Summary
Background
The rising number of children in daycare nurseries increases opportunities for the transmission of infectious diseases. Pathogens may be transmitted directly from child to child via sneezing, coughing and touching, or indirectly via the environment. Toys are among the fomites with the highest pathogen load, but their role in disease transmission is unknown.
Aim
To determine if washing and disinfection of toys can reduce sickness absence and microbial pathogen load in the nursery environment.
Methods
Twelve nurseries (caring for 587 children) were randomized to intervention and control groups. The intervention consisted of washing and disinfection of toys and linen every two weeks for three months by a commercial cleaning company. The extent and causes of sickness absence among the children were recorded in both groups before and after introduction of the intervention. Ten sampling points in each nursery were examined for bacteria and respiratory viruses.
Results
The presence of respiratory virus DNA/RNA was widespread, but very few pathogenic bacteria were found in the environment. The intervention reduced the presence of adenovirus [odds ratio (OR) 2.4, 95% confidence interval (CI) 1.1–5.0], rhinovirus (OR 5.3, 95% CI 2.3–12.4) and respiratory syncytial virus (OR 4.1, 95% CI 1.5–11.2) compared with the control group, but the intervention had no effect on sickness absence or disease patterns in the nurseries.
Conclusion
Although cleaning and disinfection of toys every two weeks can decrease the microbial load in nurseries, it does not appear to reduce sickness absence among nursery children.
Keywords: Hygiene, Cleaning, Paediatrics, Bacteria, Virus, Environment, Handwashing, Child care, Toys
Introduction
Children, especially children aged three years and under, have a high frequency of infectious disease episodes.1 Children in daycare nurseries have more infections than children cared for elsewhere,2, 3, 4 mainly because of direct transmission between children, contact or respiratory droplet transmission, and inadequate hand hygiene. Although indirect transmission of infection via the nursery environment has not been studied extensively, it is likely to play a role. Previous research on bacteria in the nursery environment has shown positive cultures in 10–60% of samples, depending on location, but almost all bacteria isolated were of low pathogenicity.5, 6, 7 Quantitative polymerase chain reaction (PCR) to determine the diversity of bacteria in the nurseries has shown that the most common bacteria in the nursery environment are coagulase-negative staphylococci (CoNS), Bacillus spp. and Pseudomonas-like bacteria, all of which rarely cause disease in healthy children.8 Less is known about viruses in the nursery environment in spite of the frequency of viral respiratory infections caused by rhinovirus, bocavirus, adenovirus and respiratory syncytial virus (RSV).9, 10, 11, 12, 13, 14 In a study of the prevalence of 16 different respiratory viruses on surfaces in 23 nurseries during the winter of 2010–2011, using the same technique as in this study, respiratory viruses were found to be widespread in the environment, especially on toys (publication pending). This study found that toys were not washed or disinfected systematically. The Danish Health Board recommends monthly cleaning,15 but it is not known whether regular cleaning of toys in nurseries can affect the pathogen load and reduce infection.
The aim of this study was to determine whether regular systematic cleaning and disinfection of toys would decrease the prevalence of bacteria and respiratory viruses in the nursery environment, and reduce sickness absence in Danish nurseries.
Materials and methods
Twelve nurseries from the municipality of Copenhagen were recruited in Autumn 2012. The number of divisions in each nursery ranged from two to six, and the number of children ranged from 25 to 120. The total number of children was 587, and their ages ranged from six months to three years. Nurseries were questioned about their policies and procedures for hygiene and cleaning of toys before commencement of the study, and randomized to intervention (N = 6) and control (N = 6) groups.
The intervention took place from January to March 2013. A commercial cleaning company (Berendsen A/S, Søborg, Denmark) collected toys and linen every two weeks from nurseries in the intervention group for cleaning in their industrial cleaning facility. Linen and toys suitable for washing machines were washed at 46°C and subsequently disinfected with Turbo Oxysan (Ecolab, Valby, Denmark). Toys that were not suitable for washing machines were either immersed in a disinfectant [Sirafan M, Ecolab (1–3% benzalkonium chloride, 1–3% didecyldimethylammonium chloride and 5–7% alcohol ethoxylates)] or cleaned manually with a microfibre cloth using the same disinfectant (Sirafan M, Ecolab). The toys were subsequently rinsed with water, air dried and returned to the nurseries. Cleaning of toys was staggered to ensure that the children had some toys to play with while others were being cleaned.
Absence data and disease patterns were recorded for each child on a daily basis from December 2012 to March 2013. The number of absent children and the reason for absence (as reported by the parents) was recorded by the staff within the following categories: respiratory infections, gastrointestinal infections, other illnesses, or day off.
Bacterial sampling and processing
Sampling was performed before and after the intervention in December 2012 and April 2013. Ten predefined sampling points (seven in the playroom and three in the toilet area) were sampled in each of the 12 nurseries. The playroom sampling points were undersides of tables, plastic toys, wooden toys, food toys, teddy bears, pillows and sofas. The toilet sampling points were toilet seats, changing mats and basin taps. The sampling area for most sampling points was 10 × 10 cm, but where it was not possible to sample a 10 × 10 cm square (e.g. on smaller toys), the whole object was swabbed. Sampling was performed using: (1) a dry, sterile, cotton-tipped swab, immersed in ox serum broth after sampling (Department of Clinical Microbiology, Herlev Hospital, Herlev, Denmark); and (2) a double-sided dipslide (25 × 80 mm) with tryptic soy agar (TSA) agar on one side and violet red bile glucose agar on the other side, with both sides containing a neutralizer against disinfectants (Model No. DPSLDTV, 3M, Copenhagen, Denmark). The dipslides were incubated for 48 h and the ox serum broth was incubated for seven days at 35–37°C. Following incubation, the ox serum broth was plated on to a blood agar plate (Item No. 677, SSI -Diagnostika, Copenhagen, Denmark) and a lactose agar plate (Item No. 694, SSI) and incubated for 24 h. Total bacteria count was determined using the TSA side of the dipslide and the supplied key from the manufacturer, and reported as colony-forming units (cfu)/cm2. The different species from both dipslides and the ox serum broth were identified using conventional identification and Matrix-Assisted Laser Desorption/Ionization-Time of Flight (MALDI-TOF) (Bruker Daltonics Scandinavia AB, Frederikssund, Denmark). MALDI-TOF was only used for potential pathogens (all faecal bacteria, Staphylococcus aureus and nasopharyngeal bacteria). Bacteria (and fungi) were divided into four groups: skin bacteria (CoNS, Micrococcus spp., Poprionibacterium spp. and S. aureus), water and soil bacteria and fungi (Acinetobacter spp., Pseudomonas-like spp., Aeromonas spp., Comamonas spp., Bacillus spp. and moulds), nasopharyngeal bacteria (Streptococcus pneumoniae, Moraxella spp. and non-haemolytic streptococci) and intestinal bacteria (all Enterobactereaceae and Enterococcus spp.). Escherichia coli and Enterococcus spp. were used as indicators of faecal contamination.
Virus sampling and processing
Respiratory viruses were sampled from the same 10 sampling points as the bacterial samples. Each point of 10 × 10 cm was sampled using a 15 × 25 mm polyester foam swab (Model 149-0264, VWR, Herlev, Denmark). Swabs were immersed in sterile RNase-free water before sampling. After sampling, each swab was placed in a 15-ml sterile plastic container with 5 ml Nuclisens Lysis buffer (bioMérieux, Marcy l'Etoile, France). On arrival at the laboratory, the tubes were placed on a shaking table for 20 min and the lysis buffer was transferred to a 3.6-ml cryotube and stored at −20°C until analysis. Virus DNA and RNA from the sample were extracted using a MiniMag apparatus and Nuclisens extraction reagents (bioMérieux). The purified DNA/RNA, eluted in 100 μl of elution buffer (bioMérieux), was stored at −80°C until quantitative PCR amplification and analysis.
Selected samples were analysed for the presence of 16 respiratory viruses: influenza A; influenza B; coronavirus NL63 229E, OC43 and HKU1; parainfluenzavirus 1, 2, 3 and 4; rhinovirus; RSV A/B; adenovirus; enterovirus; parechovirus; and bocavirus. Quantitative PCR was performed using 10 μl of extracted nucleic acids and the FTD Respiratory Pathogens 21 Plus multiplex PCR kit (Fast Track Diagnostics, Junglinster, Luxembourg) with the recommended enzyme kit (AgPath-ID One-Step RT-PCR Reagents, Life Technologies, Naerum, Denmark). PCR amplification and reading was performed using RotorGene Q (Qiagen Inc., Venlo, The Netherlands) and analysis was performed using Rotorgene Software.
Data management and statistical analysis
Total bacterial growth was read from the TSA side of the dipslide and calculated in cfu/cm2. Results from the dipslides and the ox serum broth were pooled for each sample point, and given as binary results depending on the presence or absence of the target organisms. The same binary method was used for respiratory virus detection. Logistic regression models were used to examine the odds of bacterial and viral presence before and after the intervention. Differences in total growth were analysed using Student's t-test. Differences in sickness absence between groups were analysed using a modified Chi-squared test. All calculations were performed using Statistical Package for the Social Sciences Version 22 (IBM Corp., Armonk, NY, USA). P ≤ 0.05 was considered to indicate significance.
Results
No difference in hygiene standards was found between the control group and the intervention group. No nurseries in either group had a scheduled plan for cleaning toys or washed the toys systematically.
Bacteria
Very few potentially pathogenic bacteria were found in the samples. Out of 240 samples, there were 15 potential pathogens (6%) in the pre-intervention samples (six in the control group and nine in the intervention group) and 11 (5%) potential pathogens in the post-intervention samples (eight in the control group and three in the intervention group). These were bacteria of low pathogenicity such as Enterobacter spp., E. coli and non-haemolytic streptococci. The highest prevalence was found on pillows and sofas, followed by changing mats and various toys.
Mean (± standard error) bacterial growth was 7.0 ± 0.6 cfu/cm2. Highest bacterial growth was found on teddy bears (15.0 ± 2.3 cfu/cm2), followed by sofas (13.0 ± 1.3 cfu/cm2), pillows (13.0 ± 1.9 cfu/cm2), toilet seats (5.4 ± 2.0 cfu/cm2), plastic toys (4.9 ± 2.1 cfu/cm2), wooden toys (4.7 ± 2.0 cfu/cm2), playroom tables (4.5 ± 2.0 cfu/cm2), changing mats (4.2 ± 2.1 cfu/cm2), food toys (2.7 ± 1.7 cfu/cm2) and basin taps (2.6 ± 1.7 cfu/cm2). No difference in total growth or potential pathogen presence was found between the intervention group and the control group.
Viruses
A mean of three different respiratory viruses was found at each sampling point. The prevalence rates for the different respiratory viruses are listed in Table I . The most prevalent virus was coronavirus (97% positive samples), followed by bocavirus (96%), adenovirus (73%) and rhinovirus (46%). The intervention reduced the presence of adenovirus, rhinovirus and RSV approximately two- to five-fold [odds ratio (OR) 2.4, 95% confidence interval (CI) 1.1–5.0 for adenovirus; OR 5.3, 95% CI 2.3–12.4 for rhinovirus; OR 4.1, 95% CI 1.5–11.2 for RSV] compared with the control group. On the other hand, metapneumovirus was found significantly less often in the control group than in the intervention group. The intervention had no effect on the detection of other viruses. The fomites with the highest presence of respiratory virus were pillows and sofas, followed by toys and playroom tables.
Table I.
Presence of respiratory viruses in the nursery environment before and after the intervention
| Virus | Intervention group |
Control group |
OR (95% CI) | p | ||
|---|---|---|---|---|---|---|
| Pre | Post | Pre | Post | |||
| Bocavirus | 40 (95%) | 39 (93%) | 41 (98%) | 41 (98%) | 1.0 (0.1–9.9) | 0.28 |
| Coronavirus | 39 (93%) | 40 (95%) | 42 (100%) | 42 (100%) | 1.0 (0.1–9.9) | 0.99 |
| Adenovirus | 37 (88%) | 25 (60%) | 26 (62%) | 35 (83%) | 2.4 (1.1–5.0) | 0.02a |
| Rhinovirus | 20 (48%) | 8 (19%) | 20 (48%) | 30 (71%) | 5.3 (2.3–12.4) | <0.01a |
| Parainfluenzavirus | 17 (40%) | 28 (67%) | 16 (38%) | 24 (57%) | 1.3 (0.6–3.0) | 0.50 |
| Respiratory syncytial virus A/B | 14 (33%) | 5 (12%) | 16 (38%) | 15 (36%) | 4.1 (1.5–11.2) | <0.01a |
| Metapneumovirus | 4 (10%) | 11 (26%) | 3 (7%) | 5 (12%) | 0.3 (0.1–0.7) | <0.01a |
| Enterovirus | 2 (5%) | 0 | 0 | 0 | N/A | N/A |
| Influenza A | 1 (2%) | 1 (2%) | 0 | 1 (2%) | N/A | N/A |
| Parechovirus | 1 (2%) | 0 | 2 (5%) | 0 | N/A | N/A |
| Influenza B | 0 | 1 (2%) | 0 | 3 (7%) | N/A | N/A |
OR, odds ratio; CI, confidence interval.
Numbers are positive samples (% of total), N = 42 in each group.
Significant difference (P < 0.05) between groups.
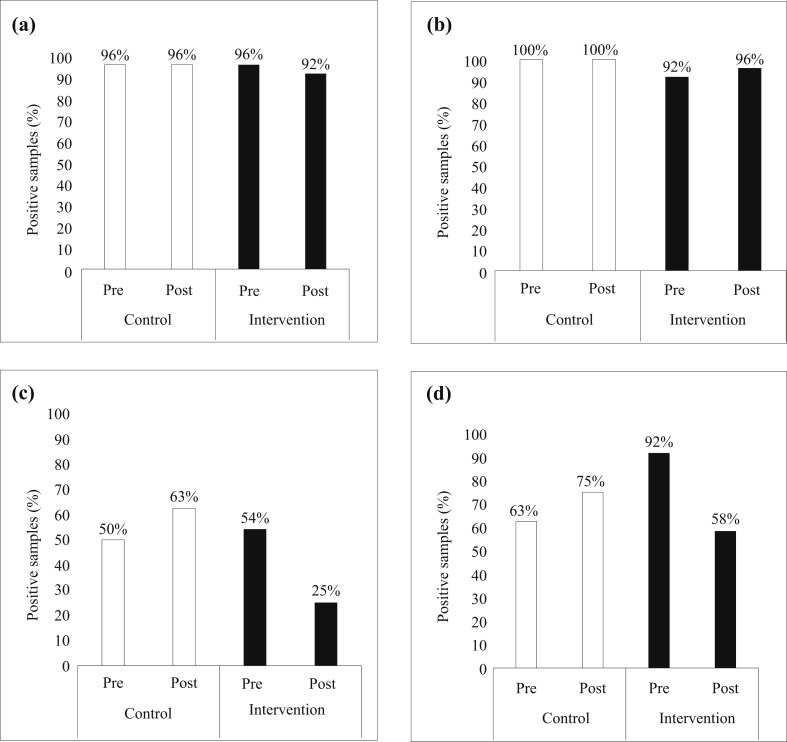
When looking at the samples from the toys alone, there was a significant decrease following the intervention in the intervention group compared with the control group for rhinovirus (OR 3.8, 95% CI 1.3–10.5; P = 0.01) and RSV (OR 5.2, 95% CI 1.1–23.8; P = 0.04), but not adenovirus. As in all samples, the presence of metapneumovirus was significantly decreased in the control group compared with the intervention group. No differences were found for the other viruses on the toys. The prevalence rates of the four most prevalent viruses on the toys are shown in Figure 1 .
Figure 1.
Presence of viruses on toys in the nursery environment. (a) Bocavirus, (b) coronavirus, (c) rhinovirus, (d) adenovirus.
Sickness absence and disease patterns
The proportion of healthy children in the control group decreased from 84% to 76% in the post-intervention period, whereas it was unchanged in the intervention group; however, the difference between the groups was not significant. Mean absence (± standard error) due to respiratory infections decreased after the intervention in both the intervention and control groups (from 6.8% ± 1.0% to 3.4% ± 0.3% in the intervention group and from 5.2% ± 0.5% to 4.1% ± 0.3% in the control group). However, gastrointestinal infections increased after the intervention (from 0.6% ± 0.2% to 2.3% ± 0.2% in the intervention group and from 0.7% ± 0.1% to 1.5% ± 0.1% in the control group). Other illnesses also increased in both groups after the intervention (from 2.8% ± 0.3% to 3.9% ± 0.4% in the intervention group and from 4.3% ± 0.4% to 4.9% ± 0.3% in the control group). No significant differences in any of the disease categories were found between the groups.
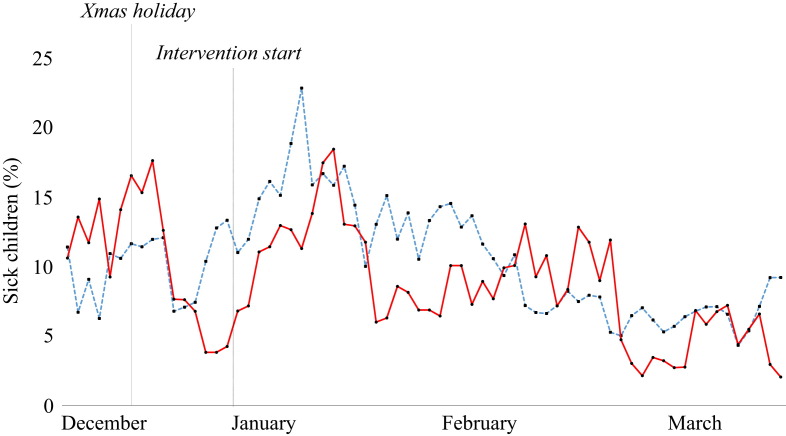
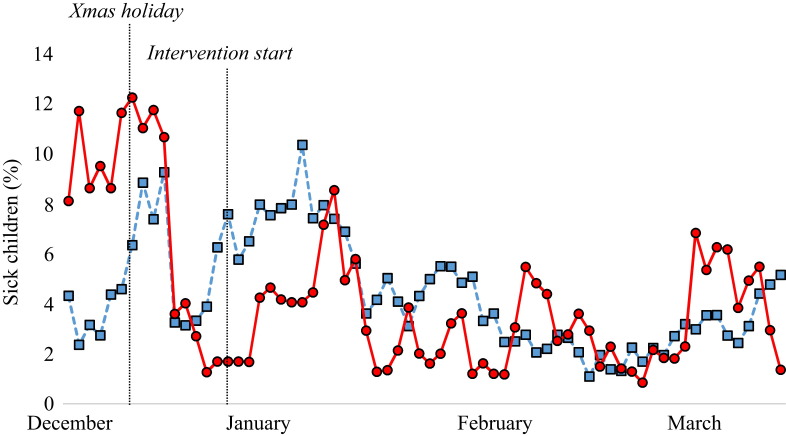
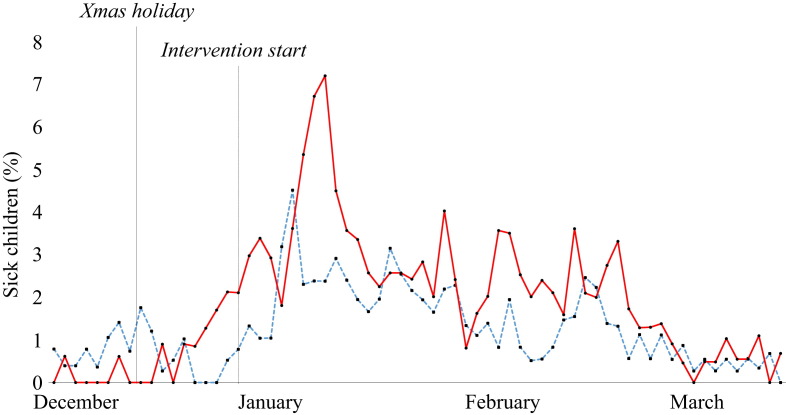
When looking at the timeline of infectious diseases in the two groups before and after the intervention, there is no striking effect of the intervention on total sickness absence, respiratory infections or gastrointestinal infections (Figure 2, Figure 3, Figure 4 ). There was a significant difference (P = 0.017) in total sickness absence between the groups, but the control group had the lowest sickness absence (10.2% vs 11.1% in the intervention group). No difference was found between the groups when analysing respiratory or gastrointestinal infections in the pre- and post-intervention periods.
Figure 2.
Sick days in the nurseries before and after the intervention. Blue line, control group; red line, intervention group.
Figure 3.
Respiratory infections in the nurseries before and after the intervention. Blue line, control group; red line, intervention group.
Figure 4.
Gastrointestinal infections in the nurseries before and after the intervention. Blue line, control group; red line, intervention group.
Discussion
The bacteria found in this study were mainly non-pathogenic; only 6% of the bacteria were potential pathogens. This is in accordance with a study by Lee et al., who investigated the bacterial diversity in a nursery through a combination of cultures and 16S rRNA sequencing.8 They found that the most prevalent bacteria cultured were Bacillus spp., Staphylococcus spp. and Pseudomonas spp., while 16S sequencing analysis was dominated by Pseudomonas spp. and Oxalobacteria spp. The present study found a very low prevalence of potential pathogens (6%), and this was lower than reported in many other studies; for example, Laborde et al. reported faecal coliform rates of 20–50% on toys, sinks and tables in toddlers' classrooms;16 and Ekanem et al. reported a faecal coliform rate of 13% on classroom objects.17 The discrepancies may be due to different sampling techniques, different sampling points and different sampling areas. It may be more efficient to sample larger areas using cloths in order to maximize the chances of finding pathogenic bacteria present in the environment.
Respiratory virus RNA/DNA was widespread in the environment, especially for viruses causing the common cold, such as rhinovirus, adenovirus, coronavirus and bocavirus. Other studies have found similar pathogens in the throats of children with respiratory infections.9, 11, 18 These studies found rhinovirus, RSV, coronavirus and adenovirus to be the most prevalent viruses. This corresponds well with the present results, where bocavirus was the most prevalent virus, followed by coronavirus, adenovirus and rhinovirus. RSV detection was lower, but this may have been because the severity of RSV infection keeps children away from nurseries. The similarity between the present findings in the environment and findings in children's throats indicates that viruses are transmitted to the environment. The weakness of the PCR technique is that viral nucleic acid is detected rather than ‘live’ viruses, and the prevalence of infectious viruses may be overestimated. To assess this, the viruses would have to be grown in cell cultures. This was not possible due to time and economic constraints. Moreover, some viruses, such as norovirus, cannot be cultured.
The main endpoint in this study was the number of days of absence due to sickness. This study did not find a decrease in total sickness absence or in sickness absence due to respiratory infection. To the authors' knowledge, this is the first study to measure the isolated effect of cleaning toys on infectious diseases in nurseries. However, a few studies have ‘touched upon’ the subject. Krilov et al. showed a significant decrease in the number of infections in a specialized preschool following the implementation of an infection control programme.19 This programme included cleaning toys, but this was combined with other measures such as hand hygiene and environmental cleaning. Numerous studies have shown the presence of coliforms and different pathogenic bacteria and viruses on toys in hospitals and other clinical settings.20, 21, 22, 23 Consequently, the general recommendations to date have been that toys should be cleaned regularly in order to prevent infections. The data from this study, however, do not support these recommendations. Possible reasons for failure to observe an effect of cleaning toys include: too great an interval between washes; not washing all toys at the same time; and the effect of person-to-person transmission overshadowing the effect of cleaning. This raises the question of whether commercial cleaning of toys is cost-effective. It is likely that the most effective ways to prevent infections in daycare nurseries are rigorous hand hygiene and other well-established infection control measures, or simply to keep the children at home for longer when they are ill.24, 25, 26, 27, 28 The latter is confirmed by the absence timelines in this study (Figure 2, Figure 3, Figure 4), which included the period before and after the Christmas holiday in December. During this holiday, the nurseries were closed for two weeks. There was a marked decrease in sickness absence after the holiday in both groups, with the percentage of children with respiratory infection decreasing from 1.6% to 0.5% in the intervention group and from 1.1% to 0.4% in the control group. If feasible, absence from nurseries might be the best way to reduce the spread of infection in children.
In conclusion, this study showed that respiratory virus DNA and RNA are widespread in the nursery environment. Fortnightly cleaning and disinfection of toys reduced the frequency of detection of some respiratory viruses, but not the bacterial load, and did not reduce the number of days of absence due to respiratory infection or sickness as a whole. As this is the first study of its kind, further studies are needed to confirm or refute the findings. Studies over a longer period of time may be necessary to control for seasonal fluctuation in infection rate and virus types.
Acknowledgements
The authors wish to thank Charlotte Foged (Rigshospitalet) and Resadije Idrizi (DTU Food) for excellent technical assistance, and Ecolab Denmark, Berendsen Denmark and 3M Denmark for the free supply of materials and cleaning.
Conflict of interest statement
Ecolab Denmark, Berendsen Denmark and 3M Denmark supplied materials and cleaning free of charge, but had no influence on the analysis of the data or the writing of the manuscript.
Funding source
This work was supported by the Danish Council for Technology and Innovation under the Ministry of Science, Innovation and Higher Education as part of the Sundhed i Børneinstitutioner innovation consortium.
References
- 1.Denny F.W., Collier A.M., Henderson F.W. Acute respiratory infections in day care. Rev Infect Dis. 1986;8:527–532. doi: 10.1093/clinids/8.4.524. [DOI] [PubMed] [Google Scholar]
- 2.Bartlett A.V., Moore M., Gary G.W., Starko K.M., Erben J.J., Meredith B.A. Diarrheal illness among infants and toddlers in day care centers. II. Comparison with day care homes and households. J Pediatr. 1985;107:503–509. doi: 10.1016/s0022-3476(85)80005-6. [DOI] [PubMed] [Google Scholar]
- 3.Uldall P. Forms of care and children's infections. 1. Occurrence and causal factors. Ugeskr Laeger. 1990;152:2345–2348. [PubMed] [Google Scholar]
- 4.Fleming D.W., Cochi S.L., Hightower A.W., Broome C.V. Childhood upper respiratory tract infections: to what degree is incidence affected by day-care attendance? Pediatrics. 1987;79:55–60. [PubMed] [Google Scholar]
- 5.Cosby C.M., Costello C.A., Morris W.C. Microbiological analysis of food contact surfaces in child care centers. Appl Environ Microbiol. 2008;74:6918–6922. doi: 10.1128/AEM.00547-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Laborde D.J., Weigle K.A., Weber D.J., Kotch J.B. Effect of fecal contamination on diarrheal illness rates in day-care centers. Am J Epidemiol. 1993;138:243–255. doi: 10.1093/oxfordjournals.aje.a116853. [DOI] [PubMed] [Google Scholar]
- 7.Staskel D.M., Briley M.E., Field L.H., Barth S.S. Microbial evaluation of foodservice surfaces in Texas child-care centers. J Am Diet Assoc. 2007;107:854–859. doi: 10.1016/j.jada.2007.02.013. [DOI] [PubMed] [Google Scholar]
- 8.Lee L., Tin S., Kelley S.T. Culture-independent analysis of bacterial diversity in a child-care facility. BMC Microbiol. 2007;7:27. doi: 10.1186/1471-2180-7-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Martin E.T., Fairchok M.P., Stednick Z.J., Kuypers J., Englund J.A. Epidemiology of multiple respiratory viruses in childcare attendees. J Infect Dis. 2013;207:982–989. doi: 10.1093/infdis/jis934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pitkaranta A., Roivainen M., Blomgren K. Presence of viral and bacterial pathogens in the nasopharynx of otitis-prone children. A prospective study. Int J Pediatr Otorhinolaryngol. 2006;70:647–654. doi: 10.1016/j.ijporl.2005.08.018. [DOI] [PubMed] [Google Scholar]
- 11.Fairchok M.P., Martin E.T., Chambers S. Epidemiology of viral respiratory tract infections in a prospective cohort of infants and toddlers attending daycare. J Clin Virol. 2010;49:16–20. doi: 10.1016/j.jcv.2010.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Boone S.A., Gerba C.P. The occurrence of influenza A virus on household and day care center fomites. J Infect. 2005;51:103–109. doi: 10.1016/j.jinf.2004.09.011. [DOI] [PubMed] [Google Scholar]
- 13.Butz A.M., Fosarelli P., Dick J., Cusack T., Yolken R. Prevalence of rotavirus on high-risk fomites in day-care facilities. Pediatrics. 1993;92:202–205. [PubMed] [Google Scholar]
- 14.Keswick B.H., Pickering L.K., DuPont H.L., Woodward W.E. Survival and detection of rotaviruses on environmental surfaces in day care centers. Appl Environ Microbiol. 1983;46:813–816. doi: 10.1128/aem.46.4.813-816.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Danish Health Board . Danish Health Board; Copenhagen: 2009. Hygiejne i Daginstitutioner. [Google Scholar]
- 16.Laborde D.J., Weigle K.A., Weber D.J., Sobsey M.D., Kotch J.B. The frequency, level, and distribution of fecal contamination in day-care center classrooms. Pediatrics. 1994;94:1008–1011. [PubMed] [Google Scholar]
- 17.Ekanem E.E., DuPont H.L., Pickering L.K., Selwyn B.J., Hawkins C.M. Transmission dynamics of enteric bacteria in day-care centers. Am J Epidemiol. 1983;118:562–572. doi: 10.1093/oxfordjournals.aje.a113661. [DOI] [PubMed] [Google Scholar]
- 18.Bonfim C.M., Nogueira M.L., Simas P.V. Frequent respiratory pathogens of respiratory tract infections in children attending daycare centers. J Pediatr (Rio J) 2011;87:439–444. doi: 10.2223/JPED.2126. [DOI] [PubMed] [Google Scholar]
- 19.Krilov L.R., Barone S.R., Mandel F.S., Cusack T.M., Gaber D.J., Rubino J.R. Impact of an infection control program in a specialized preschool. Am J Infect Control. 1996;24:167–173. doi: 10.1016/s0196-6553(96)90008-5. [DOI] [PubMed] [Google Scholar]
- 20.Akhter J., Al-Hajjar S., Myint S., Qadri S.M. Viral contamination of environmental surfaces on a general paediatric ward and playroom in a major referral centre in Riyadh. Eur J Epidemiol. 1995;11:587–590. doi: 10.1007/BF01719313. [DOI] [PubMed] [Google Scholar]
- 21.Avila-Aguero M.L., German G., Paris M.M., Herrera J.F. Toys in a pediatric hospital: are they a bacterial source? Am J Infect Control. 2004;32:287–290. doi: 10.1016/j.ajic.2003.10.018. [DOI] [PubMed] [Google Scholar]
- 22.Davies M.W., Mehr S., Garland S.T., Morley C.J. Bacterial colonization of toys in neonatal intensive care cots. Pediatrics. 2000;106:E18. doi: 10.1542/peds.106.2.e18. [DOI] [PubMed] [Google Scholar]
- 23.Fleming K., Randle J. Toys – friend or foe? A study of infection risk in a paediatric intensive care unit. Paediatr Nurs. 2006;18:14–18. [PubMed] [Google Scholar]
- 24.Koefoed B.G., Nielsen A.M., Keiding L.M. The impact of selected environmental factors on the morbidity of children in day care centers. UgeskrLaeger. 2002;164:5759–5764. [PubMed] [Google Scholar]
- 25.Lee M.B., Greig J.D. A review of enteric outbreaks in child care centers: effective infection control recommendations. J Environ Health. 2008;71:24–32. 46. [PubMed] [Google Scholar]
- 26.Sandora T.J., Taveras E.M., Shih M.C. A randomized, controlled trial of a multifaceted intervention including alcohol-based hand sanitizer and hand-hygiene education to reduce illness transmission in the home. Pediatrics. 2005;116:587–594. doi: 10.1542/peds.2005-0199. [DOI] [PubMed] [Google Scholar]
- 27.Uhari M., Mottonen M. An open randomized controlled trial of infection prevention in child day-care centers. Pediatr Infect Dis J. 1999;18:672–677. doi: 10.1097/00006454-199908000-00004. [DOI] [PubMed] [Google Scholar]
- 28.Nandrup-Bus I. Mandatory handwashing in elementary schools reduces absenteeism due to infectious illness among pupils: a pilot intervention study. Am J Infect Control. 2009;37:820–826. doi: 10.1016/j.ajic.2009.06.012. [DOI] [PubMed] [Google Scholar]