Abstract
lncRNA cancer susceptibility candidate 2 (CASC2) is a recently identified oncogenic lncRNA in different types of cancers. Our preliminary microarray data showed that lncRNA CASC2 was downregulated in the plasma of patients with rheumatoid arthritis (RA), indicating the involvement of this lncRNA in RA. In the present study, lncRNA CASC2 and IL-17 in plasma were detected by reverse transcription--quantitative PCR and ELISA, respectively. Diagnostic analyses were performed using receiver operating characteristic curves. Flow cytometry was performed to evaluate cell apoptosis. The effects of lncRNA CASC2 on IL-17 expression were determined via western blotting. lncRNA CASC2 was found to be downregulated, while IL-17 was upregulated in the plasma of RA patients when compared with these levels in the plasma of healthy controls. Plasma levels of lncRNA CASC2 and IL-17 were significantly and inversely correlated in both RA patients and healthy controls. Altered plasma levels of lncRNA CASC2 and IL-17 were able to differentiate RA patients from healthy controls. Overexpression of lncRNA CASC2 promoted, while treatment with IL-17 inhibited the apoptosis of human fibroblast-like synoviocytes (HFLSs) isolated from RA patients. Overexpression of lncRNA CASC2 inhibited IL-17 expression in HFLS, while treatment with IL-17 did not significantly affect the expression of lncRNA CASC2. Therefore, downregulation of lncRNA CASC2 is involved in RA and lncRNA CASC2 overexpression may promote the apoptosis of HFLS by downregulating IL-17.
Keywords: apoptosis, human fibroblast-like synoviocytes, IL-17, lncRNA CASC2, rheumatoid arthritis
Introduction
As a type of autoimmune disorder that mainly affects the joints, progression of rheumatoid arthritis (RA) causes progressive articular destruction and comorbidities in metabolic, vascular, psychological domains and bone (1). RA affects about 1% of the population at any stage of age, and the incidence is higher in males than in females (2). Due to the efforts made in regards to the treatment and prevention of RA, treatment outcomes of RNA have been significantly improved during the past several decades (3,4). However, sustained remission is still rare and ongoing pharmacologic therapy is always required (5). Moreover, the cause of RA remains unclear. In addition, sensitive diagnostic and prognostic biomarkers for RA are lacking.
As a type of inflammatory disease, the development of RA requires the involvement of multiple inflammatory cytokine (6). Thus, anti-inflammation is currently considered as a promising therapeutic target for RA treatment (7). As a pro-inflammatory factor, IL-17 is upregulated in RA and the overexpression of IL-17 contributes to the progression of RA through multiple aspects, such as angiogenesis (8). A growing body of literature has also shown that the development of RA also requires the participation of lncRNAs (9,10). lncRNA CASC2 has been characterized as an oncogenic lncRNA in different types of cancers (11,12). Previous studies have shown that lncRNA CASC2 can participate in cancer biology by affecting cell behaviors, such as cell proliferation, migration and invasion (11,12). However, the role of this lncRNA in other types of human diseases is unknown. Our preliminary microarray data revealed that lncRNA CASC2 is downregulated in the plasma of RA patients and showed an inverse correlation with the expression of IL-17. In the present study, it was demonstrated that downregulation of lncRNA CASC2 participates in RA and overexpression of lncRNA CASC2 may promote the apoptosis of human fibroblast-like synoviocytes by downregulating IL-17.
Materials and methods
Plasma specimens and human fibroblast-like synoviocytes
Plasma samples were obtained from 65 RA patients (patient group) and 54 healthy controls (control group) who were admitted to the First People's Hospital of Tianmen City from January 2015 to July 2018. Inclusion criteria of patients included: i) Patients diagnosed with RA for the first time; ii) patients with a complete medical record; iii) patients understood the experimental protocol and signed an informed consent. Exclusion criteria included: i) Patients diagnosed with multiple diseases; ii) patients received treatment before admission. The 54 healthy controls were selected from the Physiological Examination Center of the First People's Hospital of Tianmen City to match the age and sex distributions of the patient group. The patient group included 36 males and 29 females, and the ages ranged from 29 to 66 years, with a mean age of 47.1±5.2 years. The control group included 29 males and 25 females, and ages ranged from 27 to 68 years, with a mean age of 47.8±6.1 years. No significant differences in age and sex were found between the two groups. The present study was approved following the review of the Ethics Committee of the First People's Hospital of Tianmen City. All participants signed informed consent before participating.
Human fibroblast-like synoviocytes (HFLSs) were isolated from 2 RA patients according to the methods described by Lee et al (13). HELSs were collected from passage 3 to 5 for subsequent experiments.
Total RNA extraction and reverse transcription-quantitative PCR (RT-qPCR)
To detect the expression of lncRNA CASC2, total RNA extraction was performed using a Monarch® Total RNA Miniprep kit (New England BioLabs, Inc., Ipswich, MA, USA). cDNA was synthesized using a High-Capacity cDNA Reverse Transcription kit (Thermo Fisher Scientific, Inc., Waltham, MA, USA) at 25°C for 5 min, 50°C for 30 min and 75°C for 5 min. A Luna® Universal One-Step RT-qPCR kit (New England BioLabs, Inc.) was used to prepare all PCR reaction systems. Primers for the lncRNA CASC2 and endogenous control GAPDH were designed and synthesized by Sangon Biotech Co., Ltd. (Shanghai, China). The primer sequences were: CASC2, forward, 5′-TACAGGACAGTCAGTGGTGGTA-3′ and reverse, 5′-ACATCTAGCTTAGGAATGTGGC-3′; and GAPDH, forward, 5′-TCAAGAAGGTGGTGAAGCA-3′ and reverse, 5′-AGGTGGAGGAGTGGGTGT-3′. The qPCR reaction conditions consisted of 95°C for 1 min, followed by 40 cycles of 95°C for 10 sec and 56.5°C for 30 sec. Expression of lncRNA CASC2 was normalized to GAPDH using the 2−ΔΔCq method (14).
Enzyme-linked immunosorbent assay
To measure the plasma levels of IL-17, enzyme-linked immunosorbent assay (ELISA) was performed using Human IL-17 Quantikine ELISA Kit (cat. no. D1700; R&D Systems, Inc., Minneapolis, MN, USA). All operations were performed according to the instructions provided by R&D Systems, Inc. Plasma levels of IL-17 were normalized to pg/ml.
Cell transfection
lncRNA CASC2-expression (pcDNA3) vectors were designed and synthesized by Sangon Biotech Co., Ltd. (Shanghai, China). The genomic DNA sequence of lncRNA CASC2 was used. Cell transfections were performed using Lipofectamine® 2000 reagent (Invitrogen; Thermo Fisher Scientific, Inc.), with all steps performed according to the manufacturer's instructions. Lipofectamine 2000 reagent was first mixed with vectors to form a reagent-vector complex, followed by incubation with cells (105 cells/ml) at 37°C for 5 h for each transfection. The dose of the vectors was 10 nM. The negative control consisted of empty vector transfection. Control cells were cells treated only with Lipofectamine 2000 reagent.
Cell apoptosis assay
The overexpression rate of lncRNA CASC2 reached 200% at 24 h after transfection. Therefore, cell apoptosis was detected by cell apoptosis assay at this time point. Serum-free cell culture medium was used to prepare single-cell suspensions with a cell density of 5×104 cells/ml. The cell suspension was transferred to a 6-well plate with 2 ml in each well. Cells were cultivated at 37°C for 48 h and were then digested with 0.25% trypsin. After that, Annexin V-FITC (Dojindo Molecular Technologies, Inc., Kumamoto, Japan) and propidium iodide staining was performed at 4°C for 30 min, and apoptotic cells were detected by flow cytometry. Data were analyzed using FCS Express 6 Flow Cytometry Software (De Novo Software, Glendale, CA, USA).
Western blot analysis
To detect the expression of IL-17, total protein extraction was performed using a total protein extraction kit (cat. no. 2140; Merck KGaA, Darmstadt, Germany). Protein samples were quantified using a Pierce™ BCA Protein Assay kit (Pierce; Thermo Fisher Scientific, Inc.). After denaturing, protein samples (30 µg/lane) were subjected to 10% SDS-PAGE, followed by gel transfer to PVDF membranes. After blocking in 5% non-fat milk at 25°C for 2 h, the membranes were subjected to incubation with primary antibodies [rabbit anti-human IL-17 (1:1,200; cat. no. ab79056; Abcam, Cambridge, UK) and rabbit anti-human GAPDH (1:1,400; cat. no. ab9485; Abcam; 1:1,400) overnight at 4°C, followed by incubation with horseradish peroxidase--conjugated goat anti-rabbit IgG secondary antibody (1:1,000; cat. no. MBS435036; MyBioSource, Inc., San Diego, CA, USA) for 2 h at 22°C. ECL (Sigma-Aldrich; Merck KGaA) was used to develop signals and signals were normalized using ImageJ v.1.48 software (National Institutes of Health, Bethesda, MD, USA).
Statistical analysis
All experiments were carried out in triplicate, and data are expressed as the mean ± standard deviation. All statistical analyses were performed using GraphPad Prism 6 software (GraphPad Software, Inc., La Jolla, CA, USA). Correlations between plasma levels of lncRNA CASC2 and IL-17 were analyzed by Pearson's correlation coefficient. Receiver operating characteristic (ROC) analysis was performed to evaluate the diagnostic values of plasma lncRNA CASC2 and IL-17 for RA with RA patients as true positive cases and healthy controls as true negative cases. Comparisons between 2 groups were performed by unpaired t test. Comparisons among 3 groups were performed by one-way ANOVA followed by Tukey post hoc test. Differences were deemed statistically significant at P<0.05.
Results
lncRNA CASC2 is downregulated, while IL-17 is upregulated in the plasma of RA patients
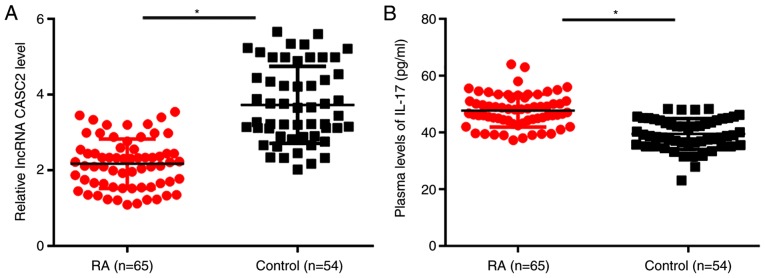
Results of qPCR showed that, compared with the healthy controls, plasma levels of lncRNA CASC2 were significantly decreased in patients with RA (P<0.05; Fig. 1A). In contrast, ELISA results showed that plasma IL-17 was upregulated in the plasma of RA patients when compared with that in the plasma of healthy controls (P<0.05; Fig. 1B).
Figure 1.
lncRNA CASC2 is downregulated, while IL-17 is upregulated in the plasma of RA patients. (A) Relative expression of lncRNA CASC2 was normalized to the patients with the lowest expression level. qPCR results showed that, compared with the healthy controls, plasma levels of lncRNA CASC2 were significantly decreased in patients with RA. (B) In contrast, ELISA results showed that plasma IL-17 was upregulated in the plasma of RA patients when compared to that noted in the plasma of healthy controls (*P<0.05). CASC2, lncRNA cancer susceptibility candidate 2; IL-17, interleukin-17; RA, rheumatoid arthritis.
Plasma levels of lncRNA CASC2 and IL-17 are significantly and inversely correlated in both RA patients and healthy controls
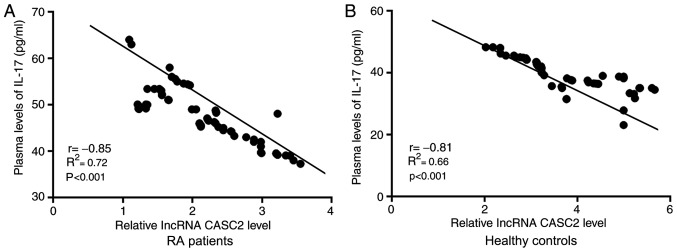
Correlations between plasma levels of lncRNA CASC2 and IL-17 were analyzed by Pearson's correlation coefficient. As shown in Fig. 2, plasma levels of lncRNA CASC2 and IL-17 were significantly and inversely correlated in both RA patients (Fig. 2A) and healthy controls (Fig. 2B).
Figure 2.
Plasma levels of lncRNA CASC2 and IL-17 are significantly and inversely correlated in both RA patients and healthy controls. Relative expression of lncRNA CASC2 was normalized to the patients with the lowest expression level. Pearson's correlation coefficient showed that plasma levels of lncRNA CASC2 and IL-17 were significantly and inversely correlated in both (A) RA patients and (B) healthy controls. CASC2, lncRNA cancer susceptibility candidate 2; IL-17, interleukin-17; RA, rheumatoid arthritis.
Altered plasma levels of lncRNA CASC2 and IL-17 differentiate RA patients from healthy controls
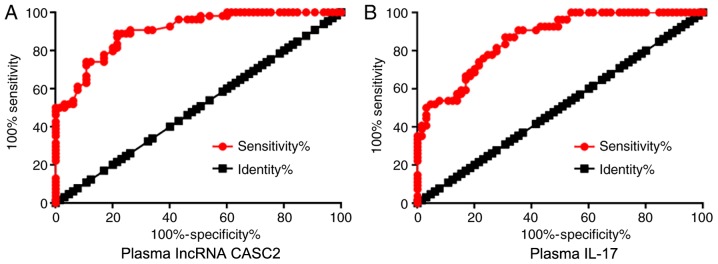
ROC curve analysis was performed to evaluate the diagnostic values of plasma lncRNA CASC2 and IL-17 for RA with RA patients as true positive cases and healthy controls as true negative cases. For plasma lncRNA CASC2, area under the curve was 0.90, with standard error of 0.026 and 95% confidence interval of 0.85–0.95 (Fig. 3A). For plasma IL-17, area under the curve was 0.86, with standard error of 0.032 and 95% confidence interval of 0.80–0.93 (Fig. 3B).
Figure 3.
Altered plasma levels of lncRNA CASC2 and IL-17 differentiate RA patients from healthy controls. ROC curve analysis showed that altered plasma levels of (A) lncRNA CASC2 and (B) IL-17 were able to differentiate RA patients from healthy controls. CASC2, lncRNA cancer susceptibility candidate 2; IL-17, interleukin-17; RA, rheumatoid arthritis.
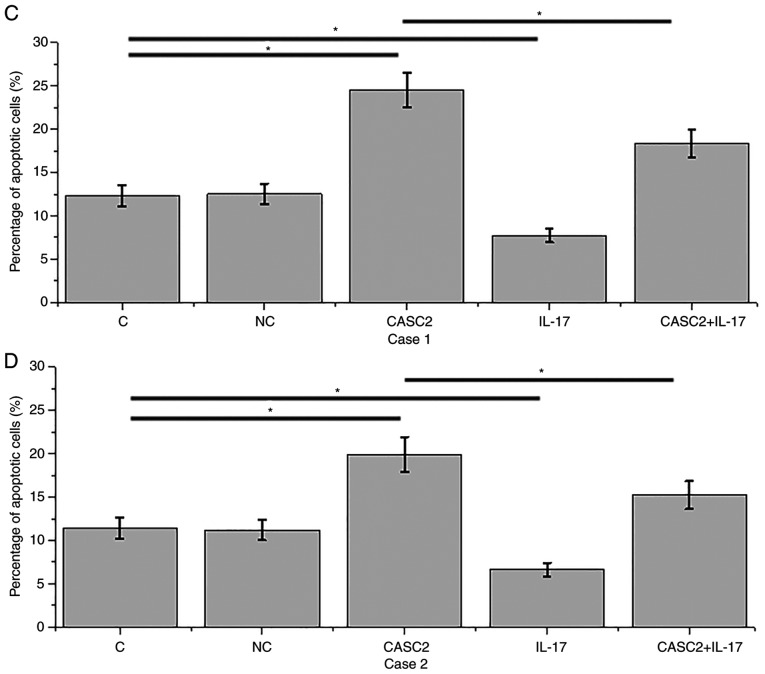
lncRNA CASC2 overexpression promotes while IL-17 inhibits the apoptosis of HFLSs
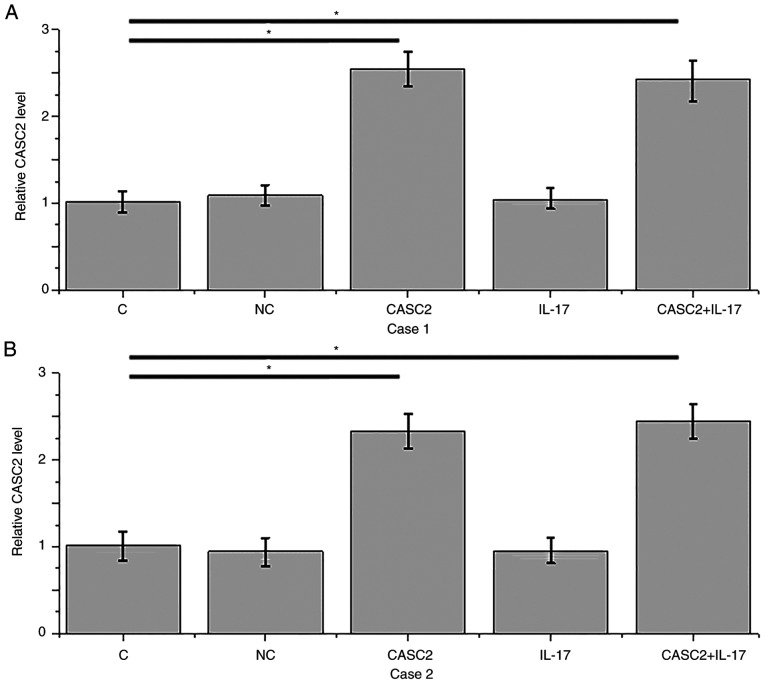
Compared with the control (C) and negative control (NC) groups, overexpression of lncRNA CASC2 was reached after transfection of HFLSs isolated from 2 RA patients (P<0.05; Fig. 4A and B). Overexpression of lncRNA CASC2 led to significant promotion of apoptosis of HFLSs isolated from 2 RA patients (Fig. 4C and D, P<0.05). In contrast, treatment with IL-17 at a dose of 10 ng/ml significantly inhibited the apoptosis of HFLSs (P<0.05; Fig. 4C and D). In addition, IL-17 treatment attenuated the promoting effect of lncRNA CASC2 overexpression on cell apoptosis (P<0.05).
Figure 4.
lncRNA CASC2 overexpression promotes while IL-17 inhibits the apoptosis of HFLSs. (A and B) Overexpression of lncRNA CASC2 was reached after transfection in HFLSs isolated from 2 RA patients. (C and D) Overexpression of lncRNA CASC2 led to a significant promotion of the percentage of apoptosis in HFLSs isolated from 2 RA patients. In contrast, treatment with IL-17 at a dose of 10 ng/ml significantly inhibited the apoptosis of HFLSs. In addition, IL-17 treatment attenuated the promoting effect of lncRNA CASC2 overexpression on cell apoptosis (*P<0.05). CASC2, lncRNA cancer susceptibility candidate 2; IL-17, interleukin-17; RA, rheumatoid arthritis; HFLSs, human fibroblast-like synoviocytes.
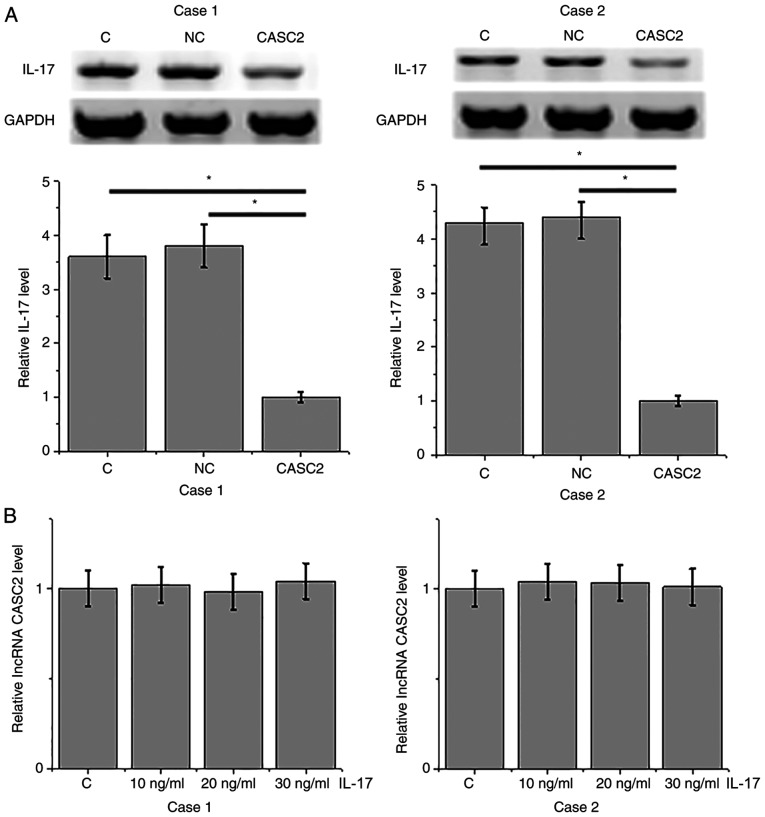
lncRNA CASC2 is a potential upstream inhibitor of IL-17 in HFLSs
Compared with the control (C) and negative control (NC) groups, overexpression of lncRNA CASC2 led to a significant inhibition of expression of IL-17 in HFLSs isolated from 2 RA patients (P<0.05; Fig. 5A). In contrast, treatment with IL-17 at doses of 10, 20 and 30 ng/ml did not significantly affect the expression of lncRNA CASC2.
Figure 5.
lncRNA CASC2 is a potential upstream inhibitor of IL-17 in HFLSs. (A) Overexpression of lncRNA CASC2 inhibited IL-17 expression in HFLSs, while (B) treatment with IL-17 did not significantly affect the expression of lncRNA CASC2 (*P<0.05). CASC2, lncRNA cancer susceptibility candidate 2; IL-17, interleukin-17; HFLSs, human fibroblast-like synoviocytes.
Discussion
lncRNA CASC2 has been characterized as an oncogenic lncRNA in different types of cancers, while the role of this lncRNA in other types of human diseases is unknown. The key finding of the preset study is that lncRNA CASC2 is downregulated in RA and the overexpression of lncRNA CASC2 inhibited IL-17 expression and promoted apoptosis of HFLSs isolated from RA patients.
A recent study showed that RA affected the expression pattern of a large set of lncRNAs (15). In another study, lncRNA HOTIAR was confirmed to be an essential regulator and clinical marker for RA (9). In addition, Yang and Chen showed that lncRNA-NR024118 interacts with shikonin to inhibit the inflammatory response in RA (10). In the present study, downregulation of lncRNA CASC2 was observed in the plasma of RA patients when compared with that noted in healthy controls. In addition, downregulation of lncRNA CASC2 was able to effectively differentiate RA patients from healthy controls. Therefore, lncRNA CASC2 may serve as a potential diagnostic marker for RA.
Overexpression of IL-17 is frequently observed in RA and upregulated IL-17 promotes RA development (8). Consistently, in the present study, the upregulation of IL-17 was observed in RA patients when compared to that noted in healthy controls. It is known that IL-17 may interact with lncRNAs to participate in biological processes (16,17). At present, the molecular mechanisms of the actions of lncRNAs in RA remain largely unknown. Importantly, a significant and inverse correlation was found between lncRNA CASC2 and IL-17 in both RA patients and healthy controls. In addition, our in vitro cell experiments also showed that overexpression of lncRNA CASC2 led to significant inhibition of the expression of IL-17 in HFLSs isolated from RA patients. These data suggest the involvement of lncRNA CASC2 in RA by inhibiting IL-17.
HFLSs participate in the development and progression of RA by producing cytokines and proteases that contribute to cartilage destruction (18). Apoptosis of HFLSs is currently considered as a potential therapeutic target for RA (19,20). Our study observed that IL-17 inhibited HFLSs apoptosis, while lncRNA CASC2 promoted HFLSs apoptosis by inhibiting IL-17. Therefore, overexpression of lncRNA CASC2 may be used to treat RA. However, the molecular mechanism of the involvement of IL-17 and CASC2 in the apoptosis of HFLSs is still unknown. Further research is warranted.
In conclusion, lncRNA CASC2 is downregulated, while IL-17 is upregulated in RA. lncRNA CASC2 overexpression promotes the apoptosis of HFLSs by downregulating IL-17, thereby suppressing the progression of RA.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during present study are available from the corresponding author on reasonable request.
Authors' contributions
CL performed all the experiments, analyzed all the data and was a major contributor in writing the manuscript. XG, SB, GZ and HW also contributed to performing the experimental tasks and aided in the drafting of the manuscript. All authors read and approved the final manuscript and agree to be accountable for all aspects of the research in ensuring that the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ethics approval and consent to participate
Ethical approval was obtained from the First People's Hospital of Tianmen City Medical Research Ethics Committee. The study followed the tenets of the Declaration of Helsinki, and informed written consent was obtained from all patients and controls.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.McInnes IB, Schett G. The pathogenesis of rheumatoid arthritis. N Engl J Med. 2011;365:2205–2219. doi: 10.1056/NEJMra1004965. [DOI] [PubMed] [Google Scholar]
- 2.McInnes IB, Schett G. Pathogenetic insights from the treatment of rheumatoid arthritis. Lancet. 2017;389:2328–2337. doi: 10.1016/S0140-6736(17)31472-1. [DOI] [PubMed] [Google Scholar]
- 3.Monti S, Montecucco C, Bugatti S, Caporali R. Rheumatoid arthritis treatment: The earlier the better to prevent joint damage. RMD Open. 2015;1(Suppl 1):e000057. doi: 10.1136/rmdopen-2015-000057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Stevenson M, Archer R, Tosh J, Simpson E, Everson-Hock E, Stevens J, Hernandez-Alava M, Paisley S, Dickinson K, Scott D, et al. Adalimumab, etanercept, infliximab, certolizumab pegol, golimumab, tocilizumab and abatacept for the treatment of rheumatoid arthritis not previously treated with disease--modifying antirheumatic drugs and after the failure of conventional disease-modifying antirheumatic drugs only: Systematic review and economic evaluation. Health Technol Assess. 2016;20:1–610. doi: 10.3310/hta20350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Smolen JS, Breedveld FC, Burmester GR, Bykerk V, Dougados M, Emery P, Kvien TK, Navarro-Compán MV, Oliver S, Schoels M, et al. Treating rheumatoid arthritis to target: 2014 update of the recommendations of an international task force. Ann Rheum Dis. 2016;75:3–15. doi: 10.1136/annrheumdis-2015-207524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Choy EH, Panayi GS. Cytokine pathways and joint inflammation in rheumatoid arthritis. N Engl J Med. 2001;344:907–916. doi: 10.1056/NEJM200103223441207. [DOI] [PubMed] [Google Scholar]
- 7.Silverstein FE, Faich G, Goldstein JL, Simon LS, Pincus T, Whelton A, Makuch R, Eisen G, Agrawal NM, Stenson WF, et al. Gastrointestinal toxicity with celecoxib vs nonsteroidal anti-inflammatory drugs for osteoarthritis and rheumatoid arthritis: The CLASS study: A randomized controlled trial. celecoxib long-term arthritis safety study. JAMA. 2000;284:1247–1255. doi: 10.1001/jama.284.10.1247. [DOI] [PubMed] [Google Scholar]
- 8.Pickens SR, Volin MV, Mandelin AM, II, Kolls JK, Pope RM, Shahrara S. IL-17 contributes to angiogenesis in rheumatoid arthritis. J Immunol. 2010;184:3233–3241. doi: 10.4049/jimmunol.0903271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Song J, Kim D, Han J, Kim Y, Lee M, Jin EJ. PBMC and exosome-derived Hotair is a critical regulator and potent marker for rheumatoid arthritis. Clin Exp Med. 2015;15:121–126. doi: 10.1007/s10238-013-0271-4. [DOI] [PubMed] [Google Scholar]
- 10.Yang KY, Chen DL. Shikonin inhibits inflammatory response in rheumatoid arthritis synovial fibroblasts via lncRNA-NR024118. Evid Based Complement Alternat Med. 2015;2015:631737. doi: 10.1155/2015/631737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pei Z, Du X, Song Y, Fan L, Li F, Gao Y, Wu R, Chen Y, Li W, Zhou H, et al. Down-regulation of lncRNA CASC2 promotes cell proliferation and metastasis of bladder cancer by activation of the Wnt/β-catenin signaling pathway. Oncotarget. 2017;8:18145–18153. doi: 10.18632/oncotarget.15210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cao Y, Xu R, Xu X, Zhou Y, Cui L, He X. Downregulation of lncRNA CASC2 by microRNA-21 increases the proliferation and migration of renal cell carcinoma cells. Mol Med Rep. 2016;14:1019–1025. doi: 10.3892/mmr.2016.5337. [DOI] [PubMed] [Google Scholar]
- 13.Lee SY, Kwok SK, Son HJ, Ryu JG, Kim EK, Oh HJ, Cho ML, Ju JH, Park SH, Kim HY. IL-17-mediated Bcl-2 expression regulates survival of fibroblast-like synoviocytes in rheumatoid arthritis through STAT3 activation. Arthritis Res Ther. 2013;15:R31. doi: 10.1186/ar4179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 15.Zhang Y, Xu YZ, Sun N, Liu JH, Chen FF, Guan XL, Li A, Wang F, Zhao QF, Wang HY, et al. Long noncoding RNA expression profile in fibroblast-like synoviocytes from patients with rheumatoid arthritis. Arthritis Res Ther. 2016;18:227. doi: 10.1186/s13075-016-1129-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Huang XD, Dai JG, Lin KT, Liu M, Ruan HT, Zhang H, Liu WG, He MX, Zhao M. Regulation of IL-17 by lncRNA of IRF-2 in the pearl oyster. Fish Shellfish Immunol. 2018;81:108–112. doi: 10.1016/j.fsi.2018.07.020. [DOI] [PubMed] [Google Scholar]
- 17.Huang W, Thomas B, Flynn RA, Gavzy SJ, Wu L, Kim SV, Hall JA, Miraldi ER, Ng CP, Rigo F, et al. DDX5 and its associated lncRNA Rmrp modulate TH17 cell effector functions. Nature. 2015;528:517–522. doi: 10.1038/nature16193. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 18.Bartok B, Firestein GS. Fibroblast-like synoviocytes: Key effector cells in rheumatoid arthritis. Immunol Rev. 2010;233:233–255. doi: 10.1111/j.0105-2896.2009.00859.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zou S, Wang C, Cui Z, Guo P, Meng Q, Shi X, Gao Y, Yang G, Han Z. β-Elemene induces apoptosis of human rheumatoid arthritis fibroblast-like synoviocytes via reactive oxygen species-dependent activation of p38 mitogen-activated protein kinase. Pharmacol Rep. 2016;68:7–11. doi: 10.1016/j.pharep.2015.06.004. [DOI] [PubMed] [Google Scholar]
- 20.Li S, Jin Z, Lu X. MicroRNA-192 suppresses cell proliferation and induces apoptosis in human rheumatoid arthritis fibroblast-like synoviocytes by downregulating caveolin 1. Mol Cell Biochem. 2017;432:123–130. doi: 10.1007/s11010-017-3003-3. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during present study are available from the corresponding author on reasonable request.