Abstract
A novel coronavirus of ferrets was first described in 1993. This coronavirus caused an enteric disease called epizootic catarrhal enteritis (ECE). Recently, a ferret systemic coronavirus (FRSCV)-associated disease was discovered. This new systemic disease resembles the dry form of feline infectious peritonitis (FIP) and has been reported in the United States and Europe. This article addresses the clinical signs, pathology, pathogenesis, diagnosis, treatment, and prevention of this ferret FIP-like disease.
Keywords: Ferret, Coronavirus, Hypergammaglobulinemia, Granuloma, Feline infectious peritonitis, Mustela putorius furo
Coronaviruses are large, enveloped, positive-stranded RNA viruses classified under the genus Coronavirus within the family Coronaviridae, order Nidovirales.1 Based on sequence homology, they are subdivided into three groups.2 Group 1 coronaviruses include some important causes of enteric disease in domestic animals, including transmissible gastroenteritis virus of swine, feline coronavirus (FCoV), and canine coronavirus. Both the ferret enteric coronavirus (FRECV) and the ferret systemic coronavirus (FRSCV) were recently identified as group 1 coronaviruses.3, 4 Ferret enteric coronavirus causes an enteric disease called epizootic catarrhal enteritis (ECE).3, 5 More recently, a new systemic coronavirus-associated disease closely resembling the granulomatous or dry form of feline infectious peritonitis (FIP) was reported in the United States and Europe.6, 7 This article focuses on coronaviral diseases of ferrets, with emphasis on the clinical signs, pathology, pathogenesis, diagnosis, treatment, and prevention of the ferret systemic coronavirus-associated disease.
Ferret epizootic enteritis
In March of 1993 a novel enteric disease was reported in domestic ferrets along the east coast of the United States.5 Initial clinical signs included lethargy, hyporexia or anorexia, and vomiting. These signs were quickly followed by a profuse, foul-smelling, bright green watery diarrhea with a high mucus content, and dehydration. During the more chronic stages of the disease, feces of affected ferrets commonly contained grainy material described as resembling birdseed. Based on the clinical presentation, the disease was commonly referred to as green slime disease. After a detailed review of the gross and microscopic lesions and the discovery of an enteric coronavirus within affected intestines, the name epizootic catarrhal enteritis was introduced.5
ECE is a highly contagious diarrheal disease with outbreaks usually involving 100% of the ferrets in the household, breeding facility, or rescue shelter. Even though the morbidity for ECE commonly reaches 100%, overall mortality rate is low (<5%). During the initial outbreaks, young ferrets commonly presented with milder, often subclinical disease, but older ferrets were more severely affected and had higher mortality rates. The disease quickly spread throughout the United States and to several other countries.5
On gross examination of ferrets with ECE, the mucosa of the affected portion of the small intestine is hyperemic, and the intestinal wall appears thin. The microscopic lesions of ECE include diffuse lymphocytic enteritis, with villus atrophy, fusion, and blunting; vacuolar degeneration and necrosis of the apical epithelium; or a combination of all these lesions.5 Immunohistochemistry using a monoclonal antibody against group 1c coronavirus antigen detected large numbers of coronavirus-infected epithelial cells.3, 5 Transmission electron microscopy identified coronavirus-like particles, approximately 120 nm in diameter, in cytoplasmic vacuoles of apical enterocytes and at the cell surface.5 Similar viral particles were observed electron-microscopically in fecal samples from multiple ferrets.5
In subsequent studies, partial sequences of the polymerase, spike, membrane protein, and nucleocapsid genes were identified using coronavirus consensus polymerase chain reaction (PCR) assays.3 Based on analyses of these data and the complete sequence of the nucleocapsid gene, the ECE-associated coronavirus was found to be a novel coronavirus most closely related to group 1 coronaviruses. This coronavirus was identified as FRECV and shown to be more similar to feline coronavirus, porcine transmissible gastroenteritis virus, and canine coronavirus than to porcine epidemic diarrhea virus and human coronavirus 229E.3 In situ hybridization using oligoprobes based on FRECV-specific sequences confirmed infection of villar epithelial cells with FRECV in ferrets with ECE.3
Based on serologic data and screening of fecal samples by PCR, FRECV seems to be widely distributed in the ferret population in the United States. However, severe clinical ECE has been reported rarely over the past couple of years.
Ferret systemic coronavirus–associated disease
An emerging systemic disease of ferrets characterized by pyogranulomatous perivasculitis and peritonitis was first recognized in Spain in 2004.6 Shortly thereafter, the disease was also described in the United States.7, 8 Clinically and pathologically, the disease closely resembled FIP. FIP is a fatal, multi-systemic, immune-mediated disease of cats caused by virulent mutants of FCoV. The FIP viruses are believed to arise spontaneously from persisting low pathogenic to nonpathogenic feline enteric coronavirus strains.9 This concept was, however, challenged in a recent publication.10 Similar to FIP, this novel disease in ferrets was characterized by positive immunohistochemical labeling of the cytoplasm of intralesional macrophages for coronaviral antigen.7, 11
Recent publications confirmed the detected coronavirus to be a novel group 1 coronavirus and named it ferret systemic coronavirus.4, 7 Partial sequence analysis showed FRSCV to be more similar to FRECV than to other group 1 coronaviruses, including FCoV.3, 4 The similarities in clinical disease and microscopic lesions between FRSCV and FIP virus suggested a similar pathogenesis for FRSCV-associated disease and FIP, but experimental proof is needed.
Clinical Signs
FRSCV-associated disease causing FIP-like lesions has been reported in mostly young ferrets, with most younger than 18 months.7 Clinical signs in ferrets are nonspecific, similar to those described in cats with the granulomatous or dry form of FIP.6, 12, 13 Common clinical signs include diarrhea, weight loss, lethargy, hyporexia or anorexia, and vomiting. These gastrointestinal signs may lead to loss of body condition and moderate to severe emaciation. Signs of central nervous system disease include hind limb paresis or paraparesis, ataxia, tremors, and seizures. Animals may present with primary neurologic disease, including head tilt and seizure activity. Less common clinical signs include sneezing, coughing, labored breathing, nasal discharge, dehydration, bruxism, systolic heart murmur, jaundice, focal areas of erythema of the skin, green colored urine, reddened rectal mucosa, and rectal prolapse.7, 14 Ocular signs have not yet been reported. On abdominal palpation, large abdominal masses, splenomegaly, and renomegaly are common findings. Peripheral lymphadenopathy has been reported in a few cases, and some ferrets also had fevers ranging from 103°F to 105.4°F.7, 14
Pathology
Gross lesions observed in ferrets with FRSCV infection closely resemble those described in cats with the dry form of feline infectious peritonitis.7, 12, 15 The most commonly observed gross lesion consists of multifocal to coalescing white to tan irregular nodules or plaques ranging from 0.5 to 2.0 cm in diameter dispersed over serosal surfaces (Fig. 1, Fig. 2, Fig. 3 ). Nodules are usually oriented along vasculature pathways. The peritoneum, particularly the intestinal serosa and the mesentery, are most commonly affected (see Fig. 1), with the mesentery being multifocally and irregularly thickened by pale white firm nodules and plaques.
Fig. 1.
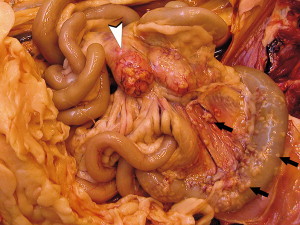
FRSCV-associated granulomatous peritonitis. Multifocal to coalescing, white firm nodules (black arrows) of varying size are widely distributed throughout the mesentery following the vasculature. The mesenteric lymph nodes (arrowhead) are enlarged and contain similar nodules that replace normal parenchyma and commonly extend through the capsule. (Courtesy of Dodd Sledge, DVM, Lansing, MI, USA.)
Fig. 2.
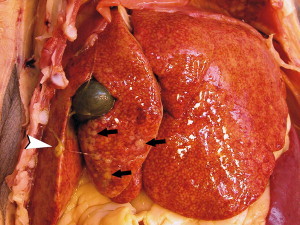
FRSCV-associated granulomatous hepatitis and fibrinous peritonitis. The hepatic capsule is covered by thick strands of fibrin (arrowhead). Multifocal, white firm nodules are randomly distributed over the hepatic serosa and commonly extend into the parenchyma. (Courtesy of Dodd Sledge, DVM, Lansing, MI, USA.)
Fig. 3.
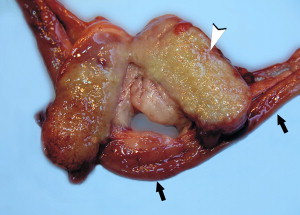
FRSCV-associated granulomatous lymphadenitis. Section of jejunum (black arrows) and cross section of a severely enlarged mesenteric lymph node (arrowhead). The lymph node parenchyma is effaced by severe, diffuse granulomatous inflammation. (Courtesy of Dodd Sledge, DVM, Lansing, MI, USA.)
Similar nodules can be commonly found on the surface or extending into the parenchyma of numerous other organs, with the liver (see Fig. 2), kidneys, spleen, and lung most commonly affected. The mesenteric lymph nodes are affected in most cases. They can be enlarged up to eight times their normal size, with a highly irregular capsular surface characterized by dozens of slightly raised white nodules. On cut surface, the normal parenchyma is often replaced by granulomatous inflammation (see Fig. 3).
Other less-specific gross lesions include the commonly observed splenomegaly, and occasional renomegaly and hepatomegaly. Based on current knowledge, ferrets with FRSCV infrequently present with serous effusions into the body cavities that are characteristic of the effusive or wet form of FIP; however, fibrinous exudate is rarely encountered (see Fig. 2). In animals with neurologic signs, gross lesions within the nervous system were rather limited. Some moderate meningeal opacity around the medulla and choroid plexuses of the fourth ventricle may be observed. On transverse sections, the choroid plexi can be slightly thickened and viscous exudates may be visible.
Histologic lesions are characterized by severe pyogranulomatous inflammation and are most commonly observed in the mesentery and along the peritoneal surface. Pyogranulomatous inflammation commonly encompasses the small intestine and focally expands or destroys the muscularis and serosa (Fig. 4 ). Pyogranulomas are characterized by central areas of necrosis composed of cellular debris and degenerative neutrophils surrounded by epithelioid macrophages with additional layers of lymphocytes and plasma cells (Fig. 5 ). Rare multinucleated giant cells have been described.
Fig. 4.
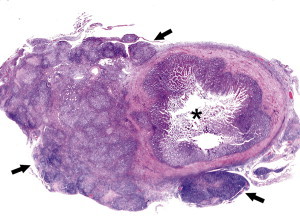
FRSCV-associated granulomatous peritonitis. The asterisk indicates the lumen of a cross section of jejunum. Circumferentially encompassing the small intestine and extending into the mesentery is severe granulomatous peritonitis (black arrows). Hematoxylin and eosin staining (orginal magnification ×4).
Fig. 5.
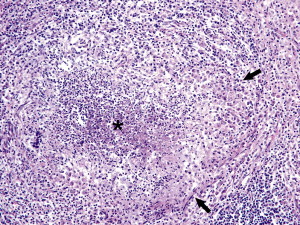
FRSCV-associated pyogranulomatous peritonitis. A higher magnification of the pyogranulomatous lesions of Fig. 4 shows the necrotic center composed of cellular debris and degenerative neutrophils (asterisk) surrounded by epithelioid macrophages (black arrows) followed by lymphocytes and plasma cells and a rim of fibroblasts. Hematoxylin and eosin staining (original magnification ×10).
Necrosis is an inconsistent feature, but microgranulomas may be composed predominantly of epithelioid macrophages. Variable degrees of fibrosis surround some granulomas. Granulomatous inflammation is often localized around vessels and frequently involves the adventitia, with inflammatory cells migrating into the medial tunics of small veins and venules (Figs. 6 and 7 ). Similar areas of multifocal pyogranulomatous inflammation commonly expand and obliterate the normal architecture of the lymph nodes and other infected organs, resulting in nephritis, pancreatitis, adrenalitis, meningitis, myocarditis, and pneumonia.
Fig. 6.
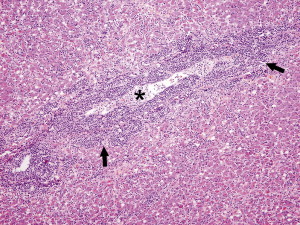
FRSCV–associated granulomatous vasculitis and hepatitis. The inflammatory reaction is centered around vessels (asterisk) and composed of macrophages admixed with neutrophils, lymphocytes, and plasma cells that form thick perivascular cuffs (black arrows) that also migrate through the vascular wall and extend into the surrounding hepatic parenchyma. Hematoxylin and eosin staining (original magnification ×10).
Fig. 7.
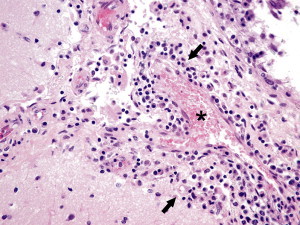
Ferret systemic coronavirus–associated granulomatous vasculitis. Granulomatous vasculitis is also observed in the brain and most commonly affects the periventricular and meningeal vessels (asterisk). Inflammatory cells (black arrows), including macrophages admixed with lymphocytes, plasma cells, and a few neutrophils, surround affected vessels and infiltrate the vascular media. Hematoxylin and eosin staining (original magnification ×40).
In animals with neurologic signs, the primary lesions may be localized entirely within the brain and consist of a severe pyogranulomatous leptomeningitis, choroiditis, ependymitis, and encephalomyelitis. The inflammatory process is centered on vessels, particularly venules (see Fig. 7), along the inner and outer surfaces of the brain, with only focal extension into the underlying parenchyma. The most severe parenchymal extension of the inflammatory reaction is usually observed periventricularly.
Immunohistochemistry using a monoclonal antibody against group 1c coronavirus antigen shows strong positive intracytoplasmic staining of macrophages within the center of pyogranulomas (Fig. 8 ).7, 8, 11
Fig. 8.
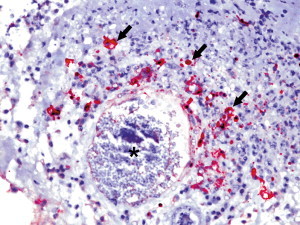
Ferret systemic coronavirus-associated granulomatous vasculitis. Large numbers of macrophages within the inflammatory reaction surrounding and infiltrating into a large vessel (asterisk) contain abundant amounts of coronaviral antigen as indicated by the red chromogen (black arrows). Immunohistochemistry against coronavirus type 1 antigen using an alkaline phosphatase detection system, hematoxylin counterstaining (original magnification ×40).
Transmission electron microscopy of areas of pyogranulomatous inflammation revealed macrophages with spherical, enveloped viral particles, 70 to 140 nm in diameter, in membrane-bound cytoplasmic vacuoles and free within the cytoplasm.7 Occasionally, circumferential spikes were observed along the outer wall of the virions.
Pathogenesis
Current knowledge of the pathogenesis of FRSCV-associated disease is rather limited. No experimental reproduction of the disease has occurred, so most of the current hypotheses are drawn from clinical observations, pathologic examinations, and genetic analysis of the ferret coronaviruses. The clinical signs of the systemic form, with which FRSCV has been associated, are certainly similar to those seen in cats affected with the granulomatous or dry form of FIP. Likewise, the gross and histopathologic lesions associated with the systemic form are nearly identical to those seen in the tissues of cats affected with the granulomatous form of FIP. The limited number of FRSCV strains analyzed thus far differ significantly from FRECV strains in the gene encoding for the spike protein, but additional FRECV and FRSCV strains must be analyzed to either substantiate or modify current data.4
Diagnosis
Because no pathognomonic clinical signs exist for FRSCV-associated disease, diagnostic testing is required to confirm the diagnosis. Typical hematologic signs include nonregenerative anemia, hyperglobulinemia, hypoalbuminemia, and thrombocytopenia. Serum protein electrophoretograms show a polyclonal hypergammaglobulinemia.7, 14, 16, 17 Differential diagnoses for hypergammaglobulinemia in domestic ferrets include Aleutian disease, lymphoma/lymphosarcoma, multiple myeloma, chronic infection (Helicobacter), or chronic inflammation from inflammatory bowel disease.7, 14, 16, 18 Counterimmunoelectrophoresis testing for anti-Aleutian disease parvovirus antibodies should be performed to exclude Aleutian disease as a differential.7, 16
Biochemical changes are variable and reflect damage to abdominal organs, such as kidneys, liver, pancreas, and the gastrointestinal tract (see Fig. 1, Fig. 2, Fig. 3). Serum chemistry abnormalities include elevated serum lipase, elevated blood urea nitrogen, elevated serum alanine transferase, elevated alkaline phosphatase, and elevated serum gamma glutamyl transferase.7 Urinalysis results have only been reported for four cases of FRSCV-associated disease, but abnormal findings include green urine, proteinuria, blood, and rare bilirubin crystals.7 The greenish color of the urine is likely caused by high levels of biliverdin. Biliverdin may be from microhemorrhage into tissues and extravascular destruction of red blood cells as part of the vasculitis and disseminated intravascular coagulation. A similar mechanism has been reported in cats with FIP.13 Radiographs may show abdominal masses, splenomegaly, and nephromegaly. Patchy densities in the lungs have been reported in at least one ferret.17
The Diagnostic Center for Population and Animal Health at Michigan State University is offering PCR-based testing to detect ferret coronavirus infections. To determine whether ferrets are shedding FRECV, fecal swabs or samples are preferred. A combination of unfixed and fixed tissues containing granulomatous lesions is used to detect ferret coronavirus in ferrets affected with the systemic form.
The two reverse transcription PCR (RT-PCR) assays amplify a portion of the spike gene. Based on available data, these assays are genotype-specific. An example of the data included in a recent article is presented in Fig. 9 .4 The authors are currently examining whether these assays also have pathotype specificity. Ultimately clinical, pathologic, and molecular diagnostic data must be combined to determine the final diagnosis.
Fig. 9.
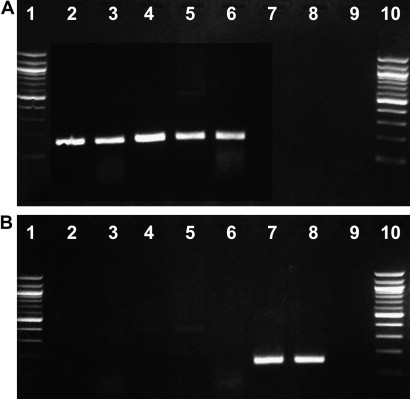
S-gene genotype-specific diagnostic reverse-transcription polymerase chain reaction on clinical samples. (A) Genotype 1–specific assay, 157-bp–positive amplicon observed only on lanes 2 through 6. (B) Genotype 2–specific assay, 147-bp–positive amplicon observed only on lanes 7 and 8. Lanes 1 and 10: 100-bp DNA ladder; Lane 2: FRSCV MSU-1–positive lung: Lane 3: FRSCV MSU-1–positive kidney; Lane 4: FRSCV WADL–positive lymph node; Lane 5: FRSCV MSU-S–positive spleen; Lane 6: FRSCV MSU-S–positive intestine; Lane 7: FRECV MSU-2–positive feces; Lane 8: FRECV 1202–positive feces; Lane 9: negative control (sterile water). MSU, Michigan State University; WADL, Washington Animal Disease Diagnostic Laboratory. (From Wise A, Kiupel M, Garner MM, et al. Comparative sequence analysis of the distal one-third of the genomes of a systemic and an enteric ferret coronavirus. Virus Res 2010;149:42–50; with permission.)
In addition to viral RNA detection using RT-PCR, the laboratory offers a serologic test to detect antibodies to ferret coronaviruses. The main purpose of this test is to determine whether ferrets were previously exposed to a ferret coronavirus. Thus far this test has been used exclusively to detect evidence of previous FRECV infection. As is the case for feline coronavirus, overinterpretation of titer levels can lead to erroneous conclusions, and therefore interpretation requires a good understanding of the limitations of this test.
Serum antibody tests are frequently used to help diagnose FIP in cats.13, 19 Feline serum antibody tests can include immunofluorescent antibody (IFA), virus neutralization, and enzyme-linked immunosorbent assay (ELISA), but IFA is most commonly used.13 In general, a high antibody titer is suggestive of FIP, and a low or negative antibody titer makes FIP less likely.13 Unfortunately, these tests are not specific for FIP. Many healthy cats will have a positive antibody titer from exposure to the feline enteric coronavirus, but most of them will not develop FIP.13, 19
In the few ferrets that have been tested for serum antibody titers with the feline IFA or ELISA test, antibody titers have been negative.7 In an attempt to improve the sensitivity and specificity of FIP testing, PCR tests have been developed. These tests detect viral RNA from the feline coronavirus. A RT-PCR test was developed recently to detect messenger RNA of the M gene of the feline coronavirus in macrophages/monocytes.20 The M gene is only expressed during viral replication; therefore, detection of replicating coronavirus in the blood is thought to be more specific for FIP in cats.20 Unfortunately, the M gene of the ferret coronavirus is very different from that of the feline coronavirus, and therefore this test will not work for ferrets. (Bernhard Kaltenboeck, DVM, PhD, personal communication, 2008).
Typical clinical signs, blood work results, and a polyclonal gammopathy on serum protein electrophoresis are suggestive of FRSCV-associated disease but are not definitive. Typical microscopic lesions are very suggestive of a diagnosis of FRSCV-associated disease; however, definitive diagnosis requires positive immunohistochemistry staining of the coronavirus antigen in macrophages within areas of granulomatous inflammation. The monoclonal antibody FIPV3-70 has been used and is recommended to detect the FRSCV antigen.7, 11 However, this antibody cross-reacts with all group 1 coronaviruses and will also detect FRECV.
Further differentiation of FRSCV from FRECV remains a major challenge. Based on sequence data from a limited number of enteric and systemic strains, FRSCV and FRECV are closely related but are genetically distinct. In fact, current data indicate that FRSCV differs significantly more from FRECV than FIPV does from FCoV. Two ferret coronavirus-specific RT-PCR assays were developed as a result of these genetic findings, and these assays are the current gold standard to differentiate FRSCV from FRECV.
Treatment
Currently no cure exists for ferrets with FRSCV-associated disease; most died of the disease or were humanely euthanized because of advanced disease. Nonetheless, some ferrets have survived for several months after diagnosis, and one of the authors (JM) has a long-term survivor of more than 3 years.7, 14, 16
FRSCV-associated disease is an immune-mediated disease, and therefore treatment is aimed at suppressing the immune system, suppressing the excessive inflammatory response, and eliminating or reducing the systemic coronavirus through immune modulation. In addition, symptomatic treatment and nutritional support can be used to try to ameliorate the clinical signs and improve the ferret's quality of life.14, 16, 17
Immune suppression
Prednisolone (Pediapred) is the main medication used to suppress the ferret's immune system.16 Prednisolone suppresses both the humoral and cell-mediated immunity (CMI).21, 22, 23, 24, 25, 26 Prednisolone decreases chemotaxis and phagocytosis by macrophages.21 The reduction in phagocytosis by macrophages and monocytes may be caused by inhibition of Fc receptors or steroid receptor–mediated events.23, 27 The inhibition of phagocytosis may help decrease the amount of circulating coronavirus in macrophages and monocytes.
Prednisolone also has potent anti-inflammatory effects. Prednisolone inhibits the release of arachidonic acid from membrane phospholipids, and this inhibition prevents the synthesis of prostaglandins, thromboxanes, and leukotrienes, which are the main mediators of inflammation.22 In addition to its immunosuppressive and anti-inflammatory actions, prednisolone may increase the ferret's appetite and make it feel better.16 A high dose of prednisolone (1–2 mg/kg twice daily) is suggested initially, with a gradual tapering of the dose over time.16
Other immunosuppressants, such as cyclophosphamide (Cytoxan) and chlorambucil (Leukeran), have been used in the treatment of felines with FIP.13, 19, 25 The goal of these two chemotherapy medications is to cause additional immunosuppression and lower the prednisolone dose; however, cyclophosphamide should not be used in ferrets because it may lower the CMI and cause the wet form of FIP to develop (John August, BVetMed, MS, MRCVS, personal communication, 2007, and Diane Addie, PhD, BVMS, MRCVS, personal communication, 2007). Azathioprine (Imuran), another immunosuppressant, has a greater effect on humoral immunity than on CMI.22 It is very myelotoxic in cats; therefore, it is not recommended for treatment of cats with FIP.19, 22, 25 However, it is used anecdotally for treating inflammatory bowel disease and Aleutian disease in ferrets; therefore, it might be worth trying azathioprine with prednisolone for additional immunosuppression, to lower the gamma globulin level, and to lower the prednisolone dose.
Immune modulation
The goal of immune modulation is to help reduce or eliminate the systemic coronavirus. Immune modulators used in feline FIP cases include a polyprenyl immunostimulant and interferon.24, 25, 26, 28, 29, 30, 31, 32 Polyprenyl immunostimulant is a new, investigational veterinary product composed of phosphorylated and linear polyisoprenols. It has been shown to increase T-helper lymphocytes and Th-1 cytokines, and has been used for the treatment of numerous viral diseases.28, 32 It enhances cell-mediated immunity through Toll-like receptors (Alfred Legendre, DVM, MS, personal communication, 2010).
In a recent pilot study, polyprenyl immunostimulant was used to treat three cats with the dry form of FIP. Two cats experienced long-term remission of more than 2 years when treated with a dose of 3 mg/kg orally, two to three times a week. The third cat was only treated for a few months with a twice-daily dose of 1 mg/kg subcutaneously, and survived for 14 months.28 Polyprenyl immunostimulant does not seem to work for cases of the wet form of FIP, but it did seem to be effective in the three cats with the dry form of FIP.28 Polyprenyl immunostimulant is available as part of a field trial through the College of Veterinary Medicine at the University of Tennessee (Tanya Kuritz, PhD, personal communication, 2009). Polyprenyl immunostimulant has a very wide margin of safety in cats, dogs, and mice, but studies are needed to assess its safety and efficacy in ferrets (Alfred Legendre, DVM, MS, personal communication, 2009).
Interferon is another immune modulator that has been studied in feline FIP.13, 24, 25, 26, 29, 30, 31, 32 Both feline interferon omega and human interferon alpha have been shown to inhibit feline coronavirus replication in vitro.13, 32 Recombinant feline interferon omega is available in some European countries and Japan and has been used to treat cats with FIP.19, 24, 25, 26 In one uncontrolled study in Japan, a recombinant feline interferon omega was used with dexamethasone or oral prednisolone to treat 12 cats with suspected FIP; 4 older cats with the wet form of FIP experienced long-term remission of greater than 2 years with this treatment.30
A recent placebo-controlled, double-blind study with feline interferon omega did not show any effect on survival time or quality of life. This study used either feline interferon omega or placebo along with antibiotics and glucocorticoids; however, the only long-term survivor (200 days) was in the interferon treatment group.31 Another feline researcher anecdotally reports one third of the cats improve with feline interferon omega treatment.26 Recombinant human interferon alpha has also been used in cats, but in one experimental study did not reduce mortality significantly.29
Vasculitis medications
Vasculitis is one of the common inflammatory lesions in cats with FIP and ferrets with FRSCV-associated disease.7, 11, 13, 17, 19, 26, 33 Recent research has shown that tumor necrosis factor alpha (TNF-α), interleukin-1 beta (IL-1β), matrix metalloproteinase-9 (MMP-9), and major histocompatibility complex II (MHC II) are all involved in the pathogenesis of vasculitis in feline FIP.33 Thus, medications that decrease these promoters of vasculitis may help improve the vasculitis, decrease the inflammation, and alleviate some of the clinical signs. Pentoxifylline is a methylxanthine derivative with hemorrheologic, immunomodulatory, and anti-inflammatory effects.34, 35, 36, 37, 38, 39, 40, 41, 42 Pentoxifylline is a nonspecific phosphodiesterase inhibitor.34, 35, 36, 37, 38, 39 Phosphodiesterase inhibition will increase cyclic adenosine monophosphate (cAMP), which increases red blood cell deformability, decreases red blood cell destruction, and improves microcirculation and tissue oxygenation. In addition, increasing cAMP decreases platelet aggregation and blood viscosity.34, 42 Furthermore, pentoxifylline also inhibits TNF-α synthesis and lowers IL-1 levels.34, 35, 36, 39, 40, 41 Plus pentoxifylline can decrease fibrosis.34, 36 Pentoxifylline has been used to treat feline FIP by some veterinarians with anecdotal improvement, but no studies have been published yet.19, 25 A suggested dose for pentoxifylline in cats is 20 to 25 mg/kg twice daily (Alice Wolf, DVM, personal communication, 2008).
Doxycycline (Vibramycin) is a bacteriostatic antibiotic in the tetracycline family. In addition to its antibiotic properties, it also has anti-inflammatory properties, including inhibition of MMPs.43, 44, 45, 46, 47, 48, 49, 50, 51 MMPs are a family of proteolytic enzymes that degrade various components of the extracellular matrix, and include collagenases, gelatinases, stromelysins, matrilysins, and membrane-type MMPs.43 Doxycycline can also inhibit TNF-α production, decrease fibrosis, and inhibit leukocyte adhesion to endothelial cells.44, 50, 51 Broad-spectrum antibiotic treatment is recommended during immunosuppressive therapy to prevent secondary bacterial infection.16, 25, 26 Doxycycline can be used for this purpose, and its inhibition of MMPs and TNF-α may help decrease damage to the blood vessels. A published dose of doxycycline for ferrets is 10 mg/kg twice daily.52
Ozagrel (Xanbon) is a thromboxane synthetase inhibitor that has been studied in cats with FIP.53 Thromboxane synthetase inhibitors suppress production of thromboxane A2, which is a potent platelet aggregating agent. Platelet aggregation is associated with progression of the vasculitis in FIP.53 The reported dose for ozagrel in cats is 5 to 10 mg/kg twice daily.53
Aspirin (Ascriptin) or acetylsalicylic acid is a nonsteroidal anti-inflammatory drug. Its main mechanism of action is inhibition of inflammatory mediators through cyclooxygenase (COX) inhibition. Aspirin decreases platelet aggregation through inhibiting prostaglandin and preventing the formation of thromboxane A2. However, it can also cause gastric irritation and ulceration, and ferrets as a species are very prone to gastric ulcers. Therefore aspirin, especially with concurrent prednisolone, would have to be used with extreme caution, if at all, in ferrets.
Prednisolone is also used to treat vasculitis.23 Prednisolone can prevent the formation of thromboxanes,22 and can also inhibit TNF-α, IL-1, MHC-II, and platelet aggregation.21, 22, 23, 54, 55 Combining prednisolone, pentoxifylline, and doxycycline may have a synergistic effect on decreasing inflammation and vasculitis.34, 48
Symptomatic treatment
Symptomatic treatment can be used to help alleviate some of the clinical signs. Common clinical signs have involved the gastrointestinal tract and have included hyporexia and anorexia, weight loss, vomiting, dehydration, and diarrhea.7, 14, 16, 17 Gastroprotectant therapy can be used to help reduce nausea and inappetence, and prevent gastric ulcers secondary to high doses of prednisolone. Sucralfate (Carafate oral suspension) can be administered orally at a dose of 75 to 100 mg/kg, 10 to 15 minutes before each feeding and before prednisolone administration.56, 57 Antacids such as cimetidine hydrochloride (Tagamet), famotidine (Pepcid AC), ranitidine (Zantac), and omeprazole (Prilosec) can be used to reduce gastric acid. Omeprazole and famotidine have the advantage of only once-a-day administration; however, cimetidine also has some beneficial effects on the immune system.57, 58, 59, 60, 61, 62 Cimetidine is a type 2 histamine receptor blocker, and can have antiviral activity, stimulate the CMI, and increase T-lymphocyte levels.24, 58, 59, 60, 61, 62 Therefore, cimetidine, 10 mg/kg, administered three times daily is the suggested antacid.
Vomiting can be controlled with either maropitant citrate (Cerenia) or metoclopramide (Reglan). Dehydration can be corrected with fluid therapy. Critical ferrets and those with excessive water loss from vomiting or diarrhea may benefit from intravenous fluids, and those with severe hypoalbuminemia may benefit from administration of colloids or a blood transfusion. For noncritical ferrets, subcutaneous fluids can be used, and most owners are capable of giving subcutaneous fluids at home.56 For ferrets that are stable, a veterinary oral electrolyte solution such as the chicken-flavored Rebound OES, or human electrolyte solutions such as grape-flavored Pedialyte or Gatorade can be used.
Diarrhea is one of the common problems and is likely caused by the pyogranulomatous inflammation in the gastrointestinal tract; however, a fecal parasite examination should be performed to rule out concurrent coccidiosis (Eimeria or Isospora species) and giardiasis.14 Pyogranulomatous intestinal inflammation can lead to maldigestion and malabsorption of nutrients and protein-losing enteropathy. Empiric treatment for diarrhea with antibiotics, probiotics, and antidiarrheals, along with immunosuppressive therapy, may be useful in these cases.
Antiulcer antibiotics such as clarithromycin (Biaxin oral suspension) in combination with amoxicillin (Amoxi-drops) or amoxicillin with clavulanic acid (Clavamox drops) can be used to treat Helicobacter mustelae in the stomach and provide broad-spectrum coverage for the intestinal tact.56, 63 Additional antibiotics such as enrofloxacin (Baytril), marbofloxacin (Zeniquin), or kanamycin (Amforol oral suspension) can be used to cover gram negative bacteria. Probiotics for cats such as Proviable KP paste or Fortiflora may help restore the normal bacterial flora and improve the diarrhea. Antidiarrheals such as kaolin-pectin (Proviable KP paste), loperamide (Imodium A-D), or activated attapulgite (Amforol oral suspension) can be useful in firming up the feces.
Nutritional support
Maintaining proper protein, fat, and calorie intake is critical during treatment. Sick ferrets usually do not eat enough food to cover their nutritional needs, and therefore need nutritional support.56, 57 Ferrets will often eat a soft food when they refuse their normal dry kibble, and therefore veterinary diets designed for recovery, such as Hill's a/d, Royal Canin's recovery RS, Carnivore Care, Iams maximum calorie, or human meat-based baby foods, such as those designed for the first and second stage of solid foods, are usually accepted by inappetant ferrets.16, 56, 57 Ferrets particularly like the turkey or chicken baby foods and Hill's a/d.16, 56 Another option is to make a gruel out of their regular food by grinding it up and adding warm water. Most of these soft foods can be hand-fed or syringe-fed frequently to maintain protein, fat, and caloric needs.
Supplementing vitamins and minerals may also be beneficial. Anemia is one of the common blood abnormalities, and therefore supplemental iron along with erythropoietin (Epogen, Procrit) may help increase the red blood cell production. Chronic diarrhea may be helped by cobalamin (vitamin B12) supplementation. Parental administration of cobalamin at a dose of 250 μg subcutaneously once a week is an empiric recommendation.64 Vitamin B complex at a dose of 1 to 2 mg/kg subcutaneously may be used to increase the ferret's appetite.56 Vitamin B complex can also be added to the subcutaneous fluids or be given orally.
Antioxidants
Antioxidants may be useful to reduce the free radicals and reactive oxygen species that are produced from the inflammatory response and hyperactive immune response. Melatonin is a natural hormone that is a potent antioxidant and free radical scavenger.65, 66, 67 Melatonin also has some anti-inflammatory effects, including decreasing TNF-α, decreasing IL-1B, and inhibiting MMPs.68, 69, 70, 71, 72, 73 Melatonin also has some immune modulation effects and antiviral effects.74, 75, 76, 77, 78, 79 Furthermore, melatonin stimulates the appetite, so it would be another way to treat the inappetence and weight loss. Melatonin is available as a liquid, a tablet, or an injectable implant for ferrets.
Another strong antioxidant is superoxide dismutase (SOD). SOD may have an additional beneficial effect on the immune system because it can increase the ratio of CD4+ to CD8+ lymphocytes in cats.80 S-adenosylmethionine (SAMe) and silybin are other antioxidant therapies that should be beneficial. SAMe can increase glutathione levels and increase hepatic cell repair and regeneration, whereas silybin is an antioxidant. Together they are reported to help protect the liver, which may be useful during long-term prednisolone use.81 Other antioxidants like vitamins A, C, and E may also be helpful.26 One possible side effect from vitamin C supplementation is the formation of calcium oxalate uroliths.82 Calcium oxalate was the third most common ferret urolith and accounted for 10.5% of all the ferret uroliths analyzed at the University of Minnesota Urolith Center, and therefore vitamin C must be used with caution.83
Prevention
Guidelines for prevention of FIP in cats can be modified for prevention of FRSCV-associated disease.24, 26 Prevention of FRSCV-associated disease currently is best accomplished by avoiding exposure to the ferret coronavirus. Unfortunately, FRECV is assumed to be ubiquitous in most multiple-ferret homes, shelters, and breeding farms.5 Nonetheless, reducing fecal contamination of the environment through disinfecting the litter boxes, cages, and bowls with sodium hypochlorite (bleach) weekly, keeping litter boxes away from food and water bowls, and vacuuming up litter around litter boxes and in the ferret room at least once a week may help.26 For breeders, isolation and early weaning of the kits may help prevent exposure.
Summary
Ferret FIP-like disease is a new and almost-always fatal disease associated with the systemic ferret coronavirus; however, it is still worth treating most cases. Treatment may temporarily improve the condition and allow the ferret and owner several months of quality life. Long-term clinical remission may even be possible in some cases. The hope is that ongoing research will lead to new and more effective treatment options for this recently recognized ferret disease.
Acknowledgments
Dr Murray would like to thank the pathologists (Drs Matti Kiupel, Mike Garner, Gaymen Helman, Shane Stiver, and Bruce Williams) and feline infectious peritonitis experts (Drs Diane Addie, John August, Alice Wolf, Richard Weiss, Bernhard Kaltenboeck, Al Legendre, and Niels Pedersen) who helped him understand the immunopathogenesis and treatment strategies for this new disease. He would also like to thank Dr Katrina Ramsell and the Web site www.catvirus.com.
References
- 1.Lai M.M.C., Perlman S., Anderson L.J. Coronaviridae. In: Knipe D.M., Howley P.M., editors. Fields virology. 5th edition. Lippincott Williams & Wilkins; Philadelphia: 2007. pp. 1305–1335. [Google Scholar]
- 2.Gonzalez J.M., Gomez-Puertas P., Cavanagh D. A comparative sequence analysis to revise the current taxonomy of the family Coronaviridae. Arch Virol. 2003;148(11):2207–2235. doi: 10.1007/s00705-003-0162-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wise A.G., Kiupel M., Maes R.K. Molecular characterization of a novel coronavirus associated with epizootic catarrhal enteritis (ECE) in ferrets. Virology. 2006;349(1):164–174. doi: 10.1016/j.virol.2006.01.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wise A., Kiupel M., Garner M.M. Comparative sequence analysis of the distal one-third of the genomes of a systemic and an enteric ferret coronavirus. Virus Res. 2010;149(1):42–50. doi: 10.1016/j.virusres.2009.12.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Williams B., Kiupel M., West K. Coronavirus associated epizootic catarrhal enteritis in ferrets. J Am Vet Med Assoc. 2000;217(4):526–530. doi: 10.2460/javma.2000.217.526. [DOI] [PubMed] [Google Scholar]
- 6.Martinez J., Ramis A.J., Reinacher M. Detection of feline infectious peritonitis virus-like antigen in ferrets. Vet Rec. 2006;158(15):523. doi: 10.1136/vr.158.15.523-b. [DOI] [PubMed] [Google Scholar]
- 7.Garner M.M., Ramsell K., Morera N. Clinicopathologic features of a systemic coronavirus-associated disease resembling feline infectious peritonitis in the domestic ferret (Mustela putorius) Vet Pathol. 2008;45(2):236–246. doi: 10.1354/vp.45-2-236. [DOI] [PubMed] [Google Scholar]
- 8.Juan-Salles C., Teifke J.P., Morera N. Pathology and immunohistochemistry of a disease resembling feline infectious peritonitis in ferrets (Mustela putorius furo) Vet Pathol. 2006;43(5):845. doi: 10.1354/vp.45-2-236. [DOI] [PubMed] [Google Scholar]
- 9.Rottier P.J.M., Nakamura K., Schellen P. Acquisition of macrophage tropism during the pathogenesis of feline infectious peritonitis is determined by mutations in the feline coronavirus spike protein. J Virol. 2005;79:14122–14130. doi: 10.1128/JVI.79.22.14122-14130.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Brown M.A., Troyer J.L., Pecon-Slattery J. Genetics and pathogenesis of feline infectious peritonitis virus. Emerg Infect Dis. 2009;15(9):1445–1452. doi: 10.3201/eid1509.081573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Martínez J., Reinacher M., Perpiñán D. Identification of group 1 coronavirus antigen in multisystemic granulomatous lesions in ferrets (Mustela putorius furo) J Comp Pathol. 2008;138:54–58. doi: 10.1016/j.jcpa.2007.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hartmann K. Feline infectious peritonitis. Vet Clin North Am Small Anim Pract. 2005;35:39–79. doi: 10.1016/j.cvsm.2004.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Pedersen N.C. A review of feline infectious peritonitis virus infection: 1963–2008. J Feline Med Surg. 2009;11:225–258. doi: 10.1016/j.jfms.2008.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Perpinan D., Lopez C. Clinical aspects of systemic granulomatous inflammatory syndrome in ferrets (Mustela putorius furo) Vet Rec. 2008;162(6):180–183. doi: 10.1136/vr.162.6.180. [DOI] [PubMed] [Google Scholar]
- 15.Weiss R.C., Scott F.W. Pathogenesis of feline infectious peritonitis: pathologic changes and immunofluorescence. Am J Vet Res. 1981;42:2036–2048. [PubMed] [Google Scholar]
- 16.Murray J. Clinical management of systemic coronavirus in domestic ferrets. In: Proceedings of the 29th Annual AAV Conference & Expo with AEMV. Savannah (GA), August 11–14, 2008. p. 51–5.
- 17.Ramsell KD, Garner MM, Maes R, et al. A disease resembling feline infectious peritonitis in the domestic ferret. In: Proceedings of the International Ferret Symposium. Portland, June 22–24, 2007.
- 18.Garner M.M. Focus on diseases of ferrets. Exotic DVM. 2003;5(3):75–80. [Google Scholar]
- 19.Addie D., Belak S., Boucraut-Baralon C. Feline infectious peritonitis. ABCD guidelines on prevention and management. J Feline Med Surg. 2009;11:594–604. doi: 10.1016/j.jfms.2009.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Simons F.A., Vennema H., Rofina J.E. A mRNA PCR for the diagnosis of feline infectious peritonitis. J Virol Methods. 2005;124:111–116. doi: 10.1016/j.jviromet.2004.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tizard I. Veterinary immunology. 4th edition. WB Saunders; Philadelphia: 1992. Drugs that affect the immune system; pp. 447–456. [Google Scholar]
- 22.Gregory C.R. Immunosuppressive agents. In: Bonagura J.D., editor. Kirk's current veterinary therapy XIII. WB Saunders; Philadelphia: 2000. pp. 509–513. [Google Scholar]
- 23.Taylor H.G., Samanta A. Treatment of vasculitis. Br J Clin Pharmacol. 1993;35:93–104. doi: 10.1111/j.1365-2125.1993.tb05674.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Addie D.D., Ishida T. Feline infectious peritonitis: therapy and prevention. In: Bonagura J.D., Twedt D.C., editors. Kirk's current veterinary therapy XIV. Saunders Elsevier; St Louis (MO): 2009. pp. 1295–1298. [Google Scholar]
- 25.Hartmann K. Diagnosis and treatment of feline infectious peritonitis. In: August J.R., editor. vol. 6. Saunders Elsevier; St. Louis (MO): 2010. pp. 62–76. (Consultations in feline internal medicine). [Google Scholar]
- 26.Addie D.D., Jarett O. Feline coronavirus infections. In: Greene C.E., editor. Infectious diseases of the dog and cat. 3rd edition. Saunders Elsevier; St Louis (MO): 2006. pp. 88–102. [Google Scholar]
- 27.Jones C.J.P., Morris K.J., Jayson M.I.V. Prednisolone inhibits phagocytosis by polymorphonuclear leucocytes via steroid receptor mediated events. Ann Rheum Dis. 1983;42(1):56–62. doi: 10.1136/ard.42.1.56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Legendre A.M., Bartges J.W. Effect of polyprenol immunostimulant on the survival times of three cats with the dry form of feline infectious peritonitis. J Feline Med Surg. 2009;11(8):624–626. doi: 10.1016/j.jfms.2008.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Weiss R.C., Cox N.R., Oostrom-Ram T. Effect of interferon or Propionibacterium acnes on the course of experimentally induced feline infectious peritonitis in specific-pathogen-free and random-source cats. Am J Vet Res. 1990;51(5):726–733. [PubMed] [Google Scholar]
- 30.Ishida T., Shibanai A., Tanaka S. Use of recombinant feline interferon and glucocorticoid in the treatment of feline infectious peritonitis. J Feline Med Surg. 2004;6(2):107–109. doi: 10.1016/j.jfms.2003.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ritz S., Egberink H., Hartmann K. Effect of feline interferon-omega on the survival times and quality of life of cats with feline infectious peritonitis. J Vet Intern Med. 2007;21(6):1193–1197. doi: 10.1111/j.1939-1676.2007.tb01937.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kennedy M.A. An update on feline infectious peritonitis. Vet Med. 2009;104(8):384–391. [Google Scholar]
- 33.Kipar A., May H., Menger S. Morphologic features and development of granulomatous vasculitis in feline infectious peritonitis. Vet Pathol. 2005;42(3):321–330. doi: 10.1354/vp.42-3-321. [DOI] [PubMed] [Google Scholar]
- 34.Marsella R. Pentoxifylline. In: Bonagura J.D., Twedt D.C., editors. Kirk's current therapy XIV. Saunders Elsevier; St Louis (MO): 2009. pp. 397–400. [Google Scholar]
- 35.Scott D.W., Miller W.H., Griffin C.E. Muller & Kirk's small animal dermatology. 6th edition. WB Saunders; Philadelphia: 2001. Dermatologic therapy; p. 241. [Google Scholar]
- 36.Mendes J.B., Campos P.P., Rocha M.A. Cilostazol and pentoxifylline decrease angiogenesis, inflammation, and fibrosis in sponge-induced intraperitoneal adhesion in mice. Life Sci. 2009;84(15–16):537–543. doi: 10.1016/j.lfs.2009.01.023. [DOI] [PubMed] [Google Scholar]
- 37.Deree J., Lall R., Melbostad H. Neutrophil degranulation and effects of phosphodiesterase inhibition. J Surg Res. 2006;133(1):22–28. doi: 10.1016/j.jss.2006.02.031. [DOI] [PubMed] [Google Scholar]
- 38.Scott D.W., Miller W.H., Griffin C.E. Muller & Kirk's small animal dermatology. 6th edition. WB Saunders; Philadelphia: 2001. Immune-mediated disorders; pp. 742–756. [Google Scholar]
- 39.Fernandes J.L., de Oliveira R.T., Mamoni R.L. Pentoxifylline reduces pro-inflammatory and increases anti-inflammatory activity in patients with coronary artery disease-a randomized placebo-controlled study. Atherosclerosis. 2008;196(1):434–442. doi: 10.1016/j.atherosclerosis.2006.11.032. [DOI] [PubMed] [Google Scholar]
- 40.Duman D.G., Ozdemir F., Birben E. Effects of pentoxifylline on TNF-alpha production by peripheral blood mononuclear cells in patients with nonalcoholic steatohepatitis. Dig Dis Sci. 2007;52(10):2520–2524. doi: 10.1007/s10620-006-9723-y. [DOI] [PubMed] [Google Scholar]
- 41.Satapathy S.K., Sakhuja P., Malhotra V. Beneficial effects of pentoxifylline on hepatic steatosis, fibrosis and necroinflammation in patients with non-alcoholic steatohepatitis. J Gastroenterol Hepatol. 2007;22(5):634–638. doi: 10.1111/j.1440-1746.2006.04756.x. [DOI] [PubMed] [Google Scholar]
- 42.Maiti R., Agrawai N.K., Dash D. Effect of pentoxifylline on inflammatory burden, oxidative stress and platelet aggregability in hypertensive type 2 diabetes mellitus patients. Vascul Pharmacol. 2007;47(2–3):118–124. doi: 10.1016/j.vph.2007.05.004. [DOI] [PubMed] [Google Scholar]
- 43.Raffetto J.D., Khalil R.A. Matrix metalloproteinases and their inhibitors in vascular remoldeling and vascular disease. Biochem Pharmacol. 2008;75(2):346–359. doi: 10.1016/j.bcp.2007.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Mulivor A.W., Lipowsky H.H. Inhibition of glycan shedding and leukocyte-endothelial adhesion in postcapillary venules by suppression of matrixmetalloprotease activity with doxycycline. Microcirculation. 2009;4:1–10. doi: 10.3109/10739680903133714. [DOI] [PubMed] [Google Scholar]
- 45.Liu J., Xiong W., Baca-Regen L. Mechanism of inhibition of matrix metalloproteinase-2 expression by doxycycline in human aortic smooth muscle cells. J Vasc Surg. 2003;38(6):1376–1383. doi: 10.1016/s0741-5214(03)01022-x. [DOI] [PubMed] [Google Scholar]
- 46.Hackmann A.E., Rubin B.G., Sanchez L.A. A randomized, placebo-controlled trial of doxycycline after endoluminal aneurysm repair. J Vasc Surg. 2008;48(3):519–526. doi: 10.1016/j.jvs.2008.03.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Iovieno A., Lambiase A., Micera A. In vivo characterization of doxycycline effects on tear metalloproteinases in patients with chronic blepharitis. Eur J Ophthalmol. 2009;19(5):708–716. doi: 10.1177/112067210901900504. [DOI] [PubMed] [Google Scholar]
- 48.Lee H.M., Ciancio S.G., Tuter G. Subantimicrobial dose doxycycline efficacy as a matrix metalloproteinase inhibitor in chronic periodontitis patients is enhanced when combined with a non-steroidal anti-inflammatory drug. J Periodontal. 2004;75(3):453–463. doi: 10.1902/jop.2004.75.3.453. [DOI] [PubMed] [Google Scholar]
- 49.Gu Y., Lee H.M., Sorsa T. Doxycycline inhibits mononuclear cell-mediated connective tissue breakdown. FEMS Immunol Med Microbiol. 2010;58(2):218–225. doi: 10.1111/j.1574-695X.2009.00625.x. [DOI] [PubMed] [Google Scholar]
- 50.Lau A.C., Duong T.T., Ito S. Inhibition of matrix metalloproteinase-9 activity improves coronary outcome in an animal model of Kawasaki disease. Clin Exp Immunol. 2009;157(2):300–309. doi: 10.1111/j.1365-2249.2009.03949.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Hori Y., Kunihiro S., Sato S. Doxycycline attenuates isoproterenol-induced mycocardial fibrosis and matrix metalloproteinase activity in rats. Biol Pharm Bull. 2009;32(10):1678–1682. doi: 10.1248/bpb.32.1678. [DOI] [PubMed] [Google Scholar]
- 52.Lewington J.H. Ferret husbandry, medicine and surgery. 2nd edition. Saunders Elsevier; Sydney (Australia): 2007. Diseases of the ferret ear, eye and nose; p. 311. [Google Scholar]
- 53.Watari T., Kaneshima T., Tsujikoto H. Effect of thromboxane synthetase inhibitor on feline infectious peritonitis in cats. J Vet Med Sci. 1998;60(5):657–659. doi: 10.1292/jvms.60.657. [DOI] [PubMed] [Google Scholar]
- 54.Moraes L.A., Paul-Clark M.J., Rickman A. Ligand-specific glucocorticoid receptor activation in human platelets. Blood. 2005;106(13):4167–4175. doi: 10.1182/blood-2005-04-1723. [DOI] [PubMed] [Google Scholar]
- 55.Debets J.M., Ruers T.J., van der Linden M.P. Inhibitory effect of corticosteroids on the secretion of tumor necrosis factor (TNF) by monocytes is dependant on the stimulus inducing TNF synthesis. Clin Exp Immunol. 1989;78(2):224–229. [PMC free article] [PubMed] [Google Scholar]
- 56.Williams B.H. Therapeutics in ferrets. Vet Clin North Am Exot Anim Pract. 2000;3(1):131–153. doi: 10.1016/S1094-9194(17)30098-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Antinoff N., Hahn K. Ferret oncology: diseases, diagnostics, and therapeutics. Vet Clin North Am Exot Anim Pract. 2004;7(3):579–625. doi: 10.1016/j.cvex.2004.05.001. [DOI] [PubMed] [Google Scholar]
- 58.Bourinbalar A.S., Fruhstorfer E.C. The effect of histamine type 2 receptor antagonists on human immunodeficiency virus (HIV) replication: identification of a new class of antiviral agents. Life Sci. 1996;59(23):365–370. doi: 10.1016/s0024-3205(96)00553-x. [DOI] [PubMed] [Google Scholar]
- 59.Kabuta H., Yamamoto S., Shingu M. The effect of cimetidine on survival of mice infected with herpes simplex virus type 2, murine encephalomyelitis and vesicular stomatitis virus infections. Kurume Med J. 1989;36(3):95–99. doi: 10.2739/kurumemedj.36.95. [DOI] [PubMed] [Google Scholar]
- 60.Hirai N., Hill N.O., Motoo Y. Antiviral and antiproliferative activities of human leukocyte interferon potentiated by cimetidine in vitro. J Interferon Res. 1985;5(3):375–382. doi: 10.1089/jir.1985.5.375. [DOI] [PubMed] [Google Scholar]
- 61.Nishiguchi S., Tamori A., Shiomi S. Cimetidine reduces impairment of cellular immunity after transcatheter arterial embolization in patients with hepatocellular carcinoma. Hepatogastroenterology. 2003;50(50):460–462. [PubMed] [Google Scholar]
- 62.Katoh J., Tauchiya K., Osawa H. Cimetidine reduces impairment of cellular immunity after cardiac operations with cardiopulmonary bypass. J Thorac Cardiovasc Surg. 1998;116(2):312–318. doi: 10.1016/s0022-5223(98)70132-1. [DOI] [PubMed] [Google Scholar]
- 63.Powers L.V. Bacterial and parasitic diseases of ferrets. Vet Clin North Am Exot Anim Pract. 2009;12(3):531–561. doi: 10.1016/j.cvex.2009.06.001. [DOI] [PubMed] [Google Scholar]
- 64.Hoppes S, Xenoulis PG, Berghoff N, et al. Serum cobalamin, folate, and methylmalonic acid concentrations in ferrets (Mustela putorius). In: Proceedings of the 29th Annual AAV Conference & Expo with AEMV. Savannah (GA), August 11–14, 2008. p. 61–3.
- 65.Reiter R.J., Melchiorri D., Sewerynek E. A review of the evidence supporting melatonin's role as an antioxidant. J Pineal Res. 1995;18(1):1–11. doi: 10.1111/j.1600-079x.1995.tb00133.x. [DOI] [PubMed] [Google Scholar]
- 66.Reiter R.J., Tan D.X., Gitto E. Pharmacological utility of melatonin in reducing oxidative cellular and molecular damage. Pol J Pharmacol. 2004;56(2):158–170. [PubMed] [Google Scholar]
- 67.Zararsiz I., Sarsilmaz W., Tas U. Protective effects of melatonin against formaldehyde-induce kidney damage in rats. Toxicol Ind Health. 2007;23(10):573–579. doi: 10.1177/0748233708089022. [DOI] [PubMed] [Google Scholar]
- 68.Maldonado M.D., Mora-Santos M., Naji L. Evidence of melatonin synthesis and release by mast cells. Possible modulatory role on inflammation. Pharmacol Res. 2009 Dec 3 doi: 10.1016/j.phrs.2009.11.014. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 69.Lahiri S., Singh P., Singh S. Melatonin protects against experimental reflux esophagitis. J Pineal Res. 2009;46(2):207–213. doi: 10.1111/j.1600-079X.2008.00650.x. [DOI] [PubMed] [Google Scholar]
- 70.Ganguly K., Swamaker S. Induction of matrix metalloproteinase-9 and -3 in nonsteroidal anti-inflammatory drug-induced acute gastric ulcers in mice: regulation by melatonin. J Pineal Res. 2009;47(1):43–55. doi: 10.1111/j.1600-079X.2009.00687.x. [DOI] [PubMed] [Google Scholar]
- 71.Swamaker S., Mishra A., Ganguly K. Matrix metalloproteinase-9 activity and expression is reduced by melatonin during prevention of ethanol-induced gastric ulcer in mice. J Pineal Res. 2007;43(10):56–64. doi: 10.1111/j.1600-079X.2007.00443.x. [DOI] [PubMed] [Google Scholar]
- 72.Hung Y.C., Chen T.Y., Lee E.J. Melatonin decreases matrix metalloproteinase-9 activation and expression and attenuates reperfusion-induced hemorrhage following transient focal cerebral ischemia in rats. J Pineal Res. 2008;45(4):459–467. doi: 10.1111/j.1600-079X.2008.00617.x. [DOI] [PubMed] [Google Scholar]
- 73.Esposito E., Mazzon E., Riccardi L. Matrix metalloproteinase-9 and metalloproteinase-2 activity and expression is reduced by melatonin during experimental colitis. J Pineal Res. 2008;45(2):166–173. doi: 10.1111/j.1600-079X.2008.00572.x. [DOI] [PubMed] [Google Scholar]
- 74.Srinivasan V., Spence D.W., Trakht I. Immunomodulation by melatonin: its significance for seasonally occurring diseases. Neuroimmunomodulation. 2008;15(2):93–101. doi: 10.1159/000148191. [DOI] [PubMed] [Google Scholar]
- 75.Reiter R.J., Calvo J.R., Karbownik M. Melatonin and its relation to the immune system and inflammation. Ann N Y Acad Sci. 2000;917:376–386. doi: 10.1111/j.1749-6632.2000.tb05402.x. [DOI] [PubMed] [Google Scholar]
- 76.Maestroni G.J. Therapeutic potential of melatonin in immunodeficiency states, viral diseases, and cancer. Adv Exp Med Biol. 1999;467:217–226. doi: 10.1007/978-1-4615-4709-9_28. [DOI] [PubMed] [Google Scholar]
- 77.Bonilla E., Valero N., Chacin-Bonilla L. Melatonin and viral infections. J Pineal Res. 2004;36(2):73–79. doi: 10.1046/j.1600-079X.2003.00105.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Huang S.H., Cao X.J., Wei W. Melatonin decreases TLR3-mediated inflammatory factor expression via inhibition of NF-kappa B activation in respiratory syncytial virus-infected RAW264.7 macrophages. J Pineal Res. 2008;45(1):93–100. doi: 10.1111/j.1600-079X.2008.00560.x. [DOI] [PubMed] [Google Scholar]
- 79.Nunes O.S., Pereira R.S. Regression of herpes viral infection symptoms using melatonin and SB-73: comparison with acyclovir. J Pineal Res. 2008;44(4):373–378. doi: 10.1111/j.1600-079X.2007.00538.x. [DOI] [PubMed] [Google Scholar]
- 80.Webb C.B., Lehman T.L., McCord K.W. Effects of an oral superoxide dismutase enzyme supplementation on indices of oxidative stress, proviral load, and CD4:CD8 ratios in asymptomatic FIV-infected cats. J Feline Med Surg. 2008;10(5):423–430. doi: 10.1016/j.jfms.2008.01.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Webster C.R., Cooper J. Therapeutic use of cytoprotective agents in canine and feline hepatobiliary disease. Vet Clin North Am Small Anim Pract. 2009;39(3):631–652. doi: 10.1016/j.cvsm.2009.02.002. [DOI] [PubMed] [Google Scholar]
- 82.Gisselman K., Langston C., Palma D. Calcium oxalate urolithiasis. Compend Contin Educ Pract Vet. 2009;31(11):496–500. [PubMed] [Google Scholar]
- 83.Osborne C.A., Albasan H., Lulich J.P. Quantitative analysis of 4468 uroliths retrieved from farm animals, exotic species, and wildlife submitted to the Minnesota Urolith center: 1981–2007. Vet Clin North Am Small Anim Pract. 2008;39:65–78. doi: 10.1016/j.cvsm.2008.09.005. [DOI] [PubMed] [Google Scholar]


