Abstract
Biosynthesis of nanoparticles inside S. aureus cells has enhanced the sensitivity of immunoassays based on the S. aureus nanoparticles. However, the current methods are limited to antigen detection by conjugating IgG antibodies on S. aureus nanoparticles. In this study, a simple way to conjugate antigens to the S. aureus nanobioparticles was developed by utilizing a cell wall binding domain (CBD) from a bacteriophage lysin PlyV12. Based on this novel design, simple agglutination tests of the IgG antibodies of Ebola virus (EBOV) nucleoprotein (NP) and Middle East Respiratory Virus (MERS) NP in rabbit sera were successfully developed by conjugating the S. aureus nanobioparticles with two fusion proteins EBOV NP- CBD and MERS NP-CBD, respectively. The conjugation was done easily by just mixing the fusion proteins with the S. aureus nanoparticles. The detection time was within 20 min without any special equipment or expertise. As far as we know, this is the first time to realize the detection of viral antibodies based on S. aureus nanoparticles.
Keywords: Virus; Antibody detection; Staphylococcus aureus; 5-cyano-2,3-ditolyl tetrazolium chloride
1. Introduction
In recent years, as an emerging field in bio-nanotechnology, the synthesis of nanomaterials by living organisms, such as yeast, fungi and bacteria, have demonstrated great potential for different applications [1], [2]. Among bacteria, protein A producing S. aureus has been an attractive vector for immunoassays of pathogens, especially for coagglutination tests of bacteria and viruses [3], because it is easy to conjugate IgG antibodies through protein A molecules on the surface of S. aureus cells. On the same time, biosynthesis of nanoparticles inside S. aureus cells could be exploited to enhance detection sensitivity. For example, biosynthesis of quantum dots inside S. aureus cells has been used for sensitive detection of H9N2 virus [2]. In our lab, we established a simple way to transform S. aureus into red and fluorescent nanobioprobes by reacting with a monotetrazolium redox dye, 5-cyano-2, 3-ditolyl tetrazolium chloride (CTC) at room temperature [4]. Comparing with the uncolored S. aureus nanobioprobes, 100 fold higher sensitivity was achieved in rapid coagglutination test of E. coli O157:H7 with the novel red nanobioprobes.
Despite tremendous progress made in this area, the current methods are still limited to antigen detection by conjugating IgG antibodies on S. aureus nanoparticles. In this study, we aim to present a simple way to conjugate antigens to the S. aureus nanobioparticles by utilizing a cell wall binding domain (CBD) from a bacteriophage lysin PlyV12, which could bind to the surface of the S. aureus nanobioparticles [5], [6]. Based on this novel design, simple agglutination tests of the IgG antibodies of Ebola virus (EBOV) nucleoprotein (NP) and Middle East respiratory syndrome coronavirus (MERS-CoV) NP were successfully developed by conjugating the S. aureus nanobioparticles with two fusion proteins EBOV NP-CBD and MERS NP-CBD.
EBOV and MERS-CoV are highly contagious pathogens which can cause Ebola virus disease and the acute respiratory infectious disease-Middle East respiratory syndrome, respectively [7], [8]. Because until now no specific antiviral drugs or vaccines have been approved for clinical use against EBOV and MERS-CoV, rapid and specific diagnosis is essential for controlling the infections. In general, virus-specific antibodies IgM or IgG with rising titers constitute a strong presumptive detection. Therefore, detection of IgM or IgG antibodies in blood are used commonly in epidemic investigation of viral infections [9], [10], [11], [12], [13], [14]. Some diagnostic methods, such as Enzyme-linked immunosorbent assay (ELISA) [9], [10], [11], [15], indirect immunofluorescence assay (IFA) [16], [17], [18], neutralization test [12], [19], [20] and protein microarray [21], have been developed for EBOV and MERS-CoV antibody detection. However, the requirement of dedicated instruments limits the application of these assays in field, which is important for fast responses during epidemics. In comparison, agglutination test is more rapid, inexpensive, simple to interpret, and most importantly requires no specialized instrument since naked eyes could read the results [3]. Therefore, agglutination test might be a good choice for EBOV or MERS-CoV antibody detection in field. In our agglutination test, the conjugation is quite simple and could be done easily by just mixing the fusion proteins with the S. aureus nanoparticles. As far as we know, this is the first time to realize the detection of viral antibodies based on S. aureus nanoparticles.
2. Materials and methods
A monoclonal antibody (clone 4B4) specific against EBOV subtype Zaire NP were developed by our lab previously. Polyclonal rabbit antisera against EBOV subtype Zaire NP and MERS Virus NP were kindly provided by Professor Zhengli Shi, from Wuhan Institute of Virology, Chinese Academy of Sciences.
2.1. Determination of the antibody titer of monoclonal antibody and polyclonal rabbit antiserum against EBOV subtype Zaire NP by ELISA
ELISAs were performed as described by Nakayama et al. using the recombinant NP antigens [9]. Briefly, ELISA plates were coated with a predetermined optimal quantity of recombinant EBOV subtype Zaire NP antigens (50 ng/well). Then purified monoclonal antibody (clone 4B4) and polyclonal rabbit antiserum diluted in series 2 fold from 1:10 to 1:81920 with PBS containing 1% BSA were added into the wells and incubated with the coated NP antigen for 30 min at 37 °C, respectively. After washing, the bound monoclonal antibody (clone 4B4) and the polyclonal rabbit antiserum were detected by using horseradish peroxidase (HRP) conjugated rabbit anti-mouse IgG and goat anti-rabbit IgG at a dilution of 1:5000, respectively. Finally, the optical density (OD) was measured at 450 nm using a microplate reader (Synergy H1, BioTek, USA). Each dilution was tested in triplicates.
2.2. Preparation of recombinant antigens
The fusion protein EBOV NP-CBD was constructed as follows. The EBOV NP gene was amplified from pET32a-EBOV NP with the primers EBOV NP-CBD-F: 5′- GCGTGCTAGCGATTCTCGTCCTCAG-3′ and EBOV NP-CBD-R:5′- AATTGAATTCTGAACCGCCTCCACCCTGATGATGTTGCAGG-3′ (restriction endonuclease sites were italized) and then digested by Nhe I and EcoR I. Subsequently, the digested fragment was inserted into plasmid pET28a-(G4S)2-EBOV CBD digested by Nhe I and EcoR I, yielding the plasmid pET28a-EBOV NP-(G4S)2-CBD. The expression vector pET28a-MERS NP-(G4S)2-CBD for fusion protein MERS NP-CBD was constructed in the same way. Then the cloned plasmids were transformed into E.coli BL21 competent cells. Both histidine (His) tagged EBOV NP-CBD and MERS NP-CBD were expressed in E.coli and purified by nickel-chelating affinity chromatography. Purity of the eluted EBOV NP-CBD and MERS NP-CBD were confirmed by SDS-PAGE and the concentrations of EBOV NP-CBD and MERS NP-CBD after dialysis were determined with a bicinchoninic acid (BCA) kit (Bi Yun Tian Biotechnology Inc, China).
2.3. Preparation of S. aureus Nanobioparticles
The S. aureus nanobioparticles were prepared from S. aureus strain CCTCC AB91118. Briefly, the S. aureus cells were grown in 5 mL of Luria-Bertani broth from the master seed and incubated at 37 °C for 12 h. Then 2 mL of the culture was transferred into 100 mL of Luria-Bertani broth and incubated at 37 °C until OD600 reached 0.6-0.8. The cells were harvested by centrifugation at 8000 rpm for 5 min and washed twice with phosphate buffer saline (PBS, pH 7.4). Following the addition of 4 mM CTC into the culture, the cells were kept at 37 °C for 1 h. Subsequently, the red colored cells were harvested by centrifugation at 6000 rpm for 10 min and washed thrice with PBS. Finally, the cells were heat treated at 80 °C for 20 min in a water bath for complete inactivation and the nanobioparticles obtained were stored at 4 °C until used.
The preparation of S. aureus nanobioparticles used for agglutination test of MERS NP IgG antibodies was similar to the above method, except that S. aureus cells were stained with another tetrazolium redox dye, triphenyl tetrazolium chloride (TTC), instead of CTC.
2.4. Testing IgG antibodies of EBOV NP (V-bottom 96-well plate)
The S. aureus nanobioparticles were sensitized with the fusion protein EBOV NP-CBD for detecting the IgG antibodies of EBOV NP. Briefly, the suspension (1 mL) of the S. aureus nanoparticles prepared above was mixed with 20 μg EBOV NP-CBD and then the mixture was incubated at 37 °C for half an hour in a shaker (180 rpm). The nanobioprobes obtained were then washed with PBS (pH 7.4) by centrifugation and resuspended in PBS to a concentration of 10% (w/v). Sodium azide was added to a final concentration of 0.02% and the nanobioprobe suspension was stored at 4 °C until used.
Before the test, the polyclonal rabbit antiserum was inactivated at 56 °C for 30 min. For the agglutination test, 25 μL of the diluted polyclonal rabbit antiserum or the diluted mAb was mixed with an equal volume of the nanobioprobes sensitized with the EBOV NP-CBD in a V-bottom 96-well plate. The mixture was incubated at 37 °C and mixed thoroughly every two minutes. A blank control reaction was simultaneously made by mixing 25 μL of the nanobioprobe suspension with 25 μL of PBS. A sample was considered positive only when it appeared obvious agglutination and the blank control did not show agglutination by naked eyes within 10 min.
2.5. The reaction among the IgG moleucles, protein A on the surface of one staphylococcal particle and antigen molecules fused with CBD on another particle
To demonstrate that the IgG antibodies of MERS NP are sandwiched between protein A on the surface of one staphylococcal particle via their Fc fragments and the MERS NP-CBD fused with another particle after agglutination, two types of S. aureus nanobioparticles, one sensitized with MERS NP-CBD and the other non-sensitized, were used. First, 50 μL of the non-sensitized S. aureus nanoparticles were mixed with 10 μL of the MERS NP polyclonal rabbit antiserum and incubated at 37 °C for an hour. Then the mixture was centrifuged at 6000 rpm for 5 min to remove the un-bound antibodies and harvest the particles pre-reacted with the IgG antibodies. The pre-reacted particles were washed thrice with PBS, and resuspended in 50 μL of PBS. Finally, 25 μL of the MERS NP-CBD sensitized S. aureus nanobioparticles were mixed with 25 μL of the pre-reacted S. aureus nanoparticles on a microscope slide. The mixture was incubated at room temperature and mixed thoroughly every two minutes. A negative control was simultaneously made by mixing 25 μL of the non-sensitized S. aureus nanobioparticles with 25 μL of the nanoparticles pre-reacted with the IgG antibodies.
2.6. Testing IgG antibodies of MERS NP (slide method)
Before the agglutination test for IgG antibodies of MERS NP, the S. aureus nanobioparticles were sensitized with the fusion protein MERS NP-CBD in a similar way as the preparation of EBOV NP sensitized S. aureus nanobioparticles. For the agglutination test, 30 μL of MERS NP polyclonal rabbit antiserum was mixed with 10 uL of the nanobioprobes sensitized with the MERS NP-CBD on a microscope slide. The mixture was incubated at room temperature and mixed thoroughly every two minutes. The blank control and negative control were simultaneously made by mixing 30 μL of PBS or negative rabbit serum, respectively, with 10 μL of the nanobioprobe suspension.
2.7. Specificity evaluation
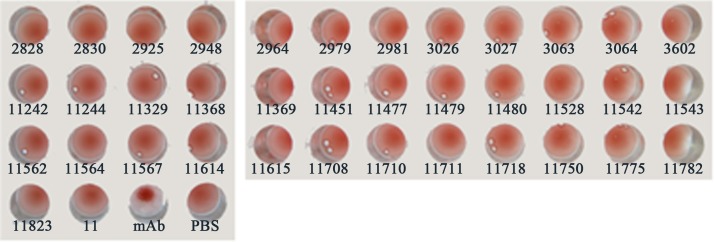
Thirty seven HCV positive sera and a serum from a suspected case with influenza-like symptoms were used to test the non-specific agglutination. All of the sera were firstly inactivated by heat at 60 °C for an hour and diluted 10 times using PBS before test. The specificity tests were performed by mixing 25 μL of a diluted serum with an equal volume of the nanobioprobes sensitized with EBOV NP-CBD. The mixture was incubated at 37 °C and mixed thoroughly every two minutes. Agglutination in 30 min was observed.
2.8. Stability testing
The suspension of the nanobioprobes sensitized with the EBOV NP-CBD was stored at 4 °C for a month. During the storage, at the end of each week, the agglutination tests were performed.
3. Results
3.1. Principle of the agglutination test for detecting the IgG antibodies of EBOV NP
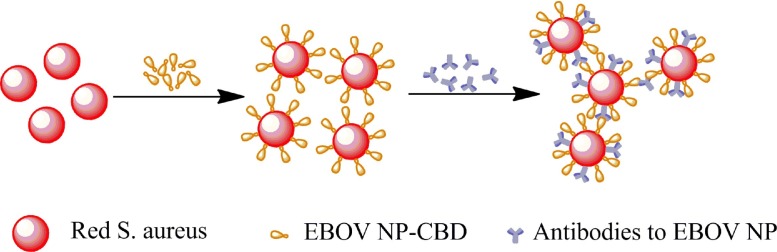
As shown in Fig. 1 , for detecting the IgG antibodies of EBOV NP, the colored S. aureus nanoparticles were sensitized with the fusion protein EBOV NP-CBD via the binding of the CBD to the surface [5]. In the presence of the IgG antibodies of EBOV NP, most of the IgG antibodies could bind to protein A on the surface of the staphylococcal particles via their Fc fragments, leaving their antigen binding domains to attach with the NP on other staphylococcal particles, which leads to agglutination.
Fig 1.
Schematic diagram of the agglutination test for detection of EBOV NP antibodies.
3.2. EBOV NP antigens could bind to the surface of S. aureus nanoparticles via fusion with CBD
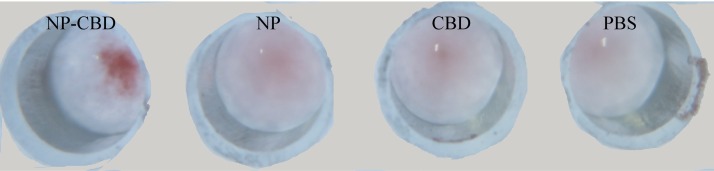
The suspension (1 mL) of the S. aureus nanoparticles were sensitized with 20 μg fusion protein EBOV NP-CBD, EBOV NP or CBD, respectively. After that, these sensitized nanobioprobes were incubated with an equal volume of mAb at a dilution of 1:100, respectively, in a V-bottom 96-well plate. As shown in Fig. 2 , EBOV NP-CBD sensitized nanobioprobes appeared obvious agglutination while EBOV NP or CBD sensitized nanobioprobes and the blank control did not show agglutination within 10 min. These results showed that EBOV NP antigens could bind to the surface of S. aureus nanoparticles via fusion with CBD.
Fig. 2.
Agglutination test of the EBOV NP antibodies with S. aureus nanoparticles sensitized with different proteins. NP-CBD: EBOV nucleoprotein fused with the cell binding domain of lysin PlyV12; NP: EBOV nucleoprotein; CBD: the cell binding domain of lysin PlyV12. PBS: phosphate saline buffer as a control.
3.3. Agglutination test for detecting the IgG antibodies of EBOV NP
The titers of monoclonal antibody and polyclonal rabbit antiserum against EBOV subtype Zaire NP tested by ELISA were 1:20480 and 1:81920, respectively. The polyclonal antibodies to EBOV NP were diluted in series 2 fold from 1:10 to 1:10240 with PBS and 25 μL from each dilution was used for the agglutination test. As shown in Fig. 3 A, the highest dilution of the antibody at which agglutination was clearly seen in 10 min was 1:1280.
Fig. 3.
Agglutination test of the EBOV NP antibodies. (A) Agglutination test of the EBOV NP polyclonal antibody. (B) Agglutination test of the EBOV NP monoclonal antibody.
Because EBOV positive sera were unavailable, in order to simulate EBOV positive sera, the mAb specific against EBOV NP were first diluted 10 fold with a human serum, and then diluted 2 fold from 1:10 to 1:640 with PBS. An aliquot of 25 μL from each dilution was used for the agglutination test. The human serum without spiking with the EBOV NP was also diluted with PBS as a negative control. As shown in Fig. 3B, the highest dilution of the antibody was 1:80, at which agglutination could be clearly seen in 10 min, while no agglutination could be seen for the negative control. Since the mAb sample was first diluted 10 fold with the human serum, the actual dilution factor is 1:800 in Fig. 3B. These results showed that EBOV NP IgG antibodies in the human serum can be detected successfully by the agglutination test.
3.4. Agglutination test for detecting the IgG antibodies of MERS NP
The polyclonal antibodies to MERS NP and negative rabbit serum were diluted 10 folds and then used for the agglutination test on a glass slide. As shown in Fig. 4 , the polyclonal rabbit antiserum sample appeared obvious agglutination, while the blank control or the negative control did not show agglutination within 10 min.
Fig. 4.
Agglutination test of the MERS NP polyclonal antibody.
3.5. Agglutination was due to the reaction among the IgG antibodies of MERS NP, protein A on the surface of one staphylococcal particle and the MERS NP-CBD fused with another particle
As shown in Fig. 5 , when the MERS NP-CBD sensitized S. aureus nanobioparticles were mixed with the S. aureus nanoparticles which have pre-reacted with the polyclonal rabbit antiserum sample, obvious agglutination could be observed while the negative control did not show agglutination within 10 min. Since there were no free antibodies in the mixture, these results demonstrate that the agglutination was caused by the reaction among the IgG antibodies of MERS NP, protein A on the surface of one staphylococcal particle and the MERS NP-CBD bound with another particle.
Fig. 5.
Agglutination between the S. aureus nanoparticles pre-reacted with the polyclonal rabbit antiserum with either the MERS NP-CBD sensitized S. aureus nanobioparticles (MERS-NP-CBD) or the non-sensitized S. aureus nanobioparticles (negative control).
3.6. Specificity evaluation
As shown in Fig. 6 , no agglutination could be observed within 30 min when the EBOV NP-CBD sensitized nanobioprobes were incubated with the 38 clinical sera tested. These results demonstrated that the nanobioprobes were quite specific.
Fig. 6.
Specificity evaluation of the agglutination test for detection of the EBOV monoclonal antibody using 38 serum samples. mAb: the well added with the EBOV monoclonal antibody; PBS: buffer used as the control.
3.7. Stability testing
As shown in Fig. 7 , EBOV NP-CBD sensitized nanobioprobes were found stable for at least one month after storage at 4 °C and the detection sensitivity did not deteriorate during the storage.
Fig. 7.
Stability test of the agglutination reagents for detection of the EBOV monoclonal antibody. A. The first week; B. After store at 4 °C for a month.
4. Discussion
In this study, we have presented a simple and rapid way to conjugate antigens to the surface of the S. aureus nanobioparticles by fusing antigens with PlyV12 CBD and successfully developed an agglutination test for the detection of IgG antibodies of EBOV NP and MERS NP. Compared with the previous agglutination tests based on latex particles for the detection of IgG antibodies, there are a few advantages using the present method.
First, physical adsorption or chemical cross-linking is normally used to sensitize the latex particles with antigens. The antigen molecules are randomly immobilized on the surface of latex particles, which could affect the subsequent antigen-antibody binding reaction. In the current method, the antigen molecules were immobilized on the S. aureus particles via the CBD fused with them, which can make sure that all the antigen molecules have a good orientation to react with the antibody. Therefore, the current method could provide better control of the antigen activity than the previous methods for detection of antibodies.
Second, to detect antibodies during the latex agglutination, the two antigen binding sites of a IgG molecule need to react with one antigen molecule on one particle and another antigen molecule on another particle in order to form agglutination, which might be difficult since the antigen molecules on the same particle will compete to attach with the two antigen binding sites of a IgG molecule. However, in the current method, no such competition exists since most of the IgG moleucles could bind to protein A on the surface of one staphylococcal particle via their Fc fragments, and bind to the antigen molecules on another particle via their Fab to form agglutination (Fig. 1 and Fig. 5). This new agglutination mode could lead to higher detection sensitivity in theory. Actually as far as we know, this is the first time that the S. aureus nanoparticles have been used in agglutination test for the detection of antibodies.
Lastly, the S. aureus nanoparticles are biologically synthesized with many biomolecules on its surface, which could be utilized to attach with different molecules and provide new possibilities for sensitization. Lysins, produced by bacteriophages to digest the bacterial cell wall for the release of progeny virions, have been considered effective anti-infective agents for control of Gram-positive bacteria [22]. Normally at the C terminus of a lysin there is a cell wall binding domain (CBD) which can specifically attach to the host cell with a high affinity. In the current study, a CBD from a phage lysin PlyV12 was utilized for conjugation of the antigen by a simple mixing and incubation step. The successful sensitization of the S. aureus nanoparticles with EBOV NP and MERS NP indicated that this strategy via CBD might be used widely for other antigens.
Fig. 5 showed that there was no non-specific agglutination caused by HCV positive sera or the suspected influenza virus serum, demonstrating good selectivity of our method. The sensitivities of the antibody agglutination tests are lower than that of ELISA. The highest dilution of the monoclonal antibody and the polyclonal rabbit antiserum antibody against EBOV subtype Zaire NP were 1:800 and 1:1280, respectively, at which agglutination could be clearly seen in 10 min (Fig. 4), while the antibody titers of the antibody and the polyclonal rabbit antiserum tested by ELISA were 1:20480 and 1:81920, respectively. However, due to their simplicity, rapidness, good stability and no needs of instrument to read results, the antibody agglutination tests based on the multifunctional S. aureus nanobioprobes could have good potential for real applications in field.
In conclusion, a simple way to conjugate antigens to the S. aureus nanobioparticles was developed by utilizing a cell wall binding domain (CBD) from a bacteriophage lysin PlyV12. Based on this novel design, simple agglutination tests of antibodies could be developed without any special instrument or expertise.
Competing interests
HP Wei declares that a patent has been filed for the colored S. aureus nanoparticles.
Acknowledgements
This work was supported financially by the Chinese Academy of Sciences, (CXJJ-15Z012) and the Key Laboratory of Emerging Infectious Diseases and Biosafety, Wuhan, China.
References
- 1.Dhillon G.S., Brar S.K., Kaur S., Verma M. Green approach for nanoparticle biosynthesis by fungi: current trends and applications. Crit. Rev. Biotechnol. 2012;32(1):49–73. doi: 10.3109/07388551.2010.550568. [DOI] [PubMed] [Google Scholar]
- 2.Xiong L.H., Cui R., Zhang Z.L., Yu X., Xie Z., Shi Y.B., Pang D.W. Uniform fluorescent nanobioprobes for pathogen detection. ACS Nano. 2014;8(5):5116–5124. doi: 10.1021/nn501174g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Picardeau M., Bertherat E., Jancloes M., Skouloudis A.N., Durski K., Hartskeerl R.A. Rapid tests for diagnosis of leptospirosis: current tools and emerging technologies. Diagn. Microbiol. Infect. Dis. 2014;78(1):1–8. doi: 10.1016/j.diagmicrobio.2013.09.012. [DOI] [PubMed] [Google Scholar]
- 4.Hu W., Zhang Y., Yang H., Yu J., Wei H. Simple and rapid preparation of red fluorescence and red color S. aureus derived nanobioparticles for pathogen detection. J. Microbiol. Methods. 2015;115:47–53. doi: 10.1016/j.mimet.2015.04.018. [DOI] [PubMed] [Google Scholar]
- 5.Dong Q., Wang J., Yang H., Wei C., Yu J., Zhang Y., Huang Y., Zhang X.E., Wei H. Construction of a chimeric lysin Ply187N-V12C with extended lytic activity against staphylococci and streptococci. Microbial Biotechnol. 2015;8(2):210–220. doi: 10.1111/1751-7915.12166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yu J., Zhang Y., Zhang Y., Li H., Yang H., Wei H. Sensitive and rapid detection of staphylococcus aureus in milk via cell binding domain of lysin. Biosens. Bioelectron. 2016;77:366–371. doi: 10.1016/j.bios.2015.09.058. [DOI] [PubMed] [Google Scholar]
- 7.Zaki A.M., van Boheemen S., Bestebroer T.M., Osterhaus A.D., Fouchier R.A. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N. Engl. J. Med. 2012;367(19):1814–1820. doi: 10.1056/NEJMoa1211721. [DOI] [PubMed] [Google Scholar]
- 8.Richardson K.J. Ebola virus disease. Adv. Emerg. Nurs. J. 2015;37(2):102–115. doi: 10.1097/TME.0000000000000063. [DOI] [PubMed] [Google Scholar]
- 9.Nakayama E., Yokoyama A., Miyamoto H., Igarashi M., Kishida N., Matsuno K., Marzi A., Feldmann H., Ito K., Saijo M., Takada A. Enzyme-linked immunosorbent assay for detection of filovirus species-specific antibodies. Clin. Vaccine Immunol. 2010;17(11):1723–1728. doi: 10.1128/CVI.00170-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Saijo M., Niikura M., Morikawa S., Ksiazek T.G., Meyer R.F., Peters C.J., Kurane I. Enzyme-linked immunosorbent assays for detection of antibodies to Ebola and Marburg viruses using recombinant nucleoproteins. J. Clin. Microbiol. 2001;39(1):1–7. doi: 10.1128/JCM.39.1.1-7.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ikegami T., Saijo M., Niikura M., Miranda M.E., Calaor A.B., Hernandez M., Manalo D.L., Kurane I., Yoshikawa Y., Morikawa S. Immunoglobulin G enzyme-linked immunosorbent assay using truncated nucleoproteins of Reston Ebola virus. Epidemiol. Infect. 2003;130(3):533–539. [PMC free article] [PubMed] [Google Scholar]
- 12.Zhao G., Du L., Ma C., Li Y., Li L., Poon V.K., Wang L., Yu F., Zheng B.J., Jiang S., Zhou Y. A safe and convenient pseudovirus-based inhibition assay to detect neutralizing antibodies and screen for viral entry inhibitors against the novel human coronavirus MERS-CoV. Virol. J. 2013;10:266. doi: 10.1186/1743-422X-10-266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yamaoka Y., Matsuyama S., Fukushi S., Matsunaga S., Matsushima Y., Kuroyama H., Kimura H., Takeda M., Chimuro T., Ryo A. Development of monoclonal antibody and diagnostic test for middle east respiratory syndrome coronavirus using cell-free synthesized nucleocapsid antigen. Front. Microbiol. 2016;7:509. doi: 10.3389/fmicb.2016.00509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tang X.C., Agnihothram S.S., Jiao Y., Stanhope J., Graham R.L., Peterson E.C., Avnir Y., Tallarico A.S., Sheehan J., Zhu Q., Baric R.S., Marasco W.A. Identification of human neutralizing antibodies against MERS-CoV and their role in virus adaptive evolution. Proc. Natl. Acad. Sci. U. S. A. 2014;111(19):E2018–E2026. doi: 10.1073/pnas.1402074111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Al-Abdallat M.M., Payne D.C., Alqasrawi S., Rha B., Tohme R.A., Abedi G.R., Al Nsour M., Iblan I., Jarour N., Farag N.H., Haddadin A., Al-Sanouri T., Tamin A., Harcourt J.L., Kuhar D.T., Swerdlow D.L., Erdman D.D., Pallansch M.A., Haynes L.M., Gerber S.I., Jordan M.-C.I.T. Hospital-associated outbreak of Middle East respiratory syndrome coronavirus: a serologic, epidemiologic, and clinical description. Clin. Infect. Dis. 2014;59(9):1225–1233. doi: 10.1093/cid/ciu359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Saijo M., Niikura M., Morikawa S., Kurane I. Immunofluorescence method for detection of Ebola virus immunoglobulin g, using HeLa cells which express recombinant nucleoprotein. J. Clin. Microbiol. 2001;39(2):776–778. doi: 10.1128/JCM.39.2.776-778.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ikegami T., Saijo M., Niikura M., Miranda M.E., Calaor A.B., Hernandez M., Manalo D.L., Kurane I., Yoshikawa Y., Morikawa S. Development of an immunofluorescence method for the detection of antibodies to Ebola virus subtype Reston by the use of recombinant nucleoprotein-expressing HeLa cells. Microbiol. Immunol. 2002;46(9):633–638. doi: 10.1111/j.1348-0421.2002.tb02745.x. [DOI] [PubMed] [Google Scholar]
- 18.Corman V.M., Muller M.A., Costabel U., Timm J., Binger T., Meyer B., Kreher P., Lattwein E., Eschbach-Bludau M., Nitsche A., Bleicker T., Landt O., Schweiger B., Drexler J.F., Osterhaus A.D., Haagmans B.L., Dittmer U., Bonin F., Wolff T., Drosten C. Assays for laboratory confirmation of novel human coronavirus (hCoV-EMC) infections. Euro Surveill. 2012;17(49) doi: 10.2807/ese.17.49.20334-en. [DOI] [PubMed] [Google Scholar]
- 19.Macneil A., Reed Z., Rollin P.E. Serologic cross-reactivity of human IgM and IgG antibodies to five species of Ebola virus. PLoS Negl. Trop. Dis. 2011;5(6):e1175. doi: 10.1371/journal.pntd.0001175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Perera R.A., Wang P., Gomaa M.R., El-Shesheny R., Kandeil A., Bagato O., Siu L.Y., Shehata M.M., Kayed A.S., Moatasim Y., Li M., Poon L.L., Guan Y., Webby R.J., Ali M.A., Peiris J.S., Kayali G. Seroepidemiology for MERS coronavirus using microneutralisation and pseudoparticle virus neutralisation assays reveal a high prevalence of antibody in dromedary camels in Egypt, June 2013. Euro Surveill. 2013;18(36):20574. doi: 10.2807/1560-7917.es2013.18.36.20574. [DOI] [PubMed] [Google Scholar]
- 21.Reusken C., Mou H., Godeke G.J., van der Hoek L., Meyer B., Muller M.A., Haagmans B., de Sousa R., Schuurman N., Dittmer U., Rottier P., Osterhaus A., Drosten C., Bosch B.J., Koopmans M. Specific serology for emerging human coronaviruses by protein microarray. Euro Surveill. 2013;18(14):20441. doi: 10.2807/1560-7917.es2013.18.14.20441. [DOI] [PubMed] [Google Scholar]
- 22.Fischetti V.A., Nelson D., Schuch R. Reinventing phage therapy: are the parts greater than the sum? Nat. Biotechnol. 2006;24(12):1508–1511. doi: 10.1038/nbt1206-1508. [DOI] [PubMed] [Google Scholar]