Abstract
To determine the spectrum of pathogens causing acute febrile respiratory illness in human immunodeficiency virus (HIV)-infected adus, we re-analyzed data from a prospective surveillance study involving 50 outpatients (90% of whom received highly active antiretroviral therapy). Nasopharyngeal samples were tested for 23 respiratory viruses by multiplex reverse-transcriptase polymerase chain reaction (PCR) and for atypical bacteria by PCR. Sputum cultures and serological testing were performed. Viruses accounted for 64% of infections. After influenza (22 cases), humanmetapneumovirus infection (6 cases) was most common and was associated with bronchospasm. Bacterial infections occurred in 6 patients (3 of whom had concurrent viral infection). Over 80% of patients received antibiotics. Rapid testing to identify specific viral pathogens could aid in patient management and reduce unnecessary antibiotic exposure.
Defects in humoral and cellular immunity associated with human immunodeficiency virus (HIV) infection potentially alter the course and severity of common viral infections [1]. Although highly active antiretroviral therapy (HAART) may partially restore these deficiencies, HIV-infected persons may remain at increased risk for morbidity from respiratory illnesses, especially if their ability to generate antigen-specific responses remains impaired. The high prevalence of smoking and chronic lung diseases among these individuals may further predispose HIV-infected patients to respiratory tract infections [2]. Despite the potential public health impact of viral infections in this population, there is virtually no information on the causes, risk factors, and outcomes of respiratory infection in the HAART era.
Several respiratory viruses have recently been discovered, including human metapneumovirus (hMPV) and coronaviruses [3]. HMPV, in particular, is an important cause of severe respiratory illness, particularly in children, elderly individuals, and immunocompromised individuals [4, 5]. There are few studies that have examined its role in HIV-infected adults, but hMPV, like respiratory syncytial virus (RSV) [6], may potentially be important, given its predilection for causing disease in immunocompromised hosts.
The development of a new technology, 23-plex polymerase chain reaction (PCR) coupled to microarray hybridization, has allowed us to reanalyze specimens and data collected during a prospective surveillance study involving HIV-infected outpatients who consulted health care services for fever and respiratory symptoms in the HAART era [7]. Furthermore, we extended our studies to search for 3 common atypical bacterial pathogens—Mycoplasma pneumoniae, Chlamydophila pneumoniae, and Legionella pneumophila—and conventional bacteria in sputum. Our aim was to identify the full range of pathogens responsible for respiratory tract infection in HIV-infected outpatients.
Methods. Consecutive patients who presented for febrile respiratory syndromes from November 2003 through April 2006 were recruited from a large university-based clinic (Montreal Chest Hospital, McGill University Health Centre; Montreal, Canada) serving ∼1200 HIV-infected adults. The protocol was approved by the research ethics board. Respiratory infections were defined as a documented temperature >138°C associated with 1 or more of the following clinical symptoms: cough, tachypnea, dyspnea, wheezing, stridor, rhinorrhea, sore throat, and myalgias. Fifty seven patients were screened, and 50 were enrolled. Five patients were ineligible because of undocumented temperature; 2 refused nasopharyngeal aspiration; 42 completed a follow-up visit.
After informed consent, demographic and clinical data and vital signs were recorded. Additional investigations, such as chest and sinus radiographs, complete blood counts, and blood cultures, were performed at the discretion of the treating physician. An acute serum sample was collected during the initial consultation. Information on the duration and outcome of the episode and a convalescent serum sample were obtained 2 weeks later.
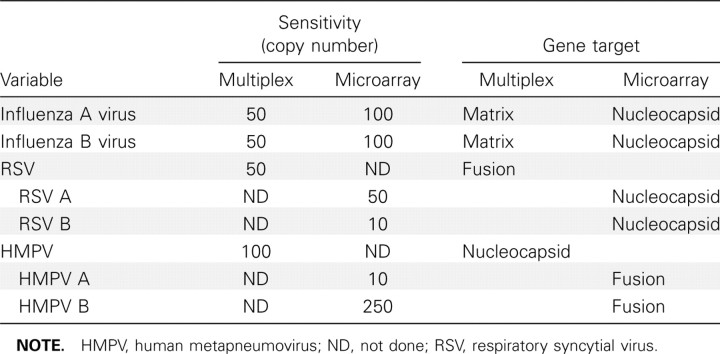
Nasopharyngeal aspirates were used to collect respiratory specimens from November 2003 through April 2004, after which combined nasal and throat swabs were used as described [7]. In the original study, we found similar agreement between nasal pharyngeal aspirates and swabs [7]. No PCR inhibitory substances were detected in our clinical samples. Samples were stored at 4°C for up to 4 days and then shipped to a central laboratory, where they were aliquoted and stored at –80°C until analyses at the end of the study. All respiratory samples were originally tested for influenza A and B virus, RSV, and hMPV using an in-house real-time multiplex reverse-transcriptase PCR assay, as described elsewhere [8]. Using aliquots of the original samples that were stored frozen, we now tested for adenoviruses groups A, B, C, and E; rhinovirus A and B; influenza A and B virus; hMPV A and B; RSV A and B; parainfluenzaviruses types 1–3; coronaviruses HKU1, 229E, NL63 and OC43; and enteroviruses A-D by multiplex PCR followed by primer extension and microarray hybridization in an integrated molecular diagnostic device, the INFINITI analyzer (AutoGenomics) [9]. A comparison of the sensitivities and gene targets of the 2 assays is shown in table 1.
table 1.
Comparison of the In-House Multiplex Polymerase Chain Reaction (PCR) Assay with 23-Plex PCR Microarray for Selected Respiratory Viral Pathogens
Paired acute and convalescent serum samples were tested by standard complement fixation assays for influenza A and B virus, RSV, parainfluenzaviruses 1–3, and adenoviruses. A 4-fold increase in titer was considered to be diagnostic for acute infection.
Nasopharyngeal samples were tested for the presence of the rRNA genes of M. pneumoniae, C. pneumoniae, and L. pneumophila. Also, a sputum sample (not induced) was taken from all individuals with productive cough and sent to the local microbiology laboratory for bacterial culture and antibiotic susceptibility.
Baseline characteristics were compared between patients with different infections using x2 or Fisher's exact test for proportions and Wilcoxon-Mann-Whitney U test for continuous variables. Data were analyzed using SAS software, version 9.02 (SAS Institute).
Results. Fifty patients met study criteria and consented to participate. The median CD4+ T cell count was 325 cells/µL (range, 2–808 cells/µL), median HIV RNA level was <50 copies/mL (range, <50–166,991 cells/µL), and 90% of participants were receiving HAART. Subjects who were not receiving HAART were either undergoing treatment interruption (3 patients) or had a CD4+ T cell count >350 cells/µL (2 patients).
Forty-nine of 50 patients had a sufficient respiratory specimen for PCR testing. Identification of a pathogen was made in 34 patients (68%) using 1 or more methods, including isolation of virus in 28 (56%), concomitant viral and bacterial isolation in 4 (8%), and isolation of bacteria in 2 (4%). Thus, viral infections were contributory in 64% of patients. The use of 23-plex PCR detected an additional 11 viral infections, compared with the number of infections detected by the in-house PCR, increasing the diagnostic yield by 22%, compared with that of our previous analyses.
All but 1 previously identified influenza A virus infection (10 cases) and all influenza B virus infections (10) were detected with the new assay, with 1 additional case each of influenza A and B virus infection found, bringing the total number of influenza infections to 22. The viral load in the 2 additional cases was very low (high cycle threshold number). The 1 case of influenza A that was not detected with the new assay was in a patient who also had serological test results that were positive for influenza A virus, suggesting that this was truly a positive result. Other viruses detected were hMPV A (in 3 patients) and hMPV B (in 3 patients, including 1 with concomitant influenza B virus infection), RSV (2 patients), parainfluenzavirus-2 (2 patients), parainfluenzavirus-3 (1 patient), and HKU-1 coronavirus (1 patient with concomitant influenza A virus infection). Previously, no cases of hMPV, RSV, parainfluenzavirus-2, or coronavirus infection were detected. All cases were detected from October through April in all years.
No isolated cases of M. pneumoniae, C. pneumoniae or L. pneumophila infection were detected in the respiratory specimens. One patient with parainfluenzavirus-3 infection detected by both multiplex PCR and serological testing also had test results that were positive for both C. pneumonaie and M. pneumoniae. Another patient with influenza B virus detected on multiplex testing also had M. pneumoniae infection.
Four of 12 patients with sputum cultures had documented bacterial pathogens: Streptococcus pneumoniae in 3 (including 2 patients with bacteremia, one with hMPV isolated and another with influenza A virus isolated) and Neisseria meningitidis in 1 (with a clinical diagnosis of pneumonia but normal chest radiograph findings).
Demographic and clinical characteristics of the study population, by the group of pathogens isolated, are shown in table 2. Of note, individuals with influenza infection and those with undiagnosed infection had lower CD4+ cell counts than did individuals with other etiologies (P = .02).
table 2.
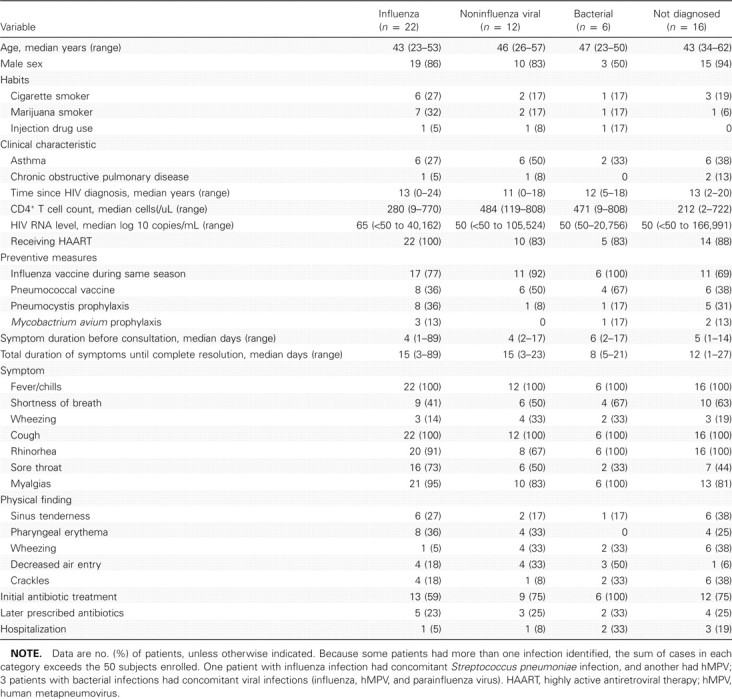
Demographic and Clinical Characteristics of Patients According to Etiology of Respiratory Illness Identified
Because the details related to cases of influenza have previously been published [7], and because the detection of the additional cases did not alter our previous findings, we focused on the characteristics of patients with hMPV infection, the second most isolated pathogen, differentiating it from influenza. All patients with hMPV infection and influenza were receiving HAART. However, patients with hMPV infection had higher median CD4+ cell counts (528 vs 280 cells/uL; P= .04). All patients had received the influenza vaccine, compared with 77% of those with influenza. Patients with hMPV infection were more likely than others to have a history of asthma (50% vs 32%). The only distinguishing clinical characteristic of hMPV infection was that it was associated with self-reported wheezing (50% vs 14%; P = .09), which was confirmed on physical examination (50% vs 5%; P = .02). No patient with isolated hMPV infection had pneumonia diagnosed on the basis of chest radiographs. Eighty-three percent of patients with hMPV infection received antibiotic therapy at some time, which is a rate that is similar to that for influenza, despite the most common clinical diagnoses being common cold or a flu-like illness. None of the patients who received a diagnosis of hMPV infection required hospitalization, compared with 1 patient (5%) with influenza.
Discussion. Almost no information exists on the etiology of respiratory tract infection in HIV-positive outpatients in the HAART era. Our previous study revealed the importance of influenza in this setting [7], which we have again confirmed. With the inclusion of 2 additional detected cases, influenza accounted for 42% of all observed respiratory tract illness in our population. Because of the increased sensitivity and spectrum of the multiplex detection device employed, we now have expanded our findings to highlight the frequency of a diverse number of respiratory tract viruses, including, to our knowledge for the first time, the demonstration that hMPV is an important pathogen, accounting for 12% of infections. HMPV mainly circulated concurrently with influenza B virus in the late winter months.
The only other published study to examine the role of viral pathogens among HIV-infected patients in the HAART era is a prospective study from the Swiss Cohort, involving a population with much more-advanced disease [10]. Viral PCR testing of bronchoalveolar lavage specimens from 59 patients undergoing investigation for opportunistic infection was performed. Unlike our patients, only 48% were receiving HAART, with median CD4+ cell counts of 55 cells/uL. Viruses were identified in 19% of patinets, whereas 36% had concomitant bacterial or opportunistic infections.
There are no studies that examine the role of hMPV infection in adults with HIV infection. As with previous reports from other settings, we observed that hMPV infections presented with nonspecific symptoms, such as fever and myalgias [4]. However, symptoms suggestive of lower respiratory tract involvement predominated, with cough, dyspnea, and wheezing in the vast majority of cases. Indeed, wheezing on physical examination was a distinguishing characteristic of hMPV infection. With the exception of 1 case of streptococcal pneumonia and bacteremia, no other complications were observed. However, >80% of those patients with hMPV infection received antibiotics despite an absence of radiographic signs of pneumonia, suggesting that the presence of lower respiratory tract symptoms may have reduced the threshold for use of antibiotics. When unwarranted, over-prescription of antibiotics may lead to unwanted adverse effects and the development of antimicrobial resistance [11].
Patients with hMPV infection were actually less immunosuppressed than were those experiencing influenza. Given that wheezing predominated, our findings suggest that viral-mediated inflammation may underpin the clinical symptoms associated with hMPV infection. Indeed, hMPV infection has been associated with airway hyperresponsiveness in both animal models [12] and humans [13]. The incidence of asthma has increased among HIV-infected children and young adults who receive HAART, and it has been speculated that this may be driven by immune reconstitution, because asthma has been associated with higher CD4+ cell counts [14]. Indeed, a large proportion of the patients with hMPV infection in our study were asthmatic. Adult asthmatics have been shown to display exaggerated interferon γ responses to hMPV and RSV [15]. Thus, the potential link between hMPV and asthma in individuals with HIV infection warrants further exploration. Because all patients with hMPV infection had also received vaccination against influenza, a vaccine that is arguably more effective in individuals with higher CD4+ cell counts, the higher frequency of hMPV could also have been attributable to competing risks of viral infection other than influenza, once it has been effectively prevented.
Our study focused on a relatively small number of outpatients with fever and did not include all patients with respiratory symptoms; therefore, we cannot provide a measure of incidence for the various respiratory infections. We also cannot exclude that we may have missed more-severe viral or bacterial infections, particularly in individuals with pneumonia who presented directly to the hospital. Thus, we were unable to determine the etiologic role of respiratory viruses in more-severe disease. Finally, the lack of HIV-uninfected and asymptomatic control groups limits our ability to determine how the detection rates of respiratory pathogens in our study sample differed from those in the general population.
Although respiratory viral illnesses may be mild in nature, they account for a large proportion of respiratory tract infections among HIV-infected outpatients. Rapid multiplex PCR testing to identify specific viral pathogens could aid in the management of such cases (eg, by guiding specific influenza treatments) and reduce antibiotic exposure that may contribute to the development of antimicrobial resistance. There is clearly a need to develop effective antiviral therapies and vaccines of which HIV-infected persons would be the primary beneficiaries.
Acknowledgments
We thank the patients of the Immunodeficiency Service at the Montreal Chest Institute and their physicians, Drs James Allan, Norbert Gilmore, Richard Lalonde, Roger Leblanc, John MacLeod, Martin Potter, Pierre Rene, and Jean-Pierre Routy, without whom this study would not have been possible; Rosa Morales and Sahar Saeed, for study coordination and for their help in preparing the manuscript; Sylvie Deslandes, for excellent technical assistance; and AutoGenomics, for providing reagents necessary to conduct this study.
Footnotes
Potential conflict of interest: none reported.
Presented in part: 16th Conference on Retroviruses and Opportunistic Infections, Canada, February 2009 (abstract 797).
Financial support: Canadian Foundation for AIDS Research (CANFAR) and infrastructure funding from Le Fonds de la recherche en santé du Québec (FRSQ). M.K. is the recipient of a “checheur-boursier clincien” salary award from the FRSQ. G.B. is the recipient of a Canada Research Chair on Emerging Viruses and Antiviral Resistance from the Canadian Institutes of Health Research.
References
- 1.Beck JM, Rosen MJ, Peavy HH. Pulmonary complications of HIV infection: report of the Fourth NHLBI Workshop. Am J Respir Crit Care Med. 2001;164:2120–26. doi: 10.1164/ajrccm.164.11.2102047. [DOI] [PubMed] [Google Scholar]
- 2.Conley LJ, Bush TJ, Buchbinder SP, Penley KA, Judson FN, Holmberg SD. The association between cigarette smoking and selected HIV-related medical conditions. AIDS. 1996;10:1121–6. [PubMed] [Google Scholar]
- 3.Sloots TP, Whiley DM, Lambert SB, Nissen MD. Emerging respiratory agents: new viruses for old diseases? J Clin Virol. 2008;42:233–43. doi: 10.1016/j.jcv.2008.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Boivin G, Abed Y, Pelletier G, et al. Virological features and clinical manifestations associated with human metapneumovirus: a new paramyxovirus responsible for acute respiratory-tract infections in all age groups. J Infect Dis. 2002;186:1330–4. doi: 10.1086/344319. [DOI] [PubMed] [Google Scholar]
- 5.Madhi SA, Ludewick H, Abed Y, Klugman KP, Boivin G. Human metapneumovirus-associated lower respiratory tract infections among hospitalized human immunodeficiency virus type 1 (HIV-1)-infected and HIV-1-uninfected African infants. Clin Infect Dis. 2003;37:1705–10. doi: 10.1086/379771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Murphy D, Rose RC., 3rd Respiratory syncytial virus pneumonia in a human immunodeficiency virus-infected man. JAMA. 1989;261:1147. doi: 10.1001/jama.1989.03420080061014. [DOI] [PubMed] [Google Scholar]
- 7.Klein MB, Lu Y, DelBalso L, Cote S, Boivin G. Influenzavirus infection is a primary cause of febrile respiratory illness in HIV-infected adults, despite vaccination. Clin Infect Dis. 2007;45:234–40. doi: 10.1086/518986. [DOI] [PubMed] [Google Scholar]
- 8.Boivin G, Cote S, Dery P, De Serres G, Bergeron MG. Multiplex realtime PCR assay for detection of influenza and human respiratory syncytial viruses. J Clin Microbiol. 2004;42:45–51. doi: 10.1128/JCM.42.1.45-51.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Raymond F, Carbonneau J, Boucher N, et al. Comparison of automated microarray detection with real-time PCR assays for detection of respiratory viruses in specimens obtained from children. J Clin Microbiol. 2009;47:743–50. doi: 10.1128/JCM.01297-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Garbino J, Inoubli S, Mossdorf E, et al. Respiratory viruses in HIVinfected patients with suspected respiratory opportunistic infection. AIDS. 2008;22:701–5. doi: 10.1097/QAD.0b013e3282f470ac. [DOI] [PubMed] [Google Scholar]
- 11.Goossens H, Ferech M, Vander Stichele R, Elseviers M. Outpatient antibiotic use in Europe and association with resistance: a cross-national database study. Lancet. 2005;365:579–87. doi: 10.1016/S0140-6736(05)17907-0. [DOI] [PubMed] [Google Scholar]
- 12.Hamelin ME, Prince GA, Gomez AM, Kinkead R, Boivin G. Human metapneumovirus infection induces long-term pulmonary inflammation associated with airway obstruction and hyperresponsiveness in mice. J Infect Dis. 2006;193:1634–42. doi: 10.1086/504262. [DOI] [PubMed] [Google Scholar]
- 13.Jartti T, van den Hoogen B, Garofalo RP, Osterhaus AD, Ruuskanen O. Metapneumovirus and acute wheezing in children. Lancet. 2002;360:1393–4. doi: 10.1016/S0140-6736(02)11391-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Foster SB, Paul ME, Kozinetz CA, Macias CG, Shearer WT. Prevalence of asthma in children and young adults with HIV infection. J Allergy Clin Immunol. 2007;119:750–2. doi: 10.1016/j.jaci.2007.01.002. [DOI] [PubMed] [Google Scholar]
- 15.Douville RN, Bastien N, Li Y, Simons FE, HayGlass KT. Adult asthmatics display exaggerated IFNgamma responses to human metapneumovirus and respiratory syncytial virus. Biochem Cell Biol. 2007;85:252–8. doi: 10.1139/O07-005. [DOI] [PubMed] [Google Scholar]