Abstract
Porcine epidemic diarrhea virus (PEDV), a member of the Coronaviridae family, causes acute diarrhea, vomiting, dehydration, and high mortality rates in neonatal piglets. Severe outbreaks of PEDV variants have re-emerged in Asia and North America since 2010, causing tremendous economic losses to the swine industry. The lack of effective therapeutic treatment promotes the research for new antivirals. Lithium chloride (LiCl) has been reported as a potential antiviral drug for certain viruses. In this study, the antiviral effect of LiCl on PEDV in Vero cells was evaluated. Real-time quantitative PCR and indirect immunofluorescence assay indicated that LiCl effectively inhibited the entry and replication of PEDV in Vero cells. The expression of viral RNA and protein of PEDV in Vero cells was suppressed in a dose-dependent manner by LiCl. Moreover, addition of LiCl inhibited both early and late cell apoptosis induced by PEDV. Our data implied that LiCl could be a potential antiviral drug against PEDV infection. Further studies are required to explore the antiviral effect of lithium chloride on PEDV infection in vivo.
Keywords: Porcine epidemic diarrhea virus, Lithium chloride, Antiviral activity, Apoptosis
Highlights
-
•
LiCl has antiviral activity against PEDV in vitro.
-
•
The anti-PEDV activity of LiCl may be mediated by inhibiting cell apoptosis induced by PEDV.
-
•
LiCl could be a potential antiviral drug against PEDV infection.
1. Introduction
Porcine epidemic diarrhea virus (PEDV), the etiological agent of porcine epidemic diarrhea (PED), belongs to the family Coronaviridae and causes acute watery diarrhea, vomiting, dehydration, and high mortality rates in neonatal piglets (Lee, 2015.). PEDV is an enveloped virus with a single-stranded positive sense RNA genome of approximately 28 kb in size, and it possesses four structural proteins: the glycosylated spike (S), envelope (E), membrane (M) proteins and the unglycosylated nucleocapid (N) protein (Jung and Saif, 2015). PEDV has circulated throughout Europe and Asia during the past four decades, and has caused devastating impacts on world pig-rearing industry (Collin et al., 2015). Currently, vaccination is the primary measure for prevention of PEDV. However, genetic variants have been identifying in Asia, Europe and North America since 2010 (Hanke et al., 2015; Horie et al., 2016; Huang et al., 2013; Lee and Lee, 2014; Li et al., 2012; Sung et al., 2015; Trujillo-Ortega et al., 2016; Vui et al., 2015; Wang et al., 2013;), which makes current traditional inactivated and attenuated PEDV vaccines are less effective against circulating PEDV variants (Choi et al., 2009; Lee, 2015). The lack of effective therapeutic treatment underlines the importance of research for new antivirals.
Lithium chloride (LiCl) has been used as a drug for treatment of bipolar disorder, depression, Alzheimer's disease and diabetes (Curran and Ravindran, 2014; Lavoie et al., 2015; Munoz-Montano et al., 1997). Antiviral effects of LiCl have been extensively investigated. LiCl can inhibit the infection of some DNA and RNA viruses, such as herpes simplex virus (Skinner et al., 1980), canine parvovirus (Zhou et al., 2015), porcine parvovirus (Chen et al., 2015), pseudorabies herpesvirus (Sui et al., 2010), porcine reproductive and respiratory syndrome virus (Hao et al., 2015), feline calicivirus (Wu et al., 2015), infectious bronchitis virus, an avian coronavirus (Li et al., 2009) and transmissible gastroenteritis virus (TGEV), a porcine coronavirus (Ren et al., 2011). Both PEDV and TGEV are classified into group I of the genus Alphacoroavirus. However, it remains unknown whether LiCl has inhibitory effects on PEDV infection. In this study, we demonstrated the inhibitory effect of LiCl on PEDV infection. Moreover, the effect of LiCl on the cell apoptosis induced by PEDV was investigated. These data indicated that LiCl might be a potential compound for reducing PEDV infection.
2. Materials and methods
2.1. Cells, virus and drug
Vero cells (African green monkey kidney cells) (CCL-81, ATCC, USA) were maintained in Dulbecco's modified Eagle's medium (DMEM, Gibco) supplemented with 10% fetal bovine serum (FBS, Gibco), 100 U/ml of penicillin and 100 μg/ml of streptomycin at 37 °C in 5% CO2 incubator. PEDV Chinese isolate CH/ZMDZY/11 was propagated in Vero cells as described (Wang et al., 2013). Lithium chloride (LiCl) was purchased from Sigma-Aldrich (Sigma, St. Louis, MO, USA) and was dissolved in DMEM and sterilized by passage through a 0.22 μm filter.
2.2. Virus titration
Vero cells were seeded into 96-well plates 24 h before infection. Cell lysates from the experiments were serially diluted in DMEM and were added to the Vero cells. After incubation at 37 °C for 1 h, the cells were washed three times with PBS then incubated at 37 °C for 72 h, the 50% tissue culture infectious dose (TCID50) was calculated by the method of Reed and Muench.
2.3. Cytotoxicity assay for LiCl
Cytotoxic effect of LiCl was determined using the Cell Counting Kit-8 (Dojindo, Kumamoto, Japan) according to the manufacturer's instructions. Briefly, Vero cells in 96-well plate were treated with LiCl at a series of concentrations (5, 10, 15, 20, 30, 40, 50 mM) in serum-free DMEM for 48 h. Mock-treated Vero cells served as a control. After washing with PBS, the cells were incubated with 80 μL medium and 10 μL of CCK-8 solution at 37 °C for 1 h. The absorbance was measured at a wavelength of 450 nm using a microplate reader (Thermo Scientific, USA). The relative cell viability rate was calculated as (mean OD450 drug)/(mean OD450 control) × 100%. LiCl concentrations below the 50% cytostatic concentration (CC50) were defined as non-toxic concentrations.
2.4. Effect of LiCl on viral infection
In order to investigate whether LiCl affects infection of PEDV to Vero cells, Vero cells were treated with a mixture of nontoxic concentrations (1, 5, 10, 15 mM) of LiCl and PEDV (10−3 TCID50/cell) at 37 °C for 40 h. As a control, cells were infected with the same dose of PEDV without LiCl. Subsequently, the antiviral efficacy was evaluated by analysis of viral RNA levels, protein expression level and virus loads.
2.5. Effect of LiCl on viral attachment
To evaluate the effect of LiCl on attachment of PEDV to Vero cells, Vero cells were treated with a mixture of nontoxic concentrations (1, 5, 10, 15 mM) of LiCl and PEDV (10−3 TCID50/cell) at 4 °C for 1 h, which allowed the viruses to bind to the surface of cells but not enter the cells. As a control, cells were infected with the same dose of PEDV without LiCl treatment. After removing the drug and the unbound viruses by washing with cold DMEM, the Vero cell were subjected to three freeze-thaw cycles in preparation for measuring viral RNA levels and virus loads.
2.6. Effect of LiCl on viral entry
To determine whether LiCl affects entry of PEDV into Vero cells, Vero cells were infected with PEDV (10−3 TCID50/cell) at 4 °C for 1 h. After removing the unbound viruses by washing with cold DMEM, the cells were incubated with nontoxic concentrations (1, 5, 10, 15 mM) of LiCl at 37 °C for 40 h. As a control, cells were infected with the same dose of PEDV without LiCl treatment. Subsequently, the antiviral efficacy was evaluated by analysis of viral RNA levels and virus loads.
2.7. Effect of LiCl on viral replication
To evaluate whether LiCl affects replication of PEDV to Vero cells, Vero cells were infected with PEDV (1 × 10−3 TCID50/cell) at 37 °C for 1 h. After removing the unbound viruses with DMEM, the cells were incubated with nontoxic concentrations (1, 5, 10, 15 mM) of LiCl at 37 °C for 40 h. As a control, cells were infected with the same dose of PEDV without LiCl treatment. Subsequently, the antiviral efficacy was evaluated by analysis of viral RNA levels and viral loads.
2.8. Real-time quantitative PCR
To determine the inhibitory effects of LiCl on PEDV infection, real-time quantitative PCR was performed. PEDV-infected Vero cells treated by LiCl were subjected to RNA extraction with a QIAamp Viral RNA Mini Kit (Qiagen, Germany). Subsequent reverse transcription was performed using random primers and the amplification included 42 °C for 1 h and a final extension at 72 °C for l0 min. The reverse transcription products were subjected to real-time PCR. Real-time quantitative PCR, targeting the S gene of PEDV was carried out with the primers shown in Table 1 , using a Lightcycler 1.5 Real Time PCR System (Roche, Germany) and SYBR Premix Ex Taq (TaKaRa, Janpan) according to the instructions of the manufacturer. The relative RNA expression levels were calculated by the 2−∆∆Ct method, using beta-actin as an internal control for normalization. The mean RNA level of the mock-treated group was set at 1.00.
Table 1.
Sequences of the primers used for real-time quantitative PCR.
| Primer pairs | PCR product in length (bp) |
|---|---|
| Sense 5′-TAACAAAACACTTGATGAGATT-3′ | S (250) |
| Antisense 5′-CCCACCACGGCCACTTGATATATG-3′ | |
| Sense 5′- AGCAAGCAGGAGTATGACGAGT −3′ | Beta-actin (93) |
| Antisense 5′- CAAGAAAGGGTGTAACGCAACT −3′ |
2.9. Indirect immunofluorescence assay (IFA)
The IFA was performed to confirm the inhibitory effects of LiCl on PEDV replication. After washing with PBS, the Vero cells from the PEDV replication stage assay that had been treated with nontoxic concentrations of LiCl were fixed with 4% paraformaldehyde in PBS for 15 min and then permeabilized with 0.2% Triton X-100 in PBS for 10 min. After washing, the Vero cells were incubated with rabbit anti-S antibody (1:100) for 1 h. Subsequently, FITC-conjugated goat anti-rabbit IgG (1:100) (Boster, China) was used as secondary antibody. After washing three times with PBS, nuclear staining with 4′, 6-diamidino-2-phenylindole (DAPI) was performed for reference protein expression according to instructions of the manufacturer (Boster, China). After washing, fluorescence was observed under a Nikon ECLIPSE TS100 fluorescence microscope (Nikon, Japan).
2.10. Fluorescence analysis of cell apoptosis
Cell apoptosis was analyzed with Annexin V-FITC/PI Kit (Nanjing Keygen Biotech, China). Annexin V has a high affinity for the membrane phosphatidylserine which was translocated from the inner face of plasma membrane to the cell surface at early stage of cell apoptosis, and FITC-labeled Annexin V could be used as a fluorescent probe to detect cell early apoptosis by fluorescence microscopy. Propidium iodide (PI) can pass through the membrane of apoptotic and necrotic cells, and can be used to detect cellular late apoptosis. A combination of Annexin V and PI was used to distinguish cellular early and late apoptosis. Briefly, FITC-labeled Annexin V (50 μl/well) and PI (50 μl/well) was added to the cells infected by LiCl-treated PEDV, or PEDV-infected cells treated with LiCl, and incubated at room temperature for 15 min in the dark prior to fluorescence observation. Fluorescence was observed under a Nikon ECLIPSE TS100 fluorescence microscope (Nikon, Japan).
2.11. Statistical analysis
All experiments were performed in triplicate, and the results were reported as the mean ± standard deviation (SD). The significance of differences between experimental groups was determined using an unpaired t-test and a one-way ANOVA using Prism 6.0 software. A p-value < 0.05 was selected to indicate significance.
3. Results
3.1. Cytotoxicity of LiCl on Vero cells
The cytotoxic effect of LiCl on Vero cells was determined by using the CCK8 kit according to the instruction s of the manufacture. The relative cell viability was above 90% after treatment with LiCl at concentrations of 5, 10 and 15 mM for 48 h, whereas the viability was under 50% after treatment with 20, 30, 40, 50 and 50 mM LiCl for 48 h (Fig. 1 ). A LiCl concentration under the 50% cytostatic concentration (CC50) that inhibited the proliferation of exponentially growing cells by 50% was defined as a non-toxic concentration. At a concentration of 15 mM LiCl, no difference of cell morphology compared with mock-treated cells was observed (data not shown), therefore 1-15 mM was chosen as the nontoxic concentration range of LiCl for the subsequent antiviral assays.
Fig. 1.
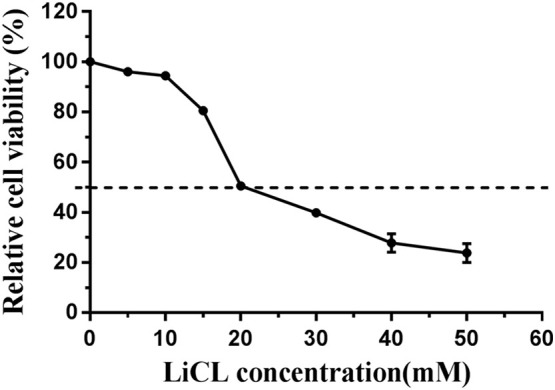
The cytotoxic effect of LiCl treatment on Vero cells. The cells were treated with 0, 5, 10, 15, 20, 30, 40 and 50 mM LiCl for 48 h. The relative cell viability rate was calculated as (mean OD450 drug)/(mean OD450nm control) × 100%. Data are expressed as the mean ± S.D. of three independent experiments. The dotted line indicates the 50% cytostatic concentration (CC50).
3.2. LiCl inhibits PEDV infection
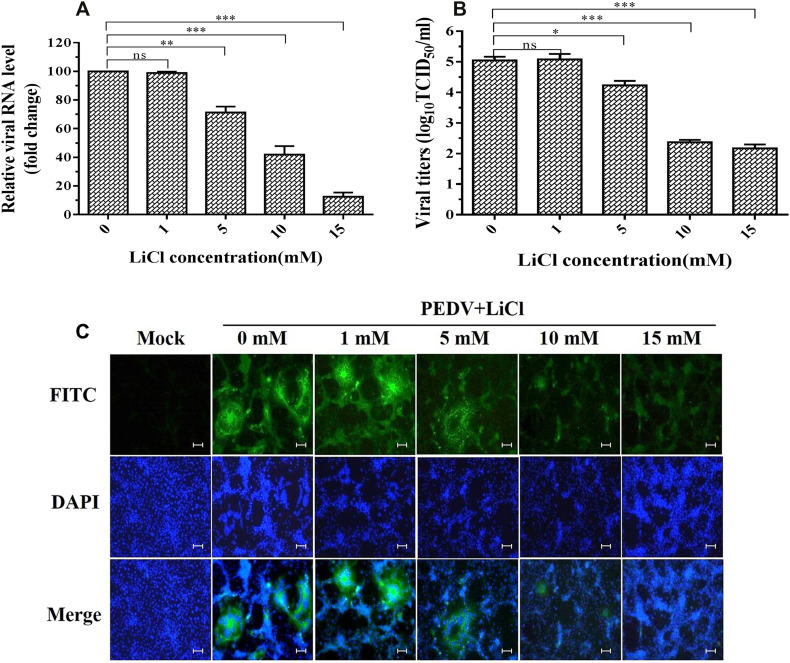
To investigate the antiviral activity of LiCl against PEDV, a mixture of PEDV with LiCl in an appropriate concentration (1, 5, 10, 15 mM) was added to the cultured Vero cells. The inhibitory effects of LiCl were evaluated in different level. In RNA level detected by real-time qPCR assays, the mean relative viral mRNA level of mock-treated cells and cells treated with 1, 5, 10, 15 mM LiCl was 100.00%, 98.99%, 71.32%, 41.90% and 12.59% (with mock-treated cells set as 100%), respectively (Fig. 2A). Viral titers of PEDV from mock-treated cells and those treated with 1, 5, 10, 15 mM were 5.05, 5.08, 4.23, 2.37, and 2.17 log10TCID50/ml, respectively (Fig. 2B). In indirect immunofluorescence assays (IFA), mock-treated Vero cells produced stronger fluorescent signals at 40 h after infection with PEDV. The fluorescence signals declined after treatment with 5, 10, 15 mM LiCl in a dose dependent manner (Fig. 2C).
Fig. 2.
Inhibitory effects of LiCl on PEDV infection. A. The viral load in Vero cells treated with different concentrations of LiCl, determined by qPCR. The relative viral RNA level determined by real-time qPCR. B. The viral titer (log10TCID50/ml) in Vero lysates calculated by the method of Reed and Muench. C. PEDV replication in Vero cells determined by IFA. Green fluorescence represents the PEDV distribution, and the blue fluorescence represents the nuclear distribution. Values represent the mean ± SD for three independent experiments. ns, no significant difference; *P < 0.05; **P < 0.001; ***P < 0.0001. Scale bar, 200 μm. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
3.3. LiCl inhibits PEDV entry and replication
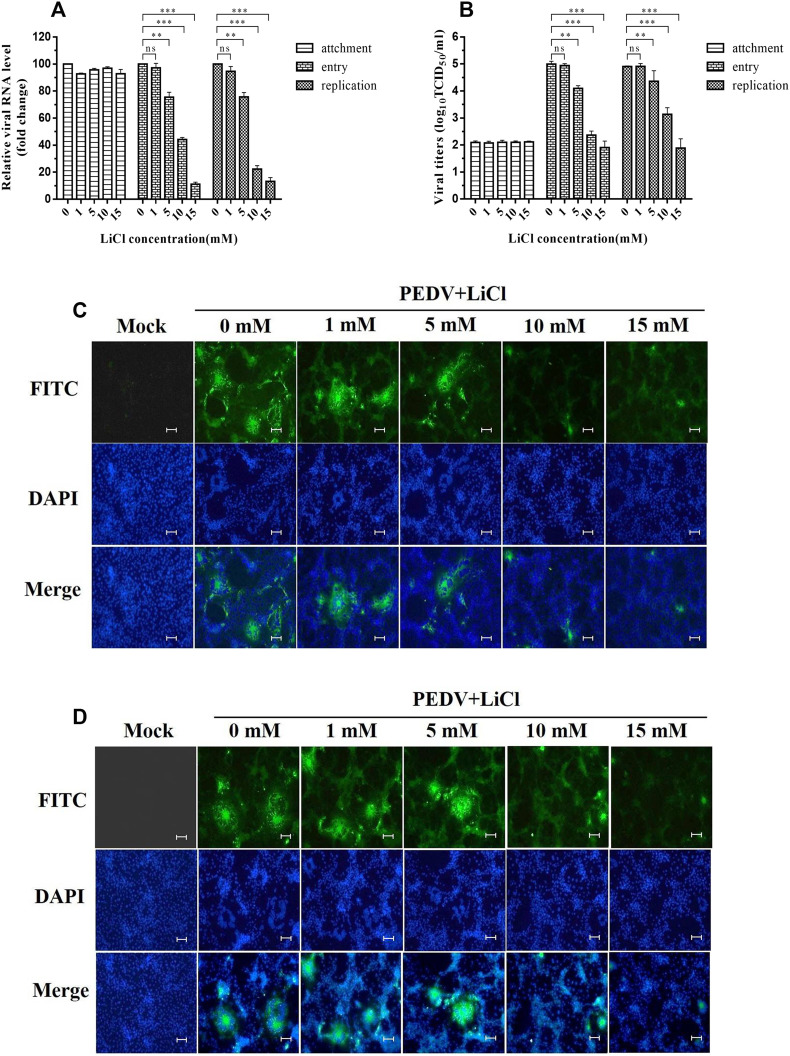
In order to determine which step in the viral life cycle LiCl affects, viral attachment, entry, and replication assays were performed in Vero cells. In viral attachment tests, the relative levels of viral RNA in mock-treated cells and those treated with 1, 5, 10, and 15 mM LiCl were 100%, 92.87%, 96.03%, 97.5%, 92.5%, respectively, and the viral titers of mock-treated cells and those treated with 1, 5, 10, and 15 mM LiCl were 2.1, 2.08, 2.1, 2.11, 2.03, respectively. The result showed that treatment with LiCl at attachment stage did not significantly affect the relative PEDV genomic RNA levels compared with mock-treated Vero cells, indicating that LiCl had no effect on PEDV attachment to Vero cells (Fig. 3A and B).
Fig. 3.
Inhibitory Effects of LiCl on different stages of PEDV infection. A. Relative viral RNA levels of cells treated with LiCl at the viral attachment, entry and replication stages as determined by real-time PCR. B. Viral titers (log10TCID50/ml) of cells treated with LiCl at the viral attachment, entry and replication stages as calculated by the method of Reed and Muench. C. Inhibitory effects of LiCl on PEDV observed at the entry stage using immunofluorescence assays (IFA). D. Inhibitory effects of LiCl on PEDV observed at the replication stage using immunofluorescence assay (IFA). Values represent the mean ± SD for three independent experiments. Green fluorescence represents the PEDV distribution, and the blue fluorescence represents the nuclear distribution. ns, no significant difference; *P < 0.05; **P < 0.01; ***P < 0.001. Scale bar, 200 μm. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
In viral entry tests, the relative levels of viral S gene RNA in mock-treated cells and those treated with 1, 5, 10, and 15 mM LiCl were 100.00%, 97.28%, 75.58% 44.23% and 11.20%, respectively (Fig. 3A). The viral titers of mock-treated cells and those treated with 1, 5, 10, and 15 mM LiCl were 4.99, 4.94, 4.10, 2.3, and 1.91 log10TCID50/ml, respectively (Fig. 3B). These results indicated that LiCl inhibited PEDV entry into cells in a dose-dependent manner.
In viral replication tests, the relative levels of viral RNA in mock-treated cells and those treated with 1, 5, 10, and 15 mM LiCl were 100.00%, 94.687%, 75.78%, 22.39% and 13.20%, respectively (Fig. 3A). The viral titers of mock-treated cells and those treated with 1, 5, 10, and 15 mM LiCl were 4.92, 4.91, 4.36, 3.14 and 1.89 log10TCID50/ml, respectively (Fig. 3B). These results indicated that LiCl inhibited PEDV replication in infected cells in a dose-dependent manner.
The results of indirect immunofluorescence assays (IFA) also confirmed that LiCl inhibited PEDV infection at entry and replication stages. At each stage, mock-treated Vero cells produced stronger fluorescent signals than those LiCl-treated Vero cells after infection with PEDV. The fluorescence signals declined after treatment with 5, 10, 15 mM LiCl in a dose dependent manner (Fig. 3C and D).
3.3.1. Lithium chloride inhibits Vero cell apoptosis caused by PEDV infection
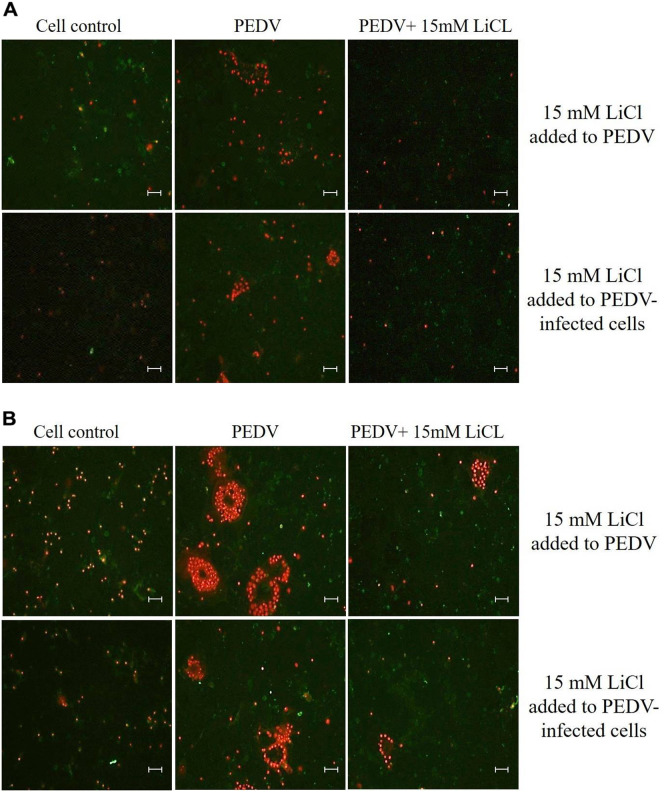
Apoptosis was detected by Annexin V-FITC/PI kit, FITC-labeled Annexin V was used to detect cell early apoptosis and PI was used to detect cell late apoptosis. The results showed that PEDV induced early apoptosis and late apoptosis in infected cells at 24 and 48 h post infection. LiCl inhibited early and late apoptosis of cells induced by PEDV infection and replication in a dose–dependent manner as shown by IFA. LiCl at a concentration of 15 mM significantly inhibited early and late apoptosis of cells induced by PEDV (Fig. 4 ).
Fig. 4.
The effect of LiCl on cell apoptosis analyzed by fluorescence staining. The cell apoptosis was analyzed after the cells were infected by LiCl-treated PEDV or after PEDV-infected cells were treated with LiCl at maximum non-toxic concentration. The green fluorescence signals are designated as the index of early apoptosis and the red fluorescence signals are designated as the index of late apoptosis. A representative staining picture regarding the effect of LiCl with maximum non-toxic concentration on the early apoptosis (24 h post-infection) and late apoptosis (48 h post-infection) are shown in Panel A and Panel B, respectively. Scale bar, 200 μm. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
4. Discussion
Porcine epidemic diarrhea virus (PEDV) is the predominant cause of severe enteropathogenic diarrhea in swine, and causes substantial economic losses in the pig industry. Re-emergence of epidemic PED indicates that PEDV is effectively able to escape from the current vaccination protocols, biosecurity and control systems (Jung and Saif, 2015). Vaccination is a fundamental strategy for controlling and eradicating PED during epidemic or endemic outbreaks, but the efficacy is limited (Baek et al., 2016; Shen et al., 2015). Until now there is no recorded clinically effective antiviral chemotherapeutic agent for treatment of disease caused by PEDV, even several compounds were demonstrated having antiviral activity in PEDV-infected Vero cells (Deejai et al., 2017; Yang et al., 2015; Lee et al., 2015; Kwon et al., 2013; Choi et al., 2009). So it is important to develop an effective strategy against PEDV infection.
Previous studies have shown that LiCl can inhibit the infection of coronaviruses (Li et al., 2009; Ren et al., 2011). However, it remains unknown whether LiCl has inhibitory effects on PEDV, a porcine coronavirus. In this study, we demonstrated the inhibitory effect of LiCl on PEDV infection. LiCl inhibits PEDV infection of Vero cells. After LiCl treatment, both viral RNA and viral protein levels decreased. When effects of LiCl on the stages of PEDV life cycles were evaluated, the real-time PCR, IFA and Viral titration results showed that LiCl significantly inhibited the entry and replication of PEDV in Vero cells in a dose-dependent manner. However LiCl had no effect on attachment of PEDV to Vero cells at every concentration tested, indicating that it did not directly affect the attachment of the virus to the cell receptor.
Apoptosis is considered to be a host innate defense mechanism that eliminates virus-infected cells. However, some viruses possess the ability to actively induce apoptosis to facilitate the release and dissemination of viral progeny to neighboring cells for further invasion, which are important biological parts in viral pathogenesis and disease processes that promote cell death and tissue injury (DeDiego et al., 2011; Favreau et al., 2012; Lan et al., 2013). PEDV induced caspase-independent apoptosis through activation of mitochondrial apoptosis-inducing factor has been demonstrated (Kim and Lee, 2014). In this study, we re-confirmed that PEDV infection induced the appearance of apoptotic cells as shown by IFA, and most apoptotic cells were at a stage of late apoptosis at 48 h post-infection. LiCl had a potent inhibitory effect on PEDV-induced early and late cell apoptosis. This is in accordance with the results from TGEV, another porcine coronavirus (Ren et al., 2011). Regarding the action mechanisms of LiCl on cell infection by PEDV, our data implies that LiCl may inhibit PEDV infection via exerting an anti-apoptotic effect, since a caspase-independent mitochondrial apoptosis-mediated pathway plays a central role in PEDV-induced apoptosis to facilitate viral replication. This is conflict with the results of the expression of caspase-3 was inhibited by LiCl in TGEV-infected cells. Apoptosis-associated caspase activation of TGEV and SARS-CoV has been reported. Maybe LiCl can act on multiple factors in the apoptosis pathway or PEDV induces apoptosis through different pathway. However the exact action mechanism of LiCl on PEDV-induced apoptosis needs to be defined.
In conclusion, this study has demonstrated that PEDV infection was inhibited in a dose-dependent manner by LiCl treatment. The antiviral effect of LiCl was at the phase of PEDV entry and replication in Vero cells. These results indicate that LiCl has potential as an effective anti-PEDV drug. Further research will be required to explore whether LiCl has an antiviral effect and the mechanism of the antiviral effect of LiCl on PEDV infection in vivo.
Competing interests
We confirm that there are no known conflicts of interest associated with this publication.
Acknowledgements
This research was supported by the Program for Science&Technology Innovation Team in the Universities of Henan Province (No. 14IRTSTHN015); Key Scientific Research Project of Henan Higher Education (No. 16A230002); International cooperation and exchange project for Zhengzhou city (No.·153PGJHZ203).
References
- Baek P.S., Choi H.W., Lee S., Yoon I.J., Lee Y.J., Lee du S., Lee S., Lee C. Efficacy of an inactivated genotype 2b porcine epidemic diarrhea virus vaccine in neonatal piglets. Vet. Immunol. Immunopathol. 2016;174:45–49. doi: 10.1016/j.vetimm.2016.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y., Yan H., Zheng H., Shi Y., Sun L., Wang C., Sun J. Antiviral effect of lithium chloride on infection of cells by porcine parvovirus. Arch. Virol. 2015;160:1015–1020. doi: 10.1007/s00705-015-2352-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi H.J., Kim J.H., Lee C.H., Ahn Y.J., Song J.H., Baek S.H., Kwon D.H. Antiviral activity of quercetin 7-rhamnoside against porcine epidemic diarrhea virus. Antivir. Res. 2009;81:77–81. doi: 10.1016/j.antiviral.2008.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collin E.A., Anbalagan S., Okda F., Batman R., Nelson E., Hause B.M. An inactivated vaccine made from a U.S. field isolate of porcine epidemic disease virus is immunogenic in pigs as demonstrated by a dose-titration. BMC Vet. Res. 2015;11:62. doi: 10.1186/s12917-015-0357-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curran G., Ravindran A. Lithium for bipolar disorder: a review of the recent literature. Expert. Rev. Neurother. 2014;14:1079–1098. doi: 10.1586/14737175.2014.947965. [DOI] [PubMed] [Google Scholar]
- DeDiego M.L., Nieto-Torres J.L., Jiménez-Guardeño J.M., Regla-Nava J.A., Alvarez E., Oliveros J.C., Zhao J., Fett C., Perlman S., Enjuanes L. Severeacute respiratory syndrome coronavirus envelope protein regulates cell stress response and apoptosis. PLoS Pathog. 2011;7 doi: 10.1371/journal.ppat.1002315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deejai N., Roshorm Y.M., Kubera A. Antiviral compounds against nucleocapsid protein of porcine epidemic diarrhea virus. Anim. Biotechnol. 2017;28:120–130. doi: 10.1080/10495398.2016.1232268. [DOI] [PubMed] [Google Scholar]
- Favreau D.J., Meessen-Pinard M., Desforges M., Talbot P.J. Human coronavirus-induced neuronal programmed cell death is cyclophilinddependent and potentially caspase dispensable. J. Virol. 2012;86:81–93. doi: 10.1128/JVI.06062-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanke D., Jenckel M., Petrov A., Ritzmann M., Stadler J., Akimkin V., Blome S., Pohlmann A., Schirrmeier H., Beer M., Hoper D. Comparison of porcine epidemic diarrhea viruses from Germany and the United States, 2014. Emerg. Infect. Dis. 2015;21:493–496. doi: 10.3201/eid2103.141165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hao H.P., Wen L.B., Li J.R., Wang Y., Ni B., Wang R., Wang X., Sun M.X., Fan H.J., Mao X. LiCl inhibits PRRSV infection by enhancing Wnt/beta-catenin pathway and suppressing inflammatory responses. Antivir. Res. 2015;117:99–109. doi: 10.1016/j.antiviral.2015.02.010. [DOI] [PubMed] [Google Scholar]
- Horie M., Kabemura M., Masatani T., Matsuu A., Ozawa M. Isolation and molecular characterization of porcine epidemic diarrhea viruses collected in Japan in 2014. Arch. Virol. 2016;161:2189–2195. doi: 10.1007/s00705-016-2900-1. [DOI] [PubMed] [Google Scholar]
- Huang Y.W., Dickerman A.W., Pineyro P., Li L., Fang L., Kiehne R., Opriessnig T., Meng X.J. Origin, evolution, and genotyping of emergent porcine epidemic diarrhea virus strains in the United States. MBio. 2013;4:e00737–00813. doi: 10.1128/mBio.00737-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung K., Saif L.J. Porcine epidemic diarrhea virus infection: etiology, epidemiology, pathogenesis and immunoprophylaxis. Vet. J. 2015;204:134–143. doi: 10.1016/j.tvjl.2015.02.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim Y., Lee C. Porcine epidemic diarrhea virus induces caspase-independent apoptosis through activation of mitochondrial apoptosis-inducing factor. Virology. 2014;460–461:180–193. doi: 10.1016/j.virol.2014.04.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwon H.J., Ryu Y.B., Kim Y.M., Song N., Kim C.Y., Rho M.C., Jeong J.H., Cho K.O., Lee W.S., Park S.J. In vitro antiviral activity of phlorotannins isolated from Ecklonia cava against porcine epidemic diarrhea coronavirus infection and hemagglutination. Bioorg. Med. Chem. 2013;21:4706–4713. doi: 10.1016/j.bmc.2013.04.085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lan Y., Zhao K., Wang G., Dong B., Zhao J., Tang B., Lu H., Gao W., Chang L., Jin Z., Gao F., He W. Porcine hemagglutinating encephalomyelitis virus induces apoptosis in a porcine kidney cell line via caspase-dependent pathways. Virus Res. 2013;176:292–297. doi: 10.1016/j.virusres.2013.05.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lavoie J., Hebert M., Beaulieu J.M. Looking beyond the role of glycogen synthase kinase-3 genetic expression on electroretinogram response: what about lithium? Biol. Psychiatry. 2015;77:e15–17. doi: 10.1016/j.biopsych.2014.07.024. [DOI] [PubMed] [Google Scholar]
- Lee C. Porcine epidemic diarrhea virus: an emerging and re-emerging epizootic swine virus. Virol. J. 2015;12:193. doi: 10.1186/s12985-015-0421-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee S., Lee C. Outbreak-related porcine epidemic diarrhea virus strains similar to US strains, South Korea, 2013. Emerg. Infect. Dis. 2014;20:1223–1226. doi: 10.3201/eid2007.140294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee J.H., Park J.S., Lee S.W., Hwang S.Y., Young B.E., Choi H.J. Porcine epidemic diarrhea virus infection: inhibition by polysaccharide from Ginkgo biloba exocarp and mode of its action. Virus Res. 2015;195:148–152. doi: 10.1016/j.virusres.2014.09.013. [DOI] [PubMed] [Google Scholar]
- Li J., Yin J., Sui X., Li G., Ren X. Comparative analysis of the effect of glycyrrhizin diammonium and lithium chloride on infectious bronchitis virus infection in vitro. Avian Pathol. 2009;38:215–221. doi: 10.1080/03079450902912184. [DOI] [PubMed] [Google Scholar]
- Li W., Li H., Liu Y., Pan Y., Deng F., Song Y., Tang X., He Q. New variants of porcine epidemic diarrhea virus, China, 2011. Emerg. Infect. Dis. 2012;18:1350–1353. doi: 10.3201/eid1808.120002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munoz-Montano J.R., Moreno F.J., Avila J., Diaz-Nido J. Lithium inhibits Alzheimer's disease-like tau protein phosphorylation in neurons. FEBS Lett. 1997;411:183–188. doi: 10.1016/s0014-5793(97)00688-1. [DOI] [PubMed] [Google Scholar]
- Ren X., Meng F., Yin J., Li G., Li X., Wang C., Herrler G. Action mechanisms of lithium chloride on cell infection by transmissible gastroenteritis coronavirus. PLoS One. 2011;6 doi: 10.1371/journal.pone.0018669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen H., Zhang C., Guo P., Liu Z., Zhang J. Effective inhibition of porcine epidemic diarrhea virus by RNA interference in vitro. Virus Genes. 2015;51:252–259. doi: 10.1007/s11262-015-1242-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skinner G.R., Hartley C., Buchan A., Harper L., Gallimore P. The effect of lithium chloride on the replication of herpes simplex virus. Med. Microbiol. Immunol. 1980;168:139–148. doi: 10.1007/BF02121762. [DOI] [PubMed] [Google Scholar]
- Sui X., Yin J., Ren X. Antiviral effect of diammonium glycyrrhizinate and lithium chloride on cell infection by pseudorabies herpesvirus. Antivir. Res. 2010;85:346–353. doi: 10.1016/j.antiviral.2009.10.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sung M.H., Deng M.C., Chung Y.H., Huang Y.L., Chang C.Y., Lan Y.C., Chou H.L., Chao D.Y. Evolutionary characterization of the emerging porcine epidemic diarrhea virus worldwide and 2014 epidemic in Taiwan. Infect. Genet. Evol. 2015;36:108–115. doi: 10.1016/j.meegid.2015.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trujillo-Ortega M.E., Beltran-Figueroa R., Garcia-Hernandez M.E., Juarez-Ramirez M., Sotomayor-Gonzalez A., Hernandez-Villegas E.N., Becerra-Hernandez J.F., Sarmiento-Silva R.E. Isolation and characterization of porcine epidemic diarrhea virus associated with the 2014 disease outbreak in Mexico: case report. BMC Vet. Res. 2016;12:132. doi: 10.1186/s12917-016-0763-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vui D.T., Thanh T.L., Tung N., Srijangwad A., Tripipat T., Chuanasa T., Nilubol D. Complete genome characterization of porcine epidemic diarrhea virus in Vietnam. Arch. Virol. 2015;160:1931–1938. doi: 10.1007/s00705-015-2463-6. [DOI] [PubMed] [Google Scholar]
- Wang X.M., Niu B.B., Yan H., Gao D.S., Yang X., Chen L., Chang H.T., Zhao J., Wang C.Q. Genetic properties of endemic Chinese porcine epidemic diarrhea virus strains isolated since 2010. Arch. Virol. 2013;158:2487–2494. doi: 10.1007/s00705-013-1767-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu H., Zhang X., Liu C., Liu D., Liu J., Tian J., Qu L. Antiviral effect of lithium chloride on feline calicivirus in vitro. Arch. Virol. 2015;160:2935–2943. doi: 10.1007/s00705-015-2534-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang J.L., Ha T.K., Dhodary B., Pyo E., Nguyen N.H., Cho H., Kim E., Oh W.K. Oleanane triterpenes from the flowers of Camellia japonica inhibit porcine epidemic diarrhea virus (PEDV) replication. J. Med. Chem. 2015;58:1268–1280. doi: 10.1021/jm501567f. [DOI] [PubMed] [Google Scholar]
- Zhou P., Fu X., Yan Z., Fang B., Huang S., Fu C., Hong M., Li S. Antiviral effect of lithium chloride on infection of cells by canine parvovirus. Arch. Virol. 2015;160:2799–2805. doi: 10.1007/s00705-015-2577-x. [DOI] [PMC free article] [PubMed] [Google Scholar]