Natural killer (NK) cells can be isolated from peripheral blood and expanded using cytokines in vitro in autologous and allogeneic settings as adoptive immunotherapy for hematologic malignancies and solid cancers.1–5 We have reported an effective in vitro NK-cell expansion method using cytokines without feeder cells.6–8 In addition, NK cells kill target cells by antibody-dependent cellular cytotoxicity (ADCC) in which an antibody such as an anti-CD20 monoclonal antibody (rituximab) crosslinks to the Fc receptor (CD16) on NK cells.9,10 Therefore, we have conducted a phase I study of therapy using ex vivo expanded auto-NK cells from patients’ peripheral blood mononuclear cells (PBMC) combined with rituximab-containing chemotherapy for relapsed CD20-positive malignant lymphoma patients.
Patients with relapsed or refractory CD20-positive malignant lymphoma in whom prior standard chemotherapy had failed were enrolled in this study. Eligibility criteria included age between 20 and 80 years, Eastern Cooperative Oncology Group (ECOG) performance status of not more than 3, expected life span of more than three months, and absence of severe active infection or severe organ dysfunction. This study was an open label, non-randomized phase I clinical trial with dose escalation of NK cells. The primary end point was evaluation of the safety of infusion of adoptive NK cells by our expansion method in vitro. The secondary end point was clinical response. This study was approved by the ethics committee of Tokyo Women’s Medical University and was registered in the University Hospital Medical Information Network (UMIN) Clinical Trail Registry as ID: UMIN000014072. This study was designed and conducted in accordance with the Declaration of Helsinki and the Ethical Guidelines for Clinical Research (Ministry of Health, Labor and Welfare, Japan), and was approved by the Certified Committee for Regenerative Medicine.
Peripheral blood mononuclear cells were obtained from the peripheral blood (20-50 mL) of each lymphoma patient. The PBMC (1×106/mL) with IL-15 (10 ng/mL) (PeproTech Inc., Rocky Hill, NJ, USA), IL-2 (5 ng/mL) (R & D Systems, Minneapolis, MN, USA), and anti-CD3 mAb (OKT3, 10-1,000 ng/mL, Janssen Pharmaceutical Company, Tokyo, Japan) with tacrolimus (0.1 ng/mL, Fujisawa, Osaka, Japan) and dalteparin sodium (Fragmin, 5-10 U/mL, Pfizer Japan, Tokyo, Japan) in culture medium SCGM (CeeGenix, Freiburg, Germany) that was produced under good manufacturing practice (GMP) were cultured with 2.5% auto-serum in a culture bag without feeder cells for three weeks.7,8 Cell cultures were split into approximately half and a quarter after 3-4 days of culture, and fresh medium, cytokines and reagents were added. The first three patients, the second three patients, and the third three patients were infused with 1×106/kg, 3×106/kg and 10×106/kg of patients’ body weight of expanded auto-CD56+CD3–NK cells, respectively. When two patients have developed a non-hematologic grade 4 adverse event within four weeks after infusion in the cohort, this study should be stopped and the tolerable cell dose for the previous cohort will be decided as a maximum cell dose. Quality control tests, including tests for sterility, mycoplasma contamination and endotoxin, were performed.
Clinical response was assessed with the Response Evaluation Criteria in Solid Tumor (RECIST version 1.1). Adverse events were assessed according to the Common Terminology Criteria for Adverse Events (CTCAE version 4.0). The fluorescence intensity of the cells was analyzed using a Navios Flow Cytometer (Beckman Coulter). The cytolytic activities of expanded NK cells were tested against 51Cr-labeled K562 (an erythroleukemic cell line) cells. Statistical analysis was performed using Student t-test.
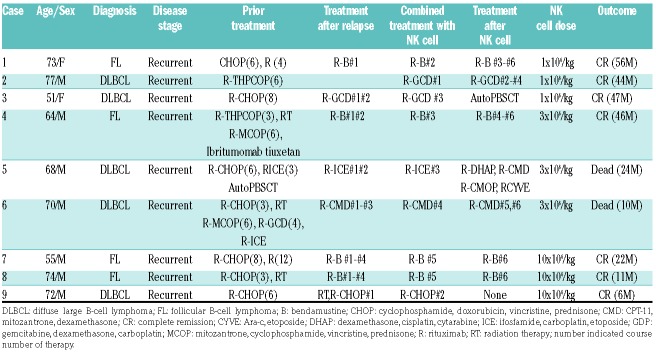
Patients’ characteristics are shown in Table 1. Nine patients were enrolled in this study between August 2014 and October 2018. Median age was 70 years (range, 51-77 years). Four patients had follicular B-cell lymphoma and five patients had diffuse large B-cell lymphoma. All of the patients had relapsed after multiple cycles of R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, prednisone)-like regimens; all patients received rituximab-combined salvage chemotherapy. Expanded auto-NK cells were infused intravenously for more than 60 minutes one day after rit-uximab administration.
Table 1.
Patients’ characteristics.
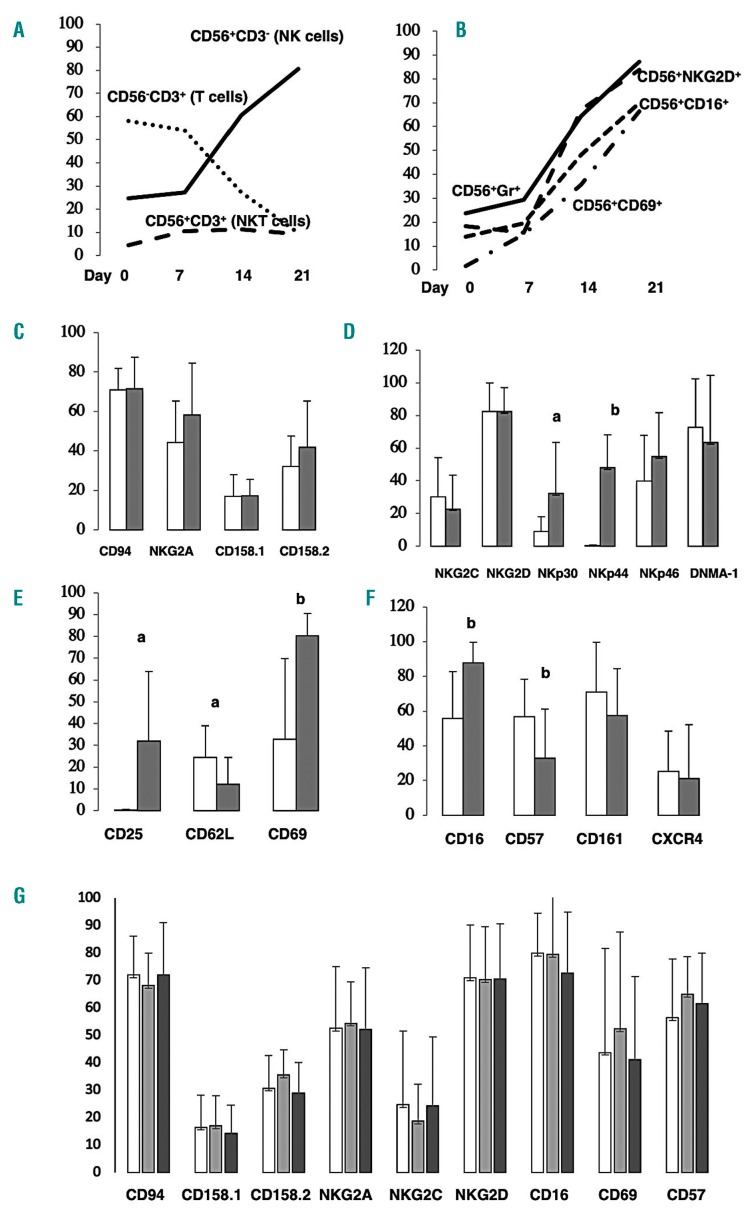
The proportion of CD56+CD3– NK cells increased up to 80.5% after three weeks of culture using a representative patient’s PBMC (Case 7, Figure 1A). These expanded NK cells expressed stimulatory NK-cell receptors NKG2D, CD16, CD69 and intracellular cytotoxic molecule granzyme (Figure 1B). Finally, we could obtain 551.4±420.0×106, 274.7±143.6 fold (50.7±19.1%) CD56+CD3– NK cells from 1×107 unmanipulated PBMC under GMP conditioned medium with 2.5% patient’s auto-serum without feeder cells [mean±standard deviation (SD), n=9] (Table 2). These products contained 26.2±11.6% of CD56+CD3+ NKT cells. The expanded NK cells had a very high level of cytolytic activity against the K562 leukemic cell line with specific lysis of more than 50% (58.1±13.1%, mean ±SD, n=9) under the condition of an effector:target ratio (E:T ratio) of 10:1 (Table 2). Expanded CD56+CD3– NK cells expressed the same levels of inhibitory NK receptors (Figure 1C) but expressed significantly higher levels of stimulatory NK-cell receptors, including NKp30, NKp46 (Figure 1D) and activation markers CD25 and CD69 (Figure 1E), and CD16 (Figure 1F), than the levels of these receptors on CD56+CD3– NK cells in PBMC before culture were noted.
Figure 1.
Time course profiles of expanded cells during culture without feeder cells, expression of various receptors on expanded CD56+CD3–natural killer (NK) cells and on CD56+CD3– NK cells in patients’ peripheral blood mononuclear cells (PBMC) before and after NK-cell infusion. Time course profiles of CD56+CD3– cells (NK cells), CD56–CD3+ (T cells) and CD56+CD3+ cells (NKT cells) (A) and CD56+NKG2D+, CD56+CD16+, CD56+CD69+ cells and CD56+granzyme+ cells (B) during expansion using a representative patient’s PBMC (Case 7). Proportions of NK receptors on CD56+CD3– NK cells before expansion (white bars) and after expansion (gray bars). Inhibitory NK receptors (C), stimulatory NK cell receptors (D), activation markers (E), and others (F). Proportions of NK receptors on CD56+CD3– NK cells in patients’ PBMC before NK-cell infusion (white bars), seven days after infusion (gray bars), and 14 days after infusion (black bars)(G). There were significant differences between before and after expansion (%, a=P<0.05; b=P<0.01. Bars indicate means ± standard deviation, n=9).
Table 2.
Expansion of natural killer cells from peripheral blood mononuclear cells (PBMC) of lymphoma patient.
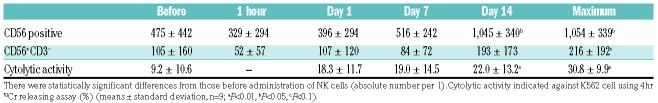
The absolute number of CD56-positive cells in patients was significantly increased two weeks after infusion compared with that before adoptive transfer of auto-expanded NK cells (1,045±340, mean±SD, n=9; P<0.01) (Table 3). The absolute maximum number of CD56+CD3– NK cells during a period of two weeks tended to increase compared with that before NK-cell infusion (216±192, mean±SD, n=9; P<0.1) (Table 3). PBMC from lymphoma patients two weeks after infusion had increased cytolytic activity compared with that before NK-cell infusion (19.0±14.5 on day 7, 22.0±13.2 on day 14, max. 30.8±9.9 during the 2-week period; mean±SD, n=9; P<0.01) (Table 3). We tried to show proportions of NK receptors on CD56+CD3– NK cells in patients’ PBMC before NK-cell infusion, seven days after infusion and 14 days after infusion. However, we could not show any differences among them before and after NK-cell infusion (Figure 1G).
Table 3.
Absolute number of CD56-expressing cells and cytolytic activity in lymphoma patients before and after infusion of natural killer (NK) cells.
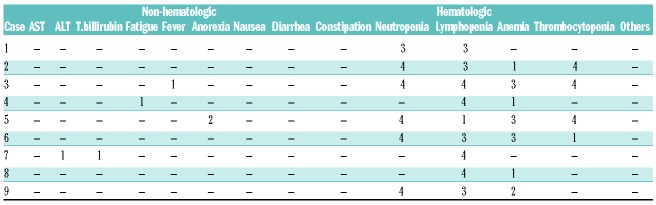
During a median follow-up period of 28 months (range, 6-56 months), seven patients maintained complete response and two patients died with disease progression 24 months and 10 months after infusion, respectively. The median duration of complete response was 44 months (range, 6-56 months) (Table 1). There were several hematologic adverse events related to chemotherapy, but there were no severe non-hematologic adverse events (Table 4).
Table 4.
Adverse events according to Common Terminology Criteria for Adverse Events Ver.4.0.
Pievani et al. reported the addition of rituximab increases the killing activity of cytokine-induced killer (CIK) cells that contained 47±8% of CD3+CD56+ NKT cells, 49±9% of CD3+CD56– T cells, and only 4±3% of CD3–CD56+ NK cells against B-cell non-Hodgkin lymphoma (NHL) targets in vitro.11 Therefore, it was suggested that rituximab could be used in vivo to enhance cytokine-induced killer cell therapeutic activity for B-cell NHL. Deng et al. observed a synergistic cytotoxicity of expanded NK cells against CD20+ B-lymphoma cell lines and greater production of IFN-γ and TNF-a when combined with rituximab in vitro.12 In vitro expanded NK cells show anti-lymphoma activity in SCID mice that had been intravenously injected with Raji human B-cell lymphoma cells, and the addition of rituximab enhanced their efficacy by triggering ADCC.13 It was reported that the addition of rituximab induced NK-mediated ADCC and augmented killing of CD20+ target cells when using ex vivo expanded NK cells with K562-mbIL15-41BBL cells and IL-2 in vitro.14 Therefore, the combination of adoptive transfer of expanded NK cells and rituximab could be a promising cellular therapy for CD20-expressing malignant lymphoma.
Tarek et al. reported that, in the neuroblastoma setting, HLA class I expression on NB cells selectively inhibited licensed NK-cell activity, permitting primarily unlicensed NK cells to mediate ADCC. Therefore, unlicensed NK cells expressing KIR for non–self-HLA ligands may be more effective in tumor eradication.15 Infused autologous NK cell with high expression level of NKG2A (approx. 60%) in this study may be relatively naïve immunologically.
In summary, we conducted a phase I study of ex vivo expanded NK-cell therapy using patients’ autologous PBMC combined with rituximab-containing chemotherapy for relapsed CD20-positive malignant lymphoma patients to enhance ADCC. Adoptive infusion of expanded auto-CD56+CD3– NK cells with higher cytolytic activities and higher expression levels of NKp30, NKp44 and CD16 compared with those before culture up to 10×106/kg is safe and feasible. Seven of the nine patients maintained complete response with a median duration of 44 months (range, 6-56 months) after NK-cell infusion. However, it is difficult to evaluate the exact contribution of NK-cell infusion for clinical response because eight of the nine patients in this study received chemotherapy after NK-cell infusion. However, ex vivo expanded and activated autologous NK-cell therapy combined with rituximab-containing chemotherapy for relapsed CD20-positive malignant lymphoma patients may be one of the promising options among various adoptive cellular therapies including CAR-T/NK.
Acknowledgments
We thank Ms. R. Tsuruya and Ms. Y. Maruyama for their assistance.
Footnotes
Funding: this study was supported in part by a Grant-in-Aid from the Ministry of Health, Labor and Welfare of Japan, the Mitsubishi Foundation and Tella Corporation .
Information on authorship, contributions, and financial & other disclosures was provided by the authors and is available with the online version of this article at www.haematologica.org.
References
- 1.Locatelli F, Moretta F, Brescia L, Merli P. Natural killer cells in the treatment of high-risk acute leukaemia. Semin Immunol. 2014;26(2):173–179. [DOI] [PubMed] [Google Scholar]
- 2.Johnson JK, Miller JS. Current strategies exploiting NK-cell therapy to treat haematologic malignancies. Int J Immunogenet. 2018. July 16 [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 3.Fang F, Xiao W, Tian Z. Challenges of NK cell-based immunotherapy in the new era. Front Med. 2018;12(4):440–450. [DOI] [PubMed] [Google Scholar]
- 4.Guillerey C, Huntington ND, Smyth MJ. Targeting natural killer cells in cancer immunotherapy. Nat Immunol.2016; 17(9):1025–1036. [DOI] [PubMed] [Google Scholar]
- 5.Cooley S, Parham P, Miller JS. Strategies to activate NK cells to prevent relapse and induce remission following hematopoietic stem cell transplantation. Blood. 2018;131(10):1053–1062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tanaka J, Toubai T, Tsutsumi Y, et al. Cytolytic activity and regulatory functions of inhibitory NK cell receptor-expressing T cells expanded from granulocyte colony-stimulating factor-mobilized peripheral blood mononuclear cells. Blood. 2004;104(3):768–774. [DOI] [PubMed] [Google Scholar]
- 7.Tanaka J, Sugita J, Shiratori S, et al. Expansion of NK cells from cord blood with antileukemic activity using GMP-compliant substances without feeder cells. Leukemia. 2012;26(5):1149–1152. [DOI] [PubMed] [Google Scholar]
- 8.Tanaka J, Sugita J, Shiratori S, Shigematsu A, Imamura M. Dasatinib enhances the expansion of CD56+CD3− NK cells from cord blood. Blood. 2012;119(25):6175–6176. [DOI] [PubMed] [Google Scholar]
- 9.Smith MR. Rituximab (monoclonal anti-CD20 antibody): mechanisms of action and resistance. Oncogene. 2003;22(47):7359–7368. [DOI] [PubMed] [Google Scholar]
- 10.Lim SH, Beers SA, French RR, Johnson PW, Glennie MJ, Cragg MS. Anti-CD20 monoclonal antibodies: historical and future perspectives. Haematologica. 2010;95(1):135–143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pievani A, Belussi C, Klein C, Rambaldi A, Golay J, Introna M. Enhanced killing of human B-cell lymphoma targets by combined use of cytokine-induced killer cell (CIK) cultures and anti-CD20 antibodies. Blood. 2011;117(2):510–518. [DOI] [PubMed] [Google Scholar]
- 12.Deng X, Terunuma H, Nieda M, Xiao W, Nicol A. Synergistic cytotoxicity of ex vivo expanded natural killer cells in combination with monoclonal antibody drugs against cancer cells. Int Immunopharmacol. 2012;14(4):593–605. [DOI] [PubMed] [Google Scholar]
- 13.Lim O, Lee Y, Chung H, et al. GMP-compliant, large-scale expanded allogeneic natural killer cells have potent cytolytic activity against cancer cells in vitro and in vivo. PLoS One. 2013;8(1):e53611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wang W, Erbe AK, Alderson KA, et al. Human NK cells maintain licensing status and are subject to killer immunoglobulin-like receptor (KIR) and KIR-ligand inhibition following ex vivo expansion. Cancer Immunol Immunother. 2016;65(9):1047–1059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tarek N, Le Luduec JB, Gallagher MM, et al. Unlicensed NK cells target neuroblastoma following anti-GD2 antibody treatment. J Clin Invest. 2012;122(9):3260–3270. [DOI] [PMC free article] [PubMed] [Google Scholar]