Abstract
There has been considerable investigation of host-microbial interactions in patients with chronic rhinosinusitis (CRS) in hopes of elucidating mechanisms of disease and better treatment. Most attention has been paid to bacterial infection and potential underlying defects in innate immunity. Bacterial biofilm is present in most patients with CRS undergoing surgical intervention, and its presence is associated with more severe disease and worse surgical outcomes. A role for viral or fungal infection in patients with CRS is less clear. There is no evidence for a primary defect in mucociliary clearance in most patients with CRS. Decreased levels of certain antimicrobial proteins, most notably lactoferrin, have been found in sinus secretions, whereas levels of other antimicrobial proteins have been found to be normal. No primary defects in Toll-like receptors have been found in patients with CRS, although a 50% reduced expression of Toll-like receptor 9 was reported in patients with recalcitrant nasal polyps. A polymorphism in a bitter taste receptor was recently associated with refractory CRS and persistent Pseudomonas aeruginosa infection. A downregulation of innate immunity by maladaptive TH2 tissue inflammation has also been described in patients with recalcitrant nasal polyps, suggesting a link to persistent infection. To date, an effective means of restoring host-microbial balance and mitigating disease in patients with CRS remains elusive.
Key words: Chronic rhinosinusitis, host, microbial, biofilm, innate, immunity, antimicrobial
Abbreviations used: AFRS, Allergic fungal rhinosinusitis; BPI, Bactericidal/permeability-increasing protein; CD, Crohn disease; Cp110, Centrosomal protein 110; CRS, Chronic rhinosinusitis; CRSsNP, Chronic rhinosinusitis without nasal polyps; CRSwNP, Chronic rhinosinusitis with nasal polyposis; CSLM, Confocal scanning laser microscopy; DMBT1, Deleted in malignant brain tumor 1; EMCRS, Eosinophilic mucin chronic rhinosinusitis; FISH, Fluorescence in situ hybridization; hBD, Human β-defensin; HC, Healthy control subject; IBD, Inflammatory bowel disease; IESA, Intraepithelial Staphylococcus aureus; IL-22R, IL-22 receptor; LBP, LPS-binding protein; MBL, Mannose-binding lectin; NO, Nitric oxide; NOD, Nucleotide-binding oligomerization domain; NP, Nasal polyp; PCD, Primary ciliary dyskinesia; PLUNC, Palate lung and nasal epithelium clone; PNEC, Cultured primary nasal epithelial cell; SEB, Staphylococcal enterotoxin B; SEM, Scanning electron microscopy; SLPI, Secretory leukocyte proteinase inhibitor; SP-A, Surfactant protein A; SP-D, Surfactant protein D; TEM, Transmission electron microscopy; TLR, Toll-like receptor
Discuss this article on the JACI Journal Club blog: www.jaci-online.blogspot.com.
Host-microbial interactions play a critical role in CRS disease initiation and perpetuation. This article aims to summarize knowledge of host-microbial interactions elucidated in relation to normal sinus physiology and pathology of patients with chronic rhinosinusitis (CRS), including the subsets regarded as chronic rhinosinusitis without nasal polyps (CRSsNP), chronic rhinosinusitis with nasal polyposis (CRSwNP), and allergic fungal rhinosinusitis (AFRS).1
Most studies of innate immunity and host-microbial interactions in patients with CRS have focused on patients with “refractory” or “recalcitrant” disease. Refractory CRS has been defined on the basis of failure to stabilize after surgery, antibiotics, saline rinses, and topical steroid treatment.2 Somewhat differently, “recalcitrant CRS” has been defined based on recurrence of nasal polyps (NPs) after polyp surgery.3 These definitions are noteworthy because patients with refractory polyposis, for example, might have more evidence of infection, whereas patients with recalcitrant polyposis might have little or no evidence of infection but more evidence for maladaptive TH2-biased mucosal inflammation.
Microbiology of CRS
Role of viruses
Viral upper respiratory tract infections are potentially highly relevant to CRS. The average healthy adult person experiences 1 to 3 common colds per year (http://www.niaid.nih.gov/topics/commoncold/pages/overview.aspx). In healthy subjects the onset and time course of cold symptoms and levels of viral mRNA detectable in nasal secretions over 21 days have been mapped out after experimental rhinovirus infection.4, 5 Patients with asthma or chronic obstructive pulmonary disease manifest a significantly higher peak rhinovirus 16 viral load and duration of symptoms. Asthmatic patients also manifest a corresponding 10-fold decreased induction of type I (β) and type III (λ1 and λ2/3) interferons.6 Given the similarities between asthma and CRS at the tissue level and the fact that many CRS exacerbations occur during the viral season,7 it is plausible that a similar defect exists in patients with CRS. However, experimental rhinovirus infection has not been studied in patients with CRS. The innate antiviral response to rhinovirus infection involves activation of type I interferons through interferon-regulatory factor 1 gene activation, an increase in nitric oxide (NO) production, and epithelial production of human β-defensin (hBD) 2, IL-8, and RANTES.8, 9 IL-17A was found to augment production of hBD-2 and IL-8 but downregulate production of RANTES in this model. The chemokine CXCL10 (interferon-inducible protein 10) is also induced.10
There has not been a study of the incidence of rhinovirus infection in patients with CRS. A study by Jang et al11 reported that 21% of patients with CRS have detectable rhinovirus infection. This study examined nasal lavage fluid and turbinate epithelial cells (collected with a Rhino-probe mucosal curette; Rhino-Probe, Arlington Scientific, Arlington, Tex) from 39 patients with CRS and 27 healthy control subjects (HCs). Using an RT-PCR–based assay, they found that lavage fluid from all patients with CRS and HCs and turbinate epithelial cells from HCs were negative for picornavirus, whereas 8 (21%) of 39 epithelial cell samples from patients with CRS were positive. Further examination revealed that all 8 patients with CRS with positive results had positive results for rhinovirus. It is unclear whether these represented subclinical infections because patients were studied at only 1 time point.
In an in vitro experiment Wang et al12 infected NPs and nasal turbinate epithelial cells from 16 patients with CRSwNP and sphenoid sinus and turbinate epithelial cells from 19 HCs with rhinovirus (rhinovirus 16). No significant differences in rates of infection or induction of IL-6 or IL-8 were found.12 Our group found that cultured airway epithelial cells from patients with CRSsNP had an exaggerated response to stimulation with the combination of double-stranded RNA (a Toll-like receptor [TLR] 3 agonist and surrogate for viral infection) plus cigarette smoke extract, with exaggerated production of RANTES and hBD-2.13
Finally, although studies are quite limited, there is a lack of evidence for persistence of viral infection in patients with CRS. Again using PCR methodology, Wood et al14 found no evidence for common respiratory tract viruses, including parainfluenza 1, 2, and 3; respiratory syncytial virus; human metapneumovirus; adenovirus; rhinovirus; coronavirus; bocavirus; cytomegalovirus; or influenza A or B virus in sinus mucosal samples from 13 patients with CRS.14
Whether upper respiratory tract viruses could contribute causally to the inception of CRS analogous to their hypothesized role in asthma pathogenesis remains unexplored.15
Bacterial involvement in patients with CRS
Bacteriology of CRS determined by using conventional culture techniques
Studies with conventional culture techniques in children with CRS cultured in the absence of antibiotic treatment reported positive cultures in roughly 60% of cases, with the most common pathogens being Haemophilus influenzae, Streptococcus pneumoniae, and Moraxella catarrhalis (reviewed by Meltzer et al1). Studies by Brook et al16, 17 using special techniques to optimize recovery of anaerobic bacteria identified these bacteria in roughly 80% of children with CRS.
Prospective studies in adults identified a positive bacterial culture in a variable percentage of patients with CRS (reviewed by Meltzer et al1). Coagulase-negative Staphylococcus species was the most common aerobic isolate in several studies, often accompanied by Staphylococcus aureus and viridians streptococci. Organisms associated with acute bacterial rhinosinusitis were cultured in some cases. In several studies gram-negative enteric rods, including Pseudomonas aeruginosa, Klebsiella pneumoniae, Proteus mirabilis, Enterobacter species, and Escherichia coli were also isolated. These organisms are rarely found in middle meatus cultures from healthy subjects. More recent studies of intraoperative sinus cultures with simultaneous analysis of cultures and biofilm reported positive cultures in 72.6% to 80% of cases, with a predominance of S aureus and P aeruginosa in the isolates.18, 19, 20
The frequency with which anaerobic organisms have been recovered from adults with CRS has varied widely, with anaerobes found mainly by investigators using special techniques to optimize their recovery.21, 22 Several species, including pigmented Prevotella, Fusobacterium, Bacteroides, and Peptostreptococcus species, were isolated. In support of a role for anaerobic bacteria in chronic maxillary sinusitis, Finegold et al22 found recurrence of signs and symptoms to be twice as frequent when cultures yielding anaerobic bacterial counts of greater than 103 cfu/mL. Further supportive evidence came from the detection of IgG antibodies to anaerobic organisms commonly recovered from sinus aspirates, namely Fusobacterium nucleatum and Prevotella intermedia. Antibody levels to these organisms decreased in the patients who responded to therapy but did not decrease in those in whom therapy failed.23 Recent studies with molecular techniques have shed new light on the potential role of anaerobes in patients with CRS (see below).
Role of atypical bacterial infection in patients with CRS
Studies with conventional culture techniques or molecular techniques to overcome issues of detection of nonculturable bacteria suggest that atypical mycobacterial infection is rare in patients with CRS but should be sought in patients with refractory CRS (see additional text in this article's Online Repository at www.jacionline.org).
Bacterial biofilm in patients with CRS
Biofilm formation is an important survival mechanism for microorganisms through attachment to surfaces.24 Formation of biofilm is a complex process controlled by different genetic pathways depending on growth conditions and exposure to membrane-targeting antibiotics.25 Furthermore, biofilm-associated bacteria are known to have enhanced resistance to antimicrobial agents relative to floating (planktonic) bacteria.26 Biofilm formation on sinonasal mucosal surfaces was first described in 200427 and later in several other studies.28, 29, 30, 31, 32, 33
Multiple techniques for biofilm detection have been described and are discussed in the additional text in this article's Online Repository. Table E1 in this article's Online Repository at www.jacionline.org summarizes the results of several studies of biofilm (including fungal biofilm) in patients with CRS, including the techniques used for biofilm identification. Most studies have not subcategorized patients as having CRSsNP, CRSwNP, AFRS, or eosinophilic mucin chronic rhinosinusitis (EMCRS). One study34 found biofilm to be present in only 2 of 12 patients with NPs, a lower prevalence than reported in most other studies of CRS. When the studies using scanning electron microscopy (SEM), transmission electron microscopy (TEM), or confocal scanning laser microscopy (CSLM) are taken in total (excluding the study confined to patients with NPs), the prevalence of biofilm in the CRS case series summarized in Table E1 was 56.3%.
Is the presence of biofilm of prognostic value?
The presence of bacterial biofilm, as determined by using CSLM, was found to be associated with more severe sinus disease preoperatively (worse radiologic and symptom scoring) and worse sinus symptom and nasal endoscopy scores 16 months after surgery.30, 35 Single-organism H influenzae biofilm was associated with mild clinical and radiographic disease and normalization of sinus mucosa a short time after surgery. In contrast, polymicrobial biofilm or biofilm containing S aureus was associated with more severe disease and a poorer postoperative course.36 The presence of bacterial biofilm was strongly associated with persistent mucosal inflammation after endoscopic sinus surgery (53% of biofilm-positive patients vs 0% of biofilm-negative patients).31 One study showed that the likelihood of detecting bacteria with biofilm-forming capacity increases in relation to prior endoscopic sinus surgery, possibly reflecting the severity of their disease.37 Similarly, Zhang et al38 found an association of biofilm forming-capacity in vitro from clinical samples with prior sinus surgeries and nasal steroid use in the month before sample collection but no association with the presence of NPs, allergy, or Samter triad and an association with positive culture results at least suggesting that biofilm might be of greater relevance in patients with nonpolypoid CRS. In summary, these studies suggest that that mucosal biofilm is a marker of more severe mucosal disease and a predictor of poorer outcome after sinus surgery. On the basis of animal models of rhinosinusitis, biofilm formation might reflect the severity, chronicity, or both of sinus infection.39
Intracellular bacteria in patients with CRS
Intracellular intraepithelial Staphylococcus aureus (IESA) was first reported in sinus epithelium by Corriveau et al40 using a peptide nucleic acid fluorescence in situ hybridization (FISH) assay. In this study IESA was found in the epithelium of patients with CRS but was also seen in some HCs, raising the question of its pathologic significance. In a recent study using a similar S aureus FISH probe with propidium iodide counterstain and CSLM, IESA was detected in 56% of patients with CRS undergoing sinus surgery but none of 8 HCs.41 Simultaneous analysis revealed the presence of S aureus biofilm in 100% of the IESA-positive tissues and 50% of the tissue from IESA-negative patients with CRS. The CSLM-FISH/propidium iodide technique has the advantage of being able to detect IESA and S aureus biofilm in the same tissue sample.41, 42
Presently, it is unclear whether the presence of IESA has any prognostic or pathologic significance beyond that afforded by the presence of biofilm, and there are no studies linking the presence of IESA to any specific defects in epithelial innate immunity.
Bacteriology of patients with CRS using molecular techniques
The evolving view of bacterial involvement in patients with CRS has expanded beyond that of infection with individual pathogens to considerations of increased bacterial burden, biofilm formation and alterations in the microbial community (ie, the microbiome).
The microbiome is the universe of culturable and nonculturable microorganisms present in a specific ecologic niche, such as the gastrointestinal tract or the sinus mucosa. Analysis of the microbiome has gained much recent attention as a novel means of studying host-microbial relations in various organs, including the respiratory tract and sinuses. This has been made possible by the advent of broad-platform molecular techniques, including microarrays and sequencing methods, allowing for identification of the full microbiome based on microbial RNA. The most commonly used technique is pyrosequencing to identify bacterial species based on the conserved 16S eubacterial 16S ribosomal gene.43 Techniques to study the microbial community are rapidly emerging but include methods to quantify bacterial diversity, representation of specific bacterial species, and bacterial “load” or “burden.”
Stephenson et al20 used pyrosequencing with modified 16S eubacterial primers to amplify the 600-bp region of 16S rRNA genes. Eighteen patients with CRS (83% with NPs) were compared with 9 control subjects. Conventional cultures identified bacterial growth in 82% of patients with CRS (the percentage positive in control subjects was not mentioned), with an average of 1.4 isolates per sample. In contrast, the results of pyrosequencing were positive in 100%, with a mean of 10 and a range of 1 to 20 isolates per sample. The most prevalent organisms in patients with CRS were anaerobic bacteria, Diaphorobacter and Peptoniphilus species, whereas coagulase-negative Staphylococcus species were identified in 50% of samples. S aureus and Corynebacterium and Propionibacterium species were also identified in control subjects.
Feazel et al44 obtained middle meatus swabs and compared bacterial culture and pyrosequencing results in 15 patients with CRS (including 2 with NPs) versus 5 HCs. Standard bacteriologic cultures were positive for all 15 patients with CRS and 5 HCs, with an average of 2.8 isolates reported per subject (range, 1-5 isolates). No statistically significant differences were found between the number of isolates obtained from patients with CRS and control subjects. The most commonly cultured organisms were coagulase-negative staphylococci (75%), S aureus (50%), and Propionibacterium acnes (30%). By using pyrosequencing, the most prevalent DNA sequence types detected were coagulase-negative staphylococci (100%), Corynebacterium species (85.7%), P acnes (76.2%), and S aureus (66.7%). In general, cultured organisms comprised less than 15% of the organisms identified by means of sequencing, but bacteria identified by using culture were identified by means of pyrosequencing in most cases. Some of the nonspecific CRS culture results, such as “mixed gram-negative rods,” were associated with pyrosequencing detection of moderate mixed anaerobes representing 75% of the total sequences identified. This confirms a long-held contention of some investigators that studies reporting a low prevalence of anaerobes in CRS samples might have had methodologic flaws related to sample collection, bacterial culture, or both.
In the study by Feazel et al,44 S aureus was selectively enriched in patients with CRS by means of culture (10/16 patients with CRS vs 0/5 HCs). DNA sequences representative of S aureus were detected in a similar percentage of patients with CRS (11/16 [69%]) and HCs (3/5 [60%]), but a greater abundance (bacterial burden) of S aureus sequences was found in patients with CRS. In this study both antibiotic exposure and asthma were associated with reduced microbial diversity and increased S aureus abundance.
Role of fungal colonization and fungal biofilm in patients with CRS
Using a special technique of “mucus preservation” (sample collection on saline-moistened nonstick gauze to prevent adsorption of mucus into gauze and prompt transport of samples to the laboratory for staining), Ponikau et al45 reported that 93% of patients with CRS had detectable fungal hyphae in mucus obtained from diseased sinuses. Virtually all patients with CRS and HCs also had a positive fungal culture of nasal secretions, with a broad array of fungi isolated. The latter finding was confirmed by an independent group of investigators who found a positive fungal stain or culture in 87% of patients with CRS and 91.3% of HCs.46 Gosepath et al47 analyzed tissue specimens from patients with CRS using a highly sensitive PCR assay and detected fungal DNA in all 27 patients with CRS with both a universal fungal primer and a primer specific for Alternaria species. In contrast, although the universal primer detected fungal DNA in 10 of 15 HCs, none of these subjects had positive results with the Alternaria species–specific primer.
Fungal biofilm has also been described in patients with CRS. Healy et al48 used specific bacterial FISH DNA probes and a panfungal probe to detect biofilm in mucosal samples from 11 patients with EMCRS and AFRS or CRS. Three mucosal samples from control subjects were also collected. Samples were also analyzed by using epifluorescent microscopy. Bacterial biofilm was demonstrated in 9 of 11 samples and 2 of 3 control specimens, and fungal elements were detected in association with bacterial biofilm in the majority of cases.
Foreman et al49 analyzed 50 patients with CRS and 10 control subjects for the simultaneous presence of bacterial and fungal biofilm by using bacteria-specific FISH probes and a universal probe for fungus. They identified bacterial biofilm in 72% and fungal biofilm in 22% of patients with CRS. In this study there were too few patients to compare the prevalence of fungal biofilm in patients with EMCRS, AFRS, and CRS.
Barrier function in sinus epithelium and its relevance to CRS
Epithelial barrier function is important for maintaining mucosal hydration and preventing penetration of foreign particles, including microbes, into the subepithelial layer. Defective epithelial barrier function has been found to be a key risk factor for development of atopic dermatitis, leading to increased transepidermal water loss and possibly contributing directly to increased susceptibility to S aureus skin infection.50, 51
Tight junction proteins are essential to normal epithelial barrier function. They are composed of a branching network of sealing strands, with each strand acting independently. The key tight junction proteins are the claudins and the occludins. In the intestine tight junction proteins efficiently restrict most microbes from penetrating into the subepithelial tissue.52 Acute reversible changes in barrier function have been associated with intestinal viral or bacterial infections, including rotavirus and enteropathogenic E coli.53 Whether primary defects in epithelial barrier function increase susceptibility to inflammatory bowel disease (IBD) is not presently established.53
No primary defects in epithelial tight junction proteins have been described in patients with CRS or NPs. Soyka at al54 found that NPs have decreased transtissue resistance and an irregular, patchy decreased expression of the tight junction proteins occludin and zonula occludens 1. These effects are likely secondary to tissue inflammation because it was shown that in vitro culture of HCs or NP epithelial cells in the presence of IL-4 resulted in downregulation of occludin and zonula occludens 1 expression, whereas culturing with IFN-γ caused an upregulation of these proteins.
Unlike tight junctions, less is known of the role of adherens junction proteins and desmosomal proteins in epithelial barrier function. Decreased expression of the epithelial adherens junction protein E-cadherin was described in NPs and linked to other manifestations of epithelial-mesenchymal transition.55 Decreased expression of the desmosomal junction proteins desmoglein 2 and desmoglein 3 was reported.56 In the latter study culture of human bronchial epithelial cells with TNF-α or IL-13 downregulated desmoglein 2 expression, suggesting that the defect in desmoglein expression in NPs might be secondary to the underlying inflammatory process.56
Mucociliary transport and its relevance to CRS
Mucociliary clearance is an essential process in normal sinus function. Impairments in mucociliary clearance lead to mucostasis, bacterial colonization, biofilm formation, and CRS. Genetically engineered mice that lack normal cilia spontaneously have bacterial sinusitis.57, 58
The classic example of abnormal ciliary function is primary ciliary dyskinesia (PCD; also known as immotile cilia syndrome). PCD is a rare, usually autosomal recessive disorder characterized by sinopulmonary disease, laterality defects (eg, situs inversus), and male infertility. PCD has traditionally been diagnosed based on ultrastructural defects in cilia, which are identified in about 90% of patients with PCD and involve the outer dynein arms, inner dynein arms, or both.59 Mutational analyses have discovered that 38% of patients with PCD carry mutations of the dynein genes DNAI1 and DNAH5.59 Because it is estimated that normal ciliary function involves more than 1000 gene products, additional mutations accounting for the remaining 62% of patients with PCD are likely to be uncovered. Recently, screening of 146 unrelated PCD families identified patients in 6 families with reduced outer dynein arms carrying mutations in CCDC103.60 The CCDC103 gene product might function as a tightly bound, axoneme-associated protein. A somewhat similar clinical syndrome known as Young syndrome is characterized by obstructive azoospermia with normal sperm structure and function and recurrent sinopulmonary infections.61 Unlike patients with PCD, no mutations in ciliary structural genes have been identified in patients with Young syndrome, and the problem has been presumed to be caused by an abnormality in mucus.62
Several studies have established that CRS is associated with an acquired reduction in mucociliary clearance.63, 64, 65, 66, 67 Mucociliary clearance typically normalizes after clearance of infection and restoration of normal sinus drainage.63, 65, 66 This has been confirmed by in vitro studies showing that sinonasal mucosal explants from patients with CRS show blunted ciliary beat frequency responses to cholinergic and adrenergic stimulation but normal responses within 36 hours of culturing.68 Lai et al69 reported increased expression of the centrosomal protein 110 (Cp110) in inflamed sinus tissue from patients with CRS. Cp110 is known to prevent the terminal step in ciliary maturation. They further showed that Cp110 could be induced in vitro by proinflammatory cytokines in cultured sinus epithelial cells with concomitant inhibition of ciliogenesis. In contrast, there is no evidence for a primary defect in mucociliary clearance to account for CRS. To date, genetic association studies of patients with refractory CRS have not uncovered any gene polymorphisms that would suggest a primary defect in mucociliary clearance.70
Host defects in the innate immune response associated with CRS
Key antimicrobial proteins and peptides in host innate immunity
Composition of sinus mucosal secretions
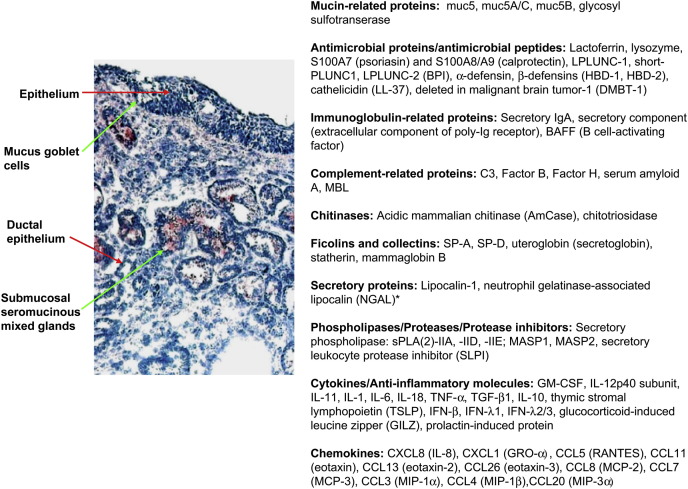
Fig 1 13, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84 illustrates the general structure of the sinus epithelium, submucosal mixed seromucinous glands, and stroma and summarizes the many proteins and peptides produced by sinus mucosal cells.
Fig 1.
Structure of the sinus epithelium, submucosal mixed seromucinous glands, and stroma and summary of proteins and peptides produced by sinus mucosal cells of potential relevance to innate or adaptive immune responses. Seromucinous glands are stained positively for CXCL1 (GRO-α). Adapted from references 13 and 71 to 83. *The NGAL protein was observed in the epithelium, infiltrating inflammatory cells, and submucosal gland of NPs but was rarely detected in normal inferior turbinate tissue.84 MBL-associated serine protease (MASP) 1 and 2 and MBL are soluble proteins that can arise in sinus secretions from plasma.
Submucosal seromucinous mixed glands comprise a series of ducts with interconnecting serous and mucous tubules that terminate in acini.85 Alcian blue can be used to stain acidic mucoglycoproteins within epithelial goblet cells and submucosal glands, whereas serous glands are not stained.86 Serous glands are reported to be the major site of production of antimicrobial peptides, although many of the antimicrobial peptides illustrated are produced by both surface epithelial cells and submucosal glands. Protease-activated receptor 2 has been demonstrated on submucosal glands, and a heightened glandular secretory response to protease-activated receptor 2 agonists, including house dust mite extract, has been described in patients with CRS.87
Lysozyme
Lysozyme catalyzes the breakdown of bacterial cell wall peptidoglycan by cleaving β[1-4]glycosidic linkages between N-acetylmuramic acid and N-acetylglucosamine (http://www.ncbi.nlm.nih.gov/gene/4069). Lysozyme localizes primarily to serous cells of submucosal mixed glands and neutrophils, with weak staining of the mucus cells of submucosal mixed glands and goblet cells.71, 88 Decreased immunostaining for lysozyme was reported in longstanding cases of CRS89; however, more recent studies reported increased rather than decreased immunostaining in patients with CRS.71, 88, 90 In the study by Woods et al,90 increased immunoreactivity for lysozyme was found in mucosal biopsy specimens of both patients with CRSsNP and those with CRSwNP, including low-level immunostaining in the epithelium.
Lactoferrin
Similar to lysozyme, lactoferrin is detected primarily in serous cells of submucosal glands.88 Although Zhang et al88 reported an increase in lactoferrin immunostaining in patients with CRS, Psaltis et al91 reported a decrease in lactoferrin at both the mRNA and protein levels in patients with CRS and an even further decrease in expression in the presence of bacterial biofilm.92
Secretory leukocyte proteinase inhibitor
Secretory leukocyte proteinase inhibitor (SLPI) protects epithelial tissues from serine proteases, including trypsin, leukocyte elastase, and cathepsin G (http://www.ncbi.nlm.nih.gov/gene/6590). It is a cationic protein with a very high affinity for leukocyte elastase.93 It also has antibacterial and other immunomodulatory properties and is the third most abundant antimicrobial protein in the upper airways.94 The antibacterial properties of SLPI are thought to reside in the N-terminal domain of the protein, where its activity could be mediated by its cationic charge.94 SLPI has antibacterial activity against P aeruginosa, S aureus, Staphylococcus epidermidis, and Candida albicans.94
The role of SLPI in innate immunity and control of inflammation is complex. Furthermore, it has been shown that the decreased levels of SLPI found in Pseudomonas species–infected lungs of patients with cystic fibrosis is due to degradation by neutrophil elastase.95 SLPI has been recovered from purulent maxillary sinus secretions in patients with maxillary sinusitis,96 but there have been no studies linking abnormalities of SLPI to CRS.
Antimicrobial peptides defensins, cathelicidins, DMBT-1, and S-100 proteins
Defensins and cathelicidins are the main families of antimicrobial peptides present in airway secretions and expressed by the airway epithelium.97 They represent small (3-5 kDa) cationic peptides that are either produced constitutively or induced by microbial products (including TLR ligands), cytokines, or growth factors.
The α-defensins are produced mainly by neutrophils and intestinal Paneth cells, whereas hBD-1, hBD-2, hBD-3 and hBD-4 are primarily produced by epithelial cells.97 In epithelial cells hBD-1 production is constitutive, whereas hBD-2, hBD-3, and hBD-4 production is inducible. Negligible expression of hBD-2 and hBD-3 was found in normal nasal mucosa.98 Reduced expression of hBD-2 was found in epithelial cells isolated from NPs.3
In human subjects only 1 cathelicidin (hCAP-18) is expressed. In neutrophils the cathelicidin proform hCAP-18 is proteolytically processed by proteinase 3, resulting in the release of LL-37.99 In skin serine proteinases control cleavage of hCAP-18 to LL-37.100 LL-37 was detected in epithelial cells, submucosal glands, and inflammatory cells in nasal tissue explants and induced by culture with fungal allergens from Aspergillus and Alternaria species.101, 102 No clear pattern of deficiency in the production of LL-37 in sinus tissue has been demonstrated in patients with CRS.102
Bactericidal/permeability-increasing protein and Plunc family proteins
A family of proteins, including bactericidal/permeability-increasing protein (BPI) and LPS-binding protein (LBP), has antimicrobial effects against gram-negative bacteria. Both bind to bacterial LPS, causing growth arrest and inhibition of LPS-induced activation of inflammatory responses.103, 104 Although BPI and LBP are found in airway secretions, there has been little study of their levels in nasal or sinus secretions.
Plunc proteins, members of the palate lung and nasal epithelium clone (PLUNC) family, are structurally related to LBP and BPI. Recent studies suggest physiologically relevant concentrations of PLUNC inhibit P aeruginosa biofilm formation in vitro without acting directly as a bactericide.105, 106 This finding suggests that PLUNC protein might have relevance to patients with refractory CRS.
SPLUNC1 and LPLUNC2 were found to be differentially expressed in serous and mucous cells, respectively, of submucosal glands in uncinate tissue. Decreased SPLUNC1 and LPLUNC2 levels were found in NPs relative to those seen in healthy control uncinate tissues, likely reflecting the decreased number of submucosal glands in NPs because their levels correlated with decreased lactoferrin levels.107 Similarly, Wu et al108 found PLUNC protein levels to be reduced in NPs and somewhat increased in patients with CRS relative to HCs, which is consistent with other studies showing an increased number of submucosal glands in patients with CRSsNP relative to HCs.109
DMBT1 (gp-340)
The deleted in malignant brain tumor 1 (DMBT1) gene encodes alternatively spliced proteins referred to as gp-340 (DMBT1gp340) and salivary agglutinin (DMBT1SAG). The former is secreted in bronchoalveolar lining fluid, whereas the latter is present in saliva, but the 2 molecules are identical. DMBT1 interacts with and agglutinates several gram-negative and gram-positive bacteria, including Streptococcus mutans, a bacterium responsible for dental caries. DMBT1 interacts with surfactant protein A (SP-A) and surfactant protein D (SP-D) and a variety of other host proteins involved in innate immunity or wound healing. A precise role for DMBT1 in human disease has yet to be elucidated. DMBT-1 is overexpressed in NPs mainly in submucosal glands.110
Ficolins and collectins (SP-A, SP-D, conglutinin, and mannose-binding lectin)
Ficolins and collectins (“collagenous lectins”) are soluble innate pattern recognition receptors that have an innate function resembling that of antibodies in adaptive immunity.111 Subunits of ficolins and collectins recognize carbohydrate arrays of their microbial targets through globular trimeric carbohydrate-recognition domains. SP-A, ficolins, and mannose-binding lectin (MBL) also share structural similarity with C1q, and ficolins and MBL (but not SP-A) activate complement. Whether collectins interact with immune cells through receptors is unclear.111
There are no studies of ficolin in the upper airway. SP-A expression was reported to be increased in sinus mucosal biopsy specimens in patients with CRSsNP112 and also present in NPs.113 However, Woodworth et al72 found comparable expression of SP- A and SP-D in HCs and patients with CRSwNP. In these studies SP-A and SP-D immunoreactivity was found in epithelial cells and submucosal seromucinous glands. One other study reported reduced immunostaining for SP-D in nasal submucosal glands and a lack of induction of SP-D by Aspergillus and Alternaria species allergens in cultured nasal explants from patients with nonallergic fungal eosinophilic sinusitis.114 In contrast, these allergens induced SP-D production in HCs and patients with CRS. Ramanathan et al3 showed that culturing human sinonasal epithelial cells in the presence of the TH2 cytokines IL-4 or IL-13 for 36 hours reduced expression of antimicrobial innate immune genes by using real-time PCR, ELISA, and flow cytometry, including TLR9, hBD-2, and SP-A. In these experiments IL-4 and IL-13 reduced expression of these innate factors in both HCs and patients with CRS. However, given that mucosal SP-A expression is increased in patients with CRSwNP, the significance of in vitro suppression of SP-A by IL-4/IL-13 is unclear.
MBL, also referred to as mannose-binding protein, is a calcium-dependent serum protein that binds carbohydrate determinants on the surfaces of a wide range of pathogens (viruses, bacteria, fungi, and protozoa), thereby activating the complement cascade or acting directly as an opsonin.115 MBL is a member of the collectin family of proteins and has structural similarity to complement component C1. MBL deficiency is associated with an increased incidence of upper respiratory tract infections in children, presumably because their adaptive immune systems are still immature. However, a study of MBL deficiency (defined by expression of an MBL deficiency allele) in 9245 Danish adults found no significant differences in infectious disease prevalence or mortality in MBL-deficient subjects versus control subjects.116 However, there has been a paucity of studies of MBL levels or genotype in a population of patients with CRS. One study found increased serum levels of both C3 and MBL in patients with CRSsNP and those with CRSwNP.117
Complement components
The complement component C3 and serum amyloid A protein are produced by sinonasal epithelial cells.73 Serum amyloid A functions as an acute-phase opsonin against gram-negative bacteria, including E coli and P aeruginosa, through an outer membrane protein A family member.118 No differences were found in expression of C3 or serum amyloid A in patients with CRS compared with that seen in HCs.73 Increased expression of factor B, C3, and C5 mRNA was found in tissues from patients with AFRS and those with CRSwNP compared with that seen in HCs,119 but the functional significance of this is unclear.
NO
NO is constitutively produced at high levels in sinus epithelium by virtue of high constitutive levels of inducible nitric oxide synthase (or NOS2A).120 The NO concentration in a healthy maxillary sinus (9.1 ± 3.8 ppm) exceeds that necessary for antibacterial effects in vitro 121 and is vastly higher than that produced in the nose or lungs (normal exhaled NO value, <50 ppb). The antimicrobial effects of NO in the sinuses might also relate to stimulation of increased ciliary beat frequency and complex reactivities between NO radical superoxide, metals, and thiols.122 NO production is also downstream of innate signaling through bitter taste receptors (see below).
Recent experiments have demonstrated an NO-responsive quorum-sensing mechanism in Vibrio harveyi 123 and Shewanella oneidensis 124 that regulates biofilm formation. In these bacteria NO stimulates biofilm formation by controlling levels of the bacterial cyclic diguanosine monophosphate.124 Whether a similar mechanism exists in other pathogens remains to be explored, but it has been reported that low NO levels (0.9-2.0 μmol/L) stimulate biofilm formation in S aureus.125
Innate signaling mechanisms through epithelial cells
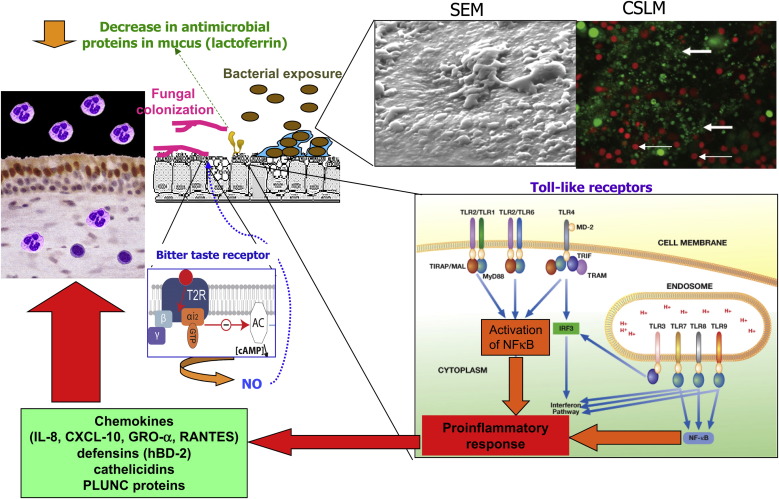
Multiple innate pattern recognition receptor pathways are either known or potentially relevant to sinus epithelium, including transmembrane TLRs and intracellular nucleotide-binding oligomerization domain (NOD) receptors,126, 127 dectin receptors, and bitter taste receptors. Table E2 in this article's Online Repository at www.jacionline.org summarizes the pattern recognition receptors involved in microbial recognition by airway epithelial cells, their microbial ligands, and abnormalities described in patients with CRS. Fig 2 18, 29, 128 summarizes some of the salient features of host-microbial interactions involved in triggering innate immune responses in patients with CRS.
Fig 2.
Salient features of host-microbial interactions involved in triggering innate immune responses in patients with CRS. TLR signaling pathways induce proinflammatory cytokine and chemokine production. Bitter taste receptor is activated by a quorum-sensing molecule from P aeruginosa and stimulates production of NO, which then stimulates mucocociliary clearance and has direct antimicrobial effects. Depiction of the intracellular TLR signaling pathways was adapted from the IAVI Report (http://www.iavireport.org/Back-Issues/Pages/IAVI-Report-9(4)-TollBridgetoImmunity.aspx). Upper right inset (left panel), SEM of bacterial biofilm showing characteristic glycocalyx and water channels. The photograph was used with permission from Sanclement et al.29Right panel, CSLM image (×63 magnification) of a patient with CRS stained with the BacLight LIVE/DEAD kit (Invitrogen, Molecular Probes, Carlsbad, Calif) demonstrating a bacterial biofilm comprised of many intensely fluorescing live bacteria organized in clusters (large arrow). Small arrows designate the larger live and dead epithelial cells. Used with permission from Psaltis et al.18 The bitter taste receptor depicted in the small middle inset was adapted from Fenech C, Patrikainen L, Kerr DS, Grall S, Liu Z, Laugerette F, et al. Ric-8A, a Gα protein guanine nucleotide exchange factor potentiates taste receptor signaling. Front Cell Neurosci 2009;3:11.128
Microbial recognition through TLR receptors
Sinonasal epithelial cells express TLRs 1 through 10.74, 129 TLR2, TLR3, TLR4, and TLR9 signaling has been demonstrated in sinonasal epithelial cells.13, 130 TLR ligation in airway epithelial cells results in activation of specific intracellular signaling pathways (reviewed by Bals and Hiemstra131), leading to production of (1) innate antimicrobial peptides and (2) cytokines and chemokines that amplify innate responses (eg, neutrophil infiltration) and activate adaptive immune responses. The TLR2 receptor has the greatest diversity of ligands and recognizes a wide array of gram-positive and gram-negative bacteria, as well as fungi, in part because of formation of heterodimers with TLR1 or TLR6.132 The TLR4 pathway is extremely important in host responses to gram-negative bacterial infection in the airway, and polymorphisms in TLR4 have been associated with more gram-negative bacterial infections in patients in an intensive care unit.133 The response of human tracheobronchial epithelium to LPS requires the TLR4 “coreceptor” CD14.134
Studies of TLR signaling pathways have focused on patients with “refractory” CRS unresponsive to medical management or those with “recalcitrant” NPs. Increased expression of TLR2 was found in patients with recalcitrant CRS.135 An exaggerated response to TLR3 plus cigarette smoke extract was found in cultured epithelial cells from patients with CRSsNP, with excess production of RANTES and hBD-2.13 Reduced baseline expression of TLR9 was found in cultured epithelial cells from patients with refractory NPs,130 and this might be linked to the effects of locally produced TH2 cytokines.3
Microbial recognition through dectin receptors
Dectin-1 is a type II transmembrane protein with a C-type lectin–like carbohydrate recognition domain, a transmembrane region, and a cytoplasmic tail containing an immunoreceptor tyrosinase activation motif.136 Dectin-1 binds specifically to β-1,3 glucans and induces intracellular signaling. Dectin-1 and TLR2/TLR6 signaling combine to enhance the responses triggered by each receptor.137, 138 Dectin-1 deficiency has been associated with mucocutaneous candidiasis.139 Although dectin receptors are regarded as receptors on myeloid cells, Sun et al140 recently showed that dectin-1 receptors could be induced on airway epithelial cells by Alternaria species in a TLR2-dependent manner with induction of TNF-α, GM-CSF, IL-8, hBD-2, and hBD-9. Although fungal colonization and mucosal responses to fungi have received much attention, dectin-1 has not yet been studied in patients with CRS.
Microbial recognition through NOD-like receptors
NOD2 is a member of the NOD-like receptor protein family that initiate inflammatory responses when exposed to ligands derived from bacterial components intracellularly.141, 142 Polymorphisms in NOD2 (CARD15) lead to impaired NOD2 function and increased susceptibility to Crohn disease (CD), a condition marked by excessive inflammatory responses to normal bacterial flora. The ligand for NOD2 is muramyl dipeptide, the “minimal bioactive peptidoglycan motif common to all bacteria.”143 NOD2 ordinarily downregulates responses to TLR stimulation,141 and it has been suggested that NOD2 polymorphisms in patients with CD result in a decrease in negative regulation of TLR responses and a pathologic increase in responses to normal gut flora. Alternatively, it has been suggested that impaired activation of the NOD2 pathway might facilitate invasion of the intestinal epithelial cells through commensal or pathogenic bacteria.142
NOD2 defects in patients with IBD are expressed widely in bodily tissues, raising the question as to whether there is any association between IBD and CRS. One study found a prevalence of chronic “sinonasal disease” of 48% in patients with IBD, with a higher prevalence in patients with CD (53%) versus those with ulcerative colitis (37%) and an even higher prevalence of chronic sinonasal disease in patients with CD with obstructive bowel complications (68% vs 27%), with 23% of these patients reporting CRS.144 Another study that examined this association found a similar prevalence of CRS in patients with IBD as in the general population, although the proportion of patients with CRS having nasal polyposis was higher among patients with IBD.145 There are no studies linking genetic polymorphisms in NOD1, NOD2, or NOD pathway genes with CRS.
Microbial recognition through bitter taste receptors
Lee et al146 recently discovered that one of the bitter taste receptors, T2R38, is activated by a quorum-sensing molecule from P aeruginosa associated with biofilm formation. Bitter taste receptors are a family of G protein–coupled receptors that signal by inducing a transient intracellular calcium flux and stimulating ciliary beat frequency. Activation of the receptor induces production of NO and increases ciliary beat frequency in sinus epithelial cells. A common polymorphism (TAS2R38 variant) was discovered that is associated with reduced signaling, NO production, and ciliary beat frequency and increased growth of P aeruginosa in air-liquid cultures of human airway epithelial cells. The effect of TAS2R38 on the killing of P aeruginosa was shown to be NO dependent. The TAS2R38 genotype correlated with the presence of sinonasal gram-negative infection in patients with CRS, suggesting a mechanistic link between a deficiency in innate signaling and increased bacterial infection.
Adaptive immune antimicrobial signaling mechanisms: IL-17A and IL-22 signaling pathways
IL-17A and IL-22 are “signature” cytokines of TH17 cells involved in host defense against extracellular pathogens, including fungi, bacteria, and some parasites.139 IL-22 is considered an “essential guardian of mucosal immunity against extracellular bacteria in the lung and gut.”147 In intestinal epithelial cells IL-22 stimulates the production of a wide variety of antibacterial proteins, stimulates mucin 1 production under inflammatory conditions, and enhances epithelial regeneration with goblet cell restitution.148 IL-22 deficiency in mice is associated with a more severe form of gram-negative enteric infection with Citrobacter rodentium, with increased intestinal epithelial damage, systemic bacterial burden, and mortality. In this model IL-22 mediates production of Reg family antimicrobial proteins, including RegIIIb and RegIIIc, by colonic epithelial cells. Both T cells, including CD4+ cells and innate lymphoid cells, and dendritic cells produce IL-22.149, 150, 151 A subset of CD4+ helper T cells that produce IL-22 but not IL-17 has been identified in human subjects and termed TH22 cells.151
IL-22 also protects the lungs from gram-negative Klebsiella pneumonia infection.152 In conjunction with IL-17A or IL-17F, IL-22 synergistically induces expression of hBD-2 and S100A9 and additively enhances the expression of S100A7 and S100A8.153 IL-22–mediated effects on airway epithelial cells require signaling through the IL-22 receptor (IL-22R) and activation of signal transducer and activator of transcription 3.154 Il-23, produced mainly by phagocytes and dendritic cells,155 is a crucial upstream regulator of IL-22 and IL-17 production.152, 156 Although IL-22 and IL-17A act synergistically or additively to promote the expression of genes involved in mucosal defense, TH17 and TH22 cells likely serve different roles in mucosal immunity and autoimmunity.147, 154
IL-17A and IL-22 also have an established role in antifungal immunity at mucosal surfaces,157 which would suggest that defects in the signaling pathways of these cytokines in the sinus mucosa might be associated with difficulty handling fungal infection. However, this has not been studied.
To date, no definite defect in IL-17A or IL-22 signaling has been identified in patients with CRS. Using a genome-wide association study, Endam et al158 identified a polymorphism in IL-22R in association with refractory CRS, but no functional studies were performed to explore this relationship. Reduced IL-22R1 expression in sinus mucosal epithelial cells was found in patients with recalcitrant NPs, and a relationship was found between reduced expression of IL-22R1 and a higher rate of recurrence of NPs after sinus surgery.159
Role of bacteria or fungi in maladaptive TH2 responses in patients with CRS
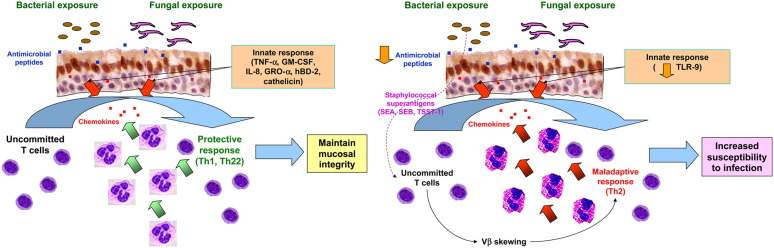
It has long been recognized that CRS is a disease in which the local tissue inflammatory response might be strongly biased toward TH2 inflammation despite a lack of systemic evidence for allergic disease. This is particularly true in patients with CRSwNP160 but also true, to a lesser degree, in patients with CRSsNP.161, 162, 163, 164 There is evidence that links colonizing microorganisms to this maladaptive TH2 “local allergic” response in patients with CRSwNP (schematized in Fig 3 ). This has been shown in cultured airway epithelial cells and dispersed T lymphocytes from NPs.
Fig 3.
Normal host response to microbial infection (left panel) versus maladaptive TH2 response (right panel). The maladaptive TH2 response has been best demonstrated in patients with NPs and pertains to patients with CRSwNP.
Fungi are commonly detected in the attached mucus of sinus tissues in patients with CRS47, 165 and can induce eosinophil activation and degranulation.166 Certain fungi, particularly Alternaria and Candida species, were shown to induce production of IL-5 and IL-13, as well as IFN-γ, in peripheral blood lymphocytes from patients with CRS. It was hypothesized that this maladaptive response would account for an eosinophilic mixed TH1/TH2 mucosal immune response.165 Fungal allergens also elicit modest production of IL-5 and IL-13 from dispersed NP T lymphocytes.167 However, a recent study did not demonstrate consistent IL-5 production in response to Alternaria species in patients with CRS from Utah.168 A small randomized placebo-controlled trial of intranasal amphotericin B reported improvement in radiographic mucosal thickening, nasal endoscopy, and eosinophilic inflammation.169 However, 2 other randomized placebo-controlled trials with intranasal antifungal treatment did not demonstrate a significant clinical benefit.170, 171 In addition, antifungal treatment did not alter the cytokine or chemokine production profiles of nasal inflammatory cells.172
Mucosal colonization with S aureus has been found in 64% of patients with CRSwNP compared with roughly 30% of healthy subjects or patients with CRSsNP.173 In a study of 13 patients with massive polyposis, 55% of patients were found to have enterotoxin-producing S aureus in the nasal mucus adjacent to polyps.174 It was further shown that T lymphocytes isolated from the polyps showed a skewing of Vβ use with enrichment for Vβ known to respond to staphylococcal superantigens.174 This finding was later confirmed by another group.175, 176 IgE antibodies directed against staphylococcal superantigens were found in NP homogenates in 27.8% of patients with NPs and 53.8% of patients with NPs with coexisting asthma.173 Finally, staphylococcal enterotoxin B (SEB) was found to induce robust production of IL-5 and IL-13 in dispersed NP T lymphocytes.167 These studies suggest that colonizing S aureus might be a major driver of the local TH2 inflammatory response in patients with CRSwNP.
Staphylococcal superantigens, such as SEB, have been shown to impair oral tolerance and promote allergy in a murine model of food allergy.177 SEB has also been shown to augment allergic inflammation in a murine model of eosinophilic CRS, leading to formation of nasal polyposis.55
Downregulation of epithelial innate immunity by maladaptive TH2 tissue inflammation
Given the strong adaptive TH2-type chronic inflammation characteristic of patients with CRSwNP, investigations were undertaken to ascertain whether TH2-type inflammation modulated innate immune function. Ramanathan et al130 first demonstrated that cultured primary nasal epithelial cells (PNECs) from HCs and patients with CRSwNP express surface TLR9 and respond to CpG (a TLR9 agonist) by increasing production of hBD-2 and IL-8. They found that TLR9 expression on PNECs from patients with CRSwNP was reduced by 50% compared to control PNECs. Culturing control PNECs in the presence of the TH1 cytokine IFN-γ increased TLR9 expression by 49%, whereas culturing in the presence of the TH2 cytokines IL-4 or IL-13 decreased TLR9 expression by 46.6%. Because this group previously reported reduced epithelial expression of IL-22R1 in patients with recalcitrant NPs, it would be interesting to know whether TH2 cytokines would induce this effect.
Is there an inappropriate or heightened response to colonizing “commensal” organisms in patients with CRS?
Epithelial surfaces have adapted specialized mechanisms for coping with potential intruders. These not only protect the host from infection but also provide beneficial effects in terms of digestion (in the gut) and immune maturation (gut and respiratory tract). Mechanisms have also evolved to cope with “commensals” in the gut and presumably also in the oral mucosa and nasal epithelium, where bacterial colonization is normal. It has been suggested that CRS might have a proinflammatory response to common colonizing organisms, such as coagulase-negative Staphylococcus species.178 Could it be that the “proinflammatory” state of CRS relates to a lack of tolerance to seemingly innocuous “commensal” organisms? We do not know, but this represents an exciting area for future investigation.
Summary/conclusions
There is limited information indicating that patients with CRS have an increased incidence of rhinovirus infection and some suggestion that they might have an exaggerated response to viral infection, but there is no evidence for persistent viral infection in the majority of cases. It would be instructive to map the time course and innate immune response to experimental rhinovirus infection and ascertain whether patients with CRS have increased susceptibility, increased viral burden, or an inappropriate innate immune response.
A role for bacterial infection or colonization in CRS pathogenesis is well established. Bacteria are cultured and bacterial biofilm is present in the majority of patients undergoing surgical intervention. Diseased sinus tissue also has an increased bacterial burden, most convincingly demonstrated for S aureus, and bacterial biofilm with other organisms, including P aeruginosa, is often present. The majority of patients with CRS also have fungal hyphae detectable in mucus extracted from diseased sinuses, and fungal biofilm is detectable in some cases, usually in association with bacterial biofilm. The presence of bacterial biofilm, particularly polymicrobial biofilm or biofilm containing S aureus, has prognostic value and is associated with more severe sinus disease preoperatively and worse symptom and nasal endoscopy scores after surgery.
Mucociliary clearance is essential for normal sinus function. Active disease is associated with a reduction in mucociliary clearance but normalization of mucociliary clearance after clearance of infection and restoration of normal sinus. There is no evidence for a primary defect in mucociliary clearance to account for CRS, except in the distinct clinical syndrome of PCD.
A defect in local host innate immunity has long been suggested as a pathogenic mechanism of CRS to account for CRS, especially because systemic immune function is normal in the vast majority of cases. Decreased levels of the antimicrobial proteins, most notably lactoferrin, have been found most often in sinus secretions, whereas levels of other antimicrobial proteins and peptides have been reported to be normal.
Multiple innate pattern recognition receptor pathways, notably TLRs and intracellular NOD receptors and bitter taste receptors, are critical in host/microbial interactions in the sinuses. No primary defects have been found in these pathways to account for refractory CRS, although a 50% reduction in the expression of TLR9 has been found in patients with refractory NPs.
A downregulation of epithelial innate immunity by maladaptive TH2 tissue inflammation has been demonstrated in patients with recalcitrant CRSwNP. In vitro studies have shown that IL-4 and IL-13 decrease TLR9 expression on cultured epithelial cells by nearly 50% and also reduce expression of hBD-2 and SP-A. Colonizing fungi might play a role in maladaptive TH2 responses and thereby promote tissue eosinophilia. Colonization with enterotoxin-producing S aureus is a driver of the local TH2 response in patients with CRSwNP because it induces a local expansion of T lymphocytes and production of enterotoxin-specific IgE antibodies.
To date, an effective means of restoring host-microbial balance and mitigating disease in patients with CRS remains elusive. There are few studies on the effectiveness of medical treatment alone at eradicating bacterial infection and bacterial biofilm in patients with CRS. Clinical trials of antifungal rinses have been generally unsuccessful. Surgical removal of diseased sinus tissue with restoration of sinus ventilation and use of culture-directed antibiotics remain the best approaches to treatment. There is hope that further elucidation of the genetic underpinnings of CRS and the host-microbial interactions present will provide greater insight into disease pathogenesis and more effective treatment.
Footnotes
Disclosure of potential conflict of interest: D. L. Hamilos has consultant arrangements with and has received grants from Merck and received royalties from UpToDate.
Role of atypical bacterial infection in patients with CRS
One study using sensitive molecular techniques (PCR) found no evidence for Mycoplasma pneumoniae, Chlamydia pneumoniae, or Legionella pneumophila in mucosal samples from 11 patients with CRS.E1 Recent studies used pyrosequencing to potentially overcome issues of detection of nonculturable bacteria in mucosal samples from patients with CRS; did not detect Mycoplasma, Ureaplasma, and Chlamydia species; and found Legionella, Mycobacterium, and Nocardia species in only rare cases.E2, E3
Tichenor et alE4 identified 8 patients with CRS with refractory symptoms who had positive cultures for atypical mycobacteria over a 10-year period. Mycobacterium avium molecular fingerprints clonally related to the patients' isolates were also identified in the plumbing of 3 of 8 households sampled, suggesting that at least in these 3 cases, indoor plumbing and sinus irrigations might have been responsible for the infection.
Solyar et alE5 reported on a series of 37 patients with refractory CRS who had a positive mycobacterial culture over a 7-year period. The most frequent isolates were Mycobacterium abscessus (57.1%), Mycobacterium avium-intracellulare complex (14.3%), and Mycobacterium chelonae (14.3%). It was reported that 5.3% of outpatient-acquired and 2.6% of inpatient-acquired cultures were positive for atypical mycobacteria. Some evidence of improvement in CRS was noted after treatment with antimycobacterial antibiotics. These studies suggest that atypical mycobacterial infection is rare in patients with CRS but should be sought in patients with refractory CRS.
Techniques for bacterial biofilm detection in patients with CRS
See Table E2 for a summary of studies of bacterial or fungal biofilm in patients with CRS.
SEM and TEM have the advantage of high magnification and are the only techniques that provide ultrastructural confirmation of biofilm presence. Technical artifacts can arise because of sample dehydration and surface distortion. Disadvantages of SEM include small sample size, potentially introducing sampling artifacts. CSLM has the advantage that specimens can be imaged without fixation or dehydration, and specific bacteria or fungi can be stained with fluorescent markers. The FISH assay uses either universal bacterial probes, such as EUB338,E6 or species-specific primers based on unique sequences in the 16S ribosomal RNA gene. The LIVE/DEAD BacLight Bacterial Viability Kit (Invitrogen, Carlsbad, Calif) uses 2 nucleic acid stains: green-fluorescent SYTO 9 stain, which labels both live and dead bacteria, and red-fluorescent propidium iodide, which penetrates only bacteria or cells with damaged membranes. CSLM is used to confirm the presence of biofilm on the mucosal surface. No bacteria-specific probes are used.
A direct comparison of techniques in a sheep model of rhinosinusitis concluded that CSLM is more objective for documenting biofilm presence than SEM or TEM because of the inherent flaws, sampling error, and subjectivity involved in SEM and TEM.E7 Another study determined that the BacLight method was more specific than CSLM/FISH for biofilm detection in sinus tissues.E8, E9 Hochstim et alE6 reported that bacterial biofilm can be detected by using routine hematoxylin-eosin staining, with good agreement with FISH/CSLM as the control standard. However, this technique is suspect because of its reliance on identification of small basophilic bacterial clusters on the epithelial surface. A concern is whether any of the techniques other than SEM or TEM are sufficiently rigorous to define biofilm because they do not provide ultrastructural confirmation of the presence of biofilm.
Biofilm-forming capacity in vitro
The Calgary Biofilm Assay is an in vitro test used to assess the ability of a bacterial isolate to form biofilm. The bacteria are cultured on a 96-well plate with round pegs positioned over the plate. A semiquantitative analysis using uptake of crystal violet in the biofilm has been developed.E10 The LIVE/DEAD BacLight Bacterial Viability Kit (see above) can also be used to assess biofilm formation in vitro on 98-well chamber slides.E11 Prince et alE12 used the Calgary Biofilm Detection Assay to examine in 159 patients with CRS with mucopurulence and found that 28.6% of patients had biofilm-forming capacity. Polymicrobial cultures, P aeruginosa, and/or S aureus comprised 71% of samples. Postsurgical cases had a higher prevalence of biofilm-forming capacity than surgery-naive patients (30.7% vs 15%).
Table E1.
Summary of studies of bacterial or fungal biofilm in patients with CRS
| Study | Method of biofilm detection | Specific bacterial species identified | Patients with CRS (no. positive/total studied) | Healthy subjects (no. positive/total studied) |
|---|---|---|---|---|
| Cryer et al, 2004E13 | SEM | No specific probes used | 4/16 | — |
| Ferguson and Stolz, 2005E14 | TEM | Pseudomonas aeruginosa | 2/4 | — |
| Sanclement et al, 2005E15 | SEM/TEM | No specific probes used | 24/30 | 0/4 |
| Sanderson et al, 2006E16 | FISH/CSLM | Streptococcus pneumoniae, Staphylococcus aureus, Haemophilus influenzae, and Pseudomonas aeruginosa | 14/18 | 2/5 |
| Psaltis et al, 2007E17 | CSLM | 17/38 | 0/9 | |
| Foreman et al, 2009E18 | FISH/CSLM |
Staphylococcus aureus Haemophilus influenzae Pseudomonas aeruginosa Universal fungal probe |
36/50 (11/50 positive for fungi) | 0/10 |
| Hekiert et al, 2009E19 | SEM | No specific probes used | 17/60 | — |
| Galli et al, 2008E20 | SEM | Haemophilus influenzae | 10/24 | 0/20 |
| Singhal et al, 2010E21 | FISH/CSLM | No specific probes used | 36/51 | — |
| Zernotti et al, 2010E22 | CSLM (in vivo) | No specific probes used | 2/12 patients with CRS/NP | 0/10 |
| Foreman et al, 2010E9 | LIVE/DEAD BacLight Bacterial Viability Kit using CSLM compared with FISH/CSLM | No specific probes used | 15/20 Positive for biofilm with each technique but 18/20 positive by ≥1 technique | |
| Hochstim et al, 2010E6 | Hematoxylin and eosin staining plus FISH/CSLM with bacteria-specific probe EUB338 | No specific probes used Universal bacteria-specific probe EUB338∗ | 15/24 | 1/10 |
EUB338 is a “universal” bacterial probe used in FISH.
Table E2.
Pattern recognition receptors involved in microbial recognition by airway epithelial cells, their microbial ligands, and abnormalities described in patients with CRS†, ∗
| Receptor | Ligand | Relevant pathogens | Key innate signaling events in airway epithelial cells | Abnormalities described in patients with CRS |
|---|---|---|---|---|
| TLR2 | Peptidoglycan, lipoteichoic acid, and lipoprotein from gram-positive bacteria, lipoarabinomannan from mycobacteria, and zymosan from yeast cell wall | Gram-positive and gram-negative bacteria; fungi (Candida species and Aspergillus fumigatus) | Increased production of hBD-2 and IL-8 | Increased expression of TLR2 in patients with recalcitrant CRSE23 |
| TLR3 | Viral double-stranded RNA (dsRNA); polyinosine-polycytidylic acid (poly[I:C]) is a synthetic analog of dsRNA. | Rhinovirus, other viruses | Increased type I and type III interferon levels; chemokines (IL-8, GRO-α, RANTES, CXCL10); hBD-2, hBD-3 | Exaggerated response to TLR3 plus cigarette smoke extract with excess production of RANTES and hBD-2E24 |
| TLR4 (including CD14 and MD2 on cell surface) | LPS (facilitated by LPS-binding protein)E25 | Gram-negative bacteria; Candida species and Aspergillus fumigatusE26 | NF-κB and activation of proinflammatory cytokine genes, including IL-8 and hBD-2 | Reduced level of expression of TLR4, TLR7, and MyD88 in patients with CRSsNP compared with control subjectsE27 |
| TLR7/8 | Single-stranded RNAs (natural ligands); small synthetic molecules: imidazoquinolines and nucleoside analogs | Viruses | NF-κB and activation of proinflammatory cytokine genes | Reduced level of expression of TLR4, TLR7, and MyD88 in patients with CRSsNP compared with control subjectsE27 |
| TLR9 | Specific unmethylated CpG oligonucleotide (ODN) sequences (CpG DNA)‡ | Bacteria | Production of IL-8E28 | Decreased baseline expression of TLR9 in patients with CRSwNP; decreased TLR9 in cultured AECs in response to IL-4 and IL-13E29 |
| Dectin receptors | β-Glucans from fungi | Aspergillus fumigatus | Production of TNF-α, GM-CSF, IL-8, hBD-2, and hBD-9 | None |
| Bitter taste receptors | Functional responses to pathogen-derived quorum-sensing molecules | Pseudomonas aeruginosa | NO production, stimulation of mucociliary clearance and direct antibacterial effects | TAS2R38 genotype correlated with Pseudomonas aeruginosa infection in patients with CRSE30 |
| NOD1 (intracellular receptor) | iE-DAP D-Lactyl-l-Ala-g-Glu-meso-DAP-Gly Heptanolyl-g-Glu-meso-DAP-Ala GM-tripeptide |
Recognition of bacterial/viral PAMPs | NF-κB and activation of proinflammatory cytokine genes | None |
| NOD2 (intracellular receptor) | Muramyl dipeptide (MDP), a component of peptidoglycan MurNAc-I-Ala-g-d-Glu-I-Lys |
Recognition of bacterial/viral PAMPs | NF-κB and activation of proinflammatory cytokine genes | None |
TLR ligand information source: http://www.invivogen.com/tlr2-ligands.
AEC, Airway epithelial cells; MyD88, myeloid differentiation pathway response gene (88); NF-κB, nuclear factor κB; PAMP, peptidoglycan recognition protein.
TLR1, TLR5, TLR6, and TLR10 are not included because there are no reports of abnormalities in these TLRs associated with CRS.
Bacterial DNA containing unmethylated CpG dinucleotides.
References
- 1.Meltzer E.O., Hamilos D.L., Hadley J.A., Lanza D.C., Marple B.F., Nicklas R.A. Rhinosinusitis: establishing definitions for clinical research and patient care. J Allergy Clin Immunol. 2004;114(Suppl):155–212. doi: 10.1016/j.jaci.2004.09.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Desrosiers M. Refractory chronic rhinosinusitis: pathophysiology and management of chronic rhinosinusitis persisting after endoscopic sinus surgery. Curr Allergy Asthma Rep. 2004;4:200–207. doi: 10.1007/s11882-004-0027-z. [DOI] [PubMed] [Google Scholar]
- 3.Ramanathan M., Jr., Lee W.K., Spannhake E.W., Lane A.P. Th2 cytokines associated with chronic rhinosinusitis with polyps down-regulate the antimicrobial immune function of human sinonasal epithelial cells. Am J Rhinol. 2008;22:115–121. doi: 10.2500/ajr.2008.22.3136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mosser A.G., Vrtis R., Burchell L., Lee W.M., Dick C.R., Weisshaar E. Quantitative and qualitative analysis of rhinovirus infection in bronchial tissues. Am J Respir Crit Care Med. 2005;171:645–651. doi: 10.1164/rccm.200407-970OC. [DOI] [PubMed] [Google Scholar]
- 5.Mallia P., Message S.D., Gielen V., Contoli M., Gray K., Kebadze T. Experimental rhinovirus infection as a human model of chronic obstructive pulmonary disease exacerbation. Am J Respir Crit Care Med. 2011;183:734–742. doi: 10.1164/rccm.201006-0833OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Contoli M., Message S.D., Laza-Stanca V., Edwards M.R., Wark P.A., Bartlett N.W. Role of deficient type III interferon-lambda production in asthma exacerbations. Nat Med. 2006;12:1023–1026. doi: 10.1038/nm1462. [DOI] [PubMed] [Google Scholar]
- 7.Rank M.A., Wollan P., Kita H., Yawn B.P. Acute exacerbations of chronic rhinosinusitis occur in a distinct seasonal pattern. J Allergy Clin Immunol. 2010;126:168–169. doi: 10.1016/j.jaci.2010.03.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sanders S.P., Proud D., Permutt S., Siekierski E.S., Yachechko R., Liu M.C. Role of nasal nitric oxide in the resolution of experimental rhinovirus infection. J Allergy Clin Immunol. 2004;113:697–702. doi: 10.1016/j.jaci.2004.01.755. [DOI] [PubMed] [Google Scholar]
- 9.Wiehler S., Proud D. Interleukin-17A modulates human airway epithelial responses to human rhinovirus infection. Am J Physiol Lung Cell Mol Physiol. 2007;293:L505–L515. doi: 10.1152/ajplung.00066.2007. [DOI] [PubMed] [Google Scholar]
- 10.Zaheer R.S., Koetzler R., Holden N.S., Wiehler S., Proud D. Selective transcriptional down-regulation of human rhinovirus-induced production of CXCL10 from airway epithelial cells via the MEK1 pathway. J Immunol. 2009;182:4854–4864. doi: 10.4049/jimmunol.0802401. [DOI] [PubMed] [Google Scholar]
- 11.Jang Y.J., Kwon H.J., Park H.W., Lee B.J. Detection of rhinovirus in turbinate epithelial cells of chronic sinusitis. Am J Rhinol. 2006;20:634–636. doi: 10.2500/ajr.2006.20.2899. [DOI] [PubMed] [Google Scholar]
- 12.Wang J.H., Kwon H.J., Chung Y.S., Lee B.J., Jang Y.J. Infection rate and virus-induced cytokine secretion in experimental rhinovirus infection in mucosal organ culture: comparison between specimens from patients with chronic rhinosinusitis with nasal polyps and those from normal subjects. Arch Otolaryngol Head Neck Surg. 2008;134:424–427. doi: 10.1001/archotol.134.4.424. [DOI] [PubMed] [Google Scholar]
- 13.Yamin M., Holbrook E.H., Gray S.T., Harold R., Busaba N., Sridhar A. Cigarette smoke combined with Toll-like receptor 3 signaling triggers exaggerated epithelial regulated upon activation, normal T-cell expressed and secreted/CCL5 expression in chronic rhinosinusitis. J Allergy Clin Immunol. 2008;122:1145–1153. doi: 10.1016/j.jaci.2008.09.033. [DOI] [PubMed] [Google Scholar]
- 14.Wood A.J., Antoszewska H., Fraser J., Douglas R.G. Is chronic rhinosinusitis caused by persistent respiratory virus infection? Int Forum Allergy Rhinol. 2011;1:95–100. doi: 10.1002/alr.20030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jackson D.J., Lemanske R.F., Jr. The role of respiratory virus infections in childhood asthma inception. Immunol Allergy Clin North Am. 2010;30:513–522. doi: 10.1016/j.iac.2010.08.004. vi. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Brook I. Bacteriologic features of chronic sinusitis in children. JAMA. 1981;246:967–969. [PubMed] [Google Scholar]
- 17.Brook I., Yocum P., Shah K. Aerobic and anaerobic bacteriology of concurrent chronic otitis media with effusion and chronic sinusitis in children. Arch Otolaryngol Head Neck Surg. 2000;126:174–176. doi: 10.1001/archotol.126.2.174. [DOI] [PubMed] [Google Scholar]
- 18.Psaltis A.J., Ha K.R., Beule A.G., Tan L.W., Wormald P.J. Confocal scanning laser microscopy evidence of biofilms in patients with chronic rhinosinusitis. Laryngoscope. 2007;117:1302–1306. doi: 10.1097/MLG.0b013e31806009b0. [DOI] [PubMed] [Google Scholar]
- 19.Oncel S., Pinar E., Sener G., Calli C., Karagoz U. Evaluation of bacterial biofilms in chronic rhinosinusitis. J Otolaryngol Head Neck Surg. 2010;39:52–55. [PubMed] [Google Scholar]
- 20.Stephenson M.F., Mfuna L., Dowd S.E., Wolcott R.D., Barbeau J., Poisson M. Molecular characterization of the polymicrobial flora in chronic rhinosinusitis. J Otolaryngol Head Neck Surg. 2010;39:182–187. [PubMed] [Google Scholar]
- 21.Brook I., Frazier E.H. Correlation between microbiology and previous sinus surgery in patients with chronic maxillary sinusitis. Ann Otol Rhinol Laryngol. 2001;110:148–151. doi: 10.1177/000348940111000210. [DOI] [PubMed] [Google Scholar]
- 22.Finegold S.M., Flynn M.J., Rose F.V., Jousimies-Somer H., Jakielaszek C., McTeague M. Bacteriologic findings associated with chronic bacterial maxillary sinusitis in adults. Clin Infect Dis. 2002;35:428–433. doi: 10.1086/341899. [DOI] [PubMed] [Google Scholar]
- 23.Brook I., Yocum P. Immune response to Fusobacterium nucleatum and Prevotella intermedia in patients with chronic maxillary sinusitis. Ann Otol Rhinol Laryngol. 1999;108:293–295. doi: 10.1177/000348949910800313. [DOI] [PubMed] [Google Scholar]
- 24.Donlan R.M., Costerton J.W. Biofilms: survival mechanisms of clinically relevant microorganisms. Clin Microbiol Rev. 2002;15:167–193. doi: 10.1128/CMR.15.2.167-193.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chiang W.C., Pamp S.J., Nilsson M., Givskov M., Tolker-Nielsen T. The metabolically active subpopulation in Pseudomonas aeruginosa biofilms survives exposure to membrane-targeting antimicrobials via distinct molecular mechanisms. FEMS Immunol Med Microbiol. 2012;65:245–256. doi: 10.1111/j.1574-695X.2012.00929.x. [DOI] [PubMed] [Google Scholar]
- 26.Hoyle B.D., Costerton J.W. Bacterial resistance to antibiotics: the role of biofilms. Prog Drug Res. 1991;37:91–105. doi: 10.1007/978-3-0348-7139-6_2. [DOI] [PubMed] [Google Scholar]
- 27.Cryer J., Schipor I., Perloff J.R., Palmer J.N. Evidence of bacterial biofilms in human chronic sinusitis. ORL J Otorhinolaryngol Relat Spec. 2004;66:155–158. doi: 10.1159/000079994. [DOI] [PubMed] [Google Scholar]
- 28.Ferguson B.J., Stolz D.B. Demonstration of biofilm in human bacterial chronic rhinosinusitis. Am J Rhinol. 2005;19:452–457. [PubMed] [Google Scholar]
- 29.Sanclement J.A., Webster P., Thomas J., Ramadan H.H. Bacterial biofilms in surgical specimens of patients with chronic rhinosinusitis. Laryngoscope. 2005;115:578–582. doi: 10.1097/01.mlg.0000161346.30752.18. [DOI] [PubMed] [Google Scholar]
- 30.Singhal D., Psaltis A.J., Foreman A., Wormald P.J. The impact of biofilms on outcomes after endoscopic sinus surgery. Am J Rhinol Allergy. 2010;24:169–174. doi: 10.2500/ajra.2010.24.3462. [DOI] [PubMed] [Google Scholar]
- 31.Hochstim C.J., Masood R., Rice D.H. Biofilm and persistent inflammation in endoscopic sinus surgery. Otolaryngol Head Neck Surg. 2010;143:697–698. doi: 10.1016/j.otohns.2010.07.017. [DOI] [PubMed] [Google Scholar]
- 32.Perloff J.R., Palmer J.N. Evidence of bacterial biofilms on frontal recess stents in patients with chronic rhinosinusitis. Am J Rhinol. 2004;18:377–380. [PubMed] [Google Scholar]
- 33.Harvey R.J., Lund V.J. Biofilms and chronic rhinosinusitis: systematic review of evidence, current concepts and directions for research. Rhinology. 2007;45:3–13. [PubMed] [Google Scholar]
- 34.Zernotti M.E., Angel Villegas N., Roques Revol M., Baena-Cagnani C.E., Arce Miranda J.E., Paredes M.E. Evidence of bacterial biofilms in nasal polyposis. J Investig Allergol Clin Immunol. 2010;20:380–385. [PubMed] [Google Scholar]
- 35.Psaltis A.J., Weitzel E.K., Ha K.R., Wormald P.J. The effect of bacterial biofilms on post-sinus surgical outcomes. Am J Rhinol. 2008;22:1–6. doi: 10.2500/ajr.2008.22.3119. [DOI] [PubMed] [Google Scholar]
- 36.Foreman A., Wormald P.J. Different biofilms, different disease? A clinical outcomes study. Laryngoscope. 2010;120:1701–1706. doi: 10.1002/lary.21024. [DOI] [PubMed] [Google Scholar]
- 37.Prince A.A., Steiger J.D., Khalid A.N., Dogrhamji L., Reger C., Eau Claire S. Prevalence of biofilm-forming bacteria in chronic rhinosinusitis. Am J Rhinol. 2008;22:239–245. doi: 10.2500/ajr.2008.22.3180. [DOI] [PubMed] [Google Scholar]
- 38.Zhang Z., Kofonow J.M., Finkelman B.S., Doghramji L., Chiu A.G., Kennedy D.W. Clinical factors associated with bacterial biofilm formation in chronic rhinosinusitis. Otolaryngol Head Neck Surg. 2011;144:457–462. doi: 10.1177/0194599810394302. [DOI] [PubMed] [Google Scholar]
- 39.Ha K.R., Psaltis A.J., Tan L., Wormald P.J. A sheep model for the study of biofilms in rhinosinusitis. Am J Rhinol. 2007;21:339–345. doi: 10.2500/ajr.2007.21.3032. [DOI] [PubMed] [Google Scholar]
- 40.Corriveau M.N., Zhang N., Holtappels G., Van Roy N., Bachert C. Detection of Staphylococcus aureus in nasal tissue with peptide nucleic acid-fluorescence in situ hybridization. Am J Rhinol Allergy. 2009;23:461–465. doi: 10.2500/ajra.2009.23.3367. [DOI] [PubMed] [Google Scholar]
- 41.Tan N.C., Foreman A., Jardeleza C., Douglas R., Tran H., Wormald P.J. The multiplicity of Staphylococcus aureus in chronic rhinosinusitis: correlating surface biofilm and intracellular residence. Laryngoscope. 2012;122:1655–1660. doi: 10.1002/lary.23317. [DOI] [PubMed] [Google Scholar]
- 42.Tan N.C., Tran H.B., Foreman A., Jardeleza C., Vreugde S., Wormald P.J. Identifying intracellular Staphylococcus aureus in chronic rhinosinusitis: a direct comparison of techniques. Am J Rhinol Allergy. 2012;26:444–449. doi: 10.2500/ajra.2012.26.3822. [DOI] [PubMed] [Google Scholar]
- 43.Ronaghi M., Elahi E. Pyrosequencing for microbial typing. J Chromatogr B Analyt Technol Biomed Life Sci. 2002;782:67–72. doi: 10.1016/s1570-0232(02)00693-1. [DOI] [PubMed] [Google Scholar]
- 44.Feazel L.M., Robertson C.E., Ramakrishnan V.R., Frank D.N. Microbiome complexity and Staphylococcus aureus in chronic rhinosinusitis. Laryngoscope. 2012;122:467–472. doi: 10.1002/lary.22398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ponikau J.U., Sherris D.A., Kern E.B., Homburger H.A., Frigas E., Gaffey T.A. The diagnosis and incidence of allergic fungal sinusitis. Mayo Clin Proc. 1999;74:877–884. doi: 10.4065/74.9.877. [DOI] [PubMed] [Google Scholar]
- 46.Braun H., Stammberger H., Buzina W., Freudenschuss K., Lackner A., Beham A. [Incidence and detection of fungi and eosinophilic granulocytes in chronic rhinosinusitis] Laryngorhinootologie. 2003;82:330–340. doi: 10.1055/s-2003-39777. [DOI] [PubMed] [Google Scholar]
- 47.Gosepath J., Brieger J., Vlachtsis K., Mann W.J. Fungal DNA is present in tissue specimens of patients with chronic rhinosinusitis. Am J Rhinol. 2004;18:9–13. [PubMed] [Google Scholar]
- 48.Healy D.Y., Leid J.G., Sanderson A.R., Hunsaker D.H. Biofilms with fungi in chronic rhinosinusitis. Otolaryngol Head Neck Surg. 2008;138:641–647. doi: 10.1016/j.otohns.2008.02.002. [DOI] [PubMed] [Google Scholar]
- 49.Foreman A., Boase S., Psaltis A., Wormald P.J. Role of bacterial and fungal biofilms in chronic rhinosinusitis. Curr Allergy Asthma Rep. 2012;12:127–135. doi: 10.1007/s11882-012-0246-7. [DOI] [PubMed] [Google Scholar]
- 50.Schittek B. The antimicrobial skin barrier in patients with atopic dermatitis. Curr Probl Dermatol. 2011;41:54–67. doi: 10.1159/000323296. [DOI] [PubMed] [Google Scholar]
- 51.O'Regan G.M., Sandilands A., McLean W.H., Irvine A.D. Filaggrin in atopic dermatitis. J Allergy Clin Immunol. 2009;124(Suppl 2):R2–R6. doi: 10.1016/j.jaci.2009.07.013. [DOI] [PubMed] [Google Scholar]
- 52.Guttman J.A., Finlay B.B. Tight junctions as targets of infectious agents. Biochim Biophys Acta. 2009;1788:832–841. doi: 10.1016/j.bbamem.2008.10.028. [DOI] [PubMed] [Google Scholar]
- 53.Marchiando A.M., Graham W.V., Turner J.R. Epithelial barriers in homeostasis and disease. Annu Rev Pathol. 2010;5:119–144. doi: 10.1146/annurev.pathol.4.110807.092135. [DOI] [PubMed] [Google Scholar]
- 54.Soyka M.B., Wawrzyniak P., Eiwegger T., Holzmann D., Treis A., Wanke K. Defective epithelial barrier in chronic rhinosinusitis: the regulation of tight junctions by IFN-gamma and IL-4. J Allergy Clin Immunol. 2012;130:1087–1096.e10. doi: 10.1016/j.jaci.2012.05.052. [DOI] [PubMed] [Google Scholar]
- 55.Shin H.W., Cho K., Kim D.W., Han D.H., Khalmuratova R., Kim S.W. Hypoxia-inducible factor 1 mediates nasal polypogenesis by inducing epithelial-to-mesenchymal transition. Am J Respir Crit Care Med. 2012;185:944–954. doi: 10.1164/rccm.201109-1706OC. [DOI] [PubMed] [Google Scholar]
- 56.Zuckerman J.D., Lee W.Y., DelGaudio J.M., Moore C.E., Nava P., Nusrat A. Pathophysiology of nasal polyposis: the role of desmosomal junctions. Am J Rhinol. 2008;22:589–597. doi: 10.2500/ajr.2008.22.3235. [DOI] [PubMed] [Google Scholar]
- 57.Blatt E.N., Yan X.H., Wuerffel M.K., Hamilos D.L., Brody S.L. Forkhead transcription factor HFH-4 expression is temporally related to ciliogenesis. Am J Respir Cell Mol Biol. 1999;21:168–176. doi: 10.1165/ajrcmb.21.2.3691. [DOI] [PubMed] [Google Scholar]
- 58.Ostrowski L.E., Yin W., Rogers T.D., Busalacchi K.B., Chua M., O'Neal W.K. Conditional deletion of dnaic1 in a murine model of primary ciliary dyskinesia causes chronic rhinosinusitis. Am J Respir Cell Mol Biol. 2010;43:55–63. doi: 10.1165/rcmb.2009-0118OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zariwala M.A., Knowles M.R., Omran H. Genetic defects in ciliary structure and function. Annu Rev Physiol. 2007;69:423–450. doi: 10.1146/annurev.physiol.69.040705.141301. [DOI] [PubMed] [Google Scholar]
- 60.Panizzi J.R., Becker-Heck A., Castleman V.H., Al-Mutairi D.A., Liu Y., Loges N.T. CCDC103 mutations cause primary ciliary dyskinesia by disrupting assembly of ciliary dynein arms. Nat Genet. 2012;44:714–719. doi: 10.1038/ng.2277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Handelsman D.J., Conway A.J., Boylan L.M., Turtle J.R. Young's syndrome. Obstructive azoospermia and chronic sinopulmonary infections. N Engl J Med. 1984;310:3–9. doi: 10.1056/NEJM198401053100102. [DOI] [PubMed] [Google Scholar]
- 62.de Iongh R., Ing A., Rutland J. Mucociliary function, ciliary ultrastructure, and ciliary orientation in Young's syndrome. Thorax. 1992;47:184–187. doi: 10.1136/thx.47.3.184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Elwany S., Hisham M., Gamaee R. The effect of endoscopic sinus surgery on mucociliary clearance in patients with chronic sinusitis. Eur Arch Otorhinolaryngol. 1998;255:511–514. doi: 10.1007/s004050050109. [DOI] [PubMed] [Google Scholar]
- 64.Passali D., Ferri R., Becchini G., Passali G.C., Bellussi L. Alterations of nasal mucociliary transport in patients with hypertrophy of the inferior turbinates, deviations of the nasal septum and chronic sinusitis. Eur Arch Otorhinolaryngol. 1999;256:335–337. doi: 10.1007/s004050050158. [DOI] [PubMed] [Google Scholar]
- 65.Dal T., Onerci M., Caglar M. Mucociliary function of the maxillary sinuses after restoring ventilation: a radioisotopic study of the maxillary sinus. Eur Arch Otorhinolaryngol. 1997;254:205–207. doi: 10.1007/BF00879275. [DOI] [PubMed] [Google Scholar]
- 66.Kaluskar S.K. Pre- and postoperative mucociliary clearance in functional endoscopic sinus surgery. Ear Nose Throat J. 1997;76:884–886. [PubMed] [Google Scholar]
- 67.Shone G.R., Yardley M.P., Knight L.C. Mucociliary function in the early weeks after nasal surgery. Rhinology. 1990;28:265–268. [PubMed] [Google Scholar]
- 68.Chen B., Antunes M.B., Claire S.E., Palmer J.N., Chiu A.G., Kennedy D.W. Reversal of chronic rhinosinusitis-associated sinonasal ciliary dysfunction. Am J Rhinol. 2007;21:346–353. doi: 10.2500/ajr.2007.21.3029. [DOI] [PubMed] [Google Scholar]
- 69.Lai Y., Chen B., Shi J., Palmer J.N., Kennedy D.W., Cohen N.A. Inflammation-mediated upregulation of centrosomal protein 110, a negative modulator of ciliogenesis, in patients with chronic rhinosinusitis. J Allergy Clin Immunol. 2011;128:1207–1215. doi: 10.1016/j.jaci.2011.09.001. [DOI] [PubMed] [Google Scholar]
- 70.Mfuna-Endam L., Zhang Y., Desrosiers M.Y. Genetics of rhinosinusitis. Curr Allergy Asthma Rep. 2011;11:236–246. doi: 10.1007/s11882-011-0189-4. [DOI] [PubMed] [Google Scholar]
- 71.Fukami M., Stierna P., Veress B., Carlsoo B. Lysozyme and lactoferrin in human maxillary sinus mucosa during chronic sinusitis. An immunohistochemical study. Eur Arch Otorhinolaryngol. 1993;250:133–139. doi: 10.1007/BF00171697. [DOI] [PubMed] [Google Scholar]
- 72.Woodworth B.A., Lathers D., Neal J.G., Skinner M., Richardson M., Young M.R. Immunolocalization of surfactant protein A and D in sinonasal mucosa. Am J Rhinol. 2006;20:461–465. doi: 10.2500/ajr.2006.20.2892. [DOI] [PubMed] [Google Scholar]
- 73.Lane A.P., Truong-Tran Q.A., Myers A., Bickel C., Schleimer R.P. Serum amyloid A, properdin, complement 3, and toll-like receptors are expressed locally in human sinonasal tissue. Am J Rhinol. 2006;20:117–123. [PMC free article] [PubMed] [Google Scholar]
- 74.Vandermeer J., Sha Q., Lane A.P., Schleimer R.P. Innate immunity of the sinonasal cavity: expression of messenger RNA for complement cascade components and toll-like receptors. Arch Otolaryngol Head Neck Surg. 2004;130:1374–1380. doi: 10.1001/archotol.130.12.1374. [DOI] [PubMed] [Google Scholar]
- 75.Cole A.M., Dewan P., Ganz T. Innate antimicrobial activity of nasal secretions. Infect Immun. 1999;67:3267–3275. doi: 10.1128/iai.67.7.3267-3275.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Casado B., Pannell L.K., Iadarola P., Baraniuk J.N. Identification of human nasal mucous proteins using proteomics. Proteomics. 2005;5:2949–2959. doi: 10.1002/pmic.200401172. [DOI] [PubMed] [Google Scholar]
- 77.Schleimer R.P., Kato A., Kern R., Kuperman D., Avila P.C. Epithelium: at the interface of innate and adaptive immune responses. J Allergy Clin Immunol. 2007;120:1279–1284. doi: 10.1016/j.jaci.2007.08.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Kato A., Peters A., Suh L., Carter R., Harris K.E., Chandra R. Evidence of a role for B cell-activating factor of the TNF family in the pathogenesis of chronic rhinosinusitis with nasal polyps. J Allergy Clin Immunol. 2008;121:1385–1392. doi: 10.1016/j.jaci.2008.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Richer S.L., Truong-Tran A.Q., Conley D.B., Carter R., Vermylen D., Grammer L.C. Epithelial genes in chronic rhinosinusitis with and without nasal polyps. Am J Rhinol. 2008;22:228–234. doi: 10.2500/ajr.2008.22.3162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Park S.K., Cho H.W., Heo K.W., Hur D.Y., Lee H.K. Role of acidic mammalian chitinase and chitotriosidase in nasal polyps. Otolaryngol Head Neck Surg. 2009;141:462–466. doi: 10.1016/j.otohns.2009.06.013. [DOI] [PubMed] [Google Scholar]
- 81.Zhang X.H., Lu X., Long X.B., You X.J., Gao Q.X., Cui Y.H. Chronic rhinosinusitis with and without nasal polyps is associated with decreased expression of glucocorticoid-induced leucine zipper. Clin Exp Allergy. 2009;39:647–654. doi: 10.1111/j.1365-2222.2008.03198.x. [DOI] [PubMed] [Google Scholar]
- 82.Tieu D.D., Peters A.T., Carter R.G., Suh L., Conley D.B., Chandra R. Evidence for diminished levels of epithelial psoriasin and calprotectin in chronic rhinosinusitis. J Allergy Clin Immunol. 2010;125:667–675. doi: 10.1016/j.jaci.2009.11.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Lu X., Wang N., Long X.B., You X.J., Cui Y.H., Liu Z. The cytokine-driven regulation of secretoglobins in normal human upper airway and their expression, particularly that of uteroglobin-related protein 1, in chronic rhinosinusitis. Respir Res. 2011;12:28. doi: 10.1186/1465-9921-12-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Woo H.J., Min J.K., Bai C.H., Song S.Y., Kang H.J., Lee H.M. Expression of neutrophil gelatinase-associated lipocalin in nasal polyps. Arch Otolaryngol Head Neck Surg. 2008;134:1182–1186. doi: 10.1001/archotol.134.11.1182. [DOI] [PubMed] [Google Scholar]
- 85.Wu X., Peters-Hall J.R., Ghimbovschi S., Mimms R., Rose M.C., Pena M.T. Glandular gene expression of sinus mucosa in chronic rhinosinusitis with and without cystic fibrosis. Am J Respir Cell Mol Biol. 2011;45:525–533. doi: 10.1165/rcmb.2010-0133OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Malekzadeh S., Hamburger M.D., Whelan P.J., Biedlingmaier J.F., Baraniuk J.N. Density of middle turbinate subepithelial mucous glands in patients with chronic rhinosinusitis. Otolaryngol Head Neck Surgery. 2002;127:190–195. doi: 10.1067/mhn.2002.126800. [DOI] [PubMed] [Google Scholar]
- 87.Cho H.J., Lee H.J., Kim S.C., Kim K., Kim Y.S., Kim C.H. Protease-activated receptor 2-dependent fluid secretion from airway submucosal glands by house dust mite extract. J Allergy Clin Immunol. 2012;129:529–535. doi: 10.1016/j.jaci.2011.11.024. [DOI] [PubMed] [Google Scholar]
- 88.Zhang L., Han D., Wang Z. [Lysozyme and lactoferrin in human uncinate process mucosa during chronic sinusitis] Zhonghua Er Bi Yan Hou Ke Za Zhi. 1998;33:219–221. [PubMed] [Google Scholar]
- 89.Ishida M., Matsunaga T., Uda H. An immunohistological study of nasal and paranasal mucosa of patients with relapsing chronic sinusitis. Rhinology. 1984;22:115–118. [PubMed] [Google Scholar]
- 90.Woods C.M., Lee V.S., Hussey D.J., Irandoust S., Ooi E.H., Tan L.W. Lysozyme expression is increased in the sinus mucosa of patients with chronic rhinosinusitis. Rhinology. 2012;50:147–156. doi: 10.4193/Rhino11.229. [DOI] [PubMed] [Google Scholar]
- 91.Psaltis A.J., Bruhn M.A., Ooi E.H., Tan L.W., Wormald P.J. Nasal mucosa expression of lactoferrin in patients with chronic rhinosinusitis. Laryngoscope. 2007;117:2030–2035. doi: 10.1097/MLG.0b013e31812e01ab. [DOI] [PubMed] [Google Scholar]
- 92.Psaltis A.J., Wormald P.J., Ha K.R., Tan L.W. Reduced levels of lactoferrin in biofilm-associated chronic rhinosinusitis. Laryngoscope. 2008;118:895–901. doi: 10.1097/MLG.0b013e31816381d4. [DOI] [PubMed] [Google Scholar]
- 93.Thompson R.C., Ohlsson K. Isolation, properties, and complete amino acid sequence of human secretory leukocyte protease inhibitor, a potent inhibitor of leukocyte elastase. Proc Natl Acad Sci U S A. 1986;83:6692–6696. doi: 10.1073/pnas.83.18.6692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Weldon S., Taggart C.C. Innate host defense functions of secretory leucoprotease inhibitor. Exp Lung Res. 2007;33:485–491. doi: 10.1080/01902140701756547. [DOI] [PubMed] [Google Scholar]
- 95.Weldon S., McNally P., McElvaney N.G., Elborn J.S., McAuley D.F., Wartelle J. Decreased levels of secretory leucoprotease inhibitor in the Pseudomonas-infected cystic fibrosis lung are due to neutrophil elastase degradation. J Immunol. 2009;183:8148–8156. doi: 10.4049/jimmunol.0901716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Fryksmark U., Jannert M., Ohlsson K., Tegner H. Antileukoprotease in patients with maxillary sinusitis. Rhinology. 1985;23:247–251. [PubMed] [Google Scholar]
- 97.Hiemstra P.S. The role of epithelial beta-defensins and cathelicidins in host defense of the lung. Exp Lung Res. 2007;33:537–542. doi: 10.1080/01902140701756687. [DOI] [PubMed] [Google Scholar]
- 98.Claeys S., de Belder T., Holtappels G., Gevaert P., Verhasselt B., van Cauwenberge P. Human beta-defensins and toll-like receptors in the upper airway. Allergy. 2003;58:748–753. doi: 10.1034/j.1398-9995.2003.00180.x. [DOI] [PubMed] [Google Scholar]
- 99.Sorensen O.E., Follin P., Johnsen A.H., Calafat J., Tjabringa G.S., Hiemstra P.S. Human cathelicidin, hCAP-18, is processed to the antimicrobial peptide LL-37 by extracellular cleavage with proteinase 3. Blood. 2001;97:3951–3959. doi: 10.1182/blood.v97.12.3951. [DOI] [PubMed] [Google Scholar]
- 100.Yamasaki K., Schauber J., Coda A., Lin H., Dorschner R.A., Schechter N.M. Kallikrein-mediated proteolysis regulates the antimicrobial effects of cathelicidins in skin. FASEB J. 2006;20:2068–2080. doi: 10.1096/fj.06-6075com. [DOI] [PubMed] [Google Scholar]
- 101.Ooi E.H., Wormald P.J., Carney A.S., James C.L., Tan L.W. Fungal allergens induce cathelicidin LL-37 expression in chronic rhinosinusitis patients in a nasal explant model. Am J Rhinol. 2007;21:367–372. doi: 10.2500/ajr.2007.21.3025. [DOI] [PubMed] [Google Scholar]
- 102.Ooi E.H., Wormald P.J., Carney A.S., James C.L., Tan L.W. Human cathelicidin antimicrobial peptide is up-regulated in the eosinophilic mucus subgroup of chronic rhinosinusitis patients. Am J Rhinol. 2007;21:395–401. doi: 10.2500/ajr.2007.21.3048. [DOI] [PubMed] [Google Scholar]
- 103.Hirata M., Zhong J., Wright S.C., Larrick J.W. Structure and functions of endotoxin-binding peptides derived from CAP18. Prog Clin Biol Res. 1995;392:317–326. [PubMed] [Google Scholar]
- 104.Elsbach P. The bactericidal/permeability-increasing protein (BPI) in antibacterial host defense. J Leukoc Biol. 1998;64:14–18. doi: 10.1002/jlb.64.1.14. [DOI] [PubMed] [Google Scholar]
- 105.Gakhar L., Bartlett J.A., Penterman J., Mizrachi D., Singh P.K., Mallampalli R.K. PLUNC is a novel airway surfactant protein with anti-biofilm activity. PLoS One. 2010;5:e9098. doi: 10.1371/journal.pone.0009098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Bartlett J.A., Gakhar L., Penterman J., Singh P.K., Mallampalli R.K., Porter E. PLUNC: a multifunctional surfactant of the airways. Biochem Soc Trans. 2011;39:1012–1016. doi: 10.1042/BST0391012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Seshadri S., Lin D.C., Rosati M., Carter R.G., Norton J.E., Suh L. Reduced expression of antimicrobial PLUNC proteins in nasal polyp tissues of patients with chronic rhinosinusitis. Allergy. 2012;67:920–928. doi: 10.1111/j.1398-9995.2012.02848.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Wu M.M., Sun H., Nan Q. Expression and clinical significance of PLUNC protein in nasal polyp and chronic sinusitis tissue. Ear Nose Throat J. 2012;91:282–285. [PubMed] [Google Scholar]
- 109.Malekzadeh S., McGuire J.F. The new histologic classification of chronic rhinosinusitis. Curr Allergy Asthma Rep. 2003;3:221–226. doi: 10.1007/s11882-003-0043-4. [DOI] [PubMed] [Google Scholar]
- 110.Liu Z., Kim J., Sypek J.P., Wang I.M., Horton H., Oppenheim F.G. Gene expression profiles in human nasal polyp tissues studied by means of DNA microarray. J Allergy Clin Immunol. 2004;114:783–790. doi: 10.1016/j.jaci.2004.04.052. [DOI] [PubMed] [Google Scholar]
- 111.Palaniyar N. Antibody equivalent molecules of the innate immune system: parallels between innate and adaptive immune proteins. Innate Immun. 2010;16:131–137. doi: 10.1177/1753425910370498. [DOI] [PubMed] [Google Scholar]
- 112.Lee H.M., Kang H.J., Woo J.S., Chae S.W., Lee S.H., Hwang S.J. Upregulation of surfactant protein A in chronic rhinosinusitis. Laryngoscope. 2006;116:328–330. doi: 10.1097/01.mlg.0000194223.22763.5f. [DOI] [PubMed] [Google Scholar]
- 113.Wang X., Zhao C., Liu M. [Expression and significance of surfactant A in nasal polyps of chronic rhinosinusitis] Lin Chuang Er Bi Yan Hou Tou Jing Wai Ke Za Zhi. 2010;24:652–654. [PubMed] [Google Scholar]
- 114.Ooi E.H., Wormald P.J., Carney A.S., James C.L., Tan L.W. Surfactant protein d expression in chronic rhinosinusitis patients and immune responses in vitro to Aspergillus and Alternaria in a nasal explant model. Laryngoscope. 2007;117:51–57. doi: 10.1097/01.mlg.0000243196.75418.6f. [DOI] [PubMed] [Google Scholar]
- 115.Koch A., Melbye M., Sorensen P., Homoe P., Madsen H.O., Molbak K. Acute respiratory tract infections and mannose-binding lectin insufficiency during early childhood. JAMA. 2001;285:1316–1321. doi: 10.1001/jama.285.10.1316. [DOI] [PubMed] [Google Scholar]
- 116.Dahl M., Tybjaerg-Hansen A., Schnohr P., Nordestgaard B.G. A population-based study of morbidity and mortality in mannose-binding lectin deficiency. J Exp Med. 2004;199:1391–1399. doi: 10.1084/jem.20040111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Cui Y.H., Zhang F., Xiong Z.G., You X.J., Gao Q.X., Liu Z. Increased serum complement component 3 and mannose-binding lectin levels in adult Chinese patients with chronic rhinosinusitis. Rhinology. 2009;47:187–191. [PubMed] [Google Scholar]
- 118.Shah C., Hari-Dass R., Raynes J.G. Serum amyloid A is an innate immune opsonin for Gram-negative bacteria. Blood. 2006;108:1751–1757. doi: 10.1182/blood-2005-11-011932. [DOI] [PubMed] [Google Scholar]
- 119.Schlosser R.J., Mulligan R.M., Casey S.E., Varela J.C., Harvey R.J., Atkinson C. Alterations in gene expression of complement components in chronic rhinosinusitis. Am J Rhinol Allergy. 2010;24:21–25. doi: 10.2500/ajra.2010.24.3399. [DOI] [PubMed] [Google Scholar]
- 120.Lundberg J.O., Farkas-Szallasi T., Weitzberg E., Rinder J., Lidholm J., Anggaard A. High nitric oxide production in human paranasal sinuses. Nat Med. 1995;1:370–373. doi: 10.1038/nm0495-370. [DOI] [PubMed] [Google Scholar]
- 121.Lundberg J.O. Nitric oxide and the paranasal sinuses. Anat Rec (Hoboken) 2008;291:1479–1484. doi: 10.1002/ar.20782. [DOI] [PubMed] [Google Scholar]
- 122.De Groote M.A., Fang F.C. NO inhibitions: antimicrobial properties of nitric oxide. Clin Infect Dis. 1995;21(Suppl 2):S162–S165. doi: 10.1093/clinids/21.supplement_2.s162. [DOI] [PubMed] [Google Scholar]
- 123.Henares B.M., Higgins K.E., Boon E.M. Discovery of a nitric oxide responsive quorum sensing circuit in Vibrio harveyi. ACS Chem Biol. 2012;7:1331–1336. doi: 10.1021/cb300215t. [DOI] [PubMed] [Google Scholar]
- 124.Plate L., Marletta M.A. Nitric oxide modulates bacterial biofilm formation through a multicomponent cyclic-di-GMP signaling network. Mol Cell. 2012;46:449–460. doi: 10.1016/j.molcel.2012.03.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Jardeleza C., Foreman A., Baker L., Paramasivan S., Field J., Tan L.W. The effects of nitric oxide on Staphylococcus aureus biofilm growth and its implications in chronic rhinosinusitis. Int Forum Allergy Rhinol. 2011;1:438–444. doi: 10.1002/alr.20083. [DOI] [PubMed] [Google Scholar]
- 126.Janeway C.A., Jr., Medzhitov R. Innate immune recognition. Annu Rev Immunol. 2002;20:197–216. doi: 10.1146/annurev.immunol.20.083001.084359. [DOI] [PubMed] [Google Scholar]
- 127.Medzhitov R. Recognition of microorganisms and activation of the immune response. Nature. 2007;449:819–826. doi: 10.1038/nature06246. [DOI] [PubMed] [Google Scholar]
- 128.Fenech C., Patrikainen L., Kerr D.S., Grall S., Liu Z., Laugerette F. Ric-8A, a Gα protein guanine nucleotide exchange factor potentiates taste receptor signaling. Front Cell Neurosci. 2009;3:11. doi: 10.3389/neuro.03.011.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Dong Z., Yang Z., Wang C. Expression of TLR2 and TLR4 messenger RNA in the epithelial cells of the nasal airway. Am J Rhinol. 2005;19:236–239. [PubMed] [Google Scholar]
- 130.Ramanathan M., Jr., Lee W.K., Dubin M.G., Lin S., Spannhake E.W., Lane A.P. Sinonasal epithelial cell expression of toll-like receptor 9 is decreased in chronic rhinosinusitis with polyps. Am J Rhinol. 2007;21:110–116. doi: 10.2500/ajr.2007.21.2997. [DOI] [PubMed] [Google Scholar]
- 131.Bals R., Hiemstra P.S. Innate immunity in the lung: how epithelial cells fight against respiratory pathogens. Eur Respir J. 2004;23:327–333. doi: 10.1183/09031936.03.00098803. [DOI] [PubMed] [Google Scholar]
- 132.Farhat K., Riekenberg S., Heine H., Debarry J., Lang R., Mages J. Heterodimerization of TLR2 with TLR1 or TLR6 expands the ligand spectrum but does not lead to differential signaling. J Leukoc Biol. 2008;83:692–701. doi: 10.1189/jlb.0807586. [DOI] [PubMed] [Google Scholar]
- 133.Agnese D.M., Calvano J.E., Hahm S.J., Coyle S.M., Corbett S.A., Calvano S.E. Human toll-like receptor 4 mutations but not CD14 polymorphisms are associated with an increased risk of gram-negative infections. J Infect Dis. 2002;186:1522–1525. doi: 10.1086/344893. [DOI] [PubMed] [Google Scholar]
- 134.Becker M.N., Diamond G., Verghese M.W., Randell S.H. CD14-dependent lipopolysaccharide-induced beta-defensin-2 expression in human tracheobronchial epithelium. J Biol Chem. 2000;275:29731–29736. doi: 10.1074/jbc.M000184200. [DOI] [PubMed] [Google Scholar]
- 135.Lane A.P., Truong-Tran Q.A., Schleimer R.P. Altered expression of genes associated with innate immunity and inflammation in recalcitrant rhinosinusitis with polyps. Am J Rhinol. 2006;20:138–144. [PMC free article] [PubMed] [Google Scholar]
- 136.Brown G.D., Herre J., Williams D.L., Willment J.A., Marshall A.S., Gordon S. Dectin-1 mediates the biological effects of beta-glucans. J Exp Med. 2003;197:1119–1124. doi: 10.1084/jem.20021890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Gantner B.N., Simmons R.M., Canavera S.J., Akira S., Underhill D.M. Collaborative induction of inflammatory responses by dectin-1 and Toll-like receptor 2. J Exp Med. 2003;197:1107–1117. doi: 10.1084/jem.20021787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Dennehy K.M., Brown G.D. The role of the beta-glucan receptor Dectin-1 in control of fungal infection. J Leukoc Biol. 2007;82:253–258. doi: 10.1189/jlb.1206753. [DOI] [PubMed] [Google Scholar]
- 139.Engelhardt K.R., Grimbacher B. Mendelian traits causing susceptibility to mucocutaneous fungal infections in human subjects. J Allergy Clin Immunol. 2012;129:294–307. doi: 10.1016/j.jaci.2011.12.966. [DOI] [PubMed] [Google Scholar]
- 140.Sun W.K., Lu X., Li X., Sun Q.Y., Su X., Song Y. Dectin-1 is inducible and plays a crucial role in Aspergillus-induced innate immune responses in human bronchial epithelial cells. Eur J Clin Microbiol Infect Dis. 2012;31:2755–2764. doi: 10.1007/s10096-012-1624-8. [DOI] [PubMed] [Google Scholar]
- 141.Strober W., Kitani A., Fuss I., Asano N., Watanabe T. The molecular basis of NOD2 susceptibility mutations in Crohn's disease. Mucosal Immunol. 2008;1(Suppl 1):S5–S9. doi: 10.1038/mi.2008.42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Walsh D., McCarthy J., O'Driscoll C., Melgar S. Pattern recognition receptors—molecular orchestrators of inflammation in inflammatory bowel disease. Cytokine Growth Factor Rev. 2012;24:91–104. doi: 10.1016/j.cytogfr.2012.09.003. [DOI] [PubMed] [Google Scholar]
- 143.Girardin S.E., Boneca I.G., Viala J., Chamaillard M., Labigne A., Thomas G. Nod2 is a general sensor of peptidoglycan through muramyl dipeptide (MDP) detection. J Biol Chem. 2003;278:8869–8872. doi: 10.1074/jbc.C200651200. [DOI] [PubMed] [Google Scholar]
- 144.Book D.T., Smith T.L., McNamar J.P., Saeian K., Binion D.G., Toohill R.J. Chronic sinonasal disease in patients with inflammatory bowel disease. Am J Rhinol. 2003;17:87–90. [PubMed] [Google Scholar]
- 145.Chandra R.K., Lin D., Tan B., Tudor R.S., Conley D.B., Peters A.T. Chronic rhinosinusitis in the setting of other chronic inflammatory diseases. Am J Otolaryngol. 2011;32:388–391. doi: 10.1016/j.amjoto.2010.07.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Lee R.J., Xiong G., Kofonow J.M., Chen B., Lysenko A., Jiang P. T2R38 taste receptor polymorphisms underlie susceptibility to upper respiratory infection. J Clin Invest. 2012;122:4145–4159. doi: 10.1172/JCI64240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Laurence A., O'Shea J.J., Watford W.T. Interleukin-22: a sheep in wolf's clothing. Nat Med. 2008;14:247–249. doi: 10.1038/nm0308-247. [DOI] [PubMed] [Google Scholar]
- 148.Mizoguchi A. Healing of intestinal inflammation by IL-22. Inflamm Bowel Dis. 2012;18:1777–1784. doi: 10.1002/ibd.22929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Zheng Y., Valdez P.A., Danilenko D.M., Hu Y., Sa S.M., Gong Q. Interleukin-22 mediates early host defense against attaching and effacing bacterial pathogens. Nat Med. 2008;14:282–289. doi: 10.1038/nm1720. [DOI] [PubMed] [Google Scholar]
- 150.Taube C., Tertilt C., Gyulveszi G., Dehzad N., Kreymborg K., Schneeweiss K. IL-22 is produced by innate lymphoid cells and limits inflammation in allergic airway disease. PLoS One. 2011;6:e21799. doi: 10.1371/journal.pone.0021799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Basu R., O'Quinn D.B., Silberger D.J., Schoeb T.R., Fouser L., Ouyang W. Th22 cells are an important source of IL-22 for host protection against enteropathogenic bacteria. Immunity. 2012;37:1061–1075. doi: 10.1016/j.immuni.2012.08.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Aujla S.J., Chan Y.R., Zheng M., Fei M., Askew D.J., Pociask D.A. IL-22 mediates mucosal host defense against Gram-negative bacterial pneumonia. Nat Med. 2008;14:275–281. doi: 10.1038/nm1710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Liang S.C., Tan X.Y., Luxenberg D.P., Karim R., Dunussi-Joannopoulos K., Collins M. Interleukin (IL)-22 and IL-17 are coexpressed by Th17 cells and cooperatively enhance expression of antimicrobial peptides. J Exp Med. 2006;203:2271–2279. doi: 10.1084/jem.20061308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Sonnenberg G.F., Fouser L.A., Artis D. Border patrol: regulation of immunity, inflammation and tissue homeostasis at barrier surfaces by IL-22. Nat Immunol. 2011;12:383–390. doi: 10.1038/ni.2025. [DOI] [PubMed] [Google Scholar]
- 155.Langrish C.L., McKenzie B.S., Wilson N.J., de Waal Malefyt R., Kastelein R.A., Cua D.J. IL-12 and IL-23: master regulators of innate and adaptive immunity. Immunol Rev. 2004;202:96–105. doi: 10.1111/j.0105-2896.2004.00214.x. [DOI] [PubMed] [Google Scholar]
- 156.Oppmann B., Lesley R., Blom B., Timans J.C., Xu Y., Hunte B. Novel p19 protein engages IL-12p40 to form a cytokine, IL-23, with biological activities similar as well as distinct from IL-12. Immunity. 2000;13:715–725. doi: 10.1016/s1074-7613(00)00070-4. [DOI] [PubMed] [Google Scholar]
- 157.Zelante T., Iannitti R., De Luca A., Romani L. IL-22 in antifungal immunity. Eur J Immunol. 2011;41:270–275. doi: 10.1002/eji.201041246. [DOI] [PubMed] [Google Scholar]
- 158.Endam L.M., Bosse Y., Filali-Mouhim A., Cormier C., Boisvert P., Boulet L.P. Polymorphisms in the interleukin-22 receptor alpha-1 gene are associated with severe chronic rhinosinusitis. Otolaryngol Head Neck Surg. 2009;140:741–747. doi: 10.1016/j.otohns.2008.12.058. [DOI] [PubMed] [Google Scholar]
- 159.Ramanathan M., Jr., Spannhake E.W., Lane A.P. Chronic rhinosinusitis with nasal polyps is associated with decreased expression of mucosal interleukin 22 receptor. Laryngoscope. 2007;117:1839–1843. doi: 10.1097/MLG.0b013e31811edd4f. [DOI] [PubMed] [Google Scholar]
- 160.Hamilos D.L., Leung D.Y., Wood R., Cunningham L., Bean D.K., Yasruel Z. Evidence for distinct cytokine expression in allergic versus nonallergic chronic sinusitis. J Allergy Clin Immunol. 1995;96:537–544. doi: 10.1016/s0091-6749(95)70298-9. [DOI] [PubMed] [Google Scholar]
- 161.Harlin S.L., Ansel D.G., Lane S.R., Myers J., Kephart G.M., Gleich G.J. A clinical and pathologic study of chronic sinusitis: the role of the eosinophil. J Allergy Clin Immunol. 1988;81:867–875. doi: 10.1016/0091-6749(88)90944-x. [DOI] [PubMed] [Google Scholar]
- 162.Demoly P., Crampette L., Mondain M., Campbell A.M., Lequeux N., Enander I. Assessment of inflammation in noninfectious chronic maxillary sinusitis. J Allergy Clin Immunol. 1994;94:95–108. doi: 10.1016/0091-6749(94)90076-0. [DOI] [PubMed] [Google Scholar]
- 163.Baroody F.M., Hughes C.A., McDowell P., Hruban R., Zinreich S.J., Naclerio R.M. Eosinophilia in chronic childhood sinusitis. Arch Otolaryngol Head Neck Surg. 1995;121:1396–1402. doi: 10.1001/archotol.1995.01890120054010. [DOI] [PubMed] [Google Scholar]
- 164.Ponikau J.U., Sherris D.A., Kephart G.M., Kern E.B., Gaffey T.A., Tarara J.E. Features of airway remodeling and eosinophilic inflammation in chronic rhinosinusitis: is the histopathology similar to asthma? J Allergy Clin Immunol. 2003;112:877–882. doi: 10.1016/j.jaci.2003.08.009. [DOI] [PubMed] [Google Scholar]
- 165.Shin S.H., Ponikau J.U., Sherris D.A., Congdon D., Frigas E., Homburger H.A. Chronic rhinosinusitis: an enhanced immune response to ubiquitous airborne fungi. J Allergy Clin Immunol. 2004;114:1369–1375. doi: 10.1016/j.jaci.2004.08.012. [DOI] [PubMed] [Google Scholar]
- 166.Inoue Y., Matsuwaki Y., Shin S.H., Ponikau J.U., Kita H. Nonpathogenic, environmental fungi induce activation and degranulation of human eosinophils. J Immunol. 2005;175:5439–5447. doi: 10.4049/jimmunol.175.8.5439. [DOI] [PubMed] [Google Scholar]
- 167.Okano M., Fujiwara T., Haruna T., Kariya S., Makihara S., Higaki T. Role of fungal antigens in eosinophilia-associated cellular responses in nasal polyps: a comparison with enterotoxin. Clin Exp Allergy. 2011;41:171–178. doi: 10.1111/j.1365-2222.2010.03584.x. [DOI] [PubMed] [Google Scholar]
- 168.Orlandi R.R., Marple B.F., Georgelas A., Durtschi D., Barr L. Immunologic response to fungus is not universally associated with rhinosinusitis. Otolaryngol Head Neck Surg. 2009;141:750–756. doi: 10.1016/j.otohns.2009.09.016. e1-2. [DOI] [PubMed] [Google Scholar]
- 169.Ponikau J.U., Sherris D.A., Weaver A., Kita H. Treatment of chronic rhinosinusitis with intranasal amphotericin B: a randomized, placebo-controlled, double-blind pilot trial. J Allergy Clin Immunol. 2005;115:125–131. doi: 10.1016/j.jaci.2004.09.037. [DOI] [PubMed] [Google Scholar]
- 170.Weschta M., Rimek D., Formanek M., Polzehl D., Podbielski A., Riechelmann H. Topical antifungal treatment of chronic rhinosinusitis with nasal polyps: a randomized, double-blind clinical trial. J Allergy Clin Immunol. 2004;113:1122–1128. doi: 10.1016/j.jaci.2004.03.038. [DOI] [PubMed] [Google Scholar]
- 171.Ebbens F.A., Scadding G.K., Badia L., Hellings P.W., Jorissen M., Mullol J. Amphotericin B nasal lavages: not a solution for patients with chronic rhinosinusitis. J Allergy Clin Immunol. 2006;118:1149–1156. doi: 10.1016/j.jaci.2006.07.058. [DOI] [PubMed] [Google Scholar]
- 172.Ebbens F.A., Georgalas C., Luiten S., van Drunen C.M., Badia L., Scadding G.K. The effect of topical amphotericin B on inflammatory markers in patients with chronic rhinosinusitis: a multicenter randomized controlled study. Laryngoscope. 2009;119:401–408. doi: 10.1002/lary.20064. [DOI] [PubMed] [Google Scholar]
- 173.Van Zele T., Gevaert P., Watelet J.B., Claeys G., Holtappels G., Claeys C. Staphylococcus aureus colonization and IgE antibody formation to enterotoxins is increased in nasal polyposis. J Allergy Clin Immunol. 2004;114:981–983. doi: 10.1016/j.jaci.2004.07.013. [DOI] [PubMed] [Google Scholar]
- 174.Bernstein J.M., Ballow M., Schlievert P.M., Rich G., Allen C., Dryja D. A superantigen hypothesis for the pathogenesis of chronic hyperplastic sinusitis with massive nasal polyposis. Am J Rhinol. 2003;17:321–326. [PubMed] [Google Scholar]
- 175.Tripathi A., Kern R., Conley D.B., Seiberling K., Klemens J.C., Harris K.E. Staphylococcal exotoxins and nasal polyposis: analysis of systemic and local responses. Am J Rhinol. 2005;19:327–333. [PubMed] [Google Scholar]
- 176.Conley D.B., Tripathi A., Seiberling K.A., Suh L.A., Harris K.E., Paniagua M.C. Superantigens and chronic rhinosinusitis II: analysis of T-cell receptor V beta domains in nasal polyps. Am J Rhinol. 2006;20:451–455. doi: 10.2500/ajr.2006.20.2880. [DOI] [PubMed] [Google Scholar]
- 177.Ganeshan K., Neilsen C.V., Hadsaitong A., Schleimer R.P., Luo X., Bryce P.J. Impairing oral tolerance promotes allergy and anaphylaxis: a new murine food allergy model. J Allergy Clin Immunol. 2009;123:231–238.e4. doi: 10.1016/j.jaci.2008.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Sachse F., von Eiff C., Becker K., Steinhoff M., Rudack C. Proinflammatory impact of Staphylococcus epidermidis on the nasal epithelium quantified by IL-8 and GRO-alpha responses in primary human nasal epithelial cells. Int Arch Allergy Immunol. 2008;145:24–32. doi: 10.1159/000107463. [DOI] [PubMed] [Google Scholar]
References
- Lee R.E., Kaza S., Plano G.V., Casiano R.R. The role of atypical bacteria in chronic rhinosinusitis. Otolaryngol Head Neck Surg. 2005;133:407–410. doi: 10.1016/j.otohns.2005.04.022. [DOI] [PubMed] [Google Scholar]
- Stephenson M.F., Mfuna L., Dowd S.E., Wolcott R.D., Barbeau J., Poisson M. Molecular characterization of the polymicrobial flora in chronic rhinosinusitis. J Otolaryngol Head Neck Surg. 2010;39:182–187. [PubMed] [Google Scholar]
- Feazel L.M., Robertson C.E., Ramakrishnan V.R., Frank D.N. Microbiome complexity and Staphylococcus aureus in chronic rhinosinusitis. Laryngoscope. 2012;122:467–472. doi: 10.1002/lary.22398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tichenor W.S., Thurlow J., McNulty S., Brown-Elliott B.A., Wallace R.J., Jr., Falkinham J.O., 3rd Nontuberculous Mycobacteria in household plumbing as possible cause of chronic rhinosinusitis. Emerg Infect Dis. 2012;18:1612–1617. doi: 10.3201/eid1810.120164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solyar A., Lee A.S., Przybyszewski B., Lanza D.C. Atypical mycobacterium detection in refractory chronic rhinosinusitis. Otolaryngol Head Neck Surg. 2012;146:1012–1016. doi: 10.1177/0194599812437333. [DOI] [PubMed] [Google Scholar]
- Hochstim C.J., Choi J.Y., Lowe D., Masood R., Rice D.H. Biofilm detection with hematoxylin-eosin staining. Arch Otolaryngol Head Neck Surg. 2010;136:453–456. doi: 10.1001/archoto.2010.62. [DOI] [PubMed] [Google Scholar]
- Ha K.R., Psaltis A.J., Tan L., Wormald P.J. A sheep model for the study of biofilms in rhinosinusitis. Am J Rhinol. 2007;21:339–345. doi: 10.2500/ajr.2007.21.3032. [DOI] [PubMed] [Google Scholar]
- Psaltis A.J., Ha K.R., Beule A.G., Tan L.W., Wormald P.J. Confocal scanning laser microscopy evidence of biofilms in patients with chronic rhinosinusitis. Laryngoscope. 2007;117:1302–1306. doi: 10.1097/MLG.0b013e31806009b0. [DOI] [PubMed] [Google Scholar]
- Foreman A., Singhal D., Psaltis A.J., Wormald P.J. Targeted imaging modality selection for bacterial biofilms in chronic rhinosinusitis. Laryngoscope. 2010;120:427–431. doi: 10.1002/lary.20705. [DOI] [PubMed] [Google Scholar]
- Stepanovic S., Vukovic D., Dakic I., Savic B., Svabic-Vlahovic M. A modified microtiter-plate test for quantification of staphylococcal biofilm formation. J Microbiol Methods. 2000;40:175–179. doi: 10.1016/s0167-7012(00)00122-6. [DOI] [PubMed] [Google Scholar]
- Jurcisek J.A., Dickson A.C., Bruggeman M.E., Bakaletz L.O. In vitro biofilm formation in an 8-well chamber slide. J Vis Exp. 2011;(47) doi: 10.3791/2481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prince A.A., Steiger J.D., Khalid A.N., Dogrhamji L., Reger C., Eau Claire S. Prevalence of biofilm-forming bacteria in chronic rhinosinusitis. Am J Rhinol. 2008;22:239–245. doi: 10.2500/ajr.2008.22.3180. [DOI] [PubMed] [Google Scholar]
- Cryer J., Schipor I., Perloff J.R., Palmer J.N. Evidence of bacterial biofilms in human chronic sinusitis. ORL J Otorhinolaryngol Relat Spec. 2004;66:155–158. doi: 10.1159/000079994. [DOI] [PubMed] [Google Scholar]
- Ferguson B.J., Stolz D.B. Demonstration of biofilm in human bacterial chronic rhinosinusitis. Am J Rhinol. 2005;19:452–457. [PubMed] [Google Scholar]
- Sanclement J.A., Webster P., Thomas J., Ramadan H.H. Bacterial biofilms in surgical specimens of patients with chronic rhinosinusitis. Laryngoscope. 2005;115:578–582. doi: 10.1097/01.mlg.0000161346.30752.18. [DOI] [PubMed] [Google Scholar]
- Sanderson A.R., Leid J.G., Hunsaker D. Bacterial biofilms on the sinus mucosa of human subjects with chronic rhinosinusitis. Laryngoscope. 2006;116:1121–1126. doi: 10.1097/01.mlg.0000221954.05467.54. [DOI] [PubMed] [Google Scholar]
- Psaltis A.J., Ha K.R., Beule A.G., Tan L.W., Wormald P.J. Confocal scanning laser microscopy evidence of biofilms in patients with chronic rhinosinusitis. Laryngoscope. 2007;117:1302–1306. doi: 10.1097/MLG.0b013e31806009b0. [DOI] [PubMed] [Google Scholar]
- Foreman A., Psaltis A.J., Tan L.W., Wormald P.J. Characterization of bacterial and fungal biofilms in chronic rhinosinusitis. Am J Rhinol Allergy. 2009;23:556–561. doi: 10.2500/ajra.2009.23.3413. [DOI] [PubMed] [Google Scholar]
- Hekiert A.M., Kofonow J.M., Doghramji L., Kennedy D.W., Chiu A.G., Palmer J.N. Biofilms correlate with TH1 inflammation in the sinonasal tissue of patients with chronic rhinosinusitis. Otolaryngol Head Neck Surg. 2009;141:448–453. doi: 10.1016/j.otohns.2009.06.090. [DOI] [PubMed] [Google Scholar]
- Galli J., Calo L., Ardito F., Imperiali M., Bassotti E., Passali G.C. Damage to ciliated epithelium in chronic rhinosinusitis: what is the role of bacterial biofilms? Ann Otol Rhinol Laryngol. 2008;117:902–908. doi: 10.1177/000348940811701207. [DOI] [PubMed] [Google Scholar]
- Singhal D., Psaltis A.J., Foreman A., Wormald P.J. The impact of biofilms on outcomes after endoscopic sinus surgery. Am J Rhinol Allergy. 2010;24:169–174. doi: 10.2500/ajra.2010.24.3462. [DOI] [PubMed] [Google Scholar]
- Zernotti M.E., Angel Villegas N., Roques Revol M., Baena-Cagnani C.E., Arce Miranda J.E., Paredes M.E. Evidence of bacterial biofilms in nasal polyposis. J Investig Allergol Clin Immunol. 2010;20:380–385. [PubMed] [Google Scholar]
- Lane A.P., Truong-Tran Q.A., Schleimer R.P. Altered expression of genes associated with innate immunity and inflammation in recalcitrant rhinosinusitis with polyps. Am J Rhinol. 2006;20:138–144. [PMC free article] [PubMed] [Google Scholar]
- Yamin M., Holbrook E.H., Gray S.T., Harold R., Busaba N., Sridhar A. Cigarette smoke combined with Toll-like receptor 3 signaling triggers exaggerated epithelial regulated upon activation, normal T-cell expressed and secreted/CCL5 expression in chronic rhinosinusitis. J Allergy Clin Immunol. 2008;122:1145–1153.e3. doi: 10.1016/j.jaci.2008.09.033. [DOI] [PubMed] [Google Scholar]
- Dentener M.A., Vreugdenhil A.C., Hoet P.H., Vernooy J.H., Nieman F.H., Heumann D. Production of the acute-phase protein lipopolysaccharide-binding protein by respiratory type II epithelial cells: implications for local defense to bacterial endotoxins. Am J Respir Cell Mol Biol. 2000;23:146–153. doi: 10.1165/ajrcmb.23.2.3855. [DOI] [PubMed] [Google Scholar]
- Roeder A., Kirschning C.J., Rupec R.A., Schaller M., Korting H.C. Toll-like receptors and innate antifungal responses. Trends Microbiol. 2004;12:44–49. doi: 10.1016/j.tim.2003.11.003. [DOI] [PubMed] [Google Scholar]
- Zhang Q., Wang C.S., Han D.M., Sy C., Huang Q., Sun Y. Differential expression of Toll-like receptor pathway genes in chronic rhinosinusitis with or without nasal polyps. Acta Otolaryngol. 2013;133:165–173. doi: 10.3109/00016489.2012.717713. [DOI] [PubMed] [Google Scholar]
- Akhtar M., Watson J.L., Nazli A., McKay D.M. Bacterial DNA evokes epithelial IL-8 production by a MAPK-dependent, NF-kappaB-independent pathway. FASEB J. 2003;17:1319–1321. doi: 10.1096/fj.03-0950fje. [DOI] [PubMed] [Google Scholar]
- Ramanathan M., Jr., Lee W.K., Spannhake E.W., Lane A.P. Th2 cytokines associated with chronic rhinosinusitis with polyps down-regulate the antimicrobial immune function of human sinonasal epithelial cells. Am J Rhinol. 2008;22:115–121. doi: 10.2500/ajr.2008.22.3136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee R.J., Xiong G., Kofonow J.M., Chen B., Lysenko A., Jiang P. T2R38 taste receptor polymorphisms underlie susceptibility to upper respiratory infection. J Clin Invest. 2012;122:4145–4159. doi: 10.1172/JCI64240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bals R., Hiemstra P.S. Innate immunity in the lung: how epithelial cells fight against respiratory pathogens. Eur Respir J. 2004;23:327–333. doi: 10.1183/09031936.03.00098803. [DOI] [PubMed] [Google Scholar]
- Ooi E.H., Wormald P.J., Tan L.W. Innate immunity in the paranasal sinuses: a review of nasal host defenses. Am J Rhinol. 2008;22:13–19. doi: 10.2500/ajr.2008.22.3127. [DOI] [PubMed] [Google Scholar]