Abstract
The increasing incidence and prevalence of asthma in many parts of the world continue to make it a global health concern. The heterogeneous nature of the clinical manifestations and therapeutic responses of asthma in both adult and pediatric patients indicate that it may be more of a syndrome rather than a specific disease entity. Numerous triggering factors including viral infections, allergen and irritant exposure, and exercise, among others, complicate both the acute and chronic treatment of asthma. Therapeutic intervention has focused on the appreciation that airway obstruction in asthma is composed of both bronchial smooth muscle spasm and variable degrees of airway inflammation characterized by edema, mucus secretion, and the influx of a variety of inflammatory cells. The presence of only partial reversibility of airflow obstruction in some patients indicates that structural remodeling of the airways may also occur over time. Choosing appropriate medications depends on the disease severity (intermittent, mild persistent, moderate persistent, severe persistent), extent of reversibility, both acutely and chronically, patterns of disease activity (exacerbations related to viruses, allergens, exercise, etc), and the age of onset (infancy, childhood, adulthood). (J Allergy Clin Immunol 2003;111:S502-19.)
Keywords: Airway hyperresponsiveness, allergy, asthma, β-agonists, exacerbation, exercise-induced asthma, IgE, inflammation, inhaled corticosteroids, virus
Abbreviations: COX , Cyclo-oxygenase; EIB , Exercise-induced bronchospasm; GERD , Gastroesophageal reflux disease; GR , Glucocorticoid receptor; ICS , Inhaled corticosteroids; MDI , Metered-dose inhaler; NSAID , Nonsteroidal anti-inflammatory drug; PEF , Peak expiratory flow; RSV , Respiratory syncytial virus
Definition
Despite the marked heterogeneity of the asthma phenotype, a consensus definition for asthma has been developed that recognizes this disorder to be a chronic inflammatory disorder of the airways in which many cells and cellular elements play a role, in particular, mast cells, eosinophils, T lymphocytes, neutrophils, and epithelial cells. In susceptible individuals, this inflammation causes recurrent episodes of wheezing, breathlessness, chest tightness, and cough, particularly at night and/or in the early morning. These episodes are usually associated with widespread but variable airflow obstruction that is often reversible either spontaneously or with treatment. The inflammation also causes an associated increase in the existing bronchial hyperresponsiveness to a variety of stimuli.
From this definition, a number of key points regarding the recognition, treatment, and underlying causes of asthma deserve to be highlighted:
-
•
Asthma, whatever the severity, is a chronic inflammatory disorder of the airway; this characteristic has implications for the diagnosis, prevention, and treatment of the disease.
-
•
Airway inflammation may be variably associated with changes in airway hyperresponsiveness, airflow limitation, respiratory symptoms, and disease chronicity.
-
•
Airway inflammation may be acutely and chronically associated with the development of airflow limitation as the result of bronchoconstriction, airway edema, mucus secretion, and, in some patients, airway wall remodeling.
-
•
Airway inflammation with histopathologic features found in adult asthmatic patients may begin during early childhood in high-risk individuals.
-
•
Atopy, the genetic predisposition for the development of an antigen-specific, IgE-mediated response to common aeroallergens, is the strongest identifiable predisposing factor for the development of asthma.
Pathophysiology
Genetics
The genetics of asthma have recently been extensively reviewed.1 At present, most investigators would agree that there is a major hereditary contribution to the underlying causes of asthma and allergic diseases. However, the inheritance pattern of asthma demonstrates that it is a “complex genetic disorder” such as is seen in hypertension, atherosclerosis, arthritis, and diabetes mellitus. As such, asthma cannot be classified simply as having an autosomal dominant, recessive, or sex-linked mode of inheritance.
Studies evaluating disease-causing or disease-modifying genes have demonstrated linkage to the following chromosomes or chromosomal regions: 5q31 [total IgE and eosinophil levels; cytokines (interleukins -4, -5, and -13); CD14 (endotoxin receptor important in the initiation of the innate immune response)]; 6 [major histocompatibility complex, tumor necrosis factor complex (asthma inflammation)]; 11q13 [β-chain of the high-affinity IgE receptor]; 12q [asthma]; and 13q [atopy and asthma]; among others.1 Recently, the ADAM33 gene, which encodes a protein-processing enzyme known as a metalloprotease, was found to be commonly associated with asthma.2
In addition, determining the extent of polymorphic variation in treatment response genes, termed pharmacogenetics, has come to the forefront in asthma research.3 Thus far, the primary focus of this research activity has been directed toward the characterization of genes related to β-adrenergic response,4 the 5-lipoxygenase pathway,5 and the glucocorticoid receptor.3 It is feasible that asthma treatment programs in the future may be individualized, based on the nature of each patient's polymorphic variations for genes that are found to significantly influence therapeutic responses both acutely and chronically.
Airway obstruction
Clinical symptoms and the resulting pathophysiology in asthma are the direct extension of airway obstruction. In appreciating airway obstruction and its effect on lung physiology and patient symptoms, a number of factors need to be considered. First, airway obstruction can be intermittent, persistent, and/or progressive. Second, airway obstruction can be totally, partially, or not reversible. Third, airflow obstruction can be the end result of multiple structural and/or physiologic factors that individually or collectively contribute to airway narrowing. Finally, the precise contribution of each of these features varies among asthmatic patients and contributes to the diversity in clinical manifestations including the severity of the disease and the therapeutic response to various medications.
Airway smooth muscle spasm
One of the characteristic features of asthma is airway hyperresponsiveness, which means that acute airflow obstruction occurs to several stimuli, and the resulting contractile response leads to excessive airway narrowing. Bronchial smooth muscle spasm probably is instrumental in this excessive reactivity, but there are many factors in the airway that regulate or contribute to airway smooth muscle tone. For example, the airway contains a number of resident cells (mast cells, alveolar macrophages, airway epithelium and endothelium) as well as immigrating inflammatory cells (eosinophils, lymphocytes, neutrophils, basophils, and, possibly, platelets). These cells are capable of secreting a variety of mediators, such as histamine, the cysteinyl leukotrienes (LTC4, LTD4, and LTE4), prostaglandin D2, and platelet-activating factor, which can directly contract the bronchial smooth muscle. In addition, the recruited cells can generate inflammatory mediators, which cause the airway smooth muscle to become more contractible to bronchospasm mediators.
Airway smooth muscle is also under neuroregulatory control and is innervated by the vagus nerve. Either through the direct activation of this nerve or through reflex mechanisms, the secretion of acetylcholine will lead to bronchial muscle contraction. Furthermore, there are neuroregulator mediators, such as substance P and neurokinins, which can determine airway smooth muscle tone.
Airway mucosal edema
Many of the same mediators that lead to bronchial smooth muscle contraction, for example, histamine, cysteinyl leukotrienes, and bradykinin, can increase capillary membrane permeability to cause muco-sal edema. These changes in the airway tissues will also lead to airway flow obstruction.
Mucus hypersecretion
One of the characteristic features of severe asthma is overproduction of mucus. Mucus can mechanically narrow the airway lumen and, in severe asthma, form tenacious plugs that will obliterate the airway. The development of mucous plugging of the airway occurs either in severe, prolonged attacks of asthma or in patients with chronic disease. The end result is a further compromise of the airway lumen.
Inflammation
Airway inflammation is a characteristic feature of asthma and contributes significantly to many features of this disease, including airflow obstruction, bronchial hyperresponsiveness, and the initiation of the injury-repair process (remodeling) found in some patients. The pattern of inflammation varies considerably and depends on the stage of the disease: acute, chronic, or remodeling (see below). The degree of airway inflammation varies with the severity and chronicity of the disease and may also determine the responsiveness of the patient to treatment.
Tissues from patients who died of status asthma show a characteristic pattern of airway inflammation that includes denudation of airway epithelium, mucous plugging of segmental bronchi and bronchioles, collagen deposition beneath the basement membrane, edema of the submucosa, infiltration by inflammatory cells [neutrophils (seen more frequently in exacerbations that are sudden and severe) and eosinophils], and smooth muscle hypertrophy/hyperplasia. Gradations of these responses are seen as the disease severity moves from mild asthma to a chronic, persistent process.
Many inflammatory cells contribute to airway inflammation in asthma, including activated mast cells, lymphocytes, particularly the TH2 subpopulation of cells, which release a family of proinflammatory cytokines including IL-4, IL-5, and IL-13. These cytokines will assist in the recruitment and activation of eosinophils. Lymphocytes, along with epithelial cells, will generate chemokines, including RANTES and eotaxin, which appear to be essential for the recruitment of eosinophils.
Another critical step in this process is the activation of endothelial adhesion proteins, particularly those of the immunoglobulin superfamily, ICAM-1 and VCAM-1. These proteins will combine with specific receptors on inflammatory cells, for example, neutrophils, eosinophils, and lymphocytes, to reduce their flow in the vessel and assist in cell movement to the airway (Fig 1).
Fig. 1.
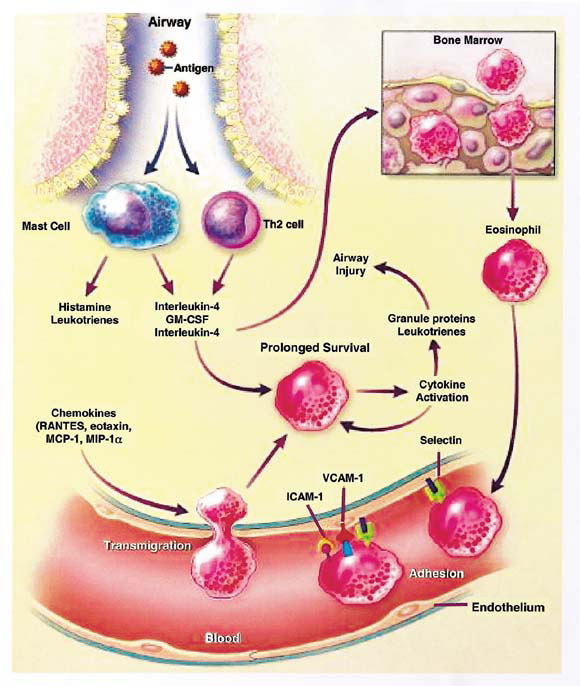
One mechanism that initiates airway inflammation in antigen exposure in sensitized individuals. Antigen interaction with mast cell-bound, specific-IgE antibody results in release of preformed (histamine) and generated (leukotrienes) mediators along with cytokines [interleukins -4 and -5 and granulocyte macrophage-colony stimulating factor (GM-CSF)]. These various compounds can induce localized inflammatory cell influx and activation through the upregulation of various chemokines and adhesion molecules and recruitment of bone marrow cells (eg, eosinophils). (Modified and reproduced with permission from Busse WW, Lemanske RF Jr. N Engl J Med 2001;344:350-62.)
Remodeling
Recent evidence has demonstrated that some patients with asthma will have irreversible airflow obstruction.6 This process has been termed airway remodeling and represents an injury-repair process of the airway tissue. A number of components of airway remodeling in asthma have been identified including airway smooth muscle hypertrophy, mucus gland and goblet cell hyperplasia, angiogenesis (vascular hyperplasia), and collagen deposition in the airway. These histologic features appear to be permanent and do not reverse with treatment.
Although the consequences of airway remodeling are appreciated, the processes involved in its regulation have yet to be fully defined. Nonetheless, the process appears to be under the control of mediators quite distinct from those involved in the acute inflammatory response. For example, in remodeling, the generation and presence of growth factors appear more critical and lead to the above structural changes in the airway tissue. Thus, the shift or transition to remodeling from allergic inflammation suggests a new family of mediators with actions on smooth muscle growth, collagen deposition, blood vessel proliferation, and mucous gland hyperplasia. Collectively, these new data provide a picture of asthma that begins with an acute cellular inflammatory response that may then evolve into a more chronic process in which structural changes occur in the airway to further affect airway hyperresponsiveness and airflow obstruction.7
Airway hyperresponsiveness
One of the characteristic features of asthma is airway hyperresponsiveness to a variety of inhaled substances (eg, methacholine) or in association with exposure to cold air, exercise, irritants, or with hyperventilation.8 Many factors contribute to hyperresponsiveness seen in asthma including genetic polymorphisms, airway architecture (ie, edema, smooth muscle hypertrophy, and deposition of collagen material), age, and time of the day (nighttime versus daytime). Airway hyperresponsiveness, if demonstrable during infancy and early childhood, may be a risk factor for the subsequent development of clinical asthma.9 Although airway inflammation contributes to this physiologic aberration in asthma, multiple other factors that influence airway caliber play a role as well.10 It is important to emphasize that airway hyperresponsiveness is not unique to asthma. A positive methacholine challenge test is diagnostic for airway hyperresponsiveness (which can be seen in atopics, patients with cystic fibrosis, and other chronic lung diseases and even in normal individuals for a few weeks after a viral respiratory tract infection), not asthma per se. The potential usefulness of this test is if it is negative (eg, in the evaluation of chronic cough), since it is unusual for a patient with clinical asthma to have a level of airway responsiveness that would fall into the normal range.
Classification
Disease severity
Asthma can be classified on the basis of etiologic factors, severity, and the pattern of airflow limitation. Since asthma is a heterogenous disorder, multiple causative factors no doubt exist for both its inception and symptom exacerbation once the disease is established. Factors underlying inception can range from viral respiratory tract infections in infancy (eg, respiratory syncytial virus11) to occupational exposure in adults.12 Factors underlying asthma exacerbations include allergen exposure in sensitized individuals, viral infections, exercise, irritants, and ingestion of nonsteroidal anti-inflammatory agents, among others. Exacerbating factors may include one or all of these exposures and vary both among and within patients.
Conventional assessments of asthma severity have combined evaluations of symptoms, amounts of β2-agonist use to treat symptoms, and lung function. Based on these types of assessments, the severity of a patient's asthma before treatment has been classified by expert panels into intermittent and three levels of persistent disease: mild, moderate, and severe (Figs 2 and 3).13 When a patient is already receiving treatment, the classification of severity should be based on the clinical features pres-ent and the step of the daily medication that the patient is currently receiving. Thus, a patient with ongoing symptoms of moderate persistent asthma, despite receiving the appropriate maintenance treatment for this step (Fig. 2, Fig. 3), should be regarded as having moderate persistent asthma. It should be emphasized that this classification scheme pertains to disease severity chronically; patients with only intermittent symptoms (eg, viral-induced asthma in young children) can still have severe deterioration in lung function during acute exacerbations. Importantly, individuals of low income, the medically underserved, inner-city populations, or certain cultural groups have increased risk for more severe asthma.14, 15
Fig. 2.

Stepwise approach for treating infants and young children (5 years of age and younger) with acute or chronic asthma (reproduced from www.nhlbi.nih.gov/guidelines/asthma/asthsumm.htm ).
Fig. 3.

Stepwise approach for treating asthma in adults and children older than 5 years of age (reproduced from www.nhlbi.nih.gov/guidelines/asthma/asthsumm.htm ).
Precipitating factors
Allergens
Allergen exposure is important in host allergic sensitization and as a common precipitant of asthmatic symptoms in both children and adults. The development of allergic disease involves first, the process of sensitization [allergen-specific IgE antibody formation in a genetically predisposed (atopic) individual], and second, the expression and targeting of this response to various organ systems (eg, nose, skin, lung, and so forth). In asthma, the target organ is obviously the lung, but immunoinflammatory events in the upper airway may contribute to loss of asthma control as well.16
The formation of antigen-specific IgE antibody to aeroallergens (eg, mites, trees, grasses, animal dander) does not usually occur until 2 to 3 years of life. Thus, aeroallergen-induced asthma is uncommon during the first year of life but begins to increase in prevalence during later childhood and adolescence and peaks in the second decade of life. Once established in genetically predisposed individuals, IgE-mediated reactions are a major contributor both to acute asthmatic symptoms and chronic airway inflammation. Chronic low-level exposure to indoor allergens and dust mite and cockroach in particular may play a major role in both asthma pathogenesis and subsequent provocation of symptoms.17 Although a wide variety of inhaled allergens can provoke asthma symptoms, sensitization to house dust mite,18 cockroach,19 Alternaria, 20 and, possibly, cat21 are important in the pathogenesis of asthma. Paradoxically, recent data would suggest that exposure to cats or dogs during early life may actually protect against the development of asthma.22 The features of these allergens to the development of asthma are not fully established but may relate to their enzymatic as well as antigenic activity.23 Alternaria exposure, in particular, may produce severe acute asthmatic symptoms, as sensitivity to Alternaria has been implicated as a risk factor for sudden respiratory arrest in adolescents and young adults with asthma.24 Although food allergy may produce bronchospasm along with skin and/or gastrointestinal symptoms, it is very unusual for food allergy to produce an isolated respiratory reaction.25
Infections
Respiratory tract infections caused by viruses,26, 27 Chlamydia, 28, 29, 30 and Mycoplasma 31 have been implicated in the pathogenesis of asthma. Of these respiratory pathogens, viruses have been demonstrated to be associated with asthma in at least three ways. First, during infancy, certain viruses have been implicated as potentially being responsible for the inception of the asthmatic phenotype. The virus most convincingly demonstrated in this regard has been respiratory syncytial virus (RSV).11, 32 However, since nearly every child has been infected at least once with this virus by 2 years of age, additional genetic, environmental, or developmental factors must contribute to the propensity of this particular virus to be linked with childhood asthma.33, 34
Second, in patients with established asthma, particularly children, viral upper respiratory tract infections play a significant role in producing acute exacerbations of airway obstruction that may result in frequent outpatient visits or in hospitalizations.26, 35, 36, 37 Rhinovirus, the common cold virus, is the most frequent cause of exacerbations, but other viruses including parainfluenza, RSV, influenza, and coronavirus also have been implicated, albeit to a lesser extent. The increased tendency for viral infections to produce lower airway symptoms in asthmatic individuals may be related, at least in part, to interactions among allergic sensitization, allergen exposure, and viral infections acting as cofactors in the induction of acute episodes of airflow obstruction.38, 39
Third, and paradoxically, infections have been considered to have the potential of actually preventing the development of allergic respiratory tract diseases, including asthma. Interest in this area increased after the advancement of the “hygiene hypothesis,”40 which proposed that increasing family size coincident with an increased number of infections may protect against these developments. On the basis of a progressively broader interpretation of this initial hypothesis, a number of other epidemiologic and biological factors have been evaluated regarding their ability to influence the development of allergic sensitization and/or asthma.40
For infections with other microbial agents, recent attention has focused on Chlamydia 41, 42 and Mycoplasma 31 as potential contributors to both exacerbations and the severity of chronic asthma in terms of loss of lung function or medication requirements. Finally, infections involving the upper airways (ie, sinusitis) have been considered to contribute to asthma control instability, evoking the concept of a unified airway16 in relation to inflammatory responses and alterations in airway physiology.
Treatment for infection-associated asthma depends on the microbe involved and the age of the patient. For virus-induced asthma exacerbations, oral corticosteroids are the most effective form of therapy. For disease severity or chronicity that is related to either Chlamydia or Mycoplasma , treatment with macrolide antibiotics may be considered.43
Exercise
Exercise is one of the more common precipitants of airway obstruction in asthmatic patients. The symptoms of exercise-induced bronchospasm (EIB) may include any or all of the following: wheezing, coughing, shortness of breath, and, in children, chest pain or discomfort. The symptoms are most intense for 5 to 10 minutes and usually resolve within 15 to 30 minutes after exercise cessation.
Given a sufficient exercise stimulus (80% of maximal heart rate for 5 to 10 minutes), the clinical pattern of EIB is fairly characteristic. Bronchodilation is the initial response to exercise, which occurs in both normal individuals and subjects with asthma, and may be mediated by the release of catecholamines. This response is transient, peaks midexercise, and returns to baseline at the end of exercise. Progressive bronchospasm then ensues, with maximal obstruction occurring 5 to 10 minutes after the cessation of exercise. Spontaneous remission usually follows, such that pulmonary function returns to baseline within 30 to 60 minutes. Under most circumstances, the degree of bronchoconstriction is rarely severe enough to be life-threatening, and such a situation almost invariably reflects advanced untreated disease or confounding triggering factors (ie, concomitant allergen or irritant exposure), or both. EIB is more apt to occur after short (4 to 10 minutes) periods of intense exercise, although obstruction has been shown to occur after exercise durations up to 30 minutes. Some individuals with EIB are capable of “running through” their symptoms. That is, despite continued exercise in the presence of acute asthma, gradual spontaneous resolution of bronchospasm occurs, giving these subjects a “second wind.”
To make the diagnosis of EIB, objective documentation of airflow obstruction after an exercise challenge test or a convincing history with appropriate response to prophylactic or rescue medication is required. Exercise challenge testing must be of sufficient intensity and duration to be able to accurately diagnose the condition, keeping in mind that such confounding problems as vocal cord dysfunction may need to be considered in the differential diagnosis.44 Classically, after an appropriate exercise stimulus, decreases in peak flow or FEV1 by 10% are highly suspicious of, and decreases by 15% are diagnostic of, EIB.
Nonsteroidal anti-inflammatory drugs
Approximately 5% to 10% of asthmatic patients will have an acute worsening of symptoms to nonsteroidal anti-inflammatory drugs (NSAIDs).45 The aspirin triad, asthma, nasal polyps, and aspirin sensitivity, is usually found in adult asthmatic patients. The response to aspirin, or other NSAIDs, begins within 1 hour of aspirin ingestion and is associated with profound rhinorrhea, eye lacrimation, and, potentially, severe bronchospasm. Patients sensitive to aspirin usually are reactive to all other NSAIDs, and variations in the frequency and severity of adverse responses appear to depend on the potency of each drug within this class of compounds to inhibit the activity of the cyclo-oxygenase (COX)-1 enzyme. The use of COX-2 inhibitors in aspirin-sensitive patients is usually not a problem in the majority of patients46; rofecoxib has recently been shown to have an excellent safety profile in this regard.47
The sensitivity to NSAIDs is not IgE-mediated but involves the modulation of eicosanoid production. It has been suggested that NSAIDs act by reducing the formation of prostaglandins that help maintain normal airway function while increasing the formation of asthma-provoking eicosanoids, including hydroxyeicosatetraenoic acids and large quantities of cysteinyl leukotrienes.45 In addition, there is evidence that mast cell activation occurs and its mediators can be detected in nasal secretions during an episode of aspirin-induced asthma.48
A precise phenotype for patients at risk for aspirin-induced responses has yet to be fully described, but overexpression of leukotriene C4 synthase has been associated with this syndrome.49 However, this syndrome should be of concern in any asthmatic patient with nasal polyposis, chronic sinusitis, and eosinophilia, although the polyposis and sinusitis may precede the onset of recognized NSAID sensitivity by years.
Gastroesophageal reflux
The true incidence of gastro-esophageal reflux disease (GERD) in asthma, and as a causative factor in disease severity, has yet to be established. However, it has been estimated that as many as 45% to 65% of adults and children with asthma have GERD. The mechanisms by which GERD affects asthma are also not established but may include microaspiration or irritation of the esophagus with reflex bronchospasm. Although often asymptomatic in its presentation, many patients have nighttime exacerbations or difficult-to-control symptoms. Confirmation of the importance of GERD to asthma often requires endoscopy and 24-hour monitoring of intraesophageal pH levels with concomitant measures of peak expiratory flow rates. Recognition of this factor in asthma severity is important since effective therapy is currently available.50
Psychosocial factors
The role of psychosocial factors, or “stress,” has undergone an important reevaluation both in terms of a disease risk factor and a concomitant component of severity. In addition to patient stress acting in an autocrine fashion, recent evidence has shown that parental stress is a risk factor for asthma expression in some children. The mechanisms by which this occurs have not been defined but may include the promotion of allergic inflammation.51
Diagnosis
Objective parameters
Asthma is an obstructive lung disease (defined by a decreased FEV1/FVC ratio) but differs from other obstructive lung diseases (emphysema, cystic fibrosis, and so forth) in that diffusing capacity is normal and the airway obstruction is usually reversible (partially or completely). Measures of pulmonary function are essential to assessing the severity of asthma, and are also useful to monitor the course of asthma and a patient's response to therapy. Spirometry is recommended in the initial assessment of most patients with suspected asthma. Subsequent measurement of peak expiratory flow (PEF) at home may be helpful guides to assess symptoms, to alert to worsening of airflow obstruction, and to monitor therapeutic responses.
Abnormalities in pulmonary function are a measure and reflection of the degree of airflow obstruction and reflect the consequence of asthma on airway mechanics. Typical spirometric abnormalities during an exacerbation of symptoms include reductions of FEV1, PEF, FEV1:forced vital capacity ratio, and an increase in the FEV1 (>12% to 15%) in response to a bronchodilator. However, failure to demonstrate an improvement with bronchodilator should not be interpreted as absolute evidence of irreversible disease of the airways, but rather that the major component of obstruction is inflammation, not bronchospasm. Demonstration of the extent of reversibility often requires the administration of corticosteroids. Other abnormalities in lung volumes include a decreased vital capacity and an increase in functional residual capacity, total lung capacity, and residual volume (up to 300% to 600% of predicted normal value during an acute attack). Additional abnormalities in parameters of pulmonary function include a decrease in frequency-dependent compliance (a sensitive indicator of obstruction of the small airways), increased airway resistance, and a decrease in its reciprocal, specific airway conductance.
Simple pulmonary function tests (such as PEF or FEV1) performed routinely in an outpatient setting are useful methods to monitor the course of asthma. To help manage asthma at home, a system of PEF zones can be used which correlate PEF measurements and variability with appropriate levels of medication to control asthma.52 Action plans targeting symptom control versus PEF values have also been utilized, and the superiority or lack thereof of each approach in achieving and maintaining asthma control recently have been evaluated (www.nhlbi.nih.gov/guidelines/asthma/asthsumm.htm ). The consensus opinion was that current evidence neither supports nor refutes the benefits of written action plans based on PEF monitoring compared with symptom-based plans in improving health care utilization, symptoms, or lung function. In patients with moderate to severe persistent asthma, home peak flow monitoring should be considered because it may enhance clinician-patient communication and may increase patient and care giver awareness of the disease status and control.
Bronchoprovocation
Methacholine
Airway hyperresponsiveness is a physiologic characteristic of asthma, and its presence can be helpful in establishing the diagnosis. Although hyperresponsiveness is not diagnostic of asthma, its absence is a strong indication that the condition under evaluation is unlikely to be asthma. Airway hyperresponsiveness can be identified and quantified in pulmonary testing facilities by using bronchial challenge or provocation techniques, which are helpful in establishing the presence of asthma when baseline lung functions are normal and the diagnosis is in question. The most commonly employed methods used to evaluate airway hyperresponsiveness include inhalation provocation with methacholine (direct stimuli) and exercise challenge (indirect stimuli). Direct stimuli act upon the relevant receptors on bronchial smooth muscle stimulating airway muscle contraction directly. Indirect stimuli result in airway smooth muscle contraction through one or more intermediate mechanisms, including local or central neuronal reflexes, activation of resident (eg, mast cells through non-IgE-dependent mediator release) or inflammatory cells, or others.8 Bronchoprovocation studies with methacholine are more sensitive but less specific than is exercise challenge for diagnosing asthma. In addition, indirect airway responsiveness correlates better with asthma severity, asthma symptoms, and airway inflammation.8
To perform methacholine bronchoprovocation, changes in pulmonary function (ie, fall in FEV1) are measured with serial spirometry after the patient inhales incremental doses of methacholine. People with asthma respond to bronchoprovocation with greater degrees of airflow obstruction than do normal subjects. The concentration at which patients respond, that is, provocative concentration causing a 20% fall in FEV1 (PC20), defines the level of bronchial responsiveness. Bronchial provocation can be helpful in the differential diagnosis of asthma when the history, physical findings, and baseline lung functions are not adequate to confirm the clinical diagnosis of asthma, that is, cough variant asthma and the evaluation of exercise-induced dyspnea.
Exercise
To establish a diagnosis of EIB, an exercise challenge can be performed.53 With exercise, there is loss in airway heat and water, which then provokes bronchospasm. To simulate these conditions in a laboratory, patients exercise for 4 to 8 minutes to achieve 50% or more of the patient's maximum predicted oxygen consumption. In the formal laboratory setting, an exercise challenge is often done with treadmill exercise to raise the patient's heart rate to that which produced 80% to 90% of oxygen utilization for 6 to 8 minutes. Pulmonary function measurements of the FEV1 are determined before and after exercise and at 5-minute intervals for 20 to 30 minutes.
Alternatively, a patient can run outdoors (or perform the amount and type of exercise associated with symptoms) for 4 to 8 minutes at a brisk pace. PEF can be monitored after this challenge. This type of challenge can be helpful because it recreates the conditions associated with induction of respiratory symptoms. Most exercise physiologists would consider a decrease of >10% consistent with, and a decrease of >15% diagnostic of, exercise-induced bronchospasm.53
Other physiological alterations
Chest radiography
In patients with newly diagnosed asthma, a chest radiograph is often obtained to rule out coexisting diseases; however, abnormalities related to asthma are unusual.54 During acute exacerbations, there is commonly hyperinflation and mucous plugging with resulting atelectasis. Occasionally in severe asthma, a pneumothorax or pneumomediastinum may occur. A chest radiograph in these situations provides helpful information if there is significant compromise of the lung space or assisted ventilation is required. The benefit of newer techniques, such as high-resolution computerized axial tomography, in the diagnosis and treatment of asthma, has not been fully assessed.
Peripheral white blood cell counts
Although peripheral white blood cell counts add little to overall asthma treatment, the finding of peripheral blood eosinophilia can either assist in the diagnosis of asthma or provide a surrogate marker of asthma severity. In children, increased absolute eosinophils counts may be a predictor of future asthma risk.55
Arterial blood gases
Arterial blood gas measurements reflect the consequence of airflow obstruction on arterial oxygenation and levels of carbon dioxide.56 With the development of airflow obstruction in asthma, there is an uneven distribution of inspired air, which is reflected in ventilation/perfusion ratio. During a mild to moderate exacerbation of asthma, hypoxia develops and becomes more severe as the airflow obstruction intensifies.
Blood gas analyses should be obtained in patients with an acute exacerbation of asthma and severely impaired pulmonary function tests when there is a failure to respond to therapy in a 30- to 60-minute period, if there is a history of frequent hospitalizations for asthma, or of multiple emergency department visits in the preceding hours or days. The earliest abnormalities in arterial blood gas levels are respiratory alkalosis and hypocarbia, with the partial pressure of oxygen remaining normal. With increasing severity of asthma, hypoxia intensities and alterations in carbon dioxide and pH should be evaluated closely. The appearance of a “normal” PCO 2 suggests patient fatigue, whereas acidemia and an increased PCO 2 indicate impending respiratory failure. Therefore, a patient with a severe exacerbation can progress through stages of hypoxemia with a respiratory alkalosis, to hypoxemia with normal pH and PCO 2, to the stage of impending respiratory failure. Recognizing this possible progression is essential so that the treating clinician does not get a false sense of security when blood gas findings consistent with a “moderate exacerbation” are present.
Differential diagnosis
Infants and children
Wheezing, a symptom principally associated with asthma, is a common clinical presentation in infancy and childhood. Approximately 20% of all children have at least one wheezing illness by 1 year of age, nearly 33% by 3 years of age, and almost 50% by 6 years of age.57 The majority of these episodes are triggered by viral respiratory tract infections.58 However, multiple other causes for wheezing in this age group also must be considered including cystic fibrosis, anatomic abnormalities (eg, vascular ring, tracheomalacia, bronchomalacia), foreign body aspiration, and gastroesophageal reflux, among others. Once these “nonasthmatic” wheezing illnesses have been excluded, the characterization of various wheezing phenotypes and their subsequent risk for the later development of asthma can be determined.
By age 6 years, based on the time of onset and pattern of wheezing symptoms, children have been grouped into at least three wheezing phenotypes: transient wheezers (present in the first 3 years, gone by age 3 years), persistent wheezers (present in the first 3 years and still present beyond age 3 years), and late-onset wheezers (not pres-ent in the first 3 years, but symptoms begin between 3 and 6 years of age).59 Transient wheezing is associated with diminished lung function (possibly related to reduced lung size) at birth that, over time, tends to normalize.59 Late-onset wheezing is associated with an increased tendency toward allergic sensitization and relatively stable lung function at least over the first decade of life.59 Persistent wheezing is more commonly seen in children with asthmatic parents55; those who have significant lower respiratory tract illnesses with respiratory syncytial virus11; and in the Southwestern part of the United States, those with allergic sensitization to the mold Alternaria .20 Importantly, children who have persistent wheezing tend to have near normal levels of lung function at birth that decrease significantly during the first 5 to 10 years of life. Thus, if prevention or attenuation of this loss of lung function is possible, early recognition and treatment of children who will have persistent wheezing appears to be critical.
To assist clinicians in identifying children at high risk of development of asthma, an asthma risk index was recently developed on the basis of results obtained in a large cohort of children followed from birth through adolescence.55 Children with a history of recurrent wheezing (>3 episodes in past year, one physician-diagnosed) and either one major criteria (parental history of asthma, physician-diagnosed atopic dermatitis, or aeroallergen sensitivity), or two minor criteria (peripheral eosinophilia ≥4%, food sensitivity, wheezing unrelated to infections) have a 65% chance of having asthma at age 6 years. If none of these criteria are present, the chance of a child having asthma at this age is <5%. Current clinical trials are being performed to determine if children with positive asthma risk indexes who are identified and treated in early childhood can have the incidence of development of asthma reduced and/or the decrease in lung function prevented.
Adults
Like children, the cardinal clinical features of asthma include cough, wheeze, and shortness of breath. These are nonspecific symptoms, and other respiratory problems need to be considered in the diagnosis of asthma. Furthermore, because asthma may be intermittent in its severity, abnormalities in the physical examination and in lung functions may be absent at the time of evaluation. Therefore, the diagnosis of asthma will require recognition of important historical items, the physical examination, which also evaluates for the presence of coexisting illnesses, and the use of pulmonary functions to altered reversible airflow obstruction or the presence of bronchial hyperresponsiveness.
Many of the same factors that can cause wheezing in children and masquerade for asthma are found in adults. These include upper airway obstruction, foreign bodies, tracheal compression, and luminal or extraluminal tracheal disease. Of these, the most frequent confounding problem is vocal cord dysfunction (VCD). Patients with VCD have acute respiratory distress, loud audible wheezing, and measures of airflow obstruction on pulmonary function tests. In some patients, the clinical picture can be further complicated if VCD and asthma coexist. The two other common causes of chronic obstructive lung disease in adults, emphysema and chronic bronchitis, can be differentiated from asthma on the basis of abnormal diffusing capacities in emphysema, cough and sputum production in chronic bronchitis, and a smoking history in both. It should be noted that asthma may coexist with both of these conditions, making the finding of reversibility challenging to interpret in some cases.
Eosinophilia is characteristic feature of asthma. A number of pulmonary diseases have wheezing, pulmonary infiltration, and eosinophilia. These include allergic bronchopulmonary aspergillosis, chronic eosinophilic pneumonia, and Churg-Strauss syndrome. The presence of recurrent or persistent infiltrates on chest radiography are indications that the patient does not have uncomplicated asthma.
Therapy
Medications
β2-Adrenergic agonists
β2-Adrenergic drugs are the most potent and rapidly acting bronchodilators in clinical use today. Their availability in multiple forms (short, intermediate and long-acting) and delivery systems (metered-dose inhalers [MDIs], nebulizer solutions, oral liquids and tablets, respirable powders) gives them wide clinical versatility (see Figs 4 and 5).
Fig. 4.
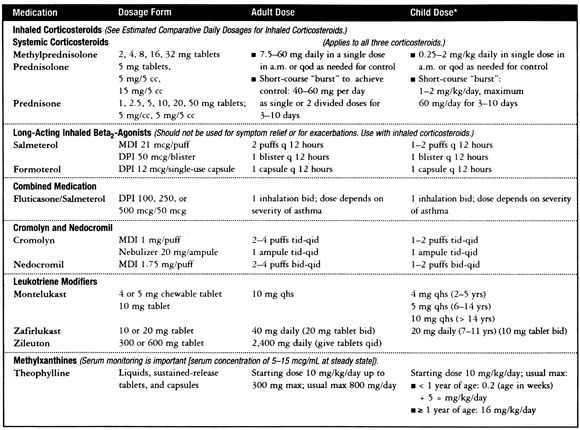
Dosages for long-term control medications (reproduced from www.nhlbi.nih.gov/guidelines/ asthma/asthsumm.htm).
Fig. 5.

Estimated comparative daily dosages of inhaled corticosteroids (reproduced from www.nhlbi.nih.gov/guidelines/asthma/asthsumm.htm ).
In addition to relaxing airway smooth muscle, β2-agonists enhance mucociliary clearance, decrease vascular permeability, and may modulate mediator release from mast cells.60 Side effects of selective β2-agonists include tremor, tachycardia, and increased anxiety, but these effects are minimal when β-agonists are administered by inhalation.60
For acute rescue treatment of asthma exacerbations, intermediate-acting (albuterol, terbutaline, pirbuterol) β2-agonists may be used every 4 to 6 hours by aerosolized delivery devices (MDIs or nebulizers). A need for more frequent administration than twice weekly for symptom relief (ie, rescue use) should alert the treating physician that the patient's underlying disease process (ie, inflammation) needs more aggressive and appropriate intervention (Fig. 2, Fig. 3).
The long-acting β-agonists salmeterol and formoterol are also effective for the treatment of moderate to severe persistent asthma.61 Long-acting β-agonists should not be used as monotherapy in patients requiring daily controller medications.62 However, in patients receiving inhaled corticosteroids (ICS) whose asthma is suboptimally controlled, these agents produce better overall asthma control when added to this baseline dose of ICS than what can be achieved by doubling the dose of the ICS.63, 64 The one outcome measure that seems to differ in this regard is frequency of asthma exacerbations, in which both increasing the dose of an ICS and/or adding a long-acting β-agonist to a baseline dose of ICS provide significant improvement.65 Once asthma control is optimized after the introduction of a long-acting β-agonist in this clinical setting, reduction in dose but not elimination of the ICS can safely be instituted in the majority of patients.66
Interest in the development of levalbuterol, the (R) enantiomer of racemic (RS) albuterol, arose from data in animal models that suggested the (S) enantiomer may produce adverse effects.67 Despite its approved use down to 6 years of age, the relative advantages of using levalbuterol in place of racemic albuterol (both in terms of efficacy and safety) have been questioned by a number of investigators.68, 69, 70, 71, 72 Based on these conflicting opinions, consensus recommendations regarding its use as a substitute for racemic albuterol will not be possible until further studies can address these controversial issues.
Theophylline
Theophylline, a methylxanthine, is a bronchodilator medication that may have mild anti-inflammatory effects as well.73 Sustained-release the-ophylline preparations and aminophylline can be used as controller medications in asthma in both children and adults.74, 75 Due to its low cost, it is used in many countries to treat mild disease. Although, it can be used as an add-on therapy to low or high doses of inhaled glucocorticoids when further asthma control is needed, it is less effective in this capacity than the addition of a long acting β2-agonist.76
Serum levels of theophylline, due to liver metabolism, may be markedly affected by a number of variables including age, diet, disease states, and drug interactions, all of which contribute to the complexity of using this medication.73 In addition, theophylline may produce a number of dose-related side effects. Gastrointestinal symptoms may be intolerable to some patients, even well within the usual therapeutic drug levels. For children taking theophylline, a concern to parents and teachers is the suggestion that theophylline might adversely affect school performance, although many studies have not been able to substantiate this association.73 Nonetheless, avoiding prescribing theophylline to children with preexisting behavior and/or school difficulties seems reasonable.
Cromolyn and nedocromil
Cromolyn sodium and nedocromil sodium are two structurally different anti-inflammatory medications for the treatment of chronic asthma that have similar properties. They are rapidly absorbed from the lungs and are impressively safe. Both medications are not bronchodilators but have been shown to inhibit inflammatory cell activation and mediator release, early and late allergen-induced bronchoconstriction, and reduce airway hyperresponsiveness.77, 78 The mechanism of action of these agents may be related to their effects on airway epithelial chloride channels79 or their effects on local neuronal reflexes. Cromolyn has been shown to be effective in both adult80 and pediatric patients.81 Both can be modestly effective prophylactically in the attenuation of exercise-induced bronchospasm,82 albeit less so than β2-agonists. Both agents may be useful for prophylaxis before relevant allergen exposure in sensitized patients. For long-term treatment of persistent asthma in children, treatment with ICS has been shown to be superior to nedocromil in achieving overall asthma control.83
Leukotriene antagonists
Leukotrienes are biologically active fatty acids derived from the oxidative metabolism of arachidonic acid, an integral part of the cell membrane. The cysteinyl leukotrienes (LTC4, LTD4, and LTE4) can be generated by eosinophils, mast cells and alveolar macrophages and combine with specific receptors, CysLT1 and CysLT2. The majority of actions of cysteinyl leukotrienes are generated by interaction with the CysLT1 receptor which can lead to airway smooth muscle contraction, leukocyte chemotaxis, and increases in vascular permeability. The actions of leukotrienes can be prevented by inhibition of cysteinyl leukotriene synthesis [5-lipoxygenase inhibitors (zileuton)] or antagonists or leukotriene receptors (zafirlukast and montelukast). Of these compounds, the receptor antagonists are now most frequently used in asthma treatment.84, 85
The receptor antagonists have been shown to inhibit exercise-provoked bronchospasm, to modify the airway response to inhaled antigens, and to improve airway function in patients with chronic asthma. In adult patients with asthma, leukotriene receptor antagonists can improve airflow obstruction between 8% and 13%, reduce the need for β-agonists, and reduce asthma exacerbations. In head-to-head trials with inhaled cortico-steroids, the leukotriene receptor antagonists are less effective in terms of improvement in lung function and reduction in exacerbations. However, when added to a baseline dose of inhaled corticosteroids, this class of compounds has the potential of improving overall asthma control. The convenience of once-per-day oral dosing (montelukast) and safety has been an appeal for some patients. With increasing experience in clinical response profiles and pharmacogenetic variability, more precise recommendations on prescribing priority and placement should be possible.
Glucocorticoids
Glucocorticosteroids are the most potent anti-inflammatory agents available for the treatment of asthma.86 Their efficacy is related to many factors including a diminution in inflammatory cell function and activation, stabilization of vascular leakage, a decrease in mucus production, and an increase in β-adrenergic response. Glucocorticoids produce their effect on various cells by binding to intracellular glucocorticoid receptors (GR), which go on to regulate transcription of certain target genes. Steroid-bound GR form dimers that bind to DNA glucocorticoid response elements (GREs), resulting in increased transcription, an increase in mRNA, and increased synthesis of proteins. However, in asthma, it is more likely that control of inflammation comes from repression of gene transcription.
ICS have the potential for producing systemic side effects that are dependent on the dose and potency of the corticosteroid as well as its bioavailability, absorption in the gut, first-pass metabolism in the liver, and the half-life of its systemically absorbed (from lung and possibly gut) fraction.
Although ICS, when used in recommended doses, have minimal adverse effects (with the exception of oral candidiasis when oral hygiene is suboptimal), concern has been raised that the use of these agents in children may be associated with growth suppression.87 However, recent data have been reassuring. Two long-term studies have demonstrated that although some reductions in growth velocity will occur (about 1 to 1.5 cm/y) within the first few months of therapy (using recommended doses),83 long-term treatment should not influence the attainment of predicted adult heights in the majority of children.88 However, since the use of low-dose ICS may rarely adversely affect growth and the use of high doses of ICS can be associated with more significant long-term consequences, dose reductions should be attempted whenever possible by using various forms of adjunctive controller medications.
The goals in using corticosteroids in the treatment of asthma are similar to those in using other classes of medications and can be viewed simplistically in two ways. First, to gain control of the underlying disease process, or to achieve a state of disease remission (eg, in status asthmaticus, or in patients with chronic stable asthma who have had significant diminutions in lung function over more prolonged time periods). Second, to maintain this control (remission) for as long as possible with the least amount of side effects. These initial actions usually require high-dose ICS or, more frequently, systemic corticosteroids. Remittive therapy usually requires an oral corticosteroid burst [eg, 0.5 to 1.0 mg/kg per day prednisone for 5 days (usual adult maximum is 40 to 60 mg/d)]. The duration of the burst will vary considerably among patients, but the goal should be to maximize lung function, minimize symptoms, and reduce the use of rescue medications. At the same time, an ICS medication should be started at doses sufficient to maintain the initial remission for prolonged periods of time. The dose and type of medication may be influenced by age (delivery systems, side effects), cost, and the familiarity of the clinician with the nuances of the various available products. It should be emphasized that the abrupt discontinuation of ICS is an important cause of asthma exacerbations.
Treatment
Acute asthma
Exacerbations of asthma (asthma attacks) can occur for a variety of reasons (viral respiratory infections, allergen exposure, aspirin ingestion, or withdrawal of medications, particularly inhaled corticosteroids). Treatment of these exacerbations will depend on the age of the patient and the severity of the episode at the time of evaluation (Fig 6).56, 89 Mild exacerbations may be treated at home (Fig 7) with clinician-generated, symptom-based action plans; acute, severe exacerbations of asthma are potentially life-threatening and require critical assessment and appropriate therapy.
Fig. 7.

Home treatment of asthma exacerbations (reproduced from Murphy S, Bleecker ER, Boushey H, et al, editors. Guidelines for the diagnosis and management of asthma. National Asthma Education and Prevention Program. II, 1-150. 1997. Bethesda, Md: National Institutes of Health).
To determine the severity of an asthma exacerbation, a number of factors can be evaluated from history, examination and assessment of lung function (Fig 6).
Fig. 6.

Estimated severity of asthma exacerbations (reproduced from Murphy S, Bleecker ER, Boushey H, et al, editors. Guidelines for the diagnosis and management of asthma. National Asthma Education and Prevention Program. II, 1-150. 1997. Bethesda, Md: National Institutes of Health).
For patients with exacerbations significantly severe to require outpatient evaluation (Fig 8), a brief history and physical examination are necessary and appropriate before beginning treatment. Of historic importance are the severity of symptoms, current medications (including recent corticosteroid use), onset of symptoms, and prior hospitalization or emergency department visits. In addition to a physical examination to determine vital signs and auscultation of the chest for breath sounds/wheezing or “silent chest,” careful attention should be directed toward alertness, cyanosis, and use of accessory muscles of respiration (retractions or abdominal breathing in children). Collectively, these features can provide objective insight into the level of asthma severity.
Assessment of lung function by using peak flow meters or spirometry (FEV1 and FVC) is essential to most precisely determine the baseline level of airflow obstruction and the ultimate response to therapy. With severe airflow obstruction (FEV1 <40%) or a history of severe respiratory compromise, an arterial blood gas value can assess oxygenation and carbon dioxide concentrations as an indicator of impending respiratory failure.
Initial treatment consists of administering oxygen [maintain oxygen saturation 90% (95% children)] and inhaled β2-agonists (Fig 8). Although rapid-acting inhaled β2-agonists are generally administered by nebulization, equivalent bronchodilation with a more rapid onset, fewer side effects, and less time in the emergency department can be achieved with an MDI with a spacer.90 For airflow obstruction not responding adequately to bronchodilator delivery with an MDI, continuous nebulizer therapy has been shown to be more effective compared with similar therapy administered intermittently.91 The administration of subcutaneous or intramuscular epinephrine should be reserved for emergency situations in which aerosolized delivery of β2-agonists is not possible or when acute airflow obstruction is part of a more generalized anaphylactic reaction.
Fig. 8.
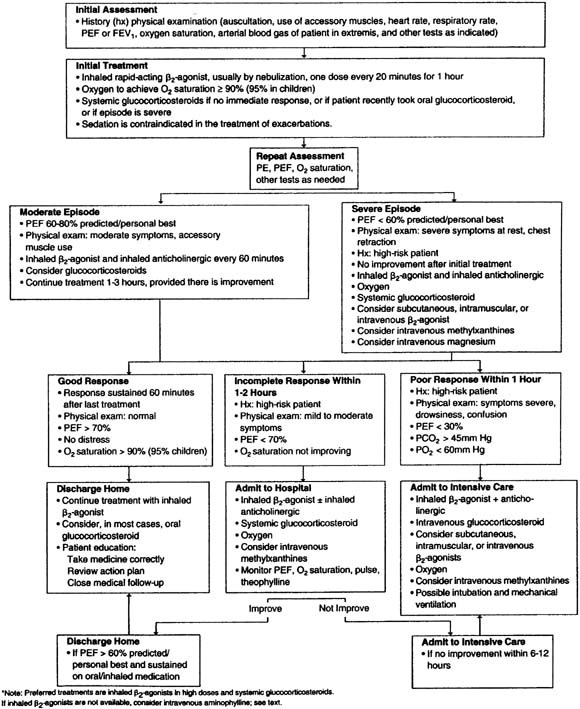
Hospital-based treatment of asthma exacerbations (reproduced from Murphy S, Bleecker ER, Boushey H, et al, editors. Guidelines for the diagnosis and management of asthma. National Asthma Education and Prevention Program. II, 1-150. 1997. Bethesda, Md: National Institutes of Health).
The use of additional bronchodilator therapy (eg, ipratropium bromide or theophylline) in the setting of acute asthmatic exacerbations also has been evaluated. A combination of β2-agonists and ipratropium bromide (anticholinergic agent) may produce better bronchodilation than either drug alone and is associated with lower hospitalization rates in both adult and pediatric patients.92 The efficacy of intravenous aminophylline in acute severe asthma has been evaluated, and the accumulated evidence in the majority of patients does not support its routine use in this setting because of its high risk/benefit ratio.93
Treatment with glucocorticosteroids (inhaled, oral, and/or parenteral) is the mainstay of treatment for asthma exacerbations. From the variety of approaches that have been evaluated for the treatment of exacerbations ranging from mild to severe, the overall results indicate the importance of treating each patient individually based on their prior pattern of therapeutic response and/or the nature and severity of the current clinical presentation that is being addressed. For home treatment of mild exacerbations, increasing doses of baseline ICS or intermittent intervention with high doses of ICS may be efficacious in averting the progression of symptoms, particularly in children.94 In patients in the emergency department, treatment of both children and adults with high doses of ICS who are not currently receiving corticosteroid therapy may reduce the risk of subsequent hospitalization.95
For more moderate to severe exacerbations, systemic corticosteroids are usually required because they enhance the speed of resolution of symptoms and significantly improve a variety of outcome measures. They should be considered integral to the treatment of these episodes, especially if:
-
•
The initial rapid-acting inhaled β2-agonist dose has failed to achieve lasting improvement
-
•
The exacerbation developed even though the patient was taking oral corticosteroids
-
•
Previous exacerbations required oral corticosteroids.
Systemic corticosteroids require at least 4 hours to produce clinical improvement.13 A meta-analysis has suggested that doses of corticosteroids equivalent to 60 to 80 mg methylprednisolone or 300 to 400 mg hydrocortisone per day are adequate for hospitalized patients, and even 40 mg methylprednisolone or 200 mg hydrocortisone is probably adequate.96, 97 There are no convincing data on the proper duration of oral prednisone treatment, although a 10- to 14-day course in adults and a 3- to 5-day course in children are usually considered appropriate.13 Current evidence suggests that there is no benefit to tapering the dose of oral prednisone either in the short term98 or over several weeks.99
Chronic asthma
In formulating a strategy for the treatment of chronic asthma, goals of therapy need to be developed and include what defines asthma control. The following criteria may not be achieved in every patient but provide reasonable criteria for treatment52:
-
•
Minimal (or none) chronic symptoms including nocturnal symptoms
-
•
Reduce frequency of exacerbations, including the need for emergency room visits and hospitalizations
-
•
Minimize the need for acute rescue therapy such as inhaled β2-agonists
-
•
Establish a normal lifestyle with no limitations on activities including exercise
-
•
Normalize pulmonary functions
-
•
Minimal to no adverse effects from medications
Although the selection of pharmacologic treatment is determined and dependent upon many factors, it is, in general, based on the severity of asthma. Because asthma is a variable but chronic disease (or syndrome), specific treatment will need to be adjusted both acutely or during exacerbations and chronically to maintain adequate symptom control and minimize side effects and cost over more prolonged periods of time.
To accomplish these goals, a stepwise approach has been adapted for treatment (Fig. 2, Fig. 3).52 (www.nhlbi.nih.gov/guidelines/asthma/asthsumm.htm ) The basis of the stepwise approach is to increase the number, frequency, and dose of medications with increasing asthma severity until the patient's disease has been put into remission. Usually, the initial treatment is given at a high level but appropriate to asthma severity. When control is achieved, a careful stepdown in therapy is considered to maintain disease remission with the fewest number of medications and the least number of side effects from the various treatments.
Asthma severity has been divided into intermittent and persistent, which is further divided into mild, moderate, and severe (Fig. 2, Fig. 3). Placement of patients into various steps is based on the features of asthma at the time of the initial evaluation (when patients have not yet received medications for their asthma) or based on their asthma features and/or medication requirements to maintain maximum disease control. The classification of intermittent asthma does not indicate a level of severity, since patients in this category may only have intermittent symptoms but, when symptoms develop, they may be abrupt and severe in nature.
Summary
Asthma is a complex genetic disorder that is characterized by airway inflammation and reversible airflow obstruction. It is further distinguished by multiple phenotypes that may differ on the basis of age of onset, triggering factors, and patterns of severity both during acute exacerbations and on a more chronic basis as reflected by variably reversible loss of lung function. As a result of this clinical heterogeneity, treatment approaches need to be individualized and modified to obtain and maintain adequate symptom and disease control over time. Although current therapy is targeted at development of secondary and tertiary prevention strategies, ongoing research is evaluating the prospects of primary prevention as well.
Footnotes
Supported by NIH Grants 1PO1AI50500, 2P50HL56396, and 1RO1HL61879.
Reprint requests: Robert F. Lemanske, Jr, MD, University of Wisconsin Hospital, 600 Highland Ave, K4/916, Madison, WI 53792.
References
- 1.Cookson WO. Asthma genetics. Chest. 2002;121(3 Suppl):7S–13S. doi: 10.1378/chest.121.3_suppl.7s-a. [DOI] [PubMed] [Google Scholar]
- 2.Van Eerdewegh P, Little RD, Dupuis J, Del Mastro RG, Falls K, Simon J. Association of the ADAM33 gene with asthma and bronchial hyperresponsiveness. Nature. 2002;418:426–430. doi: 10.1038/nature00878. [DOI] [PubMed] [Google Scholar]
- 3.Fenech A, Hall IP. Pharmacogenetics of asthma. Br J Clin Pharmacol. 2002;53:3–15. doi: 10.1046/j.0306-5251.2001.01509.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Israel E, Drazen JM, Liggett SB, Boushey HA, Cherniack RM, Chinchilli VM. The effect of polymorphisms of the b2-adrenergic receptor on the response to regular use of albuterol in asthma. Am J Respir Crit Care Med. 2000;162:75–80. doi: 10.1164/ajrccm.162.1.9907092. [DOI] [PubMed] [Google Scholar]
- 5.Palmer LJ, Silverman ES, Weiss ST, Drazen JM. Pharmacogenetics of asthma. Am J Respir Crit Care Med. 2002;165:861–866. doi: 10.1164/ajrccm.165.7.2109096. [DOI] [PubMed] [Google Scholar]
- 6.Vignola AM, Kips J, Bousquet J. Tissue remodeling as a feature of persistent asthma. J Allergy Clin Immunol. 2000;105:1041–1053. doi: 10.1067/mai.2000.107195. [DOI] [PubMed] [Google Scholar]
- 7.Richter A, Puddicombe SM, Lordan JL, Bucchieri F, Wilson SJ, Djukanovic R. The contribution of interleukin (IL)-4 and IL-13 to the epithelial-mesenchymal trophic unit in asthma. Am J Respir Cell Mol Biol. 2001;25:385–391. doi: 10.1165/ajrcmb.25.3.4437. [DOI] [PubMed] [Google Scholar]
- 8.Cockcroft DW. How best to measure airway responsiveness. Am J Respir Crit Care Med. 2001;163:1514–1515. doi: 10.1164/ajrccm.163.7.2103055b. [DOI] [PubMed] [Google Scholar]
- 9.Palmer LJ, Rye PJ, Gibson NA, Burton PR, Landau LI, LeSouef PN. Airway responsiveness in early infancy predicts asthma, lung function, and respiratory symptoms by school age. Am J Respir Crit Care Med. 2001;163:37–42. doi: 10.1164/ajrccm.163.1.2005013. [DOI] [PubMed] [Google Scholar]
- 10.James AL, Pare PD, Hogg JC. The mechanics of airway narrowing in asthma. Am Rev Respir Dis. 1989;139:242–246. doi: 10.1164/ajrccm/139.1.242. [DOI] [PubMed] [Google Scholar]
- 11.Stein RT, Sherrill D, Morgan WJ, Holberg CJ, Halonen M, Taussig LM. Respiratory syncytial virus in early life and risk of wheeze and allergy by age 13 years. Lancet. 1999;354:541–545. doi: 10.1016/S0140-6736(98)10321-5. [DOI] [PubMed] [Google Scholar]
- 12.Malo JL, Chan-Yeung M. Occupational asthma. J Allergy Clin Immunol. 2001;108:317–328. doi: 10.1067/mai.2001.116432. [DOI] [PubMed] [Google Scholar]
- 13.Clark TJH, Cagnani CB, Bousquet J. 2002. Global initiative for asthma. Global strategy for asthma management and prevention; pp. 1–176. NIH Publication No. 02-3659. [Google Scholar]
- 14.Adams RJ, Fuhlbrigge A, Guilbert T, Lozano P, Martinez F. Inadequate use of asthma medication in the United States: results of the asthma in America national population survey. J Allergy Clin Immunol. 2002;110:58–64. doi: 10.1067/mai.2002.125489. [DOI] [PubMed] [Google Scholar]
- 15.Hessel PA, Mitchell I, Tough S, Green FHY, Cockcroft DW, Kepron W. Risk factors for death from asthma. Ann Allergy Asthma Immunol. 1999;83:362–368. doi: 10.1016/s1081-1206(10)62832-3. [DOI] [PubMed] [Google Scholar]
- 16.Simons FER. Allergic rhinobronchitis: the asthma-allergic rhinitis link. J Allergy Clin Immunol. 1999;104:534–540. doi: 10.1016/s0091-6749(99)70320-9. [DOI] [PubMed] [Google Scholar]
- 17.Duff AL, Platts-Mills TAE. Allergens and asthma. Pediatr Clin North Am. 1992;39:1277–1291. doi: 10.1016/s0031-3955(16)38445-0. [DOI] [PubMed] [Google Scholar]
- 18.Sporik R, Holgate ST, Platts-Mills TA, Cogswell JJ. Exposure to house-dust mite allergen (Der p I) and the development of asthma in childhood: a prospective study. N Engl J Med. 1990;323:502–507. doi: 10.1056/NEJM199008233230802. [DOI] [PubMed] [Google Scholar]
- 19.Rosenstreich DL, Eggleston P, Kattan M, Baker D, Slavin RG, Gergen P. The role of cockroach allergy and exposure to cockroach allergen in causing morbidity among inner-city children with asthma. N Engl J Med. 1997;336:1356–1363. doi: 10.1056/NEJM199705083361904. [DOI] [PubMed] [Google Scholar]
- 20.Halonen M, Stern DA, Lohman C, Wright AL, Brown MA, Martinez FD. Two subphenotypes of childhood asthma that differ in maternal and paternal influences on asthma risk. Am J Respir Crit Care Med. 1999;160:564–570. doi: 10.1164/ajrccm.160.2.9809038. [DOI] [PubMed] [Google Scholar]
- 21.Platts-Mills TA, Vaughan JW, Blumenthal K. Serum IgG and IgG4 antibodies to Fel d 1 among children exposed to 20 microg Fel d 1 at home: relevance of a nonallergic modified TH2 response. Int Arch Allergy Immunol. 2001;124:126–129. doi: 10.1159/000053689. [DOI] [PubMed] [Google Scholar]
- 22.Ownby DR, Johnson CC, Peterson EL. Exposure to dogs and cats in the first year of life and risk of allergic sensitization at 6 to 7 years of age. JAMA. 2002;288:963–972. doi: 10.1001/jama.288.8.963. [DOI] [PubMed] [Google Scholar]
- 23.Platts-Mills TA, Vervloet D, Thomas WR, Aalberse RC, Chapman MD. Indoor allergens and asthma: report of the Third International Workshop. J Allergy Clin Immunol. 1997;100:S2–S24. doi: 10.1016/s0091-6749(97)70292-6. [DOI] [PubMed] [Google Scholar]
- 24.O'Hollaren MT, Yunginger JW, Offard KP. Exposure to an aeroallergen as a possible precipitating factor in respiratory arrest in young patients with asthma. N Engl J Med. 1991;324:359–363. doi: 10.1056/NEJM199102073240602. [DOI] [PubMed] [Google Scholar]
- 25.James JM, Bernhisel-Broadbent J, Sampson HA. Respiratory reactions provoked by double-blind food challenges in children. Am J Respir Crit Care Med. 1994;149:59–64. doi: 10.1164/ajrccm.149.1.8111598. [DOI] [PubMed] [Google Scholar]
- 26.Folkerts G, Busse WW, Nijkamp FP, Sorkness R, Gern JE. Virus-induced airway hyperresponsiveness and asthma. Am J Respir Crit Care Med. 1998;157:1708–1720. doi: 10.1164/ajrccm.157.6.9707163. [DOI] [PubMed] [Google Scholar]
- 27.Lemanske RF, Jr, Lemen RJ, Gern JE. Infections in childhood. In: Barnes PJ, Grunstein MM, Leff AR, editors. Lippincott-Raven Publishers; Philadelphia, Pa: 1997. pp. 1207–1216. (Asthma). [Google Scholar]
- 28.Hahn DL, Dodge RW, Golubjatnikov R. Association of Chlamydia pneumoniae (strain TWAR) infection with wheezing, asthmatic bronchitis, and adult-onset asthma. JAMA. 1991;266:225–230. [PubMed] [Google Scholar]
- 29.Cook PJ, Davies P, Tunnicliffe W, Ayres JG, Honeybourne D, Wise R. Chlamydia pneumoniae and asthma. Thorax. 1998;53:254–259. doi: 10.1136/thx.53.4.254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cunningham AF, Johnston SL, Julious SA, Lampe FC, Ward ME. Chronic Chlamydia pneumoniae infection and asthma exacerbations in children. Eur Respir J. 1998;11:345–349. doi: 10.1183/09031936.98.11020345. [DOI] [PubMed] [Google Scholar]
- 31.Kraft M, Cassell GH, Henson JE, Watson H, Williamson J, Marmion BP. Detection of Mycoplasma pneumoniae in the airways of adults with chronic asthma. Am J Respir Crit Care Med. 1998;158:998–1001. doi: 10.1164/ajrccm.158.3.9711092. [DOI] [PubMed] [Google Scholar]
- 32.Sigurs N, Bjarnason R, Sigurbergsson F, Kjellman B. Respiratory syncytial virus bronchiolitis in infancy is an important risk factor for asthma and allergy at age 7. Am J Respir Crit Care Med. 2000;161:1501–1507. doi: 10.1164/ajrccm.161.5.9906076. [DOI] [PubMed] [Google Scholar]
- 33.Gern JE, Lemanske RF, Jr, Busse WW. Early life origins of asthma. J Clin Invest. 1999;104:837–843. doi: 10.1172/JCI8272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wennergren G, Kristjansson S. Relationship between respiratory syncytial virus bronchiolitis and future obstructive airway diseases. Eur Respir J. 2001;18:1044–1058. doi: 10.1183/09031936.01.00254101. [DOI] [PubMed] [Google Scholar]
- 35.Johnston SL, Pattemore PK, Sanderson G, Smith S, Lampe F, Josephs L. Role of virus infections in children with recurrent wheeze or cough. Thorax. 1993;48:1055–1060. [Google Scholar]
- 36.Johnston SL, Pattemore PK, Sanderson G, Smith S, Lampe F, Josephs L. Community study of role of viral infections in exacerbations of asthma in 9-11 year old children. BMJ. 1995;310:1225–1229. doi: 10.1136/bmj.310.6989.1225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Johnston SL, Pattemore PK, Sanderson G, Smith S, Campbell MJ, Josephs LK. The relationship between upper respiratory infections and hospital admissions for asthma: a time-trend analysis. Am J Respir Crit Care Med. 1996;154:654–660. doi: 10.1164/ajrccm.154.3.8810601. [DOI] [PubMed] [Google Scholar]
- 38.Green RM, Custovic A, Sanderson G. Synergism between allergens and viruses and risk of hospital admission with asthma: case-control study. BMJ. 2002;324:763. doi: 10.1136/bmj.324.7340.763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rakes GP, Arruda E, Ingram JM, Hoover GE, Zambrano JC, Hayden FG. Rhinovirus and respiratory syncytial virus in wheezing children requiring emergency care: IgE and eosinophil analyses. Am J Respir Crit Care Med. 1999;159:785–790. doi: 10.1164/ajrccm.159.3.9801052. [DOI] [PubMed] [Google Scholar]
- 40.Strachan DP. Family size, infection and atopy: the first decade of the “hygiene hypothesis.”. Thorax. 2000;55(Suppl 1):S2–S10. doi: 10.1136/thorax.55.suppl_1.s2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Von Hertzen LC. Role of persistent infection in the control and severity of asthma: focus on Chlamydia pneumoniae. Eur Respir J. 2002;19:546–556. doi: 10.1183/09031936.02.00254402. [DOI] [PubMed] [Google Scholar]
- 42.Martin RJ, Kraft M, Chu HW, Berns EA, Cassell GH. A link between chronic asthma and chronic infection. J Allergy Clin Immunol. 2001;107:595–601. doi: 10.1067/mai.2001.113563. [DOI] [PubMed] [Google Scholar]
- 43.Kraft M, Cassell GH, Pak J, Martin RJ. Mycoplasma pneumoniae and Chlamydia pneumoniae in asthma: effect of clarithromycin. Chest. 2002;121:1782–1788. doi: 10.1378/chest.121.6.1782. [DOI] [PubMed] [Google Scholar]
- 44.Rundell KW, Judelson DA, Williams SD. Diagnosis of exercise-induced asthma in the elite athlete. In: Rundell KW, Wilber RL, Lemanske RF Jr, editors. Human Kinetics Publishers, Inc; Champaign, Ill: 2002. pp. 181–210. (Exercise-induced asthma: Pathophysiology and treatment). [Google Scholar]
- 45.Szczeklik A, Stevenson DD. Aspirin-induced asthma: advances in pathogenesis and management. J Allergy Clin Immunol. 1999;104:5–13. doi: 10.1016/s0091-6749(99)70106-5. [DOI] [PubMed] [Google Scholar]
- 46.Szczeklik A, Nizankowska E, Bochenek G, Nagraba K, Mejza F, Swierczynska M. Safety of a specific COX-2 inhibitor in aspirin-induced asthma. Clin Exp Allergy. 2001;31:219–225. doi: 10.1046/j.1365-2222.2001.01075.x. [DOI] [PubMed] [Google Scholar]
- 47.Martin-Garcia C, Hinojosa M, Berges P, Camacho E, Garcia-Rodriguez R, Alfaya T. Safety of a cyclooxygenase-2 inhibitor in patients with aspirin-sensitive asthma. Chest. 2002;121:1812–1817. doi: 10.1378/chest.121.6.1812. [DOI] [PubMed] [Google Scholar]
- 48.Fischer AR, Rosenberg MA, Lilly CM, Callery JC, Rubin P, Cohn J. Direct evidence for role of the mast cell in the nasal response to aspirin in aspirin-sensitive asthma. J Allergy Clin Immunol. 1994;94:1046–1056. doi: 10.1016/0091-6749(94)90123-6. [DOI] [PubMed] [Google Scholar]
- 49.Sanak M, Simon HU, Szczeklik A. Leukotriene C4 synthase promoter polymorphism and risk of aspirin- induced asthma. Lancet. 1997;350:1599–1600. doi: 10.1016/s0140-6736(05)64015-9. [DOI] [PubMed] [Google Scholar]
- 50.Harding SM. Gastroesophageal reflux and asthma: insight into the association. J Allergy Clin Immunol. 1999;104:251–259. doi: 10.1016/s0091-6749(99)70360-x. [DOI] [PubMed] [Google Scholar]
- 51.Wright RJ, Rodriguez M, Cohen S. Review of psychosocial stress and asthma: an integrated biopsychosocial approach. Thorax. 1998;53:1066–1074. doi: 10.1136/thx.53.12.1066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Murphy S, Bleecker ER, Boushey H, Brown C, Buist AS, Busse W. National Institutes of Health; Bethesda, Md: 1997. Guidelines for the diagnosis and management of asthma; pp. 1–150. National Asthma Education and Prevention Program, editor. II. [Google Scholar]
- 53.Anderson SD, Beck KC, Davis MS, Dempsey JA, Derchak PA, Freed AN. Human Kinetics; Champaign, Ill: 2002. Exercise-induced asthma: Pathophysiology and treatment. [Google Scholar]
- 54.Gershel JC, Goldman HS, Stein REK, Shelov SP, Ziprkowski M. The usefulness of chest radiographs in first asthma attacks. N Engl J Med. 1983;309:336–339. doi: 10.1056/NEJM198308113090603. [DOI] [PubMed] [Google Scholar]
- 55.Castro-Rodríguez JA, Holberg CJ, Wright AL, Martinez FD. A clinical index to define risk of asthma in young children with recurrent wheezing. Am J Respir Crit Care Med. 2000;162:1403–1406. doi: 10.1164/ajrccm.162.4.9912111. [DOI] [PubMed] [Google Scholar]
- 56.Corbridge TC, Hall JB. The assessment and management of adults with status asthmaticus. Am J Respir Crit Care Med. 1995;151:1296–1316. doi: 10.1164/ajrccm.151.5.7735578. [DOI] [PubMed] [Google Scholar]
- 57.Wright AL, Taussig LM, Ray CG, Harrison HR, Holberg CJ. The Tucson Children's Respiratory Study, II: lower respiratory tract illness in the first year of life. Am J Epidemiol. 1989;129:1232–1246. doi: 10.1093/oxfordjournals.aje.a115243. [DOI] [PubMed] [Google Scholar]
- 58.Cypcar D, Busse WW. Role of viral infections in asthma. Immunol Allergy Clin North Am. 1993;13:745–766. [Google Scholar]
- 59.Martinez FD, Wright AL, Taussig LM. Asthma and wheezing in the first six years of life. N Engl J Med. 1995;332:133–138. doi: 10.1056/NEJM199501193320301. [DOI] [PubMed] [Google Scholar]
- 60.Nelson HS. Drug therapy: beta-adrenergic bronchodilators. N Engl J Med. 1995;333:499–506. doi: 10.1056/NEJM199508243330807. [DOI] [PubMed] [Google Scholar]
- 61.Kips JC, Pauwels RA. Long-acting inhaled beta(2)-agonist therapy in asthma. Am J Respir Crit Care Med. 2001;164:923–932. doi: 10.1164/ajrccm.164.6.2010107. [DOI] [PubMed] [Google Scholar]
- 62.Lazarus SC, Boushey HA, Fahy JV, Chinchilli VM, Lemanske RF, Jr, Sorkness CA. Long-acting beta2-agonist monotherapy vs continued therapy with inhaled corticosteroids in patients with persistent asthma: a randomized controlled trial. JAMA. 2001;285:2583–2593. doi: 10.1001/jama.285.20.2583. [DOI] [PubMed] [Google Scholar]
- 63.Greening AP, Wind P, Northfield M, Shaw G. Added salmeterol versus higher-dose corticosteroid in asthma patients with symptoms on existing inhaled corticosteroid. Lancet. 1994;344:219–224. doi: 10.1016/s0140-6736(94)92996-3. [DOI] [PubMed] [Google Scholar]
- 64.Woolcock A, Lundback B, Ringdal N, Jacques LA. Comparison of addition of salmeterol to inhaled steroids with doubling of the dose of inhaled steroids. Am J Respir Crit Care Med. 1996;153:1481–1488. doi: 10.1164/ajrccm.153.5.8630590. [DOI] [PubMed] [Google Scholar]
- 65.Pauwels RA, Löfdahl CG, Postma DS, Tattersfield AE, O'Byrne P, Barnes PJ. Effect of inhaled formoterol and budesonide on exacerbations of asthma. N Engl J Med. 1997;337:1405–1411. doi: 10.1056/NEJM199711133372001. [DOI] [PubMed] [Google Scholar]
- 66.Lemanske RF, Jr, Sorkness CA, Mauger EA. Inhaled corticosteroid reduction and elimination in patients with persistent asthma receiving salmeterol: a randomized controlled trial. JAMA. 2001;285:2594–2603. doi: 10.1001/jama.285.20.2594. [DOI] [PubMed] [Google Scholar]
- 67.Jenne JW. The debate on S-enantiomers of b-agonists: tempest in a teapot or gathering storm? J Allergy Clin Immunol. 1998;102:893–895. doi: 10.1016/s0091-6749(98)70324-0. [DOI] [PubMed] [Google Scholar]
- 68.Asmus MJ, Hendeles L, Weinberger M, Ahrens RC, Bisgaard H, Lotvall J. Levalbuterol has not been established to have therapeutic advantage over racemic albuterol. J Allergy Clin Immunol. 2002;110:325. doi: 10.1067/mai.2002.126375. [DOI] [PubMed] [Google Scholar]
- 69.Baramki D, Koester J, Anderson AJ, Borish L. Modulation of T-cell function by (R)- and (S)-isomers of albuterol: anti-inflammatory influences of (R)-isomers are negated in the presence of the (S)-isomer. J Allergy Clin Immunol. 2002;109:449–454. doi: 10.1067/mai.2002.122159. [DOI] [PubMed] [Google Scholar]
- 70.Chowdhury BA. Comparative efficacy of levalbuterol and racemic albuterol in the treatment of asthma. J Allergy Clin Immunol. 2002;110:324. doi: 10.1067/mai.2002.126374. [DOI] [PubMed] [Google Scholar]
- 71.Ahrens R, Weinberger M. Levalbuterol and racemic albuterol: are there therapeutic differences? J Allergy Clin Immunol. 2001;108:681–684. doi: 10.1067/mai.2001.119407. [DOI] [PubMed] [Google Scholar]
- 72.Lotvall J, Palmqvist M, Arvidsson P, Maloney A, Ventresca GP, Ward J. The therapeutic ratio of R-albuterol is comparable with that of RSalbuterol in asthmatic patients. J Allergy Clin Immunol. 2001;108:726–731. doi: 10.1067/mai.2001.119152. [DOI] [PubMed] [Google Scholar]
- 73.Weinberger M, Hendeles L. Theophylline in asthma. N Engl J Med. 1996;334:1380–1394. doi: 10.1056/NEJM199605233342107. [DOI] [PubMed] [Google Scholar]
- 74.Tinkelman DG, Reed CE, Nelson HS, Offord KP. Aerosol beclomethasone dipropionate compared with theophylline as primary treatment of chronic, mild to moderately severe asthma in children. Pediatrics. 1993;92:64–77. [PubMed] [Google Scholar]
- 75.Reed CE, Offord KP, Nelson HS, Li JT, Tinkelman DG, American Academy of Allergy AaIBD-TSG Aerosol beclomethasone dipropionate spray compared with theophylline as primary treatment for chronic mild-to-moderate asthma. J Allergy Clin Immunol. 1998;101:14–23. doi: 10.1016/s0091-6749(98)70187-3. [DOI] [PubMed] [Google Scholar]
- 76.Davies B, Brooks G, Devoy M. The efficacy and safety of salmeterol compared to theophylline: meta-analysis of nine controlled studies. Respir Med. 1998;92:256–263. doi: 10.1016/s0954-6111(98)90105-6. [DOI] [PubMed] [Google Scholar]
- 77.Hoag JE, McFadden ER., Jr Long-term effect of cromolyn sodium on nonspecific bronchial hyperresponsiveness: a review. Ann Allergy. 1991;66:53–63. [PubMed] [Google Scholar]
- 78.Schwartz HJ, Blumenthal M, Brady R, Braun S, Lockey R, Myers D. A comparative study of the clinical efficacy of nedocromil sodium and placebo: how does cromolyn sodium compare as an active control treatment? Chest. 1996;109:945–952. doi: 10.1378/chest.109.4.945. [DOI] [PubMed] [Google Scholar]
- 79.Alton EWFW, Kingsleigh-Smith DJ, Munkonge FM, Smith SN, Lindsay ARG, Gruenert DC. Asthma prophylaxis agents alter the function of an airway epithelial chloride channel. Am J Respir Cell Mol Biol. 1996;14:380–387. doi: 10.1165/ajrcmb.14.4.8600943. [DOI] [PubMed] [Google Scholar]
- 80.Bel EH, Timmers MC, Hermans J, Dijkman JH, Sterk RJ. The long-term effects of nedocromil sodium and beclomethasone dipropionate on bronchial responsiveness to methacholine in nonatopic asthmatic subjects. Am Rev Respir Dis. 1990;141:21–28. doi: 10.1164/ajrccm/141.1.21. [DOI] [PubMed] [Google Scholar]
- 81.Shapiro GG, Sharpe M, DeRouen TA, Pierson WE, Furukawa CT, Virant FS. Cromolyn versus triamcinalone acetonide for youngsters with moderate asthma. J Allergy Clin Immunol. 1991;88:742–748. doi: 10.1016/0091-6749(91)90181-m. [DOI] [PubMed] [Google Scholar]
- 82.Kelly K, Spooner CH, Rowe BH. Nedocromil sodium vs. sodium cromoglycate for preventing exercise- induced bronchoconstriction in asthmatics. Cochrane Database Syst Rev. 2000;4:CD002731. doi: 10.1002/14651858.CD002731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Childhood Asthma Management Program Research Group Long-term effects of budesonide or nedocromil in children with asthma. N Engl J Med. 2000;343:1054–1063. doi: 10.1056/NEJM200010123431501. [DOI] [PubMed] [Google Scholar]
- 84.Horwitz RJ, McGill KA, Busse WW. The role of leukotriene modifiers in the treatment of asthma. Am J Respir Crit Care Med. 1998;157:1363–1371. doi: 10.1164/ajrccm.157.5.9706059. [DOI] [PubMed] [Google Scholar]
- 85.Drazen JM, Israel E, O'Byrne PM. Treatment of asthma with drugs modifying the leukotriene pathway. N Engl J Med. 1999;340:197–206. doi: 10.1056/NEJM199901213400306. [DOI] [PubMed] [Google Scholar]
- 86.Barnes PJ. Mechanisms of action of glucocorticoids in asthma. Am J Respir Crit Care Med. 1996;154:521–527. doi: 10.1164/ajrccm/154.2_Pt_2.S21. [DOI] [PubMed] [Google Scholar]
- 87.Allen DB. Safety of inhaled corticosteroids in children. Pediatr Pulmonol. 2002;33:208–220. doi: 10.1002/ppul.10040. [DOI] [PubMed] [Google Scholar]
- 88.Agertoft L, Pedersen S. Effect of long-term treatment with inhaled budesonide on adult height in children with asthma. N Engl J Med. 2000;343:1064–1069. doi: 10.1056/NEJM200010123431502. [DOI] [PubMed] [Google Scholar]
- 89.Keogh KA, Macarthur C, Parkin PC, Stephens D, Arseneault R, Tennis O. Predictors of hospitalization in children with acute asthma. J Pediatr. 2001;139:273–277. doi: 10.1067/mpd.2001.116282. [DOI] [PubMed] [Google Scholar]
- 90.Cates CJ, Rowe BH, Bara A. Holding chambers versus nebulisers for beta-agonist treatment of acute asthma. Cochrane Database Syst Rev. 2002;2:CD000052. doi: 10.1002/14651858.CD000052. [DOI] [PubMed] [Google Scholar]
- 91.Reisner C, Kotch A, Dworkin G. Continuous versus frequent intermittent nebulization of albuterol in acute asthma: a randomized, prospective study. Ann Allergy Asthma Immunol. 1995;75:41–47. [PubMed] [Google Scholar]
- 92.Rodrigo GJ, Rodrigo C. The role of anticholinergics in acute asthma treatment: an evidence-based evaluation. Chest. 2002;121:1977–1987. doi: 10.1378/chest.121.6.1977. [DOI] [PubMed] [Google Scholar]
- 93.Parameswaran K, Belda J, Rowe BH. Addition of intravenous aminophylline to beta2-agonists in adults with acute asthma. Cochrane Database Syst Rev. 2000;4:CD002742. doi: 10.1002/14651858.CD002742. [DOI] [PubMed] [Google Scholar]
- 94.Volovitz B, Nussinovitch M, Finkelstein Y, Harel L, Varsano I. Effectiveness of inhaled corticosteroids in controlling acute asthma exacerbations in children at home. Clin Pediatr (Phila) 2001;40:79–86. doi: 10.1177/000992280104000203. [DOI] [PubMed] [Google Scholar]
- 95.Edmonds ML, Camargo CA, Pollack CV, Rowe BH. Early use of inhaled corticosteroids in the emergency department treatment of acute asthma. Cochrane Database Syst Rev. 2000;3:CD002308. doi: 10.1002/14651858.CD002308. [DOI] [PubMed] [Google Scholar]
- 96.Manser R, Reid D, Abramson M. Corticosteroids for acute severe asthma in hospitalised patients. Cochrane Database Syst Rev. 2000;2:CD001740. doi: 10.1002/14651858.CD001740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Rowe BH, Spooner C, Ducharme FM, Bretzlaff JA, Bota GW. Early emergency department treatment of acute asthma with systemic corticosteroids. Cochrane Database Syst Rev. 2000;2:CD002178. doi: 10.1002/14651858.CD002178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.O'Driscoll BR, Kalra S, Wilson M, Pickering CA, Carroll KB, Woodcock AA. Double-blind trial of steroid tapering in acute asthma. Lancet. 1993;341:324–327. doi: 10.1016/0140-6736(93)90134-3. [DOI] [PubMed] [Google Scholar]
- 99.Lederle FA, Pluhar RE, Joseph AM, Niewoehner DE. Tapering of corticosteroid therapy following exacerbation of asthma: a randomized, double-blind, placebo-controlled trial. Arch Intern Med. 1987;147:2201–2203. [PubMed] [Google Scholar]


