apoptosis.
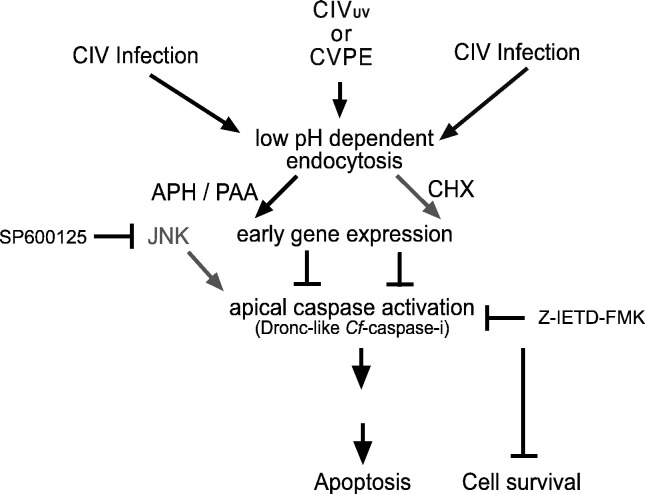
Fig. 11.
Model for apoptosis regulation in CIV-infected C. fumiferana, CF-124T cells. Virion proteins induce apoptosis via a putative C. fumiferana apical caspase (designated Cf-caspase-i) after endocytic entry. Endocytosis is required for CIV infection. Note: processes compromised by addition of inhibitor are indicated in gray. During CIV infection, early gene expression inhibits Cf-caspase-i activity and reduces apoptosis. Blocking early viral expression raises activation of Cf-caspase-i activity and consequent apoptosis. JNK and apical caspase inhibitors each hinder CIV-induced apoptosis in C. fumiferana cells (Fig. 2, Fig. 3, Fig. 4). In this model, Z-IETD-FMK inhibits a DRONC-like caspase (Cf-caspase-i) resulting in cell survival; additionally, JNK inhibitor SP600125 impedes DRONC-mediated apoptosis, thus promoting cell survival.
An “extrinsic” pathway has been postulated for lepidopteran, S. frugiperda cells (IPLB-SF-21), based on the induction of apoptosis by mammalian FADD and TNFα, which utilize caspase-8 in mammalian systems (Vucic et al., 1997, Maguire et al., 2000). A putative apical caspase designated Sf-caspase-X is inhibited by Z-IETD-FMK in S. frugiperda cells (Manji and Friesen, 2001). Our data show that Z-IETD-FMK also blocks CVPE-induced apoptosis (Fig. 3) and suggest that CVPE-induced apoptosis in C. fumiferana cells is mediated through a putative apical caspase, designated C. fumiferana initiator caspase (Cf-caspase-i). There is some evidence that caspase-8 homologue (DREDD) and FADD homologue (DFADD) detected in Drosophila may be contributing to the apoptotic process in insects (Chen et al., 1998, Hu and Yang, 2000); however, this is not firmly established. On the other hand, it is well established that DRONC plays a central role in Drosophila apoptosis as well as metamorphosis and could well be the common conduit for all caspase-dependent apoptosis in insect systems (Waldhuber et al., 2005, Hay and Guo, 2006). It should also be noted that CVPE as well as CIV induce metamorphic arrest and mortality in the cotton boll weevil (Bilimoria, 2001). Given this and the relationship between the JNK pathway and DRONC in Drosophila, we postulate that Z-IETD-FMK suppresses apoptosis in the CIV-CF cell system by inhibiting putative DRONC-like Cf-caspase-i (Fig. 11).
We have provided strong evidence suggesting inhibition of apoptosis by one or more viral early gene products. CIV virion protein extract (CVPE) and UV-irradiated CIV induced strong apoptosis as observed by blebbing and TUNEL assays, but induction of apoptosis with infectious CIV was muted. Induction by CVPE, CIVUV, or CIV in the presence of CHX suggests that CIV gene expression or DNA replication was not required for apoptosis (Fig. 1, Fig. 2, Fig. 7). Suppression of apoptosis under conditions that permit early but not late gene expression (Fig. 9) and enhancement of apoptosis under conditions that block early gene expression (Fig. 10) further support these findings. Muted apoptosis with infectious CIV (Fig. 7) may be due to expression of iap-like genes documented in the CIV genome, viz ORF 157L (L), ORF 193R (IDL), ORF 332L (IL), and ORF 284R (L) (Jakob et al., 2001, D'Costa et al., 2004). The first three of these ORFs are similar to iap genes in Cydia pomonella granulosis virus (CpGV; (Crook et al., 1993) and have the following motifs, respectively: ring finger, BIR repeat profile, and bipartite nuclear localization signal. ORF 284R is similar to an ASFV gene coding for a polypeptide (p27) with an IAP domain (Chacon et al., 1995) and is expressed exclusively at late times in CIV infections (D'Costa et al., 2004). The temporal status of ORFs 157L, 193R, and 332L are not known, but published data do not preclude an immediate-early status for ORF 193R or ORF 332L (D'Costa et al., 2004). IAP genes, dIAP 1–3, are described in Drosophila where they inhibit caspase-9 homologue, DRONC, and Drosophila tumor necrosis factor receptor-associated factor 1, dTraf1. Our data do not preclude early expression of a CIV gene product other than iap that inhibits apical caspase activity and either blocks apoptosis or recruits cellular factors for this purpose (Saito et al., 2006).
Taken together, our results show that, in response to entry of CIV or virion proteins, C. fumiferana cells undergo apoptosis through direct or indirect activation of JNK (and therefore the MAP kinase pathway) and caspases, possibly involving the putative apical caspase, Cf-caspase-i. However, upon infection with CIV, this response is rapidly and significantly inhibited by viral early gene expression. Excessive viral load overwhelms control mechanisms, resulting in profuse, immediate apoptosis. Fig. 11 depicts a working model incorporating our data. This is the first report showing that endocytosis and JNK-like activity are essential components of the apoptotic pathway in a member of the family Iridoviridae and that apoptosis in this virus group is suppressed by early viral expression. This is also the first report of JNK signal requirement during apoptosis induction by an insect virus.
Materials and methods
Virus rearing and purification
Chilo iridescent virus was raised in larvae of the greater wax moth Galleria mellonella and purified by sucrose gradient centrifugation as described previously (Henderson et al., 2001).
Cell culture
IPRI-CF-124T (CF) cells (Bilimoria and Sohi, 1977) from the spruce budworm C. fumiferana and BRL-AG-3A (AG) cells (Stiles et al., 1992) from the boll weevil A. grandis were cultured in Corning 25-cm2 flasks using Hink's TNM-FH medium supplemented with 10% fetal bovine serum (HyClone laboratories) and incubated at 28 °C. CF and AG cells were typically subcultured at 6-day intervals at a ratio of 1:10 (Henderson et al., 2001).
Virus infections
IPRI-CF-124T cells (7.5 × 105 cells/ml) in 24-well plates (Corning) were inoculated with suspensions of purified CIV at 10 μg/ml in unsupplemented medium. TCID50 assays developed in our laboratory indicated that 10 μg/ml of purified CIV represents 109 IU/ml (D'Costa, S. M., Perales, M., and Bilimoria, S. L., unpublished data). Cells were washed and inoculum was adsorbed as described previously (D'Costa et al., 2001). Infected cultures were incubated at 21 °C. Mock-infected cells were treated as above with inoculum devoid of virus.
Preparation of CIV protein extract
CHAPS [3-(3-chloamidopropyl) dimethylamino-1-propanesulfonate] (Sigma-Aldrich, St. Louis, MO) were used to prepare CIV protein extract (CVPE) from purified virions by a procedure modified from Cerutti and Devauchelle (1980) and described by Paul et al. (2007).
Blebbing assay
Blebbing assays were carried out as described by Paul et al. (2007). Actinomycin D (4 μg/ml) was used as positive control and heat-inactivated CVPE (65 °C for 30 min) was the negative control. Experiments were performed in triplicate and percentages of cells showing blebs were calculated.
UV-irradiated virus
CIV (2 ml at 1.6 mg/ml) was added to 60-mm2 plates (Corning) and exposed to UV radiation (254 nm) using a 60-watt germicidal UV lamp (Contamination Control Inc.) at a distance of 10 cm for 45 min. UV inactivation of CIV was confirmed by testing CF cells infected with normal and UV-irradiated CIV (CIVUV) for viral DNA replication by dot blot analysis (D'Costa et al., 2001) and quantification of blots by scintillation counting. CF cells were infected with CIVUV and CIV at normal (10 μg/ml) and excess (400 μg/ml; CIVXS) doses. To determine the effect of infection with CIVUV vs. CIV, 10 μg/ml of each virus preparation was added to 24-well plates seeded with CF cells, incubated at 21 °C for 18 h and observed by phase-contrast microscopy for cytopathic effect (hypertrophy, syncytia formation, granularity or lysis) or apoptotic blebbing.
Caspase inhibitor treatments
Pancaspase inhibitor Z-VAD-FMK or caspase-8 inhibitor (Z-IETD-FMK; Calbiochem, CA) was used as appropriate to inhibit caspase activities. CF cells were seeded in either 24-well or 60-well plates (Corning). Cells were incubated with pancaspase inhibitor (50 μM), caspase-8 inhibitor (50 μM), or JNK inhibitor SP600125 (25 nM) for 1 h at 28 °C. These cells were then challenged with either CVPE (10 μg/ml), CIV (400 μg/ml), or CIVUV (10 μg/ml) and incubated for an additional 24 h. The percent of cell population undergoing blebbing was monitored as described above.
Apical caspase enzyme assay
IPRI-CF-124T cells (7.5 × 105 cells/ml) in 24-well plates (Corning) were inoculated with CVPE (10 μg/ml), CIV (10 μg/ml), or CIVUV (10 μg/ml) for 24 h at 21 °C. Caspase-8 assay was carried out using the Caspase-Glo Caspase-8 Assay Kit (Promega, WI) as per manufacturer's protocol. Relative luminescence was measured with a Reporter microplate luminometer (Turner Designs, CA).
TUNEL assay
The TUNEL assay (terminal deoxynucleotidyl transferase [TdT]-mediated dUTP nick-end labeling) was carried out using the TdT-FragEL DNA fragmentation detection kit, Calbiochem (Cambridge, MA) as described by Paul et al. (2007). Five randomly selected fields were counted per slide, and percentages of TUNEL-positive cells (brown nuclei) were calculated from a sample size of at least 100 cells.
Ammonium chloride (AC) and bafilomycin A1 (Bf)-mediated inhibition of endocytosis
CF cells (6.3 × 105 cells/ml complete TNM-FH) were seeded at 400 μl per well in Corning 24-well trays. Cells were pretreated with ammonium chloride (10 mM) or bafilomycin A1 (1 μM) for 30 min prior to addition of CVPE (10 μg/ml) and UV-treated CIV (10μg/ml; see above) then incubated at 28 °C for 24 h. The concentrations of AC and Bf were maintained at 10 mM and 1 μM, respectively, through the treatment phase.
CIV replication
CF cells (6.3 × 105 cells/ml complete TNM-FH) were seeded at 400 μl per well in Corning 24-well trays. Parallel sets of cells were pretreated with ammonium chloride (10 mM) or bafilomycin A (1 μM) for 30 min prior to CIV infection or not treated. Cells were incubated at 21 °C for 48 h. Hypertrophy, syncytia formation, granularity, or lysis indicated CPE. Cell monolayers were lysed with 0.5 M NaOH. Suspensions were neutralized with 10 M ammonium acetate and blotted on nitrocellulose membrane using a dot-blot apparatus (Bio-Rad). Dot blotting was carried out as described previously (D'Costa et al., 2001).
Preparation of polysterene-linked UV-treated CIV
UV-treated CIV (100 μg/ml) was attached to 12 mg polysterene beads (average diameter 10 μ) per manufacturer's protocol (Polysciences, Inc., PA) except that sodium azide was omitted to prevent cell toxicity. Beads were extensively washed after conjugation with virus. Viral attachment to beads was confirmed using the assay for a kinase demonstrated in CIV particles (Monnier and Devauchelle, 1980) and recently detected in CVPE (Paul et al., 2007). The amount of CIVUV conjugated with beads was determined using the Bradford assay. Interaction of bead-bound CIV was confirmed with kinase assays on whole cells. CIVUV–bead conjugate (10 μg protein/ml) and gamma 32P-ATP (10 μM) were added to cells. (ATP cannot traverse the cell membrane.) Cells were incubated at 28 °C for 1 h and washed thoroughly. Staurosporine (1 μM) and genistein (50 μM) were added to stop all kinase activity after incubation. The cell lysate was run on SDS–PAGE and analyzed by phosphorimaging. Interaction of CIVUV–bead conjugate with the cell surface was confirmed by the presence of at least one phosphorylated cellular protein. CIVUV (10 μg/ml) and CIVUV–bead conjugate (10 μg protein/ml) were added to CF cells, incubated at 28 °C for 24 h, and assayed for cell blebbing by staining for cells with Gram's safranin for 2 min.
Acknowledgments
This work was supported in part by grants to S.L.B. from the Texas Advanced Research Program (003644-0148) and the Texas Advanced Technology Program (003644-0046). Support was also provided by the Office of the Vice President for Research, Office of the Provost, and the Department of Biological Sciences at Texas Tech University. NSC and ERP were partially supported by Research Awards from the Department of Biological Sciences, and SMD was partially supported by a Summer Research Award from the Graduate School, Texas Tech University. We thank Dr. John Zak for support and encouragement and Dr. Randy Allen for use of the luminometer.
References
- Bilimoria, S.L., 2001. Use of viral proteins for controlling the cotton boll weevil and other insect pests. United States Patent no. 6200561.
- Bilimoria S.L., Sohi S.S. Development of an attached strain from a continuous insect cell line. In Vitro. 1977;13:461–466. doi: 10.1007/BF02615136. [DOI] [PubMed] [Google Scholar]
- Carrascosa A.L., Bustos M.J., Nogal M.L., Gonzalez de Buitrago G., Revilla Y. Apoptosis induced in an early step of African swine fever virus entry into vero cells does not require virus replication. Virology. 2002;294:372–382. doi: 10.1006/viro.2001.1348. [DOI] [PubMed] [Google Scholar]
- Cerutti M., Devauchelle G. Inhibition of host macromolecular synthesis in cells infected with an invertebrate virus. Arch. Virol. 1980;63:297–303. doi: 10.1007/BF01315036. [DOI] [PubMed] [Google Scholar]
- Chacon M.R., Almazan F., Nogal M.L., Vinuela E., Rodriguez J.F. The African swine fever virus IAP homolog is a late structural polypeptide. Virology. 1995;214:670–674. doi: 10.1006/viro.1995.0083. [DOI] [PubMed] [Google Scholar]
- Chen P., Rodriguez A., Erskine R., Thach T., Abrams J.M. Dredd, a novel effector of the apoptosis activators reaper, grim, and hid in Drosophila. Dev. Biol. 1998;201:202–216. doi: 10.1006/dbio.1998.9000. [DOI] [PubMed] [Google Scholar]
- Chinchar V.G., Bryan L., Wang J., Long S., Chinchar G.D. Induction of apoptosis in frog virus 3-infected cells. Virology. 2003;306:303–312. doi: 10.1016/s0042-6822(02)00039-9. [DOI] [PubMed] [Google Scholar]
- Connolly J.L., Dermody T.S. Virion disassembly is required for apoptosis induced by reovirus. J. Virol. 2002;76:1632–1641. doi: 10.1128/JVI.76.4.1632-1641.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crook N.E., Clem R.J., Miller L.K. An apoptosis-inhibiting baculovirus gene with a zinc finger-like motif. J. Virol. 1993;67:2168–2174. doi: 10.1128/jvi.67.4.2168-2174.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D'Costa S.M., Yao H., Bilimoria S.L. Transcription and temporal cascade in Chilo iridescent virus infected cells. Arch. Virol. 2001;146:2165–2178. doi: 10.1007/s007050170027. [DOI] [PubMed] [Google Scholar]
- D'Costa S.M., Yao H.J., Bilimoria S.L. Transcriptional mapping in Chilo iridescent virus infections. Arch. Virol. 2004;149:723–742. doi: 10.1007/s00705-003-0232-4. [DOI] [PubMed] [Google Scholar]
- Essbauer S., Ahne W. The epizootic haematopoietic necrosis virus (Iridoviridae) induces apoptosis in vitro. J. Vet. Med., B Infect. Dis. Vet. Public Health. 2002;49:25–30. doi: 10.1046/j.1439-0450.2002.00531.x. [DOI] [PubMed] [Google Scholar]
- Hacker J.K., Hardy J.L. Adsorptive endocytosis of California encephalitis virus into mosquito and mammalian cells: a role for G1. Virology. 1997;235:40–47. doi: 10.1006/viro.1997.8675. [DOI] [PubMed] [Google Scholar]
- Hay S., Kannourakis G. A time to kill: viral manipulation of the cell death program. J. Gen. Virol. 2002;83:1547–1564. doi: 10.1099/0022-1317-83-7-1547. [DOI] [PubMed] [Google Scholar]
- Hay B.A., Guo M. Caspase-dependent cell death in Drosophila. Annu. Rev. Cell Dev. Biol. 2006;22:623–650. doi: 10.1146/annurev.cellbio.21.012804.093845. [DOI] [PubMed] [Google Scholar]
- Henderson C.W., Johnson C.L., Lodhi S.A., Bilimoria S.L. Replication of Chilo iridescent virus in the cotton boll weevil, Anthonomus grandis, and development of an infectivity assay. Arch. Virol. 2001;146:767–775. doi: 10.1007/s007050170145. [DOI] [PubMed] [Google Scholar]
- Hu S., Yang X. dFADD, a novel death domain-containing adapter protein for the Drosophila caspase DREDD. J. Biol. Chem. 2000;275:30761–30764. doi: 10.1074/jbc.C000341200. [DOI] [PubMed] [Google Scholar]
- Hu G.B., Cong R.S., Fan T.J., Mei X.G. Induction of apoptosis in a flounder gill cell line by lymphocystis disease virus infection. J. Fish Dis. 2004;27:657–662. doi: 10.1111/j.1365-2761.2004.00588.x. [DOI] [PubMed] [Google Scholar]
- Igaki T., Kanda H., Yamamoto-Goto Y., Kanuka H., Kuranaga E., Aigaki T., Miura M. Eiger, a TNF superfamily ligand that triggers the Drosophila JNK pathway. EMBO J. 2002;21:3009–3018. doi: 10.1093/emboj/cdf306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- IJkel W., Westenberg M., Goldbach R.W., Blissard G.W., Vlak J.M., Zuidema D. A novel baculovirus envelope fusion protein with a proprotein convertase cleavage site. Virology. 2000;275:30–41. doi: 10.1006/viro.2000.0483. [DOI] [PubMed] [Google Scholar]
- Imajoh M., Sugiura H., Oshima S. Morphological changes contribute to apoptotic cell death and are affected by caspase-3 and caspase-6 inhibitors during red sea bream iridovirus permissive replication. Virology. 2004;322:220–230. doi: 10.1016/j.virol.2004.02.006. [DOI] [PubMed] [Google Scholar]
- Iyer L.M., Balaji S., Koonin E.V., Aravind L. Evolutionary genomics of nucleo-cytoplasmic large DNA viruses. Virus Res. 2006;117:156–184. doi: 10.1016/j.virusres.2006.01.009. [DOI] [PubMed] [Google Scholar]
- Jakob N.J., Muller K., Bahr U., Darai G. Analysis of the first complete DNA sequence of an invertebrate iridovirus: coding strategy of the genome of Chilo iridescent virus. Virology. 2001;286:182–196. doi: 10.1006/viro.2001.0963. [DOI] [PubMed] [Google Scholar]
- Jan J.T., Griffin D.E. Induction of apoptosis by Sindbis virus occurs at cell entry and does not require virus replication. J. Virol. 1999;73:10296–10302. doi: 10.1128/jvi.73.12.10296-10302.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanda H., Miura M. Regulatory roles of JNK in programmed cell death. J. Biochem. ( Tokyo ) 2004;136:1–6. doi: 10.1093/jb/mvh098. [DOI] [PubMed] [Google Scholar]
- LaCount D.J., Friesen P.D. Role of early and late replication events in induction of apoptosis by baculoviruses. J. Virol. 1997;71:1530–1537. doi: 10.1128/jvi.71.2.1530-1537.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lei K., Davis R.J. JNK phosphorylation of Bim-related members of the Bcl2 family induces Bax-dependent apoptosis. Proc. Natl. Acad. Sci. U. S. A. 2003;100:2432–2437. doi: 10.1073/pnas.0438011100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lien S., Pastor R., Sutherlin D., Lowman H.B. A substrate-phage approach for investigating caspase specificity. Protein J. 2004;23:413–425. doi: 10.1023/b:jopc.0000039555.92058.51. [DOI] [PubMed] [Google Scholar]
- Liu Y., Cai Y., Zhang X. Induction of caspase-dependent apoptosis in cultured rat oligodendrocytes by murine coronavirus is mediated during cell entry and does not require virus replication. J. Virol. 2003;77:11952–11963. doi: 10.1128/JVI.77.22.11952-11963.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Long G., Pan X., Kormelink R., Vlak J.M. Functional entry of baculovirus into insect and mammalian cells is dependent on clathrin-mediated endocytosis. J. Virol. 2006;80:8830–8833. doi: 10.1128/JVI.00880-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maguire T., Harrison P., Hyink O., Kalmakoff J., Ward V.K. The inhibitors of apoptosis of Epiphyas postvittana nucleopolyhedrovirus. J. Gen. Virol. 2000;81:2803–2811. doi: 10.1099/0022-1317-81-11-2803. [DOI] [PubMed] [Google Scholar]
- Manji G.A., Friesen P.D. Apoptosis in motion. An apical, P35-insensitive caspase mediates programmed cell death in insect cells. J. Biol. Chem. 2001;276:16704–16710. doi: 10.1074/jbc.M010179200. [DOI] [PubMed] [Google Scholar]
- McLaughlin R.E., Scott H.A., Bell M.R. Infection of the boll weevil by Chilo iridescent virus. J. Invertebr. Pathol. 1972;19:285–290. [Google Scholar]
- Mizutani T., Kobayashi M., Eshita Y., Shirato K., Kimura T., Ako Y., Miyoshi H., Takasaki T., Kurane I., Kariwa H., Umemura T., Takashima I. Involvement of the JNK-like protein of the Aedes albopictus mosquito cell line, C6/36, in phagocytosis, endocytosis and infection of West Nile virus. Insect Mol. Biol. 2003;12:491–499. doi: 10.1046/j.1365-2583.2003.00435.x. [DOI] [PubMed] [Google Scholar]
- Monnier C., Devauchelle G. Enzyme activities associated with an invertebrate iridovirus: protein kinase activity associated with iridescent virus type 6 (Chilo iridescent virus) J. Virol. 1980;35:444–450. doi: 10.1128/jvi.35.2.444-450.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paran N., Geiger B., Shaul Y. HBV infection of cell culture: evidence for multivalent and cooperative attachment. EMBO J. 2001;20:4443–4453. doi: 10.1093/emboj/20.16.4443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paul E.R., Chitnis N.S., Henderson C.W., Kaul R.J., D'Costa S.M., Bilimoria S.L. Induction of apoptosis by iridovirus virion protein extract. Arch. Virol. 2007;152:1353–1364. doi: 10.1007/s00705-007-0945-x. [DOI] [PubMed] [Google Scholar]
- Ramsey-Ewing A., Moss B. Apoptosis induced by a postbinding step of vaccinia virus entry into Chinese hamster ovary cells. Virology. 1998;242:138–149. doi: 10.1006/viro.1997.8985. [DOI] [PubMed] [Google Scholar]
- Riesgo-Escovar J.R., Jenni M., Fritz A., Hafen E. The Drosophila Jun-N-terminal kinase is required for cell morphogenesis but not for DJun-dependent cell fate specification in the eye. Genes Dev. 1996;10:2759–2768. doi: 10.1101/gad.10.21.2759. [DOI] [PubMed] [Google Scholar]
- Saito K., Meyer K., Warner R., Basu A., Ray R.B., Ray R. Hepatitis C virus core protein inhibits tumor necrosis factor alpha-mediated apoptosis by a protective effect involving cellular FLICE inhibitory protein. J. Virol. 2006;80:4372–4379. doi: 10.1128/JVI.80.9.4372-4379.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sluss H.K., Han Z., Barrett T., Davis R.J., Ip Y.T. A JNK signal transduction pathway that mediates morphogenesis and an immune response in Drosophila. Genes Dev. 1996;10:2745–2758. doi: 10.1101/gad.10.21.2745. [DOI] [PubMed] [Google Scholar]
- Stiles B., McDonald I.C., Gerst J.W., Adams T.S. Initiation and characterization of five embryonic cell lines from the cotton boll weevil, Anthonomus grandis, in a commercial serum-free medium. In Vitro Cell. Dev. Biol. 1992;28A:355–363. [Google Scholar]
- Vucic D., Seshagiri S., Miller L.K. Characterization of reaper- and FADD-induced apoptosis in a lepidopteran cell line. Mol. Cell. Biol. 1997;17:667–676. doi: 10.1128/mcb.17.2.667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waldhuber M., Emoto K., Petritsch C. The Drosophila caspase DRONC is required for metamorphosis and cell death in response to irradiation and developmental signals. Mech. Dev. 2005;122:914–927. doi: 10.1016/j.mod.2005.04.003. [DOI] [PubMed] [Google Scholar]
- Williams T., Barbosa-Solomieu V., Chinchar V.G. A decade of advances in iridovirus research. Adv. Virus Res. 2005;65:173–248. doi: 10.1016/S0065-3527(05)65006-3. [DOI] [PubMed] [Google Scholar]
- Wojda I., Kowalski P., Jakubowicz T. JNK MAP kinase is involved in the humoral immune response of the greater wax moth larvae Galleria mellonella. Arch. Insect Biochem. Physiol. 2004;56:143–154. doi: 10.1002/arch.20001. [DOI] [PubMed] [Google Scholar]