Abstract
Acute renal impairment in coronavirus-associated severe acute respiratory syndrome.
Background
Severe acute respiratory syndrome (SARS) is a newly emerged infection from a novel coronavirus (SARS-CoV). Apart from fever and respiratory complications, acute renal impairment has been observed in some patients with SARS. Herein, we describe the clinical, pathologic, and laboratory features of the acute renal impairment complicating this new viral infection.
Methods
We conducted a retrospective analysis of the plasma creatinine concentration and other clinical parameters of the 536 SARS patients with normal plasma creatinine at first clinical presentation, admitted to two regional hospitals following a major outbreak in Hong Kong in March 2003. Kidney tissues from seven other patients with postmortem examinations were studied by light microscopy and electron microscopy.
Results
Among these 536 patients with SARS, 36 (6.7%) developed acute renal impairment occurring at a median duration of 20 days (range 5–48 days) after the onset of viral infection despite a normal plasma creatinine level at first clinical presentation. The acute renal impairment reflected the different prerenal and renal factors that exerted renal insult occurring in the context of multiorgan failure. Eventually, 33 SARS patients (91.7%) with acute renal impairment died. The mortality rate was significantly higher among patients with SARS and acute renal impairment compared with those with SARS and no renal impairment (91.7% vs. 8.8%) (P < 0.0001). Renal tissues revealed predominantly acute tubular necrosis with no evidence of glomerular pathology. The adjusted relative risk of mortality associated with the development of acute renal impairment was 4.057 (P < 0.001). By multivariate analysis, acute respiratory distress syndrome and age were the most significant independent risk factors predicting the development of acute renal impairment in SARS.
Conclusion
Acute renal impairment is uncommon in SARS but carries a high mortality. The acute renal impairment is likely to be related to multi-organ failure rather than the kidney tropism of the virus. The development of acute renal impairment is an important negative prognostic indicator for survival with SARS.
Keywords: severe acute respiratory syndrome, coronavirus, acute renal failure, acute tubular necrosis, acute respiratory distress syndrome, mortality
A novel mysterious pneumonia appeared in the Guangdong Province of China in November 2002. It did not receive much attention until widespread outbreak occurred, first in southern Asia and then across the world. In March 2003, the World Health Organization (WHO) coined the new disease as severe acute respiratory syndrome (SARS). SARS is a highly contagious and predominantly pneumonic illness caused by a novel coronavirus (CoV) now commonly known as SARS-CoV1 , 2. Since its recognition in March 2003, SARS has spread to 30 countries and has infected over 8000 people, resulting in almost 700 deaths worldwide3. In Hong Kong, 1755 subjects were infected and 299 died. Over 25% of the victims were health care workers, and the fear of this disease had created devastating socioeconomic effects in Hong Kong and worldwide.
Although acute respiratory failure due to diffuse alveolar damage was associated with the highest mortality4, lungs were not the only organs involved. Severe diarrhea and hepatic dysfunction were the presenting symptoms in some patients with SARS5. Among patients with confirmed CoV-associated SARS, we encountered a significant number of patients with acute renal dysfunction. Some even progressed to renal failure and required dialysis. Herein, we describe the demographic, clinical, laboratory, and radiologic features of this SARS cohort with acute renal impairment [acute renal failure (ARF)] and study any risk factor associated with the development of ARF.
METHODS
Patients and data collection
The analysis included all confirmed SARS cases with normal plasma creatinine (normal range 60 to 120 μmol/L) at first clinical presentation, admitted to the Princess Margaret Hospital and the Queen Mary Hospital, Hong Kong, between March 1 and May 31, 2003. SARS was initially diagnosed based on criteria issued by the WHO6. The hospital records were reviewed following approval of the institutional ethical committee. Additional radiologic and laboratory information were retrieved from the hospital computerized Clinical Management System. Demographic data and symptom profiles were recorded in all patients by a standard form on admission. Chest radiograph was mandatory with supplementary chest computerized-axial tomography (CT) when clinically indicated. Blood pressure, temperature, and pulse rate were monitored regularly after admission. Initial laboratory investigations included complete blood count, arterial blood gas, lactate dehydrogenase (LDH), creatine phosphokinase, liver, and renal function tests. Serial monitoring of the laboratory profile was performed according to the clinical progress of individual patient. Chest radiograph was taken on alternate days. Either nasopharyngeal aspirates or nasal/throat swabs were taken for CoV testing by polymerase chain reaction (PCR). Paired sera were collected on admission and on day 14 for determination of a fourfold rise of IgG anti-CoV antibodies.
Definitions
Acute renal impairment was defined as an elevation of plasma creatinine by at least 30% of the baseline value on admission (documented by three consecutive blood samplings) or above 160 μmol/L following a normal plasma creatinine on admission. Acute respiratory distress syndrome (ARDS) was defined according to the criteria of the American-European Consensus Conference as an illness characterized by impaired oxygenation, with a ratio of the partial pressure of arterial oxygen (PaO2) to the fraction of inspired oxygen (FiO2) (≤200 mm Hg), regardless of whether or how much positive end-expiratory pressure is used to support, and the presence of bilateral infiltrates on chest radiographs and pulmonary artery occlusion pressure ≤18 mm Hg with no clinical evidence of left atrial hypertension7.
Dialysis treatment
For those patients who required subsequent dialysis treatment for ARF, either peritoneal dialysis or hemofiltration was performed depending on the availability of equipment. Indications for dialysis included increased uremia, electrolyte and acid-base imbalance, and fluid retention. Peritoneal dialysis was administered using an acute peritoneal catheter. Hourly 1 L exchange of peritoneal dialysate was performed manually. For blood purification, pump-driven continuous venovenous hemofiltration was used. The replacement solution contained self-admixtured sodium bicarbonate and sodium chloride solution. Citrate was used for anticoagulation. Calcium chloride was infused through another central line. The hourly ultrafiltration rate was set at 1500 mL and the net ultrafiltration was adjusted by titrating the volume of saline replacement administered via a separate line.
Renal specimen
Due to the special infection-control precaution of handling deceased subjects with SARS, postmortem examination was not performed routinely. Postmortem was performed in a designated pathology laboratory. Kidney specimens from autopsy of seven other SARS subjects (not from the above study group) were retrieved for standard examination of light microscopy and electron microscopy.
Kidney tissues were further studied by in situ hybridization for detection of SARS-CoV using techniques described8. Briefly, the probe was labeled with digoxigenin by nick translation (DIG-Nick Translation Mix) (Roche, Indianapolis, IN, USA). Cell-block preparations from Vero cell culture with and without inoculation with SARS-CoV were used as positive and negative controls, respectively. The signal specificity was also assessed by (1) digestion of the sections with RNase A, (2) omission of the probes in the hybridization mixture, and (3) tissue sections from three autopsy cases of non-SARS patients were included as negative controls.
Statistics
We compared risk factors associated with ARF and normal renal function with the χ2 or Fisher's exact test for categorical variables. Laboratory data were expressed as median and range and analyzed with Mann-Whitney test. Cox proportional hazards model was used in the calculation of hazard ratio (relative risk) of mortality and development of ARF for each risk factor. Significant risk factors identified by univariate analyses were further analyzed in multivariate model to identify independent risk factors associated with the development of ARF. Due to a relatively smaller population of SARS patients with ARF as compared with those with normal renal function, risk factors with P value less than 0.01, accompanied with 99% confidence interval, was chosen as statistically significant level unless specified otherwise. The least squares mean plasma creatinine and the time for doubling the plasma creatinine concentration in different groups of SARS with ARF were calculated from prediction polynomial equations of mixed linear model. We used the standard SPSS 11.0 statistical package for all analyses.
RESULTS
Patient demographic data
During the study period, 544 patients with confirmed SARS were admitted to the Princess Margaret and Queen Mary Hospitals. Eight patients were excluded from the study as they had chronic renal impairment due to chronic glomerulonephritis or diabetes nephropathy. Among the remaining 536 patients, thirty-six patients (25 males and 11 females) (6.7%) with a median age of 53.5 years (34 to 77) were subsequently found to have elevated plasma creatinine levels during the later clinical course. The median time from the onset of viral infection to the development of ARF in these 36 patients was 20 days (range, 5 to 48 days). The peak plasma creatinine levels ranged from 136 to 1083 μmol/L, with a median value of 274 μmol/L. Fourteen of these 36 patients (39%) had plasma bicarbonate lower than 24 mmol/L (normal 24 to 26 mmol/L) prior to or at the time of detection of ARF. Oliguria (urine output <500 mL/day) occurred in half of these 36 patients. Those patients with ARF were older (P <0.001) and had higher systolic blood pressure (P = 0.005) than other SARS patients with no ARF. Comorbidity factors included hypertension (10 patients), diabetes mellitus (5 patients), ischemic heart disease (2 patients), hepatitis B–associated chronic hepatitis (one patient), bronchiectasis (one patient), stroke (one patient), hypertrophic obstructive cardiomyopathy (one patient), and sinoatrial node dysfunction (one patient).
Most of these 36 patients had fever on presentation (97.2%). High temperature (≥39°C) was observed in 22.2% of patients. Other common complaints included palpitation (86.1%), chills (63.8%), malaise (66.7%), cough (44.4%), dyspnoea (41.6%), anorexia (33.3%), and myalgia (30.6%). Rigor only occurred in ten patients (27.8%). Cough was usually nonproductive and only nine patients (25%) had production of sputum. Other notable symptoms included headache (19.4%), dizziness (13.8%), and diarrhea (13.8%). The clinical presentations were similar to previous described9. The mean systolic and diastolic blood pressures on admission blood pressure were 136 mm Hg and 72.5 mm Hg, respectively. Elevated systolic (>140 mm Hg) and diastolic (>90 mm Hg) blood pressures were found in 12 (33.3%) and 3 (8.3%) patients, respectively. Five patients had systolic blood pressure exceeding 160 mm Hg.
Laboratory findings
Semiquantitative urinalysis was performed in 26 patients when they developed ARF. Proteinuria by dipstick was found in 22 patients (84.6%). Heavy proteinuria was not a common finding (trace in 11 patients, + in seven patients, ++ in two patients, and +++ in three patients). The laboratory profiles on admission are shown in Table 1 . Notably, hyponatremia (sodium < 135 mmol/L) was found in 28 patients (77.7%). Absolute neutropenia was only found in four patients (11.1%) yet the majority of the patients had lymphopenia (75.0%). In these 36 patients, the nadir hemoglobin and nadir leukocyte counts fell to 8.7 g/dL (3.4 to 15.5) and 5.35 × 109/L (1.8 to 10.5), respectively. The nadir absolute neutrophil and lymphocyte counts dropped to 3.95 × 109/L (0.8 to 9.9) and 0.3 × 109/L (0.1 to 0.7), respectively. At clinical presentation, all patients had radiographic abnormalities on chest radiograph. Among the 36 patients with ARF, the right lung involvement was more prevalent than the left lung (41.7% vs. 13.9%), which was similar to the patients with normal renal function (47.8% vs. 26.6%) (P = 0.332). However, the remaining patients with ARF (44.4%) had significantly higher bilateral and diffuse infiltrations compared with SARS patients with normal renal function (21.0%) (P = 0.002). Nasopharyngeal aspirates or nasal/throat swabs were positive for CoV by PCR analysis in 19 patients (52.7%) and another six patients (16.7%) had detected IgG against CoV. SARS-CoV infection was confirmed in the remaining 11 patients by subsequent fourfold rise in antibody titer.
Table 1.
Various laboratory and clinical parameters of patients with acute renal impairment and normal kidney function
| Variable | Renal impairment (N = 36) | Normal renal function (N = 500) | P value |
|---|---|---|---|
| Age years | 53.5 (34–77) | 38.0 (18–96) | <0.001 |
| Systolic blood pressure mm Hg | 130.5 (105–200) | 125.0 (96–210) | 0.013 |
| Diastolic blood pressure mm Hg | 70.0 (54–100) | 70.0 (40–109) | 0.942 |
| Hemoglobin g/dLa | 14.0 (8.4–16.9) | 13.2 (7.6–18) | 0.035 |
| White blood cells (×109/L)a | 6.5 (1.8–16.3) | 5.30 (1.55–27.3) | 0.005 |
| Neutrophil (×109/L)a | 5.00 (0.8–15.5) | 3.90 (0.7–26.3) | 0.004 |
| Lymphocyte (×109/L)a | 0.80 (0.3–2.8) | 0.80 (0.2–3.1) | 0.556 |
| Platelet (×109/L)a | 151.0 (79–285) | 163.0 (41–893) | 0.671 |
| Prothrombin time seconda | 11.9 (10.2–14.1) | 12.0 (0.4–120) | 0.391 |
| Activated partial thromboplastin time seconda | 33.2 (23.3–87) | 32.2 (14–120) | 0.092 |
| Plasma sodium mmol/La | 132.0 (126–140) | 134.0 (121–144) | 0.001 |
| Plasma potassium mmol/La | 3.7 (2.8–6.1) | 3.6 (2.2–8.1) | 0.634 |
| Plasma urea mmol/La | 4.6 (1.8–16) | 3.4 (0.3–18.6) | <0.001 |
| Plasma creatinine μmol/La | 93.5 (48–128) | 70.0 (40–123) | <0.001 |
| Plasma albumin g/La | 34.0 (23–44) | 38.0 (20–50) | <0.001 |
| Plasma alanine aminotranferase U/La | 49.0 (12–193) | 26.0 (3–587) | 0.001 |
| Plasma lactate dehydrogenase U/La | 311.0 (148–990) | 218.5 (30–1669) | <0.001 |
| Nadir hemoglobin g/dL | 8.7 (3.4–15.5) | 11.3 (3.9–15.8) | <0.001 |
| Nadir white blood cells (×109/L) | 5.35 (1.8–10.5) | 4.00 (0.8–20.5) | 0.003 |
| Nadir lymphocyte (×109/L) | 0.30 (0.1–0.7) | 0.40 (0–7.3) | <0.001 |
| Plasma peak creatine phosphokinase U/L | 222.0 (26–22974) | 96.0 (11–7154) | <0.001 |
| Symptomatic days before admission | 1.0 (1–3) | 3.0 (1–15) | 0.071 |
Measurement at first admission.
When compared with those with normal renal function during the course of SARS, patients who subsequently developed ARF had lower plasma sodium (P = 0.001) and albumin (P < 0.001), elevated alanine aminotransferase (P = 0.001), and raised plasma LDH on admission (P < 0.001). On the other hand, diastolic blood pressure, hemoglobin on admission, lymphocyte count on admission, platelet count on admission, prothrombin time, activated partial thromboplastin time, plasma potassium, and symptomatic days before admission did not differ between two groups of patients. Patients who subsequently developed ARF had higher peak plasma creatine phosphokinase concentration after admission (P < 0.001).
Plasma creatinine concentration
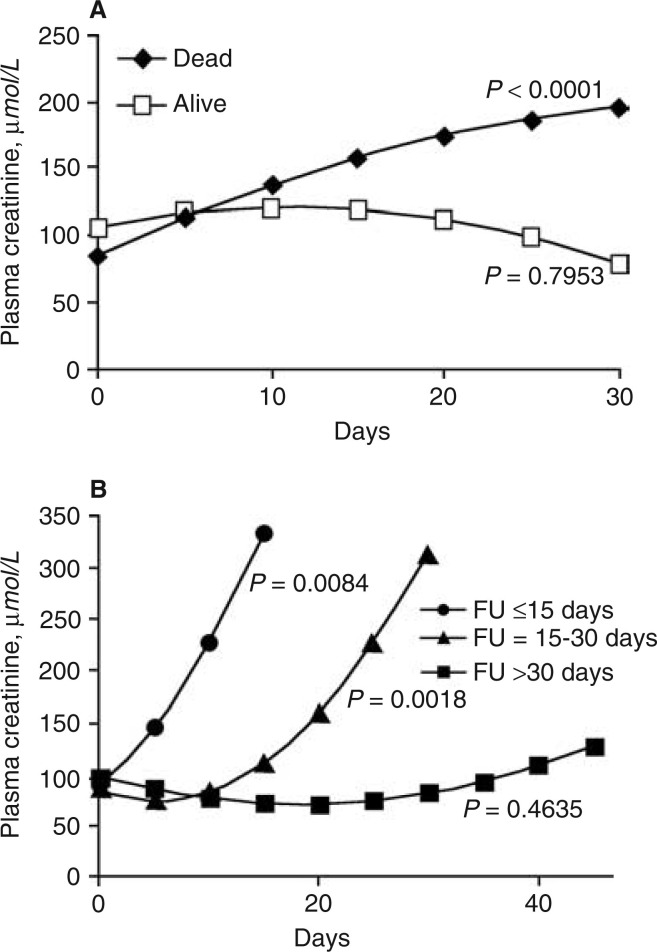
The serial median plasma creatinine of these 36 SARS patients with ARF is shown in Figure 1 . The median duration from the onset of viral infection to the development of ARF was 20 days (range, 5 to 48 days). The least squares mean creatinine value over time for these 36 SARS patients with ARF is shown in Figure 2a . Patients who subsequent died (N = 33) had a progressive rise of plasma creatinine and the doubling time for the plasma creatinine value at admission was calculated to be 18.3 days, using prediction equation of mixed linear model. In contrast, those who were alive (N = 3) had a predicted fall of plasma creatinine from day 10 after admission. Analysis of the serial measurements of plasma creatinine among those 33 deceased patients is depicted in Figure 2b. The rise of plasma creatinine was rapid in those who succumbed early in their illness. Using a prediction equation of mixed linear model, the average times to double the plasma creatinine value on admission for those survived for less than 15 days and those survived between 16 and 30 days after admission were 7.2 days and 20.6 days, respectively. Patients with ARF who succumbed within 15 days after hospitalization had lower plasma albumin (P < 0.01), higher plasma LDH (P < 0.01), and higher plasma creatine phosphokinase (P < 0.01) on admission than those with ARF who succumbed only after day 15 from initial hospitalization.
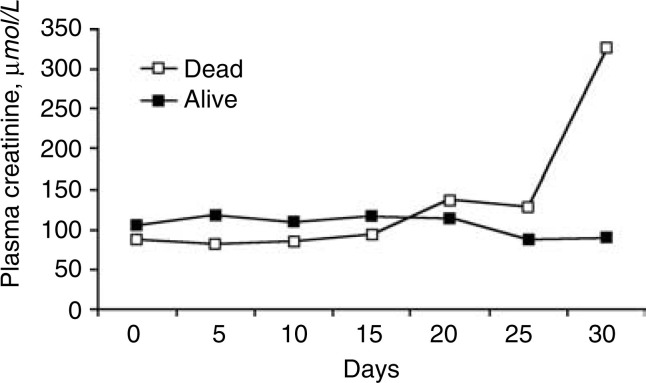
Figure 1.
Serial measurement of plasma creatinine (expressed as median) of these 36 severe acute respiratory syndrome (SARS) patients complicated with acute renal failure (ARF).
Figure 2.
Plasma creatinine concentrations. (A) The least squares mean plasma creatinine concentration over time for alive patients (N = 3) and deceased patients (N = 33) deducted from polynomial equations of mixed linear model. (B) The least squares mean plasma creatinine concentration deducted from polynomial equations of mixed linear model for patients who died within 15 days (N = 12), within 15 to 30 days (N = 19), and after 30 days (N = 2). FU is the duration of hospitalization.
Treatment protocol and other nonrenal complications
On admission, all patients were initially treated with broad-spectrum antibiotics, including intravenous cefepime (2 g three times a day) and oral clarithromycin (500 mg twice a day) for 3 to 5 days to target bacterial pathogens incriminated in the etiology of severe community-acquired pneumonia. Subsequently, 34 patients (94.4%) received intravenous ribavirin according to a standard regime10. Corticosteroids were administered in 34 patients (94.4%) when the chest radiograph showed increasing infiltrates or the blood gases revealed mild desaturation. The majority of them (31/36) also received pulse dose of methylprednisolone. Percentage of patients receiving ribavirin and corticosteroids did not differ between patients with and without ARF (92% and 91.2%, respectively).
Seventy-two (13.4%) of our 536 SARS patients developed ARDS. Thirty-three (45.8%) of these 72 patients also developed ARF. In contrast, only three (0.65%) of the 464 SARS patients with no ARDS developed ARF (Fisher's exact test, P < 0.0001). Of the SARS patients with ARF, 11.4% had raised plasma creatine phosphokinase (>1500 U/L). The percentage was significantly higher than that of SARS patients with no ARF (1.1%) (Fisher's exact test, P = 0.002).
All 36 patients with ARF were cared in the intensive care unit. Indications for admission included fluid retention, oxygen desaturation, or rising plasma creatinine. Eighteen patients had profound hypotension necessitating inotropic support. Ten patients received acute supportive dialysis: four by continuous hemofiltration and the remaining six by peritoneal dialysis. The remaining 26 patients developed ARF late in their illness with established multiorgan failure, including ARDS. Dialysis was not performed in these patients in view of severe multiorgan failure. Eventually, 33 SARS patients (91.7%) with ARF died. The mortality rate was significantly higher among patients with SARS and ARF compared with those with SARS and no renal impairment (91.7% vs. 8.8%) (P < 0.0001).
Relative risk factors of mortality or development of ARF in SARS
On univariate analysis, the risk factors associated with mortality were age (P < 0001), systolic blood pressure (P = 0.032), high leukocyte count on admission (P = 0.029), low nadir leukocyte count (P < 0.001), low nadir lymphocyte count (P < 0.001), low plasma albumin on admission (P < 0.001), raised alanine aminotransferase (P = 0.004), the peak serum creatinine kinase phosphatase (P < 0.001), use of inotropes (P < 0.001), ARDS (P < 0.001), and ARF (P < 0.001) Table 2 . Table 3 summarizes independent risk factors predicting mortality in SARS by multivariate analysis. On multivariate analysis, only age, plasma albumin on admission, ARDS, and the development of ARF were significant risk factors. The adjusted relative risks of mortality associated with the development of ARDS and ARF were 3.286 (P = 0.004) and 4.057 (P < 0.001), respectively. A moderate correlation was found between the development of ARDS and ARF in the multivariate model for predicting mortality in SARS (correlation coefficient r = 0.61, P < 0.001).
Table 2.
Risk factors of mortality in severe acute respiratory syndrome (SARS) by univariate analysis
| Relative risk (99% CI) | P value | |
|---|---|---|
| Use of inotropes | 15.77 (7.539–32.99) | <0.001 |
| Acute renal impairment | 16.91 (8.368–34.16) | <0.001 |
| Acute respiratory distress syndrome | 10.28 (4.854–21.77) | <0.001 |
| Systolic blood pressure | 1.014 (0.997–1.031) | 0.032 |
| White blood cells | 1.066 (0.988–1.150) | 0.029 |
| Lymphocyte count | 0.529 (0.209–1.339) | 0.077 |
| Nadir white blood cells | 1.160 (1.057–1.272) | <0.001 |
| Nadir lymphocyte count | 0.016 (0.001–0.217) | <0.001 |
| Plasma albumin on admission | 0.890 (0.841–0.942) | <0.001 |
| Peak plasma creatine phosphokinase > 1500a | 6.689 (1.699–26.34) | <0.001 |
| Plasma alanine aminotransferase on admission | 1.005 (1.001–1.009) | 0.004 |
| Age | 1.049 (1.029–1.068) | <0.001 |
Patients with creatine phosphokinase less than 1500 serve as the reference group.
Table 3.
Independent risk factor predicting mortality in severe acute respiratory syndrome (SARS) by multivariate analysis
| Adjusted relative risk (99% CI) | P value | |
|---|---|---|
| Acute renal impairment | 4.057 (1.461–11.27) | <0.001 |
| Acute respiratory distress syndrome | 3.286 (1.141–9.463) | 0.004 |
| Age | 1.033 (1.008–1.058) | 0.001 |
| Plasma albumin on admission | 0.936 (0.876–0.999) | 0.009 |
On univariate analysis, the risk factors associated with the development of ARF in SARS patients were age (P < 0.001), systolic blood pressure (P = 0.032), low nadir leukocyte count (P = 0.029), low nadir lymphocyte count (P = 0.006), low plasma albumin on admission (P < 0.001), raised alanine aminotransferase (P = 0.004), the peak serum creatinine kinase phosphatase (P < 0.001), the use of inotropes (P < 0.001), and the complication of ARDS (P < 0.001) Table 4 . On multivariate analysis, ARDS was the most significant independent risk factor predicting the development of ARF in SARS (adjusted relative risk 37.91 (99% CI 7.969 to 180.4) (P < 0.001) Table 5 . As a strong correlation was found between the use of inotropes and ARDS, it was not included in the multivariate model to avoid imprecise estimation of relative risk (correlation coefficient 0.79, P < 0.0001).
Table 4.
Risk factors of the development of acute renal impairment in severe acute respiratory syndrome (SARS) by univariate analysis
| Relative risk (99% CI) | P value | |
|---|---|---|
| Use of inotropes | 124.2 (19.01–811.4) | <0.001 |
| Acute respiratory distress syndrome | 46.60 (9.800–221.6) | <0.001 |
| Systolic blood pressure | 1.017 (0.997–1.038) | 0.032 |
| White blood cells | 1.047 (0.943–1.164) | 0.257 |
| Lymphocyte count | 0.717 (0.233–2.205) | 0.446 |
| Nadir white blood cells | 1.115 (0.980–1.267) | 0.029 |
| Nadir lymphocyte count | 0.039 (0.002–0.812) | 0.006 |
| Plasma albumin on admission | 0.904 (0.842–0.971) | <0.001 |
| Peak plasma creatine phosphokinase > 1500a | 12.65 (3.039–52.64) | <0.001 |
| Plasma alanine transferase on admission | 1.005 (1.001–1.010) | 0.004 |
| Age | 1.044 (1.019–1.069) | <0.001 |
Patients with creatine phosphokinase less than 1500 serve as the reference group.
Table 5.
Independent risk factor predicting the development of acute renal impairment in severe acute respiratory syndrome (SARS) by multivariate analysis
| Adjusted relative risk (99% CI) | P value | |
|---|---|---|
| Acute respiratory distress syndrome | 37.91 (7.969–180.4) | <0.001 |
| Age | 1.037 (1.007–1.067) | 0.001 |
Renal pathology finding
The pathological findings of kidney specimens from autopsy in another seven SARS subjects are summarized in Table 6 . Four patients had ARF despite a normal plasma creatinine concentration on first clinical presentation. Under light microscopy, some degree of benign hypertensive nephrosclerosis and autolysis were noted in most cases. However, glomerular pathology was not observed in the kidneys. Acute tubular necrosis of varying degrees was found in all seven renal specimens. In case 3 with comorbid disease of myelodysplastic syndrome, abnormal myeloid infiltrate was noted in the kidney. No interstitial infiltrate or interstitial nephritis was detected in all other cases. Viral inclusion was not evident. Careful ultrastructural examination of both glomerular and tubular epithelial cells failed to reveal virus or viral-like particles. Immunofluorescence studies on frozen sections were not performed because of high infectivity, but no electron-dense immune deposits were detected with ultrastructural examination. In situ hybridization failed to demonstrate SARS-CoV in these seven kidney samples. All seven cases demonstrated diffuse alveolar damage in their lungs. Kidney specimens from the four subjects who subsequently developed significant impairment of renal function showed moderate to severe acute tubular necrosis.
Table 6.
Clinical and pathologic findings of seven severe acute respiratory syndrome (SARS) patients undergoing postmortem examination
| Case number | Gender/age | SARS confirmed by polymerase chain reaction | Kidney confirmed by light microscopy | Benign nephrosclerosis | Viral particles by electron microscopy | Autolysis | Plasma creatinine at presentation μmol/L | Plasma creatinine at the time of death μmol/L | Postmortem cause of death |
|---|---|---|---|---|---|---|---|---|---|
| 1 | M/69 | Yes | Moderate acute tubular necrosis | + | Not detected | Yes | 100 | 258 | Diffuse alveolar damage |
| 2 | M/64 | Yes | Moderate acute tubular necrosis | + | Not detected | Yes | 88 | 209 | Diffuse alveolar damage |
| 3a | M/76 | Yes | Severe acute tubular necrosis | + | Not detected | Yes | 153 | 179 | Diffuse alveolar damage |
| 4 | F/81 | Yes | Mild acute tubular necrosis | + | Not detected | Minimal | 107 | 131 | Diffuse alveolar damage |
| 5 | M/44 | Yes | Severe acute tubular necrosis | - | not detected | yes | 107 | 315 | Diffuse alveolar damage |
| 6 | M/79 | Yes | Moderate acute tubular necrosis | + | not detected | yes | 179 | 238 | Diffuse alveolar damage |
| 7 | F/91 | Yes | Minimal | + | not detected | yes | 75 | 97 | Diffuse alveolar damage |
Heavy abnormal myeloid infiltrate in the kidney (the patient had history of myelodysplastic syndrome).
DISCUSSION
SARS is a new infectious disease with a formidable morbidity and mortality. A wide range of nonrespiratory symptoms has subsequently been reported suggesting other organ involvements including the liver and gastrointestinal tract during the course of the disease4, 10, 11. With the exception of a few chronic dialysis patients complicated by SARS12, 13, 14, impaired renal function with raised plasma creatinine is not a common finding in most SARS patients at the time of first clinical presentation. A preliminary study of 12 patients with SARS who developed ARF reports a high mortality rate of 77%[abstract; Tsai HB, et al, J Am Soc Nephrol 14:513A, 2003).
In our retrospective analysis of 536 SARS patients, 6.7% of this cohort had raised plasma creatinine concentration during the later course of the infection despite normal value at first presentation. Although more sophisticated assessment of renal function was not performed due to the high infectivity of body fluid, ARF with significant kidney dysfunction was suggested by the peak median plasma creatinine concentration of 274 μmol/L. More intriguingly, these patients only developed ARF at a median duration of 20 days from the onset of viral infection that coincided with the late viremic or the hyperimmune response phase of the infection15. Hence, it is totally not surprising that ARF was not reported in previous studies in which only clinicopathologic features at first clinical presentation were presented1, 4, 16, 17, 18. Patients who subsequently developed renal impairment had clinical presentations similar to the symptomatology of SARS patients in previous reports. Severe diarrhea, an important symptom in a patient cohort that might have acquired the infection through the contamination of the sewage systems16, only occurred in 13.8% of our cohort.
The etiology of ARF is likely to be diverse and multifactorial. Broad-spectrum antibiotics, which were sometimes administered in high doses, may cause interstitial nephritis. It should be clarified that identical antibiotic regimes were given to our two groups of SARS patients (with and without ARF). Eosinophilia was not detected in patients with ARF. Despite the fact that ribavirin is predominantly excreted by the kidneys, nephrotoxicity has not been previously reported. In our seven postmortem subjects, we detected no distinct renal pathology and did not find CoV ultrastructurally. However, the ultrastructural interpretation was limited by autolysis of the tissues and a direct involvement of the kidneys by the CoV could not be entirely ruled out. The detection of PCR fragments of CoV in urine from 21% to 50% of SARS patients between the second and third week of the viral infection16 still highlights a possibility of kidney tropism of the CoV. Nevertheless, the normal glomerular histology with absence of electron-dense deposits, mild proteinuria, and absence of active cellular cast all tend to negate the existence of an active immune-mediated glomerulonephritis. Moreover, the in situ hybridization study also failed to detect SARS-CoV in kidney tissues and suggested that persistent infection in these fatal cases was less likely.
On the other hand, refractory hypotension occurred commonly in those patients who developed multiorgan failures. Inotropic support in forms of adrenaline/noradrenaline infusion was administered to half of these patients. Bacterial sepsis, not uncommon in these patients on prolonged ventilatory support and multiorgan failure, could also contribute to the development of ARF. The fall in glomerular filtration secondary to hypotension, vasoconstriction and sepsis plausibly contributed to the development of acute tubular necrosis, evidenced by the histologic finding of the renal tissues in our study.
The poor survival of chronic dialysis patients complicated by SARS was not surprising11, 12, 13 as patients with chronic illness recovered poorly following SARS17 , 19. However, this cohort of patients with ARF demonstrated an extremely poor prognosis. The mortality was in excess of 90% and higher than those who developed ARDS requiring ventilatory support. The ARF reflects that the renal insult occurred in the context of multiorgan failure. As our clinical findings suggest the relative importance of prerenal factors in the development of ARF in these SARS patients, strict precautions should be exercised to ensure that these patients receive adequate hemodynamic support. Hypotension and sepsis must be treated aggressively as the complicating renal impairment is always associated with high mortality. In our multivariate analysis, ARDS is the most significant independent risk factors predicting the development of ARF in SARS despite other parameters (systolic blood pressure, low nadir leukocyte count, low nadir lymphocyte count, low plasma albumin on admission, and the peak serum creatinine kinase phosphatase) are implicated in the development of ARF by univariate analysis.
CONCLUSION
ARF is uncommon in SARS yet carries a high mortality. It occurs in patients complicated with acute respiratory distress syndrome (ARDS) or multiorgan failure. The development of ARF is an important negative prognostic indicator for survival with SARS.
REFERENCES
- 1.Peiris J.S., Lai S.T., Poon L.L., et al. Coronavirus as a possible cause of severe acute respiratory syndrome. Lancet. 2003;361:1319–1325. doi: 10.1016/S0140-6736(03)13077-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Rota P.A., Oberste M.S., Monroe S.S., et al. Characterization of a novel coronavirus associated with severe acute respiratory syndrome. Science. 2003;300:1394–1399. doi: 10.1126/science.1085952. [DOI] [PubMed] [Google Scholar]
- 3.World Health Organization . 2003. http://www.who.int/csr/sars/country/2003_05_22/en/ (accessed. [Google Scholar]
- 4.Tsang K.W., Ho P.L., Ooi G.C., et al. A cluster of cases of severe acute respiratory syndrome in Hong Kong. N Engl J Med. 2003;348:1977–1985. doi: 10.1056/NEJMoa030666. [DOI] [PubMed] [Google Scholar]
- 5.Peiris J.S., Chu C.M., Cheng V.C., et al. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: A prospective study. Lancet. 2003;361:1767–1772. doi: 10.1016/S0140-6736(03)13412-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.World Health Organization: Case definitions for surveillance of severe acute respiratory syndrome (SARS). http://www.who.int/csr/sars/infectioncontrol (revised May 1, 2003, and accessed May 10, 2003)
- 7.Bernard G.R., Artigas A., Brigham K.L., et al. The American-European Consensus Conference on ARDS: Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med. 1994;149:818–824. doi: 10.1164/ajrccm.149.3.7509706. [DOI] [PubMed] [Google Scholar]
- 8.To K.F., Tong J.H., Chan P.K., et al. Tissue and cellular tropisms of the coronavirus associated with severe acute respiratory syndrome—An in-situ hybridization study of fatal cases. J Pathol. 2004;202:157–163. doi: 10.1002/path.1510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tsui P.T., Kwok M.L., Yuen H., Lai S.T. Severe acute respiratory syndrome: Clinical outcome and prognostic correlates. Emerg Infect Dis. 2003;9 doi: 10.3201/eid0909.030362. 1064–1049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.So L.K., Lau A.C., Yam L.C., et al. Development of a standard treatment protocol for severe acute respiratory syndrome. Lancet. 2003;361:1615–1617. doi: 10.1016/S0140-6736(03)13265-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Leung W.K., To K.F., Chan P.K., et al. Enteric involvement of severe acute respiratory syndrome-associated coronavirus infection. Gastroenterology. 2003;125:1011–1017. doi: 10.1016/S0016-5085(03)01215-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tang H.L., Cheuk A., Chu K.H., et al. Severe acute respiratory syndrome in haemodialysis patients: A report of two cases. Nephrol Dial Transplant. 2003;18:2178–2181. doi: 10.1093/ndt/gfg454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Li P.K., Kwan B.C., Leung C.B., et al. Severe acute respiratory syndrome in a haemodialysis patient. Am J Kidney Dis. 2003;42:1069–1074. doi: 10.1016/j.ajkd.2003.07.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wong P.N., Yee W., Mak S.K., et al. The clinical presentation and outcome of severe acute respiratory syndrome in dialysis patients. Am J Kidney Dis. 2003;42:1075–1081. doi: 10.1016/j.ajkd.2003.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lai K.N., Tsang K.W., Seto W.H., Ooi C.G. Clinical, laboratory, and radiologic manifestation of SARS. Curr Infect Dis Report. 2004;6:213–219. doi: 10.1007/s11908-004-0011-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Peiris J.S., Chu C.M., Cheng V.C., et al. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: A prospective study. Lancet. 2003;361:1761–1767. doi: 10.1016/S0140-6736(03)13410-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lee N., Hui D., Wu A., et al. A major outbreak of severe acute respiratory syndrome in Hong Kong. N Engl J Med. 2003;348:1986–1994. doi: 10.1056/NEJMoa030685. [DOI] [PubMed] [Google Scholar]
- 18.Ksiazek T.G., Erdman D., Goldsmith C.S., et al. A novel coronavirus associated with severe acute respiratory syndrome. N Engl J Med. 2003;348:1953–1966. doi: 10.1056/NEJMoa030781. [DOI] [PubMed] [Google Scholar]
- 19.Booth C.M., Matukas L.M., Tomlinson G.A., et al. Clinical features and short-term outcomes of 144 patients with SARS in the greater Toronto area. JAMA. 2003;289:2861–2863. doi: 10.1001/jama.289.21.JED30036. [DOI] [PubMed] [Google Scholar]