Abstract
Traditional Chinese Medicine (TCM) is widely used in clinical research due to its low toxicity, low number of side effects, and low cost. Many components of common fruits and vegetables play well-documented roles as chemopreventive or chemotherapeutic agents that suppress tumorigenesis. Anthraquinones are commonly extracted from the Polygonaceae family of plants, e.g., Rheum palmatum and Rheum officinale. Some of the major chemical components of anthraquinone and its derivatives, such as aloe-emodin, danthron, emodin, chrysophanol, physcion, and rhein, have demonstrated potential anticancer properties. This review evaluates the pharmacological effects of emodin, a major component of Aloe vera. In particular, emodin demonstrates anti-neoplastic, anti-inflammatory, anti-angiogenesis, and toxicological potential for use in pharmacology, both in vitro and in vivo. Emodin demonstrates cytotoxic effects (e.g., cell death) through the arrest of the cell cycle and the induction of apoptosis in cancer cells. The overall molecular mechanisms of emodin include cell cycle arrest, apoptosis, and the promotion of the expression of hypoxia-inducible factor 1α, glutathione S-transferase P, N-acetyltransferase, and glutathione phase I and II detoxification enzymes while inhibiting angiogenesis, invasion, migration, chemical-induced carcinogen-DNA adduct formation, HER2/neu, CKII kinase, and p34cdc2 kinase in human cancer cells. Hopefully, this summary will provide information regarding the actions of emodin in cancer cells and broaden the application potential of chemotherapy to additional cancer patients in the future.
Keywords: angiogenesis, apoptosis, cell cycle arrest, emodin, traditional Chinese medicine (TCM)
1. Introduction
Numerous researchers have reported the use of phytochemical compounds such as anthraquinone emodin extracts from traditional Chinese medicines (TCM), including Polygonum multiflorum [1], [2], P Cuspidatum [3], [4], Rumex patientia [5], Rhamnus catharticus, Rhamnus orbiculatus [6], Aloe vera [7], Acorus tatarinowii [8], Cassia obtusifolia [9], Cassia occidentalis [10], Rheum palmatum [11], Rheum officinale [12], Eriocaulon buergerianum [13], Dendrobium thyrsiflorum [14], Fibraurea tinctoria [15],Coptis chinensis [16],Scutellaria baicalensis [16], Isatis indigotica [17], and Rumex chalepensis [18]. Studies on the use of TCM have noted lipid regulation activities and anti-inflammatory, antimicrobial, antiviral, antitumor, and antioxidant effects. To learn more about the therapeutic functions of TCM, experiments are needed to identify the functional ingredients and ascertain the molecular mechanisms of these compounds. Recent research is paying more attention to TCM because it may have future applications in clinical medicine. In particular, rhubarb (Rheum palmatum) is one of the oldest and most famous Chinese herbal medicines and is still used in various herbal remedies and therapeutic applications. Based on current reports and investigation, we believe rhubarb has clinical potential.
Rhubarb is a well-known treatment for many diseases in TCM [19], [20]. Anthraquinones extracted from the rhubarb rhizome exhibit antidiabetic properties, suggesting a metabolic role in the insulin-stimulated glucose transport pathway [21]. Both in vitro and in vivo studies have reported the antimicrobial activities of extracts from Sapindus mukorossi and Rheum emodin against Helicobacter pylori [22]. Moreover, the antioxidant and anticancer potential of Rheum emodin rhizome extracts have demonstrated therapeutic value [23]. Extracts from Rheum palmatum have a high level of inhibitory activity against anti-Severe acute respiratory syndrome (SARS) coronavirus 3C-like protease effects [24]. A polysaccharide extracted from Rheum tanguticum has been shown to affect 2,4,6-trinitrophene sulphonic acid (TNBS)-induced colitis and CD4+ T cells in rats [25]. Rhubarb has also demonstrated protective effects against experimental severe acute pancreatitis [26]. A study on anti-Oketsu activity indicates that rhubarb II has inhibitory effects against allergies [27]. Hexane extracts from Rheum undulatum not only decreases cell viability, thereby triggering apoptotic cell death in oral cancer, but also decreases the expression of specificity protein (Sp1) and its downstream protein, survivin [28].
The effects of rhubarb extracts on experimental chronic renal failure (CRF) indicate that it can reduce proteinuria and the severity glomerulosclerosis within remnant kidneys in rats [29]. Treatment of menopausal symptoms using an extract from the roots of Rhapontic rhubarb (plus the results of in vitro and in vivo experiments) indicate estrogenic actions, especially estrogen receptor β (ERβ)-mediated effects [30]. Oligostilbenes from rhubarb also inhibit low-density lipoprotein and high-density lipoprotein oxidation humans [31], suggesting a pivotal role in the prevention of lipoprotein oxidation.
2. Active ingredients found in the Polygonaceae family
Emodin (1,3,8-trihydroxy-6-methylanthraquinone) (Fig. 1 ) is an active ingredient in the root and rhizome of Rheum palmatum (Polygonaceae) [11]. This herb has been used in TCM for the treatment of gallstones, inflammation, hepatitis, and osteomyelitis and is also a known vasorelaxant and diuretic [32]. It reportedly has antibacterial, anti-inflammatory, antiviral, anti-ulcerogenic, anticancer, immunosuppressive [33], [34], [35], [36], and chemopreventive effects [37]. Emodin has also been reported to exert inhibitory effects on cell death in the human lung squamous carcinoma CH27 cell line [36], andhuman promyeloleukemic HL-60 cells induce apoptosis by activating the caspase-3 cascade independently of reactive oxygen species (ROS) production [38]. Emodin-induced apoptosis in human cervical cancer Bu 25TK cells occurs through poly (ADP-ribose) polymerase cleavage and the activation of caspase-9, but caspase-8 is not activated [39]. Moreover, emodin triggers apoptosis in human hepatoma HepG2/C3A, PLC/PRF/5, and SK-HEP-1 cells through a p53-dependent pathway [40]. In addition, emodin enhances arsenic trioxide-induced apoptosis by generating ROS and inhibiting survival signaling [41], and gene expression alteration occurs in HeLa cells through the redox-dependent enhancement of arsenic cytotoxicity [42]. Our laboratory has proven that Aloe-emodin affects the expression of cytokines and the functions of leukocytes in Sprague Dawley rats [134]. Emodin affects murine myelomonocytic leukemia WEHI-3 cells in vitro and enhances phagocytosis in leukemic mice in vivo [135].
Fig. 1.
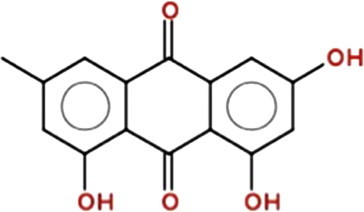
Chemical structure of emodin.
Emodin downregulates androgen receptors and inhibits the cellular growth of prostate cancer [43]. Emodin inhibits the adhesion of human breast cancer (MDA-MB-231), human cervix epithelioid carcinoma (HeLa), and human hepatocarcinoma (HepG2) tumor cells by suppressing lipid raft coalescence and interfering with integrin clustering and focal adhesion complex (FAC) formation [44]. Likewise, it has been demonstrated that emodin could act as a Janus-activated kinase 2 inhibitor and have cytotoxic activities against multiple myeloma in humans [45]. Emodin selectively inhibits the interleukin–6-induced JAK2/STAT3 pathway and induces apoptosis in myeloma cells via the downregulation of myeloid cell leukemia 1 (Mcl-1) cells [45]. In local ischemic myocardium, emodin mediates protection from acute myocardial infarction through the inhibition of inflammation and apoptosis [46].
3. Pharmacological mechanisms against various types of cancer cells
Emodin has shown significant anticancer activities in several tumor cells, both in vivo and in vitro, while its molecular anticancer mechanisms have not been well explored. This review discusses emodin’s pharmacological activities and the mechanisms that induce cell death in many types of human cancer cells, both in vitro and in vivo. Research findings on emodin-induced cytotoxicity and its protective effects are described below.
3.1. HER2/neu expression
Previously published reports in the literature confirm that emodin and its derivatives inhibit p185neu tyrosine kinase via the suppression of HER2/neu-transformed phenotypes (e.g., by inducing cellular transformations and metastasis-associated potential) [47]. In breast cancer, the emodin derivative, azide methyl anthraquinone, induces mitochondrion-dependent apoptosis in HER2/neu-overexpressing MDA-MB-453 cells and lung adenocarcinoma Calu-3 cells and blocks HER2/neu binding to Hsp90. Azide methyl anthraquinone also induces the proteasomal degradation of HER2/neu in MDA-MB-453 and Calu-3 cells in vitro [48].
3.2. CKII and p34cdc2 kinase
Emodin inhibits the activity of casein kinase II (CKII) by acting as a competitor at ATP-binding sites. [49]. CKII is involved in the proliferation of human U87 astroglioma cells via stimulation of basal phospholipase D (PLD) activity. [50]. Emodin reportedly induces apoptosis in human tongue squamous cancer SCC-4 cells through ROS and mitochondria-dependent pathways in vitro [51]. Aloe-emodin, which is extracted from the rhizome of Rheum palmatum, downregulates MMP-2 through a p38 Mitogen-activated protein kinase (MAPK)-Nuclear factor-κB (NF-kB)–dependent pathway, thereby leading to the inhibition of invasion by nasopharyngeal carcinoma cells (NPC-TW 039 and NPC-TW 076) [52].
3.3. Oncogenes
It is well documented that nuclear factor-kappaB (NF-κB) plays an important role in the transcription of tumor cells [53], [54]. It has been reported that emodin inhibits the proliferation and induction of apoptosis in pancreatic cancer cell lines (SW1990/GZ and SW1990). Emodin not only downregulates NF-κB under unstimulated conditions, but it also inhibits gemcitabine-induced NF-κB protein expression [53]. Aloe-emodin also purportedly induces antiproliferative activities through p53- and p21-dependent apoptotic pathway in the human hepatoma HepG2 and Hep3B cell lines [55]. An attractive target of oncogene-based anticancer drugs derived from natural herbal plants (like emodin), Polygonum cuspidatum exhibits strongly selective activities against src-HER2/neu and ras-oncogenes. In other words, emodin might be a oncogenetic signal for the inhibition of transduction [56].
3.4. Hypoxia-inducible factor 1α
Heterodimer hypoxia-inducible factor 1α (HIF-1) consists of a β subunit that is constitutively expressed and an oxygen-regulated α subunit. HIF-1 regulates genes that participate in angiogenesis, iron metabolism, glucose metabolism, and cell proliferation/survival [57]. The activity of HIF-1, especially its α subunit, is controlled by the posttranslational modification of the amino acid residues in its subunits [57]. HIF-1 plays a key role in the cellular response to tumor hypoxia that poses a major problem to successful radiotherapy and chemotherapy. The targeting of HIF-1 is now considered to be a pivotal and efficient strategy for treating neurodegenerative maladies like Alzheimer’s (AD), Parkinson’s (PD), Huntington’s Disease (HD), amyotrophic lateral sclerosis (ALS), etc. [58]. It has also been reported that emodin diminishes hypoxia-induced embryotoxicity by upregulating HIF-1 and intracellular superoxide dismutases in whole cultured mouse embryos [59]. As a novel inhibitor of HIF-1, emodin is an adjunct that boosts the efficacy of cytotoxic drugs used for the treatment of prostate cancer DU-145 cells, demonstrating overactivated HIF-1 and potent multidrug resistance (MDR) [60].
3.5. N-acetyltransferase activity
Our previous studies have demonstrated how emodin and aloe-emodin inhibit N-acetyltransferase (NAT) activity and gene expression in mouse leukemia L1210 cells [61], human melanoma cells (A375.S2) [62], and strains of H pylori in peptic ulcer patients [63], [64].
3.6. Cell cycle arrest
The cell cycle is classified into the G0/G1, S, and G2/M phases; if an agent induces apoptosis, then those will be sug-G1 phase [65]. In clinic settings, some anticancer agents can induce cell cycle arrest (arrest during the G0/G1, S, and/or G2/M phase) [65], [66]. It has been reported that emodin and docosahexaenoic acid (DHA) increase arsenic trioxide interferon–α-induced cell death in human T-cell leukemia virus type 1 (HTLV-I)–transformed cells via ROS generation and the inhibition of Akt and activator protein 1 (AP-1) [67]. Emodin inhibits the growth of hepatocellular carcinomas, such as Huh7, Hep3B, and HepG2, through anticancer pathways (e.g., G2/M arrest and increased expression levels of the involved genes, both at the mRNA and protein levels) [68]. Emodin also reportedly inhibits vascular endothelial growth factor-A–induced angiogenesis [69]. Other investigators have demonstrated how emodin induces apoptosis through the p53-dependent pathway in human hepatocellular carcinoma cells [40], as well as growth arrest and death through ROS and p53 in human vascular smooth muscle cells [70].
Aloe-emodin also induces G2/M arrest in human promyelocytic leukemia HL-60 cells [71], cervical cancer HeLa cells [72], and through activated alkaline phosphatase in human oral cancer KB cells in vitro [73]. It has also been reported that aloe-emodin induces apoptosis through protein 53 (p53)-dependent apoptotic pathways in human bladder cancer T24 cells [74]. Aloe-emodin induces destabilization of caspase-8 and -10-associated RING protein (CARP) mRNA, indicating that caspase-8–mediated p53-independent apoptosis in human carcinoma cells [75] and human nasopharyngeal carcinoma cells induces caspase-3, -8, and -9–mediated activation of the mitochondrial death pathway [76]. Still, the antiproliferative activity of aloe-emodin occurs via p53- and p21-dependent apoptotic pathways in human hepatoma HepG2 cell lines [55], [77]. Other evidence indicates that aloe-emodin and emodin inhibit schisandrin B in gastric cancer cells in vitro [78].
3.7. Apoptosis
It is well documented that the best strategy for killing cancer cells is via the induction of apoptosis [79] and that the best way for chemotherapeutic agents to kill cancer cells is to trigger apoptosis in tumors [79], [80]. In human hepatoma Huh-7 cells, apoptosis is mediated by the downregulation of calpain-2 and ubiquitin-protein ligase E3A [81]. Emodin has strong antioxidative and anticancer actions and abrogates cisplatin-induced nephrotoxicity in rats [82]. Other reports have cited the antitumor and apoptosis-promoting properties of emodin, an anthraquinone derivative, against pancreatic cancer in mice by inhibiting Akt activation [12]. Emodin enhances apoptosis in cisplatin-induced gallbladder carcinomas in a ROS-dependent manner and suppresses survivin expression [83]. Emodin downregulates X-linked inhibitor of apoptosis protein (XIAP) expression [84] and inhibits NF-κB against human pancreatic cancer [53], thereby enhancing the antitumor efficacy. Emodin induces apoptosis in the mouse microglial BV-2 cell line via Tribbles homolog 3 (TRB3) and eliminates inflammatory microglia, thereby exerting neuroprotective effects [85].
Emodin induces ROS generation and the activation of the ATM-p53-Bax–dependent signaling pathway in human lung adenocarcinoma A549 cells [86]. It has been reported that emodin exerts potential anticancer effects in pancreatic cancer cells by downregulating the expression of survivin and β-catenin [87]. Emodin also demonstrates potential as an anti-atherosclerosis agent by inhibiting the proliferation of Tumor necrosis factor (TNF)-alpha–induced human aortic smooth muscle cells (HASMC) through mitochondrial- and caspase-dependent apoptotic pathways [88]. Emodin induces apoptosis via the caspase-3–dependent pathway in human renal proximal tubule HK-2 cells [89] and inhibits human prostate cancer LNCaP cell proliferation via androgen receptor and p53-p21 pathways [90]; pyrazole emodin derivatives inhibit the growth of and induce apoptosis in human hepatocellular carcinoma HepG2 cells [91]. Pyrazole emodin derivative also induce apoptosis in human cervical cancer cells via the activation of caspase-3 and -9 and the cleavage of poly (ADP-ribose) polymerase [39]. Aloe-emodin induces apoptosis in human lung nonsmall carcinoma H460 cells through Cyclic Adenosine monophosphate (cAMP)-dependent protein kinase, protein kinase C, Bcl-2, caspase-3, and the p38 signaling pathway and induces human lung squamous cell carcinoma CH27 cell death via the Bax and Fas death pathways [92], [93]. Emodin not only successfully suppresses acute graft rejection in vivo, thereby prolonging the survival of the recipient rats by inhibiting hepatocellular apoptosis and modulating Th1/Th2 balance [94], but also mediates protection against acute myocardial infarction [46] in local ischemic myocardium. Emodin can reverse gemcitabine resistance in pancreatic cancer cells via mitochondria-dependent pathways in vitro [95].
3.8. Glutathione S-transferase and glutathione peroxidase
The function of glutathione S-transferase has implications in cell growth and oxidative stress as well as disease progression and prevention, which are present in subcellular compartments (e.g., cytosol, mitochondria, endoplasmic reticulum, nucleus, plasma membrane) [96]. Glutathione peroxidase (GPx), a selenoenzyme, plays a key role in the protection of organisms from oxidative damage by catalyzing the reduction of harmful hydroperoxides using thiol cofactors [97]. The function of GPx is to regulate hydroperoxide levels, but it might have dual roles [98], [99]. The role of glutathione and glutathione-dependent enzymes in antioxidative processes is the maintenance and regulation of cell status, glutathionylation, and deglutathionylation, redox-dependent signaling, and apoptosis [100].
Emodin also demonstrates hepatoprotective effects against CCl4-induced liver injury [101]. Emodin induces apoptosis in Dalton’s lymphoma cells in association with the modulation of hydrogen peroxide-metabolizing antioxidant enzymes [102]. Emodin affects the mitochondrial capacity of ATP generation and antioxidant components as well as susceptibility against ischemia-reperfusion injury in rat hearts, although there is a sex difference [103]. Emodin also reportedly demonstrates antioxidant actions in vivo [104] and myocardial protective effects [105].
3.9. Carcinogenesis
Novel functions of emodin have been reported, namely that emodin enhances the repair of UV- and cisplatin-induced DNA damage and might even promote nucleotide excision repair (NER) capabilities in human fibroblast cells (WI38) [106] and human tongue cancer SCC-4 cells following DNA damage and the inhibition of DNA repair genes [107]. Emodin also demonstrates a proven ability to inhibit mutagenicity and the formation of 1-nitropyrene–induced DNA adducts in Escherichia coli PQ37 [108].
3.10. Gene expression
Several studies have reported that emodin affects the gene expression of human breast carcinoma BCap-37 cells [109] and downregulates the expression of transient receptor potential vanilloid 1 (TRPV1) ion channel protein mRNA and its functions in Dorsal root ganglion (DRG) neurons in vitro, thereby inhibiting inflammatory stimuli-induced hyperalgesia [110]. Emodin-mediated cytotoxicity in human lung adenocarcinoma H1650 (CRL-5883), human bronchioloalveolar carcinoma A549, lung squamous cell carcinoma H520, and H1703 cells is suppressed by Excision repair cross-complementary 1 (ERCC1) and Rapid Application Development (Rad)51 expression via extracellular regulated protein kinase 1/2 (ERK1/2) inactivation [111]. It has also been reported that emodin induces DNA damage and inhibits the expression of DNA repair genes in human tongue cancer SCC-4 cells [107]. Studies also show that emodin induces toxicological effects to the murine testicular gene expression profile [112] and inhibits the cytotoxic actions of tumor necrosis factor [113]. On the other hand, it has also been reported that emodin inhibits the migration and invasion in human tongue cancer SCC-4 cells due to the inhibition of the gene expression of matrix metalloproteinase (MMP)-9 [114].
3.11. Glutathione S-transferase P expression
Glutathione S-transferase P (GSTP) has been reported to regulate the S-glutathionylation of specific clusters of main proteins; it also plays a negative modulating role in some kinase pathways through ligand or protein interactions. GSTP is ubiquitously expressed in human tissue [115] and is linked to two cell-signaling functions critical to survival. It can sequester and negatively regulate c-jun N-terminal kinase (JNK) [116]. Catalytic reversal of S-glutathionylation is well characterized, but the role of GSTP in catalyzing the forward reaction contributes to the glutathionylation cycle [116].
Emodin reportedly induces neuroprotective effects in rat cortical neurons against β-amyloid–induced neurotoxicity [117]. Emodin induces apoptosis via an ROS-dependent mitochondrial signaling pathway in human lung adenocarcinoma A549 cells [118]. Emodin inhibits invasiveness, suppresses MMP-9 expression through the suppression of AP-1 and NF-κB in human cancer HSC5 cells (skin squamous cell carcinoma) and MDA-MB-231 cells (human breast cancer cell line) [119]. Likewise, emodin effectively suppresses hyaluronic acid (HA)-induced matrix metalloproteinase (MMP) secretion and the invasion of glioma through the inhibition of focal adhesion kinase (FAK), extracellular regulated protein kinase (ERK)1/2, and Akt/protein kinase B (PKB) activation and the partial inhibition of the transcriptional activities of activator protein-1 (AP-1) and nuclear factor-κB (NF-κB) [33].
3.12. Angiogenesis
Therapeutic antiangiogenesis is widely viewed as a useful approach for the treatment of cancer, cardiovascular diseases, bone fractures, rheumatoid arthrities, and other diseases [120]. In tumor formation, angiogenesis plays a vital role in development, reproduction, and wound repair. Many studies describe natural and synthetic compounds with antiangiogenic activities, attracting notice to their potential applications in cancer prevention and treatment [121]. Emodin reportedly inhibits tumor-associated angiogenesis through the inhibition of ERK phosphorylation [122] and inhibits vascular endothelial growth factor-A–induced angiogenesis by blocking receptor-2 (KDR/Flk-1) phosphorylation [69]. Vascular endothelial growth factor (VEGF) has been studied for its role as a stimulant in angiogenesis and vascular permeability. Several studies show that emodin and its anthraquinone derivatives inhibit the angiogenesis and proliferation [123] of primary cultured bovine aortic endothelial cells in the absence or presence of basic fibroblast growth factor (bFGF) or the presence of VEGF in a dose-dependent manner [124], [125]. Likewise, emodin inhibits VEGF receptors in human colon cancer cells [126], upregulates urokinase plasminogen activator (uPA) and plasminogen activator inhibitor-1, and promotes wound healing in human fibroblasts [127]. Emodin has been used in cancer therapies for the treatment of autoimmune diseases with anti-VEGF or anti-VEGFR (receptor) effects [69], [126]. It has also been reported that emodin induces antiproliferative and antimetastatic effects in human pancreatic cancer SW1990 cells [128]. In human neuroblastoma SH-SY5Y cells, emodin inhibits the level of MMP, thus inhibiting migration and invasion in vitro [129].
3.13. Drug resistance
The overexpression of multidrug resistance (MDR) in tumor cells poses a serious obstacle to successful chemotherapy [130]. Treating cancer with chemotherapeutic agents and radiation leads to complications, such as the development of tumor resistance to therapy (radio- or chemoresistance). Emodin might sensitize tumor cells to radiation therapy and chemotherapeutic agents by inhibiting the pathways that lead to treatment resistance. Emodin has also been found to protect against therapy-associated toxicities [131]. Emodin induces the mechanisms that involve the ROS-mediated suppression of MDR and HIF-11 [60]. Our studies demonstrate emodin’s cytotoxic and protective effects in rat C6 glioma cells: the survival effects involve Mdr1a, MRP2, MRP3, MRP6, and NF-κB [132]. Emodin may be involved in reducing the glutathione level and downregulating MDR-related protein 1 (MRP1) expression in gallbladder SGC996 cancer cells. In tumor-bearing mice, it has also been indicated that co-treatment with emodin/cisplatin suppresses tumor growth in vivo by increasing cancer cell apoptosis and downregulating MRP1 expression [61], [133].
4. Conclusion
Despite the fact that TCM research has been greatly accelerated with the advent of new technologies, we still need to work hard to gain stronger evidence that confirms the clinical applications of herbal medicines. Based on our observations and the results of previously reported studies, emodin can act as an anticancer agent against many human cancer cell lines through its effects across multiple signaling pathways. Over these past several years, our laboratory has evaluated agents that affect cell cycle arrest, apoptosis, metastasis, and angiogenesis in human cancer cell lines, both in vitro and in vivo, in addition to tumor cell growth, invasion, migration, and metastasis that are also involved in angiogenesis. Based on these observations regarding the effects of emodin, these findings may offer information that could be used in the design of novel therapeutic agents that inhibit tumor cells. Accordingly, we also summarize the pharmacology of emodin as a possible anticancer agent (Fig. 2 ).
Fig. 2.
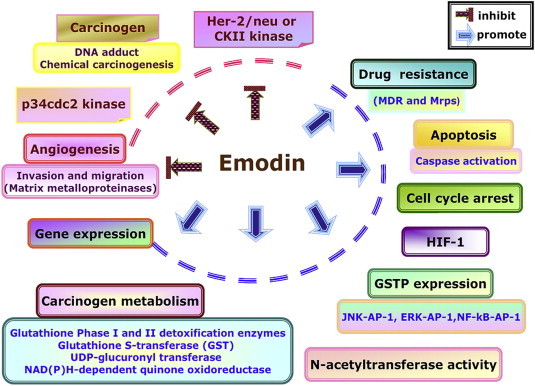
The pharmacology of emodin as a possible anti-cancer therapy.
References
- 1.Wang M., Zhao R., Wang W., Mao X., Yu J. Lipid regulation effects of Polygoni Multiflori Radix, its processed products and its major substances on steatosis human liver cell line L02. J Ethnopharmacol. 2012;139:287–293. doi: 10.1016/j.jep.2011.11.022. [DOI] [PubMed] [Google Scholar]
- 2.Rao G.X., Xue Y.M., Hui T.T., Wang W.J., Zhang Q.L. Studies on the chemical constituents of the leaves of Polygonum multiflorum. Zhong Yao Cai. 2009;32:891–893. [PubMed] [Google Scholar]
- 3.Shin J.A., Shim J.H., Jeon J.G., Choi K.H., Choi E.S., Cho N.P. Apoptotic effect of Polygonum Cuspidatum in oral cancer cells through the regulation of specificity protein 1. Oral Dis. 2011;17:162–170. doi: 10.1111/j.1601-0825.2010.01710.x. [DOI] [PubMed] [Google Scholar]
- 4.Lee M.H., Kao L., Lin C.C. Comparison of the antioxidant and transmembrane permeative activities of the different Polygonum cuspidatum extracts in phospholipid-based microemulsions. J Agric Food Chem. 2011;59:9135–9141. doi: 10.1021/jf201577f. [DOI] [PubMed] [Google Scholar]
- 5.Liu J., Xia Z.T., Zhou G.R., Zhang L.L., Kong L.Y. Study on the chemical constituents of Rumex patientis. Zhong Yao Cai. 2011;34:893–895. [PubMed] [Google Scholar]
- 6.Locatelli M., Epifano F., Genovese S., Carlucci G., Koncic M.Z., Kosalec I. Anthraquinone profile, antioxidant and antimicrobial properties of bark extracts of Rhamnus catharticus and R. orbiculatus. Nat Prod Commun. 2011;6:1275–1280. [PubMed] [Google Scholar]
- 7.Naqvi S., Ullah M.F., Hadi S.M. DNA degradation by aqueous extract of Aloe vera in the presence of copper ions. Indian J Biochem Biophys. 2010;47:161–165. [PubMed] [Google Scholar]
- 8.Zhu M., Tan N., Ji C., Xu J., He W., Zhang Y. Chemical constituents from petroleum ether fraction of ethanol extract of Acorus tatarinowii. Zhongguo Zhong Yao Za Zhi. 2010;35:173–176. [PubMed] [Google Scholar]
- 9.Yang Y.C., Lim M.Y., Lee H.S. Emodin isolated from Cassia obtusifolia (Leguminosae) seed shows larvicidal activity against three mosquito species. J Agric Food Chem. 2003;51:7629–7631. doi: 10.1021/jf034727t. [DOI] [PubMed] [Google Scholar]
- 10.Arya V., Yadav S., Kumar S., Yadav J.P. Antioxidant activity of organic and aqueous leaf extracts of Cassia occidentalis L. in relation to their phenolic content. Nat Prod Res. 2011;25:1473–1479. doi: 10.1080/14786419.2010.545351. [DOI] [PubMed] [Google Scholar]
- 11.Wang J.B., Zhao H.P., Zhao Y.L., Jin C., Liu D.J., Kong W.J. Hepatotoxicity or hepatoprotection? Pattern recognition for the paradoxical effect of the Chinese herb Rheum palmatum L. in treating rat liver injury. PLoS One. 2011 doi: 10.1371/journal.pone.0024498. 6:e24498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wei W.T., Chen H., Ni Z.L., Liu H.B., Tong H.F., Fan L. Antitumor and apoptosis-promoting properties of emodin, an anthraquinone derivative from Rheum officinale Baill, against pancreatic cancer in mice via inhibition of Akt activation. Int J Oncol. 2011;39:1381–1390. doi: 10.3892/ijo.2011.1147. [DOI] [PubMed] [Google Scholar]
- 13.Fang J.J., Ye G., Chen W.L., Zhao W.M. Antibacterial phenolic components from Eriocaulon buergerianum. Phytochemistry. 2008;69:1279–1286. doi: 10.1016/j.phytochem.2007.11.014. [DOI] [PubMed] [Google Scholar]
- 14.Xing Y.M., Chen J., Cui J.L., Chen X.M., Guo S.X. Antimicrobial activity and biodiversity of endophytic fungi in Dendrobium devonianum and Dendrobium thyrsiflorum from Vietnam. Curr Microbiol. 2011;62:1218–1224. doi: 10.1007/s00284-010-9848-2. [DOI] [PubMed] [Google Scholar]
- 15.Su C.R., Chen Y.F., Liou M.J., Tsai H.Y., Chang W.S., Wu T.S. Anti-inflammatory activities of furanoditerpenoids and other constituents from Fibraurea tinctoria. Bioorg Med Chem. 2008;16:9603–9609. doi: 10.1016/j.bmc.2008.09.023. [DOI] [PubMed] [Google Scholar]
- 16.Tjong Y., Ip S., Lao L., Fong H.H., Sung J.J., Berman B. Analgesic effect of Coptis chinensis rhizomes (Coptidis Rhizoma) extract on rat model of irritable bowel syndrome. J Ethnopharmacol. 2011;135:754–761. doi: 10.1016/j.jep.2011.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lin C.W., Tsai F.J., Tsai C.H., Lai C.C., Wan L., Ho T.Y. Anti-SARS coronavirus 3C-like protease effects of Isatis indigotica root and plant-derived phenolic compounds. Antiviral Res. 2005;68:36–42. doi: 10.1016/j.antiviral.2005.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hasan A., Ahmed I., Jay M., Voirin B. Flavonoid glycosides and an anthraquinone from Rumex chalepensis. Phytochemistry. 1995;39:1211–1213. doi: 10.1016/0031-9422(95)00071-e. [DOI] [PubMed] [Google Scholar]
- 19.Lu C.C., Yang J.S., Huang A.C., Hsia T.C., Chou S.T., Kuo C.L. Chrysophanol induces necrosis through the production of ROS and alteration of ATP levels in J5 human liver cancer cells. Mol Nutr Food Res. 2010;54:967–976. doi: 10.1002/mnfr.200900265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chiang J.H., Yang J.S., Ma C.Y., Yang M.D., Huang H.Y., Hsia T.C. Danthron, an anthraquinone derivative, induces DNA damage and caspase cascades-mediated apoptosis in SNU-1 human gastric cancer cells through mitochondrial permeability transition pores and Bax-triggered pathways. Chem Res Toxicol. 2011;24:20–29. doi: 10.1021/tx100248s. [DOI] [PubMed] [Google Scholar]
- 21.Lee M.S., Sohn C.B. Anti-diabetic properties of chrysophanol and its glucoside from rhubarb rhizome. Biol Pharm Bull. 2008;31:2154–2157. doi: 10.1248/bpb.31.2154. [DOI] [PubMed] [Google Scholar]
- 22.Ibrahim M., Khan A.A., Tiwari S.K., Habeeb M.A., Khaja M.N., Habibullah C.M. Antimicrobial activity of Sapindus mukorossi and Rheum emodi extracts against H pylori: in vitro and in vivo studies. World J Gastroenterol. 2006;12:7136–7142. doi: 10.3748/wjg.v12.i44.7136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rajkumar V., Guha G., Ashok Kumar R. Antioxidant and anti-cancer potentials of Rheum emodi rhizome extracts. Evid Based Complement Alternat Med. 2011:697986. doi: 10.1093/ecam/neq048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Luo W., Su X., Gong S., Qin Y., Liu W., Li J. Anti-SARS coronavirus 3C-like protease effects of Rheum palmatum L. extracts. Biosci Trends. 2009;3:124–126. [PubMed] [Google Scholar]
- 25.Liu L., Wang Z.P., Xu C.T., Pan B.R., Mei Q.B., Long Y. Effects of Rheum tanguticum polysaccharide on TNBS-induced colitis and CD4+T cells in rats. World J Gastroenterol. 2003;9:2284–2288. doi: 10.3748/wjg.v9.i10.2284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhao Y.Q., Liu X.H., Ito T., Qian J.M. Protective effects of rhubarb on experimental severe acute pancreatitis. World J Gastroenterol. 2004;10:1005–1009. doi: 10.3748/wjg.v10.i7.1005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Matsuda H., Tomohiro N., Hiraba K., Harima S., Ko S., Matsuo K. Study on anti-Oketsu activity of rhubarb II: anti-allergic effects of stilbene components from Rhei undulati Rhizoma (dried rhizome of Rheum undulatum cultivated in Korea) Biol Pharm Bull. 2001;24:264–267. doi: 10.1248/bpb.24.264. [DOI] [PubMed] [Google Scholar]
- 28.Choi E.S., Cho S.D., Jeon J.G., Cho N.P. The apoptotic effect of the hexane extract of Rheum undulatum L. in oral cancer cells through the down-regulation of specificity protein 1 and survivin. Lab Anim Res. 2011;27:19–24. doi: 10.5625/lar.2011.27.1.19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhang G., el Nahas A.M. The effect of rhubarb extract on experimental renal fibrosis. Nephrol Dial Transplant. 1996;11:186–190. [PubMed] [Google Scholar]
- 30.Vollmer G., Papke A., Zierau O. Treatment of menopausal symptoms by an extract from the roots of rhapontic rhubarb: the role of estrogen receptors. Chin Med. 2010;5:7. doi: 10.1186/1749-8546-5-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ngoc T.M., Hung T.M., Thuong P.T., Na M., Kim H., Ha do T. Inhibition of human low density lipoprotein and high density lipoprotein oxidation by oligostilbenes from rhubarb. Biol Pharm Bull. 2008;31:1809–1812. doi: 10.1248/bpb.31.1809. [DOI] [PubMed] [Google Scholar]
- 32.Teng Z.H., Zhou S.Y., Ran Y.H., Liu X.Y., Yang R.T., Yang X. Cellular absorption of anthraquinones emodin and chrysophanol in human intestinal Caco-2 cells. Biosci Biotechnol Biochem. 2007;71:1636–1643. doi: 10.1271/bbb.70025. [DOI] [PubMed] [Google Scholar]
- 33.Kim M.S., Park M.J., Kim S.J., Lee C.H., Yoo H., Shin S.H. Emodin suppresses hyaluronic acid-induced MMP-9 secretion and invasion of glioma cells. Int J Oncol. 2005;27:839–846. [PubMed] [Google Scholar]
- 34.Kuo Y.C., Meng H.C., Tsai W.J. Regulation of cell proliferation, inflammatory cytokine production and calcium mobilization in primary human T lymphocytes by emodin from Polygonum hypoleucum Ohwi. Inflamm Res. 2001;50:73–82. doi: 10.1007/s000110050727. [DOI] [PubMed] [Google Scholar]
- 35.National Toxicology Program. NTP toxicology and carcinogenesis studies of EMODIN (CAS NO. 518-82-1): feed studies in F344/N rats and B6C3F1 mice. Natl Toxicol Program Tech Rep Ser. 2001;493:1–278. [PubMed] [Google Scholar]
- 36.Lee H.Z. Effects and mechanisms of emodin on cell death in human lung squamous cell carcinoma. Br J Pharmacol. 2001;134:11–20. doi: 10.1038/sj.bjp.0704205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Koyama J., Morita I., Tagahara K., Nobukuni Y., Mukainaka T., Kuchide M. Chemopreventive effects of emodin and cassiamin B in mouse skin carcinogenesis. Cancer Lett. 2002;182:135–139. doi: 10.1016/s0304-3835(02)00100-3. [DOI] [PubMed] [Google Scholar]
- 38.Chen Y.C., Shen S.C., Lee W.R., Hsu F.L., Lin H.Y., Ko C.H. Emodin induces apoptosis in human promyeloleukemic HL-60 cells accompanied by activation of caspase 3 cascade but independent of reactive oxygen species production. Biochem Pharmacol. 2002;64:1713–1724. doi: 10.1016/s0006-2952(02)01386-2. [DOI] [PubMed] [Google Scholar]
- 39.Srinivas G., Anto R.J., Srinivas P., Vidhyalakshmi S., Senan V.P., Karunagaran D. Emodin induces apoptosis of human cervical cancer cells through poly(ADP-ribose) polymerase cleavage and activation of caspase-9. Eur J Pharmacol. 2003;473:117–125. doi: 10.1016/s0014-2999(03)01976-9. [DOI] [PubMed] [Google Scholar]
- 40.Shieh D.E., Chen Y.Y., Yen M.H., Chiang L.C., Lin C.C. Emodin-induced apoptosis through p53-dependent pathway in human hepatoma cells. Life Sci. 2004;74:2279–2290. doi: 10.1016/j.lfs.2003.09.060. [DOI] [PubMed] [Google Scholar]
- 41.Yi J., Yang J., He R., Gao F., Sang H., Tang X. Emodin enhances arsenic trioxide-induced apoptosis via generation of reactive oxygen species and inhibition of survival signaling. Cancer Res. 2004;64:108–116. doi: 10.1158/0008-5472.can-2820-2. [DOI] [PubMed] [Google Scholar]
- 42.Wang X.J., Yang J., Cang H., Zou Y.Q., Yi J. Gene expression alteration during redox-dependent enhancement of arsenic cytotoxicity by emodin in HeLa cells. Cell Res. 2005;15:511–522. doi: 10.1038/sj.cr.7290321. [DOI] [PubMed] [Google Scholar]
- 43.Cha T.L., Qiu L., Chen C.T., Wen Y., Hung M.C. Emodin down-regulates androgen receptor and inhibits prostate cancer cell growth. Cancer Res. 2005;65:2287–2295. doi: 10.1158/0008-5472.CAN-04-3250. [DOI] [PubMed] [Google Scholar]
- 44.Huang Q., Shen H.M., Shui G., Wenk M.R., Ong C.N. Emodin inhibits tumor cell adhesion through disruption of the membrane lipid raft-associated integrin signaling pathway. Cancer Res. 2006;66:5807–5815. doi: 10.1158/0008-5472.CAN-06-0077. [DOI] [PubMed] [Google Scholar]
- 45.Muto A., Hori M., Sasaki Y., Saitoh A., Yasuda I., Maekawa T. Emodin has a cytotoxic activity against human multiple myeloma as a Janus-activated kinase 2 inhibitor. Mol Cancer Ther. 2007;6:987–994. doi: 10.1158/1535-7163.MCT-06-0605. [DOI] [PubMed] [Google Scholar]
- 46.Wu Y., Tu X., Lin G., Xia H., Huang H., Wan J. Emodin-mediated protection from acute myocardial infarction via inhibition of inflammation and apoptosis in local ischemic myocardium. Life Sci. 2007;81:1332–1338. doi: 10.1016/j.lfs.2007.08.040. [DOI] [PubMed] [Google Scholar]
- 47.Zhang L., Lau Y.K., Xi L., Hong R.L., Kim D.S., Chen C.F. Tyrosine kinase inhibitors, emodin and its derivative repress HER-2/neu-induced cellular transformation and metastasis-associated properties. Oncogene. 1998;16:2855–2863. doi: 10.1038/sj.onc.1201813. [DOI] [PubMed] [Google Scholar]
- 48.Yan Y.Y., Zheng L.S., Zhang X., Chen L.K., Singh S., Wang F. Blockade of Her2/neu binding to Hsp90 by emodin azide methyl anthraquinone derivative induces proteasomal degradation of Her2/neu. Mol Pharm. 2011;8:1687–1697. doi: 10.1021/mp2000499. [DOI] [PubMed] [Google Scholar]
- 49.Yim H., Lee Y.H., Lee C.H., Lee S.K. Emodin, an anthraquinone derivative isolated from the rhizomes of Rheum palmatum, selectively inhibits the activity of casein kinase II as a competitive inhibitor. Planta Med. 1999;65:9–13. doi: 10.1055/s-1999-13953. [DOI] [PubMed] [Google Scholar]
- 50.Ahn B.H., Min G., Bae Y.S., Min D.S. Phospholipase D is activated and phosphorylated by casein kinase-II in human U87 astroglioma cells. Exp Mol Med. 2006;38:55–62. doi: 10.1038/emm.2006.7. [DOI] [PubMed] [Google Scholar]
- 51.Lin S.Y., Lai W.W., Ho C.C., Yu F.S., Chen G.W., Yang J.S. Emodin induces apoptosis of human tongue squamous cancer SCC-4 cells through reactive oxygen species and mitochondria-dependent pathways. Anticancer Res. 2009;29:327–335. [PubMed] [Google Scholar]
- 52.Lin M.L., Lu Y.C., Chung J.G., Wang S.G., Lin H.T., Kang S.E. Down-regulation of MMP-2 through the p38 MAPK-NF-kappaB-dependent pathway by aloe-emodin leads to inhibition of nasopharyngeal carcinoma cell invasion. Mol Carcinog. 2010;49:783–797. doi: 10.1002/mc.20652. [DOI] [PubMed] [Google Scholar]
- 53.Liu A., Chen H., Tong H., Ye S., Qiu M., Wang Z. Emodin potentiates the antitumor effects of gemcitabine in pancreatic cancer cells via inhibition of nuclear factor-κB. Mol Med Report. 2011;4:221–227. doi: 10.3892/mmr.2011.414. [DOI] [PubMed] [Google Scholar]
- 54.Meng G., Liu Y., Lou C., Yang H. Emodin suppresses lipopolysaccharide-induced pro-inflammatory responses and NF-kappaB activation by disrupting lipid rafts in CD14-negative endothelial cells. Br J Pharmacol. 2010;161:1628–1644. doi: 10.1111/j.1476-5381.2010.00993.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kuo P.L., Lin T.C., Lin C.C. The antiproliferative activity of aloe-emodin is through p53-dependent and p21-dependent apoptotic pathway in human hepatoma cell lines. Life Sci. 2002;71:1879–1892. doi: 10.1016/s0024-3205(02)01900-8. [DOI] [PubMed] [Google Scholar]
- 56.Chang C.J., Ashendel C.L., Geahlen R.L., McLaughlin J.L., Waters D.J. Oncogene signal transduction inhibitors from medicinal plants. Vivo. 1996;10:185–190. [PubMed] [Google Scholar]
- 57.Zhang Z., Yan J., Chang Y., ShiDu Yan S., Shi H. Hypoxia inducible factor-1 as a target for neurodegenerative diseases. Curr Med Chem. 2011;18:4335–4343. doi: 10.2174/092986711797200426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Wang R., Zhou S., Li S. Cancer therapeutic agents targeting hypoxia-inducible factor-1. Curr Med Chem. 2011;18:3168–3189. doi: 10.2174/092986711796391606. [DOI] [PubMed] [Google Scholar]
- 59.Yon J.M., Baek I.J., Lee B.J., Yun Y.W., Nam S.Y. Emodin and [6]-gingerol lessen hypoxia-induced embryotoxicities in cultured mouse whole embryos via upregulation of hypoxia-inducible factor 1alpha and intracellular superoxide dismutases. Reprod Toxicol. 2011;31:513–518. doi: 10.1016/j.reprotox.2011.02.011. [DOI] [PubMed] [Google Scholar]
- 60.Huang X.Z., Wang J., Huang C., Chen Y.Y., Shi G.Y., Hu Q.S. Emodin enhances cytotoxicity of chemotherapeutic drugs in prostate cancer cells: the mechanisms involve ROS-mediated suppression of multidrug resistance and hypoxia inducible factor-1. Cancer Biol Ther. 2008;7:468–475. doi: 10.4161/cbt.7.3.5457. [DOI] [PubMed] [Google Scholar]
- 61.Chung J.G., Li Y.C., Lee Y.M., Lin J.P., Cheng K.C., Chang W.C. Aloe-emodin inhibited N-acetylation and DNA adduct of 2-aminofluorene and arylamine N-acetyltransferase gene expression in mouse leukemia L 1210 cells. Leuk Res. 2003;27:831–840. doi: 10.1016/s0145-2126(03)00017-1. [DOI] [PubMed] [Google Scholar]
- 62.Lin S.Y., Yang J.H., Hsia T.C., Lee J.H., Chiu T.H., Wei Y.H. Effect of inhibition of aloe-emodin on N-acetyltransferase activity and gene expression in human malignant melanoma cells (A375.S2) Melanoma Res. 2005;15:489–494. doi: 10.1097/00008390-200512000-00002. [DOI] [PubMed] [Google Scholar]
- 63.Wang H.H., Chung J.G., Ho C.C., Wu L.T., Chang S.H. Aloe-emodin effects on arylamine N-acetyltransferase activity in the bacterium Helicobacter pylori. Planta Med. 1998;64:176–178. doi: 10.1055/s-2006-957399. [DOI] [PubMed] [Google Scholar]
- 64.Chung J.G., Wang H.H., Wu L.T., Chang S.S., Chang W.C. Inhibitory actions of emodin on arylamine N-acetyltransferase activity in strains of Helicobacter pylori from peptic ulcer patients. Food Chem Toxicol. 1997;35:1001–1007. doi: 10.1016/s0278-6915(97)87269-9. [DOI] [PubMed] [Google Scholar]
- 65.Mason E.F., Rathmell J.C. Cell metabolism: an essential link between cell growth and apoptosis. Biochim Biophys Acta. 2011;1813:645–654. doi: 10.1016/j.bbamcr.2010.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Medema R.H., Macurek L. Checkpoint control and cancer. Oncogene. 2011 doi: 10.1038/onc.2011.451. [DOI] [PubMed] [Google Scholar]
- 67.Brown M., Bellon M., Nicot C. Emodin and DHA potently increase arsenic trioxide interferon-alpha-induced cell death of HTLV-I-transformed cells by generation of reactive oxygen species and inhibition of Akt and AP-1. Blood. 2007;109:1653–1659. doi: 10.1182/blood-2006-04-015537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Hsu C.M., Hsu Y.A., Tsai Y., Shieh F.K., Huang S.H., Wan L. Emodin inhibits the growth of hepatoma cells: finding the common anti-cancer pathway using Huh7, Hep3B, and HepG2 cells. Biochem Biophys Res Commun. 2010;392:473–478. doi: 10.1016/j.bbrc.2009.10.153. [DOI] [PubMed] [Google Scholar]
- 69.Kwak H.J., Park M.J., Park C.M., Moon S.I., Yoo D.H., Lee H.C. Emodin inhibits vascular endothelial growth factor-A-induced angiogenesis by blocking receptor-2 (KDR/Flk-1) phosphorylation. Int J Cancer. 2006;118:2711–2720. doi: 10.1002/ijc.21641. [DOI] [PubMed] [Google Scholar]
- 70.Wang X., Zou Y., Sun A., Xu D., Niu Y., Wang S. Emodin induces growth arrest and death of human vascular smooth muscle cells through reactive oxygen species and p53. J Cardiovasc Pharmacol. 2007;49:253–260. doi: 10.1097/FJC.0b013e318033dfb3. [DOI] [PubMed] [Google Scholar]
- 71.Chen H.C., Hsieh W.T., Chang W.C., Chung J.G. Aloe-emodin induced in vitro G2/M arrest of cell cycle in human promyelocytic leukemia HL-60 cells. Food Chem Toxicol. 2004;42:1251–1257. doi: 10.1016/j.fct.2004.03.002. [DOI] [PubMed] [Google Scholar]
- 72.Guo J.M., Xiao B.X., Liu Q., Zhang S., Liu D.H., Gong Z.H. Anticancer effect of aloe-emodin on cervical cancer cells involves G2/M arrest and induction of differentiation. Acta Pharmacol Sin. 2007;28:1991–1995. doi: 10.1111/j.1745-7254.2007.00707.x. [DOI] [PubMed] [Google Scholar]
- 73.Xiao B., Guo J., Liu D., Zhang S. Aloe-emodin induces in vitro G2/M arrest and alkaline phosphatase activation in human oral cancer KB cells. Oral Oncol. 2007;43:905–910. doi: 10.1016/j.oraloncology.2006.11.002. [DOI] [PubMed] [Google Scholar]
- 74.Lin J.G., Chen G.W., Li T.M., Chouh S.T., Tan T.W., Chung J.G. Aloe-emodin induces apoptosis in T24 human bladder cancer cells through the p53 dependent apoptotic pathway. J Urol. 2006;175:343–347. doi: 10.1016/S0022-5347(05)00005-4. [DOI] [PubMed] [Google Scholar]
- 75.Lin M.L., Lu Y.C., Su H.L., Lin H.T., Lee C.C., Kang S.E. Destabilization of CARP mRNAs by aloe-emodin contributes to caspase-8-mediated p53-independent apoptosis of human carcinoma cells. J Cell Biochem. 2011;112:1176–1191. doi: 10.1002/jcb.23031. [DOI] [PubMed] [Google Scholar]
- 76.Lin M.L., Lu Y.C., Chung J.G., Li Y.C., Wang S.G., N G.S. Aloe-emodin induces apoptosis of human nasopharyngeal carcinoma cells via caspase-8-mediated activation of the mitochondrial death pathway. Cancer Lett. 2010;291:46–58. doi: 10.1016/j.canlet.2009.09.016. [DOI] [PubMed] [Google Scholar]
- 77.Lu G.D., Shen H.M., Ong C.N., Chung M.C. Anticancer effects of aloe-emodin on HepG2 cells: cellular and proteomic studies. Proteomics Clin Appl. 2007;1:410–419. doi: 10.1002/prca.200600798. [DOI] [PubMed] [Google Scholar]
- 78.Liu X.N., Zhang C.Y., Jin X.D., Li Y.Z., Zheng X.Z., Li L. Inhibitory effect of schisandrin B on gastric cancer cells in vitro. World J Gastroenterol. 2007;13:6506–6511. doi: 10.3748/wjg.v13.i48.6506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Dive C., Evans C.A., Whetton A.D. Induction of apoptosis—new targets for cancer chemotherapy. Semin Cancer Biol. 1992;3:417–427. [PubMed] [Google Scholar]
- 80.Sen S., D'Incalci M. Apoptosis. Biochemical events and relevance to cancer chemotherapy. FEBS Lett. 1992;307:122–127. doi: 10.1016/0014-5793(92)80914-3. [DOI] [PubMed] [Google Scholar]
- 81.Jeon W., Jeon Y.K., Nam M.J. Apoptosis by aloe-emodin is mediated through down-regulation of calpain-2 and ubiquitin-protein ligase E3A in human hepatoma Huh-7 cells. Cell Biol Int. 2012;36:163–167. doi: 10.1042/CBI20100723. [DOI] [PubMed] [Google Scholar]
- 82.Ali B.H., Al-Salam S., Al Husseini I.S., Al-Lawati I., Waly M., Yasin J. Abrogation of cisplatin-induced nephrotoxicity by emodin in rats. Fundam Clin Pharmacol. 2011 doi: 10.1111/j.1472-8206.2011.01003.x. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 83.Wang W., Sun Y., Li X., Li H., Chen Y., Tian Y. Emodin potentiates the anticancer effect of cisplatin on gallbladder cancer cells through the generation of reactive oxygen species and the inhibition of survivin expression. Oncol Rep. 2011;26:1143–1148. doi: 10.3892/or.2011.1390. [DOI] [PubMed] [Google Scholar]
- 84.Wang Z.H., Chen H., Guo H.C., Tong H.F., Liu J.X., Wei W.T. Enhanced antitumor efficacy by the combination of emodin and gemcitabine against human pancreatic cancer cells via downregulation of the expression of XIAP in vitro and in vivo. Int J Oncol. 2011;39:1123–1131. doi: 10.3892/ijo.2011.1115. [DOI] [PubMed] [Google Scholar]
- 85.Zhou X., Wang L., Wang M., Xu L., Yu L., Fang T. Emodin-induced microglial apoptosis is associated with TRB3 induction. Immunopharmacol Immunotoxicol. 2011;33:594–602. doi: 10.3109/08923973.2010.549135. [DOI] [PubMed] [Google Scholar]
- 86.Lai J.M., Chang J.T., Wen C.L., Hsu S.L. Emodin induces a reactive oxygen species-dependent and ATM-p53-Bax mediated cytotoxicity in lung cancer cells. Eur J Pharmacol. 2009;623:1–9. doi: 10.1016/j.ejphar.2009.08.031. [DOI] [PubMed] [Google Scholar]
- 87.Guo Q., Chen Y., Zhang B., Kang M., Xie Q., Wu Y. Potentiation of the effect of gemcitabine by emodin in pancreatic cancer is associated with survivin inhibition. Biochem Pharmacol. 2009;77:1674–1683. doi: 10.1016/j.bcp.2009.02.021. [DOI] [PubMed] [Google Scholar]
- 88.Heo S.K., Yun H.J., Park W.H., Park S.D. Emodin inhibits TNF-alpha-induced human aortic smooth-muscle cell proliferation via caspase- and mitochondrial-dependent apoptosis. J Cell Biochem. 2008;105:70–80. doi: 10.1002/jcb.21805. [DOI] [PubMed] [Google Scholar]
- 89.Wang C., Wu X., Chen M., Duan W., Sun L., Yan M. Emodin induces apoptosis through caspase 3-dependent pathway in HK-2 cells. Toxicology. 2007;231:120–128. doi: 10.1016/j.tox.2006.11.064. [DOI] [PubMed] [Google Scholar]
- 90.Yu C.X., Zhang X.Q., Kang L.D., Zhang P.J., Chen W.W., Liu W.W. Emodin induces apoptosis in human prostate cancer cell LNCaP. Asian J Androl. 2008;10:625–634. doi: 10.1111/j.1745-7262.2008.00397.x. [DOI] [PubMed] [Google Scholar]
- 91.Wang X.D., Gu L.Q., Wu J.Y. Apoptosis-inducing activity of new pyrazole emodin derivatives in human hepatocellular carcinoma HepG2 cells. Biol Pharm Bull. 2007;30:1113–1116. doi: 10.1248/bpb.30.1113. [DOI] [PubMed] [Google Scholar]
- 92.Yeh F.T., Wu C.H., Lee H.Z. Signaling pathway for aloe-emodin-induced apoptosis in human H460 lung nonsmall carcinoma cell. Int J Cancer. 2003;106:26–33. doi: 10.1002/ijc.11185. [DOI] [PubMed] [Google Scholar]
- 93.Lee H.Z., Hsu S.L., Liu M.C., Wu C.H. Effects and mechanisms of aloe-emodin on cell death in human lung squamous cell carcinoma. Eur J Pharmacol. 2001;431:287–295. doi: 10.1016/s0014-2999(01)01467-4. [DOI] [PubMed] [Google Scholar]
- 94.Lin S.Z., Chen K.J., Tong H.F., Jing H., Li H., Zheng S.S. Emodin attenuates acute rejection of liver allografts by inhibiting hepatocellular apoptosis and modulating the Th1/Th2 balance in rats. Clin Exp Pharmacol Physiol. 2010;37:790–794. doi: 10.1111/j.1440-1681.2010.05378.x. [DOI] [PubMed] [Google Scholar]
- 95.Liu D.L., Bu H., Li H., Chen H., Guo H.C., Wang Z.H. Emodin reverses gemcitabine resistance in pancreatic cancer cells via. Int J Oncol. 2012;40:1049–1057. doi: 10.3892/ijo.2011.1285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Raza H. Dual localization of glutathione S-transferase in the cytosol and mitochondria: implications in oxidative stress, toxicity and disease. FEBS J. 2011;278:4243–4251. doi: 10.1111/j.1742-4658.2011.08358.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Bhabak K.P., Mugesh G. Functional mimics of glutathione peroxidase: bioinspired synthetic antioxidants. Acc Chem Res. 2010;43:1408–1419. doi: 10.1021/ar100059g. [DOI] [PubMed] [Google Scholar]
- 98.Lubos E., Loscalzo J., Handy D.E. Glutathione peroxidase-1 in health and disease: from molecular mechanisms to therapeutic opportunities. Antioxid Redox Signal. 2011;15:1957–1997. doi: 10.1089/ars.2010.3586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Brigelius-Flohe R., Kipp A. Glutathione peroxidases in different stages of carcinogenesis. Biochim Biophys Acta. 2009;1790:1555–1568. doi: 10.1016/j.bbagen.2009.03.006. [DOI] [PubMed] [Google Scholar]
- 100.Kalinina E.V., Chernov N.N., Aleud R., Novichkova M.D., Saprin A.N., Berezov T.T. Current views on antioxidative activity of glutathione and glutathione-depending enzymes. Vestn Ross Akad Med Nauk. 2010:46–54. [PubMed] [Google Scholar]
- 101.Lee B.H., Huang Y.Y., Duh P.D., Wu S.C. Hepatoprotection of emodin and Polygonum multiflorum against CCl(4)-induced liver injury. Pharm Biol. 2012;50:351–359. doi: 10.3109/13880209.2011.604335. [DOI] [PubMed] [Google Scholar]
- 102.Singh K.B., Trigun S.K. Apoptosis of Dalton's lymphoma due to in vivo treatment with emodin is associated with modulations of hydrogen peroxide metabolizing antioxidant enzymes. Cell Biochem Biophys. 2011 doi: 10.1007/s12013-011-9305-2. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 103.Du Y., Ko K.M. Effects of emodin treatment on mitochondrial ATP generation capacity and antioxidant components as well as susceptibility to ischemia-reperfusion injury in rat hearts: single versus multiple doses and gender difference. Life Sci. 2005;77:2770–2782. doi: 10.1016/j.lfs.2005.03.027. [DOI] [PubMed] [Google Scholar]
- 104.Chiu P.Y., Mak D.H., Poon M.K., Ko K.M. In vivo antioxidant action of a lignan-enriched extract of Schisandra fruit and an anthraquinone-containing extract of Polygonum root in comparison with schisandrin B and emodin. Planta Med. 2002;68:951–956. doi: 10.1055/s-2002-35661. [DOI] [PubMed] [Google Scholar]
- 105.Yim T.K., Wu W.K., Mak D.H., Ko K.M. Myocardial protective effect of an anthraquinone-containing extract of Polygonum multiflorum ex vivo. Planta Med. 1998;64:607–611. doi: 10.1055/s-2006-957531. [DOI] [PubMed] [Google Scholar]
- 106.Chang L.C., Sheu H.M., Huang Y.S., Tsai T.R., Kuo K.W. A novel function of emodin: enhancement of the nucleotide excision repair of UV- and cisplatin-induced DNA damage in human cells. Biochem Pharmacol. 1999;58:49–57. doi: 10.1016/s0006-2952(99)00075-1. [DOI] [PubMed] [Google Scholar]
- 107.Chen Y.Y., Chiang S.Y., Lin J.G., Yang J.S., Ma Y.S., Liao C.L. Emodin, aloe-emodin and rhein induced DNA damage and inhibited DNA repair gene expression in SCC-4 human tongue cancer cells. Anticancer Res. 2010;30:945–951. [PubMed] [Google Scholar]
- 108.Su H.Y., Cherng S.H., Chen C.C., Lee H. Emodin inhibits the mutagenicity and DNA adducts induced by 1-nitropyrene. Mutat Res. 1995;329:205–212. doi: 10.1016/0027-5107(95)00045-k. [DOI] [PubMed] [Google Scholar]
- 109.Huang Z., Chen G., Shi P. Effects of emodin on the gene expression profiling of human breast carcinoma cells. Cancer Detect Prev. 2009;32:286–291. doi: 10.1016/j.cdp.2008.12.003. [DOI] [PubMed] [Google Scholar]
- 110.Sui F., Huo H.R., Zhang C.B., Yang N., Guo J.Y., Du X.L. Emodin down-regulates expression of TRPV1 mRNA and its function in DRG neurons in vitro. Am J Chin Med. 2010;38:789–800. doi: 10.1142/S0192415X1000824X. [DOI] [PubMed] [Google Scholar]
- 111.Ko J.C., Su Y.J., Lin S.T., Jhan J.Y., Ciou S.C., Cheng C.M. Suppression of ERCC1 and Rad51 expression through ERK1/2 inactivation is essential in emodin-mediated cytotoxicity in human non-small cell lung cancer cells. Biochem Pharmacol. 2010;79:655–664. doi: 10.1016/j.bcp.2009.09.024. [DOI] [PubMed] [Google Scholar]
- 112.Oshida K., Hirakata M., Maeda A., Miyoshi T., Miyamoto Y. Toxicological effect of emodin in mouse testicular gene expression profile. J Appl Toxicol. 2011;31:790–800. doi: 10.1002/jat.1637. [DOI] [PubMed] [Google Scholar]
- 113.Harhaji L., Mijatovic S., Maksimovic-Ivanic D., Popadic D., Isakovic A., Todorovic-Markovic B. Aloe emodin inhibits the cytotoxic action of tumor necrosis factor. Eur J Pharmacol. 2007;568:248–259. doi: 10.1016/j.ejphar.2007.04.029. [DOI] [PubMed] [Google Scholar]
- 114.Chen Y.Y., Chiang S.Y., Lin J.G., Ma Y.S., Liao C.L., Weng S.W. Emodin, aloe-emodin and rhein inhibit migration and invasion in human tongue cancer SCC-4 cells through the inhibition of gene expression of matrix metalloproteinase-9. Int J Oncol. 2010;36:1113–1120. doi: 10.3892/ijo_00000593. [DOI] [PubMed] [Google Scholar]
- 115.Tew K.D., Manevich Y., Grek C., Xiong Y., Uys J., Townsend D.M. The role of glutathione S-transferase P in signaling pathways and S-glutathionylation in cancer. Free Radic Biol Med. 2011;51:299–313. doi: 10.1016/j.freeradbiomed.2011.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Tew K.D., Townsend D.M. Regulatory functions of glutathione S-transferase P1-1 unrelated to detoxification. Drug Metab Rev. 2011;43:179–193. doi: 10.3109/03602532.2011.552912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Liu T., Jin H., Sun Q.R., Xu J.H., Hu H.T. Neuroprotective effects of emodin in rat cortical neurons against beta-amyloid-induced neurotoxicity. Brain Res. 2010;1347:149–160. doi: 10.1016/j.brainres.2010.05.079. [DOI] [PubMed] [Google Scholar]
- 118.Su Y.T., Chang H.L., Shyue S.K., Hsu S.L. Emodin induces apoptosis in human lung adenocarcinoma cells through a reactive oxygen species-dependent mitochondrial signaling pathway. Biochem Pharmacol. 2005;70:229–241. doi: 10.1016/j.bcp.2005.04.026. [DOI] [PubMed] [Google Scholar]
- 119.Huang Q., Shen H.M., Ong C.N. Inhibitory effect of emodin on tumor invasion through suppression of activator protein-1 and nuclear factor-kappaB. Biochem Pharmacol. 2004;68:361–371. doi: 10.1016/j.bcp.2004.03.032. [DOI] [PubMed] [Google Scholar]
- 120.Folkman J. Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat Med. 1995;1:27–31. doi: 10.1038/nm0195-27. [DOI] [PubMed] [Google Scholar]
- 121.Cao Y. Antiangiogenic cancer therapy. Semin Cancer Biol. 2004;14:139–145. doi: 10.1016/j.semcancer.2003.09.018. [DOI] [PubMed] [Google Scholar]
- 122.Kaneshiro T., Morioka T., Inamine M., Kinjo T., Arakaki J., Chiba I. Anthraquinone derivative emodin inhibits tumor-associated angiogenesis through inhibition of extracellular signal-regulated kinase 1/2 phosphorylation. Eur J Pharmacol. 2006;553:46–53. doi: 10.1016/j.ejphar.2006.09.026. [DOI] [PubMed] [Google Scholar]
- 123.He Z.H., He M.F., Ma S.C., But P.P. Anti-angiogenic effects of rhubarb and its anthraquinone derivatives. J Ethnopharmacol. 2009;121:313–317. doi: 10.1016/j.jep.2008.11.008. [DOI] [PubMed] [Google Scholar]
- 124.Cardenas C., Quesada A.R., Medina M.A. Evaluation of the anti-angiogenic effect of aloe-emodin. Cell Mol Life Sci. 2006;63:3083–3089. doi: 10.1007/s00018-006-6399-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Wang X.H., Wu S.Y., Zhen Y.S. Inhibitory effects of emodin on angiogenesis. Yao Xue Xue Bao. 2004;39:254–258. [PubMed] [Google Scholar]
- 126.Lu Y., Zhang J., Qian J. The effect of emodin on VEGF receptors in human colon cancer cells. Cancer Biother Radiopharm. 2008;23:222–228. doi: 10.1089/cbr.2007.0425. [DOI] [PubMed] [Google Scholar]
- 127.Radha K.S., Madhyastha H.K., Nakajima Y., Omura S., Maruyama M. Emodin upregulates urokinase plasminogen activator, plasminogen activator inhibitor-1 and promotes wound healing in human fibroblasts. Vascul Pharmacol. 2008;48:184–190. doi: 10.1016/j.vph.2008.02.002. [DOI] [PubMed] [Google Scholar]
- 128.Liu A., Chen H., Wei W., Ye S., Liao W., Gong J. Antiproliferative and antimetastatic effects of emodin on human pancreatic cancer. Oncol Rep. 2011;26:81–89. doi: 10.3892/or.2011.1257. [DOI] [PubMed] [Google Scholar]
- 129.Lu H.F., Lai K.C., Hsu S.C., Lin H.J., Kuo C.L., Liao C.L. Involvement of matrix metalloproteinases on the inhibition of cells invasion and migration by emodin in human neuroblastoma SH-SY5Y cells. Neurochem Res. 2009;34:1575–1583. doi: 10.1007/s11064-009-9946-3. [DOI] [PubMed] [Google Scholar]
- 130.Wesolowska O. Interaction of phenothiazines, stilbenes and flavonoids with multidrug resistance-associated transporters, P-glycoprotein and MRP1. Acta Biochim Pol. 2011;58:433–448. [PubMed] [Google Scholar]
- 131.Garg A.K., Buchholz T.A., Aggarwal B.B. Chemosensitization and radiosensitization of tumors by plant polyphenols. Antioxid Redox Signal. 2005;7:1630–1647. doi: 10.1089/ars.2005.7.1630. [DOI] [PubMed] [Google Scholar]
- 132.Kuo T.C., Yang J.S., Lin M.W., Hsu S.C., Lin J.J., Lin H.J. Emodin has cytotoxic and protective effects in rat C6 glioma cells: roles of Mdr1a and nuclear factor kappaB in cell survival. J Pharmacol Exp Ther. 2009;330:736–744. doi: 10.1124/jpet.109.153007. [DOI] [PubMed] [Google Scholar]
- 133.Wang W., Sun Y.P., Huang X.Z., He M., Chen Y.Y., Shi G.Y. Emodin enhances sensitivity of gallbladder cancer cells to platinum drugs via glutathion depletion and MRP1 downregulation. Biochem Pharmacol. 2010;79:1134–1140. doi: 10.1016/j.bcp.2009.12.006. [DOI] [PubMed] [Google Scholar]
- 134.Yu C.S., Yu F.S., Chan J.K., Li T.M., Lin S.S., Chen S.C. Aloe-emodin affects the levels of cytokines and functions of leukocytes from Sprague-Dawley rats. In Vivo. 2006;20:505–509. [PubMed] [Google Scholar]
- 135.Chang Y.C., Lai T.Y., Yu C.S., Chen H.Y., Yang J.S., Chueh F.S. Emodin induces apoptotic death in murine myelomoocytic leukemia WEHI-3 cells in vitro and enhances phagocytosis in leukemia mice in vivo. Evid Based Complement Alternat Med. 2011;2011:523596. doi: 10.1155/2011/523596. [PubMed - in process] [DOI] [PMC free article] [PubMed] [Google Scholar]


