Abstract
Rotavirus A (RVA) Alp11B was detected from a neonatal Peruvian alpaca presenting with diarrhea, and the Alp11B VP7, VP4, VP6, NSP4, and NSP5 genes were sequenced. The partial genotype constellation of this strain, RVA/Alpaca-wt/PER/Alp11B/2010, was determined to be G35-P[50]-I13-E16-H6.
Keywords: Rotavirus, Alpaca, Camelid, Genotyping
Highlights
-
•
Genomic constellation of strain Alp11B from alpaca
-
•
Novel rotavirus A VP4 and VP7 genotypes
-
•
Camelids rotavirus strains
Rotaviruses (RV) are non-enveloped double-stranded RNA viruses in the Reoviridae family and in the Rotavirus genus, and are classified into eight species (A-H) and two candidate species (I and J) (Matthijnssens et al., 2012, Mihalov-Kovács et al., 2015, Bányai et al., 2017). Species A rotavirus (RVA) is a major cause of dehydrating diarrhea in humans and animals worldwide (Estes and Greenberg, 2013). The RVA genome consists of 11 segments of double-stranded RNA (dsRNA) encoding six structural proteins (VP1–4, VP6, and VP7) and five or six nonstructural proteins (NSP1-NSP5/6) depending on the strain (Estes and Greenberg, 2013). The RVA genomic classification nomenclature is based on all 11 segments of dsRNA (Matthijnssens et al., 2008, Matthijnssens et al., 2011). Currently, there are 35 VP7 (G), 50 VP4 (P), 26 VP6 (I), 21 VP1 (R), 19 VP2 (C), 19 VP3 (M), 30 NSP1 (A), 21 NSP2 (N), 21 NSP3 (T), 27 NSP4 (E), and 21 NSP5/6 (H) genotypes (Rotavirus Classification Workgroup (RCWG), 2016). In this study, we described two new RVA VP4 and VP7 genotypes in strain RVA/Alpaca-wt/PER/Alp11B/2010 from a Peruvian alpaca.
During the first week of February 2010, a diarrhea outbreak occurred that resulted in high rates of morbidity and mortality among neonatal alpacas in a community in Silli, Peru (Rojas et al., 2016a). This community is located in the southern highlands of Peru (14°24′45.3″S, 71°11′32.6″ W; ~ 4000 m above sea level) in the province of Canchis in the state of Cusco. The animals were subjected to postmortem examinations at the Laboratory of Histology, Embryology and Veterinary Pathology, Universidad Nacional Mayor de San Marcos, Peru. Intestinal lavage samples were obtained during necropsy by thoroughly washing the intestines with warm water, and then these samples were analyzed for E. coli, Clostridium ssp. Eimeria spp., Cryptosporidium spp., coronavirus and RVA. The sample Alp11b was positive only for RVA (Rojas et al., 2016a). The importation of these alpaca specimens was approved by the Brazilian Institute of Environment (IBAMA; Brasília, DF, Brazil; license 14BR012948/DF 02/20/2014).
For RVA detection the lavage samples were diluted in 10% v/v using sterile phosphate-buffered saline and clarified by low speed centrifugation at 2500g for 5 min. Total RNA was extracted from 300 μL of the supernatant using the Totally RNA® Kit, according to the manufacturer's instructions (Applied Biosystems/Ambion, Austin, USA). RVA detection was performed by RT-PCR with primers that amplified a small conserved portion of the VP6 gene (Rojas et al., 2016b). Once RVA was detected, the sample was subjected to additional RT-PCR amplifications to identify the VP4, VP7, VP6, NSP4, and NSP5 genotypes using specific primers (Appendix) that were previously published or designed based on RVA sequences available in GenBank. Overlapping sequences were assembled and edited using SeqMan, EditSeq, and MegAlign in the Lasergene software package (DNASTAR, Madison, WI).
Phylogenetic analysis was performed with MEGA software version 7.0.14 (Kumar et al., 2016). Dendrograms were constructed using the Maximum Likelihood method based on the Kimura two-parameter model. The Kimura two-parameter was chosen using the Find Best DNA/Protein Model tool on the MEGA software. Statistical significance was estimated by bootstrap analysis with 1000 pseudoreplicates. The sequences of our strain were compared to the sequences of the RVA strains obtained from GenBank. Genotypes were assigned to each genome segment by the web-based automated rotavirus genotyping tool RotaC (http://rotac.regatools.be) (Maes et al., 2009). Sequences of Alp11B strain were aligned and compared to that of each corresponding gene of RVA strains obtained from GenBank, by using MegAlign, which are available in the Lasergene software package (DNASTAR, Madison, WI). Multiple alignments were done by using the complete open reading frame (ORF) for each gene segment. Nucleotide and amino acid identities were determined with the MegAlign p-distance algorithm. Sequences generated in this study were deposited into GenBank under accession numbers KM276820, KM276822, KY971955, KY971977 and KY972004.
The genotypes of the VP6, NSP4, and NSP5 genes in Alp11B were identified as I13, E16 and H6, respectively. The VP6 gene was closely related to the human strain Ecu534 (88.5% nucleotide identity). The NSP4 gene was closely related to the vicuña strain C75 (91.1% nucleotide identity), and the NSP5 gene was 100% identical to strain SA44, which is also from a Peruvian alpaca, and closely related to the bat strain 4754 (96.4% nucleotide identity) (Fig. 1 ).
Fig. 1.
Phylogenetic trees constructed from partial nucleotide sequences of VP6, NSP4, and NSP5 genes of RVA strain RVA/Alpaca-wt/PER/Alp11B/2010/G35P[50] and other RVA strains. Distances were corrected with the Kimura two-parameter model. Phylogenetic trees were constructed by the Maximum Likelihood method. Statistical support was provided by bootstrapping 1000 pseudoreplicates. Bootstrap values above 75% are given at branch nodes. The distance scale is in substitutions/site. Black triangles indicate Alp11B genes.
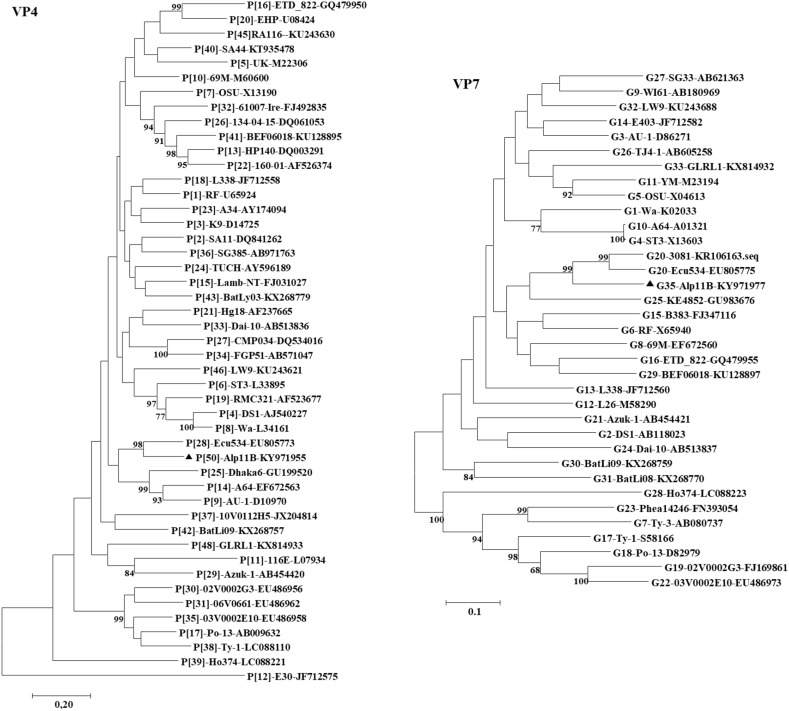
The nucleotide sequences of the VP4 and VP7 genes from Alp11B were not related to any RVA strain available in GenBank (Fig. 2 ). The highest nucleotide identity found for the VP4 gene was 76.7% with the human strain Ecu534, and the highest nucleotide identity for the VP7 sequence was 79.8% also with strain Ecu534. Both of these identity values were below the 80% cutoff value proposed by the RCWG (Matthijnssens et al., 2008). The VP4 and VP7 nucleotide sequences were submitted to the RCWG for further analysis and were assigned novel P[50] and G35 genotypes, respectively. However, the VP7 sequence was a borderline new genotype, and it would be possible that there might be some cross reactivity at the serological level with G20.
Fig. 2.
Maximum likelihood phylogenetic trees based on nucleotide sequences of the full-length VP4 and VP7 ORFs. Distances were corrected with the Kimura two-parameter model. Bootstrap statistics show genetic relationships between the Peruvian alpaca strain RVA/Alpaca-wt/PER/Alp11B/2010/G35P[50] and known human and animal rotavirus strains obtained from GenBank. Bootstrap values above 75% are given at branch nodes. The distance scale is in substitutions/site. Black triangles indicate Alp11B genes.
Few South American camelid (SAC) RVA strains have been characterized, but those that have showed great genotype diversity. The vicuña strain RVA/vicuña-wt/ARG/C75/2010/G8P[14] (Badaracco et al., 2013) and the guanaco strains RVA/Guanaco-wt/ARG/Chubut/1999/G8P[14] and RVA/Guanaco-wt/ARG/RíoNegro/1998/G8P[1] possess a bovine-like genome constellation, G8-P[1/14]-I2 (Matthijnssens et al., 2009). The guanaco strains also possess E12 and H3 NSP4 and NSP5 genotypes, respectively. The vicuña strain possesses the unique NSP4-E16 genotype, but the NSP5 gene was not characterized. In contrast, the VP4 and VP7 genes of the alpaca strains RVA/Alpaca-tc/Per/K′ayra/3368-10/2011/G3P[11] and RVA/Alpaca-tc/Per/K′ayra/3386-10/2011/G3P[x] showed 75% and 96% identity to porcine and human RVA strains suggesting that they resulted from interspecies transmission (Garmendia et al., 2015), but their NSP4 and NSP5 genes have not been characterized. Only one alpaca strain has been fully characterized, RVA/Alpaca-tc/PER/SA44/2014, and it bears the unique constellation G3-P[40]-I8-R3-C3-M3-A9-N3-T3-E3-H6. This unique genetic makeup suggests that strain SA44 emerged from multiple reassortment events between bat, equine, and human-like RVA strains (Rojas et al., 2016b).
Strain Alp11B and strain SA44 were detected from the same location in Peru, and Alp11B also has a unique genetic constellation, G35-P[50]-E16-H6 with high identity to camelid, bat, and human-like RVA strains. The NSP4-E16 genotype was also found in vicuña strain C75 from a camelid in Argentina, thus it could be a common genotype in camelids. The NSP5-H6 genotype of Alp11B was related to alpaca strain SA44 and bat strain 4754, but VP6-I13 is a rare genotype that has only been found in the human strain Ecu534, which was detected in Ecuador in 2006 (Solberg et al., 2009). The VP4 and VP7 genes Alp11B represent two new genotypes with the highest identities to the Ecu534 strain. Because of limited sample, the entire genome constellation of Alp11B could not be characterized and the origin of strain Alp11B remains unclear. Moreover, because the sequences were obtained directly from the clinical sample we could not exclude the possibility of mixed infection with two or more RVA strains in the sample.
Acknowledgments
Acknowledgements
This study was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, grant number 303864/2014-1), and the Fundação Carlos Chagas de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ, grant number E-26/201.374/2014), Brazil. The funders were not involved in the study design, data collection, data interpretation, or the decision to submit the work for publication. We thank Soluza dos Santos Gonçalves for technical assistance.
Soluza dos Santos Gonçalves is a recipient of a fellowship from FAPERJ E-26/202.625/2016.
Conflicts of interest
The authors declare that there are no conflicts of interest.
Footnotes
Supplementary data to this article can be found online at http://dx.doi.org/10.1016/j.meegid.2017.08.019.
Appendix A. Supplementary data
Supplementary material
References
- Badaracco A., Matthijnssens J., Romero S., Heylen E., Zeller M., Garaicoechea L., Van Ranst M., Parreño V. Discovery and molecular characterization of a group A rotavirus strain detected in an Argentinean vicuña (Vicugna vicugna) Vet. Microbiol. 2013;161:247–254. doi: 10.1016/j.vetmic.2012.07.035. http://dx.doi.org/10.1016/j.vetmic.2012.07.035 [DOI] [PubMed] [Google Scholar]
- Bányai K., Kemenesi G., Budinski I., Földes F., Zana B., Marton S., Varga-Kugler R., Oldal M., Kurucz K., Jakab F. Candidate new rotavirus species in Schreiber's bats, Serbia. Infect. Genet. Evol. 2017;48:19–26. doi: 10.1016/j.meegid.2016.12.002. http://dx.doi.org/10.1016/j.meegid.2016.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Estes M.K., Greenberg H.B. Rotaviruses. In: Knipe D.M., Howley P.M., Cohen J.I., Griffin D.E., Lamb R.A., Martin M.A., Racaniello V.R., Rizman B., editors. Fields Virology. 6th edition. Vol 1. Wolters Kluver Health/Lippincott, Williams and Wilkins; Philadelphia: 2013. pp. 1347–1401. [Google Scholar]
- Garmendia A.E., Lopez W., Ortega N., Chamorro M.J. Molecular characterization of rotavirus isolated from alpaca (Vicugna pacos) crias with diarrhea in the Andean Region of Cusco, Peru. Vet. Microbiol. 2015;180:109–112. doi: 10.1016/j.vetmic.2015.07.022. http://dx.doi.org/10.1016/j.vetmic.2015.07.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar S., Stecher G., Tamura K. MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016;33:1870–1874. doi: 10.1093/molbev/msw054. http://dx.doi.org/10.1093/molbev/msw054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maes P., Matthijnssens J., Rahman M., Van Ranst M. RotaC: a web-based tool for the complete genome classification of group A rotaviruses. BMC Microbiol. 2009;9:238. doi: 10.1186/1471-2180-9-238. http://dx.doi.org/10.1186/1471-2180-9-238 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matthijnssens J., Ciarlet M., Rahman M., Attoui H., Bányai K., Estes M.K., Gentsch J.R., Iturriza-Gómara M., Kirkwood C.D., Martella V., Mertens P.P., Nakagomi O., Patton J.T., Ruggeri F.M., Saif L.J., Santos N., Steyer A., Taniguchi K., Desselberger U., Van Ranst M. Recommendations for the classification of group A rotaviruses using all 11 genomic RNA segments. Arch. Virol. 2008;153:1621–1629. doi: 10.1007/s00705-008-0155-1. http://dx.doi.org/10.1007/s00705-008-0155-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matthijnssens J., Potgieter C.A., Ciarlet M., Parreño V., Martella V., Bányai K., Garaicoechea L., Palombo E.A., Novo L., Zeller M., Arista S., Gerna G., Rahman M., Van Ranst M. Are human P[14] rotavirus strains the result of interspecies transmissions from sheep or other ungulates that belong to the mammalian order Artiodactyla? J. Virol. 2009;83:2917–2929. doi: 10.1128/JVI.02246-08. http://dx.doi.org/10.1128/JVI.02246-08 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matthijnssens J., Ciarlet M., McDonald S.M., Attoui H., Bányai K., Brister J.R., Buesa J., Esona M.D., Estes M.K., Gentsch J.R., Iturriza-Gómara M., Johne R., Kirkwood C.D., Martella V., Mertens P.P., Nakagomi O., Parreño V., Rahman M., Ruggeri F.M., Saif L.J., Santos N., Steyer A., Taniguchi K., Patton J.T., Desselberger U., Van Ranst M. Uniformity of rotavirus strain nomenclature proposed by the Rotavirus Classification Working Group (RCWG) Arch. Virol. 2011;156:1397–1413. doi: 10.1007/s00705-011-1006-z. http://dx.doi.org/10.1007/s00705-011-1006-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matthijnssens J., Otto P.H., Ciarlet M., Desselberger U., Van Ranst M., Johne R. VP6-sequence-based cutoff values as a criterion for rotavirus species demarcation. Arch. Virol. 2012;157:1177–1182. doi: 10.1007/s00705-012-1273-3. http://dx.doi.org/10.1007/s00705-012-1273-3 [DOI] [PubMed] [Google Scholar]
- Mihalov-Kovács E., Gellért Á., Marton S., Farkas S.L., Fehér E., Oldal M., Jakab F., Martella V., Bányai K. Candidate new rotavirus species in sheltered dogs, Hungary. Emerg. Infect. Dis. 2015;21:660–663. doi: 10.3201/eid2104.141370. http://dx.doi.org/10.3201/eid2104.141370 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rojas M., Gonçalves J.L., Dias H.G., Manchego A., Pezo D., Santos N. Whole-genome characterization of a Peruvian alpaca rotavirus isolate expressing a novel VP4 genotype. Vet. Microbiol. 2016;196:27–35. doi: 10.1016/j.vetmic.2016.10.005. http://dx.doi.org/10.1016/j.vetmic.2016.10.005 [DOI] [PubMed] [Google Scholar]
- Rojas M., Manchego A., Rocha C.B., Fornells L.A., Silva R.C., Mendes G.S., Dias H.G., Sandoval N., Pezo D., Santos N. Outbreak of diarrhea among preweaning alpacas (Vicugna pacos) in the southern Peruvian highland. J. Infect. Dev. Ctries. 2016;10:269–274. doi: 10.3855/jidc.7398. http://dx.doi.org/10.3855/jidc.7398 [DOI] [PubMed] [Google Scholar]
- Rotavirus Classification Workgroup (RCWG) Newly Assigned Genotypes—Update April 20 2016. 2016. https://rega.kuleuven.be/cev/viralmetagenomics/virus-classification (accessed 27.04.17)
- Solberg O.D., Hasing M.E., Trueba G., Eisenberg J.N. Characterization of novel VP7, VP4, and VP6 genotypes of a previously untypeable group A rotavirus. Virology. 2009;385:58–67. doi: 10.1016/j.virol.2008.11.026. http://dx.doi.org/10.1016/j.virol.2008.11.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material