Abstract
Background
Caralluma europaea (C. europaea) is a medicinal plant used in Moroccan popular medicine. Objective of the Study. The present work was aimed at identifying the chemical composition and the antioxidant and antiproliferative activities of hydroethanolic and bioactive compound classes of C. europaea) is a medicinal plant used in Moroccan popular medicine. Materials and Methods. The chemical composition was analyzed using HPLC. The antioxidant power was determined using both DPPH and FRAP assays. The antiproliferative activity was effectuated against cancerous cells using WST-1.
Results
The chemical analysis showed the presence of bioactive constituents such as quercetin, myricetin, and hesperetin. The polyphenol and flavonoid contents were estimated at 51.42 mg GA/g and 20.06 mg EQ/g, respectively. The EC50 values of FRAP assay of hydroethanolic, flavonoid, saponin, and mucilage extracts were 5.196 mg/ml, 4.537 mg/ml, 3.05 mg/ml, and 6.02 mg/ml, respectively. The obtained IC50 values with the DPPH test were 1.628 mg/ml, 1.05 mg/ml, 1.94 mg/ml, and 9.674 mg/ml, respectively. Regarding MDA-MB-231, saponins were highly effective even with the lowest concentration (15.62 μg/ml). The flavonoids decreased the cell viability with IC50 values of 43.62 ± 0.06 μg/ml). The flavonoids decreased the cell viability with IC50 values of 43.62 ± 0.06 μg/ml). The flavonoids decreased the cell viability with IC50 values of 43.62 ± 0.06
Conclusion
The present results suggest that C. europaea) is a medicinal plant used in Moroccan popular medicine.
1. Introduction
Traditional medicine is attracting more interest and its use has increased over the past three decades. According to the WHO, more than 80% of African people still rely on popular medicine for their benefit care including cancer treatment [1]. Nowadays, many pharmaceutical agents have been discovered in medicinal plants. However, the potential of plants as sources for the production of new drugs is largely untapped [2]. Medicinal plants play an important role in folk medicine, and they have been described in worldwide pharmacopoeia [3].
Caralluma europaea is a wild species frequently used in Moroccan traditional medicine for its presumed anticancer activity [4]. C. europaea belongs to the Apocynaceae family, and it is widely spread in Southern Jordan and the North African coast of the Mediterranean Sea [5]. In Morocco, C. europaea is used in traditional medicine for its anti-inflammatory, antipyretic, antinociceptive, antihyperglycemic, antidiabetic, antitrypanosomal, antiulcer, neuroprotective, antiobesogenic, antiatherosclerotic, and antiparasitic activities [6]. Several scientific studies have confirmed the antioxidant, antimicrobial, and anti-inflammatory activities of Caralluma europaea [7–9].
Nowadays, the world witnesses a great revolution in cancer treatment due to the large development of novel pathways of treatment. Although that resistance to classical agents continues to be a major problem in cancer therapies [10]. It was earlier reported that the medicinal plants play an alternative role in breast cancer treatment [11, 12].
A few chemical constituents were reported from C. europaea, including flavone glycosides such as luteolin 4′-neohesperidoside, luteolin-3′-O-(6′-O-sinapoylglucoside)-4′-O-neohesperidoside, and luteolin-3′-O-(6′-O-feruloylglucoside)-4′-O-neohesperidoside; monoterpenoids; α-terpinene; linalool; and terpinolene [5, 8].
The objective of the present study was to screen the phytochemical composition and the antioxidant and antiproliferative effects of both aerial parts hydroethanolic and natural bioactive classes of C. europaea using the methods described in the next section.
2. Materials and Methods
2.1. Plant Material
Caralluma europaea was collected in February 2016, from the Imouzzer region at the Middle Atlas Mountains, Morocco, using GPS coordinates (33°44′ North, 5°01′ West). The plant was authenticated by Professor Bari Amina, and a voucher specimen, 18I4C001, has been deposited in the herbarium of the Department of Biology, Faculty of Sciences Dhar El Mahraz, Fez University.
2.2. Preparation of Hydroethanolic Extract
The aerial parts of the plants were washed with water. After drying in the shade, the material was ground into a fine powder using a grinding apparatus. 10 g of the powder was extracted with 100 ml of hydroethanolic solution (7 : 3 v/v ethanol: distilled water) for 45 min. The mixture was filtered, then concentrated at low temperature, and finally preserved at −80°C until use.
2.3. Phytochemical Screening
C. europaea was screened for potential presence of flavonoids, alkaloids, quinones, catechic tannins, gallic tannins, mucilages, and saponins using the protocols described in [13–16].
2.4. Determination of Total Antioxidant Compounds
2.4.1. Total Polyphenol Content
The polyphenolic content (TPC) was determined using Folin-Ciocalteu reagent as previously described in the literature. The results were expressed as mg gallic acid equivalent (mg GAE)/g dry extract [17].
2.4.2. Total Flavonoid Content
The flavonoid content (TFC) was determined according to the method in earlier data. The results were expressed as mg quercetin equivalent (mg QE)/g dry extract [18].
2.5. HPLC Analysis
Polyphenolic extract of Caralluma europaea was analyzed using high-performance liquid chromatography (Agilent Technologies) equipped with a quaternary pump. The sample was detected by using a UV detector operating at 280 nm. A volume of 10 ml of the extract was injected over a C18 ZORBAX Eclipse Plus (serial number: USUXBO2265) (46 ∗ 150 nm; 5 μm) column, at a flow rate of 1 ml/min, and the column temperature was maintained at 25°C. The mobile phase was composed of acidified water 0.1% (A) and acetonitrile (B) with a total running time of 65 min.
2.6. Determination of Antioxidant Activity
2.6.1. Free Radical-Scavenging Ability (DPPH)
The test of DPPH was performed using the methods reported in [19], with slight modifications. A solution of the DPPH was prepared by dissolving 100 ml of methanol and 2.4 mg DPPH. 980 μl of DPPH solution (60 μM) was mixed with 20 μl of an extract with concentrations ranging from 10 to 50 μg ml−1. After 2 h of incubation, the absorbance was determined at 517 nm. BHT (10 to 50 μg ml−1) was used as a positive control. The percentage of inhibition of DPPH was calculated using the formula given as follows:
| (1) |
where IP is the inhibition percentage, A0 is the absorbance of the DPPH solution without extract, and A is the absorbance of the DPPH solution with the extract.
2.6.2. Ferric Reducing Antioxidant Power (FRAP)
The FRAP method determines the capacity of antioxidants to reduce Fe+3 to Fe+2. 1 ml of extract was dissolved with 2.5 ml of phosphate-buffered saline and 2.5 ml of potassium ferricyanide (1%). After incubation for 20 min at 50°C, 2.5 ml of trichloroacetic acid (10%) was added. The obtained solution was centrifuged for 10 min at 3000 rpm. Then, 2.5 ml of the supernatant was combined with 0.5 ml of FeCl3 (0.1%) and 2.5 ml of distilled water. The absorbance was determined spectrophotometrically at 700 nm. Ascorbic acid was considered as a reference [20].
2.7. Extraction of Some Bioactive Compound Classes from Caralluma europaea
2.7.1. Flavonoids
The powder of C. europaea (30 g) was macerated with 100 ml of methanol (MeOH) for 72 h at room temperature. The mixture was filtered and concentrated at 60°C. The obtained residue was dissolved in distilled water and then extracted again successively three times with 3 × 30 ml (chloroform, diethyl ether, n-hexane, ethyl acetate, and n-butanol) to get respective extracts. The flavonoids contained in the last phase were recovered and then subjected to evaporation at 55°C in order to obtain the dry residue [21].
2.7.2. Saponins
10 g of the powder of C. Europaea was initially defatted with petroleum ether for 2 h. Then, the extraction was carried out with 300 ml of ethanol for 24 h. The obtained extract was fractionated with petroleum ether and distilled water in equal proportions. 150 ml of the aqueous portion was added to n-butanol. The n-butanol fraction was separated, the remained solution was concentrated, and then the saponins were dried in an oven [22].
2.7.3. Mucilages
10 g of the powder of C. Europaea was macerated with distilled water for 5 or 6 h, boiled for 30 min, and kept aside for 1 h. The material was filtered through a muslin cloth. Acetone was added to the filtrate in order to precipitate the mucilage. The mucilage was separated and dried at a temperature under 50°C. The dried mucilage was powdered and stored for further use [23].
2.8. Evaluation of Antiproliferative Activity
2.8.1. Cell Culture
The antiproliferative activity of natural compounds from the aerial parts of C. europaea was tested against two human breast cancer cell lines, MDA-MB-231 and MCF-7, provided by Dr. L'Houcine Ouafik (Laboratoire de Transfert d'Oncologie, Marseille). The cells grew in DMEM medium supplemented with glutamine (1%), fetal calf serum (10%), and a mixture of streptomycin/penicillin (1%). Cells were preserved at 37°C.
2.8.2. In Vitro Antiproliferative Activity Assay
The antiproliferative activity was evaluated using WST-1 test (disodium mono{4-[3-(4-iodophenyl)-2-(4-nitrophenyl)-2H-tetrazol]-3-ium-5-yl]benzene-1,3-disulfonate}) [24]. The concentrations tested for their antiproliferative effects ranged from 1.562 μg/ml to 500 μg/ml. Cells of a subconfluent culture were harvested and centrifuged at 100 rpm for 5 min. The prepared cells were seeded in 96-well plates, at a cellular density of 8000 cells/well. Cells were incubated in a humidified atmosphere for 24 h at 37°C (5% CO2). After this, 100 μl of fresh medium containing serial concentrations was added to cells for further incubation of 48 h at 37°C.
At the end of the treatment period, the medium was removed, and 10 μl of WST-1 reagent was added to each well. The plates were incubated again (4 h at 37°C). Cell viability was performed by absorbance reading of each well at 450 nm using a Wallac Victor X3 multiplate reader. Untreated cells were considered as a negative control. Mitomycin was considered a positive control. The IC50 value responsible for 50% of cell growth inhibition was determined by plotting the inhibition percentage versus the concentrations (μg/ml).
2.9. Statistical Analysis
Analysis of the dose-response curves and the IC50 values was done using GraphPad Prism 5. Calculation of confidence limits and significance testing were made at the level of p=0.05 (p value <0.05 was considered significant).
3. Results
3.1. Qualitative Phytochemical Screening
The results of the phytochemical analysis of hydroethanolic extracts of C. europaea revealed the presence of flavonoids, catechic tannins, triterpene saponins, mucilages, coumarins, oses, and holosides and the absence of quinones and alkaloids (Table 1).
Table 1.
Phytochemical screening of C. europaea.
| Compounds | Results |
|---|---|
| Flavonoids | + |
| Catechic tannins | ++ |
| Triterpenes saponins | ++ |
| Mucilages | ++ |
| Coumarins | + |
| Oses and holosides | ++ |
| Quinones | − |
| Alkaloids | − |
++: abundantly present; +: present; −: undetected.
3.2. Total Polyphenol and Flavonoids Content
The phenolic content of aerial parts of C. europaea was determined from the calibration curve (y = 1.575x + 0.022; R2 = 0.959). The value was 51.42 ± 0.003 mg gallic acid equivalent/g of extract. Regarding the total flavonoid content (y = 0.814x + 0.021; R2 = 0.953), the value was 20.06 ± 0.007 mg QE/g of extract (Figure 1). The obtained results in this study showed a significant level of phenolic compounds contained in aerial parts of C. europaea.
Figure 1.
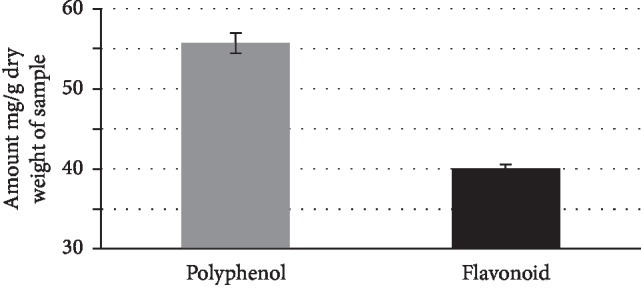
Total polyphenols and flavonoids contents of C. europaea hydroethanolic extract.
3.3. HPLC Analysis
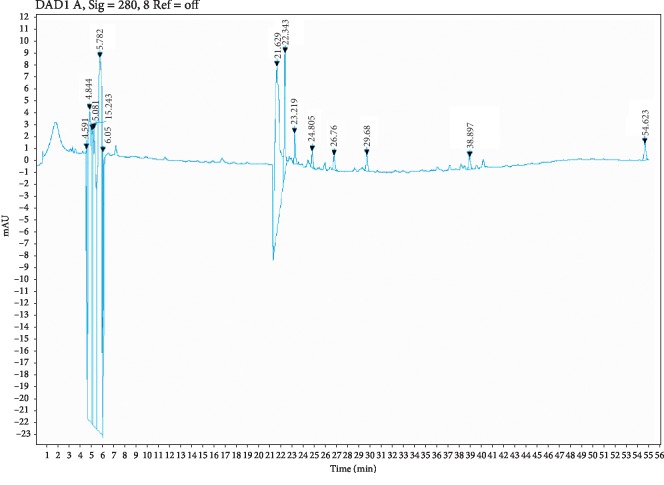
Five compounds were identified in polyphenols extract of Caralluma europaea using HPLC: hesperetin (TR = 24.805 min), quercetin (TR = 23.219 min), myricetin (TR = 22.34 min), ferulic acid (TR = 22.629 min), and gallic acid (TR = 6.051 min) (Figure 2).
Figure 2.
HPLC chromatogram of the main compounds identified in the polyphenolic extract of C. europaea.
According to HPLC analysis, the rate of polyphenols contained in Caralluma europaea extract ranged from 0.034 to 2.772 μg/ml. The ferulic acid concentration was determined at 2.772 μg/ml, followed by quercetin with 0.77 μg/ml. The concentrations of myricetin, gallic acid, and hesperetin were estimated at 0.350 μg/ml, 0.135 μg/ml, and 0.034 μg/ml, respectively (Table 2).
Table 2.
Amounts of the identified compounds in polyphenols extract of Caralluma europaea expressed in μg/ml.
| Phenolic compounds | Concentrations (μg/ml) |
|---|---|
| Hesperetin | 0.034 |
| Quercetin | 0.77 |
| Myricetin | 0.350 |
| Ferulic acid | 2.772 |
| Gallic acid | 0.135 |
3.4. Evaluation of Antioxidant Activity Using FRAP and DPPH Assay
The antioxidant activity of C. europaea extract was determined using two methods: DPPH and FRAP tests. The results vary according to the used methods.
3.4.1. Evaluation of Antioxidant Activity Using FRAP Assay
As indicated in Figure 3, all extracts showed lower antioxidant activities than the standard (ascorbic acid) (Figure 4). The EC50 values of hydroethanolic, flavonoids, saponins, and mucilages fractions were 5.196 mg/ml, 4.537 mg/ml, 3.05 mg/ml, and 6.02 mg/ml, respectively.
Figure 3.
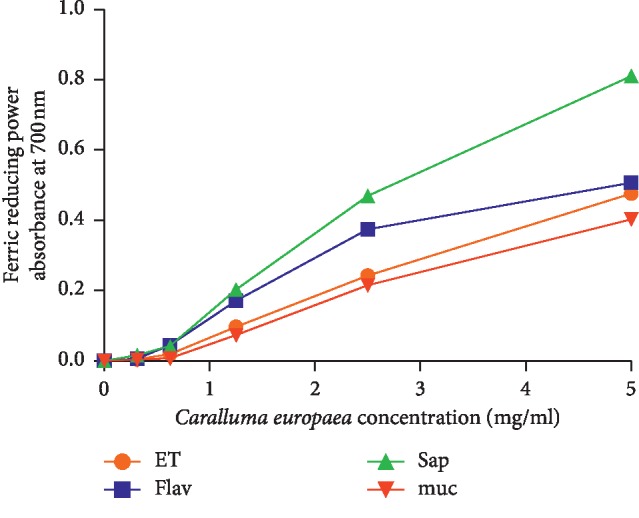
Antioxidant activity of hydroethanolic extract (ET), flavonoids (Flav), saponins (Sap), and mucilages (muc) of C. europaea using FRAP assay.
Figure 4.
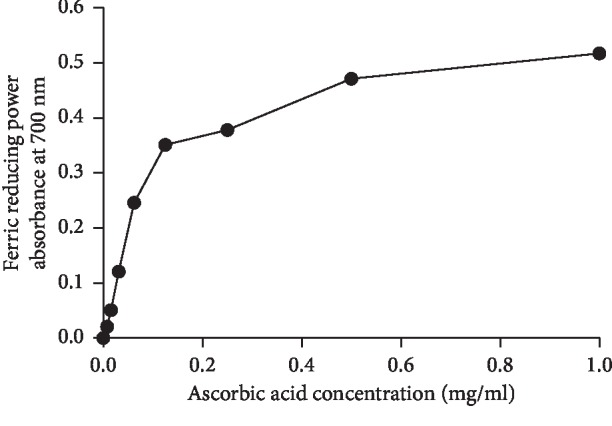
Antioxidant activity of ascorbic acid using FRAP assay.
3.4.2. Evaluation of Antioxidant Activity Using DPPH Assay
As shown in Figure 5, the percentage of free radical inhibition of hydroethanolic extracts such as flavonoids, saponins, and mucilages was lower than that of ascorbic acid (Figure 6). The antioxidant activity of the flavonoids fraction (IC50 = 1.51 mg/ml) was more pronounced compared to the other hydroethanolic extracts (IC50 = 1.628 mg/ml), saponins (IC50 = 1.94 mg/ml), and mucilages (IC50 = 9.674 mg/ml). The statistical analysis showed a significant difference between the IC50 values of all tested extracts and that of the ascorbic acid (IC50 = 0.23 mg/ml) (p value <0.05).
Figure 5.
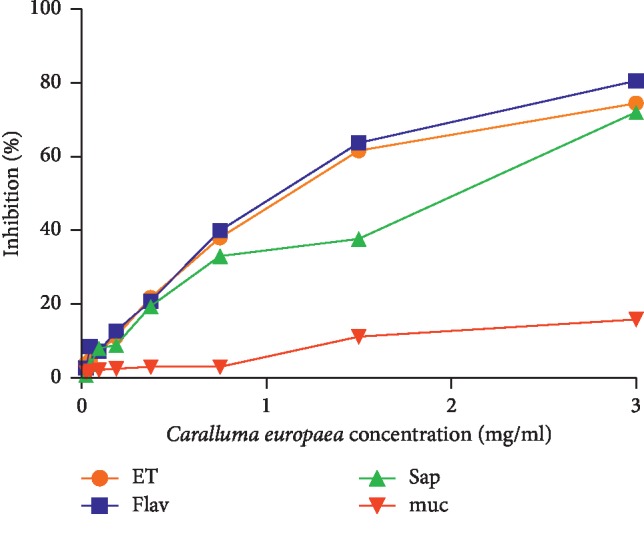
Inhibition percentage of free radicals of hydroethanolic extract (ET), flavonoids (Flav), saponins (Sap), and mucilages (muc) of C. europaea using DPPH assay.
Figure 6.
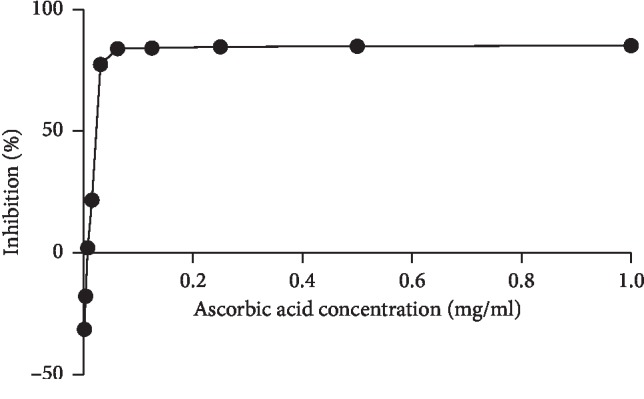
Inhibition percentage of free radicals of ascorbic acid using DPPH assay.
3.5. Extraction of Natural Antioxidants from C. europaea
Genus of Caralluma is characterized by the presence of bioactive components such as glycosides and steroids, known for their various biological activities [25–27]. In order to investigate the potential antiproliferative effect of C. europaea natural compounds, we proceeded with the extraction of saponins, flavonoids, and mucilage. The hydroethanolic extract was also tested for its antiproliferative effect. The extraction yields of various fractions were reported in Table 3.
Table 3.
Yield percentage of C. europaea bioactive classes.
| Compounds | Yields (%) |
|---|---|
| Flavonoids | 1.97 |
| Saponins | 2.44 |
| Mucilages | 41.3 |
3.6. Antiproliferative Activity
The antiproliferative effect of hydroethanolic extract of C. europaea and its fractions against MDA-MB-231 cell lines was evaluated using the WST-1 assay. Figure 7 shows the effect of hydroethanolic extract and its fractions at different concentrations (15.6 to 500 μg/ml) on MDA-MB-231 cell viability. Flavonoids fraction showed a remarkable antiproliferative effect with an IC50 value of 43.62 ± 0.06 μg/ml. The saponins fraction significantly inhibited cell proliferation with the studied range of concentrations even with the lowest value (15.62 μg/ml). However, hydroethanolic extract and mucilage fraction were ineffective with the current used concentrations.
Figure 7.
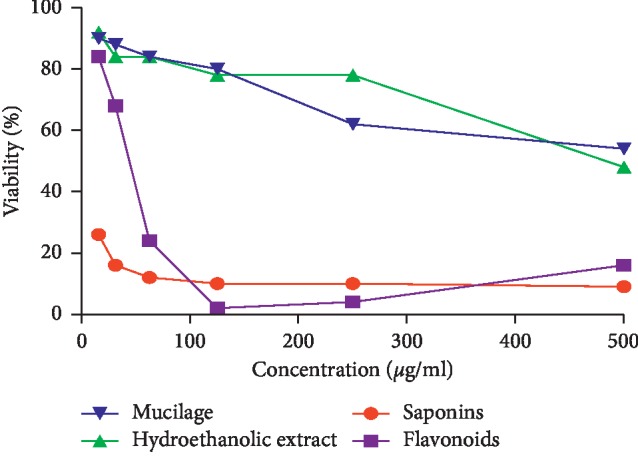
Antiproliferative effect of hydroethanolic extract of C. europaea and its fractions against MDA-MB-231 cell lines.
Saponins fraction of C. europaea extracts induced an important inhibited effect on both MDA-MB-231 (Figure 8) and MCF cell lines (Figure 9) even with the lowest concentration (1.562 μg/ml). Saponins clearly exhibited significant antiproliferative activity on both cell lines, with IC50 values of 5.097 μg/ml and 4.195 μg/ml on MCF7 and MDA-MB-231 cell lines, respectively. Flavonoids were ineffective with the tested range of concentrations. There was no significant difference between the IC50 values of saponin fractions on MCF7 no MDA-MB-231 (p > 0.05).
Figure 8.
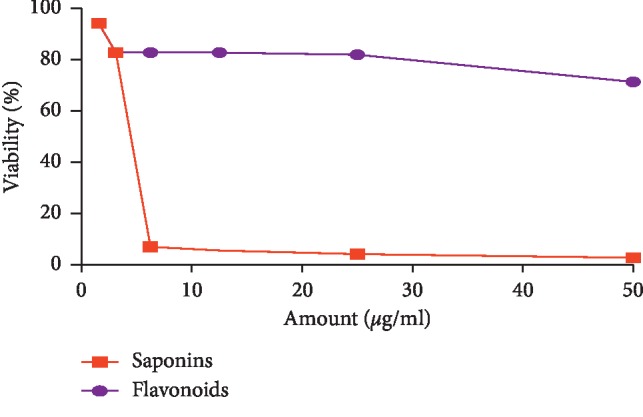
Antiproliferative effect of C. europaea saponin and flavonoid fractions against MDA-MB-231 cell lines.
Figure 9.
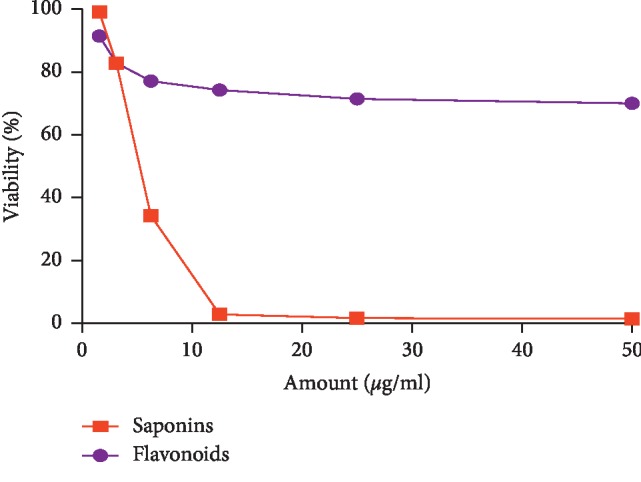
Antiproliferative effect of C. europaea saponin and flavonoid fractions against MCF7 cell lines.
4. Discussion
Cancer is one of the most lethal diseases worldwide, requiring effective prevention and treatment measures. Therefore, considerable research has been conducted to develop novel natural chemotherapeutic agents. In medicine, plant-derived compounds can be used as traditional, modern drugs, or chemical units for synthetic drugs. Antiproliferative activity models provide data to select extracts of plants with antitumoral potential for future studies [28].
Genus Caralluma arouses great interest in scientific fields due to its attractive immunostimulatory and pharmacological activities [29]. These activities could be due to its phytochemicals including glycoside and pregnane steroid [26, 27]. In Morocco, Caralluma europaea is widely used in folk medicine for its presumed anticancer activity [30]. However, no scientific data about its potential anticancer activity against cancerous cell lines were reported to the best of our knowledge.
The hydroethanolic extract of C. europaea was investigated for its phytochemical compounds, the total phenolic and flavonoid contents, and the antioxidant and antiproliferative activities. The qualitative phytochemical study showed the presence of different classes of bioactive secondary metabolites, such as flavonoids, triterpene saponins, oses, and holosides, previously described to have various medicinal activities [31]. Regarding the studied activities, our results were in accordance with those reported for C. sinaica [32].
It is worth noting that C. europaea is rich in phenolic compounds; the total phenolic content of C. europaea hydroethanolic extract was 51.42 ± 0.003 mg GAE/g dry of extract. These values were significantly higher than those of the methanolic and aqueous extract of C. adscendens, being 21.0 ± 0.59 and 18.8 ± 0.98 mg GAE/g dry extract, respectively [33].
Phenolic compounds are ubiquitous plant metabolites and the major group of compounds that contribute to the antioxidant properties; they can play an important role in the initiation of deleterious free radical actions [34]. Species of the genus Caralluma have been known for their richness in phenolic compounds [35, 36].
Our study also demonstrated that the hydroethanolic extract of C. europaea was rich in flavonoids compounds. The total flavonoids in the hydroethanolic extract of C. europaea were 20 ± 0.007 mg QE/g dry. This content is still relatively higher compared to methanolic and aqueous extracts of C. adscendens with values of 3.7 ± 1.16 and 3.1 ± 0.54 mg QE/g dry weight, respectively [33].
Flavonoids are the most important natural phenolic compounds and are thought to be responsible for the antioxidant and the antiproliferative activities of natural products [37, 38]. Many species of Caralluma have been known for the presence of interesting amounts of bioactive compounds [39, 40].
Flavonoids and polyphenols are natural antioxidants, known to be very active scavengers of many free radicals [41]. DPPH test is a method for estimating free radical-scavenging activity of antioxidants. The FRAP test determines the ability of these complexes to break the free radical chain by donating an atom of hydrogen [42, 43]. In the current study, the antioxidant activity of C. europaea hydroethanolic extract was determined using DPPH and FRAP assays. Using the DPPH method, all tested extracts of C. europaea showed a lower antioxidant power compared to ascorbic acid. The IC50 values of hydroethanolic, flavonoids, saponins, and mucilages extracts were 1.628 mg/ml, 1.51 mg/ml, 1.94 mg/ml, and 9.674 mg/ml, respectively, which were significantly lower than that of the ascorbic acid (0.23 mg/ml). Furthermore, the obtained IC50 values with FRAP test concerning hydroethanolic, flavonoids, saponins, and mucilages fractions were 5.196 mg/ml, 4.537 mg/ml, 3.05 mg/ml, and 6.02 mg/ml, respectively. These results can be attributed to the phenolic contents of the aerial parts of C. europaea.
Antiproliferative effect of hydroethanolic extract C. europaea and its fractions was tested against two human breast cancer cell lines, MDA-MB231 and MCF-7. The tested concentration on the MDA-MB-231 cell line ranged from 15.6 to 500 μg/ml. The flavonoids fraction reduced cell viability in a dose-dependent manner after 48 h of treatment, with an IC50 value of 43.62 ± 0.006 μg/ml. Antitumor activity of this extract can be attributed to the identified compounds with the HPLC method, as previously reported, such as hesperetin, quercetin, myricetin, and ferulic acid. With the same range of concentration, saponins fraction seems to be very effective against cell lines; however, mucilage fraction and hydroethanolic extract were ineffective. Antiproliferative effects of saponins and flavonoids fractions were tested again with lower concentrations ranging from 1.562 to 50 μg/ml on MCF7 and MDA-MB- 231 cell lines. Saponins clearly exhibited significant antiproliferative activity on both cell lines, with IC50 values of 5.097 μg/ml and 4.195 μg/ml against MCF7 and MDA-MB-231 cell lines, respectively.
Potential antiproliferative and anticancer activities of some species of Caralluma were reported in many earlier research works. The methanolic extract of Caralluma acutangula exhibited effects with concentrations varying between 0 and 50 μg/ml on MCF-7 (breast cancer) and HEPG2 (hepatocellular carcinoma) [44]. The methanolic extracts of Caralluma acutangula possess an interesting antiproliferative activity on HeLa (cervix cancer), MCF7 (breast cancer), and HEPG2 (hepatocellular carcinoma) cells [45].
Antiproliferative activity of plants may be attributed to the presence of alkaloids, glycosides, saponins, and polyacetylenes [46]. A previous phytochemical study on Caralluma species suggested the presence of glycosides and saponins [47]. Saponins are natural compounds that have several pharmacological activities like antiproliferative properties [48, 49], with pronounced effects on malignant tumor cells [9].
Numerous in vitro studies have reported the antiproliferative activity of saponins on breast cancer, cervical colon, and hepatic carcinomas. It was reported that several signaling pathways have been identified to be responsible for cell death [49, 50]. The antiproliferative effect of saponins resulted in the activation of different signaling pathways [49], whereas the induction of apoptosis is the most involved pathway [51]. Saponins have an important effect on some experimental tumors, such as estrogen receptor-negative mammary carcinogenesis [52].
In the present research work, flavonoids showed a significant antiproliferative effect with IC50 values of 43.62 ± 0.06 μg/ml. This class of molecules possesses a large range of pharmacological activities including antiproliferative activity. The flavonoids constitute the most important group of polyphenolic compounds, which have been investigated for their antiangiogenic, antiproliferative, and antiestrogenic effects [53]. Several works have reported that the flavonoids possess antiestrogenic activity and prevent the development of hormone-dependent cancers [54].
5. Conclusion
The findings of the present work revealed that the extract of aerial parts and the natural bioactive classes of C. europaea exhibit promoting antioxidant and antiproliferative activities. Therefore, C. europaea may constitute an important source of bioactive compounds which may lead to conceptualizing new effective drugs against cancer as well as diseases due to free radicals.
Acknowledgments
The authors extend their appreciation to the Deanship of Scientific Research at King Saud University for funding this work through research group no. RG-1440-009. The authors are grateful for the support of the project H2020-MSCA-RISE-Marie Sk∤odowska-Curie Actions (MSCA), Research and Innovation Staff Exchange (RISE); project acronym: VAHVISTUS; project number: 734759.
Contributor Information
Mohammed Bourhia, Email: bourhiamohammed@gmail.com.
Riaz Ullah, Email: rullah@ksu.edu.sa.
Data Availability
The data used to support the findings of this work are included within the article.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- 1.World Health Organization. Fifty-Sixth World Health Assembly A56/18. Geneva, Switzerland: World Health Organization; 2003. https://www.who.int/whr/2003/en/ [Google Scholar]
- 2.Ramawat K. G., Mérillon J.-M. Bioactive Molecules and Medicinal Plants. Berlin, Germany: Springer-Verlag; 2008. [Google Scholar]
- 3.Hmamouchi M. Les plantes médicinales et aromatiques marocaines : utilisation, biologie, écologie, chimie, pharmacologie, toxicologie, lexiques. Bibliographie du patrimoine culturel immatériel. 1999;1999 http://www.idpc.ma/view/documentation/bibliopci:35?titleinitial=h&num=3. [Google Scholar]
- 4.Zazouli S., Chigr M., Jouaiti A. Effect of polar and nonpolar solvent on total phenolic and antioxidant activity of roots extracts of Caralluma europaea. Der Pharma Chemica. 2016;8(11):191–196. [Google Scholar]
- 5.Zito P., Sajeva M., Bruno M., et al. Essential oil composition of stems and fruits of caralluma europaea N.E.Br. (Apocynaceae) Molecules. 2010;15(2):627–638. doi: 10.3390/molecules15020627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Adnan M., Jan S., Mussarat S., et al. A review on ethnobotany, phytochemistry and pharmacology of plant genus Caralluma R. Br. Journal of Pharmacy and Pharmacology. 2014;66(10):1351–1368. doi: 10.1111/jphp.12265. [DOI] [PubMed] [Google Scholar]
- 7.Prabakaran R., Kalimuthu K. Antibacterial activity of the whole plant of Caralluma Nilagiriana Kumari et Subba Rao—an endemic medicinal plant species. International Journal of Pharma and Bio Sciences. 2013;4:B42–B48. [Google Scholar]
- 8.Issiki Z., Issiki Z., Moundir C., et al. Toxicological evaluation of the aqueous extract of Caralluma europaea and its immunomodulatory and inflammatory activities. Pharmacognosy Research. 2017;9(4):390–395. doi: 10.4103/pr.pr_24_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fuchs H., Bachran D., Panjideh H., et al. Saponins as tool for improved targeted tumor therapies. Current Drug Targets. 2009;10(2):140–151. doi: 10.2174/138945009787354584. [DOI] [PubMed] [Google Scholar]
- 10.Li M., Tse L. A., Chan W.-C., et al. Evaluation of breast cancer risk associated with tea consumption by menopausal and estrogen receptor status among Chinese women in Hong Kong. Cancer Epidemiology. 2016;40:73–78. doi: 10.1016/j.canep.2015.11.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zafar A., Singh S., Naseem I. Cytotoxic activity of soy phytoestrogen coumestrol against human breast cancer MCF-7 cells: insights into the molecular mechanism. Food and Chemical Toxicology. 2017;99:149–161. doi: 10.1016/j.fct.2016.11.034. [DOI] [PubMed] [Google Scholar]
- 12.Adebayo I. A., Arsad H., Samian M. R. Antiproliferative effect on breast cancer (Mcf7) of Moringa oleifera seed extracts. African Journal of Traditional, Complementary and Alternative Medicines. 2017;14(2):282–287. doi: 10.21010/ajtcam.v14i2.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.N’Guessan K., Kadja B., Zirihi G. N., Traoré D., Aké-Assi L. Screening phytochimique de quelques plantes médicinales ivoiriennes utilisées en pays Krobou (Agboville, Côte-d’Ivoire) Sciences & Nature. 2009;6(1) doi: 10.4314/scinat.v6i1.48575. [DOI] [Google Scholar]
- 14.Diallo A. Etude de la Phytochimie et des Activites Biologiques de Syzygium guineense Willd. (Myrtaceae) Bamako, Mali: University of Bamako; 2005. [Google Scholar]
- 15.Karumi Y., Onyeyili P. A., Ogugbuaja V. O. Identification of active principles of Momordica balsamina (balsam apple) leaf extract. Journal of Medical Sciences Pakistan. 2004;4(3) [Google Scholar]
- 16.Dohou N., Yamni K., Tahrouch S., Idrissi Hassani L. M., Alain B., Najib G. Screening phytochimique d’une endémique ibéro-marocaine, Thymelaea lythroides. Bulletin de la Société de pharmacie de Bordeaux. 2003;142:61–78. [Google Scholar]
- 17.Singleton V. L., Orthofer R., Lamuela-Raventós R. M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Oxidants and Antioxidants Part A. 1999;299:152–178. doi: 10.1016/s0076-6879(99)99017-1. [DOI] [Google Scholar]
- 18.Arabshahi-Delouee S., Urooj A. Antioxidant properties of various solvent extracts of mulberry (Morus indica L.) leaves. Food Chemistry. 2007;102(4):1233–1240. doi: 10.1016/j.foodchem.2006.07.013. [DOI] [Google Scholar]
- 19.Brand-Williams W., Cuvelier M. E., Berset C. Use of a free radical method to evaluate antioxidant activity. LWT—Food Science and Technology. 1995;28(1):25–30. doi: 10.1016/s0023-6438(95)80008-5. [DOI] [Google Scholar]
- 20.Oyaizu M. Studies on products of browning reaction: antioxidative activities of products of browning reaction prepared from glucosamine. The Japanese Journal of Nutrition and Dietetics. 1986;44(6):307–315. doi: 10.5264/eiyogakuzashi.44.307. [DOI] [Google Scholar]
- 21.Feknous S., Saidi F., Said R. M. Extraction, caractérisation et identification de quelques métabolites secondaires actifs de la mélisse (Melissa officinalis L.) Nature & Technology. 2014;11:7–13. [Google Scholar]
- 22.Bouchelta A., Boughdad A., Blenzar A. Effets biocides des alcaloïdes, des saponines et des flavonoïdes extraits de Capsicum frutescens L. (Solanaceae) sur Bemisia tabaci (Gennadius) (Homoptera : aleyrodidae) Biotechnology, Agronomy, Society and Environment. 2005;9(4):259–269. [Google Scholar]
- 23.Ameena K., Dilip C., Krishnan P., Sankar C., Simi S. Isolation of the mucilages from Hibiscus rosasinensis linn. and Okra (Abelmoschus esculentus linn.) and studies of the binding effects of the mucilages. Asian Pacific Journal of Tropical Medicine. 2010;3(7):539–543. doi: 10.1016/s1995-7645(10)60130-7. [DOI] [Google Scholar]
- 24.Piastowska-Ciesielska A. W., Kozłowski M., Wagner W., Domińska K., Ochędalski T. Effect of an angiotensin II type 1 receptor blocker on caveolin-1 expression in prostate cancer cells. Archives of Medical Science. 2013;4(4):739–744. doi: 10.5114/aoms.2012.30955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Abdel-Sattar E., Harraz F. M., Al-ansari S. M. A., et al. Acylated pregnane glycosides from Caralluma tuberculata and their antiparasitic activity. Phytochemistry. 2008;69(11):2180–2186. doi: 10.1016/j.phytochem.2008.05.017. [DOI] [PubMed] [Google Scholar]
- 26.Malladi S., Nadh Ratnakaram V., Suresh Babu K., Pullaiah T. Phytochemical screening of caralluma lasiantha isolation of C21 pregnane steroid. Oriental Journal of Chemistry. 2017;33(2):963–967. doi: 10.13005/ojc/330248. [DOI] [Google Scholar]
- 27.Monte L. G., Santi-Gadelha T., Reis L. B., et al. Lectin of Abelmoschus esculentus (okra) promotes selective antitumor effects in human breast cancer cells. Biotechnology Letters. 2014;36(3):461–469. doi: 10.1007/s10529-013-1382-4. [DOI] [PubMed] [Google Scholar]
- 28.Cardellina J. H. Evolving strategies for the selection, dereplication and prioritization of antitumor and HIV-inhibitory natural products extracts. In: Bohlin L., Bruhn J. G., editors. Bioassay Methods in Natural Product Research And Drug Development. Dordrecht, Netherlands: Springer; 1999. pp. 25–35. [Google Scholar]
- 29.Sireesha M., Venkata Nadh R., Babu K., Sreenivasulu M. Phytochemical library of caralluma genus. International Journal of Research in Pharmaceutical Sciences. 2018;9(4):1201–1213. [Google Scholar]
- 30.Bellakhdar J., Claisse R., Fleurentin J., Younos C. Repertory of standard herbal drugs in the Moroccan pharmacopoea. Journal of Ethnopharmacology. 1991;35(2):123–143. doi: 10.1016/0378-8741(91)90064-k. [DOI] [PubMed] [Google Scholar]
- 31.Halliwell B. Antioxidants in human health and disease. Annual Review of Nutrition. 1996;16(1):33–50. doi: 10.1146/annurev.nu.16.070196.000341. [DOI] [PubMed] [Google Scholar]
- 32.Habibuddin M., Daghriri H. A., Humaira T., Qahtani M. S. A., Hefzi A. A. H. Antidiabetic effect of alcoholic extract of Caralluma sinaica L. on streptozotocin-induced diabetic rabbits. Journal of Ethnopharmacology. 2008;117(2):215–220. doi: 10.1016/j.jep.2008.01.021. [DOI] [PubMed] [Google Scholar]
- 33.Maheshu V., Priyadarsini D. T., Sasikumar J. M. Antioxidant capacity and amino acid analysis of Caralluma adscendens (Roxb.) Haw var. fimbriata (wall.) Grav. & Mayur. aerial parts. Journal of Food Science and Technology. Oct. 2014;51(10):2415–2424. doi: 10.1007/s13197-012-0761-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wang H., Gan D., Zhang X., Pan Y. Antioxidant capacity of the extracts from pulp of Osmanthus fragrans and its components. LWT—Food Science and Technology. 2010;43(2):319–325. doi: 10.1016/j.lwt.2009.08.003. [DOI] [Google Scholar]
- 35.Dhayalan P., Krishnasamy R., Periyasamy S. In vitro antioxidant and preliminary phytochemical studies of Caralluma fimbriata wall. International Journal of Pharmaceutical Research. 2011;4(2):44–48. [Google Scholar]
- 36.Devi S. G., Dhamotharan R. Preliminary studies on phytochemical screening and in vitro antioxidant activities of Caralluma fimbriata. World Journal of Pharmaceutical Research. 2016;5(4):1097–1107. [Google Scholar]
- 37.Pyo Y.-H., Lee T.-C., Logendra L., Rosen R. T. Antioxidant activity and phenolic compounds of Swiss chard (Beta vulgaris subspecies cycla) extracts. Food Chemistry. 2004;85(1):19–26. doi: 10.1016/s0308-8146(03)00294-2. [DOI] [Google Scholar]
- 38.Crozier A., Clifford M., Ashihara H. Plant Secondary Metabolites: Occurrence, Structure and Role in the Human Diet. Oxford, UK: Blackwell Publishing Ltd.; 2006. [Google Scholar]
- 39.Kamil M., Jayaraj A. F., Ahmad F., et al. Separation of flavonoids from Caralluma arabica using high-speed counter-current chromatography. Journal of Pharmacy and Pharmacology. 2000;52:p. 265. [Google Scholar]
- 40.Tambe D. A., Chaudhari T. B., Chaudhari S. R. Phyto-pharmacology of Caralluma adscendens roxb: a review. Pharmacognosy Journal. 2010;2(14):33–38. doi: 10.1016/s0975-3575(10)80069-0. [DOI] [Google Scholar]
- 41.Bravo L. Polyphenols: chemistry, dietary sources, metabolism, and nutritional significance. Nutrition Reviews. 1998;56(11):317–333. doi: 10.1111/j.1753-4887.1998.tb01670.x. [DOI] [PubMed] [Google Scholar]
- 42.Kubola J., Siriamornpun S. Phenolic contents and antioxidant activities of bitter gourd (Momordica charantia L.) leaf, stem and fruit fraction extracts in vitro. Food Chemistry. 2008;110(4):881–890. doi: 10.1016/j.foodchem.2008.02.076. [DOI] [PubMed] [Google Scholar]
- 43.Ouedrhiri W., Bouhdida S., Balouiria M., et al. Chemical composition of Citrus aurantium L. Leaves and zest essential oils, their antioxidant, antibacterial single and combined effects. Journal of Chemical and Pharmaceutical Research. 2015;7(1):78–84. [Google Scholar]
- 44.Al-Faifi Z. I. A., Masrahi Y. S., Aly M. S., Al-Turki T. A. In vitro anticancer activity of caralluma acutangula (decne.) N.E.Br. extract. International Journal of Pharmaceutical Sciences Review and Research. 2016;38(12):59–63. [Google Scholar]
- 45.Almehdar H., Abdallah H. M., Osman A.-M. M., Abdel-sattar E. A. In vitro cytotoxic screening of selected Saudi medicinal plants. Journal of Natural Medicines. 2011;66(2):406–412. doi: 10.1007/s11418-011-0589-8. [DOI] [PubMed] [Google Scholar]
- 46.Orech F., Akenga T., Ochora J., Friis H., Aagaard-Hansen J. Potential toxicity of some traditional leafy vegetables consumed in Nyang’oma division, Western Kenya. African Journal of Food, Agriculture, Nutrition and Development. 2005;5(1):p. 9. [Google Scholar]
- 47.Ramesh M., Nageshwar Rao Y., Kumar M. R., Venkata Narasimha Appa Rao A., Prabhakar M. C., Reddy B. M. Antinociceptive and anti-inflammatory activity of carumbelloside-I isolated from Caralluma umbellata. Journal of Ethnopharmacology. 1999;68(1–3):349–352. doi: 10.1016/s0378-8741(99)00122-1. [DOI] [PubMed] [Google Scholar]
- 48.Kim M.-O., Moon D.-O., Choi Y. H., et al. Platycodin D induces apoptosis and decreases telomerase activity in human leukemia cells. Cancer Letters. 2008;261(1):98–107. doi: 10.1016/j.canlet.2007.11.010. [DOI] [PubMed] [Google Scholar]
- 49.Podolak I., Galanty A., Sobolewska D. Saponins as cytotoxic agents: a review. Phytochemistry Reviews. 2010;9(3):425–474. doi: 10.1007/s11101-010-9183-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Weng A., Melzig M. F., Bachran C., Fuchs H. Enhancement of saporin toxicity against U937 cells by gypsophila saponins. Journal of Immunotoxicology. 2008;5(3):287–292. doi: 10.1080/15376510802312266. [DOI] [PubMed] [Google Scholar]
- 51.Wang S.-R., Fang W.-S. Pentacyclic triterpenoids and their saponins with apoptosis-inducing activity. Current Topics in Medicinal Chemistry. 2009;9(16):1581–1596. doi: 10.2174/156802609789909821. [DOI] [PubMed] [Google Scholar]
- 52.Liby K., Risingsong R., Royce D. B., et al. Prevention and treatment of experimental estrogen receptor-negative mammary carcinogenesis by the synthetic triterpenoid CDDO-methyl ester and the rexinoid LG100268. Clinical Cancer Research. 2008;14(14):4556–4563. doi: 10.1158/1078-0432.ccr-08-0040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zhang S., Yang X., Morris M. E. Flavonoids are inhibitors of breast cancer resistance protein (ABCG2)-Mediated transport. Molecular Pharmacology. 2004;65(5):1208–1216. doi: 10.1124/mol.65.5.1208. [DOI] [PubMed] [Google Scholar]
- 54.Collins B. M., McLachlan J. A., Arnold S. F. The estrogenic and antiestrogenic activities of phytochemicals with the human estrogen receptor expressed in yeast. Steroids. 1997;62(4):365–372. doi: 10.1016/s0039-128x(96)00246-2. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data used to support the findings of this work are included within the article.