Highlights
-
•
Targeting specific organelles treat or decrease symptoms of several diseases.
-
•
Manipulation of nanomaterials characteristics can improve the intracellular delivery.
-
•
Approaches to escape to endocytic route and non-endocytic route as an alternative.
-
•
Strategies to reach the target, maintaining the original drug bioavailability.
Abstract
The intracellular delivery of nanomaterials and drugs has been attracting increasing research interest, mainly because of their important effects and functions in several organelles. Targeting specific organelles can help treat or decrease the symptoms of diabetes, cancer, infectious, and autoimmune diseases. Tuning biological and chemical properties enables the creation of functionalized nanomaterials with enhanced intracellular uptake, ability to escape premature lysosome degradation, and to reach a specific target. Here, we provide an update of recent advances in the intracellular delivery mechanisms that could help drugs reach their target more efficiently.
Teaser
This review provides an update of recent advances in intracellular delivery and reports mechanisms that could help drugs reach their target efficiently, resulting in smarter drugs that reach their target still with the original bioavailability.
Introduction
Developments in nanomedicine delivery have provided new perspectives of the design and synthesis of efficient nanocarriers and multifunctional nanomaterials. Initially, research focused on nanocarrier biocompatibility and toxicity, whereas second-generation nanomaterials aimed to have an optimized surface, to provide more stability, stealth, and targeting capabilities. The most recent models support the ‘smart nanomedicine’ idea, improving targeting mechanisms and theranostic abilities [1]. This nanotechnological advance is important because it enables drugs to cross physiological barriers to reach their target sites safely and sustainably [2]. In fact, nanomaterials provide a stable biocompatible environment to encapsulate drugs, promoting their controlled release and efficient absorption 3, 4. They also improve the duration of the therapeutic effects and minimize adverse effects by driving drugs toward the site of action and increasing the concentration of a drug in the area of pathology in a specific way 5, 6. Depending on the accumulation of the delivery system in the tissue, cell, or in a specific subcellular compartment of interest, targeting approaches can be primary, secondary, or tertiary, respectively [7].
Subcellular targeting is vital for efficient, specific treatments and, thus, specific barriers must be overcome. The importance of organelle targeting increases when drugs efficiently treat or decreases symptoms of diseases such as cancer, Alzheimer’s, diabetes, infectious and autoimmune diseases [8]. Specifically, the intracellular environment contains compounds, responsible for cell growth, proliferation, differentiation, and death, that are, therefore, promising drug targets. Thus, target sites can be distributed throughout the cytoplasm, nucleus, mitochondria, endoplasmic reticulum (ER), and Golgi complex [9]. However, endosomes and lysosomes, which have a low pH and are rich in enzymes, can result in drug degradation or nonspecific distribution 10, 11. Thus, modulation of the size, charge, and surface composition of a nanomaterial can dictate the internalization pathway, enabling the nanomaterial to evade lysosomes and interact with its target organelle [12]. Indeed, the potential of nanomaterials to overcome such barriers has led to the development of platforms capable of improving their bioavailability [13].
Therefore, there is a need to fully understand the pathways of nanomaterial uptake and intracellular mechanisms to create smarter drugs that can reach the target site with their original bioavailability [14]. Here, we review each step between drug administration and the target site to identify all the challenges that must be overcome for successful drug delivery. This review will help researchers design more efficient nanomaterials, resulting in enhanced drug uptake.
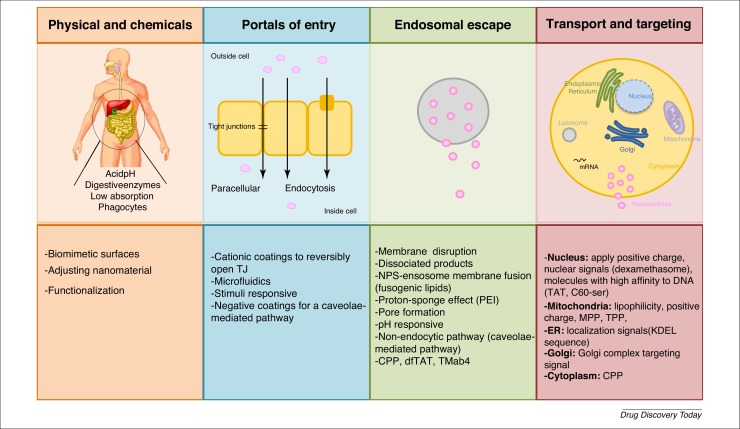
Physical and chemical barriers
There are three fundamental steps during the production of nanomaterials for drug delivery that must be taken into account: (1) the nanomaterial must be able to survive the harsh conditions of the gastrointestinal tract (GIT); (2) once internalized, it must reach its target site and; (3) the pharmacokinetics of the drug must be maintained.
Nanomaterials can be administrated by several routes, with the oral route being the most convenient [15]. However, for efficient oral delivery, nanomaterials must pass through the oral cavity, which is rich in proteins, mucosal compounds, and bacterial flora, followed by the stomach, which is highly acidic, and finally the intestine, where the nanomaterials will be subjected to the activity of digestive enzymes, the presence of a mucus layer (formed of the glycoprotein mucin), and tight junctions (TJs). Once they have crossed the endothelial barrier, the nanomaterials reach the blood, where they can be taken up unspecifically by phagocytic cells or by specific target cells [16]. In addition, target sites for a drug can be distributed throughout the cytoplasm, nucleus, mitochondria, and lysosomes [9].
The GIT is the main barrier to the successful absorption of nanomaterials, particularly in terms of drug degradation because of enzymes, such as proteolytic enzymes in lysosomes, brush-border peptidases in the villi, and pancreatic proteases in the duodenal region, and bacterial flora within the gut, which also contains mucin. Epithelial cells also constitute a physiological barrier, because they are bond by TJs. Nanomaterials be modified physicochemically in response to low pH, high temperatures, reactions with surrounding molecules, in addition to changes to the weight and charge of the nanomaterial, which will also influence their absorption and degradation [17]. An additional barrier is the intestinal epithelium, which controls the passage of drugs into the blood. Its surface area, 99% of which is covered by enterocytes and microvilli, significant impacts absorption [18]. Morphologically, intestinal crypts comprise absorptive enterocytes and secretory cells (e.g., goblet cells, enteroendocrine cells, and Paneth cells). In addition, there are isolated lymphoid follicles and Peyer’s patches, which contain specialized M cells 3, 17.
Lastly, to reach their target site for their original bioavailability retained, nanomaterials must overcome the mononuclear phagocyte system, avoid becoming nonspecifically distributed, which can lead to drug resistance, and escape from endosomes [19]. As discussed in-depth below, the mononuclear phagocyte system can be overcome by modulating certain characteristics of a nanomaterial, while its nonspecific distribution can be avoided by the use of functionalization or addition of biomimetic surfaces; drug efflux pumps and consequent drug resistance can be avoid by the use of CD44 targeting and drugs can escape endosomes by disrupting their membrane [11].
Cellular internalization
After reaching its target cell, the internalization of a nanomaterial involves a set of processes, including pre-absorption, uptake, translocation, and reaching its final target 3, 20. Initially, the nanomaterial activates its own transport into the cell via receptor recognition by the ligands that it contains.
Transport mechanisms
There are several possible pathways for the internalization of a nanomaterial. Here, we focus on the paracellular pathway and endocytosis, because these are the pathways most used by nonfunctionalized and functionalized nanomaterials,
The paracellular route is mainly exploited by nanomaterials of a particular nature, size, or with an absence of receptors. The nanomaterial is transported through two juxtaposed cells via aquaporins and TJs. Aquaporins are proteins that facilitate water transport across the luminal space to the basolateral membrane [21], whereas TJs maintain the structural integrity of the membrane. TJ are gaps between cells and contain occludin and claudin-2 proteins, which aids paracellular transport because of their permeability [22]. The presence of TJs and a low surface area limit the transport of nanomaterials, because they cannot pass through the intestinal barrier via this route. The TJs must be reversibly opened by chemically modifying the nanomaterial using permeation enhancers, such as chitosan, polyacrylate, and thiolated polymers (anionic and cationic polymers) [23]. Thus, the paracellular pathway mainly transports hydrophilic drugs 3, 6. A recent study used a cationic cyclodextrin-polyethylenimine 2k conjugate complexed with anionic mRNA encoding HIV gp120 to open TJs and enhance the paracellular transport of the nanomaterial [24].
Thiolated polymers, known as thiomers, are hydrophilic polymers that are conjugated with hydrophilic macromolecules for enhanced uptake. Thiomers have the ability to mimic the mechanism of secreted mucus glycoproteins, covalently binding through disulfide bonds with cysteine from the mucus, enhancing their mucoadhesion and inhibiting enzyme actions [25]. Thus, thiomers remain concentrated at the absorption membrane as a result of the protection of their thiol groups and efflux inhibition [26]. Thiomers reduce oxidized glutathione (GSSG) to GSH at the same time that thiol groups are oxidized to disulfide bonds, attaching the nanomaterial to the mucosa glycoproteins [27]. Then, GSH binds to the cysteine active site of protein tyrosine phosphatase (PTP) via a disulfide bond, preventing PTP from remove the phosphate group from the tyrosine residues. This results in the TJs opening, improving the permeation properties of the nanomaterial and facilitating its diffusion [28]. An early study showed PTP inhibition using sodium orthovanadate, which led to tyrosine phosphorylation at TJs and improved the permeability in bovine corneal epithelial cells [29]. TJs are responsible for the uptake of hydrophilic nanomaterials throughout the paracellular route and their association with thiomers increases their apparent permeability 30, 31.
By contrast, functionalized and some nonfunctionalized nanomaterials are typically endocytosed. Endocytosis involves engulfment in membrane invaginations and the formation of intracellular vesicles 9, 11. This pathway represents the classic mechanism of nanomaterial uptake, and can be divided into five types. Even when the nanomaterial is already inside the cell, drugs can be pumped out via drug efflux pumps, resulting in drug resistance [32].
Phagocytosis is a receptor-mediated process, where large particles are engulfed by the cellular membrane. It is limited to macrophages, dendritic cells, and M cells 33, 34. By contrast, pinocytosis is the uptake of fluids and solutes and can be subclassified as macropinocytosis, clathrin- and caveolae-mediated uptake, and clathrin- and caveolae-independent uptake. In macropinocytosis, a large volume of fluid containing nanomaterials is engulfed. It is an active and actin-dependent process and, thus, requires actin to grow and cholesterol to extend the membrane 33, 35. It is also nonspecific receptor mediated and, thus, does not require specific ligand–receptor binding or a specific cellular type. However, cellular stimulation is required to initiate the process [36].
Clathrin-mediated endocytosis is a receptor-mediated uptake mechanism, under the control of dynamin. Dynamin is a GTPase protein that leads to the division of clathrin pits from the membrane, resulting in the formation of small vesicles (60–200 nm) coated in clathrin. After internalization, clathrin is removed by auxilin and HSC70 and vesicles are fused with Rab5 early endosomes 33, 37. This mechanism is seen as a housekeeping mechanism that degrades unwanted proteins and maintains cellular homeostasis; it is a route usually involving low-density lipoproteins, transferrins, and ligand–receptor complexes [36]. Pharmacological inhibitors have been used to analyze the topology of endocytosis 38, 39. Steinbach and co-workers used chlorpromazine (specific for clathrin), nystatin (specific for caveolin), and LY294002 (specific for macropinocytosis) to show that clathrin-mediated endocytosis was the predominant mechanism for the internalization of nanoparticles (NPs) [39], as have other studies 37, 40. Yet, when nanomaterials are decorated with certain ligands, such as folic acid and cholesterol, caveolin-mediated endocytosis is the mechanism used [11], as demonstrated using albumin by Chanthick and colleagues [41]. Small caveolae-coated vesicles (50–80 nm), also under the control of dynamin, are formed through flask-shaped invaginations in a nonspecific uptake process. Therefore, caveolin is an integral membrane protein responsible for the formation of caveolae and, consequently, the formation of invaginations through caveolin-1 expression. Its morphology and functions differ depending on the cell type. During internalization, caveolae are endocytosed together with vesicles. Caveolae are similar to lipid rafts, because they also comprise cholesterol and sphingolipids, as well as glycosylphosphatidylinositol (GPI) and several kinases. In contrast to clathrin, caveolae-mediated endocytosis requires a signal to begin. Endocytosis starts with tyrosine phosphorylation of the associated protein, which leads to depolymerization and enlistment of actin [36]. This leads to escape from the endolysosome and to the direct exocytosis of drug carriers 33, 35.
Finally, clathrin- and caveolin-independent transport occurs via nonspecific endocytosis, without the need for a receptor; it is also characterized as dynamin in- and dependent pathway 3, 36. In such cases, EIPA is usually used as a transport inhibitor [42].
A recent promising biomanufacturing advance for the intracellular delivery of nanomaterials was microfluidics, a membrane disruption method. Microfluidics is a technology that improves the quality of the delivery system and establishes a structure–function relationship of a nanomaterial, with additional advantages of a uniform-sized emulsion droplet and the understanding of intracellular delivery barriers. However, it is not yet clear whether it is possible to scale-up this system for whole-body use [43]. The microfluidic device passes between single cells creating transient pores in the plasmatic membrane, which enhance the intracellular delivery via mechanoporation [44]. Its efficiency has already been demonstrated for the intracellular delivery of NPs loaded with dexamethasone; the authors also studied the effect of NP design on the fate of stem cells [45]. Others have used this approach to prevent the rapid clearance and consequent loss of microbubbles using flow-focusing microfluidic devices, in real time and near to the therapeutic site [46].
Nanomaterial characteristcs and their influence
Internalization and intracellular transport mechanisms are affected by properties of the nanomaterial being used (Fig. 1 ). Different characteristics (size, charge, shape, or elasticity) affect the interaction between the nanomaterial and the cell membrane, causing altered signaling events and activation of different pathways of adhesion and internalization. Thus, the desired result, that is, more effective and specifically targeted delivery at a predetermined rate and time, can be obtained by controlling these parameters 1, 15.
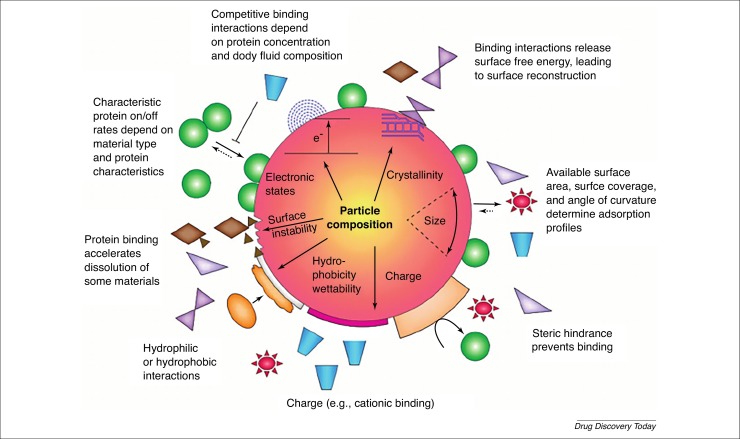
Figure 1.
Effects of a protein corona surrounding a nanoparticle. The corona constitutes a primary nanobio-interface that determines the fate of the nanoparticle and can have deleterious effects on the interactive proteins. Pre-existing or initial material characteristics contribute to the formation of the corona in biological environments. Characteristic protein attachment and/or detachment rates, competitive binding interactions, steric hindrance by detergents and adsorbed polymers, and the protein profile of the body fluid lead to dynamic changes of the corona. The corona can change when particles move from one biological compartment to another. Reproduced, with permission, from Ref. [47].
Size
Size is a vital parameter because it determines the endocytosis pathway and which cells take up the particle. Particles <10 nm are mainly removed by renal clearance [48]. When orally administrated, 10–100-nm particles have higher cellular uptake efficiency, because they are taken up in Peyer’s patches, and then absorbed into the systemic circulation, avoiding rapid renal clearance. By contrast, larger particles are removed by the mononuclear phagocyte system (MPS), and those that are 200–300 nm become concentrated in the spleen 49, 50. During internalization, larger particles (>1 μm) enter the cell via macropinocytosis; those that are >120 nm enter via the clathrin-dependent pathway; those that are 50–100 nm enter via the caveolae-dependent pathway and smaller particles (<50 nm) are internalized via the clathrin- and caveolae-independent pathway. Thus, internalized nanomaterials are correlated with the size of vacuole formed [20].
A penetration enhancer can be added to macromolecules with high molecular weight, such as dimethyl palmitoyl ammonio propanesulfonate (PPS), a zwitterionic surfactant. According to microscopy studies, PPS allows macromolecules to diffuse via the paracellular and transcellular pathways, improving their bioavailability by 45-fold [51]. In addition, polyacrylate, chitosan [52], phytic acid [53], and self-assembling lipid-like peptides [54] also work as enhancers, as well as thiomers and lectins [55]. These enhancers have the ability to reversibly open TJs, increasing the paracellular transport of macromolecular drugs because of the downregulation of occludin and claudin (TJ proteins), and the synergism of reductive effects of Ca2+ 56, 57. In terms of the latter, the cations bind with more affinity to polyacrylate derivatives, inhibiting lumen enzymes (e.g., trypsin). Thus, the polyacrylate-Ca2+ complex triggers the opening of TJs 58, 59. It was also demonstrated that cyclodextrin improves macromolecular absorption via pulmonary delivery [60]. Finally, an alternative strategy is the use of palmitoyl glycol chitosan hydrogels [61] or the development of liposomes with enhancers to delivery hydrophilic macromolecules [62].
Surface composition
The surface of a nanomaterial is also important because it is in contact with the cell, and this contact can be affected by the hydrophobicity or hydrophilicity of the nanomaterial surface. Thus, this parameter determines the cellular internalization and intracellular transport and influences the circulation time and nanocarrier association with, and recognition by, cells. Hydrophobic particles can be delivered to immune cells and penetrate the bilayer membrane, becoming easily internalized 11, 34. Polymers are added to nanomaterials to render their surface more inert and hydrophilic to avoid immune clearance and increase the blood circulation time. (Poly)ethylene glycol (PEG)ylation is a strategy to render nanomaterials invisible to macrophages or phagocytes and to prolong their half-life [63]. PEG chains are covalently conjugated to nanomaterials, rendering them hydrophilic [64]. PEG can capture water molecules and form a natural barrier around the nanomaterial, preventing the proteins from being adsorbed, which is known as the ‘stealth’ effect [65].
Another strategy is to use biomimetic surfaces, whereby the nanomaterial surface is modified with a natural membrane coating to avoid phagocytic clearance, prolong drug circulation, and improve the biocompatibility. Markers, such as CD47 [66] or even leukocytes, are attached to the surface of the nanomaterial to camouflage it, in that the body recognizes it as ‘self’ [67].
Surface charge
The surface charge of the nanomaterial can also be manipulated. For example, intestinal cell membranes are negatively charged because of the presence of phospholipids; thus, positively charged nanomaterials interact more strongly with such membranes, enhancing their internalization [9]. A typical example is polyethyleneimine (PEI), which is commonly used to permeate membranes and enable nanomaterials to enter the cell [68]. In terms of transport, positively charged particles utilize the clathrin-mediated pathway and macropinocytosis [69], whereas negatively charged particles tend to utilize the caveolae-mediated pathway 23, 41. Neutral or slightly negatively charged particles are considered the most appropriate for long circulation, because they are not susceptible to MPS. By contrast, nanomaterials that are highly positively or negatively charged are taken up by macrophages 18, 23, 70.
Shape
Shape can also dictate the path of the nanomaterial and can change over time or after receiving external stimuli to enable a long circulation and overcome diffusion barriers. It is an important factor in flow margination, the avoidance of the immune system, and tissue accumulation. Spherical particles are better internalized by macrophages, whereas asymmetrical nanomaterials are more advantageous for sensing, self-assembly, tissue engineering, immunoengineering, and therapeutic and diagnostic delivery 1, 34. Thus, one strategy is to create elliptical particles to escape macrophages and then expose it to a stimulus, to change it into a sphere for improved internalization 20, 71.
Elasticity and solubility
Finally, increasing elasticity also increases blood residence time and avoids clearance by the immune system. More elastic nanomaterials use macropinocytosis, whereas less elastic ones are internalized via clathrin-dependent mechanisms 34, 72.
Another popular method is to chemically modify nanomaterials to change the fluidity and/or solubility of the mucus for oral administration. For example, it is possible to add fatty acids or to substitute amino acids to obtain increased solubility. The addition of fatty acids is proportional to the increased solubility, which can increase 16-fold with linoleic acid [73]. A similar effect was also observed with capric, lauric, and oleic acids. These fatty acids increase hydrophilic transport and reduce transepithelial electrical resistance, indicating the opening of TJs [74]. Paracellular transport has been related, in several papers, to the transport of poorly absorbed drugs. A study revealed the relation between paracellular transport and fatty acids and their effects on the absorption of mannitol, a paracellular marker, and on morphology changes of TJs [75]. Researchers added 20 amino acids to ribonuclease (RNase) Sa at position 76, and confirmed that asparagine, glutamine, and threonine had the most significant effects on protein solubility [76].
In short, various characteristics (Table 1 ) influence the uptake, renal clearance, drug stability, and circulation time of nanomaterials, as well as the drug release. These properties can even be changed via internal (pH, redox, temperature, or enzymes) and external stimuli (ultrasound, magnetic, light, or electrical) 9, 15. Stimuli-responsive nanocarriers, already reviewed elsewhere [77], are a smart approach for intracellular drug delivery. Recently, authors inclusively created porous silica nanocarriers sensitive to three stimuli (proteases, redox, and pH) for drug delivery [78].
Table 1.
The influence of nanomaterial characteristics on cellular uptake and internalization pathways and strategies for enhancing their uptake
| Characteristic | Cellular uptake | Internalization pathway | Strategy | |
|---|---|---|---|---|
| Size | <10 nm | Renal clearance | – | Add a penetration enhancer (PPS, polyacrylate, chitosan, phytic acid, self-assembling lipid-like peptides, thiomers, and lectins) to macromolecules to open TJs |
| 10–10 nm | Peyer’s patches | <50 nm: clathrin and caveolae independent; 50–100 nm: caveolae dependent | ||
| 100–200 nm | Removed by MPS | 120 nm: clathrin dependent | ||
| 200–300 nm | Spleen | Macropinocytosis | ||
| Surface composition | Hydrophobic | Immune cells | N/A | Avoid phagocytic clearance, prolong drug circulation, and improve biocompatibility by (i) adding hydrophilic polymers (PEGylation); or (ii) having biomimetic surfaces (CD47, leukocytes) |
| Charge | Positive | Highly positive: taken up by macrophages | Clathrin and macropinocytosis | Positively charged nanomaterials interact more strongly with negatively charged intestinal cell membrane |
| Neutral | Appropriate for long circulation residency | N/A | ||
| Negative | Highly negative: taken up by macrophages; slightly negative: appropriate for long circulation residency | Caveolae mediated | ||
| Shape | Spherical | Macrophages | N/A | Elliptical particles that escape macrophages and, after exposure to stimuli, change to a spherical form for better internalization |
| Asymmetrical | N/A | N/A | ||
| Elasticity and solubility | Soft |
Avoid immune system |
Macropinocytosis |
Add fatty acids and/or substitute amino acids |
| Hard | N/A | Clathrin-dependent mechanisms | ||
Blanco and co-workers summarized the main effects of NP size, shape, and surface charge among different organs, including lungs, liver, spleen, and kidneys [11]. It is also necessary to analyze the presence of nanomaterials in the target tissue or organ. This is currently possible using methods such as magnetic resonance, ultrasound, fluorescence positron emission tomography (PET), phosphorescence, X-ray, optical coherence tomography, photoacoustic and Raman, fluorescence and photoacoustic approaches. By contrast, intracellular delivery can be sensed by electrochemistry or electrophysiology methods, as reviewed elsewhere 79, 80.
Manipulation of the different characteristics of a nanomaterial can help functionalized and nonfunctionalized nanomaterials to reach their targets, as discussed below.
Nonfunctionalized nanomaterials
Nonfunctionalized nanomaterials are less specific carriers, because they do not have a specific target, limiting their functionality as well as rendering them less stable [81]. However, these carriers can delivery drugs to or into cells, as is the case for nanocrystals, NPs and macromolecules. These can be taken up by M cells, enterocytes of the intestinal epithelium, or even by phagocytes [82]. Nonfunctionalized nanomaterials are usually used for the negative control of functionalized nanomaterials.
Nanocrystals
Nanocrystals are solid particles formed by a set of atoms that can be structured with an aspect ratio of 10–100. Cellulose nanocrystals (CNCs) are elongated and rigid, and are formed by the acidic hydrolysis of cellulose [83]. Nanocrystals have the ability to be loaded with high amounts of drug, which makes them an ideal way of obtaining pharmacological effects [82]. CNCs with PEI have been developed to delivery small interfering (si)RNA for gene silencing. As an antitumor strategy, this formulation protected the siRNA and facilitated its delivery into tumor cells via endosomal escape or the ‘proton sponge effect’ [84]. In another study, the effect of unmodified CNCs and their cationic derivatives was evaluated in mitochondria stress. The authors revealed that these compounds induced reactive oxygen species (ROS) production and an immune response [83]. The beneficial effect of nonfunctionalized CNCs was also demonstrated by adding them to β-lactoglobulin to prolong stability at low pH [85]. In addition, the surface pH influences the affinity, agglomeration, and aggregation of nanomaterials. According to some results, surface pH affects phagocytosis and cellular homeostasis, whereby TF-2 was able to trigger inflammatory responses at acid pH whereas TF-1, with a basic pH, suppressed the release of cytokines [86]. In terms of cellular internalization, nanocrystals enter cells through passive diffusion [87] or even by endocytosis 88, 89.
Nanoparticles
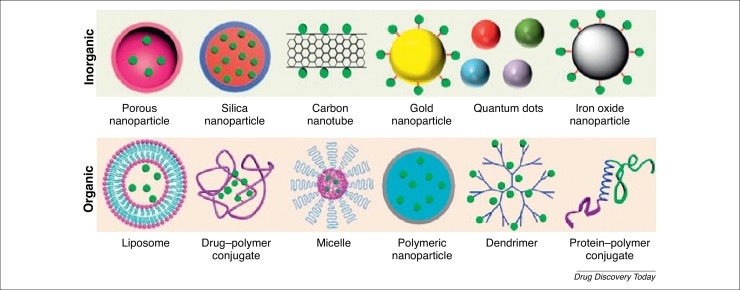
NPs (1–1000 nm) are nontoxic, safe carriers that can be divided into inorganic [e.g., porous NPs, silica NPs, carbon nanotubes, gold NPs (AuNPs), quantum dots, and iron oxide NPs] and organic (e.g., liposomes, drug–polymer conjugates, micelles, polymeric NPs, dendrimers, and protein–polymer conjugates) [90] (Fig. 2 ). For instance, porous NPs are characterized by a high loading capacity, chemical and physical robustness, low toxicity, and easy and cheap production. They can be divided into mesoporous silica NPs (MSNs) and porous silicon NPs (PSiNP) 91, 92. In both types, it is possible to avoid the prior release of drugs by controlling the binding affinity with cargos. Gatekeepers (e.g., AuNPs) can be attached to the pore outlets via covalent bonds that can be broken by an externally applied stimulus (e.g., light, magnetic field, ultrasound, or temperature) or by an internal stimulus inherent to the treated pathology (excluding acid or basic environments and the presence of enzymes) [13]. For instance, silica-encapsulated solid lipid (SESL) particles were developed for cinnarizine (CIN) delivery to overcome the limited oral absorption of this weakly basic drug because of its pH dependency under intestinal conditions [93]. Another example of inorganic NPs are QDs, which can be rapidly taken up by macrophages, and have the ability to move around the cytoplasm and target nuclear histones via nuclear pore complexes [94]. Despite their small size (2–10 nm), this ability is due to the charge associated with the QDs. QDs are also theranostic, showing the potential to promote drug delivery into cells [95]. Such intracellular drug delivery has been demonstrated with nonfunctionalized AuNPs, whereby unmodified AuNPs (i.e., without surface modification) were shown to interact cytoplasmically with pancreatic tumor cells [96].
Figure 2.
Examples of inorganic and organic nanoparticles used in medicine. Adapted, with permission, from Ref. [90].
Altering the charge and surface characteristics of NPs can maximize their internalization and intracellular transport. Cationic liposomes effects have been previously studied [97] and are a frequent choice for gene delivery [98]. A recent study combined Trp3 protein with cationic liposomes to improve the efficiency of plasmid DNA (pDNA) delivery to the nucleus of mammalian cells [99]. Liposomes, as a main nonviral vector, can be produced for pDNA delivery into cells via macropinocytosis [100]. Liposomes comprise 3–4 nm of lamellar phospholipid bilayers, with a hydrophilic center and a hydrophobic part of the membrane containing amphiphilic molecules. Liposomes are taken up by monocytes and macrophages, which is an advantage if the aim is to modulate immunity 16, 101. However, they occasionally induce complement activation, which can lead to hypersensitivity reactions [102].
Polymeric NPs are solid colloidal particles in which the drug is entrapped, encapsulated, or absorbed in biocompatible polymeric materials to improve drug absorption across biological membranes [103]. Polymeric materials can be biodegradable [such as poly(lactide) (PLA), poly(lactide-co-glycolide) copolymers (PLGA), poly (ε-caprolactone) (PCL) and poly(amino acids)], nonbiodegradable [such as poly(methyl methacrylate) (PMMA), polyacrylamide, polystyrene, and polyacrylates] or natural (such as chitosan, dextran, heparin, alginate, gelatin, and albumin) [1]. Polymeric NPs are widely used for drug delivery in several cells, including macrophages 104, 105, and are also able to reach organelles, such as mitochondria and nucleus [106]. They appear to be a promising strategy for use in treating inflammatory bowel diseases [107], diabetes [108], cancer 109, 110, and in ophthalmological conditions [111].
In drug–polymer conjugates, polymeric NPs are conjugated with drugs via either direct covalent linkage or noncovalent interactions. Normally, the two components are connected by a stable or cleavable linker, especially if the drug has to reach the intracellular target of the conjugate [112]. A recent study by Liang et al. provided an example of a stable polymer–drug conjugate with enhanced anticancer effects for treating colon cancer. The authors chemically conjugated the amine group of gemcitabine (GEM; 2′,2′difluorodeoxycytidine) with the PLA carboxylic acid group of a methoxy (m)PEG-PLA polymer block. PEG avoids protein absorption and the defense system of the body, while the hydrophobic PLA renders the conjugate biocompatible, biodegradable, and nontoxic [113].
Micelles are organic NPs that comprise amphiphilic copolymers (15–80 nm) and are usually formed by self-assembly in a liquid [102]. They have a hydrophilic shell, which prevents interactions with the solvent and renders the loaded micelles stable in aqueous solution, and a hydrophobic core, which can hold therapeutic drug molecules [101]. Micelles have attracted interest for drug administration owing to their high stability and solubilization capacity because of their hydrophilic shell, controlled drug release, and low toxicity. A recent study proposed a micelle formulation of PLGA-PEG-PLGA to prevent cancer metastasis. The authors showed that a derivative of ursolic acid can act as an anticancer agent, inducing apoptosis by targeting the nucleus and mitochondria of cancer cells [106].
Macromolecules
Despite their size, macromolecules can also diffuse across membranes, as shown, for example, across skin layers [114]. Their delivery can also be achieved with an external stimulus. For instance, controlled release can be achieved using near-infrared (NIR)-light irradiation in AuNPs associated with macromolecules and light-responsive microcapsules, resulting in the heating and disruption of the capsules [115]. There are also other ways to delivery macromolecules, such as by mechanoporation, where pores are formed to enable access [116]. A common strategy is the conjugation of macromolecules to mucoadhesive polymers, such as chitosan, to open TJs and facilitate paracellular transport [117].
The mechanisms of nonfunctionalized nanomaterial internalization are not yet well established, and more studies are required. Therefore, the use of functionalized nanomaterials is an alternative approach for several biomedical applications.
Functionalized nanomaterials
Functionalized nanomaterials are directed to a specific target, improving their effect and minimizing adverse effects by driving drugs towards the exact site of action. The upgrading of their absorption, distribution, metabolism, and excretion (ADME) properties enhances their therapeutic efficiency 16, 20.
Active targeting of nanomaterials refers to when drugs reach the right target via site-specific targeting. This means that there is an interaction between the targeting ligands on the nanocarrier and the receptors on the cell membranes. This recognition is specific, promoting particle internalization 120, 121. Thus, modifying the surface of nanomaterials with particular ligands is one way to improve their cellular uptake and endosomal escape [39]. Table 2 summarizes receptors targeted in this approach, including CD31, integrin β3 and transferrin [122], TLR2, TLR3 and TLR9 [123], αvβ3 integrin [119], transferrin [124], EGFR [125], CD44 [126], IGFR [127],FcRn [128], CD163 [129], biotin [130], folate [131], and vitamin B12 [132].
Table 2.
Examples of receptor and ligands used in intracellular drug deliverya
| Receptor | Drug | Nanocarrier | Material and/or ligand | Disease | Size | Refs |
|---|---|---|---|---|---|---|
| αvβ3 integrin | c(RGDyC) or CTX | Nanochains | Dextran, IONPs | Glioma | 100 nm | [21] |
| Biotin | QUE and DOX | Polymeric NPs | PEG-PCL | Breast cancer | 105.8 nm | [118] |
| CD163 | Calcein | Liposomes | PEG | Inflammatory and malignant processes | 46 nm | [117] |
| CD31, integrin β3, transferrin | DOX, DIR or DID | Liposomes | SSL, RGD-SSL, 7PEP-SSL | Sarcoma | 100 nm | [110] |
| CD44 | SLM, PTX | Polymeric NPs | PLGA, HA | Breast cancer | 150 nm | [114] |
| EGFR | PTX, parthenolide | Micelles | PEG2000-DSPE, vitamin E-TPGS | Lung cancer | 15 nm | [113] |
| FcRn | Insulin | Polymeric NPs | PLA-PEG, IgG | Diabetes | 63 nm | [116] |
| Folate | Nucleic acids | Polymeric NPs | aminoglycoside-derived | Breast and bladder cancer | < 200 nm | [119] |
| IGFR | Insulin-sodium oleate complex | Polymeric NPs | PLGA, Eudragit FS30D | Diabetes | 213 nm | [115] |
| TLR2, TLR3, TLR9 | MALP-2, poly(I:C), ODN | Polymeric NPs | PLGA, Eudragit FS30D | Genitorectal viral infection | 10 Mm | [111] |
| Transferrin | Cyclosporine A | Polymeric NPs | PLGA, xanthanoid gambogic acid | N/A | 100 nm | [112] |
| VB12 | Insulin | Polymeric NPs | Dextran | Diabetes | 192 nm | [120] |
Abbreviations: 7PEP-SSL, 7PEP functionalized SSL; c(RGDyC), cyclic pentapeptide; CTX, chlorotoxin; FcRn, Neonatal Fc receptor; IGFR, insulin-like growth factor; IONPs, iron oxide nanoparticles; MALP-2, macrophage-activating lipoprotein; ODN, CpG oligodeoxynucleotides; PEG2000-DSPE, 2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[amino(polyethylene glycol)-2000; PEG-PCL, poly(ethylene glycol)-b-poly(ε-caprolactone); poly(I,C), polyinosine-polycytidylic acid; PTX, paclitaxel; QUE, Quercetin; RGD-SSL, RGD functionalized SSL; SLM, salinomycin; SSL, sterically stabilized liposomes; TLR, Toll-like receptors; Vitamin E-TPGS, vitamin E D-alpha tocopheryl polyethyleneglycol succinate.
Cell-penetrating peptides (CPPs) are a group of peptides (10–30 amino acids) with the ability to promote molecular transport and are important for enhancing the intracellular uptake. CPPs can be conjugated via covalent binding to protein drugs and it is believed that endocytosis is involved in their translocation. Given that they are typically positively charged, CPPs, such as glycosaminoglycans (GAGs) and sialic acids, interact electrostatically with negatively charged cells, such as glycosaminoglycans (GAGs) and sialic acids, at the beginning of translocation [133]. A CPP-adaptor fusion protein, TAT-calmodulin (TAT-CaM), has been developed that results in efficient intracellular delivery and endosomal escape. In this example, there is reversible and high-affinity noncovalent binding that delivers the cargo into the cytoplasm of eukaryotic cells [134]. Other researchers have used analogs of the CPP transportan10 conjugated with NPs to transport siRNA, concluding that the pH change leads to more efficient delivery, given that the positive charges are responsible for the stability of the complex [135].
Another approach to nanomaterial functionalization is drug conjugation with lectins, which have a nonimmunological origin, facilitating the interaction with cells and drug diffusion. Given that they contain glycans, lectins can bind specifically and noncovalently to proteins and lipids via glycosylation [136]. Glycans act as receptor-like structures allowing lectins to bind directly to epithelial cells, via cytoadhesion, which can result in the transcytosis of larger molecules [137]. A previous study reported that 15 out of 32 lectins analyzed [e.g., Erythrina crista-galli lectin (ECL), Lycopersicon esculentum lectin (LEL), and wheat germ agglutinin (WGA)] bound to the apical membranes of airway epithelium and were taken up by endocytosis [138]. WGA was analyzed further and shown to have cytoadhesive and cytoinvasive properties, which facilitated the transport of high-molecular-weight molecules. This lectin binds to N-acetyl-glucosamine and sialic acid and improves the binding and uptake of protein-based drugs 139, 140.
Thus, an approach to developing functionalized nanomaterials is to study the target tissue and/or cell for principal receptors or receptors that are overexpressed. Nanomaterials can then be functionalized with ligands (i.e., antibodies and peptides) that will interact with those receptors. In addition, dual ligands can also be used to achieve a synergetic effect [141]. Polysaccharide-nanosystems association as well as sialyl LewisX, hyaluronic acid (HA), heparin, dextran, fucoidan, chitosan, and cyclodextrin are ligands that contribute to local drug delivery, stealthness, enhanced affinity, and stabilization against aggregation [142].
Intracellular transport of nanoparticles
Once inside the cell, nanomaterials have three options: (i) be captured by endosomes and consequently degraded by lysosomes; (ii) escape into the cytoplasm; or (iii) be recycled [39]. However, nanomaterials must escape to reach their intended intracellular target. Thus, endosomal escape is crucial for the intracellular delivery of a drug [9].
Endocytic route and how to escape it
Nanomaterials are trafficked into endosomes via the endolysosomal route. Early endosomes with pH 6 mature to late endosomes with pH 5, which can then fuse with lysosomes, which contain particular digestive enzymes (Fig. 3 ) 9, 12. In addition, endosomal trafficking and maturation can be controlled by peptide association, pH manipulation, or the disperal of drugs into the cytosol 143, 144. Endosome lysis, proton-sponge effect, transport of dissociated products, pore formation, membrane disruption, and fusion of nanomaterials with the limiting membrane are also strategies descibed to escape endosomes [19].
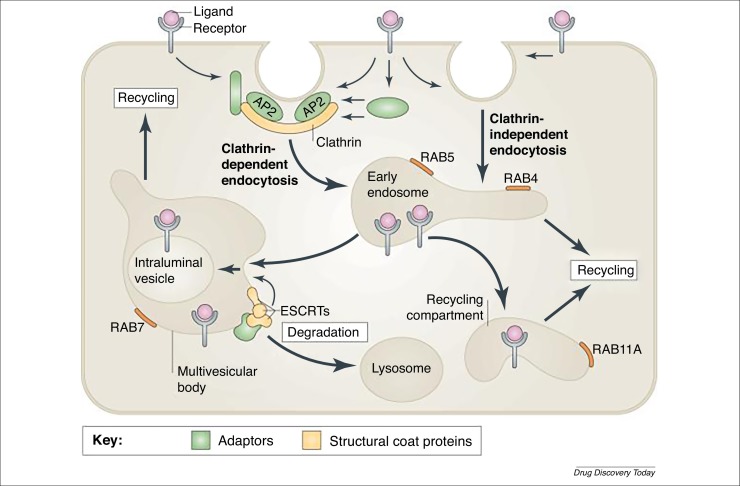
Figure 3.
Pathways of receptor endocytosis. Endocytosis involves the capture of transmembrane proteins and their extracellular ligands into cytoplasmic vesicles that are pinched off from the plasma membrane. The best-studied pathway of receptor internalization is mediated by clathrin-coated pits. These are small areas of the plasma membrane that are covered on the cytoplasmic surface with clathrin triskelions, which comprise three clathrin heavy chains and three clathrin light chains assembled into a polyhedral clathrin lattice. Receptors are recruited to clathrin-coated pits by directly interacting with the clathrin coat adaptor complex AP2 or by binding to other adaptor proteins, which in turn interact with the clathrin heavy chain and/or AP2. Clathrin-coated pits invaginate inwards with the help of several accessory proteins and pinch off to form clathrin-coated vesicles in a process that requires the GTPase dynamin. Several clathrin-independent pathways of endocytosis also exist, although the precise mechanisms and structural components involved in these pathways are not well understood. Endocytic vesicles derived from both clathrin-dependent and -independent endocytosis fuse with early endosomes. Endosomal trafficking is controlled by several Rab proteins, which are small GTP-binding proteins of the Ras superfamily. Each GTP-bound Rab protein resides in a particular type of endosome and functions by recruiting specific effector proteins. Following their internalization into early RAB5-containing endosomes, receptors can rapidly recycle back to the plasma membrane by a RAB4-dependent mechanism, traffic to the recycling compartment that contains RAB11A, or remain in endosomes, which mature into multivesicular bodies (MVBs) and late endosomes. MVBs are defined by the presence of intraluminal vesicles (ILVs), which are formed by inward membrane invagination involving endosomal sorting complex required for transport (ESCRT) complexes. Early-to-late endosome maturation involves the acquisition of RAB7 and the removal of endosomal components that are capable of, and necessary for, recycling. In the MVBs, cargo destined for degradation is incorporated into ILVs. Fusion of late endosomes and MVBs with lysosomes carrying proteolytic enzymes results in cargo degradation. Reproduced, with permission, from Ref. [145].
The proton-sponge effect causes acidification via the translocation of protons into the endosome, causing osmosis, lysis, and release of content. Polyamine-based (e.g., PEI 20, 36) polymers have a pKa of approximately 5–7, which leads to the increased influx of ions via the inflow of water and consequently endosomal membrane rupture. In NP-endosome membrane fusion, fusogenic lipids are incorporated in the carriers. Fusogenic lipids comprise mainly alanine and glycine, as well as certain hydrophobic residues. For instance, 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine (DOPE) is acid responsive, such that the acid pH drives glutamate and aspartate protonation, resulting in a new hexagonal conformational. This facilitates the insertion of fusogenic lipids between juxtaposed membranes and distroys the lipid packing 20, 36. In pore formation, after peptides bind in α-helical structure, they are obliquely inserted into the hydrophobic core of endosomal membrane, which stabilizes the transmembrane pore, and are then oriented via their hydrophilic structures. However, this does not happen during membrane disruption. In this instance, peptides bind to the hydrophilic part of phospholipids and undergo reorientation of their hydrophobic residues, resulting in an amphipathic α-helical structure, which allows peptides to cover the membrane like a carpet and causing disintegration by micellization of the membrane [36].
Berguig and colleagues created antibody–peptide drug conjugates to suppress tumor growth and prolong the drug half life with low-dose cytotoxicity. The polymer comprise two blocks: the first improved the pharmacokinetics and the second, which was pH responsive, enabled endosomal escape. By incorporating the second block, the authors were able to form micelles at physiological pH and disrupt the membrane at endosomal pH, promoting endosomal escape and intracellular delivery of the first block [146]. Other examples include hydrophobic amino acid R groups to aid viral escape from endosomes [147]; Ca2+–siRNA nanocomplexes, where electrotastic interactions lead to siRNA release into the cytoplasm [148]; and TMab4 variants, which resulted in a threefold improved endosomal escape via membrane pores [144]. In addition, research using a disulfide-bonded dimer of TAT (dfTAT), which is found in the cytosol and nucleus, showed that it is essential for not only cell penetration, but also membrane leakage. In this study, Erazo-Oliveras and co-workers demonstrated that dfTAT interacting with bis(monoacylglycero)phosphate (BMP), present in late endosomes, mediated endosomal leakage [112].
Nonendocytic route as an alternative
When nanomaterials escape endosomes, they enter the cytoplasm or exit through the ER or Golgi complex. The cytoplasmic route is generally avoided because most drugs must enter the blood circulation to exert their pharmacological functions [146]. In addition to the difficulty in exiting cells from the basolateral side, nanomaterials also face degradation factors, such as enzymes, in cytoplasm [10].
Those nanomaterials that have an intracellular destiny utilize ER and Golgi routes as well as the retrograde trafficking pathway. This route is responsible for recycling several molecules, such as receptors, which are targeted by the Golgi complex [149]. ER and Golgi complexs are important for both secretory and endocytic pathways, and are also an option to escape the acidic lysosomal environment [12]. Yu and colleagues found a trafficking pathway between the ER and plasmatic membrane (PM), and then from the PM to ER via the Golgi complex. This trafficking was controlled by retromer-mediated endocytic recycling in the Golgi pathway [150]. Binding to the class A receptor enables internalization by caveolae-mediated endocytosis or the lipid raft-mediated pathway [11]. In early endosomes, the nanocarrier content is delivered into the ER, Golgi complex, mitochondria, or even outside the cells [151]. Syntaxin-6 [present in the trans-Golgi network (TGN) and early endosomes] has an important role in retrograde trafficking because of its tyrosine-based sorting motif, which regulates post-Golgi transport and the delivery of certain components, such as caveolin-1, into the membrane. The inhibition of syntaxin-6 proportionally decreases caveolae-mediated endocytosis. Thus, the transport of lipidic classes depends on syntaxin-6 [152]. In addition, syntaxin-6 also interferes with retrograde trafficking of mannose 6-phosphate receptors (in pancreatic β cells) and insulin-responsive membrane proteins (in endocrine cells) to lysosomes, the TGN, and the insulin-responsive compartment. Du and collaborators demonstrated the movement of epidermal growth factor receptor (EGFR) to the Golgi complex via microtubule-dependent movement and its fusion with the complex via syntaxin-6. They also verified that the translocation is necessary for future fusion with the nucleus, which represents a pathway from the cell surface to the Golgi complex or even the nucleus [153]. Thus, this pathway would be preferred for nucleus-targeted delivery rather than diffusing throughout the cytoplasm [20].
Preferred intracellular targets
The final step in the drug delivery process is the most important, given that it is crucial to ensure that the drugs are delivered their site of action with their original pharmacodynamics and bioavailability intact [9].
The delivered drug can target several intracellular components (Fig. 4 ), although the main target organelles are the nucleus, mitochondria, cytoplasm, ER, and Golgi complex 10, 154.
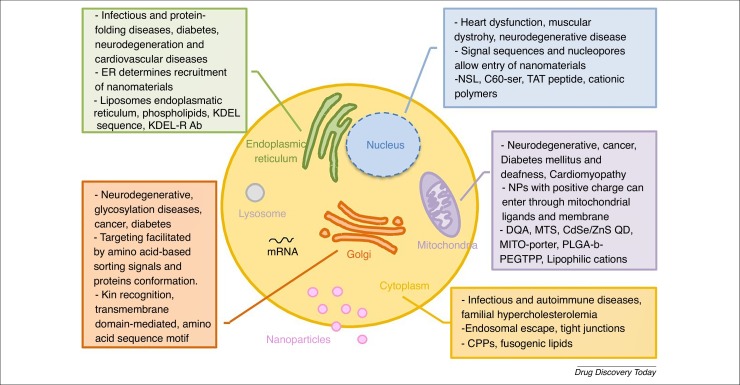
Figure 4.
Possible intracellular sites of action of delivered drugs. The figure details the main target organelles, their associated diseases, characteristics relevant for successful drug internalization and transport, and the nanomaterials commonly used for drug delivery. For definitions of abbreviations, please see the main text.
Nucleus
The nucleus is organized as a double-layered lipidic envelope with nuclear pore complexes (NPCs) (size ∼10 nm) that regulate flow of molecules in and out of the nucleus, and an underlying nuclear lamina. In practice, molecules >9 nm and a molecular weight higher >40 KDa cannot diffuse passively through NPCs. Large molecules need carriers to actively pass through the membrane, such as nuclear localization sequences (NLS). NLS present SV40 sequences containing basic residues (Pro-Lys-Lys-Lys-Arg-Lys-Val) that are recognized by importins, a class of karyopherin-β proteins responsible for nuclear import 155, 156. This organelle controls DNA transcription, reproduction, metabolism, and the cell cycle, and important functions such as electron transport, ATP synthesis, ROS generation, genetic programming, and cellular signaling regulation, can also be affected. It is possible to use nanomaterials smaller than nucleus pores by applying a positive charge to the nuclear membrane 10, 20, establishing interactions with nuclear signals or even using molecules with high affinity for DNA, such as C60-ser or the TAT peptide [7]. The nucleus a crucial role in the treatment of heart diseases, dystrophy, neurodegenerative diseases, cancer and infection, in which gene mutations are common. Inducing nuclear membrane permeabilization or using nanomaterials coated with molecules that are recognized by nuclear receptors present in the cytoplasm (e.g., dexamethasone, which is identified by glucocorticoid receptors) are alternate ways to interfere with DNA transcription and genetic programming [156]. Various systems have been developed for gene delivery, which is the most common way of targeting the nucleus [157]. For instance, micelles with two synergetic anticancer drugs co-encapsulated into HA-vitamin E succinate were internalized via caveolae-mediated endocytosis and accumulated in the nucleus. Cell apoptosis was confirmed, as was reverse multidrug resistance via CD44 targeting [158]. PLGA NPs have also been developed coated with AS1411 aptamer (Apt) and loaded with superparamagnetic iron oxide NPs (SPIONs) with doxorubicin (DOX). Apt is used because it targets nucleolin. Nucleolin proteins are present in the nucleus and have an important role in angiogenesis and cell proliferation and adhesion. Thus, the combination of Apt with DOX is useful in the treatment with cancer, given that is a DNA intercalating agent [159]. Izeddin et al. developed a technique to track protein molecules in the nucleus and determine transcription factor (TF) dynamics. They found that c-Myc and P-TEFb were global and local explorers of the nucleus, respectively. The control of TFs influences gene regulation, and protein reactions and functions in the nucleus, which can be exploited for drug development [160]. Another interesting approach is the use of NPs containing a PEG-benzoic imine-oligo-L-lysine/iridium (III) metallodrug complex with pH-activated size reduction. Thus, at pH 7, they are large and protected by a PEG shell, whereas, at the acid pH of endosomes, NPs remain small because of the dissociation and detachment of PEG. This results in smaller NPs that present oligo-L-lysine that affects nuclear localization, favoring translocation into the nucleus [161].
Mitochondria
Mitochondria are characterized by an external membrane, an intermembrane space, an internal membrane (rich in protein), and the mitochondrial matrix with its own DNA. They are responsible for cellular homeostasis and, thus, are a good target to interfere with via oxidative phosphorylation, apoptosis, production of ROS, intracellular signaling, and metabolism. Thus, it is unsurprising that mitochondria contribute to obesity, Alzheimer’s disease, neurodegeneration, cancer, diabetes mellitus, and cardiomyopathy, given that polymorphisms or mutations that occur in mitochondrial DNA result in deficient energy production. Therefore, lipophilicity and positive charges are required for mitochondrial targeting. The internal negative membrane potential of this organelle is a possible target for positively charged particles [7]. Mitochondriotropic molecules can be used to accumulate nanomaterials inside the cell, because they are amphiphilic. These molecules have the ability to escape endosomes and deliver their cargo into mitochondria [10]. Nanoformulations can be guided by mitochondrial processing proteinases (MPP), which process mitochondrial targeting sequences (MTSs) [162]. DQA, MTS, CdSe/ZnS QD, MITO-porter, PLGA-b-PEGTPP, and lipophilic cations are potential methods for use in this approach [7]. Recently, mitochondria targeting of cancer cells was attempted using a HA protective shell encapsulated with gold nanostars (AuNs) and DOX. The formulation was shown to be internalized via the CD44 receptor. Coated to the AuNs was triphenylphosphine-modified α-helical pro-apoptotic peptide (TPP-KLA), which localizes to mitochondria, interrupt their membranes, inducing dysfunction and, thus, apoptosis [163]. Folate-coated chitosan NPs loaded ursolic acid have also been proposed as an antibreast cancer drug. It was confirmed that these NPs were internalized via a folate-receptor mediated endocytotic pathway. After destroying the permeability of lysosomal membrane, NPs were localized into mitochondria, resulting in the overproduction of ROS, destroying the mitochondrial membrane potential and causing apoptosis of the cancer cells [164]. Several polymeric conjugations are now available, especially for cancer. Chitosan has also been used to reach mitochondria by conjugating it with mitochondria-targeting ligand triphenylphosphine (TPP) to deliver ionidamine and siRNA and to trigger the mitochondrial apoptosis pathway [165]. Thus, the most appropriate way to target mitochondria and the metabolic pathway is to coat nanomaterials with mitochondrial targeting signals and/or peptides conjugated with proapoptotic drugs.
Endoplasmic reticulum
The ER is organized in membrane structures called ‘cisternae’ separated by intermembrane spaces. It is in this organelle that protein folding and lipid biosynthesis occur. It is also responsible for controlling drug detoxification, apoptosis, immune responses, and signaling pathways. It is also is where peptides are loaded to the major histocompatibility complex (MHC) class I and antigens are delivered, thus it acts as a presentation site [10]. Protein transport is mediated by a KDEL sequence that works as a localization signal, which is recognized by its receptor (KDEL-R), expressed in ER. Thus, the ER is an appropriate pharmaceutical target, using a biomimetic approach. Another method for ER targeting is the use of localization signals or antibodies against ER receptors that lead to ER stress, protein misfolding and aggregation, calcium homeostasis disturbance, and exposure to free radicals [7]. Associated with ER dysfunctions are infectious and protein-folding diseases, diabetes, neurodegeneration, and cardiovascular diseases [154]. Murugan and colleagues developed mesoporous silica NP pores loaded with topotecan and coated with poly(acrylic acid)-chitosan conjugated with quercetin. This approach resulted in breast cancer cell death because of structural changes in organelles such as the ER, nucleus, and mitochondria [166]. For the treatment of diabetes, the imaging O2 •− using two photon fluorescent probes (ER-BZT) followed by treatment with metformin has been proposed. Given that the production of ROS leads to ER stress and disease formation [167], it was found that the natural compound jaceosidin contributes to sarco-ER Ca2+-ATPase 2b (SERCA2b) upregulation, decreasing ER stress [168]. Targeting the liposomal ER, phospholipids, KDEL sequence, and KDEL-R antibodies are reviewed elsewhere [7].
Golgi complex
Only 4% of the intracellular delivery of nanomaterials target this organelle, which comprises a system of tubular membranes oriented in a cis or trans direction [154]. The main function of the Golgi complex is N- or O-glycosylation, and protien transport and sorting, in addition to its influence on lipid biosynthesis [169]. The TGN is in the center of both the endocytic and exocytic pathways, with an important role in straightening lipids and proteins. It is normally associated with ER, given that different cargos are transported from there to the cis-Golgi network and then to TNG, where cargos are sorted and forwarded to their destinations [170]. Matsuto and colleagues demonstrated that BICD2 enables Rab6A binding to the Golgi complex. They also showed that Rab6A and BICD2 are responsible for the fusion between Golgi tubules and the ER in retrograde trafficking [171]. It is unsurprising that Golgi complex dysfunctions contributes to various diseases, such as Alzheimer’s, cancer, neurodegenerative diseases, and diabetes [154]. In 2011, a Golgi complex-targeting signal was discovered in a coronavirus responsible for severe acute respiratory syndrome. It was shown that, in the cytoplasmic tail, there is a beta-hairpin structural motif that controls Golgi complex localization [172]. Exploiting this motif could be a way of improving the targeting of the Golgi complex. Banfield has also described several mechanisms of protein retention in the Golgi complex, such as kin recognition, transmembrane domain-mediated and amino acid sequence motifs, which could also be targeted for drug delivery [169].
Cytoplasm
The cytoplasm is an intracellular space that contains various organelles, and can be a target or a way to reach specific organelles. However, in terms of the latter, drugs are diffusing and interact randomly with organelles. For instance, siRNA acts in the cytoplasm, as do nanomaterials that have high metabolic stability [173]. SH3 and SH2 of SLAP can be delivered into the cytoplasm to inhibit T cell receptors, which can be useful for the treatment of autoimmune diseases [174]. Typically, this organelle is targeted when infectious and autoimmune diseases and familial hypercholesterolemia occur [8]. There are several approaches available that allow the delivery of nanomaterials into the cytoplasm, such as CPPs [175] and cationic nanomaterials [176]. Liposomes with fusogenic properties [177] or that are pH sensitive have also been studied as drug carriers [178], given that they have the ability to destabilize endosomal membranes and release drugs into the cytoplasm [9]. The use of polyampholyte NPs and freeze concentration has been suggested to deliver proteins into the cytoplasm [174], as has the application of a faded electric treatment for siRNA delivery [179] via connectosomes and diffusion across TJs [180], bubble liposomes and ultrasound [181]. Enzyme-sensitive or redox-responsive linkers responsible for degradation of cell membrane have also been reported [7].
Given the nanomaterial parameters described above, the most ideal route of delivery remains the oral route [81]. Currently 43 drugs have been approved as nanopharmaceuticals (reviewed in [182]). For example, for intracellular applications, Feraheme™ (Ferumoxytol) was approved in 2009 by the US Food and Drug Administration (FDA) to treat the iron deficiency anemia in adults with chronic kidney disease. This nanopharmaceutical comprises SPIONS coated with dextran, and the iron is released inside macrophages, from where it is then added to the intracellular storage iron pool or transferred to plasma transferrin. In 2014, Plegridy® (Biogen), a polymer-protein conjugate (PEGylated IFN beta-1a) with improved protein stability due to PEGylation, was approved for the treatment of multiple sclerosis. This drug binds to type I interferon receptors on the surface of cells and causes intracellular events that lead to the regulation of interferon-responsive gene expression. Similarly, in 2015, Onivyde® (liposomal Irinotecan) was approved by the FDA for treating pancreatic cancer, because it increases the intracellular uptake of irinotecan and reduces the systemic toxicity arising from adverse effects (www.fda.gov/).
Concluding remarks
Functionalized nanomaterials are currently the most appropriate and successful way for a drug to reach a specific target and have reduced adverse effects and increased efficiency. However, there are several parameters that must be taken into account, such as their size, charge, composition, and shape, because these are correlated with cell uptake, long circulation times, renal clearance, and drug stability. As discussed above, there are various obstacles that must be overcome for successful drug delivery, culminating in the final step characterized by endosomal escape, organelle location, and membrane translocation. Some drugs require delivery to a specific organelle, increasing the delivery challenge. It is evident that organelle dysfunction is the origin of several diseases; therefore, one treatment strategies is to target a set of organelles to reach the desired therapeutic effect.
Future nanoformulations should aim to exploit the most successful approaches described above to result in safe, effective and targeted drug delivery. As detailed in Fig. 5 , the ideal nanoformulation would have a positive surface charge and ligand affinity to improve endocytosis, enabling them to follow the retrograde pathway to avoid lysosomal degradation or to escape to endosomes. Finally, a biomimetic strategy could be a way to improve intracellular transport and/or exocytosis. Nevertheless, we require a better understanding of the mechanisms underlying intracellular transport and the integrity of nanomaterials during and after drug release.
Figure 5.
The challenges to, and strategies for, the enhanced intracellular delivery of nanomaterials. For definitions of abbreviations, please see the main text.
Acknowledgments
This article is a result of the project NORTE-01-0145-FEDER-000012, supported by Norte Portugal Regional Operational Programme (NORTE 2020), under the PORTUGAL 2020 Partnership Agreement, through the European Regional Development Fund (ERDF). This work was financed by FEDER – Fundo Europeu de Desenvolvimento Regional funds through the COMPETE 2020 – Operacional Programme for Competitiveness and Internationalisation (POCI), Portugal 2020, and by Portuguese funds through FCT – Fundação para a Ciência e a Tecnologia/Ministério da Ciência, Tecnologia e Ensino Superior in the framework of the project “Institute for Research and Innovation in Health Sciences” (POCI-01-0145-FEDER-007274). C.A. and M.H.C. would like to thank to the FCT for financial support (SFRH/BD/117598/2016 and SFRH/BD/ 131587/2017).
Biographies

Cláudia Azevedo is a PhD candidate at INEB7 (UP), where she is currently pursuing her doctorate in functionalized nanoparticles for drug delivery under the supervision of Bruno Sarmento. She was awarded her BSc in biosciences in 2011 from the Universidade Católica do Porto. She obtained her MSc in molecular oncology from the Instituto de Ciências Biomédicas Abel Salazar, UP in 2013; her MSc thesis focused on cancer metabolism. In June 2014, Cláudia was awarded an MSc Research Fellowship by the Instituto de Tecnologia Química Biológica (ITQB) for a project bioengineering exosomes to treat gliomas. At present she is pursuing her doctorate under the supervision of Professor Bruno Sarmento in functionalized nanoparticles for drug delivery.

Helena Macedo is a PhD candidate at INEB7 (UP). In December 2016, Helena was awarded an Integrated MSc in biomedical engineering with specialization in clinical engineering from the Universidade do Minho. During her MSc, she held a curricular internship in the 3Bs Research Institute, Portugal, where she carried out a project focusing on the development of alginate and chitosan membranes for cardiovascular applications. For part of her MSc thesis, she investigated the potential of L-arginine as a prebiotic to prevent or treat periodontitis.

Bruno Sarmento was awarded his PhD in pharmaceutical nanotechnology at the University of Porto (UP) in 2007, in collaboration with Queens University (Canada), University of Copenhagen (Denmark) and University of Santiago de Compostela (Spain). From 2007 to 2012, he held a post-doctoral position at UP, in collaboration with the University of Copenhagen, investigating insulin intestinal absorption mechanisms using in vitro models. In 2012, he became an associated researcher at INEB, where he established an independent research group. He was appointed as an assistant researcher at i3S (Institute for Investigation and Innovation in Health)/INEB in 2016. He is also currently a visiting professor at the Post-Graduate School, UniOeste (Brazil). So far, he has (co)supervised/co-supervised numerous students and has published extensively in international peer-reviewed (ISI) journals, books, and conference proceedings. He has also given more than 50 presentations at national and international meetings and has been awarded several distinctions. Dr Sarmento’s current research focuses on the development of functionalized nanomedicines and their application in the pharmaceutical and biomedical fields, with a particular interest in nanoformulations of biopharmaceutical drugs for use in diabetes, cancer, and infectious diseases. He has also specialized in mucosal tissue engineering models to validate functionalized nanomedicines and to perform in vitro–in vivo correlations. So far, he has supervised/co-supervised 8 Post-Docs, 28 PhD students (14 completed) and 19 MSc students (17 completed), and 6 researcher assistants. He published more than 200 papers in international peer reviewed (ISI) journals, most in top journals (Q1 in Pharmaceutical Sciences; Q1 in Nanoscience and Nanotechnology; total citations 4310; H-index 35), 34 book chapters and more than 180 proceedings. He edited 5 books, participated in more than 50 invited/selected talks in national and international meetings and was awarded several distinctions. His current research is focused on the development of functionalized nanomedicines and their application in the pharmaceutical and biomedical fields. In particular, nanoformulations of biopharmaceutical drugs with interest in diabetes, cancer and infectious diseases. He has also specialized in mucosal tissue engineering models to validate functionalized nanomedicines and to perform in vitro/in vivo correlation.
References
- 1.Banik B.L. Polymeric nanoparticles: the future of nanomedicine. WIREs Nanomed. Nanobiotechnol. 2016;8:271–299. doi: 10.1002/wnan.1364. [DOI] [PubMed] [Google Scholar]
- 2.Rekha M.R., Sharma C.P. Oral delivery of therapeutic protein/peptide for diabetes −Future perspectives. Int. J. Pharm. 2013;440:48–62. doi: 10.1016/j.ijpharm.2012.03.056. [DOI] [PubMed] [Google Scholar]
- 3.Pereira C. Targeting membrane transporters and receptors as a mean to optimize orally delivered biotechnological based drugs through nanoparticle delivery systems. Curr. Pharm. Biotechnol. 2014;15:650–658. doi: 10.2174/1389201015666140915152330. [DOI] [PubMed] [Google Scholar]
- 4.Araujo F. Antihyperglycemic potential of incretins orally delivered via nano and microsystems and subsequent glucoregulatory effects. Curr. Pharm. Biotechnol. 2014;15:609–619. doi: 10.2174/1389201015666140915150312. [DOI] [PubMed] [Google Scholar]
- 5.Moulari B. Nanoparticle targeting to inflamed tissues of gastrointestinal tract. Curr. Drug Deliv. 2013;10:9–17. doi: 10.2174/1567201811310010004. [DOI] [PubMed] [Google Scholar]
- 6.Sarmento B. Polymer-based delivery systems for oral delivery of peptides and proteins. In: Jorgensen L., Nielsen H.M., editors. Delivery Technologies for Biopharmaceutics: Peptides, Proteins, Nucleic Acids and Vaccines. John Wiley & Sons; 2009. pp. 207–227. [Google Scholar]
- 7.Parodi A. Enabling cytoplasmic delivery and organelle targeting by surface modification of nanocarriers. Nanomedicine. 2015;10:1923–1940. doi: 10.2217/nnm.15.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sakhrani N.M., Padh H. Organelle targeting: third level of drug targeting. Drug Des. Devel. Ther. 2013;7:585–599. doi: 10.2147/DDDT.S45614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jhaveri A., Torchilin V. Intracellular delivery of nanocarriers and targeting to subcellular organelles. Expert Opin. Drug Deliv. 2016;13:49–70. doi: 10.1517/17425247.2015.1086745. [DOI] [PubMed] [Google Scholar]
- 10.Ma X. Future of nanotherapeutics: targeting the cellular sub-organelles. Biomaterials. 2016;97:10–21. doi: 10.1016/j.biomaterials.2016.04.026. [DOI] [PubMed] [Google Scholar]
- 11.Blanco E. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat. Biotechnol. 2015;9:941–951. doi: 10.1038/nbt.3330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fan W. Intracellular transport of nanocarriers across the intestinal epithelium. Drug Discov. Today. 2016;21:856–863. doi: 10.1016/j.drudis.2016.04.007. [DOI] [PubMed] [Google Scholar]
- 13.Lopes M. Dual chitosan/albumin-coated alginate/dextran sulfate nanoparticles for enhanced oral delivery of insulin. J. Control. Release. 2016;232:29–41. doi: 10.1016/j.jconrel.2016.04.012. [DOI] [PubMed] [Google Scholar]
- 14.Mann S.K. Quantifying Nanoparticle Internalization Using a High Throughput Internalization Assay. Pharm. Res. 2016;33:2421–2432. doi: 10.1007/s11095-016-1984-3. [DOI] [PubMed] [Google Scholar]
- 15.Bennet D., Kim S. Polymer nanoparticles for smart drug delivery. In: Sezer A.D., editor. Application of Nanotechnology in Drug Delivery. Nanotechnology and Nanomaterials; 2014. pp. 257–309. [Google Scholar]
- 16.Duschl A. Nanomedicine. In: Boraschi D., Penton-Rol G., editors. Immune Rebalancing - Future of Immunosuppression. Elsevier; 2016. pp. 251–274. [Google Scholar]
- 17.Lopes M.A. Intestinal absorption of insulin nanoparticles: contribution of M cells. Nanomedicine. 2014;10:1139–1151. doi: 10.1016/j.nano.2014.02.014. [DOI] [PubMed] [Google Scholar]
- 18.Pawar V.K. Targeting of gastrointestinal tract for amended delivery of 3Q4 protein/peptide therapeutics: Strategies and industrial perspectives. J. Control. Release. 2014;196:168–183. doi: 10.1016/j.jconrel.2014.09.031. [DOI] [PubMed] [Google Scholar]
- 19.Stewart M.P. In vitro and ex vivo strategies for intracellular delivery. Nature. 2016;538:183–192. doi: 10.1038/nature19764. [DOI] [PubMed] [Google Scholar]
- 20.Sun W. Leveraging physiology for precision drug delivery. Physiol. Rev. 2017;97:189–225. [Google Scholar]
- 21.Agarwal S.K., Gupta A. Aquaporins: the renal water channels. Indian J. Nephrol. 2008;18:95–100. doi: 10.4103/0971-4065.43687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.DiMarco R.L. Improvement of paracellular transport in the Caco-2 drug screening model using protein-engineered substrates. Biomaterials. 2017;129:152–162. doi: 10.1016/j.biomaterials.2017.03.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Alai M.S. Application of polymeric nanoparticles and micelles in insulin oral delivery. J. Food Drug Anal. 2015;23:351–358. doi: 10.1016/j.jfda.2015.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Li M. Enhanced intranasal delivery of mRNA vaccine by overcoming the nasal epithelial barrier via intra- and paracellular pathways. J. Control. Release. 2016;228:9–19. doi: 10.1016/j.jconrel.2016.02.043. [DOI] [PubMed] [Google Scholar]
- 25.Bernkop-Schnürch A. Springer; 2009. Oral Delivery of Macromolecular Drugs: Barriers, Strategies and Future Trends. [Google Scholar]
- 26.Rahmat D. HEC-cysteamine conjugates: influence of degree of thiolation on efflux pump inhibitory and permeation enhancing properties. Int. J. Pharm. 2012;422:40–46. doi: 10.1016/j.ijpharm.2011.10.024. [DOI] [PubMed] [Google Scholar]
- 27.Clausen A.E. The Role of glutathione in the permeation enhancing effect of thiolated polymers. Pharm. Res. 2002;19:602–608. doi: 10.1023/a:1015345827091. [DOI] [PubMed] [Google Scholar]
- 28.Bernkop-Schnürch A. Thiomers: a new generation of mucoadhesive polymers. Adv. Drug Deliv. Rev. 2005;57:1569–1582. doi: 10.1016/j.addr.2005.07.002. [DOI] [PubMed] [Google Scholar]
- Chen Y.-M. The role of protein tyrosine phosphorylation in the cell-cell junctions and intercellular permeability of post-confluent bovine corneal epithelial cells. Taiwan J. Ophthalmol. 2013;3:37–41. [Google Scholar]
- 30.Palazzo C. Mucoadhesive properties of low molecular weight chitosan- or glycol chitosan- and corresponding thiomer-coated poly(isobutylcyanoacrylate) core-shell nanoparticles. Eur. J. Pharm. Biopharm. 2017;117:315–323. doi: 10.1016/j.ejpb.2017.04.020. [DOI] [PubMed] [Google Scholar]
- 31.Mahmood A. An in-vitro exploration of permeation enhancement by novel polysulfonate thiomers. Int. J. Pharm. 2015;496:304–313. doi: 10.1016/j.ijpharm.2015.10.013. [DOI] [PubMed] [Google Scholar]
- 32.Dartier J. ATP-dependent activity and mitochondrial localization of drug efflux pumps in doxorubicin-resistant breast cancer cells. Biochim. Biophys. Acta. 2017;1861:1075–1084. doi: 10.1016/j.bbagen.2017.02.019. [DOI] [PubMed] [Google Scholar]
- 33.Fonte P. Polymer-based nanoparticles for oral insulin delivery: revisited approaches. Biotechnol. Adv. 2015;33:1342–1354. doi: 10.1016/j.biotechadv.2015.02.010. [DOI] [PubMed] [Google Scholar]
- 34.Agarwal R., Roy K. Intracellular delivery of polymeric nanocarriers: a matter of size, shape, charge, elasticity and surface composition. Ther. Deliv. 2013;4:705–723. doi: 10.4155/tde.13.37. [DOI] [PubMed] [Google Scholar]
- 35.Woitiski C.B. Strategies toward the improved oral delivery of insulin nanoparticles via gastrointestinal uptake and translocation. Biodrugs. 2008;22:223–237. doi: 10.2165/00063030-200822040-00002. [DOI] [PubMed] [Google Scholar]
- 36.Mundra V., Mahato R.I. Design of nanocarriers for efficient cellular uptake and endosomal release of small molecule and nucleic acid drugs: learning from virus. Front. Chem. Sci. Eng. 2014;8:387–404. [Google Scholar]
- 37.Forte M. Polystyrene nanoparticles internalization in human gastric adenocarcinoma cells. Toxicol. In Vitro. 2016;31:126–136. doi: 10.1016/j.tiv.2015.11.006. [DOI] [PubMed] [Google Scholar]
- 38.Iversen T.-G. Endocytosis and intracellular transport of nanoparticles: Present knowledge and need for future studies. Nano Today. 2011;6:176–185. [Google Scholar]
- 39.Steinbach J.M. Cell penetrating peptide-modified poly(lactic-co-glycolic acid) nanoparticles with enhanced cell internalization. Acta Biomater. 2016;30:49–61. doi: 10.1016/j.actbio.2015.11.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Surassmo S. Surface modification of PLGA nanoparticles by carbopol to enhance mucoadhesion and cell internalization. Colloids Surf. B. 2015;130:229–236. doi: 10.1016/j.colsurfb.2015.04.015. [DOI] [PubMed] [Google Scholar]
- 41.Chanthick C. Caveolae-mediated albumin transcytosis is enhanced in dengue-infected human endothelial cells: A model of vascular leakage in dengue hemorrhagic fever. Sci. Rep. 2016;6:31855. doi: 10.1038/srep31855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Forte M. Polystyrene nanoparticles internalization in human gastric adenocarcinoma cells. Toxicol. In Vitro. 2016;31:126–136. doi: 10.1016/j.tiv.2015.11.006. [DOI] [PubMed] [Google Scholar]
- 43.Chan H.F. Can microfluidics address biomanufacturing challenges in drug/gene/cell therapies? Regen Biomater. 2016;3:87–98. doi: 10.1093/rb/rbw009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Szeto G.L. Microfluidic squeezing for intracellular antigen loading in polyclonal B-cells as cellular vaccines. Sci. Rep. 2015;5:10276. doi: 10.1038/srep10276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hasani-Sadrabadi M.M. Enhanced osteogenic differentiation of stem cells via microfluidics synthesized nanoparticles. Nanomedicine. 2015;11:1809–1819. doi: 10.1016/j.nano.2015.04.005. [DOI] [PubMed] [Google Scholar]
- 46.Dixon A.J. Enhanced intracellular delivery of a model drug using microbubbles produced by a microfluidic device. Ultrasound Med. Biol. 2013;39:1267–1276. doi: 10.1016/j.ultrasmedbio.2013.01.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Nel A.E. Understanding biophysicochemical interactions at the nano-bio interface. Nat. Mater. 2009;8:543–557. doi: 10.1038/nmat2442. [DOI] [PubMed] [Google Scholar]
- 48.Soo Choi H. Renal clearance of quantum dots. Nat. Biotechnol. 2007;25:1165–1170. doi: 10.1038/nbt1340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wahajuddin Arora S. Superparamagnetic iron oxide nanoparticles: magnetic nanoplatforms as drug carriers. Int. J. Nanomed. 2012;7:3445–3471. doi: 10.2147/IJN.S30320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Howe S.E. The uptake of soluble and particulate antigens by epithelial cells in the mouse small intestine. PLoS One. 2014;9:e86656. doi: 10.1371/journal.pone.0086656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Gupta V. A permeation enhancer for increasing transport of therapeutic macromolecules across the intestine. J. Control. Release. 2013;172:541–549. doi: 10.1016/j.jconrel.2013.05.002. [DOI] [PubMed] [Google Scholar]
- 52.Wang J. Mechanism of surface charge triggered intestinal epithelial tight junction opening upon chitosan nanoparticles for insulin oral delivery. Carbohydr. Polym. 2017;157:596–602. doi: 10.1016/j.carbpol.2016.10.021. [DOI] [PubMed] [Google Scholar]
- 53.Fu Q. The effect of phytic acid on tight junctions in the human intestinal Caco-2 cell line and its mechanism. Eur. J. Pharm. Sci. 2015;80:1–8. doi: 10.1016/j.ejps.2015.09.009. [DOI] [PubMed] [Google Scholar]
- 54.Karavasili C. Bioactive Self-assembling lipid-like peptides as permeation enhancers for oral drug delivery. J. Pharm. Sci. 2015;104:2304–2311. doi: 10.1002/jps.24484. [DOI] [PubMed] [Google Scholar]
- 55.Dünnhaupt S. Thiolated nanocarriers for oral delivery of hydrophilic macromolecular drugs. Carbohydr. Polym. 2015;117:577–584. doi: 10.1016/j.carbpol.2014.09.078. [DOI] [PubMed] [Google Scholar]
- 56.Yeh T.-H. Mechanism and consequence of chitosan-mediated reversible epithelial tight junction opening. Biomaterials. 2011;32:6164–6173. doi: 10.1016/j.biomaterials.2011.03.056. [DOI] [PubMed] [Google Scholar]
- 57.Chi Y. Connexin43 hemichannels contributes to the disassembly of cell junctions through modulation of intracellular oxidative status. Redox Biol. 2016;9:198–209. doi: 10.1016/j.redox.2016.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Lueben H.L. Mucoadhesive polymers in peroral peptide drug delivery. IV. Polycarbophil and chitosan are potent enhancers of peptide transport across intestinal mucosae in vitro. J. Control. Release. 1997;45:15–23. [Google Scholar]
- 59.Borchard G. The potential of mucoadhesive polymers in enhancing intestinal peptide drug absorption. III: effects of chitosan-glutamate and carbomer on epithelial tight junctions in vitro. J. Control. Release. 1996;39:131–138. [Google Scholar]
- 60.Zhang H. Improvement of pulmonary absorption of poorly absorbable macromolecules by hydroxypropyl-β-cyclodextrin grafted polyethylenimine (HP-β-CD-PEI) in rats. Int. J. Pharm. 2015;489:294–303. doi: 10.1016/j.ijpharm.2015.05.020. [DOI] [PubMed] [Google Scholar]
- 61.Martin L. The release of model macromolecules may be controlled by the hydrophobicity of palmitoyl glycol chitosan hydrogels. J. Control. Release. 2002;80:87–100. doi: 10.1016/s0168-3659(02)00005-6. [DOI] [PubMed] [Google Scholar]
- 62.Parmentier J. In vitro evaluation of liposomes containing bio-enhancers for the oral delivery of macromolecules. Eur. J. Pharm. Biopharm. 2010;76:394–403. doi: 10.1016/j.ejpb.2010.09.002. [DOI] [PubMed] [Google Scholar]
- 63.Huang H. A porphyrin-PEG polymer with rapid renal clearance. Biomaterials. 2016;76:25–32. doi: 10.1016/j.biomaterials.2015.10.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Roberts M.J. Chemistry for peptide and protein PEGylation. Adv. Drug Deliv. Rev. 2012;64(Suppl):116–127. doi: 10.1016/s0169-409x(02)00022-4. [DOI] [PubMed] [Google Scholar]
- 65.Schöttler S. Protein adsorption is required for stealth effect of poly(ethylene glycol)- and poly(phosphoester)-coated nanocarriers. Nat. Nano. 2016;11:372–377. doi: 10.1038/nnano.2015.330. [DOI] [PubMed] [Google Scholar]
- 66.Qie Y. Surface modification of nanoparticles enables selective evasion of phagocytic clearance by distinct macrophage phenotypes. Sci. Rep. 2016;6:26269. doi: 10.1038/srep26269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Evangelopoulos M. Cell source determines the immunological impact of biomimetic nanoparticles. Biomaterials. 2016;82:168–177. doi: 10.1016/j.biomaterials.2015.11.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Bera A., Jewrajka S.K. Tailoring polyamide thin film composite nanofiltration membranes by polyethyleneimine and its conjugates for the enhancement of selectivity and antifouling property. RSC Adv. 2016;6:4521–4530. [Google Scholar]
- 69.Noh S.M. Enhanced cellular delivery and transfection efficiency of plasmid DNA using positively charged biocompatible colloidal gold nanoparticles. Biochim. Biophys. Acta. 2007;1770:747–752. doi: 10.1016/j.bbagen.2007.01.012. [DOI] [PubMed] [Google Scholar]
- 70.Xiao K. The effect of surface charge on in vivo biodistribution of PEG-oligocholic acid based micellar nanoparticles. Biomaterials. 2011;32:3435–3446. doi: 10.1016/j.biomaterials.2011.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Yoo J.W., Mitragotri S. Polymer particles that switch shape in response to a stimulus. Proc. Natl. Acad. Sci. U. S. A. 2010;107:11205–11210. doi: 10.1073/pnas.1000346107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Banquy X. Effect of mechanical properties of hydrogel nanoparticles on macrophage cell uptake. Soft Matter. 2009;5:3984–3991. [Google Scholar]
- 73.O’Reill J.R. The effect of mixed micellar systems, bile salt/fatty acids, on the solubility and intestinal absorption of clofazimine (B663) in the anaesthetised rat. Int. J. Pharm. 1994;109:147–154. [Google Scholar]
- 74.Pabla D. Intestinal permeability enhancement of levothyroxine sodium by straight chain fatty acids studied in MDCK epithelial cell line. Eur. J. Pharm. Sci. 2010;40:466–472. doi: 10.1016/j.ejps.2010.05.002. [DOI] [PubMed] [Google Scholar]
- 75.Aspenström-Fagerlund B. Fatty acids increase paracellular absorption of aluminium across Caco-2 cell monolayers. Chemico-Biol. Interact. 2009;181:272–278. doi: 10.1016/j.cbi.2009.06.016. [DOI] [PubMed] [Google Scholar]
- 76.Trevino S.R. Amino acid contribution to protein solubility: Asp, Glu, and Ser contribute more favorably than the other hydrophilic amino acids in RNase Sa. J. Mol. Biol. 2007;366:449–460. doi: 10.1016/j.jmb.2006.10.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Mura S. Stimuli-responsive nanocarriers for drug delivery. Nat. Mater. 2013;12:991–1003. doi: 10.1038/nmat3776. [DOI] [PubMed] [Google Scholar]
- 78.Qiu L. Triple-stimuli (protease/redox/pH) sensitive porous silica nanocarriers for drug delivery. Sens. Actuators B. 2017;240:1066–1074. [Google Scholar]
- 79.Park S.-M. Towards clinically translatable in vivo nanodiagnostics. Nat. Rev. Mater. 2017;2:17014. doi: 10.1038/natrevmats.2017.14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Wang D. Advanced optoelectronic nanodevices and nanomaterials for sensing inside single living cell. Optics Commun. 2017;395:3–15. [Google Scholar]
- 81.Rajendran L. Subcellular targeting strategies for drug design and delivery. Nat. Rev. Drug Discov. 2010;9:29–42. doi: 10.1038/nrd2897. [DOI] [PubMed] [Google Scholar]
- 82.Müller R.H. State of the art of nanocrystals—special features, production, nanotoxicology aspects and intracellular delivery. Eur. J. Pharm. Biopharm. 2011;78:1–9. doi: 10.1016/j.ejpb.2011.01.007. [DOI] [PubMed] [Google Scholar]
- 83.Sunasee R. Cellulose nanocrystal cationic derivative induces NLRP3 inflammasome-dependent IL-1β secretion associated with mitochondrial ROS production. Biochem. Biophys. Rep. 2015;4:1–9. doi: 10.1016/j.bbrep.2015.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Ndong Ntoutoume G.M.A. PEI-cellulose nanocrystal hybrids as efficient siRNA delivery agents—Synthesis, physicochemical characterization and in vitro evaluation. Carbohydr. Polym. 2017;164:258–267. doi: 10.1016/j.carbpol.2017.02.004. [DOI] [PubMed] [Google Scholar]
- 85.Scheuble N. Tailored interfacial rheology for gastric stable adsorption layers. Biomacromolecules. 2014;15:3139–3145. doi: 10.1021/bm500767c. [DOI] [PubMed] [Google Scholar]
- 86.Boonrungsiman S. Shape and surface properties of titanate nanomaterials influence differential cellular uptake behavior and biological responses in THP-1 cells. Biochem. Biophys. Rep. 2017;9:203–210. doi: 10.1016/j.bbrep.2016.12.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Junyaprasert V.B., Morakul B. Nanocrystals for enhancement of oral bioavailability of poorly water-soluble drugs. Asian J. Pharm. Sci. 2015;10:13–23. [Google Scholar]
- 88.Li J. Cellular internalization of LiNbO3 nanocrystals for second harmonic imaging and the effects on stem cell differentiation. Nanoscale. 2016;8:7416–7422. doi: 10.1039/c6nr00785f. [DOI] [PubMed] [Google Scholar]
- 89.Murugan K. Cellular internalisation kinetics and cytotoxic properties of statistically designed and optimised neo-geometric copper nanocrystals. Mater. Sci. Eng. C. 2017;78:376–388. doi: 10.1016/j.msec.2017.04.087. [DOI] [PubMed] [Google Scholar]
- 90.Tang L., Cheng J. Nonporous silica nanoparticles for nanomedicine application. Nano Today. 2013;8:290–312. doi: 10.1016/j.nantod.2013.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Baeza A. Recent advances in porous nanoparticles for drug delivery in antitumoral applications: inorganic nanoparticles and nanoscale metal-organic frameworks. Expert Opin. Drug Deliv. 2016;14:783–796. doi: 10.1080/17425247.2016.1229298. [DOI] [PubMed] [Google Scholar]
- 92.Santos H.A. Porous silicon nanoparticles for nanomedicine: preparation and biomedical applications. Nanomedicine. 2014;9:535–554. doi: 10.2217/nnm.13.223. [DOI] [PubMed] [Google Scholar]
- 93.Yasmin R. Synergistic role of solid lipid and porous silica in improving the oral delivery of weakly basic poorly water soluble drugs. Eur. J. Pharm. Sci. 2017;96:508–514. doi: 10.1016/j.ejps.2016.10.026. [DOI] [PubMed] [Google Scholar]
- 94.Nabiev I. Nonfunctionalized nanocrystals can exploit a cell’s active transport machinery delivering them to specific nuclear and cytoplasmic compartments. Nano Lett. 2007;7:3452–3461. doi: 10.1021/nl0719832. [DOI] [PubMed] [Google Scholar]
- 95.Volkov Y. Quantum dots in nanomedicine: recent trends, advances and unresolved issues. Biochem. Biophys. Res. Commun. 2015;468:419–427. doi: 10.1016/j.bbrc.2015.07.039. [DOI] [PubMed] [Google Scholar]
- 96.Sobhan M.A. Non-specific internalization of laser ablated pure gold nanoparticles in pancreatic tumor cell. Colloids Surf. B. 2012;92:190–195. doi: 10.1016/j.colsurfb.2011.11.046. [DOI] [PubMed] [Google Scholar]
- 97.Li Y. Relationships between liposome properties, cell membrane binding, intracellular processing, and intracellular bioavailability. AAPS J. 2011;13:585–597. doi: 10.1208/s12248-011-9298-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Ran R. Enhanced gene delivery efficiency of cationic liposomes coated with PEGylated hyaluronic acid for anti P-glycoprotein siRNA: a potential candidate for overcoming multi-drug resistance. Int. J. Pharm. 2014;477:590–600. doi: 10.1016/j.ijpharm.2014.11.012. [DOI] [PubMed] [Google Scholar]
- 99.Alves R.F. Recombinant protein-based nanocarriers and their association with cationic liposomes: characterization and in vitro evaluation. Colloids and Surfaces A. 2017;513:1–10. [Google Scholar]
- 100.Kim B.K. Efficient delivery of plasmid DNA using cholesterol-based cationic lipids containing polyamines and ether linkages. Int. J. Mol. Sci. 2014;15:7293–7312. doi: 10.3390/ijms15057293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Wang Y., Chen L. Quantum dots, lighting up the research and development of nanomedicine. Nanomedicine. 2011;7:385–402. doi: 10.1016/j.nano.2010.12.006. [DOI] [PubMed] [Google Scholar]
- 102.Knudsen K.B. In vivo toxicity of cationic micelles and liposomes. Nanomedicine. 2015;11:467–477. doi: 10.1016/j.nano.2014.08.004. [DOI] [PubMed] [Google Scholar]
- 103.Desai P.P. Overcoming poor oral bioavailability using nanoparticle formulations −opportunities and limitations. Drug Discov. Today Technol. 2012;9:e87–e95. doi: 10.1016/j.ddtec.2011.12.001. [DOI] [PubMed] [Google Scholar]
- 104.Lee S.Y., Cho H.-J. Dopamine-conjugated poly(lactic-co-glycolic acid) nanoparticles for protein delivery to macrophages. J. Colloid Interface Sci. 2017;490:391–400. doi: 10.1016/j.jcis.2016.11.078. [DOI] [PubMed] [Google Scholar]
- 105.Vene E. Opsonisation of nanoparticles prepared from poly(β-hydroxybutyrate) and poly(trimethylene carbonate)-b-poly(malic acid) amphiphilic diblock copolymers: Impact on the in vitro cell uptake by primary human macrophages and HepaRG hepatoma cells. Int. J. Pharm. 2016;513:438–452. doi: 10.1016/j.ijpharm.2016.09.048. [DOI] [PubMed] [Google Scholar]
- 106.Chen X. PLGA-PEG-PLGA triblock copolymeric micelles as oral drug delivery system: In vitro drug release and in vivo pharmacokinetics assessment. J. Colloid Interface Sci. 2017;490:542–552. doi: 10.1016/j.jcis.2016.11.089. [DOI] [PubMed] [Google Scholar]
- 107.Lee S. Zein-alginate based oral drug delivery systems: protection and release of therapeutic proteins. Int. J. Pharm. 2016;515:300–306. doi: 10.1016/j.ijpharm.2016.10.023. [DOI] [PubMed] [Google Scholar]
- 108.Lin Y.-H. Novel nanoparticles for oral insulin delivery via the paracellular pathway. Nanotechnology. 2007;18:1–11. [Google Scholar]
- 109.Quinteros D.A. Novel Polymeric nanoparticles intended for ophthalmic administration of acetazolamide. J. Pharm. Sci. 2016;105:3183–3190. doi: 10.1016/j.xphs.2016.06.023. [DOI] [PubMed] [Google Scholar]
- 110.Lv Y. Novel multifunctional pH-sensitive nanoparticles loaded into microbubbles as drug delivery vehicles for enhanced tumor targeting. Sci. Rep. 2016;6:29321. doi: 10.1038/srep29321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Yousry C. Studying the influence of formulation and process variables on Vancomycin-loaded polymeric nanoparticles as potential carrier for enhanced ophthalmic delivery. Eur. J. Pharm. Sci. 2017;100:142–154. doi: 10.1016/j.ejps.2017.01.013. [DOI] [PubMed] [Google Scholar]
- 112.Erazo-Oliveras A. The late endosome and its lipid BMP act as gateways for efficient cytosolic access of the delivery agent dfTAT and its macromolecular cargos. Cell Chem. Biol. 2016;23:1–10. doi: 10.1016/j.chembiol.2016.03.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Lianga T.-J. Gemcitabine-based polymer-drug conjugate for enhanced anticancer effect in colon cancer. Int. J. Pharm. 2016;513:564–571. doi: 10.1016/j.ijpharm.2016.09.018. [DOI] [PubMed] [Google Scholar]
- 114.Römgens A.M. Diffusion profile of macromolecules within and between human skin layers for (trans)dermal drug delivery. J. Mechan. Behav. Biomed. Mater. 2015;50:215–222. doi: 10.1016/j.jmbbm.2015.06.019. [DOI] [PubMed] [Google Scholar]
- 115.Carregal-Romero S. NIR-light triggered delivery of macromolecules into the cytosol. J. Control. Release. 2012;159:120–127. doi: 10.1016/j.jconrel.2011.12.013. [DOI] [PubMed] [Google Scholar]
- 116.Matsumoto D. Mechanoporation of living cells for delivery of macromolecules using nanoneedle array. J. Biosci. Bioeng. 2016;122:748–752. doi: 10.1016/j.jbiosc.2016.05.006. [DOI] [PubMed] [Google Scholar]
- 117.Sheng J. Enhancing Insulin Oral Absorption by Using Mucoadhesive Nanoparticles Loaded with LMWP-linked Insulin Conjugates. J. Control. Release. 2016;233:181–190. doi: 10.1016/j.jconrel.2016.05.015. [DOI] [PubMed] [Google Scholar]
- 118.Lee S.Y., Cho H.-J. Amine-functionalized poly(lactic-co-glycolic acid) nanoparticles for improved cellular uptake and tumor penetration. Colloids Surf. B. 2016;148:85–94. doi: 10.1016/j.colsurfb.2016.08.050. [DOI] [PubMed] [Google Scholar]
- 119.Zhang Y. Functionalized magnetic nanochains with enhanced MR imaging: a novel nanosystem for targeting and inhibition of early glioma. Colloids Surf. B. 2016;140:437–445. doi: 10.1016/j.colsurfb.2016.01.006. [DOI] [PubMed] [Google Scholar]
- 120.Li N. Active targeting co-delivery system based on pH-sensitive methoxy-poly (ethylene glycol)2K-poly(e-caprolactone)4K-poly(glutamic acid)1 K for enhanced cancer therapy. J. Colloid Interface Sci. 2016;472:90–98. doi: 10.1016/j.jcis.2016.03.039. [DOI] [PubMed] [Google Scholar]
- 121.Li X. Orally active-targeted drug delivery systems for proteins and peptides. Expert Opin. Drug Deliv. 2014;11:1435–1447. doi: 10.1517/17425247.2014.924500. [DOI] [PubMed] [Google Scholar]
- 122.Wang D. The modulation of tumor vessel permeability by thalidomide and its impacts on different types of targeted drug delivery systems in a sarcoma mouse model. J. Control. Release. 2016;238:186–196. doi: 10.1016/j.jconrel.2016.07.014. [DOI] [PubMed] [Google Scholar]
- 123.Zhu Q. Large intestine-targeted, nanoparticle-releasing oral vaccine to control genitorectal viral infection. Nat. Med. 2012;18:1291–1297. doi: 10.1038/nm.2866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Saini P. The next generation noncompetitive active polyester nanosystems for transferrin receptor-mediated peroral transport utilizing gambogic acid as a ligand. Sci. Rep. 2016;6:29501. doi: 10.1038/srep29501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Gill K.K. EGFR targeted delivery of paclitaxel and parthenolide co-loaded in PEG-Phospholipid micelles enhance cytotoxicity and cellular uptake in non-small cell lung cancer cells. J. Drug Deliv. Sci. Technol. 2016;36:150–155. [Google Scholar]
- 126.Muntimadugua E. CD44 targeted chemotherapy for co-eradication of breast cancer stem cells and cancer cells using polymeric nanoparticles of salinomycin and paclitaxel. Colloids Surf. B. 2016;143:532–546. doi: 10.1016/j.colsurfb.2016.03.075. [DOI] [PubMed] [Google Scholar]
- 127.Sun S. pH-sensitive poly(lactide-co-glycolide) nanoparticle composite microcapsules for oral delivery of insulin. Int. J. Nanomed. 2015;10:3489–3498. doi: 10.2147/IJN.S81715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Pridgen E.M. Transepithelial transport of Fc-targeted nanoparticles by the neonatal Fc receptor for oral delivery. Sci. Transl. Med. 2013;5 doi: 10.1126/scitranslmed.3007049. 213ra167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Etzerodt A. Efficient intracellular drug-targeting of macrophages using stealth liposomes directed to the hemoglobin scavenger receptor CD163. J. Control. Release. 2012;160:72–80. doi: 10.1016/j.jconrel.2012.01.034. [DOI] [PubMed] [Google Scholar]
- 130.Lv L. Quercetin and doxorubicin co-encapsulated biotin receptor-targeting nanoparticles for minimizing drug resistance in breast cancer. Oncotarget. 2016;7:32184–32199. doi: 10.18632/oncotarget.8607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Godeshala S. Folate receptor-targeted aminoglycoside-derived polymers for transgene expression in cancer cells. Bioeng. Transl. Med. 2016;1:220–231. doi: 10.1002/btm2.10038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Chalasani K.B. Effective oral delivery of insulin in animal models using vitamin B12-coated dextran nanoparticles. J. Control. Release. 2007;122:141–150. doi: 10.1016/j.jconrel.2007.05.019. [DOI] [PubMed] [Google Scholar]
- 133.Kristensen M. Applications and challenges for use of cell-penetrating peptides as delivery vectors for peptide and protein cargos. Int. J. Mol. Sci. 2016;17:E185. doi: 10.3390/ijms17020185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Salerno J.C. Novel cell penetrating peptide-adaptors effect intracellular delivery and endosomal escape of protein cargos. J. Cell Sci. 2016;129:893–897. doi: 10.1242/jcs.182113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Pärnaste L. The formation of nanoparticles between small interfering RNA and amphipathic cell-penetrating peptides. Mol. Ther. Nucleic Acids. 2017;7:1–10. doi: 10.1016/j.omtn.2017.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Gavrovic-Jankulovic M., Prodanovic R. Drug delivery: plant lectins as bioadhesive drug delivery systems. J. Biomater. Nanobiotechnol. 2011;2:614–621. [Google Scholar]
- 137.Lehr C.-M. Lectin-mediated drug delivery: the second generation of bioadhesives. J. Control. Release. 2000;65:19–29. doi: 10.1016/s0168-3659(99)00228-x. [DOI] [PubMed] [Google Scholar]
- 138.Yi S.M. Lectin binding and endocytosis at the apical surface of human airway epithelia. Gene Ther. 2001;8:1826–1832. doi: 10.1038/sj.gt.3301598. [DOI] [PubMed] [Google Scholar]
- 139.Gabor F. Lectin-mediated drug delivery: binding and uptake of BSA-WGA conjugates using the Caco-2 model. Int. J. Pharm. 2002;237:227–239. doi: 10.1016/s0378-5173(02)00049-2. [DOI] [PubMed] [Google Scholar]
- 140.Wirth M. Lectin-mediated drug delivery: influence of mucin on cytoadhesion of plant lectins in vitro. J. Control. Release. 2002;79:183–191. doi: 10.1016/s0168-3659(01)00538-7. [DOI] [PubMed] [Google Scholar]
- 141.Bayyurt B. Encapsulation of two different TLR ligands into liposomes confer protective immunity and prevent tumor development. J. Control. Release. 2017;247:134–144. doi: 10.1016/j.jconrel.2017.01.004. [DOI] [PubMed] [Google Scholar]
- 142.Silva A.K.A. Polysaccharide nanosystems for future progress in cardiovascular pathologies. Theranostic. 2014;4:579–591. doi: 10.7150/thno.7688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Torchilin V. Intracellular delivery of protein and peptide therapeutics. Drug Discov. Today Technol. 2008;5:95–103. doi: 10.1016/j.ddtec.2009.01.002. [DOI] [PubMed] [Google Scholar]
- 144.Kim J.-S. Endosomal acidic pH-induced conformational changes of a cytosol-penetrating antibody mediate endosomal escape. J. Control. Release. 2016;235:165–175. doi: 10.1016/j.jconrel.2016.05.066. [DOI] [PubMed] [Google Scholar]
- 145.Sorkin A., Zastrow M.V. Endocytosis and signalling: intertwining molecular networks. Nat. Rev. Mol. Cell Biol. 2009;10:609–622. doi: 10.1038/nrm2748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Berguig G.Y. Intracellular delivery system for antibody-peptide drug conjugates. Mol. Ther. 2015;23:907–917. doi: 10.1038/mt.2015.22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Lönn P. Enhancing endosomal escape for intracellular delivery of macromolecular biologic therapeutics. Sci. Rep. 2016;6:32301. doi: 10.1038/srep32301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Goldshtein M. Mechanisms of cellular uptake and endosomal escape of calcium-siRNA nanocomplexes. Int. J. Pharm. 2016;515:46–56. doi: 10.1016/j.ijpharm.2016.10.009. [DOI] [PubMed] [Google Scholar]
- 149.Carney D.W. Structural optimization of a retrograde trafficking inhibitor that protects cells from infections by human polyoma-and papillomaviruses. Bioorg. Med. Chem. 2014;22:4836–4847. doi: 10.1016/j.bmc.2014.06.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Yu J. WLS Retrograde transport to the endoplasmic reticulum during Wnt secretion. Dev. Cell. 2014;29:277–291. doi: 10.1016/j.devcel.2014.03.016. [DOI] [PubMed] [Google Scholar]
- 151.Kou L. The endocytosis and intracellular fate of nanomedicines: Implication for rational design. Asian J. Pharm. Sci. 2013;8:1–10. [Google Scholar]
- 152.Jung J.-J. Regulation of intracellular membrane trafficking and cell dynamics by syntaxin-6. Biosci. Rep. 2012;32:383–391. doi: 10.1042/BSR20120006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Du Y. Syntaxin 6-mediated Golgi translocation plays an important role in nuclear functions of EGFR through microtubule-dependent trafficking. Oncogene. 2014;33:756–770. doi: 10.1038/onc.2013.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Maity A.R., Stepensky D. Delivery of drugs to intracellular organelles using drug delivery systems: analysis of research trends and targeting efficiencies. Int. J. Pharm. 2015;496:268–274. doi: 10.1016/j.ijpharm.2015.10.053. [DOI] [PubMed] [Google Scholar]
- 155.Sun Q. Inhibiting cancer cell hallmark features through nuclear export inhibition. Signal Transduct. Target. Ther. 2016;1:16010. doi: 10.1038/sigtrans.2016.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Tammam S.N. How successful is nuclear targeting by nanocarriers? J. Controlled Release. 2016;229:140–153. doi: 10.1016/j.jconrel.2016.03.022. [DOI] [PubMed] [Google Scholar]
- 157.Chen K. A gene delivery system containing nuclear localization signal: Increased nucleus import and transfection efficiency with the assistance of RanGAP1. Acta Biomater. 2017;48:215–226. doi: 10.1016/j.actbio.2016.11.004. [DOI] [PubMed] [Google Scholar]
- 158.Ma W. Co-assembly of doxorubicin and curcumin targeted micelles for synergistic delivery and improving anti-tumor efficacy. Eur. J. Pharm. Biopharm. 2017;112:209–223. doi: 10.1016/j.ejpb.2016.11.033. [DOI] [PubMed] [Google Scholar]
- 159.Mosafer J. In vitro and in vivo evaluation of anti-nucleolin-targeted magnetic PLGA nanoparticles loaded with doxorubicin as a theranostic agent for enhanced targeted cancer imaging and therapy. Eur. J. Pharm. Biopharm. 2017;113:60–74. doi: 10.1016/j.ejpb.2016.12.009. [DOI] [PubMed] [Google Scholar]
- 160.Izeddin I. Single-molecule tracking in live cells reveals distinct target-search strategies of transcription factors in the nucleus. Elife. 2014;3:e02230. doi: 10.7554/eLife.02230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Fan Y. pH-activated size reduction of large compound nanoparticles for in vivo nucleus-targeted drug delivery. Biomaterials. 2016;85:30–39. doi: 10.1016/j.biomaterials.2016.01.057. [DOI] [PubMed] [Google Scholar]
- 162.Marcus D. Heterologous mitochondrial targeting sequences can deliver functional proteins into mitochondria. Int. J. Biochem. Cell Biol. 2016;81:48–56. doi: 10.1016/j.biocel.2016.10.013. [DOI] [PubMed] [Google Scholar]
- 163.Chen S. Mitochondria-targeting ‘Nanoheater’ for enhanced photothermal/chemo-therapy. Biomaterials. 2017;17:92–104. doi: 10.1016/j.biomaterials.2016.11.056. [DOI] [PubMed] [Google Scholar]
- 164.Jin H. Folate-chitosan nanoparticles loaded with ursolic acid confer anti-breast cancer activities in vitro and in vivo. Sci. Rep. 2016;6:30782. doi: 10.1038/srep30782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Zhang B.-F. Mitochondria apoptosis pathway synergistically activated by hierarchical targeted nanoparticles co-delivering siRNA and lonidamine. Biomaterials. 2015;61:178–189. doi: 10.1016/j.biomaterials.2015.05.027. [DOI] [PubMed] [Google Scholar]
- 166.Murugan C. Combinatorial nanocarrier based drug delivery approach for amalgamation of anti-tumor agents in breast cancer cells: an improved nanomedicine strategies. Sci. Rep. 2016;6:34053. doi: 10.1038/srep34053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Xiao H. A new endoplasmic reticulum-targeted two-photon fluorescent probe for imaging of superoxide anion in diabetic mice. Biosens. Bioelectron. 2017;91:449–455. doi: 10.1016/j.bios.2016.12.068. [DOI] [PubMed] [Google Scholar]
- 168.Ouyang Z. A natural compound jaceosidin ameliorates endoplasmic reticulum stress and insulin resistance via upregulation of SERCA2b. Biomed. Pharmacother. 2017;89:1286–1296. doi: 10.1016/j.biopha.2017.03.023. [DOI] [PubMed] [Google Scholar]
- 169.Banfield D.K. Mechanisms of Protein Retention in the Golgi. Cold Spring Harbor Perspect. Biol. 2011;3:a005264. doi: 10.1101/cshperspect.a005264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Matteis M.A.D., Luini A. Exiting the Golgi complex. Nat. Rev. Mol. Cell Biol. 2008;9:273–284. doi: 10.1038/nrm2378. [DOI] [PubMed] [Google Scholar]
- 171.Matsuto M. Reconstitution of the targeting of Rab6A to the Golgi apparatus in semi-intact HeLa cells: A role of BICD2 in stabilizing Rab6A on Golgi membranes and a concerted role of Rab6A/BICD2 interactions in Golgi-to-ER retrograde transport. Biochim. Biophys. Acta. 2015;1853:2592–2609. doi: 10.1016/j.bbamcr.2015.05.005. [DOI] [PubMed] [Google Scholar]
- 172.Cohen J.R. Identification of a Golgi complex-targeting signal in the cytoplasmic tail of the severe acute respiratory syndrome coronavirus envelope protein. J. Virol. 2011;85:5794–5803. doi: 10.1128/JVI.00060-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Tanaka K. Cytoplasm-responsive nanocarriers conjugated with a functional cell-penetrating peptide for systemic siRNA delivery. Int. J. Pharm. 2013;455:40–47. doi: 10.1016/j.ijpharm.2013.07.069. [DOI] [PubMed] [Google Scholar]
- 174.Kim J.-H. Intracellular cytoplasm-specific delivery of SH3 and SH2 domains of SLAP inhibits TcR-mediated signaling. Biochem. Biophys. Res. Commun. 2015;460:603–608. doi: 10.1016/j.bbrc.2015.03.076. [DOI] [PubMed] [Google Scholar]
- 175.Zou X. Cytoplasmic delivery and selective, multicomponent labeling with oligoarginine-linked protein tags. Bioconjug. Chem. 2015;26:460–465. doi: 10.1021/bc500550z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Kozielski K.L. Bioreducible cationic polymer-based nanoparticles for efficient and environmentally triggered cytoplasmic siRNA delivery to primary human brain cancer cells. ACS Nano. 2014;8:3232–3241. doi: 10.1021/nn500704t. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Atif S.M. Fusogenic potential of sperm membrane lipids: Nature’s wisdom to accomplish targeted gene delivery. FEBS Lett. 2006;580:2183–2190. doi: 10.1016/j.febslet.2006.03.015. [DOI] [PubMed] [Google Scholar]
- 178.Kim S.K. Targeted delivery of EV peptide to tumor cell cytoplasm using lipid coated calcium carbonate nanoparticles. Cancer Lett. 2013;334:311–318. doi: 10.1016/j.canlet.2012.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Hasan M. Faint electric treatment-induced rapid and efficient delivery of extraneous hydrophilic molecules into the cytoplasm. J. Control. Release. 2016;228:20–25. doi: 10.1016/j.jconrel.2016.02.048. [DOI] [PubMed] [Google Scholar]
- 180.Gadok A.K. Connectosomes for direct molecular delivery to the cellular cytoplasm. J. Am. Chem. Soc. 2016;138:12833–12840. doi: 10.1021/jacs.6b05191. [DOI] [PubMed] [Google Scholar]
- 181.Negishi Y. Delivery of siRNA into the cytoplasm by liposomal bubbles and ultrasound. J. Control. Release. 2008;132:124–130. doi: 10.1016/j.jconrel.2008.08.019. [DOI] [PubMed] [Google Scholar]
- 182.Weissig V. Nanopharmaceuticals (part 1): products on the market. Int. J. Nanomed. 2014;9:4357–4373. doi: 10.2147/IJN.S46900. [DOI] [PMC free article] [PubMed] [Google Scholar]