Highlights
-
•
Dynamic cerebral blood flow autoregulation is impaired on the first day following cardiac surgery.
-
•
Autoregulation index before and after surgery is predictive of post-operative delirium.
-
•
Patients with impaired dynamic cerebral autoregulation at 24 h and 7 days after surgery had a higher incidence of delirium.
Keywords: Cardiac surgery, Cardiopulmonary bypass, Cerebral blood flow, Left ventricular ejection fraction
Abstract
Objective
We investigated the potential association of cerebral autoregulation (CA) with postoperative delirium (PD), a common complication of cardiac surgery with cardiopulmonary bypass (CPB).
Methods
In patients undergoing coronary artery bypass graft (CABG) surgery with CPB, cerebral blood flow velocity (CBFV) and blood pressure (BP) were continuously recorded during 5-min preoperatively (T1), after 24 h (T2), and 7 days after procedure (T3). Prospective multivariate logistic regression analysis was performed to determine the independent risk factors of PD. Autoregulation index (ARI) was calculated from the CBFV response to a step change in BP derived by transfer function analysis.
Results
In 67 patients, mean age 64.3 ± 9.5 years, CA was depressed at T2 as shown by values of ARI (3.9 ± 1.7), compared to T1 (5.6 ± 1.7) and T3 (5.5 ± 1.8) (p < 0.001). Impaired CA was found in 37 (55%) patients at T2 and in 7 patients (20%) at T3. Lower ARI at T1 and T2 were predictors of PD (p = 0.003).
Conclusion
Dynamic CA was impaired after CABG surgery with CPB and was a significant independent risk factor of PD.
Significance
Assessment of CA before and after surgery could have considerable potential for early identification of patients at risk of PD, thus reducing poor outcomes and length of stay.
Clinical trials registration: www.clinicaltrials.gov (NCT02143544, April 30, 2014).
1. Introduction
Post-operative delirium (PD), defined as a disorder of consciousness, characterized by fluctuations in cognition, apathy and non-organized thinking, occurring over a short period of time, is a common complication of heart surgery with cardiopulmonary bypass (CPB) and has been associated with morbimortality, as well as with higher healthcare costs (Inouye, 2006, Deiner and Silverstein, 2009, Brown, 2014). Though delirium may resolve during the hospital stay, what may be more serious is its association with long-term functional and cognitive impairment (Saczynski et al., 2012). Early and accurate prediction of PD could help more effective prevention and treatment.
Cardiac surgery with CPB can provoke fluctuations in cerebral blood flow (CBF) that can lead to cerebral ischemia or hyper-perfusion (Scolletta et al., 2015, Caldas et al., 2017). Excessive cerebral microembolic load, endothelial injury, a compromised blood-brain barrier, impairment of cerebral autoregulation and changes in cerebral oxygenation are potential factors for CBF disturbances, during or after CABG, and might also contribute to delirium susceptibility (Moody et al., 1995, Plaschke et al., 2010, Siepe et al., 2011, Brown, 2014, Hori et al., 2014, Mailhot et al., 2016). However, the role of CBF regulatory mechanisms, both before, during and after surgery, is still unknown and the mechanisms underlying PD are not elucidated.
Cerebral autoregulation (CA) defines the brain's capacity to provide stable CBF despite fluctuations in mean arterial blood pressure (BP) over the range of 60–150 mmHg. Impairment of CA can lead to disturbances in CBF that could play a role in the occurrence of PD due to brain ischemia or hyper-perfusion causing edema and capillary damage (Paulson et al., 1990). CA assessments are generally classified as being ‘static’ or ‘dynamic’ and can be performed by measuring CBF velocity of the middle cerebral arteries (MCA) with transcranial Doppler (TCD), simultaneously with BP (Tiecks et al., 1995). Static CA represents the CBF dependence on BP under steady-state conditions. Dynamic CA reflects the transient response of CBF to sudden fluctuations in BP (Aaslid et al., 1989). Worsening in CA efficiency has been shown to be associated with a wide range of cerebrovascular complications (Panerai, 2008). Impaired cerebral microcirculation has been reported in patients with PD and the high vulnerability of these patients to changes on cerebral parenchyma make CA a potential factor that could be used to explain the occurrence of PD (Hála, 2007, Otomo et al., 2013). Therefore, impairment on CA may be a marker for the development of delirium.
We performed a prospective study to explore the potential association of PD with the temporal evolution of dynamic CA in high-risk surgical patients after CABG surgery with CPB.
2. Methods
2.1. Subjects
The current study is a single center, observational and prospective that was performed at the Heart Institute, University of Sao Paulo, Brazil, from May 2014 to July 2015. Patients who underwent elective coronary artery bypass graft surgery with CPB were considered eligible to participate if: (i) age higher than 18 years, (ii) ‘European System for Cardiac Operative Risk Evaluation’ (EuroSCORE) ≥ 6 and/or left ventricular ejection fraction (LVEF) ≤ 40% on transthoracic echocardiography, and (iii) written informed consent.
Exclusion criteria were surgery without CPB, death before the seventh day post-surgery, absence of temporal acoustic window bilaterally or poor quality recordings. The protocol was approved by the Ethics Committee for the Analysis of Research Projects (CAPPesq) of HCFMUSP (approval number: 835.731) and written informed consent was provided by all participants.
2.2. Procedure
Preoperative medication utilized was Midazolam (0.1–0.2 mg/kg administered thirty minutes before the procedure). Then anesthesia was induced with fentanyl (3–5 μg/kg), midazolam (0.05 mg/kg), etomidate (0.2–0.3 mg/kg), and pancuronium bromide (0.1 mg/kg). Anesthesia was continued with isoflurane in oxygen and fentanyl as necessary. During CPB, additional doses of midazolam and pancuronium were administered as needed. All patients were operated under CPB and moderate hypothermia; myocardial protection was achieved with intermittent antegrade blood cardioplegia. Blood tests (hemoglobin, hematocrit, lactate, arterial and venous blood gas) from all patients were collected before CPB, after and during ICU stay. ‘Sequential Organ Failure Assessment’ (SOFA) score was adopted to determine the extent of a person's organ function or rate of failure (Vincent et al., 1996). This score is based on six different items, one each for the respiratory, cardiovascular, hepatic, coagulation, renal and neurological systems, that were evaluated daily (Vincent et al., 1996). Follow-up after hospital discharge was performed by telephone until the 30th postoperative day to monitor untoward events such as death or need for hospitalization during this period.
Systemic and cerebral physiological monitoring was performed at rest one day before the procedure (T1), the day after surgery (T2) and 7 days (T3) after surgery. Transcranial Doppler (TCD) evaluation of the MCA blood flow was carried out using bilateral 2 MHz pulsed, range-gated probes (‘DWL, Dopplerbox, Germany’), held in place using a helmet, with insonation depth from 50 to 55 mm. Where only one MCA could be insonated, that side was used in subsequent analyses.
BP was continuously measured non-invasively at T1 and T3 using finger arterial volume clamping (‘Finometer PRO; Finapres Medical Systems, Amsterdam, Netherlands’) with the servo-adjust switched off once stable waveforms were recorded with the servo-adjust on. At T2, BP was measured invasively with an arterial catheter (‘Philips Monitor MP50, Germany’).
End-tidal CO2 (EtCO2) was continuously measured with an infrared capnograph (‘Dixtal, dx1265 Capnogard, Manaus, Brazil’) via a closely fitted mask and was documented at 1 min intervals. Patients were also monitored with a pulmonary artery catheter with continuous measurement of cardiac output (‘Swan-Ganz CCO V; Edwards Lifesciences, Irvine, CA’).
Signals were sampled (100 Hz) and stored for offline analysis. All recordings were visually inspected and the BP signal was calibrated using the systolic and diastolic values of radial sphygmomanometry. All signals were filtered with an eighth-order Butterworth low-pass filter with a cut-off frequency of 20 Hz. The beginning and end of each cardiac cycle were detected in the BP signal, and mean BP, CBF velocity (CBFV) and heart rate were obtained for each heartbeat. Beat-to-beat parameters were interpolated with a third-order polynomial and resampled at 5 Hz to generate signals with a uniform time base.
Dynamic CA was calculated by transfer function analysis (TFA), using spontaneous fluctuations of BP as input and corresponding variation in CBFV as output in recordings lasting five minutes, as described previously (Panerai, 1998, Caldas et al., 2017). Welch method was adopted for smoothing spectral estimates obtained with the fast Fourier transform (102.4 s segments, 50% superposition). Using the inverse fast Fourier transform, the CBFV response to a step change in BP was also derived and compared with 10 template curves proposed by Tiecks et al. (1995) to estimate the autoregulation index (ARI) (Panerai et al. 1998). Measurements were rejected if coherence did not reach 0.5 for any frequency below 0.25 Hz. Coherence is part of TFA, and represents the fraction of output power (CBFV variability) that can be linearly explained by the corresponding input power (ABP variability) at each frequency (Claassen and Meel-van, 2016). This parameter varies between zero and 1, and can be interpreted similarly to a correlation coefficient. Coherence will approach zero if the signal-to-noise ratio of measurements is too low, if the input-output relationship is non-linear, or if there are other variables influencing CBFV. In general, the 95% confidence limit of coherence is used to determine acceptance of parameters derived by TFA (Claassen and Meel-van, 2016).
Baseline cerebral hemodynamic parameters are reported as the average over a 5-min baseline recording. Impaired dynamic CA was defined as ARI < 4 (Patel et al., 2016).
2.3. Assessment of post-surgical delirium
Patients underwent twice a day assessment for delirium by the Confusion Assessment Method (CAM) from post-procedure day 1, until discharge from ICU by a trained nurse. In intubated patients, assessment of delirium was achieved with use of the CAM for the ICU, a validated nonverbal version of this score (Ely et al., 2001).
2.4. Neuropsychological assessment
Two cognitive tests were assessed before and six months after surgery. The Mini Mental State Examination (MMSE) can detect cognitive deficits with a score of less than 24, with a maximum value of 30 (Folstein et al., 1975). ‘The Montreal Cognitive Assessment’ (MoCA, Version 7.1) was developed to detect mild cognitive impairment for score values under 26, again, with a maximum range of 30 (Nasreddine et al., 2005). These tests were administered by a trained nurse who was blinded to all other data and measurements performed in these patients.
2.5. Statistical analysis
Kolmogorov-Smirnov test was used to assess of normality and one-sample test, parametric or non-parametric tests were used as appropriate. Fisher’s exact text was used with categorical variables. Results are expressed as mean ± SD or median (IQR). Inter-hemispherical differences in parameters were tested with the paired Student’s t-test or Wilcoxon non-parametric test. In the absence of differences, values for the right and left MCAs were averaged. Changes in ARI at T1, T2 and T3 were assessed with repeated measures ANOVA. Logistic regression with both categorical and continuous independent variables was used to build predictive models for the occurrence of PD. The binary classification (CAM ICU + vs. CAM ICU −) was used as outcome variable in a way that “no delirium” and “postoperative delirum” were coded as 0 and 1, respectively. Logistic regression models for the presence or absence of PD were constructed using those variables that showed statistically significant differences between groups before surgery, during the cardiac surgery and in the first postoperative day as inputs, adopting forward stepwise variable selection. Statistical analyses were made using ‘SPSS’ version 22.0 (‘SPSS Inc., Chicago, IL’). A p-value < 0.05 was considered as statistically significant in all analyses.
3. Results
One hundred and one patients were recruited. Patients were excluded due to surgery without CPB (n = 7), death before the seventh day post-surgery (n = 6), absence of a temporal acoustic window bilaterally (n = 10), or poor quality recordings (n = 11). The 67 patients (51 male) included in the study were aged 64.3 ± 9.5 years. EuroSCORE was 5 (3–7) and echocardiogram LVEF was 40% (35–45). Patient demographics and baseline characteristics are presented in Table 1 .
Table 1.
Demographic and baseline characteristics.
| Variables | Patients (n = 67) |
|---|---|
| Male (n, %) | 51 (76.6%) |
| Age (years) | 64.3 ± 9.5 |
| Education (years) | 6 (2–10) |
| Risk factors | |
| LVEF (%) | 40 (35–45) |
| EuroSCORE | 5 (3–7) |
| Previous cardiac surgery (n, %) | 0 |
| Previous myocardial infarction (n, %) | 55 (82.1%) |
| Hypertension (n, %) | 53 (79.1%) |
| Peripheral vascular disease (n, %) | 5 (7.5%) |
| Smoking (n, %) | 16 (23.9%) |
| Previous smoking (n, %) | 32 (47.8%) |
| Dyslipidemia (n, %) | 46 (68.7%) |
| Diabetes (n, %) | 33 (49.3%) |
| Atrial fibrillation (n, %) | 5 (7.5%) |
| Previous stroke (n, %) | 5 (7.5%) |
| Valvular disease (n, %) | 9 (13.4%) |
| Obesity (BMI > 30 kg/m2) (n, %) | 10 (14.9%) |
| Left coronary trunk lesion > 50% (n, %) | 21 (31.3%) |
| Medication (n) | |
| ACE inhibitor/ ARB | 50 (74.6%) |
| Beta blocker | 54 (80.6%) |
| Surgery | |
| CABG + valve (n, %) | 3 (4.5%) |
| Length of CPB (min) | 92.5 ± 27.8 |
| IABP (n, %) | 34 (50.7%) |
LVEF, left ventricular ejection fraction; EuroSCORE, European System for Cardiac Operative Risk Evaluation; BMI, body mass index; ACE, angiotensin converting enzyme; ARB, angiotensin receptor blockers; CABG, coronary artery bypass graft; CPB, cardiopulmonary bypass; IABP, intra-aortic balloon pump.
3.1. Diagnosis of PD: Associated clinical and systemic hemodynamic characteristics
Patient characteristics regarding predisposing and precipitating variables are presented in Table 2 . PD was diagnosed in 17 patients; demographic and baseline characteristics associated with this condition were age (p = 0.035) and MoCA (p = 0.013). Blood tests and systemic hemodynamic parameters are given as Supplementary Data (Table S1) for different time periods, from before CPB to 24 h in ICU. Patients who had diagnosis of PD had higher CO2 GAP, lower cardiac output, during both surgery and before CPB, than patients without PD (Supplementary Table S1). The day after surgery, patients with PD had higher SOFA score (12 (9–15) vs. 10 (8–10), p = 0.004), amount of fluid input (941.2 ± 583.1 mL vs. 480.0 ± 390.7 mL; p = 0.003), hydric balance (604.3 ± 696.7 mL vs. 218.3 ± 6 26.9 mL; p = 0.037) (Supplementary Table S1) and total dose of norepinephrine (0.18 μg/kg/min (0.0–0.66) vs. 0.025 μg/kg/min (0.00–0.44; p = 0.002) when compared with patients without PD (Table 3 ).
Table 2.
Distribution of parameters according to the occurrence of delirium, as assessed by the CAM ICU.
| Variable | CAM ICU – | CAM ICU + | P |
|---|---|---|---|
| (N = 50) | (N = 17) | ||
| Male (n, %) | 38 (78.0%) | 13 (76.5%) | 1.000 |
| Age (years) | 63 ± 10 | 68 ± 7 | 0.035 |
| Risk factors | |||
| LVEF (%) | 40 (35–40) | 40 (37.5–47.5) | 0.293 |
| EuroSCORE | 5 (3–6) | 6 (4–7) | 0.121 |
| Education (years) | 6 (2–10) | 5 (1–8) | 0.127 |
| MoCA before surgery | 21.5 ± 4.1 | 18.3 ± 5.2 | 0.013 |
| MMSE before surgery | 26 (23–29) | 23 (18.5–23) | 0.228 |
| MoCA after surgery | 21.6 ± 4.1 | 19.0 ± 6.3 | 0.127 |
| MMSE after surgery | 28 (25–29) | 23 (18.5–23) | 0.275 |
| Atrial fibrillation n (%) | 4 (8.0%) | 1 (5.9%) | 1.000 |
| Previous myocardial infarction n (%) | 41 (82.0%) | 14 (82.3%) | 0.643 |
| Hypertension n (%) | 38 (76.0%) | 15 (88.2%) | 0.240 |
| Peripheral vascular disease n (%) | 6 (12.0%) | 1 (5.9%) | 0.669 |
| Smoking n (%) | 14 (28.0%) | 2 (11.8%) | 0.322 |
| Previous smoking n (%) | 23 (46.0%) | 9 (5.2%) | 0.621 |
| Dyslipidemia n (%) | 37 (74.0%) | 9 (52.9%) | 0.106 |
| Diabetes n (%) | 22 (44.0%) | 11 (67.7%) | 0.140 |
| Previous stroke n (%) | 4 (8.0%) | 1 (5.9%) | 1.000 |
| Obesity (BMI > 30 kg/m2) n (%) | 7 (14.0%) | 3 (17.6%) | 0.706 |
| Left coronary trunk lesion > 50% n (%) | 14 (28.0%) | 7 (41.2%) | 0.312 |
| Valvular disease n (%) | 7 (14.0%) | 2 (11.8%) | 1.000 |
| Medication | |||
| ACE inhibitor/ARB n (n%) | 40 (80.0%) | 10 (58.8%) | 0.110 |
| Beta blocker n(%) | 40(80.0%) | 14 (82.4%) | 1.000 |
| Surgery | |||
| CABG + valve n (%) | 2 (4.0%) | 1 (5.9%) | 0.591 |
| IABP n (%) | 25 (50.0%) | 9 (52.9%) | 1.000 |
| Surgery (hours:min) | 05:21 ± 1:00 | 5:01 ± 0:54 | 0.296 |
| Length of CPB (min) | 97 ± 30 | 90 ± 21 | 0.386 |
| Aortic cross-clamp duration (min) | 79 ± 26 | 66 ± 17 | 0.068 |
| Hydric balance (milliliters) | 2913.82 ± 1217.916 | 3138.7 ± 1014.81 | 0.456 |
| RBC transfusion n (%) | 17 (34%) | 17 (34%) | 0.770 |
CAM ICU, Confusion Assessment Method for the ICU; LVEF, left ventricular ejection fraction; EuroSCORE, European System for Cardiac Operative Risk Evaluation; MMSE, Mini Mental State Examination; MoCA, Montreal Cognitive Assessment; BMI, body mass index; ACE, angiotensin converting enzyme; ARB, angiotensin receptor blockers; CABG, coronary artery bypass graft; CPB, cardiopulmonary bypass; IABP, intra-aortic balloon pump.
Table 3.
Clinical outcomes in patients with and without delirium as assessed by CAM ICU.
| CAM ICU + N = 17 |
CAM ICU – N = 50 |
p | |
|---|---|---|---|
| Duration of mechanical ventilation (min) | 49.5 ± 70.1 | 13.7 ± 30.7 | 0.002 |
| Death 30 days (n) | 2 | 2 | 0.264 |
| Duration of dobutamine use during ICU stay (hour) | 57.4 ± 54.2 | 95.5 ± 82.9 | 0.008 |
| Use of norepinephrine during ICU stay (hour) | 161.0 ± 132.4 | 75.5 ± 107.9 | 0.002 |
| Acute kidney injury (n) | 12 | 19 | 0.012 |
| SARS | 0 | 1 | 1.000 |
| Seizure | 2 | 1 | 0.062 |
| Tachyarrhythmia (n) | 11 | 14 | 0.010 |
| Cardiogenic shock (n) | 5 | 4 | 0.040 |
| Vasoplaegic shock (n) | 3 | 2 | 0.099 |
| Infecction | 13 | 31 | 0.379 |
| Length of ICU stay (days) | 10.65 ± 7.55 | 7.74 ± 6.16 | 0.009 |
| Norepinephrine (μg/kg/min) | 0.18 (0.0–0.66) | 0.025 (0.00–0.44) | 0.002 |
| SOFA | 12 (9–15) | 10 (8–10) | 0.004 |
ICU, intensive care unit; SARS, Severe Acute Respiratory Syndrome; SOFA, Sequential Organ Failure Assessment.
Clinical outcomes, including infection, duration of mechanical ventilation, duration of drug use, acute kidney injury, and length of ICU stay are given in Table 3.
3.2. Dynamic CA
The CBFV and ARI values were not different between the right and left MCAs; the mean values averaged for both hemispheres were used in further comparisons. Significant differences were observed in the heart rate, BP, CBFV and ARI values for the three-different time-points studied (T1-T3) (Supplementary Table S2). The BP values at T2 were lower when compared with T1 and T3 (p < 0.001) (Supplementary Table S2). On the other hand, the CBFV at T2 was the higher than at T1 or T3 (Supplementary Table S2) (p < 0.001).
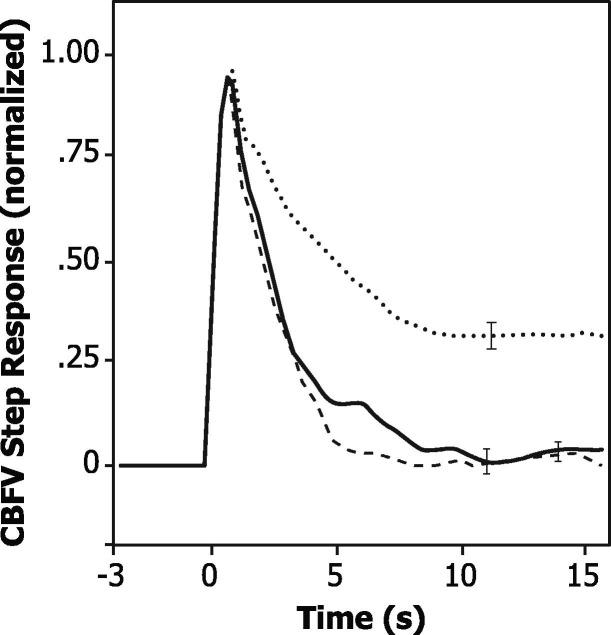
The ARI values at T2 (3.9 ± 1.7) were markedly reduced in comparison with the corresponding values at T1 (5.6 ± 1.7) and T3 (5.5 ± 1.8) (p < 0.001). Before surgery, dynamic CA was impaired (ARI < 4) in 12 (17.9%) patients, but this number increased to 37 patients at T2 (55.2%) with 14 patients (35.8%) still manifesting impaired CA at T3 (p < 0.001) (Supplementary Table S2). These differences in ARI were confirmed by the corresponding CBFV step responses (Fig. 1 ), reflecting the effect of a sudden BP change, with a much slower recovery towards baseline at T2 in comparison with T1 and T3.
Fig. 1.
Population averages (n = 67) of normalized CBFV responses to a step change in blood pressure for assessments before surgery (continuous line), 24 h (dashed line) and 7 days (dotted line) after surgery. Error bars represent the largest ± 1 SE at the point of occurrence. As shown by Tiecks et al. (1995), each CBFV step response corresponds to a value of the autoregulation index (ARI). For the curves above, the corresponding values of ARI are 5.6 ± 1.7 (before surgery), 3.9 ± 1.7 (24 h) and 5.5 ± 1.8 (7 days).
In patients with PD, 13 (76.5%) had impaired dynamic CA, and only 4 (23.5%) had normal CA (p = 0.041) at T2. At T3, 14 patients had persistence of impaired autoregulation, 9 (64.2%) of whom had delirium (Table 4 , p = 0.001).
Table 4.
Distribution of cerebral hemodynamic parameters according to the occurrence of delirium.
| Variable | CAM ICU – N = 50 |
CAM ICU + N = 17 |
P | |
|---|---|---|---|---|
| Before surgery (T1) | ||||
| MAP (mmHg) | 92.2 ± 13.2 | 92.6 ± 14.5 | 0.900 | |
| HR (bpm) | 64.0 ± 10.8 | 68.64 ± 10.3 | 0.126 | |
| CBFV (cm/s) | 60.3 ± 15.4 | 54.6 ± 14.3 | 0.183 | |
| ETCO2 (mmHg) | 33.9 ± 4.5 | 33.6 ± 3.6 | 0.819 | |
| ARI | 5.9 ± 1.5 | 4.8 ± 1.9 | 0.021 | |
| ARI < 4 (n, %) | 7 (14.0%) | 6 (35.3%) | 0.077 | |
| First day post-surgery (T2) | ||||
| MAP (mmHg) | 81.4 ± 12.0 | 83.2 ± 15.2 | 0.608 | |
| HR (bpm) | 102.7 ± 14.16 | 103.2 ± 18.0 | 0.915 | |
| CBFV (cm/s) | 78.2 ± 21.5 | 62.6 ± 17.4 | 0.004 | |
| ETCO2 (mmHg) | 33.2 ± 3.5 | 34.9 ± 4.4 | 0.105 | |
| ARI | 4.3 ± 1.5 | 3.1 ± 1.8 | 0.010 | |
| ARI < 4 (n, %) | 24 (48.0%) | 13 (76.5%) | 0.041 | |
| 7 days post-surgery (T3) | ||||
| MAP (mmHg) | 89.0 ± 8.5 | 91.5 ± 9.2 | 0.309 | |
| HR (bpm) | 89.0 ± 13.7 | 92.1 ± 15.7 | 0.450 | |
| CBFV (cm/s) | 72.9 ± 17.6 | 58.4 ± 18.9 | 0.005 | |
| ETCO2 (mmHg) | 31.9 ± 3.4 | 32.3 ± 3.5 | 0.684 | |
| ARI | 5.9 ± 1.5 | 4.5 ± 2.4 | 0.031 | |
| ARI < 4 (n, %) | 5 (10.0%) | 9 (52.9%) | 0.001 | |
ARI, autoregulation index; CAM ICU, Confusion Assessment Method for the ICU; CBFV, cerebral blood flow velocity. ETCO2, end- tidal CO2; HR, heart rate; MAP, mean arterial pressure.
Predictive cerebral autoregulation characteristics associated with delirium on univariate analysis were lower ARI at T1 (4.8 ± 1.9 vs. 5.9 ± 1.5; p = 0.021), at T2 (3.1 ± 1.8 vs. 4.3 ± 1.5; p = 0.01) and at T3 (4.5 ± 2.4 vs. 5.9 ± 1.5; p = 0.031) (Table 4). At T2 (p = 0.004) and at T3 (p = 0.005), CBFV was lower in patients with PD compared with those without PD (Table 4).
Predictive multiple logistic regression models of PD showed that before surgery, lower ARI index and lower MoCA score were significant factors to PD occurrence (Table 5 ). During surgery, and at the first day after surgery on ICU, the predictive models showed that the ARI index, amount of fluid input and SOFA score were significant factors for predicting PD occurrence (Table 5). Noteworthy, predictive models after surgery proved more accurate than corresponding models before surgery. Of the 17 patients who were positive for PD (Table 4), 7 were detected correctly before surgery and 9 were true-positives after surgery. For the true negatives, the corresponding value was 46, for both models, leading to the accuracy values given in Table 5.
Table 5.
Predictors of delirium occurrence estimated from multiple logistic regression analysis.
| Variable | Beta coeff | SE | Wald | Odds ratio | CI 95% Lower - Upper | Accuracy (%) | P |
|---|---|---|---|---|---|---|---|
|
79.1 | ||||||
| MoCA | −0.159 | 0.069 | 4.52 | 0.853 | 0.745–0.976 | 0.021 | |
| ARI T1 | −0.377 | 0.178 | 4.45 | 0.686 | 0.484 – 0.973 | 0.035 | |
| Constant | 4.129 | ||||||
|
82.1 | ||||||
| Total fluid input | 0.002 | 0.001 | 9.04 | 1.002 | 1.001–1.004 | 0.003 | |
| ARI T2 | −0.688 | 0.207 | 6.89 | 0.503 | 0.301–0.503 | 0.009 | |
| SOFA | 0.411 | 0.262 | 3.92 | 1.508 | 1.004–2.264 | 0.048 | |
| Constant | 4817 | ||||||
MoCA, Montreal Cognitive Assessment; ARI, autoregulation index; T1 before surgery; T2, 24 h after surgery; SOFA, Sequential Organ Failure Assessment. SE, standard error
4. Discussion
To the best of our knowledge, this study is the first to report the evolution of serial TCD assessments of dynamic CA in high-risk patients undergoing cardiac surgery with CPB, including a pre-surgical assessment. Two main findings are noteworthy. First, dynamic CA is reduced on the first day following surgery, with the majority of patients showing ARI values indicative of CA impairment. Second, although dynamic CA recovered in most patients after seven days, those patients with impaired dynamic CA at 24 h and 7 days had a higher incidence of PD. Taken together, these findings suggest that poor CA might be involved in the occurrence of neurological complications of cardiac surgery. Of considerable relevance, predictive models before and after surgery indicate that assessment of CA could help to identify patients at higher risk of PD, eventually contributing to better prevention and reduction in the number and duration of episodes of delirium.
This study adds to the existing literature on cerebral hemodynamic abnormalities in cardiac surgery. CBFV and CA have been assessed during cardiac surgery (Ono et al., 2012, Easley et al., 2013), but the effects on dynamic CA before and after cardiac surgery have not been reported. These studies suggested that CA might be impaired during cardiac surgery, but our results demonstrated that CA impairment is also present in 57% of patients up to 24 h post-surgery.
The cerebral autoregulation 24 h and 7 days after cardiac surgery was not studied previously, with the majority of studies only reporting measurements intra-surgery and a few hours after cardiopulmonary bypass. Given the prospective nature of our study, and the fact that the temporal evolution of cerebral autoregulation was not known at the time of planning, we elected to perform measurements both at 24 h and 7 days post-surgery. As discussed below, our novel findings about the temporal evolution of dynamic CA and its association with the occurrence of PD have considerable potential for the detection and prevention of this complication of cardiac surgery.
4.1. Cerebral autoregulation during cardiopulmonary bypass
Previous studies have shown alterations in cerebral hemodynamics and CA in patients undergoing CPB (Ono et al., 2012, Easley et al., 2013). Impaired CA might contribute to brain ischemic damage when arterial pressure is low such as during CPB, and could explain the reported association with perioperative stroke and mortality (Ono et al., 2012, Ono et al., 2014).
Studies focusing on cardiac surgery have provided conflicting results. Christiansen et al. demonstrated intact dynamic CA in eight patients, six hours after cardiac surgery, in agreement with Preisman et al., based on 12 patients assessed 45-min after surgery (Preisman et al., 2005, Christiansen et al., 2015). These results may have been influenced by the relatively small sample size and the use of transfer function analysis limited to gain and phase (Panerai et al., 1998, Panerai, 2008). In our study, we calculated CA by ARI (Tiecks et al., 1995). The ARI incorporates all the gain and phase information, which are then aggregated and summarized into a single value. In this way, if smaller differences in gain and/or phase are not enough to lead to statistically significant differences, their aggregated effect, as part of the non-linear transformation involved in estimating the ARI (Tiecks et al., 1995) and lead to the ARI being significantly different. On the other hand, based on a larger patient group, Hori et al reported that reduced CA efficacy was associated with delirium, but in their study CA was assessed only during CPB (Hori et al., 2014). Our larger study, using ARI as a more robust index of CA (incorporating both gain and phase information), confirms that dynamic CA is impaired following cardiac surgery. Uniquely however, we also report that there is substantial improvement after seven days.
4.2. Post-surgical delirium
Delirium is an acute condition of consciousness and attention that has a fluctuating course common after cardiac procedure, and is associated with additional new cognitive decline, post-operative stroke, dementia, increased morbidity, length-of-stay, hospital readmission and mortality (Davis et al., 2012, Martin et al., 2012, Saczynski et al., 2012, Brown, 2014). In our study, the incidence of PD was only 25.4%, compared to other reports in the literature citing rates of up to 70% of patients affected after heart surgery (Brown, 2014). The relatively low rate of delirium in our case might have resulted from adherence to strict surgical guidelines and post-surgical nursing care protocols. Nevertheless, basing delirium diagnosis only on clinical signs detected by nurses and physicians, has been shown to have low sensitivity, and might under-recognize hypoactive forms of delirium (Inouye et al., 2001).
Of considerable interest is the finding that the incidence of delirium was related with dynamic CA impairment after surgery. In agreements with our results, Schramm et al. (2012) showed that impaired CA is associated with delirium in patients with severe sepsis. This observation deserves further investigation due to its clinical relevance and potential implications for post-surgical care. It is possible to speculate that delirium was caused by factor(s), yet to be identified, in a sub-group of patients with impaired dynamic CA.
Predictive models that could identify patients at a higher risk of delirium before surgery can be an important tool to physicians who could then instigate more strict controls of systemic and cerebral hemodynamics, closer monitoring of anesthesia and surgical procedures and other non-pharmacological therapies such as, family stay, before delirium settles in. Furthermore, the early prediction of delirium in intensive care patients facilitates the use of non-drug preventive measures in high risk patients, such as improvement of orientation, cognitive stimulation, early mobilization, and listening to music. It also facilitates drug interventions in high risk patients, such as the administration of prophylactics, for example haloperidol. These interventions aim to improve patients’ cognition or have a systemic effect, although the evidence of beneficial preventive measures with drugs and nursing interventions in critically ill patients is limited at this moment.
The identification of cerebral hemodynamic parameters that could pinpoint this high risk sub-group would be worth pursuing in future investigations.
The association we found between the occurrence of PD and depressed CA is likely to be complex and deserves further investigation. The majority of our patients had impaired CA after cardiac surgery with CPB (Caldas et al., 2017), but not all of these patients developed PD. Despite the predictive value of ARI, there are other contributing factors (Table 4) that also played a role in the development of delirium. Identifying the exact underlying mechanisms whereby disturbed CA leads to PD with CPB surgery should be a priority for treatment of these patients.
Factors previously reported to have an association with PD included aging, prior stroke, cognitive decline, and reduced CBFV (Inouye, 2006, Rudolph et al., 2009, Caplan et al., 2014, Bostwick, 2004). Our results add to the existing literature about predisposing factors for delirium, we found that lower ARI before surgery was a risk factor and predictor of PD after cardiac surgery. Under normal physiological conditions, CA counteracts the changes in systemic blood pressure that occur during daily activities and several studies have shown a link between ARI impairment and cerebrovascular conditions (Panerai, 2008, Salinet et al., 2014, Ma et al., 2016), but ARI analysis before cardiac surgery has not been previously reported. Further studies are needed to elucidate the mechanisms underlying this finding, nevertheless, identifying this group of patients is an important first step in critical care of the perioperative period to prevent PD or reduce its duration.
Our results confirmed the influence of age and CBFV as co-factors in the occurrence of PD, as well as the contribution of cognition, assessed by the MoCA. However, we did not confirm the association with pre-morbid stroke, but this is likely to reflect lack of power as only five patients had a prior stroke.
The potential consequences of delirium after cardiac surgery have been reported in several studies (Martin et al., 2012, Saczynski et al., 2012, Otomo et al., 2013, Brown et al., 2016). In agreement with these data, our group of patients with PD had longer duration of mechanical ventilation, prolonged use of drugs such as dobutamine and norepinephrine, with more patients having acute kidney injury, tachyarrhythmia and cardiogenic shock, and the longer lengths of ICU stay.
PD adversely affects both early- and long-term outcomes, thus risk reduction in high risk patients, and early detection and treatment of those affected, is important. PD is a marker of impaired cerebral microcirculation and patients developing delirium may have a more vulnerable central nervous system (Otomo et al., 2013). Evidence of this susceptibility is provided by previous neurophysiological studies demonstrating major differences in the EEG of patients with PD following cardiothoracic surgery (van der Kooi et al., 2015). Importantly, the contribution of CA has considerable potential to improve existing models for the prediction of PD that have not included cerebral hemodynamic parameters (Rudolph et al., 2009, Luetz et al., 2016, Mailhot et al., 2016) and therefore warrants further investigation.
4.3. Study limitations
CBFV measurements with TCD can only provide reliable estimates of CBF if the diameter of the insonated artery (MCA) remains constant. This is likely to be the case during 5-minute baseline measurements obtained at rest (Serrador et al., 2000). Additionally, a permeable acoustic window is needed for ultrasound to penetrate the skull, a well-known limitation of TCD, which mainly affects post-menopausal women. In our case, 10 patients had to be rejected due to the absence of temporal windows, which agrees with the 10% rejection figure found in international studies (Alexandrov and Bornstein, 2006).
Our patients could also be affected by carotid artery disease. Several studies have shown that both ARI and phase are depressed in patients with significant carotid artery stenosis (Panerai et al., 1998, Panerai, 2008). None of the patients studied had symptoms of advanced carotid artery disease, but we cannot exclude the possibility that values of ARI could have been biased by the presence of asymptomatic carotid artery disease. Differences in the method of measurement of ABP, with the Finometer being used at T1 and T3 and an arterial line at T2, could also be seen as a potential limitation. However, a detailed previous study showed excellent agreement between the two methods for assessment of dynamic CA (Sammons et al., 2007).
Finally, the lack of a reference standard patient interview by a psychiatrist in the assessment of delirium was an important limitation that needs to be acknowledged. Nonetheless, the assessment was performed by a trained nurse.
5. Conclusions
In patients undergoing cardiac surgery with CPB, there was a strong association between dynamic CA and the occurrence of PD. Dynamic CA was impaired in the majority of patients in the first day after surgery. Assessment of cerebral hemodynamics before and after surgery could have considerable potential for early identification of patients at risk of PD thus reducing poor outcomes and lengths of stay. Further work is needed to elucidate the underlying mechanisms of impaired dynamic CA and to minimize the effects of impaired CA in patients undergoing cardiac surgery.
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
The abstract was presented as an e-poster at the 36th International Symposium on Intensive Care and Emergency Medicine (ISICEM) – Brussels March 2016).
Supplementary data to this article can be found online at https://doi.org/10.1016/j.clinph.2018.11.008.
Appendix A. Supplementary material
The following are the Supplementary data to this article:
References
- Aaslid R., Lindegaard K.F., Sorteberg W., Nornes H. Cerebral autoregulation dynamics in humans. Stroke. 1989;20(1):45–52. doi: 10.1161/01.str.20.1.45. [DOI] [PubMed] [Google Scholar]
- Alexandrov A.V., Bornstein N.M. Advances in neurosonology 2005. Stroke. 2006;37(2):299–300. doi: 10.1161/01.STR.0000201891.41764.e5. [DOI] [PubMed] [Google Scholar]
- Bostwick J.M. Delirium in old age. Clin Neurophysiol. 2004;115(6):1487–1488. [Google Scholar]
- Brown C.H. Delirium in the cardiac surgical ICU. Curr Opin Anaesthesiol. 2014;27(2):117–122. doi: 10.1097/ACO.0000000000000061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown C.H., Laflam A., Max L., Lymar D., Neufeld K.J., Tian J. The impact of delirium after cardiac surgery on postoperative resource utilization. Ann Thorac Surg. 2016;101(5):1663–1669. doi: 10.1016/j.athoracsur.2015.12.074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caldas J.R., Haunton V.J., Panerai R.B., Hajjar L.A., Robinson T.G. Cerebral autoregulation in cardiopulmonary bypass surgery: a systematic review. Interact Cardiovasc Thorac Surg. 2017;16:1–10. doi: 10.1093/icvts/ivx357. [DOI] [PubMed] [Google Scholar]
- Caplan G.A., Lan Z., Newton L., Kvelde T., McVeigh C., Hill M.A. Transcranial Doppler to measure cerebral blood flow in delirium superimposed on dementia. A cohort study. J Am Med Dir Assoc. 2014;15(5):355–360. doi: 10.1016/j.jamda.2013.12.079. [DOI] [PubMed] [Google Scholar]
- Christiansen C.B., Berg R.M., Plovsing R., Ronit A., Holstein-Rathlou N.H., Yndgaard S. Dynamic cerebral autoregulation after cardiopulmonary bypass. Thorac Cardiovasc Surg. 2015;26 doi: 10.1055/s-0035-1566128. [DOI] [PubMed] [Google Scholar]
- Claassen J.A., Meel-van den Abeelen A.S., Simpson D.M., Panerai R.B. (CARNet) international CARN. Transfer function analysis of dynamic cerebral autoregulation: A white paper from the International Cerebral Autoregulation Research Network. J Cereb Blood Flow Metab. 2016;36(4):665–680. doi: 10.1177/0271678X15626425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis D.H.J., Muniz Terrera G., Keage H., Rahkonen T., Oinas M., Matthews F.E. Delirium is a strong risk factor for dementia in the oldest-old: a population-based cohort study. Brain J Neurol. 2012;135:2809–2816. doi: 10.1093/brain/aws190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deiner S., Silverstein J.H. Postoperative delirium and cognitive dysfunction. Br J Anaesth. 2009;103(Suppl 1):i41–i46. doi: 10.1093/bja/aep291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Easley R.B., Kibler K.K., Brady K.M., Joshi B., Ono M., Brown C. Continuous cerebrovascular reactivity monitoring and autoregulation monitoring identify similar lower limits of autoregulation in patients undergoing cardiopulmonary bypass. Neurol Res. 2013;35(4):344–354. doi: 10.1179/1743132812Y.0000000145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ely E., Inouye S.K., Bernard G.R. Delirium in mechanically ventilated patients: validity and reliability of the confusion assessment method for the intensive care unit (cam-icu) JAMA. 2001;286(21):2703–2710. doi: 10.1001/jama.286.21.2703. [DOI] [PubMed] [Google Scholar]
- Folstein M.F., Folstein S.E., McHugh P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189–198. doi: 10.1016/0022-3956(75)90026-6. [DOI] [PubMed] [Google Scholar]
- Hála M. Pathophysiology of postoperative delirium: systemic inflammation as a response to surgical trauma causes diffuse microcirculatory impairment. Med Hypotheses. 2007;68(1):194–196. doi: 10.1016/j.mehy.2006.07.003. [DOI] [PubMed] [Google Scholar]
- Hori D., Brown C., Ono M., Rappold T., Sieber F., Gottschalk A. Arterial pressure above the upper cerebral autoregulation limit during cardiopulmonary bypass is associated with postoperative delirium. Br J Anaesth. 2014;113(6):1009–1017. doi: 10.1093/bja/aeu319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inouye S.K. Delirium in older persons. N Engl J Med. 2006;354(11):1157–1165. doi: 10.1056/NEJMra052321. [DOI] [PubMed] [Google Scholar]
- Inouye S.K., Foreman M.D., Mion L.C., Katz K.H., Jr, LMC. Nurses’ recognition of delirium and its symptoms: comparison of nurse and researcher ratings. Arch Intern Med. 2001;161(20):2467–2473. doi: 10.1001/archinte.161.20.2467. [DOI] [PubMed] [Google Scholar]
- Luetz A., Weiss B., Boettcher S., Burmeister J., Wernecke K.-D., Spies C. Routine delirium monitoring is independently associated with a reduction of hospital mortality in critically ill surgical patients: a prospective, observational cohort study. J Crit Care. 2016;35:168–173. doi: 10.1016/j.jcrc.2016.05.028. [DOI] [PubMed] [Google Scholar]
- Ma H., Guo Z.N., Liu J., Xing Y., Zhao R., Yang Y. Temporal course of dynamic cerebral autoregulation in patients with intracerebral hemorrhage. Stroke. 2016;47(3):674–681. doi: 10.1161/STROKEAHA.115.011453. [DOI] [PubMed] [Google Scholar]
- Mailhot T., Cossette S., Lambert J., Cournoyer A., Denault A.Y. Cerebral oximetry as a biomarker of postoperative delirium in cardiac surgery patients. J Crit Care. 2016;34:17–23. doi: 10.1016/j.jcrc.2016.02.024. [DOI] [PubMed] [Google Scholar]
- Martin B.J., Buth K.J., Arora R.C., Baskett R.J. Delirium: a cause for concern beyond the immediate postoperative period. Ann Thorac Surg. 2012;93(4):1114–1120. doi: 10.1016/j.athoracsur.2011.09.011. [DOI] [PubMed] [Google Scholar]
- Moody D.M., Brown W.R., Challa V.R., Stump D.A., Reboussin D.M., Legault C. Brain microemboli associated with cardiopulmonary bypass: a histologic and magnetic resonance imaging study. Ann Thorac Surg. 1995;59(5):1304–1307. doi: 10.1016/0003-4975(95)00057-r. [DOI] [PubMed] [Google Scholar]
- Nasreddine Z.S., Phillips N.A., Bedirian V., Charbonneau S., Whitehead V., Collin I. The montreal cognitive assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695–699. doi: 10.1111/j.1532-5415.2005.53221.x. [DOI] [PubMed] [Google Scholar]
- Ono M., Brady K., Easley R.B., Brown C., Kraut M., Gottesman R.F. Duration and magnitude of blood pressure below cerebral autoregulation threshold during cardiopulmonary bypass is associated with major morbidity and operative mortality. J Thorac Cardiovasc Surg. 2014;147(1):483–489. doi: 10.1016/j.jtcvs.2013.07.069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ono M., Joshi B., Brady K., Easley R.B., Zheng Y., Brown C. Risks for impaired cerebral autoregulation during cardiopulmonary bypass and postoperative stroke. Br J Anaesth. 2012;109(3):391–398. doi: 10.1093/bja/aes148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Otomo S., Maekawa K., Goto T., Baba T., Yoshitake A. Pre-existing cerebral infarcts as a risk factor for delirium after coronary artery bypass graft surgery. Interact Cardiovasc Thorac Surg. 2013;17(5):799–804. doi: 10.1093/icvts/ivt304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panerai R.B. Assessment of cerebral pressure autoregulation in humans–a review of measurement methods. Physiol Meas. 1998;19(3):305–338. doi: 10.1088/0967-3334/19/3/001. [DOI] [PubMed] [Google Scholar]
- Panerai R.B. Cerebral autoregulation: from models to clinical applications. Cardiovasc Eng Dordr Neth. 2008;8(1):42–59. doi: 10.1007/s10558-007-9044-6. [DOI] [PubMed] [Google Scholar]
- Panerai R.B., White R.P., Markus H.S., Evans D.H. Grading of cerebral dynamic autoregulation from spontaneous fluctuations in arterial blood pressure. Stroke. 1998;29(11):2341–2346. doi: 10.1161/01.str.29.11.2341. [DOI] [PubMed] [Google Scholar]
- Patel N., Panerai R.B., Haunton V., Katsogridakis E., Saeed N.P., Salinet A. The Leicester cerebral haemodynamics database: normative values and the influence of age and sex. Physiol Meas. 2016;37(9):1485–1498. doi: 10.1088/0967-3334/37/9/1485. [DOI] [PubMed] [Google Scholar]
- Paulson O.B., Strandgaard S., Edvinsson L. Cerebral autoregulation. Cerebrovasc Brain Metab Rev. 1990;2(2):161–192. [PubMed] [Google Scholar]
- Plaschke K., Fichtenkamm P., Schramm C., Hauth S., Martin E., Verch M. Early postoperative delirium after open-heart cardiac surgery is associated with decreased bispectral EEG and increased cortisol and interleukin-6. Intensive Care Med. 2010;36(12):2081–2089. doi: 10.1007/s00134-010-2004-4. [DOI] [PubMed] [Google Scholar]
- Preisman S., Marks R., Nahtomi-Shick O., Sidi A. Preservation of static and dynamic cerebral autoregulation after mild hypothermic cardiopulmonary bypass. Br J Anaesth. 2005;95(2):207–211. doi: 10.1093/bja/aei147. [DOI] [PubMed] [Google Scholar]
- Rudolph J.L., Jones R.N., Levkoff S.E., Rockett C., Inouye S.K., Sellke F.W. Derivation and validation of a preoperative prediction rule for delirium after cardiac surgery. Circulation. 2009;119(2):229–236. doi: 10.1161/CIRCULATIONAHA.108.795260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saczynski J.S., Marcantonio E.R., Quach L., Fong T.G., Gross A., Inouye S.K. Cognitive trajectories after postoperative delirium. N Engl J Med. 2012;367(1):30–39. doi: 10.1056/NEJMoa1112923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salinet A.S., Panerai R.B., Robinson T.G. The longitudinal evolution of cerebral blood flow regulation after acute ischaemic stroke. Cerebrovasc Dis Extra. 2014;4(2):186–197. doi: 10.1159/000366017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sammons E.L., Samani N.J., Smith S.M., Rathbone W.E., Bentley S., Potter J.F. Influence of noninvasive peripheral arterial blood pressure measurements on assessment of dynamic cerebral autoregulation. J Appl Physiol. 2007;103(1):369–375. doi: 10.1152/japplphysiol.00271.2007. [DOI] [PubMed] [Google Scholar]
- Schramm P., Klein K.U., Falkenberg L., Berres M., Closhen D., Werhahn K.J. Impaired cerebrovascular autoregulation in patients with severe sepsis and sepsis-associated delirium. Crit Care. 2012;16(5):R181. doi: 10.1186/cc11665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scolletta S., Taccone F.S., Donadello K. Brain injury after cardiac surgery. Minerva Anestesiol. 2015;81(6):662–677. [PubMed] [Google Scholar]
- Serrador J.M., Picot P.A., Rutt B.K., Shoemaker J.K., Bondar R.L. MRI measures of middle cerebral artery diameter in conscious humans during simulated orthostasis. Stroke J Cereb Circ. 2000;31(7):1672–1678. doi: 10.1161/01.str.31.7.1672. [DOI] [PubMed] [Google Scholar]
- Siepe M., Pfeiffer T., Gieringer A., Zemann S., Benk C., Schlensak C. Increased systemic perfusion pressure during cardiopulmonary bypass is associated with less early postoperative cognitive dysfunction and delirium. Eur J Cardio-Thorac Surg. 2011;40(1):200–207. doi: 10.1016/j.ejcts.2010.11.024. [DOI] [PubMed] [Google Scholar]
- Tiecks F.P., Lam A.M., Aaslid R., Newell D.W. Comparison of static and dynamic cerebral autoregulation measurements. Stroke. 1995;26(6):1014–1019. doi: 10.1161/01.str.26.6.1014. [DOI] [PubMed] [Google Scholar]
- van der Kooi A.W., Zaal I.J., Klijn F.A., Koek H.L., Meijer R.C., Leijten F.S. Delirium detection using EEG: what and how to measure. Chest. 2015;147(1):94–101. doi: 10.1378/chest.13-3050. [DOI] [PubMed] [Google Scholar]
- Vincent J.L., Moreno R., Takala J., Willatts S., De Mendonça A., Bruining H. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med. 1996;22(7):707–710. doi: 10.1007/BF01709751. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.