Abstract
This report demonstrates that a rapid decrease of peripheral T cell subsets is a unique characteristic in patients with SARS during acute infection, although total white blood cell counts, red blood cell counts, and platelet counts remain relatively normal. In recovering patients, a rapid and dramatic restoration of peripheral T cell subsets was seen in the periphery. Although the underlying mechanism of the acute decrease of peripheral T cell subsets observed in patients with SARS during the acute stage remains unknown, this clinical characteristic can facilitate an earlier and more accurate diagnosis of SARS.
Severe acute respiratory syndrome (SARS) is a serious respiratory illness caused by a new member of the coronavirus family [1–3]. Since emerging in China's Guangdong province in November 2002, SARS has spread to at least 28 countries and regions in Asia, Europe, and North America, infecting >8000 people and killing >800 (as of 11 July 2003). The incubation period of SARS is usually 2–10 days, and the clinical manifestation of SARS is characterized by a high fever (temperature >38°C); chills; rigors; myalgias; dry, nonproductive cough; and dizziness. At the onset of fever, the majority of patients demonstrate air-space consolidation, with either unilateral or bilateral involvement [4–9]. In patients who experience clinical deterioration, serial chest radiographs show progression of pulmonary infiltrates ∼7–10 days after presentation [4–10]. The fatality rate seems to vary in different age groups and is currently estimated to be 13.2% for patients <60 years of age and 43.3% for patients ⩾60 of age [11].
Through the collaborative effort of scientists worldwide, exceedingly rapid progress has been made in the identification and sequencing of SARS's etiologic agent, a novel coronavirus that is genetically and serologically distinct from known strains of the same family [1–3, 12–14]. Recent studies of cynomolgus monkeys inoculated with this new coronavirus have revealed symptoms and histopathological findings similar to those in humans with SARS [15], providing further evidence that the coronavirus in question is the causative agent of SARS. In addition, the genetic characterizations of multiple strains that have been identified in various regions around the world seem to suggest that this novel coronavirus does not mutate as quickly as HIV or hepatitis C virus, raising optimism about the development of therapies and vaccines in the future. However, the underlying mechanism by which this new coronavirus causes SARS remains unknown.
Subjects, materials, and methods. Between March and April 2003, we enrolled 98 patients who were in the acute stage of SARS and 25 patients who had recovered from SARS, according toWorld Health Organization criteria (http://www.who.int/csr/sars/casedefinition/en), at the Department of Infectious Diseases, Peking Union Medical College Hospital, Beijing. Patients were treated at the hospital under the guidelines of the ethical review committee. Patients who were merely suspected of having SARS or who were deemed to probably have SARS were not included in the study. Patients in the acute stage of SARS were defined as those in the first week of the illness; recovered patients were defined as those free from the illness after ∼1 month of recovery. (Most patients with SARS take ∼1 month to recover, with the resolution of all symptoms observed during the acute phase. Once recovered, patients regain a normal body temperature, experience no cough or chest pain, and have normal chest radiograph findings.) The average age (±SD) of the patients in the acute stage of SARS was 40 ± 16, whereas that of patients who had recovered from SARS was 41 ± 15. All of these individuals, it was confirmed, had come into close contact with people with SARS. The patients' temperatures were between 38°C and 40°C at the time of diagnosis. Fifty-seven percent of the patients had developed dry cough or light-white sputum, 36% had shortness of breath, and 62% had hypoxia. Chest radiographs demonstrated air-space consolidation, with unilateral involvement in 64% of patients and bilateral involvement in 29% of patients. Most of the patients had relatively normal liver and renal functions. In addition, all of the participating patients were antibody and antigen negative for HIV- 1, cytomegalovirus (CMV), and Epstein-Barr virus (EBV). None of the patients received any kind of treatment before blood samples were collected.
For purposes of comparison, blood samples were also obtained from 32 HIV-1-positive patients, 11 CMV-positive patients, and 6 EBV-positive patients. All of these patients were singly infected with their respective viruses and were in the acute (or early) stage of infection. The HIV-1-positive individuals selected for the study were asymptomatic and were not receiving any antiretroviral treatment. Their CD4 T cell counts were >400 cells/mL of blood at the time of diagnosis. The acute status was determined by an incomplete or evolving pattern in serum antibody reaction against HIV-1 antigens (bioMérieux and Genelabs), as described elsewhere [16, 17]. Acute CMV infections were identified using an indirect immunofluorescence assay that detected pp65 CMV matrix protein, and acute EBV infections were detected by anti- EBV IgM in serum using a commercial detection kit. Additional blood samples were obtained from 56 healthy individuals who were on the hospital staff or who were blood donors.
Peripheral blood CD3, CD4, and CD8 T cell; B cell; and NK cell counts were measured directly from 100 µL of whole blood [16, 18, 19]. Cocktails of monoclonal antibodies (MAbs) (Beckman- Coulter) specific for CD3, CD4, and CD8 cells were used. For B cells, a mouse anti-human CD19 MAb was used, whereas for NK cells, a mouse anti-human CD16 and CD56 MAb was used. The same isotype control MAb was used. All of these antibodies were purchased from Immunotech. After incubation for 20 min at room temperature, the red blood cells were lysed by the addition of 500 µL of Optilyse C (Beckman-Coulter), followed by incubation for 8 min at room temperature. After being washed thoroughly with PBS, antibody-stained cells were analyzed by use of flow cytometry (EPICS-XL flow cytometer; Beckman-Coulter).
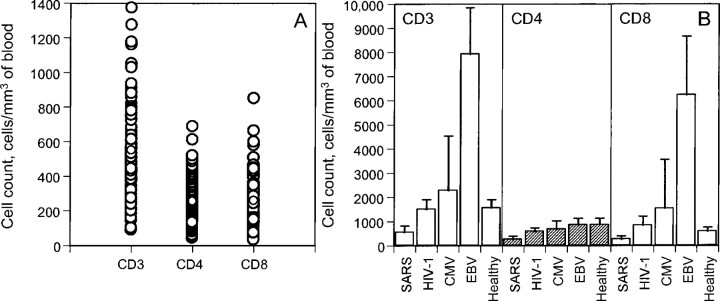
Results. Using flow cytometry, we compared the absolute numbers and percentages of CD3, CD4, and CD8 T cells in samples from the 98 patients in the acute stage of SARS with the T cell counts of healthy individuals, as well as with the counts of those infected singly by HIV-1, CMV, or EBV. As shown in figure 1, patients with SARS clearly experienced T cell loss in peripheral blood during the acute stage of infection. The mean absolute CD3, CD4, and CD8 T cell counts in healthy individuals was 1555, 858, and 584 cells/mm3, respectively, whereas those in patients with SARS were markedly lower, at 549, 254, and 258 cells/mm3, respectively (P < .0001) (figure 1). The ratio of CD4 cells to CD8 cells among patients in the acute stage of SARS was 0.98. Among the 98 patients in the acute stage of SARS, 38 had CD4 counts <200 cells/mm3. In some patients with SARS, absolute CD3 T cell counts were as low as 89 cells/mm3, and CD4 and CD8 counts were as low as 43 and 39 cells/mm3, respectively (figure 1A).
Figure 1.
Comparison of peripheral changes in CD3, CD4, and CD8 T cell counts in patients who have severe acute respiratory syndrome (SARS) (A) and among healthy individuals and those infected with the SARS virus, HIV-1, cytomegalovirus (CMV), or Epstein-Barr virus (EBV) (B).
Marked differences have also been observed in T cell subsets between patients in the early stage of SARS and individuals infected with HIV-1, CMV, or EBV. In contrast to the severe loss of T cell subsets found in patients with SARS, patients infected with HIV, CMV, or EBV undergo proliferative responses, which result in higher T cell percentages and absolute counts (figure 1B) than are seen in healthy individuals. InCMVand EBV-infected patients in particular, increases in absolute CD3 and CD8 T cell counts were more profound than those in HIV-1 infected patients (figure 1B) [20–24]. For instance, the mean absolute CD3 and CD8 T cell counts in patients infected with EBV were 7923 and 6224 cells/mm3, respectively, which were 5–10-fold higher than those in healthy individuals and 13–23-fold higher than those in patients with SARS. The absolute CD4 T cell counts in patients with HIV-1, CMV, or EBV infection, however, decreased modestly during the acute phase of infection (data not shown).
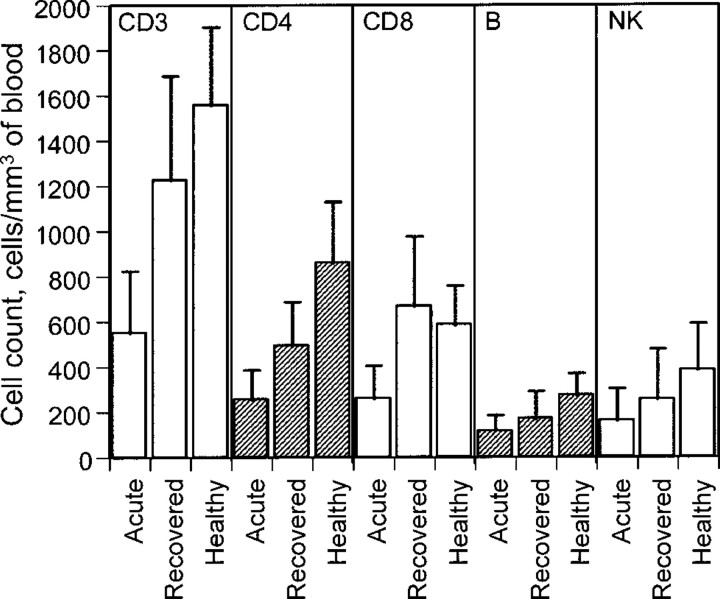
A comparison of lymphocyte subsets was also made between patients in the acute stage of SARS and those who had recovered from the illness. As shown in figure 2, in the 25 subjects studied, there was a rapid and significant restoration of CD3, CD4, and CD8 T cell; B cell; and NK cell counts ∼21 days after the onset of illness. Absolute cell counts almost doubled for all of the cell subsets studied (figure 2), although the percentage did not change significantly. The trend in the normalization of peripheral lymphocyte counts usually coincided with the improving clinical condition of patients with SARS.
Figure 2.
Comparison of peripheral changes in CD3, CD4, and CD8 T cell; B cell; and NK cell counts in patients with severe acute respiratory syndrome during acute infection, in patients who have recovered from the illness, and in healthy individuals.
Discussion. In this report, we describe a systematic, crosssectional study of the changes of peripheral lymphocyte subsets in patients with SARS and compare the changes with those in patients infected with HIV-1, CMV, or EBV. Our results demonstrate for the first time that there is rapid and generalized lymphopenia in patients with SARS during the acute phase of infection, which is in distinct contrast to the proliferative responses seen in HIV-1-, CMV-, or EBV-infected patients. In patients who recovered from the initial SARS illness, an equally rapid and dramatic restoration of CD3, CD4, and CD8 T cell; B cell; and NK cell counts was seen in the peripheral blood.
It is intriguing to see the human immune system respond in a drastically different manner to different viral infections. Whereas HIV-1, CMV, and EBV infections lead to proliferative lymphocyte responses, other viruses—including the SARS coronavirus, the measles virus [25], avian influenza virus H5N1 [26], swine foot-and-mouth disease virus [27], and respiratory syncytial virus [28]—are associated with generalized lymphopenia. It is believed that in measles infection, the acute lymphopenia is likely due to the apoptosis of uninfected cells [25], whereas in H5N1, swine foot-and-mouth disease virus, and respiratory syncytial virus infection, the underlying mechanism is much less clear [26–28]. Currently, we know little about how the SARS coronavirus precipitates such severe lymphopenia in such a short period of time. It is possible that tissue sequestration, cell destruction in peripheral blood, or bone marrow failure singly or together play a critical role. However, given the rapid restoration of lymphocytes in peripheral blood in recovered patients, it seems that tissue sequestration may play the most important role determining lymphocyte count in peripheral blood in patients with SARS. The exact mechanism of lymphocyte sequestration, however, remains unclear.We can only speculate, on the basis of the clinical syndrome of patients with SARS, that lymphocyte infiltration and sequestration are likely occurring in the lungs, gastrointestinal tract, or lymphoid tissues. The matter of finding concrete evidence that would support this hypothesis, and indeed of gaining an understanding of the underlying mechanism behind SARS virus-induced lung and gastrointestinal sequelae, awaits resolution in future studies.
References
- 1. Peiris JS, Lai ST, Poon LL, et al. Coronavirus as a possible cause of severe acute respiratory syndrome. Lancet. 2003361:1319–25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Drosten C, Gunther S, Preiser W, et al. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N Engl J Med. 2003348:1967–76 [DOI] [PubMed] [Google Scholar]
- 3. Ksiazek TG, Erdman D, Goldsmith CS, et al. A novel coronavirus associated with severe acute respiratory syndrome. N Engl J Med. 2003348:1953–66 [DOI] [PubMed] [Google Scholar]
- 4. Tsang KW, Ho PL, Ooi GC, et al. A cluster of cases of severe acute respiratory syndrome in Hong Kong. N Engl J Med. 2003348:1977–85 [DOI] [PubMed] [Google Scholar]
- 5. Peiris J, Chu CM, Cheng V, et al. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: a prospective study. Lancet. 2003361:1767–72 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Lee N, Hui D, Wu A, et al. A major outbreak of severe acute respiratory syndrome in Hong Kong. N Engl J Med. 2003348:1986–94 [DOI] [PubMed] [Google Scholar]
- 7. Poutanen SM, Low DE, Henry B, et al. Identification of severe acute respiratory syndrome in Canada. N Engl J Med. 2003348:1995–2005 [DOI] [PubMed] [Google Scholar]
- 8. Booth CM, Matukas LM, Tomlinson GA, et al. Clinical features and short-term outcomes of 144 patients with SARS in the greater Toronto area. JAMA. 2003289:2801–9 [DOI] [PubMed] [Google Scholar]
- 9. Nicholls JM, Poon LLM, Lee KC, et al. Lung pathology of fatal severe acute respiratory syndrome. Lancet. 2003361:1773–8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Wong KT, Antonio GE, Hui DS, et al. Severe acute respiratory syndrome: radiographic appearances and pattern of progression in 138 patients. Radiology. 2003228:401–6 [DOI] [PubMed] [Google Scholar]
- 11. Donnelly CA, Ghani AC, Leung GM, et al. Epidemiological determinants of spread of causal agent of severe acute respiratory syndrome in Hong Kong. Lancet. 2003361:1761–6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Holmes KV. SARS-associated coronavirus. N Engl J Med. 2003348:1948–51 [DOI] [PubMed] [Google Scholar]
- 13. Rota PA, Oberste MS, Monroe SS, et al. Characterization of a novel coronavirus associated with severe acute respiratory syndrome. Science. 2003300:1394–9 [DOI] [PubMed] [Google Scholar]
- 14. Marra MA, Jones SJ, Astell CR, et al. The genome sequence of the SARS-associated coronavirus. Science. 2003300:1399–404 [DOI] [PubMed] [Google Scholar]
- 15. Fouchier RA, Kuiken T, Schutten M, et al. Aetiology: Koch's postulates fulfilled for SARS virus. Nature. 2003423:240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Li TS, Tubiana R, Fillet AM, Autran B, Katlama C. Negative result of cytomegalovirus blood culture with restoration of CD4+ T cell reactivity to cytomegalovirus after HAART in an HIV-1-infected patient. J Acquir Immune Defic Syndr Hum Retrovirol. 199920:514–5 [DOI] [PubMed] [Google Scholar]
- 17. Anand K, Ziebuhr J, Wadhwani P, Mesters JR, Hilgenfeld R. Coronavirus main proteinase (3CLpro) structure: basis for design of anti- SARS drugs. Science. 2003300:1763–7 [DOI] [PubMed] [Google Scholar]
- 18. Autran B, Carcelain G, Li TS, et al. Positive effects of combined antiretroviral therapy on CD4+ T cell homeostasis and function in advanced HIV disease. Science. 1997277:112–6 [DOI] [PubMed] [Google Scholar]
- 19. Li TS, Tubiana R, Katlama C, Calvez V, Ait Mohand H, Autran B. Long-lasting recovery in CD4 T-cell function and viral-load reduction after highly active antiretroviral therapy in advanced HIV-1 disease. Lancet. 1998351:1682–6 [DOI] [PubMed] [Google Scholar]
- 20. Looney RJ, Falsey A, Campbell D, et al. Role of cytomegalovirus in the T cell changes seen in elderly individuals. Clin Immunol. 199990:213–9 [DOI] [PubMed] [Google Scholar]
- 21. Kuijpers TW, Vossen MT, Gent MR, et al. Frequencies of circulating cytolytic/ CD45RA+CD27−/ CD8+ T lymphocytes depend on infection with CMV. J Immunol. 2003170:4342–8 [DOI] [PubMed] [Google Scholar]
- 22. Gratama JW, Kardol M, Naipal AM, et al. The influence of cytomegalovirus carrier status on lymphocyte subsets and natural immunity. Clin Exp Immunol. 198769:16–24 [PMC free article] [PubMed] [Google Scholar]
- 23. Callan MF. The evolution of antigen-specific CD8+ T cell responses after natural primary infection of humans with Epstein-Barr virus. Viral Immunol. 200316:3–16 [DOI] [PubMed] [Google Scholar]
- 24. Khanna R, Burrows SR, Moss DJ. Immune regulation in Epstein-Barr virus-associated diseases. Microbiol Rev. 199559:387–405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Okada H, Kobune F, Sato TA, et al. Extensive lymphopenia due to apoptosis of uninfected lymphocytes in acute measles patients. Arch Virol. 2000145:905–20 [DOI] [PubMed] [Google Scholar]
- 26. Zitzow LA, Rowe T, Morken T, Shieh WJ, Zaki S, Katz JM. Pathogenesis of avian influenza A (H5N1) viruses in ferrets. J Virol. 200276:4420–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Bautista EM, Ferman GS, Golde WT. Induction of lymphopenia and inhibition of T cell function during acute infection of swine with foot and mouth disease virus (FMDV). Vet Immunol Immunopathol. 200392:61–73 [DOI] [PubMed] [Google Scholar]
- 28. O'Donnell DR, Carrington D. Peripheral blood lymphopenia and neutrophilia in children with severe respiratory syncytial virus disease. Pediatr Pulmonol. 200234:128–30 [DOI] [PubMed] [Google Scholar]