Abstract
Objectives
Filoviruses such as Ebola virus and Marburg virus cause a severe haemorrhagic fever syndrome in humans for which there is no specific treatment. Since filoviruses use a complex route of cell entry that depends on numerous cellular factors, we hypothesized that there may be drugs already approved for human use for other indications that interfere with signal transduction or other cellular processes required for their entry and hence have anti-filoviral properties.
Methods
We used authentic filoviruses and lentiviral particles pseudotyped with filoviral glycoproteins to identify and characterize such compounds.
Results
We discovered that amiodarone, a multi-ion channel inhibitor and adrenoceptor antagonist, is a potent inhibitor of filovirus cell entry at concentrations that are routinely reached in human serum during anti-arrhythmic therapy. A similar effect was observed with the amiodarone-related agent dronedarone and the L-type calcium channel blocker verapamil. Inhibition by amiodarone was concentration dependent and similarly affected pseudoviruses as well as authentic filoviruses. Inhibition of filovirus entry was observed with most but not all cell types tested and was accentuated by the pre-treatment of cells, indicating a host cell-directed mechanism of action. The New World arenavirus Guanarito was also inhibited by amiodarone while the Old World arenavirus Lassa and members of the Rhabdoviridae (vesicular stomatitis virus) and Bunyaviridae (Hantaan) families were largely resistant.
Conclusions
The ion channel blockers amiodarone, dronedarone and verapamil inhibit filoviral cell entry.
Keywords: haemorrhagic fever, cationic amphiphiles, calcium channels
Introduction
Members of the Filoviridae family, which includes Marburg virus (MARV) and several species of Ebola virus, are enveloped filament-shaped viruses with a non-segmented negative-sense RNA genome. Infection of humans causes a severe haemorrhagic fever syndrome.1 Most cases occur in resource-poor settings in sub-Saharan Africa, with case fatality rates in the range of 42%–90%.
Monocytes, macrophages and dendritic cells are thought to be sites of viral replication during the early and later stages of infection2,3 yet viral amplification also occurs in endothelial cells and many other tissues.3–5 Consistent with rapid spread to a broad range of cell types and tissues in humans, filoviruses have been found to readily enter into a broad range of cultured cell types, with the notable exception of lymphocytes.3,6 It has emerged over recent years that the filoviral route of cell entry is complex.7 Interaction between the target cell and the incoming virion is mediated on the viral side by heterotrimers of the filoviral glycoprotein (GP) subunits 1 (GP1) and 2 (GP2) that are generated by the furin-mediated cleavage of a single precursor GP (GP0) and remain linked by a disulfide bond.8,9 Numerous factors on the surface of host cells have been reported to interact with GP1/GP2. These include the folate receptor,10 members of the Tyro3 family of receptor tyrosine kinases (Axl, Dtk, Mer)11 and T cell immunoglobulin and mucin domain 1,12 although the relevance of the folate receptor for filovirus entry was later disputed.13,14 In addition, numerous C-type lectins, calcium-dependent carbohydrate-binding molecules, including the asialoglycoprotein receptor,15 DC-SIGN and DC-SIGNR,16,17 human macrophage galactose-type C-type lectin18 and liver and lymph node sinusoidal endothelial cell C-type lectin19 have been reported to interact with GP1 and promote filovirus entry.
Following host cell binding, filoviruses are taken up into the cell most likely through macropinocytosis.20,21 The low pH-dependent cathepsin B/L-mediated cleavage of filovirus GP is then required for fusion with a late endosomal membrane.22 In addition, the endosomal cholesterol transporter Niemann–Pick C1 (NPC1) acts as an essential intracellular receptor.23,24 This intricate multistep route of viral entry is likely to require some form of coordination, which may take advantage of cellular signalling, as has been shown for coxsackie group B viruses25 and hepatitis C virus.26 Indeed, the phosphoinositide-3 kinase/Akt pathway as well as protein kinase C and small cellular GTPases have been implicated in filovirus entry.27
Although (as discussed below) several experimental anti-filoviral approaches have been reported, no vaccine or specific antiviral treatment has yet been established. Given low case numbers and geographical restriction to resource-limited regions, industry-sustained drug development programmes are unlikely. Nonetheless, there is a clear unmet clinical need from naturally occurring cases as well as an additional potential need given that filoviruses are considered to be CDC category A bioterrorism agents (www.bt.cdc.gov/bioterrorism). Given the complex filoviral route of entry, we hypothesized that drugs which perturb cellular signalling networks and are approved for other indications might also interfere with filovirus entry. We performed a small-scale screen and found that amiodarone, a multi-ion channel inhibitor used clinically as an anti-arrhythmic agent, acts as a potent and specific inhibitor of filoviral cell entry. The concentrations of amiodarone that are required for filovirus inhibition are well within the range that is achieved in serum during anti-arrhythmic therapy in humans, i.e. 1.5–2.5 μg/mL.28
Materials and methods
Compounds
Pharmacological agents were purchased from Sanofi-Aventis (Frankfurt, Germany) (amiodarone), Sigma-Aldrich (Taufkirchen, Germany) [amiodarone, dronedarone, epigallocatechin-3-gallate (EGCG), ethosuximide, lidocaine, metoprolol], Merck Millipore (Darmstadt, Germany) [Z-Phe-Tyr[t-Bu]-diazomethylketone (FYdmk)], Toronto Research Chemicals (Toronto, Canada) (artesunate, cyclosporine A, erlotinib, fingolimod, imatinib, rapamycin, sorafenib, sotrastaurin, sunitinib), Abbott (Wiesbaden, Germany) (verapamil), Roche Pharma (Grenzach-Wyhlen, Germany) (bevacizumab), Janssen-Cilag (Neuss, Germany) (haloperidol) and Astellas Pharma (Munich, Germany) (theophylline).
Cell culture
All immortalized cell lines were cultured in Dulbecco's Modified Eagle Medium (DMEM) with 10% fetal calf serum (FCS), non-essential amino acids, l-glutamine and penicillin/streptomycin at 37°C and 5% CO2. Primary human umbilical vein endothelial cells were isolated from umbilical cords as previously described29 and cultivated in EGM-2 medium (Lonza, Basel, Switzerland) containing 2% FCS and penicillin/streptomycin. Primary human hepatocytes were isolated using a modified two-step collagenase (Roche P, Mannheim, Germany) perfusion technique as previously reported30 and cultured in Williams' medium E (all Biochrom AG, Berlin, Germany) supplemented with insulin (1 μM), dexamethasone (1 μM), penicillin/streptomycin, sodium pyruvate, HEPES buffer, l-glutamine and 5% FCS. The generation of macrophages from primary human monocytes has recently been described.31 Briefly, Ficoll gradient-purified human monocytes from peripheral blood mononuclear cells were cultured in RPMI 1640 medium with 7.5% human fibrin-depleted plasma and 1% penicillin/streptomycin for 24 h. Cells were then resuspended in X-Vivo 10 medium supplemented with 1% human fibrin-depleted plasma and 1% penicillin/streptomycin, seeded and allowed to differentiate for at least 7 days. The medium was changed every 2–3 days.
DNA constructs
The plasmids CSFlucW2 encoding firefly luciferase in the context of a gutted lentiviral genome, HIV gag-pol, pcDNA3.1 and vesicular stomatitis virus (VSV) G-protein (VSV-G) that are the basis for the production of lentiviral pseudoparticles have previously been described,31,32 as has the plasmid pCAGGS-MARVGP.33 The plasmids pVR1012-GP(Z), pVR1012-GP(R), pVR1012-GP(S) and pVR1012-GP(IC) encoding the GP of Zaire Ebola virus (EBOV) as well as Reston, Sudan and Ivory Coast (Tai Forrest) Ebola virus, respectively, were a kind gift from Drs Gary Nabel and Anthony Sanchez (NIH, Bethesda, USA). The plasmid pCMV-LassaGPC encoding the GP of Lassa virus was a kind gift from Dr Francois-Loic Cosset (Inserm U758, Lyon, France). The plasmid pWRG/HTN-M(x) encoding the GP of Hantaan virus was a kind gift from Dr Jay Hooper (USAMRIID, Fort Detrick, USA). The plasmid INH-95551 encoding the GP of the New World arenavirus Guanarito was a kind gift from Drs Hyeryun Choe (Harvard Medical School, Boston, USA) and Stefan Kunz (University of Lausanne, Switzerland).
Pseudoparticle production and transduction
Pseudoparticles were generated by co-transfection of plasmids encoding: (i) a gutted lentiviral genome containing the firefly luciferase gene (CSFlucW2); (ii) HIV gag-pol; and (iii) the desired envelope GP. The day before transfection, 8 × 105 293T cells were seeded in a 35 mm well. The next day, a total of 1 μg of DNA was transfected per well using polyethylenimine. The medium was replaced after 6 h. Supernatants were harvested at 48 h and 72 h after transfection, pooled and filtered (0.45 μm pore size), and then used immediately or stored at 4°C (<1 week) or −80°C (>1 week). Pseudoparticle transduction assays were performed in a 12-well format using 8 × 104 EAhy cells per well. Seeding numbers were adjusted for other cell lines. Transductions were carried out with polybrene (4 μg/mL) and with or without drugs. After 6 h, the medium was exchanged. After 72 h, cells were lysed and luciferase was measured as previously described.34 To separate the virus from the drug, we used an Amicon Ultra-15 centrifugal device with a 100 kDa cut-off (Millipore). In this device, pre-treated pseudovirus was washed with 10 mL of drug-free media three times by centrifugation at 3000 rpm.
Authentic filovirus production and infection
All work with the infectious Mayinga strain of EBOV (GenBank accession number NC002549) was performed under the highest safety precautions in the biosafety level 4 (BSL-4) facility at the Institute of Virology, Philipps-University Marburg, Germany.
Vero E6 cells were infected with EBOV at a multiplicity of infection (moi) of 1 TCID50/cell.35 EBOV particles were purified from the supernatant of infected cells at 72 h post-infection by centrifugation over a sucrose cushion for 90 min at 30 000 rpm, 4°C in a Beckman ultracentrifuge using a SW32 rotor (Beckman Coulter, Palo Alto, CA, USA). Pellets were resuspended in PBS. Amiodarone was mixed with concentrated EBOV particles with an moi of 1.5 TCID50/cell and transferred to EAhy cells. After 1 h of incubation, the inoculum was removed and replaced by fresh DMEM supplemented with 3% FCS. At 20 h post-infection, cells were fixed in 4% paraformaldehyde for 48 h, removed from the BSL-4 laboratory and subjected to quantitative immunofluorescence analysis. Immunostaining of infected cells was performed as described below.
Immunofluorescence staining
Immunofluorescence analyses were carried out as previously described.33 For the detection of nucleocapsid (NP)-induced inclusion bodies in EBOV-infected EAhy cells (after fixation of the cells with 4% paraformaldehyde in DMEM for 48 h), we used a monoclonal anti-EBOV NP antibody and a secondary FITC-coupled goat anti-mouse antibody.
Statistical analysis
Numerical comparisons between groups were made by unpaired two-sided Student's t-test. Differences with P values below 0.05 are referred to as significant. P < 0.05 and P < 0.01 are indicated by single and double asterisks throughout. GraphPad Prism software (La Jolla, CA, USA) was used to fit concentration–response curves and determine the 50% inhibitory concentration (IC50).
Results
Identification of amiodarone as an inhibitor of filovirus GP-driven cell entry
Based on a literature review, we selected 15 drugs for the initial screen. Drugs were chosen that met several of the following selection criteria: (i) a known safety profile in humans; (ii) known to perturb cell signalling processes; (iii) recognized antiviral properties against viruses other than members of the Filoviridae;36–41 and (iv) coverage of a broad range of indications and drug classes. Of these 15 drugs, 14 are approved by the EMA and FDA for various indications; one drug, the protein kinase C inhibitor sotrastaurin, is in advanced clinical development as an immunosuppressive agent for use in organ transplant recipients. Human endothelium-derived EAhy cells were transduced with pseudoparticles bearing either EBOV GP or MARV GP in the presence or absence of drugs. Drug concentrations tested were based on serum levels achievable in humans. Four concentrations were tested per drug and two rounds of screening were performed. The cathepsin B and L inhibitor FYdmk, known to potently inhibit EBOV GP-mediated cell entry, was included as a positive control (Figure 1). While most drugs had no major effect on filovirus GP-mediated cell entry, amiodarone was found to reduce the detectable signal by over 99%, and thus more strongly than FYdmk, which achieved 86%–98% inhibition.
Figure 1.
Amiodarone markedly reduces filoviral GP-mediated cell entry. EAhy cells were transduced with pseudoparticles bearing EBOV or MARV GP in the presence of the drugs indicated. Out of four concentrations tested, the highest concentration without apparent signs of toxicity as judged by cell morphology is shown. The first and second numbers in the brackets indicate the drug concentration used in μg/mL and in μM, respectively. The cathepsin inhibitor FYdmk, known to inhibit filovirus GP-mediated cell entry, was included as a positive control. Columns represent the means of n = 2 independent measurements. #, since bevacizumab is an antibody, its concentration is given only in μg/mL.
Concentration–response relationship of amiodarone inhibition of filoviral cell entry
Amiodarone-mediated inhibition of EBOV and MARV GP-bearing pseudoparticles showed an IC50 of 1.38 μg/mL (2.02 μM) and 1.18 μg/mL (1.73 μM), respectively (Figure 2a). We then exposed EAhy cells stably expressing firefly luciferase to different concentrations of amiodarone to test for cytotoxic and anti-proliferative effects. A 50% signal reduction (CC50) was seen at 16.69 μg/mL (25.9 μM) (Figure 2b). This corresponds to a calculated therapeutic index of 12 and 14 for EBOV and MARV, respectively. In a ‘worst case’ scenario (assuming that the true IC50 is 1 × SD above and CC50 1 × SD below our determination), the index is still >5 for both EBOV and MARV.
Figure 2.
Amiodarone toxicity and concentration–response curves. (a) EAhy cells were transduced with pseudoparticles bearing EBOV (upper panel) or MARV (lower panel) GP in the presence of increasing concentrations of amiodarone. Individual data points represent the mean ± SD of n = 3 independent replicates from a representative of three independent experiments. The IC50 ± SD is indicated. The x-axis indicating the amiodarone concentration has a log scale. (b) EAhy cells stably expressing firefly luciferase were exposed to the indicated concentrations of amiodarone for 6 h. Then drug-containing medium was replaced with fresh medium without the drug. After 72 h, the cells were lysed, luciferase activity was measured and the CC50 was determined. Individual data points represent the mean ± SD of n = 3 independent replicates from a representative of three independent experiments.
Amiodarone inhibition of authentic Ebola virus
To assess the effect of amiodarone on authentic filovirus entry, EAhy cells were exposed to purified EBOV in the presence of different concentrations of amiodarone for 1 h. The medium was exchanged, and 20 h later cells were fixed with paraformaldehyde and stained for EBOV NP protein. DAPI was used as a counterstain. There was a clear concentration-dependent reduction in the percentage of NP-positive cells while the overall cell number was unchanged (Supplementary Data). Significant inhibition was observed at amiodarone concentrations of 0.5 μg/mL or higher (Figure 3). On averaging all the experiments performed, the IC50 was 0.25 μg/mL, corresponding to a calculated therapeutic index of above 50. Of note, the apparently flatter slope of the concentration response curve is most likely due to the fact that experiments performed on separate occasions were averaged. Thus, authentic filoviruses may be more sensitive to amiodarone inhibition than pseudoparticles bearing filoviral GP.
Figure 3.
Amiodarone inhibition of authentic EBOV infection. EAhy cells were exposed to purified Zaire Ebola virus in the presence or absence of amiodarone at the indicated concentrations. After 20 h cells were fixed and stained for Ebola NP protein. The percentage of cells infected with authentic EBOV in the absence of drug was set to 100%. Means ± SD from n = 3 independent experiments are shown. Significant inhibition is indicated by asterisks (*P < 0.05, **P < 0.01).
Amiodarone inhibition of cell entry mediated by other viral GPs
Next we aimed to assess whether amiodarone inhibits entry driven by the GPs of all filovirus species and whether entry mediated by the GPs from other viral families may be affected. Pseudoparticles bearing GP of the Ebola virus species Tai Forest, Reston, Sudan and Zaire, as well as MARV, were similarly inhibited by amiodarone (Figure 4a). Thus, amiodarone is a blocker of cell entry mediated by viral GPs from across the Filoviridae family. Among a broader range of human pathogenic enveloped viruses that enter target cells through endocytosis and in a pH-dependent manner, we observed a significant inhibition of pseudoviruses bearing the GP of the New World arenavirus Guanarito (Figure 4b). VSV-G- (Rhabdoviridae family), Lassa virus GP- (Old World arenavirus) and Hantaan (Bunyaviridae family) virus GP-pseudotyped lentiviruses were affected to a lesser extent. Since all pseudoviruses have the same lentiviral NP and reporter genome, these differences strongly support the fact that amiodarone exerts its inhibitory effect on viral cell entry, i.e. a replication cycle step up to the point of fusion of the viral envelope with a cellular membrane.
Figure 4.
Effect of amiodarone on diverse viral GPs. (a) EAhy cells were transduced with pseudoparticles bearing GPs of different filovirus species or VSV-G as indicated in the figure. Individual data points represent the mean of n = 3 independent replicates from a representative of three independent experiments. (b) EAhy cells were transduced with pseudoparticles bearing GPs from the indicated viruses in the presence or absence of amiodarone. Means ± SD of n = 3 independent replicates from a representative of three independent experiments are shown. Significant inhibition is indicated by asterisks (*P < 0.05, **P < 0.01).
Effects of amiodarone pre-treatment of target cells
We next tested the effect of target cell pre-treatment. Amiodarone was present either 1 h before the cells were washed three times and then EBOV or MARV pseudoparticles were added to the cells, or during the 6 h when the cells were exposed to the pseudoparticles, or both (Figure 5a). We observed the most marked inhibition when the drug was present throughout the experiment, i.e. from 1 h prior to addition of pseudoviruses until 6 h after, followed by the set-up where amiodarone was present only during the cell entry phase. In the case of MARV, there was also significant inhibition when the drug was present only before the pseudoparticles were added to the cells. This is suggestive of a cell-directed mechanism of action. However, the effect on the target cells appears to be reversible upon removal of the drug. Moreover, when EBOV pseudovirus was incubated with a high concentration (4.5 μg/mL) of amiodarone and then washed in 100 kDa filter devices in order to remove drug, we observed a subsignificant 52% reduction of infectivity relative to VSV-G pseudoviruses (Supplementary Data). Next we assessed whether EAhy cells could be adapted to amiodarone by growing them in the presence of 1.5 μg/mL amiodarone for 2 weeks prior to experimentation. Cell growth was largely unaltered and there were no obvious morphological alterations. We observed a comparable degree of inhibition of EBOV and MARV pseudoparticles, suggesting that prolonged exposure to amiodarone does not lead to compensatory changes in the host cell (Figure 5b).
Figure 5.
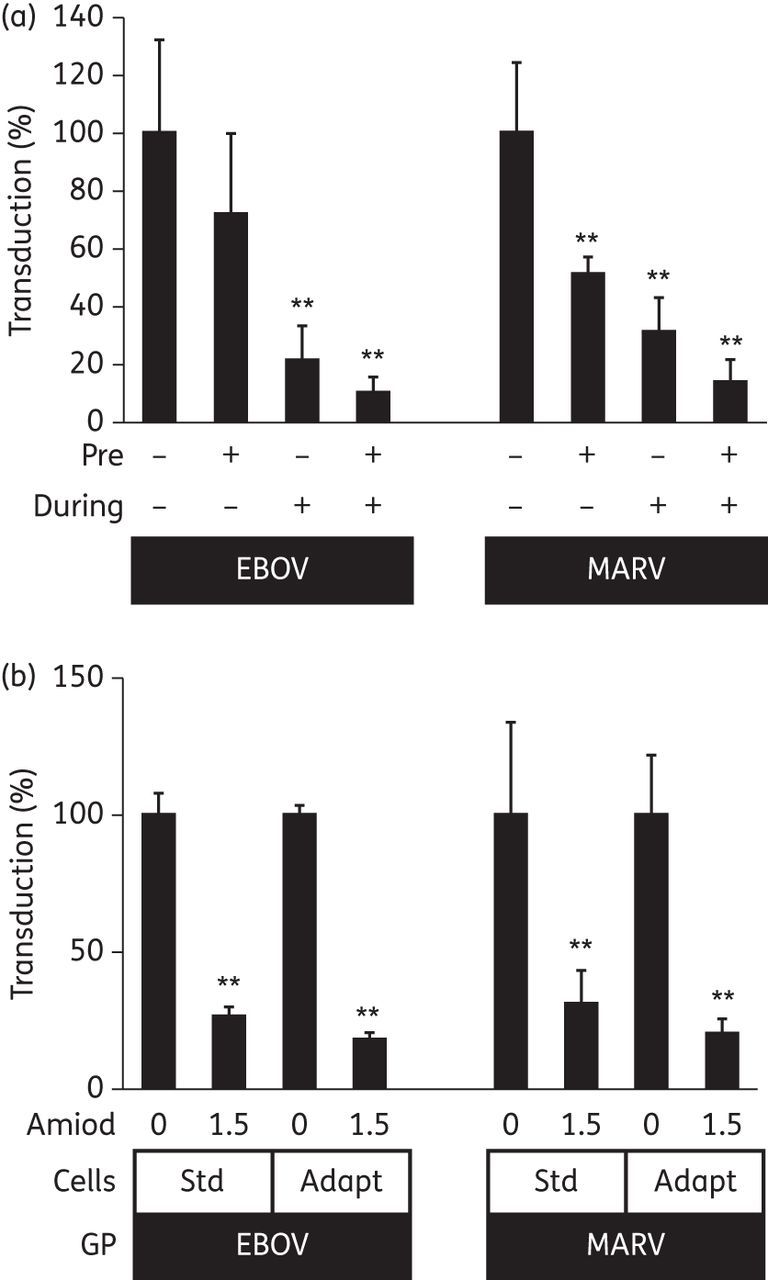
Effect of target cell pre-treatment. (a) EAhy cells were transduced with pseudoparticles bearing EBOV or MARV GPs. Amiodarone (1.5 μg/mL) was present in the cell culture medium 1 h before (Pre) and/or during (During) the 6 h of infection as indicated. Means ± SD of n = 6 independent replicates from a representative of two independent experiments are shown. (b) Standard EAhy cells (Std) or EAhy cells that had previously been cultured in the presence of amiodarone (1.5 μg/mL) (Adapt) were transduced with pseudoparticles bearing EBOV or MARV GPs in the presence or absence of amiodarone (1.5 μg/mL). Means ± SD of n = 4 independent replicates from a representative of two independent experiments are shown. Significant inhibition is indicated by asterisks (*P < 0.05, **P < 0.01).
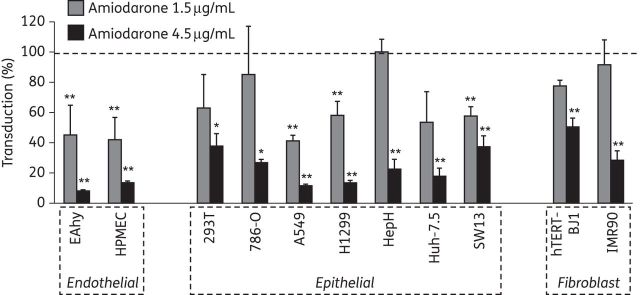
Effect of amiodarone on filoviral entry into different cell lines and primary cells
We next evaluated the amiodarone inhibition of cell entry mediated by filoviral GP in a range of different target cells. There was significant inhibition in all cell lines tested, although there appeared to be a trend towards a more pronounced effect on cell lines of endothelial origin compared with epithelial cell lines, with cell lines of fibroblast origin appearing least sensitive (Figure 6). Filoviral GP-mediated entry into primary human umbilical vein endothelial cells and in macrophages derived from primary human monocytes was significantly inhibited at amiodarone concentrations below 1 μg/mL for both MARV and EBOV GP while amiodarone had no effect on the transduction of primary hepatocytes (Supplementary Data). These cell type-specific differences are also in keeping with a host cell-targeted mechanism of action of amiodarone.
Figure 6.
Amiodarone inhibition of EBOV GP-mediated entry into different cell types. The indicated cell lines were transduced with pseudoparticles bearing EBOV GP in the presence or absence of amiodarone. Values are normalized to absence of the drug and to the signal detected with VSV-G in order to control for effects on cell proliferation. Means ± SD of n = 3 independent replicates from a representative of two independent experiments are shown. Significant inhibition is indicated by asterisks (*P < 0.05, **P < 0.01).
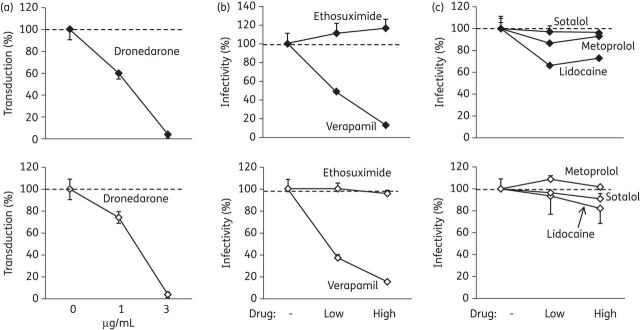
The multi-channel inhibitor dronedarone as well as some calcium channel inhibitors share the anti-filoviral effect of amiodarone
Amiodarone inhibits or otherwise modulates a number of cellular targets that include potassium channels, sodium channels, calcium channels and α- and β-adrenergic receptors.42 We used more specific pharmacological agents to test whether the observed anti-filoviral effect was associated with any of the known pharmacodynamic properties of amiodarone. Dronedarone, a newer drug with a pharmacodynamic profile similar to amiodarone, showed a marked concentration-dependent inhibition of MARV and EBOV GP-bearing pseudoparticles similar to amiodarone (Figure 7a). The L-type calcium channel blocker verapamil similarly inhibited filovirus GP-mediated entry, although higher absolute and molar concentrations were required (Figure 7b). Conversely, the selective T-type calcium channel blocker ethosuximide had no effect. The sodium channel blocker lidocaine, the β1-selective adrenoreceptor antagonist metoprolol, and the non-selective β-blocker and potassium channel blocker sotalol were also without detectable effects (Figure 7c). Drug concentrations were chosen based on plasma levels reported to be achievable in humans. Relevant toxicity or anti-proliferative effects at the concentrations used were excluded using an EAhy cell line stably expressing a luciferase reporter (Supplementary Data). Thus, several agents that are either non-specific ion channel inhibitors (amiodarone, dronedarone) or inhibitors of L-type calcium channels (verapamil) inhibit cell entry mediated by filoviral GPs, while other agents possessing some of the other multiple pharmacodynamic effects of amiodarone do not.
Figure 7.
Effect of specific ion channel blockers on filoviral entry. (a) EAhy cells were transduced with EBOV (upper panels) or MARV (lower panels) pseudoparticles in the presence of the indicated concentrations of dronedarone. Individual data points represent the mean ± SD of n = 3 independent replicates from a representative of three independent experiments. (b) Transduction in the presence of calcium channel blockers. For each drug, a lower and a higher concentration was used as follows: ethosuximide, 100 and 300 μg/mL; verapamil, 20 and 60 μg/mL. Individual data points represent the mean ± SD of n = 3 independent replicates from a representative of three independent experiments. (c) Transduction in the presence of the sodium channel blocker lidocaine (5 and 15 μg/mL), the β-receptor blocker metoprolol (0.2 and 0.6 μg/mL) and the potassium channel blocker/β-receptor blocker sotalol (3 and 9 μg/mL). Individual data points represent the mean ± SD of n = 3 independent replicates from a representative of three independent experiments.
Discussion
In this paper we describe the identification of amiodarone and other drugs that act as inhibitors of ion channels (dronedarone, verapamil) as inhibitors of filovirus cell entry. We showed that amiodarone at concentrations that are routinely reached in human serum when amiodarone is employed clinically acts as a host cell-targeting agent that blocks viral entry.
In the absence of approved treatments, the management of filoviral haemorrhagic fever is largely limited to supportive care and containment. Mortality is very high in resource-poor settings, and even when modern intensive care is available, treatment is challenging.43 Several potential approaches have been suggested based on in vitro or animal data. These include monoclonal antibodies isolated from survivors,44 lipid-stabilized small-interfering RNAs directed against viral genes45 and a recombinant VSV-based vaccine.46 However, it will be difficult to transfer these highly experimental approaches into routine clinical care. Very recently, the oestrogen receptor modulator clomiphene, which is licensed for ovarian stimulation in humans, has been reported to inhibit filovirus infection, possibly by perturbing the role of NPC1 in filoviral entry.47,48 Amiodarone is available both as an oral and an intravenous formulation and has been used for decades for both the long-term and short-term treatment of cardiac arrhythmias. As a generic drug, its use would be feasible in both resource-rich and resource-poor settings. It is known to have a narrow therapeutic window, but serum levels to treat cardiac arrhythmias are in the range of 1.5–2.5 μg/mL, which is 6- to 10-fold above the in vitro IC50 we observed for authentic filoviruses.28 The known side effects of amiodarone (which include ocular and pulmonary toxicity, thyroid dysfunction and pro-arrhythmic effects) are generally manageable, especially in the short term. Importantly, they would clearly be acceptable if the drug at the same time increased the odds of survival in the setting of acute filoviral haemorrhagic fever. Given that filoviral haemorrhagic fever involves the spread of infection to a broad range of cells and tissues, it is plausible that a potent entry inhibitor would limit systemic spread and hence might reduce mortality. However, clinical studies would be needed to determine this.
At this point, we do not know the exact mechanism by which amiodarone and the other anti-arrhythmic agents studied inhibit filoviral cell entry. Several observations suggest that amiodarone acts on the host cell rather than the incoming virion: (i) preincubation of target cells with the drug also had an inhibitory effect on MARV; (ii) the most pronounced effect was seen when the drug was present before and during infection; and (iii) the inhibitory effect was cell type dependent in the sense that it was detectable to a variable extent on most cell types tested, with the notable exception of primary human hepatocytes. However, there was also a trend to reduced infectivity when pseudoviruses were pre-treated with amiodarone and then washed before being added to the target cell. This might suggest that either we were unable to completely separate the drug from the virus or that amiodarone inhibition requires some form of ternary interaction between drug, filovirus and host cell.
There are at least two hypotheses for how amiodarone may render the host cell less susceptible to filoviral entry: On the one hand, both amiodarone and dronedarone have been reported to perturb a late step in endosomal processing, resulting in lipid accumulation and gross alterations in endosomal structure, a phenotype thought reminiscent of Niemann–Pick type C disease.49,50 Besides filoviruses, New World arenavirus Guanarito was also strongly inhibited by amiodarone while other viruses tested were not. Since both are thought to enter target cells via the late endosome, this would be consistent with an amiodarone-mediated perturbation of this compartment.22,51,52 Indeed Shoemaker et al. have reported that clomiphene and other related inhibitors of filoviral entry they have identified have in common that they are cationic amphiphiles causing a Niemann–Pick-like accumulation of cholesterol in the endosomes.47 This could be due to inhibition of acid sphingomyelinase by a broad range of cationic amphiphiles.53 Amiodarone, dronedarone and verapamil, with pKa values of 8.5, 9.8 and 8.8, respectively,49,54 all fit into that category. Moreover, entry through late endosomes has also been suggested for hantaviruses, one of which was modestly inhibited by amiodarone in our research.55 In a different study, an inhibitory effect of very high concentrations of amiodarone on severe acute respiratory syndrome coronavirus was reported.50 That paper suggests that the inhibition of infection occurs after fusion of the viral envelope with the late endosomal membrane and delivery of the viral genome to the cytosol. Since a block at a post-fusion step would be expected to affect all pseudoviruses equally, this does not explain our observation that some pseudoviruses are inhibited by amiodarone while others are not.
The second hypothesis is that the inhibition of filoviral entry by anti-arrhythmic agents is linked to their primary pharmacodynamic properties. Amiodarone is a pleiotropic drug known to target potassium channels, sodium channels, calcium channels, and α- and β-adrenergic receptors. When testing other agents with a narrower range of molecular target, only the L-type calcium channel blocker verapamil exhibited an anti-filoviral effect. These findings may suggest a role for calcium currents in filoviral entry. While their role is more obvious in excitable tissues such as muscle and neuronal cells, L-type calcium channels are also present in many non-excitable cells including endothelial and epithelial cells.56,57 Calcium fluxes can cause rapid shifts in intracellular calcium levels and are an important part of cellular signalling processes, most notably maybe the phospholipase C/1,4,5-inositol trisphosphate/diacylglycerol pathway.58 Thus, interference with the calcium-mediated cell signalling events required for the orchestration of filoviral entry is another plausible mechanism. Interestingly, it has very recently been reported that gabapentin, verapamil and other inhibitors of voltage-gated calcium channels, as well as the siRNA-mediated knockdown of calcium channel subunits, inhibit infection by the New World arenavirus Junin.59
Ongoing work aims to test these hypotheses and determine the exact mechanism of filovirus inhibition by amiodarone and other anti-arrhythmic agents. The work presented here shows that a limited-scale screening of drugs with a known safety profile in humans can reveal novel antiviral properties that may be useful in addressing rare yet severe infections for which drug discovery programmes will likely not be initiated. The chances of identifying potentially useful agents may be best for viruses that employ complex cell entry routes such as filoviruses. The discovery of such unexpected antiviral properties in known drugs with cellular targets may at the same time offer novel insights into the molecular mechanisms of the virus–host interaction and new therapeutic approaches.
Funding
This work was supported by an ‘Emmy Noether’ Fellowship from Deutsche Forschungsgemeinschaft to T. V. H. (HA4394/2-1).
Transparency declarations
The authors declare that they have no conflicts of interest relevant to this work.
Supplementary Material
Acknowledgements
We thank Hyeryun Choe, Francois-Loic Cosset, Jay Hooper, Stefan Kunz, Gary Nabel, Charles Rice and Anthony Sanchez for providing reagents and Sandra Westhaus for help with data analysis.
References
- 1.Feldmann H, Geisbert TW. Ebola haemorrhagic fever. Lancet. 2011;377:849–62. doi: 10.1016/S0140-6736(10)60667-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Geisbert TW, Hensley LE, Larsen T, et al. Pathogenesis of Ebola hemorrhagic fever in cynomolgus macaques: evidence that dendritic cells are early and sustained targets of infection. Am J Pathol. 2003;163:2347–70. doi: 10.1016/S0002-9440(10)63591-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Geisbert TW, Young HA, Jahrling PB, et al. Pathogenesis of Ebola hemorrhagic fever in primate models: evidence that hemorrhage is not a direct effect of virus-induced cytolysis of endothelial cells. Am J Pathol. 2003;163:2371–82. doi: 10.1016/S0002-9440(10)63592-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Baskerville A, Fisher-Hoch SP, Neild GH, et al. Ultrastructural pathology of experimental Ebola haemorrhagic fever virus infection. J Pathol. 1985;147:199–209. doi: 10.1002/path.1711470308. [DOI] [PubMed] [Google Scholar]
- 5.Geisbert TW, Jaax NK. Marburg hemorrhagic fever: report of a case studied by immunohistochemistry and electron microscopy. Ultrastructural Pathol. 1998;22:3–17. doi: 10.3109/01913129809032253. [DOI] [PubMed] [Google Scholar]
- 6.Ito H, Watanabe S, Takada A, et al. Ebola virus glycoprotein: proteolytic processing, acylation, cell tropism, and detection of neutralizing antibodies. J Virol. 2001;75:1576–80. doi: 10.1128/JVI.75.3.1576-1580.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Takada A. Filovirus tropism: cellular molecules for viral entry. Front Microbiol. 2012;3:34. doi: 10.3389/fmicb.2012.00034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Volchkov VE, Feldmann H, Volchkova VA, et al. Processing of the Ebola virus glycoprotein by the proprotein convertase furin. Proc Natl Acad Sci USA. 1998;95:5762–7. doi: 10.1073/pnas.95.10.5762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jeffers SA, Sanders DA, Sanchez A. Covalent modifications of the Ebola virus glycoprotein. J Virol. 2002;76:12463–72. doi: 10.1128/JVI.76.24.12463-12472.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chan SY, Empig CJ, Welte FJ, et al. Folate receptor-alpha is a cofactor for cellular entry by Marburg and Ebola viruses. Cell. 2001;106:117–26. doi: 10.1016/s0092-8674(01)00418-4. [DOI] [PubMed] [Google Scholar]
- 11.Shimojima M, Takada A, Ebihara H, et al. Tyro3 family-mediated cell entry of Ebola and Marburg viruses. J Virol. 2006;80:10109–16. doi: 10.1128/JVI.01157-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kondratowicz AS, Lennemann NJ, Sinn PL, et al. T-cell immunoglobulin and mucin domain 1 (TIM-1) is a receptor for Zaire Ebolavirus and Lake Victoria Marburgvirus. Proc Natl Acad Sci USA. 2011;108:8426–31. doi: 10.1073/pnas.1019030108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Simmons G, Rennekamp AJ, Chai N, et al. Folate receptor alpha and caveolae are not required for Ebola virus glycoprotein-mediated viral infection. J Virol. 2003;77:13433–8. doi: 10.1128/JVI.77.24.13433-13438.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sinn PL, Hickey MA, Staber PD, et al. Lentivirus vectors pseudotyped with filoviral envelope glycoproteins transduce airway epithelia from the apical surface independently of folate receptor alpha. J Virol. 2003;77:5902–10. doi: 10.1128/JVI.77.10.5902-5910.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Becker S, Spiess M, Klenk HD. The asialoglycoprotein receptor is a potential liver-specific receptor for Marburg virus. J Gen Virol. 1995;76:393–9. doi: 10.1099/0022-1317-76-2-393. [DOI] [PubMed] [Google Scholar]
- 16.Alvarez CP, Lasala F, Carrillo J, et al. C-type lectins DC-SIGN and L-SIGN mediate cellular entry by Ebola virus in cis and in trans. J Virol. 2002;76:6841–4. doi: 10.1128/JVI.76.13.6841-6844.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Simmons G, Reeves JD, Grogan CC, et al. DC-SIGN and DC-SIGNR bind ebola glycoproteins and enhance infection of macrophages and endothelial cells. Virology. 2003;305:115–23. doi: 10.1006/viro.2002.1730. [DOI] [PubMed] [Google Scholar]
- 18.Takada A, Fujioka K, Tsuiji M, et al. Human macrophage C-type lectin specific for galactose and N-acetylgalactosamine promotes filovirus entry. J Virol. 2004;78:2943–7. doi: 10.1128/JVI.78.6.2943-2947.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gramberg T, Hofmann H, Moller P, et al. LSECtin interacts with filovirus glycoproteins and the spike protein of SARS coronavirus. Virology. 2005;340:224–36. doi: 10.1016/j.virol.2005.06.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mulherkar N, Raaben M, de la Torre JC, et al. The Ebola virus glycoprotein mediates entry via a non-classical dynamin-dependent macropinocytic pathway. Virology. 2011;419:72–83. doi: 10.1016/j.virol.2011.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Nanbo A, Imai M, Watanabe S, et al. Ebolavirus is internalized into host cells via macropinocytosis in a viral glycoprotein-dependent manner. PLoS Pathogens. 2010;6:e1001121. doi: 10.1371/journal.ppat.1001121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chandran K, Sullivan NJ, Felbor U, et al. Endosomal proteolysis of the Ebola virus glycoprotein is necessary for infection. Science. 2005;308:1643–5. doi: 10.1126/science.1110656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Carette JE, Raaben M, Wong AC, et al. Ebola virus entry requires the cholesterol transporter Niemann-Pick C1. Nature. 2011;477:340–3. doi: 10.1038/nature10348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cote M, Misasi J, Ren T, et al. Small molecule inhibitors reveal Niemann-Pick C1 is essential for Ebola virus infection. Nature. 2011;477:344–8. doi: 10.1038/nature10380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Coyne CB, Bergelson JM. Virus-induced Abl and Fyn kinase signals permit coxsackievirus entry through epithelial tight junctions. Cell. 2006;124:119–31. doi: 10.1016/j.cell.2005.10.035. [DOI] [PubMed] [Google Scholar]
- 26.Ploss A, Evans MJ. Hepatitis C virus host cell entry. Curr Opinion Virol. 2012;2:14–9. doi: 10.1016/j.coviro.2011.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Saeed MF, Kolokoltsov AA, Freiberg AN, et al. Phosphoinositide-3 kinase-Akt pathway controls cellular entry of Ebola virus. PLoS pathogens. 2008;4:e1000141. doi: 10.1371/journal.ppat.1000141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Goldschlager N, Epstein AE, Naccarelli G, et al. Practical guidelines for clinicians who treat patients with amiodarone. Practice Guidelines Subcommittee, North American Society of Pacing and Electrophysiology. Arch Int Med. 2000;160:1741–8. doi: 10.1001/archinte.160.12.1741. [DOI] [PubMed] [Google Scholar]
- 29.Kirsch T, Woywodt A, Beese M, et al. Engulfment of apoptotic cells by microvascular endothelial cells induces proinflammatory responses. Blood. 2007;109:2854–62. doi: 10.1182/blood-2006-06-026187. [DOI] [PubMed] [Google Scholar]
- 30.Vondran FW, Katenz E, Schwartlander R, et al. Isolation of primary human hepatocytes after partial hepatectomy: criteria for identification of the most promising liver specimen. Artificial Organs. 2008;32:205–13. doi: 10.1111/j.1525-1594.2007.00524.x. [DOI] [PubMed] [Google Scholar]
- 31.Gierer S, Bertram S, Kaup F, et al. The spike protein of the emerging betacoronavirus EMC uses a novel coronavirus receptor for entry, can be activated by TMPRSS2, and is targeted by neutralizing antibodies. J Virol. 2013;87:5502–11. doi: 10.1128/JVI.00128-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Flint M, von Hahn T, Zhang J, et al. Diverse CD81 proteins support hepatitis C virus infection. J Virol. 2006;80:11331–42. doi: 10.1128/JVI.00104-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mittler E, Kolesnikova L, Strecker T, et al. Role of the transmembrane domain of Marburg virus surface protein GP in assembly of the viral envelope. J Virol. 2007;81:3942–8. doi: 10.1128/JVI.02263-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ciesek S, Westhaus S, Wicht M, et al. Impact of occludin intra- and inter-species variation on its co-receptor function for authentic hepatitis C virus particles. J Virol. 2011;85:7613–21. doi: 10.1128/JVI.00212-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hierholzer JC, Killington RA. Virus isolation and quantitation. London: Academic Press; 1996. [Google Scholar]
- 36.Ciesek S, Steinmann E, Wedemeyer H, et al. Cyclosporine A inhibits hepatitis C virus nonstructural protein 2 through cyclophilin A. Hepatology. 2009;50:1638–45. doi: 10.1002/hep.23281. [DOI] [PubMed] [Google Scholar]
- 37.Ciesek S, von Hahn T, Colpitts CC, et al. The green tea polyphenol, epigallocatechin-3-gallate, inhibits hepatitis C virus entry. Hepatology. 2011;54:1947–55. doi: 10.1002/hep.24610. [DOI] [PubMed] [Google Scholar]
- 38.Lupberger J, Zeisel MB, Xiao F, et al. EGFR and EphA2 are host factors for hepatitis C virus entry and possible targets for antiviral therapy. Nature Med. 2011;17:589–95. doi: 10.1038/nm.2341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Randhawa PS, Farasati NA, Huang Y, et al. Viral drug sensitivity testing using quantitative PCR: effect of tyrosine kinase inhibitors on polyomavirus BK replication. Am J Clin Pathol. 2010;134:916–20. doi: 10.1309/AJCP7JYHJN1PGQVC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gazina EV, Harrison DN, Jefferies M, et al. Ion transport blockers inhibit human rhinovirus 2 release. Antiviral Research. 2005;67:98–106. doi: 10.1016/j.antiviral.2005.05.003. [DOI] [PubMed] [Google Scholar]
- 41.Nemerow GR, Cooper NR. Infection of B lymphocytes by a human herpesvirus, Epstein-Barr virus, is blocked by calmodulin antagonists. Proc Natl Acad Sci USA. 1984;81:4955–9. doi: 10.1073/pnas.81.15.4955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kodama I, Kamiya K, Toyama J. Cellular electropharmacology of amiodarone. Cardiovascular Research. 1997;35:13–29. doi: 10.1016/s0008-6363(97)00114-4. [DOI] [PubMed] [Google Scholar]
- 43.van Paassen J, Bauer MP, Arbous MS, et al. Acute liver failure, multiorgan failure, cerebral oedema, and activation of proangiogenic and antiangiogenic factors in a case of Marburg haemorrhagic fever. Lancet Infect Dis. 2012;12:635–42. doi: 10.1016/S1473-3099(12)70018-X. [DOI] [PubMed] [Google Scholar]
- 44.Maruyama T, Rodriguez LL, Jahrling PB, et al. Ebola virus can be effectively neutralized by antibody produced in natural human infection. J Virol. 1999;73:6024–30. doi: 10.1128/jvi.73.7.6024-6030.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Geisbert TW, Lee AC, Robbins M, et al. Postexposure protection of non-human primates against a lethal Ebola virus challenge with RNA interference: a proof-of-concept study. Lancet. 2010;375:1896–905. doi: 10.1016/S0140-6736(10)60357-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Geisbert TW, Daddario-DiCaprio KM, Williams KJ, et al. Recombinant vesicular stomatitis virus vector mediates postexposure protection against Sudan Ebola hemorrhagic fever in nonhuman primates. J Virol. 2008;82:5664–8. doi: 10.1128/JVI.00456-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Shoemaker CJ, Schornberg KL, Delos SE, et al. Multiple cationic amphiphiles induce a Niemann-Pick C phenotype and inhibit Ebola virus entry and infection. PloS One. 2013;8:e56265. doi: 10.1371/journal.pone.0056265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Johansen LM, Brannan JM, Delos SE, et al. FDA-approved selective estrogen receptor modulators inhibit Ebola virus infection. Sci Trans Med. 2013;5:190ra79. doi: 10.1126/scitranslmed.3005471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Piccoli E, Nadai M, Caretta CM, et al. Amiodarone impairs trafficking through late endosomes inducing a Niemann-Pick C-like phenotype. Biochem Pharmacol. 2011;82:1234–49. doi: 10.1016/j.bcp.2011.07.090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Stadler K, Ha HR, Ciminale V, et al. Amiodarone alters late endosomes and inhibits SARS coronavirus infection at a post-endosomal level. American J Resp Cell Mol Biol. 2008;39:142–9. doi: 10.1165/rcmb.2007-0217OC. [DOI] [PubMed] [Google Scholar]
- 51.Rojek JM, Sanchez AB, Nguyen NT, et al. Different mechanisms of cell entry by human-pathogenic Old World and New World arenaviruses. J Virol. 2008;82:7677–87. doi: 10.1128/JVI.00560-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Rojek JM, Kunz S. Cell entry by human pathogenic arenaviruses. Cellular Microbiol. 2008;10:828–35. doi: 10.1111/j.1462-5822.2007.01113.x. [DOI] [PubMed] [Google Scholar]
- 53.Kornhuber J, Tripal P, Reichel M, et al. Identification of new functional inhibitors of acid sphingomyelinase using a structure-property-activity relation model. J Med Chem. 2008;51:219–37. doi: 10.1021/jm070524a. [DOI] [PubMed] [Google Scholar]
- 54.Hasegawa J, Fujita T, Hayashi Y, et al. pKa determination of verapamil by liquid-liquid partition. J Pharmaceutical Sci. 1984;73:442–5. doi: 10.1002/jps.2600730405. [DOI] [PubMed] [Google Scholar]
- 55.Jin M, Park J, Lee S, et al. Hantaan virus enters cells by clathrin-dependent receptor-mediated endocytosis. Virology. 2002;294:60–9. doi: 10.1006/viro.2001.1303. [DOI] [PubMed] [Google Scholar]
- 56.Dorkkam N, Wongdee K, Suntornsaratoon P, et al. Prolactin stimulates the L-type calcium channel-mediated transepithelial calcium transport in the duodenum of male rats. Biochem Biophys Res Commun. 2013;430:711–6. doi: 10.1016/j.bbrc.2012.11.085. [DOI] [PubMed] [Google Scholar]
- 57.Moccia F, Berra-Romani R, Tanzi F. Update on vascular endothelial Ca(2+) signalling: a tale of ion channels, pumps and transporters. World J Biol Chem. 2012;3:127–58. doi: 10.4331/wjbc.v3.i7.127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Clapham DE. Calcium signaling. Cell. 2007;131:1047–58. doi: 10.1016/j.cell.2007.11.028. [DOI] [PubMed] [Google Scholar]
- 59.Lavanya M, Cuevas CD, Thomas M, et al. siRNA screen for genes that affect Junin virus entry uncovers voltage-gated calcium channels as a therapeutic target. Sci Trans Med. 2013;5:204ra131. doi: 10.1126/scitranslmed.3006827. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.