Highlights
-
•
Glycyrrhizin inhibits PRRSV proliferation.
-
•
Glycyrrhizin mainly interferes with internalization stage in PRRSV life cycle.
-
•
Glycyrrhizin does not directly inactivate PRRSV particles.
Keywords: Porcine reproductive and respiratory syndrome virus (PRRSV), Glycyrrhizin, Proliferation, Penetration
Abstract
Glycyrrhizin is a natural component extracted from the roots of Glycyrrhiza glabra. In this study, we investigated the antiviral activity of glycyrrhizin against porcine reproductive and respiratory syndrome virus (PRRSV), an Arterivirus that has been devastating the swine industry worldwide since the late 1980s. Our results showed that treatment with glycyrrhizin significantly reduced PRRSV proliferation and PRRSV-encoded protein expression in a dose-dependent manner. Mechanistically, glycyrrhizin mainly inhibits the penetration stage, and has little effect on the steps of adsorption or release of PRRSV in its life cycle. Furthermore, we were able to exclude a direct inhibitory action of glycyrrhizin on PRRSV particles. Given these results, glycyrrhizin may be a candidate component for a novel porcine reproductive and respiratory syndrome (PRRS) control strategy.
Porcine reproductive and respiratory syndrome (PRRS) is an economically important infectious disease characterized by severe reproductive failure in sows and respiratory distress in piglets and growing pigs (Lunney et al., 2010, Meng, 2012). The causative agent, PRRS virus (PRRSV), is a single-stranded positive-sense RNA virus classified within the Arteriviridae family (Nelsen et al., 1999, Snijder et al., 2013). Since its emergence in the late 1980s, PRRS has been a threat to the global swine industry (Neumann et al., 2005).
At present, vaccination is the most prevalent way of controlling PRRSV infection. Unfortunately, the current commercially vaccines fail to provide sustainable disease control and prevention (Huang and Meng, 2010, Murtaugh and Genzow, 2011, Yoo et al., 2004). Antiviral therapeutics provide a pivotal tool for combating viral infections, especially for cases in which existing vaccines fail to match the circulating virus. Therefore, an alternative measure to control PRRSV, such as pharmacological intervention, is needed. Traditional Chinese medicines provide a valuable source for promising antiviral drugs. Many natural compounds and compositions have been proven to have antiviral activities, including against PRRSV, such as flavaspidic acid AB (Yang et al., 2013), ouabain, valinomycin, bufalin (Karuppannan et al., 2012), and morpholino oligomer (Opriessnig et al., 2011).
Glycyrrhizin, a triterpene saponine, is a constituent of licorice root. Previous studies have demonstrated that glycyrrhizin has the ability to inhibit the proliferation of many viruses, such as hepatitis B virus (HBV) (Sato et al., 1996), hepatitis C virus (HCV) (Matsumoto et al., 2013), herpes simplex virus (HSV) (Huang et al., 2012), SARS coronavirus (Cinatl et al., 2003), and influenza viruses (Wolkerstorfer et al., 2009). In the present study, we aimed to assess the antiviral activity of glycyrrhizin against PRRSV and further elucidate the exact phase of the viral reproductive cycle that is affected by glycyrrhizin.
We first tested the cytotoxicity of glycyrrhizin (glycyrrhizic acid ammonium salt, Sigma–Aldrich) to MARC-145 cells, the permissive cells for PRRSV infection in vitro, by MTT assay as described previously (Yang et al., 2013). As shown in Fig. 1A, no appreciable cytotoxicity in MARC-145 cells could be observed at concentrations ranging from 200 to 800 μM. To test whether glycyrrhizin has antiviral activity against PRRSV, MARC-145 cells were pretreated with glycyrrhizin at the indicated concentrations and then infected with PRRSV strain WUH3 (at 0.1 PFU/cell). Plaque assays were performed to determine virus titers of the infected cells at the indicated times post-infection. As shown in Fig. 1 B, treatment with glycyrrhizin resulted in a notably significant reduction of PRRSV titer in a dose-dependent manner at all tested time points. To further confirm the reduction of virus titer induced by treatment with glycyrrhizin, we also tested the expression of PRRSV nonstructural protein 2 (Nsp2) using western blot with monoclonal antibody against the Nsp2 of PRRSV strain WHU3 (Wang et al., 2013a). The results showed that the expression of PRRSV Nsp2 was notably significantly reduced following treatment with glycyrrhizin, and this inhibition also occurred in a dose-dependent manner (Fig. 1C). Moreover, the inhibition of Nsp2 expression by glycyrrhizin could be observed at all examined time points (Fig. 1D). These results indicate that treatment with glycyrrhizin inhibits PRRSV proliferation.
Fig. 1.
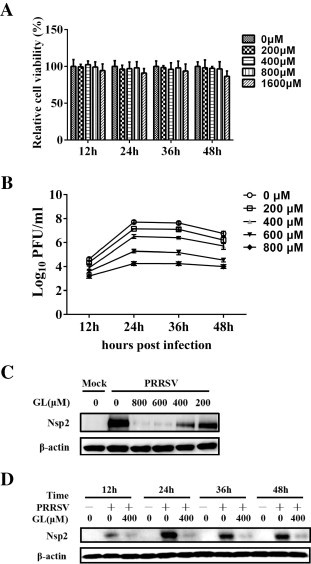
Glycyrrhizin has antiviral activity against PRRSV. (A) Determination of cytotoxicity of glycyrrhizin by MTT assay. MARC-145 cells were incubated with various concentrations of glycyrrhizin or the control PBS for 48 h prior to the MTT assay. (B) MARC-145 cells were pretreated with glycyrrhizin at the indicated concentrations for 2 h prior to PRRSV infection (0.1 PFU/cell). The infected cells were cultured in the presence of different concentrations of glycyrrhizin, and collected at 12, 24, 36 and 48 hpi for plaque assays. Plaque forming units were counted and the viral titer was determined as plaque forming units per ml (PFU/ml). Differences between glycyrrhizin treated groups and PBS treated control group were statistically significant (P < 0.01). (C) MARC-145 cells were treated with glycyrrhizin (GL) at the indicated concentrations for 2 h, then mock-infected or infected with PRRSV (0.1 PFU/cell) and cultured in the presence of glycyrrhizin. At 24 hpi, cells were collected for western blot analysis using a specific antibody against PRRSV Nsp2 protein, with β-actin used as a loading control. (D) MARC-145 cells were pretreated with 400 μM of glycyrrhizin for 2 h prior to PRRSV infection (0.1 PFU/cell), then cultured in the presence of glycyrrhizin. At 12, 24, 36 and 48 hpi, cells were harvested for western blot analysis using a monoclonal antibody against PRRSV Nsp2. The data in this figure are representative of three independent experiments.
Suggested mechanisms of glycyrrhizin antiviral activity include direct effects on the adsorption, penetration and particle maturation steps of the virus reproductive cycle. To further explore the possible mechanism of action through which glycyrrhizin inhibits PRRSV infection, we first attempted to investigate the growth kinetics of PRRSV. MARC-145 cells were infected with PRRSV at 0.1 PFU/cell. Both the cells and culture medium were titrated by plaque assay at 0 hpi and every 2 h between 6 and 26 hpi, as well as 36 and 48 hpi. The results showed that infectious PRRSV could be detected in the supernatant at 10 hpi (Fig. 2 A), suggesting that progeny virus particles were released after 8 hpi. It should be noted that there is slight difference between the virus titers of Figs. 1B and 2A. We speculated that this difference was resulted from the different cell treatment methods because cells were washed three times with PBS in the experiment of Fig. 1B when compared to Fig. 2A.
Fig. 2.
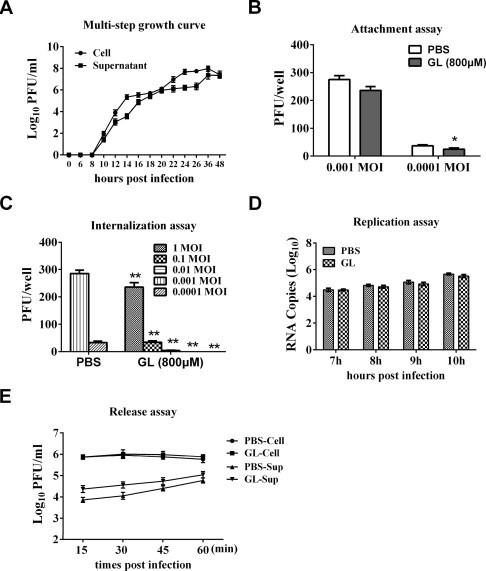
Glycyrrhizin inhibits PRRSV infection by interfering with viral internalization. (A) MARC-145 cells were infected with PRRSV at 0.1 PFU/cell. At the indicated time points after infection, cells and culture medium were collected and titrated with a plaque assay. Results are representative of three independent experiments. (B) MARC-145 cells were pre-chilled at 4 °C for 1 h, and the medium was replaced by a mixture of glycyrrhizin (800 μM) or PBS and PRRSV (0.001, 0.0001 PFU/cell). After incubation at 4 °C for additional 2 h, the mixture includes free virus and glycyrrhizin was removed. The cell monolayer was washed with ice-cold PBS three times, covered with overlay medium, incubated at 37 °C under 5% CO2 for a further 72 h before being examined by a plaque assay. Significant differences compared with PBS treated control group are denoted by *P < 0.05. (C) MARC-145 cells were pre-chilled at 4 °C for 1 h, then were incubated for another 2 h at 4 °C with PRRSV at different doses (1, 0.1, 0.01, 0.001, 0.0001 PFU/cell). The virus-containing medium was replaced by fresh medium containing glycyrrhizin (800 μM) or PBS, and the temperature was shifted to 37 °C to allow virus entry. After 3 h, the cells were carefully washed with PBS three times, and covered with overlay medium. After incubation at 37 °C for 72 h, the cells were examined by a plaque assay. **P < 0.01 (for glycyrrhizin treated group compared to PBS control with the same infectious dose of virus). (D) MARC-145 cells were incubated with PRRSV (0.1 PFU/cell) for 6 h, then the cell-free virus particles were removed, and cells were cultured in fresh medium containing glycyrrhizin (800 μM). At 7, 8, 9 and 10 hpi, the infected cells were collected for real-time RT-PCR to detect the level of negative-sense PRRSV RNA. (E) MARC-145 cells were infected with PRRSV (0.1 PFU/cell) for 18 h. Then the inoculums were removed and the cells were cultured in fresh medium containing glycyrrhizin (800 μM). At 15, 30, 45 and 60 min after medium switching, both the infected cells and culture medium were harvested and titrated by a plaque assay. Results (B), (C), (D), and (E) are the mean ± SD from three independent experiments performed in triplicate. Student’s t-test was used to analyze differences between two experimental groups. A P-value less than 0.05 was considered significant and a P-value less than 0.01 was considered highly significant.
Subsequently, a plaque reduction assay was applied to evaluate the effect of glycyrrhizin on PRRSV adsorption. Briefly, MARC-145 cells cultured in 6-well plates were pre-chilled at 4 °C for 1 h, and then the medium was replaced by a mixture of glycyrrhizin (800 μM) and PRRSV (0.001, 0.0001 PFU/cell). After incubation at 4 °C for another 2 h (to permit binding, but prevent viral internalization), the free virus was removed. The cell monolayer was washed with ice-cold phosphate-buffered saline (PBS) three times, covered with overlay medium, incubated at 37 °C under 5% CO2 for a further 72 h, and examined by a plaque assay. The results of Fig. 2B showed that glycyrrhizin reduced virus infection by 15% at 0.001 PFU/cell and approximately 30% at 0.0001 PFU/cell through inhibiting PRRSV attachment to MARC-145 cells.
Next, we evaluated the effect of glycyrrhizin on PRRSV internalization. MARC-145 cells cultured in 6-well plates were pre-chilled at 4 °C for 1 h, then were incubated for another 2 h at 4 °C with different concentrations of PRRSV (1.0, 0.1, 0.01, 0.001, 0.0001 PFU/cell). The virus-containing medium was replaced by fresh medium containing glycyrrhizin (800 μM) or PBS, and the temperature was shifted to 37 °C to allow virus entry. Three hours later, the cells were carefully washed with PBS to remove the non-internalized virus particles and glycyrrhizin or PBS and the cells were overlaid with 1.8% (w/v) Bacto agar mixed 1:1 with 2× DMEM containing 0.05 mg/ml neutral red. After incubation at 37 °C for 72 h, plaques were counted directly. The results showed that infectious virus titers were reduced by approximately 1000-fold when glycyrrhizin was added during the penetration stage (Fig. 2C), demonstrating that glycyrrhizin inhibited PRRSV internalization to MARC-145 cells. The titers of three PBS control groups (0.01, 0.1, and 1.0 PFU/cell) are not shown because of the countless viral plaques, while the viral plaques of GL-treated groups are zero at 0.001 and 0.0001 PFU/cell. Previous work has shown that glycyrrhizin suppresses virus penetration by modifying the fluidity of cellular membranes, and through this mechanism reduces HIV-1 and IAV infection (Harada, 2005, Wolkerstorfer et al., 2009). It has been demonstrated that PRRSV makes entry into MARC-145 cells through a microfilament-dependent endocytic mechanism (Kreutz and Ackermann, 1996). Thus, glycyrrhizin most likely inhibits PRRSV penetration by interfering with the cellular membranes, eventually leading to a reduction in endocytosis. Further experiments will be needed to confirm this hypothesis.
To determine whether glycyrrhizin inhibits PRRSV at the replication step, MARC-145 cells were incubated with PRRSV (0.1 PFU/cell) for 6 h, then the cell-free virus particles were removed and the cells were cultured in fresh medium containing glycyrrhizin (800 μM). At 7, 8, 9 and 10 hpi, the infected cells were collected and total RNA was isolated using Tri-Zol reagent following the manufacturer’s instructions (Invitrogen). PRRSV negative-sense RNA was quantified by real-time RT-PCR as previously described (Zhang et al., 2006). No noticeable differences in PRRSV RNA levels could be detected between cells treated with glycyrrhizin and the control cells treated with PBS, leading to the conclusion that glycyrrhizin does not inhibit PRRSV at the replication step (Fig. 2D).
To determine the role of glycyrrhizin on PRRSV release, MARC-145 cells were infected with PRRSV (0.1 PFU/cell). After approximately 18 h, the inoculums were removed and the cells were cultured in fresh medium containing glycyrrhizin (800 μM). At 15, 30, 45 and 60 min after medium switching, both the infected cells and culture medium were harvested and titrated by a plaque assay. As shown in Fig. 2E, no noticeable differences were observed in PRRSV titers between glycyrrhizin-treated and PBS-treated cells. However, compared with the PBS control, the supernatants of cells treated with glycyrrhizin had a slight increase in virus titer. We speculate that the residual glycyrrhizin in the supernatants interferes with the penetration process, resulting in a slight increase of virus titer. Taken together, these results suggest that glycyrrhizin does not inhibit PRRSV release.
Previous studies have also shown that glycyrrhizin directly inactivates HSV-1 (Pompei et al., 1979) and coxsackie virus (Wang et al., 2013b). To investigate whether glycyrrhizin exhibits direct inactivation of PRRSV, a virucidal activity assay was performed as described previously (Wang et al., 2013b). PRRSV was resuspended in 100 μl culture medium and treated with glycyrrhizin at 200, 400 and 800 μM in a 37 °C water bath for 60 min. MARC-145 cells were cultured in 6-well plates and pre-chilled at 4 °C for 1 h, then the pretreated virus (final PFU/cell = 0.001) was added (final concentrations of glycyrrhizin applied to MARC-145 cells were 20, 40 and 80 μM for glycyrrhizin-treated samples). After incubation at 4 °C for an additional 2 h, the cells were washed with ice-cold PBS, covered with overlay medium, incubated at 37 °C for a further 72 h, and examined by a plaque assay. The results showed that co-incubation of PRRSV with glycyrrhizin had no effect on the ability of PRRSV to infect MARC-145 cells (Data not shown), demonstrating that glycyrrhizin does not directly inactivate PRRSV particles.
Collectively, our work identified a new anti-PRRSV drug that mainly interferes with the internalization stage in the PRRSV life cycle. There are multiple mechanisms by which glycyrrhizin could reduce virus yields. For example, glycyrrhizin inhibits the release of infectious HCV particles and modifies HBV-related antigens expression (Matsumoto et al., 2013, Sato et al., 1996). Furthermore, it has been intravenously used for the treatment of chronic hepatitis B in Japan and improves liver function with occasional complete recovery from hepatitis (Sato et al., 1996), supporting its safety. Thus, glycyrrhizin could potentially serve as an agent for preventing PRRSV infection. Further studies in live animals will be necessary to confirm the value of inhibiting PRRSV infection in vivo.
Acknowledgements
This work was supported by the National Natural Sciences Foundation of China (31490602, 31201890, 31328023), the National Key Basic Research Program of China (973 Program) (2014CB542703), and the Keygrant Project of Chinese Ministry of Education (No. 313025).
References
- Cinatl J., Morgenstern B., Bauer G., Chandra P., Rabenau H., Doerr H.W. Glycyrrhizin, an active component of liquorice roots, and replication of SARS-associated coronavirus. Lancet. 2003;361:2045–2046. doi: 10.1016/S0140-6736(03)13615-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harada S. The broad anti-viral agent glycyrrhizin directly modulates the fluidity of plasma membrane and HIV-1 envelope. Biochem. J. 2005;392:191–199. doi: 10.1042/BJ20051069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang W., Chen X., Li Q., Li P., Zhao G., Xu M., Xie P. Inhibition of intercellular adhesion in herpex simplex virus infection by glycyrrhizin. Cell Biochem. Biophys. 2012;62:137–140. doi: 10.1007/s12013-011-9271-8. [DOI] [PubMed] [Google Scholar]
- Huang Y.W., Meng X.J. Novel strategies and approaches to develop the next generation of vaccines against porcine reproductive and respiratory syndrome virus (PRRSV) Virus Res. 2010;154:141–149. doi: 10.1016/j.virusres.2010.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karuppannan A.K., Wu K.X., Qiang J., Chu J.J., Kwang J. Natural compounds inhibiting the replication of Porcine reproductive and respiratory syndrome virus. Antiviral Res. 2012;94:188–194. doi: 10.1016/j.antiviral.2012.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kreutz L.C., Ackermann M.R. Porcine reproductive and respiratory syndrome virus enters cells through a low pH-dependent endocytic pathway. Virus Res. 1996;42:137–147. doi: 10.1016/0168-1702(96)01313-5. [DOI] [PubMed] [Google Scholar]
- Lunney J.K., Benfield D.A., Rowland R.R. Porcine reproductive and respiratory syndrome virus: an update on an emerging and re-emerging viral disease of swine. Virus Res. 2010;154:1–6. doi: 10.1016/j.virusres.2010.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsumoto Y., Matsuura T., Aoyagi H., Matsuda M., Hmwe S.S., Date T., Watanabe N., Watashi K., Suzuki R., Ichinose S., Wake K., Suzuki T., Miyamura T., Wakita T., Aizaki H. Antiviral activity of glycyrrhizin against hepatitis C virus in vitro. PLoS One. 2013;8:e68992. doi: 10.1371/journal.pone.0068992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meng X.J. Emerging and Re-emerging Swine Viruses. Transbound. Emerg. Dis. 2012 doi: 10.1111/j.1865-1682.2011.01291.x. [DOI] [PubMed] [Google Scholar]
- Murtaugh M.P., Genzow M. Immunological solutions for treatment and prevention of porcine reproductive and respiratory syndrome (PRRS) Vaccine. 2011;29:8192–8204. doi: 10.1016/j.vaccine.2011.09.013. [DOI] [PubMed] [Google Scholar]
- Nelsen C.J., Murtaugh M.P., Faaberg K.S. Porcine reproductive and respiratory syndrome virus comparison: divergent evolution on two continents. J. Virol. 1999;73:270–280. doi: 10.1128/jvi.73.1.270-280.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neumann E.J., Kliebenstein J.B., Johnson C.D., Mabry J.W., Bush E.J., Seitzinger A.H., Green A.L., Zimmerman J.J. Assessment of the economic impact of porcine reproductive and respiratory syndrome on swine production in the United States. J. Am. Vet. Med. Assoc. 2005;227:385–392. doi: 10.2460/javma.2005.227.385. [DOI] [PubMed] [Google Scholar]
- Opriessnig T., Patel D., Wang R., Halbur P.G., Meng X.J., Stein D.A., Zhang Y.J. Inhibition of porcine reproductive and respiratory syndrome virus infection in piglets by a peptide-conjugated morpholino oligomer. Antiviral Res. 2011;91:36–42. doi: 10.1016/j.antiviral.2011.04.012. [DOI] [PubMed] [Google Scholar]
- Pompei R., Flore O., Marccialis M.A., Pani A., Loddo B. Glycyrrhizic acid inhibits virus growth and inactivates virus particles. Nature. 1979;281:689–690. doi: 10.1038/281689a0. [DOI] [PubMed] [Google Scholar]
- Sato H., Goto W., Yamamura J., Kurokawa M., Kageyama S., Takahara T., Watanabe A., Shiraki K. Therapeutic basis of glycyrrhizin on chronic hepatitis B. Antiviral Res. 1996;30:171–177. doi: 10.1016/0166-3542(96)00942-4. [DOI] [PubMed] [Google Scholar]
- Snijder E.J., Kikkert M., Fang Y. Arterivirus molecular biology and pathogenesis. J. Gen. Virol. 2013;94:2141–2163. doi: 10.1099/vir.0.056341-0. [DOI] [PubMed] [Google Scholar]
- Wang D., Cao L., Xu Z., Fang L., Zhong Y., Chen Q., Luo R., Chen H., Li K., Xiao S. MiR-125b reduces porcine reproductive and respiratory syndrome virus replication by negatively regulating the NF-kappaB pathway. PLoS One. 2013;8:e55838. doi: 10.1371/journal.pone.0055838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J., Chen X., Wang W., Zhang Y., Yang Z., Jin Y., Ge H.M., Li E., Yang G. Glycyrrhizic acid as the antiviral component of Glycyrrhiza uralensis Fisch. against coxsackievirus A16 and enterovirus 71 of hand foot and mouth disease. J. Ethnopharmacol. 2013;147:114–121. doi: 10.1016/j.jep.2013.02.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolkerstorfer A., Kurz H., Bachhofner N., Szolar O.H. Glycyrrhizin inhibits influenza A virus uptake into the cell. Antiviral Res. 2009;83:171–178. doi: 10.1016/j.antiviral.2009.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Q., Gao L., Si J., Sun Y., Liu J., Cao L., Feng W.H. Inhibition of porcine reproductive and respiratory syndrome virus replication by flavaspidic acid AB. Antiviral Res. 2013;97:66–73. doi: 10.1016/j.antiviral.2012.11.004. [DOI] [PubMed] [Google Scholar]
- Yoo D., Welch S.K., Lee C., Calvert J.G. Infectious cDNA clones of porcine reproductive and respiratory syndrome virus and their potential as vaccine vectors. Vet. Immunol. Immunopathol. 2004;102:143–154. doi: 10.1016/j.vetimm.2004.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y.J., Stein D.A., Fan S.M., Wang K.Y., Kroeker A.D., Meng X.J., Iversen P.L., Matson D.O. Suppression of porcine reproductive and respiratory syndrome virus replication by morpholino antisense oligomers. Vet. Microbiol. 2006;117:117–129. doi: 10.1016/j.vetmic.2006.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]


