Abstract
Mammalian orthoreoviruses (MRVs) have a wide range of geographic distribution and have been isolated from humans and various animals. This study describes the isolation, molecular characterization and analysis of pathogenicity of MRV variant B/03 from wild short-nosed fruit bats. Negative stain electron microscopy illustrated that the B/03 strain is a non-enveloped icosahedral virus with a diameter of 70 nm. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) migration patterns showed that the B/03 viral genome contains 10 segments in a 3:3:4 arrangement. The isolate belongs to MRV serotype 1 based on S1 gene nucleotide sequence data. BALB/c mice experimentally infected with B/03 virus by intranasal inoculation developed severe respiratory distress with tissue damage and inflammation. Lastly, B/03 virus has an increased transmission risk between bats and humans or animals.
Keywords: Mammalian orthoreoviruses, Genome sequencing, Pathogenicity, Reassortment, Bat
Highlights
-
•
A novel MRV strain was isolated from wild short-nosed fruit bats in China.
-
•
Phylogenetic analyses suggested B/03 is a serotype 1 MRV of reassortment strain.
-
•
Pathogenesis of B/03 strain was tested in BALB/c mice.
1. Introduction
Mammalian orthoreovirus (MRV) belongs to the genus Orthoreovirus, which includes non-enveloped double-stranded RNA viruses, each with a genome comprising 10 genetic segments divided into three size classes (Attoui et al., 2011). Four major MRV serotypes have been characterized by neutralization assays, and all inhibit hemagglutination: type 1 Lang (T1L), type 2 Jones (T2J), type 3 Dearing (T3D) and type 4 Ndelle (T4N) (Kohl et al., 2012, Attoui et al., 2001a, Attoui et al., 2001b). MRV isolates were obtained from hosts with or without clinical signs of disease, and the virus can infect a broad range of mammals (Dermody et al., 2013). MRVs are ubiquitous mammalian pathogens, infecting nearly all mammalian hosts, including humans and other animal species (Steyer et al., 2013, Decaro et al., 2005, Attoui et al., 2011).
Infected bats are associated with an increasing number of emerging and re-emerging viruses, including the Hendra virus (HeV), Nipah virus (NiV), Ebola virus (EBOV) and SARS coronavirus. Infected bats threaten public health because they exist in large populations and travel across wide geographical distances (Wong et al., 2007, Calisher et al., 2006). However, reports on the detection and isolation of orthoreovirus from bats are limited. In 1968, the first orthoreovirus in bats, Nelson Bay virus (NBV), was isolated from the blood of fruit bats in Australia. In 1999, the second bat-borne orthoreovirus, Pulau virus (PulV), was isolated from fruit bat urine collected on Tioman Island, Malaysia. Since then, bat-borne orthoreoviruses have received much attention. Additional orthoreoviruses (MelV, KamV, Xi-River, Broome viruses, Kampar, Sikamat, HK23629/07, RpMRV-YN2012, Cangyuan virus) have been isolated from or detected in bats and in humans who were likely in contact with bats (Chua et al., 2007, Chua et al., 2008, Du et al., 2010, Thalmann et al., 2010, Cheng et al., 2009, Chua et al., 2011, Wang et al., 2015, Hu et al., 2014).
Recently, several groups have reported MRV infection in bats that resulted in visible pathology within tissues (Kohl et al., 2012, Lelli et al., 2013). The authors speculated that bat-to-human interspecies transmission was possible, but no substantial evidence to support this hypothesis was provided. In this study, we report the characterization of a novel MRV strain (called “B/03”) isolated from healthy, wild short-nosed fruit bats in Guangdong province, China. The whole genome sequence of strain B/03 was determined. Its evolution and evidence of genetic reassortment were analyzed by sequence comparison using phylogenetic analysis. Furthermore, we evaluated the pathogenicity of B/03 virus using four-week-old female BALB/c mice.
2. Materials and methods
2.1. Virus, cells and animals
MRV strain MPC/04 was isolated from masked palm civets in Guangdong Province in southern China by our laboratory and caused a potentially fatal infection of the inoculated host mouse (Li et al., 2015). Vero E6 cells were obtained from the ATCC (ATCC® CRL-1586™) and grown at 37 °C in 5% CO2 in DMEM supplemented with 2 mM glutamine, 5% fetal calf serum and antibiotics. Four-week-old female BALB/c mice were obtained from the experimental animal center of Harbin Veterinary Research Institute (HVRI). All animals were housed in the animal facility at HVRI under standard conditions in accordance with institutional guidelines.
2.2. Viral isolation and identification
Thirty tissue samples from short-nosed fruit bats were collected from Shaoguan city of China's Guangdong province and homogenized. The homogenate was filtered through a 0.22 μm pore-size filter and used to inoculate confluent monolayers of Vero E6 cells. Blind passages were performed until a cytopathic effect (CPE) was observed. The infected cells were plaque purified, and the virus was propagated in Vero E6 cultures. Virus was collected from infected cells by three freeze-thaw cycles. Aliquots were stored at − 80 °C. One aliquot was titrated on Vero E6 cells to estimate a titer by plaque assay. If CPE was not observed after 4 passages, the result of virus isolation was considered negative. The infected cells were prepared for negative stain and thin section examination by electron microscopy (EM).
In addition, an indirect immunofluorescence assay (IFA) was used to detect MRV proteins in infected cell cultures. Briefly, after washing with PBS, cells were fixed with 4% paraformaldehyde and incubated with 1% BSA for 1 h. Then, the cells were incubated with a mouse anti-MRV (T3D) antibody, followed by a goat anti-mouse IgG-FITC secondary antibody (SANTA CRUZ, USA). After washing, fluorescence was observed under an AMG EVOS F1 inverted microscope. Normal mouse sera, diluted 1:50, was used as a negative control.
2.3. Electropherotype
Viral dsRNA was extracted from purified virus particles using TRIZOL Reagent according to the manufacturer's protocol. Double strand RNA (dsRNA) segments were separated by electrophoresis in 8% (w/v) polyacrylamide slab gels. Approximately 30 μl of each sample was loaded into the gels, and electrophoresis was performed at 120 V for 4 h at room temperature.
2.4. PCR, nucleotide sequencing, and phylogenetic analysis
To further characterize the virus, primers were designed with Primer Premier 5.0 software based on published sequences. All information regarding the primers is provided in Table 1 . RT-PCR was performed using the One Step RT-PCR kit (Qiagen) as described in previous reports (Li et al., 2015). The whole genome of B/03 was amplified and sequenced by the Sanger method; then, the sequence data were assembled using the Seqman program and manually edited. The sequence of B/03 was compared with other published MRV sequences. Phylogenetic analyses were performed using the Neighbor-Joining (NJ) method with the Kimura 2-parameter model in MEGA 5.
Table 1.
Primers used in this study for amplification of the full-length genome of B/03 strain.
| Gene | Primer | Start | Sequence (5′-3′) |
|---|---|---|---|
| L1 | L1a | 9 | TTCCACGACAATGTCATCCA |
| L1b | 1019 | AGTTCGCGCGCTTTCTTATC | |
| L1c | 951 | GGGAGTCATGCCATTGTCCA | |
| L1d | 1964 | TGAATCATGTTCTGCATTCC | |
| L1e | 1886 | CTGCATCCATTGTAAATGACGAGTC | |
| L1f | 2339 | GCTATGTCATATTTCCATCCGAATTC | |
| L1g | 1930 | GCTAGGCCGATATCGGGAATGCAG | |
| L1h | 2278 | CTTGAGATTAGCTCTAGCATCTTCTG | |
| L1i | 2212 | CCAAGGTGACGACGGACTGA | |
| L1j | 2901 | CGCTCGTCCAGATTTCGTAG | |
| L1k | 2806 | AACGCAGATTATCGCAGGTG | |
| L1l | 3839 | CACGACCCATGGTAGACTCA | |
| L2 | L2a | 13 | ATGGCGAACGTY(T/C)TGGGGR(A/G)GTGAG |
| L2b | 817 | GGACGTTGGCTGAGAATTGCTCTA | |
| L2c | 735 | GGTCAGY(T/C)GCR(A/G)CCTCCTTATATT | |
| L2d | 1684 | CACCACGGGCAACATCATAAC | |
| L2e | 1577 | CCGTTAGTCATAGAGCCCTGGATT | |
| L2f | 2681 | CGGCCCCCAAAGACAACATA | |
| L2g | 2578 | CAGCCGAGTGGM(A/C)TGY(T/C)TGGAA | |
| L2h | 3346 | CTGGATTAGCGTTAGACCCGAC | |
| L2i | 3301 | GGAATCTAY(C/T)ACM(A/C)ATGCAGGC | |
| L2j | 3895 | GAGGGACRR(A/G)TGAGTTACAGAGG | |
| L3 | L3a | 13 | GATGAAGCGGATTCCAAGGA |
| L3b | 1148 | AAAACCCCGTGTGCCTATTC | |
| L3c | 1054 | GGGGGCTAATCCGCTAATGTT | |
| L3d | 2179 | GCGCCATAACGAATCTGAGAG | |
| L3e | 2125 | GTGGGCTGAAATTATTCATAGATACTGG | |
| L3f | 3489 | GCATTAGCGTACTGACGTGGATCATA | |
| L3g | 3439 | CGCTTACCCATACATGCTGC | |
| L3h | 3901 | GATGAATCGGCCCAACTAGC | |
| M1 | M1a | 1 | GCTATTCGCGGTCATGGC |
| M1b | 1485 | CCTGTCATCATGCGGAATGAG | |
| M1c | 1382 | GAGCAK(T/G)GCGGTTATGGAR(G/A)AT | |
| M1d | 1764 | TGCGCR(G/A)CTAGTR(A/G)GCATACAT | |
| M1e | 1590 | CATTCGCTCATGCCGATAGTG | |
| M1f | 2304 | GATGAAGCGCGTACGTAGTCTTAG | |
| M2 | M2a | 2 | GCTAATCTGCTGACCGTCACTC |
| M2b | 2199 | TGTGCCTGCATCCCTTAACC | |
| M3 | M3a | 1 | GCGGTCGGTCGACGCTAAAGTGACCGTGGTCATGGCTTCATTCAAGGG |
| M3b | 2241 | GCAGGGGATCCGATGAATGGGGGTCGGGAAGGCTTAAGGG | |
| S1 | S1a | 9 | CGCCTATGGATGCATCTCTCA |
| S1b | 400 | CAAAGTGGATGTTCGTCCAGTGA | |
| S1c | 340 | ACCACGAGTTGACAGTCTGGAT | |
| S1d | 1436 | CGCGCTAGATTCACCTCACATT | |
| S2 | S2a | 1 | GCTATTCGCTGGTCAGTTATGGC |
| S2b | 1331 | GATGAATGTGTGGTCAGTCGTGAG | |
| S3 | S3a | 1 | GCTAAAGTCACACCTGTCGTCGTC |
| S3b | 1198 | GATGATTAGGCGTCACCCACCAC | |
| S4 | S4a | 1 | GCGAATTCGCTATTTTTGCCTCTTCCCAGA |
| S4b | 1215 | CAS4TGCCTGCAGATGAATGAAGCCTGTCCCACGTC |
2.5. Virulence studies
A total of 100 four-week-old female BALB/c mice were randomly divided into ten groups of 10. The animals in groups 1–8 were infected by intranasal (i.n.), intracranial (i.c.), intraperitoneal (i.p.) or intragastric (i.g.) inoculation with 106 and 107 PFU-purified B/03 virus diluted in PBS. Animals in group 9 received 107 PFU of MPC/04 (i.n.). Simultaneously, mice were inoculated with PBS (i.n.) as a control. For 30 days, all mice were monitored daily for clinical signs of disease. Tissues were harvested from mice euthanized by CO2 narcosis for analysis of viral replication and pathology.
A histopathological scoring system was used to characterize pathological lesions in detail. Standard histopathological procedures were used to record all observed lesions. Samples were fixed in buffered formalin and embedded in paraffin wax. Sections (4 μm) were stained with hematoxylin and eosin (H–E) for histopathological examination. A scoring system for gross pathology and histopathology was developed, and the severity of lesions ranged from 0 (no lesion) to 3 (severe lesion). Data from both scoring systems were analyzed using the Kruskal-Wallis non-parametric mean comparison test (Elliott and Hynan, 2011), and differences were considered significant at P < 0.05.
Organs from duplicate animals were collected at each time point, suspended in 9 vol of PBS and disrupted by sonication for 4 min on ice. Titration was performed by virus plaque assay. Infectious titers were calculated per gram of tissue. The data were plotted as the mean values with variation shown as ± 1 standard error.
2.6. Ethics statement
The animal experiments were approved by the Animal Ethics Committee of HVRI of the Chinese Academy of Agricultural Sciences (CAAS) and performed in accordance with animal ethics guidelines and approved protocols. The Animal Ethics Committee approval number was SYXK (Hei) 2011-022.
3. Results
3.1. Virus isolation and visualization
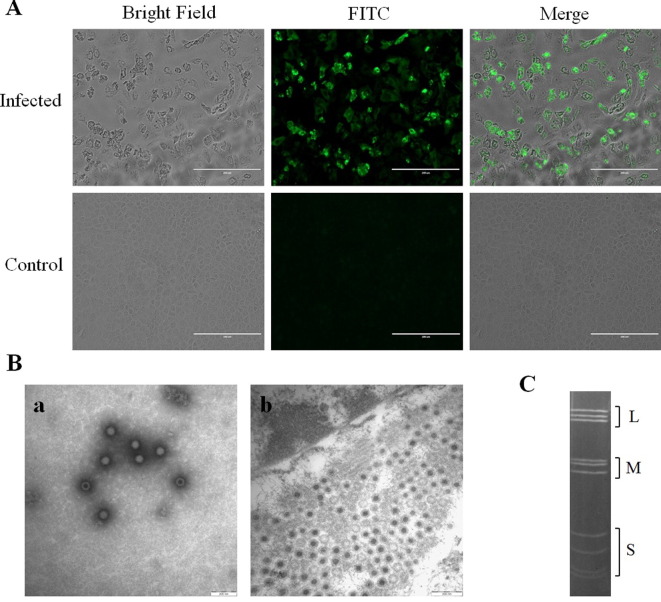
After four passages of the isolated virus on Vero E6 cells, a distinct CPE was observed in infected cells, characterized by granulating, shrinking, rounding, seining, and detaching, as determined by IFA using serum from mice immunized with T3D (Fig. 1A). MRV particles in infected Vero cells were also examined by EM techniques. As shown in Fig. 1B, negative stain EM showed multiple virus-like particles with non-enveloped icosahedral formation in the homogenates. Ultra-thin sections of infected Vero cells showed typical electrondense virus particles organized in a paracrystalline pattern within the cytoplasm. Polyacrylamide gel electrophoresis (PAGE) migration patterns of the genome segments showed that B/03 virus contains 10 segments in a 3:3:4 arrangement, typical of reoviruses (Fig. 1C).
Fig. 1.
(A) Indirect immunofluorescence detection of MRV in Vero E6 cells infected with strain B/03. The cells were fixed with 4% paraformaldehyde, blocked with 1% BSA, washed, and incubated with mouse anti-MRV (T3D) antibody. The cells were then incubated with FITC-labeled goat anti-mouse IgG secondary antibody. (B) Electron micrographs. a: Negative staining of cell culture supernatant. Non-enveloped reoviral-like particles with double-layered capsid structure were observed (diameter = approximately 70 nm). b: Ultra-thin sections of infected Vero E6 cells displayed typical contrast-rich virus particles, organized as paracrystalline structures within the cytosol. (C) SDS-PAGE demonstrating electrophoresis pattern. L, M and S represent large, medium and small segments, respectively. dsRNA segments were separated by electrophoresis in 8% (w/v) polyacrylamide slab gels.
3.2. Sequencing and phylogenetic analysis
The complete genome sequence of B/03 virus was generated, and comparative analysis with other MRV strains was performed. The complete sequences of 10 segments were submitted to a GenBank database under accession numbers KX263307–KX263316. The complete genome of B/03 virus is 22,875 bp, and sizes of segments 1 through 10 are as follows: L1, 3804 bp; L2, 3870 bp; L3, 3828 bp; M1, 2211 bp; M2, 2127 bp; M3, 2166 bp; S1, 1413 bp; S2, 1257 bp; S3, 1101 bp; and S4, 1098 bp. The inferred lengths of eight structural proteins and three nonstructural proteins are as follows: λ1 (1275 aa), λ2 (1289 aa), λ3 (1267 aa), μNS (721 aa), μ1 (708 aa), μ2 (736 aa), σ1 (470 aa), σ1s (119 aa), σ2 (418 aa), σNS (366 aa), and σ3 (365 aa).
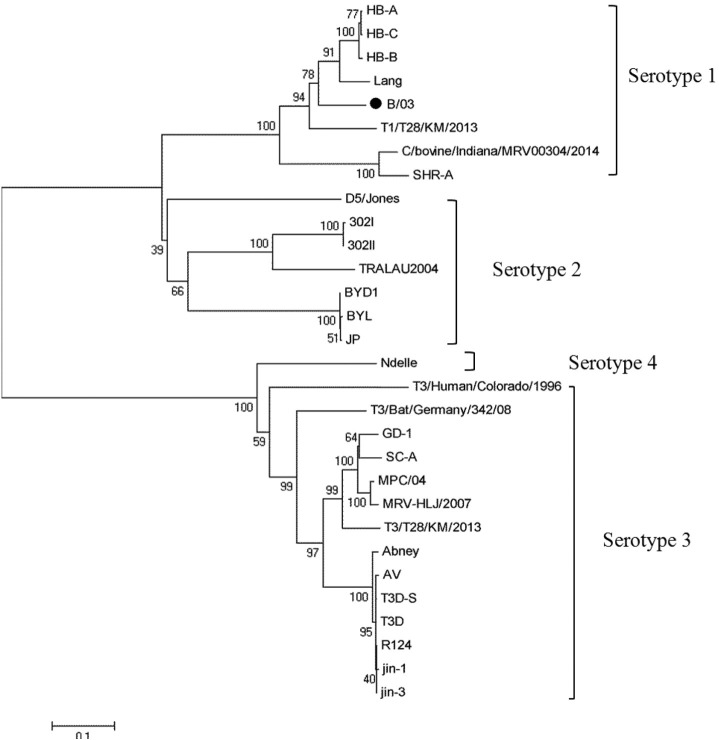
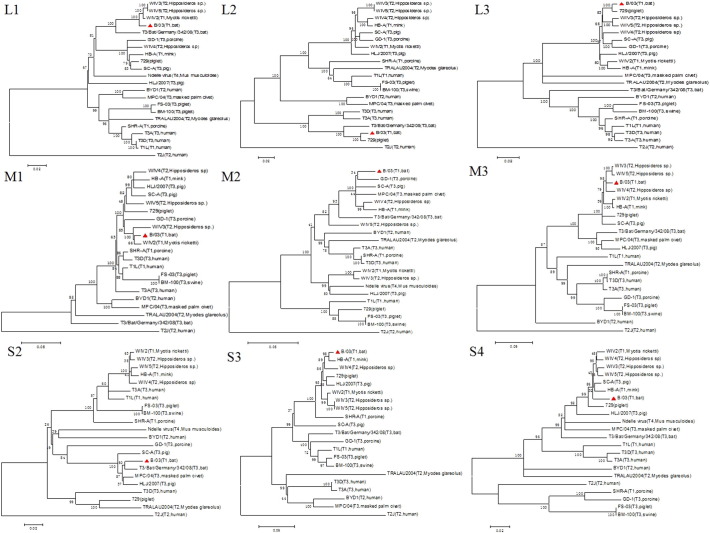
Pairwise nucleotide and inferred amino acid comparison between strain B/03 and other MRV strains were performed for all ten segments (Table 2 ). Nucleotide sequence alignments indicated that the L1, M1, M2, M3, and S1 genes from B/03 virus were highly related to those of WIV2–4 virus (98.63, 98.19, 97.04, 98.29, and 98.80%, respectively). The L2, L3 and S4 genes had highest identities (97.54–99.03%) with pig strains 729 and SC-A. The S2 segment of strain B/03 was most similar to strain MPC/04 on both the nucleotide and inferred amino acid level. Segment S3 of strain B/03 was most similar to strains HB-C and HB-A (isolated from minks) on both nucleotide and amino acid levels. The MRV S1 gene encodes the viral attachment protein σ1, which is unique to each MRV prototype strain and determines the serotype. When compared to the S1 sequences available in GenBank, the B/03 strain shares higher identity with that of MRV-1 than with those of MRV-2, MRV-3 or MRV-4 (Fig. 2 ). Phylogenetic analysis of the L1, L2, L3, M1, M2, M3, S2, S3 and S4 genome segments for the B/03 strain and most related whole-genome strains available in GenBank is shown in Supplementary 1.
Table 2.
Mammalian orthoreoviruses with the highest nucleotide and amino acid sequence identity to B/03 virus.
| B/03 genes |
B/03 proteins |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Identity (%) | Isolate | Serotype | Host | Country | Identity (%) | Isolate | Serotype | Host | Country | ||
| L1 | 98.63 | WIV2 | T1 | Myotis ricketti | China | λ3 | 99.53 | WIV2 | T1 | Myotis ricketti | China |
| L2 | 98.32 | 729 | unknown | pig | Austria | λ2 | 99.30 | 729 | unknown | pig | Austria |
| L3 | 99.03 | 729 | unknown | pig | Austria | λ1 | 99.84 | WIV5 | T2 | Hipposideros sp. | China |
| M1 | 98.19 | WIV2 | T1 | Myotis ricketti | China | μ2 | 99.18 | Lang | T1 | human | USA |
| M2 | 97.04 | WIV4 | T2 | Hipposideros sp. | China | μ1 | 99.86 | MRV2Tou05 | T2 | human | France |
| M3 | 98.29 | WIV3 | T2 | Hipposideros sp. | China | μNS | 99.72 | WIV3 | T2 | Hipposideros sp. | China |
| S1 | 98.80 | WIV2 | T1 | Myotis ricketti | China | σ1 | 98.72 | WIV2 | T1 | Myotis ricketti | China |
| S2 | 97.14 | MPC/04 | T3 | masked palm civet | China | σ2 | 98.33 | MPC/04 | T3 | masked palm civet | China |
| S3 | 98.55 | HB-C | T1 | mink | China | σNS | 99.73 | HB-A | T1 | mink | China |
| S4 | 97.54 | SC-A | T3 | pig | China | σ3 | 97.81 | WIV3 | T2 | Hipposideros sp. | China |
Fig. 2.
Phylogenetic trees based on the S1 gene of B/03 virus from this study and 29 previously published reovirus sequences from GenBank. The Neighbor-joining method was used for construction of the phylogenetic tree with bootstrap values of 1000 replicates shown at the branches. The scale bar represents the p-distance.
Supplementary 1.
Phylogenetic analysis of the L1, L2, L3, M1, M2, M3, S2, S3 and S4 genome segments for the B/03 strain andmost relatedwhole-genome strains from GenBank. Neighbor-joining was used for the construction of phylogenetic tree with bootstrap values of 1000 replicates shown at the branches. The scale bar represents the p-distance.
3.3. Virulence studies
3.3.1. Clinical features of experimentally infected BALB/c mice
To determine the potential route of infection, BALB/c mice were inoculated in four ways with different doses of purified virus B/03 (106 or 107 PFU). Mice infected by i.n. inoculation in groups 1 (106 PFU B03), 2 (107 PFU B03) and 9 (107 PFU MPC/04) exhibited clinical signs (noticeable respiratory distress and body weight loss), and the time of death and clinical manifestations of infection varied with dose. The highest dose of B/03 virus caused disease and resulted in death in 5 of 10 mice starting at 7 days post-infection (Fig. 3 ). The same dose of MPC/04 virus also induced clinical symptoms and death in mice. Mice infected with 106 PFU of B03 virus also manifested respiratory crackling and death in 2 of 10 mice starting at 8 days post-infection. In contrast, all of the mice exposed to the same doses by either i.c. i.p. or i.g. inoculation did not exhibit any signs of respiratory distress or changes in body weight during the course of the experiment, and no histological changes were observed in their organs at any time point. The control mice remained healthy throughout the trial. These experiments showed that respiratory infection with B/03 virus induces disease in BALB/c mice.
Fig. 3.
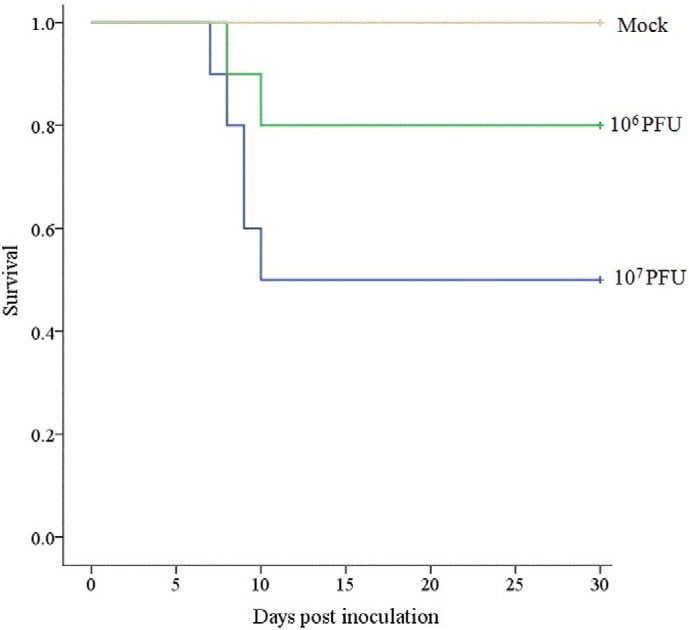
Survival of BALB/c mice infected with different doses of B/03 (106 or 107 PFU) via the intranasal route. Survival of infected mice was monitored daily for a total of 30 days. The time to death of BALB/c mice and clinical symptoms of infection varied with dose. Other routes of infection did not cause fatality (not show).
3.3.2. Histopathological changes
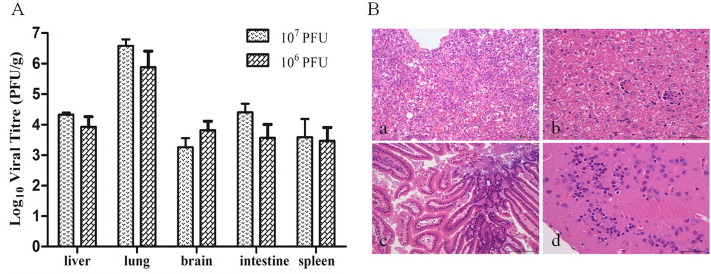
To assess the gross pathological consequences of infection, the liver, lung, brain, intestine, and spleen were subjected to histopathological examination using standard procedures. All surviving animals were euthanized at 30 dpi for inclusion in this analysis. Samples were paraffin-embedded, sectioned and stained with hematoxylin and eosin. Infection of the lung produced signs of inflammation associated with alveolar thickening and lymphocytic infiltration (Fig. 4A). A composite analysis using the abovementioned histopathological scoring system was performed (Table 3 ). These data indicate that the B/03 strain induces histopathological changes associated with disease. No statistically significant differences were observed in mice inoculated by other routes or in control animals.
Fig. 4.
(A) Lung pathology BALB/c mice infected with B/03 virus. Panel (a) and (b) show the B/03-infected lung with 107 and 106 PFU of B/03 virus at 7 dpi. Panel (c) shows images of lungs from uninfected control mice. (B) Viral titers in the liver, lung, brain, intestine and spleen of BALB/c mice (i.n. inoculation) with 106 and 107 PFU of B/03 virus at 7 dpi.
Table 3.
Average scores for the histopathological changes observed in infected BALB/c mice.
| Groups | Liver | Lung | Brain | Intestine | Spleen | Total |
|---|---|---|---|---|---|---|
| 107 PFU B/03 | 1.03 | 2.57 | 0.13 | 1.7 | 1.23 | 6.66 |
| 106 PFU B/03 | 1.07 | 2.2 | 0.0 | 1.83 | 0.67 | 5.77 |
| 107 PFU MPC/04 | 1.67 | 2.77 | 0.37 | 1.53 | 1.07 | 7.41 |
| Control | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
3.3.3. Viral titers in the tissues
To analyze viral replication in different organs of infected mice, viral titers were quantified in tissues 7 days after infection with 106 and 107 PFU of B/03 virus. Animals inoculated with 107 PFU of B/03 virus had higher viral titers (up to 107 PFU/g) than those of mice inoculated with 106 PFU. Higher levels of viral RNA were detected in the lungs than in the brain, liver, intestine or spleen (Fig. 4B). However, the inoculated tissue (i.c., i.p. and i.g.) with 107 PFU had 3–5 times less virus in each organ compared to those of i.n. inoculated mice (data not shown). No virus was detected in the control animals.
4. Discussion
The most recent disease outbreaks have been associated with zoonotic transmission events, and newly emerging viruses have originated from wildlife (Moratelli and Calisher, 2015, O'Shea et al., 2014). Thus, surveillance and evaluation of viruses prevalent in wildlife are of special interest. Bats are the natural host reservoir for a number of high-impact zoonotic viruses. More than 200 viruses belonging to 27 families were isolated from or detected in bats. A few of these viruses have been responsible for human disease, including Ebola virus (Leroy et al., 2005), Middle East respiratory syndrome coronavirus (MERS-CoV) (Ithete et al., 2013), Severe Acute Respiratory Syndrome coronaviruses (SARS-CoV) (Ge et al., 2013), Nipah and Hendra viruses (Halpin et al., 2000, Marsh et al., 2012). Several bat orthoreovirus isolates have been obtained from bats in recent years (Moratelli and Calisher, 2015, Chua et al., 2007, Pritchard et al., 2006, Du et al., 2010). A novel bat reassortant MRV, RpMRV-YN2012, obtained from least horseshoe bats in China resulted from a reassortment of MRVs known to infect humans and animals (Wang et al., 2015). Six MRV strains were isolated from Hipposideros and Myotis and grouped into MRV serotypes 1, 2, or 3 based on the S1 gene sequence (Yang et al., 2015). Three novel MRVs were isolated from European bats, with rather mild or clinically unapparent infections in their hosts (Kohl et al., 2012). Considering the diversity and wide distribution of bats and the potential for transmission of bat viruses to humans and other animals, continued surveillance of MRVs in all host species is urgently needed. In this study, one strain of MRV, B/03, was isolated by in vitro cell culture from thirty wild short-nosed fruit bat samples from Shaoguan city of China's Guangdong province. The isolate was serially propagated in cell culture and characterized by cell culture CPE, immunofluorescence staining, Electropherotype, EM and entire genome sequencing.
MRV genomes undergo multiple types of genomic alteration, including intragenic rearrangement and reassortment, in both laboratory and natural conditions (Dermody et al., 2013). To molecularly characterize B/03 virus, we amplified and sequenced the three large (L1–L3), three medium (M1–M3), and four small (S1–S4) viral genes. Based on sequence comparison and phylogenetic analysis, we conclude that the B/03 isolate is a novel type 1 bat orthoreovirus, and it might have originated from gene segment mixing during infection with more than one MRV strain in nature. The potential function of these genes may be important for understanding pathogenic mechanisms and should be studied further.
MRVs were traditionally believed to be causative agents of mild respiratory and enteric diseases without significant clinical impact (Steyer et al., 2013). However, several recent studies have suggested that MRV can cause serious illness and even death in humans and other mammals, characterized by upper respiratory tract infection, diarrhea and encephalitis (Tyler et al., 2004, Ouattara et al., 2011). Indeed, the pathogenesis of reovirus infections has been most extensively studied using both suckling and adult mice, and infections lead to systemic viral replication, morbidity, and mortality (Dermody et al., 2013, Doyle et al., 2015, Organ and Rubin, 1998). This study aimed to use BALB/c mice to study MRV B/03 pathogenesis. BALB/c mice were infected by intranasal (i.n.), intracranial (i.c.), intraperitoneal (i.p.) or intragastric (i.g.) inoculation with different doses of B/03 virus. We found that mice are susceptible to MRV B/03 infection by intranasal inoculation. The highest dose of B/03 virus (107 PFU) induced signs of disease on day 5 after infection and resulted in the death of 5 of 10 mice starting at 7 days post-infection. We observed approximately 50% mortality in mice that underwent i.n. inoculation with 107 PFU of B/03 virus. In a previous study, we showed that the MPC/04 strain (isolated from masked palm civets) is pathogenic to four-week-old female BALB/c mice with approximately 70% mortality and pronounced pathological changes in tissues of mice inoculated (i.n.) with 107 PFU (Li et al., 2015). In this study, infection with B/03 virus caused obvious lesions in the lungs due to tissue damage and inflammation associated with alveolar thickening and lymphocytic infiltration, as well as accumulation of cellular debris and distended bronchioles and alveoli. Our study shows that the B/03 strain is pathogenic to BALB/c mice. Additional studies regarding viral replication, pathogenesis and host interactions are needed to better understand the pathogenesis of this virus. These findings parallel those found by others in rats (Gauvin et al., 2013, Morin et al., 1996).
To characterize viral replication, we analyzed viral titers in the lung, brain, liver, intestine and spleen after i.n. infection with B/03 virus. Viral loads in the lung were higher (100-fold) than those in other tissues, and this observation was associated with the ability of the virus to cause acute respiratory distress. These data are consistent with pathological changes in the lung. Mice in the 107 PFU inoculation group displayed severe acute respiratory symptoms and died starting at day 7 dpi. Our results indicate that B/03 virus replicates to higher levels in the lung than in other organs, a finding that is in agreement with the induction of acute respiratory distress in infected mice. We also provide evidence that these novel MRV strains are pathogenic to mice, leading to lethal respiratory disease.
Considering B/03 may have resulted from a reassortment of bat, mink, and/or human MRV strains, which can cause severe disease in humans and animals, it is necessary to identify pathogenicity in animal hosts. Our data confirm that mice were infected with B/03 virus via the respiratory route, causing a potentially fatal respiratory infection. Further work is required to understand the full zoonotic potential and pathogenesis of B/03 virus.
The following is the supplementary data related to this article.
Acknowledgments
This work was supported by funds from the National Natural Science Foundation of China (31402204), the State Key Laboratory of Veterinary Biotechnology (SKLVBP201421), and the Basic Scientific Research Operation Cost of State-leveled Public Welfare Scientific Research Courtyard (0302014011).
Contributor Information
Shengwang Liu, Email: SWLIU@hvri.ac.cn.
Liandong Qu, Email: qld@hvri.ac.cn.
References
- Attoui H., Biagini P., Stirling J., Mertens P.P., Cantaloube J.F., Meyer A., de Micco P., de Lamballerie X. Sequence characterization of Ndelle virus genome segments 1, 5, 7, 8, and 10: evidence for reassignment to the genus Orthoreovirus, family Reoviridae. Biochem. Biophys. Res. Commun. 2001;287:583–588. doi: 10.1006/bbrc.2001.5612. [DOI] [PubMed] [Google Scholar]
- Attoui H., Biagini P., Stirling J., Mertens P.P., Cantaloube J.F., Meyer A. Sequence characterization of Ndelle virus genome segments 1, 5, 7, 8, and 10: evidence for reassignment to the genus Orthoreovirus, family Reoviridae. Biochem. Biophys. Res. Commun. 2001;287:583–588. doi: 10.1006/bbrc.2001.5612. [DOI] [PubMed] [Google Scholar]
- Attoui, H., Mertens, P.P.C., Becnel, J., Belaganahalli, S., Bergoin, M., Brussaard, C.P., 2011. Orthoreovirus, Reoviridae. In: Virus Taxonomy. Classification and Nomenclature of Viruses: Ninth Report of the International Committee on the Taxonomy of Viruses. London: Elsevier Academic Press. p, 546–554. http://www.elsevier.com/books/virus-taxonomy/king/978-0-12-384684-6.
- Calisher C.H., Childs J.E., Field H.E., Holmes K.V., Schountz T. Bats: important reservoir hosts of emerging viruses. Clin. Microbiol. Rev. 2006;19:531–545. doi: 10.1128/CMR.00017-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng P., Lau C.S., Lai A., Ho E., Leung P., Chan F., Wong A., Lim W. A novel reovirus isolated from a patient with acute respiratory disease. J. Clin. Virol. 2009;45:79–80. doi: 10.1016/j.jcv.2009.03.001. [DOI] [PubMed] [Google Scholar]
- Chua K.B., Crameri G., Hyatt A., Yu M., Tompang M.R., Rosli J., McEachern J., Crameri S., Kumarasamy V., Eaton B.T., Wang L.F. A previously unknown reovirus of bat origin is associated with an acute respiratory disease in humans. Proc. Natl. Acad. Sci. U. S. A. 2007;104:11424–11429. doi: 10.1073/pnas.0701372104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chua K.B., Voon K., Crameri G., Tan H.S., Rosli J., McEachern J.A., Suluraju S., Yu M., Wang L.F. Identification and characterization of a new orthoreovirus from patients with acute respiratory infections. PLoS One. 2008;3 doi: 10.1371/journal.pone.0003803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chua K.B., Voon K., Yu M., Keniscope C., Abdul Rasid K., Wang L.F. Investigation of a potential zoonotic transmission of orthoreovirus associated with acute influenza-like illness in an adult patient. PLoS One. 2011;6 doi: 10.1371/journal.pone.0025434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Decaro N., Campolo M., Desario C., Ricci D., Camero M., Lorusso E., Elia G., Lavazza A., Martella V., Buonavoglia C. Virological and molecular characterization of a mammalian orthoreovirus type 3 strain isolated from a dog in Italy. Vet. Microbiol. 2005;109:19–27. doi: 10.1016/j.vetmic.2005.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dermody T.S., Parker J.S., Sherry B. Orthoreoviruses. In: Knipe D.M., Howley P.M., editors. Fields Virology. sixth ed. Wolters Kluwer/Lippincott Williams & Wilkins; Philadelphia: 2013. pp. 1304–1346. [Google Scholar]
- Doyle J.D., Stencel-Baerenwald J.E., Copeland C.A., Rhoads J.P., Brown J.J., Boyd K.L., Atkinson J.B., Dermody T.S. Diminished reovirus capsid stability alters disease pathogenesis and littermate transmission. PLoS Pathog. 2015;11 doi: 10.1371/journal.ppat.1004693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du L., Lu Z., Fan Y., Meng K., Jiang Y., Zhu Y., Wang S., Gu W., Zou X., Tu C. Xi River virus, a new bat reovirus isolated in southern China. Arch. Virol. 2010;155:1295–1299. doi: 10.1007/s00705-010-0690-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elliott A.C., Hynan L.S. A: SAS(®) macro implementation of a multiple comparison post hoc test for a Kruskal-Wallis analysis. Comput. Methods Prog. Biomed. 2011;102:75–80. doi: 10.1016/j.cmpb.2010.11.002. [DOI] [PubMed] [Google Scholar]
- Gauvin L., Bennett S., Liu H., Hakimi M., Schlossmacher M., Majithia J., Brown E.G. Respiratory infection of mice with mammalian reoviruses causes systemic infection with age and strain dependent pneumonia and encephalitis. Virol. J. 2013;10:67. doi: 10.1186/1743-422X-10-67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ge X.Y., Li J.L., Yang X.L., Chmura A.A., Zhu G., Epstein J.H., Mazet J.K., Hu B., Zhang W., Peng C., Zhang Y.J., Luo C.M., Tan B., Wang N., Zhu Y., Crameri G., Zhang S.Y., Wang L.F., Daszak P., Shi Z.L. Isolation and characterization of a bat SARS-like coronavirus that uses the ACE2 receptor. Nature. 2013;503:535–538. doi: 10.1038/nature12711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halpin K., Young P.L., Field H.E., Mackenzie J.S. Isolation of Hendra virus from pteropid bats: a natural reservoir of Hendra virus. J. Gen. Virol. 2000;81:1927–1932. doi: 10.1099/0022-1317-81-8-1927. [DOI] [PubMed] [Google Scholar]
- Hu T.S., Qiu W., He B., Zhang Y., Yu J., Liang X., Zhang W.D., Chen G., Zhang Y.G., Wang Y.Y., Zheng Y., Feng Z.L., Hu Y.H., Zhou W.G., Tu C.C., Fan Q.S., Zhang F.Q. Characterization of a novel orthoreovirus isolated from fruit bat, China. BMC Microbiol. 2014;14:293. doi: 10.1186/s12866-014-0293-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ithete N.L., Stoffberg S., Corman V.M., Cottontail V.M., Richards L.R., Schoeman M.C., Drosten C., Drexler J.F., Preiser W. Close relative of human Middle East respiratory syndrome coronavirus in bat, South Africa. Emerg. Infect. Dis. 2013;19:1697–1699. doi: 10.3201/eid1910.130946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kohl C., Lesnik R., Brinkmann A., Ebinger A., Radonic A., Nitsche A., Muhldorfer K., Wibbelt G., Kurth A. Isolation and characterization of three mammalian orthoreoviruses from European bats. PLoS One. 2012;7 doi: 10.1371/journal.pone.0043106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lelli D., Moreno A., Lavazza A., Bresaola M., Canelli E., Boniotti M.B., Cordioli P. Identification of mammalian orthoreovirus type 3 in Italian bats. Zoonoses Public Health. 2013;60:84–92. doi: 10.1111/zph.12001. [DOI] [PubMed] [Google Scholar]
- Leroy, E.M., Kumulungui, B., Pourrut, X., Rouquet, P., Hassanin, A., Yaba, P., Délicat, A., Paweska, J.T., Gonzalez, J.P., Swanepoel, R., 2005. 438:575–576. doi:http://dx.doi.org/10.1038/438575a. [DOI] [PubMed]
- Li Z.J., Shao Y.H., Liu C.G., Liu D.F., Guo D.C., Qiu Z., Tian J., Zhang X.Z., Liu S.W., Qu L.D. Isolation and pathogenicity of the mammalian orthoreovirus MPC/04 from masked civet cats. Infect. Genet. Evol. 2015;36:55–61. doi: 10.1016/j.meegid.2015.08.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marsh G.A., de Jong C., Barr J.A., Tachedjian M., Smith C., Middleton D., Yu M., Todd S., Foord A.J., Haring V., Payne J., Robinson R., Broz I., Crameri G., Field H.E., Wang L.F. Cedar virus: a novel henipavirus isolated from Australian bats. PLoS Pathog. 2012;8 doi: 10.1371/journal.ppat.1002836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moratelli R., Calisher C.H. Bats and zoonotic viruses: can we confidently link bats with emerging deadly viruses? Mem. Inst. Oswaldo Cruz. 2015;110:1–22. doi: 10.1590/0074-02760150048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morin M.J., Warner A., Fields B.N. Reovirus infection in rat lungs as a model to study the pathogenesis of viral pneumonia. J. Virol. 1996;70:541–548. doi: 10.1128/jvi.70.1.541-548.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Organ E.L., Rubin D.H. Pathogenesis of reovirus gastrointestinal and hepatobiliary disease. Curr. Top. Microbiol. Immunol. 1998;233:67–83. doi: 10.1007/978-3-642-72095-6_4. [DOI] [PubMed] [Google Scholar]
- O'Shea T.J., Cryan P.M., Cunningham A.A., Fooks A.R., Hayman D.T., Luis A.D., Peel A.J., Plowright R.K., Wood J.L. Bat flight and zoonotic viruses. Emerg. Infect. Dis. 2014;20:741–745. doi: 10.3201/eid2005.130539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ouattara L.A., Barin F., Barthez M.A., Bonnaud B., Roingeard P., Goudeau A., Castelnau P., Vernet G., Paranhos-Baccala G., Komurian-Pradel F. Novel human reovirus isolated from children with acute necrotizing encephalopathy. Emerg. Infect. Dis. 2011;17:1436–1444. doi: 10.3201/eid1708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pritchard L.I., Chua K.B., Cummins D., Hyatt A., Crameri G., Eaton B.T., Wang L.F. Pulau virus; a new member of the Nelson Bay orthoreovirus species isolated from fruit bats in Malaysia. Arch. Virol. 2006;151:229–239. doi: 10.1007/s00705-005-0644-4. [DOI] [PubMed] [Google Scholar]
- Steyer A., Gutiérrez-Aguire I., Kolenc M., Koren S., Kutnjak D., Pokorn M., Poljšak-Prijatelj M., Racki N., Ravnikar M., Sagadin M., Fratnik Steyer A., Toplak N. High similarity of novel orthoreovirus detected in a child hospitalized with acute gastroenteritisto mammalian orthoreoviruses found in bats in Europe. J. Clin. Microbiol. 2013;51:3818–3825. doi: 10.1128/JCM.01531-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thalmann C.M., Cummins D.M., Yu M., Lunt R., Pritchard L.I., Hansson E., Crameri S., Hyatt A., Wang L.F. Broome virus, a new fusogenic orthoreovirus species isolated from an Australian fruit bat. Virology. 2010;402:26–40. doi: 10.1016/j.virol.2009.11.048. [DOI] [PubMed] [Google Scholar]
- Tyler K.L., Barton E.S., Ibach M.L., Robinson C., Campbell J.A., O'Donnell S.M., Valyi-Nagy T., Clarke P., Wetzel J.D., Dermody T.S. Isolation and molecular characterization of a novel type 3 reovirus from a child with meningitis. J. Infect. Dis. 2004;189:1664–1675. doi: 10.1086/383129. [DOI] [PubMed] [Google Scholar]
- Wang L.H., Fu S.H., Cao L., Lei W.W., Cao Y.X., Song J.D., Tang Q., Zhang H.L., Feng Y., Yang W.H., Liang G.D. Isolation and identification of a natural reassortant mammalian orthoreovirus from least horseshoe bat in China. PLoS One. 2015;10 doi: 10.1371/journal.pone.0118598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong S., Lau S., Woo P., Yuen K.Y. Bats as a continuing source of emerging infections in humans. Rev. Med. Virol. 2007;17:67–91. doi: 10.1002/rmv.520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang X.L., Tan B., Wang B., Li W., Wang N., Luo C.M., Wang M.N., Zhang W., Li B., Peng C., Ge X.Y., Zhang L.B., Shi Z. Isolation and identification of bat viruses closely related to human, porcine, and mink orthoreoviruses. J. Gen. Virol. 2015;96:3525–3531. doi: 10.1099/jgv.0.000314. [DOI] [PMC free article] [PubMed] [Google Scholar]