Abstract
Background. In both military and civilian settings, transmission of respiratory pathogens may be due to person-to-person and environmental contributions. This possibility was explored in a military training setting, where rates of febrile respiratory illness (FRI) often reach epidemic levels.
Methods. Population size and FRI rates were monitored over 10 months in the units of 50–90 individuals. Some units were open to the influx of potentially infectious convalescents (hereafter referred to as “open units,” and some were closed to such an influx (hereafter referred to as “closed units”). Virologic testing and polymerase chain reaction analysis were used to detect adenovirus on surface structures.
Results. The odds ratio (OR) associated with FRI in closed units, compared with open units, was 1.13 (95% confidence interval [CI], 0.99–1.28). The OR in units with a population greater than the median size, compared with units with a population lower than the median size was 1.38 (95% CI, 1.23–1.55). Between 5% and 9% of surface samples obtained from selected units harbored viable adenovirus.
Conclusions. FRI rates were not reduced in units that were closed to potentially contagious individuals. These findings imply that the primary source of the pathogen is likely environmental rather than human, and they underscore what is known about other virus types. Diligence in identifying the relative roles of different transmission routes is suggested for civilian settings similar to those described in the current study.
Among the means of controlling transmission of disease, protecting groups of people by limiting their interactions may be highly effective. For example, such “social distancing” has been shown to have a role in reducing the spread of pandemic influenza [1, 2]. In the present study, we investigate the usefulness of social distancing in an environment in which high rates of febrile respiratory illness (FRI) are endemic.
The dynamics and persistence of respiratory pathogens, specifically those associated with FRI in the military recruit training setting, have never been well understood [3]. A characteristic of this setting is the constant influx of potentially infectious and susceptible individuals, a situation that also exists in certain civilian settings, such as educational institutions [4], hospitals, elder care facilities [5, 6], and places where groups of children gather [7–9]. Previous work has identified other potential contributors to pathogen transmission, including crowding [10–17], the type of pathogen [13, 18–22], population age and health [5, 6], sanitation [14, 16], and the size, materials, and ventilation of buildings [16, 23–25].
Russell et al. [3] found a temporal association between FRI rates and the frequency of potentially infectious military recruits circulating between training units and inferred that this may be a primary source and route of adenovirus transmission and persistence. In a study conducted by Greenwood et al. [26] in the 1930s, epidemics of disease could be manipulated in mice populations by regulating the influx of susceptible persons. Arlander et al. [27] suggested that the mechanism demonstrated in the mice populations evaluated by Greenwood et al. [26] could play a crucial role in FRI epidemics at military recruit training centers and wherever new individuals are continually being introduced into a relatively dense population.
In the present study, we achieved a large measure of experimentally manipulated social distance within a military recruit population. Because the primary means of transmission was presumed to be person-to-person transmission, we expected that groups that were socially distanced from potentially infectious individuals who were new to the group would incur rates of illness lower than those noted for groups that were not socially distanced. An interaction between social distancing and population size was also anticipated. In addition, we were able to examine an alternative hypothesis arising from research on the effects of contamination with environmental pathogens on illness rates [3, 28–31].
Several viral pathogens responsible for respiratory illness have been shown likely to be transmitted via building surfaces in civilian settings [29]. Adenovirus type 4 (Ad4) is known to survive on surfaces in the buildings where the military recruit population lives [3], providing a potentially robust source of infection. The present study weighs the relative significance of these different modes and sources of pathogen transmission, as well as their association with FRI.
Methods
Setting. Military recruit training entails 2 days of medical and administrative in-processing, followed by assignment of incoming recruits to distinct housing units. These units train together, and they have very little interaction with other units or groups of people, unless they visit the medical clinic. In essence, each unit is “cohorted” into one specific open-bay housing unit.
Recruits who are unable to train because they are sick or lack physical conditioning are removed from training and are placed in either the medical convalescent unit (MCU) or the physical conditioning unit (PCU). At the time of release (when a patient exhibits no symptoms and has adequate physical conditioning), recruits from both of these units typically join a group of new recruits who are already in training and have reached the same point in training that the returning recruits had reached at the time of their removal.
In the present study, some housing units were closed to the introduction of any new recruits after being formed (hereafter referred to as “closed units”). Recruits from closed units who were sick did report to the medical clinic, and if their symptoms did not require placement in the MCU, the recruits returned to their unit after seeing their provider. Other units (hereafter referred to as “open units”) remained open to the introduction of convalescent recruits or recruits temporarily receiving additional conditioning training, as per normal routine. For each decrease in the number of recruits in these open units, a replacement recruit was usually provided from the MCU or PCU. The study was conducted during the first 4 weeks of recruit training, which historically is the period associated with the highest incidence of FRI. The institutional review board of the Naval Health Research Center classified this protocol as a nonresearch public health endeavor.
Data acquisition. Data on 13,114 male military recruits (mean age, 19 years) at the Marine Corps Recruit Depot in San Diego, California (there are no female recruits at this facility), were collected between February 2004 and March 2005. These recruits were distributed among a total of 196 housing units. Oropharyngeal swab specimens were obtained from all recruits discharged from the MCU or the PCU, and they were processed to detect the presence or absence of adenovirus, as described elsewhere [5]. The density and size of each unit, as well as unit-specific cases of FRI, were recorded. “FRI” was defined either by a body temperature of >38°C and 1 respiratory symptom or by the presence of nonfebrile pneumonia. The FRI rates provided in the present article denote the number of cases of FRI per 100 persons per week, averaged over the 4 weeks during which each housing group was measured.
Squares of 30 × 30 cm on selected surfaces in housing units and in the medical clinic were swabbed with one Dacron swab, which was stored in a tube of viral transport medium (MicroTest M4 Transport; Remel). In the case of irregular objects, contours likely to be touched by people were judiciously swabbed with one swab. The samples were tested by polymerase chain reaction (PCR) performed using the same method [32] used for the samples obtained from humans, and these samples were also cultured as described elsewhere [3]. Three such sample collection efforts were conducted. In the first sample collection, 96 surface sites in swabbed, and the specimens were cultured in a unit that had been vacant for 3 days after 4 weeks of occupation, during which time the FRI attack rate in the unit was 33%. In the second collection, 80 samples were collected from 11 different surface sites from 2 randomly chosen units that had been vacant for 1 week. In the third collection, 57 samples obtained from 10 different surface sites in the medical clinic were collected.
Statistical analysis. Mean FRI rates were calculated from the count of FRI cases for each unit type (closed or open) and from unit population size. A χ2 test was performed to compare FRI rates of different unit population sizes for open units, and logistic regression analysis was performed to evaluate the interaction between type of unit, population size, and overall occurrence of FRI.
Results
When averaged over the 4 weeks that each open unit was tracked, FRI rates were 3.46 cases/100 person-weeks and were greater than the levels considered to denote an epidemic (i.e., ≥1.5 cases/100 person-weeks) in 88% of the units. It has been estimated that 81% of the FRI cases were due to Ad4 (Naval Health Research Center Tri-Service FRI Surveillance, unpublished data).
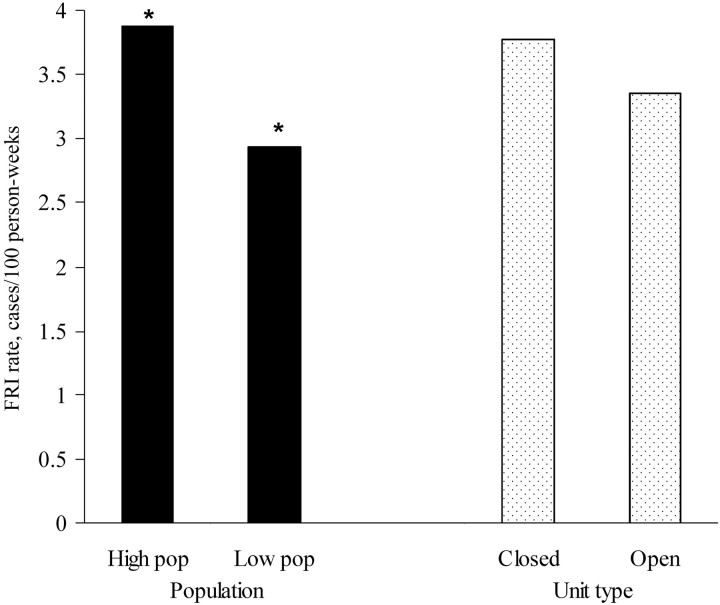
Case numbers and rates. The closed units, which had an average of 3.92 cases/100 person-weeks over 4 weeks (i.e., 329 cases among 2099 subjects), had slightly higher FRI rates than the open units, which had an average of 3.48 cases/100 person-weeks over 4 weeks (i.e., 1586 cases among 11,389 subjects). However, a logistic regression of the odds of an FRI diagnosis, calculated for the type of unit and the population size, showed that the difference was not statistically significant (adjusted odds ratio [OR], 1.13 [95% confidence interval {CI}, 0.99–1.28]; χ2 = 3.17; P = .075) (figure 1).
Figure 1.
Contrast of populations with a size above the median (High pop) and those with a size below the median (Low pop). The rates of febrile respiratory illness (FRI) for these populations were significantly different. Although the units closed to an influx of potentially infectious convalescing persons (hereafter referred to as “closed units”) generally had higher FRI rates than the units open to such an influx (hereafter referred to as “open units”), the difference was not significant. Logistic regression of the odds of an FRI diagnosis, in terms of the population size and unit type, showed that the difference in population size remained even after adjusting for unit type.
Oropharyngeal samples obtained from 762 people discharged from the MCU and from 395 people discharged from the PCU were processed by PCR for the detection of Ad4. Ten percent of the samples from the group discharged from the MCU had evidence of Ad4, as did 6% of samples from the group discharged from the PCU.
Population sizes and FRI case rates and numbers. The effect of population size was addressed first by removing the experimental (i.e., closed) units from the analysis, to gain a picture of the association between FRI and population in the absence of an experimental manipulation. In the 166 open units alone, where the population size ranged from 44 to 88 people/unit, there were 1602 FRI cases (among 11,389 people). The average FRI rate in open units ranged from 0.44 to 8.97 cases/100 person-weeks over 4 weeks. Recruits in units with a population size greater than the median value had significantly higher odds of FRI than did units with a population size less than the median value (OR, 1.38 [95% CI, 1.23–1.55]; χ2 = 30.13; P < .001). The Pearson r correlation between unit population size and FRI was 0.55 (P < .01).
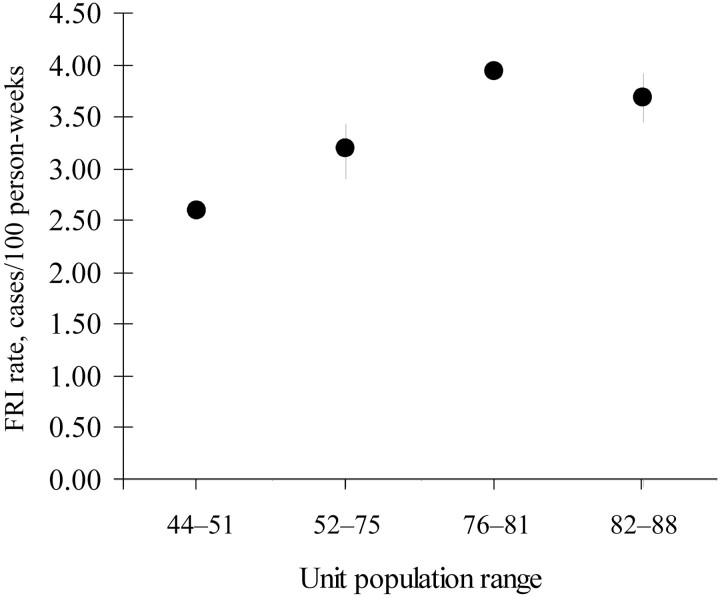
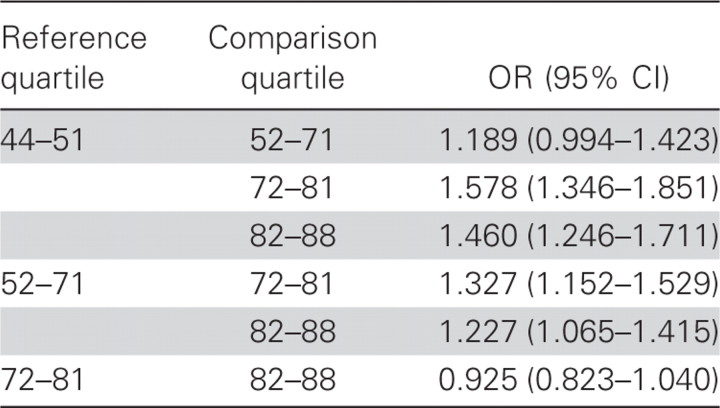
Second, when both types of units (open and closed) were included, logistic regression performed for a unit population size greater than the median versus a unit population size less than the median showed that the former had higher odds of an FRI diagnosis than the latter, even with adjustment for the type of unit (adjusted OR, 1.40 [95% CI, 1.23–1.54]; χ2 = 35.34; P < .001) (figure 1). With the use of quartiles as a guide, 4 groups were constructed using both closed and open units, according to the population-size ranges of the units: group 1 consisted of units with 44–51 people; group 2, units with 52–75 people; group 3, units with 76–81 people; and group 4, units with 82–88 people. A comparison of these groups is shown in figure 2. The logistic regression analysis of the effect of quartile-based groupings of population sizes on odds of an FRI diagnosis, adjusted for type of unit, showed that groups 1 and 2 were each different from groups 3 and 4, but groups 1 and 2 were not statistically different from each other, nor were groups 3 and 4 different from each other (table 1). The mean FRI rate for each group is shown in figure 2.
Figure 2.
Febrile respiratory illness (FRI) rates and 95% confidence intervals of units grouped according to 4 population size ranges formed around the quartiles.
Table 1.
Logistic regression analysis of effect of quartile group on odds of an FRI diagnosis, adjusted for type of unit.
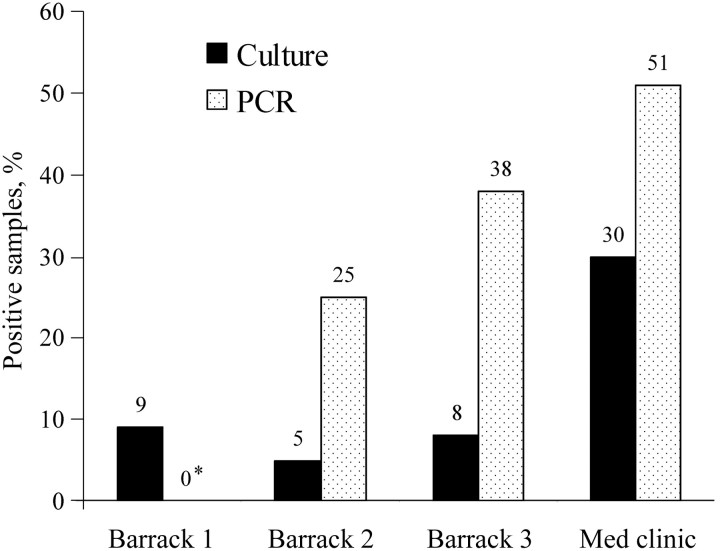
Surface samples obtained from the environment. In the unit that was vacant for 3 days, 96 sites were swabbed and the resulting specimens cultured; of these sites, 9% were found to harbor viable virus. In 1 of the 2 units that was vacant for 1 week, 5% of the samples were culture positive and 25% were PCR positive. In the other unit that was vacant for 1 week, 8% of the samples were culture positive and 38% were PCR positive. Thirty percent of the samples obtained from the 57 sites in the medical clinic were culture positive, and 51% were PCR positive (figure 3). Specific sites in the housing units with relatively higher concentrations of Ad4 were the pillows, bedposts, mattresses, and lockers, and, in the clinic, these sites were the restroom door, sink, waiting room floor and walls, and examination chairs.
Figure 3.
Percentage of surface samples in each barrack that were found to be positive for adenovirus type 4 by means of culture and polymerase chain reaction (PCR) analysis. The figures above the bars denote the number of samples. The asterisk (*) denotes that the DNA test for barrack 1 is unavailable. Barrack 1 was an open-bay unit that was vacant for 3 days, Barracks 2 and 3 were open-bay units that were vacant for 1 week, and the medical clinic (Med clinic) was operating routinely. Multiple sites within each building were sampled.
Discussion
The original hypothesis was rejected. There was not a statistically significant difference between the FRI rates in the open and closed units as a whole, although the tendency was for the closed units to exhibit higher rates. The rejection of the hypothesis suggests that the primary route of transmission of FRI is not via the MCU/PCU recycling protocol (i.e., not via person-to-person contact between unit members and members newly introduced to the unit [i.e., potentially infectious convalescents]). The social distancing instituted in this setting was not successful in decreasing FRI rates. These results should be placed in the context of our finding of the existence of substantial environmental pathogen contamination in the housing units. The lessons may be similar for any setting in which environmental transmission of pathogens is likely.
Ten percent of MCU discharges and 6% of PCU discharges had Ad4 in the oropharynx. Either such percentages are insufficient to influence overall rates in a unit, where two-thirds of the population is assumed to be susceptible [3], or these individuals are not sufficiently “infectious” in their convalescent state. However, as described by Russell et al. [3], by the end of 6 weeks, virtually 100% of the people in the units under surveillance had anti-Ad4 titers. It is also known that 60%–80% of the FRI cases that occur at this institution are due to Ad4. Other pathogens, such as influenza viruses A and B, coronavirus, rhinovirus, and enterovirus, have each been shown to be responsible for very low percentages of the total FRI rate (Naval Health Research Center Tri-Service FRI Surveillance, unpublished data), and it is very unlikely that they play significant role.
In general, respiratory illness is believed to be primarily caused by direct person-to-person aerosolized transmission. More recent studies in civilian settings [28, 30, 31] have questioned this assumption, and the Centers for Disease Control and Prevention recommends disinfection of environmental surfaces possibly exposed to respiratory pathogens in health care settings [33]. The spread of a pathogen via an indirect surface route makes sense if it is shown that the pathogen can persist on the surface for a sufficient duration, as Boone and Gerba [29] have shown, with hand contact an essential mode of transmission [34]. Of the viral pathogens responsible for respiratory illness, coronavirus [35, 36], influenza [37], rhinovirus [30], and adenovirus [16, 23] are likely to be transmissible via environmental surfaces in different conditions of room air [29, 38–40]. Despite the fact that our study was not designed for prospective control and measurement of environmental pathogens, the findings of these other studies place ours in a context.
Ad4 is endemic in military training facilities, and its source is not incoming recruits. There are 2 observations supporting this position. First, after the military adenovirus vaccine program ended in 1999, Ad4 was found in an increasing majority of FRI cases [41–43], and, recently, it was found to represent nearly all FRI cases [41, 44]. Genome typing of Ad4 isolates recovered from 8 different training sites revealed that 4 of the 7 known genomic variants of Ad4 individually dominate adenovirus populations for years at a time at particular training sites, virtually always to the exclusion of any other genome type [21]. The occurrence of these genome types is not correlated between sites, obviating a shared external source (e.g., incoming recruits).
Second, in Army training centers, all recruits depart for a 2-week period at the end of December. On the recruits' return, FRI cases are seen, but adenovirus is not considered to be an etiology for the first couple of weeks. Studies have shown that local staff do not harbor the virus; however, inevitably, year after year, FRI rates due to the same endemic adenovirus strain resume expected levels [41]. Generally, the rate slowly begins to increase after 1 month, and it does not return to expected levels until ∼2 months after the beginning of January. Russell et al. [3] similarly found, at another training center, that none of the 271 recruits studied who were tested within 1 day of arriving at the center had oropharyngeal evidence of viable Ad4, and only 1 had evidence of Ad4 DNA. Thus, they are not coming to the center with the prevailing Ad4 virus and especially not with the Ad4 genome type specific to the training center. It is likely that the pathogen reservoir is the environment.
A growing number of studies in both military and civilian settings have demonstrated the relevance of environmental transmission of pathogens that cause respiratory illness [29, 45]. In the setting of military recruit housing, studies performed by Artenstein and Miller's group and other investigators [39, 45, 46] and, more recently, by Echavarria et al. [47] demonstrated a strong association between the presence of Ad4 contamination of the air and adenovirus-associated FRI rates. Russell et al. [3] had uncovered very similar findings for air samples, as well as for surface samples. Such results strongly implicate the air as the source of transmission in the setting studied. In the present study, 4 separate collections of samples from random surfaces at buildings clearly show the existence of Ad4 on many of the structures' various surfaces (up to 50% of the surface sites examined).
Furthermore, although the recruits arrived at the camp without oropharyngeal evidence of Ad4, 21% of the subjects in the study by Russell et al. [3] had Ad4 DNA detected in swab specimens collected from their hands by the second day of training. Apparently, recruits are exposed to FRI-inducing pathogens almost immediately on arrival at the training center. It is possible that these structures are contaminated by infectious individuals and are not themselves infectious. However, because these are the structures where the subjects live and receive medical care, and because it is known that adenovirus is transmissible via surface-to-hand contact [29], these findings suggest that environmental sources of infection are critical to the propagation and maintenance of high FRI prevalence rates in this setting. This early exposure could have played a role in the negligible subsequent effects of cohorting.
Nevertheless, it is curious that Ad4 can persist so robustly on surfaces for days to weeks yet can take 1–2 months to return to normal prevalence levels at Army centers after the winter break. Survival on surfaces may explain the persistence of Ad4 and its type-specific niche at a training center, but, because it is also clear that Ad4 can be transmitted by person-to-person contact, we must account for personal interactions. It is quite likely that recruits who are not already infected by Ad4 pick it up within their own units or while visiting the medical clinic (from its surfaces or other infectious recruits) and then bring it back to the unit. Person-to-person transmission is then facilitated by crowding, as the statistically significant association between population size and FRI rates indicate (figure 1). A similar effect of crowing should be expected for any civilian setting involving close quarters.
The association between population size and illness rates is actually rather complex. For example, Breese et al. [13] presented evidence that respiratory illness in the Navy in World War II was related to the number of people per room and not to population density. On the other hand, at a naval recruit training center, Miller [19] found that newer, larger barracks with more space per recruit were associated with lower FRI rates, even though the number of recruits per room was equivalent in the old and new barracks. He suggested that some of these apparent contradictions may be due to the nature of the pathogen responsible and the time at which an outbreak occurs during training. For example, data produced by Breese et al. [13] show that, in the first 2 weeks of training, correlations between illness and density were relatively higher than those between illness and population size; however, the reverse was the case for the second 4 weeks. Thus, although it is natural to assume that there is some association between the number of people in a space and the rates of illness, it is advisable to keep in mind that population size and density may interact in different ways with the type of pathogen and the amount of time that a unit of people have been together (for comparison, see the article by Bernstein [11]).
In the present study, for example, when units are grouped into quartiles of population sizes, FRI rates increase with each quartile from the first (lowest population) to the third, and they then level off, with no difference noted between the third and fourth quartiles. Larger populations increase the likelihood of person-to-person spread, but they also increase the likelihood of maintaining and increasing the environmental reservoir, which may be very robust. Teasing out the relative influence of these factors would require a prospective study that we did not perform here, with even greater control than that found by Russell et al. [3]
In summary, we found no statistically significant effect of a social distancing procedure in which some units were closed to an influx of potentially infectious individuals. The hypothesis of the present study assumed that the primary mode of transmission was from person to person. However, on the basis of our negative results and the evidence of fomite habitation of the virus presented in the current article and in other studies [3, 21, 41], environmental contamination of pathogens is implicated as playing a larger role in the spread of respiratory disease in this setting than was previously assumed. An environmental reservoir likely initiates transmission cycles.
Although our data do not establish a cause-and-effect association, both person-to-person and environmental transmission routes are expected to become more significant as population sizes increase. Although the setting here was a military institution and the pathogen was one virus in particular, the findings of this study could have implications for the numerous civilian settings and other pathogens described in the introduction. This observation should be particularly relevant for group-type living conditions. It should also be particularly relevant for pathogens that can be spread by fomites. It has been recommended that, when this is the case, a program of frequent hand washing should be instituted [48].
Influenza, for example, is a pathogen identified as being transmissible via environmental surfaces [12, 40, 49, 50]. Given this knowledge, even though social distancing would be primary in plans for emergency control of transmission in a population during a respiratory disease pandemic, it may be advisable to (1) consider the effect of potential surface contamination and (2) be cognizant of the dynamics of suspected person-to-person transmission routes. These, at least, are the lessons in the context of our study. Unsuspected environmental routes may contribute significantly to transmission, and highly suspected routes of person-to-person transmission may not be so.
In general, assumptions regarding person-to-person transmission of respiratory pathogens may require further examination. Distancing of groups of people according to their work, school, or housing situations requires assessment of transmission dynamics with respect to the infectiousness and survival of the pathogen, the disease's infectivity and pathogenicity, the circulation of susceptible people, crowding, and pathogen contamination of the environment.
Acknowledgments
We are indebted to the command and staff of the Marine Corps Recruit Depot, San Diego (MCRD-SD), and the MCRD-SD Branch Medical Clinic, under the command of the Naval Medical Center, San Diego, without whose cooperation this study would not have been possible. We are especially grateful to Lieutenant Colonel Owens, Captain Adam, and Major Fuller, as well as the many housing group commanders and drill instructors of MCRD-SD, as well as Captain Ziemke, Lieutenant Payton, and Hospitalman Sandoval, Hospitalman Mendez, and Hospital Corpsman 2nd Class Gadomski at the MCRD-SD Branch Medical Clinic. We also thank the professionals at the Molecular Laboratory at the Naval Health Research Center, especially Angel Osuna, Marina Irvine, and Ryan Ortiguerra, for their analysis of the samples.
Footnotes
Potential conflicts of interest: none reported.
Financial support: US Department of Defense and the Global Emerging Infections Surveillance and Response System (award 61102A-M0101-60501). This study is supported by the Department of Defense under work unit no. 60501.
This research has been conducted in compliance with all applicable federal regulations governing the protection of human subjects in research (protocol NHRC.1999.0002). The views expressed in this article are those of the authors and do not reflect the official policy or position of the Department of the Navy, the Department of Defense, or the US Government.
References
- 1.Glass RJ, Glass LM, Beyeler WE, Min HJ. Targeted social distancing design for pandemic influenza. Emerg Infect Dis. 2006;12:1671–81. doi: 10.3201/eid1211.060255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Caley P, Philp DJ, McCracken K. Quantifying social distancing arising from pandemic influenza. J R Soc Interface. 2008;5:631–9. doi: 10.1098/rsif.2007.1197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Russell KL, Broderick MP, Franklin SE, et al. Transmission dynamics and prospective environmental sampling of adenovirus in a military recruit setting. J Infect Dis. 2006;194:877–85. doi: 10.1086/507426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Feikin DR, Moroney JF, Talkington DF, et al. An outbreak of acute respiratory disease caused by Mycoplasma pneumoniae and adenovirus at a federal service training academy: new implications from an old scenario. Clin Infect Dis. 1999;29:1545–50. doi: 10.1086/313500. [DOI] [PubMed] [Google Scholar]
- 5.Gross PA, Rodstein M, LaMontagne JR, et al. Epidemiology of acute respiratory illness during an influenza outbreak in a nursing home: a prospective study. Arch Intern Med. 1988;148:559–61. [PubMed] [Google Scholar]
- 6.Morris RD, Munasinghe RL. Geographic variability in hospital admission rates for respiratory disease among the elderly in the United States. Chest. 1994;106:1172–81. doi: 10.1378/chest.106.4.1172. [DOI] [PubMed] [Google Scholar]
- 7.Centers for Disease Control and Prevention. Outbreak of pharyngoconjunctival fever at a summer camp—North Carolina, 1991. Infect Control Hosp Epidemiol. 1992;13:499–500. [PubMed] [Google Scholar]
- 8.Gerber SI, Erdman DD, Pur SL, et al. Outbreak of adenovirus genome type 7d2 infection in a pediatric chronic-care facility and tertiary-care hospital. Clin Infect Dis. 2001;32:694–700. doi: 10.1086/319210. [DOI] [PubMed] [Google Scholar]
- 9.Porter JD, Teter M, Traister V, Pizzutti W, Parkin WE, Farrell J. Outbreak of adenoviral infections in a long-term paediatric facility, New Jersey, 1986/87. J Hosp Infect. 1991;18:201–10. doi: 10.1016/0195-6701(91)90144-w. [DOI] [PubMed] [Google Scholar]
- 10.Azizi BH, Zulkifli HI, Kasim S. Indoor air pollution and asthma in hospitalized children in a tropical environment. J Asthma. 1995;32:413–8. doi: 10.3109/02770909409077752. [DOI] [PubMed] [Google Scholar]
- 11.Bernstein SH. Observations on the effects of housing on the incidence and spread of common respiratory diseases among Air Force recruits. Am J Hyg. 1957;65:162–71. doi: 10.1093/oxfordjournals.aje.a119863. [DOI] [PubMed] [Google Scholar]
- 12.Boone SA, Gerba CP. The occurrence of influenza A virus on household and day care center fomites. J Infect. 2005;51:103–9. doi: 10.1016/j.jinf.2004.09.011. [DOI] [PubMed] [Google Scholar]
- 13.Breese BB, Stanbury J, Upham H-V, Calhoun AJ, Van Buren RL, Kennedy AS. Influence of crowding on respiratory illness in a large naval training station. War Medicine. 1945;7:143–6. [Google Scholar]
- 14.Brodkey C, Gaydos JC. United States Army guidelines for troop living space: a historical review. Mil Med. 1980;145:418–21. [PubMed] [Google Scholar]
- 15.Cardoso MR, Cousens SN, de Goes Siqueira LF, Alves FM, D'Angelo LA. Crowding: risk factor or protective factor for lower respiratory disease in young children? BMC Public Health. 2004;4:19. doi: 10.1186/1471-2458-4-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lee T, Jordan NN, Sanchez JL, Gaydos JC. Selected nonvaccine interventions to prevent infectious acute respiratory disease. Am J Prev Med. 2005;28:305–16. doi: 10.1016/j.amepre.2004.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Musher DM. How contagious are common respiratory tract infections? N Engl J Med. 2003;348:1256–66. doi: 10.1056/NEJMra021771. [DOI] [PubMed] [Google Scholar]
- 18.Jordan WS., Jr Occurrence of adenovirus infections in civilian populations. AMA Arch Intern Med. 1958;101:54–9. doi: 10.1001/archinte.1958.00260130068006. [DOI] [PubMed] [Google Scholar]
- 19.Miller LF. Acute respiratory infections in naval personnel. Mil Med. 1964;129:526–32. [PubMed] [Google Scholar]
- 20.Dudding BA, Top FH, Jr, Winter PE, Buescher EL, Lamson TH, Leibovitz A. Acute respiratory disease in military trainees: the adenovirus surveillance program, 1966–1971. Am J Epidemiol. 1973;97:187–98. doi: 10.1093/oxfordjournals.aje.a121499. [DOI] [PubMed] [Google Scholar]
- 21.Kajon AE, Moseley JM, Metzgar D, et al. Molecular epidemiology of adenovirus type 4 infections in US military recruits in the postvaccination era (1997–2003) J Infect Dis. 2007;196:67–75. doi: 10.1086/518442. [DOI] [PubMed] [Google Scholar]
- 22.Schmitz H, Wigand R, Heinrich W. Worldwide epidemiology of human adenovirus infections. Am J Epidemiol. 1983;117:455–66. doi: 10.1093/oxfordjournals.aje.a113563. [DOI] [PubMed] [Google Scholar]
- 23.Tumwesigire SG, Barton T. Environmental risk factors for acute respiratory infections among children of military personnel in Uganda. East Afr Med J. 1995;72:290–4. [PubMed] [Google Scholar]
- 24.Milton AH, Rahman M. Respiratory effects and arsenic contaminated well water in Bangladesh. Int J Environ Health Res. 2002;12:175–9. doi: 10.1080/09603120220129346. [DOI] [PubMed] [Google Scholar]
- 25.Lemon HM, Wise H, Hamburger M. Bacterial content of air in army barracks: results. War Medicine. 1944;6:92–101. [Google Scholar]
- 26.Greenwood M. Epidemiology: historical and experimental. Baltimore: Johns Hopkins; 1932. [Google Scholar]
- 27.Arlander TR, Pierce WE, Edwards EA, Peckinpaugh RO, Miller LF. Epidemiology and prevention of acute respiratory disease in naval recruits. IV. An epidemiologic study of respiratory illness patterns in Navy and Marine Corps recruits. Am J Public Health Nations Health. 1965;55:67–80. doi: 10.2105/ajph.55.1.67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Barker J, Stevens D, Bloomfield SF. Spread and prevention of some common viral infections in community facilities and domestic homes. J Appl Microbiol. 2001;91:7–21. doi: 10.1046/j.1365-2672.2001.01364.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Boone SA, Gerba CP. Significance of fomites in the spread of respiratory and enteric viral disease. Appl Environ Microbiol. 2007;73:1687–96. doi: 10.1128/AEM.02051-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Aitken C, Jeffries DJ. Nosocomial spread of viral disease. Clin Microbiol Rev. 2001;14:528–46. doi: 10.1128/CMR.14.3.528-546.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Goldmann DA. Transmission of viral respiratory infections in the home. Pediatr Infect Dis J. 2000;19:S97–102. doi: 10.1097/00006454-200010001-00002. [DOI] [PubMed] [Google Scholar]
- 32.Metzgar D, Osuna M, Kajon AE, Hawksworth AW, Irvine M, Russell KL. Abrupt emergence of diverse species B adenoviruses at US military recruit training centers. J Infect Dis. 2007;196:1–9. doi: 10.1086/522970. [DOI] [PubMed] [Google Scholar]
- 33.Centers for Disease Control and Prevention, Healthcare Infection Control Practices Advisory Committee. Guidelines for environmental infection control in health-care facilities. MMWR Recomm Rep. 2003;52:1–42. [PubMed] [Google Scholar]
- 34.Maki DG, Alvarado CJ, Hassemer CA, Zilz MA. Relation of the inanimate hospital environment to endemic nosocomial infection. N Engl J Med. 1982;307:1562–6. doi: 10.1056/NEJM198212163072507. [DOI] [PubMed] [Google Scholar]
- 35.Abad FX, Villena C, Guix S, Caballero S, Pintó RM, Bosch A. Potential role of fomites in the vehicular transmission of human astroviruses. Appl Environ Microbiol. 2001;67:3904–7. doi: 10.1128/AEM.67.9.3904-3907.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Dowell SF, Simmerman JM, Erdman DD, et al. Severe acute respiratory syndrome coronavirus on hospital surfaces. Clin Infect Dis. 2004;39:652–7. doi: 10.1086/422652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hota B. Contamination, disinfection, and cross-colonization: are hospital surfaces reservoirs for nosocomial infection? Clin Infect Dis. 2004;39:1182–9. doi: 10.1086/424667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Abad FX, Pintó RM, Bosch A. Survival of enteric viruses on environmental fomites. Appl Environ Microbiol. 1994;60:3704–10. doi: 10.1128/aem.60.10.3704-3710.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Miller WS, Artenstein MS. Aerosol stability of three acute respiratory disease viruses. Proc Soc Exp Biol Med. 1967;125:222–7. doi: 10.3181/00379727-125-32054. [DOI] [PubMed] [Google Scholar]
- 40.Bean B, Moore BM, Sterner B, Peterson LR, Gerding DN, Balfour HH., Jr Survival of influenza viruses on environmental surfaces. J Infect Dis. 1982;146:47–51. doi: 10.1093/infdis/146.1.47. [DOI] [PubMed] [Google Scholar]
- 41.Russell KL, Hawksworth AW, Ryan MA, et al. Vaccine-preventable adenoviral respiratory illness in US military recruits, 1999–2004. Vaccine. 2006;24:2835–42. doi: 10.1016/j.vaccine.2005.12.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Erdman DD, Xu W, Gerber SI, et al. Molecular epidemiology of adenovirus type 7 in the United States, 1966–2000. Emerg Infect Dis. 2002;8:269–77. doi: 10.3201/eid0803.010190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Sanchez JL, Binn LN, Innis BL, et al. Epidemic of adenovirus-induced respiratory illness among US military recruits: epidemiologic and immunologic risk factors in healthy, young adults. J Med Virol. 2001;65:710–8. doi: 10.1002/jmv.2095. [DOI] [PubMed] [Google Scholar]
- 44.Blasiole DA, Metzgar D, Daum LT, et al. Molecular analysis of adenovirus isolates from vaccinated and unvaccinated young adults. J Clin Microbiol. 2004;42:1686–93. doi: 10.1128/JCM.42.4.1686-1693.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Artenstein MS, Miller WS, Lamson TH, Brandt BL. Large-volume air sampling for meningococci and adenoviruses. Am J Epidemiol. 1968;87:567–77. doi: 10.1093/oxfordjournals.aje.a120847. [DOI] [PubMed] [Google Scholar]
- 46.Artenstein MS, Miller WS, Rust JH, Jr, Lamson TH. Large-volume air sampling of human respiratory disease pathogens. Am J Epidemiol. 1967;85:479–85. doi: 10.1093/oxfordjournals.aje.a120710. [DOI] [PubMed] [Google Scholar]
- 47.Echavarria M, Kolavic SA, Cersovsky S, et al. Detection of adenoviruses (AdV) in culture-negative environmental samples by PCR during an AdV-associated respiratory disease outbreak. J Clin Microbiol. 2000;38:2982–4. doi: 10.1128/jcm.38.8.2982-2984.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ryan MA, Christian RS, Wohlrabe J. Handwashing and respiratory illness among young adults in military training. Am J Prev Med. 2001;21:79–83. doi: 10.1016/s0749-3797(01)00323-3. [DOI] [PubMed] [Google Scholar]
- 49.Ansari SA, Springthorpe VS, Sattar SA, Rivard S, Rahman M. Potential role of hands in the spread of respiratory viral infections: studies with human parainfluenza virus 3 and rhinovirus 14. J Clin Microbiol. 1991;29:2115–9. doi: 10.1128/jcm.29.10.2115-2119.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Brady MT, Evans J, Cuartas J. Survival and disinfection of parainfluenza viruses on environmental surfaces. Am J Infect Control. 1990;18:18–23. doi: 10.1016/0196-6553(90)90206-8. [DOI] [PubMed] [Google Scholar]