Abstract
Meliacine (MA), an antiviral principle present in partially purified leaf extracts of Melia azedarach L., prevents the development of herpetic stromal keratitis (HSK) in mice by diminishing the viral load in the eye and the severity of lesions caused by a virus-induced immunopathological reaction.
The tetranortriterpenoid 1-cinnamoyl-3,11-dihydroxymeliacarpin (CDM), obtained from MA purification, displays anti-herpetic activity and impedes nuclear factor κB (NF-κB) activation in HSV-1 infected conjunctival cells.
To extend our understanding about CDM biological properties, we investigated its anti-HSV-1 activity as well as the effect on NF-κB activation and cytokine secretion induced by viral (HSV-1) and no-viral (LPS) stimuli, in corneal cells and macrophages.
CDM exerted a potent anti-HSV-1 effect on corneal cells and inhibited NF-κB translocation to the nucleus, leading to a decrease in IL-6 production. Besides, CDM seemed to modulate IL-6 and TNF-α responses in macrophages, whether they were infected with HSV-1 or stimulated with LPS. However, CDM did not affect NF-κB activation in these cells, suggesting that an alternative NF-κB cell signaling pathway would be involved in the modulation of cytokine production.
We conclude that, in addition to its antiviral effect, CDM would be acting as an immunomodulating compound which would be responsible for the improvement of murine HSK already reported.
Keywords: Antiviral, Immunomodulatory, NF-κB, Cytokine, HSV, Medicinal plants
1. Introduction
A great variety of ethnomedicinal plants are being studied as a source of natural products useful in the development of novel drugs. It has been established that many of them inhibit several steps of the viral replication cycle of many DNA and/or RNA viruses (Chattopadhyay and Naik, 2007).
We have reported that meliacine (MA), an antiviral principle present in partially purified leaf extracts of Melia azedarach L., exerts an antiviral action on the development of herpetic stromal keratitis (HSK) in mice by causing a significant decrease in the viral load in the eye of Herpes simplex virus type 1 (HSV-1) infected animals, as well as in the incidence and severity of lesions due to a virus-induced immunopathological reaction (Pifarré et al., 2002). Bioassay guided purification of MA led to the isolation of the limonoid 1-cinnamoyl-3,11-dihydroxymeliacarpin (CDM) that reduces both vesicular stomatitis virus (VSV) and HSV-1 multiplication in Vero cells (Alché et al., 2003). We have found that a late step in VSV and HSV-1 multiplication cycles is hindered by CDM since glycoproteins (g) B, gC and gD of HSV-1, as well as gG of VSV, are confined to the Golgi apparatus when CDM is added after infection (Barquero et al., 2004, Barquero et al., 2006).
HSV-1-induced ocular disease occurs as a result of a primary infection in the corneal epithelium and then, cells like macrophages intervene in clearing the virus from the infected eye and in the development of the immunologically driven stromal keratitis (Bauer et al., 2002, Biswas and Rouse, 2005). Besides, conjunctival cells are also involved in amplifying the inflammatory processes in the eye (Kase et al., 2004).
It has been shown that activation of nuclear factor κB (NF-κB) plays a pivotal role in triggering an immune inflammatory response to a range of stimuli, including viral infections, such as HIV-1, human T cell leukemia virus type 1, hepatitis B and influenza viruses. It has been reported that HSV-1 induces a strong nuclear translocation of NF-κB in human cell lines that could have several functions: to promote viral replication, prevent virus-induced apoptosis, and mediate the immune response to the invading pathogen (Gregory et al., 2004, Hiscott et al., 2001).
A great number of plant-derived substances, such as sesquiterpenes, di- and triterpenes, that prevent NF-κB activation, have been described (Nam, 2006). We have found that CDM is able to block HSV-1 induced activation of NF-κB by inhibiting its translocation to the nucleus of infected human conjunctival cells (NHC), and postulated that CDM would be able to abolish murine HSK by controlling viral spread and the associated immunopathology as well (Barquero et al., 2006).
The aim of the present study was to determine whether CDM displays an antiviral activity in infected corneal cells, the target of HSV-1 multiplication in vivo, as well as its effect on the translocation of NF-κB to the nucleus. Since NF-κB plays a particularly important role as far as expression of cytokines is concerned (Santoro et al., 2003), we also evaluated the modulating effect of CDM on the production of different cytokines in corneal cells and macrophages.
2. Materials and methods
2.1. Cells and viruses
Human Corneal-Limbal Epithelial (HCLE) cells were kindly provided by Dr Ilene K. Gipson and Dr. Pablo Argüeso (The Schepens Eye Research Institute, Harvard Medical School, Boston, USA) and grown in GIBCO Keratinocyte Serum Free Medium, supplemented with 25 μg/ml bovine pituitary extract (BPE), 0.2 ng/ml epidermal growth factor (EGF), and 0.4 mM CaCl2, and maintained in low calcium DMEM/F12.
Murine macrophage cell line J774A.1 was kindly provided by Dr. Osvaldo Zabal (INTA–Castelar, Buenos Aires) and grown in RPMI 1640 medium supplemented with 10% inactivated fetal bovine serum (FBS) (RPMI 10%) and maintained in RPMI supplemented with 2% inactivated FBS (RPMI 2%).
Murine L929 cells and Vero cells were grown in Eagle's minimal essential medium supplemented with 10% inactivated FBS (MEM 10%), and maintained in MEM supplemented with 1.5% inactivated FBS (MEM 1.5%).
The HSV-1 KOS strain was propagated at low multiplicity and used for in vitro experiments.
2.2. Reagents
LPS from E. coli serotype 055: B5 was obtained from Sigma. The rabbit polyclonal anti-p65 and anti-IκBα antibodies, and the mouse monoclonal antibody anti-gD of HSV-1 were obtained from Santa Cruz Biotechnology, USA. The monoclonal anti-calnexin antibody was obtained from Chemicon. The anti-actin (Merck) antibody was kindly provided by Dr. Viviana Castilla, Laboratory of Virology, School of Sciences, University of Buenos Aires. Secondary goat anti-rabbit FluoroLink™ Cy™2 and anti-mouse FluoroLink™ Cy™3 antibodies were purchased from GE Healthcare Bio-Sciences, Argentina. Peroxidase-conjugated goat anti-rabbit or anti-mouse antibodies were obtained from ICN Immunobiological.
2.3. Antiviral compound
CDM was purified from leaves of M. azedarach L., as described by Alché et al. (2003), solubilized in MEM 1.5% to a final concentration of 1 mg/ml (1.5 mM), and stored at −20 °C.
2.4. Cytotoxicity assay
Cell viability in the presence of the compound was determined using the cleavage of the tetrazolium salt MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) (Sigma) by the mitochondrial enzyme succinate dehydrogenase to give a blue product (formazan) (Denizot and Lang, 1986). HCLE cells were seeded at a concentration of 104 cells/well in 96-well plates and grown at 37 °C for 24 h. The culture medium was replaced by DMEM/F12 medium containing CDM at various concentrations by triplicate, and cells were further grown for 24 h. After that, we added 0.01 ml MTT (5 mg/ml in distilled water) to cells in culture medium. After MTT cleavage (2 h at 37 °C), formazan product was solubilized by the addition of 0.2 ml of ethanol. The absorbance of each well was measured on an Eurogenetics MPR-A 4i microplate reader using a test wavelength of 570 nm and a reference wavelength of 630 nm. Results were expressed as a percentage of absorbance of treated cell cultures with respect to untreated ones.
2.5. Antiviral activity
HCLE and J774A.1 cells grown in 96-well tissue culture plates were infected with HSV-1 at a multiplicity of infection (m.o.i.) of 0.067 PFU/cell. After 1 h adsorption at 37 °C, the inoculum was removed and medium containing different concentrations of CDM was added, by triplicate. The plates were incubated at 37 °C in 4% CO2 atmosphere until 100% cell death was observed microscopically in untreated infected control cells, approximately at 24 h post-infection (p.i.). After cell disruption by three cycles of freezing and thawing, supernatants were harvested and pooled. Virus yields were titrated by plaque assay in Vero cells and the effective concentration (EC50) values were calculated as the concentration of CDM required to reduce the yield of infectious virus by 50% relative to the untreated virus control.
2.6. Acridine orange staining of living cells
HCLE cells grown on coverslips were treated with 40 μM CDM for 2 h at 37 °C and stained with acridine orange (1 μg/ml) for 15 min at 37 °C. Then, cells were washed twice with cold PBS, mounted on PBS and visualized on an Olympus BX51 with epifluorescence optics.
2.7. Indirect immunofluorescence assay (IFI)
Subconfluent cells grown on glass coverslips in 24-well plates were fixed with methanol for 10 min at −20 °C. After three washes with PBS, the coverslips were inverted on a drop of diluted primary antibody for 30 min at 37 °C, and then returned to culture dishes and subjected to three additional washes with PBS. Afterwards, cells were incubated with diluted secondary antibody for 30 min at 37 °C.
Finally, coverslips were rinsed, mounted and photographed with an Olympus FB300 confocal microscope or an Olympus BX51 with epifluorescence optics.
2.8. Western blot analysis
Whole extracts from cells grown in 24-well plates for 24 h were loaded on 10% sodium dodecyl sulphate-polyacrilamide gel electrophoresis (SDS-PAGE) and transferred onto polyvinylidene fluoride (PVDF) membranes for 60 min at 75 mA. Membranes were blocked in PBS containing 5% unfitted milk overnight and then incubated with diluted primary antibodies for 2 h at 37 °C. After washing, membranes were incubated with diluted peroxidase-conjugated antibodies for 1.5 h at 37 °C. The immunoreactive bands were visualized using an enhanced chemiluminesence system (ECL, PerkinElmer). Calnexin and actin were used as loading controls.
2.9. Cytokine determination
Cells were frozen and thawed, and then, supernatants were harvested, centrifuged at 1000 rpm for 10 min, and cytokines were quantified by ELISA, or in a biological assay by triplicate.
Human TNF-α and IL-6, and mouse IL-6 were quantified by commercial ELISA sets (BD OptEIATM, Becton Dickinson, USA) according to manufacturer instructions.
Measurement of mouse TNF-α bioactivity was performed with the L929 cell-based bioassay (Decker et al., 1987), with minor modifications. L929 cells were grown in 96-well culture plates (2 × 104 cells/well) for 24 h at 37 °C. Supernatants were removed and substituted with the samples to be assayed for TNF-α content in successive twofold dilutions and incubated at 38.5 °C with 5 μg/ml of Actinomycin D (AcD) (Sidus, Argentina), for 22 h. Cells were fixed in 10% formaldehyde and stained with crystal violet 0.05%. To assess TNF-α activity, light absorbance at 580 nm of the eluted crystal violet from the samples was measured and compared to a mouse recombinant TNF-α standard dilution series (Sigma). The bioassay was specific for TNF-α since the activity was neutralized by an antibody against TNF-α.
2.10. Statistical analysis
Student's t-test was used for statistical analysis of all data.
3. Results
3.1. Antiviral activity of CDM in a corneal cell line
It is well known that HSV-1 establishes infection usually in the epithelial layer of the cornea (Deshpande et al., 2004). Considering that CDM suppressed HSV-1 replication in Vero and human conjunctival cells, we decided to investigate the anti-HSV-1 effect of CDM in HCLE cells where HSV-1 multiplied reaching a maximum viral titer of 5.3 × 106 at 24 h p.i. (Alché et al., 2003, Barquero et al., 2006).
When HCLE cells infected with HSV-1 were treated with different concentrations of CDM, a dose-dependent inhibition of viral yields was observed, and an EC50 value of 0.78 μM was calculated. Besides, CDM proved to have no cytotoxic effect at all the concentrations tested (Fig. 1A).
Fig. 1.
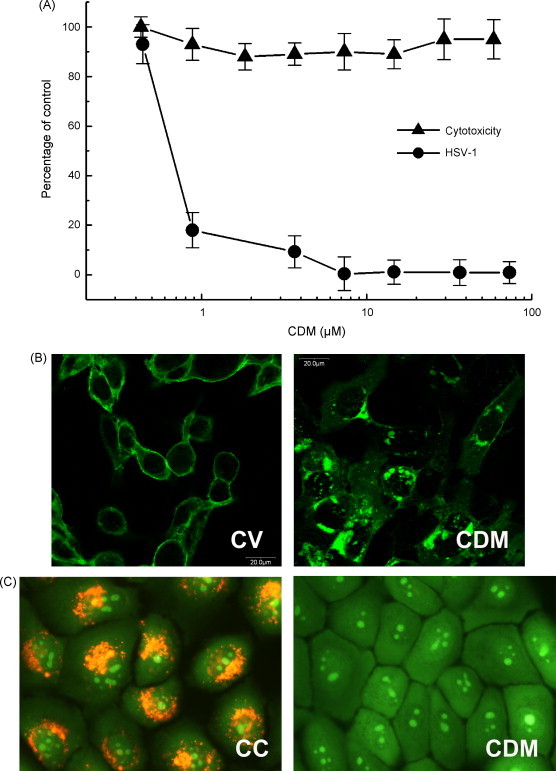
Antiviral activity of CDM in HCLE cells. (A) HSV-1 infected cells were treated with different concentrations of CDM. After 24 h, supernatants were harvested and titrated by plaque assay and cytotoxicity of CDM was determined in uninfected treated cultures by the MTT assay. Data are expressed as the mean ± S.D. of two separate experiments. (B) HSV-1-infected cells were treated with CDM or not (CV) and after 13 h p.i. the intracellular localization of gD was done by IFI staining. Magnification 400×. (C) Cells grown on coverslips treated with CDM or not (CC) were stained with acridine orange and visualized on an epifluorescence microscope. Magnification 400×.
We have previously reported that CDM displays its antiviral action by affecting the trafficking of gB, gC and gD of HSV-1 in Vero and NHC cells when supplied after infection (Barquero et al., 2006). In order to study the effect of CDM on the transport of viral glycoproteins in corneal cells, HCLE cells were infected with HSV-1 (m.o.i. = 2) and treated with CDM (40 μM). When a total IFI staining was performed using an anti-gD monoclonal antibody, we found that gD of HSV-1 exhibited a perinuclear localization associated with the Golgi apparatus (Fig. 1B).
It has been already shown that the inhibition of viral glycoprotein transport may be related to the perturbation of the acidic pH of intracellular organelles (Sidhu et al., 1999). Although the mechanism by which CDM affects the exocytic pathway is still unraveled, we have shown that CDM provokes the basification of the pH of the endosomal vesicles in Vero cells (Barquero et al., 2004).
Now, we demonstrated that CDM also modified the endosomal pH of HCLE cells. The vital fluorescence microscopic study revealed that untreated cells exhibited a bright orange punctate fluorescence concentrated in low-pH vesicles. Nevertheless, the pH of acidic intracellular vesicles from CDM-treated cells was markedly affected since only a faint granular fluorescence was observed after 2 h of treatment (Fig. 1C).
In summary, CDM induced HCLE cytoplasmic alkalinization and exerted a potent anti-HSV-1 effect by confining HSV-1 glycoproteins to the Golgi apparatus.
3.2. Effect of CDM on NF-κB nuclear translocation, IκBα degradation and cytokine production induced by HSV-1 in corneal cells
We have previously shown that CDM blocks HSV-1 induced activation of NF-κB by inhibiting its translocation to the nucleus of conjunctival cells (Barquero et al., 2006). Hence, to examine the effect of CDM on NF-κB intracellular localization, we infected HCLE cells with HSV-1 (m.o.i. = 1) and, at 24 h p.i., cells were fixed and processed for an IFI staining using a rabbit polyclonal anti-p65 antibody.
By visual inspection of the images, we observed that p65 remained in the cytoplasm of uninfected cells, whereas HSV-1 induced NF-κB translocation to the nucleus of HCLE cells.
These qualitative observations were corroborated by calculating the percentage of translocation as the number of cells with fluorescence in the nucleus relative to the total cell number, in 50 cells per coverslip. NF-κB nuclear translocation occurred in the majority of infected cells (92%), whereas viral-induced NF-κB activation was efficiently impaired by CDM, since only 20% of HCLE treated-infected cells retained p65 in their nuclei (Fig. 2A).
Fig. 2.
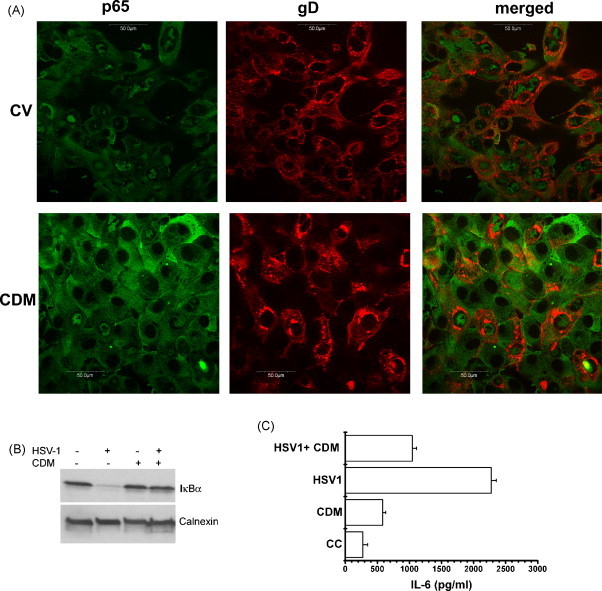
Effect of CDM on NF-κB nuclear translocation, IκBα degradation and cytokine production induced by HSV-1 in HCLE cells. (A) HSV-1-infected cells were treated with CDM or not (CV). Double IFI staining was performed by adding anti-p65 and anti-HSV-1 gD antibodies to methanol fixed cells. NF-κB translocation and gD expression were analyzed by confocal microscopy. Magnification 600×. (B) HSV-1 infected cells were treated or not with CDM. After 24 h, cells were lysed and subjected to SDS-PAGE, followed by immunoblotting with antibodies against IκBα. (C) Cells were infected with HSV-1 and treated or not with CDM. After 24 h, IL-6 was determined by ELISA. Data are expressed as the mean ± S.D. of two separate experiments.
Together with NF-κB, we also detected HSV-1 gD as a marker of viral infection, by a double IFI staining. We found that nuclear p65 fluorescence in the nuclei of infected cells was associated with gD expression. When HCLE infected cells were treated with CDM, both p65 and gD were detected coincidently in a few and scattered foci of infection (Fig. 2A).
Thus, the inhibition of NF-κB translocation in treated-infected cells could be ascribed to the restraint of viral propagation caused by CDM rather than to an intrinsic effect on NF-κB.
Since the translocation of NF-κB to the nucleus is preceded by the proteolytic degradation of its inhibitor in the cytoplasm, IκBα, we determined whether the inhibition of NF-κB activation by CDM was concomitant to IκBα degradation.
HCLE cells infected with HSV-1 (m.o.i. = 1) were treated or not with 40 μM of CDM, and, at 24 h p.i., cells were processed through Western blot analysis by using a rabbit polyclonal anti IκBα antibody. We found that IκBα was almost completely degraded after infection with HSV-1; as expected, the addition of CDM hampered HSV-1 induced persistent IκBα degradation. Besides, we observed that CDM didn’t induce IκBα degradation in uninfected cells (Fig. 2B).
It has been already reported that HSV-1 is able to activate NF-κB in human telomerase-immortalized corneal epithelial cells (HUCL) concomitant with a transcriptional expression of IL-6 and TNF-α, among other cytokines (Li et al., 2005). However, HCLE cells failed to produce TNF-α after infection with HSV-1 (Michelini et al., 2007). To assess the biological relevance of the inhibition of NF-κB translocation provoked by CDM on cytokine production, we measured its effect on IL-6 secretion.
Supernatants harvested from HCLE cells infected with HSV-1 (m.o.i. = 1) treated or not with CDM (40 μM), were used to quantify IL-6 by ELISA, in triplicate. We found that the production of IL-6 was ten fold higher than that of uninfected cells (p < 0.005). This increase was significantly reduced by half when CDM was added to HSV-1 infected cells (p < 0.005). In uninfected cells, no significant differences between IL-6 release from non-treated and CDM-treated cells were detected (Fig. 2C).
Taken together, these results suggest that NF-κB translocation prevented as a consequence of the anti-HSV-1 activity of CDM, would lead to a decrease in IL-6 production.
3.3. Effect of CDM on NF-κB nuclear translocation, IκBα degradation and cytokine production in macrophages
Although corneal epithelial cells constitute the first line of defence after ocular HSV-1 infection, macrophages also play a crucial role as one of the dominant cell infiltrates in the infected cornea (Mott et al., 2007). Thus, we decided to evaluate the effect of CDM in J774A.1 cells infected with HSV-1.
Consequently, we first investigated the antiviral action of CDM, finding that it inhibited HSV-1 multiplication with an EC50 value of 12.05 μM, without cytotoxic effect (data not shown).
As it has been described in other cell lines, a similar pattern of NF-κB translocation and IκBα degradation was observed in J774A.1 cells after HSV-1 infection (Gregory et al., 2004). By means of an IFI staining, we found that p65 fluorescence remained in the cytoplasm of uninfected cells, whereas HSV-1 induces translocation to the nucleus in 82% of infected J774A.1 cells, which correlated with IκBα degradation observed through an immunoblot assay. When CDM was added, the percentage of cells exhibiting NF-κB translocation to the nucleus dropped to 17% and, as expected, IκBα degradation was inhibited in CDM-treated infected cells (Fig. 3A and B).
Fig. 3.
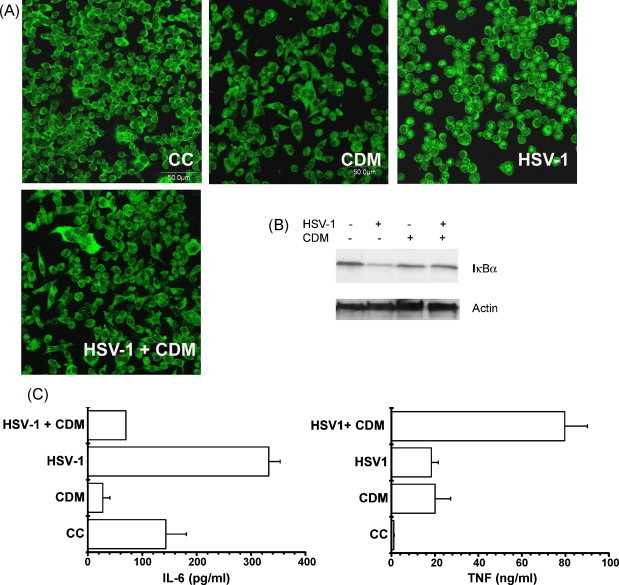
Effect of CDM on NF-κB nuclear translocation, IκBα degradation and cytokine production induced by HSV-1 in J774A.1 cells. (A) Cells were infected with HSV-1 and treated or not with CDM after virus adsorption. IFI staining was performed by adding anti-p65 antibodies to methanol fixed cells and NF-κB translocation was analyzed by confocal microscopy. Magnification 400×. (CC) non-infected cells; (CDM) non-infected CDM treated cells; (HSV-1) HSV-1 infected cells; (HSV-1 + CDM) HSV-1 infected cells treated with CDM. (B) Cells were infected with HSV-1 and treated or not with CDM after virus adsorption. After 24 h, cells were lysed and subjected to SDS-PAGE, followed by immunoblotting with antibodies against IκBα. (C) Cells were infected with HSV-1 and treated or not with CDM. After 24 h, TNF-α was quantified through a biological assay and IL-6 was determined by ELISA. Data are expressed as the mean ± S.D. of two separate experiments.
It is well established that HSV-1 infection triggers pro-inflammatory cytokine production in macrophages (Biswas and Rouse, 2005; Melchjorsen et al., 2006). Thus, we investigated the effect of CDM on IL-6 and TNF-α secretion by ELISA and a biological assay, respectively.
In the case of uninfected cells, levels of IL-6 and TNF-α accumulated in the culture medium for 24 h varied between control and CDM-treated J774A.1 cells. While the concentration of IL-6 was significantly lowered in the supernatants from CDM-treated cells with respect to untreated J774A.1 cells (p < 0.01), the TNF-α yield belonging to CDM-treated cells was much higher than that accumulated in control cells (p < 0.05) (Fig. 3C).
As shown in Fig. 3C, HSV-1 infection induced macrophages to secrete increased amounts of IL-6 and TNF-α in comparison with uninfected cells (p < 0.01 and p < 0.05, respectively). However, CDM-treated cells failed to produce IL-6 in response to HSV-1 infection (p < 0.005), whereas a strong enhancement in TNF-α secretion was observed (p < 0.05).
Hence, CDM seemed to modulate the response of both cytokines in J774A.1 cells, whether infected or not.
Since it has been previously shown that the addition of increasing concentrations of MA to LPS-stimulated mouse peritoneal macrophages synergized the amount of TNF-α released in a dose-dependent manner, we used LPS as a stimulus instead of HSV-1 to evaluate solely the immunomodulating effect of CDM (Petrera and Coto, 2003).
J774A.1 cells were stimulated with LPS (10 μg/ml) in the presence or absence of CDM (40 μM) for 30 min at 37 °C. When cells were stained for NF-κB p65 subunit detection, we observed that CDM did not inhibit LPS induced-NF-κB translocation (Fig. 4A). Besides, CDM did not affect the kinetic of IκBα degradation induced by LPS. CDM alone neither activated NF-κB nor led to IκBα degradation (Fig. 4A and B).
Fig. 4.
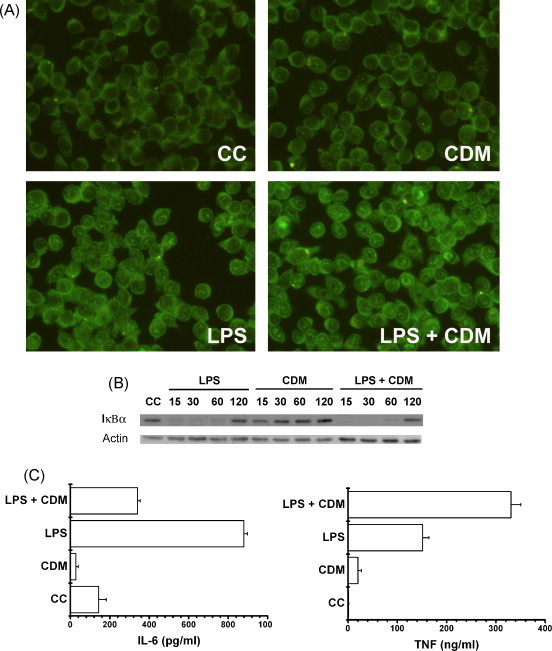
Effect of CDM on NF-κB nuclear translocation, IκBα degradation and cytokine production induced by LPS in J774A.1 cells. (A) Cells were stimulated with 10 μg/ml LPS for 30 min, and treated or not with CDM. The localization of p65 was detected by IFI staining in methanol fixed cells. Magnification 400×. (CC) Control cells; (CDM) non-stimulated cells treated with CDM; (LPS) LPS-stimulated cells; (LPS + CDM) LPS-stimulated cells treated with CDM. (B) J774A.1 cells were stimulated with LPS and treated or not with CDM. After 15, 30, 60 and 120 min, cells were lysed and subjected to SDS-PAGE, followed by immunoblotting with antibodies against IκBα. (C) Cells were stimulated with LPS for 8 h, and treated or not with CDM. TNF-α yield was quantified through a biological assay and IL-6 was determined by ELISA. Data are expressed as the mean ± S.D. of two separate experiments.
Taking into account that CDM did not affect LPS-induced NF-κB translocation, it was important to determine if CDM was able to affect IL-6 and TNF-α yields. When J774A.1 cells were stimulated with LPS during 8 h, the release of IL-6 and TNF-α was enhanced significantly (p < 0.001 and p < 0.01, respectively) (Fig. 4C). Nevertheless, CDM considerably reduced the release of IL-6 (p < 0.001), and strongly enhanced TNF-α production (p < 0.01), after 8 h of treatment of LPS-stimulated J774A.1 cells (Fig. 4C).
In these cells, CDM inhibited NF-κB activation and IκBα degradation depending on the fact that macrophages were induced with LPS or infected with HSV-1, while it modulated IL-6 and TNF-α production regardless the stimuli used.
4. Discussion
Since a long time, medicinal plants have been used to treat viral infections. The chemical diversity, structural complexity, lack of substantial toxic effects, and broad spectrum of antiviral activity of natural products, make them ideal candidates for new therapeutics (Mukhtar et al., 2008). In fact, terpenoids isolated from medicinal plants have attracted attention because many of them exhibit specific antiviral effect against HSV-1 and 2, and the coronavirus Sars-CoV, in vitro (Wen et al., 2007). Triterpenoids and limonoids isolated from Meliaceae plants proved to inhibit HSV-1, HIV-1 and RSV multiplication (Chen et al., 2007, Joycharat et al., 2008; Manners, 2007).
The murine model of HSV-induced ocular disease provides valuable information related to the viral and immune stages of the HSK. We have previously reported that topical administration of MA in the corneas of HSV-1 infected mice exerts its therapeutic effect by reducing viral load as well as by abolishing the ocular inflammatory reaction and neovascularization, displaying, in addition to its antiviral activity, an eventual immunomodulatory effect (Pifarré et al., 2002). In another report, we described the chemical nature of the antiviral principle (CDM) and its mechanism of action in Vero and NHC cells infected with HSV-1 (Barquero et al., 2004, Barquero et al., 2006).
In the present paper, we have shown that CDM displayed a potent antiviral effect in HCLE cells (Fig. 1A), 25-fold higher in comparison to that obtained in Vero cells (Alché et al., 2003). This is important because HSV-1 replication in the eye occurs almost exclusively in corneal epithelial cells (Biswas and Rouse, 2005). The direct anti-HSV-1 activity of CDM achieved when the compound is supplied after infection has already been demonstrated: CDM affects gB, gC and gD glycoprotein exocytic pathway from NHC and Vero cells, confining them to the Golgi complex (Barquero et al., 2006). Now, we demonstrate that CDM also affects the trafficking of, at least, gD glycoprotein of HSV-1 by exerting an analogous antiviral effect in HCLE cells (Fig. 1B). Furthermore, the hindrance of HSV-1 multiplication by CDM is responsible for the inhibition of NF- κB translocation in corneal cells (Fig. 2A). Taken together, these results would explain the reduction of virus yield in the eyes of MA-treated infected mice (Pifarré et al., 2002).
In recent past, a number of studies have explored immunomodulatory properties of plant extracts having antiviral properties (Mukhtar et al., 2008). A methanolic extract of Capparis spinosa L. buds up-regulates the expression of proinflammatory cytokines, such as IL-12, IFN-γ and TNF-α, besides its anti-HSV activity (Arena et al., 2008). Likewise, pure compounds from Plantago genus enhance the activity of lymphocyte proliferation and secretion of IFN-γ (Chiang et al., 2003).
The development of murine HSK involves production of proinflammatory cytokines, chemokines and neovascularization, and takes place between 5 and 7 days after infection, once the virus was cleared from the eye. Interestingly, MA prevents ocular disease in HSV-1 infected mice when administered from 4 days p.i. onwards, suggesting the presence of a compound with immunomodulating activity (Pifarré et al., 2002). In this sense, now we have found that CDM modulates cytokine production in vitro (Fig. 2, Fig. 3, Fig. 4).
IL-6 and TNF-α are some of the numerous cytokines known to be upregulated following HSV-1 ocular infection (Biswas and Rouse, 2005). The significant reduction in IL-6 secretion detected in supernatants from CDM treated-infected epithelial cells and macrophages would be related to the inhibition of NF-κB translocation by CDM, as a result of its antiviral activity (Fig. 1, Fig. 2, Fig. 3). On the contrary, the strong increase of TNF-α secretion in J774A.1 CDM-treated infected cells observed in the absence of NF-κB activation (Fig. 3C) would be exerted through an alternative NF-κB cell signaling pathway, such as the activation of distinct mitogen-activated protein (MAP) kinase cascades (Diao et al., 2005, Zachos et al., 1999). This possibility might be supported by the fact that CDM enhanced levels of TNF-α production, decreased IL-6 secretion and did not impede NF-κB translocation when J774A.1 cells were induced with LPS rather than infected with HSV-1 (Fig. 4).
It has been demonstrated that both cytokines play a different role in corneal angiogenesis: while IL-6 induces the production of vascular endothelial growth factor (VEGF), TNF-α may counter the induction effect of VEGF on vascular endothelial cells, resulting in a blockade of neovascularization in injured corneas in vivo (Biswas et al., 2006, Fujita et al., 2007). If this is the case, we would argue that the strong increase of TNF-α secretion together with IL-6 reduction due to CDM could be responsible for the inhibition of neovascularization registered when mice were infected in the cornea with HSV-1 and treated with MA.
We hypothesize that the improvement of HSK reported with MA may be a consequence of the antiviral and immunomodulating effects observed in vitro with CDM. Further studies on the antiangiogenic and immunomodulating effects of CDM will be needed to decipher its mechanism of action, since the availability of a compound gathering together both biological properties will make it a viable candidate for a new and novel anti-HSV agent.
Acknowledgements
The authors wish to thank Isabel Paz and Guillermo Assad Ferek for their technical assistance. This work was supported by grants from the Agencia Nacional de Promoción Científica y Técnica (ANPCyT) (PICT 38260/05) and CONICET (PIP 6033). Dr. A.A. Barquero, Dr. M.S. Maier and Dr. L.E. Alché are Research Members of the National Research Council of Argentina (CONICET).
Author contribution: C.B., A.B. and L.A. designed research; C.B. performed research; A.B. contributed with acridine orange staining and H.D.C. with TNF-α bioassay; M.M. provided pure CDM; C.B., A.B. and L.A. analyzed data; C.B., A.B. and L.A. wrote the paper.
References
- Alché L.E., Assad Ferek G., Meo M., Coto C.E., Maier M.S. An antiviral meliacarpin from leaves of Melia azedarach L. Z. Naturforsch. 2003;58c:215–219. doi: 10.1515/znc-2003-3-413. [DOI] [PubMed] [Google Scholar]
- Arena A., Bisignano G., Pavone B., Tomaino A., Bonina F.P., Saija A., Cristani M., D’Arrigo M., Trombetta D. Antiviral and immunomodulatory effect of a lyophilized extract of Capparis spinosa L. buds. Phytother. Res. 2008;22:313–317. doi: 10.1002/ptr.2313. [DOI] [PubMed] [Google Scholar]
- Barquero A.A., Alché L.E., Coto C.E. Block of vesicular stomatitis virus endocytic and exocytic pathways by 1-cinnamoyl-3, 11-dihydroxymeliacarpin, a tetranortriterpenoid of natural origin. J. Gen. Virol. 2004;85:483–493. doi: 10.1099/vir.0.19343-0. [DOI] [PubMed] [Google Scholar]
- Barquero A.A., Michelini F.M., Alché L.E. 1-cinnamoyl-3,11-dihydroxymeliacarpin is a natural compound with antiviral and nuclear factor-κB modulating properties. Biochem. Biophys. Res. Co. 2006;344:955–962. doi: 10.1016/j.bbrc.2006.03.226. [DOI] [PubMed] [Google Scholar]
- Bauer D., Schmitz A., Van Rooijen N., Steuhl K., Heiligenhaus A. Conjunctival macrophage-mediated influence of the local and systemic immune response after corneal herpes simplex virus-1 infection. Immunology. 2002;107:118–128. doi: 10.1046/j.1365-2567.2002.01477.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biswas P.S., Rouse B.T. Early events in HSV keratitis-setting the stage for a blinding disease. Microbes Infect. 2005;7:799–810. doi: 10.1016/j.micinf.2005.03.003. [DOI] [PubMed] [Google Scholar]
- Biswas P., Banerjee K., Kinchington P., Rouse B. Involvement of IL-6 in the paracrine production of VEGF in ocular HSV-1 infection. Exp. Eye Res. 2006;82:46–54. doi: 10.1016/j.exer.2005.05.001. [DOI] [PubMed] [Google Scholar]
- Chattopadhyay D., Naik T.N. Antivirals of ethnomedicinal origin: structure-activity relantionship and scope. Mini-Rev. Med. Chem. 2007;7:275–301. doi: 10.2174/138955707780059844. [DOI] [PubMed] [Google Scholar]
- Chen J.L., Kernan M.R., Jolad S.D., Stoddart C.A., Bogan M., Cooper R. Dysoxylins A-D, tetranortriterpenoids with potent anti-rsv activity from Dysoxylum gaudichaudianum. J. Nat. Prod. 2007;70:312–315. doi: 10.1021/np060398y. [DOI] [PubMed] [Google Scholar]
- Chiang L.C., Ng L.T., Chiang W., Chang M.Y., Lin C.C. Immunomodulatory activities of flavonoids, monoterpenoids, triterpenoids, iridoid glycosides and phenolic compounds of Plantago species. Planta Med. 2003;69:600–604. doi: 10.1055/s-2003-41113. [DOI] [PubMed] [Google Scholar]
- Decker T., Lohmann-Mathess M.L., Gifford G.E. Cell-associated tumor necrosis factor (TNF) as a killing mechanism of activated cytotoxic macrophages. J. Immunol. 1987;138:957–962. [PubMed] [Google Scholar]
- Denizot F., Lang R. Rapid colorimetric assay for cell growth and survival. J. Immunol. Methods. 1986;89:271–277. doi: 10.1016/0022-1759(86)90368-6. [DOI] [PubMed] [Google Scholar]
- Deshpande S., Banerjee K., Biswas P.S., Rouse B.T. Herpetic eye disease: immunopathogenesis and therapeutic measures. Expert Rev. Mol. Med. 2004;2004:1–14. doi: 10.1017/S1462399404007604. [DOI] [PubMed] [Google Scholar]
- Diao L., Zhang B., Xuan C., Sun S., Yang K., Tang Y., Qiao W., Chen Q., Geng Y., Wang C. Activation of c-Jun N-terminal kinase (JNK) pathway by HSV-1 immediate early protein ICP0. Exp. Cell Res. 2005;308:196–210. doi: 10.1016/j.yexcr.2005.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujita S., Saika S., Kao W.W., Fujita K., Miyamoto T., Ikeda K., Nakajima Y., Ohnishi Y. Endogenous TNF alpha suppression of neovascularization in corneal stroma in mice. Invest. Ophthalmol. Vis. Sci. 2007;48:3051–3055. doi: 10.1167/iovs.06-1083. [DOI] [PubMed] [Google Scholar]
- Gregory D., Hargett D., Holmes D., Money E., Bachenheimer S. Efficient replication by herpes simplex virus type 1 involves activation of the IkappaB kinase-IkappaB-p65 pathway. J. Virol. 2004;78:13582–13590. doi: 10.1128/JVI.78.24.13582-13590.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hiscott J., Kwon H., Genin P. Hostile takeovers: viral appropriation of the NF-kappaB pathway. J. Clin. Invest. 2001;107:143–151. doi: 10.1172/JCI11918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joycharat N., Greger H., Hofer O., Saifah E. Flavaglines and triterpenoids from the leaves of Aglaia forbesii. Phytochemistry. 2008;69:206–211. doi: 10.1016/j.phytochem.2007.06.016. [DOI] [PubMed] [Google Scholar]
- Kase S., Aoki K., Harada T., Harada C., Ohgami K., Shiratori K., Nishi S., Ohno S., Yoshida K. Activation of nuclear factor-kappa B in conjunctiva with epithelial scraping of the Mouse cornea and human epidemia keratoconjunctivitis. Br. J. Ophtalmol. 2004;88:947–949. doi: 10.1136/bjo.2003.024646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H., Zhang J., Kumar A., Zheng M., Atherton S.S., Yu F.X. Herpes simplex virus 1 infection induces the expression of proinflammatory cytokines, interferons and TLR7 in human corneal epithelial cells. Immunology. 2005;117:167–176. doi: 10.1111/j.1365-2567.2005.02275.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manners G. Citrus limonoids: analysis, bioactivity, and biomedical prospects. J. Agric. Food Chem. 2007;55:8285–8294. doi: 10.1021/jf071797h. [DOI] [PubMed] [Google Scholar]
- Melchjorsen J., Sirén J., Julkunen I., Paludan S., Matikainen S. Induction of cytokine expression by herpes simplex virus in human monocyte-derived macrophages and dendritic cells is dependent on virus replication and is counteracted by ICP27 targeting NF-kappaB and IRF-3. J. Gen. Virol. 2006;87:1099–1108. doi: 10.1099/vir.0.81541-0. [DOI] [PubMed] [Google Scholar]
- Michelini F.M., Berra A., Alché L.E. The in vitro immunomodulatory activity of a synthetic brassinosteroid analogue World account for the improvement of herpetic stromal keratitis in mice. J. Steroid. Biochem. 2007;108:164–170. doi: 10.1016/j.jsbmb.2007.10.002. [DOI] [PubMed] [Google Scholar]
- Mott K., Brick D.J., van Rooijen N., Ghiasi H. Macrophages are important determinants of acute ocular HSV-1 infection in immunized mice. Invest. Ophthalmol. Vis. Sci. 2007;48:5605–5615. doi: 10.1167/iovs.07-0894. [DOI] [PubMed] [Google Scholar]
- Mukhtar M., Arshad M., Ahmad M., Pomerantz R.J., Wigdahl B., Parveen Z. Antiviral potentials of medicinal plants. Virus Res. 2008;131:111–120. doi: 10.1016/j.virusres.2007.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nam N. Naturally occurring NF-κB inhibitors. Mini-Rev. Med. Chem. 2006;6:945–951. doi: 10.2174/138955706777934937. [DOI] [PubMed] [Google Scholar]
- Petrera E., Coto C.E. Effect of meliacine, a plant derived antiviral, on tumor necrosis factor alpha. Fitoterapia. 2003;74:77–83. doi: 10.1016/s0367-326x(02)00294-0. [DOI] [PubMed] [Google Scholar]
- Pifarré M.P., Berra A., Coto C.E., Alché L.A. Therapeutic action of Meliacine, a plant-derived antiviral, on HSV-induced ocular disease in mice. Exp. Eye Res. 2002;75:327–334. doi: 10.1016/s0014-4835(02)92027-0. [DOI] [PubMed] [Google Scholar]
- Santoro M.G., Rossi A., Amici C. NF-kappaB and virus infection: who controls whom. EMBO J. 2003;22:2552–2560. doi: 10.1093/emboj/cdg267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sidhu G., Singh A., Sundarrajan R.N., Sundar S.V., Maheshwari R.K. Role of vacuolar H+-ATPase in interferon-induced inhibition of viral glycoprotein transport. J. Interferon Cytokine Res. 1999;19:1297–1303. doi: 10.1089/107999099312975. [DOI] [PubMed] [Google Scholar]
- Wen C., Kuo Y., Jan J., Liang P., Wang S., Liu H., Lee C., Chang S., Kuo C., Lee S., Hou C., Hsiao P., Chien S., Shyur L., Yang N. Specific plant terpenoids and lignoids possess potent antiviral activities against severe acute respiratory syndrome coronavirus. J. Med. Chem. 2007;50:4087–4095. doi: 10.1021/jm070295s. [DOI] [PubMed] [Google Scholar]
- Zachos G., Clements B., Conner J. Herpes simplex virus type 1 infection stimulates p38/c-Jun N-terminal mitogen-activated protein kinase pathways and activates transcription factor AP-1. J. Biol. Chem. 1999;274:5097–5103. doi: 10.1074/jbc.274.8.5097. [DOI] [PubMed] [Google Scholar]


