Summary
Bronchiolitis and preschool recurrent wheeze (PSRW) are common paediatric problems causing significant morbidity and mortality in the first years of life. Respiratory syncytial virus (RSV) and rhinoviruses are the commonest pathogens associated with these illnesses. Why some infants are severely affected, requiring admission to hospital, whilst others experience a simple cold is not fully understood: research has suggested that the innate immune response to these viruses is important. The innate immune system has many components and activation or deficiency in one or many areas may explain the different clinical presentations and disease severities that can occur in these infants. This review will summarize the recent evidence highlighting how RSV and rhinoviruses may modulate the innate immune response in both bronchiolitis and PSRW, and discuss how these illnesses affect the long-term development of the infant lung and the possible susceptibility to persistent airway disease.
Keywords: innate immune response, bronchiolitis, respiratory syncytial virus, preschool recurrent wheeze, rhinovirus, asthma
Bronchiolitis
Acute viral bronchiolitis is a major cause of morbidity and mortality in infants.1, 2 Bronchiolitis, which literally means inflammation of the bronchioles, was first described more than 150 years ago as ‘congestive catarrhal fever’ to illustrate the clinical pattern of fever, rhinorrhoea, wheezing, crepitations, dyspnoea and difficulty feeding. It was not until 1957 that the epidemiology and viral aetiology were first described.3
Respiratory syncytial virus (RSV) is the main virus to cause bronchiolitis and is associated with over 75% of cases where a pathogen has been isolated.4 Other viruses, including adenovirus, influenza, parainfluenza, rhinovirus, human metapneumovirus (HMPV), the newly isolated coronaviruses (NL63 and HKU1) and human bocavirus may also cause similar clinical features.5 It is not uncommon for two or three viruses to be isolated in the same patient, during a single episode of bronchiolitis, with the incidence of dual infection varying between 5% and 10% in infants with severe bronchiolitis.6 There is some indication that dual infection with HMPV and RSV may be associated with more severe disease and the need for ventilation.7 However, in the sickest infants, who require ventilation for bronchiolitis, clinical and/or cellular and cytokine data of bronchoalveolar lavage (BAL) fluid do not distinguish dual infection (RSV and HMPV) from single RSV infection.8 Bacterial infections can also be found, especially in those with severe disease.9 Thorburn et al. over three winters, found that 70 of 165 (42.2%) children who required ventilation for RSV-positive bronchiolitis had lower airway secretions positive for bacteria on admission to PICU.9 Bacterial co-infection, i.e. a bacterial colony count >105 cfu/ml, was found in 36 of 165 (21.8%), with the commonest pathogens being community-acquired organisms, Haemophilus influenzae and Staphylococcus aureus. Those with bacterial co-infection required ventilation for longer than those with RSV infection only (p < 0.01).
Most (95%) infants will encounter RSV infection at least once before their second birthday but, in the majority, this will manifest only as an upper respiratory tract infection.10 In about 40% there will be signs and symptoms of infection of the lower airways with cough and wheeze, but the infant will continue to feed normally (mild bronchiolitis).11 In Europe and the USA hospitalization with bronchiolitis occurs at rates of 30 per 1000 for children aged under 1 year.12 In some infants, supplemental feeding by nasogastric tube or intravenous infusion will be needed to maintain daily fluid intake (moderate bronchiolitis), whilst others will also need additional oxygen (severe bronchiolitis).13 Approximately 2–3% of all patients hospitalized with bronchiolitis develop respiratory failure requiring admission to paediatric intensive care units (life-threatening bronchiolitis), but fortunately deaths from bronchiolitis are rare, with the rate of post-neonatal deaths in the UK falling from 21.47 per 100 000 in 1979 to 1.82 per 100 000 in 2000.14 Peak hospitalization rates for RSV bronchiolitis occur at 2 months of age15 and it is not uncommon for these infants to be re-infected again the following year.16 Subsequent infections with RSV are usually milder in severity.
The primary site of infection for viruses in bronchiolitis is the lung, but extrapulmonary manifestations may also occur, including seizures, apnoeas, electrolyte imbalance, hepatitis and myocarditis.1, 17 Currently, there is little evidence to show that a viraemia occurs with RSV infection and these extrapulmonary manifestations are attributed to direct spread or aspiration of infected droplets.
Preschool recurrent wheeze
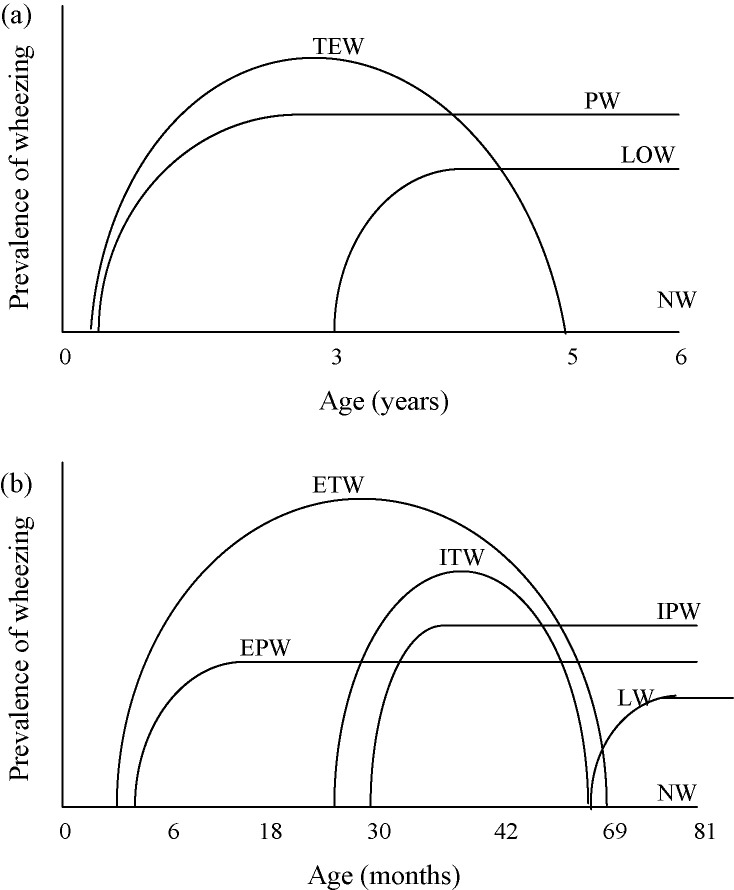
Preschool recurrent wheeze (PSRW) is a collection of discrete phenotypic disorders, each with different aetiologies, in children who are not yet attending school, i.e. younger than 5–6 years old, where wheeze is the predominant symptom. The classification of PSRW into different phenotypes has arisen as a result of studies investigating the relationship between PSRW and the development of asthma in later life. Four main phenotypes were determined in the Tucson Children’s Respiratory Study group.18 This was a population-based cohort of >1000 infants enrolled at birth between 1980 and 1984 and followed-up by respiratory-based questionnaires for 18–20 years (at 2, 3, 6, 11, 13, 16 and 18 years), plus a review at 6 years to determine the markers of atopy, i.e. the presence of eczema, allergic rhinitis and allergy sensitization using skin prick testing. The groups were (Fig. 1a):
-
1.
Transient early wheezers – infants who started to wheeze in the first 3 years of life with no wheezing in the previous 12 months at age 6 years.
-
2.
Late-onset wheezers – infants who did not wheeze during the first 3 years but wheezed in the previous 12 months at age 6 years.
-
3.
Persistent wheezers – infants who wheezed during the first 3 years and continued to wheeze in the previous 12 months at age 6 years.19
-
4.
Never wheezed – infants who had never wheezed in the first 6 years of life.
Figure 1.
The wheezing phenotypes. (a) Tucson classification of four wheezing phenotypes. TEW: transient early wheezers; LOW, late-onset wheezers; PW, persistent wheezers; NW, never wheezed. (b) Avon Longitudinal Study of Parents and Children (ALSPAC) classification of six wheezing phenotypes. ETW, early-onset transient wheezers; ITW, intermediate-onset transient wheezers; EPW, early-onset persistent wheezers; IPW, intermediate-onset persistent wheezers; LW, late-onset wheezers; NW, never wheezed.
Stein et al., in 1997, investigated the differences between the Tucson-defined wheezing phenotypes and concentrations of circulating IgE at ages 6 and 11 years: for transient wheezers or late-onset wheezers there was no association between concentrations of circulating IgE at ages 6 and 11 years and later development of atopy, but there was an association for the persistent wheezers.20
Recently the classification of wheezing phenotypes has been adapted. The ALSPAC group (Avon Longitudinal Study of Parents and Children) has shown that the development of wheeze, especially in the first 3½ years, is not as well defined as in the Tucson phenotypes.21 The ALSPAC study22 prospectively followed 14 541 infants who had an expected date of delivery between 01/04/1991 and 31/12/1992. Questionnaires on wheezing were sent to all parents regarding their children at 6, 18, 30, 42 and 81 months after birth. Each mother was asked whether their child had had any wheezing with whistling during the study periods 0–6, 6–18, 18–30, 30–42 and 69–81 months. Skin prick tests were performed at 7½ years and lung function testing was performed at 8½ years. Six wheezing phenotypes were determined (Fig. 1b):
-
1.
Never wheezed – no wheezing at any of the five time points.
-
2.
Early-onset transient wheezers – wheezed at 0–18 months but not at 69–81months.
-
3.
Intermediate-onset transient wheeze – no wheeze at 0–18 months, wheeze at 18–42 months and no wheeze at 69–81 months.
-
4.
Early-onset persistent wheeze – wheeze at 0–18 months and 69–81 months.
-
5.
Intermediate-onset persistent wheeze – no wheeze at 0–18 months, wheeze at 18–42 months and wheeze at 69–81 months.
-
6.
Late-onset wheeze – onset of wheeze after 42 months and before 81 months.
These wheezing phenotypes have been used in several longitudinal birth cohorts to help determine the pathophysiology of PSRW.
What is wheeze?
Parents will report at least one episode of wheeze in 30% of all preschool children.19 However, parents will often describe ‘rattly breathing’ as ‘wheeze’. They are referring to movement of mucus in the larger airways rather than describing turbulent airflow through constricted lower airways.23 These rattles or ‘ruttles’ can mask accurate parental reporting of wheeze, but the two should be easily differentiated on auscultation. Wheeze rarely occurs in isolation in children and cough ± shortness of breath are also commonly found. A Danish study24 investigated approximately 3000 children aged 5 years using a parental questionnaire addressing respiratory symptoms in the previous 12 months. It reported that during this time 19.4% had one episode of wheeze, 14.4% had more than one episode of wheeze and, of those with recurrent wheeze, 25.7% also had recurrent cough. In this cohort, the risk of recurrent wheeze increased with male gender, decreasing level of maternal education, birth gestation outside 39–41 weeks, maternal history of atopy and parental smoking.
Viral aetiology of wheezing
Acute viral infections are common in infants with on average two to five lower respiratory tract infections in the first year of life.25 These can cause wheezing on their own but can also exacerbate any underlying diseases that predispose the infant to wheezing (Table 1 ). Viral-induced wheezing may require admission to hospital, but generally the illness is mild and symptoms resolve with age, so obtaining accurate information regarding the viral aetiology is difficult. In an Australian community-based cohort study,25 nasopharyngeal aspirates (NPAs) were taken from recruited infants (aged under 1 year) within 48 h of respiratory symptoms to investigate the viral aetiology of all acute respiratory infections identified over 1 year. Acute respiratory infections (ARIs) were subdivided into upper respiratory tract infections (URTIs) and lower respiratory tract infections (LRTIs), and these were further subdivided into wheezing (wLRTIs) and non-wheezing phenotypes. NPAs were analysed using a panel of 17 respiratory viruses which included rhinovirus, picornoviruses (enterovirus, echovirus, coxsackie virus), coronavirus 229E and OC43, RSV, influenza A and B, parainfluenza 1–3, human metapneumovirus (HMPV), Chlamydia pneumoniae and Mycoplasma pneumoniae. There were 984 episodes of ARI in 263 recruited children; 5.9% of all children had no ARI but most infants had between two and five episodes (median 4, range 0–11 episodes). One-third (329 of 984; 33.4%) of the ARIs were LRTIs, with 95 of 329 LRTIs (28.9%) associated with wheeze. A virus was identified in 673 of the 984 episodes (69%), with 473 (48.5%) due to rhinovirus and 106 (10.9%) due to RSV. The other viruses each caused <55 episodes of ARI. Dual infections were identified in 101 of 673 (10.3%) and three viruses in six of 673 (0.6%). Rhinoviruses were the most commonly detected virus type in wLRTIs (45.3%) compared to 16.8% for RSV. The other viruses each contributed < 5% of the total wLRTIs. Only 17 of the 984 episodes required admission to hospital: in five of these 17 the pathogen was RSV and in three rhinovirus. This study demonstrates that in infants aged under 2 years, rhinovirus is an important cause of mild recurrent wheeze whereas RSV, while being less common, may account for more severe disease. Rhinovirus is also important in older children as it was the commonest viral cause of exacerbations of asthma in older children.26
Table 1.
Differential diagnosis of recurrent wheezing in preschool infants
| System | Disease |
|---|---|
| Respiratory | Asthma, recurrent viral infections, cystic fibrosis, fibrosing alveolitis, chronic lung disease, bronchiolitis obliterans |
| Cardiovascular | AVSD, VSD, thoracic outlet obstruction |
| Gastrointestinal | GORD, cow’s milk intolerance, Congenital diaphragmatic hernia |
| Immune | |
| Tracheolaryngomalacia, TOF |
AVSD, atrioventricular septal defect; VSD, ventricular septal defect; GORD, gastro-oesophageal reflux disease; TOF, tracheo-oesophageal fistula.
Role of respiratory viruses in initiating preschool recurrent wheeze
The COAST (Childhood Origin of Asthma) study27 prospectively evaluated the contribution of viral respiratory infections to the development of recurrent wheezing during infancy in a high-risk cohort. It enrolled 289 newborns with at least one parent with respiratory allergies and/or a history of asthma from November 1998 to May 2000 and followed them for 1–3 years. All infants had nasopharyngeal mucus samples collected during clinic visits at 2, 4, 6, 9 and 12 months and during times of acute respiratory illness. These were analysed for RSV, rhinovirus, influenza A and B, parainfluenza 1–4, adenovirus and picornavirus. In the third year of life, there were 180 wheezy illness episodes among 76 infants, with a virus identified in 62%. The rates of detection of the individual viruses were comparable to other studies, with most illness episodes due to rhinovirus (76 of 180; 42%) and each of the other viruses contributing significantly fewer (RSV 8.5%, parainfluenza 8%, adenovirus 1%, influenza 1%, rhinovirus 1%, picornavirus 0.5%). Wheezing due to rhinovirus during the first year was the strongest single predictor of subsequent third-year wheezing, with 63% of infants with wheezing due to rhinovirus infection in the first year continuing to wheeze in years 1–3 compared to 20% with wheezing in the first year due to other viruses (OR 6.6).27 For all viral aetiologies there was a direct association between severity of respiratory illness in the first year (especially if associated with wheezing) and subsequent year 3 wheezing. However, frequency of respiratory illnesses in the first year did not predict wheezing at 3 years. This suggests that the strongest predictor of PSRW is a severe wheezing illness with rhinovirus in the first year of life. Other risk factors for recurrent wheezing in infants include prematurity, birthweight <2nd or >99th centile, family history of atopy, male gender, overcrowding, older siblings, attending day care, maternal smoking (especially during pregnancy) and reduced lung function at 2 weeks of age.
Association between bronchiolitis, preschool recurrent wheeze, asthma and later lung function
Three reviews including a total of 22 studies (Table 2 ) have looked at the association between RSV bronchiolitis in infancy and subsequent recurrent wheezing.28, 29, 30 The first review in 2000 by Bont et al. 28 included 13 prospective follow-up studies with a total of 1161 infants followed for up to 14 years after initial infection in the first 2 years of life. In 11 of the 13 studies where cases were hospitalized, i.e. moderate to severe bronchiolitis, they found a strong association between bronchiolitis in infancy and subsequent development of wheezing. Semple and Smyth29 reviewed the literature in 2006 and found five additional studies published since the Bont review, again showing the same association. In 2007, a systematic review by Perez-Yarza et al. 30 investigated whether RSV respiratory infections in the first 36 months of life were associated with the development of episodes of recurrent wheezing/asthma. This review included 12 studies, five of which were not included in the review by Semple and Smyth. There were four main outcomes:
-
1.
Parental reporting of recurrent wheezing was reported in eight of 12 studies and five of these showed no association between RSV bronchiolitis and parental reporting of wheezing. For the remaining three, there was an association between RSV bronchiolitis in early life and the subsequent development of recurrent wheezing but the quality of some of these studies was questioned. The study by Stein et al. 31 from the Tucson group was a well-designed prospective population-based study and showed that children with bronchiolitis had an increased risk of wheezing until 10 years of age, with most resolving by the age of 13 years, and there was no association with atopy.
-
2.
Physician-diagnosed recurrent wheezing was reported in three of 12 studies. All three showed an association between RSV bronchiolitis and physician-diagnosed recurrent wheezing which decreased with increasing age and was not associated with atopy.
-
3.
Physician-diagnosed asthma was reported in eight of 12 studies with six of these showing an association. The longest study was by Korppi et al. 32 who followed up infants for 18–20 years and found an increased risk of asthma or allergen sensitization in those children who had been hospitalized with RSV-positive bronchiolitis but were not atopic. These young adults had reduced forced expiratory volume in 1 s (FEV1) and forced vital capacity (FVC).
-
4.
Allergen sensitization, defined as a positive skin prick test and/or food-specific IgE levels, was reported in eight of 12 studies. In five of these the data were not conclusive. The study by Sigurs et al. 33 found an association between RSV bronchiolitis and the development of allergic asthma in later life with the presence of raised serum IgE antibodies or positive skin prick tests post RSV bronchiolitis in children aged 13 years. All children in this study had been hospitalized with bronchiolitis.
Table 2.
Summary of studies prospectively following infants post bronchiolitis. PFT, pulmonary function test
| Author | Year published | Bont et al. Review28 | Semple and Smyth review29 | Perez Yarza et al. review30 | Hospital admissions | Controls | Follow-up years | Outcome |
Associated with parental atopy | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Wheeze | Asthma | Lung function | |||||||||
| Eisen | 1963 | Y | Y | N | Y | N | 14 | Y 49% | N | ||
| Sims | 1978/1981 | Y/Y | Y | N | Y | Y | 8 | 56% | Reduced | N | |
| Mok | 1982 | Y | Y | N | Y | Y | 7 | Y | Reduced | N | |
| Korppi | 2004 | Y | Y | Y | Y | non-atopic | 20 | No difference | Reduced | N/A | |
| Gurwitz | 1981 | Y | Y | N | Y | N | 8 | 52% | Reduced | N/A | |
| Pulan | 1982 | Y | Y | N | Y | Y | 10 | 42% | Reduced | N | |
| Bont | 2000 | Y | Y | N | N | Y | 8 | 44% | N | ||
| Hall | 1984 | Y | Y | N | Y | Y | 8 | 45% | Reduced | N | |
| Welliver | 1993 | Y | Y | N | Y | N | 2 | 53% | Nasal IgE predicts | N/A | |
| Sly | 1989 | Y | Y | N | Y | N | 5 | 71% | N | ||
| Murray | 1992/1997 | Y/Y | Y | N | Y | N | 5 and 10 | 43% | Reduced | N | |
| Osundwa | 1993 | Y | Y | Y | Y | Y | 2 | 44% | N | ||
| Sigurs | 1995/2000/2005 | Y/Y/N | Y | Y/Y/Y | Y | Y | 3, 7, 13 | 60%, 30%, 43% | Y | ||
| Renzi | 1997 | N | Y | N | Y | N | 3 months | 65% | Th2 cytokines | N/A | |
| Bont | 2004 | N | Y | N | Y | N | 3 | Wheeze if reduced PFT | N | ||
| Stein | 1999 | N | Y | Y | N | Y | 13 | Up to 11 but not 13 years | N | ||
| Bont | 2000/2004 | Y/N | Y | N | Y | Y | 1 and 3 | IL-10 associated | N | ||
| Schauer | 2002 | Y | Y | Y | Y | Y | 1 | Y | N | ||
| Fjaerli | 2005 | N | N | Y | Y | Y | 7 | 48% | 60% | N/A | |
| Henderson | 2005 | N | N | Y | Y | Y | 7 | Y OR 2.3 | Y OR 2.5 | N/A | |
| Singleton | 2003 | N | N | Y | Y | N | 5 | N | Y RR 3.1 | N/A | |
| Weber | 1999 | N | N | Y | Y | N | 3 | Y | N/A | ||
| Juntti | 2003 | N | N | Y | N | Y | 5–10 | Y OR 0.9 | Y OR 13.4 | N | N |
In a small subgroup analysis of the Tucson Birth Cohort,34 22 infants with RSV were compared to 22 infants with non-RSV bronchiolitis, and the impact of non-RSV bronchiolitis on long-term adult respiratory morbidity was evaluated. They found, in a multivariate analysis, that those infants admitted to hospital with non-RSV bronchiolitis had an eight-fold increased risk of asthma compared with those infants with RSV bronchiolitis. There was no difference in lung function or bronchial hyperreactivity between the two groups at a median of 19 years post viral illness. The analysis of the causative virus was carried out before the availability of rapid antigen diagnostic tests.
In summary, these studies strongly suggest that RSV bronchiolitis is associated with subsequent wheezing throughout childhood with associated abnormal lung function persisting at least until early adult life. Whether RSV infection is associated with atopy or asthma is much less clear.
Further information about the outcomes of infants with PSRW is available from studies which did not specifically address associations with bronchiolits.35, 36 The Manchester Asthma and Allergy Study group35 used the Tucson predefined wheezing phenotypes of transient early wheezers, late-onset wheezers and persistent wheezers and assessed infant lung function at 3 and 5 years. They found that the risk of becoming a persistent wheezer, compared to a transient early wheezer, increased with increasing specific airway resistance (sRaw) at age 3 years (OR 5.2, 95% CI 1.3–22.0; p < 0.02). For those who had not wheezed as infants but later developed wheeze (late-onset wheezers) there was no association between lung function at age 3 or 5 years and development of recurrent wheeze after 3 years. So, although sRaw at 3 years could be used to distinguish the transient early and the persistent wheezers, it could not be used to predict which infants were to become late-onset wheezers.
Using a whole population birth cohort from the Isle of Wight,36 infants with different wheezing phenotypes were followed up for 10 years with review at 1, 2, 4 and 10 years. Analysis showed that 37% of transient early wheezers still wheezed at 10 years (persistent wheezers). These persistent wheezers had significantly more physician-diagnosed asthma in early life (p < 0.005 at 2 years), hospital admissions, use of inhaled therapies and enhanced bronchial hyperresponsiveness than early transient wheezers. However, both transient early and persistent wheezers had similar lung function tests at 10 years with reduced FEV1 and FEV1/FVC ratio compared to non-wheezers. Late-onset wheezers had normal lung function at age 10 years and lower prevalence of adverse health outcomes.
Both these studies35, 36 highlight that, as with RSV bronchiolitis, it is the development of disease in infants early in life (under 3 years) that is associated with more persistent effects on the airways rather than disease which develops later. This suggests that virus–host interactions early in life may be critical in setting the course of future illnesses.
The association between RSV and recurrent wheezing is not understood. Viral infection during a critical period in airway development may have a direct causative effect or there may be a genetic predisposition for infection or impaired respiratory reserve such that RSV, by causing bronchiolitis, identifies a small proportion of infants who will later develop recurrent wheezing or atopy.
Burrows and co-workers,37 more than 30 years ago, made the observation that children with respiratory illness were more likely to have reduced lung function tests as young adults and to develop chronic obstructive lung disease. Data from the Tucson Birth Cohort38 have shown that lung function in early adulthood, measured at a mean of 10.9 years, 16.8 years and 21.7 years, could be predicted by maximum expiratory flow at functional residual capacity (VmaxFRC) in infancy (mean 2.3 months). Infants in the lowest percentile quarter for VmaxFRC had lower FEV1/FVC ratio, FEF25–75 and FEV1 up to the age of 22 years, even after adjustment for height, weight, age and sex, than those in the upper three quartiles. Over the following years the rate of decline in FEV1 to early adulthood was greater for those infants who had had viral-induced wheeze than for those with wheeze suggestive of asthma or those without any respiratory symptoms.39 These observations may be accounted for in a number of ways. It may be that both early and later symptom complexes (including asthma) are manifestations of the same airways abnormality or disease. Early onset may either be a result of environmental factors or predisposition due to inherent susceptibility. This is slightly different from the hypothesis that, although the airways disorder/disease is the same at all ages, viral infections at this age in high-risk individuals present as wheezing disorders, i.e. RSV infection in a predisposed infant will cause bronchiolitis but in a lower risk infant will cause an upper respiratory tract infection alone. A very different explanation for these findings is that the events associated with early-onset illness cause either airway damage and remodelling of the lung and/or modulate the immune response to viruses and that these changes lead to later airway disease. Unravelling these mechanisms is currently the focus of a number of research groups worldwide.
This review of the literature has shown that infants encounter the majority of viral infections predisposing them to bronchiolits and/or PSRW during the first 2 years of life. It appears that during this time the airways are most vulnerable to damage, perhaps triggering airway remodelling and subsequent disease of the airways. In vitro and animal models suggest that viral infections have to occur in a critical time period in a susceptible infant to increase their risk of developing asthma in early childhood. Although the association between bronchiolitis, PSRW, asthma and chronic respiratory disease is still not fully elucidated, respiratory viruses appear to play a key role. We will now consider what part the innate immune response to these viruses, as the host’s first line of defence to these pathogens in the lung, plays in these diseases.
Innate immune activation and deficiency in bronchiolitis and preschool recurrent wheeze
The respiratory innate immune system comprises both cellular and non-cellular components, which limit the spread of any invading pathogens within the lung and activate the adaptive immune response to mount a specific response. Individual respiratory viruses will activate various components of the innate immune system in a variety of ways and many have evolved mechanisms to hide or utilize the host response to prolong survival. Research has shown that an over-exuberant response or deficiency in these areas due either to a constitutive abnormality or as a result of the infecting virus itself, may explain the development of severe respiratory disease in some. It may also explain why many of these infants subsequently develop recurrent wheezing and obstructive lung disease/asthma in later life. RSV is especially interesting as, despite the presence of maternally acquired antibodies, infection and re-infection can occur throughout childhood.
Airway epithelial cells
The primary site of infection and defence against the respiratory viruses causing bronchiolitis and exacerbations of PSRW is the airway epithelium. Respiratory viruses may damage the airway by direct infection and necrosis of the airway epithelium or, indirectly, by the production of a pro-inflammatory response, including several factors that promote airway and alveolar development. Infection of epithelium cells by RSV during bronchiolitis has been shown to activate their NF-κβ pathway. This leads to the induction of inflammatory chemokines and cytokines, e.g. CCL-5, CCL-2, CCL-3, CXCL-8,40 CXCL-6,41 tumour necrosis factor (TNF)-α42 and CXCL-10,43 with the subsequent recruitment of neutrophils, macrophages, dendritic cells and lymphocyte subsets into the airways. RSV prevents AEC from apoptosis by downregulating the tumour suppressor gene p53, which normally inhibits cell regulation. This causes prolonged survival of airway epithelial cells (AECs) and allows continuing viral replication.44
AECs are also thought to be the site of primary infection for rhinoviruses and studies have shown that rhinovirus can induce increased production of pro-inflammatory cytokines, chemokines and adhesion molecules.45, 46, 47
Macrophages/monocytes
Macrophages present pathogens to other cells and have a key effector role. They also have an immune suppressor effect in both the innate and adaptive immune responses to dampen down excess inflammation. Once macrophages are recruited into the lung in bronchiolitis, there is activation of the NF-κβ48 pathway, resulting in the production of pro-inflammatory mediators – tumour necrosis factor (TNF)-α, CXCL-6 and CXCL-12. In mice depleted of alveolar macrophages and then exposed to RSV there was significantly less release of inflammatory mediators and activation and recruitment of natural killer (NK) cells into the airways. However, there was little change in adaptive immune responses or long-term outcome.49 Alveolar macrophage effects are specifically caused when the RSV F protein binds to macrophages via Toll-like receptor 4 (TLR-4).50 TLR-4, as part of the universal pattern recognition system, is found on all mammalian cells, although the exact interaction between RSV and TLRs has not been clarified for all cell types. RSV infection also upregulates the expression of TLR-3 in macrophages and causes them to produce chemokines such as CCL-5.51
In vitro work has shown that rhinovirus can infect macrophages.52 Although only a small proportion of macrophages supported viral replication, infection-induced TNF-α release had a time- and dose-dependent effect. This effect in macrophages, like for RSV, was dependent on activation of the NF-κβ pathway.
Monocytes express the integrin B3 receptor, which is important for monocyte migration, adhesion and signalling, an early step in the innate immune response to respiratory viruses. An analysis of a subgroup of the children recruited into the COAST study53 showed that within the ITGB3 gene, two of 13 SNPs were associated with raised IgE at the age of 1 year, three of 13 with wheezing at 5 years and two of 13 with asthma at 6 years. The authors speculate that infants with this polymorphism may have deficiencies in monocyte recruitment, causing delayed viral clearance and thus contributing to an increased severity of PSRW and bronchiolitis.
Dendritic cells
Dendritic cells (DCs) first encounter RSV in an immature state; capture of RSV then allows them to differentiate into mature DCs and migrate back to the lymph nodes where they cause the differentiation of naïve T cells and then the proliferation of that subpopulation. Although not yet shown in vivo, in vitro infection of DCs leads to the expression of viral proteins, upregulation of co-stimulatory molecules and increased apoptosis.54 Human cord blood DCs infected in vitro with RSV showed an increased production of interleukin (IL)-10, IL-11 and prostaglandin (PG)E2 and a lower production of IL-12 compared to those infected with influenza. This shows that, although DCs have been implicated in the pathogenesis of both bronchiolitis and PSRW, their response is virus specific.55
The plasmocytoid dendritic cells (pDCs) are a specialized subset of DCs found in the circulating blood and peripheral lymph tissue. They are the major source of interferon (IFN)-α and -β production upon stimulation by virus or bacteria. IFNs play a pivotal role in inhibiting viral replication. RSV infection of pDCs has been shown to inhibit host IFN production and decrease the capacity of pDCs to activate naïve T cells, resulting in low levels of T-cell proliferation and impaired cytokine production.56 Compared to other viruses, RSV is a poor inducer of IFN-α/-β and is thus more resistant to antiviral effects. These effects are mediated via the non-structural proteins NS1 and NS2 of RSV which antagonize these antiviral IFN-induced responses.57
Natural killer cells
Natural killer cells (NK) are large granular lymphocytes that are important in the early stages of microbial infection. NK cells contain granzymes which contain serine proteases thought to be important in activating cell death pathways. Chemokines produced by infected AECs are crucial in recruiting NK cells into the lungs in bronchiolitis.58 In a mouse model of RSV bronchiolitis, NK cells were recruited into the lungs in the first 3 days of infection and were an important source of IFN-γ.59 IFN-γ allows CD8 T cells to differentiate into effector cytotoxic T cells to help in viral clearance. When RSV-infected mice were depleted of NK cells, they had prolonged shedding of the virus, showing that NK cells are important in viral clearance.60 The response to RSV was evaluated in older mice who showed diminished NK cell activity and cytotoxic T-cell response, demonstrating that age-related or acquired immune dysfunction in the NK pathway leads to a more severe response to infection.58 Clinical studies of NK cells are difficult due to the problems in isolating them. In infants with severe RSV disease, NK cells have an increased expression of granzyme B, suggesting they are activated and modulating cell death pathways.61
Neutrophils
During RSV bronchiolitis the predominant cell in the airways is the neutrophil, contributing to >75% of the recruited cells.62 Their role in the disease pathogenesis is unclear but they have been shown to be activated in the airways of infants with bronchiolitis, to be associated with increased production of cytokines40 (such as CXCL-963) and to have prolonged survival due to delayed apoptosis.64 Intriguingly, CXCL-9, a type 2 cytokine thought to be produced by neutrophils, is known to induce mucus production by AECs and cause goblet cell hyperplasia and chemokine secretion both by neutrophils and AECs. Neutrophils are also the predominant cell in viral exacerbations of asthma, so understanding their role in bronchiolitis may help to determine their role in these other airway diseases.
Toll-like receptors
Toll-like receptors (TLRs) are important pattern recognition molecules found on all mammalian cells. They help to initiate the innate immune response and mediate signalling between the innate and the adaptive immune system. TLR-4 null mice infected with RSV showed impaired pulmonary NK cell and CD14+ cell trafficking, deficient NK cell function, reduced CXCL-12 and impaired virus clearance.65 In comparison, TLR-3 null mice infected with RSV had increased mucus production and CXCL-13 and CXCL-5, and an accumulation of eosinophils in the airways.66 TLR-4 mutations, Asp299 and Gly399, have been shown to be associated with severe RSV bronchiolitis.67 However, the role of TLRs in the pathogenesis of bronchiolitis is still to be elucidated. Mutations and variations in the gene expression of TLR-4 and other TLRs may explain the variations in clinical presentation among infants with bronchiolitis.
Surfactant proteins
Surfactant proteins SPA and D are produced by alveolar and airway epithelial cells and help prevent RSV infecting its targets by binding to RSV F and G.68 BAL fluid from infants ventilated with RSV show reduced levels of SPA, B and D.69 Polymorphisms in both SPA and D have been linked to severe bronchiolitis70, 71 and SPA-deficient mice have more severe RSV bronchiolitis than wild-type siblings.72 This is due to longer viral persistence in the lungs and implicates surfactant proteins in viral clearance.
Cytokines
Many cytokines, with both pro- and anti-inflammatory roles, are produced by all cells of the innate immune system. RSV infection provokes an intense release of pro-inflammatory cytokines, especially CXCL-6 and TNF-α, which decline as the infant recovers.41 IFN-γ has been inversely associated with severe bronchiolitis. In a mouse study73 comparing wild-type and IFN-γ-deficient mice, IFN-γ production in the former during initial infection prevented re-infection with RSV and subsequent development of bronchial hyperresponsiveness, unlike in the IFN-γ-deficient mice.73 The COAST study74 looked at the innate immune system at birth and followed the infant cohort through to 3 years of age to see if any abnormalities were predictive of subsequent respiratory disease. There was an inverse relationship between neonatal cord blood phytohaemagglutinin (PHA)-induced IFN-γ responses and the number of moderate and severe viral infections per year. Vigorous secretion of IFN-γ in the cord blood was associated with fewer viral infections in the first three years of life. There was no association with other PHA-induced responses, including CXCL-5, CXCL-10 and CXCL-13. Differences in IFN-γ secretion were found to be gender specific with boys having greater responses than girls.75 This suggests that antenatal factors are important in determining susceptibility to PSRW and later development of asthma by reducing production of IFN-γ.
Placental production of cytokines may protect both the immunological function of the placenta and the developing baby. Abnormalities in the feto-placental unit may predict those infants at subsequent risk of severe bronchiolitis or PSRW. To investigate this Macaubas et al. 76 collected cord blood from 2860 live births and followed up 2602 of these infants to the age of 6 years. At 6 years of age they found an association between having had detectable concentrations of CXCL-4 and IFN-α at birth in cord blood and lower risk of physician-diagnosed asthma (OR 0.6, 95% CI 0.37–0.99) and current wheeze (OR 0.55, 95% CI 0.32–0.93). In this cohort, lower production of CXCL-4 and IFN-α in the feto-placental unit increased the susceptibility to PSRW/asthma.
Chemokines
The CC chemokines, which include CCL-5 and CCL-3, are important pro-inflammatory mediators produced by infected AECs. Both are ligands for the receptor CCR-5. A promoter SNP in CCR-5 is associated with severe RSV disease (OR 1.25; p = 0.01).77
The CXCL chemokines include CXCL-8, a powerful neutrophil chemoattractant found in large amounts in the BAL of infants with severe RSV bronchiolitis. The amount of CXCL-8 in the airways correlates with severity of RSV bronchiolitis.43, 78 Hull et al. 79 showed that one SDP in particular, SNP il-8–251, correlated with increased CXCL-8 production and more severe RSV disease.
CXCL-10 is a relatively little studied chemokine but is found in large quantities (four times that of CXCL-8 and 140 times that of CCL-3) in the airways of infants with severe RSV bronchiolitis.43 It is secreted by neutrophils, monocytes and T cells in response to IFN-γ and it acts to recruit monocytes into the lung to help phagocytose apoptotic neutrophils. It could be speculated that deficiencies in the CXCL-10 pathway lead to the lung being exposed to harmful effects from apoptotic neutrophils not being phagocytized and instead releasing their contents into the lung milieu.
RSV glycoprotein (G protein) has been found to mimic the chemokine CXCL-1, which mediates the chemotaxis of leukocytes expressing the receptor CX3CR1. In samples taken from infants with RSV bronchiolitis there were significantly lower levels of CX3CR1 in CD8 T cells in the convalescent phase (6–8 weeks post infection) than in healthy controls. This suggests that there is ongoing RSV infection or inflammation. Since CXC3CR1 levels reflect efficient cytotoxic T-cell function, their lower level in convalescence indicate an impaired immune response.80
Summary
Bronchiolitis and PSRW have a huge socioeconomic impact on infants and their families in the preschool years. Bronchiolitis is a well defined clinical condition whereas PSRW is a heterogeneous group of wheezy disorders with at least four defined phenotypes based on the age that wheeze appears, disappears or persists.
Respiratory viruses, especially RSV and rhinovirus, cause on average two to five respiratory tract infections per year in infants and play a significant role in causing both bronchiolitis and PSRW.25 Although both viruses preferentially infect the respiratory tract, their disease phenotypes are different. The majority of cases of bronchiolitis are caused by RSV whereas rhinovirus tends to cause mild recurrent wheeze.
There is a strong association between the development of recurrent wheeze and previous bronchiolitis but the evidence is still not clear whether either subsequently leads to the development of asthma. The earlier and the more severe the initial viral respiratory tract infection, the greater the likelihood of subsequent respiratory pathology and abnormal lung function. Early abnormalities in lung function are predictive of the development of later airways disease.
The pathogenesis of both bronchiolitis and PSRW is not fully understood but it is clear that the innate immune response plays an important role in limiting viral infections. Both cellular and non-cellular components of the innate immune system are activated in bronchiolitis and PSRW. Defects in some components of the innate immune system have been shown to be associated with the development of more severe disease or the host’s inability to clear the respiratory virus.
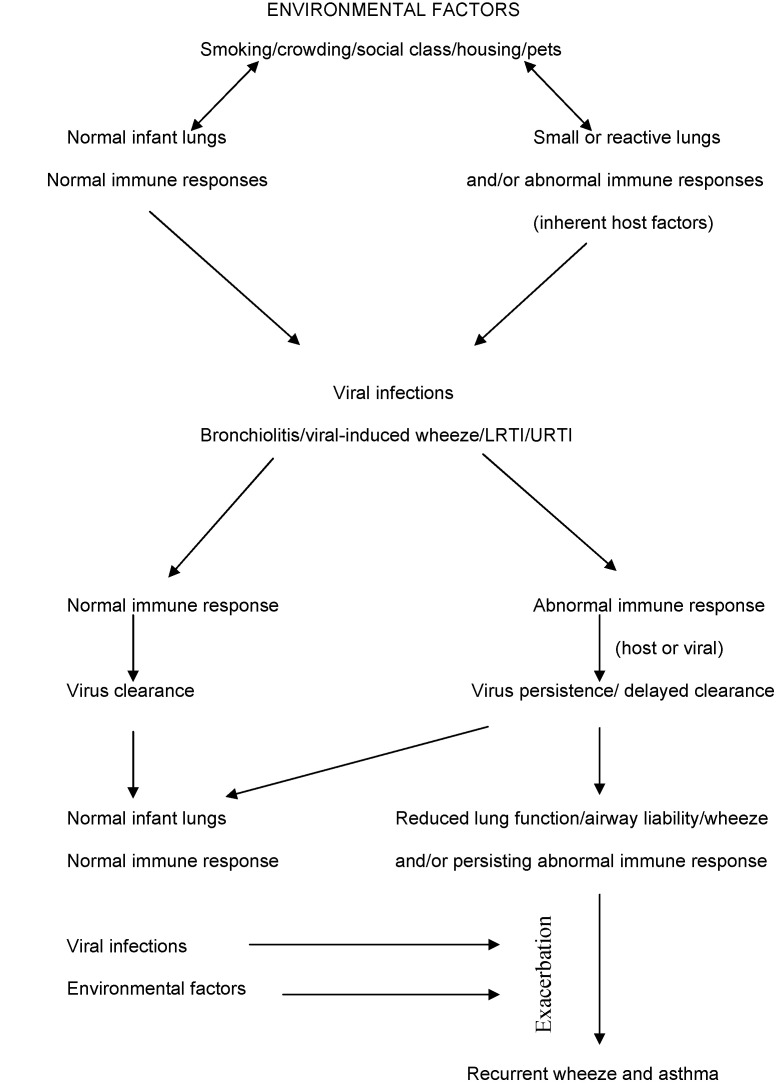
In summary (Fig. 2 ), many factors contribute to the development of respiratory disease. This review shows that an intact innate immune system is needed to maintain healthy airways and protect them from viral infections during a critical period of lung development in infancy. Defects, either due to an inherent defect or as a direct result of the virus, increase severity of bronchiolitis and PSRW. This then increases the risk of subsequent respiratory disease, either into childhood or early adulthood, causing significant morbidity and mortality.
Figure 2.
Interactions between respiratory viruses, environmental factors, bronchiolitis, preschool recurent wheeze, the innate immune system and the subsequent development of airways disease. LRTI, lower respiratory tract infection; URTI, upper respiratory tract infection.
Practice points
-
1.
RSV causes at least 70% of cases of bronchiolitis; 1–2% of all affected infants require hospitalization.
-
2.
RSV can activate the innate immune system by infecting airway epithelial cells, macrophages and dendritic cells, leading to upregulation of function and production of cytokines. It also activates neutrophils and NK cells.
-
3.
Deficiencies in TLRs, SPA and D, and IL-8 role in the innate immune system have been shown to be associated with severe RSV disease.
-
4.
Rhinovirus is a common cause of both upper and lower respiratory tract illness, especially PSRW: 30% of all infants under 3 years experience at least one episode of wheezing.
-
5.
Rhinovirus has been shown to infect both airway epithelial cells and macrophages, leading to their activation, and differences in innate immune response have been shown between infants who develop asthma and those who are non-atopic.
-
6.
Deficiencies in cytokine production at birth are associated with subsequent risk of wheezing.
-
7.
In the long term there is an increased risk of recurrent wheezing post RSV bronchiolitis. A significant number of infants with PSRW will continue to wheeze into adolescence. This may represent a different phenotype from those who wheeze transiently.
References
- 1.Smyth R.L., Openshaw P.J. Bronchiolitis. Lancet. 2006;368:312–322. doi: 10.1016/S0140-6736(06)69077-6. [DOI] [PubMed] [Google Scholar]
- 2.Murray C.J., Lopez A.D. Global mortality and disability and contribution of risk factors. Global Burden of Disease study. Lancet. 1997;34:1436–1442. doi: 10.1016/S0140-6736(96)07495-8. [DOI] [PubMed] [Google Scholar]
- 3.Chanock R.M., Finberg L. Recovery from infants with respiratory illness of a virus related to chimpanzee coryzal agent (CCA). Epidemiological aspects of infection in infants and young children. Am J Hyg. 1957;66:291–300. doi: 10.1093/oxfordjournals.aje.a119902. [DOI] [PubMed] [Google Scholar]
- 4.Jartti T., Lehtinen P., Vuorinen T. Respiratory picornoviruses and respiratory syncytial virus as causative agents of acute wheezing in children. Emerg Infect Dis. 2004;10:1095–1101. doi: 10.3201/eid1006.030629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kahn J.S. Newly discovered respiratory viruses: significance and implications. Curr Opin Pharmacol. 2007;7:1–6. doi: 10.1016/j.coph.2007.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pierangeli A., Gentile M., Di Marco P. Detection and typing of molecular techniques of respiratory viruses in children hospitalised for acute respiratory infection in Rome, Italy. J Med Virol. 2007;79:463–468. doi: 10.1002/jmv.20832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Semple M.G., Cowell A., Dove W. Dual infection of infants with human metapneumovirus and human respiratory syncytial virus is strongly associated with severe bronchiolitis. J Infect Dis. 2005;191:382–386. doi: 10.1086/426457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.McNamara P.S., Flanagan B.F., Smyth R.L., Hart C.A. Impact of HMPV and RSV co-infection in severe bronchiolitis. Pediatr Pulmonol. 2007;42:740–743. doi: 10.1002/ppul.20649. [DOI] [PubMed] [Google Scholar]
- 9.Thorburn K., Harigopal S., Reddy V., Taylor N., Van Saene H.K. High incidence of pulmonary bacterial co-infection in children with severe respiratory syncytial virus (RSV) bronchiolitis. Thorax. 2006;61:611–615. doi: 10.1136/thx.2005.048397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Openshaw P.J., Dean G.S., Culley F.J. Links between RASV bronchiolitis and childhood asthma, clinical and research approaches. Pediatr Infect Dis. 2003;22(Suppl 2):s58–s64. doi: 10.1097/01.inf.0000053887.26571.eb. [DOI] [PubMed] [Google Scholar]
- 11.Manoha C., Espinosa S., Aho S.L., Huet F., Pothier P. Epidemiological and clinical features of hMPV, RSV and RV infections in young children. J Clin Virol. 2007;38:221–226. doi: 10.1016/j.jcv.2006.12.005. [DOI] [PubMed] [Google Scholar]
- 12.Simoes E.A., Carbonell-Estrany X. Impact of severe disease caused by respiratory syncytial viral infection in children living in developed countries. Pediatr Infect Dis J. 2003;22(Suppl 2):s13–s18. doi: 10.1097/01.inf.0000053881.47279.d9. [DOI] [PubMed] [Google Scholar]
- 13.Fletcher J.N., Smyth R.L., Thomas H.M., Ashby D., Hart C.A. Respiratory syncytial genotypes and disease severity among children in hospital. Arch Dis Child. 1997;77:508–511. doi: 10.1136/adc.77.6.508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Panickar J.R., Dodd R.S., Smyth R.L., Couriel J.M. Trends in respiratory illness in children in England and Wales from 1968 to 2000. Thorax. 2005;60:586–590. doi: 10.1136/thx.2005.044750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kim H.W., Arrobio J.O., Brandt C.D. Epidemiology of RSV infection in Washington. Importance of the virus in different respiratory tract disease syndromes and temporal distribution of infection. Am J Epidemiol. 1973;98:216–225. doi: 10.1093/oxfordjournals.aje.a121550. [DOI] [PubMed] [Google Scholar]
- 16.Glezen W.P., Taber L.H., Frank A.L., Kasel J.A. Risk of primary infection and reinfection with respiratory syncytial virus. Am J Dis Child. 1986;140:543–546. doi: 10.1001/archpedi.1986.02140200053026. [DOI] [PubMed] [Google Scholar]
- 17.Eisenhut M. Extra pulmonary manifestations of severe respiratory syncytial virus infection – a systematic review. Crit Care. 2006;10:R107. doi: 10.1186/cc4984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Taussig L.M., Wright A.L., Morgan W.J., Harrison H.R., Ray C.G. The Tucson Children’s Respiratory Study. 1. Design and implementation of a prospective study of acute and chronic respiratory illness in children. Am J Epidemiol. 1989;129:1219–1231. doi: 10.1093/oxfordjournals.aje.a115242. [DOI] [PubMed] [Google Scholar]
- 19.Martinez F.D., Wright A.L., Taussig L.M., Holberg C.J., Halonen M., Morgan W.J. Asthma and wheezing in the first six years of life. N Engl J Med. 1995;332:133–138. doi: 10.1056/NEJM199501193320301. [DOI] [PubMed] [Google Scholar]
- 20.Stein R.T., Holberg C.J., Morgan W.J. Peak flow variability, metacholine responsiveness and atopy as markers for detecting different wheezing phenotypes in childhood. Thorax. 1997;52:946–952. doi: 10.1136/thx.52.11.946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sheriff A., Peters T.J., Henderson J., Strachan D., and the ALSPAC study group Risk factor associations in children followed longitudinally from birth to 3½ years. Int J Epidemiol. 2001;30:1473–1484. doi: 10.1093/ije/30.6.1473. [DOI] [PubMed] [Google Scholar]
- 22.Henderson J., Sherriff A., Farrow A., Ayres J.G. Household chemicals, persistent wheezing and lung function: effect modification by atopy? Eur Respir J. 2008;31:547–554. doi: 10.1183/09031936.00086807. [DOI] [PubMed] [Google Scholar]
- 23.Elphick H.E., Ritson S., Rogers H., Everard M.L. When a wheeze is not a wheeze; acoustic analysis of breath sounds. Eur Respir J. 2000;16:593–597. doi: 10.1034/j.1399-3003.2000.16d04.x. [DOI] [PubMed] [Google Scholar]
- 24.Hermann C., Westergaard T., Pedersen B.V., Wohlfahrt J., Host A., Melbye M. A comparison of risk factors for wheeze and recurrent cough in preschool children. Am J Epidemiol. 2005;162:345–350. doi: 10.1093/aje/kwi212. [DOI] [PubMed] [Google Scholar]
- 25.Kusel M.M., de Klerk N.H., Holt P.G., Kebadze T., Johnson S.L., Sly P.D. Role of respiratory viruses in acute upper and lower respiratory tract illness in the first year of life: a birth cohort. Paediatr Infect Dis J. 2006;25:680–686. doi: 10.1097/01.inf.0000226912.88900.a3. [DOI] [PubMed] [Google Scholar]
- 26.Johnston S.L., Pattemore P.K., Sanderson G. Community study of viral infections in exacerbations of asthma in 9–11 year old children. BMJ. 1995;310:1225–1229. doi: 10.1136/bmj.310.6989.1225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lemanske R.F., Jackson D.J., Gangnon R.E. Rhinovirus infection during infancy predicts subsequent childhood wheezing. J Allergy Clin Immunol. 2005;116:571–577. doi: 10.1016/j.jaci.2005.06.024. [DOI] [PubMed] [Google Scholar]
- 28.Bont L., Stein M., Kimpen J.L. Long term consequences of RSV bronchiolitis. Paediatr Respir Rev. 2000;1:221–227. doi: 10.1053/prrv.2000.0052. [DOI] [PubMed] [Google Scholar]
- 29.Semple MG, Smyth RL. Lessons from hRSV bronchiolitis and acute wheezing episodes in infants. In: Exacerbations of asthma. Johnston SL, O’Bryne PM. (Eds.), Informa Healthcare, Oxon, UK, chapter 11, pp 187–203.
- 30.Perez-Yarza E., Moreno A., Lazaro P., Mejias A., Ramilo O. The association between RSV infection and development of childhood asthma: a systematic review. Paediatr Infect Dis J. 2007;26:733–739. doi: 10.1097/INF.0b013e3180618c42. [DOI] [PubMed] [Google Scholar]
- 31.Stein R.T., Sherrill D., Morgan W.J. RSV in early life and risk of wheeze and allergy by 13 years. Lancet. 1999;354:541–545. doi: 10.1016/S0140-6736(98)10321-5. [DOI] [PubMed] [Google Scholar]
- 32.Korppi M., Piipo-Salolainen E., Korhonen K., Remes S. Respiratory morbidity 20 years after RSV infection in infancy. Pediatr Pulmonol. 2004;38:155–160. doi: 10.1002/ppul.20058. [DOI] [PubMed] [Google Scholar]
- 33.Sigurs N., Gustafsson P.M., Bjarnason R. Severe RSV bronchiolitis in infancy and asthma and allergy at age 13. Am J Respir Crit Care Med. 2005;171:137–141. doi: 10.1164/rccm.200406-730OC. [DOI] [PubMed] [Google Scholar]
- 34.Piipo-Salolainen E., Korppi M., Korhonen K., Remes S. Adult asthma after non-respiratory synctyial virus bronchiolitis in infancy: subgroup analysis of the 20 year prospective follow up study. Pediatr Int. 2007;49:190–195. doi: 10.1111/j.1442-200X.2007.02340.x. [DOI] [PubMed] [Google Scholar]
- 35.Lowe L.A., Simpson A., Woodcock A., Morris J., Murray C.S., Xustovic A Wheeze phenotypes and lung function in preschool children. Am J Respir Crit Care. 2005;171:231–237. doi: 10.1164/rccm.200406-695OC. [DOI] [PubMed] [Google Scholar]
- 36.Kurukulaaratchy R.J., Fenn M.H., Waterhouse L.M., Matthews S.M., Holgate S.T., Arshad S.H. Characterisation of wheezing phenotypes in the first 10 years of life. Clin Exp Allergy. 2003;33:573–578. doi: 10.1046/j.1365-2222.2003.01657.x. [DOI] [PubMed] [Google Scholar]
- 37.Burrows B., Knudson R.J., Lebowitz M.D. The relationship of childhood respiratory illness to adult obstructive airway disease. Am Rev Respir Dis. 1977;115:751–760. doi: 10.1164/arrd.1977.115.5.751. [DOI] [PubMed] [Google Scholar]
- 38.Stern D.A., Morgan W.J., Wright A.L., Guerra S., Martinez F.D. Poor airway function in early infancy and lung function by age 22 years: a non-selective longitudinal cohort study. Lancet. 2007;370:758–764. doi: 10.1016/S0140-6736(07)61379-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Brey U., Silverman M., Kraemer R., Jackson A.C. High frequency respiratory impedance measured by forced oscillation technique in infants. Am J Respir Crit Care Med. 1998;158:363–370. doi: 10.1164/ajrccm.158.2.9703038. [DOI] [PubMed] [Google Scholar]
- 40.McNamara P.S., Flanagan B.F., Hart C.A., Smyth R.L. Production of chemokines in the lungs of infants with severe respiratory syncytial virus bronchiolitis. J Infect Dis. 2005;191:1225–1232. doi: 10.1086/428855. [DOI] [PubMed] [Google Scholar]
- 41.McNamara P.S., Flanagan B.F., Selby A., Hart C.A., Smyth R.L. Pro- and anti-inflammatory responses in respiratory syncytial virus bronchiolits. Eur Respir J. 2004;23:106–112. doi: 10.1183/09031936.03.00048103. [DOI] [PubMed] [Google Scholar]
- 42.Bitko V., Velaquez A., Yang L. Transcriptional induction of multiple cytokines by human respiratory syncytial virus requires activation of NF-kappa-B and is inhibited by sodium salicyclate and aspirin. Virology. 1997;2322:369–378. doi: 10.1006/viro.1997.8582. [DOI] [PubMed] [Google Scholar]
- 43.McNamara P.S., Flanagan B.F., Hart C.A., Smyth R.L. Production of chemokines in the lungs of infants with severe RSV bronchiolitis. J Infect Dis. 2005;191:1225–1232. doi: 10.1086/428855. [DOI] [PubMed] [Google Scholar]
- 44.Groskreutz D.J., Monick M.M., Yarovinsky T.O. RSV decreases p53 protein to prolong survival of airway epithelial cells. J Immunol. 2007;179:2741–2747. doi: 10.4049/jimmunol.179.5.2741. [DOI] [PubMed] [Google Scholar]
- 45.Johnston S.L., Papi A., Bates P.J., Mastronarde J.G., Monick M.M., Hunninghake G.W. Low grade rhinovirus infection induces a prolonged release of IL-8 in pulmonary epithelium. J Immunol. 1998;160:6172–6181. [PubMed] [Google Scholar]
- 46.Papi A., Johnston S.L. Rhinovirus infection induces expression of its own receptor intracellular adhesion molecule 1 (ICAM 1) via increased NK-κβ mediated transcription. J Biol Chem. 1999;274:9707–9720. doi: 10.1074/jbc.274.14.9707. [DOI] [PubMed] [Google Scholar]
- 47.Schroth M.K., Grimm E., Frindt P. Rhinovirus replication causes RANTES production in primary bronchial epithelial cells. Am J Respir Cell Mol Biol. 1999;20:1220–1228. doi: 10.1165/ajrcmb.20.6.3261. [DOI] [PubMed] [Google Scholar]
- 48.Haerberle H.A., Takizawa R., Casola A. Respiratory syncytial virus activation of nuclear factor-kappa B in the lung involves alveolar macrophages and Toll like receptor 4-dependant pathways. J Infect Dis. 2002;1869:1199–1206. doi: 10.1086/344644. [DOI] [PubMed] [Google Scholar]
- 49.Pribul P.K., Harker J., Wang B. Alveolar macrophages are a major determinant of early responses to viral ling infection but do not influence subsequent disease development. J Virol. 2008;82(9):4441–4448. doi: 10.1128/JVI.02541-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kurt-Jones E.A., Popova L., Kwinn L. Pattern recognition receptors TLR4 and CD14 mediate responses to respiratory syncytial virus. Nat Immunol. 2000;1:398–401. doi: 10.1038/80833. [DOI] [PubMed] [Google Scholar]
- 51.Rudd B.D., Burstein E., Duckett C.S., Li X., Lukacs N.W. Differential role for TLR3 in respiratory syncytial virus induced chemokines expression. J Virol. 2005;796:3350–3357. doi: 10.1128/JVI.79.6.3350-3357.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Laza-Stanca V., Stanciu L.A., Message S.D., Edwards M.R., Gern J.E., Johnston S.L. Rhinovirus replication in human macrophages induces NF-κβ dependant tumour necrosis factor alpha production. J Virol. 2006;80:8248–8258. doi: 10.1128/JVI.00162-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Thompson E.E., Pan L.P., Ostrovnaya I. Integrin β3 genotype influences asthma and allergy phenotypes in the first 6 years of life. J Allergy Clin Immunol. 2007;119:1423–1429. doi: 10.1016/j.jaci.2007.03.029. [DOI] [PubMed] [Google Scholar]
- 54.de Graff P.M., de Jong E.C., Van Capel T.M. Respiratory syncytial virus infection of monocyte derived dendritic cells decreases their capacity to activate CD4 T cells. J Immunol. 2005;1759:5904–5911. doi: 10.4049/jimmunol.175.9.5904. [DOI] [PubMed] [Google Scholar]
- 55.Bartz H., Buning-Pfaue F., Turkel O., Schauer U. Respiratory syncytial virus induces prostaglandin E2, Il-10 and Il-11 generation in antigen presenting cells. Clin Exp Immunol. 2002;1293:438–445. doi: 10.1046/j.1365-2249.2002.01927.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Schlender J., Hornung V., Finke S. Inhibition of Toll like receptor 7 and 9 mediated alpha/beta interferon production in human plasmocytoid dendritic cells by respiratory syncytial virus and measles virus. J Virol. 2005;799:5507–5515. doi: 10.1128/JVI.79.9.5507-5515.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Schlender J., Bossert B., Bucholz U., Conzelmann K.-K. Bovine respiratory syncytial virus non-structural proteins NS1 and NS2 cooperatively antagonise alpha/beta interferon induced antiviral response. J Virol. 2000;74:8234–8242. doi: 10.1128/jvi.74.18.8234-8242.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Liu B., Kimura Y. Local immune response to respiratory Syncytial virus infection is diminished in senescence-accelerated mice. J Gen Virol. 2007;88:2552–2558. doi: 10.1099/vir.0.83089-0. [DOI] [PubMed] [Google Scholar]
- 59.Hussell T., Openshaw P.J. Intracellular IFN-gamma expression in natural killer cells preceded CD8+ T cell recruitment during respiratory syncytial virus infection. J Gen Virol. 1998;791:2593–2601. doi: 10.1099/0022-1317-79-11-2593. [DOI] [PubMed] [Google Scholar]
- 60.Harrop J.A., Anderson J.J., Hayes P., Serin N., Scott R. Characteristics of the pulmonary natural killer (NK) cell response to respiratory syncytial virus infection in the BALB/C mice. Immunol Infect Dis. 1994;4:179. [Google Scholar]
- 61.Bem R.A., Bos A.P., Bots M. Activation of the Granzyme pathway in children with severe respiratory syncytial virus infection. Pediatr Res. 2008 Feb 27 doi: 10.1203/PDR.0b013e31816fdc32. Epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.McNamara P.S., Ritson P., Selby A., Hart C.A., Smyth R.L. Bronchoalveolar lavage cellularity in infants with severe respiratory Syncytial virus bronchiolitis. Arch Dis Child. 2003;88:922–926. doi: 10.1136/adc.88.10.922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.McNamara P.S., Flanagan B.F., Baldwin L.M., Newland P., Hart C.A., Smyth R.L. Interleukin-9 production in the lungs of infants with severe respiratory syncytial virus bronchiolitis. Lancet. 2004;363:1031–1037. doi: 10.1016/S0140-6736(04)15838-8. [DOI] [PubMed] [Google Scholar]
- 64.Lindemans C.A., Coffer P.F., Schellens I.M., de Graff P.M., Kimpen J.L., Koenderman L. Respiratory syncytial virus inhibits granulocyte apoptosis through a phosphatidylinositol 3-kinase and NF-kappa B dependant mechanism. J Immunol. 2006;176:5529–5537. doi: 10.4049/jimmunol.176.9.5529. [DOI] [PubMed] [Google Scholar]
- 65.Haynes L.M., Moore M.D., Kurt-Jones E.A., Finberg R.W., Anderson L.J., Tripp R.A. Involvement of Toll like receptor 4 expression in infants with respiratory syncytial virus. J Virol. 2001;75:10730–10737. doi: 10.1128/JVI.75.22.10730-10737.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Rudd B.D., Smit J.J., Flavell R.A. Deletion of TLR3 alters the pulmonary immune environment and mucus production during respiratory syncytial virus infection. J Immunol. 2006;1763:1937–1942. doi: 10.4049/jimmunol.176.3.1937. [DOI] [PubMed] [Google Scholar]
- 67.Tal G., Mandelberg A., Dalal I. Association between common Toll like receptor 4 mutations and severe respiratory syncytial virus disease. J Infect Dis. 2004;189:2057–2063. doi: 10.1086/420830. [DOI] [PubMed] [Google Scholar]
- 68.Hickling T.P., Bright H., Wing K. A recombinant trimetric surfactant protein D carbohydrate recognition domain inhibits respiratory syncytial virus infection in vitro and in vivo. Eur J Immunol. 1999;29:3478–3484. doi: 10.1002/(SICI)1521-4141(199911)29:11<3478::AID-IMMU3478>3.0.CO;2-W. [DOI] [PubMed] [Google Scholar]
- 69.Kerr M.H., Paton J.Y. Surfactant protein levels in severe respiratory syncytial virus infection. Am J Respir Crit Car Med. 1999;159:1115–1118. doi: 10.1164/ajrccm.159.4.9709065. [DOI] [PubMed] [Google Scholar]
- 70.Lahti M., Lofgren J., Matilda R. Surfactant protein D polymorphism associated with severe respiratory syncytial virus infection. Pediatr Res. 2002;51:696–699. doi: 10.1203/00006450-200206000-00006. [DOI] [PubMed] [Google Scholar]
- 71.Lofgren J., Ramet M., Renko M., Marttila R., Hallman M. Association between surfactant protein A gene locus and severe respiratory syncytial virus infection in infants. J Infect Dis. 2002;185:283–289. doi: 10.1086/338473. [DOI] [PubMed] [Google Scholar]
- 72.Levine A.M., Gwozdz J., Stark J., Bruno M., Whitsett J., Korfhagen T. Surfactant protein A enhances respiratory Syncytial virus clearance in vitro. J Clin Invest. 1999;103:1015–1021. doi: 10.1172/JCI5849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lee Y.M., Miyahara N., Takeda K. IFN-gamma production during initial infection determines the outcome with RSV. Am J Respir Crit Care Med. 2008;177:208–218. doi: 10.1164/rccm.200612-1890OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Lemanske R.F. The childhood origins of asthma (COAST) study. Pediatr Allergy Immunol. 2002;13(Suppl 15):38–43. doi: 10.1034/j.1399-3038.13.s.15.8.x. [DOI] [PubMed] [Google Scholar]
- 75.Uekert S.J., Akan G., Evans M.D. Sex-related differences in immune development and the expression of atopy in early childhood. J Allergy Clin Immunol. 2006;118:1375–1381. doi: 10.1016/j.jaci.2006.09.008. [DOI] [PubMed] [Google Scholar]
- 76.Macaubas C., de Klerk N.H., Holt B.J. Association between antenatal cytokine production and the development of atopy and asthma at age 6 years. Lancet. 2003;362:1192–1197. doi: 10.1016/s0140-6736(03)14542-4. [DOI] [PubMed] [Google Scholar]
- 77.Hull J., Rowlands K., Lockhart E. Variants of the chemokines receptor CCR5 are associated with severe bronchiolitis caused by respiratory syncytial virus. J Infect Dis. 2003;188:904–907. doi: 10.1086/377587. [DOI] [PubMed] [Google Scholar]
- 78.Smyth R.L., Mobbs K.J., O’SHea U., Ashby D., Hart C.A. Respiratory syncytial virus bronchiolitis, disease severity, interleukin -8 and virus genotype. Paediatr Pulmonol. 2002;33:339–346. doi: 10.1002/ppul.10080. [DOI] [PubMed] [Google Scholar]
- 79.Hull J., Thomson A., Kwiatkowski D. Association of respiratory syncytial virus bronchiolitis with the interleukin 8 gene region in UK families. Thorax. 2000;55:1023–1027. doi: 10.1136/thorax.55.12.1023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Ceprika A.M., Gagro A., Bace A. Expression of chemokines receptor CX3XC1 in infants with respiratory syncytial virus bronchiolitis. Pediatr Allergy Immunol. 2008;19:148–156. doi: 10.1111/j.1399-3038.2007.00611.x. [DOI] [PubMed] [Google Scholar]