Abstract
Porcine epidemic diarrhea virus (PEDV) is the predominant cause of severe entero-pathogenic diarrhea in swine. The lack of effective therapeutical treatment underlines the importance of research for new antivirals. In this study, we identified Q7R, which actively inhibited PEDV replication with a 50% inhibitory concentration (IC50) of 0.014 μg/mL. The 50% cytotoxicity concentration (CC50) of Q7R was over 100 μg/mL and the derived therapeutic index was 7142. Several structural analogues of Q7R, quercetin, apigenin, luteolin and catechin, also showed moderate anti-PEDV activity. Antiviral drugs and natural compounds revealed ribavirin, interferon-α, coumarin and tannic acid have relative weaker efficacy compared to Q7R. Q7R did not directly interact with or inactivate PEDV particles and affect the initial stage of PEDV infection by interfering of PEDV replication. Also, the effectiveness of Q7R against the other two viruses (TGEV, PRCV) was lower compared to PEDV. Q7R could be considered as a lead compound for development of anti-PEDV drugs to may be used to during the early stage of PEDV replication and the structure-activity data of Q7R may usefully guideline to design other related antiviral agents.
Keywords: Porcine epidemic diarrhea virus, Antiviral activity, Quercetin 7-rhamnoside, Houttuynia cordat
1. Introduction
Porcine epidemic diarrhea virus (PEDV), family Coronaviridae, causes porcine epidemic diarrhea, an enteric disease characterized by acute watery diarrhea, dehydration, vomiting, and high mortality in nursery piglets (Debouck and Pensaert, 1980). Infection with this virus has become a serious issue in the swine industry and outbreaks have lead to serious economic losses in many countries (Hofmann and Wyler, 1987). Unfortunately, now there are no effective commercial vaccines or specific treatments available and the only measures to control the disease are those directed to preventing the entrance of the virus on the farm.
Medicinal plants are increasingly being pursued as suitable alternative sources for discovery of antiviral agents (Briskin, 2000, Cowan, 1999, Jassim and Naji, 2003, Vlietinck and Vanden Berghe, 1991, Williams, 2001). Recent studies have demonstrated that Houttuynia cordata Thunb. (Saururaceae) is effective in treating anaphylaxis and cancer (Kwon et al., 2003, Li et al., 2005, Lu et al., 2006). It has also been shown that the essential oil prepared from fresh plants of H. cordata possessed direct inhibitory activity against herpes simplex virus type 1 (HSV-1), influenza virus and human immunodeficiency virus type 1 (HIV-1), without showing cytotoxicity (Lu et al., 2006). In particular, quercetin, a flavonoid present in H. cordata, has been reported to have inhibitory effects on several viruses (Mucsi and Pragai, 1985). Although a variety of pharmacological activities associated with chemicals in H. cordata has been demonstrated, antiviral activities against PEDV have not been reported.
In this study we examine the positional effects of the hydroxyl group within basic flavonoid structure, and the type and position of attached sugars against PEDV replication. Furthermore, to elucidate the action of Q7R on PEDV multiplication in more detail, we investigated the effect of Q7R on the infection cycle of PEDV through time–of–addition study, RT-PCR analysis, effects of Q7R on the infectivity of PEDV particles.
2. Materials and methods
2.1. Compounds
Tamiflu (F. Hofmann-La Roche Ltd., Switzerland), relenza (Laboratoire GlaxoSmithKline, France) and lamivudine (GlaxoSmithKline Australia Pty Ltd., Australia) were purchased from a pharmacy in Korea as prescribed by a medical doctor. Acriflavine, acycloguanosine, azidovudine caffeine, cumarin, glycyrrhizin, interferon-α, riboflavin, tannic acid, quercetin, apigenin, luteolin, catechin, quercetrin, genistin, hesperidin, rutin and sulforhodamine B (SRB) were purchased from Sigma–Aldrich (St. Louis, MO, USA). All chemicals were a reagent grade.
2.2. Viruses and cell lines
Vero (african green monkey kidney cell line; ATCC CCR-81), ST (pig testis cell line; ATCC CRL-1746), TGEV (transmissible gastroenteritis virus; ATCC VR-763) and PRCV (porcine respiratory coronavirus; ATCC VR-2384) were kindly provided by ATCC (American Type Culture Collection, Manassas, VA, USA). PEDV CV 777 (porcine epidemic diarrhea virus) was obtained from national veterinary research & quarantine service in Korea. Vero and ST cell lines were maintained in minimal essential medium (MEM) supplemented with 10% fetal bovine serum (FBS) and 0.01% antibiotic-antimycotic. Antibiotic-antimycotic, trypsin-EDTA, FBS and MEM were supplied by Gibco BRL (Grand Island, NY, USA). The tissue culture plates were purchased from Falcon (BD Biosciences, NJ, USA).
2.3. Bioassay-guided fractionation and isolation of active principles
The aerial parts of H. cordata were collected at Gyeongsangnam-do Agricultural Research & Extension Services, Korea, in August 2004. The dried and ground materials (1 kg) were extracted three times with MeOH (3 × 10 L) at room temperatures for 3 days, and this extract was performed to SRB-based cytotoxicity and antiviral activity assay (Table 1 ). The MeOH extract (247 g; IC50, 82.2 μg/mL; CC50, 1036.0 μg/mL) was separated into two fractions using EtOAc as the non-aqueous phase. The concentrated EtOAc fraction (16.0 g; IC50, 4.8 μg/mL; CC50, 62.1 μg/mL) was loaded on a silica gel (Merck, 230–400 mesh, 300 g) column, and eluted with a step-gradient of CHCl3:MeOH (100:0, 98.5:1.5, 97:3, 93.75:6.25, 87.5:12.5, 25:75, 50:50 and 0:100; each 1 L) to afford eight fractions. The active fraction (CHCl3:MeOH, 50:50 fraction; 7.97 g; IC50, <1 μg/mL; CC50, >100 μg/mL) was separated using Sephadex LH-20 (Amersham pharmacia biotech AB, Sweden, 18–111 mesh, 200 g), eluting successively with 80% methanol/water (v/v = 80/20; each 0.02 L), and 70 fractions were obtained. For further purification, the active fraction (combination of fractions from 41 to 43, 1.22 g; IC50, <1 μg/mL; CC50, >100 μg/mL) was subjected to analytical HPLC (YMC-Pack ODS-MA; i.d. 250 mm × 10 mm; eluent, 25% aq. Acetonitrile; flow rate, 1.5 mL/min; UV, 220 nm), and gave the pure compounds 1 (840 mg; IC50, 0.014 μg/mL; CC50, >100 μg/mL). Based on electron spray ionization-mass spectrometry, 1H NMR and 13C NMR spectral data, the purified compound was verified as quercetin 7- rhamnoside (Awaad et al., 2006).
Table 1.
Antiviral activity of Houttuynia cordata aerial part-derived materials against porcine epidemic diarrhea virus in Vero cells.
| Test material | IC50a | CC50b | TIc |
|---|---|---|---|
| Methanolic extract | 82.2 | 1036.0 | 12.6 |
| Haxane fraction | 4.5 ± 0.53 | 64.7 | 14.4 |
| Ethyl acetate fraction | 4.8 ± 0.37 | 62.1 | 13.1 |
| Butanol fraction | 46.3 ± 7.9 | >100 | >2.2 |
Results are presented as the mean IC50 values obtained from three independent experiments carried out in triplicate ±S.D.
Concentration required to inhibit virus-induced CPE by 50% (μg/mL).
Concentration required to reduce cell growth by 50% (μg/mL).
Therapeutic index = CC50/IC50.
2.4. Assays of antiviral activity
One day before infection, Vero cells were seeded onto a 96-well culture plate at a concentration of 2 × 104 cells per well. Next day, medium was removed and then washed with 1 × phosphate buffered saline (PBS). Infectivity of virus stock was determined by the SRB method using cytopathic effect (CPE) reduction and was determined as infectivity of the virus by SRB ID50 (50% infective dose). Following this, 0.09 mL of diluted virus suspension of PEDV containing CCID50 (50% cell culture infective dose) of the virus stock to produce a appropriate cytopathic effects within 2 days after infection and 0.01 mL of medium supplemented with typsin-EDTA containing an appropriate concentration of the compounds were added. The antiviral activity of each test material was determined with a 10-fold diluted concentration ranging from 0.1 to 100 μg/mL. Three wells were used as virus controls (virus-infected non-drug-treated cells) while three wells were used as cell controls (non-infected non-drug-treated cells). The culture plates were incubated at 37 °C in 5% CO2 for 2 days. After washing 1 times with 1 × PBS, 100 μl of cold (−20 °C) 70% acetone were added to each well and left for 30 min at −20 °C. 70% acetone was removed and 96-well plates were left at dry oven for 30 min. 100 μl of 0.4% (w/v) SRB in 1% acetic acid solution were added to each well and left at room temperature for 30 min. Unbound SRB was removed and the plates were washed 5 times with 1% acetic acid before oven drying and were then left in a dry oven for 1 day. Bound SRB was solubilized with 100 μl of 10 mM unbuffered tris-base solution and plates were left on a table for 30 min. The absorbance was read at 540 nm by using a VERSAmax microplate reader (Molecular Devices, Palo Alto, CA, USA) with a reference absorbance at 620 nm. To calculate the IC50 values, the results were transformed to percentage of controls and the IC50 values were graphically obtained from the dose-response curves. The percent protection achieved by the test compound in PEDV-infected cells was calculated by the following formula:
where (ODt)PEDV is the optical density measured with a given concentration of the test compound in PEDV-infected cells; (ODc)PEDV is the optical density measured for the control untreated PEDV-infected cells; and (ODc)mock is the optical density measured for control untreated mock-infected cells. The concentration achieving 50% protection according to the formula above was defined as the 50% inhibitory concentration (IC50). The therapeutic index was defined as CC50/IC50.
To test the effect of Q7R on the infectivity of PEDV particles, PEDV was pre-incubated with Q7R of 10 μg/mL for 1 h at 4 °C and Vero cells were infected with pretreated or untreated PEDV for 1 h at 37 °C. Unbound virus was removed and washed 2 times with PBS, ant then cells were incubated in infection medium supplemented with or without Q7R of 10 μg/mL at 37 °C. Antiviral activity was determined by SRB assay after 2 days.
The time-of-addition effect of Q7R was examined according to previously described procedures with minor modifications (Chiang et al., 2003a, Chiang et al., 2003b). Vero cells were seeded onto 96-well culture plates at density of 2 × 104 cells per well and incubated for 1 day. After washing with 1 × PBS, each 10 μg/mL of the Q7R and ribavirin was then added onto the cells at either before (−1 h), during (0 h) or after (1, 2, 4, 6, 8, 12 and 24 h) the period of PEDV infection. After 2 days, antiviral activity was carried out as above described. Ribavirin was used as positive control.
2.5. Cytotoxicity assay
The cytotoxicity was evaluated by modified the SRB assay previously described (Lin et al., 1999). Vero cells were seeded onto a 96-well culture plate at a concentration of 2 × 104 cells per well. Next day, medium was removed and the 96-well plates were replaced with media containing the serially diluted compounds and the cells were further incubated for 48 h. The culture medium was removed and washed with 1 × PBS. The next step was conducted by antiviral activity assay obove described. To calculate the CC50 values, the results were transformed to percentage of controls and the CC50 values were graphically obtained from the dose-response curves.
2.6. RT-PCR analysis
Vero cells were seeded onto a 96-well culture plate at a concentration of 2 × 104 cells per well. The next day, medium was removed and the cells were washed with 1 × PBS. Then, 0.09 mL of diluted virus suspension and 0.01 mL of medium supplemented with typsin-EDTA containing Q7R or ribavirin of 10 μg/mL were added. After incubation at 37 °C in 5% CO2 for 24 or 48 h, the next step was performed. Total RNA was extracted from the cultured cells by the acid guanidinium thiocyanate-phenol-chloroform extraction method (Chomczynski and Mackey, 1995).
Total RNA was dissolved in 0.1% diethylpyrocarbonate (Sigma)-treated water. The amount of RNA was determined by measuring spectrometric absorbance at 260 nm. To check purity of the 18S ribosomal RNA, RNA of 2 μg/mL were loaded on a 0.9% RNA gel in 1UMOPS buffer, and the 18S ribosomal RNA bands were compared.
First-strand cDNA was synthesized from total RNA in an RNase-free condition. The reaction was performed with 5 μg of total RNA using a ProSTAR1 First-Strand RT-PCR kit (Stratagene, La Jolla, CA, USA), according to the manufacturer's instructions. PCR was performed in a GeneAmp PCR system 2700 (PerkinElmer/Cetus, Norwalk, CT, USA) using the first-strand cDNA and Taq polymerase (TaKaRa Shuzo, Kyoto, Japan) as follows for each primer: membrane (M) gene of PEDV, 5′-CGGTTCTATTCCCGTTGATG-3′ and 5′-CCACAACCGAATGCTATTGA-3′; β-actin, 5′-GCCATGTACGTTGCTATCCAGGCTG-3′ and 3′-AGCCGTGGCCATCTCTTGCTCGAAG- 5′. PCR-amplified products were separated on 1.5% agarose gels containing 0.1 μg/mL ethidium bromide and visualized under UV light.
3. Results
3.1. Measurement of antiviral activities of various compounds and structure-activity relationship analysis for flavonoids
Current antiviral drugs, natural compounds and flavonoids were further studied for their inhibitory effects on replication of the PEDV and cytotoxicity in Vero cells, among which ribavirin, tannic acid, coumarin and interferon-α exhibited the activities with IC50 of 4.1, 47.4, 9 μg/mL and 0.52 unit, respectively. Their CC50 were 423.3, 229.3, 83.3 μg/mL and >100 unit, and the therapeutic indices were 103.2, 4.8, 9.2 and >192.3, respectively. Quercetin, apigenin, luteolin and catechin showed anti-PEDV activity with IC50 of less than 12 μg/mL and CC50 of 365.2, >50, 6.7 and >100 μg/mL, respectively. Furthermore, Q7R showed the highest TI (>7142.86) for PEDV tested with IC50 of 0.014 μg/mL and CC50 of >100 μg/mL (Table 2 ).
Table 2.
Antiviral activity of various compounds against porcine epidemic diarrhea virus in Vero cells.
| Test drugs | IC50a | CC50b | TIc | |
|---|---|---|---|---|
| Antiviral drugs | Ribavirin | 4.1 ± 4.2 | 423.3 | 103.2 |
| Interferon-α | 0.52 ± 0.5 unit | >100 unit | >192.3 | |
| Natural compounds | Coumarin | 47.4 | 229.3 | 4.8 |
| Tannic acid | 9 | 83.3 | 9.2 | |
| Flavonoids | Quercetin | 1.7 ± 0.8 | 365.2 | 214.8 |
| Apigenin | 0.1 ± 0.1 | >50 | >370.4 | |
| Luteolin | 0.2 ± 0.2 | 6.7 | 32.7 | |
| Catechin | 11.1 ± 7.1 | >100 | >9.0 | |
| Quercetin 7-rhanmoside | 0.014 ± 0.005 | >100 | >7142.86 | |
Results are presented as the mean IC50 values obtained from three independent experiments carried out in triplicate ± S.D.
Concentration required to inhibit virus-induced CPE by 50% (μg/mL).
Concentration required to reduce cell growth by 50% (μg/mL).
Therapeutic index = CC50/IC50.
On the basis of the above results, the relationship between flavonoid structure and antiviral activity against PEDV was further evaluated. Among the flavonoids tested, flavones (i.e. apigenin and luteolin), flavonol (i.e. Q7R) and flavan (i.e. catechin), which all have o-dihydroxy functional groups at C-3′ and C-4′, showed significant anti-PEDV activity. In addition, flavonols Q7R possessed one rhamnoside at position 7 with a 3′,4′-dihydroxyl group was also found to have an antiviral activity than higher that of the 3′,4′-dihydroxylated flavonoids. But, the anti-PEDV activity of quercetin with five hydroxyl groups decreased (IC50 value of less than 50%). This indicates that sugars groups at C-7 of A-ring are an important feature for the anti-PEDV activity of flavonoids.
3.2. The effects of Q7R on the infectivity of PEDV particles
To observe the effects of Q7R on the infectivity of PEDV particles, this exam was performed. Antiviral activity of pre-incubation with Q7R and ribavirin resulted in 14.9%, 0.9%, respectively. In contrast, continuous presence of Q7R during infection led to a significant increase in antiviral activity. A similar result was obtained in control infections with treated ribavirin (Fig. 1 ), but antiviral activity was shown to be lower than that of Q7R. This indicates that Q7R does not interact with the particles of PEDV, as pre-exposure of the virus to Q7R did not alter the infectivity of PEDV particles.
Fig. 1.
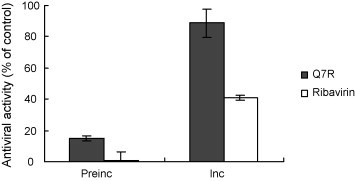
The effects of Q7R on the infectivity of PEDV particles. PEDV particles were incubated with Q7R of 10 μg/mL for 1 h at 4 °C. Afterwards, Vero cells were incubated with Q7R-treated or untreated virus for 1 h at 37 °C. Unbound virus was removed by extensive washing and infection was continued by cultivating cells in infection medium with or without Q7R of 10 μg/mL at 37 °C. Antiviral activity was determined by titration using SRB assays 2 days post-infection. Preinc, pre-incubation was expressed incubation without Q7R of 10 μg/mL after washing; Inc., incubation was expressed incubation with Q7R of 10 μg/mL after washing.
3.3. Time course of compound addition
The Q7R was added at different periods (before, during, and after) of PEDV infection. Results showed that the Q7R suppressed PEDV infection, when added just after the virus inoculation (0 h) and also early after the virus inoculation (1, 2 and 4 h). Ribavirin also showed a weak effect on PEDV infection. The inhibitory either amount of level of Q7R was higher than 85% (Fig. 2 ) but that of ribavirin was lower than 50%. However, the inhibitory rate of two compounds declined to 40% or less when added at either prior (−1 h) or post (6, 8, 12 and 24 h) infection. This observation indicated that Q7R affects the initial stage of PEDV infection.
Fig. 2.
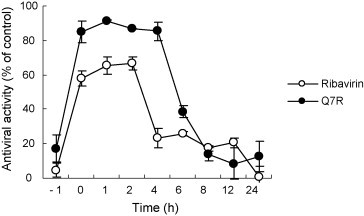
Time-of-addition effect of Q7R and ribavirin on PEDV replication in Vero cells. Ten μg/mL of each compound was added at either before (−1 h), during (0 h), or after (1, 2, 4, 6, 8, 12 and 24 h) virus infection. After 2 days, inhibition was evaluated by SRB method and expressed as the inhibition rate. Each value is the result of mean ± S.D. of three independent experiments.
3.4. RT-PCR analysis
Further evidence of the inhibitory effects of Q7R on infection PEDV and viral replication in Vero cells was provided by PCR analysis. The RNA extraction was performed at 24 and 48 h after PEDV infection. Q7R (10 μg/mL) decreased the intensity of the product band of PEDV at 24 and 48 h after infection, but ribavirin (10 μg/mL) did not decrease the intensity of the product band of PEDV at 48 h after infection (Fig. 3 ).
Fig. 3.
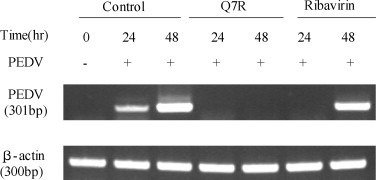
RT-PCR analysis. Replication of PEDV from Vero cells before and at 24 and 48 h after infection by PEDV in the presence of Q7R (10 μg/mL) or ribavirin (10 μg/mL) or vehicle alone (control, 0.1% DMSO), as detected by RT-PCR.
3.5. Antiviral activities in related viruses
The two viruses selected for these experiments included TGEV and PRCV in ST cells. When the antiviral activity and cytotoxicity of Q7R or ribavirin was examined by SRB assay, the two compounds did not exhibit any cytotoxicity at the highest concentrations tested (Table 3 ). The IC50 value of Q7R and ribavirin in TGEV was 63.31 and 55.09 μg/mL, respectively, while that in PRCV was 59.76 and 61.6 μg/mL, respectively. Furthermore, TI value was no great different between the two viruses.
Table 3.
Antiviral activity of quercetin 7-rhamnoside against related viruses.
| TGEV |
PRCV |
||||
|---|---|---|---|---|---|
| Test drugs | CC50a | IC50b | TIc | IC50b | TIc |
| Ribavirin | >100 | 55.09 ± 0.8 | >1.82 | 61.6 ± 1.91 | >1.62 |
| Quercetin 7-rhamnoside | >100 | 63.31 ± 1.07 | >1.58 | 59.76 ± 5.70 | >1.67 |
Results are presented as the mean IC50 values obtained from three independent experiments carried out in triplicate ± S.D.
Concentration required to inhibit virus-induced CPE by 50% (μg/mL).
Concentration required to reduce cell growth by 50% (μg/mL).
Therapeutic index = CC50 / IC50.
4. Discussion
Several veterinary coronavirus vaccines are currently available, but their efficacy is variable. The vaccine for prevention of infectious bronchitis virus (IBV), which infects chickens, is effective (Ladman et al., 2002), while the canine and porcine vaccines are only partially effective (Pratelli et al., 2004). Ribavirin has been used as treat against various DNA and RNA virus infections, although virus-acquired resistance to it was isolated from various virus populations and observed in some patients (Jason and Craig, 2006).
Antiviral studies have shown that essential oil prepared from fresh plants of H. cordata has an inhibitory effect on herpes simplex virus type 1 (HSV-1), influenza virus and human immunodeficiency virus type 1 (HIV-1), without showing cytotoxicity (Lu et al., 2006). Quercetin has been reported to have inhibitory effects on several viruses (Mucsi and Pragai, 1985). These studies indicate that H. cordata possesses compound exhibited antiviral effects. In this study, Q7R is an effective antiviral compound against PEDV. Q7R did not have a marked inhibitory effect on the growth of TGEV or PRCV-infected cells. The antiviral activity of this compound appeared to be strongly influenced by the strain of the coronaviruse tested. Ribavirin, which is a broad-spectrum nucleoside analogue, exhibited expected antiviral activity. This broad-spectrum antiviral drug was previously shown to be inhibitory to 16 DNA and RNA viruses in cell cultures (Sidwell et al., 1972). Trials of ribavirin in this study showed that the drug had favorable effects on antiviral activity in Vero cells infected with PEDV. In TGEV or PRCV, ribavirin had a weaker influence on survival of in Vero cells after infection with TGEV or PRCV. We were however able to ascertain that ribivirin does possess some antiviral activities.
To elucidate the action of Q7R on PEDV multiplication in more detail, we investigated the effect of Q7R on single steps during the infection cycle of PEDV. As a result, it can be concluded that Q7R does not interact with the particles of PEDV, as pre-exposure of the virus to Q7R did not alter the infectivity of PEDV particles (Fig. 1). Based on the results of the time-course study, pre-incubation of the Vero cells with Q7R did not protect the cells from PEDV infection (Fig. 2). Furthermore, Q7R can inhibit PEDV infection only when it was added on, during, and early stages after the virus inoculation (1, 2, 4 h), but not after 6 h or later (Fig. 2). This suggests that the mode of action is not deriven from inhibiting the absorption of virus but results from inhibition at an early stage of viral replication after infection (Fig. 1, Fig. 2).
Among the flavonoids tested, only Q7R possessed significant activity against PEDV. In a previous report, rutin (quercetin-3-rutinoside) did not express antiviral activity whereas quercitrin (quercetin 3-rhamnoside) possessed similar activity to quercetin against human herpesviruses and adenoviruses (Chiang et al., 2003a, Chiang et al., 2003b). They found that the antiviral activity among the flavonoid glycosides containing the quercetin moiety might be correlated with the species of sugar group at the 3 position. Our results suggest that the antiviral activity of flavonoids against PEDV is correlated with the species of sugar group at the 7 position.
In conclusion, the present study described that Q7R possesses strong anti-PEDV activity among the flavonoids. It will be interesting to further investigate the antiviral activity of the Q7R in preventing various PEDV-mediated injuries in in vivo pathological situations. Further studies will be required to explore the detailed antiviral mechanism of Q7R.
Acknowledgements
This work was supported by a grant from Research Institutes of Bioscience & Biotechnology (KRIBB), Republic of Korea. This research was also partially supported by a grant from Biogreen 21 program Rural Development Administration, by the Ministry for Food, Agriculture, Foresty and Fisheries, Republic of Korea.
References
- Awaad A.S., Maitland D.J., Soliman G.A. Hepatoprotective activity of Schouwia thebica web. Bioorg. Med. Chem. Let. 2006;16:4624–4628. doi: 10.1016/j.bmcl.2006.06.011. [DOI] [PubMed] [Google Scholar]
- Briskin D.P. Medicinal plants and phytomedicines Linking plant biochemistry and physiology to human health. Plant Physiol. 2000;124:507–514. doi: 10.1104/pp.124.2.507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiang L.C., Chiang W., Liu M.C., Lin C.C. In vitro antiviral activities of Caesalpinia pulcherrima and its related flavonoids. J. Antimicrob. Chemother. 2003;52:194–198. doi: 10.1093/jac/dkg291. [DOI] [PubMed] [Google Scholar]
- Chiang L.C., Chiang W., Chang M.Y., Lin C.C. In vitro cytotoxic, antiviral and immunomodulatory effects of Plantago major and Plantago asiatica. Am. J. Chin. Med. 2003;31:225–234. doi: 10.1142/S0192415X03000874. [DOI] [PubMed] [Google Scholar]
- Chomczynski P., Mackey K. Substitution of chloroform by bomochloropropane in the single-step method of RNA isolation. Anal. Biochem. 1995;225:163–164. doi: 10.1006/abio.1995.1126. [DOI] [PubMed] [Google Scholar]
- Cowan M.M. Plant products as antimicrobial agents. Clin. Microbiol. Rev. 1999;12:564–582. doi: 10.1128/cmr.12.4.564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Debouck P., Pensaert M. Experimental infection of pigs with a new porcine enteric coronavirus CV 777. Am. J. Vet. Res. 1980;41:219–223. [PubMed] [Google Scholar]
- Hofmann M., Wyler R. Study of the occurrence of epizootic viral diarrhea in swine in Switzerland. Schweiz. Arch. Tierheilkd. 1987;129:437–442. [PubMed] [Google Scholar]
- Jason D.G., Craig E.C. Mechanisms of action of ribavirin against distinct viruses. Rev. Med. Virol. 2006;16:37–48. doi: 10.1002/rmv.483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jassim S.A., Naji M.A. Novel antiviral agents: a medicinal plant perspective. J. Appl. Microbiol. 2003;95:412–427. doi: 10.1046/j.1365-2672.2003.02026.x. [DOI] [PubMed] [Google Scholar]
- Kwon K.B., Kim E.K., Shin B.C., Seo E.A., Yang J.Y., Ryu D.G. Herba houttuyniae extract induces apoptotic death of human promyelocytic leukemia cells via caspase activation accompanied by dissipation of mitochondrial membrane potential and cytochrome c release. Exp. Mol. Med. 2003;35:91–97. doi: 10.1038/emm.2003.13. [DOI] [PubMed] [Google Scholar]
- Ladman B.S., Pope C.R., Ziegler A.F., Swieczkowski T., Callahan C.J., Davison S., Gelb J., Jr. Protection of chickens after live and inactivated virus vaccination against challenge with nephropathologenic infectious bronchitis virus PA/Wolgemuth/98. Avian Dis. 2002;46:938–944. doi: 10.1637/0005-2086(2002)046[0938:POCALA]2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- Li G.Z., Chai O.H., Lee M.S., Han E.H., Kim H.T., Song C.H. Inhibitory effects of Houttuynia cordata water extracts on anaphylactic reaction and mast cell activation. Biol. Pharm. Bull. 2005;28:1864–1868. doi: 10.1248/bpb.28.1864. [DOI] [PubMed] [Google Scholar]
- Lin Z.X., Hoult J.R.S., Raman A. Sulforhodamine B assay for measuring proliferation of a pigmented melanocyte cell line and its application to the evolution of crude drugs used in the treatment of vitiligo. J. Ethnopharmacol. 1999;66:141–150. doi: 10.1016/s0378-8741(98)00199-8. [DOI] [PubMed] [Google Scholar]
- Lu H.M., Liang Y.Z., Yi L.Z., Wu X.J. Anti-inflammatory effect of Houttuynia cordata injection. J. Ethnopharmacol. 2006;104:245–249. doi: 10.1016/j.jep.2005.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mucsi I., Pragai B.M. Inhibition of virus multiplication and alteration of cyclic AMP level in cell cultures by flavonoids. Experientia. 1985;41:930–931. doi: 10.1007/BF01970018. [DOI] [PubMed] [Google Scholar]
- Pratelli A., Tinelli A., Decaro N., Martella V., Camera M., Tempesta M., Martini M., Carmichael L.E., Buonavoglia C. Safety and efficacy of a modified-live canine coronavirus vaccine in dogs. Vet. Microbiol. 2004;99:43–49. doi: 10.1016/j.vetmic.2003.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sidwell R.W., Huffman J.H., Khare G.P., Allen L.B. Broad-spectrum antiviral activity of Virazole: 1-beta-d-ribofuranosyl-1,2,4-triazole-3-carboxamide. Science. 1972;177:705–706. doi: 10.1126/science.177.4050.705. [DOI] [PubMed] [Google Scholar]
- Vlietinck A.J., Vanden Berghe D.A. Can ethnopharmacology contribute to the development of antiviral drugs. J. Ethnopharmacol. 1991;32:141–153. doi: 10.1016/0378-8741(91)90112-q. [DOI] [PubMed] [Google Scholar]
- Williams J.E. Review of antiviral and immunomodulating properties of plants of the Peruvian rainforest with a particular emphasis on Una de Gato and Sangre de Grado. Altern. Med. Rev. 2001;6:567–579. [PubMed] [Google Scholar]


