Abstract
Objective
Familial combined hyperlipidemia (FCHL) is common among survivors of a premature myocardial infarction. FCHL patients are characterized by visceral obesity, fatty liver, and insulin resistance. The aim of the present study was to determine the incidence and determinants of type 2 diabetes (T2D) in a longitudinal cohort of FCHL pedigrees.
Research design and methods
FCHL patients, their unaffected relatives and spouses included in our baseline cohort in 1998–2005 (n=596) were re-invited to determine the incidence of self-reported T2D (that was confirmed by medical records), used as the primary outcome measure. The Fatty Liver Index (FLI) and Homeostasis Model Assessment Insulin Resistance (HOMA2-IR) were used as markers of fatty liver and insulin resistance, respectively. A subset of the original cohort underwent ultrasound of the liver, and subcutaneous and visceral fat in 2002–2005 (n=275; ‘ultrasound subcohort’).
Results
Follow-up data (median: 15 years) was acquired for 76%. The incidence rate of T2D was significantly higher in FCHL patients compared with spouses (19.2 per 1000 person-years vs 2.8 per 1000 person-years; HR : 6.3, 95% CI: 2.4 to 16.8), whereas no differences were observed between unaffected relatives and spouses (HR: 0.9, 95% CI: 0.3 to 2.6). Cox’s proportional hazard regression analyses showed that baseline HOMA2-IR and FLI≥60, but not waist circumference, BMI, or the FCHL affected state, were independently associated with incident T2D. Similar results were obtained in the ultrasound subcohort (median follow-up: 11 years), in which baseline HOMA2-IR and fatty liver (assessed by ultrasound) were independently associated with incident T2D.
Conclusion
This study further corroborates the suggestion that the liver plays a central role in the pathogenesis of cardiometabolic complications in FCHL. It supports periodical screening for T2D in this high-risk population.
Keywords: insulin resistance, liver fat, hyperlipidemia, epidemiology
Significance of this study.
What is already known about this subject?
Patients with familial combined hyperlipidemia (FCHL) are characterized by fatty liver and insulin resistance. There is conflicting evidence whether the incidence of type 2 diabetes (T2D) is higher in patients with FCHL.
What are the new findings?
This 15-year longitudinal study in a relatively large FCHL cohort shows that FCHL patients have a substantially greater risk to develop T2D than their spouses (HR: 6.3). Fatty liver and insulin resistance are baseline predictors of incident T2D.
How might these results change the focus of research or clinical practice?
These findings further emphasize the prominent role of the liver in explaining the cardiometabolic abnormalities in FCHL and support periodical screening for T2D in this high-risk population.
Introduction
Familial combined hyperlipidemia (FCHL) is a complex, genetic dyslipidemia that was first described more than four decades ago among survivors of a premature myocardial infarction.1 The increased number of apolipoprotein B containing lipoproteins (very low-density lipoprotein (VLDL), intermediate-density lipoprotein (IDL), and low-density lipoprotein (LDL)) in FCHL is the consequence of VLDL overproduction combined with an impaired clearance of VLDL remnants, IDL, and LDL particles. Several genetic variants that affect particle clearance have been identified in FCHL.2
Stable isotope studies have shown that hepatic VLDL overproduction is driven by insulin resistance and hepatic fat accumulation.3 4 We have previously shown that fatty liver, the first stage of non-alcoholic fatty liver disease, is highly prevalent among FCHL patients.5 Furthermore, 25%–35% of the variance in fatty liver can be explained by genetic factors in FCHL pedigrees.6
Since both insulin resistance and fatty liver are risk factors for the development of type 2 diabetes (T2D),7 8 it appears likely that FCHL patients are also prone to develop T2D. However, the presence of T2D in the index patients is an exclusion criterion for the diagnosis of FCHL,9 which could lead to an enrichment of T2D-protective genes in FCHL pedigrees. One example of such a gene is glucokinase regulatory protein (GCKR). The GCKR variant that increases plasma lipids and protects from T2D10 is more common in FCHL.11 It is therefore difficult to predict whether FCHL patients are predisposed to or protected from T2D.
We have previously attempted to address this issue in a cohort of FCHL patients and their spouses and suggested that the incidence of T2D is higher in FCHL patients.12 A strong inference on the higher incidence of T2D and its baseline predictors in FCHL was, however, limited by the small population size (n=110 in total) and a relatively short follow-up period (5 years).12 Longer follow-up data of a larger cohort are warranted particularly because another FCHL cohort did not find a higher 10-year incidence of T2D in comparison to healthy controls.13
More recently, we presented the first data of a cohort of FCHL patients, unaffected relatives, and spouses (n=596) that was followed over a 15-year period.14 The aim of the present study, therefore, was to assess the incidence of T2D and baseline determinants thereof in this larger cohort.
Methods
Study participants
The details of this cohort have recently been described elsewhere.14 In short, between 1998 and 2005, FCHL patients, their unaffected relatives, and spouses (n=596), derived from 48 families, visited the research ward after 2 weeks withdrawal of any lipid-modifying medication, 3 days abstinence from alcohol, and fasting since the evening prior to their visit. The affected state, that is being an FCHL patient, was based on plasma lipid apolipoprotein B>1.2 g/L and triglycerides>1.5 mmol/L.9 Family members who did not fulfil these criteria were classified as unaffected relative. Of note, all subgroups (ie, FCHL patients, their unaffected relatives, and spouses) were present in up to three consecutive generations.
Between 2002 and 2005, a subset of the initial, overall cohort (n=275) was re-invited for more detailed phenotyping, including blood withdrawal and ultrasound of the subcutaneous and visceral fat compartments, the liver, and the carotid arteries,5 15 further referred to as the ‘ultrasound subcohort’. Participants received similar instructions as for the baseline visit before attending the research ward, with the only exception that they were now asked to abstain from alcohol 3 weeks prior to the visit.5
Between 2014 and 2016, all participants of the initial, overall cohort were re-invited to undergo a basic set of measurements, including anthropometrics, blood pressure and blood withdrawal. Participants were invited in the same order as in the baseline visit to limit differences in follow-up duration between participants as much as possible. All participants were asked to fill in a health questionnaire. Those who were not able or willing to visit the research ward were asked to complete the questionnaire only.
Measurements
Measurement of height, weight, waist circumference, blood pressure, whole blood glucose, plasma insulin, lipids, and alanine aminotransferase (ALT) levels at baseline were similar in the overall cohort and the ultrasound subcohort and have been described in detail elsewhere.16 17 Plasma gamma glutamyl transferase (GGT) levels were measured at baseline in the overall cohort by an enzymatic reaction using a Cobas 8000 (Roche). The Friedewald formula was used to calculate LDL cholesterol.18 The homeostasis model assessment insulin resistance (HOMA2-IR) was calculated using the calculator developed by Levy et al.19 The fatty liver index (FLI) was calculated from body mass index (BMI), waist circumference, and serum GGT and triglycerides levels. A FLI≥60 is indicative of fatty liver.20
At follow-up, glucose levels were measured in (NaFl) plasma on a Cobas 8000 (Roche).
Ultrasound measurements
In the ultrasound subcohort, all individuals additionally underwent ultrasound of the abdomen to quantify subcutaneous (SAT-US) and visceral adipose tissue (VAT-US) compartments, and to assess the presence of fatty liver (ATL 9 HDI US system, Bothell, Washington, DC, USA).5 SAT-US and VAT-US were determined at the end-expiratory stage of normal respiration at the level at which waist circumference was determined. SAT-US was defined as the depth from skin to abdominal muscles (ie, linea alba) and VAT-US as the distance from the peritoneum to the anterior of the vertebrate body.5 21 VAT-US strongly correlates with VAT determined by computed tomography.20 Standardized images of the liver and right kidney were recorded and examined by an independent radiologist. Fatty liver was diagnosed in the presence of increased echogenicity, posterior beam attenuation, and/or decreased visualization of hepatic blood vessels.5
Assessment of incident T2D
Since not all participants visited the research ward at the follow-up visit, the diagnosis of T2D was determined by questionnaires in all participants. Clinical T2D cases and year of diagnosis were subsequently confirmed by review of the medical records and used as the primary outcome measure of this study. The incidence of T2D was subsequently expressed as the incidence rate (per 1000 person-years).
Newly diagnosed T2D was defined as fasting plasma glucose levels≥7.0 mmol/L in those who did visit the research ward at follow-up.
Statistical analysis
Data are presented as mean±SD, or as median with IQR in case of non-normal distribution. Age-adjusted and sex-adjusted differences in baseline characteristics between FCHL patients and unaffected relatives (as dummy variables) versus spouses were analysed with linear and logistic regression for continuous and dichotomous variables, respectively. Skewed variables (ie, serum triglycerides and ALT, HOMA2-IR, and alcohol intake) were log transformed prior to analyses.
Cox’s proportional hazard models were used to calculate the HR and 95% CI for incident T2D in FCHL patients and their unaffected relatives, entered as dummy variables. The spouses were used as a reference group. Follow-up time was defined as time from baseline visit (ie, either the 1998–2005 visit for the overall cohort or the 2002–2005 visit for the ultrasound subcohort) until clinical T2D diagnosis or time from baseline until the present follow-up for censored cases. In the overall cohort, age, sex, alcohol intake at baseline, baseline waist circumference (or alternatively BMI), baseline HOMA2-IR, and baseline FLI were additionally entered in the model. A sensitivity analysis was conducted in which clinically diagnosed T2D and newly diagnosed T2D were combined as an outcome measure.
In the ultrasound subcohort, a similar model was constructed in which waist circumference was replaced by SAT-US and VAT-US, and FLI by fatty liver assessed by ultrasound. Since all individuals in this cohort had been instructed to abstain from alcohol 3 weeks prior to the measurements, alcohol intake was not added as a potential confounder to this model.
All analyses were performed using SPSS V.25.0.
Results
Study population
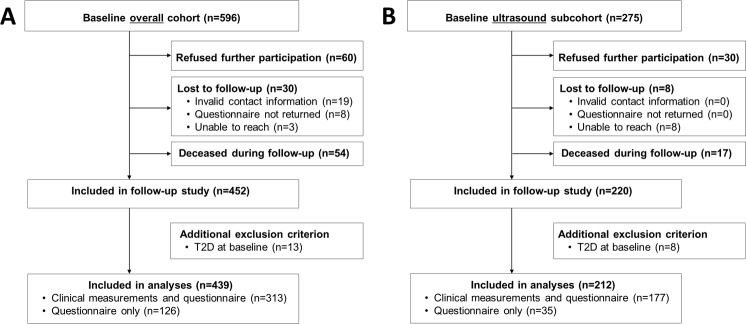
Of the 596 individuals in the overall cohort, follow-up was acquired for 452 (76%) (figure 1A). The median follow-up duration was 15 years (IQR: 14–16 years). Individuals who refused participation (n=60), were lost to follow-up (n=30), or died during follow-up (n=54) were significantly older and more (abdominally) obese, had higher systolic blood pressure and glucose levels, and more frequently used antihypertensive drugs at baseline when compared with those who were included in the follow-up evaluation (table 1). Importantly, the proportion of spouses, unaffected relatives, and FCHL patients did not differ between both groups (table 1).
Figure 1.
Flow chart of overall FCHL cohort that was invited for 15-year follow-up study (panel A) and a subcohort that underwent baseline ultrasound measurements (panel B). See the Methods section for more details. FCHL, familial combined hyperlipidemia; T2D, type 2 diabetes.
Table 1.
Comparison of baseline characteristics between participants included in this follow-up study and those who refused, were lost to follow-up, or deceased
| Follow-up | Refused/lost to follow-up/deceased | |
| Male/female, n | 203/249 | 72/72 |
| Spouse/unaffected relative/FCHL, n (%) | 113/244/95 (25/54/21) | 40/77/27 (28/53/19) |
| Age, years | 43±13 | 51±17* |
| Smoking, n (%) | 136 (30) | 42 (29) |
| BMI, kg/m² | 25.7±4.0 | 26.5±4.6* |
| Waist circumference, cm | 91.0±12.2 | 94.7±13.8* |
| Total cholesterol, mmol/L | 5.6±1.3 | 5.8±1.4 |
| HDL cholesterol, mmol/L | 1.0±0.2 | 1.0±0.3 |
| LDL cholesterol, mmol/L | 3.9±1.2 | 4.1±1.3 |
| Triglycerides, mmol/L | 1.3 (0.9–1.9) | 1.4 (1.0–2.0) |
| Apolipoprotein B, g/L | 1.1±0.3 | 1.1±0.3 |
| Lipid lowering medication, n (%) | 71 (16) | 30 (22) |
| Systolic BP, mm Hg | 132±19 | 140±25* |
| Diastolic BP, mm Hg | 84±11 | 86±13 |
| Anti-hypertensive medication, n (%) | 53 (12) | 39 (27)* |
| Glucose, mmol/L | 4.9 (4.6–5.3) | 5.1 (4.7–5.5)* |
Data are expressed as mean ± SD or as medians with IQR between parentheses.
*P<0.05, analysed with a Student’s T-test, Mann-Whitney U test, and χ2 test in case of normally distributed, non-normally distributed, and dichotomous variables, respectively.
BMI, body mass index; BP, blood pressure; FCHL, familial combined hyperlipidemia; HDL, high-density lipoprotein; LDL, low-density lipoprotein.
Thirteen participants were subsequently excluded because of the presence of T2D at baseline. Of the remaining 439 participants, 313 visited the research ward (which included completion of the questionnaire) and 126 completed the questionnaire only (figure 1A).
Of the 275 participants who underwent baseline ultrasound measurements between 2002 and 2005 (ultrasound subcohort), follow-up was acquired for 220 (80%). The median follow-up duration was 11 years (IQR: 11–12 years). Eight participants were excluded because of presence of T2D at baseline (figure 1B).
Baseline characteristics of the overall cohort and ultrasound subcohort
Baseline characteristics of FCHL patients (n=84), unaffected relatives (n=245) and their spouses (n=110) are presented in table 2. FCHL patients were characterized by a higher BMI, waist circumference, systolic and diastolic blood pressure, (off-treatment) plasma lipids (except for high-density lipoprotein (HDL) cholesterol, which was lower), whole blood glucose levels, and HOMA2-IR when compared with spouses. Furthermore, plasma ALT levels and the prevalence of fatty liver (ie, FLI ≥60) were significantly higher in FCHL patients than in spouses, at a background of a similar alcohol intake (table 2). Unaffected relatives were significantly younger than spouses. After age-adjustment and sex-adjustment, only the frequency of use of lipid-modifying drugs was higher in unaffected relatives (table 2).
Table 2.
Baseline characteristics of overall and ultrasound cohort
| Overall cohort (n=439) (15-year follow-up) |
Ultrasound subcohort (n=212) (11-year follow-up) |
|||||
| Spouses | Unaffected relatives | FCHL patients | Spouses | Unaffected relatives | FCHL patients | |
| Male/female, n | 51/59 | 98/147 | 44/40 | 29/30 | 62/49 | 26/16 |
| Age, years | 46±13 | 39±13* | 48±13 | 48±11 | 41±14* | 53±13 |
| BMI, kg/m2 | 25.2±3.6 | 25.0±3.9 | 27.8±3.9† | 25.4±4.1 | 25.7±4.3 | 27.8±3.8† |
| Waist, cm | 90±11 | 88±12 | 98±11† | 91.1±13.0 | 90.5±11.7 | 98.9±8.7† |
| SAT-US, cm | – | – | – | 2.5±1.1 | 2.5±1.2 | 3.1±1.0† |
| VAT-US, cm | – | – | – | 7.6±2.4 | 7.4±2.4† | 9.0±2.1† |
| Systolic BP, mm Hg | 131±19 | 129±18 | 138±20† | 127±18 | 131±17† | 140±16† |
| Diastolic BP, mm Hg | 83±11 | 83±11 | 88±10† | 83±11 | 84±10 | 89±9† |
| Total cholesterol, mmol/L | 5.3±1.0 | 5.2±1.1 | 6.9±1.1† | 5.4±0.9 | 5.4±1.7 | 6.9±1.1† |
| HDL cholesterol, mmol/L | 1.1±0.2 | 1.0±0.2 | 0.9±0.2† | 1.1±0.3 | 1.0±0.3† | 0.9±0.2† |
| LDL cholesterol, mmol/L | 3.7±1.0 | 3.6±1.1 | 4.8±1.1† | 3.7±0.9 | 3.6±1.2 | 5.0±1.1† |
| Triglycerides, mmol/L | 1.0 (0.8–1.5) | 1.1 (0.9–1.4) | 2.3 (1.8–2.8)† | 1.2 (0.8–1.7) | 1.1 (0.9–1.6)† | 2.0 (1.8–2.9)† |
| Apolipoprotein B, g/L | 1.0±0.2 | 1.0±0.2 | 1.5±0.3† | 1.0±0.3 | 1.0±0.3 | 1.4±0.2† |
| Lipid-modifying drug, n (%) | 3 (3) | 21 (9)† | 38 (45)† | 2 (3) | 17 (15)† | 19 (46)† |
| Glucose, mmol/L | 4.9±0.5 | 4.8±0.5 | 5.3±0.7† | 4.9±0.5 | 4.9±0.5 | 5.3±0.6† |
| HOMA2-IR | 0.5 (0.3–0.9) | 0.6 (0.5–0.9) | 1.0 (0.7–1.6)† | 0.7 (0.3–1.2) | 0.8 (0.3–1.2) | 1.3 (0.9–1.9)† |
| Alcohol intake, U/week | 4.0 (0.5–10.0) | 3.8 (0.5–10.0) | 5.0 (0.5–10.0) | 6.0 (0–12.0) | 8.0 (1.0–14.0) | 4.0 (0.4–14.0) |
| ALT, U/L | 17 (12–23) | 17 (12–23) | 25 (18–35)† | 17 (13–23) | 17 (13–24) | 22 (19–29)† |
| Fatty liver‡, n (%) | 24 (22) | 50 (21) | 51 (61)† | 10 (17) | 32 (29)† | 22 (52)† |
Data are presented as absolute numbers, mean±SD, or median (IQR).
*P<0.05 versus spouses, analysed with a Student’s T-test.
†P<0.05 versus spouses, analysed with linear regression with adjustment for age and sex.
‡Determined by the fatty liver index ≥60 (overall cohort) or ultrasound (ultrasound subcohort).
ALT, alanine aminotransferase; BMI, body mass index; BP, blood pressure; FCHL, familial combined hyperlipidemia; HDL, high-density lipoprotein; HOMA2-IR, homeostasis model assessment insulin resistance; LDL, low-density lipoprotein; SAT-US, subcutaneous adipose tissue determined by ultrasound; VAT-US, visceral adipose tissue determined by ultrasound.
In the ultrasound subcohort, FCHL patients (n=42) displayed similar differences as in the overall cohort when compared with spouses (n=59) (table 2). In addition, SAT-US and VAT-US were both higher in FCHL patients. The prevalence of fatty liver (assessed by ultrasound) was also higher in FCHL patients in this subcohort. VAT-US, systolic blood pressure, and plasma triglycerides were significantly higher in unaffected relatives (n=111) compared with spouses after adjustment for age and sex. The prevalence of fatty liver (assessed by ultrasound) was also significantly higher (table 2).
Of interest, the proportion of individuals with fatty liver was comparable between the overall cohort and the ultrasound subcohort, despite the use of two different methods (FLI≥60 vs ultrasound): 22% versus 17% in spouses, 21% versus 29% in unaffected relatives, and 61% versus 52% in FCHL patients in the overall cohort and ultrasound subcohort, respectively (table 2).
Incidence of clinical T2D
After 15 years of follow-up, T2D was reported (and confirmed by medical records) in 21 of 84 FCHL patients (25.0%; incidence rate: 19.2 per 1000 person-years), 10 of 245 unaffected relatives (4.1%; incidence rate: 2.8 per 1000 person-years), and 5 of 110 spouses (4.5%; incidence rate: 3.1 per 1000 person-years) (online supplementary table 1). The risk to develop T2D was statistically significantly higher in FCHL patients when compared with their spouses, who were used as reference group (HR: 6.3, 95% CI: 2.4 to 16.8), but not in unaffected relatives versus spouses (HR: 0.9, 95% CI: 0.3 to 2.6) (figure 2). Three individuals (two FCHL patients and one unaffected relative) who visited the research ward at follow-up were newly diagnosed with T2D, defined as fasting plasma glucose levels≥7.0 mmol/L at the follow-up visit (online supplementary table 1).
Figure 2.
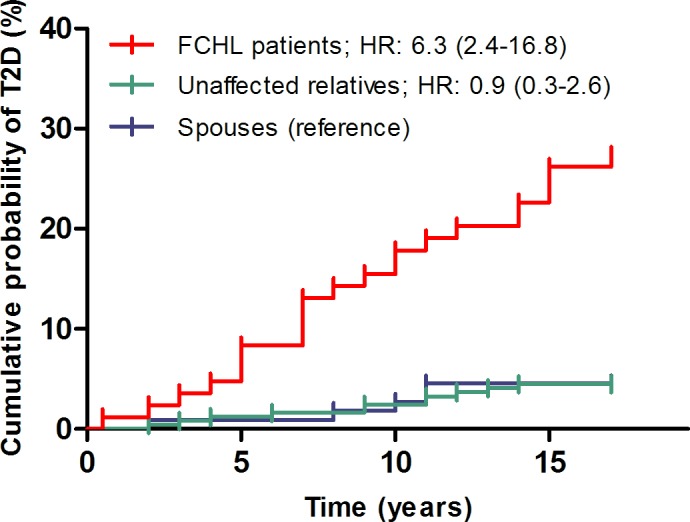
Cumulative incidence of type 2 diabetes (T2D) in patients with familial combined hyperlipidemia (FCHL), their unaffected relatives and spouses. Data are presented as HR with 95% CI, calculated with Cox's proportional hazard regression models.
bmjdrc-2019-001107supp001.pdf (57.1KB, pdf)
Adjustment for potential confounders (age, sex, and alcohol intake) did not affect the outcomes (table 3, models 2–3). To examine which factors explained the high incidence of clinical T2D in FCHL, waist circumference, HOMA2-IR and FLI≥60 (all assessed at baseline) were subsequently entered in the regression model (table 3, models 2, 4–6). In the final model, the difference in the incidence of T2D between FCHL patients and spouses was diminished by 48% and no longer statistically significant (HR: 3.0, 95% CI: 0.8-11.2; table 3, model 6). In this model, a higher HOMA2-IR at baseline and the presence of fatty liver (ie FLI ≥ 60) at baseline were independently associated with incident T2D (exponential beta: 1.7, 95% CI: 1.3-2.2 for HOMA2-IR and HR: 6.5, 95%CI: 1.7-25.4 for FLI ≥ 60). Replacement of waist circumference by BMI did not affect the outcomes (data not shown). Addition of use of lipid-modifying drugs at baseline to the model further attenuated the difference between FCHL patients and spouses (HR: 1.7, 95%CI: 0.4-7.2), without affecting the associations of HOMA2-IR and FLI ≥ 60 with incident T2D (data not shown). Baseline use of lipid-modifying drugs was significantly associated with incident T2D (HR: 3.6, 95%CI: 1.4-8.5). Of note, repeat analysis of the crude model in only those individuals who did not receive lipid-modifying drugs at baseline showed that these FCHL patients are also at increased risk to develop T2D (HR: 5.1; 95% CI: 1.5-17.2).
Table 3.
Cox’s proportional hazard regression models of incident type 2 diabetes in overall cohort and ultrasound subcohort
| Model, adjusted for | Unaffected relatives versus spouses | FCHL patients versus spouses | ||||
| HR | 95% CI | P value | HR | 95% CI | P value | |
| Overall cohort | ||||||
| Crude | 0.9 | 0.3 to 2.6 | 0.89 | 6.3 | 2.4 to 16.8 | <0.001 |
| Age+sex | 1.1 | 0.4 to 3.2 | 0.89 | 6.1 | 2.3 to 16.2 | <0.001 |
| Model 2+alcohol | 1.4 | 0.4 to 4.7 | 0.56 | 6.3 | 2.1 to 19.2 | 0.001 |
| Model 3+waist | 1.4 | 0.4 to 4.8 | 0.56 | 4.7 | 1.5 to 14.6 | 0.007 |
| Model 4+HOMA2-IR | 2.2 | 0.6 to 8.5 | 0.24 | 3.9 | 1.0 to 14.7 | 0.04 |
| Model 5+FLI ≥ 60 | 2.0 | 0.5 to 7.5 | 0.32 | 3.0 | 0.8 to 11.2 | 0.11 |
| Ultrasound subcohort | ||||||
| Crude | 3.7 | 0.4 to 30.9 | 0.23 | 8.2 | 1.0 to 69.4 | 0.05 |
| Age+sex | 3.3 | 0.4 to 27.8 | 0.27 | 7.3 | 0.9 to 63.1 | 0.07 |
| Model 2+VAT US+SAT-US | 2.6 | 0.3 to 22.6 | 0.38 | 4.8 | 0.5 to 42.7 | 0.16 |
| Model 3+HOMA2-IR | 2.7 | 0.3 to 24.1 | 0.37 | 4.6 | 0.2 to 39.8 | 0.17 |
| Model 4+fatty liver* | 2.1 | 0.2 to 19.3 | 0.51 | 4.0 | 0.5 to 35.9 | 0.21 |
*Determined by ultrasound.
FLI, fatty liver index; HOMA2-IR, homeostasis model assessment insulin resistance; SAT-US, subcutaneous adipose tissue determined by ultrasound; VAT-US, visceral adipose tissue determined by ultrasound.
Finally, in a sensitivity analysis in which clinically diagnosed and newly diagnosed T2D cases were combined as an outcome measure, similar outcomes were observed (online supplementary table 2).
In the ultrasound subcohort, the 11-year incidence was also higher in FCHL patients than in spouses (HR: 8.2, 95% CI: 1.0 to 69.4), but not in unaffected relatives compared with spouses (HR: 3.7, 95% CI: 0.4 to 30.9). Adjustment for potential confounders (age, sex) and mediators (SAT-US, VAT-US, HOMA-IR, and fatty liver determined by ultrasound) attenuated the association of the FCHL affected state with incident T2D (HR: 4.0, 95% CI: 0.5 to 35.9; table 3, models 2–5). Again, a higher HOMA2-IR at baseline and the presence of fatty liver (assessed by ultrasound) at baseline explained the high incidence in FCHL patients (exponential beta: 3.0, 95% CI: 1.1 to 7.8 for HOMA2-IR and HR: 6.9, 95% CI: 1.1 to 42.2 for fatty liver determined by ultrasound).
Combination of clinically diagnosed and newly diagnosed T2D yielded very similar outcomes (online supplementary table 2).
Discussion
To date, contrasting results have been reported on whether FCHL patients are prone to develop T2D in comparison to controls.12 13 The present study shows that one out of four FCHL patients developed T2D after 15 years of follow-up. The six times higher risk to develop T2D compared with their spouses was explained by insulin resistance and fatty liver at baseline. Similar results were obtained in the ultrasound subcohort, in which the presence of fatty liver and the subcutaneous and visceral fat compartments were assessed by ultrasound.
Our results are in line with a recent meta-analysis that showed that fatty liver is a risk factor for future T2D.7 In contrast to previous studies,22 23 we did not observe an association between abdominal obesity and incident T2D. Visceral obesity is a strong determinant of hepatic fat accumulation,24 which also explains why it is incorporated (as waist circumference) in the FLI algorithm.20 We do, however, not believe that inclusion of waist circumference in the FLI formula explains the absent association between abdominal obesity and future T2D in this cohort, since similar outcomes were obtained when fatty liver was assessed by ultrasound and waist circumference was replaced by SAT-US and VAT-US in the ultrasound cohort.
In the present study, use of lipid-modifying drugs at baseline was associated with incident T2D. Previous studies have convincingly shown that statins increase T2D risk, although with a high number needed to harm (≈ 200).25 It is therefore unlikely that statin use is a major determinant of the onset T2D in this relatively small cohort of FCHL patients. Indeed, repeat analysis including only those FCHL patients who did not receive lipid-modifying treatment showed a similar increased risk of incident T2D (although some of these individuals have received treatment in the follow-up period14). Altogether it is more likely that the contribution of baseline use of lipid-modifying drugs to incident T2D is at least in part explained by collinearity with the FCHL affected state, since use of lipid-modifying drugs was particularly confined to FCHL patients (table 2).
This study corroborates the suggestion that the liver plays a central role in the pathogenesis of cardiometabolic complications in FCHL.26 Stable isotope studies have shown that HOMA-IR is associated with hepatic insulin resistance (r=0.59),27 which in turn is associated with intrahepatic triglycerides (r=0.60).28 We and others have shown that HOMA2-IR and fatty liver are determinants of the lipid phenotype expression (in particular the hypertriglyceridemic phenotype) in FCHL.5 16 29 The current study shows that HOMA2-IR and fatty liver are also determinants of incident T2D in FCHL. Our findings also support to perform periodical screening for incident T2D in this high-risk population, in agreement with current guidelines.30
This study has several strengths and limitations. Its main strength is that this is one of the largest FCHL cohorts with long-term follow-up on clinically relevant endpoints. Moreover, for a substantial subcohort we have not only a surrogate marker, but also ultrasound data on the presence of fatty liver. One main limitation is that the group of spouses, used as a reference in this study, and the absolute numbers of incident T2D cases were relatively small resulting in limited statistical power and wide CIs. Second, we used a FLI≥60 as a surrogate of fatty liver in the overall cohort, which has a high positive likelihood ratio to rule in fatty liver.20 As hitherto mentioned, the inclusion of BMI, waist circumference, and plasma triglycerides in the FLI algorithm complicates the assessment of other baseline factors associated with incident T2D in this cohort, in particular abdominal obesity and the FCHL affected state (which includes elevated plasma triglycerides). These limitations have been overcome by the use of ultrasound as an alternative measure of fatty liver in a subset of the overall cohort, which yielded strikingly similar outcomes. Ultrasound has good test characteristics to diagnose moderate and severe hepatic fat accumulation.31 Third, since we realized that not all participants would visit our research facility during the follow-up visit, we decided to use the self-reported, clinical diagnosis of T2D as a primary outcome measure in all participants, which may be subjective to several sources of bias. Although we confirmed all T2D diagnoses by checking the medical records, there still could be recall bias. Furthermore, it is conceivable that FCHL patients, but not unaffected relatives and spouses, have received periodical checks of serum glucose levels as a part of their cardiovascular risk surveillance, which would increase the likelihood of a T2D diagnosis in this specific group (detection bias). We do, however, not believe that both sources of bias have had a major influence on the outcomes of the current study, since we detected only three newly-diagnosed T2D (of which two in FCHL patients) in those individuals who visited the research ward at follow-up. Detection bias should have resulted in a ‘catch-up’ of newly-diagnosed T2D in those who did not receive periodical screening, that is, non-affect relatives and spouses. This was clearly not the case.
In conclusion, the present longitudinal study shows that FCHL patients have a high risk to develop future T2D, which is explained by insulin resistance and fatty liver at baseline. This study further corroborates the suggestion that the liver plays a central role in the pathogenesis of cardiometabolic complications in FCHL and supports periodical screening for T2D in this high-risk population.
Footnotes
Contributors: MCGJB and MMJvG were responsible for the study concept and design and participated in the interpretation of the data. NS, StD, SvH, BH, and JL were involved in recruitment of participants and clinical measurements. SM and DdB were reponsible for the laboratory measurements at the Central Diagnostic Laboratory. All authors critically revised the manuscript and approved the final version to be published.
Funding: This work was supported by the Biobanking and Biomolecular Research Infrastructure the Netherlands (BBMRI-NL CP2013- 80).
Competing interests: None declared.
Patient consent for publication: Not required.
Ethics approval: The study protocol was approved by the Medical Ethics Committee from Maastricht University. All individuals gave written informed consent.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement: Data are available upon reasonable request.
References
- 1.Goldstein JL, Schrott HG, Hazzard WR, et al. . Hyperlipidemia in coronary heart disease. II. genetic analysis of lipid levels in 176 families and delineation of a new inherited disorder, combined hyperlipidemia. J Clin Invest 1973;52:1544–68. 10.1172/JCI107332 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Brouwers MCGJ, van Greevenbroek MMJ, Stehouwer CDA, et al. . The genetics of familial combined hyperlipidaemia. Nat Rev Endocrinol 2012;8:352–62. 10.1038/nrendo.2012.15 [DOI] [PubMed] [Google Scholar]
- 3.Malmström R, Packard CJ, Caslake M, et al. . Defective regulation of triglyceride metabolism by insulin in the liver in NIDDM. Diabetologia 1997;40:454–62. 10.1007/s001250050700 [DOI] [PubMed] [Google Scholar]
- 4.Adiels M, Taskinen M-R, Packard C, et al. . Overproduction of large VLDL particles is driven by increased liver fat content in man. Diabetologia 2006;49:755–65. 10.1007/s00125-005-0125-z [DOI] [PubMed] [Google Scholar]
- 5.Brouwers MCGJ, Bilderbeek-Beckers MAL, Georgieva AM, et al. . Fatty liver is an integral feature of familial combined hyperlipidaemia: relationship with fat distribution and plasma lipids. Clin Sci 2007;112:123–30. 10.1042/CS20060190 [DOI] [PubMed] [Google Scholar]
- 6.Brouwers MCGJ, Cantor RM, Kono N, et al. . Heritability and genetic loci of fatty liver in familial combined hyperlipidemia. J Lipid Res 2006;47:2799–807. 10.1194/jlr.M600312-JLR200 [DOI] [PubMed] [Google Scholar]
- 7.Mantovani A, Byrne CD, Bonora E, et al. . Nonalcoholic fatty liver disease and risk of incident type 2 diabetes: a meta-analysis. Diabetes Care 2018;41:372–82. 10.2337/dc17-1902 [DOI] [PubMed] [Google Scholar]
- 8.Lillioja S, Mott DM, Spraul M, et al. . Insulin resistance and insulin secretory dysfunction as precursors of non-insulin-dependent diabetes mellitus. prospective studies of Pima Indians. N Engl J Med 1993;329:1988–92. 10.1056/NEJM199312303292703 [DOI] [PubMed] [Google Scholar]
- 9.Sniderman AD, Castro Cabezas M, Ribalta J, et al. . A proposal to redefine familial combined hyperlipidaemia -- third workshop on FCHL held in Barcelona from 3 to 5 may 2001, during the scientific sessions of the European Society for clinical investigation. Eur J Clin Invest 2002;32:71–3. 10.1046/j.1365-2362.2002.00941.x [DOI] [PubMed] [Google Scholar]
- 10.Vaxillaire M, Cavalcanti-Proença C, Dechaume A, et al. . The common P446L polymorphism in GCKR inversely modulates fasting glucose and triglyceride levels and reduces type 2 diabetes risk in the DESIR prospective General French population. Diabetes 2008;57:2253–7. 10.2337/db07-1807 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Weissglas-Volkov D, Aguilar-Salinas CA, Sinsheimer JS, et al. . Investigation of variants identified in Caucasian genome-wide association studies for plasma high-density lipoprotein cholesterol and triglycerides levels in Mexican dyslipidemic study samples. Circ Cardiovasc Genet 2010;3:31–8. 10.1161/CIRCGENETICS.109.908004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Brouwers MCGJ, van der Kallen CJH, Schaper NC, et al. . Five-Year incidence of type 2 diabetes mellitus in patients with familial combined hyperlipidaemia. Neth J Med 2010;68:163–7. [PubMed] [Google Scholar]
- 13.Skoumas I, Ioakeimidis N, Vlachopoulos C, et al. . Statin therapy and risk of diabetes mellitus in aging patients with heterozygous familial hypercholesterolemia or familial combined hyperlipidemia: a 10-year follow-up. Angiology 2018;69:242–8. 10.1177/0003319717718331 [DOI] [PubMed] [Google Scholar]
- 14.Luijten J, van Greevenbroek MMJ, Schaper NC, et al. . Incidence of cardiovascular disease in familial combined hyperlipidemia: a 15-year follow-up study. Atherosclerosis 2019;280:1–6. 10.1016/j.atherosclerosis.2018.11.013 [DOI] [PubMed] [Google Scholar]
- 15.Brouwers MCGJ, Reesink KD, van Greevenbroek MMJ, et al. . Increased arterial stiffness in familial combined hyperlipidemia. J Hypertens 2009;27:1009–16. 10.1097/HJH.0b013e32832991fa [DOI] [PubMed] [Google Scholar]
- 16.Brouwers MCGJ, van Greevenbroek MMJ, Vermeulen VMM-J, et al. . Five-Year follow-up of waist circumference, insulin and ALT levels in familial combined hyperlipidaemia. Clin Sci 2007;113:375–81. 10.1042/CS20070101 [DOI] [PubMed] [Google Scholar]
- 17.Voors-Pette C, de Bruin TW. Excess coronary heart disease in familial combined hyperlipidemia, in relation to genetic factors and central obesity. Atherosclerosis 2001;157:481–9. 10.1016/S0021-9150(00)00752-8 [DOI] [PubMed] [Google Scholar]
- 18.Tremblay AJ, Morrissette H, Gagné J-M, et al. . Validation of the Friedewald formula for the determination of low-density lipoprotein cholesterol compared with beta-quantification in a large population. Clin Biochem 2004;37:785–90. 10.1016/j.clinbiochem.2004.03.008 [DOI] [PubMed] [Google Scholar]
- 19.Levy JC, Matthews DR, Hermans MP. Correct homeostasis model assessment (HOMA) evaluation uses the computer program. Diabetes Care 1998;21:2191–2. 10.2337/diacare.21.12.2191 [DOI] [PubMed] [Google Scholar]
- 20.Bedogni G, Bellentani S, Miglioli L, et al. . The fatty liver index: a simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol 2006;6:33 10.1186/1471-230X-6-33 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Stolk RP, Wink O, Zelissen PM, et al. . Validity and reproducibility of ultrasonography for the measurement of intra-abdominal adipose tissue. Int J Obes Relat Metab Disord 2001;25:1346–51. 10.1038/sj.ijo.0801734 [DOI] [PubMed] [Google Scholar]
- 22.Ohlson LO, Larsson B, Svärdsudd K, et al. . The influence of body fat distribution on the incidence of diabetes mellitus. 13.5 years of follow-up of the participants in the study of men born in 1913. Diabetes 1985;34:1055–8. 10.2337/diab.34.10.1055 [DOI] [PubMed] [Google Scholar]
- 23.Meisinger C, Döring A, Thorand B, et al. . Body fat distribution and risk of type 2 diabetes in the general population: are there differences between men and women? the MONICA/KORA Augsburg cohort study. Am J Clin Nutr 2006;84:483–9. 10.1093/ajcn/84.3.483 [DOI] [PubMed] [Google Scholar]
- 24.Nielsen S, Guo Z, Johnson CM, et al. . Splanchnic lipolysis in human obesity. J Clin Invest 2004;113:1582–8. 10.1172/JCI21047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ridker PM, Pradhan A, MacFadyen JG, et al. . Cardiovascular benefits and diabetes risks of statin therapy in primary prevention: an analysis from the JUPITER trial. Lancet 2012;380:565–71. 10.1016/S0140-6736(12)61190-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Brouwers MCGJ, de Graaf J, van Greevenbroek MMJ, et al. . Novel drugs in familial combined hyperlipidemia: lessons from type 2 diabetes mellitus. Curr Opin Lipidol 2010;21:530–8. 10.1097/MOL.0b013e32833ea9ec [DOI] [PubMed] [Google Scholar]
- 27.Abdul-Ghani MA, Matsuda M, Balas B, et al. . Muscle and liver insulin resistance indexes derived from the oral glucose tolerance test. Diabetes Care 2007;30:89–94. 10.2337/dc06-1519 [DOI] [PubMed] [Google Scholar]
- 28.Korenblat KM, Fabbrini E, Mohammed BS, et al. . Liver, muscle, and adipose tissue insulin action is directly related to intrahepatic triglyceride content in obese subjects. Gastroenterology 2008;134:1369–75. 10.1053/j.gastro.2008.01.075 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Veerkamp MJ, de Graaf J, Stalenhoef AFH. Role of insulin resistance in familial combined hyperlipidemia. Arterioscler Thromb Vasc Biol 2005;25:1026–31. 10.1161/01.ATV.0000160612.18065.29 [DOI] [PubMed] [Google Scholar]
- 30.Piepoli MF, Hoes AW, Agewall S, et al. . 2016 European Guidelines on cardiovascular disease prevention in clinical practice: The Sixth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of 10 societies and by invited experts)Developed with the special contribution of the European Association for Cardiovascular Prevention & Rehabilitation (EACPR). Eur Heart J 2016;37:2315–81. 10.1093/eurheartj/ehw106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Saadeh S, Younossi ZM, Remer EM, et al. . The utility of radiological imaging in nonalcoholic fatty liver disease. Gastroenterology 2002;123:745–50. 10.1053/gast.2002.35354 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjdrc-2019-001107supp001.pdf (57.1KB, pdf)