Highlights
-
•
Different stages of the spread of infectious agents were analyzed.
-
•
Short-range airborne route is potentially very important.
-
•
Short-range airborne route may be controlled by masks and personalized ventilation.
-
•
Displacement ventilation may not be applicable to control respiratory diseases.
Key Words: Respiratory droplet, indoor air, coughing, droplet dispersion, infection control, environmental ventilation
Abstract
Background
Since the 2003 severe acute respiratory syndrome epidemic, scientific exploration of infection control is no longer restricted to microbiologists or medical scientists. Many studies have reported on the release, transport, and exposure of expiratory droplets because of respiratory activities. This review focuses on the airborne spread of infectious agents from mucus to mucus in the indoor environment and their spread as governed by airflows in the respiratory system, around people, and in buildings at different transport stages.
Methods
We critically review the literature on the release of respiratory droplets, their transport and dispersion in the indoor environment, and the ultimate exposure of a susceptible host, as influenced by airflows.
Results
These droplets or droplet nuclei are transported by expired airflows, which are sometimes affected by the human body plume and use of a face mask, as well as room airflow. Room airflow is affected by human activities such as walking and door opening, and some droplets are eventually captured by a susceptible individual because of his or her inspired flows; such exposure can eventually lead to long-range spread of airborne pathogens. Direct exposure to the expired fine droplets or droplet nuclei results in short-range airborne transmission. Deposition of droplets and direct personal exposure to expired large droplets can lead to the fomite route and the droplet-borne route, respectively.
Conclusions
We have shown the opportunities for infection control at different stages of the spread. We propose that the short-range airborne route may be important in close contact, and its control may be achieved by face masks for the source patients and use of personalized ventilation. Our discussion of the effect of thermal stratification and expiratory delivery of droplets leads to the suggestion that displacement ventilation may not be applicable to hospital rooms where respiratory infection is a concern.
Since the 2003 severe acute respiratory syndrome epidemic, the 2009 H1N1 influenza pandemic, and the 2014 Middle East respiratory syndrome epidemic, scientific exploration of infection control is no longer restricted to microbiologists or medical scientists. Fluid mechanics has played a role in understanding the mechanism of transmission and in developing engineering interventions; for example, the studies of airflow dynamics by Yu et al1 provided plausible evidence of airborne transmission of severe acute respiratory syndrome. Airborne spread of infectious agents is directly relevant to the airborne route, and indirectly to the droplet-borne and fomite routes. Breathing, talking, sneezing, and coughing are major sources of some respiratory pathogens. Up to 40,000 droplets are expelled at a velocity of 100 m/s during a sneeze,2 and a cough can generate approximately 3,000 droplet nuclei.3 We now understand to some degree where and how respiratory droplets are formed and the pathogen content in each size of droplet. Turbulence and coherent structures in the airflow, mostly invisible, transport respiratory droplets between people. For example, vortex structures in coughing probably carry particles over long distances.4 Our body's thermal plumes can bring fine droplet nuclei upward, and vortices generated during door opening and wakes behind walking individuals can transport contaminated air out of an isolation room. Turbulence generated by supply air jets causes mixing and dilution of room air. Understanding these airflows is crucial to minimizing spread of infectious agents and infection transmission.
Here, we review the release of respiratory droplets, their transport and dispersion in the indoor environment, and the ultimate exposure of a susceptible host, as influenced by airflows. Microbial survival in the environment is beyond the scope of this article.
Release, transport, and exposure
Release of droplets from mucus to mouth
If we understand the mechanism of where and how respiratory droplets are generated, we may have opportunities to suppress them at the source. Knowing the number and size of respiratory droplets is also crucial.
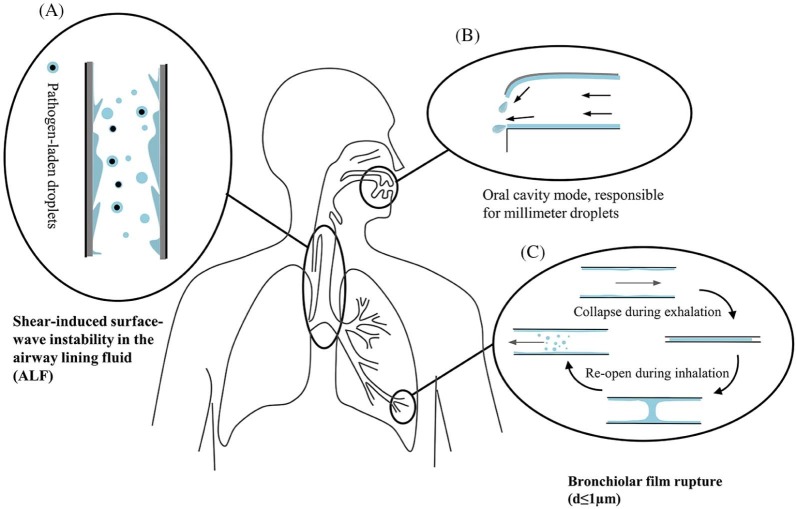
First, the content of infectious agents expelled by an infected person depends largely on the location within the respiratory tract where the droplets originate. Pathogenic microorganisms tend to be found in certain locations, particularly the tonsils and the larynx, and seldom at the front of the mouth.5 Three droplet size distribution modes have been proposed: the bronchiolar fluid film burst mode, containing droplets produced during normal breathing (d ≤ 1 µm); the laryngeal mode, most active during voicing and coughing (d ≥ 1 µm); and the oral cavity mode, active during speech and coughing, producing droplets ≥100 µm.6, 7 The oral cavity is among the sources of expiratory droplets, especially larger ones5, 8, 9; the droplet formation mechanism in the oral cavity is shown in Figure 1 . Large droplets from the trachea produced during coughing might not be released into the environment because they readily deposit within the head airways. Johnson et al6 found that droplets ≥20 µm only originate from the oral cavity. Droplets generated during breathing may originate from both the upper and lower airways, but the latter seems to make the major contribution because of the film rupture mechanism.12, 13
Fig 1.
Schematic diagram revealing the origin and generation mechanism of respiratory droplets. (A) Instability of the airway lining fluid10, (B) oral cavity model (drawn based on the atomization mechanism described in Morawska5), and (C) film rupture.11
Second, 2 major mechanisms exist for droplet formation in the respiratory tract (Fig 1). One is the instability caused by the shear stress on the mucus-air interface that leads to the avalanche of mucus and droplet formation. The biphasic airway lining fluid has an overall thickness ranging from 5-100 µm. A critical air speed is required to initiate the instability, which varies according to mucus thickness, its viscoelastic properties, and surface tension at the mucus-air interface. Coughing is one mechanism for mucus clearance, during which air speed as high as 200 m/s can be attained14 and interfacial shearing is peaked within the trachea.15 This mechanism has traditionally been considered an exhalation process during coughing and sneezing; however, it was found to be also plausible around the first bifurcations during inhalation.15, 16 Recent studies include the effect of viscoelastic properties and surface tension on the onset of instabilities (eg, Vasudevan and Lange17, 18) and the effect on the size distribution and volume concentration of bioaerosols produced.19
During normal tidal breathing, however, the shear force provided by the respiration airflow is not sufficient to induce instabilities. The mechanism for droplet formation during normal breathing relates to the reopening of collapsed terminal airways at the beginning of inspiration. Almstrand et al20 examined the production of exhaled particles after varying degrees of airway closure. Concentrations of exhaled particles showed a 2- to 18-fold increase after exhalations to residual volume, compared with exhalations where no airway closure was shown. Malashenko et al21 defined a critical capillary number (Ca = µU/σ, where µ is the dynamic viscosity of the liquid, U is the axial speed of the air-liquid meniscus propagation, and σ is the surface tension between the lining fluid and the air) above which droplets may be formed during normal breathing. In addition, experiments simulating the film droplet formation process showed that small fluid films generate droplets as efficiently as large films, and droplets may well be generated from films with diameters <1 mm (ie, the diameter of terminal bronchioles).22
Third, the reported number and size of released droplets vary significantly. In terms of the total mass of saliva, 1.1-6.7 mg of saliva were collected on a mask during a single cough, and 18.7 mg were collected while counting from 1-100.9, 23 There were 1-320 droplets per liter of exhaled air found for breathing, 24-23,600 found for coughing, and 4-600 found for speaking.7, 23, 24, 25, 26, 27, 28, 29, 30, 31
Several factors account for the significant inconsistencies between the existing data (eg, individual differences, imperfect measurement techniques, and effect of evaporation). Humidity level is crucial in the measurement of droplet and droplet nuclei sizes.29 Various measurement methods have been used, such as microscopic observations,8 optical particle counting,25 aerodynamic particle counting and scanning mobility particle sizing,29 interferometric Mie imaging,30 and laser aerosol particle spectrometry.31 Lindsley et al31 found that individuals infected with influenza virus produce a significantly greater volume of aerosol during clinical illness compared with during the asymptomatic stage (P = .0143). This enhancement in aerosol generation during illness may play an important role in influenza virus transmission.
Finally, we are interested in the quantity of pathogens in each size category of aerosols. The size of viruses varies from 0.02-0.3 µm, and the size of bacteria varies from 0.5-10 µm in their naked form. It is anticipated that small viral pathogens travel readily within the lungs and between individuals and their environment in small droplet nuclei. The influenza virus RNA detected by quantitative polymerase chain reaction in human exhaled breath suggests that it may be contained in fine particles generated during tidal breathing.32, 33 Lindsley et al34 measured the content of influenza virus in aerosol particles from human coughs. Thirty-five percent of the influenza RNA detected were contained in particles >4 µm in aerodynamic diameter, whereas 23% were contained in particles 1-4 µm and 42% were in particles <1 µm, suggesting that much of the viral RNA is contained within particles in the respirable size range. Mycobacterium tuberculosis, the causative agent of tuberculosis, has been reported in small droplet nuclei, and patients have generated bacteria-laden aerosols in a diameter range of 0.65-4.7 µm during coughing.35
Spread of droplets from the mouth and nose to the indoor environment
When the expiratory flow is weak during the full respiratory cycle, the body plume also plays a role. Weak expiratory flows (eg, those blocked by the use of a face mask) may be captured by the body plume (Fig 2 ). Expiratory flows are also where most droplet nuclei are formed under typical room conditions. Exhaled flow rate over time may be represented as a sinusoidal function for breathing, a constant for talking, and a combination of gamma probability distribution functions for coughing.36, 37 The peak velocities of coughing and breathing can be 6-22 m/s (>10 m/s on average) and 1-5 m/s, respectively.30, 36, 38 The differences in reported initial velocities are mainly attributed to different measurement techniques and individual variability.
Fig 2.
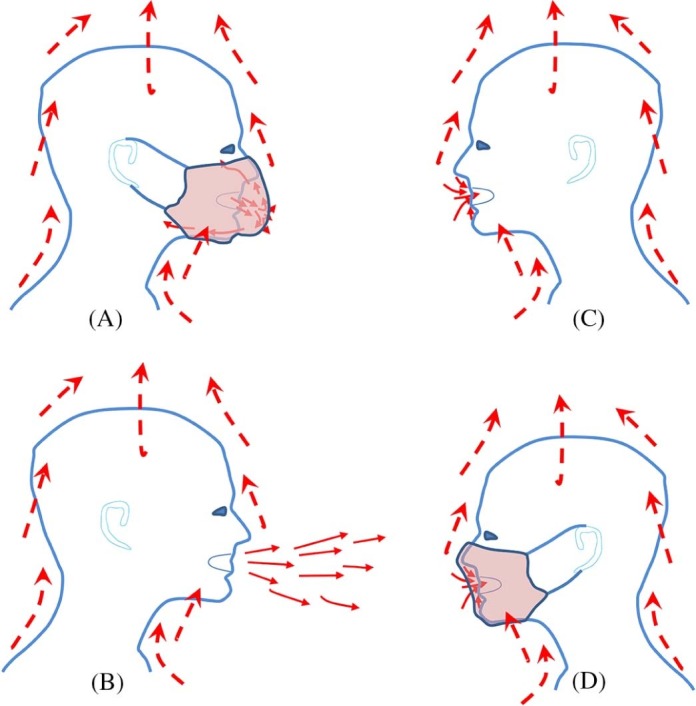
Escaped microbial aerosols of an infected individual with a mask (A) and without a mask (B) as affected by the body plume and inhalation of the airborne infectious agent(s) of a nearby individual without a mask (C) and with a mask (D).
Among all respiratory activities, coughing has probably been studied the most. The Schlieren technique using human volunteers reveals the turbulent cough jet with a leading vortex,39 and the cough has properties similar to a starting jet or puff.40 The vortex ring structure produced during coughing is also important in particle dispersion.4 An aerosol-laden jet, led by a characteristic vortex, can penetrate an impressive distance into the surrounding ambient air before finally mixing out.41 Xie et al42 found that expired droplets can travel 1.5-2 m. The presence of turbulence greatly enhances droplet spread.43
Water droplets with sizes on the order of 1 µm evaporate within a few milliseconds, water droplets of 10 µm survive for up to a few tenths of a second, and large droplets of 100 µm can survive for almost a minute. The transient process from a droplet to a droplet nucleus can be ignored when studying the movement of small droplets,44 whereas evaporation barely affects large droplets because they deposit soon after release. However, medium-sized droplets (eg, 50 µm in diameter) are most sensitive to humidity.43 Deposition occurs either by gravitational sedimentation, turbulent eddy impaction, or diffusional deposition. Large droplets were first defined as droplets >100 µm by Wells.45 In respiratory exhalation flows, the largest droplets that would completely evaporate before traveling 2 m are between 60 and 100 µm.42 These large droplets are carried >6 m by exhaled air at a velocity of 50 m/s (sneezing), >2 m at a velocity of 10 m/s (coughing), and <1 m at a velocity of 1 m/s (breathing). Particles of diameters 1-3 µm remained suspended almost indefinitely, whereas those with a diameter of 10 µm took 17 minutes to fall 3 m to the floor, those with a diameter of 20 µm took 4 minutes, and those with a diameter of 100 µm took 10 seconds.46 For small droplets, gravitational sedimentation is negligible; however, turbulent eddy impact or diffusional deposition may occur. Small droplets are subject to the ambient velocity field, and their evaporation clearly assists in transporting them a significant distance.47
Spread of droplets in the indoor environment
The transport of expiratory droplets can be considered in terms of 2 stages, with the primary being the expiratory flow, followed by secondary dispersion via room airflow. The airflow in buildings is typically designed to be <0.25 m/s on average for thermal comfort. Typical airflows are turbulent, and they are affected by many parameters, such as air distribution systems,48, 49, 50 room furniture setup, body thermal plumes,51, 52, 53 and human conversational behavior and activities.54, 55, 56 These influences are illustrated in Figure 3 . However, droplet size seems to be the most important factor affecting dispersion and deposition.5 Size of droplets affects their dispersion and deposition on surfaces and the survival of microorganisms within the droplets. Physical characteristics of the indoor environment, such as temperature and relative humidity and design of the ventilation system, are also important.5 The survival of pathogens inside the droplets is likewise subject to various environmental conditions and has been reviewed by Tang.58
Fig 3.
Droplet transport in an isolation room by expired airflow, thermal plume51, door vortices (adapted with permission from Elsever57), human walking54, 2-way buoyancy airflow, and ventilation airflow.
Here we specifically discuss the effect of human walking and body plumes. The airflow behind a human body is complex because of the complexity of body shape and movement. Generally, there are 2 distinct wake regions: an unsteady bluff-body wake behind the torso and a region of unsteady vortex shedding behind the legs, which is dominated by a jet of air formed between the legs.54 Asignificant downwash that occurs behind the body has the effect of laterally spreading the lower portions of the wake.59, 60
Using large eddy simulation, Choi and Edwards55, 61 found that backward transport (opposite to the direction of walking) can also occur because of downwash effects and tip vortex formation. Wake-induced transport of material in the direction of the walking motion continues because of inertial effects, even after the person stops. When the walking effect is combined with hinged door opening, the latter is the dominant transport mechanism, and human-induced wake motion enhances compartment-to-compartment transport. In addition, when isolation room air has a temperature different from that of the corridor, the 2-way airflow effect at the openings plays an important role in aerosol dispersal.62
The body thermal plume starts from the feet as a laminar flow and grows in both its velocity and thickness upward along the human body. It is important in the individual microenvironment and inhalation.51, 52, 53 The plume becomes fully turbulent at the middle chest level. It reaches a maximum velocity (0.2-0.3 m/s) approximately 0.5 m above the head. The thickness of the plume can reach 15 cm in the breathing zone, so airflow from the lower part of the human body is drawn into the mouth during inhalation, which makes up approximately two-thirds of total inhaled air. The total air flux in the plume is in the 20-35 L/s range.63 The rising thermal plume entrains and transports pollutants when the pollution source is on the floor, leading to a higher concentration in the microenvironment, particularly in the breathing zone of the standing or seated person, more so than in the ambient environment.64, 65 When the cough of a source patient penetrates the area around another person's lower body, the thermal plume can bring the fine droplet nuclei upward.
The thermal plume also can act as an air curtain to protect the person from the penetration of airflow expired by other people.66 In displacement ventilation, the reduction in plume buoyancy caused by stratification is substantial.63 In downward ventilation, the thermal plume can be preserved at head height if it meets the downward air at 0.25 m/s,50 which compromises the transport dominated by the thermal plume. Many factors influence the thermal plume (eg, gestures, clothing insulation, the blocking effect of a table, movement of people).51, 67, 68 When walking at a speed of >0.2 m/s, the effect of the thermal plume would give way to the human aerodynamic wake.54 It is worth mentioning that plumes induced by other heat sources also contribute to pollutant transport. In the smallpox outbreak in Meschede, Germany,69 a radiator in the index patient's room introduced an upward plume flow because of a partially open window, resulting in the spread of smallpox.
Exposure of susceptible hosts to respiratory droplets
A susceptible host can be close to a patient (eg, during conversation) or at a distance from a patient (eg, sharing the same classroom) but sitting sufficiently far away.
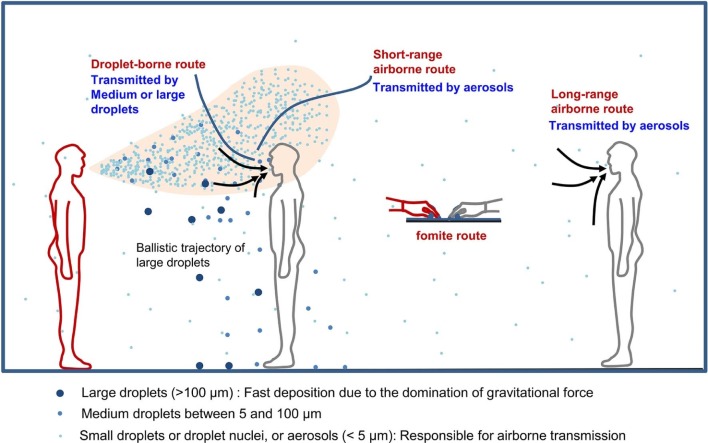
For 2 people in close contact, exposure can be caused by the direct spray route during which large droplets are deposited directly on the mucous membranes of the susceptible host (large droplet route) or by direct inhalation of fine droplets or droplet nuclei (airborne route). The latter is referred to as the short-range airborne route because exposure occurs when the 2 individuals are in close contact. For both the large droplet route and the short-range airborne route, expired droplets from the infected person can penetrate the thermal plume of the susceptible host, reaching the mucus or inhalation zone of the susceptible individual (Fig 2).66
When the susceptible individual is sufficiently far from an infected individual, direct inhalation of the contaminated room air is referred to as the airborne route. The infection risk of the susceptible host caused by inhaled droplets depends on the quantity of pathogen he or she carries and on the site at which the droplets deposit within the respiratory tract. Inhaled particles can deposit in different regions of the respiratory tract (eg, head airway region, tracheobronchial region, pulmonary region). Deposition mechanisms include inertial impaction (limited to large particles), settling (most important in small airways), Brownian motion of submicrometer particles, and interception.70 Recent studies on airflow and particle transport in the human respiratory tract were reviewed by Kleinstreuer and Zhang.71 Airflows are complex in the nasal cavities and oral airways; particles deposit largely at stagnation points, disrupting axial particle motion. According to the International Commission on Radiological Protection model72 for adults engaged in light work, total deposition is dominated by deposition in the head airways of particles >1 µm; the number of particles >10 µm that can penetrate the head airways is negligible.
We define 3 major routes of droplet exposure (Fig 4 ): the direct spray route, the long-range airborne route, and the fomite route, which is not discussed here. The direct spray route can be divided into 2 subroutes in terms of size and destination of the expiratory droplets and droplet nuclei: the short-range airborne route (<10 µm) and the droplet-borne route (>10 µm). This is basically in line with the definitions from the U.S. Centers for Disease Control and Prevention73; however, we distinguish the short-range and long-range airborne routes.
Fig 4.
Illustration of different transmission routes. Small droplets (<5 µm), sometimes called aerosols, are responsible for the short-range airborne route, long-range airborne route, and indirect contact route; large droplets are responsible for the direct spray route and indirect contact route.
The definition of the transmission route of a specific pathogen also must account for its virulence and infectious dose, and different modes are not mutually exclusive. The infectious dose of a pathogen is the number of microorganisms required to cause an infection. Data from research performed on biological warfare agents suggest that both bacteria and viruses can produce disease with as few as 1-100 infectious units (eg, brucellosis: 10-100 infectious units, Q fever: 1-10 infectious units, tularemia: 10-50 bacterial cells, smallpox: 10-100 infectious units, viral hemorrhagic fevers: 1-10 viral particles).74 There remains considerable controversy over the relative importance of the alternative modes of transmission of influenza virus. Brankston et al74 concluded in a review that natural influenza virus transmission in humans generally occurs over short distances, rather than over long distances, whereas Tellier75, 76 concluded that aerosol transmission occurs at appreciable rates. Weber and Stilianakis77 found that contact, large droplet, and small droplet (aerosol) transmission are all potentially important modes of transmission for influenza virus. Our purpose here is not to make conclusions about the relative importance of each route but to comment on the impact of airflows on the spread of infectious agents.
Relevance to infection control
Respiratory infection could be reduced or eliminated by interruptions in 3 phases: release of pathogen at the source, transport of pathogen by air or by surface touch, and protection of the susceptible person.
Prevention of droplet release at origin by saline inhalation
There are 2 ways of altering mucus properties.17, 18 The first is to lower the mucus viscosity and increase elasticity and surface tension for total suppression, and the second is to enlarge droplet size by decreasing the elasticity and surface tension and increasing the viscosity. The latter approach is preferred because the droplets generated would be smaller and more dangerous if full suppression was not achieved. Edwards et al26 found that delivering approximately 1 g of isotonic saline orally via nebulized aerosols (droplets 5.6 µm in diameter) reduced the total amount of expired aerosols (among super-producing individuals) by approximately 72% over a 6-hour period. In vitro tests using a simulated cough machine indicated that a mucus mimetic nebulized with saline produces a larger droplet size after the forced convection of air over its surface than when air is forced over the mucus mimetic alone (ie, without saline nebulization). In a subsequent study, Clark et al78 report that delivering isotonic saline aerosols (5.6-µm droplets) into the endotracheal tube of anesthetized bull calves showed a dose-responsive effect on exhaled bioaerosols; 6 minutes of treatment resulted in a decrease of up to 50% of exhaled aerosols for at least 120 minutes, compared with the pretreatment case. Inhaling safe surface-active materials, such as isotonic saline, to suppress exhaled bioaerosols was reviewed and recommended for controlling airborne transmission79; however, more studies are required to clearly elucidate the potential of this new approach.
Use of masks for infected individuals and for susceptible individuals
Two reviews80, 81 highlight the limited evidence base supporting the efficacy of face masks in reducing influenza virus transmission. They suggested that surgical masks may reduce infectiousness, rather than protect against infection, especially when airborne transmission is important. Influenza viruses (with sizes in the 80- to 120-nm range) and other viruses of similar size are capable of penetrating the mask in either direction. The N95 respirators are efficient in removing very fine droplet nuclei, but face masks are not. However, face masks, if worn by an infected person, can suppress the expired jets (Fig 2A) and reduce the close contact transmission via both the droplet-borne and short-range airborne routes.
Environmental ventilation for the long-range airborne route
A multidisciplinary systematic review49 suggested that ventilation rate and airflow patterns contribute directly to the airborne spread of infectious agents; however, the minimum ventilation rate for effective airborne transmission control is unknown at present. The current minimum requirement is 12 air changes per hour for negative-pressure airborne isolation rooms.82, 83 Natural ventilation may offer a low-cost alternative.83, 84 The current negative-pressure isolation rooms with a ceiling supply and bottom return system are recommended, but gaseous and fine particles were found to be removed more efficiently by ceiling-level exhausts, and large particles were removed mainly by deposition, rather than by ventilation.85 Displacement ventilation has been recommended as a more energy-efficient approach in nonhospital settings. However, in the case of the isolation room, the stable thermal stratification zone may cause the lock-up phenomenon to occur86 if the exhaled pollutant is not caught by the thermal plume penetrating into the upper zone, resulting in a longer residence time of pollutants.87 Displacement ventilation can create what might be referred to as inversion clouds in rooms. Because deposition is the main mechanism for removing large droplets,85 floor cleaning in hospitals is absolutely necessary.
Personalized ventilation for the short-range airborne route
This may be a less well-known technology in the infection control community. Its principle is based on detectable jets of air with a high momentum directed at a person's face.88, 89 It may not be effective when the mobility of the subject is considered. An air supply pillow was suggested for hospital use.90 The personalized ventilation (PV) system can be supplemented with a general ventilation system in the room. Experiments with PV, together with vertical ventilation from ceiling-mounted terminals, show increased efficiency of personal protection by a factor of up to 35.90 A combination of PV and the personalized exhaust method was suggested.91
Conclusions
By reviewing the airborne spread of infectious agents from mucus to mucus in the indoor environment, we have shown the opportunities for infection control at different stages of the spread. We propose that the short-range airborne route may be important in close contact, and its control may be achieved by face masks for the source patients and the use of PV. Our discussion of the effect of thermal stratification and expiratory delivery of droplets leads to the suggestion that displacement ventilation may not be applicable to hospital rooms where respiratory infection is a concern. The saline inhalation method was discussed after a discussion of the mechanisms of droplet formation and origin.
Footnotes
Funding/Support: This research was funded by the Research Grants Council General Research Fund (HKU7142/12) and the Natural Science Foundation of China (51278440). Publication of this supplement is primarily supported by RB, Montvale, New Jersey, with additional support from MicroBioTest, a division of Microbac Laboratories, Inc., Sterling, Virginia. The City University of New York (CUNY) and the University of Ottawa, Ottawa, Canada, are academic sponsors. Editorial support was provided by Ashley O'Dunne, PhD; Shannon O'Sullivan, ELS; and Alanna Franchetti, ELS of Medergy (Yardley, PA), and funded by RB.
Conflicts of Interest: None to report.
References
- 1.Yu I.T.S., Li Y., Wong T.W., Tam W., Chan A.T., Lee J.H. Evidence of airborne transmission of the severe acute respiratory syndrome virus. N Engl J Med. 2004;350:1731–1739. doi: 10.1056/NEJMoa032867. [DOI] [PubMed] [Google Scholar]
- 2.Cole E.C., Cook C.E. Characterization of infectious aerosols in health care facilities: an aid to effective engineering controls and preventive strategies. Am J Infect Control. 1998;26:453–464. doi: 10.1016/S0196-6553(98)70046-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Fitzgerald D., Haas D. Mycobacterium tuberculosis. In: Mandell G.L., Bennett J.E., Dolin R., editors. Principles and practice of infectious diseases. 6th ed. Churchill Livingstone; Philadelphia (PA): 2005. pp. 2852–2886. [Google Scholar]
- 4.Hunt J.C.R., Delfos R., Eames I., Perkins R.J. Vortices, complex flows and inertial particles. Flow Turbul Combust. 2007;79:207–234. [Google Scholar]
- 5.Morawska L. Droplet fate in indoor environments, or can we prevent the spread of infection? Indoor Air. 2006;16:335–347. doi: 10.1111/j.1600-0668.2006.00432.x. [DOI] [PubMed] [Google Scholar]
- 6.Johnson G.R., Morawska L., Ristovski Z.D., Hargreaves M., Mengersen K., Chao C.Y.H. Modality of human expired aerosol size distributions. J Aerosol Sci. 2011;42:839–851. doi: 10.1016/j.jaerosci.2008.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Morawska L., Johnson G.R., Ristovski Z.D., Hargreaves M., Mengersen K., Corbett S. Size distribution and sites of origin of droplets expelled from the human respiratory tract during expiratory activities. J Aerosol Sci. 2009;40:256–269. doi: 10.1016/j.jaerosci.2008.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Duguid J.P. The numbers and the sites of origin of the droplets expelled during expiratory activities. Edinburgh Med J. 1945;52:385–401. [PMC free article] [PubMed] [Google Scholar]
- 9.Xie X., Li Y., Sun H., Liu L. Exhaled droplets due to talking and coughing. J R Soc Interface. 2009;6(Suppl 6):S703–14. doi: 10.1098/rsif.2009.0388.focus. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Edwards D., Fiegel J., Dehaan W., Brande M., Man J., Clarke R. Novel inhalants for control and protection against airborne infections. Respir Drug Deliv. 2006;1:41–48. [Google Scholar]
- 11.Johnson G.R., Morawska L. The mechanism of breath aerosol formation. J Aerosol Med. 2009;22:229–237. doi: 10.1089/jamp.2008.0720. [DOI] [PubMed] [Google Scholar]
- 12.Almstrand A.C., Ljungström E., Lausmaa J., Bake B., Sjövall P., Olin A.C. Airway monitoring by collection and mass spectrometric analysis of exhaled particles. Anal Chem. 2008;81:662–668. doi: 10.1021/ac802055k. [DOI] [PubMed] [Google Scholar]
- 13.Fabian P., Brain J., Houseman E.A., Gern J., Milton D.K. Origin of exhaled breath particles from healthy and human rhinovirus-infected subjects. J Aerosol Med Pulm Drug Deliv. 2011;24:137–147. doi: 10.1089/jamp.2010.0815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ross B.B., Gramiak R., Rahn H. Physical dynamics of the cough mechanism. J Appl Physiol. 1955;8:264–268. doi: 10.1152/jappl.1955.8.3.264. [DOI] [PubMed] [Google Scholar]
- 15.Chowdhary R., Singh V., Tattersfield A.E., Sharma S.D., Kar S., Gupta A.B. Relationship of flow and cross-sectional area to frictional stress in airway models of asthma. J Asthma. 1999;36:419–426. doi: 10.3109/02770909909087284. [DOI] [PubMed] [Google Scholar]
- 16.Wang Y., Liu Y.X., Sun X.Z., Yu S., Gao F. Numerical analysis of respiratory flow patterns within human upper airway. Acta Mech Sin. 2009;25:737–746. [Google Scholar]
- 17.Vasudevan M., Lange C.F. Property dependence of onset of instability in viscoelastic respiratory fluids. Int J Eng Sci. 2005;43:1292–1298. [Google Scholar]
- 18.Vasudevan M., Lange C.F. Surface tension effects on instability in viscoelastic respiratory fluids. Math Biosci. 2007;205:180–194. doi: 10.1016/j.mbs.2006.08.009. [DOI] [PubMed] [Google Scholar]
- 19.Anwarul Hasan M.D., Lange C.F., King M.L. Effect of artificial mucus properties on the characteristics of airborne bioaerosol droplets generated during simulated coughing. J Nonnewton Fluid Mech. 2010;165:1431–1441. [Google Scholar]
- 20.Almstrand A.C., Bake B., Ljungström E., Larsson P., Bredberg A., Mirgorodskaya E. Effect of airway opening on production of exhaled particles. J Appl Phys. 2010;108:584–588. doi: 10.1152/japplphysiol.00873.2009. [DOI] [PubMed] [Google Scholar]
- 21.Malashenko A., Tsuda A., Haber S. Propagation and breakup of liquid menisci and aerosol generation in small airways. J Aerosol Med. 2009;22:341–353. doi: 10.1089/jamp.2008.0696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Holmgren H., Ljungstrom E. Influence of film dimensions on film droplet formation. J Aerosol Med. 2012;25:47–53. doi: 10.1089/jamp.2011.0892. [DOI] [PubMed] [Google Scholar]
- 23.Zhu S., Kato S., Yang J.-H. Study on transport characteristics of saliva droplets produced by coughing in a calm indoor environment. Build Environ. 2006;41:1691–1702. [Google Scholar]
- 24.Fairchild C.I., Stampfer J.F. Particle concentration in exhaled breath. Am Ind Hyg Assoc J. 1987;48:948–949. doi: 10.1080/15298668791385868. [DOI] [PubMed] [Google Scholar]
- 25.Papineni R.S., Rosenthal F.S. The size distribution of droplets in the exhaled breath of healthy human subjects. J Aerosol Med. 1997;10:105–116. doi: 10.1089/jam.1997.10.105. [DOI] [PubMed] [Google Scholar]
- 26.Edwards D.A., Man J.C., Brand P., Katstra J.P., Sommerer K., Stone H.A. Inhaling to mitigate exhaled bioaerosols. Proc Natl Acad Sci USA. 2004;101:17383–17388. doi: 10.1073/pnas.0408159101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Duguid J.P. The size and the duration of air-carriage of respiratory droplets and droplet-nuclei. J Hyg (Lond) 1946;44:471–479. doi: 10.1017/s0022172400019288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Loudon R.G., Roberts R.M. Cough frequency in patients with respiratory disease. Am Rev Respir Dis. 1967;96:1137–1143. doi: 10.1164/arrd.1967.96.6.1137. [DOI] [PubMed] [Google Scholar]
- 29.Yang S.H., Lee G.W.M., Chen C.M., Wu C.C., Yu K.P. The size and concentration of droplets generated by coughing in human subjects. J Aerosol Med. 2007;20:484–494. doi: 10.1089/jam.2007.0610. [DOI] [PubMed] [Google Scholar]
- 30.Chao C.Y.H., Wan M.P., Morawska L., Johnson G.R., Ristovski Z.D., Hargreaves M. Characterization of expiration air jets and droplet size distributions immediately at the mouth opening. J Aerosol Sci. 2009;40:122–133. doi: 10.1016/j.jaerosci.2008.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lindsley W.G., Pearce T.A., Hudnall J.B., Davis K.A., Davis S.M., Fisher M.A. Quantity and size distribution of cough-generated aerosol particles produced by influenza patients during and after illness. J Occup Environ Hyg. 2012;9:443–449. doi: 10.1080/15459624.2012.684582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Fabian P., McDevitt J.J., Dehaan W.H., Fung R.O.P., Cowling B.J., Chan K.H. Influenza virus in human exhaled breath: an observational study. PLoS ONE. 2008;3 doi: 10.1371/journal.pone.0002691. e2691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Milton D.K., Fabian M.P., Cowling B.J., Grantham M.L., McDevitt J.J. Influenza virus aerosols in human exhaled breath: particle size, culturability, and effect of surgical masks. PLoS Pathog. 2013;9:e1003205. doi: 10.1371/journal.ppat.1003205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lindsley W.G., Blachere F.M., Thewlis R.E., Vishnu A., Davis K.A., Cao G. Measurements of airborne influenza virus in aerosol particles from human coughs. PLoS ONE. 2010;5 doi: 10.1371/journal.pone.0015100. e15100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fennelly K.P., Martyny J.W., Fulton K.E., Orme I.M., Cave D.M., Heifets L.B. Cough-generated aerosols of Mycobacterium tuberculosis: a new method to study infectiousness. Am J Respir Crit Care Med. 2004;169:604–609. doi: 10.1164/rccm.200308-1101OC. [DOI] [PubMed] [Google Scholar]
- 36.Gupta J.K., Lin C.H., Chen Q. Flow dynamics and characterization of a cough. Indoor Air. 2009;19:517–525. doi: 10.1111/j.1600-0668.2009.00619.x. [DOI] [PubMed] [Google Scholar]
- 37.Gupta J.K., Lin C.H., Chen Q.Y. Characterizing exhaled airflow from breathing and talking. Indoor Air. 2010;20:31–39. doi: 10.1111/j.1600-0668.2009.00623.x. [DOI] [PubMed] [Google Scholar]
- 38.Kwon S.-B., Park J., Jang J., Cho Y., Park D.-S., Kim C. Study on the initial velocity distribution of exhaled air from coughing and speaking. Chemosphere. 2012;87:1260–1264. doi: 10.1016/j.chemosphere.2012.01.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tang J.W., Liebner T.J., Craven B.A., Settles G.S. A schlieren optical study of the human cough with and without wearing masks for aerosol infection control. J R Soc Interface. 2009;6(Suppl 6):S727–36. doi: 10.1098/rsif.2009.0295.focus. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Vansciver M., Miller S., Hertzberg J. Particle image velocimetry of human cough. Aerosol Sci Technol. 2011;45:415–422. [Google Scholar]
- 41.Settles G.S. Fluid mechanics and homeland security. Annu Rev Fluid Mech. 2006;38:87–110. [Google Scholar]
- 42.Xie X., Li Y., Chwang A.T.Y., Ho P.L., Seto W.H. How far droplets can move in indoor environments—revisiting the Wells evaporation–falling curve. Indoor Air. 2007;17:211–225. doi: 10.1111/j.1600-0668.2007.00469.x. [DOI] [PubMed] [Google Scholar]
- 43.Wei J., Li Y. Enhanced spread of expiratory droplets by turbulence in a cough jet. Build Environ. 2015;93:86–96. [Google Scholar]
- 44.Chen C., Zhao B. Some questions on dispersion of human exhaled droplets in ventilation room: answers from numerical investigation. Indoor Air. 2010;20:95–111. doi: 10.1111/j.1600-0668.2009.00626.x. [DOI] [PubMed] [Google Scholar]
- 45.Wells W.F. On air-borne infection study II. Droplets and droplet nuclei. Am J Epidemiol. 1934;20:611–618. [Google Scholar]
- 46.Knight V. Viruses as agents of airborne contagion. Ann NY Acad Sci. 1980;353:147–156. doi: 10.1111/j.1749-6632.1980.tb18917.x. [DOI] [PubMed] [Google Scholar]
- 47.Eames I., Tang J.W., Hunt J.C.R., Li Y. Murder, death and disease. Math Today. 2009;Feb:234–237. [Google Scholar]
- 48.Qian H., Li Y., Nielsen P.V., Hyldgaard C.E., Wong T.W., Chwang A.T. Dispersion of exhaled droplet nuclei in a two-bed hospital ward with three different ventilation systems. Indoor Air. 2006;16:111–128. doi: 10.1111/j.1600-0668.2005.00407.x. [DOI] [PubMed] [Google Scholar]
- 49.Li Y., Leung G.M., Tang J.W., Yang X., Chao C.Y., Lin J.Z. Role of ventilation in airborne transmission of infectious agents in the built environment—a multidisciplinary systematic review. Indoor Air. 2007;17:2–18. doi: 10.1111/j.1600-0668.2006.00445.x. [DOI] [PubMed] [Google Scholar]
- 50.Nielsen P.V. Control of airborne infectious diseases in ventilated spaces. J R Soc Interface. 2009;6(Suppl 6):S747–55. doi: 10.1098/rsif.2009.0228.focus. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Clark R.P., Edholm O.G. Arnold; London, England: 1985. Man and his thermal environment. [Google Scholar]
- 52.Murakami S. Analysis and design of micro-climate around the human body with respiration by CFD. Indoor Air. 2004;14:144–156. doi: 10.1111/j.1600-0668.2004.00283.x. [DOI] [PubMed] [Google Scholar]
- 53.Gao N.P., Niu J.L. CFD study of the thermal environment around a human body: a review. Indoor Built Environ. 2005;14:5–16. [Google Scholar]
- 54.Edge B.A., Paterson E.G., Settles G.S. Computational study of the wake and contaminant transport of a walking human. J Fluid Eng. 2005;127:967–977. [Google Scholar]
- 55.Choi J.I., Edwards J.R. Large-eddy simulation of human-induced contaminant transport in room compartments. Indoor Air. 2012;22:77–87. doi: 10.1111/j.1600-0668.2011.00741.x. [DOI] [PubMed] [Google Scholar]
- 56.Tang J.W., Eames I., Li Y., Taha Y.A., Wilson P., Bellingan G. Door-opening motion can potentially lead to a transient breakdown in negative-pressure isolation conditions: the importance of vorticity and buoyancy airflows. J Hosp Infect. 2005;61:283–286. doi: 10.1016/j.jhin.2005.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Tang J.W., Li Y., Eames I., Chan P.K.S., Ridgway G.L. Factors involved in the aerosol transmission of infection and control of ventilation in healthcare premises. J Hosp Infect. 2006;64:100–114. doi: 10.1016/j.jhin.2006.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Tang J.W. The effect of environmental parameters on the survival of airborne infectious agents. J R Soc Interface. 2009;6(Suppl 6):S737–46. doi: 10.1098/rsif.2009.0227.focus. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Poussou S.B., Mazumdar S., Plesniak M.W., Sojka P.E., Chen Q. Flow and contaminant transport in an airliner cabin induced by a moving body: model experiments and CFD predictions. Atmos Environ. 2010;44:2830–2839. [Google Scholar]
- 60.Hang J., Li Y., Ching W.H., Wei J., Jin R., Liu L. Potential airborne transmission between two isolation cubicles through a shared anteroom. Build Environ. 2015;89:264–278. doi: 10.1016/j.buildenv.2015.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Choi J.I., Edwards J.R. Large eddy simulation and zonal modeling of human-induced contaminant transport. Indoor Air. 2008;18:233–249. doi: 10.1111/j.1600-0668.2008.00527.x. [DOI] [PubMed] [Google Scholar]
- 62.Chen C., Zhao B., Yang X., Li Y. Role of two-way airflow owing to temperature difference in severe acute respiratory syndrome transmission: revisiting the largest nosocomial severe acute respiratory syndrome outbreak in Hong Kong. J R Soc Interface. 2011;8:699–710. doi: 10.1098/rsif.2010.0486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Craven B.A., Settles G.S. A computational and experimental investigation of the human thermal plume. J Fluid Eng. 2006;128:1251–1258. [Google Scholar]
- 64.Lewis H.E., Foster A.R., Mullan B.J., Cox R.N., Clark R.P. Aerodynamics of human microenvironment. Lancet. 1969;293:1273–1277. doi: 10.1016/s0140-6736(69)92220-x. [DOI] [PubMed] [Google Scholar]
- 65.Rim D., Novoselac A. Transport of particulate and gaseous pollutants in the vicinity of a human body. Build Environ. 2009;44:1840–1849. [Google Scholar]
- 66.Liu L. 2011. Expiratory droplet exposure between individuals in a ventilated room. PhD thesis; Hong Kong: The University of Hong Kong. [Google Scholar]
- 67.Zukowska D., Melikov A., Popiolek Z. Impact of personal factors and furniture arrangement on the thermal plume above a sitting occupant. Build Environ. 2012;49:104–116. [Google Scholar]
- 68.Spitzer I.M., Marr D.R., Glauser M.N. Impact of manikin motion on particle transport in the breathing zone. J Aerosol Sci. 2010;41:373–383. [Google Scholar]
- 69.Wehrle P.F., Posch J., Richter K.H., Henderson D.A. An airborne outbreak of smallpox in a German hospital and its significance with respect to other recent outbreaks in Europe. Bull World Health Organ. 1970;43:669–679. [PMC free article] [PubMed] [Google Scholar]
- 70.Hinds W.C. 2nd ed. Wiley; New York (NY): 1999. Aerosol technology: properties, behavior, and measurement of airborne particles. [Google Scholar]
- 71.Kleinstreuer C., Zhang Z. Annual Reviews; Palo Alto (CA): 2010. Airflow and particle transport in the human respiratory system. Annual review of fluid mechanics. [Google Scholar]
- 72.International Commission on Radiological Protection (ICRP) Elsevier Science; Tarrytown (NY): 1994. Human respiratory tract model for radiological protection, annals of the ICRP, publication 66. [PubMed] [Google Scholar]
- 73.Garner J.S. Hospital Infection Control Practices Advisory Committee. Guideline for isolation precautions in hospitals. Infect Control Hosp Epidemiol. 1996;17:53–80. doi: 10.1086/647190. [DOI] [PubMed] [Google Scholar]
- 74.Brankston G., Gitterman L., Hirji Z., Lemieux C., Gardam M. Transmission of influenza A in human beings. Lancet Infect Dis. 2007;7:257–265. doi: 10.1016/S1473-3099(07)70029-4. [DOI] [PubMed] [Google Scholar]
- 75.Tellier R. Review of aerosol transmission of influenza A virus. Emerg Infect Dis. 2006;12:1657–1662. doi: 10.3201/eid1211.060426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Tellier R. Aerosol transmission of influenza A virus: a review of new studies. J R Soc Interface. 2009;6(Suppl 6):S783–90. doi: 10.1098/rsif.2009.0302.focus. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Weber T.P., Stilianakis NI. Inactivation of influenza A viruses in the environment and modes of transmission: a critical review. J Infect. 2008;57:361–373. doi: 10.1016/j.jinf.2008.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Clark R.E.A., Katstra J., Man J.C., Dehaan W. Pulmonary delivery of anti-contagion aerosol to diminish exhaled bioaerosols and airborne infectious disease. Am J Infect Control. 2005;33:e85. [Google Scholar]
- 79.Fiegel J., Clarke R., Edwards D.A. Airborne infectious disease and the suppression of pulmonary bioaerosols. Drug Discov Today. 2006;11:51–57. doi: 10.1016/S1359-6446(05)03687-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Cowling B.J., Zhou Y., Ip D.K.M., Leung G.M., Aiello A.E. Face masks to prevent transmission of influenza virus: a systematic review. Epidemiol Infect. 2010;138:449–456. doi: 10.1017/S0950268809991658. [DOI] [PubMed] [Google Scholar]
- 81.Bin-Reza F., Lopez Chavarrias V., Nicoll A., Chamberland M.E. The use of masks and respirators to prevent transmission of influenza: a systematic review of the scientific evidence. Influenza Other Respir Viruses. 2012;6:257–267. doi: 10.1111/j.1750-2659.2011.00307.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Centers for Disease Control and Prevention (CDC) U.S. Department of Health and Human Services; Atlanta (GA): 2003. Guidelines for environmental infection control in health-care facilities. [Google Scholar]
- 83.World Health Organization (WHO) 2007. Infection prevention and control of epidemic- and pandemic-prone acute respiratory diseases in health care: WHO interim guidelines. Geneva, Switzerland: World Health Organization. [PubMed] [Google Scholar]
- 84.Escombe A.R., Oeser C.C., Gilman R.H., Navincopa M., Ticona E., Pan W. Natural ventilation for the prevention of airborne contagion. PLoS Med. 2007;4:309–317. doi: 10.1371/journal.pmed.0040068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Qian H., Li Y. Removal of exhaled particles by ventilation and deposition in a multibed airborne infection isolation room. Indoor Air. 2010;20:284–297. doi: 10.1111/j.1600-0668.2010.00653.x. [DOI] [PubMed] [Google Scholar]
- 86.Stymne H., Sandberg M., Mattsson M. The12th AIVC Conference “Air Movement and Ventilation Control within Buildings”. 1991. Dispersion pattern of contaminants in a displacement ventilated room – implications for demand control; pp. 173–189. Ottawa, Canada; September 24-27. [Google Scholar]
- 87.Li Y., Nielsen P.V., Sandberg M. Displacement ventilation in hospital environments. ASHRAE J. 2011;53:86–88. [Google Scholar]
- 88.Pantelic J., Sze-To G.N., Tham K.W., Chao C.Y.H., Khoo Y.C.M. Personalized ventilation as a control measure for airborne transmissible disease spread. J R Soc Interface. 2009;6(Suppl 6):S715–26. doi: 10.1098/rsif.2009.0311.focus. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Alain M., Kamel G., Nesreen G. A simplified combined displacement and personalized ventilation model. HVAC&R Res. 2012;18:737–749. [Google Scholar]
- 90.Nielsen P.V., Jiang H., Polak M. The 10th International Conference on Air Distribution in Rooms. 2007. Bed with integrated personalized ventilation for minimizing cross infection. Helsinki, Finland; 13-15 June. [Google Scholar]
- 91.Yang J., Sekhar S.C., Cheong K.W.D., Raphael B. Performance evaluation of a novel personalized ventilation–personalized exhaust system for airborne infection control. Indoor Air. 2015;25:176–187. doi: 10.1111/ina.12127. [DOI] [PubMed] [Google Scholar]