Highlights
► We investigated RNA interference (RNAi) as antiviral agent against rabies using two small interfering RNAs (siRNAs) targeting rabies virus (RV) nucleoprotein (N) and polymerase (L) genes. ► The antiviral potential of siRNAs delivered using adenoviruses in BHK-21 cells showed marked inhibition in RV multiplication, RV titer knockdown of RV gene transcripts. ► Mice treated with adenoviruses expressing siRNAs showed 66.6% and 33.3% protection with adenoviruses expressing siRNAs against RV-N and RV-L genes, respectively against lethal rabies virus challenge.
Abbreviations: Ad, adenovirus; BHK-21, baby hamster kidney-21; CNS, central nervous system; CVS-11, challenge virus standard-11; EGFP, enhanced green fluorescent protein; ffu, fluorescent foci unit; HEK-293, human embryonic kidney-293; L, large polymerase; LD50, lethal dose 50%; MOI, multiplicity of infection; N, nucleoprotein; PV-11, Pasteur virus-11; RFP, red fluorescent protein; RNAi, RNA interference; RABV, rabies virus; shRNA, small hairpin RNA; siRNA, small interfering RNA
Keywords: Rabies, RNAi, Adenovirus, HEK-293 cell line, BHK-21 cell line, Transfection, Mice
Abstract
To investigate the potential of RNA interference (RNAi) as antiviral agent against rabies, two small interfering RNAs (siRNAs) targeting rabies virus (RABV) nucleoprotein (N) and polymerase (L) genes were designed and evaluated. Both siRNAs knockdown or silenced the target RABV genes as evaluated in a plasmid based transient expression model. For efficient delivery, adenoviruses expressing the siRNAs were constructed and antiviral potential of the delivered siRNAs was investigated in BHK-21 cells. When cells treated with adenoviruses expressing siRNAs were challenged with RABV, there was 88.35 ± 2.4% and 41.52 ± 9.3% reduction in RABV multiplication in infected cells with siRNAs targeting RABV-N and L genes, respectively. Relative quantification of RABV transcripts using real-time PCR revealed knockdown of both RABV-N and L gene transcripts, however, significant reduction was observed only with adenovirus expressing siRNA against RABV-N. When mice treated intracerebrally with adenoviruses expressing siRNAs were challenged peripherally with lethal RABV by the intramuscular route in masseter muscle, there was 66.6% and 33.3% protection with adenoviruses expressing siRNAs against RABV-N and L genes, respectively. These results demonstrated that adenovirus expressing siRNA against RABV-N efficiently inhibited the RABV multiplication both, in vitro and in vivo and conferred significant protection against lethal RABV challenge. This supported the hypothesis that RNAi, based on siRNA targeting RABV-N gene can prevent RABV infection and holds the potential of RNAi as an approach to prevent rabies infection.
1. Introduction
Rabies is a dreadful disease caused by a virus belonging to genus Lyssavirus of family Rhabdoviridae. Rabies virus (RABV) infection is considered to be one of the oldest and most fatal neurological diseases known to mankind. Present practice of rabies control in animals and humans is to take prophylactic pre-exposure vaccination. Post-exposure vaccination is obligatory in all cases as treatment after human exposure to infected animal bites (Wiktor et al., 1984). Sometimes, due to unnoticed exposure or vaccination failure, the disease is diagnosed after the onset of clinical symptoms and death becomes unavoidable due to unavailability of any effective line of treatment for clinical cases of rabies. Therefore, efforts are being made to develop an effective antiviral strategy against rabies with the potential to suppress virus infection in cells and clear the infection without damaging the cell. The various approaches include strategy based on RNA interference (RNAi). Considering the potential of RNAi as antiviral agent, few groups have evaluated RNAi based antiviral agents against rabies (Brandao et al., 2007, Israsena et al., 2009).
RNAi mediated by small interfering RNAs (siRNAs) is a powerful technology allowing the silencing of genes with great specificity and potency. It is a highly conserved natural mechanism described for plants, fungi, worms, insects, and mammals (Ma et al., 2007, for review). The gene silencing mechanism is mediated by short RNA, named siRNAs, whereby an RNA-induced silencing complex forms an activated complex that target mRNA with sequence complementary to the siRNAs. This renders the mRNA transcripts inert and degradable resulting in a specific knockdown of the targeted gene expression. The sequence-specific knockdown of viral genes in infected cells generated great interest in development of siRNAs-based antiviral agents. siRNA-based antiviral activity has been reported for many RNA and DNA viruses in vitro and in vivo, including, human immunodeficiency virus, poliovirus, hepatitis B virus, hepatitis C virus, human papilloma virus, dengue virus, hepatitis delta virus, murine gamma herpesviruses, severe acute coronavirus and respiratory syncytial virus infections (Sliva and Schnierle, 2010, for review).
In the present study, we report siRNA-based RNAi to inhibit RABV multiplication. The siRNAs targeting RABV-N and L genes were designed and delivered using an adenoviral vector and inhibition of RABV multiplication was evaluated in vitro in BHK-21 cells. The protection against lethal RABV challenge was evaluated in vivo in mice after intracerebral treatment with adenoviruses expressing siRNAs targeting RABV genes.
2. Materials and methods
2.1. Cells and virus
Baby hamster kidney-21 (BHK-21) and human embryonic kidney-293 (HEK-293) cells, used in this study, were procured from National Centre for Cell Science (NCCS), Pune, India and grown at 37 °C under 5% CO2 in Dulbecco's modified minimum essential medium (DMEM, Hyclone), supplemented with 10% fetal bovine serum (FBS, Hyclone) and 50 μg/ml gentamicin.
The mouse brain-adapted RABV challenge virus standard-11 (CVS-11) strain was used to amplify RABV genes in RT-PCR and for in vivo challenge in mice. The RABV Pasteur virus-11 (PV-11) strain adapted to BHK-21 was used for in vitro challenge studies in BHK-21 cells.
2.2. Construction of RABV target gene reporter plasmids
The target gene reporter plasmids, pDsRed-N and pDsRed-L, were constructed for expressing RABV-N and L protein, respectively, with C-terminal fusion with red fluorescent protein (RFP). For construction of pDsRed-N, the full-length (1380 bp) RABV-N gene was amplified by RT-PCR with primers (Supplementary Table 1) using cDNA prepared from total RNA isolated from RABV-CVS-11-infected mouse brain and inserted into HindIII and BamHI sites in pDSRed-N1 vector (Clontech). Similarly, pDsRed-L was constructed by amplifying 636 bp partial RABV-L polymerase gene fragment containing siRNA target site by RT-PCR using primers (Supplementary Table 1) and cloned into XhoI and BamHI sites in pDSRed-N1 vector. The expression of red fluorescent protein (RFP) fusion proteins (N-RFP and L-RFP) was confirmed in transfected HEK-293 cells.
2.3. Construction of silencing constructs encoding short hairpin RNAs (shRNAs)
The siRNAs targeting RABV-N and L genes were designed using algorithm following sequence-motif and thermodynamics guidelines (Ui-Tei et al., 2008). Two siRNA sequences (si-N and si-L) along with antisense and loop sequences were synthesized as oligonucleotides (Supplementary Table 1), annealed and cloned into shRNA expressing plasmid pMCS-eGFP-containing enhanced green fluorescent protein (EGFP) as reporter gene. One scrambled siRNA (si-C) with no homology with the RABV genome was also cloned as shRNA for negative control siRNA (Supplementary Table 1).
2.4. Co-transfection of RABV reporter target gene and shRNA expression plasmids
To evaluate the efficiency of siRNAs silencing RABV genes, HEK-293 cells were transiently co-transfected with two plasmids: a reporter gene construct (encoding either RABV-N or L gene fused to RFP) and a silencing construct encoding a gene-specific siRNA (si-N or si-L) or negative control siRNA (si-C). HEK-293 cells were co-transfected using Lipofectomine 2000 reagent (Invitrogen) in a 24-well plate and analyzed for RFP and EGFP expression 48 h post-transfection using Olympus IX51 fluorescent microscope. Cells with EGFP expression indicated expression of shRNAs, while cells with RFP expression indicated RABV-N or L target reporter gene expression. Cells with an apparent absence of red fluorescence indicated gene silencing.
2.5. Construction of recombinant adenoviruses expressing shRNAs
Recombinant adenoviruses expressing different siRNAs (si-N, si-L or si-C) were constructed using pSilencer adeno System (Ambion). Briefly, sense and antisense oligonucleotides having cohesive restriction endonuclease site ends (Supplementary Table 1) were annealed and cloned into the Adeno shuttle vector. The recombinant Adeno shuttle vector was co-transfected with linear adenovirus LacZ backbone plasmid into HEK-293 cells and recombinant adenovirus was produced. For in vitro and in vivo studies, different recombinant adenoviruses, namely, Ad-N, Ad-L and Ad-Neg were expanded in HEK-293 cells and purified using the AdEasy virus purification kit (Stratagene). Purified adenovirus infectious unit titers were determined in HEK-293 cells by ten-fold limiting dilution and X-gal staining following the manufacturer's instructions (Ambion). To characterize recombinant adenoviruses, the viral genomic DNAs from Ad-N, Ad-L and Ad-Neg were isolated using the QIAamp DNA mini kit (Qiagen) and a 691 bp fragment containing the shRNA sequence was amplified by PCR using primers (pSiAdeno-F and R, Supplementary Table 1) and sequenced to verify the integrity of the siRNA.
2.6. Transduction of BHK-21 cells with adenovirus
To analyze the anti-rabies effect of different siRNAs in vitro, the siRNAs were delivered into BHK-21 cells by treating with recombinant adenoviruses. Briefly, BHK-21 cells were infected with 100 multiplicity of infection (MOI) of different adenoviruses in presence of polybrene (6 μg/ml) for 6 h. After 24 h treatment, transduction efficiency was analyzed by X-gal staining.
2.7. RABV infection of BHK-21 cells and fluorescent antibody detection
The efficiency of different siRNAs to inhibit RABV multiplication was evaluated in adenovirus-treated BHK-21 cells after in vitro challenge with RABV-PV-11. Briefly, 5 × 105 BHK-21 cells were infected with different adenoviruses in a 6-well plate for 24 h and then infected with 0.01 MOI of RABV-PV-11. At 6 h post-adsorption, the unadsorbed RABV in the inoculums was removed and infected cell monolayer was washed three times with PBS. At 48 h post-infection, the infected cell culture supernatant was harvested and stored at −80 °C for virus titer determination. The infected cell monolayer was fixed with 80% acetone and RABV in infected cells was detected by direct fluorescent antibody staining using rabies anti-nucleocapsid FITC-labeled antibody (BioRad) following the manufacturer's instructions. The cell monolayer was nuclear counterstained with Prolong gold antifade reagent with DAPI (Invitrogen).
The RABV titer in infected cell culture supernatant was determined by fluorescent foci unit (ffu) assay. Briefly, serial 10-fold dilutions of virus supernatant, harvested at 48 h post-infection, was mixed with 1 × 104 BHK-21 cells in a flat-bottom 96-well plate in triplicate and incubated at 37 °C. Two days after infection, infected cells were fixed with 80% acetone and plaques were visualized by staining with rabies anti-nucleocapsid FITC-labeled antibody and ffu titer was determined.
2.8. RNA isolation, cDNA synthesis and real-time PCR
To evaluate the efficiency of different siRNAs to silence expression of target RABV genes, a quantitative determination of RABV mRNAs was done using real-time PCR. Different adenovirus-treated BHK-21 cells (5 × 105) in a 6-well plate were infected with RABV-PV-11 at 0.01 MOI. At 24 h post-infection, total RNA was isolated using Trizol LS reagent (Invitrogen) and mRNA transcripts in total RNA were reverse transcribed into cDNAs using oligo dT primer and RevertAid M-MuLV reverse transcriptase (Fermentas) following the manufacturer's protocol. The RABV-N and L gene transcripts in 1:10 diluted cDNAs were quantified using gene specific real-time primers (Supplementary Table 1) using Kapa SYBR fast qPCR kit (Kapa Biosystems) following the manufacturer's instructions. For relative quantification, GAPDH gene transcripts were used as reference gene expression for normalization and n-fold changes in target mRNA transcript levels were determined by using the method described earlier (Pfaffl, 2001).
2.9. Adenovirus inoculation of mice and lethal RABV challenge
Swiss albino mice (3–4 weeks old) of either sex were grouped into four groups with twelve mice each. Three groups of mice (n = 12, each) were injected intracerebrally with 1 × 109 infectious adenovirus particles of either Ad-N or Ad-L or Ad-Neg in 50 μl volume. One group of mice (n = 12) was injected with saline and kept as negative control. All groups of mice were challenged by intramuscular route in masseter muscle with 20 LD50 of lethal RABV-CVS-11 in 50 μl volume following the method of NIH test for potency (Wilbur and Aubert, 1996). All the challenged mice were observed for 16 days for development of rabies-specific paralytic symptoms or death and percent survival was calculated.
2.10. Statistical analysis
The RABV titer and gene transcripts were compared by statistical analysis using one-way ANOVA followed by Dunnett's test for treatment versus control. Comparison of survival was done by Log-rank test following Mantel-Haenszel method using GraphPad Prism version 4.03 (GraphPad). All data were presented as the mean ± SEM. Differences between the groups with p ≤ 0.05 were considered statistically significant.
3. Results
3.1. Silencing effect of siRNAs targeting RABV-N and L genes
Two different siRNAs, targeting RABV-N mRNA nt 1120–1138 and L mRNA nt 1203–1221 were designed and sequences were cloned into shRNA expression plasmid pMCS-eGFP. This generated silencing constructs expressing siRNA against RABV-N or L genes. Since pMCS-eGFP plasmid also included EGFP as reporter gene it was easy to identify cells with the shRNA expressing plasmid. We first examined whether these siRNAs could specifically knockdown or silence the expression of the corresponding RABV-N and L proteins. The HEK-293 cells were transiently co-transfected with target reporter gene plasmids (pDsRed-N or pDsRed-L) and siRNAs expressing plasmids. The RFP-tagged version of RABV-N or L protein (N-RFP or L-RFP) was detected in all transfected cells. The cells containing the target reporter gene plasmid and specific siRNA silencing construct corroborated with the reduction in RFP expression (indicating reduction in target reporter gene expression) compared to controls (co-transfected with negative control siRNA expressing plasmid) (Fig. 1 ). This confirmed that there was expression of the siRNA which effectively silenced the target gene in a gene-specific manner.
Fig. 1.
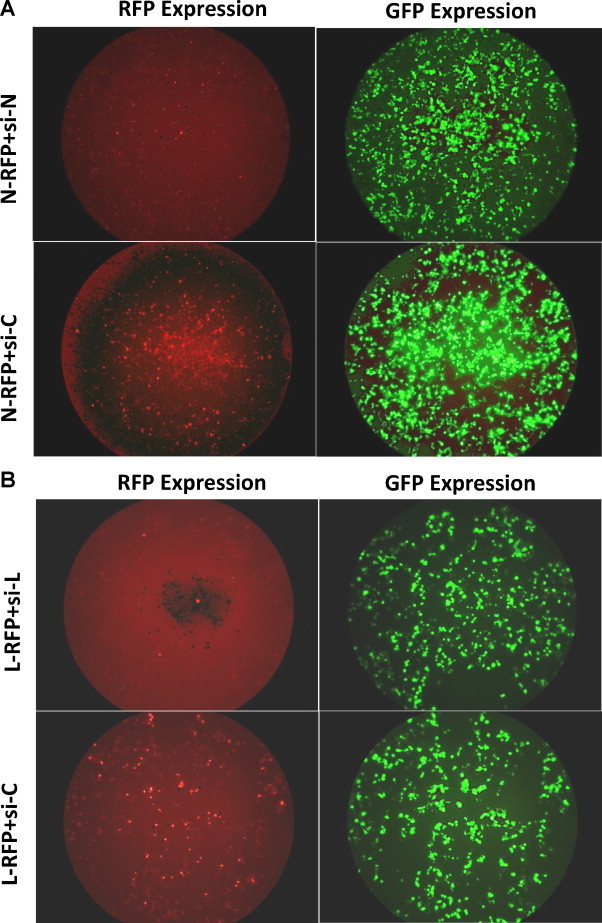
Evaluation of the silencing effect of siRNAs targeting RABV-N and L genes using a transient co-transfection model. HEK-293 cells were transiently co-transfected with target gene reporter plasmid (pDsRed-N or pDsRed-L) expressing RABV-N or L protein with C-terminal fusion with red fluorescent protein (N-RFP or L-RFP) and silencing constructs expressing siRNA (si-N or si-L or si-C). At 48 h post-transfection, cells were analyzed for RFP and EGFP expression. Cells with EGFP expression indicated presence of shRNA expressing plasmid while cells with RFP expression indicated RABV-N or RABV-L target reporter protein expression. Cells with an apparent absence of red fluorescence indicated gene silencing.
3.2. Production and characterization of recombinant adenoviruses expressing shRNAs
In order to develop an adenoviral siRNA delivery system, the siRNA sequences in shRNA form were cloned into the adeno shuttle vector and then inserted into the genome of adenovirus defective in the E1 early gene. The integrity of siRNA sequences in the recombinant adenoviruses (Ad-N or Ad-L or Ad-Neg) was confirmed by amplifying a 691 bp DNA fragment flanking the shRNA from the genomic DNA isolated from different recombinant adenoviruses and nucleotide sequences were determined (Supplementary Fig. 1). The infectivity titers of purified recombinant adenoviruses Ad-N, Ad-L and Ad-Neg were estimated in HEK-293 cells by X-gal staining and found to be 1.0 × 1011, 5.6 × 1010 and 3.14 × 1010 per ml, respectively. Since the adenovirus backbone contained LacZ gene, the infected HEK-293 cells had β-galactocidase activity which gave blue plaques after X-gal staining (Supplementary Fig. 2).
Before analyzing antiviral effect of the siRNAs in BHK-21 cells delivered by the adenoviral vector, it was necessary to determine the amount of recombinant adenovirus needed to yield the highest level of siRNA expression without cellular toxicity. The BHK-21 cells were treated with different MOI of adenoviruses in presence and absence of polybrene (results not shown) and it was found that treatment of BHK-21 cells with 100 MOI of adenovirus in presence of polybrene (6 μg/ml) was sufficient to transduce a high proportion of cells (>95%) without toxicity (Supplementary Fig. 3).
3.3. Effect of siRNAs delivered through adenoviral vector on RABV multiplication in vitro
To assess the effectiveness of siRNAs delivered by recombinant adenoviruses to inhibit RABV multiplication, adenovirus-treated BHK-21 cells were infected with RABV-PV-11. At 48 h post-infection, the RABV multiplication was analyzed both in infected cell monolayer (for cell-associated virus) and in infected cell culture supernatant (cell-released virus). The presence of RABV, as evident after fluorescent foci staining, revealed reduction in RABV multiplication with both the siRNAs compared to control siRNA (Fig. 2 ). The reduction with Ad-N (adenovirus expressing siRNA against RABV-N) was more pronounced compared to Ad-L.
Fig. 2.
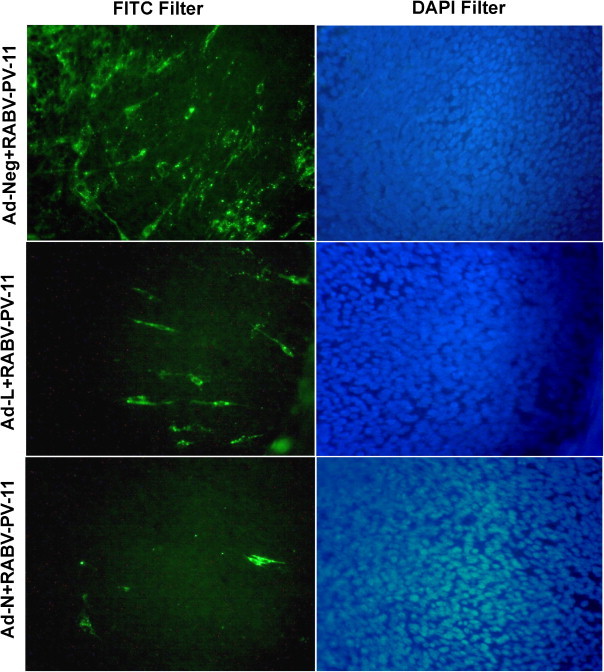
Inhibition of RABV multiplication in BHK-21 cells treated with different adenoviruses expressing siRNAs. BHK-21 cells were first treated with different adenoviruses expressing siRNAs and then challenged with 0.01 MOI of RABV-PV-11 strain. At 48 h post-challenge, presence of RABV in infected cells was detected by staining with anti-rabies nucleocapsid FITC-labeled antibody and counterstained with DAPI. The stained cells were observed with fluorescent microscope and representative fluorescent-field images (with FITC filter, left column) and relative nuclear stained-field images (with DAPI filter, right column) were recorded.
To estimate the inhibitory effect of siRNAs on RABV multiplication more precisely, their ability to limit infectious virus production was analyzed by RABV ffu count in infected cell culture supernatant. The Ad-N- and Ad-L-treated BHK-21 cells showed 6 ± 1.58 × 103 and 30 ± 7.90 × 103 ffu/ml of RABV, respectively, while Ad-Neg-treated BHK-21 cells showed 55 ± 14.43 × 103 ffu/ml of RABV. These results are average of four independent experiments. Collectively, there was significant reduction (88.35 ± 2.4%) in RABV ffu count with Ad-N expressing siRNA against RABV-N while reduction was 41.52 ± 9.3%, however non-significant, with Ad-L expressing siRNA against RABV-L (Fig. 3 ). These results clearly indicated that both siRNAs, namely, si-N and si-L delivered using adenoviral vector could inhibit RABV multiplication in BHK-21 cells in which the inhibitory effect was more pronounced (>2-fold higher) with si-N compared to si-L.
Fig. 3.
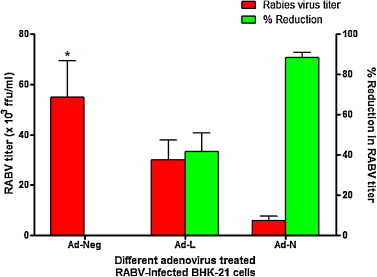
Inhibition of RABV production in BHK-21 cells treated with different adenoviruses expressing siRNAs. BHK-21 cells were first treated with different adenoviruses expressing siRNAs and then infected with 0.01 MOI of RABV-PV-11 strain. The infected cell culture supernatant was harvested at 48 h post-infection and RABV titers were measured by the rabies fluorescent foci unit (ffu) assay. Data shown are mean ± SEM from four independent experiments. *Group differs for RABV titers in comparison with control (Ad-Neg), p < 0.05 (One-way ANOVA, Dunnett's method).
To assess the ability of siRNAs to knockdown the expression of RABV genes, relative levels of RABV-N and L transcripts in different adenovirus-treated and RABV-infected BHK-21 cells were quantified by real-time PCR after normalization using GAPDH housekeeping gene and analyzed. The results from six independent experiments demonstrated that the RABV-N and L transcripts were significantly decreased to 1442-fold and 380-fold in BHK-21 cells treated with Ad-N compared to control. The effect was less pronounced (1.4-fold reduction) for both the transcripts in BHK-21 cells-treated with Ad-L (Fig. 4 ). This indicated that both adenoviruses (Ad-L and Ad-N) were effective in delivering siRNAs in BHK-21 cells and causing reduction in levels of target mRNA transcripts, however, the effect was significant only with Ad-N expressing siRNA against RABV-N.
Fig. 4.
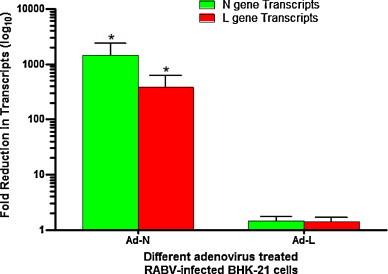
Relative levels of RABV-N and L transcripts in different adenovirus-treated and RABV-PV-11-infected BHK-21 cells quantified by real-time PCR. BHK-21 cells were first treated with different adenoviruses expressing siRNAs and then infected with RABV-PV-11 strain at a MOI of 0.01. At 24 h post-infection, the total RNA was isolated from infected cells and transcribed into cDNA using oligo-dT primer. The RABV-N and L gene transcripts in cDNAs were quantified using gene-specific primers in real-time PCR. GAPDH gene transcript was used as reference for normalization. Data presented are from six independent experiments and n-fold changes in target mRNA transcript levels were determined. *One-way ANOVA statistical analysis on log10-transformed data, followed by comparison with control using Dunnett's method.
3.4. In vivo evaluation of anti-rabies effect of different siRNA delivered through adenoviral vector
To assess whether siRNAs could inhibit RABV multiplication in vivo, we used an established murine model of RABV infection. Different adenovirus treated mice were challenged with 20 LD50 of lethal RABV CVS-11 by intramuscular route in masseter muscle and percent protection was determined. All the control mice and mice treated with adenovirus expressing control siRNA developed rabies-specific symptoms and died within 6–10 days post-challenge with median survival of 6 and 7 days, respectively. The median survival increased to 11.5 days and protection to 33.3% in a group of mice treated with Ad-L (adenovirus expressing siRNA against RABV-L). Treatment of mice with Ad-N (adenovirus expressing siRNA against RABV-N) demonstrated 66.6% protection (Fig. 5 ). The mice that survived did not develop symptoms and remained healthy till the entire period of experimentation. The result indicated that treatment of mice with adenovirus expressing siRNA against RABV-L conferred modest protection against lethal rabies challenge. The protection increased significantly in mice treated with adenovirus expressing siRNA against RABV-N gene indicating effectiveness of si-N over si-L against rabies in vivo.
Fig. 5.
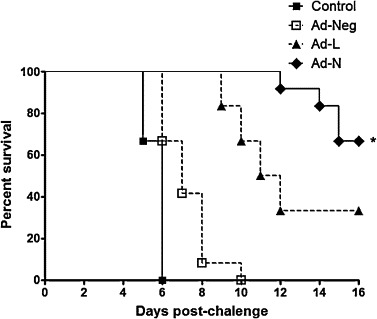
Percent survival of mice treated with different adenoviruses expressing siRNAs and challenged with lethal RABV challenge virus standard -11 (CVS-11) strain. Mice (n = 12, per group) were treated intracerebrally with different adenoviruses (Ad-N or Ad-L or Ad-Neg) or control (saline) and challenged by the intramuscular route in masseter muscle with 20 LD50 of lethal RABV-CVS-11 strain. The percent survival post-challenge was monitored. Ad-N-treated mice group differs from control or Ad-Neg-treated for survival (p < 0.05; Log-rank).
4. Discussion
RABV, once introduced through the broken skin or mucous membrane, replicates in the myocytes for hours or weeks and then migrates to nerves (Fekadu and Shaddock, 1984, Murphy, 1985). After development of infection in spinal cord or brain stem neurons, RABV disseminates rapidly throughout the central nervous system (CNS) by fast axonal transport along neuroanatomical connections. Under natural conditions, RABV infection of the CNS causes only relatively mild neuropathological changes without prominent evidence of neuronal death (Iwasaki and Tobita, 2002). Altogether, these observations have led to the concept that the neurological disease in rabies results from neuronal dysfunction rather than neuronal cell death. With this fact, if a strategy is developed to inhibit the RABV multiplication in CNS to block the pathogenic process of rabies and therefore disease progression and development may be prevented in infected individuals.
RABV has approximately 12 kb long single-stranded RNA genome consisting of a 50 nucleotides long leader followed by nucleoprotein (N), phosphoprotein (P), matrix (M), glycoprotein (G) and large polymerase (L) genes that encode proteins, namely N, P, M and G and L. There are few reports on in vitro evaluation of RNAi on rabies targeting viral structural genes like, N, and G (Israsena et al., 2009) where targeting N gene was found to be effective. RABV-G remained a unsuccessful target for RNAi because RABV-G is considered to be non-essential for virus replication in vitro in cultured cells and required only for trans-synaptic viral spread between neurons in vivo (Etessami et al., 2000, Yan et al., 2002). A modified RABV-G has been used to deliver siRNA across the blood–brain barrier (Kumar et al., 2007). Other targets for RNAi on rabies were non-structural genes, like, P (Israsena et al., 2009) and L (Brandao et al., 2007) with limited antiviral potential.
To design siRNAs that remain effective requires regions of the viral genome that are conserved among different strains of virus. Consequently, we selected RABV-N and L as our target genes for designing siRNAs as these are conserved and essential for virus multiplication. Moreover, while selecting siRNAs, the conserved region on RABV genome with no potential region for secondary structure formation was considered using siRNA designing algorithm. Since screening of effective siRNAs with infectious viruses is tedious, time-consuming and requires intense safety conditions, a simple method for the screening of potent siRNAs was used in the present study. We demonstrated that transient co-transfection of a reporter gene construct and a silencing construct was an appropriate method to screen potent siRNAs by inducing cleavage of the mRNA fused to red fluorescent protein (RFP) reporter by gene-specific siRNA. This provided a simple model for assessing the siRNA efficiency for gene silencing by analysis (reduced expression) of the fusion protein expression with fluorescent microscope. The si-N and si-L consistently inhibited the expression of RFP fusion version of RABV-N or L proteins in three independent experiments and indicated potent siRNAs for further studies. Screening of effective siRNAs using transient co-transfection of plasmids has been reported previously for targets other than RABV genes (Sanchez et al., 2005, Meng et al., 2007, Ong et al., 2008).
Delivery of siRNA remained a significant obstacle to the development of siRNA-based therapeutics. The different approaches that are being used to efficiently deliver siRNAs to neuronal tissue included the use of non-viral (Posadas et al., 2010, for review) and viral vectors (Sliva and Schnierle, 2010, for review). For in vivo delivery of siRNA against rabies, a technology is required that could deliver the RNAi therapeutic to neuronal cells. Adenovirus and lentivirus based viral vectors carry out precisely this task and have become a major delivery system for siRNA into nerve tissues (Sliva and Schnierle, 2010). The first study employing an adenoviral vector for in vivo RNAi was published in 2002 for an application in the central nervous system (Xia et al., 2002). In this study also, we selected adenovirus vector system to deliver siRNA into brain cells.
The efficiency of different siRNAs delivered by recombinant adenoviruses was analyzed in BHK-21 cells after challenging with RABV-PV-11. Since RABV-PV-11 strain is adapted to BHK-21 cells, it served as a model to analyze the efficiency of siRNAs in vitro. RNAi functions by identifying and degrading mRNA that shares sequence complementarity with the siRNA. Therefore, real-time PCR analysis was used to analyze the effect of si-N or si-L on RABV-N or L mRNA expression, respectively. The level of RABV-N mRNA in the Ad-N-treated, RABV-infected cells was significantly reduced (1442-fold) compared to the Ad-L-treated cells (1.43-fold). GAPDH was used as a cellular mRNA control and showed no effect of the siRNA treatment. Further, the level of RABV-L mRNA in Ad-N-treated cells was also reduced (380-fold), indicating an interrelated effect. This may be due to the fact that RABV-N performs its effect on RABV multiplication in two ways; RABV-N is a major structural protein and also plays a regulatory role in the switch from gene expression (mRNA production) to genome replication. The intracellular ratio of leader RNA to N protein has been reported to regulate the switch from viral gene transcription to genome replication (Wunner, 1991). To confirm the antiviral potential of RABV-N-specific siRNAs, we monitored the presence of viral progeny in the supernatants of siRNA-treated-RABV infected cells. The level of fold-decrease in RABV-N expression was correlated with decrease in infective virus titer in Ad-N treated cells. The decrease in infective RABV titer in Ad-N-treated cells was 88.4% compared to 41.5% in Ad-L-treated cells.
The efficiency of siRNAs was also analyzed to inhibit RABV multiplication in vivo in mice. The protective efficacy of different siRNAs against lethal RABV challenge was evaluated by injecting recombinant adenoviruses carrying appropriate siRNAs intracerebrally in mice and challenging with mouse adapted RABV-CVS-11 strain. Mice treated with Ad-N, which showed maximum inhibitory effect on RABV multiplication in vitro in BHK-21 cells also demonstrated significantly high protection (66.6%). In contrast, mice treated with Ad-L, which was less effective in vitro, were moderately protected (33.3%). The control siRNA-expressing adenovirus could not demonstrate protection, indicating lethal nature of the challenge virus. Recent studies have also indicated that short RABV-N cDNA and artificial microRNAs could interfere with RABV replication in cells (Wunner et al., 2004, Israsena et al., 2009) and synthetic siRNA against RABV-N could reduce RABV multiplication in BHK-21 cells (Brandao et al., 2007). Intracranial injection of siRNA in mice has shown immense potential in suppressing encephalitis caused by Chandipura virus belonging to family Rhabdoviridae (Kumar and Arankalle, 2010) and two flaviviruses, JEV (Japanese encephalitis virus) and WNV (West Nile virus) (Kumar et al., 2006).
On the basis of the results in this report, we concluded that, (i) siRNA against RABV-N could knockdown expression of RABV mRNAs and subsequently inhibiting RABV production efficiently, and (ii) inhibitory effect of siRNA against RABV-N interferes with viral gene expression, which significantly increased protection against lethal RABV challenge. These findings have significant implications for the use of siRNA as therapeutic agent against RABV infection. However, to attain absolute protection other siRNA delivery methods (e.g., lentiviral), route (intravenous crossing blood–brain barrier), dose (single or multiple siRNAs) and time of therapeutic administration of siRNAs have to be carefully optimized in mice and in non-human primates before recommending for therapeutic use.
Acknowledgements
This study was generously supported in part by a project “Evaluation of anti-rabies effect of small interfering RNA (siRNA) delivered through viral vector” Grant No. BT/PR9941/AGR/36/22/2007 from Department of Biotechnology (DBT), Government of India and Indian Council of Agricultural Research (ICAR), India.
Footnotes
Supplementary data associated with this article can be found, in the online version, at doi:10.1016/j.virusres.2011.08.004.
Appendix A. Supplementary data
References
- Brandao P.E., Castilho J.G., Fahl W., Carnieli P., Jr., Oliveira R., Macedo C.I., Carrieri M.L., Kotait I. Short-interfering RNAs as antivirals against rabies. Braz. J. Infect. Dis. 2007;11:224–225. doi: 10.1590/s1413-86702007000200011. [DOI] [PubMed] [Google Scholar]
- Etessami R., Conzelmann K.K., Fadai-Ghothi B., Natelson B., Tsiang H., Ceccaldi P.E. Spread and pathogenic characteristics of a G-deficient rabies virus recombinant: an in vitro and in vivo study. J. Gen. Virol. 2000;81:2147–2153. doi: 10.1099/0022-1317-81-9-2147. [DOI] [PubMed] [Google Scholar]
- Fekadu M., Shaddock J.H. Peripheral distribution of virus in dogs inoculated with two strains of rabies virus. Am. J. Vet. Res. 1984;45:724–729. [PubMed] [Google Scholar]
- Israsena N., Supavonwong P., Ratanasetyuth N., Khawplod P., Hemachudha T. Inhibition of rabies virus replication by multiple artificial microRNAs. Antiviral Res. 2009;84:76–83. doi: 10.1016/j.antiviral.2009.07.012. [DOI] [PubMed] [Google Scholar]
- Iwasaki Y., Tobita M. In: Pathology. Rabies, Jackson A.C., Wunner W.H., editors. Academic Press; San Diego: 2002. pp. 283–306. [Google Scholar]
- Kumar P., Lee S.K., Shankar P., Manjunath N. A single siRNA suppresses fatal encephalitis induced by two different flaviviruses. PLoS Med. 2006;3:505–514. doi: 10.1371/journal.pmed.0030096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar P., Wu H., McBride J.L., Jung K.E., Kim M.H., Davidson B.L., Lee S.K., Shankar P., Manjunath N. Transvascular delivery of small interfering RNA to the central nervous system. Nature. 2007;448:39–43. doi: 10.1038/nature05901. [DOI] [PubMed] [Google Scholar]
- Kumar S., Arankalle V.A. Intracranial administration of P gene siRNA protects mice from lethal Chandipura virus encephalitis. PLoS One. 2010;5(1):e8615. doi: 10.1371/journal.pone.0008615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma Y., Chan C.Y., He M.L. RNA interference and antiviral therapy. World J. Gastroenterol. 2007;13:5169–5179. doi: 10.3748/wjg.v13.i39.5169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meng H., Cui F.Y., Chen X.H., Pan Y.C. Inhibition of gene expression directed by small interfering RNAs in infectious bronchitis virus. Acta Virol. 2007;51:265–269. [PubMed] [Google Scholar]
- Murphy F.A. Rabies pathogenesis brief review. Arch. Virol. 1985;54:279. doi: 10.1007/BF01314774. [DOI] [PubMed] [Google Scholar]
- Ong S.P., Chu J.J.H., Ng M.L. Inhibition of West Nile virus replication in cells stably transfected with vector-based shRNA expression system. Virus Res. 2008;135:292–297. doi: 10.1016/j.virusres.2008.04.014. [DOI] [PubMed] [Google Scholar]
- Pfaffl M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001;29:2003–2007. doi: 10.1093/nar/29.9.e45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Posadas I., Guerra F.J., Cena V. Nonviral vectors for the delivery of small interfering RNAs to the CNS. Nanomedicine (London) 2010;5:1219–1236. doi: 10.2217/nnm.10.105. [DOI] [PubMed] [Google Scholar]
- Sanchez A.B., Perez M., Cornu T., Torre J.C. RNA interference-mediated virus clearance from cells both acutely and chronically infected with the prototypic arenavirus lymphocytic choriomeningitis virus. J. Virol. 2005;79:11071–11081. doi: 10.1128/JVI.79.17.11071-11081.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sliva K., Schnierle B.S. Selective gene silencing by viral delivery of short hairpin RNA. Virol. J. 2010;7:248. doi: 10.1186/1743-422X-7-248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ui-Tei K., Naito Y., Nishi K., Juni A., Saigo K. Thermodynamic stability and Watson–Crick base pairing in the seed duplex are major determinants of the efficiency of the siRNA-based off-target effect. Nucleic Acids Res. 2008;36:7100–7109. doi: 10.1093/nar/gkn902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiktor T.J., MacFarlan R.I., Foggin C.M., Koprowski H. Antigenic analysis of rabies and mokola virus from Zimbabwe using monoclonal antibodies. Dev. Biol. Stand. 1984;57:199–211. [PubMed] [Google Scholar]
- Wilbur L.A., Aubert F.A. The NIH test for potency. In: Meslin F.X., Kaplan M.M., Koprowski H., editors. Laboratory Techniques in Rabies. WHO; Geneva, Switzerland: 1996. pp. 360–368. [Google Scholar]
- Wunner W.H. The chemical composition and molecular structure of rabies viruses. In: Baer G.M., editor. The Natural History of Rabies. CRC Press; Boca Raton, FL: 1991. pp. 31–67. [Google Scholar]
- Wunner W.H., Pallatroni C., Curtis P.J. Selection of genetic inhibitors of rabies virus. Arch. Virol. 2004;149:1653–1662. doi: 10.1007/s00705-004-0299-6. [DOI] [PubMed] [Google Scholar]
- Xia H., Mao Q., Paulson H.L., Davidson B.L. siRNA-mediated gene silencing in vitro and in vivo. Nat. Biotechnol. 2002;20:1006–1010. doi: 10.1038/nbt739. [DOI] [PubMed] [Google Scholar]
- Yan X., Mohankumar P.S., Dietzschold B., Schnell M.J., Fu Z.F. The rabies virus glycoprotein determines the distribution of different rabies virus strains in the brain. J. Neuro Virol. 2002;8:345–352. doi: 10.1080/13550280290100707. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


