Summary
For almost three decades following the discovery of the human Hepatitis B Virus (HBV) the early events of virus infection (attachment to hepatocytes, specific binding to a receptor on hepatocytes) remained enigmatic. The gradual improvement of tissue culture systems for HBV has enabled the identification of viral determinants for viral infectivity and facilitated the discovery of the human sodium taurocholate co-transporting polypeptide (hNTCP) as a liver specific receptor of HBV and its satellite, the human Hepatitis Delta Virus (HDV). These findings are currently leading basic and clinical research activities in new directions. (1) Stable hNTCP-expressing cell lines have become a valuable platform to study the full HBV replication cycle from its native template, the cccDNA. (2) The suitability of NTCP complemented cell culture systems for high throughput screening approaches will facilitate identification of novel host factors involved in HBV replication (including those that determine the peculiar host specificity of HBV infection) and will enable identification and development of novel drug candidates for improved therapeutics. (3) Since NTCP is a major host-specific restriction factor for HBV and HDV, hNTCP-expressing animals provide the basis for future susceptible in vivo models. (4) The concept obtained with the entry inhibitor Myrcludex B demonstrates that NTCP is a suitable target for clinical interference with viral entry. This will foster further clinical approaches aiming at curative combination therapies.
Keywords: Hepatitis B virus entry, Hepatitis B virus receptor, NTCP, SLC10A1, Myrcludex B
NTCP acts as a functional receptor of HBV and HDV
Key point.
The human sodium taurocholate co-transporting polypeptide (hNTCP) is a bona fide receptor for HBV and HDV.
The nature of the cellular receptor mediating HBV infection remained elusive long time after the discovery the virus [1], [2], [3]. Although some candidate molecules were found to bind to the envelope proteins of HBV, none of them ever demonstrated a virus receptor activity upon expression in non-susceptible cells, which is accepted as a strong criteria regarding whether a candidate is truly a functional virus receptor. Studies of the infection biology of HBV and its satellite virus hepatitis D virus (HDV), an RNA virus using HBV envelope proteins for its packaging and cellular entry, was hampered by the lack of a convenient cell culture system. Primary cultures of human hepatocytes (PHH) [4], [5], primary tupaia hepatocytes (PTH) [6], [7], [8], and a hepatoma cell line, HepaRG, after differentiation with DMSO [9], were the only cells susceptible to HBV and HDV. PHH and PTH are scarce and their susceptibility to the virus varies depending on the source as well as isolation and culture procedures. HepaRG cells require a 2-week differentiation period before cells become susceptible. Nonetheless, elegant work using these cell culture systems over the past decades has revealed some critical aspects of HBV entry [10]: (1) Infection of HBV exhibits extraordinary species and organ specificity, indicating existence of a highly specific interaction between the virus and the hepatocytes; (2) two of HBV three envelope proteins, namely the small (S) and large (L) envelope protein, but not the middle (M) protein are responsible for HBV entry [11], [12]; (3) the antigenic loop (AGL) of S protein [13] and the preS1 domain of L-protein are essential for HBV infection [14]; (4) the N-terminal region of preS1 domain requiring an uninterrupted sequence of 75 amino acids (genotype D) [15] with a myristoylation modification at the N-terminus [16], [17] critically contributes to infectivity, among which amino acid residues 9–16 are of vital importance [18], [19], [20]. The corresponding lipopeptide of this domain blocks viral infection in vitro and in vivo [19], [20], [21], [22].
The preS1 lipopeptide as a natural ligand is useful to identify the specific receptor on hepatocytes. However, it was not an easy task to find its binding partner on hepatocytes and several non-functional molecules have been described [10]. Successful discovery of the bona fide receptor sodium taurocholate co-transporting polypeptide (NTCP) was achieved using an innovative preS1 peptide ligand combined with proteome analysis of PTH and highly efficient tracing and purification processes [23]. Biochemical studies confirmed the interaction between preS1 and the liver specific bile acids transporter. Remarkably, transfection of HepG2 cells with NTCP expression DNA vector confers susceptibility to this otherwise non-susceptible cell [23]. This finding was quickly confirmed by several important studies, including a study demonstrating that human NTCP can confer susceptibility to HDV pseudotyped with the envelope proteins of bat hepadnavirus [24], woolly monkey HBV (WMHBV) also exploits NTCP for viral entry in tupaia hepatocytes [25] and HBV/HDV exploit NTCP for species-specific entry into hepatocytes [26]. Discovery of the receptor has also engendered new perspectives on HBV and its associated diseases [27].
Key point.
hNTCP expressing hepatoma cell lines are important tools to study HBV and HDV replication.
NTCP is coded by the SLC10A1 gene. It is a bile acid transporter, responsible for hepatic uptake of the majority of conjugated bile acids from blood, and plays a critical role in bile acids homeostasis and enterohepatic circulation [28]. The expression of NTCP and its subcellular distribution is subjected to precise regulation at multiple levels. NTCP contains up to nine transmembrane segments embedded in the cell membrane; its structure remains to be elucidated. Interestingly, although the NTCP gene is conserved in mammals, disrupting NTCP in mice in short term (ca. 2 months) did not result in significant abnormalities in the animals but led to elevated serum bile acids, in particular conjugated ones [29]. A single point mutation of R252H of human NTCP interferes with its surface expression on hepatocytes. Recently, a five-year old girl bearing this mutation was identified and she exhibited mild hypotonia, growth retardation, delayed motor milestones but no severe health problem [30]. Therefore, although the long-term consequence of disrupting NTCP needs more studies, the physiological role of NTCP can probably be compensated by other, yet not defined mechanism(s), and redundant pathways may operate in the absence of NTCP. Whether there is another receptor(s) for HBV entry is not known at present, however several lines of evidence support that NTCP is well suited as a key receptor for HBV (Fig. 1 ). It is predominantly expressed in hepatocytes [31], as also shown by the hepatotropism of its preS-ligand in vivo [32]. This is consistent with the high liver specificity of HBV. Furthermore NTCP resides on the sinusoidal side of hepatocytes [33], [34], [35], which is in line with the blood transmission of the virus. There is only very low or no expression of NTCP in hepatocarcinoma cell lines like HepG2 or HuH7 and the expression of NTCP rapidly decreases after isolation of primary hepatocytes from animals [23], [36], [37], [38]. These two features at least partially explain why normal liver cancer cells are resistant to HBV infection and why the susceptibility of primary hepatocytes persists for only a few days after isolation.
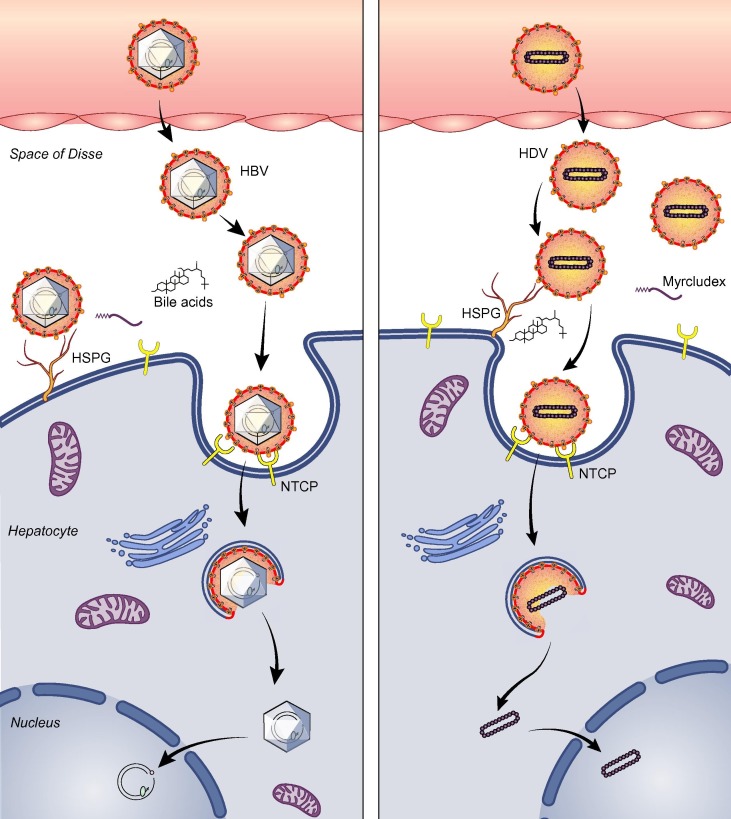
Fig. 1.
Diagram of viral entry of HBV/HDV. HBV (left) or HDV (right) travels with blood to liver, penetrates into the space of Disse and reaches the sinusoidal side of hepatocytes. The virus is enriched on the cell surface by interaction with HSPG. The viral envelope may undergo conformational changes upon binding to HSPG. Subsequently, the preS1 region of the envelope binds to the extracellular loop(s) of NTCP with high specificity. Binding of virions to NTCP may lead to endocytosis of the receptor, and ultimately the nucleocapsid (HBV) or the ribonucleoprotein complex (HDV) is realised to the nucleus and the infection is established. NTCP, sodium taurocholate cotransporting polypeptide; Myrcludex B, a lipopeptide derived from the 2–47 residues of preS1 region of HBV; HSPG, heparan sulfate proteoglycan.
Interestingly, a human population study on 1899 HBV patients from southern China (Guangdong) showed that a single mutation of NTCP, S267F ((c.800G>A, rs2296651) on SLC10A1), is associated with resistance to chronic hepatitis B [39]. The S267F is a mutation found mainly in Asian populations, with a minor frequency of ca. 10 % [40], [41], [42]. In cell cultures the mutation abolished taurocholate (TC) transport and over expression of S267F NTCP in HepG2 cells failed to support genotype D HBV and HDV infection [41]. The marked protection of this mutation against HBV infection at population level strongly argues that NTCP is a major viral receptor in human. A more recent genetic association study based on 3801 chronic hepatitis B (CHB) patients from Taiwan confirmed that the same mutation confers resistance to HBV infection and is independently associated with decreased risk of cirrhosis and HCC [42]. It is currently unclear how the mutation would reduce the risk of cirrhosis and HCC.
Interestingly, there are five HBV patients with homozygous S267F NTCP mutation in the Guangdong cohort [39] the Taiwan study also found five of such HBV patients [42]. How these patients were infected is an interesting and important question. There are several possibilities: first, cell surface molecules other than NTCP could mediate the viral infection; second, the virus was adapted to S267F-NTCP; third, the patients were genetically mosaic meaning some of their hepatocytes might still have wildtype NTCP. Clearly more work is needed to clarify these possibilities and related studies would help to deepen our understandings of HBV infection.
NTCP determines viral infection specificity at entry level
HBV only infects human, chimpanzees and northern treeshrew (Tupaia belangeri). It cannot infect non-human primates, mouse, rat and other experimental animals. However, once the HBV genome is delivered to the monkey hepatocytes, the virus can robustly replicate indicating that these cells are able to form cccDNA and replicate the virus [43]. Mouse hepatocytes bearing an HBV transgene can produce infectious virions but are unable to form cccDNA [44]. These observations support the hypothesis that the species restriction of HBV is largely at the entry level. Entry restriction is common in cross-species transmission of many viruses, for example: the infection of influenza virus among mammals and birds heavily depends on the existence of sialic acids (SA)-α-2,6-Gal–terminated saccharides or (SA)-α-2,3-Gal–terminated saccharides receptor [45]; and animal transmission of coronaviruses, including SARS-CoV and MERS-CoV is controlled by their receptors [46]. Whether NTCP is responsible for the species specificity at entry level was first examined by assessing HBV infection mediated by NTCP chimeras from crab-eating monkey (Macaca fascicularis) and human [23]. A small motif covering residues 157–165 of NTCP was identified to be critical for preS1-binding and HBV infection, highlighting the critical role of NTCP in mediating HBV infection [23], [26]. The observation that cynomolgus monkey hepatocytes cannot bind the preS1-lipopeptide in vitro and in vivo strongly supported this finding [32], [34]. The key role of residues 157–165 for HBV infection in humans has also been supported by the recent CHB population study in Taiwan, in which no mutation was found in this region in all CHB patients [42]. Interestingly, naturally occurring transmissible HBV infection (genotype D) was reported among M. fascicularis from Mauritius Island, however, the viral titre was very low and more evidence is needed to support that HBV undergoes a full life cycle in these monkeys [47].
The receptor-addressing preS1-lipopeptide can bind to primary hepatocytes from HBV non-susceptible animals, such as mouse, rat and dog [32], [34]. This finding was surprising and indicated the resistance of these animals to HBV at entry level was less likely due to the lack of a specific binding partner of the virus on the hepatocytes. Accordingly, either the binding could not lead to a productive infection, or there is difference between the isolated preS1 peptide versus the same region residing on the virions. Extensive mapping studies revealed that amino acid residues 84–87 in the first extracellular loop of NTCP restricted mice from infection of HBV at the entry level. Replacing three of these four amino acids with their human counterpart effectively render mouse NTCP a functional receptor for HBV in HepG2 cells [26], [48]. Consistent with the notion that NTCP poses the major barrier for viral infection of mice, suckling C57BL/6 mice bearing human NTCP are susceptible for HDV infection and the virus successfully undergoes most of its life cycles in the animal [49]. However, although mouse liver cells like Hepa1-6, Hep56.1D, mH274#26 supplemented with human NTCP support HDV infection, they failed to support HBV infection. These studies indicate that factors other than NTCP may be also involved in HBV infection. Although the nature of the factor(s) is still unknown, HBV infection studies based on cell fusion assays of HepG2 and mouse cells indicated that infection in mouse cells is limited by the lack of a host cell dependency factor [50]. Interestingly, a study recently indicated that the stability of viral nucleocapsids may be a determinant for cccDNA formation in murine hepatocytes (AML12) [51a]. Interestingly, this cell line, when supplemented with hNTCP supports infection with HBV [51b]. It will be important to further identify cellular factors to successfully establish a small animal model for HBV infection. On the other hand, one also has to be aware that viral infection in tissue culture can be influenced by the variables among different cell clones, the dosage of the inoculating virus and others factors that are different from the physiological conditions, therefore only viral infection of animals with defined gene alternations can provide definitive answer to the question.
NTCP complemented HepG2 cells provide a valuable platform for screening new host factors and developing antivirals against HBV/HDV infection
Key point.
hNTCP-expressing HepG2 cell lines allow high throughput approaches for drug identification.
HepG2 cells complemented with human NTCP provide an easy accessible system for HBV/HDV infection. The system is amendable for high throughput screening procedure, thus enabling studies of systemic identification of new factors involved in HBV/HDV infection and developing new antivirals. Targeted screening with HepG2-NTCP cells has identified glypican 5 (GPC5) as an entry factor for HBV/HDV [52]; viral infection based assay has found some promising drug candidates in blocking HBV/HDV infection [53], [54]. HBV can undergo its whole life cycle in HepG2-NTCP cells, from entry to establishment of the infection, and at a low efficiency of releasing of progeny virions. The covalent closed circular DNA (cccDNA) of HBV is the reservoir of chronic infection and is widely believed to be the major obstacle for eliminating the infection [55], [56]. HepG2-NTCP cells support cccDNA formation from incoming virions and are less efficient for the replenishment of cccDNA via intracellular recycling. Interestingly, although the carcinomatous nature of HepG2 cell is a concern for studies using HepG2-NTCP cells, a recent study indicates the epigenetic regulations of cccDNA in the cells are similar to those observed in PHH [57].
Entry inhibition of viral infections as a clinical concept
Key point.
hNTCP is a suitable target for drugs that interfere with HBV and HDV entry (e.g. Myrcludex B).
Therapeutic interference by entry inhibitors is an attractive clinical concept since it inhibits infection at its earliest step thereby preventing the establishment of viral genomes (e.g. cccDNA) in naïve or regenerating host cells [58]. Entry inhibition in persistently infected patients is presumably most efficient when virus spread requires entry of progeny virus into naïve cells via a receptor-mediated pathway. In addition, a fast replenishment of infected cells by naïve cells would limit the time of treatment to clear the infection. For HIV it has been shown that a direct spread from one cell-to-another can occur which might explain the therapeutic limitation of entry inhibitors [59], [60]. Nevertheless, Fuzeon® and maraviroc, two approved entry inhibitors, show antiviral activity in patients [60], [61], [62]. For HBV it has been demonstrated that spread in PHH-transplanted uPA severe combined immunodefficient (SCID) mice was completely blocked by the NTCP-specific entry inhibitor Myrcludex B. This indicates that intrahepatic spread greatly depends on an NTCP-mediated entry of progeny virus into naïve hepatocytes [63]. Moreover, cccDNA-dissemination in the liver of these animals is abrogated by cell proliferation [64], indicating that HBV depends on an extracellular virus cycle and works poorly by transferring the cccDNA template via an intercellular pathway into progeny cells. Thus, entry inhibition for HBV may become clinically important in order to block cccDNA reformation during either natural or immune-mediated/induced hepatocyte turnover.
Entry inhibition can be achieved by targeting viral structures (e.g. the neutralisation of envelope proteins at the viral surface, which are involved in receptor binding). In the course of the resolution of an acute infection, neutralising antibodies fulfil this crucial role and are therefore indispensable to clear the virus. Accordingly, antibodies recognizing envelope proteins have been developed for therapeutic purposes [65], [66], [67]. Currently, neutralising antibodies are preferentially applied in prophylactic clinical settings. For HBV and HDV such antibodies prevent reinfection of liver transplants in infected patients, are effective in post exposure prophylaxis or the prevention of mother-child transmission [68]. Long-term administrations of HBV neutralising antibodies in chronically infected patients have not been performed so far.
An alternative strategy to inhibit viral entry takes advantage of drugs that target cellular receptors/co-receptors. This approach is relatively new but has been shown to work in HIV infected patients [60]. Their mode of action is based on the sustained inhibition of de novo formation of viral templates. Although they are not curative in HIV infected patients, they theoretically bear this potential even without the involvement of adaptive immune responses. This has been recently demonstrated in an immune-deficient animal model for hepatitis C virus (HCV) infection [69]. However, the duration of treatment to gain virus elimination depends on the natural turnover rate of infected cells, the half-life time of the virus and the presence of possible viral reservoirs, which have been established in the host. Importantly, immune responses that are directed against infected cells may dramatically enhance the clearance of infected cells. Accordingly, entry inhibition may be highly synergistic, when combined with immune therapeutic approaches.
Whether a virus receptor is a suitable “drugable target” depends on different aspects. Firstly, virus spread within an organism should be subject to receptor-mediated de novo infection and should not primarily proceed by cell-to-cell transmission of viral genomes. Secondly, since virus receptors often serve important cellular functions, the drugs should not completely mess up the natural function of these molecules (clinical manageability). This can be achieved if the targeting drug blocks the receptor function at an IC50 below the concentration required for inactivation of its cellular function. Such a “therapeutic window” would allow drug dosing, which is clinically acceptable even if the target molecule displays an essential function. Alternatively, the receptor may not be an essential protein and its function might be compensated by salvage pathways. Finally, the interaction of the drug with the receptor should be highly specific in order to avoid “off-target” effects.
Inhibitors of HBV and HDV infection addressing NTCP
HBV specifically targets hepatocytes in the human liver with very high efficacy [70]. This hepatotropism is related to specific interactions of the viral envelope proteins with host factors, including heparan sulfate proteoglycans (HSPG) [71], [72], [73] and NTCP (see above). It has been shown that known substrates of NTCP as well as HSPGs interfere with HBV and HDV infection. These substances can be subdivided in different groups: firstly, natural or artificial substrates of HSPG like heparin, highly sulphated dextrans, suramin or negatively charged polymers [71], [72], [73]. They shall not be further discussed here. Secondly, substrates of NTCP, like the conjugated bile acid TC or glycocholic acid [26], [48], [74], [75]. These substrates but also bile salt homologues are transported by NTCP in a sodium dependent manner [28]. They bind into a pocket of the outward conformation of NTCP and are released (together with two sodium ions) after a substantial structural change to the inward conformation into the cytoplasm of the hepatocyte [53]. At high concentrations, conjugated bile salts inhibit HBV and HDV infection [41], [76]. The concentrations needed for infection inhibition are above the normal bile salt levels in healthy and HBV infected individuals, which probably explains the lack of therapeutic effects of bile salts in HBV/HDV infected patients [76].
In addition to endogenous substrates, xenobiotics and certain drugs have been shown to use the transporter function of NTCP to enter hepatocytes [77]. Some of these molecules have been developed as drugs and have been discovered as NTCP addressing agents in the course of their pharmacokinetic characteristics. One example is ezetimibe, which is used as an inhibitor of the primary cholesterol transporter NPC1L1 in the intestinal lumen. It is also a substrate of NTCP [28] and has been later shown to interfere with HBV and HDV entry [78]. These substances have in common that the mechanism of HBV receptor inhibition is coupled to their ability to bind the substrate in the bile salt pocket and thereby block the receptor to interact with the virus. Evidence for that comes from the finding that determinants for viral entry and bile salt transport functionally overlap [41]. Accordingly, the reported KDs of transporter activity and HBV inhibition activity are comparable. In the future, it will be interesting to see whether small molecules that bind NTCP in its substrate pocket (e.g. bile salt analogues that might covalently bind to the transporter), could act as irreversible dead-end inhibitors.
The third class of NTCP inhibitors are peptidic substrates, which bind, but are presumably not transported by NTCP. A prototypic member of this family is cyclosporin A, which was known as an NTCP-inhibitor before it was described as an HBV receptor [79]. Its antiviral activity is not restricted to the natural product cyclosporine A, but also holds up for other derivatives including non-immunosupressive variants like alisporivir [74], [80]. Like for Myrcludex B (see below), the IC50s of cyclosporin A to inhibit HBV and HDV infection is significantly lower compared to its inhibitory effect on taurocholate (TC) uptake, indicating that a therapeutic effect against HBV might be achieved at non-NTCP-saturating concentrations [74]. However, since cyclosporins are not specific for NTCP and inhibit (in addition to modulate intracellular cyclophilins) other bile salt transporters like organic anion-transporting polypeptide (OATP) at even lower concentrations [81] their therapeutic use for CHB and chronic HDV infection might be limited.
HBV L-protein derived lipopeptides as highly specific inhibitors of NTCP
Key point.
hNTCP expression in non-susceptible hosts provides the basis for the development of animal models for HBV and HDV and the understanding of host specificity.
Due to the high sequence homologies in their receptor binding site, the N-terminal preS1-domains of primate HBVs, like WMHBV or Gorilla HBV and even the recently discovered Bat-hepatitis B viruses are assumed to specifically bind their respective host Ntcps. This interaction only occurs after a preceding interaction to and possibly activation by HSPGs. Remarkably, virions do not expose the N-terminal preS-1 NTCP binding site 9-NPLGFFP-15 on their surface [53]. Accordingly, HBV may hardly be recognized and inhibited by monoclonal antibodies recognizing this motif [82]. Moreover, chronically infected patients with high loads of preS-containing virions and subviral particles do not show elevated serum bile salt levels indicating that the NTCP may not or only partially blocked by the presence of these particles [83]. In contrast, synthetic preS1-derived lipopeptides that contain the NPLGFFP-motif are potent NTCP-binders and block the bile salt transporter function [26], [41], [75]. Such peptides have been characterized extensively with regard to their specific activities and the role of the fatty acid moiety which is required for anchoring the peptide in the membrane close to the NTCP binding site [9], [18], [19], [20], [21], [22], [84], [85], [86]. A lead substance of these peptides, which is presently in clinical development, is Myrcludex B. It efficiently inhibits HBV and HDV infection of hepatocytes at already picomolar concentrations [18]. Notably, Myrcludex B as a peptidic drug of 47 amino acids contains preS-specific epitopes including the essential receptor binding motif 9-NPLGFFP-15. It also contains C-terminally located epitopes (e.g. 19-DPAF-22), which may induce highly neutralising antibodies [87]. Accordingly, Myrcludex B therapy may induce anti-preS-specific antibodies that might help to eliminate HBV or HDV from the liver.
In contrast to the small substrate molecules mentioned above, preS-derived lipopeptides (including Myrcludex B, as the clinically used lead substance) are allosteric inhibitors of NTCP, which irreversibly bind and inactivate the HBV receptor function of the molecule. These peptides also inhibit the bile acid transporter function, but only at ca. 500-fold higher concentrations [26], [75]. Functional recovery of NTCP is slow and probably only possible when the preS/receptor complex is dissociated after endocytic turnover or replacement by de novo synthesis [21]. Whether functional recycling of NTCP after formation of a preS-complex can occur is presently unknown. The reason for the remarkable differences in the IC50s of e.g. Myrcludex B between bile salt transporter (IC50 ca. 47 nM) activity and HBV/HDV infection inhibition (ca. 0.1 nM) is still unclear. However, a plausible assumption is that the HBV L-protein forms multimeric (e.g. trimeric) complexes with NTCP (as it has been shown for many other enveloped viruses). Such a complex could be functionally inactivated, if only one out of three binding sites is occupied by the peptidic fragment. In fact, NTCP has been shown to form multimeric complexes [88]. From a therapeutic point of view, this allows to inactivate the receptor function of NTCP at doses that do not significantly affect its natural function as a bile salt transporter.
Therapeutic efficacy of Myrcludex B in HBV and HDV infected patients
A phase I safety trial revealed that Myrcludex B, the first in class entry inhibitor addressing the NTCP-receptor of HBV was well tolerated in healthy volunteers at intravenously applied dose of up to 20 mg. Pharmacokinetic studies revealed that doses of >10 mg subcutaneously resulted in NTCP saturation >80% for at least 15 h (Blank et al., under revision). The proof of clinical efficacy of Myrcludex B in chronically HBV infected patients has been achieved in a phase IIa clinical trial (Bogolomov et al., AASLD meeting 2014). A sub study of this trial was performed in HBV/HDV co-infected individuals (Bogolomov et al., under revision). The trial performed in HBeAg-negative HBV monoinfected patients shows a reduction in HBV serum levels by a mean of 101.45 within 12 weeks of treatment in the 10 mg arm. Assuming that Myrcludex B only interferes with de novo infection of cells and that the rate of virus production per infected cells remains unchanged, this indicates a substantial reduction of the number of infected cells during treatment. However, this has to be proven directly in future studies by immunohistochemistry of liver biopsies. In the sub study of 24 HDV co-infected HBV patients (Bogolomov et al., under revision) Myrcludex B was applied either alone at 2 mg daily, or in combination with PegIFN for 24 weeks. Myrcludex B was well tolerated in all patients, with only slightly increased conjugated bile salt levels. Within 24 weeks HDV RNA declined by >1 log in all cohorts and even became negative in two patients each in the Myrcludex B and PegIFN control arm. Remarkably, negativation was observed in five of seven evaluable patients of the Myrcludex B/PegIFN combination arm, indicating a synergistic effect both drugs.
Taken together these first proof of concept studies using Myrcludex B demonstrate that targeting NTCP as a key molecule of hepatocyte specific entry of HBV is a feasible clinical concept in future therapeutic approaches aiming at the functional cure of chronic Hepatitis B and Hepatitis D. It will be interesting to see if long-term inhibition of viral entry results in HBsAg loss or anti-HBsAg seroconversion and whether combination with immunomodulatory drugs like IFN-alpha can induce and/or accelerate such a process.
Financial support
Wenhui Li receives funding from the Ministry of Science and Technology (MOST), the National Natural Science Foundation (NSF) of China, and the Science and Technology Bureau of Beijing Municipal Government. Stephan Urban receives funding from the “German Center of Infectious Diseases” (DZIF), the “Deutsche Forschungsgemeinschaft” (DFG) and the “Deutsche Krebshilfe”.
Conflict of interest
Wenhui Li is a co-inventor of a patent application for compositions and uses of functional receptor for HBV/HDV virus. Stephan Urban is co-applicant and co-inventor of patents protecting Myrcludex B.
Contributor Information
Wenhui Li, Email: liwenhui@nibs.ac.cn.
Stephan Urban, Email: Stephan.Urban@med.uni-heidelberg.de.
References
Author names in bold designate shared co-first authorship
- 1.Ganem D., Prince A.M. Hepatitis B virus infection–natural history and clinical consequences. N Engl J Med. 2004;350:1118–1129. doi: 10.1056/NEJMra031087. [DOI] [PubMed] [Google Scholar]
- 2.Seeger C., Mason W.S. Molecular biology of hepatitis B virus infection. Virology. 2015;479–480:672–686. doi: 10.1016/j.virol.2015.02.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Suk-Fong Lok A. Hepatitis B: 50 years after the discovery of Australia antigen. J Viral Hepat. 2016;23:5–14. doi: 10.1111/jvh.12444. [DOI] [PubMed] [Google Scholar]
- 4.Gripon P., Diot C., Theze N., Fourel I., Loreal O., Brechot C. Hepatitis B virus infection of adult human hepatocytes cultured in the presence of dimethyl sulfoxide. J Virol. 1988;62:4136–4143. doi: 10.1128/jvi.62.11.4136-4143.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gripon P., Diot C., Corlu A., Guguen-Guillouzo C. Regulation by dimethylsulfoxide, insulin, and corticosteroids of hepatitis B virus replication in a transfected human hepatoma cell line. J Med Virol. 1989;28:193–199. doi: 10.1002/jmv.1890280316. [DOI] [PubMed] [Google Scholar]
- 6.Glebe D., Aliakbari M., Krass P., Knoop E.V., Valerius K.P., Gerlich W.H. Pre-s1 antigen-dependent infection of Tupaia hepatocyte cultures with human hepatitis B virus. J Virol. 2003;77:9511–9521. doi: 10.1128/JVI.77.17.9511-9521.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kock J., Baumert T.F., Delaney W.E., 4th, Blum H.E., von Weizsacker F. Inhibitory effect of adefovir and lamivudine on the initiation of hepatitis B virus infection in primary tupaia hepatocytes. Hepatology. 2003;38:1410–1418. doi: 10.1016/j.hep.2003.09.009. [DOI] [PubMed] [Google Scholar]
- 8.Walter E., Keist R., Niederost B., Pult I., Blum H.E. Hepatitis B virus infection of tupaia hepatocytes in vitro and in vivo. Hepatology. 1996;24:1–5. doi: 10.1002/hep.510240101. [DOI] [PubMed] [Google Scholar]
- 9.Gripon P., Rumin S., Urban S., Le Seyec J., Glaise D., Cannie I. Infection of a human hepatoma cell line by hepatitis B virus. Proc Natl Acad Sci U S A. 2002;99:15655–15660. doi: 10.1073/pnas.232137699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Glebe D., Urban S. Viral and cellular determinants involved in hepadnaviral entry. World J Gastroenterol. 2007;13:22–38. doi: 10.3748/wjg.v13.i1.22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fernholz D., Galle P.R., Stemler M., Brunetto M., Bonino F., Will H. Infectious hepatitis B virus variant defective in pre-S2 protein expression in a chronic carrier. Virology. 1993;194:137–148. doi: 10.1006/viro.1993.1243. [DOI] [PubMed] [Google Scholar]
- 12.Ni Y., Sonnabend J., Seitz S., Urban S. The pre-s2 domain of the hepatitis B virus is dispensable for infectivity but serves a spacer function for L-protein-connected virus assembly. J Virol. 2010;84:3879–3888. doi: 10.1128/JVI.02528-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sureau C., Salisse J. A conformational heparan sulfate binding site essential to infectivity overlaps with the conserved hepatitis B virus a-determinant. Hepatology. 2013;57:985–994. doi: 10.1002/hep.26125. [DOI] [PubMed] [Google Scholar]
- 14.Le Seyec J., Chouteau P., Cannie I., Guguen-Guillouzo C., Gripon P. Infection process of the hepatitis B virus depends on the presence of a defined sequence in the pre-S1 domain. J Virol. 1999;73:2052–2057. doi: 10.1128/jvi.73.3.2052-2057.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Blanchet M., Sureau C. Infectivity determinants of the hepatitis B virus pre-S domain are confined to the N-terminal 75 amino acid residues. J Virol. 2007;81:5841–5849. doi: 10.1128/JVI.00096-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bruss V., Hagelstein J., Gerhardt E., Galle P.R. Myristylation of the large surface protein is required for hepatitis B virus in vitro infectivity. Virology. 1996;218:396–399. doi: 10.1006/viro.1996.0209. [DOI] [PubMed] [Google Scholar]
- 17.Gripon P., Le Seyec J., Rumin S., Guguen-Guillouzo C. Myristylation of the hepatitis B virus large surface protein is essential for viral infectivity. Virology. 1995;213:292–299. doi: 10.1006/viro.1995.0002. [DOI] [PubMed] [Google Scholar]
- 18.Schulze A., Schieck A., Ni Y., Mier W., Urban S. Fine mapping of pre-S sequence requirements for hepatitis B virus large envelope protein-mediated receptor interaction. J Virol. 2010;84:1989–2000. doi: 10.1128/JVI.01902-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Engelke M., Mills K., Seitz S., Simon P., Gripon P., Schnolzer M. Characterization of a hepatitis B and hepatitis delta virus receptor binding site. Hepatology. 2006;43:750–760. doi: 10.1002/hep.21112. [DOI] [PubMed] [Google Scholar]
- 20.Glebe D., Urban S., Knoop E.V., Cag N., Krass P., Grun S. Mapping of the hepatitis B virus attachment site by use of infection-inhibiting preS1 lipopeptides and tupaia hepatocytes. Gastroenterology. 2005;129:234–245. doi: 10.1053/j.gastro.2005.03.090. [DOI] [PubMed] [Google Scholar]
- 21.Gripon P., Cannie I., Urban S. Efficient inhibition of hepatitis B virus infection by acylated peptides derived from the large viral surface protein. J Virol. 2005;79:1613–1622. doi: 10.1128/JVI.79.3.1613-1622.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Petersen J., Dandri M., Mier W., Lutgehetmann M., Volz T., von Weizsacker F. Prevention of hepatitis B virus infection in vivo by entry inhibitors derived from the large envelope protein. Nat Biotechnol. 2008;26:335–341. doi: 10.1038/nbt1389. [DOI] [PubMed] [Google Scholar]
- 23.Yan H., Zhong G., Xu G., He W., Jing Z., Gao Z. Sodium taurocholate cotransporting polypeptide is a functional receptor for human hepatitis B and D virus. Elife. 2012;1 doi: 10.7554/eLife.00049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Drexler J.F., Geipel A., Konig A., Corman V.M., van Riel D., Leijten L.M. Bats carry pathogenic hepadnaviruses antigenically related to hepatitis B virus and capable of infecting human hepatocytes. Proc Natl Acad Sci U S A. 2013;110:16151–16156. doi: 10.1073/pnas.1308049110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhong G., Yan H., Wang H., He W., Jing Z., Qi Y. Sodium taurocholate cotransporting polypeptide mediates woolly monkey hepatitis B virus infection of Tupaia hepatocytes. J Virol. 2013;87:7176–7184. doi: 10.1128/JVI.03533-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ni Y., Lempp F.A., Mehrle S., Nkongolo S., Kaufman C., Falth M. Hepatitis B and D viruses exploit sodium taurocholate co-transporting polypeptide for species-specific entry into hepatocytes. Gastroenterology. 2014;146:1070–1083. doi: 10.1053/j.gastro.2013.12.024. [DOI] [PubMed] [Google Scholar]
- 27.Li W. The hepatitis B virus receptor. Annu Rev Cell Dev Biol. 2015;31:125–147. doi: 10.1146/annurev-cellbio-100814-125241. [DOI] [PubMed] [Google Scholar]
- 28.Anwer M.S., Stieger B. Sodium-dependent bile salt transporters of the SLC10A transporter family: more than solute transporters. Pflugers Arch. 2014;466:77–89. doi: 10.1007/s00424-013-1367-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Slijepcevic D., Kaufman C., Wichers C.G., Gilglioni E.H., Lempp F.A., Duijst S. Impaired uptake of conjugated bile acids and Hepatitis B Virus preS1-binding in Na -taurocholate cotransporting polypeptide knockout mice. Hepatology. 2015;62:207–219. doi: 10.1002/hep.27694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Vaz F.M., Paulusma C.C., Huidekoper H., de Ru M., Lim C., Koster J. Sodium taurocholate cotransporting polypeptide (SLC10A1) deficiency: Conjugated hypercholanemia without a clear clinical phenotype. Hepatology. 2015;61:260–267. doi: 10.1002/hep.27240. [DOI] [PubMed] [Google Scholar]
- 31.Stieger B. The role of the sodium-taurocholate cotransporting polypeptide (NTCP) and of the bile salt export pump (BSEP) in physiology and pathophysiology of bile formation. Handb Exp Pharmacol. 2011:205–259. doi: 10.1007/978-3-642-14541-4_5. [DOI] [PubMed] [Google Scholar]
- 32.Schieck A., Schulze A., Gahler C., Muller T., Haberkorn U., Alexandrov A. Hepatitis B virus hepatotropism is mediated by specific receptor recognition in the liver and not restricted to susceptible hosts. Hepatology. 2013;58:43–53. doi: 10.1002/hep.26211. [DOI] [PubMed] [Google Scholar]
- 33.Stieger B., Hagenbuch B., Landmann L., Hochli M., Schroeder A., Meier P.J. In situ localization of the hepatocytic Na+/Taurocholate cotransporting polypeptide in rat liver. Gastroenterology. 1994;107:1781–1787. doi: 10.1016/0016-5085(94)90821-4. [DOI] [PubMed] [Google Scholar]
- 34.Meier A., Mehrle S., Weiss T.S., Mier W., Urban S. Myristoylated PreS1-domain of the hepatitis B virus L-protein mediates specific binding to differentiated hepatocytes. Hepatology. 2013;58:31–42. doi: 10.1002/hep.26181. [DOI] [PubMed] [Google Scholar]
- 35.Schulze A., Mills K., Weiss T.S., Urban S. Hepatocyte polarization is essential for the productive entry of the hepatitis B virus. Hepatology. 2012;55:373–383. doi: 10.1002/hep.24707. [DOI] [PubMed] [Google Scholar]
- 36.Kullak-Ublick G.A., Glasa J., Boker C., Oswald M., Grutzner U., Hagenbuch B. Chlorambucil-taurocholate is transported by bile acid carriers expressed in human hepatocellular carcinomas. Gastroenterology. 1997;113:1295–1305. doi: 10.1053/gast.1997.v113.pm9322525. [DOI] [PubMed] [Google Scholar]
- 37.Liang D., Hagenbuch B., Stieger B., Meier P.J. Parallel decrease of Na(+)-taurocholate cotransport and its encoding mRNA in primary cultures of rat hepatocytes. Hepatology. 1993;18:1162–1166. [PubMed] [Google Scholar]
- 38.Rippin S.J., Hagenbuch B., Meier P.J., Stieger B. Cholestatic expression pattern of sinusoidal and canalicular organic anion transport systems in primary cultured rat hepatocytes. Hepatology. 2001;33:776–782. doi: 10.1053/jhep.2001.23433. [DOI] [PubMed] [Google Scholar]
- 39.Peng L., Zhao Q., Li Q., Li M., Li C., Xu T. The p.Ser267Phe variant in SLC10A1 is associated with resistance to chronic hepatitis B. Hepatology. 2015;61:1251–1260. doi: 10.1002/hep.27608. [DOI] [PubMed] [Google Scholar]
- 40.Pan W., Song I.S., Shin H.J., Kim M.H., Choi Y.L., Lim S.J. Genetic polymorphisms in Na+-taurocholate co-transporting polypeptide (NTCP) and ileal apical sodium-dependent bile acid transporter (ASBT) and ethnic comparisons of functional variants of NTCP among Asian populations. Xenobiotica. 2011;41:501–510. doi: 10.3109/00498254.2011.555567. [DOI] [PubMed] [Google Scholar]
- 41.Yan H., Peng B., Liu Y., Xu G., He W., Ren B. Viral entry of hepatitis B and D viruses and bile salts transportation share common molecular determinants on sodium taurocholate cotransporting polypeptide. J Virol. 2014;88:3273–3284. doi: 10.1128/JVI.03478-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hu H.H., Liu J., Lin Y.L., Luo W.S., Chu Y.J., Chang C.L. The rs2296651 (S267F) variant on NTCP (SLC10A1) is inversely associated with chronic hepatitis B and progression to cirrhosis and hepatocellular carcinoma in patients with chronic hepatitis B. Gut. 2015 doi: 10.1136/gutjnl-2015-310686. [DOI] [PubMed] [Google Scholar]
- 43.Lucifora J., Vincent I.E., Berthillon P., Dupinay T., Michelet M., Protzer U. Hepatitis B virus replication in primary macaque hepatocytes: crossing the species barrier toward a new small primate model. Hepatology. 2010;51:1954–1960. doi: 10.1002/hep.23602. [DOI] [PubMed] [Google Scholar]
- 44.Guidotti L.G., Matzke B., Schaller H., Chisari F.V. High-level hepatitis B virus replication in transgenic mice. J Virol. 1995;69:6158–6169. doi: 10.1128/jvi.69.10.6158-6169.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kuiken T., Holmes E.C., McCauley J., Rimmelzwaan G.F., Williams C.S., Grenfell B.T. Host species barriers to influenza virus infections. Science. 2006;312:394–397. doi: 10.1126/science.1122818. [DOI] [PubMed] [Google Scholar]
- 46.Lu G., Wang Q., Gao G.F. Bat-to-human: spike features determining ’host jump’ of coronaviruses SARS-CoV, MERS-CoV, and beyond. Trends Microbiol. 2015;23:468–478. doi: 10.1016/j.tim.2015.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Dupinay T., Gheit T., Roques P., Cova L., Chevallier-Queyron P., Tasahsu S.I. Discovery of naturally occurring transmissible chronic hepatitis B virus infection among Macaca fascicularis from Mauritius Island. Hepatology. 2013;58:1610–1620. doi: 10.1002/hep.26428. [DOI] [PubMed] [Google Scholar]
- 48.Yan H., Peng B., He W., Zhong G., Qi Y., Ren B. Molecular determinants of hepatitis B and D virus entry restriction in mouse sodium taurocholate cotransporting polypeptide. J Virol. 2013;87:7977–7991. doi: 10.1128/JVI.03540-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.He W., Ren B., Mao F., Jing Z., Li Y., Liu Y. Hepatitis D virus infection of mice expressing human sodium taurocholate co-transporting polypeptide. PLoS Pathog. 2015 doi: 10.1371/journal.ppat.1004840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Lempp F.A., Mutz P., Lipps C., Wirth D., Bartenschlager R., Urban S. Evidence that hepatitis B virus replication in mouse cells is limited by the lack of a host cell dependency factor. J Hepatol. 2016;64:556–564. doi: 10.1016/j.jhep.2015.10.030. [DOI] [PubMed] [Google Scholar]
- 51.(a) Cui X., Guo J.T., Hu J. Hepatitis B virus covalently closed circular DNA formation in immortalized mouse hepatocytes associated with nucleocapsid destabilization. J Virol. 2015;89:9021–9028. doi: 10.1128/JVI.01261-15. [DOI] [PMC free article] [PubMed] [Google Scholar]; (b) Lempp F.A., Qu B., Wang Y.X., Urban S., Hepatitis B. Virus infection of a mouse hepatic cell line reconstituted with human sodium taurocholate cotransporting polypeptide. J Virol. 2016 doi: 10.1128/JVI.02832-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Verrier E.R., Colpitts C.C., Bach C., Heydmann L., Weiss A., Renaud M. A targeted functional RNA interference screen uncovers glypican 5 as an entry factor for hepatitis B and D viruses. Hepatology. 2016;63:35–48. doi: 10.1002/hep.28013. [DOI] [PubMed] [Google Scholar]
- 53.Urban S., Bartenschlager R., Kubitz R., Zoulim F. Strategies to inhibit entry of HBV and HDV into hepatocytes. Gastroenterology. 2014;147:48–64. doi: 10.1053/j.gastro.2014.04.030. [DOI] [PubMed] [Google Scholar]
- 54.Yan H., Liu Y., Sui J., Li W. NTCP opens the door for hepatitis B virus infection. Antiviral Res. 2015;121:24–30. doi: 10.1016/j.antiviral.2015.06.002. [DOI] [PubMed] [Google Scholar]
- 55.Zeisel M.B., Lucifora J., Mason W.S., Sureau C., Beck J., Levrero M. Towards an HBV cure: state-of-the-art and unresolved questions-report of the ANRS workshop on HBV cure. Gut. 2015;64:1314–1326. doi: 10.1136/gutjnl-2014-308943. [DOI] [PubMed] [Google Scholar]
- 56.Guo J.T., Guo H. Metabolism and function of hepatitis B virus cccDNA: Implications for the development of cccDNA-targeting antiviral therapeutics. Antiviral Res. 2015;122:91–100. doi: 10.1016/j.antiviral.2015.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Tropberger P., Mercier A., Robinson M., Zhong W., Ganem D.E., Holdorf M. Mapping of histone modifications in episomal HBV cccDNA uncovers an unusual chromatin organization amenable to epigenetic manipulation. Proc Natl Acad Sci U S A. 2015;112:E5715–E5724. doi: 10.1073/pnas.1518090112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Teissier E., Penin F., Pecheur E.I. Targeting cell entry of enveloped viruses as an antiviral strategy. Molecules. 2011;16:221–250. doi: 10.3390/molecules16010221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Iwami S., Takeuchi J.S., Nakaoka S., Mammano F., Clavel F., Inaba H. Cell-to-cell infection by HIV contributes over half of virus infection. Elife. 2015;4 doi: 10.7554/eLife.08150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Henrich T.J., Kuritzkes D.R. HIV-1 entry inhibitors: recent development and clinical use. Curr Opin Virol. 2013;3:51–57. doi: 10.1016/j.coviro.2012.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.von Recum H.A., Pokorski J.K. Peptide and protein-based inhibitors of HIV-1 co-receptors. Exp Biol Med (Maywood) 2013;238:442–449. doi: 10.1177/1535370213480696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Eggink D., Berkhout B., Sanders R.W. Inhibition of HIV-1 by fusion inhibitors. Curr Pharm Des. 2010;16:3716–3728. doi: 10.2174/138161210794079218. [DOI] [PubMed] [Google Scholar]
- 63.Lutgehetmann M., Mancke L.V., Volz T., Helbig M., Allweiss L., Bornscheuer T. Humanized chimeric uPA mouse model for the study of hepatitis B and D virus interactions and preclinical drug evaluation. Hepatology. 2012;55:685–694. doi: 10.1002/hep.24758. [DOI] [PubMed] [Google Scholar]
- 64.Lutgehetmann M., Volz T., Kopke A., Broja T., Tigges E., Lohse A.W. In vivo proliferation of hepadnavirus-infected hepatocytes induces loss of covalently closed circular DNA in mice. Hepatology. 2010;52:16–24. doi: 10.1002/hep.23611. [DOI] [PubMed] [Google Scholar]
- 65.Corti D., Bianchi S., Vanzetta F., Minola A., Perez L., Agatic G. Cross-neutralization of four paramyxoviruses by a human monoclonal antibody. Nature. 2013;501:439–443. doi: 10.1038/nature12442. [DOI] [PubMed] [Google Scholar]
- 66.Corti D., Lanzavecchia A. Broadly neutralizing antiviral antibodies. Annu Rev Immunol. 2013;31:705–742. doi: 10.1146/annurev-immunol-032712-095916. [DOI] [PubMed] [Google Scholar]
- 67.Marasco W.A., Sui J. The growth and potential of human antiviral monoclonal antibody therapeutics. Nat Biotechnol. 2007;25:1421–1434. doi: 10.1038/nbt1363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Katz L.H., Paul M., Guy D.G., Tur-Kaspa R. Prevention of recurrent hepatitis B virus infection after liver transplantation: hepatitis B immunoglobulin, antiviral drugs, or both? Systematic review and meta-analysis. Transpl Infect Dis. 2010;12:292–308. doi: 10.1111/j.1399-3062.2009.00470.x. [DOI] [PubMed] [Google Scholar]
- 69.Mailly L., Xiao F., Lupberger J., Wilson G.K., Aubert P., Duong F.H. Clearance of persistent hepatitis C virus infection in humanized mice using a claudin-1-targeting monoclonal antibody. Nat Biotechnol. 2015;33:549–554. doi: 10.1038/nbt.3179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Asabe S., Wieland S.F., Chattopadhyay P.K., Roederer M., Engle R.E., Purcell R.H. The size of the viral inoculum contributes to the outcome of hepatitis B virus infection. J Virol. 2009;83:9652–9662. doi: 10.1128/JVI.00867-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Lamas Longarela O., Schmidt T.T., Schoneweis K., Romeo R., Wedemeyer H., Urban S. Proteoglycans act as cellular hepatitis delta virus attachment receptors. PLoS One. 2013;8 doi: 10.1371/journal.pone.0058340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Leistner C.M., Gruen-Bernhard S., Glebe D. Role of glycosaminoglycans for binding and infection of hepatitis B virus. Cell Microbiol. 2008;10:122–133. doi: 10.1111/j.1462-5822.2007.01023.x. [DOI] [PubMed] [Google Scholar]
- 73.Schulze A., Gripon P., Urban S. Hepatitis B virus infection initiates with a large surface protein-dependent binding to heparan sulfate proteoglycans. Hepatology. 2007;46:1759–1768. doi: 10.1002/hep.21896. [DOI] [PubMed] [Google Scholar]
- 74.Nkongolo S., Ni Y., Lempp F.A., Kaufman C., Lindner T., Esser-Nobis K. Cyclosporin A inhibits hepatitis B and hepatitis D virus entry by cyclophilin-independent interference with the NTCP receptor. J Hepatol. 2014;60:723–731. doi: 10.1016/j.jhep.2013.11.022. [DOI] [PubMed] [Google Scholar]
- 75.Konig A., Doring B., Mohr C., Geipel A., Geyer J., Glebe D. Kinetics of the bile acid transporter and hepatitis B virus receptor Na+/taurocholate cotransporting polypeptide (NTCP) in hepatocytes. J Hepatol. 2014;61:867–875. doi: 10.1016/j.jhep.2014.05.018. [DOI] [PubMed] [Google Scholar]
- 76.Veloso Alves Pereira I., Buchmann B., Sandmann L., Sprinzl K., Schlaphoff V., Dohner K. Primary biliary acids inhibit hepatitis D virus (HDV) entry into human hepatoma cells expressing the sodium-taurocholate cotransporting polypeptide (NTCP) PLoS One. 2015;10 doi: 10.1371/journal.pone.0117152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Doring B., Lutteke T., Geyer J., Petzinger E. The SLC10 carrier family: transport functions and molecular structure. Curr Top Membr. 2012;70:105–168. doi: 10.1016/B978-0-12-394316-3.00004-1. [DOI] [PubMed] [Google Scholar]
- 78.Lucifora J., Esser K., Protzer U. Ezetimibe blocks hepatitis B virus infection after virus uptake into hepatocytes. Antiviral Res. 2013;97:195–197. doi: 10.1016/j.antiviral.2012.12.008. [DOI] [PubMed] [Google Scholar]
- 79.Kim R.B., Leake B., Cvetkovic M., Roden M.M., Nadeau J., Walubo A. Modulation by drugs of human hepatic sodium-dependent bile acid transporter (sodium taurocholate cotransporting polypeptide) activity. J Pharmacol Exp Ther. 1999;291:1204–1209. [PubMed] [Google Scholar]
- 80.Watashi K., Sluder A., Daito T., Matsunaga S., Ryo A., Nagamori S. Cyclosporin A and its analogs inhibit hepatitis B virus entry into cultured hepatocytes through targeting a membrane transporter, sodium taurocholate cotransporting polypeptide (NTCP) Hepatology. 2014;59:1726–1737. doi: 10.1002/hep.26982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Treiber A., Schneiter R., Hausler S., Stieger B. Bosentan is a substrate of human OATP1B1 and OATP1B3: inhibition of hepatic uptake as the common mechanism of its interactions with cyclosporin A, rifampicin, and sildenafil. Drug Metab Dispos. 2007;35:1400–1407. doi: 10.1124/dmd.106.013615. [DOI] [PubMed] [Google Scholar]
- 82.Bremer C.M., Sominskaya I., Skrastina D., Pumpens P., El Wahed A.A., Beutling U. N-terminal myristoylation-dependent masking of neutralizing epitopes in the preS1 attachment site of hepatitis B virus. J Hepatol. 2011;55:29–37. doi: 10.1016/j.jhep.2010.10.019. [DOI] [PubMed] [Google Scholar]
- 83.Sugita T., Amano K., Nakano M., Masubuchi N., Sugihara M., Matsuura T. Analysis of the serum bile Acid composition for differential diagnosis in patients with liver disease. Gastroenterol Res Pract. 2015;2015 doi: 10.1155/2015/717431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Kim D.H., Ni Y., Lee S.H., Urban S., Han K.H. An anti-viral peptide derived from the preS1 surface protein of hepatitis B virus. BMB Rep. 2008;41:640–644. doi: 10.5483/bmbrep.2008.41.9.640. [DOI] [PubMed] [Google Scholar]
- 85.Lempp F.A., Urban S. Inhibitors of hepatitis B virus attachment and entry. Intervirology. 2014;57:151–157. doi: 10.1159/000360948. [DOI] [PubMed] [Google Scholar]
- 86.Barrera A., Guerra B., Notvall L., Lanford R.E. Mapping of the hepatitis B virus pre-S1 domain involved in receptor recognition. J Virol. 2005;79:9786–9798. doi: 10.1128/JVI.79.15.9786-9798.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Glebe D. Attachment sites and neutralising epitopes of hepatitis B virus. Minerva Gastroenterol Dietol. 2006;52:3–21. [PubMed] [Google Scholar]
- 88.Bijsmans I.T., Bouwmeester R.A., Geyer J., Faber K.N., van de Graaf S.F. Homo- and hetero-dimeric architecture of the human liver Na(+)-dependent taurocholate co-transporting protein. Biochem J. 2012;441:1007–1015. doi: 10.1042/BJ20111234. [DOI] [PubMed] [Google Scholar]