Highlights
-
•
We analyzed the anti-CCHFV potential of the chloroquine and chlorpromazine molecules.
-
•
They inhibited CCHFV in Vero and Huh7 cells: selectivity index ranging from 3 to 35.
-
•
We analyzed the anti-CCHV potential of the combinations.
-
•
Combinations of ribavirin and chloroquine or chlorpromazine were synergistic.
Keywords: Antiviral, Crimean-Congo hemorrhagic fever virus, Ribavirin, Chloroquine, Chlorpromazine
Abstract
Crimean-Congo hemorrhagic virus (CCHFV) causes hemorrhagic fever with high case mortality rates and is endemic in south-eastern Europe, Africa, and Asia. The limited catalog of specific treatment, highlight the necessity to look for additional therapeutic solutions.
Previous experiments suggested that CCHFV enters the cells via a clathrin dependent pathway. Therefore, we have evaluated the potential anti-CCHFV activity of several molecules targeting this entry possibility. We identified two molecules chloroquine and chlorpromazine. Neutralization and virus yield reduction assays were tested in Vero E6 and Huh7 cells on two different CCHFV strains. Several combinations, including ribavirin, were assayed to test a potential synergistic effect.
The two molecules inhibited CCHFV, and depending on the virus and the cell lines, the 50% inhibitory concentration (IC50) values for chloroquine and chlorpromazine ranged from 28 to 43 and 10.8–15.7 μM, respectively. Time-of-addition studies demonstrated that these molecules had a direct effect on CCHFV infectivity and spread. The antiviral activity of the two molecules was still effective even when added up to 6 h post-infection and up to 24 h. The selectivity index ranging from 3 to 35 lead us to evaluate combinations with ribavirin. Combinations of ribavirin and chloroquine or chlorpromazine were synergistic against CCHFV. Though the low chlorpromazine selectivity index suggests the need for a chemical improvement, our present study highlights chloroquine as the main drug having the potential for drug repurposing.
1. Introduction
Crimean-Congo hemorrhagic virus (CCHFV) is an enveloped, segmented negative-stranded RNA virus belonging to the Nairovirus genus of the Bunyaviridae family. The virus is endemic in western areas of the former Soviet Union and in southeastern and southwestern Europe; eastern and central Asia (Bente et al., 2013). Hyalomma spp. ticks are considered to be the most important vector for human transmission in the epidemiology of CCHF. Human to human transmission was documented after contact with blood and other body fluids. Human infection with CCHFV often results in severe hemorrhagic disease characterized by extensive ecchymosis, visceral bleeding, hepatic dysfunction (Ergönül, 2006) with high case mortality rates. The average mortality rate is often cited at 30% (Bente et al., 2013).
In a CCHFV infection context the current medical management is largely based on the treatment of symptoms. Currently, the only drug used to treat CCHFV infection is the ribavirin. Ribavirin antiviral therapy have to be initiated as soon as possible to be effective (Tasdelen Fisgin et al., 2009). However, there is still a debate on the ribavirin therapeutic beneficial treatment (Soares-Weiser et al., 2010). Other treatments such as type I interferon (Van Eeden et al., 1985); corticosteroid therapy (Dokuzoguz et al., 2013), or the efficient polyclonal anti-CCHFV human immunoglobulin treatment, (Vassilenko et al., 1990), were tested without removing the need of new antiviral molecules.
In the present study, we aimed at characterizing the in vitro anti CCHFV efficacy of chloroquine and chlorpromazine.
Chloroquine was used to prevent and treat malaria (Rolain et al., 2007). It was also shown that chloroquine is a broadspectrum molecule displaying an in vitro antiviral activity against a range of RNA viruses (Rolain et al., 2007). The mechanism of action of chloroquine is multiple, depending on the pathogen, it likely acts via the endosome, lysosome and golgi vesicle pH increase (Savarino et al., 2003). Chlorpromazine a well known anti-psychotic drug showed antiviral activity against adenovirus (Diaconu et al., 2010), Ebola virus (Bhattacharyya et al., 2010) and coronavirus (De Wilde et al., 2014). The mechanism of action of chlorpromazine is known to interfere with the formation of clathrin coated pits inhibiting clathrin-mediated endocytosis (Wang et al., 1993).
In this study, chloroquine and chlorpromazine were found to inhibit CCHFV replication in vitro, even if added up to 6 h post infection. Moreover when these two molecules were in combination to ribavirin, a synergistic effect was observed in Vero E6 cells. Further studies on the anti-CCHFV efficacy of these two FDA-approved drugs should be investigated in KO mice models.
2. Materials and methods
2.1. Cells and viruses
Vero E6 (African green monkey kidney, ATCC CRL-1586) and Huh7 (hepatocarcinoma cell line; CelluloNet, Cat N°120, Lyon, France) cells were grown at 37 °C in DMEM (Gibco®, Life technologies™) supplemented with 10% inactivated fetal calf serum (FCSi, Gibco®, Life technologies™), 1% NEAA 100× (Gibco®, Life technologies™) and 1% penicillin–streptomycin (PS) (10,000 U penicillin/ml; 10,000 U streptomycin/ml; Gibco®, Life technologies™).
All work involving infectious viruses was carried out in a biosafety level 4 (BSL-4) facility.
The stocks of 86–07 (IbAr10200, Nigeria) and 87–07 (ArD39554, Mauritania) CCHFV strains were prepared in Vero E6 cells and titrated by plaque formation on Vero E6 cells as previously described (Peyrefitte et al., 2010).
2.2. Toxicology study
Chloroquine (C6628; Sigma), chlorpromazine (C8138; Sigma) and ribavirin (R9644; Sigma) were dissolved in water and were stored in 100 mM stock solutions at −20 °C until use.
Confluent cell monolayers of Vero E6 and Huh7 cells were incubated in DMEM supplemented with 2.5% FCSi with different concentrations of the molecules, ranging from 0 to 1000 μM of chloroquine and chlorpromazine. Cytotoxic effects were monitored at 4, 24, 48 and 72 h of treatment. Cell viability was measured using the Cell proliferation kit (MTT; Roche) according to the manufacturer’s instructions. The 50% cytotoxic concentrations (CC50) of chloroquine and chlorpromazine were then determined. Three independent experimentations were performed in duplicate. The selectivity index (SI) which correspond to the relative effectiveness of the drugs in inhibiting viral replication compared to inducing cell death was then determined.
2.3. Inhibition of virus infection by inhibitor treatment
2.3.1. Plaque reduction assay
For CCHFV studies, Vero E6 or Huh7 cells were seeded in 12 well plates (BD Falcon) at a density of 4 × 105 cells per well. After an overnight incubation at 37 °C, infections with CCHFV (strains 86–07; 87–07) were carried out at a MOI of 0.025. Final molecule concentrations tested ranged from 0 to 250 μM (CQ) and from 0 to 50 μM (CPZ). The 250 μl of CCHFV inoculum was removed after a 1 h incubation at 37 °C then the cells were further incubated at 37 °C after the addition of 2 ml of 3.2% carboxymethylcellulose (CMC, Sigma–Aldrich) in DMEM/2.5% FCSi solution with molecules. At day 3, viral titers were determined as described by Peyrefitte et al. (2010), except for the revelation step in which True Blue (KPL) reagent was used instead of DAB.
To test the antiviral effect of chloroquine and chlorpromazine, three conditions were assayed pre-treatment (1 h before the infection step), concurrent treatment (during the virus infection step) and permanent treatment (before and during the virus infection step). Three independent experimentations were performed in duplicate.
The 50% inhibitory concentration (IC50) and the CC50 were calculated with GraphPad Prism 6 software using the non-linear regression model.
2.3.2. Effect of addition time
The antiviral effect of chloroquine and chlorpromazine on CCHFV was performed in 12 well plates. After an overnight incubation at 37 °C, Vero E6 cells (4 × 105 cells seeded per well) were infected using the 86–07 CCHFV strain at a MOI of 0.005. Final molecule concentrations were tested at 50 μM for chloroquine and 10 μM for chlorpromazine. The molecules were added after the 1 h viral adsorption step at 37 °C (t0) and then after 1, 3, 6 or 24 h post-infection (p.i.). Supernatants were harvested at 0, 1, 3, 6, 24 and 48 h p.i., then virus titers were determined by plaque reduction assay as described in 2.3.1 Section. Four independent experimentations were performed in triplicate.
2.3.3. Analysis of the anti-CCHFV effect of molecule combinations
To test if chloroquine or chlorpromazine can be combined with ribavirin, their potential synergistic effect was determined by the isobologram and combination-index methods, derived from the median-effect principle of Chou and Talalay (1984) using the CompuSyn software. Data obtained from the virus growth inhibitory experiments were used to perform these analysis. Vero E6 cells were seeded in 12 well plates (BD Falcon) at a density of 4 × 105 cells per well. After an overnight incubation at 37 °C, cells were infected using CCHFV (strains 86–07) at a MOI of 0.025. Molecules combinations were based on the comparison of their IC50. The molecule combinations were performed at a constant ratio. A 5:8 chloroquine/ribavirin and a 2:5 chlorpromazine/ribavirin combination ratio were applied with molecules concentrations ranging from 10 to 160 μM (CQ), 6.25–100 μM (ribavirin), and 2.5–40 μM (CPZ). The combinations were added concomitantly to the CCHFV infection step and maintained during all the experience. The viral inoculum was removed after 1 h at 37 °C and the cells were further incubated at 37 °C after the addition of 2 ml of 3.2% carboxymethylcellulose (CMC, Sigma Aldrich) in DMEM/2.5% FCSi solution. At 72 h p.i., viral titers were determined as described by Peyrefitte et al. (2010), except for the revelation step in which True Blue (KPL) reagent was used instead of DAB.
The combinations were tested and compared to three theoretical fractional virus inhibition values (Fa), i.e., 0.5 corresponding to a 50% theoretical inhibition, 0.75 (a 75% theoretical inhibition) and 0.9 (a 90% theoretical inhibition). Three independent experimentations were performed in duplicate.
The isobologram method is a graphical representation of the pharmacologic interaction among the molecules. When the combination given values were shown to be under the theoretical additive effects line (represented in the graph by the three segments linking the IC50, the IC75 and the IC90 of the two molecules) they were considered synergic molecules whereas values above the threshold were considered antagonistic molecules.
3. Results
3.1. Dose dependent inhibition of 86–07 and 87–07 CCHFV strains
Chloroquine was found to reduce the amount of 86–07 CCHFV infectious particles released from infected cells in a dose dependent manner displaying an IC50 of 39.4 μM (Fig. 1 a and Table 1 ). Interestingly, chloroquine efficacy was confirmed using another CCHFV strain, 87–07, showing an IC50 of 28.1 μM. An antiviral effect was also observed with chlorpromazine using the two CCHFV strains. A higher inhibiting efficacy was observed using chlorpromazine when compared to chloroquine; the IC50 ranged from 10.6 to 15.8 μM (Fig. 1b). However, due to a weaker cytotoxic effect, the chloroquine relative efficacy (SI) appeared higher than the chlorpromazine one (Table 1).
Fig. 1.
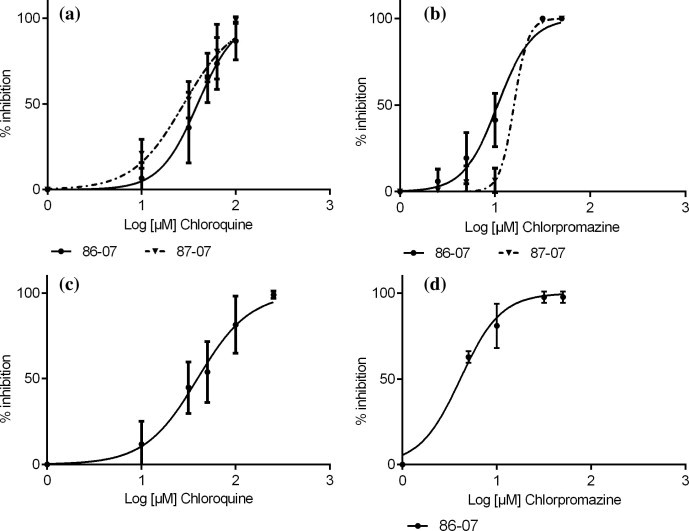
In vitro determination of IC50 by a plaque reduction assay. The 50% inhibitory concentration (IC50) for chloroquine and chlorpromazine was determined in duplicate in 12 well plates on monolayers of Vero E6 (a and b), or Huh7 (c and d) cells. Two CCHFV strains were used, 86–07 strain (circle), 87–07 strain (triangle). Plaques were enumerated after immunostaining. The dose response curves were sigmoid, and log (inhibitor) vs response curve was performed using GraphPad Prism 6, GraphPad software, www.graphpad.com.
Table 1.
IC50 and CC50 using Vero E6 and Huh7 cells and CCHFV strains 86–07 and 87–07.
| Vero E6 |
Huh7 |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CCHFV strain |
86–07 |
87–07 |
||||||||||
| Molecule | IC50 (μM) | p Value | CC50 (μM) | SI | IC50 (μM) | p Value | CC50 (μM) | SI | IC50 (μM) | p Value | CC50 (μM) | SI |
| Chloroquine | 39.4 (±0.031) | 1000 | 26.9 | 28.1 (±0.042) | nsa | 1000 | 35.6 | 43.4 | nsb | 926 | 21.3 | |
| Chlorpromazine | 10.6 (±0.031) | 30 | 2.8 | 15.8 (±0.051) | nsa | 30 | 1.9 | 4.3 | ⁎⁎,b | 30 | 7.1 | |
IC50: Inhibitory concentration (μM).
Cell viability was assayed at 1 d p.i.
SI: Selectivity index, CC50/IC50.
p Value: ns: non significative, ⁎: p < 0.05, ⁎⁎: p < 0.005.
86.07 compared to 87–07.
Vero E6 compared to Huh7.
The liver being a key target organ for CCHFV, the chloroquine and chlorpromazine efficacies were also evaluated using Huh7 hepatocytes. Infecting these cells with 86–07 CCHFV strain, the chloroquine IC50 was 43.4 μM which is close to the one showed in Vero E6 (Fig. 1c and Table 1). Interestingly, the chlorpromazine efficacy was stronger in Huh7 than in Vero E6 cells (Fig. 1d and Table 1).
3.2. Effect of the pre, concurrent and post-infection molecule addition
Permanent treatment with either chloroquine or chlorpromazine highlighted the efficacy of the two inhibitors. We investigated antiviral activity of chloroquine and chlorpromazine when added before (pre-treatment) and during (concurrent) the CCHFV infection.
The chloroquine efficacy was similar in the concurrent and permanent conditions (Table 2 ). The pre-treatment was shown not to improve chloroquine efficacy. Similar observations were obtained using chlorpromazine (Table 2).
Table 2.
Effect of molecule addition time on potency and efficacy of chloroquine and chlorpromazine.
| Conditions | Efficacy |
|||||
|---|---|---|---|---|---|---|
| Chloroquine |
Chlorpromazine |
|||||
| IC50 (μM) | SD | p Value | IC50 (μM) | SD | p Value | |
| Pretreatment | 88.9 | 0.11 | ⁎⁎,b | 16.3 | 0.08 | ⁎,b |
| Concurrent | 41.2 | 0.02 | nsa | 6.7 | 0.02 | nsa |
| Permanent | 37.2 | 0.031 | 8.5 | 0.04 | ||
IC50: Inhibitory concentration (μM).
ns: Non significative.
Permanent compared to concurrent.
Permanent compared to pre-treatment.
p < 0.05.
p < 0.005.
When, chloroquine and chlorpromazine were added at several time post infection, i.e., 1, 3, 6 and 24 h p.i. at a concentration close to the IC50, obtained in the permanent treatment conditions, at 24 h p.i. the CCHFV titers were reduced by ∼1 log10 when chloroquine was added up to 6 h p.i. (Fig. 2 a) and reduced by ∼1.5 log10 when chlorpromazine was added up to 6 h p.i. (Fig. 2b.). At 48 h p.i. CCHFV titers were still reduced when chloroquine was added up to 6 h p.i. (∼0.5 log10 reduction) and chlorpromazine was added up to 24 h p.i. (∼1 log10 reduction).
Fig. 2.
Effect of period of addition of molecules on virus yield infection. Virus yield inhibitions were performed on Vero E6 cells. Cells were infected with CCHFV 86–07, MOI 0.005. Chloroquine (a) or chlorpromazine (b) were only added during the post infection step. Five conditions were investigated; molecules were added just after the infection step (t0), 1, 3, 6, 24 h.p.i. Supernatants and cells were harvested 24 and 48 h.p.i., and the viral yield in supernatant was determined by plaque titration. When p-value <0.001 “*” was added.
3.3. Synergistic inhibition of CCHFV using ribavirin in combination to chloroquine or chlorpromazine
Because ribavirin, chloroquine and chlorpromazine are described to have distinct modes of action, we wondered whether the combination of these molecules might improve their antiviral activity. We performed assays in which Vero E6 cells were infected with CCHFV then subsequently treated with ribavirin in combination to either chloroquine or chlorpromazine at constant concentration ratios (Chou and Talalay, 1984).
As shown in Fig. 3 a, when Vero E6 cells were treated with the CQ/ribavirin combination, the observed CCHFV inhibition values were all below the additive theoretical lines. Therefore, a significant synergistic antiviral activity was observed for the chloroquine molecule in combination with ribavirin. Similar results were obtained using the ribavirin/chlorpromazine combination (Fig. 3b).
Fig. 3.
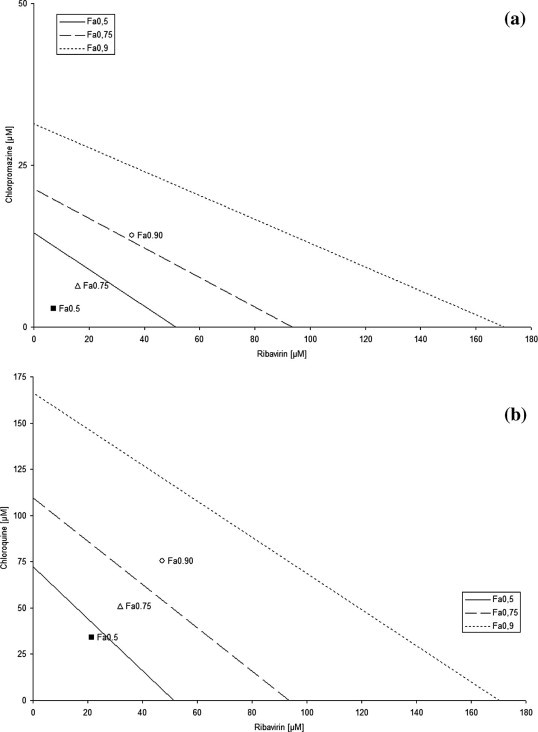
Isobolograms of in vitro drug combinations. Vero E6 cells were seeded in 12-well plates, allowed to attach for 24 h and infected with a standard dose of CCHFV with graded concentrations of ribavirin, chloroquine, chlorpromazine in combination for 72 h. After the determination of IC50 value for each drug, combinations were done using a constant drug ratio (IC50 ratio). Combination studies were done using ribavirin/CQ ratio of 5:8; ribavirin/CPZ of 5:2 over the range of drug concentrations tested. Normalized Fa0.5 (50% inhibition), Fa0.75 (75% inhibition) and Fa0.90 (90% inhibition)-isobolograms of constant dose ratios showing interaction between chloroquine and ribavirin; chlorpromazine and ribavirin. The oblique lines between X and Y axis indicates the theoretical doses that produce an additive effect (Fa0.50: straight line; Fa0.75: dash line; Fa0.90: dot line). Interaction between drugs inducing 50, 75 and 90% of inhibition are represented by square (Fa0.5), open triangle (Fa0.75) and open circle (Fa0.90). A point below the theoretical line represents a synergistic interaction, A point above the theoretical line represents antagonistic interaction.
4. Discussion
The worldwide circulation of CCHFV mainly in countries in the south of the 50th parallel north (Estrada-Pena et al., 2012) together with the CCHFV human case number increase in Turkey (Ince et al., 2014).and the lack of efficient specific treatment emphasized the need of new antiviral molecules development.
Despite a high mortality rate, the current medical management is largely supportive. Until now ribavirin is the only antiviral treatment implemented in CCHF, which is still debated (Soares-Weiser et al., 2010). A strategy to reduce the time of drug development could be the recycling use of existing drugs in other conditions. In this work, we took advantage of the CCHFV entry pathway discovery (Simon et al., 2009, Garrison et al., 2013). We investigated the anti CCHFV activity of approved molecules known to inhibit endocytic pathways. From initial in vitro antiviral testing (data not shown), we have identified the antiviral effect of two molecules known to inhibit the clathrin/pH dependent endocytic pathway.
The results presented in this in vitro study showed that chloroquine and chlorpromazine are effective antiviral molecules for CCHFV. The IC50 of chloroquine and chlorpromazine inhibition of CCHFV were 39.4 and 10.6 μM respectively.
Chloroquine was found to be the most selective CCHFV inhibitor with a selective index of more than 20. Chloroquine is a weak base that is known to affect acid vesicles leading to dysfunction of several enzymes including acid hydrolases inhibiting post-translational modification (Randolph et al., 1990), therefore potentially affecting pH-dependent entry of viruses (Gonzalez-Dunia et al., 1998).
Chloroquine has been in use for many years for the treatment of malaria and for the long term prophylaxis of inflammatory diseases. Ocular toxicity (Kazi et al., 2013), cardiotoxicity (Tönnesmann et al., 2013) and oral mucosa hyperpigmentation (De Andrade et al., 2013) are known complication of long term use of chloroquine. Reversible side effect can be observed for mucosal pigmentation after months of discontinuation of chloroquine treatment (Armor et al., 2002). Cardiomyopathy side effect can be lessened after cessation of chloroquine treatment and prescription of a combined therapy of candesartan and carvedilol treatment (Lopez-Ruiz and Uribe, 2014). Contrary to hyperpigmentation and cardiomyopathy, retinopathy is an irreversible side effect of long term chloroquine treatment and can appear even after usage of chloroquine has stopped (Kazi et al., 2013). Nevertheless, daily dose of the chloroquine seems to be relevant for the development of ocular side effect, the risk of retinopathy is lowered if this dose not exceed 4 mg/kg/day (Lerman et al., 2009) or is minimal when the daily dose not exceed 250 mg (Armor et al., 2002).
Previously, chloroquine was proposed to be used against several viral infections (Rolain et al., 2007). The antiviral efficacy of this molecule was shown on different cell culture with IC50 related to each pathogens. Chloroquine was shown to inhibit Chikungunya virus (IC50 10 μM) (Khan et al., 2010), Dengue virus (Farias et al., 2013), coronavirus (IC50 8.8 μM) (Keyaerts et al., 2004), MERS-CoV (IC50 3.0 μM) (De Wilde et al., 2014), influenza virus (IC50 3.6 μM) (Ooi et al., 2006), or Henipavirus (IC50 2 μM) (Porotto et al., 2009). Chloroquine was also used to inhibit HIV 1 in lineages of monocytes and lymphocytes (Sperber et al., 1993) which are known to be HIV 1 target cells. In adults a 10 mg/kg CQ oral dose is followed by a 263 ng/ml plasma peak concentration (Walker et al., 1983), which is close to the chloroquine IC50. Moreover chloroquine is known to accumulate in CCHFV target organs, such liver and spleen (IPCS Inchem). In this study, a similar chloroquine SI was assessed in two permissive cells (Vero E6) and human target cells (Huh7). The chloroquine post infection addition was inefficient for influenza viruses (Di Trani et al., 2007), while was efficient 2 h post CHIKV infection (Khan et al., 2010). In the same way, CCHFV titer was still reduced when chloroquine was added up to 6 h p.i., suggesting that chloroquine blocks a post entry step in CCHFV replication cycle. In this case, rapid elevation of endosomal pH and abrogation of virus endosome fusion might be the primary mechanism by which virus infectivity is inhibited by chloroquine (Poole and Ohkuma, 1981). The abolishment of the released of sialyltransferase and the Trans-Golgi Network acidification could account for another inhibitory mechanism (Berger et al., 2001). Plasma chloroquine concentration might be influenced by molecule dosage, but a 10 mg/kg first dose is rapidly detectable with a plasma peak concentration until 263 ng/ml (Walker et al., 1983), with a chloroquine accumulation in CCHFV organ targets, such liver or spleen.
The second molecule tested, chlorpromazine, an approved psychotic drug, was described by others to inhibit the clathrin dependent pathway by reducing the number of coated pits-associated receptors at the cell surface (Wang et al., 1993). Chlorpromazine can be associated with pigmentation of the oral mucosa (Seymour and Rudralingham, 2008). But hepatotoxicity in humans characterized by elevated serum alkaline phosphatases or biliary cirrhosis is the main chlorpromazine side effect (Gandhi et al., 2013). Hyperglycaemia and hypothermia are also side effect related with chlorpromazine treatment (Bonaccorsi et al., 1964).
Records of the chlorpromazine used as an antiviral molecule were associated to adenovirus (Diaconu et al., 2010), Ebola virus (Bhattacharyya et al., 2010) and more recently, coronavirus treatment (De Wilde et al., 2014). Depending on chlorpromazine posology, its plasma concentration can reach up to 300 ng/ml in treated patients (Rivera-Calimlim et al., 1979).
In this study, the chlorpromazine inhibitory effect was observed in all treatment conditions. Interestingly, chlorpromazine exhibited a marked inhibitory effect on Huh7 cell line, whose caveolae-1 pathway is missing, strongly suggesting the involvement of clathrin mediated endocytosis (Blanchard et al., 2006). The CCHFV titer was still markedly reduced when chlorpromazine was added up to 24 h p.i., suggesting another post entry inhibitory effect, likely at the assembly and budding already identified for Sindbis and vesicular stomatitis virus (Schlesinger and Cahill, 1989). However the low chlorpromazine selectivity index diminished its potential use in clinic particularly if this drug is used alone.
Ribavirin and chloroquine or chlorpromazine combination assays demonstrated synergistics effects in all tested conditions. It was expected because ribavirin, chloroquine and chlorpromazine had different antiviral mechanisms. Thus, these experiments suggest that these combinatorial treatment could represent a strategy to control CCHFV infection.
Such promising in vitro results should be confirmed by a drug efficacy evaluation in animal models. Since 2010, two mice models IFNAR−/− and STAT1−/− were used to assess a ribavirin effect against CCHFV infection (Bereczky et al., 2010, Bente et al., 2010). The observed effects were 60% survivals in STAT1−/− mice and 14% in IFNAR−/− mice (Oestereich et al., 2014). However, Oestereich et al. (2014) demonstrated that ribavirin was not able to prevent the lethal pathophysiological cascade (Oestereich et al., 2014). It would be relevant to investigate in the near future these molecules compared to others in protective assays alone or in combination to ribavirin using KO mouse models.
Acknowledgments
This work was partly funded by IRBA, DGA.
References
- Armor M.F., Carr R.E., Easterbrook M., Farjo A.A., Mieler W.F. Recommendations on screening for chloroquine and hydroxychloroquine retinopathy: a report by the American Academy of Ophthalmology. Am. Acad. Ophthalmol. 2002;109(7):1377–1382. doi: 10.1016/s0161-6420(02)01168-5. [DOI] [PubMed] [Google Scholar]
- Bente D.A., Alimonti J.B., Shieh W.-J., Camus G., Ströher U., Zaki S., Jones S.M. Pathogenesis and immune response of Crimean-Congo hemorrhagic fever virus in STAT-1 knockout mouse model. J. Virol. 2010;84(21):1089–11100. doi: 10.1128/JVI.01383-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bente D.A., Forrester N.L., Watts D.M., McAuley A.J., Whitehouse C.A., Bray M. Crimean-Congo hemorrhagic fever: history, epidemiology, pathogenesis, clinical syndrome and genetic diversity. Antiviral Res. 2013;100:159–189. doi: 10.1016/j.antiviral.2013.07.006. [DOI] [PubMed] [Google Scholar]
- Bereczky S., Lindegren G., Karlberg H., Akerström A., Klingström J., Mirazimi A. Crimean-Congo hemorrhagic fever virus infection is lethal for adult type I interferon receptor-knockout mice. J. Gen. Virol. 2010;91:1473–1477. doi: 10.1099/vir.0.019034-0. [DOI] [PubMed] [Google Scholar]
- Berger E.G., Berger B., Hochli M., Dinter A. Colocalization of beta 1,4 galactosyltransferase with mannose 6-phosphate receptor in monensin-induced TGN derived structures. Histochem. Cell Biol. 2001;115(2):157–168. doi: 10.1007/s004180000231. [DOI] [PubMed] [Google Scholar]
- Bhattacharyya S., Warlfield K.L., Ruthel G., Bavari S., Aman M.J., Hope T.J. Ebola virus uses clathrin-mediated endocytosis as an entry pathway. Virology. 2010;401:18–28. doi: 10.1016/j.virol.2010.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blanchard E., Belouzard S., Goueslain L., Wakita T., Dubuisson J., Wychowski C., Rouillé Y. Hepatitis C virus entry depends on clathrin-mediated endocytosis. J. Virol. 2006;80(14):6964–6972. doi: 10.1128/JVI.00024-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonaccorsi A., Garattini S., Jori A. Studies on the hyperglycaemia induced by chlorpromazine in rats. Br. J. Pharmacol. Chemother. 1964;23(1):93–100. doi: 10.1111/j.1476-5381.1964.tb01570.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chou T.C., Talalay P. Quantitative analysis of dose effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv. Enzyme Regul. 1984;22:27–55. doi: 10.1016/0065-2571(84)90007-4. [DOI] [PubMed] [Google Scholar]
- De Andrade B.A., Fonseca F.P., Pires F.R., Mesquita A.T., Falci S.G., Dos Santos Silva A.R., Vargas P.A., Jorge J., de Almeida O.P. Hard palate hyperpigmentation secondary to chronic chloroquine therapy: report of five cases. J. Cutan. Pathol. 2013;40(9):833–838. doi: 10.1111/cup.12182. [DOI] [PubMed] [Google Scholar]
- De Wilde A.H., Jochmans D., Posthuma C.C., Zevenhoven-Dobbe J.C., Van Nieuwkoop S., Bestebroer T.M., Van Den Hoogen B.G., Neyts J., Snijder E.J. Screening of an FDA-approved compound library identifies four small-molecule inhibitors of Middle East respiratory syndrome coronavirus replication in cell culture. Antimicrob. Agents Chemother. 2014;58(8):4875–4884. doi: 10.1128/AAC.03011-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Trani L., Savarino A., Campitelli L., Norelli S., Puzelli S., D’Ostilio D., Vignolo E., Donatelli I., Cassone A. Different pH requirements are associated with divergent inhibitory effects of chloroquine on human and avian influenza A viruses. Virol. J. 2007;4(39):1–8. doi: 10.1186/1743-422X-4-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diaconu I., Cerullo V., Escutenaire S., Kanerva A., Bauerschmitz G.J., Hernadez-Alcoceba R., Pesonen S., Hemminki A. Human adenovirus replication in immunocompetent Syrian hamsters can be attenuated with chlorpromazine or cidofovir. J. Gene Med. 2010;12:435–445. doi: 10.1002/jgm.1453. [DOI] [PubMed] [Google Scholar]
- Dokuzoguz B., Celikbas A.K., Gök S.E., Baykam N., Eroglu M.N., Ergönul O. Severity scoring index for Crimean-Congo hemorrhagic fever and the impact of ribavirin and corticosteroids on fatality. Clin. Infect. Dis. 2013;57(9):1270–1274. doi: 10.1093/cid/cit527. [DOI] [PubMed] [Google Scholar]
- Ergönül O. Crimean-Congo haemorrhagic fever. Lancet Infect. Dis. 2006;6:203–214. doi: 10.1016/S1473-3099(06)70435-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Estrada-Pena A., Palomar A.M., Santibanez P., Sanchez N., Habela M.A., Portillo A., Romero L., Oteo J.A. Crimean-Congo hemorrhagic fever virus in ticks, Southwestern Europe, 2010. Emerg. Infect. Dis. 2012;18(1):179–180. doi: 10.3201/eid1801.111040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farias K.J.S., Machado P.R.L., Da Fonseca B.A.L. Chloroquine inhibits dengue virus type 2 replication in Vero cells but not in C6/36 cells. Sci. World J. 2013;2013:282734. doi: 10.1155/2013/282734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gandhi A., Guo T., Shah P., Moorthy B., Ghose R. Chlorpromazine induced hepatotoxicity during inflammation is mediated by TIRAP-dependent signaling pathway in mice. Toxicol. Appl. Pharmacol. 2013;266(3):430–438. doi: 10.1016/j.taap.2012.11.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garrison A.R., Radoshitzky S.R., Kota K.P., Pegoraro G., Ruthel G., Khun J.H., Altamura L.A., Kwilas S.A., Bavari S., Haucke V., Schmaljohn C.S. Crimean-Congo hemorrhagic fever virus utilizes a clathrin and early endosome dependent entry pathway. Virology. 2013;444:45–54. doi: 10.1016/j.virol.2013.05.030. [DOI] [PubMed] [Google Scholar]
- Gonzalez-Dunia D., Cubitt B., De la Torre J.C. Mechanism of borna disease virus entry into cells. J. Virol. 1998;72(1):783–788. doi: 10.1128/jvi.72.1.783-788.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ince Y., Yasa C., Metin M., Sonmez M., Meram E., Benkli B., Ergonul O. Crimean-Congo hemorrhagic fever infections reported by ProMED. Int. J. Infect. Dis. 2014;26:44–46. doi: 10.1016/j.ijid.2014.04.005. [DOI] [PubMed] [Google Scholar]
- IPCS Inchem. <http://www.inchem.org/documents/pims/pharm/chloroqu.htm> (accessed 05.22.14).
- Kazi M.S., Saurabh K., Rishi P., Rishi E. Delayed onset chloroquine retinopathy presenting 10 years after long term usage of chloroquine. Middle East Afr. J. Ophthalmol. 2013;20(1):89–91. doi: 10.4103/0974-9233.106404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keyaerts E., Vijgen L., Maes P., Neyts J., Van Ranst M. In vitro inhibition of severe acute respiratory syndrome coronavirus by chloroquine. Biochem. Biophys. Res. Commun. 2004;323:264–268. doi: 10.1016/j.bbrc.2004.08.085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan M., Santhosh S.R., Tiwari M., Lakshmana Rao P.V., Parida M. Assessment of in vitro prophylactic and therapeutic efficacy of chloroquine against chikugunya virus in Vero cells. J. Med. Virol. 2010;82:817–824. doi: 10.1002/jmv.21663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lerman M.A., Karimbux N., Guze K.A., Woo S.B. Pigmentation of the hard palate. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2009;107(1):8–12. doi: 10.1016/j.tripleo.2008.07.022. [DOI] [PubMed] [Google Scholar]
- Lopez-Ruiz N., Uribe C.E. Chloroquine cardiomyopathy: beyond ocular adverse effects. BMJ Case Rep. 2014 doi: 10.1136/bcr-2014-205751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oestereich L., Rieger T., Neumann M., Bernreuther C., Lehmann M., Krasemann S., Wurr S., Emmerich P., De Lamballerie X., Ölschlager S., Günther S. Evaluation of antiviral efficacy of ribavirin, arbidol, and T-705 (favipiravir) in a mouse model for Crimean-Congo hemorrhagic fever. PLoS Negl. Trop. Dis. 2014;8(5):e2804. doi: 10.1371/journal.pntd.0002804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ooi E.E., Chew J.S.W., Loh J.P., Chua R.C.S. In vitro inhibition of human influenza A virus replication by chloroquine. Virol. J. 2006;3(39):1–3. doi: 10.1186/1743-422X-3-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peyrefitte C.N., Perret M., Garcia S., Rodrigues R., Bagnaud A., Lacote S., Crance J.-M., Vernet G., Garin D., Bouloy M., Paranhos-Baccala G. Differential activation profiles of Crimean-Congo hemorrhagic fever virus and Dugbe virus infected antigen presenting cells. J. Gen. Virol. 2010;91:189–198. doi: 10.1099/vir.0.015701-0. [DOI] [PubMed] [Google Scholar]
- Poole B., Ohkuma S. Effect of weak bases on the intralysosomal pH in mouse peritoneal macrophages. J. Cell Biol. 1981;90:665–669. doi: 10.1083/jcb.90.3.665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Porotto M., Orefice G., Yokoyama C.C., Mungall B.A., Realubit R., Sganga M.L., Aljofan M., Whitt M., Glickman F., Moscona A. Simulating Henipavirus multicycle replication in a screening assay leads to identification of a promising candidate for therapy. J. Virol. 2009;83(10):5148–5155. doi: 10.1128/JVI.00164-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Randolph V.B., Winkler G., Stollar V. Acidotropic amines inhibit proteolytic processing of flavivirus prM protein. Virology. 1990;174(2):450–458. doi: 10.1016/0042-6822(90)90099-d. [DOI] [PubMed] [Google Scholar]
- Rivera-Calimlim L., Griesbach P.H., Perlmutter R. Plasma chlorpromazine concentrations in children with behavioral disorders and mental illness. Clin. Pharmacol. Ther. 1979;26(1):114–121. doi: 10.1002/cpt1979261114. [DOI] [PubMed] [Google Scholar]
- Rolain J.M., Colson P., Raoult D. Recycling of chloroquine and its hydroxyl analogue to face bacterial, fungal and viral infections in the 21st century. Int. J. Antimicrob. Agents. 2007;30:297–308. doi: 10.1016/j.ijantimicag.2007.05.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Savarino A., Boelaert J.R., Cassone A., Majori G., Cauda R. Effects of chloroquine on viral infections: an old drug against today’s diseases? Lancet Infect. Dis. 2003;3:722–727. doi: 10.1016/S1473-3099(03)00806-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlesinger M.J., Cahill D. Verapamil and chlorpromazine inhibit the budding of Sindbis and vesicular stomatitis viruses from infected chicken embryo fibroblasts. Virology. 1989;168(1):187–190. doi: 10.1016/0042-6822(89)90421-2. [DOI] [PubMed] [Google Scholar]
- Seymour R.A., Rudralingham M. Oral and dental adverse drug reactions. Periodontology 2000. 2008;46:9–26. doi: 10.1111/j.1600-0757.2008.00226.x. [DOI] [PubMed] [Google Scholar]
- Simon M., Johansson C., Mirazimi A. Crimean-Congo hemorrhagic fever virus entry and replication is clathrin, pH, and cholesterol dependent. J. Gen. Virol. 2009;90:210–215. doi: 10.1099/vir.0.006387-0. [DOI] [PubMed] [Google Scholar]
- Soares-Weiser K., Thomas S., Thomson G., Garner P. Ribavirin for Crimean-Congo hemorrhagic fever: systematic review and meta-analysis. BMC Infect. Dis. 2010;10:207. doi: 10.1186/1471-2334-10-207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sperber K., Kalb T.H., Stecher V.J., Banerjee R., Mayer L. Inhibition of human immunodeficiency virus type 1 replication by hydroxychloroquine in T cells and monocytes. AIDS Res. Hum. Retroviruses. 1993;9(1):91–98. doi: 10.1089/aid.1993.9.91. [DOI] [PubMed] [Google Scholar]
- Tasdelen Fisgin N., Ergonul O., Doganci L., Tulek N. The role of ribavirin in the therapy of Crimean-Congo hemorrhagic fever: early use is promising. Eur. J. Clin. Microbiol. Infect. Dis. 2009;28:929–933. doi: 10.1007/s10096-009-0728-2. [DOI] [PubMed] [Google Scholar]
- Tönnesmann E., Kandolf R., Lewalter T. Chloroquine cardiomyopathy – a review of the literature. Immunopharmacol. Immunotoxicol. 2013;35(3):434–442. doi: 10.3109/08923973.2013.780078. [DOI] [PubMed] [Google Scholar]
- Van Eeden P.J., Van Eeden S.F., Joubert J.R., King J.B., Van de Wal B.W., Michell W.L. A nosocomial outbreak of Crimean-Congo haemorrhagic fever at Tygerberg Hospital. Part II. Management of patients. S. Afr. Med. J. 1985;68(10):718–721. [PubMed] [Google Scholar]
- Vassilenko S.M., Vassilev T.L., Bozadjiev L.G., Bineva I.L., Kazarov G.Z. Specific intravenous immunoglobulin for Crimean-Congo haemorrhagic fever. Lancet. 1990;335(8692):791–792. doi: 10.1016/0140-6736(90)90906-l. [DOI] [PubMed] [Google Scholar]
- Walker O., Dawodu A.H., Adeyokunnu A.A., Salako L.A., Alvan G. Plasma chloroquine and desethylchloroquine concentrations in children during and after chloroquine treatment for malaria. Br. J. Clin. Pharmacol. 1983;16(6):701–705. doi: 10.1111/j.1365-2125.1983.tb02244.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang L.-H., Rothberg K.G., Anderson R.G.W. Mis-assembly of clathrin lattices on endosomes reveals a regulatory switch for coated pit formation. J. Cell Biol. 1993;123(5):1107–1117. doi: 10.1083/jcb.123.5.1107. [DOI] [PMC free article] [PubMed] [Google Scholar]


