Abstract
Porcine epidemic diarrhea virus (PEDV) and porcine group A rotavirus (PGAR) are the main causative agents of acute diarrhea in piglets. In South Korea, PGAR is prevalent in piglets naturally infected with PEDV. Piglets naturally co-infected with PEDV and PGAR appeared to have severe and prolonged diarrhea that was distinct from that commonly observed. The aim of this study was to determine the impact of PGAR co-infection on PEDV pathogenicity in piglets. Thirty-six colostrum-deprived, one-day old, Large White-Duroc crossbred pigs were randomly divided into four equal groups: PEDV, PEDV/PGAR, PGAR, and control groups. The piglets were euthanized at 1, 2, or 3 days post-inoculation (DPI) to measure the villous height:crypt depth (VH:CD) ratio and to collect fecal samples for RT-PCR and virus isolation. No significant differences in mean VH:CD ratio and clinical symptoms (diarrhea, vomiting, dehydration, and anorexia) were observed between the PEDV/PGAR-infected and PEDV-infected groups of piglets at 1, 2 and 3 DPI; however, at 2 and 3 DPI, PGAR was detected in all fecal samples by RT-PCR and virus isolation. These findings failed to detect any interaction between PEDV and porcine rotavirus in the small intestines of piglets, suggesting that concurrent infection of PGAR may not synergistically enhance intestinal villous atrophy of piglets with PEDV disease. We propose that the severe diarrhea exhibited in PEDV and PGAR co-infected piglets may be more associated with the immunity level of the host rather than to any synergistic effect of PGAR on PEDV enteritis.
Keywords: Porcine epidemic diarrhea virus, Porcine group A rotavirus, Co-infection, Synergistic effect, Pathogenicity
Porcine epidemic diarrhea virus (PEDV), a member of the genus Coronavirus (family Coronaviridae, Order Nidovirales), causes highly contagious diarrhea in pigs of all ages. The disease leads to significant morbidity and mortality in neonatal piglets. PEDV infections are characterized by acute destruction of intestinal villous enterocytes and villous atrophy within the jejunum and ileum (Pensaert and de Bouck, 1978). PEDV has become an economic concern, mainly in South Korea as well as in Japan and China (Chae et al., 2000).
Porcine group A rotavirus (PGAR) is a causative agent of acute diarrhea in neonatal and preweaning piglets. Intestinal lesions by PGAR resemble those of PEDV and another porcine enteric coronavirus, transmissible gastroenteritis virus (TGEV), but result in milder diarrhea (Paul and Stevenson, 1999). Outbreaks of PGAR are sporadic, whereas PEDV and TGEV infections are explosive throughout porcine herds. It is believed that porcine rotavirus diseases may be associated with host immune and environmental factors (Paul and Stevenson, 1999).
A recent research study reported that the concurrent infection rate of PEDV and PGAR in Korean piglets was high (43.2%), indicating that PGAR is prevalent in piglets naturally infected with PEDV (Song et al., 2006). In some Korean pig herds, PEDV-induced diarrhea in neonatal piglets with PGAR co-infection seemed to be more severe and prolonged when compared to commonly observed diarrhea. Gross and histological findings also showed that the piglets diagnosed with PEDV and PGAR co-infection had more severe atrophic enteritis than those diagnosed with PEDV infection or PGAR infection (personal observation, D.S. Song). PGAR can predispose the host to developing concurrent enterotoxigenic Esherichia coli, Clostridium perfrigens, and coccidian infections, which can increase the severity of the diarrhea (Paul and Stevenson, 1999). It was therefore speculated that concurrent infection of PGAR might affect PEDV pathogenicity in neonatal piglets. The aim of this study was to determine the impact of PGAR co-infection on PEDV pathogenicity by subjecting piglets experimentally co-infected by PEDV and PGAR to evaluation of clinical signs (severity in diarrhea, dehydration, and dehydration) and histological morphometric analysis (villous height: crypt depth) in mid-jejunum where PEDV lesions were evident.
The PEDV strain wild type DR13 (passage level 4 (P4)) was used as inoculum after the fourth passage in tissue culture and the PGAR strain Gottfried were used after the 3 passage in MA-104 cells (Song et al., 2003, Song et al., 2006). PEDV strain DR13 (P4) was a virulent strain that caused watery diarrhea in one-day old piglets within 1 day after oral inoculation in a preliminary study. For PGAR strain Gottfried, the virus showed less virulence compared to the PEDV strain DR13 by causing mild to moderate diarrhea in one-day old piglets between 2 and 3 day after oral infection. Piglets infected with PGAR showed mild to moderate villous atrophy in mid-jejunum as well as in distal/ileal intestines at 2 and 3 day after infection. Each virus was titrated by analyzing the growth of Vero cells or MA-104 cells inoculated with 10-fold serial dilutions of the PEDV and PGAR virus stocks, respectively, following the Reed and Muench method (Reed and Muench, 1938).
Thirty-six colostrum-deprived, one-day old, Large White-Duroc crossbred pigs were randomly divided into four equal groups: PEDV, PEDV/PGAR, PGAR, and control. The piglets were acquired from pregnant sows that had not been vaccinated with any PEDV or porcine rotavirus before parturition. Of the three treatment groups, 27 piglets were inoculated orally with either 3 ml of virus stock of the PEDV strain Dr13 (105 tissue culture infective doses 50% [TCID50]/ml), 3 ml of virus stock of the PGAR strain Gottfried (104 TCID50/ml), or with 3 ml of both PEDV and GAR at a 1:1 ratio. For the PEDV/PGAR group, the piglets were infected orally with PEDV at 12 h after inoculation with PGAR so that PEDV would replicate in intestinal epithelial cells with an established PGAR infection and at the time, PGAR group piglets were inoculated with PGAR. The experiment was designed in this way to evaluate the potential effect of a secondary PGAR infection on the intestinal villous atrophy induced by a primary PEDV infection. Nine control piglets were similarly treated with uninfected cell culture medium. All piglets were maintained in stainless steel isolates (three per isolator), and fed a commercial sterile milk substitute. Animals were examined three times daily for clinical signs. Three piglets from each group were euthanized by electrocution at 1, 2, and 3 days post-inoculation (DPI) for tissue collection and analysis. All piglets were sedated with an intravenous injection of sodium pentobarbital, and euthanized by electrocution, as described previously (Anonymous, 2001). Complete necropsies were performed on all the pigs, and all organs were grossly and histologically examined. Three intestinal samples (from a 15 cm sample of the intermediate jejunal zone) were collected and fixed in 10% neutral buffered formalin for histological morphometric analysis.
RT-PCR was performed as described previously, with slight modifications (Song et al., 2006). Viral RNA was extracted from fecal samples using Trizol LS reagent (Gibco BRL) according to the manufacturer’s instructions, and then precipitated with ethanol to collect RNA. The forward and reverse primers used to detect PEDV were 5′-TTCTGAGTCACGAACAGCCA-3′ (1466-1485) and 5′-CATATGCAGCCTGCTCTGAA-3′ (2097-2116), respectively, which amplified a 651-base pair (bp) region from the spike gene of PEDV. The forward and reverse primers used to detect PGAR were 5′-AAAGATGCTAGGGACAAAATTG-3′ (57-78) and 5′-TTCAGATTGTGGAGCTATTCCA-3′ (344-365), respectively, which amplified a 309- bp region from the segment 6 region of group A rotavirus.
First-strand cDNA synthesis was performed as follows: 1 μL of viral RNA (5 ng/μL) was supplemented in a total reaction volume of 20 μL with 1 × RT buffer (50 mM Tris–HCl, 8 mM MgCl2, 30 mM KCl, 1 mM dithiothreitol (pH 8.3)), 0.5 mM of each deoxynucleotide triphosphate (dNTP), 2.5 μM random hexanucleotide mixture, 20 U of RNase inhibitor, and 50 U of Moloney murine leukemia virus reverse transcriptase. After incubation for 45 min at 42 °C, the mixture was heated for 5 min at 99 °C to denature the products, and then stabilized at 4 °C. The composition of the PCR mixture (150 μL) was 30 μL of cDNA (5 ng/μL), 2 μL of each primer (250 nM), 15 μL of 10 × PCR buffer (10 mM Tris–HCl, 40 mM KCl, 1.5 mM MgCl2 (pH 8.3)), 1.2 μL of each dNTP (0.2 mM), 29 μL of 2.5 units of Taq polymerase, and 67.2 μL of distilled water. The PCR reaction for both PEDV and PGAR was carried out under the following conditions in a thermal cycler (Applied-Biosystem): 1 cycle of 5 min at 94 °C; 35 cycles of denaturation at 94 °C for 30 s, annealing at 53 °C for 1 min, and elongation at 72 °C for 1 min. The PCR products were analyzed by electrophoresis.
Three sections of formalin-fixed jejunum were obtained from each virus-infected and control piglet for morphometric analysis. Only well-oriented sections were measured, and care was taken to ensure that only transverse sections, cut perpendicularly from villous enterocytes to the muscularis mucosa, were included. Villous height and crypt depth were estimated by measuring 10 villi and crypts throughout the section. Villous height and crypt depth (VH:CD) ratios were calculated according to a previous report (Jung et al., 2006b).
Differences in VH:CD ratio between infected and control piglets were assessed by the Kruskal–Wallis test using Statistical Analysis Systems software (SAS Institute Inc., Cary, NC). A value of P < 0.05 was considered to be statistically significant.
Clinical symptoms in PEDV or PGAR-infected piglets ranged from diarrhea to vomiting, anorexia and dehydration. In general, piglets from the PEDV and PEDV/PGAR groups showed more severe diarrhea and dehydration than piglets from the PGAR group. Eighteen piglets (18/18, 100%) from the PEDV and PEDV/PGAR groups exhibited severe watery diarrhea and intermittent vomiting from 1 DPI. Anorexia was generally observed at 1, 2, and 3 DPI, although not in all cases. Moreover, severe dehydration was prevalent in most piglets at 2 and 3 DPI. No differences in the severity of clinical signs were observed between the PEDV and PEDV/PGAR piglets. PGAR-infected piglets, however, showed moderate diarrhea at 2 and 3 DPI, and displayed better health compared to the PEDV and PEDV/PGAR piglets. No clinical signs were observed in the control piglets at each DPI.
The results are summarized in Table 1 . All fecal samples collected from PEDV-infected piglets at 1, 2, and 3 DPI were positive for PEDV by both RT-PCR and virus isolation. Of the piglets in the PEDV/PGAR group, 2 of 3 fecal samples collected at DPI 1 were positive for PGAR by judged by both RT-PCR and virus isolation. At DPI 2 and 3, however, all fecal samples were positive for PGAR by both RT-PCR and virus isolation. Fecal samples collected from piglets in the PGAR group at DPI 1 were negative for PGAR by both RT-PCR and virus isolation. At DPI 2, however, 2 of 3 samples were positive for PGAR by both RT-PCR and virus isolation, and at DPI 3, all fecal samples were positive for PGAR.
Table 1.
Diarrhea and detection of porcine epidemic diarrhea virus (PEDV) and porcine group A rotavirus (PGAR) by RT-PCR and virus isolation in the fecal samples from piglets infected with PEDV, PEDV/PGAR, or PGAR, or in control piglets at different days post-inoculation (DPI)
| Group | DPI |
||||||||
|---|---|---|---|---|---|---|---|---|---|
| Diarrhea |
RT-PCR |
Virus isolation |
|||||||
| 1 | 2 | 3 | 1 | 2 | 3 | 1 | 2 | 3 | |
| PEDV | 3/3a | 3/3 | 3/3 | 3/3 | 3/3 | 3/3 | 3/3 | 3/3 | 3/3 |
| PEDV, PGAR | 3/3 | 3/3 | 3/3 | 3, 2b/3, 3 | 3, 3b/3, 3 | 3, 3b/3, 3 | 3, 2c/3, 3 | 3, 3c/3, 3 | 3, 3c/3, 3 |
| PGAR | 0/3 | 2/3 | 3/3 | 0/3 | 2/3 | 3/3 | 0/3 | 2/3 | 3/3 |
| Control | 0/3 | 0/3 | 0/3 | 0/3 | 0/3 | 0/3 | 0/3 | 0/3 | 0/3 |
Number of positive pigs/number of pigs tested.
Number of PGAR-positive pigs by RT-PCR. For the PEDV/PGAR group, PEDV was infected orally 12 h after inoculation with PGAR.
Number of PGAR-positive pigs by virus isolation. For the PEDV/PGAR group, PEDV was infected orally 12 h after inoculation with PGAR.
PEDV/PGAR and PEDV infected piglets had severe atrophic enteritis at 1, 2, and 3 DPI, whereas PGAR infected piglets exhibited mild villous atrophy at 1 DPI, and moderate lesions at 2 and 3 DPI (Fig. 1 ). No histological lesions were detected in the control piglets. There was no autolysis in the intestines of all pig groups. The mean VH:CD ratios of the PEDV/PGAR and PEDV groups were significantly decreased at 1, 2, and 3 DPI compared to control piglets; however, the mean VH:CD ratios of the PEDV/PGAR piglets were not significantly different from those of the PEDV piglets, but were significantly lower than those of the PGAR piglets (P < 0.05) (Fig. 1). At 1 DPI, the mean VH:CD ratio of the PGAR group piglets was similar to that of the control, but was significantly decreased at 2 and 3 DPI. The mean VH:CD ratios of the PGAR piglets were significantly greater than those of the PEDV/PGAR and PEDV piglets at 2 and 3 DPI (P < 0.05).
Fig. 1.
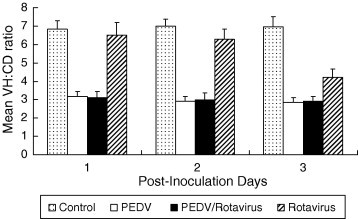
Mean villous height and crypt depth (VH:CD) ratios in the jejunum. The mean VH:CD ratio of piglets co-infected with porcine epidemic diarrhea virus (PEDV) and porcine group A rotavirus (PGAR) was significantly decreased at 1, 2, and 3 days post-inoculation compared to negative control piglets; however, the mean VH:CD ratios of PEDV/PGAR piglets were not significantly different from those of PEDV piglets. Each value represents the mean (standard deviation).
This study was designed to determine whether there is any interaction between PEDV and porcine group A rotavirus (PGAR) in the small intestine of neonatal piglets showing classical PEDV symptoms such as vomiting and the highest susceptibility to the viruses. The results suggest that concurrent infection of PGAR does not synergistically enhance intestinal villous atrophy of piglets with PEDV-induced diarrhea. No significant differences in mean VH:CD ratio or clinical symptoms were observed between PEDV/PGAR and PEDV group piglets at 1, 2 and 3 DPI; however, at 2 and 3 DPI, PGAR was detected in fecal samples by RT-PCR and virus isolation. This indicates that PGAR concurrently replicated with PEDV in the intestinal epithelial cells at the specified time points. We collected the middle jejunal intestine to evaluate the changes of PEDV pathogenicity using morphometric analysis (VH:CD). Intestinal villous atrophy in pigs with PEDV infection was observed more in middle jejunum than in distal jejunum or ileum, whereas less for PGAR infection. However, rotaviruses infect different regions of the intestines depending on the difference in the virulence of each virus strain, and the infected regions include the middle jejunum as well as the distal jejunum or ileum (Bridger, 1994, Hall et al., 1993). The piglets with PGAR Gottfried strain infection also showed marked villous atrophy in the middle jejunal intestine at 3 DPI (Fig. 1). We guess that the PGAR Gottfried strain could affect replication and distribution of PEDV in villous epithelial cells of the middle intestines of the pigs co-infected with PEDV.
The severity of intestinal villous atrophy by PEDV may depend on the type of virus strain used to inoculate the piglets. The PEDV strain DR13 (P4) was virulent enough to cause watery diarrhea in piglets within 24 h after oral infection. However, the DR13 strain was not as virulent as the one described in previous reports, where the VH:CD ratio of experimentally PEDV-infected piglets decreased to less than 3 at 3 DPI (Jung et al., 2006b, Jung et al., 2006c). Virulence and inoculum dose of the PEDV DR13 strain were considered to be appropriate at evaluation of severity of villous atrophy (VH:CD ratio). In our study, equivalent doses of the two viruses (3 × 105 TCID50/pig for PEDV and 3 × 104 TCID50/pig for PGAR in the study) were not used. A difference in the inoculum dose between these two viruses could affect the outcome. However, the effects were considered to be minor to get the same results, because the inoculum dose (3 × 104 TCID50) of PGAR showed enough clinical symptoms and villous atrophy in PGAR only-infected group.
Porcine group A rotavirus infection, as well as PEDV disease, is prevalent in neonatal piglets in South Korea and Japan (Katsuda et al., 2006, Song et al., 2006). Piglets naturally co-infected with PEDV and PGAR seem to have severe and prolonged diarrhea that is distinct from that commonly observed (personal observation, D.S. Song). PGAR infection is known to be associated with host immune and environmental factors (Paul and Stevenson, 1999). Rotavirus diarrhea is more prolonged and severe in immuno-compromised animals and humans (Eiden et al., 1985, Liakopoulou et al., 2005, Paul and Stevenson, 1999). Considering that there was no interaction between PEDV and PGAR in the present study, we speculate that the severe diarrhea observed in PEDV and PGAR co-infected piglets may be more a consequence of the immunity level of the host rather than the result of any synergistic effect of PGAR on PEDV-induced enteritis. A previous study using a virulent PEDV strain demonstrated that immunosuppression due to the transplacental infection with porcine circovirus type 2 (PCV2) probably potentiates PEDV pathogenicity in piglets by causing more severe intestinal villous atrophy compared to PEDV only-infected pigs (Jung et al., 2006a). The piglets co-infected with PCV2 and PEDV and piglets infected with PEDV alone were histologically different with respect to their VH:CD ratios at 2 and 3 PID (Jung et al., 2006b).
Although most Korean pig herds have been vaccinated against PEDV and porcine rotavirus, the disease continues to resist eradication in South Korea. The present investigation could not demonstrate any interaction between PEDV and porcine group A rotavirus in the small intestine of piglets. However, further studies, such as those designed to evaluate cytokine changes and titre the virus contents of the small intestine of piglets, will be needed to confirm any synergistic relationship between these two viruses. In the present study, we propose that concurrent infection with porcine group A rotavirus does not affect PEDV pathogenicity in piglets.
References
- Anonymous 2000 report of the AMVA panel on euthanasia. Journal of American Veterinary Medical Association. 2001;218:669–696. doi: 10.2460/javma.2001.218.669. [DOI] [PubMed] [Google Scholar]
- Bridger J.C. A definition of bovine rotavirus virulence. Journal of General Virology. 1994;75:2807–2812. doi: 10.1099/0022-1317-75-10-2807. [DOI] [PubMed] [Google Scholar]
- Chae C., Kim O., Choi C., Min K., Cho W.-S., Kim J., Tai J.H. Prevalence of porcine epidemic diarrhoea virus and transmissible gastroenteritis virus infection in Korean pigs. Veterinary Record. 2000;147:606–608. doi: 10.1136/vr.147.21.606. [DOI] [PubMed] [Google Scholar]
- Eiden J., Losonsky G.A., Johnson J., Yolken R.H. Rotavirus RNA variation during chronic infection of immunocompromised children. Pediatric Infectious Disease. 1985;4:632–637. doi: 10.1097/00006454-198511000-00007. [DOI] [PubMed] [Google Scholar]
- Hall G.A., Bridger J.C., Parsons K.R., Cook R. Variation in rotavirus virulence: a comparison of pathogenesis in calves between two rotaviruses of different virulence. Veterinary Pathology. 1993;30:223–233. doi: 10.1177/030098589303000302. [DOI] [PubMed] [Google Scholar]
- Jung K., Ha Y., Ha S.-K., Kim J., Choi C., Park H.-K., Kim S.-H., Chae C. Identification of porcine circovirus type 2 in retrospective cases of pigs naturally infected with porcine epidemic diarrhoea virus. The Veterinary Jounral. 2006;171:166–168. doi: 10.1016/j.tvjl.2004.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung K., Kim J., Ha Y., Choi C., Chae C. The effects of transplacental porcine circovirus type 2 infection on porcine epidemic diarrhea virus-induced enteritis in preweaning piglets. The Veterinary Journal. 2006;171:445–450. doi: 10.1016/j.tvjl.2005.02.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung K., Ahn K., Chae C. Decreased activity of brush border membrane-bound digestive enzymes in small intestines from pigs experimentally infected with porcine epidemic diarrhea virus. Research in Veterinary Science. 2006;81:310–315. doi: 10.1016/j.rvsc.2006.03.005. [DOI] [PubMed] [Google Scholar]
- Katsuda K., Kohmoto M., Kawashima K., Tsunemitsu H. Frequency of enteropathogen detection in suckling and weaned pigs with diarrhea in Japan. Journal of Veterinary Diagnostic Investigation. 2006;18:350–354. doi: 10.1177/104063870601800405. [DOI] [PubMed] [Google Scholar]
- Liakopoulou E., Mutton K., Carrington D., Robinson S., Steward C.G., Goulden N.J., Cornish J.M., Marks D.I. Rotavirus as a significant cause of prolonged diarrhoeal illness and morbidity following allogeneic bone marrow transplantation. Bone Marrow Transplantation. 2005;36:691–694. doi: 10.1038/sj.bmt.1705127. [DOI] [PubMed] [Google Scholar]
- Paul P.S., Stevenson G.W. Diseases of swine. In: Straw B.E., D’Allaire S., Mengeling W.L., Taylor D.J., editors. Rotavirus and Reovirus. eighth ed. Iowa State University Press; Ames, IA, USA: 1999. pp. 255–275. [Google Scholar]
- Pensaert M.B., de Bouck P. A new coronavirus-like particle associated with diarrhea in swine. Archives of Virology. 1978;58:243–247. doi: 10.1007/BF01317606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reed L.J., Muench H. A simple method of estimating fifty percent endpoints. American Journal of Hygiene. 1938;27:493–497. [Google Scholar]
- Song D.S., Yang J.S., Oh J.S., Han J.H., Park B.K. Differentiation of a Vero cell adapted porcine epidemic diarrhea virus from Korean field strains by restriction fragment length polymorphism analysis of ORF3. Vaccine. 2003;21:1833–1842. doi: 10.1016/S0264-410X(03)00027-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song D.S., Kang B.K., Oh J.S., Ha G.W., Yang J.S., Moon H.J., Jang Y.S., Park B.K. Multiplex reverse transcription-PCR for rapid differential detection of porcine epidemic diarrhea virus, transmissible gastroenteritis virus, and porcine group A rotavirus. Journal of Veterinary Diagnostic Investigation. 2006;18:278–281. doi: 10.1177/104063870601800309. [DOI] [PubMed] [Google Scholar]


