Abstract
Development of an effective vaccine for severe acute respiratory syndrome (SARS) remains to be a priority to prevent possible re-emergence of SARS coronavirus (SARS-CoV). We previously demonstrated that the receptor-binding domain (RBD) of SARS-CoV S protein is a major target of neutralizing antibodies. This suggests that the RBD may serve as an ideal vaccine candidate. Recombinant adeno-associated virus (rAAV) has been proven to be an effective system for gene delivery and vaccine development. In this study, a novel vaccine against SARS-CoV was developed based on the rAAV delivery system. The gene encoding RBD was cloned into a pAAV-IRES-hrGFP plasmid. The immunogenicity induced by the resulting recombinant RBD-rAAV was evaluated in BALB/c mice. The results demonstrated that (1) a single dose of RBD-rAAV vaccination could induce sufficient neutralizing antibody against SARS-CoV infection; (2) two more repeated doses of the vaccination boosted the neutralizing antibody to about 5 times of the level achieved by a single dose of the immunization and (3) the level of the antibody continued to increase for the entire duration of the experiment of 5.5 months. These results suggested that RBD-rAAV is a promising SARS candidate vaccine.
Keywords: Adeno-associated virus, SARS-CoV, Spike protein, Receptor-binding domain, Neutralizing antibodies, Vaccines
Introduction
Severe acute respiratory syndrome (SARS) coronavirus (SARS-CoV) has been identified as the etiological agent of the newly emerging infectious disease SARS (Drosten et al., 2003, Ksiazek et al., 2003, Marra et al., 2003, Peiris et al., 2003, Rota et al., 2003). Although SARS appears to have been successfully contained, re-emergence of the disease cannot be ruled out. This is supported by the fact that four laboratory-acquired and four community-acquired SARS cases were reported in Singapore, Taiwan and China after the SARS outbreak (www.who.int/csr/sars/en). Therefore, development of effective vaccines against SARS is urgently needed.
SARS-CoV is a single-stranded positive RNA virus, whose genome encodes nonstructural replicase polyprotein (rep), and structural proteins such as spike (S), envelope (E), membrane (M) and nucleocapsid (N) (Anand et al., 2003, Marra et al., 2003, Rota et al., 2003). S protein recognizes and binds to host receptors, and the conformational changes induced in this viral protein would then facilitate fusion between the viral envelope and the host cell membranes, thus S plays pivotal roles in viral infection and pathogenesis (Hofmann et al., 2004, Holmes, 2003, Yu et al., 2003, Zheng et al., 2005). S protein is a type I transmembrane glycoprotein consisting of S1 and S2 domains (Bosch et al., 2003, Holmes and Enjuanes, 2003, Krokhin et al., 2003, Ying et al., 2004; Fig. 1 ). The S1 domain determines the host cell tropism by attaching to cell receptors, and the S2 domain is mainly responsible for the virus and cell membrane fusion (Bosch et al., 2003, Gallagher and Buchmeier, 2001). Angiotensin-converting enzyme 2 (ACE2) has been identified as a functional receptor for SARS-CoV (Dimitrov, 2003, Li et al., 2003, Prabakaran et al., 2004), and a 193-aa fragment containing residues 318–510 of the S1 region is found to be the minimal receptor-binding domain (RBD) (Babcock et al., 2004, Wong et al., 2003, Xiao et al., 2003; Fig. 1). A second major property of SARS-CoV S protein is that it induces neutralizing antibodies and protective immunity (Bisht et al., 2004, Buchholz et al., 2004, Bukreyev et al., 2004, Yang et al., 2004). Yang et al. (2004) reported that a DNA vaccine encoding S protein was able to induce T-cell and neutralizing-antibody responses (neutralizing antibody titers range from 1:50 to 1:150). They proved that protection of mice against SARS-CoV challenge was mediated by neutralizing antibodies but not a T-cell-dependent mechanism. Traggiai et al. (2004) demonstrated that protective immunity in mice could be achieved by sole administration of neutralizing monoclonal antibodies specific for S protein. These data suggest that an effective SARS vaccine can be developed based on its ability to induce neutralizing antibodies. We previously reported that RBD of SARS-CoV S protein was able to elicit potent neutralizing antibodies in rabbits (He et al., 2004a). Furthermore, RBD has also been found to be an immunogenic domain of SARS-CoV, which induced neutralizing antibodies in SARS-CoV infected patients (He et al., 2004b). Therefore, S protein, especially RBD in the S1 domain, is a good target for developing vaccines against SARS (He et al., 2004b, Yang et al., 2004).
Fig. 1.
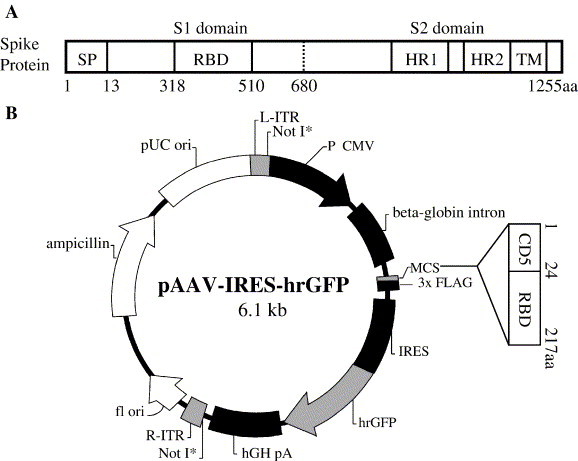
Schematic diagram of S protein of SARS-CoV and pRBD-AAV. The structure of SARS-CoV S protein (A). In the S1 domain, SP and RBD range from 1 to 13 aa (13 aa) and from 318 to 510 aa (193 aa), respectively. The S2 domain contains functional domains HR1, HR2 and TM. DNA encoding 24 aa of the CD5 signal peptide and 193 aa RBD of SARS-CoV S protein was amplified and ligated into the pAAV-IRES-hrGFP plasmid to construct the recombinant pRBD-AAV vector (B).
Adeno-associated virus (AAV) is a powerful delivery vector that is widely used in gene therapy (Chao et al., 2002, Kaspar et al., 2002, Tsai et al., 2002). With high transduction efficiency, minimal viral toxicity, broad range of infectivity and little pathogenesis in humans, the AAV system has been successfully applied in studying gene and immune therapies for many diseases (Keir et al., 1999, Lieber, 2003). Furthermore, the long-term gene transfer and expression in a variety of cell types and organs have made this system an ideal choice for developing recombinant vaccines (Tomar et al., 2003).
In the present study, we used AAV as a vector to deliver a gene expressing RBD of SARS-CoV S protein (RBD-rAAV). The effects of single dose and multiple doses of the RBD-rAAV vaccination were evaluated in BALB/c mice for inducing SARS-CoV-specific antibodies, especially neutralizing antibodies.
Results
Generation of the recombinant AAV in transfected 293T cells
SARS-CoV S protein is a glycoprotein consisting of an S1 and an S2 domain. The S1 domain contains the signal peptide (SP) at the N-terminus and RBD at aa 318 to 510. The S2 domain contains heptad repeat 1 and 2 (HR1 and HR2) and a transmembrane domain TM (Fig. 1A). In the construction of the pRBD-AAV vector, the nucleotide sequence encoding SP was replaced by the CD5 and the latter was ligated to the RBD sequence of SARS-CoV S, and the chimeric gene was subcloned into the multiple cloning sites (MCS) of the pAAV-IRES-hrGFP plasmid (Stratagene, USA) (Fig. 1B). The resulting pRBD-AAV plasmid, or the blank pAAV plasmid, was co-transfected into 293T cells together with pAAV-RC and pHelper plasmids to produce AAV, which harbors the chimeric CD5-RBD gene (RBD-rAAV) or that which does not harbor the chimeric gene (blank AAV). Intense fluorescence of the humanized recombinant green fluorescent protein (hrGFP) in RBD-AAV (Fig. 2A) and blank AAV (Fig. 2B) was observed in the transfected cells under a fluorescence microscopy at 72 h post-transfection, illustrating a high transfection efficiency of the viral plasmids in 293T cells. The yield of either the RBD-rAAV or the blank AAV from the respective transfected cells harvested from 30 × 100 mm tissue culture dishes was purified and the titer was estimated by real-time quantitative PCR to be 5 × 1012 viral particles/ml.
Fig. 2.
RBD-rAAV production in 293T cells. After 293T cells were transfected by pRBD-AAV (A) or blank pAAV (B) together with their helper plasmids for 72 h, intense fluorescence of hrGFP was observed under fluorescence microscopy (40×).
Transduction efficiency of the recombinant AAV
Fig. 3 compares the transduction of RBD-rAAV and blank AAV in 293T and HeLa cells. In this experiment, 293T and HeLa cells were inoculated with serial dilution of the viruses and after 72 h of incubation, the extent of infection with either virus was assessed by the level of GFP (hrGFP) expressed by these cells, using fluorescence microscopy and flow cytometry analysis. Fluorescence microscopy shows that 293T cells infected with the rAAVs (Fig. 3A) are more numerous than HeLa cells (Fig. 3B). As shown in Fig. 3C, flow cytometry analysis estimates that that 98.4% of 293T cells was infected with RBD-rAAV and 98.2% was infected with blank AAV and this is substantially higher than the levels of infection achieved in HeLa cells with RBD-rAAV (22.6%) or blank AAV (21.7%). These results confirm the findings by Romano et al. (2000), showing that the infection efficiency of the AAV vector differs for different cells.
Fig. 3.
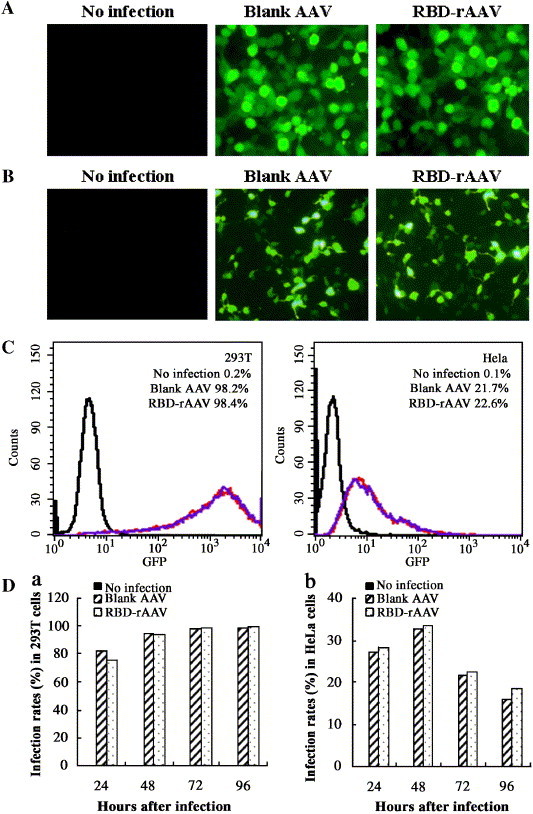
Detection of RBD-rAAV transduction efficiency by fluorescence microscopy and flow cytometry analysis. The infection of RBD-rAAV and blank AAV in 293T (A) and HeLa cells (B) was detected at 72 h post-infection under fluorescence microscopy (100×). The infection rates of the AAVs in these two cell lines were further measured at 72 h post-infection by a flow cytometry analysis (C). The top right of each histogram shows the mean percentage of GFP positive cells in analyzed cell populations from two independent experiments. Black lines indicate the non-infected cells and red lines indicate the blank AAV-infected cells, while blue lines indicate the RBD-rAAV infected cells. The infection rates of RBD-rAAV and blank AAV in 293T (a) and HeLa cells (b) were further tested at different time points from 1 to 4 days (D).
In another experiment, percentage of infected cells was enumerated at different times after infection (Fig. 3D). The results showed that percentage of RBD-rAAV-infected 293T cells detected at different times closely parallels the percentage of blank AAV-infected 293T cells (a), as does the percentage of HeLa cells infected with either of these viruses (b). This shows that the RBD insert did not affect the efficiencies of the rAAV infection and expression.
Expression of the RBD protein by the recombinant RBD-rAAV
Expression of RBD in RBD-rAAV infected HeLa cells was detected by an immunofluorescence assay (IF) and a Western blot assay. The IF results showed that a monoclonal antibody specific to SARS-CoV S1 (McAb-S1) reacted with the cells infected by RBD-AAV (Fig. 4A), but not with those infected by blank AAV (Fig. 4B). The expression of RBD was also detected by Western blot in lysate of cells infected with RBD-rAAV but not in lysate of cells infected with blank AAV (Fig. 4C). These results demonstrated that RBD-rAAV was able to efficiently express the inserted RBD gene in the infected cells.
Fig. 4.
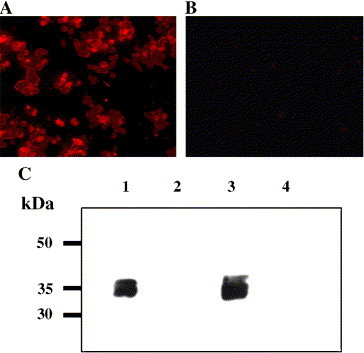
Detection of the RBD expression in RBD-rAAV infected cells by an immunofluorescence assay and Western blot analysis. Expression of RBD in RBD-rAAV (A) and blank AAV (B) infected HeLa cells were tested by an immunofluorescence assay. The RBD expression was also detected by Western blot (C). Lanes 1 and 3, RBD-rAAV-infected HeLa and 293T cell lysates; lanes 2 and 4, blank AAV-infected HeLa and 293T cell lysates. The molecular marker (kDa) is indicated on the left. The molecular mass of the expressed RBD protein was about ∼34 kDa.
Single dose of RBD-rAAV vaccination induced sufficient SARS-CoV-specific antibody responses with neutralizing activity in mice
Groups of 5 to 10 BALB/c mice were vaccinated with single dose of RBD-rAAV, blank AAV (negative control) or with inactivated SARS-CoV suspended in PBS or in adjuvant Alum (positive control). The animals were bled before immunization and at 1, 2 and 4 months post-vaccination. Levels of SARS-CoV-specific antibodies in these serum samples were determined by ELISA. As shown in Fig. 5A, the titers of SARS-CoV-specific antibodies in the sera from single dose of RBD-rAAV immunized mice reached 1:680 ± 120 (mean ± SE) at 1 month and slightly increased to 1:880 ± 160 at 4 months post-vaccination. By comparison, the levels of SARS-CoV antibodies induced by the inactivated virus vaccination peaked earlier (2 month post-immunization) and at a slightly low level (1:686 ± 175 with Alum, P > 0.05) or at a significantly low level (1:384 ± 64 without Alum, P < 0.05) at 2 months post-immunization. SARS-CoV neutralizing activity in the mouse sera was further tested by a neutralization assay in Vero E6 cells according to method described by Qu et al. (2005). In animals vaccinated with RBD-rAAV, Fig. 5B shows that the neutralizing antibody level rose continuously from 1:48 ± 8 at 1 month to 1:108 ± 20 at 4 months post-vaccination. By comparison, the antibody level of animals given the inactivated virus vaccination peaked earlier, at 2 months after immunization. The peak level of neutralizing antibody of the animals vaccinated with the RBD-rAAV is similar as that of vaccinated with the inactivated virus suspended in Alum (1:109 ± 18, P > 0.05), but it is almost 3-fold higher than that was given the inactivated virus suspended in PBS (1:42 ± 10, P < 0.05). No SARS-CoV neutralizing antibodies (<1:20) could be detected in the mouse sera from blank AAV immunized group. The results indicated that although the RBD-rAAV vaccination induced a lower level of neutralizing antibody than vaccination with SARS-CoV plus Alum at 1–2 months after immunization, it can deliver a prolonged effect with an increasing neutralizing antibody titer at 4 months post-vaccination, which was similar or higher than inactivated virus vaccination with or without Alum (P > 0.05).
Fig. 5.
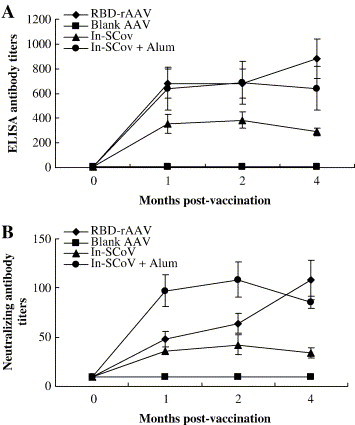
Evaluation of specific antibody responses in single dose of RBD-rAAV-vaccinated mice. The mice were i.m.-immunized respectively with one dose of RBD-rAAV, blank AAV and inactivated SARS-CoV with (In-SCoV + Alum) or without (In-SCoV) Alum. Serum samples were collected from the mice at pre-vaccination (0 month), 1, 2 and 4 months post-immunization. SARS-CoV-specific antibodies in the serum samples were detected by ELISA using the commercial SARS-CoV antibody detection kit (A). The neutralizing antibody titers in the sera were measured by a neutralization assay (B). The experiments were repeated four times for the RBD-rAAV group and two times for the other groups. The data were presented as the mean values ± SE.
Repeated vaccinations of RBD-rAAV effectively induced high levels of SARS-CoV-specific antibodies with neutralizing activity
In a further experiment, groups of 5 mice were given 2 repeated doses of RBD-rAAV, blank AAV (negative control) or with inactivated SARS-CoV with or without adjuvant Alum (positive control), and the animals were bled at different times over a period of 5.5 months following the first dose. In the animals given the RBD-rAAV vaccine, the level of the SARS-CoV-specific antibody determined by ELISA increased continuously throughout the experiment to reach a titer of 1:5120 ± 320 at 5.5 months post-vaccination (Fig. 6A). In animals given the inactivated virus, the antibody levels reached the peak (1:3584 ± 627 without Alum and 1:4754 ± 366 with Alum) at 4 months post-vaccination. The antibody level determined at the end of the experiment 5.5 months after the first dose was slightly higher for animals, which were given the RBD-rAAV vaccine than those which were given the inactivated virus, but the difference was not statistically significant (P > 0.05).
Fig. 6.
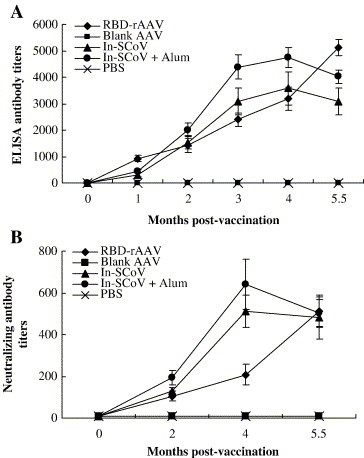
Detection of specific antibody responses in three doses of RBD-rAAV-vaccinated mice. SARS-CoV-specific antibody responses in mice vaccinated with three doses of RBD-rAAV or controls were tested by ELISA (A) and neutralization assay (B). Serum samples were collected from the mice at pre-vaccination (0 month), 1, 2, 3, 4 and 5.5 months post-immunization. The experiments were repeated two times and the data were presented as the mean values ± SE.
The neutralizing antibody level in RBD-rAAV group of animals also rose continuously throughout the duration of the experiment of 5.5 months, as did the levels of ELISA antibody, whereas the antibody level of the animals receiving the inactivated virus vaccines peaked on the 4th month. At the end of the experiment, 5.5 months after the first dose, the neutralizing antibody level of RBD-rAAV group was 1:512 ± 78, which is similar as the peak antibody levels of animals given the inactivated virus vaccine (1:512 ± 78 without Alum and 1:640 ± 121 with Alum), and it is 5 times higher than the antibody level induced by a single dose of this vaccine (see Fig. 6B).
Sera from RBD-rAAV-vaccinated mice suppressed RBD binding to the receptor ACE2
Sera from the mice immunized with RBD-rAAV were detected for the inhibitory activity against binding of RBD to SARS-CoV receptor ACE2 by a competitive assay. As shown in Fig. 7 , the sera from all of five RBD-rAAV immunized mice were able to suppress more than 90% of RBD binding to ACE2, while none of the pre-immunized sera and sera from blank AAV immunized mice inhibited the binding. These results concur with occurrence of SARS-CoV neutralizing antibody, implying that RBD-rAAV vaccination could evoke protective immunity against the virus.
Fig. 7.
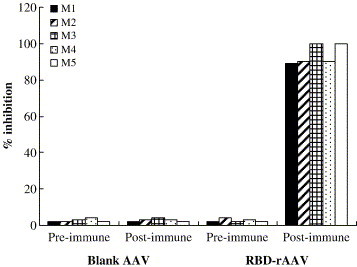
Inhibitory test of RBD binding to ACE2. Sera obtained from five mice vaccinated with 3 doses of RBD-rAAV (labeled as M1–5) at 4 months post-vaccination were detected for the inhibitory effect on the binding of RBD to ACE2 by ELISA. Sera from five mice immunized with 3 doses of blank AAV were applied to the assay as the negative control. The serum samples were tested in duplicate and the data presented here were the mean values of the two tests.
Discussion
Development of SARS vaccines is an urgent requirement in preventing re-emergence of SARS. Several approaches for developing SARS vaccines have been reported. These include inactivated vaccines (He et al., 2004a, Qu et al., 2005), DNA vaccines (Yang et al., 2004, Zeng et al., 2004), recombinant protein-based vaccines (Bisht et al., 2004, Bisht et al., 2005, He et al., 2004b), the attenuated modified vaccinia virus Ankara (MVA) based vaccine (Chen et al., 2005), the adenovirus-based vaccine (Gao et al., 2003a, Gao et al., 2003b) and the rhabdovirus-based vaccine (Faber et al., 2005). These candidate vaccines could induce SARS-CoV-specific immune responses and/or elicit protective neutralizing antibody responses in immunized animals. In this study, we constructed another SARS vaccine based on the AAV delivery system and evaluated its immunogenicity in a mouse model.
SARS-CoV S protein is known to be responsible for inducing host immune responses and virus neutralizing antibodies in SARS infection (Buchholz et al., 2004, Gao et al., 2003b, Hofmann et al., 2004, Nie et al., 2004, Yang et al., 2004, Zheng et al., 2004). It has been reported that neutralizing antibodies against SARS-CoV S protein are able to provide protection against SARS-CoV infection (Bisht et al., 2004, Bukreyev et al., 2004, Sui et al., 2004). Although multiple fragments of S protein have been shown to be able to elicit neutralizing activities (Keng et al., 2005, Zhang et al., 2004), RBD, the receptor-binding domain to the functional virus receptor ACE2, which contains multiple conformation-dependent epitopes, can effectively induce highly potent protective neutralizing antibodies (Chen et al., 2005, He et al., 2004b, He et al., 2005a). A recent study also demonstrated that the RBD region is the main target in eliciting neutralizing antibodies in inactivated SARS-CoV immunized rabbits, and an important immunogenic site in patients infected with SARS-CoV (He et al., 2005b). Therefore, RBD of SARS-CoV S protein was selected as the target immunogen in our study for development of SARS-CoV vaccines.
In the construction of the pRBD-AAV vector, the RBD gene of SARS-CoV S was codon-optimized. Codon-optimization has been used as a strategy to improve protein expression and enhance immunogenicity of DNA vaccines in many viruses and mycobacteria (Cheung et al., 2004, Ko et al., 2005, Moore et al., 2004, Ramakrishna et al., 2004, Smith et al., 2004, Wang et al., 2005a, Wang et al., 2005b). A report by Gao et al. (2003a) has shown that codon-optimization could increase the level of expression and enhance the immunogenicity of the structural genes of the human immunodeficiency virus type I (HIV-1). Frelin et al. (2004) has also indicated that codon-optimization of hepatitis C virus (HCV) genes results in an improvement in the immunogenicity of HCV-based vaccines. Although we did not compare the efficiencies between the codon-optimized RBD gene and the wild-type RBD gene in this study, the high efficiency of our RBD-rAAV vaccination may be partially attributed to that the expression and immunogenicity of RBD has been improved by the codon-optimization.
The reason why AAV was used to deliver and express the RBD gene in our study was that it has been demonstrated to be a promising tool for delivery of foreign genes due to its advantages in high virus production and broad host range without causing any known human diseases (Stilwell and Samulski, 2003, Wang et al., 2000). Our results showed that as high as 5 × 1012 viral particles/ml of RBD-rAAV could be produced by transfection of 293T cells. It was also confirmed that the recombinant RBD-rAAV was able to efficiently infect different cell lines (Fig. 3) and express the RBD protein in transduced cell lines, which was recognized by a SARS-CoV neutralizing McAb (Fig. 4).
In the mouse model, vaccination with single dose of RBD-rAAV elicited a high level of SARS-CoV-specific antibodies, especially neutralizing antibodies, reaching the mean titer of 1:108 at 4 months post-immunization, which was higher than that induced by the inactivated virus vaccination with or without Alum (Fig. 5). It was further demonstrated that the mouse sera induced by the RBD-rAAV vaccination were able to effectively suppress RBD binding to ACE2 (Fig. 7), suggesting that the neutralizing activity were mediated by inhibition of the virus binding to its receptor on the host cell membrane. It has been reported that levels of SARS-CoV neutralizing antibody found in convalescent sera of recovered SARS patients ranged from 1:40 to 1:1280 (Zheng et al., 2004). Another study also shows that the titer of 1:100 of SARS-CoV neutralizing antibody elicited by a recombinant attenuated modified vaccinia virus Ankara (MVA) encoding SARS S protein protected immunized mice from SARS-CoV challenge (Bisht et al., 2004). Our results suggested that one dose vaccination of RBD-rAAV may provide sufficient protection against the SARS-CoV infection.
It had been found that, in some viral delivery systems, i.e., adenovirus and vaccinia virus systems, the primary immunization could induce host immunity against the delivery viral particle themselves, which would affect the efficacy of subsequent repeated vaccinations (Liu and Muruve, 2003, Muruve, 2004, Nasz and Adam, 2001). However, our results showed that the primary vaccination with RBD-rAAV did not affect the repeated vaccinations. The neutralizing antibody level markedly increased after the mice were boosted twice, reaching the mean titer of 1:512 that was 5-fold higher than the mice that received one dose of vaccination (Fig. 6). This may be attributed to that the low antigenicity of the AAV delivery vector used in this study minimized the host immune response to the delivery vector itself.
Compared to the inactivated SARS-CoV immunization, SARS-CoV-specific antibodies induced by either single dose or repeated RBD-rAAV vaccinations reached the highest level at least 1–1.5 months later, but the vaccinations were able to stimulate mice continuously producing SARS-CoV-specific antibodies with an increasing neutralizing activity, at least until the end point of our experiment (4 months for single dose and 5.5 months for repeated doses), indicating that the RBD-rAAV vaccination can deliver a prolonged immune response (Fig. 5, Fig. 6). This may be due to that the gene expression of the recombinant AAV goes through a slow onset firstly, taking a course of a few days or weeks, followed by persistent gene expression for many months, which is supported by reports that the AAV may long-term express foreign genes in vivo in different organisms without resulting in significant toxicity (Monahan and Samulski, 2000, Rabinowitz and Samulski, 1998, Smith-Arica and Bartlett, 2001). However, we cannot conclude if the antibody response has reached the highest level at the study end time and how long the high level of specific antibodies will be maintained in these RBD-rAAV-vaccinated mice, because we did not monitor the antibody responses for more than 5.5 months post-immunization.
In summary, our study demonstrated that the live AAV delivery system was an effective tool in delivery and expression of the RBD gene of SARS-CoV in the mouse model. The single dose of intramuscular immunization of RBD-rAAV induced viral-specific antibody responses, whose titer was significantly higher than that of the inactivated virus vaccination. The repeated vaccination elicited a prolonged immune response, resulting in an increase of SARS-CoV neutralizing antibody in vaccinated mice up to the end point of this study. Another advantage of the vaccines based on the live AAV delivery system is that it may be used for mucosal immunization to induce local immune response. It has been reported that high titers of fecal HIV-specific IgA and neutralizing antibodies were detected in HIV-rAAV immunized mice (Xin et al., 2001). A study of RBD-rAAV intranasal vaccination is in progress to further demonstrate the feasibility of the live AAV recombinant vaccine in development of SARS vaccines.
Materials and methods
Construction of the AAV plasmid expressing RBD of SARS-CoV S protein
Gene encoding the 193-aa of RBD of SARS-CoV S protein was codon optimized using overlapping primers according to the methods described previously (Gao et al., 2004, He et al., 2004b; Fig. 1a). The codon optimized RBD gene was cloned into the pMD18-T vector (Takara, Japan), which was used as the template for the first-round PCR with the forward primer 5′-CTGCTGGGGATGCTGGTCGCTAACATCACCAACCTGTGTCCC-3′ and the reverse primer 5′-TATATCTCGAGCACGGTGGCGGGCGCGTTCAGG-3′ (XhoI restriction enzyme site underlined, no stop codon). The amplified RBD fragment was then used as the template for the second-round PCR to amplify CD5-RBD, in which the gene of the CD5 signal peptide was ligated into the 5´ end of the RBD product, using the forward primer 5′-AAGGATCCAGAAACCATGCCCATGGGGTCTCTGCAACCGCTGGCCACCTTGTACCTGCTG-3′ (BamHI restriction site underlined, start codon labeled in bold), and the same reverse primer of the first round. The amplified CD5-RBD was digested by BamHI and XhoI, and subcloned into the MCS of the pAAV-IRES-hrGFP vector, in which hrGFP from a novel marine organism was expressed as a second open reading frame, and served as an indicator for the expression of the inserted RBD gene and an evaluator for the efficiency of transfection and infection in the subsequent experiments (Fig. 1B). The constructed recombinant plasmid was named as pRBD-AAV.
Production and purification of the AAVs
The RBD-rAAV and blank AAV were generated using the AAV-Helper-Free-System following the manufacturer's instructions (Stratagene, USA). Briefly, 1 day before transfection, 293T cells were seeded into 100-mm tissue culture dishes and maintained in Dulbecco's modified Eagle's medium (DMEM) (Invitrogen, USA) supplied with 10% fetal calf serum (FCS) (Invitrogen, USA). When 293T cells (providing the adenovirus E1A gene) reached 70–80% confluence, the pRBD-rAAV or pAAV plasmid was co-transfected with the pHelper plasmid (providing the adenovirus E2A, E4, and VA RNA genes) and the pAAV-RC plasmid (providing the AAV2 rep/cap genes) by a calcium phosphate transfection method (Stratagene, USA). Transfected cells were cultured at 37°C with 5% CO2 and fresh medium was added 10–12 h later. Both culture supernatant and transfected cells were harvested 72 h after transfection.
The produced AAVs were purified according to Wu et al. (2004) with some modifications. The harvested cells were lyzed by sonication and shaken at 37°C for 1 h after adding chloroform and NaCl respectively to 10% and 1 mol/L. The supernatant was incubated with PEG8000 (final concentration 10%) on ice for 1 h. The solutions from both cell lysate and supernatant were centrifuged at 12,000 rpm for 15 min at 4°C. The pellets were re-suspended in phosphate-buffered saline (PBS) buffer (pH 7.4) and then incubated with DNase I and RNase A at the final concentration of 1 μg/ml on ice for 30 min, followed by adding an equal volume of chloroform. The aqueous phases containing the purified AAVs were collected after centrifugation at 12,000 rpm for 5 min at 4°C and stored at −80°C until use.
Titration of the AAVs
The titers of generated AAVs were determined by real-time quantitative PCR using the forward primer 5′-ATTCTGAGTCCAAGCTAGGC-3′ and the reverse primer 5′-GCTTTTGTTCCCTTTAGTGAG-3′. Briefly, 10 μl of purified virus stock was treated with protease K and sodium dodecyl sulfate (SDS) at a final concentration of 0.3 mg/ml and 0.1%, respectively. Viral DNA was extracted using DNA extraction kit (Invitrogen, USA). The viral DNA with 10-fold serial dilutions was mixed with the primers and LighterCycler FastStart Reaction SYBR Green I reagent (Roche, USA). The real-time quantification was carried out in a LightCycler System (Roche, USA) with conditions of 95°C for 10 min, 45 cycles of 95°C for 10 s, 55°C for 5 s and 72°C for 10 s, followed by 60°C for 15 s. pRBD-AAV plasmid in 10-fold serial dilutions was used as the standard control.
Detection of the AAV infectivity
The infectivity of purified AAVs was tested in 293T and HeLa cells (ATCC, USA) using fluorescence microscopy and flow cytometry analysis. Purified virus was serially diluted in serum-free medium and added to the cell cultures, which were seeded in 96-well tissue culture plates 1 day before infection. Fresh DMEM supplied with 5% FCS was added 2 h later and continued the cultures at 37°C for 1–4 days. After 72 h of culture, the cells expressing hrGFP were observed under a fluorescence microscopy (Olympus IX70, USA). Infection rates of the AAVs in different cell lines were also monitored by detecting the percentage of cells expressing hrGFP using a flow cytometer (FACSCalibur, BD, USA).
Immunofluorescence assay
An immunofluorescence assay was performed to detect the RBD expression in infected HeLa cells. Briefly, cells were infected with 1 × 1010 particles of RBD-rAAV or blank AAV. Fresh DMEM supplied with 5% FCS was added 24 h later and continued the cultures for 48 h. Infected cells were fixed with 4% paraformaldehyde and incubated with McAb S1 (kindly provided by Dr. B. Sun at the Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences) at dilution of 1:1000 for 1 h at room temperature, followed by three washes in PBS and incubation with PE-conjugated goat anti-mouse IgG (eBioscience, USA) for 30 min. After 3 washes, the cells were detected for fluorescence.
Western blot assay
The expression of the RBD protein was also detected by a Western blot analysis. Briefly, RBD-rAAV or blank AAV-infected 293T and HeLa cells were harvested at 72 h after infection. The cells were washed three times with PBS and lyzed in cell lysis buffer [50 mM Tris, pH 7.5, 150 mM NaCl, 0.1% SDS, 2 mM EDTA, 0.5% NP-40, 10% glycerol and 1× protease inhibitor cocktail (Roche, USA)]. The expressed RBD was separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to nitrocellulose membranes (Amersham Biosciences, USA). The blots were blocked overnight with 5% skim milk in PBS containing 0.1% Tween 20 (PBS-T) at 4°C, followed by incubation with the McAb-S1 at dilution of 1:2000 for 1 h at room temperature. The blots were then washed three times and incubated with HRP-conjugated goat anti-mouse IgG (DAKO, Denmark) for 1 h at room temperature. Signals were visualized with ECL Western blotting substrate reagents (Amersham Biosciences, USA) and KODAK BioMax Scientific Imaging Film (Eastman Kodak).
Animal vaccinations and sample collection
Female BALB/c mice aged 6 weeks were used for the vaccination. Mice were separated into 9 groups (5 to 10 mice/group). They were intramuscularly (i.m.) vaccinated respectively with: (1) one dose (1011 particles in 200 μl PBS) of RBD-rAAV; (2) one dose (1011 particles in 200 μl PBS) of blank AAV; (3 and 4) one dose (80 μg protein in 200 μl PBS) of inactivated SARS-CoV with or without adjuvant (Alum) prepared as described by Qu et al. (2005); (5) three doses of RBD-rAAV with the second dose at 2nd week and the third dose at 2nd month; (6) three doses of blank AAV as a control of group 5; (7 and 8) three doses of inactivated SARS-CoV with or without Alum at 1-month interval; (9) three doses of PBS as a control of groups (7) and (8). Mouse sera were collected at 0, 1, 2 and 4 months post-vaccination for group 1 to 4, and at 0, 1, 2, 3, 4 and 5.5 months post-immunization for group 5 to 9, by retro-orbital puncture under diethyl ether anesthesia.
ELISA
Specific antibodies against SARS-CoV in mouse sera were tested by an enzyme-linked immunosorbent assay (ELISA) using the commercial SARS-CoV antibody detection kit (Beijing Genomics Institute, Beijing, China). Briefly, serially diluted mouse sera were added to 96-well microtiter plates coated with the protein mixture from SARS-CoV viral lysates. The plates were incubated at 37°C for 30 min, followed by four washes with PBS-T. Bound antibodies were then reacted with HRP-conjugated goat anti-mouse IgG (DAKO, Denmark) at 37°C for 20 min. After four washes, the substrate 3,3′,5,5′-tetramethylbenzidine (TMB) (Zymed, USA) was added to the plates and the reaction was stopped by adding 1 N H2SO4. The absorbance at 450 nm was measured by an ELISA plate reader (Victor 1420 Multilabel Counter, PerkinElmer, USA).
Neutralization assay
Titers of neutralizing antibodies in mouse sera immunized with RBD-rAAV, blank AAV or inactivated virus were detected in Vero E6 cells as described by Qu et al. (2005). Briefly, Vero E6 cells were seeded at 1 × 104 cells per well in 96-well culture plates and grown at 37°C overnight. Serial 2-fold dilutions of serum samples were separately mixed with 100 TCID50 (50% tissue-culture infectious dose) of SARS-CoV GZ50 strain (GenBank accession no. AY304495), incubated at 37°C for 1 h, and added to the monolayer of Vero E6 cells. In each assay, a virus back-titration (virus in serial 2-fold dilutions with medium), virus positive control (100 TCID50) and negative cell controls with medium were also included in parallel with the neutralization test. Cytopathic effect (CPE) was observed daily and the endpoints were read and recorded up to 3 days post-infection. The neutralizing antibody titer was determined based on the highest dilution of each serum, which completely suppressed CPE induced by the virus.
Inhibition of RBD binding to SARS-CoV receptor ACE2
Inhibitory effect of mouse anti-sera on RBD binding to the receptor ACE2 was tested by an ELISA assay (He et al., 2004b, He et al., 2005a). Briefly, recombinant soluble ACE2 (R&D systems) was coated to 96-well microtiter plates at 2 μg/ml concentration in 0.1 M carbonate buffer (pH 9.6) and incubated at 4°C overnight. After blocking with 2% skim milk, 1 μg/ml of RBD-Fc was added to the wells in the presence or absence of diluted sera from RBD-rAAV and blank AAV-vaccinated mice and incubated at 37°C for 1 h. The plates were washed twice with PBS-T and incubated with HRP-conjugated goat anti-human IgG (Zymed, USA) at 37°C for 1 h, followed by adding the substrate TMB for detection.
Statistical analysis
All values were presented as means ± standard error (SE). Statistical significance between RBD-rAAV and inactivated SARS-CoV in single and repeated vaccinations was calculated by Student's t test using Stata statistical software. P values less than 0.05 were considered significant.
Acknowledgments
This study was supported by Research Fund for the Control of Infectious Diseases of the Health, Welfare and Food Bureau of the Hong Kong SAR Government and Beijing SARS Research Grant (H030230100130). We thank Dr. B. Sun (Laboratory of Molecular Cell Biology, Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences, Shanghai, China) for providing the SARS-CoV S1-specific McAb.
Contributor Information
Yusen Zhou, Email: yszhou@bmi.ac.cn.
Bo-Jian Zheng, Email: bzheng@hkucc.hku.hk.
References
- Anand K., Ziebuhr J., Wadhwani P., Mesters J.R., Hilgenfeld R. Coronavirus main proteinase (3CLpro) structure: basis for design of anti-SARS drugs. Science. 2003;300:1763–1767. doi: 10.1126/science.1085658. [DOI] [PubMed] [Google Scholar]
- Babcock G.J., Esshaki D.J., Thomas W.D., Jr., Ambrosino D.M. Amino acids 270 to 510 of the severe acute respiratory syndrome coronavirus spike protein are required for interaction with receptor. J. Virol. 2004;78:4552–4560. doi: 10.1128/JVI.78.9.4552-4560.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bisht H., Roberts A., Vogel L., Bukreyev A., Collins P.L., Murphy B.R., Subbarao K., Moss B. Severe acute respiratory syndrome coronavirus spike protein expressed by attenuated vaccinia virus protectively immunizes mice. Proc. Natl. Acad. Sci. U.S.A. 2004;101:6641–6646. doi: 10.1073/pnas.0401939101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bisht H., Roberts A., Vogel L., Subbarao K., Moss B. Neutralizing antibody and protective immunity to SARS coronavirus infection of mice induced by a soluble recombinant polypeptide containing an N-terminal segment of the spike glycoprotein. Virology. 2005;334:160–165. doi: 10.1016/j.virol.2005.01.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bosch B.J., van der Zee R., de Haan C.A., Rottier P.J. The coronavirus spike protein is a class I virus fusion protein: structural and functional characterization of the fusion core complex. J. Virol. 2003;77:8801–8811. doi: 10.1128/JVI.77.16.8801-8811.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buchholz U.J., Bukreyev A., Yang L., Lamirande E.W., Murphy B.R., Subbarao K., Collins P.L. Contributions of the structural proteins of severe acute respiratory syndrome coronavirus to protective immunity. Proc. Natl. Acad. Sci. U.S.A. 2004;101:9804–9809. doi: 10.1073/pnas.0403492101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bukreyev A., Lamirande E.W., Buchholz U.J., Vogel L.N., Elkins W.R., St Claire M., Murphy B.R, Subbarao K., Collins P.L. Mucosal immunisation of African green monkeys (Cercopithecus aethiops) with an attenuated parainfluenza virus expressing the SARS coronavirus spike protein for the prevention of SARS. Lancet. 2004;363:2122–2127. doi: 10.1016/S0140-6736(04)16501-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chao H., Sun L., Bruce A., Xiao X., Walsh C.E. Expression of human factor VIII by splicing between dimerized AAV vectors. Mol. Ther. 2002;5:716–722. doi: 10.1006/mthe.2002.0607. [DOI] [PubMed] [Google Scholar]
- Chen Z., Zhang L., Qin C., Ba L., Yi C.E., Zhang F., Wei Q., He T., Yu W., Yu J., Gao H., Tu X., Gettie A., Farzan M., Yuen K.Y., Ho D.D. Recombinant modified vaccinia virus Ankara expressing the spike glycoprotein of severe acute respiratory syndrome coronavirus induces protective neutralizing antibodies primarily targeting the receptor binding region. J. Virol. 2005;79:2678–2688. doi: 10.1128/JVI.79.5.2678-2688.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheung Y.K., Cheng S.C., Sin F.W., Xie Y. Plasmid encoding papillomavirus Type 16 (HPV16) DNA constructed with codon optimization improved the immunogenicity against HPV infection. Vaccine. 2004;23:629–638. doi: 10.1016/j.vaccine.2004.07.010. [DOI] [PubMed] [Google Scholar]
- Dimitrov D.S. The secret life of ACE2 as a receptor for the SARS virus. Cell. 2003;115:652–653. doi: 10.1016/S0092-8674(03)00976-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drosten C., Gunther S., Preiser W., van der Werf S., Brodt H.R., Becker S., Rabenau H., Panning M., Kolesnikova L., Fouchier R.A., Berger A., Burguiere A.M., Cinatl J., Eickmann M., Escriou N., Grywna K., Kramme S., Manuguerra J.C., Muller S., Rickerts V., Sturmer M., Vieth S., Klenk H.D., Osterhaus A.D., Schmitz H., Doerr H.W. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N. Engl. J. Med. 2003;348:1967–1976. doi: 10.1056/NEJMoa030747. [DOI] [PubMed] [Google Scholar]
- Faber M., Lamirande E.W., Roberts A., Rice A.B., Koprowski H., Dietzschold B., Schnell M.J. A single immunization with a rhabdovirus-based vector expressing severe acute respiratory syndrome coronavirus (SARS-CoV) S protein results in the production of high levels of SARS-CoV-neutralizing antibodies. J. Gen. Virol. 2005;86:1435–1440. doi: 10.1099/vir.0.80844-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frelin L., Ahlen G., Alheim M., Weiland O., Barnfield C., Liljestrom P., Sallberg M. Codon optimization and mRNA amplification effectively enhances the immunogenicity of the hepatitis C virus nonstructural 3/4A gene. Gene Ther. 2004;11:522–533. doi: 10.1038/sj.gt.3302184. [DOI] [PubMed] [Google Scholar]
- Gallagher T.M., Buchmeier M.J. Coronavirus spike proteins in viral entry and pathogenesis. Virology. 2001;279:371–374. doi: 10.1006/viro.2000.0757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao F., Li Y., Decker J.M., Peyerl F.W., Bibollet-Ruche F., Rodenburg C.M., Chen Y., Shaw D.R., Allen S., Musonda R., Shaw G.M., Zajac A.J., Letvin N., Hahn B.H. Codon usage optimization of HIV type 1 subtype C gag, pol, env, and nef genes: in vitro expression and immune responses in DNA-vaccinated mice. AIDS Res. Hum. Retroviruses. 2003;19:817–823. doi: 10.1089/088922203769232610. [DOI] [PubMed] [Google Scholar]
- Gao W., Tamin A., Soloff A., D'Aiuto L., Nwanegbo E., Robbins P.D., Bellini W.J., Barratt-Boyes S., Gambotto A. Effects of a SARS-associated coronavirus vaccine in monkeys. Lancet. 2003;362:1895–1896. doi: 10.1016/S0140-6736(03)14962-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao W., Rzewski A., Sun H., Robbins P.D., Gambotto A. UpGene: application of a web-based DNA codon optimization algorithm. Biotechnol. Prog. 2004;20:443–448. doi: 10.1021/bp0300467. [DOI] [PubMed] [Google Scholar]
- He Y., Zhou Y., Siddiqui P., Jiang S. Inactivated SARS-CoV vaccine elicits high titers of spike protein-specific antibodies that block receptor binding and virus entry. Biochem. Biophys. Res. Commun. 2004;325:445–452. doi: 10.1016/j.bbrc.2004.10.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He Y., Zhou Y., Liu S., Kou Z., Li W., Farzan M., Jiang S. Receptor-binding domain of SARS-CoV spike protein induces highly potent neutralizing antibodies: implication for developing subunit vaccine. Biochem. Biophys. Res. Commun. 2004;324:773–781. doi: 10.1016/j.bbrc.2004.09.106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He Y., Lu H., Siddiqui P., Zhou Y., Jiang S. Receptor-binding domain of severe acute respiratory syndrome coronavirus spike protein contains multiple conformation-dependent epitopes that induce highly potent neutralizing antibodies. J. Immunol. 2005;174:4908–4915. doi: 10.4049/jimmunol.174.8.4908. [DOI] [PubMed] [Google Scholar]
- He Y., Zhu Q., Liu S., Zhou Y., Yang B., Li J., Jiang S. Identification of a critical neutralization determinant of severe acute respiratory syndrome (SARS)-associated coronavirus: importance for designing SARS vaccines. Virology. 2005;334:74–82. doi: 10.1016/j.virol.2005.01.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hofmann H., Hattermann K., Marzi A., Gramberg T., Geier M., Krumbiegel M., Kuate S., Uberla K., Niedrig M., Pohlmann S. S protein of severe acute respiratory syndrome-associated coronavirus mediates entry into hepatoma cell lines and is targeted by neutralizing antibodies in infected patients. J. Virol. 2004;78:6134–6142. doi: 10.1128/JVI.78.12.6134-6142.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holmes K.V. SARS-associated coronavirus. N. Engl. J. Med. 2003;348:1948–1951. doi: 10.1056/NEJMp030078. [DOI] [PubMed] [Google Scholar]
- Holmes K.V., Enjuanes L. Virology. The SARS coronavirus: a postgenomic era. Science. 2003;300:1377–1378. doi: 10.1126/science.1086418. [DOI] [PubMed] [Google Scholar]
- Kaspar B.K., Vissel B., Bengoechea T., Crone S., Randolph-Moore L., Muller R., Brandon E.P., Schaffer D., Verma I.M., Lee K.F., Heinemann S.F., Gage F.H. Adeno-associated virus effectively mediates conditional gene modification in the brain. Proc. Natl. Acad. Sci. U.S.A. 2002;99:2320–2325. doi: 10.1073/pnas.042678699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keir S.D., House S.B., Li J., Xiao X., Gainer H. Gene transfer into hypothalamic organotypic cultures using an adeno-associated virus vector. Exp. Neurol. 1999;160:313–316. doi: 10.1006/exnr.1999.7236. [DOI] [PubMed] [Google Scholar]
- Keng C.T., Zhang A., Shen S., Lip K.M., Fielding B.C., Tan T.H., Chou C.F., Loh C.B., Wang S., Fu J., Yang X., Lim S.G., Hong W., Tan Y.J. Amino acids 1055 to 1192 in the S2 region of severe acute respiratory syndrome coronavirus S protein induce neutralizing antibodies: implications for the development of vaccines and antiviral agents. J. Virol. 2005;79:3289–3296. doi: 10.1128/JVI.79.6.3289-3296.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ko H.J., Ko S.Y., Kim Y.J., Lee E.G., Cho S.N., Kang C.Y. Optimization of codon usage enhances the immunogenicity of a DNA vaccine encoding mycobacterial antigen Ag85B. Infect. Immun. 2005;73:5666–5674. doi: 10.1128/IAI.73.9.5666-5674.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krokhin O., Li Y., Andonov A., Feldmann H., Flick R., Jones S., Stroeher U., Bastien N., Dasuri K.V., Cheng K., Simonsen J.N., Perreault H., Wilkins J., Ens W., Plummer F., Standing K.G. Mass spectrometric characterization of proteins from the SARS virus: a preliminary report. Mol. Cell. Proteomics. 2003;2:346–356. doi: 10.1074/mcp.M300048-MCP200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ksiazek T.G., Erdman D., Goldsmith C.S., Zaki S.R., Peret T., Emery S., Tong S., Urbani C., Comer J.A., Lim W., Rollin P.E., Dowell S.F., Ling A.E., Humphrey C.D., Shieh W.J., Guarner J., Paddock C.D., Rota P., Fields B., DeRisi J., Yang J.Y., Cox N., Hughes J.M., LeDuc J.W., Bellini W.J., Anderson L.J. A novel coronavirus associated with severe acute respiratory syndrome. N. Engl. J. Med. 2003;348:1953–1966. doi: 10.1056/NEJMoa030781. [DOI] [PubMed] [Google Scholar]
- Li W., Moore M.J., Vasilieva N., Sui J., Wong S.K., Berne M.A., Somasundaran M., Sullivan J.L., Luzuriaga K., Greenough T.C., Choe H., Farzan M. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. 2003;426:450–454. doi: 10.1038/nature02145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lieber A. AAV display–homing in on the target. Nat. Biotechnol. 2003;21:1011–1013. doi: 10.1038/nbt0903-1011. [DOI] [PubMed] [Google Scholar]
- Liu Q., Muruve D.A. Molecular basis of the inflammatory response to adenovirus vectors. Gene Ther. 2003;10:935–940. doi: 10.1038/sj.gt.3302036. [DOI] [PubMed] [Google Scholar]
- Marra M.A., Jones S.J., Astell C.R., Holt R.A., Brooks-Wilson A., Butterfield Y.S., Khattra J., Asano J.K., Barber S.A., Chan S.Y., Cloutier A., Coughlin S.M., Freeman D., Girn N., Griffith O.L., Leach S.R., Mayo M., McDonald H., Montgomery S.B., Pandoh P.K., Petrescu A.S., Robertson A.G., Schein J.E., Siddiqui A., Smailus D.E., Stott J.M., Yang G.S., Plummer F., Andonov A., Artsob H., Bastien N., Bernard K., Booth T.F., Bowness D., Czub M., Drebot M., Fernando L., Flick R., Garbutt M., Gray M., Grolla A., Jones S., Feldmann H., Meyers A., Kabani A., Li Y., Normand S., Stroher U., Tipples G.A., Tyler S., Vogrig R., Ward D., Watson B., Brunham R.C., Krajden M., Petric M., Skowronski D.M., Upton C., Roper R.L. The genome sequence of the SARS-associated coronavirus. Science. 2003;300:1399–1404. doi: 10.1126/science.1085953. [DOI] [PubMed] [Google Scholar]
- Monahan P., Samulski R. AAV vectors: is clinical success on the horizon. Gene Ther. 2000;7:24–30. doi: 10.1038/sj.gt.3301109. [DOI] [PubMed] [Google Scholar]
- Moore M.J., Dorfman T., Li W., Wong S.K., Li Y., Kuhn J.H., Coderre J., Vasilieva N., Han Z., Greenough T.C., Farzan M., Choe H. Retroviruses pseudotyped with the severe acute respiratory syndrome coronavirus spike protein efficiently infect cells expressing angiotensin-converting enzyme 2. J Virol. 2004;78:10628–10635. doi: 10.1128/JVI.78.19.10628-10635.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muruve D.A. The innate immune response to adenovirus vectors. Hum. Gene Ther. 2004;15:1157–1166. doi: 10.1089/hum.2004.15.1157. [DOI] [PubMed] [Google Scholar]
- Nasz I., Adam E. Recombinant adenovirus vectors for gene therapy and clinical trials. Acta Microbiol. Immunol. Hung. 2001;48:323–348. doi: 10.1556/AMicr.48.2001.3-4.6. [DOI] [PubMed] [Google Scholar]
- Nie Y., Wang G., Shi X., Zhang H., Qiu Y., He Z., Wang W., Lian G., Yin X., Du L., Ren L., Wang J., He X., Li T., Deng H., Ding M. Neutralizing antibodies in patients with severe acute respiratory syndrome-associated coronavirus infection. J. Infect. Dis. 2004;190:1119–1126. doi: 10.1086/423286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peiris J.S., Lai S.T., Poon L.L., Guan Y., Yam L.Y., Lim W., Nicholls J., Yee W.K., Yan W.W., Cheung M.T., Cheng V.C., Chan K.H., Tsang D.N., Yung R.W., Ng T.K., Yuen K.Y. Coronavirus as a possible cause of severe acute respiratory syndrome. Lancet. 2003;361:1319–1325. doi: 10.1016/S0140-6736(03)13077-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prabakaran P., Xiao X., Dimitrov D.S. A model of the ACE2 structure and function as a SARS-CoV receptor. Biochem. Biophys. Res. Commun. 2004;314:235–241. doi: 10.1016/j.bbrc.2003.12.081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qu D., Zheng B., Yao X., Guan Y., Yuan Z.H., Zhong N.S., Lu L.W., Xie J.P., Wen Y.M. Intranasal immunization with inactivated SARS-CoV (SARS-associated coronavirus) induced local and serum antibodies in mice. Vaccine. 2005;23:924–931. doi: 10.1016/j.vaccine.2004.07.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rabinowitz J.E., Samulski J. Adeno-associated virus expression systems for gene transfer. Curr. Opin. Biotechnol. 1998;9:470–475. doi: 10.1016/s0958-1669(98)80031-1. [DOI] [PubMed] [Google Scholar]
- Ramakrishna L., Anand K.K., Mohankumar K.M., Ranga U. Codon optimization of the tat antigen of human immunodeficiency virus type 1 generates strong immune responses in mice following genetic immunization. J. Virol. 2004;78:9174–9189. doi: 10.1128/JVI.78.17.9174-9189.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romano G., Michell P., Pacilio C., Giordano A. Latest developments in gene transfer technology: achievements, perspectives, and controversies over therapeutic applications. Stem Cells. 2000;18:19–39. doi: 10.1634/stemcells.18-1-19. [DOI] [PubMed] [Google Scholar]
- Rota P.A., Oberste M.S., Monroe S.S., Nix W.A., Campagnoli R., Icenogle J.P., Penaranda S., Bankamp B., Maher K., Chen M.H., Tong S., Tamin A., Lowe L., Frace M., DeRisi J.L., Chen Q., Wang D., Erdman D.D., Peret T.C., Burns C., Ksiazek T.G., Rollin P.E., Sanchez A., Liffick S., Holloway B., Limor J., McCaustland K., Olsen-Rasmussen M., Fouchier R., Gunther S., Osterhaus A.D., Drosten C., Pallansch M.A., Anderson L.J., Bellini W.J. Characterization of a novel coronavirus associated with severe acute respiratory syndrome. Science. 2003;300:1394–1399. doi: 10.1126/science.1085952. [DOI] [PubMed] [Google Scholar]
- Smith J.M., Amara R.R., Campbell D., Xu Y., Patel M., Sharma S., Butera S.T., Ellenberger D.L., Yi H., Chennareddi L., Herndon J.G., Wyatt L.S., Montefiori D., Moss B., McClure H.M., Robinson H.L. DNA/MVA vaccine for HIV type 1: effects of codon-optimization and the expression of aggregates or virus-like particles on the immunogenicity of the DNA prime. AIDS Res. Hum. Retroviruses. 2004;20:1335–1347. doi: 10.1089/aid.2004.20.1335. [DOI] [PubMed] [Google Scholar]
- Smith-Arica J.R., Bartlett J.S. Gene therapy: recombinant adeno-associated virus vectors. Curr. Cardiol. Rep. 2001;3:43–49. doi: 10.1007/s11886-001-0009-x. [DOI] [PubMed] [Google Scholar]
- Stilwell J.L., Samulski R.J. Adeno-associated virus vectors for therapeutic gene transfer. BioTechniques. 2003;34:148–150. doi: 10.2144/03341dd01. [DOI] [PubMed] [Google Scholar]
- Sui J., Li W., Murakami A., Tamin A., Matthews L.J., Wong S.K., Moore M.J., Tallarico A.S., Olurinde M., Choe H., Anderson L.J., Bellini W.J., Farzan M., Marasco W.A. Potent neutralization of severe acute respiratory syndrome (SARS) coronavirus by a human mAb to S1 protein that blocks receptor association. Proc. Natl. Acad. Sci. U.S.A. 2004;101:2536–2541. doi: 10.1073/pnas.0307140101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomar R.S., Matta H., Chaudhary P.M. Use of adeno-associated viral vector for delivery of small interfering RNA. Oncogene. 2003;22:5712–5715. doi: 10.1038/sj.onc.1206733. [DOI] [PubMed] [Google Scholar]
- Traggiai E., Becker S., Subbarao K., Kolesnikova L., Uematsu Y., Gismondo M.R., Murphy B.R., Rappuoli R., Lanzavecchia A. An efficient method to make human monoclonal antibodies from memory B cells: potent neutralization of SARS coronavirus. Nat. Med. 2004;10:871–875. doi: 10.1038/nm1080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsai T.H., Chen S.L., Xiao X., Liu D.W., Tsao Y.P. Gene therapy for treatment of cerebral ischemia using defective recombinant adeno-associated virus vectors. Methods. 2002;28:253–258. doi: 10.1016/s1046-2023(02)00230-x. [DOI] [PubMed] [Google Scholar]
- Wang B., Li J., Xiao X. Adeno-associated virus vector carrying human minidystrophin genes effectively ameliorates muscular dystrophy in mdx mouse model. Proc. Natl. Acad. Sci. U.S.A. 2000;97:13714–13719. doi: 10.1073/pnas.240335297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang S., Chou T.H., Sakhatskyy P.V., Huang S., Lawrence J.M., Cao H., Huang X., Lu S. Identification of two neutralizing regions on the severe acute respiratory syndrome coronavirus spike glycoprotein produced from the mammalian expression system. J. Virol. 2005;79:1906–1910. doi: 10.1128/JVI.79.3.1906-1910.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang S., Farfan-Arribas D.J., Shen S., Chou T.H., Hirsch A., He F., Lu S. Relative contributions of codon usage, promoter efficiency and leader sequence to the antigen expression and immunogenicity of HIV-1 Env DNA vaccine. Vaccine. 2005 doi: 10.1016/j.vaccine.2005.08.023. (Epub ahead of print) [DOI] [PubMed] [Google Scholar]
- Wong S.K., Li W., Moore M.J., Choe H., Farzan M. A 193-amino-acid fragment of the SARS coronavirus S protein efficiently binds angiotensin-converting enzyme 2. J. Biol. Chem. 2003;279:3197–3201. doi: 10.1074/jbc.C300520200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu X.B., Dong X.Y., Wu Z.J., Cao H., Niu D.B., Qu J.G., Wang H., Hou Y.D. A novel method for purification of recombinant adeno-associated virus vectors on a large scale. Chin. Sci. Bull. 2004;46:485–489. [Google Scholar]
- Xiao X., Chakraborti S., Dimitrov A.S., Gramatikoff K., Dimitrov D.S. The SARS-CoV S glycoprotein: expression and functional characterization. Biochem. Biophys. Res. Commun. 2003;312:1159–1164. doi: 10.1016/j.bbrc.2003.11.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xin K.Q., Urabe M., Yang J., Nomiyama K., Mizukami H., Hamajima K., Nomiyama H., Saito T., Imai M., Monahan J., Okuda K., Ozawa K., Okuda K. A novel recombinant adeno-associated virus vaccine induces a long-term humoral immune response to human immunodeficiency virus. Hum. Gene Ther. 2001;12:1047–1061. doi: 10.1089/104303401750214276. [DOI] [PubMed] [Google Scholar]
- Yang Z.Y., Kong W.P., Huang Y., Roberts A., Murphy B.R., Subbarao K., Nabel G.J. A DNA vaccine induces SARS coronavirus neutralization and protective immunity in mice. Nature. 2004;428:561–564. doi: 10.1038/nature02463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ying W., Hao Y., Zhang Y., Peng W., Qin E., Cai Y., Wei K., Wang J., Chang G., Sun W., Dai S., Li X., Zhu Y., Li J., Wu S., Guo L., Dai J., Wang J., Wan P., Chen T., Du C., Li D., Wan J., Kuai X., Li W., Shi R., Wei H., Cao C., Yu M., Liu H., Dong F., Wang D., Zhang X., Qian X., Zhu Q., He F. Proteomic analysis on structural proteins of severe acute respiratory syndrome coronavirus. Proteomics. 2004;4:492–504. doi: 10.1002/pmic.200300676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu X.J., Luo C., Lin J.C., Hao P., He Y.Y., Guo Z.M., Qin L., Su J., Liu B.S., Huang Y., Nan P., Li C.S., Xiong B., Luo X.M., Zhao G.P., Pei G., Chen K.X., Shen X., Shen J.H., Zou J.P., He W.Z., Shi T.L., Zhong Y., Jiang H.L., Li Y.X. Putative hAPN receptor binding sites in SARS-CoV spike protein. Acta Pharmacol. Sin. 2003;24:481–488. [PubMed] [Google Scholar]
- Zeng F., Chow K.Y., Hon C.C., Law K.M., Yip C.W., Chan K.H., Peiris J.S., Leung F.C. Characterization of humoral responses in mice immunized with plasmid DNAs encoding SARS-CoV spike gene fragments. Biochem. Biophys. Res. Commun. 2004;315:1134–1139. doi: 10.1016/j.bbrc.2004.01.166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang H., Wang G., Li J., Nie Y., Shi X., Lian G., Wang W., Yin X., Zhao Y., Qu X., Ding M., Deng H. Identification of an antigenic determinant on the S2 domain of the severe acute respiratory syndrome coronavirus spike glycoprotein capable of inducing neutralizing antibodies. J. Virol. 2004;78:6938–6945. doi: 10.1128/JVI.78.13.6938-6945.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng B.J., Wong K.H., Zhou J., Wong K.L., Young B.W., Lu L.W., Lee S.S. SARS-related virus predating SARS outbreak, Hong Kong. Emerg. Infect. Dis. 2004;10:176–178. doi: 10.3201/eid1002.030533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng B.J., Guan Y., He M.L., Sun H., Du L., Zheng Y., Wong K.L., Chen H., Chen Y., Lu L., Tanner J.A., Watt R.M., Niccolai N., Bernini A., Spiga O., Woo P.C., Kung H.F., Yuen K.Y., Huang J.D. Synthetic peptides outside the spike protein heptad repeat regions as potent inhibitors of SARS-associated coronavirus. Antivir. Ther. 2005;10:393–403. [PubMed] [Google Scholar]


