Abstract
The local interference of cytokine signaling mediated by siRNA-loaded nanoparticles might be a promising new therapeutic approach to dampen inflammation during pulmonary diseases. For the local therapeutic treatment of pulmonary inflammation, we produced multi-shell nanoparticles consisting of a calcium phosphate core, coated with siRNAs directed against pro-inflammatory mediators, encapsulated into poly(lactic-co-glycolic acid), and coated with a final outer layer of polyethylenimine. Nasal instillation of nanoparticles loaded with a mixture of siRNAs directed against different cytokines to mice suffering from TH1 cell-mediated lung inflammation, or of siRNA directed against NS-1 in an influenza infection model led to a significant reduction of target gene expression which was accompanied by distinct amelioration of lung inflammation in both models. Thus, this study provides strong evidence that the specific and local modulation of the inflammatory response by CaP/PLGA nanoparticle-mediated siRNA delivery could be a promising approach for the treatment of inflammatory disorders of the lung.
Key words: Pulmonary inflammation, Influenza, siRNA, Nanoparticles, Delivery
Graphical Abstract
Calcium phosphate nanoparticles functionalized with siRNA were used to treat pulmonary inflammatory diseases. Inhalation of these nanoparticles leads to reduced target gene expression and results in a decreased pathology of the lung.
Pulmonary inflammatory diseases, such as chronic obstructive pulmonary disease (COPD) or interstitial lung disease (ILD), have a high prevalence around the world and are characterized by mostly chronic inflammation and irreversible airway destruction. In addition to clinical symptoms, including coughing and dyspnea, systemic effects have been reported which are the main cause of death for patients suffering from COPD.1 The etiology of these diseases is not fully understood, but environmental factors, such as the inhalation of air pollutants, and genetic susceptibility contribute to the pathogenesis. The inhalation of irritants is followed by activation of alveolar macrophages leading to the secretion of chemokines and cytokines and thereby the continuous influx of more immune cells into the lung resulting in an inflammatory response of the airways.2 Despite ongoing research, available therapies only aim to minimize symptoms, and to date no therapy that interferes with the underlying mechanisms of inflammation has reached clinical admission. Recent advances in the use of biological treatment for other inflammatory conditions led to new approaches for the therapy of chronic pulmonary inflammation. As an altered cytokine and chemokine profile is found in patients with chronic pulmonary inflammation, therapies aimed at the neutralization of cytokines or blocking of chemokine receptors are regarded as promising approaches, and several medications are under development and reached clinical trials.3 So far, no trial has reached clinical significance and it is suggested that drugs which affect the synthesis of several cytokines may be more successful than medications targeting only one specific cytokine or its receptor.4 One new way to interfere in the cytokine signaling is the use of gene silencing by RNA interference. RNA interference, mediated by small interfering RNAs (siRNAs) of 19–23 base pairs, is a natural occurring mechanism for post-transcriptional gene silencing.5 The main limitation of RNA interference for therapeutic purposes is a successful local delivery into the target cells. The transport into the cytoplasm is difficult due to instability and susceptibility to degradation by nucleases.6 Among other delivery vehicles, calcium phosphate (CaP) nanoparticles have been shown to be highly efficient as carriers of small molecules into the cell.7 As mineral in human hard tissue, like teeth or bones, calcium phosphate is a biocompatible and degradable compound. In our previous study, CaP/poly(lactic-co-glycolic acid) (PLGA) nanoparticles were designed for the successful therapeutic delivery of siRNA to the gut.8 Following these promising results of siRNA delivery, we investigated the potential of these biodegradable, non-toxic siRNA-loaded CaP/PLGA nanoparticles for the therapeutic interference of pulmonary inflammation. We analyzed the uptake of these nanoparticles by cells present in the lung in vivo and examined the therapeutic efficiency of siRNA-loaded CaP/PLGA nanoparticles in mice suffering from TH1 cell induced lung inflammation or viral infection after nasal instillation.
Methods
Preparation of CaP/PLGA nanoparticles
The nanoparticles were prepared and functionalized as described previously.9 In brief, single shell CaP/siRNA nanoparticles were synthesized by mixing equal amounts of an aqueous solution of calcium-L-lactate (6.25 mM) and diammonium hydrogen phosphate (3.74 mM). After mixing, the calcium phosphate dispersion was mixed with solutions of either functional siRNA, scrambled siRNA (GE healthcare life sciences, Chalfont St. Giles, UK), or fluorescence-labeled oligonucleotides (Life technologies, Darmstadt, Germany) (4 mg mL−1). To encapsulate the calcium phosphate nanoparticles into the biodegradable polymer poly(D,L-lactide-co-glycolide) (PLGA) (Resomer® RG 502 H, Evonik Industries, Darmstadt, Germany), a water-in-oil-in-water (W1/O/W2) double emulsion solvent evaporation method was applied. The dispersed polyvinylalcohol (PVA)-coated nanoparticles were purified by centrifugation, shock-frozen in liquid nitrogen and lyophilized. The freeze-dried particles were resuspended in a solution of aqueous PEI (2 mg mL−1 branched 25kD, Sigma-Aldrich, St. Louis, Missouri, USA) at a ratio (w/w) of 1:2 to obtain the final nanoparticle structure. The particles were characterized by scanning electron microscopy (ESEM Quanta 400) after sputtering with gold–palladium. Dynamic light scattering was performed with a Zetasizer nanoseries instrument (Nano-ZS Malvern, laser wavelength λ = 532 nm).
Mice
BALB/c mice were purchased from Harlan Laboratories (Harlan Winkelmann GmbH, Borchen, Germany). SPC-HA mice express A/PR/34 influenza HA under the control of the specific alveolar epithelial cell promoter surfactant protein C.10 TCR-HA transgenic mice exhibit CD4+ T cells expressing an α/β-TCR specific for the MHC class II H2Ed:HA110–120-restricted epitope of the HA protein.11 All animals used in this study were housed under specific pathogen-free conditions in the Laboratory Animal Facility of the University Hospital Essen. Experiments were performed in accordance with state and federal guidelines (approved by the North Rhine-Westphalia State Agency for Nature, Environment and Consumer Protection, Germany).
Isolation of cells and flow cytometric cell sorting of lymphocytes
Single cell suspensions from the lung and liver were obtained by digestion in media containing 5% FCS, and 100 U/mL collagenase IV of clostridium histolyticum (Sigma-Aldrich, Bonn, Germany) or 200 μg/ml collagenase D (Roche, Mannheim, Germany) respectively, at 37 °C for 45 minutes. Samples were pipetted up and down every 5–10 minutes. Afterwards, the remaining tissue was squashed through 70 μm cell strainers and washed with PBS containing 2% FCS and 2 mM EDTA or erythrocyte lysis buffer, respectively. Spleens and lymph nodes were squashed through 70 μm cell strainers, washed with erythrocyte lysis buffer or PBS containing 2% FCS and 2 mM EDTA, respectively. CD4+ T cells were enriched from spleens of TCR-HA transgenic mice using autoMACS technology (Miltenyi Biotech, Bergisch-Gladbach, Germany) according to manufacturer's instructions. HA-specific CD4+ T cells were obtained by staining cells with α-CD4 and an antibody against the HA-specific TCR11 and sorting them by flow cytometry using an ARIA II Sorter (BD Bioscience, Heidelberg, Germany).
TH1 cell polarization
HA-specific CD4+ T cells were sorted, activated in vitro with plate-bound α-CD3 and soluble α-CD28 antibodies (both BD Bioscience), and were polarized into IFN-γ producing TH1 cells by adding recombinant mouse IL-12 (20 ng/μL; R&D Systems, Wiesbaden, Germany) and α-IL-4 antibody (200 ng/μL; eBioscience, San Diego, CA) to the media. At day 6, TH1 polarization was confirmed by flow cytometric staining for IFN-γ.
Induction of inflammation in SPC-HA transgenic mice and treatment with nanoparticles
5 × 105 HA-specific TH1 cells were adoptively transferred into SPC-HA transgenic mice or non-transgenic littermates. Mice were monitored daily for signs of sickness and sacrificed on day 6 after transfer for analysis. For the treatment with nanoparticles, mice were anesthetized with ketamine/xylazine, and siRNA-loaded CaP/PLGA nanoparticle suspension (50 μL, containing 5 μg siRNA) was administered by nasal instillation using a pipette.
Bronchoalveolar lavage
Mice were sacrificed, the ripcage was removed, the trachea exposed, and a 22G catheter was inserted carefully in the lower trachea; 400 μL PBS was slowly infused into the lung via the catheter and the fluid was taken out again. This procedure was repeated three times and the fluid was collected for later Luminex assay analysis.
Flow cytometric analysis
Flow cytometric analysis was performed on a Canto II flow cytometer using FACS DIVA software (BD Bioscience). Oligonucleotides were labeled with AlexaFluor647 (Life technologies), and dead cells were excluded by staining cells with 7-AAD (eBioscience, San Diego, California, USA) or the fixable viability dye eFluor780 (eBioscience). Analysis was performed using α-mouse antibodies against CD45 (30-F11), MHCII (M5/114.15.2), CD11c (N418, all Biolegend, San Diego, CA), F4/80 (BM8), CD16/CD32 (93, both eBioscience, San Diego, CA), CD11b (M1/70), B220 (RA3-6B2), CD4 (RM4–5), CD8 (53–6.7, all BD Bioscience, Heidelberg, Germany), conjugated with fluorescein isothiocyanate, phyocerythrin, phyocerythrin-cyanine7, pacific blue, or brilliant violet 510. For uptake analysis in the lung cells were divided into DCs (F4/80−MHCII+CD11c+CD11b−), macrophages (F4/80+CD11cmedCD11b+), alveolar macrophages (F4/80+CD11chiCD11bmed high auto fluorescence), epithelial cells (negative for CD45, F4/80, CD11c, CD11b, CD16/32), alveolar epithelial cells type I (as epithelial cells and T1α+) and alveolar epithelial cells type II (as epithelial cells and high in the side scatter).
RNA isolation, quantitative real-time PCR and luminex assay
Isolation of RNA from biopsy samples from the lung was performed using the RNeasy Fibrous Tissue Mini Kit (Qiagen, Hilden, Germany). Subsequently, 1 μg of RNA underwent reverse transcription using M-MLV (H-) point mutant reverse transcriptase (Promega, Mannheim, Germany). Quantitative real-time analysis was performed with a 7500 Real-Time PCR system (Applied Biosystems, Foster City, CA, USA), using the Fast SYBR Green Master Mix (Life Technologies) and specific primers for IFN-γ (5′-AGG AAC TGG CAA AAG GAT GGT GA-3′ and 5′-TGT TGC TGA TGG CCT GAT TGT CTT-3′), TNF-α (5′ CAA TGC ACA GCC TTC CTC ACA G-3′ and 5′-TAC ATA AAT AAA CCT TCC GGC CC-3′), IP-10 (Interferon gamma-induced protein 10; 5′-CTC TCC ATA CTC CCC TTT ACC C-3′ and 5′-GCT TCG GCA GTT ACT TTT GTC TCA-3′), KC (Keratinocide-derived cytokine; 5′-CAT GGC TGG GAT TCA CCT C-3′ and 5′- CAG ACC ACT TGC GAC CGA A-3′), CCL-2 (5′-CTG TGC TGA CCC CAA GAA GG-3′ and 5′-TAA GGC ATC ACA GTC CGA GTC ACA-3′), NS-1 (5′- CAG GAC ATA CTG ATG AGG ATG-3′ and 5′- GTT TCA GAG ACT CGA ACT GTG-3′) and RPS9 (5′-CTG GAC GAG GGC AAG ATG AAG C-3′ and 5′-TGA CGT TGG CGG ATG AGC ACA-3′). Relative mRNA levels were calculated with included standard curves for each individual gene and further normalization to the housekeeping gene RPS9. Cytokines from the bronchoalveolar lavage fluid were quantified using a Procarta Cytokine assay kit (Panomics, Santa Clara, CA, USA) according to the manufacturer's guidelines. The assay was run with a Luminex 200 system using Luminex IS software (Luminex Corporation, Austin, TX, USA).
In vivo life imaging
Sedated mice received a single intranasal injection (50 μL) of AlexaFluor750-oligonucleotide-loaded CaP/PLGA nanoparticles (5 μg siRNA, corresponds to 1/5*8 = 1.6 mg nanoparticles) prior to imaging using a pipette. Uptake in the lung was examined using an in vivo imaging system Caliper Lumina II (Caliper Life Sciences, Hopkinton, Massachusetts, USA) and images were analyzed using the Living Image 4.2 Software.
Preparation of tissue sections
Lungs were fixed with 4% buffered formalin, embedded in paraffin, sectioned at a thickness of 4 μm, and stained with hematoxylin and eosin. The total histological score was composed of the parameters inflammation (0–5), type II-pneumocyte hyperplasia (0–5), alveolar histiocytosis (0–3) and infiltrating neutrophils (0–3).12
Statistical analysis
Student's t test or one-way ANOVA, followed by Tukey's multiple comparisons test, was used to determine statistical significance, which was set at the level of p < 0.05.
Results
Characterization of calcium phosphate nanoparticles
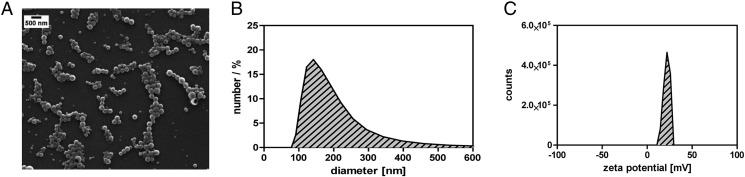
CaP/PLGA nanoparticles were prepared in a multistep process as described previously.9 The CaP core was coated with siRNA or fluorescence-labeled oligonucleotides, encapsulated into PLGA, and finally coated with polyethylenimine (PEI). This composition was previously found by our group being especially potent for the successful delivery and release of nucleic acids to cells at mucosal surfaces.8 The successful synthesis was evaluated by scanning electron microscopy (Figure 1, A ) and dynamic light scattering measurements (Figure 1, B). The final nanoparticles were homogenous and spherical in morphology and exhibited an average diameter of ~145 nm and a zetapotenti of +23 mV (Figure 1, C) confirming the colloidal stability in water.
Figure 1.
Characterization of siRNA-loaded CaP/PLGA NPs. PLGA/CaP NPs is prepared in a multi-step synthesis with a final composition of three layers surrounding the CaP core. The NPs were characterized by scanning electron microscopy (A), dynamic light scattering (B) and zeta potential (C).
CaP/PLGA nanoparticles are taken up by different cell types of the lung
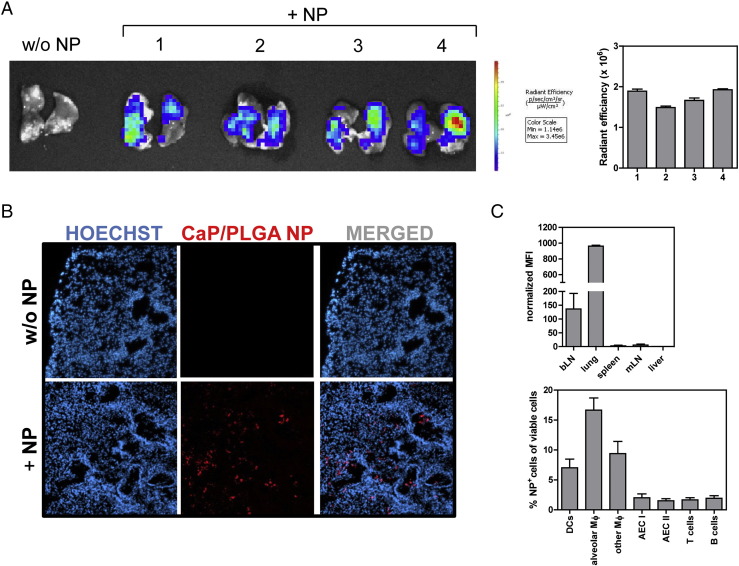
To address if siRNA-loaded CaP/PLGA nanoparticles can be used as local delivery vehicle for pulmonary diseases in vivo, the delivery of these nanoparticles to the lung after nasal instillation was evaluated. Fluorescence-labeled CaP/PLGA nanoparticles were administered to BALB/c mice once, and 1 hour post-administration, a fluorescent signal was detectable in the lungs of mice treated with nanoparticles as demonstrated by in vivo life imaging (Figure 2, A ) as well as fluorescence microscopy of tissue sections (Figure 2, B). Furthermore, the variability of the uptake of nanoparticles between different animals was low, indicating a comparable and reproducible uptake in this experimental setting (Figure 2, A).
Figure 2.
CaP/PLGA NPs are taken up in the lung after nasal instillation. Distribution of fluorescence-labeled oligonucleotide-loaded CaP/PLGA NPs was analyzed 1 h after nasal instillation by IVIS (A), fluorescence microscopy (B) and flow cytometry (C).
To further analyze the general distribution, the uptake of nanoparticles was analyzed one hour after nasal instillation in different organs by flow cytometry. The major part of the CaP/PLGA nanoparticles remained in the lung, while a weak signal was additionally detectable in bronchial lymph nodes (bLN). Almost no signal was measurable in spleen, liver and mesenteric lymph nodes (Figure 2, C), indicating a local rather than a systemic distribution. Further flow cytometric analysis of the lung demonstrated that macrophages and dendritic cells were the main cell populations to internalize the functionalized CaP/PLGA nanoparticles, while only very few epithelial cells and lymphocytes endocytosed the nanoparticles (Figure 2, C). These results confirm the local uptake of functionalized CaP/PLGA nanoparticles by the lung in vivo after nasal instillation, diminishing the possibility of side effects.
Establishment of TH1-induced lung inflammation
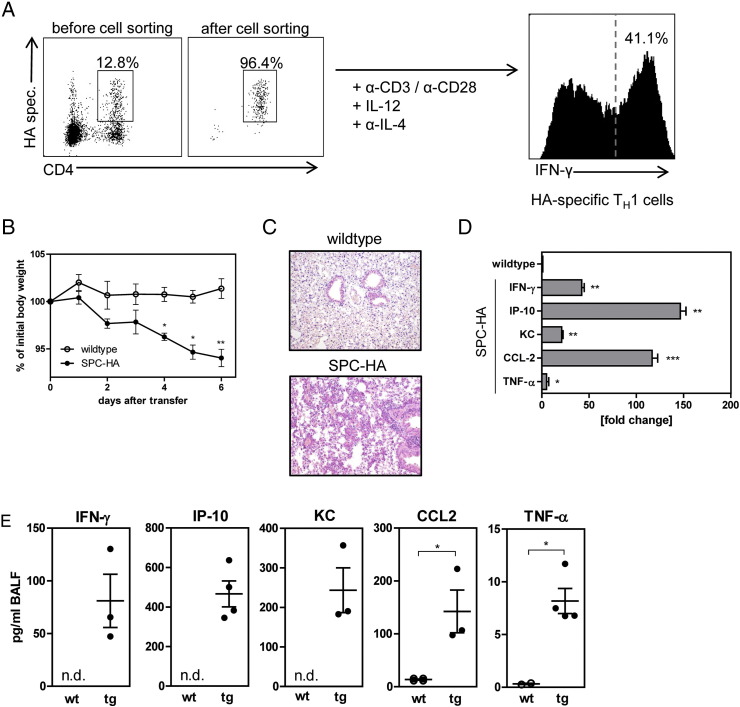
To analyze the therapeutic approach using siRNA-loaded CaP/PLGA nanoparticles and determine potential target genes, a TH1-mediated model of pulmonary inflammation was used. T cell receptor (TCR) transgenic HA-specific CD4+ T cells from TCR-HA transgenic mice were sorted and differentiated into TH1 cells in vitro. The differentiation efficiency was determined on day 6 by analysis of the IFN-γ production (Figure 3, A ). Approximately 35–45% of the CD4+ T cells produced IFN-γ confirming a TH1 phenotype. After the differentiation, 5 × 105 HA-specific TH1 cells were transferred into SPC-HA transgenic mice, which express influenza HA under the control of the specific alveolar epithelial cell promoter surfactant protein C, and mice were monitored daily for signs of sickness by measuring the body weight. Compared to non-transgenic littermates, transgenic mice started to lose weight from day 2 onwards, reaching a weight loss ~10% on day 6 (Figure 3, B). On day 6, the lungs were analyzed histopathologically and revealed an inflammation in transgenic mice with a destruction of the lung structure compared to non-transgenic mice, which exhibited no pathologic alterations (Figure 3, C).
Figure 3.
Establishment and characterization of TH1 cell induced lung inflammation. HA-specific CD4+ T cells from the spleens of TCR-HA transgenic mice were sorted by flow cytometry and differentiated into TH1 cells in vitro, success was validated by intracellular staining of IFN-γ on day 6 (A). Exemplary dot plots are shown. 5 × 105 HA-specific TH1 cells were adoptively transferred into SPC-HA transgenic mice and non-transgenic littermates. Severity of inflammation was assessed by weight loss (B) and histology on day 6 (C). Expression of pro-inflammatory cytokines and chemokines was assessed by real-time PCR (D) and Luminex analysis (E). Statistical analysis was performed using Student's t-test (*p < 0.05; **p < 0.01; ***p < 0.001; n.d., not detectable).
Cytokine and chemokine signaling plays an important role during pulmonary infection and inflammation. For instance, during COPD, several cytokines have been reported to be elevated in the fluid obtained from bronchoalveolar lavages or biopsies from the lung.13 To determine target genes for the siRNA treatment in this disease model, biopsies from the lung were analyzed for the expression of several cytokines. Diseased SPC-HA transgenic mice expressed significantly increased levels of IFN-γ, IP-10, KC, CCL-2 and TNF-α compared to healthy littermates after transfer of TH1 cells (Figure 3, D). This elevated expression was confirmed on protein level in the fluid obtained from bronchoalveolar lavages (Figure 3, E). The strongest increase compared to non-transgenic mice was detectable in the expression of IP-10 on mRNA and protein level, but also CCL-2, TNF-α, KC and IFN-γ were significantly elevated in mice suffering from TH1-induced pulmonary inflammation. The most prominent expression on mRNA level was found for IP-10, CCL-2, and IFN-γ, therefore, those genes were chosen as therapeutic targets in this disease model.
Treatment with siRNA-loaded CaP/PLGA nanoparticles abrogates pulmonary inflammation
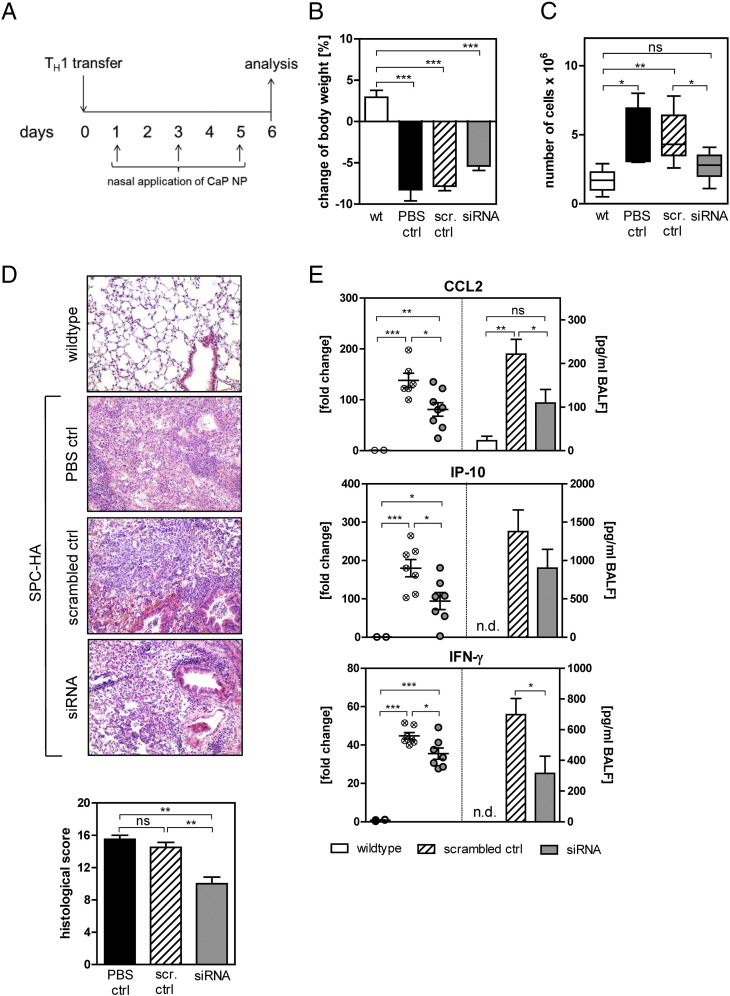
To examine the therapeutic potential of siRNA-loaded CaP/PLGA nanoparticles, SPC-HA mice were treated with a mixture of siRNAs or scrambled siRNA-loaded nanoparticles on days 1, 3 and 5 after TH1 cell transfer (Figure 4, A ). During the treatment, the weight loss was monitored daily. While non-transgenic mice did not lose any weight after the cell transfer, SPC-HA transgenic mice which were treated with scrambled siRNA-loaded nanoparticles or PBS exhibited a weight loss of 7% by day 6. Interestingly, the transgenic mice treated with specific siRNAs lost less weight than the animals of the control groups (Figure 4, B). Mice were sacrificed on day 6 and further analyzed. Importantly, mice treated with siRNA-loaded CaP/PLGA nanoparticles had a significantly reduced number of cells within the BAL fluid compared to scrambled control or PBS treated mice, indicating a less severe inflammation, as patients suffering from pulmonary inflammation often exhibit an influx of immune cells due to increased chemokine production14 (Figure 4, C). Furthermore, the lung tissue of mice treated with siRNA against CCL-2, IP-10 and IFN-γ was less severely inflamed than tissue from control animals. Less alveolar epithelial cell type II hyperplasia, a reduced accumulation of alveolar macrophages and a decreased number of neutrophils in the interstitium were found in siRNA-treated mice. This is also reflected in the significantly reduced histological score of the lung tissue (Figure 4, D). Finally, to validate if the potential beneficial effect of the siRNA-loaded CaP/PLGA nanoparticle treatment is due to the decreased expression of the target genes, biopsies of the lung and fluid from the BAL were analyzed for gene expression. Indeed, the expression of all three target genes was significantly decreased on mRNA and protein level (Figure 4, E). The expression of CCL-2 was reduced by 40% compared to the scrambled control treated animals on both, mRNA and protein level. The IP-10 expression in lung tissue was decreased by 50% on mRNA level while the down-regulation on protein level was less prominent but nevertheless reduced by 25%. The IFN-γ production was also significantly diminished by 55% on protein level. In summary, these results indicate a beneficial therapeutic effect of siRNA-loaded CaP/PLGA nanoparticles for inflammation of the lung.
Figure 4.
Treatment with PLGA/CaP NPs reduces lung inflammation. During induction of inflammation, mice were treated with siRNA-loaded CaP/PLGA NPs directed against CCL2, IP-10 and IFN-γ by nasal instillation on days 1, 3 and 5 (A). On day 6, mice were analyzed for weight loss (B), and the infiltration of cells into the lung (C). The histopathology in the lung was analyzed (D) and expression levels of target genes in lung tissue or BALF were measured by qPCR and Luminex assay (E). Data from two independent experiments are shown. Statistical analysis was performed with one-way ANOVA followed by Tukey's multiple comparison test or Student's t-test (*p < 0.05; **p < 0.01; ***p < 0.001; ns, not significant).
Treatment with siRNA-loaded CaP/PLGA nanoparticles reduces viral lung infection
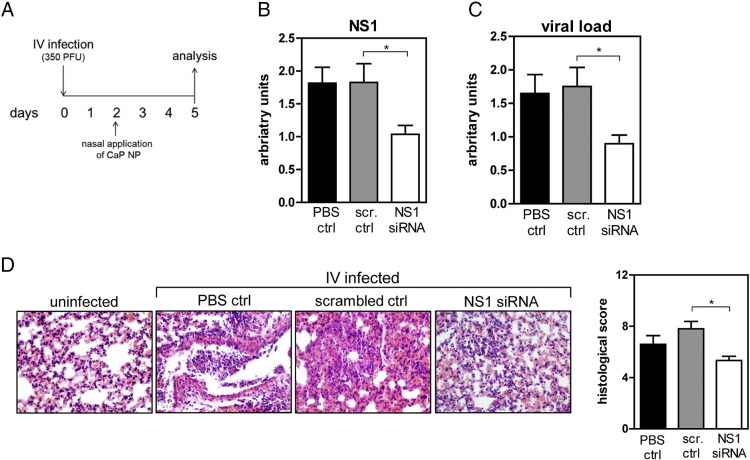
In addition to inflammatory diseases the lung is prone to viral infections. While for some viral infections vaccines are available, other diseases like the severe acute respiratory syndrome caused by a coronavirus are not curable and a vaccine does not exist.15 A therapeutic rather than a prophylactic approach to treat viral infections is the use of siRNA to interfere in the reproduction of the virus and inhibit its spreading. To verify if this interference is a possibility to reduce the viral load, CaP/PLGA nanoparticles were loaded with siRNA directed against the non-structural protein 1 (NS1) of the influenza virus. BALB/c mice were infected with the influenza virus and treated with siRNA-loaded nanoparticles two days post infection and analyzed for signs of sickness on day 5 (Figure 5, A ). To verify that the viral protein expression is altered due to the treatment with siRNA-loaded nanoparticles, the NS-1 expression was analyzed in the lung tissue. Indeed, the expression of NS-1 in the lung of infected mice which received siRNA-loaded CaP/PLGA nanoparticle was significantly decreased (Figure 5, B). In consequence, also the viral load was significantly reduced in the lung on day 5 (Figure 5, C). The histopathological analysis of the lung tissue revealed less structural damages and less infiltrated cells in mice treated with siRNA against NS1 compared to mice treated with scrambled siRNA-loaded CaP/PLGA nanoparticles or PBS. The histological score was significantly decreased by almost half in mice which received NS1 siRNA treatment (Figure 5, D). In summary, these results illustrate a promising therapeutic approach against viral infections.
Figure 5.
siRNA-loaded PLGA/CaP NPs directed against viral NS1 dampen influenza infection. Following influenza infection, mice were treated with siRNA-loaded CaP/PLGA NPs directed against NS1 (A). On day 5 post infection, lung tissues were analyzed for the expression of NS1 (B) and viral load (C) qPCR and histopathology (D) was performed. Data from three independent experiments are shown. Statistical analysis was performed by Student's t-test (*p < 0.05).
Discussion
Inflammatory conditions of the lung are widespread and heterogeneous, including interstitial lung disease (ILD), chronic obstructive pulmonary disease (COPD) and asthma. They are characterized by airflow limitation, emphysema, fibrosis or bronchitis. Despite ongoing research, currently available therapies, mainly inhalers, aim to minimize symptoms and aggravations but are not curative. Inhalers open the airway muscles by relaxation or interfere in the activation of the immune system but are unspecific. Therefore, medications to treat the underlying inflammation or infection might be an improved and promising treatment strategy. Here, we have used an inflammatory model based on CD4+ TH1 lymphocytes which are described to be important effectors of pulmonary injury, especially in ILD.16 Many diseases, including ILD and COPD, are caused by excessive, incorrect activity of genes; the ability of siRNA to modulate gene expression specifically on a molecular level offers a suitable approach for interference. Here, we used a sterile model of lung inflammation to analyze the potency of local modification of gene expression by siRNA. SiRNA directed against CCL-2, IP-10 and IFN-γ was used to reduce gene expression in inflamed lung tissue, and siRNA directed against NS-1 of the influenza virus itself to diminish viral replication. To exert its function, siRNA must be delivered to the target tissue; here, we demonstrated that CaP/PLGA nanoparticles can successfully be used as delivery vehicle to the lung tissue. The nanoparticles protect the siRNA from the environment and can be used to decrease gene expression and thereby abrogate inflammatory responses in the lung.
Calcium phosphate has previously been described as a safe and biocompatible biomedical carrier due to its natural occurrence in mammalian hard tissues.7 In this study, we have used CaP nanoparticles encapsulated within PLGA, a safe and biodegradable polymer used as pharmaceutic delivery vehicle,17 to deliver siRNA safely into target cells. PEI, a different polymer, was applied to the surface for an increased cellular uptake and endosomal escape.18 Alongside an increased uptake efficiency, PEI was reported to induce inflammatory responses in the lung19; however, in this study, no inflammatory effects were detected. This might be due to the highest uptake rate by macrophages in this study, while a study by Beyerle and colleagues demonstrated that PEI mainly affects alveolar epithelial cells.20 Other reports highlighted that the drug delivery to alveolar macrophages has been shown to be most efficient with particles in the low micrometer range,21, 22 while smaller particles have been suggested to have a toxic effect. This effect might be due to the distribution of the nanoparticles beyond the lung into the blood and brain. However, not all particles in the nanoscale behave the same, but their properties depend on the composition. Other publications noticed only a minimal translocation of nanoparticles applied to the lung.23, 24 Furthermore, compared to larger particles, particles in the nanoscale can target a variety of cells and not only macrophages,25, 26 resulting in an increased uptake and delivery of active compound. In previous publications, we have shown that this composition of nanoparticles is highly efficient to achieve an increased ability of gene silencing per nanoparticle as well as an efficient uptake and down-regulation of target genes at mucosal surfaces.8, 9
Patients suffering from acute or chronic lung disorders have increased levels of several chemokines and cytokines in sputum samples, blood or the fluid obtained from BALs.27 In our model, we regulated the expression of IFN-γ, CCL-2 and IP-10 to achieve a decreased inflammation of the lung. IFN-γ was found to be elevated in lung biopsies of patients suffering from COPD,28 and Wang and colleagues demonstrated the importance of IFN-γ in lung related diseases, as IFN-γ overexpressing mice develop pulmonary emphysema.29 In response to IFN-γ, IP-10 is secreted by several cell types and functions as chemoattractant for natural killer cells and macrophages. IP-10 is elevated in blood and sputum samples of COPD patients, and increased levels were found to correlate with disease severity.13 Similar to IP-10, CCL-2 functions as chemoattractant and is elevated in sputum samples of patients with inflammatory lung conditions.27 Studies regarding the function of CCL-2 or its receptor, CCR2, hardly exist. However, a murine antagonist against CCR2 was successfully applied to reduce influenza virus induced lung pathology.30 The treatment with CaP/PLGA nanoparticles loaded with siRNA against these three target genes led to decreased levels of gene expression in the TH1-induced model of lung inflammation, thus we have indirectly shown the successful delivery of siRNA and its release to the cytoplasm into cells from the lung, after nasal instillation. The knockdown of all three mRNAs was sufficient to decrease protein levels of the target genes and most importantly, to restrict the pulmonary inflammation. So far, biological agents have not proven successful in clinical trials for inflammatory conditions of the lung31, 32; one reason might be the different phenotypes and varying symptoms between patients. Furthermore, the inflammatory response is likely not driven by only one mediator but may be attributed to several dysregulated genes. The siRNA-loaded CaP/PLGA nanoparticles have turned out as a promising approach, as they are easily adaptable to individual patients and can target several overexpressed genes at once.
A further interesting field of application for siRNA-loaded CaP/PLGA nanoparticles is the application against lung infections by viruses. While flu caused by the influenza virus is rarely dangerous due to effective immunization, several viral infections exist with no potent vaccine available. A therapeutic rather than a prophylactic approach against viral infections might be the knockdown of viral gene expression themselves. As proof-of-principle experiment, we used siRNA-loaded CaP/PLGA nanoparticles directed against NS-1 of the influenza virus and demonstrated a reduced viral load and pathology. To our knowledge, this is the first study to use siRNA against NS-1 as a therapeutic treatment and to apply the nanoparticles locally to the lung. A previous study only exhibited the beneficial effect of intravenous, prophylactic treatment with siRNA against influenza viral infection.33
In summary, we were able to use siRNA-loaded CaP/PLGA nanoparticles covered with PEI directed against IFN-γ, IP-10 and CCL-2 to achieve a specific knockdown of the target gene expression in vivo by RNA interference. By multiple nasal instillations, we were able to diminish pulmonary inflammation caused by TH1 cells. Additionally, we demonstrated the beneficial effect of RNA interference against viral genes as therapeutic treatment. In summary, our data provide a promising approach for the specific delivery of siRNA to the lung and the treatment of pulmonary inflammation.
Footnotes
Conflict of interest: The authors declare that no conflict of interest exists.
Funding: Deutsche Forschungsgemeinschaft (EP22/35-1 and WE4472/5-1, GRK1949/1).
References
- 1.Mannino D.M., Buist A.S., Petty T.L., Enright P.L., Redd S.C. Lung function and mortality in the United States: data from the first National Health and nutrition examination survey follow up study. Thorax. 2003;58:388–393. doi: 10.1136/thorax.58.5.388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Murugan V., Peck M.J. Signal transduction pathways linking the activation of alveolar macrophages with the recruitment of neutrophils to lungs in chronic obstructive pulmonary disease. Exp Lung Res. 2009;35:439–485. doi: 10.1080/01902140902759290. [DOI] [PubMed] [Google Scholar]
- 3.Morjaria J.B., Malerba M., Polosa R. Biologic and pharmacologic therapies in clinical development for the inflammatory response in COPD. Drug Discov Today. 2010;15:396–405. doi: 10.1016/j.drudis.2010.03.001. [DOI] [PubMed] [Google Scholar]
- 4.Barnes P.J. Immunology of asthma and chronic obstructive pulmonary disease. Nat Rev Immunol. 2008;8:183–192. doi: 10.1038/nri2254. [DOI] [PubMed] [Google Scholar]
- 5.Hannon G.J. RNA interference. Nature. 2002;418:244–251. doi: 10.1038/418244a. [DOI] [PubMed] [Google Scholar]
- 6.Guo J., Fisher K.A., Darcy R., Cryan J.F., O'Driscoll C. Therapeutic targeting in the silent era: advances in non-viral siRNA delivery. Mol Biosyst. 2010;6:1143–1161. doi: 10.1039/c001050m. [DOI] [PubMed] [Google Scholar]
- 7.Sokolova V., Epple M. Inorganic nanoparticles as carriers of nucleic acids into cells. Angew Chem Int Ed Engl. 2008;47:1382–1395. doi: 10.1002/anie.200703039. [DOI] [PubMed] [Google Scholar]
- 8.Frede A., Neuhaus B., Klopfleisch R., Walker C., Buer J., Muller W. Colonic gene silencing using siRNA-loaded calcium phosphate/PLGA nanoparticles ameliorates intestinal inflammation in vivo. J Control Release. 2016;222:86–96. doi: 10.1016/j.jconrel.2015.12.021. [DOI] [PubMed] [Google Scholar]
- 9.Dördelmann G., Kozlova D., Karczewski S., Lizio R., Knauer S., Epple M. Calcium phosphate increases the encapsulation efficiency of hydrophilic drugs (proteins, nucleic acids) into poly(D,L-lactide-co-glycolide acid) nanoparticles for intracellular delivery. J Mater Chem B. 2014 doi: 10.1039/c4tb00922c. [DOI] [PubMed] [Google Scholar]
- 10.Bruder D., Westendorf A.M., Geffers R., Gruber A.D., Gereke M., Enelow R.I. CD4 T lymphocyte-mediated lung disease: steady state between pathological and tolerogenic immune reactions. Am J Respir Crit Care Med. 2004;170:1145–1152. doi: 10.1164/rccm.200404-464OC. [DOI] [PubMed] [Google Scholar]
- 11.Kirberg J., Baron A., Jakob S., Rolink A., Karjalainen K., von Boehmer H. Thymic selection of CD8+ single positive cells with a class II major histocompatibility complex-restricted receptor. J Exp Med. 1994;180:25–34. doi: 10.1084/jem.180.1.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Klopfleisch R. Multiparametric and semiquantitative scoring systems for the evaluation of mouse model histopathology--a systematic review. BMC Vet Res. 2013;9:123. doi: 10.1186/1746-6148-9-123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tangedal S., Aanerud M., Persson L.J., Brokstad K.A., Bakke P.S., Eagan T.M. Comparison of inflammatory markers in induced and spontaneous sputum in a cohort of COPD patients. Respir Res. 2014;15:138. doi: 10.1186/s12931-014-0138-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Di S.A., Caramori G., Ricciardolo F.L., Capelli A., Adcock I.M., Donner C.F. Cellular and molecular mechanisms in chronic obstructive pulmonary disease: an overview. Clin Exp Allergy. 2004;34:1156–1167. doi: 10.1111/j.1365-2222.2004.02030.x. [DOI] [PubMed] [Google Scholar]
- 15.Fehr A.R., Perlman S. Coronaviruses: an overview of their replication and pathogenesis. Methods Mol Biol. 2015;1282:1–23. doi: 10.1007/978-1-4939-2438-7_1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Semenzato G., Bortoli M., Agostini C. Applied clinical immunology in sarcoidosis. Curr Opin Pulm Med. 2002;8:441–444. doi: 10.1097/00063198-200209000-00017. [DOI] [PubMed] [Google Scholar]
- 17.Danhier F., Ansorena E., Silva J.M., Coco R., Le B.A., Preat V. PLGA-based nanoparticles: an overview of biomedical applications. J Control Release. 2012;161:505–522. doi: 10.1016/j.jconrel.2012.01.043. [DOI] [PubMed] [Google Scholar]
- 18.Boussif O., Lezoualc'h F., Zanta M.A., Mergny M.D., Scherman D., Demeneix B. A versatile vector for gene and oligonucleotide transfer into cells in culture and in vivo: polyethylenimine. Proc Natl Acad Sci U S A. 1995;92:7297–7301. doi: 10.1073/pnas.92.16.7297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gautam A., Densmore C.L., Waldrep J.C. Pulmonary cytokine responses associated with PEI-DNA aerosol gene therapy. Gene Ther. 2001;8:254–257. doi: 10.1038/sj.gt.3301369. [DOI] [PubMed] [Google Scholar]
- 20.Beyerle A., Hobel S., Czubayko F., Schulz H., Kissel T., Aigner A. In vitro cytotoxic and immunomodulatory profiling of low molecular weight polyethylenimines for pulmonary application. Toxicol In Vitro. 2009;23:500–508. doi: 10.1016/j.tiv.2009.01.001. [DOI] [PubMed] [Google Scholar]
- 21.Ahsan F., Rivas I.P., Khan M.A., Torres Suarez A.I. Targeting to macrophages: role of physicochemical properties of particulate carriers--liposomes and microspheres--on the phagocytosis by macrophages. J Control Release. 2002;79:29–40. doi: 10.1016/s0168-3659(01)00549-1. [DOI] [PubMed] [Google Scholar]
- 22.Lawlor C., O'Sullivan M.P., Sivadas N., O'Leary S., Gallagher P.J., Keane J. The application of high-content analysis in the study of targeted particulate delivery systems for intracellular drug delivery to alveolar macrophages. Mol Pharm. 2011;8:1100–1112. doi: 10.1021/mp1004178. [DOI] [PubMed] [Google Scholar]
- 23.Kreyling W.G., Semmler M., Erbe F., Mayer P., Takenaka S., Schulz H. Translocation of ultrafine insoluble iridium particles from lung epithelium to extrapulmonary organs is size dependent but very low. J Toxicol Environ Health A. 2002;65:1513–1530. doi: 10.1080/00984100290071649. [DOI] [PubMed] [Google Scholar]
- 24.Takenaka S., Karg E., Kreyling W.G., Lentner B., Möller W., Behnke-Semmler M. Distribution pattern of inhaled ultrafine gold particles in the rat lung. Inhal Toxicol. 2006;18:733–740. doi: 10.1080/08958370600748281. [DOI] [PubMed] [Google Scholar]
- 25.Davda J., Labhasetwar V. Characterization of nanoparticle uptake by endothelial cells. Int J Pharm. 2002;233:51–59. doi: 10.1016/s0378-5173(01)00923-1. [DOI] [PubMed] [Google Scholar]
- 26.Foster K.A., Yazdanian M., Audus K.L. Microparticulate uptake mechanisms of in-vitro cell culture models of the respiratory epithelium. J Pharm Pharmacol. 2001;53:57–66. doi: 10.1211/0022357011775190. [DOI] [PubMed] [Google Scholar]
- 27.Traves S.L., Culpitt S.V., Russell R.E., Barnes P.J., Donnelly L.E. Increased levels of the chemokines GROalpha and MCP-1 in sputum samples from patients with COPD. Thorax. 2002;57:590–595. doi: 10.1136/thorax.57.7.590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Di S.A., Caramori G., Capelli A., Gnemmi I., Ricciardolo F.L., Oates T. STAT4 activation in smokers and patients with chronic obstructive pulmonary disease. Eur Respir J. 2004;24:78–85. doi: 10.1183/09031936.04.00080303. [DOI] [PubMed] [Google Scholar]
- 29.Wang Z., Zheng T., Zhu Z., Homer R.J., Riese R.J., Chapman H.A. Interferon gamma induction of pulmonary emphysema in the adult murine lung. J Exp Med. 2000;192:1587–1600. doi: 10.1084/jem.192.11.1587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lin K.L., Sweeney S., Kang B.D., Ramsburg E., Gunn M.D. CCR2-antagonist prophylaxis reduces pulmonary immune pathology and markedly improves survival during influenza infection. J Immunol. 2011;186:508–515. doi: 10.4049/jimmunol.1001002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mahler D.A., Huang S., Tabrizi M., Bell G.M. Efficacy and safety of a monoclonal antibody recognizing interleukin-8 in COPD: a pilot study. Chest. 2004;126:926–934. doi: 10.1378/chest.126.3.926. [DOI] [PubMed] [Google Scholar]
- 32.Rennard S.I., Fogarty C., Kelsen S., Long W., Ramsdell J., Allison J. The safety and efficacy of infliximab in moderate to severe chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2007;175:926–934. doi: 10.1164/rccm.200607-995OC. [DOI] [PubMed] [Google Scholar]
- 33.Ge Q., Filip L., Bai A., Nguyen T., Eisen H.N., Chen J. Inhibition of influenza virus production in virus-infected mice by RNA interference. Proc Natl Acad Sci U S A. 2004;101:8676–8681. doi: 10.1073/pnas.0402486101. [DOI] [PMC free article] [PubMed] [Google Scholar]