Abstract
Improved understanding of antigenic components and their interaction with the immune system, as supported by computational tools, permits a sophisticated approach to modern vaccine design. Vaccine platforms provide an effective tool by which strategically designed peptide and protein antigens are modularized to enhance their immunogenicity. These modular vaccine platforms can overcome issues faced by traditional vaccine manufacturing and have the potential to generate safe vaccines, rapidly and at a low cost. This review introduces two promising platforms based on virus-like particle and liposome, and discusses the methodologies and challenges.
Keywords: Virus-like particle, Liposome, Vaccine design, Modular, Platform technology
1. Advancing from traditional vaccine production
Vaccination continues to be a leading defense strategy against infectious pathogens. Traditional vaccines that employ whole-cell antigens to raise an immune response have been irrefutably successful in the control or localized eradication of diseases such as poliomyelitis, measles, mumps, rubella, influenza and hepatitis A and B [1], [2], [3]. Eradication of smallpox was declared in 1980 after a global immunization effort by WHO [4]. Rinderpest was the second disease globally eradicated by traditional vaccine means as declared by the World Organization for Animal Health in 2011 [5]. Despite this success, live attenuated and inactivated vaccines possess several major drawbacks. Both live attenuated and inactivated vaccines require the production of large volumes of pathogens in the form of viruses and bacteria. This lengthy culturing process contributes to a considerable lag time between antigen production and vaccine delivery. Furthermore, it demands specialized containment facilities and poses considerable risk to the operators and environment due to the infectious nature of the material [6], [7]. Despite adequate passaging to diminish virulence, live attenuated pathogens are capable of reverting to virulent strains as evidenced with simian immunodeficiency virus [8], African horse sickness [9] and infectious bronchitis virus vaccines [10]. The genuine threat of vaccine-derived polio associated with Sabin’s oral polio vaccine has hindered immunization programs worldwide [11], [12]. Inactivated polio vaccine has less of a biosafety risk to vaccine recipients as inactivated poliovirus is incapable of replication, thereby eliminating the possibility of vaccine-derived polio. However, inactivation of microorganisms can compromise the native conformation of antigenic epitopes resulting in reduced immunogenicity [13]. Pathogens that display high levels of antigenicity owing to high mutation rates (e.g. RNA viruses such as influenza and human immunodeficiency virus [14], [15]) or existing as multiple genotypes and serotypes (e.g. rotavirus [16], [17], enterovirus [18] and the Group A Streptococcus [19]) present a challenge for developing efficacious vaccines. While this is an important consideration for all vaccine manufacturing platforms, the current timescale of traditional vaccine manufacturing highlights their inadequacy.
Outbreaks of H1N1 influenza, Middle East Respiratory Syndrome, Ebola and Zika over the last decade, are timely reminders that improved modern vaccine technology is necessary to shorten the developmental and production time of vaccines. Vaccine platform technologies, the formulation of antigens of choice with a pre-defined platform base, have the potential to address vaccine manufacturing challenges such as speed, safety and efficacy. Platforms based on virus-like particle (VLP) and liposomes are discussed, with a focus on the challenges and opportunities offered by these vaccine platform technologies.
2. Modular vaccine approach
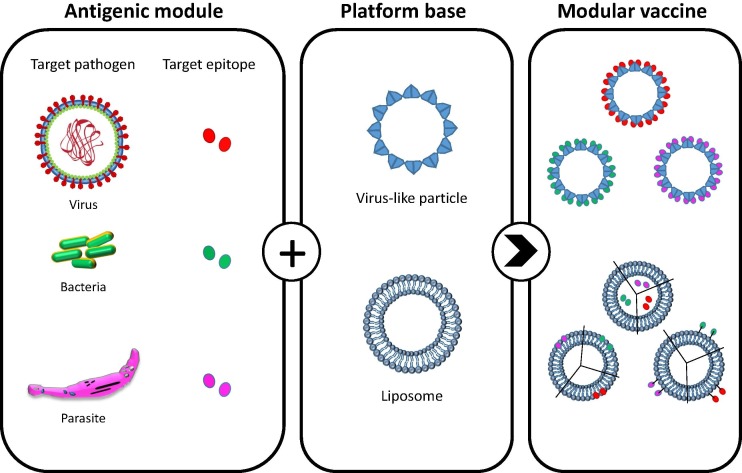
A tailorable platform that supports safe and simple manufacture of target antigens at high capacity has the potential to rapidly respond to an emerging disease. Most vaccine platform technologies consist of a platform base carrier (Fig. 1 ) that is amendable to modularization with target antigenic components of pathogens (known as modules). Independently, these components exhibit weak immunogenicity and poor stability. To harness the immunostimulatory properties of such antigens, platform carriers are engineered and developed to enhance the antigenicity but without the infectious trait of pathogens. Such engineering also allows the production of novel vaccine candidates that cannot be obtained through traditional methods (attenuation and inactivation). Basic research to determine suitable modules with antigenic potential is a prerequisite of this modular approach, yet the use of generic platforms supports streamlined and standardized vaccine development, potentially reducing the cost of development.
Fig. 1.
Modularization of target epitopes onto VLP and liposome vaccine platforms. Antigenic modules from a variety of microorganisms may be modularized onto the surface of VLPs through electrostatic interaction, chemical conjugation or genetic fusion. In liposomes, these antigenic modules may be encapsulated into the aqueous core, adsorbed into the lipid bilayer or conjugated (both covalently or non-covalently) to the vesicle surface.
A well-exploited platform is based on VLP technology. VLPs are highly ordered structures, with varying degrees of complexity, which stimulate both innate and adaptive immune responses [20], [21]. These intrinsic properties contributed to the commercialization of VLP-based vaccines against human papillomavirus (HPV), hepatitis B and E [22], [23], [24]. The self-adjuvanting properties of VLPs, due to their particulate structure and optimal size for uptake by antigen presenting cells [20], [25], makes them an attractive tool for increasing the immunogenicity of antigens. Antigens encapsulated within VLPs can also be used as vectors for drug delivery [26]. Well reported platforms based on self-assembling proteins include HPV L1 [27], Hepatitis B core [28] or surface antigen [29], [30], murine polyomavirus VP1 [31], [32] and bacteriophages MS2 [33], AP205 [34], [35] and Qβ [36]. High antigen-specific antibody titers and protective efficacies have been demonstrated across a range of peptide epitopes and protein domains modularized onto these VLP platforms. As reported, a pre-existing immunity against the VLP proteins from previous exposure to the platform does not diminish the immune response against the antigenic modules [37], [38]. Mosquirix™ (RTS,S/ASO1, GlaxoSmithKline), a protein-based malaria vaccine comprising circumsporozoite protein and Hepatitis B surface antigen, has demonstrated safety and protection in children and infants in a Phase III trial [39], and WHO has recently announced the first pilot studies in sub-Saharan Africa [40].
Liposomes are another favorable vaccine platform owing to their natural ability to induce an immune response [41]. Composed of an aqueous core and a uni- or multilamellar phospholipid bilayer, these lipid-based vesicles have immense adaptability and parameters with relation to size, charge, lipid, adjuvant composition and antigen presentation are manipulable [42]. As a result of this versatility, liposomal-based platforms are less well-defined than VLP-based platforms. Surface charge of the vesicle is reported to be an important factor that influences the immune response [42], [43], [44]. Cationic formulations are considered the most effective tools in liposomal antigen delivery due to their ability to bind antigen presenting cells through electrostatic interactions and form antigen depots at the site of injection [45], [46]. The combination of positively charged dimethyldioctadecylammonium (DDA) with the immunostimulant, trehalose-6,6-dibehenate (TDB) was engineered for the delivery of the tuberculosis antigen, Ag85B-ESAT-6 [45] and is possibly the best characterized. DDA:TDB is also considered as a potential platform for Chlamydia vaccines [47].
3. Vaccine design
The strategy for modularizing antigenic peptide or protein module onto the platform base is the key driver for inducing the protective immune response. Maintaining both the native conformational structure of the antigenic module post modularization and the integrity of the immunostimulating platform base are of equal importance. The rules to guide vaccine design are still limited. Although computational simulation tools and structure-based vaccine design are still in their infancy, they offer alternative possibilities to traditional empirical vaccine development [48], [49].
Modularization of chosen antigens onto VLPs is achieved through electrostatic interaction [50], chemical conjugation or genetic fusion [51]. Electrostatic interaction requires minimal processing but these non-covalent interactions can be weak and stability is questionable. A variety of linkage chemistries suitable for chemical conjugation result in a more permanent interaction albeit this requires more complex manufacturing processes under potentially harsh conditions that may alter protein structure. Permanent and regular module display is afforded through genetic fusion, eliminating downstream processing yet insertion sites for modules can place limitations on antigen size and may be incompatible with VLP assembly. Peptides are more amenable to VLP surface display than large protein domains although conformational structure can be compromised, ultimately affecting the quality of the immune response [52], [53], [54]. Displaying large protein domains has the added benefit of presenting multiple epitopes in the correct structure which may increase immunogenicity. However, expression of large genetically-fused antigens is a challenge owing to protein folding errors or compromised VLP formation through steric hindrance [55], [56]. To overcome these issues, strategies such as linker designs [56], antigen titration [38], [56], [57], split-intein conjugation [34], [58], [59] and a tandem core fusion strategy [60] are implemented to enable ease of large antigen modularization.
For liposomal vaccine platform, antigens can be encapsulated into the hydrophilic aqueous core [61], [62], intercalated into the lipid bilayer or surface attached [63]. Successful modularization of antigens up to 150 kDa have been reported [64], [65], [66], larger than those described for VLPs. Modularization with surface attached antigens often elicit superior immune responses in comparison to encapsulated antigens perhaps owing to intracellular processing which is possible for the latter [67]. Despite this, encapsulation protects antigens from protease degradation, facilitates longer circulation time and can generate effective immune responses [68], [69], [70]. Low encapsulation efficiency is common due to antigen loss from the vesicle during the manufacturing process which involves film extrusion and high sheer methods [71]. Incubating antigens with pre-formed liposomes in the presence of 30% v/v ethanol improves encapsulation efficiency [71], [72] and may aid a more streamlined manufacturing process whereby peptides can be encapsulated post-production. Unlike VLP technology, modules cannot be genetically fused to the carrier thus surface exposed antigens rely heavily upon bioconjugate technologies such as covalent conjugation (i.e. palmitoylation). Lipidation can compromise peptide conformation potentially resulting in altered immune responses [73]. Incorporating appropriate linkers between the module and the fatty acid to create spatial separation can address this [74], [75], [76]. As demonstrated by Lipotek Pty Ltd [77] and others [66], [78], the use of nitrilotriacetic acid (NTA) - histidine conjugation is promising, yet this remains a relatively unexplored area of liposome technology. NTA conjugation offers the opportunity of assembling entire protein domains [66] onto pre-formed liposomes whilst removing costly purification processes. Novel liposomal platforms encapsulate immunostimulants (including diphtheria toxoid and TLR9 agonists) independent of surface attached target antigen [76], [79]. This spatial segregation of antigenic components (with the immunostimulant exposed only upon intracellular processing) has been shown to enhance target specific immune responses. Table 1 summarizes manufacturing technologies for modularization.
Table 1.
Platform manufacturing technologies for modularization.
| Mechanism of Modularization | Advantages and Challenges | Platform | Disease | References |
|---|---|---|---|---|
| VLP – Molecular insertion | Simple molecular cloning Co-production of platform and module Reproducible module display Identification of insertion site Determination of suitable linkers Limitations on module size Steric hindrance with large modules |
Bacteriophage AP205 | Influenza (M2) | [84] |
| Cucumber Mosaic Virus | Alzheimer’s disease (Amyloid β) | [85] | ||
| Newcastle disease virus | [86] | |||
| Hepatitis B Core | Malaria (Circumsporozite) | [28] | ||
| Dengue virus type 2 (Envelope domain III) | [87] | |||
| Influenza (M2e) | [88] | |||
| Tuberculosis (CFP-10) | [89] | |||
| Human Papillomavirus L1 Capsid | Human respiratory syncytial virus | [27] | ||
| Murine Polyomavirus | Influenza (M2e) | [32] | ||
| Group A Streptococcus (J8) | [31] | |||
| Rotavirus (VP8*) | [38] | |||
| Tobacco mosaic virus | Poliovirus (type 3) | [90] | ||
| Foot-and-mouth disease | [91] | |||
| VLP – Conjugation | Conjugation of large modules without affecting VLP assembly Range of conjugation chemistries Quantification of conjugation efficiency Removal of unconjugated material Location of module dependent upon method of conjugation Harsh conditions alter epitope structure |
Bacteriophage AP250 | Malaria (Circumsporozite) | [34] |
| Malaria (Pfs25 / VAR2CSA), Tuberculosis (Ag58A) | [59] | |||
| Malaria (Pfs25 / CIDR) | [58] | |||
| Bacteriophage Qβ | Influenza (Hemagglutinin) | [92] | ||
| Hepatitis B Core | Influenza A (M2e) | [93] | ||
| Rabbit Haemorrhagic Disease Virus | Human papillomavirus type 16 (E6) | [94] | ||
| Liposome – Encapsulated | Module protected from proteases Longer circulation time Low encapsulation efficiency |
Cationic liposome | Leishmania | [61],[65] |
| Hepatitis E | [62] | |||
| Duck Tembusu virus | [70] | |||
| Liposome – Surface conjugation | Modularization possible on pre-formed liposomes Range of conjugation chemistries Harsh conditions alter epitope structure Determination of suitable linkers Removal of unconjugated material |
Cationic liposome | Human papillomavirus type 16 (E7) | [75] |
| DMPC-DMPG-cholesterol-MPLa | Human immunodeficiency virus type 1 (gp41) | [95] | ||
| Metallochelating liposome | Candida albicans (Heat shock protein 90) | [66] | ||
| Neutral liposome | Group A Streptococcus | [76] | ||
| Oleoyl liposome | Hepatitis C virus | [96] | ||
| Liposome – Adsorbed | Minimal preparation Lacks control of module orientation or display |
Cationic liposome | Tuberculosis (Ag85B-ESAT-6) | [97] |
| Cationic and neutral liposomes | Influenza (Hemagglutinin) | [98] | ||
DMPC, 1,2-dimyristoyl-sn-glycero-3-phosphocholine; DMPG, 1,2-dimyristoyl-sn-glycero-3-phosphorylglycerol; MPL, monophosphoryl lipid A.
4. Platform-based vaccine manufacturing
The long and complex vaccine development process (development, testing, regulatory) requires a huge investment of resources which includes time, facilities and money. Vaccine manufacturing processes are often customized and conducted in dedicated facilities for separate vaccines due to the characteristics of vaccine antigens and safety issues. These factors pose barriers for a fast response that is critical for controlling modern-day disease outbreaks that spread rapidly, as observed for H1N1 influenza in 2009 [80] and most recently Zika [81]. A platform approach for vaccine manufacturing ideally streamline the bioprocess, and shorten vaccine product development and delivery (time to market).
Platform technologies allow the standardization of upstream and downstream processes, given that the platform base remains unchanged. Certainly, processes will need optimization with modularization of different antigenic modules, but vaccine platform technologies provide flexibility and possibility for multi-product facilities. Prior knowledge, experience and production facility set-up is immensely beneficial. Merck Research Laboratories used their prior knowledge and know-hows from developing hepatitis B VLP vaccine (Recombivax) as the key decision factor when choosing to use the same host (Saccharomyces cerevisiae) for the production of HPV VLP vaccine (Gardasil) [82]. Similarly, the decision on the choice of adjuvant to formulate HPV VLP was made based on Recombivax.
The desire to lower cost of goods, thus leading to cheaper vaccines in the market has been well discussed and debated in papers and at conferences. The largest vaccine market is in developing countries, where vaccines would have a significant impact on public health, but these low-income countries face vaccine accessibility and affordability challenges. In combination with modular single-use technologies [83], modern vaccine manufacturing based on platform technologies may potentially lower capital and operating costs, resulting in affordable vaccines.
Another benefit of platform technologies is the potential reduction of regulatory burden. The level of proof and documentation required for new antigenic module on the generic platform may lessen as regulatory authorities are well informed by regulatory track records on the platform base. In the scenario of a disease outbreak, a close collaboration with regulatory authorities may lead to fast-track development of a safe and effective vaccine for the public, against an emerging pathogen.
The benefits of VLP and liposome platform technologies are many but perhaps the most significant is their potential to generate multivalent vaccines. Vaccines designed for immunization against multiple strains of an antigenically diverse pathogen are possible through display of different modules on a single platform or formulation of multiple platform products. Future work is expected to optimize the methodologies by which modules are incorporated into each platform to ensure the success of modern vaccines.
Acknowledgment
We acknowledge the funding support from the Australian Research Council (ARC Discovery Project DP160102915).
References
- 1.Cutts F.T., Lessler J., Metcalf C.J. Measles elimination: progress, challenges and implications for rubella control. Expert Rev Vaccines. 2013;12:917–932. doi: 10.1586/14760584.2013.814847. [DOI] [PubMed] [Google Scholar]
- 2.Roush S.W.M., Trudy V. Historical comparisons of morbidity and mortality for vaccine-preventable diseases in the United States. J Am Med Assoc. 2007;298:2155–2163. doi: 10.1001/jama.298.18.2155. [DOI] [PubMed] [Google Scholar]
- 3.Van Panhuis W.G.B., Shawn, Zadorozhny Vladimir, Lee Bruce Y., Eng Heather, Cross Anne. Contagious diseases in the United States from 18 88 to the present. New Engl J Med. 2013;369:2153. doi: 10.1056/NEJMms1215400. 6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Henderson D.A. The eradication of smallpox–an overview of the past, present, and future. Vaccine. 2011;29(Suppl 4):D7–D9. doi: 10.1016/j.vaccine.2011.06.080. [DOI] [PubMed] [Google Scholar]
- 5.Mariner J.H., House J.A., Mebus C.A., Sollod A.E., Chibeu D., Jones B.A. Rinderpest eradication: appropriate technology and social innovations. Sci Transl Med. 2012;337:1309–1312. doi: 10.1126/science.1223805. [DOI] [PubMed] [Google Scholar]
- 6.Uddowla S., Hollister J., Pacheco J.M., Rodriguez L.L., Rieder E. A safe foot-and-mouth disease vaccine platform with two negative markers for differentiating infected from vaccinated animals. J Virol. 2012;86:11675–11685. doi: 10.1128/JVI.01254-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Steel J., Lowen A.C., Pena L., Angel M., Solorzano A., Albrecht R. Live attenuated influenza viruses containing NS1 truncations as vaccine candidates against H5N1 highly pathogenic avian influenza. J Virol. 2009;83:1742–1753. doi: 10.1128/JVI.01920-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Whatmore A.C., Cook N., Hall G.A., Sharpe S., Rud E.W., Cranage M.P. Repair and evolution of nef in vivo modulates simian immunodeficiency virus virulence. J Virol. 1995;69:5117. doi: 10.1128/jvi.69.8.5117-5123.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Weyer C.T., Grewar J.D., Burger P., Rossouw E., Lourens C., Joone C. African horse sickness caused by genome reassortment and reversion to virulence of live, attenuated vaccine viruses, South Africa, 2004–2014. Emerg Infect Dis. 2016;22:2087–2096. doi: 10.3201/eid2212.160718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhang Y., Wang H.N., Wang T., Fan W.Q., Zhang A.Y., Wei K. Complete genome sequence and recombination analysis of infectious bronchitis virus attenuated vaccine strain H120. Virus Genes. 2010;41:377–388. doi: 10.1007/s11262-010-0517-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Nathanson N., Kew O.M. From emergence to eradication: the epidemiology of poliomyelitis deconstructed. Am J Epidemiol. 2010;172:1213–1229. doi: 10.1093/aje/kwq320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bandyopadhyay A.G., Garon J., Seib K., Orenstein W.a. Polio vaccination: past, present and future. Future Microbiol. 2015;10:791–808. doi: 10.2217/fmb.15.19. [DOI] [PubMed] [Google Scholar]
- 13.Fan Y.C., Chiu H.C., Chen L.K., Chang G.J., Chiou S.S. Formalin inactivation of japanese encephalitis virus vaccine alters the antigenicity and immunogenicity of a neutralization epitope in envelope protein domain III. PLoS Negl Trop Dis. 2015;9:e0004167. doi: 10.1371/journal.pntd.0004167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Treanor J. Influenza vaccine — outmaneuvering antigenic shift and drift. New Engl J Med. 2004;350:218–220. doi: 10.1056/NEJMp038238. [DOI] [PubMed] [Google Scholar]
- 15.Lipsitch M., O'Hagan J.J. Patterns of antigenic diversity and the mechanisms that maintain them. J R Soc Interface. 2007;4:787–802. doi: 10.1098/rsif.2007.0229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Miles M.G., Lewis K.D., Kang G., Parashar U.D., Steele A.D. A systematic review of rotavirus strain diversity in India, Bangladesh, and Pakistan. Vaccine. 2012;30(Suppl 1):A131–A139. doi: 10.1016/j.vaccine.2011.10.002. [DOI] [PubMed] [Google Scholar]
- 17.Chung J.Y., Kim M.S., Jung T.W., Kim S.J., Kang J.H., Han S.B. Detection of rotavirus genotypes in Korea 5 years after the introduction of rotavirus vaccines. J Korean Med Sci. 2015;30:1471–1475. doi: 10.3346/jkms.2015.30.10.1471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Xu M., Su L., Cao L., Zhong H., Dong N., Dong Z. Genotypes of the enterovirus causing hand foot and mouth disease in Shanghai, China, 2012–2013. PLoS ONE. 2015;10:e0138514. doi: 10.1371/journal.pone.0138514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Steer A.C., Carapetis J.R., Dale J.B., Fraser J.D., Good M.F., Guilherme L. Status of research and development of vaccines for Streptococcus pyogenes. Vaccine. 2016;34:2953–2958. doi: 10.1016/j.vaccine.2016.03.073. [DOI] [PubMed] [Google Scholar]
- 20.Keller S.A.B., Bauer Monika, Manolova Vania, Muntwiler Simone, Saudan Philippe, Bachmann Martin F. Cutting edge: limited specialization of dendritic cell subsets for MHC class II-associated presentation of viral particles. J Immunol. 2010;184:26–29. doi: 10.4049/jimmunol.0901540. [DOI] [PubMed] [Google Scholar]
- 21.Boisgerault F., Moron G., Leclerc C. Virus-like particles: a new family of delivery systems. Expert Rev Vaccines. 2002;1:101–109. doi: 10.1586/14760584.1.1.101. [DOI] [PubMed] [Google Scholar]
- 22.Kushnir N., Streatfield S.J., Yusibov V. Virus-like particles as a highly efficient vaccine platform: diversity of targets and production systems and advances in clinical development. Vaccine. 2012;31:58–83. doi: 10.1016/j.vaccine.2012.10.083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pils S., Joura E.A. From the monovalent to the nine-valent HPV vaccine. Clin Microbiol Infect. 2015;21:827–833. doi: 10.1016/j.cmi.2015.05.001. [DOI] [PubMed] [Google Scholar]
- 24.Lua L.H., Connors N.K., Sainsbury F., Chuan Y.P., Wibowo N., Middelberg A.P. Bioengineering virus-like particles as vaccines. Biotechnol Bioeng. 2014;111:425–440. doi: 10.1002/bit.25159. [DOI] [PubMed] [Google Scholar]
- 25.Manolova V.F., Flace Anna, Bauer Monika, Schwarz Katrin, Saudan Philippe, Bachmann Martin F. Nanoparticles target distinct dendritic cell populations according to their size. Eur J Immunol. 2008;38:1404–1413. doi: 10.1002/eji.200737984. [DOI] [PubMed] [Google Scholar]
- 26.Zdanowicz M., Chroboczek J. Virus-like particles as drug delivery vectors. Acta Biochim Pol. 2016;63:469–473. doi: 10.18388/abp.2016_1275. [DOI] [PubMed] [Google Scholar]
- 27.Murata Y., Lightfoote P.M., Rose R.C., Walsh E.E. Antigenic presentation of heterologous epitopes engineered into the outer surface-exposed helix 4 loop region of human papillomavirus L1 capsomeres. Virol J. 2009;6:81. doi: 10.1186/1743-422X-6-81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sällberg M., Hughes J., Jones J., Phillips T.R., Milich D.R. A malaria vaccine candidate based on a Hepatitis B virus core platform. Intervirology. 2003;45:350–361. doi: 10.1159/000067928. [DOI] [PubMed] [Google Scholar]
- 29.Shchelkunov S.N., Salyaev R.K., Pozdnyakov S.G., Rekoslavskaya N.I., Nesterov A.E., Ryzhova T.S. Immunogenicity of a novel, bivalent, plant-based oral vaccine against hepatitis B and human immunodeficiency viruses. Biotech Lett. 2006;28:959–967. doi: 10.1007/s10529-006-9028-4. [DOI] [PubMed] [Google Scholar]
- 30.Ballou W.R. The development of the RTS, S malaria vaccine candidate: challenges and lessons. Parasite Immunol. 2009;31:492–500. doi: 10.1111/j.1365-3024.2009.01143.x. [DOI] [PubMed] [Google Scholar]
- 31.Middelberg A.P., Rivera-Hernandez T., Wibowo N., Lua L.H., Fan Y., Magor G. A microbial platform for rapid and low-cost virus-like particle and capsomere vaccines. Vaccine. 2011;29:7154–7162. doi: 10.1016/j.vaccine.2011.05.075. [DOI] [PubMed] [Google Scholar]
- 32.Wibowo N., Chuan Y.P., Lua L.H.L., Middelberg A.P.J. Modular engineering of a microbially-produced viral capsomere vaccine for influenza. Chem Eng Sci. 2013;103:12–20. [Google Scholar]
- 33.Fu Y., Li J. A novel delivery platform based on Bacteriophage MS2 virus-like particles. Virus Res. 2016;211:9–16. doi: 10.1016/j.virusres.2015.08.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Janitzek C.M., Matondo S., Thrane S., Nielsen M.A., Kavishe R., Mwakalinga S.B. Bacterial superglue generates a full-length circumsporozoite protein virus-like particle vaccine capable of inducing high and durable antibody responses. Malar J. 2016;15:545. doi: 10.1186/s12936-016-1574-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pastori C., Tudor D., Diomede L., Drillet A.S., Jegerlehner A., Rohn T.A. Virus like particle based strategy to elicit HIV-protective antibodies to the alpha-helic regions of gp41. Virology. 2012;431:1–11. doi: 10.1016/j.virol.2012.05.005. [DOI] [PubMed] [Google Scholar]
- 36.Bessa J.S., Schmitz Nicole, Hinton Heather J., Schwarz Katrin, Jegerlehner Andrea, Bachmann Martin F. Efficient induction of mucosal and systemic immune responses by virus‐like particles administered intranasally: implications for vaccine design. Eur J Immunol. 2008;38:114–126. doi: 10.1002/eji.200636959. [DOI] [PubMed] [Google Scholar]
- 37.Chuan Y.P., Rivera-Hernandez T., Wibowo N., Connors N.K., Wu Y., Hughes F.K. Effects of pre-existing anti-carrier immunity and antigenic element multiplicity on efficacy of a modular virus-like particle vaccine. Biotechnol Bioeng. 2013;110:2343–2351. doi: 10.1002/bit.24907. [DOI] [PubMed] [Google Scholar]
- 38.Tekewe A., Fan Y., Tan E., Middelberg A.P., Lua L.H. Integrated molecular and bioprocess engineering for bacterially produced immunogenic modular virus-like particle vaccine displaying 18 kDa rotavirus antigen. Biotechnol Bioeng. 2017;114:397–406. doi: 10.1002/bit.26068. [DOI] [PubMed] [Google Scholar]
- 39.Penny M.A., Verity R., Bever C.A., Sauboin C., Galactionova K., Flasche S. Public health impact and cost-effectiveness of the RTS, S/AS01 malaria vaccine: a systematic comparison of predictions from four mathematical models. Lancet. 2016;387:367–375. doi: 10.1016/S0140-6736(15)00725-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.WHO. <http://www.who.int/immunization/research/development/malaria_vaccine_qa/en/>; November 2016.
- 41.Allison A.C., Gregoriadis G. Liposomes as immunological adjuvants. Nature. 1974;252:252. doi: 10.1038/252252a0. [DOI] [PubMed] [Google Scholar]
- 42.Watson D.S., Endsley A.N., Huang L. Design considerations for liposomal vaccines: influence of formulation parameters on antibody and cell-mediated immune responses to liposome associated antigens. Vaccine. 2012;30:2256–2272. doi: 10.1016/j.vaccine.2012.01.070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kraaijeveld C.A.S., Schilham M., Jansen J., Benaissa-Trouw B., Harmsen M., Van Houte A.J. The effect of liposomal charge on the neutralizing antibody response against inactivated encephalomyocarditis and Semliki Forest viruses. Clin Exp Immunol. 1984;56:509–514. [PMC free article] [PubMed] [Google Scholar]
- 44.Nakanishi T.K., Kunisawa Jun, Hayashi Akira, Tsutsumi Yasuo, Kubo Kazuyoshi, Nakagawa Shinsaku. Positively charged liposome functions as an efficient immunoadjuvant in inducing cell-mediated immune response to soluble proteins. J Control Release. 1999;61:233–240. doi: 10.1016/s0168-3659(99)00097-8. [DOI] [PubMed] [Google Scholar]
- 45.Christensen D., Agger E.M., Andreasen L.V., Kirby D., Andersen P., Perrie Y. Liposome-based cationic adjuvant formulations (CAF): past, present, and future. J Liposome Res. 2009;19:2–11. doi: 10.1080/08982100902726820. [DOI] [PubMed] [Google Scholar]
- 46.Tandrup Schmidt S., Foged C., Korsholm K.S., Rades T., Christensen D. Liposome-based adjuvants for subunit vaccines: formulation strategies for subunit antigens and immunostimulators. Pharmaceutics. 2016;8 doi: 10.3390/pharmaceutics8010007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Yu H., Jiang X., Shen C., Karunakaran K.P., Jiang J., Rosin N.L. Chlamydia muridarum T-cell antigens formulated with the adjuvant DDA/TDB induce immunity against infection that correlates with a high frequency of gamma interferon (IFN-gamma)/tumor necrosis factor alpha and IFN-gamma/interleukin-17 double-positive CD4+ T cells. Infect Immun. 2010;78:2272–2282. doi: 10.1128/IAI.01374-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.He L., Zhu J. Computational tools for epitope vaccine design and evaluation. Curr Opin Virol. 2015;11:103–112. doi: 10.1016/j.coviro.2015.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Park M.S., Park S.Y., Miller K.R., Collins E.J., Lee H.Y. Accurate structure prediction of peptide-MHC complexes for identifying highly immunogenic antigens. Mol Immunol. 2013;56:81–90. doi: 10.1016/j.molimm.2013.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gleiter S.S., Stubenrauch Kay, Lilie Hauke. Changing the surface of a virus shell fusion of an enzyme to polyoma VP1. Protein Sci. 1999;8:2562–2569. doi: 10.1110/ps.8.12.2562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Peacey M., Wilson S., Baird M.A., Ward V.K. Versatile RHDV virus-like particles: incorporation of antigens by genetic modification and chemical conjugation. Biotechnol Bioeng. 2007;98:968–977. doi: 10.1002/bit.21518. [DOI] [PubMed] [Google Scholar]
- 52.Anggraeni M.R., Connors N.K., Wu Y., Chuan Y.P., Lua L.H., Middelberg A.P. Sensitivity of immune response quality to influenza helix 190 antigen structure displayed on a modular virus-like particle. Vaccine. 2013;31:4428–4435. doi: 10.1016/j.vaccine.2013.06.087. [DOI] [PubMed] [Google Scholar]
- 53.Scarselli M.A., Aricò Beatrice, Brunelli Brunella, Savino Silvana, Di Marcello Federica, Palumbo Emmanuelle. Rational design of a meningococcal antigen inducing broad protective immunity. Sci Transl Med. 2011;3:91ra62. doi: 10.1126/scitranslmed.3002234. [DOI] [PubMed] [Google Scholar]
- 54.Dormitzer P.R., Grandi G., Rappuoli R. Structural vaccinology starts to deliver. Nat Rev Microbiol. 2012;10:807–813. doi: 10.1038/nrmicro2893. [DOI] [PubMed] [Google Scholar]
- 55.Chackerian B. Virus-like particles: flexible platforms for vaccine development. Expert Rev Vaccines. 2007;6:381–390. doi: 10.1586/14760584.6.3.381. [DOI] [PubMed] [Google Scholar]
- 56.Lua L.H., Fan Y., Chang C., Connors N.K., Middelberg A.P. Synthetic biology design to display an 18 kDa rotavirus large antigen on a modular virus-like particle. Vaccine. 2015;33:5937–5944. doi: 10.1016/j.vaccine.2015.09.017. [DOI] [PubMed] [Google Scholar]
- 57.Cohen J., Nussenzweig V., Vekemans J., Leach A. From the circumsporozoite protein to the RTS,S/AS candidate vaccine. Human Vaccines. 2010;6:90–96. doi: 10.4161/hv.6.1.9677. [DOI] [PubMed] [Google Scholar]
- 58.Brune K.D., Leneghan D.B., Brian I.J., Ishizuka A.S., Bachmann M.F., Draper S.J. Plug-and-Display: decoration of Virus-Like Particles via isopeptide bonds for modular immunization. Sci Rep. 2016;6:19234. doi: 10.1038/srep19234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Thrane S., Janitzek C.M., Matondo S., Resende M., Gustavsson T., de Jongh W.A. Bacterial superglue enables easy development of efficient virus-like particle based vaccines. J Nanobiotechnol. 2016;14:30. doi: 10.1186/s12951-016-0181-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Peyret H., Gehin A., Thuenemann E.C., Blond D., El Turabi A., Beales L. Tandem fusion of hepatitis B core antigen allows assembly of virus-like particles in bacteria and plants with enhanced capacity to accommodate foreign proteins. PLoS ONE. 2015;10:e0120751. doi: 10.1371/journal.pone.0120751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ravindran R., Maji M., Ali N. Vaccination with liposomal leishmanial antigens adjuvanted with monophosphoryl lipid-trehalose dicorynomycolate (MPL-TDM) confers long-term protection against visceral leishmaniasis through a human administrable route. Mol Pharm. 2012;9:59–70. doi: 10.1021/mp2002494. [DOI] [PubMed] [Google Scholar]
- 62.Kulkarni S.P., Thanapati S., Arankalle V.A., Tripathy A.S. Specific memory B cell response and participation of CD4+ central and effector memory T cells in mice immunized with liposome encapsulated recombinant NE protein based Hepatitis E vaccine candidate. Vaccine. 2016;34:5895–5902. doi: 10.1016/j.vaccine.2016.10.046. [DOI] [PubMed] [Google Scholar]
- 63.Bobbala S., Hook S. Is there an optimal formulation and delivery strategy for subunit vaccines? Pharm Res. 2016;33:2078–2097. doi: 10.1007/s11095-016-1979-0. [DOI] [PubMed] [Google Scholar]
- 64.Davis D., Gregoriadis G. Liposomes as adjuvants with immunopurified tetanus toxoid: Influence of liposomal characteristics. Immunology. 1987;61:229–234. [PMC free article] [PubMed] [Google Scholar]
- 65.Nagill R., Kaur S. Enhanced efficacy and immunogenicity of 78 kDa antigen formulated in various adjuvants against murine visceral leishmaniasis. Vaccine. 2010;28:4002–4012. doi: 10.1016/j.vaccine.2010.01.015. [DOI] [PubMed] [Google Scholar]
- 66.Mašek J., Bartheldyová E., Turánek-Knotigová P., Škrabalová M., Korvasová Z., Plocková J. Metallochelating liposomes with associated lipophilised norAbuMDP as biocompatible platform for construction of vaccines with recombinant His-tagged antigens: Preparation, structural study and immune response towards rHsp90. J Control Release. 2011;151:193–201. doi: 10.1016/j.jconrel.2011.01.016. [DOI] [PubMed] [Google Scholar]
- 67.Rao M., Wassef N.M., Alving C.R., Krzych U. Intracellular processing of liposome-encapsulated antigens by macrophages depends upon the antigen. Infect Immun. 1995;63:2396–2402. doi: 10.1128/iai.63.7.2396-2402.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Taki A., Smooker P. Small wonders-the use of nanoparticles for delivering antigen. Vaccines (Basel) 2015;3:638–661. doi: 10.3390/vaccines3030638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Teng X., Tian M., Li J., Tan S., Yuan X., Yu Q. Immunogenicity and protective efficacy of DMT liposome-adjuvanted tuberculosis subunit CTT3H vaccine. Hum Vaccin Immunother. 2015;11:1456–1464. doi: 10.1080/21645515.2015.1037057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ma T., Liu Y., Cheng J., Liu Y., Fan W., Cheng Z. Liposomes containing recombinant E protein vaccine against duck Tembusu virus in ducks. Vaccine. 2016;34:2157–2163. doi: 10.1016/j.vaccine.2016.03.030. [DOI] [PubMed] [Google Scholar]
- 71.Shariat S.B., Badiee Ali, Jaafari Mahmoud Reza, Mortazavi Seyed Alireza. Optimization of a method to prepare liposomes containing HER2/Neu- derived peptide as a vaccine delivery system for breast cancer. Iran J Pharm Res. 2014;13:15–25. [PMC free article] [PubMed] [Google Scholar]
- 72.Wang C.H., Huang Y.Y. Encapsulating protein into preformed liposomes by ethanol-destabilized method. Artif Cells Blood Sub, Biotechnol. 2003;31:303–312. doi: 10.1081/bio-120023160. [DOI] [PubMed] [Google Scholar]
- 73.Hickman D.T., Lopez-Deber M.P., Ndao D.M., Silva A.B., Nand D., Pihlgren M. Sequence-independent control of peptide conformation in liposomal vaccines for targeting protein misfolding diseases. J Biol Chem. 2011;286:13966–13976. doi: 10.1074/jbc.M110.186338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Muhs A.H., Hickman David T., Pihlgren Maria, Chuard Nathalie, Giriens Valerie, Meerschman Carine. Liposomal vaccines with conformation-specific amyloid peptide antigens define immune response and efficacy in APP transgenic mice. Proc Natl Acad Sci U S A. 2007;104:9810. doi: 10.1073/pnas.0703137104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Chen W., Huang L. Induction of cytotoxic T-lymphocytes and antitumor activity by a liposomal lipopeptide vaccine. Mol Pharm. 2008;5:464–471. doi: 10.1021/mp700126c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Zaman M., Ozberk V., Langshaw E.L., McPhun V., Powell J.L., Phillips Z.N. Novel platform technology for modular mucosal vaccine that protects against streptococcus. Sci Rep. 2016;6:39274. doi: 10.1038/srep39274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Tyne A.S., Chan J.G., Shanahan E.R., Atmosukarto I., Chan H.K., Britton W.J. TLR2-targeted secreted proteins from Mycobacterium tuberculosis are protective as powdered pulmonary vaccines. Vaccine. 2013;31:4322–4329. doi: 10.1016/j.vaccine.2013.07.022. [DOI] [PubMed] [Google Scholar]
- 78.Marques-Gallego P., de Kroon A.I. Ligation strategies for targeting liposomal nanocarriers. Biomed Res Int. 2014;2014:129458. doi: 10.1155/2014/129458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Hills T., Jakeman P.G., Carlisle R.C., Klenerman P., Seymour L.W., Cawood R. A rapid-response humoral vaccine platform exploiting pre-existing non-cognate populations of anti-vaccine or anti-viral CD4+ T helper cells to confirm B cell activation. PLoS ONE. 2016;11:e0166383. doi: 10.1371/journal.pone.0166383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Sullivan S.J., Jacobson R.M., Dowdle W.R., Poland G.A. 2009 H1N1 influenza. Mayo Clin Proc. 2010;85:64–76. doi: 10.4065/mcp.2009.0588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Singer M. The spread of Zika and the potential for global arbovirus syndemics. Glob Public Health. 2017;12:1–18. doi: 10.1080/17441692.2016.1225112. [DOI] [PubMed] [Google Scholar]
- 82.Buckland B.C. The process development challenge for a new vaccine. Nat Med. 2005 doi: 10.1038/nm1218. [DOI] [PubMed] [Google Scholar]
- 83.Lopes A.G. Single-use in the biopharmaceutical industry: a review of current technology impact, challenges and limitations. Food Bioprod Process. 2015;93:98–114. [Google Scholar]
- 84.Tissot A.C., Renhofa R., Schmitz N., Cielens I., Meijerink E., Ose V. Versatile virus-like particle carrier for epitope based vaccines. PLoS ONE. 2010;5:e9809. doi: 10.1371/journal.pone.0009809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Vitti A., Piazzolla G., Condelli V., Nuzzaci M., Lanorte M.T., Boscia D. Cucumber mosaic virus as the expression system for a potential vaccine against Alzheimer's disease. J Virol Methods. 2010;169:332–340. doi: 10.1016/j.jviromet.2010.07.039. [DOI] [PubMed] [Google Scholar]
- 86.Zhao Y., Hammond R.W. Development of a candidate vaccine for Newcastle disease virus by epitope display in the Cucumber mosaic virus capsid protein. Biotech Lett. 2005;27:375–382. doi: 10.1007/s10529-005-1773-2. [DOI] [PubMed] [Google Scholar]
- 87.Arora U.T., Tyagi Poornima, Swaminathan Sathyamangalam, Khanna Navin. Chimeric Hepatitis B core antigen virus-like particles displaying the envelope domain III of dengue virus type 2<Arora 20 12 .pdf>. J Nanobiotechnol. 2012;10 doi: 10.1186/1477-3155-10-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.De Filette M., Martens W., Smet A., Schotsaert M., Birkett A., Londono-Arcila P. Universal influenza A M2e-HBc vaccine protects against disease even in the presence of pre-existing anti-HBc antibodies. Vaccine. 2008;26:6503–6507. doi: 10.1016/j.vaccine.2008.09.038. [DOI] [PubMed] [Google Scholar]
- 89.Dhanasooraj D., Kumar R.A., Mundayoor S. Vaccine delivery system for tuberculosis based on nano-sized hepatitis B virus core protein particles. Int J Nanomedicine. 2013;8:835–843. doi: 10.2147/IJN.S40238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Haynes J.R.C., Cunningham Janet, Von Seefried Adolph, Lennick Michael, Garvin Robert T., Shen Shi-Hsiang. Development of a genetically–engineered, candidate polio vaccine employing the self–assembling properties of the tobacco mosaic virus coat protein. Bio/Technology. 1986;4:637. doi: 10.1038/nbt0786-637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Wu L. Expression of foot-and-mouth disease virus epitopes in tobacco by a tobacco mosaic virus-based vector∗1. Vaccine. 2003;21:4390–4398. doi: 10.1016/s0264-410x(03)00428-6. [DOI] [PubMed] [Google Scholar]
- 92.Jegerlehner A., Zabel F., Langer A., Dietmeier K., Jennings G.T., Saudan P. Bacterially produced recombinant influenza vaccines based on virus-like particles. PLoS ONE. 2013;8:e78947. doi: 10.1371/journal.pone.0078947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Jegerlehner A., Schmitz N., Storni T., Bachmann M.F. Influenza a vaccine based on the extracellular domain of M2: weak protection mediated via antibody-dependent NK cell activity. J Immunol. 2004;172:5598–5605. doi: 10.4049/jimmunol.172.9.5598. [DOI] [PubMed] [Google Scholar]
- 94.Jemon K., Young V., Wilson M., McKee S., Ward V., Baird M. An enhanced heterologous virus-like particle for human papillomavirus type 16 tumour immunotherapy. PLoS ONE. 2013;8:e66866. doi: 10.1371/journal.pone.0066866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Watson D.S., Platt V.M., Cao L., Venditto V.J., Szoka F.C., Jr. Antibody response to polyhistidine-tagged peptide and protein antigens attached to liposomes via lipid-linked nitrilotriacetic acid in mice. Clin Vaccine Immunol. 2011;18:289–297. doi: 10.1128/CVI.00425-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Takagi A., Kobayashi N., Taneichi M., Uchida T., Akatsuka T. Coupling to the surface of liposomes alters the immunogenicity of hepatitis C virus-derived peptides and confers sterile immunity. Biochem Biophys Res Commun. 2013;430:183–189. doi: 10.1016/j.bbrc.2012.11.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Hamborg M., Kramer R., Schante C.E., Agger E.M., Christensen D., Jorgensen L. The physical stability of the recombinant tuberculosis fusion antigens h1 and h56. J Pharm Sci. 2013;102:3567–3578. doi: 10.1002/jps.23669. [DOI] [PubMed] [Google Scholar]
- 98.Barnier-Quer C., Elsharkawy A., Romeijn S., Kros A., Jiskoot W. Adjuvant effect of cationic liposomes for subunit influenza vaccine: influence of antigen loading method, cholesterol and immune modulators. Pharmaceutics. 2013;5:392–410. doi: 10.3390/pharmaceutics5030392. [DOI] [PMC free article] [PubMed] [Google Scholar]