Graphical abstract
Highlights
-
•
Nanotechnology is emerging as a promising approach in the field of medicine.
-
•
Carbon nanotubes (CNTs) are efficient drug delivery systems.
-
•
Promising therapeutic efficacy of CNTs is needed to be investigated thoroughly.
-
•
Safety evaluation of CNTs is one of the most important aspects.
-
•
Targeted delivery via CNTs is an important strategy to improve therapeutic index.
-
•
f-CNTs may be used as more efficient controlled delivery system.
Abstract
Surface engineered carbon nanotubes (CNTs) are attracting recent attention of scientists owing to their vivid biomedical and pharmaceutical applications. The focus of this review is to highlight the important role of surface engineered CNTs in the highly challenging but rewarding area of nanotechnology. The major strength of this review lies in highlighting the exciting applications of CNTs to boost the research efforts, which unfortunately are otherwise scattered in the literature making the reading non-coherent and non-homogeneous.
Introduction
In the current scenario, CNTs are one of the forefronts in a diverse field of medicine, which are continuously being explored by researchers, scientists and academicians for the development of new, safe and effective nanomedicine. The CNTs are promising quasi one-dimensional nanomaterial with well ordered flat networks of fused benzene rings or hollow graphitic cylindrical tubular structure consisting of hexagonal arrangement with sp 2 hybridized carbon atoms 1, 2, 3, 4. CNTs are mainly classified into four types, based upon the structure and diameters including single-, double-, triple-, and multi-walled carbon nanotubes. The length of the tubes can be extended depending on the type of production method 5, 6, 7, 8. CNTs are one of the attractive tools in nanotechnology with surface engineered carbon nanotubes (f-CNTs), emerging as a new family of carbon nanovectors that enabled numerous biomedical applications. Functionalization plays a pivotal role in the design of CNTs-based nanomaterials by rendering them more biocompatible and less toxic with increased dispersibility in comparison to pristine CNTs. The methodology of functionalization of CNTs can be classified broadly into two categories: (a) covalent attachment of active biochemical moieties through chemical reactions onto CNTs skeleton, and (b) non-covalent adsorption or wrapping of different functional molecules 7, 9. The various types of nanocomposites have been summarized in Fig. 1 .
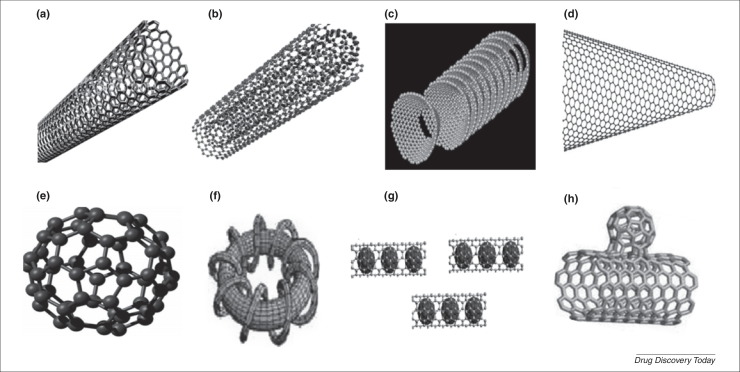
Fig. 1.
Various nanocomposites: (a) SWCNTs, (b) MWCNTs, (c) Cup-stacked CNTs, (d) Carbon nanohorns, (e) Fullerenes, (f) Carbon nanotorous, (g) Carbon nanopeapods, and (h) Carbon nanobuds (Adopted from www.google.com; accessed date: 21st September, 2014).
Cellular trafficking mechanism of carbon nanotubes
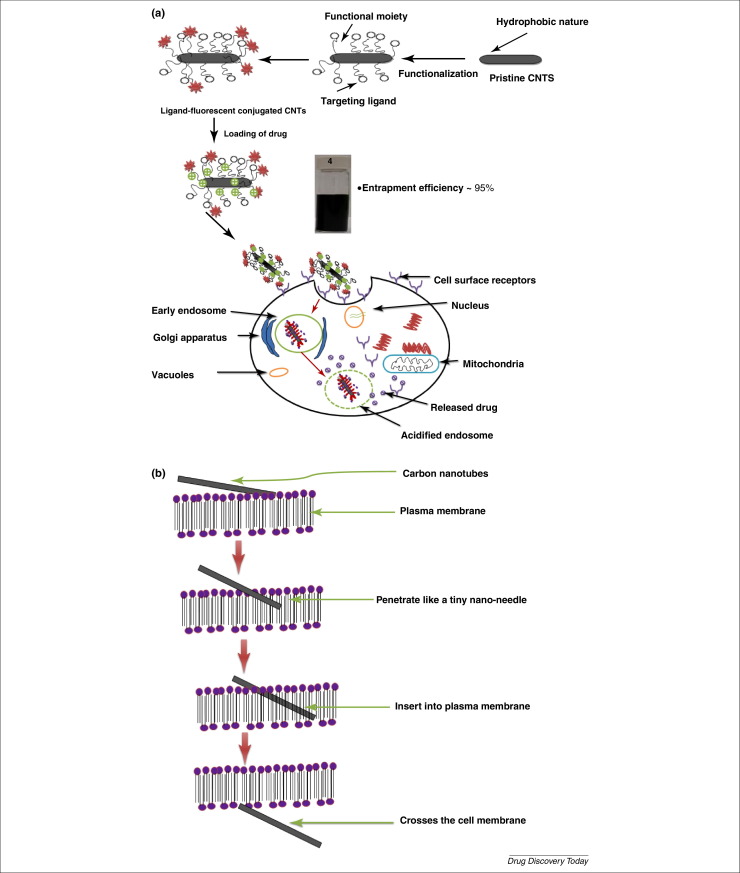
The study of cellular uptake mechanism of the surface engineered CNTs has been intensely addressed in the last two decades 10, 11. Lacerda et al. have demonstrated that the surface engineered CNTs have propensity to enter the cells via an energy-dependent, endosomally-mediated cellular internalization approach and direct cytoplasmic translocation through insertion or passive diffusion in a non-invasive manner (tiny nanoneedle mechanism) 11, 12, 13.
The f-CNTs have been easily cross the blood brain barriers (BBB) without requirement of any external transporter device owing to nano-dimension and tiny nano-needle tubular structure morphology (Fig. 2 A). The main pathways speculated by scientists for cellular uptakes of f-CNTs are: (i) phagocytosis, (ii) CNTs piercing into membrane by passive diffusion, (iii) caveolae-mediated endocytosis, (iv) clathrin-mediated endocytosis, and (v) caveolae-clathrin mediated endocytosis. The intracellular uptake mechanisms of CNTs are crucial for the controlled uptake and devising new strategies in the field of novel drug delivery 12, 13.
Fig. 2.
(A) Receptor mediated endocytosis targeted drug delivery via parenteral administration of CNTs formulation. (B) CNTs penetrate and cross the plasma cell membrane due to their tiny needle dimension.
Reports have shown that the nanotubes may enter cells rapidly with free trafficking into the cytoplasm in the first hour of internalization. This penetration of surface engineered CNTs into phagocytic and non-phagocytic cells is mediated by three alternative pathways: i) via membrane wrapping as individual tubes; ii) via direct membrane translocation of individual nanotubes; and iii) in bundles within vesicular compartments (Fig. 2 B) [13]. These exciting facts about intracellular trafficking of CNTs have opened a new door for the development of a promising delivery system for therapeutic and diagnostic agents [11].
Pharmaceutical and biomedical applications of surface engineered carbon nanotubes
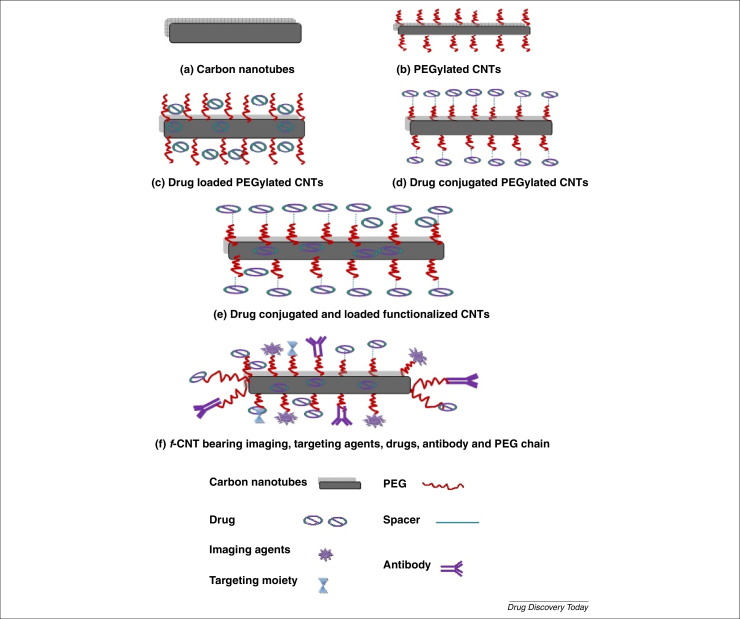
The potential of surface engineered CNTs and possible biomedical applications are discussed below and various CNTs conjugates employed in bioactive delivery shown in Fig. 3 .
Fig. 3.
CNTs conjugates employed in bioactives delivery.
Transdermal drug delivery
Transdermal drug delivery system (TDDS) represents a convincing as well as promising alternative to oral or injectable delivery of drugs. Recently, CNTs have been investigated as TDDS due to their tiny nano-needle shape structure and superior adsorption capacity. Im and co-workers prepared an electro-sensitive TDDS of CNTs using electrospinning method to control drug release with polyethylene oxide and pentaerythritol triacrylate polymers. In this TDDS, CNTs facilitated the electro-sensitive-transdermal drug delivery [14]. The advantages of CNTs in TDDS include high loading efficiency and enhanced transdermal penetration of drugs by application of a small electrical bias to create a programmable drug delivery system. The future explorations of f-CNTs in TDDS present a feasible solution to limitation of current smoking cessation and opioid withdrawal symptom treatments 15, 16.
Ophthalmic drug delivery
Several approaches have been exploited in the development of safe, biocompatible and comfortable nanovehicle for controlled ophthalmic drug delivery with enhanced patient compliance and reduced toxicity. Ophthalmic drugs have a short residence time (<5 min) and only 1–5% of applied therapy may penetrate the cornea and enter into the intraocular tissues. The CNTs-based ocular drug delivery has been found to enhance the bioavailability of few ophthalmic medications overcoming the limitation of traditional ophthalmic drug delivery methods [17]. CNTs could also be used for ocular targeting of different therapeutic agents to the different ocular sites. Unfortunately only very few reports are available in this area [18]. Application of ‘Kajal’ (also known as kohl or surma) is a common practice in Indian families. Kajal has been claimed for prevention and treatment of various eye diseases (blepharitis, cataract and conjunctivitis etc.) and also said to ward off an ‘evil eye’. Cosmetic application of CNTs is well documented in various literatures 19, 20, 21.
Central nervous system disorders
Surface engineered CNTs based delivery systems have been explored as a new platform to repair the damaged CNS tissue/cells in the field of neuroscience. The CNTs have been claimed to be able to cross the blood brain barrier (BBB) by different targeting mechanisms and act as safe and effective targeted drug delivery system 22, 23. Since the last one-decade researchers have continuously explored and assessed the CNTs as substrate for neuronal tissue growth, regeneration and its application in neuroscience research. CNTs have numerous salient features with a simple and inert molecular tubular nanoneedle structure and porosity beneficial to neural interface for trafficking of cells, sensing of microenvironments, delivery of transfection agents, and scaffolding for incorporating within host body, to provide the support for adhesion, proliferation and migration of cells. Ren and co-workers developed a dual targeted delivery system based on the oxidized PEGylated MWCNTs modified with Angipep-2 (a targeting ligand of low-density lipoprotein receptor related-protein (LRP) receptors) loaded with Doxorubicin (DOX) and systematically evaluated their in vitro and in vivo anti-glioma effect by C6 cytotoxicity and median survival time (MST) of intracranial C6 glioma bearing Balb/c mice. The combination of O-MWCNTs-PEG with angiopep-2 as targeting ligand was claimed to play a role of active dual-targeting and combination of O-MWCNTs-PEG and angiopep-2 constituted an ideal dual-targeting drug delivery to brain glioma [22].
Photodynamic therapy
Photodynamic therapy (PDT) is an effective, alternative, non-invasive, non-toxic therapy used in oncology [24]. Till date only few numbers of reports are available on the applications of surface engineered CNTs in PDT. Photosensitizer [5-aminolevulinic acid (5-ALA)] loaded PAMAM dendrimers modified MWCNTs (dMNTs) showed good biocompatibility with significant increase in the accumulation of 5-ALA in MGC-803 tumor cells leading to a potential photodynamic anti-tumor effect [25]. Chitosan wrapped Chlorine6 (Ce6), which is a photosensitizer, loaded SWCNTs revealed high cellular uptake (high anticancer effects) and low toxicity against HeLa cancer cells than free Ce6 [26].
Photothermal therapy
CNTs hold promise in photothermal therapy (PTT) due to extraordinary photon-to-thermal energy conversion efficiency with high absorption in the near infra-red (NIR) light 27, 28, 29. The SWCNTs have the ability to absorb 700–1100 nm NIR radiation and convert it into heat, which kills cancer cells [29]. Moon and co-workers demonstrated the photothermal effect of PEGylated SWCNTs upon NIR irradiation on KB tumor bearing nude mice and observed that sufficiently high thermal energy was generated from optically excited PEG-SWCNTs to destroy tumor in a non-invasive manner. The mice treated with PEG-SWCNTs and NIR irradiation showed the complete eradication of solid malignant tumor devoid of any toxicity or abnormal behavior 20 days post treatment. The issue concerned with residual SWCNTs accumulation inside the body after the treatment was resolved as nanotubes were excreted from mice's body in about 2 months through the biliary or urinary pathway in biodistribution study [27]. Hashida and co-workers reported the photothermal ablation activity of a novel SWCNTs-peptide composite with a designed peptide having a repeated structure of H-(-Lys-Phe-Lys-Ala-)7-OH[(KFKA)7] against tumor cells. The obtained results suggest the remarkable suppression of tumor growth compared with only SWCNT-(KFKA)7 injection alone or NIR irradiation alone [28].
Gene therapy
Currently viral vector system based gene delivery is most widely used to achieve high efficiency of gene expression, which concerns the safety, as viral vector can be immunogenic, oncogenic, and may be responsible for inflammation. Further, gene therapy may suffer rapid degradation of exogenous nucleic acid. Now-a-days some effective multiple non-viral delivery systems like polymeric nanoparticles, liposomes, cell penetrating peptides, cationic lipids, dendrimers and f-CNTs are being employed to efficiently deliver nucleic acids including siRNA with reduced immunogenicity. The efficiency of gene expression mediated by these nanovectors is lower as compared with the viral vector but they provide the advantages of easy up-scaling, flexibility and reduced immunogenicity. The applications of CNTs as new vectors for delivery of nucleic acids is another area of research and development which could offer several advantages for nucleic acid, siRNA, gene and protein delivery owing to their thin and long architecture that offers a large surface area to which siRNA can be bound and delivered to target site. Further, CNTs may have protective capacity, which stabilize and lead to more efficient cellular transfection and prevent the lysosomal degradation of encapsulated nucleic acids [30]. Kawaguchi and co-workers synthesized DNA-functionalized SWCNTs to improve the dispersibility as compared to acid treated SWCNTs. The binding ratio of the acid-treated SWCNTs and DNA was calculated to be 3:1 (DNA/SWCNTs) from the phosphorous content in DNA-SWCNTs [31]. In context of the enhanced gene delivery Sanz and co-workers were first to develop and demonstrate the enhanced gene delivery using lysosomotropic anti-malarial drug chloroquine loaded polyethyleneimine (PEI)-coated DWCNTs to enhance the cell transfection efficiency [32].
Treatment of infectious diseases
Infectious diseases such as tuberculosis, leishmaniasis, severe acute respiratory syndrome (SARS), flu (swine, bird and avian) and anthrax infection indicate that infectious diseases have emerged as a critical public health issue with global concerns. Surface engineered CNTs have been used in the treatment of infectious diseases due to their ability to easily conjugate drugs like amphotericin B (AmB) and dapsone. Leishmaniasis is a microbial disease in which macrophages and other cells of reticuloendothelial system (RES) serve as host as well as replication site for the parasite [33]. Conjugation of amphotericin B (AmB) to f-CNTs can modify its properties in terms of toxicity and anti-mycotic efficiency 34, 35.
Wu and co-workers explored the toxicity and antifungal activity of AmB loaded CNTs (AmB-CNTs) functionalized with fluorescein isothiocynate (FITC) for efficient nuclear localization in human Jurkat lymphoma T cells and observed that AmB-CNTs could be easily taken up by mammalian cells precluding any specific toxic effect but preserving antifungal activity [34]. CNTs surface engineered with mannose showed promising targeted delivery of AmB to J774 macrophages cells [36].
Cancer theranostics
Cancer is one of the leading causes of death worldwide. More than 99% cases of chemotherapy destroy cancer cells along-with the normal healthy cells, with serious side effects and hence strategies based on nanotechnology are being explored as ‘safe and effective’ strategy to achieve selective uptake of chemotherapeutic agent by target cancer cells [7].
A significant decrease in toxicity with preferential killing of cancer cells was achieved with dual targeted nanocarrier combining MWCNTs and iron oxide magnetic nanoparticles (MNs) conjugated with FA for antineoplastic agent, DOX [37]. Jain and co-workers observed tumor targeted delivery of DOX and reduced toxicity with DOX loaded dexamethasone mesylate anchored MWCNTs to A-549 lung epithelial cancer cells [38]. Further, CNTs surface engineered with folic acid and D-alpha tocopheryl polyethylene glycol 1000 succinate (TPGS) showed tumor targeted delivery of DOX with improved cytotoxicity toward cancer cells and enhanced cellular uptake speculated to be mediated by endocytosis and tiny nano-needle mechanism 2, 8. Fig. 3 presents the different conjugates based on CNTs employed in the delivery of anticancer bioactives varying from pristine CNTs to drug loaded CNTs. From the aforesaid account it may be concluded that the surface engineered CNTs could have promising potential in cancer therapy.
Macrophages targeting
Reports showed that CNTs may emerge as promising nanocarrier in targeting of macrophages owing to their exceptional physicochemical characteristics. AmB conjugated CNTs showed improved antileishmanial activity as compared to free AmB in J774A.1 macrophage cells [39]. Later, AmB loaded mannose conjugated CNTs showed efficient macrophage targeting in J774 macrophage cell lines with significantly reduced toxicity toward RBCs and kidney cells showing considerable reduction in the nephro- and hemolytic-toxicity, which are the major constraints in clinical use of AmB [36].
A brief summary of various bioactives delivered through surface engineered CNTs is shown in Table 1 10, 13, 22, 34, 36, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55.
Table 1.
Various bioactives delivered through functionalized CNTs for pharmaceutical and biomedical applications
| Functionalized CNTs | Cell lines used | Bioactives agents used | Physicochemical/biological parameters | Outcomes of the study | References |
|---|---|---|---|---|---|
| FITC-MWCNTs & Oxidized MWCNTs | Human Jurkat T lymphoma cells | Amphotericin B (AmB) via covalent attachment | Electron microscopy Antifungal activity |
AmB covalently linked to CNTs is taken up by mammalian cells devoid any specific toxic effect. f-CNTs are able to enter the cell by a spontaneous mechanism. | [34] |
| PL-PEG-FA & PL-PEG-FITC-SWCNTs. | HeLa cells | Cy3-labeled DNA | Atomic force microscopy Confocal laser scanning microscopy |
SWCNTs internalized in living cells can act as tiny NIR ‘heaters’ or ‘antennas’. | [10] |
| PL-PEG-RGD-SWCNT & FITC annexin V-SWCNTs. | MCF-7 breast cancer and U87 MG cancer cell lines | Doxorubicin | Fluorescence Supramolecular loading efficiency |
Enhanced uptake of DOX in case of integrin-positive U87 MG using RGD-based targeting relative to integrin-negative MCF-7 Cells. | [40] |
| PL-PEG-SWCNTs | NTera-2 | Cisplatin prodrug conjugate | Fluorescence microscopy | Cytotoxicity of the free platinum(IV) complex increases by >100-fold when the complex is attached to the surface of the functionalized SWNTs. | [41] |
| Pluronic F 127 stabilized-MWCNTs | MCF-7 human breast cancer cells | Doxorubicin through π–π stacking | Electron microscopy Confocal laser microscopy |
Enhanced cytotoxicity of DOX-MWCNTs complex over free DOX. | [42] |
|
DSPE-PEG5000-PTX-SWCNTs PEG- SWCNTs. |
4T1 tumor-bearing mice (tumor size, ∼200 mm3). | Paclitaxel (PTX) | Pharmacokinetics and biodistribution study Antitumor efficacy Pilot toxicity study |
Water-soluble SWCNTs-PTX formulation is cremophor-free. SWCNTs have shown to be safe at least in mouse models. |
[43] |
| CNTs | Bladder cancer cells | Carboplatin | Electron energy loss spectroscopy X-ray-photoelectron spectroscopy Cell proliferation Cytotoxicity |
High efficacy of carboplatin filled CNTs with reduced growth of bladder cancer cells compared to free drug CNTs. | [44] |
| Folic acid (FA)-chitosan (CHI)/alginate (ALG)-SWCNTs. | HeLa cell line | Doxorubicin through π–π stacking. | Fluorescence Cell viability and cell uptake studies |
CNTs complex found to be more selective and effective than free DOX due to targeting based on FA and release of DOX at lysosomal pH. | [45] |
| mAB-BSA adsorbed-SWCNTs. | WPDr colon cancer cell line | Doxorubicin through π–π stacking. | Electron microscopy (SEM, HRTEM) Raman spectroscopy |
Uptake study revealed that delivery efficiency was 100%. All cells could take up the SWCNT complexes. |
[46] |
| Diaminotriethylene glycol-MWCNTs. | MKN-28 cells and H22 cell lines | 10-Hydroxycamptothecin (HCPT) through π–π stacking. |
In vitro single photon emission computed tomography (SPECT). ex vivo gamma scintillation counting analysis |
MWCNTs-HCPT conjugate enhanced superior antitumor activity both in vitro and in vivo to clinical HCPT formulation. They show relatively long blood circulation and high drug accumulation at the tumor site. | [47] |
| Dapsone-MWCNTs-FITC | Subvert normal macrophage (MØ) and PMØ | Dapsone (DDS) | Confocal laser microscopy Phagocytosis assay |
Dapsone-MWCNTs did not trigger oxidative stress but caused apoptosis of PMØ predominantly after prolonged cultivation (3 days). | [48] |
| Azomethine ylide (1.3-dipolar cycloaddition)-MWCNTs. | MCF-7 Cell line. | Methotrexate (MTX) through cleavable ester bond. | Electron microscopy Cytotoxicity study |
Cytotoxic activity of CNTs conjugate was strongly dependent on the presence and type of linker. | [49] |
| EGF-SWCNTs–cisplatin–EGF | Head and Neck Squamous Carcinoma Cells (HNSCC), HN12 cells |
Cisplatin | Histological analysis Raman analysis Annexin V assay |
PEG-SWCNTs, as opposed to control SWCNTs, form more highly dispersed delivery vehicles that, when loaded with both cisplatin and EGF, inhibit growth of squamous cell tumors. | [50] |
| Sgc8c aptamer-SWCNTs | Molt-4 (Target) and U266 non-target (B lymphocyte human myeloma) cell line | Daunorubicin (anthracycline antibiotic) through π–π stacking | Cell viability Loading efficiency ∼157% Flow cytometry analysis |
Dau-aptamer-SWCNTs complex was able to selectively target Molt-4 cells compared to non target cells. | [51] |
| CNT-PEI and CNT-pyridinium-MWCNTs | Human lung cancer cells (H1299) | siRNA | Gene silencing and cytotoxicity agarose gel electrophoresis | CNT-PEI and CNT-pyridinium did not show any added value over PEI, pDMAEMA, and Lipofectamine used as reference transfection agents for siRNA silencing activity and cytotoxicity. | [52] |
| Hyaluronate tethered MWCNTs | A 549 cell line | Doxorubicin (DOX) | Pharmacodynamic and organ distribution studies Tumor growth inhibition studies Apoptosis study Cytotoxicity study |
The 99mTc-MWCNT-HA-DOX is significantly higher than both free drug and non-targeted MWCNTs in organ distribution studies on Ehlrich ascites tumor (EAT) bearing mice. The tumor-growth inhibitory effect of HA-MWCNTs-DOX was found to be 5 times higher than free DOX in chemically breast-cancer-induced model in pharmacodynamic studies devoid of any detectable cardiotoxity, hepatotoxicity, or nephrotoxicity. |
[53] |
| DOX-FA-MN-MWCNT | U87 cell lines | Doxorubicin | Electron microscopy CLSM. |
Dual targeted DOX-FA-MN-MWCNTs showed the high drug loading, pH-dependent controlled release, magnetic targeting, and ligand–receptor interactions. | [37] |
| AmB/Mannose-MWCNTs | J774 cells | Amphotericin B (AmB) | CLSM In vitro & in vivo studies macrophage uptake study |
AmBitubes showed better targeting efficiency to macrophages i.e. J 774 cell lines with reduced toxic effects associated with AmB. | [36] |
| DOX/DEX-MWCNTs | A-549 cells | Doxorubicin (DOX) | In vitro & ex vivo studies | DOX/DEX-MWCNTs found less hemolytic and more cytotoxic as compared to free DOX on A-549 lung epithelial cancer cell line. | [38] |
| Functionalized MWCNTs | Raw 264.7 and A-549 cell lines | – | Immunofluorescence microscopy | The chemical functionalization tailors the fate of CNTs by tilting the balance toward specific mechanisms of internalization, cellular processing and elimination/degradation. | [13] |
| Angiopeptide conjugated PEGylated oxidized MWCNTs | Brain capillary endothelial cells (BCEC) and C6 glioma cells | Doxorubicin (DOX) | Anti-glioma efficacy Intracellular fluorescence distribution. Fluorescence imaging |
The results showed that DOX-O-MWCNTs-PEG-ANG is a promising dual-targeting carrier to deliver DOX for the treatment of brain tumor | [22] |
| f-SWCNTs-COS-GTX-p53 | HeLa and MCF-7 cells | Gliotoxin | Cytotoxicity study Fluorescent activated cell sorter analysis |
The f-SWCNTs-COS-GTX-p53 was found to be most effective delivery vehicle with a controlled release and enhanced cytotoxicity rendered through apoptosis in HeLa cells. | [54] |
| GL-MWCNTs-DOX | HepG2 cell | Doxorubicin | Electron microscopy Flow cytometry and phase contrast microscopy Anticancer activity |
GL-MWCNTs-DOX formulation approach was found to be promising toward development of an affluent liver targeting. | [55] |
MWCNTs: Multi walled carbon nanotubes; SWCNTs: Single walled carbon nanotubes; FITC: Fluorescence isothiocynate; PEI: Polyethyleneimine; PL-PEG: Phoshpholipid-Polyethlene glycol; RGD: Arginine-glycine-aspartic acid; NGR: Asn-Gly-Arg; HCPT: Hydroxycamptothecin; NIR: Near infrared; EGF: Epidermal growth factor; PL-PEG-FA: Phospholipid polyethylene glycol-folic acid; PEI: Polyethleneimine; siRNA: small interfering ribonucleic acid; Amphotericin B: AmB; GTX: Gliotoxin; GL: Glycyrrhizinic acid; DOX: Doxorubicin.
Diagnosis
CNTs alone as well as in combination with other nanocarriers like dendrimers, nanoparticles and quantum dots are emerging as promising novel drug delivery and diagnostic nano-vector tool with minimal toxicity and immunogenicity. Delogu and co-workers assessed the echogenic property of nanotubes in vitro and found that the ultrasound signal of f-MWCNTs is higher than the pristine MWCNTs, graphene oxide and f-SWCNTs. The authors found that the MWCNTs are highly echogenic in liver and heart as examined on pig as an experimental animal model. Thus the nanotubes could show enormous potential as ultrasound contrast agents and theragnostics moiety combining diagnostic and therapeutic modalities [56].
Biological fate of carbon nanotubes
The smart/intelligent surface engineered carbon nanotubes are still having controversy about their biodegradable/non-biodegradable nature. Previous literature suggested non-biodegradable nature of CNTs and excretion through biliary pathway, especially via urine and feces. However, few investigations observed that CNTs are degraded in specific environments via natural, enzymatic catalysis, natural horseradish peroxidase (HRP) with low concentration of H2O2 (approximately 40 μM) at 4 °C over 12 weeks or in the presence of human neutrophil enzyme, myeloperoxidase. Importantly biodegraded nanotubes did not produce any inflammatory response when aspirated in to the lungs of mice 57, 58, 59. Bianco and co-workers investigated the biodegradation of SWCNTs and MWCNTs under different conditions and concluded that the oxidized MWCNTs were highly degrading in nature as compared to SWCNTs [59]. These few recent literature reports support the biodegradable nature of CNTs, otherwise previously, CNTs were considered non-biodegradable and excreted out as examined by electron microscopic studies. Additionally biocompatible nanotubes could be prepared via conjugation of phospholipids (PL) and polyethylene glycol (PEG) chains on to them, which may be a promising alternative candidate in biomedical applications 58, 59, 60.
Regulatory considerations
The toxicities associated with the CNTs are yet unclear. Several research studies demonstrated the toxicity of pristine CNTs, which could fortunately be minimized via surface engineering that renders them more biocompatible and non-immunogenic.
The specific structural properties of CNTs like needle-shape, long and thin and biopersistent nature (insoluble) may produce adverse pulmonary effect but this potential risk could be avoided by use of surface engineering of CNTs. However, till date CNTs’ eco-toxicological data are limited and controversial and also lack reliable information of exposure as per Occupational Health and Safety regulations. In contrast to ecotoxic point of view, surface of CNTs can often be modified with the different chemical reactions or by ‘grafting to’ and ‘grafting from’ make CNTs more biocompatible with better stability. Additionally, there is a significant need for research and development in the field of analytical and detection methods. Strict norms and regulations are needed in the development of new drug products for being safe, effective and economic in generally regarded as safe (GRAS) prominence.
Conclusion and future perspective
CNTs’ potentials as novel carriers in bioactives delivery make them promising candidate for the development of new pharmaceutical products in clinical relevance. Before the clinical use of CNTs based products; their pharmacological and toxicological parameters must be proved to be safe and effective. Surface of CNTs could be easily engineered with targeting and imaging agents, peptides, proteins, siRNA, nucleic acids, antibodies and chemotherapeutic drugs for aforementioned pharmaceutical and biomedical applications. These surface engineered CNTs are considered promising nanomaterial for use in many biomedical applications, including biocompatible modules for the delivery of bioactives. In conclusion, before realizing the full potential of CNTs, it is very important to generate data on their safety, efficacy, and feasibility. Although the scientists are optimistic about the possible application of CNTs yet learning the lesson from past (like thalidomide tragedy), the safety and efficacy of CNTs/CNTs based products/devices etc. must be established beyond doubt. We have witnessed the emergence of several materials/devices/products, which could not make their way to market but then that's the research. Let's keep our fingers crossed and wait for the conclusive proof regarding the possible application of CNTs, particularly in biomedical arena. No conclusion should be drawn either in haste or without conclusive evidence of safety.
Conflict of interest
The authors report no conflict of interest related to manuscript.
References
- 1.Rastogi V. Carbon nanotubes: an emerging drug carrier for targeting cancer cells. J. Drug Deliv. 2014:1–23. doi: 10.1155/2014/670815. Article ID 670815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bartelmess J. Carbon nanomaterials: multi-functional agents for biomedical fluorescence and Raman imaging. Chem. Soc. Rev. 2015 doi: 10.1039/C4CS00306C. (in press) [DOI] [PubMed] [Google Scholar]
- 3.Smith B.R. Selective uptake of single-walled carbon nanotubes by circulating monocytes for enhanced tumour delivery. Nat. Nanotechnol. 2014;9:481–487. doi: 10.1038/nnano.2014.62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gupta R. Fucosylated multiwalled carbon nanotubes for Kupffer cells targeting for the treatment of cytokine-induced liver damage. Pharm. Res. 2013;31:322–334. doi: 10.1007/s11095-013-1162-9. [DOI] [PubMed] [Google Scholar]
- 5.Brahmachari S. Biotinylated amphiphile-single walled carbon nanotubes conjugate for target-specific delivery to cancer cells. J. Mater. Chem. B. 2014;2:1160–1173. doi: 10.1039/c3tb21334j. [DOI] [PubMed] [Google Scholar]
- 6.Mehra N.K. Receptor based targeting of therapeutics. Ther. Deliv. 2013;4:369–394. doi: 10.4155/tde.13.6. [DOI] [PubMed] [Google Scholar]
- 7.Mehra N.K. A review of ligand tethered surface engineered carbon nanotubes. Biomaterials. 2014;35:1267–1283. doi: 10.1016/j.biomaterials.2013.10.032. [DOI] [PubMed] [Google Scholar]
- 8.Mehra N.K. The cancer targeting potential of D-α-tocopheryl polyethylene glycol 1000 succinate tethered multi walled carbon nanotubes. Biomaterials. 2014;35:4573–4588. doi: 10.1016/j.biomaterials.2014.02.022. [DOI] [PubMed] [Google Scholar]
- 9.Jain K. Potential and emerging trends in nanopharmacology. Curr. Opinion Pharmacol. 2014;15:97–106. doi: 10.1016/j.coph.2014.01.006. [DOI] [PubMed] [Google Scholar]
- 10.Kam N.W.S. Carbon nanotubes as multifunctional biological transporters and near-infrared agents for selective cancer cell destruction. Proc. Natl. Acad. Sci. 2005;102:11600–11605. doi: 10.1073/pnas.0502680102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lacerda L. How do functionalized carbon nanotubes land on, bind to and pierce through model and plasma membranes. Nanoscale. 2013;5:10242–10250. doi: 10.1039/c3nr03184e. [DOI] [PubMed] [Google Scholar]
- 12.Lacerda L. Carbon nanotubes cell translocation and delivery of nucleic acids in vitro and in vivo. J. Mater. Chem. 2008;18:17–22. [Google Scholar]
- 13.Lacerda L. Translocation mechanisms of chemically functionalized carbon nanotubes across plasma membranes. Biomaterials. 2012;33:3334–3343. doi: 10.1016/j.biomaterials.2012.01.024. [DOI] [PubMed] [Google Scholar]
- 14.Im J.S. The effect of carbon nanotubes on drug delivery in an electro-sensitive transdermal drug delivery systems. Biomaterials. 2010;31:1414. doi: 10.1016/j.biomaterials.2009.11.004. [DOI] [PubMed] [Google Scholar]
- 15.Degim I.T. Carbon nanotubes for transdermal drug delivery. J. Microencapsulation. 2010;27:669–681. doi: 10.3109/02652048.2010.506581. [DOI] [PubMed] [Google Scholar]
- 16.Strasinger C.L. Carbon nanotubes membranes for use in the transdermal treatment of nicotine addiction and opioid withdrawal symptoms. Res. Treatment. 2009;3:31–39. doi: 10.4137/sart.s1050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chauhan, A. and Gulsen, D. (2009) EP1534202.
- 18.Beg S. Advancement in carbon nanotubes: basics, biomedical applications and toxicity. J. Pharm. Pharmacol. 2011;63:141–163. doi: 10.1111/j.2042-7158.2010.01167.x. [DOI] [PubMed] [Google Scholar]
- 19.Sucapane A. Interactions between cultured neurons and carbon nanotubes: a nanoneuroscience vignette. J. Nanoneurosc. 2008;1:10–16. doi: 10.1166/jns.2009.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mohta A. Kajal (Kohl)-A dangerous cosmetic. Oman J. Ophthalmol. 2010;3:100–101. doi: 10.4103/0974-620X.64242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sweha F. Kohl along history in medicine and cosmetics. Hist Sci. Med. 1982;17:182–183. [PubMed] [Google Scholar]
- 22.Ren J. The targeted delivery of anticancer drugs to brain glioma by PEGylated oxidized multi-walled carbon nanotubes modified with angiopep-2. Biomaterials. 2012;33:3324–3333. doi: 10.1016/j.biomaterials.2012.01.025. [DOI] [PubMed] [Google Scholar]
- 23.Oliveira J.M. Dendrimers and derivatives as a potential therapeutic tool in regenerative medicine — A review. Progr. Polym. Sci. 2010;35:1163. [Google Scholar]
- 24.Tekade R.K. Dendrimers in oncology: an expanding horizon. Chem. Rev. 2009;109:49–87. doi: 10.1021/cr068212n. [DOI] [PubMed] [Google Scholar]
- 25.Huang P. Photosensitizer-loaded dendrimer-modified multi-walled carbon nanotubes for photodynamic therapy. J. Control. Rel. 2011;152(Suppl 1):e33-4. doi: 10.1016/j.jconrel.2011.08.105. [DOI] [PubMed] [Google Scholar]
- 26.Xiao H. Photodynamic effects of chlorine e6 attached to single wall carbon nanotubes through noncovalent interactions. Carbon. 2012;50:1681–1689. [Google Scholar]
- 27.Moon H.K. In vivo near-infrared mediated tumor destruction by photothermal effect of carbon nanotubes. ACS Nano. 2009;3:3707–3713. doi: 10.1021/nn900904h. [DOI] [PubMed] [Google Scholar]
- 28.Hashida Y. Photothermal ablation of tumor cells using a single-walled carbon nanotubes-peptide composite. J. Control. Rel. 2014;173:59–66. doi: 10.1016/j.jconrel.2013.10.039. [DOI] [PubMed] [Google Scholar]
- 29.Gannon C.J. Carbon nanotubes-enhanced thermal destruction of cancer cells in a noninvasive radiofrequency field. Cancer. 2007;110:2654–2665. doi: 10.1002/cncr.23155. [DOI] [PubMed] [Google Scholar]
- 30.Kateb B. Internalization of MWCNTs by microglia: possible application in immunotherapy of brain tumors. NeuroImage. 2007;37:S9–S17. doi: 10.1016/j.neuroimage.2007.03.078. [DOI] [PubMed] [Google Scholar]
- 31.Kawaguchi M. Dispersion stability and exothermic properties of DNA-functionalized single-walled carbon nanotubes. Int. J. Nanomed. 2011;6:636–729. doi: 10.2147/IJN.S17175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sanz V. Chloroquine-enhanced gene delivery. Carbon. 2011;49:5348. [Google Scholar]
- 33.Jain K., Jain N.K. Novel therapeutic strategies for treatment of visceral leishmaniasis. Drug Discov. Today. 2013;18:1272–1281. doi: 10.1016/j.drudis.2013.08.005. [DOI] [PubMed] [Google Scholar]
- 34.Wu W. Targeted delivery of amphotericin B to cells by using functionalized carbon nanotubes. Angew. Chem. Int. Ed. 2005;44:6358–6362. doi: 10.1002/anie.200501613. [DOI] [PubMed] [Google Scholar]
- 35.Benincasa M. Antifungal activity of amphotericin B conjugated to carbon nanotubes. ACS Nano. 2011;5:199–208. doi: 10.1021/nn1023522. [DOI] [PubMed] [Google Scholar]
- 36.Pruthi J. Macrophages targeting of amphotericin B through mannosylated multi walled carbon nanotubes. J. Drug Target. 2012;20:593–604. doi: 10.3109/1061186X.2012.697168. [DOI] [PubMed] [Google Scholar]
- 37.Lu Y.J. Dual targeted delivery of doxorubicin to cancer cells using folate-conjugated magnetic multi-walled carbon nanotubes. Colloids Surf. B: Biointerfaces. 2012;89:1–9. doi: 10.1016/j.colsurfb.2011.08.001. [DOI] [PubMed] [Google Scholar]
- 38.Lodhi N. Development and characterization of dexamethasone mesylate anchored on multi walled carbon nanotubes. J. Drug Target. 2013;21:67–76. doi: 10.3109/1061186X.2012.729213. [DOI] [PubMed] [Google Scholar]
- 39.Prajapati V.K. Targeted killing of leishmania donovani in vivo and in vitro with amphotericin B attached to functionalized carbon nanotubes. J. Antimicrob Chemother. 2011;66:874–879. doi: 10.1093/jac/dkr002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Liu Z. Supramolecular chemistry on water soluble carbon nanotubes for drug loading and delivery. ACS Nano. 2007;1:50. doi: 10.1021/nn700040t. [DOI] [PubMed] [Google Scholar]
- 41.Feazell R.P. Soluble single-walled carbon nanotubes as longboat delivery systems for platinum (IV) anticancer drug design. J. Am. Chem. Soc. 2007;129:8438. doi: 10.1021/ja073231f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ali-Boucetta H. Multiwalled carbon nanotube-doxorubicin supramolecular complexes for cancer therapeutics. Chem. Commun. 2008;4:459–461. doi: 10.1039/b712350g. [DOI] [PubMed] [Google Scholar]
- 43.Liu Z. Drug delivery with carbon nanotubes for in vivo cancer treatment. Cancer Res. 2008;68:6652. doi: 10.1158/0008-5472.CAN-08-1468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hampel S. Carbon nanotubes filled with a chemotherapeutic agent: a nanocarrier mediates inhibition of tumor cell growth. Nanomedicine. 2008;3:175–182. doi: 10.2217/17435889.3.2.175. [DOI] [PubMed] [Google Scholar]
- 45.Zhang X. Targeted delivery and controlled release of doxorubicin to cancer cells using modified single wall carbon nanotubes. Biomaterials. 2009;30:6041–6047. doi: 10.1016/j.biomaterials.2009.07.025. [DOI] [PubMed] [Google Scholar]
- 46.Heister E. Drug loading, dispersion stability, and therapeutic efficacy in targeted drug delivery with carbon nanotubes. Carbon. 2009;47:2152. [Google Scholar]
- 47.Wu W. Covalently combining carbon nanotubes with anticancer agent: preparation and antitumor activity. ACS Nano. 2009;3:2740–2750. doi: 10.1021/nn9005686. [DOI] [PubMed] [Google Scholar]
- 48.Vukovic G.D. The response of peritoneal macrophages to dapsone covalently attached on the surface of carbon nanotubes. Carbon. 2011;48:3066–3078. [Google Scholar]
- 49.Samori C. Enhanced anticancer activity of multi-walled carbon nanotubes-methotrexate conjugates using cleavable linkers. Chem. Commun. 2010;46:1494–1496. doi: 10.1039/b923560d. [DOI] [PubMed] [Google Scholar]
- 50.Bhirde A.A. Distribution and clearance of PEG-single-walled carbon nanotubes cancer drug delivery vehicles in mice. Fut. Nanomed. 2010;5:1535–1546. doi: 10.2217/nnm.10.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Taghdisi S.M. Reversible targeting and controlled release delivery of daunorubicin to cancer cells by aptamer wrapped carbon nanotubes. Eur. J. Pharm. Biopharm. 2011;77:200–206. doi: 10.1016/j.ejpb.2010.12.005. [DOI] [PubMed] [Google Scholar]
- 52.Varkouhi A.K. siRNA delivery with functionalized carbon nanotubes. Int. J. Pharm. 2011;416:419. doi: 10.1016/j.ijpharm.2011.02.009. [DOI] [PubMed] [Google Scholar]
- 53.Datir S.R. Hyaluronate tethered smart multi walled carbon nanotubes for tumor-targeted delivery of doxorubicin. Bioconj. Chem. 2011;23:2201–2213. doi: 10.1021/bc300248t. [DOI] [PubMed] [Google Scholar]
- 54.Bhatnagar I. Polymer functionalized single walled carbon nanotubes mediated drug delivery of gliotoxin in cancer cells. J. Biomed. Nanotechnol. 2014;10:120–130. doi: 10.1166/jbn.2014.1677. [DOI] [PubMed] [Google Scholar]
- 55.Chopdey P.K. Glycyrrhizin conjugated dendrimer and multi-walled carbon nanotubes for liver specific delivery of doxorubicin. J. Nanosci. Nanotechnol. 2015;15:1088–1100. doi: 10.1166/jnn.2015.9039. [DOI] [PubMed] [Google Scholar]
- 56.Delogu L.G. Functionalized multiwalled carbon nanotubes as ultrasound contrast agents. Proc. Natl. Acad. Sci. 2012;109:4116612. doi: 10.1073/pnas.1208312109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Allen B.L. Biodegradation of single-walled carbon nanotubes through enzymatic catalysis. Nano Lett. 2008;8:3899–3903. doi: 10.1021/nl802315h. [DOI] [PubMed] [Google Scholar]
- 58.Kagan V.E. Carbon nanotubes degraded by neutrophil myeloperoxidase induce less pulmonary inflammation. Nat. Nanotechnol. 2010;5:354–359. doi: 10.1038/nnano.2010.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Russier J. Oxidative biodegradation of single- and multi-walled carbon nanotubes. Nanoscale. 2011;3:893–896. doi: 10.1039/c0nr00779j. [DOI] [PubMed] [Google Scholar]
- 60.Kostarelos K. Promises, facts and challenges for carbon nanotubes in imaging and therapeutics. Nat. Nanotechnol. 2009;4:627–633. doi: 10.1038/nnano.2009.241. [DOI] [PubMed] [Google Scholar]