Summary
In this report, for surface display of viral antigen on lactobacilli, we have developed a surface antigen display system using the poly-γ-glutamate synthetase A protein (pgsA) of Bacillus subtilis as an anchoring matrix. Recombinant fusion proteins comprised of pgsA and neucleocapsid protein of PEDV were stably expressed in Lactobacillus casei. Surface location of fusion protein was verified by ELISA, immunofluoresence microscopy. Oral and intranasal inoculations of recombinant L. casei into pregnant sow and mice resulted in high levels of serum immunoglobuline G (IgG) and mucosal IgA, as demonstrated by rnELISA(recombinant N protein ELISA) using recombinant N protein. Absorbance of IgG in pregnant sow sera highly increased duration of the experiment. More importantly, the level of IgA in colostrum were increased significantly higher than that of IgG. The IgG levels of the piglets were increased after suckling colostrum secreted from sows previously inoculated recombinant L. casei. These results indicate that mucosal immunization with recombinant L. casei expressing PEDV N protein (neucleoprotein of PEDV) on its surface elicited high levels of mucosal IgA and circulation IgG immune responses against the antigen N of PEDV.
Keywords: Porcine epidemic diarrhea viral (PEDV), Lactic acid bacteria, N protein
Introduction
Bacterial cells present an attractive approach for recombinant vaccines because of their easiness of use and low costs of manufacturing and distribution. Displaying foreign antigens on the surface of bacteria may facilitate their recognition by the immune system, and mediate an immunoadjuvant effect with other bacterial surface components. The delivery of antigens to mucosal-associated lymphoid-tissues in paediatric and immune compromised populations by safe, non-invasive vectors, such as commensal lactobacilli, represents a crucial improvement to prevailing vaccination options [1]. However, most surface display systems for vaccine applications are limited in the size of foreign antigens they can display. In addition, surface display of antigens has been demonstrated. Coronaviruses are members of a family of RNA viruses that cause different pathologies in humans and animals. An important aspect of study is the finding of efficient antiviral strategies that could prevent the considerable economic losses caused by these infectious agents all over the world. Several viral proteins are important for inducing an immune response to coronaviruses: the spike protein (S), the membrane protein (M) and the nucleoprotein (N) [2]. The S glycoprotein of coronavirus envelope plays a crucial role in the early steps of infection. It mediates binding of the virus to the cell surface and the subsequent fusion between the viral and cell membranes [3]. Because the diseases caused by coronavirus continue to cause major economic losses to swine industry, and to date effective vaccines or treatments have remained elusive, there is a demand for the development of a new type of vaccine against PEDV.
For surface display of antigens on LAB(lactic acid bacteria), we have developed a expression vector using pgsA gene product as an anchoring matrix. PgsA is a synthetase complex (PGS system) of Bacillus subtilis [4], as a fusion partner for expression of heterologous antigens on the surface of L. casei525 [5]. The N gene of PEDV were inserted into this vector, and displayed on the surface of lactic acid bacteria, L. casei525. Intranasal and oral vaccination of mice and sow with the live and dead recombinant L. casei525 elicited high levels of systemic serum antibodies and local mucosal immunity against the antigens N of PEDV. The results of this study suggest a potential use for the surface expression system to construct the spike gene of swine coronaviruses and against other diarrhea pathogens and respiratory system diseases that are transmitted mucosally.
Materials and methods
Construction of minimal surface display vector
The minimal surface display plasmid, pHCE1LB-pgsA-PEDN, were constructed by PCR amplification using T7-pgsBCA, pGEM-T-PEDN as templates under control of HCE promoter, as described below. PCR-amplified 1026 bp DNA fragments with 5′CGCCATATGATGAAAAAAGAACTGAGCTTT3′ and 5′CGCGGATCCTTTAGATTTTAGTTTGTCACT3′ encoding the membrane protein pgsA was NdeI-BamHI digested and inserted into pHCE1LB (kindly provided by Dr. Jong-Soo Lee of BioLeaders Corp., Daejeon, Korea), creating plasmid pHCE1LB-pgsA. For construction of plasmid pHCE1LB-pgsA-PEDN, PCR-amplified 1326 bp DNA fragment with 5′CGCGGATCCGCTTCTGTCAGCTTT3′ and 5′CGCGGATCCTTAATTTCCTGTATCGAAG AT3′ encoding the nucleocapsid protein of PEDV was digested by BamHI and inserted into pHCE1LB-pgsA.
Lactobacillus casei525, isolated from Korean food, was transformed by electroporation (2.5 V, 400 Ω, 25 μF, 0.2 cm2 cuvette, Capacitance Extender Plus, Pulse Controller Plus, Bio-RAD, USA) with pHCE1LB-pgsA-PEDN. The lactobacilli were cultured in MRS medium (Difco, USA) supplemented with 16 μg/ml of erythromycin at 30 °C. The cells were harvested and the existence of the fusion proteins on their surface was examined.
ELISA and immunofluorescence microscopy
In order to examine the surface display of the recombinant N proteins on L. casei cells, the method of Laitinen et al. [6] was employed. Briefly, the cells were harvested after expression, and resuspended in phosphate-buffered saline (PBS) to Abs600 of 1.0. Aliquots of 1 ml from this resuspension were centrifuged at 3000 × g, for 30 min. and washed twice with PBS before being resuspended in 200 μl of PBS. Into the cell suspension 4 μl of rabbit polyclonal antibodies against PEDN prepared previously were added and incubated for 1 h. After washing, the cells were resuspended in 200 μl of PBS. Into the cell suspension, 2 μl of anti-rabbit IgG conjugated with horseradish peroxidase (Sigma, USA) was added and incubated for 1 h. The cells were washed twice with PBS. The cells were washed once with H2O2 substrate buffer containing OPD chromogen. The cells were resuspended in 1 ml of the same buffer to make the Abs600 value of 0.02–0.20. The cell suspension (100 μl) was poured into the wells of the microtitation plates. The plates were incubated 15 min at the dark at room temperature. In the same manner pgsA on the cell surface was detected to examine the fusion protein using the anti-pgsA antibody.
For immunofluorescence microscopy, cells labeled with anti-PEDV N polyclonal antibody and fluorescence isothiocyanate-conjugated anti-rabbit antibody were examined using a Carl Zeiss Axioskop 2 fluorescence microscopy. Photographs were taken with an Axiocam high-resolution camera.
SDS-PAGE and immunoblotting
In order to examine the pgsA-PEDN protein in the supernatant, SDS-PAGE described elsewhere and immunoblotting according to the method of Burnette [7] were performed using a 12% separating gel and 5% stacking gel. Gels were stained with Coomassie brilliant blue R250 (Amresco, USA). Immunoblotting was performed. Briefly, after transferring the protein to the polyvinylidene difluoride (PVDF) blotting membranes (Roche Diagnostic Corp., USA), the membranes were blocking with 5 ml of 1 M Tris–HCl, pH 8.0 solution containing 5% skim milk (Difco, USA) for 1 h. Rabbit anti-PEDN and anti-pgsA polyclonal antibodies were added in the blocking solution at a 1:1000 dilution, respectively. Following overnight incubation, the membranes were extensively washed with washing buffer (20 mM Tris–HCl, pH 8.0, 150 mM NaCl, 0.05% Tween-20). To the membranes 1:3000 dilution of biotinylated anti-rabbit antibody (Sigma, USA) was added and incubated for 3 h. After washing the membranes with washing buffer, the membranes were treated with avidin and biotin complex (Vectastain ABC Kit, Vector Lab, USA) following the manufacturer's instructions. Visualization of immunobinding was carried out with diaminobenzidine (DAB) solution (Vector Lab, USA). The typical band was observed by the naked eyes.
Immunization by recombinant N proteins in mice and sows and sampling
Five-week-old female C57BL/6 mice were obtained from SPF animal laboratory (Harbin Veterinary Research Institute, CAAS) and housed in filter-top cages. Mice were acclimated for 1 week after arrival before being used for immunization. The 11-day-old Yorkshire pregnant sows (JE IL FEED COMPANY, Korea) 1 month before farrowing, seronegative to the PEDV, were assigned to two different experiment groups. The sows were fed with a commercial diet for their age. Each group was kept in isolated facilities to prevent microbial contamination among them.
To study the possibility of the surface-displayed N proteins to induce the mucosal immunity in mice or sow, the method of Ogra et al. [8] was employed. Five-week-old female C57BL/6 mice (240 heads) were divided into 12 groups (20 heads/group). The 60 mice (five for each group) were kept for the untreated negative controls. Groups of six mice were immunized rather orally or intranasally with equal mixtures of live L. casei that express recombinant PEDN protein from plasmid pHCE1LB-pgsA-PEDN. L. casei harboring the parental plasmid pHCE1LB-pgsA was used as a negative control. The recombinant live L. casei cells displaying PEDN on their surface were resuspended in 100 μl sterile PBS at the concentration of 5 × 109 for the oral route. The suspension was administered daily via intragastric lavage on days 0–4, 7–11, 21–25 and 49–53. For the intranasal route, 2 × 109 L. casei cells in 20 μl suspension were administered into nostrils of lightly anesthetized mice on days 0–2, 7–9, 21 and 49. Blood samples were collected from the tail vein on days 0 (preimmune), 14, 28, 42, 56 and 70. Sera were prepared from the blood and stored at −20 °C until they were analyzed. To obtain bronchoalveolar and intestinal lavage samples, mice were killed on days 56 and 70. Bronchoalveolar and intestinal lavage fluids were obtained by washing the respective organs three times with 0.5 ml of ice-cold saline containing protease inhibitors. Samples were centrifuged at 2,500 × g for 20 min at 4 °C, and the supernatants were stored at −20 °C until they were analyzed.
In order to study the efficacy of the surface-displayed N proteins on L. casei on the prevention of the PEDV infection, the recombinant heat-killed L. casei cells (65 °C, 15 min, de Waard et al. [9]) surface-displaying PEDN were administered into the pregnant sows. Nine sows were used in this experiment, five sows for the treatment group and four sows for the untreatment control group. The recombinant heat-killed L. casei cells (109 cells/ml) were added to the feed at the final cell concentration of 106 cells/kg feed. The sows in the treatment group were fed with this feed throughout the experiment. The sows in the untreatment group were fed with the plain feed throughout the experiment. On days 0, 14, 42, 56 and 70, the sera were collected. The sow farrowed at around day 30. The colostrum was collected at the farrowing day. The neonates of the litter after suckling colostrum secreted from gilts previously inoculated recombinant PEDN L. casei were bled to prepare the sera.
ELISA for the detection of antigen-specific antibody
The recombinant protein ELISA was developed previously for detection of antigen-specific IgG and IgA in sera and colostrum. The method is referred to Hou et al. [10].
Plaque reduction neutralization assay
The neutralization activity of antigen-specific antibodies in sow and piglet serum and colostrum samples was tested using the plaque reduction neutralization test. Briefly, the PEDV (kPEDV-9, kindly provided by National Veterinary Research and Quarantine Services of Korea) suspension (5 × 1012 pfu/ml) was mixed with the serial twofold dilutions of antibodies (IgG purified from immunized sow and piglet sera, IgA purified from colostrum). After incubation at 37 °C for 1 h, the antibody–virus mixtures were added to Vero cell in 96-well plates, and the infection was allowed to proceed for 2 days. Then, the medium was decanted, and the cells were overlain with 0.9% noble agar dissolved in MEM, and incubated for 24 h. Finally, the virus plaques were visualized by adding 0.1% neutral red dye solution after the incubation. The control samples were from control sows and piglets. The IgA and IgG against PEDV as positive controls were kindly provided by Dr. Luo Chang-bao (China, serum neutralization titre ≥1:16)[10]. The same experiment was performed twice and the statistical comparison was made by the Mann–Whitney U-test.
Results
Construction of surface display vector and expression of PEDV viral-antigen hybrids on the surface of L. casei
To express fusion protein on the surface of L. casei, we employed pgsA gene as partner to direct the protein to the cell surface. We cloned PEDV N gene directed downstream to pgsA gene in a shuttle vector pHCE1LB. Escherichia coli harboring the plasmid was grown overnight at 37 °C. The cells were harvested, and expression of the expected chimeric protein was confirmed by immunoblotting using anti-pgsA and anti-PEDV N polyclonal antibodies (data not shown). L. casei cells were then transformed with the plasmid (pHCE1LB-pgsA-PEDN) and cultured at 30 °C. Expression of the pgsA-PEDN fusion protein was monitored by immunoblotting whole-cell lysates of serially passaged recombinant L. casei (Fig. 1 ). The pgsA-PEDN was stably expressed through serial passages and maintained its predicted molecular mass (90 kDa). When reacted with anti-PEDN and anti-pgsA polyclonal antibodies, the same bands corresponding to the interested protein size 90 kDa were exhibited strongly on the membranes.
Figure 1.
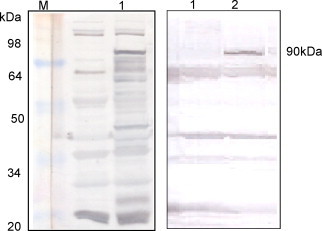
Detection of pHCE1LB-pgsA-PEDN expressed on Lactobacillus casei by Western Blot with anti-PEDN (left) and anti-pgsA (right) polyclonal antibodies. Lane 1 shows whole-cell lysates of wild-type (parental vector); lane 2 shows the recombinant L. casei. Protein bands of ∼90 kDa, corresponding to the expected size of pgsA-PEDN were detected.
To determine cellular localization of the recombinant protein on the surface of L. casei was verified by ELISA and florescence microscopy (Figure 2, Figure 3 , respectively). The ELISA color intensity increased with increasing cell density of recombinant L. casei, whereas the color intensity for L. casei host cells remained at a very low level (Fig. 2). Analyses by immunofluorescence microscope also revealed fluorescence only in the recombinant bacteria harboring pHCE1LB-pgsA-PEDN, but not in control cells harboring pHCE1LB-pgsA (Fig. 3). These results showed that PEDN protein was properly and efficiently displayed on the cell surface of L. casei using pgsA as a membrane-anchored protein display motif.
Figure 2.
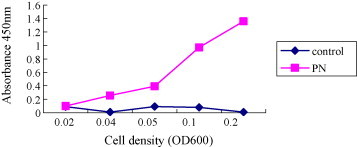
Confirmation of surface-displayed PEDN on L. casei by ELISA.
Figure 3.
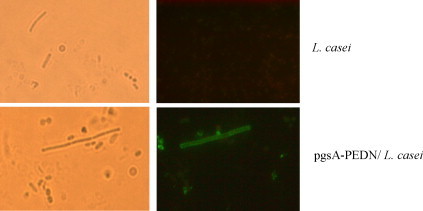
Representative immunofliuoresence images of control cells harboring pHCE1LB-pgsA and recombinant L. casei cells expressing pgsA-PEDN. Bright-field images are shown in the left.
Immunogenicity of hybrid proteins expressed on L. casei
In order to characterize the immunogenicity of PEDN surface-displayed on L. casei by mucosal administration the N proteins were administered into 5-week-old female C57BL/6 mice and pregnant sows. C57BL/6 mice (20 per group in 12 groups) were orally and intranasally administrated with 5 × 109 cells/dose and 2 × 109 cells/dose of the recombinant live L. casei, and control group with L. casei host cells. Serum samples were used for evaluating the systemic immune response by ELISA (Fig. 4 ). During the first two series of immunization, very low levels of IgG antibody were detected (Fig. 4, day 14). The higher IgG levels were detected shortly after the third immunization (day 28, p < 0.01). After the fourth immunization, further increase in IgG titer was observed (day 56, p < 0.01). The results also showed that there were no significant different between oral and intranasal routes.
Figure 4.
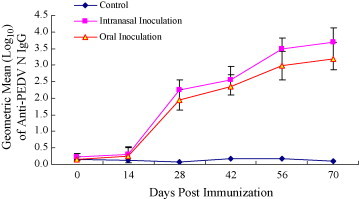
Antigenicity of surface displayed PED Ag in lactic acid bacteria display. Anti-PEDN mouse serum IgG absorbances following Intranasal, oral immunization with recombinant L. casei. Statistical comparisons between groups were made by the Mann–Whitney U-test.
To assess mucosal immune response, N protein-specific IgA levels in intestinal and bronchoalveolar lavage fluids were assessed by rnELISA. Fluids collected on days 56 and 70 after immunization were examined using the purified recombinant N protein expressed in E. coli as coating antigens. Both intranasal and oral immunization elicited N protein-specific IgA responses at the site of inoculation, as well as the remote mucosal sites (Fig. 5 ). However, only background levels of IgA were detected in control mice. To better characterize antibody responses against PEDN protein, five pregnant sows 1 month before farrowing the piglets were immunized with heat-killed recombinant PEDN L. casei. The nine pregnant sows with no previous history of PED, five of them were orally administrated, four with L. casei as control. Specific antibodies IgG were detected on days 0, 14, 42, 56 and 70 after immunization. Absorbance of IgG increased duration of the experiment (Fig. 6 ). IgG and IgA colostrum antibodies following oral immunization with recombinant L. casei were also detected by rnELISA. Absorbance IgA in colostrum (Fig. 7A , left) were increased significantly while IgG slightly (Fig. 7A, right). After suckling colostrum secreted from sows previously inoculated recombinant L. casei, IgG levels of the piglets were increased slightly (Fig. 7B).
Figure 5.
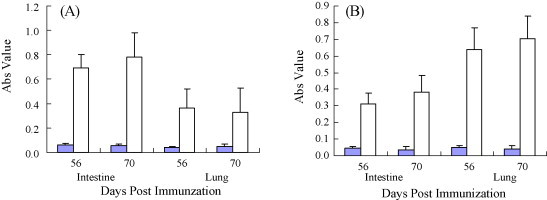
Anti-PEDVN mucosal IgA antibody responses. Intestinal and bronchoalveoar lavage fluids, harvested from mice sacrificed 56 or 70 days postimmunization, were analyzed by rnELISA. Absorbance 450 nm of samples from animals immunized orally (A) or intranasally (B) are shown. Fluids from control animals are shown as black bars. The error bars represent standard deviations.
Figure 6.
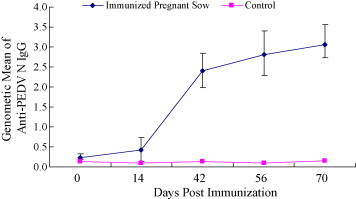
Anti-PEDN pregnant sow serum IgG absorbances following oral immunization with recombinant L. casei. Coating Ag: 100 ng/well; serum: 1:100; conjugation: 1:3000.
Figure 7.
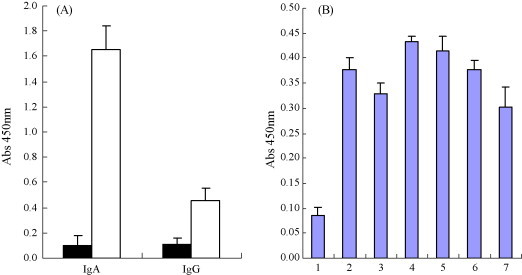
Detection of PEDN IgA and IgG levels in colostrum secreted from sow previously Inoculated recombinant L. casei (A). Coating Ag: 100 ng/well for PEDN colostrums: 1:100; conjugation: 1:3000. Colostrum from control animals are shown as black bars. The error bars represent standard deviations. PEDN IgG antibody in piglets serum on day 10 after sucking colostrums secreted from gilts previously inoculated recombinant PEDN L. casei (B). Coating Ag: 100 ng/well; serum: 1:100; conjugation: 1:3000, lane 1, negative control; lanes 2–7. The piglet sera randomly selected from offerings of the five immunized pregnant sows.
Virus neutralization by the antibodies induced by feeding sows and piglets recombinant L. casei
To confirm efficacy of the induced antibodies in inhibiting the virus, we tested whether serum and colostrum samples inhibited the infection of target cells in a plaque reduction neutralization assay. As shown in Fig. 8 , purified IgG from the sera collected from pregnant sow on day 70 after immunization and piglets on day 10 after sucking colostrums did not inhibit plaque formation by PEDV (Fig. 8A). No antiviral activity was observed with IgG purified from control animals. In contrast, the positive control IgG significantly inhibited plaque formation by 50.6% compared with controls, and the reduction was statistically significant (p < 0.01). Similarly, colostrum IgA also did not inhibit plaque formation compared with controls (Fig. 8B). But the positive control IgA significantly inhibited plaque formation by 47% (p < 0.01).
Figure 8.
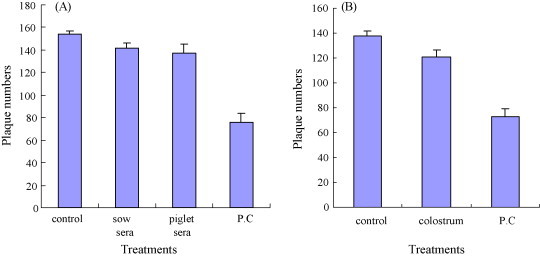
inhibition of viral plaque formation by (A) serum IgG prepared from sow fed with recombinant L. casei and piglets serum on day 10 after sucking colostrums and (B) colostrum IgA. (A) The number of plaque formed using purified IgG treatment. Control: IgG from sow sera unimmunized with recombinant L. casei; sow sera: IgG from sow sera on day 70 after immunization; piglets serum: IgG from piglet sera on day 10 after sucking colostrums; P.C: positive control IgG. (B) The number of plaque formed using purified IgA treatment. Control: IgA from sows’ colostrum unimmunized with recombinant L. casei; colostrum: IgA from immunized sows’ colostrum immunized with recombinant L. casei; P.C: positive control IgA. The assays were performed in duplicate, and the graphs are representative of two independent experiments. The error bars represent standard deviations.
Discussion
The recombinant live oral vaccines expressing pathogen-derived antigens on the surface of bacteria can be an alternative method for providing protection, because PEDV replicate mainly in the vili of the small intestines. Mucosal immunization offers a number of advantages over other routes of antigen delivery, including convenience, cost effectiveness and induction of both local and systemic immune responses [1], [5], [11], [12], [13]. The goal is to provide the first line of defense by effectively eliminating pathogens at the mucosal surface. But surface display of antigens on the bacterial surface has been problematic because large antigens perturb membrane topology. True surface exposure of antigens requires a transmenbrane anchor that is long enough to cross the cell wall [5]. At least 100 amino acids are needed to properly cross the cell wall [14].
In this report, we have developed a surface display system using the pgsA protein as the transmembrane anchor to present heterologous proteins on L. casei. We adopted pgsA protein derived from the pgsBCA enzyme complex of B. subtilis isolated from chung-kook-jang (a traditional Korean soybean paste used as a food additive). To investigate the feasibility of using lactic acid bacteria as a carrier of immunogenic peptides to mucosal immune system, we immunized mice intranasally and orally and pregnant sow orally with L. casei anchoring neucleocapsid proteins of PEDV. Although the amount of fusion PEDN proteins expressed on the surface of L. casei was not very higher than PEDN expressed in E. coli (data not shown), oral and intranasal immunization of swine and mice with these recombinant L. casei resulted in systemic and local immune responses. Mucosal delivery of vaccines induces mucosal immunity more efficiently than parenteral immunization [15]. The surfaces of the respiratory, gastrointestinal and urogenital tracts, which are not covered by skin, are referred to as mucosa [16]. In total, the mucosa covers an area that is about 200 times larger than the skin. This large area is not as resistant to penetration as the skin, because it is often limited by a thin epithelial layer without keratinization. A number of pathogens initiate infection through the mucosal surfaces. The mucosal surfaces, particularly the respiratory and digestive tracts, are exposed daily to a wide variety of foreign organisms and antigens. In addition, immunoglobulin A (IgA) is the primary immunoglobulin isotype induced at the mucosal surface. Secretory IgA (sIgA) in mucosal secretions provides protection against bacterial and viral pathogens and neutralization microbial toxins [17]. Thus, they must be very effective in dealing with the large amounts of foreign antigens. The most important part of the immediate defence against pathogens in the mucosa is the innate immune system. The mucosa and associated lymphoid tissue are an attractive target for vaccine strategies, because they harbor the early stages of infection [8]. The nasal cavity is already established as a potential alternative to the parenteral route for administering peptide antigens and drugs. Nasal administration of antigens could produce effectives for the protection of upper respiratory tract and other distant mucosal sites, as well as systemic immunity [18], [19], [20], [21], [22], [23].
Protection against neonatal infectious agents such as TGEV and PEDV is currently best obtained by passive immunization of piglets after induction of colostral antibodies in the sow [24], [25], [26]. Colostral antibodies can be induced by the oral delivery of protective antigens activating the gut-associated lymphoid-tissues (GALT) of sows [27], [26]. For this purpose, pregnant sow were orally administrated with recombinant L. casei before farrowing. The higher IgA levels in these sow colostrum were detected by PEDN ELISA, although their IgG levels were relatively lower. Furthermore, the piglets sucked these colostrum were showed a certain extent IgG level. Since swine are born without immunoglobulins, immunoprotection for newborn piglets mainly consists of passive immunity through colostral immunoglobulins from immunized dam [28]. Thus, passive immunity from colostrum is of primary important in piglets for protection against infectious enteric diseases. But protect immunity need to further research for virus challenge. Parenteral immunization is highly effective in protecting against systemic infection, in part due to the production of circulating IgG and its transudation into many extravascular sites, especially during inflammatory processes. However, parenteral immunization can fail to protect against invasion of mucosa. This failure is attributable to insufficient IgG transudation into mucosal secretions and the inability of systemic immunogen presentation to induce mucosal IgA production, the principal isotype present in external secretions. In contrast, mucosal immunization elicits both mucosal IgA and circulating IgG [29]. On mucosal surface, IgA represents a first line of defence by neutralizing invading pathogens [30]. Our observation support the concept of a mucosal immune system in which mucosally situated IgA and IgG plasma cell progenitors are stimulated selectively by mucosal immunization and predict that mucosal immunization can provoke both mucosal and circulating antibody responses better than parenteral immunization [29].
In conclusion, we have demonstrated that nucleocapsid protein of coronavirus PEDV exposed on the surface of non-pathogenic strain L. casei, resisting gastric acidity, delivered orally to animals, elicits both systemic and mucosal immune responses, although their antibodies cannot neutralize PEDV. In this study, we still constructed S1 gene of PEDV in L. casei, but the level of its expression on the surface of L. case was much lower than that of N gene (data not shown). Considering that the S protein as a major target for the development of animal coronavirus vaccine [5], [33], a vaccine that can induce mucosal immunity will include the S protein. In this regard, additional studies will be needed to further research to solve some problems on the S expression. For a successful immune response against an antigen, it is necessary that the antigen is present in sufficient quantities and in a form that can be recognized by antigen presenting cells of immune system. Moreover, not every gene can be expressed efficiently in host organism. Expression systems and experimental approaches are useful for overproduction of proteins. The results of our study at least indicate that the pgsA protein is capable of presenting foreign epitopes (size 1 kb or so) to the immune system and its recombinant fusion proteins on the surface of lactic acid bacteria can induce a specific anti-foreign epitope immune response after oral or intranasal administration. Furthermore, the pgsA-based chimeric surface display system expressed by L. casei can be optimized by using optical promoter. The immunogenicity of N antigens expressed by such strains may present significant opportunities for PEDV vaccine development.
During the last few years significant progress has already been made towards the development of an oral vaccine based on lactic acid bacteria as delivery vehicles [31]. By combining the knowledge generated in the different disciplines, molecular biology, immunology, ecology and gastroenterology, even more progress is expected to be made towards an understanding of the underlying principles of oral immunization and the importance of live bacterial carriers therein [32]. The elucidation and characterization at the DNA level of bacterial surface proteins involved in adherence to mucosal tissues has provided new and challenging opportunities which allow us to better understand the interaction between intestinal Lactobacilli and mucosal tissues. The recent development of molecular biological techniques that permit construction of lactic acid bacteria presenting foreign antigens in different of the immune system, offers for the first time opportunities to further explore the potential of these safe food bacteria as vaccine vehicles [9].
Despite the rapid advancement of our knowledge and concurrent development of techniques, a number of problems will have to be solved before the first LAB vaccine can be marketed. Although it is possible accurately determine the levels of antigen expression in the test tube, we cannot measure gene expression in the gut. Therefore, methods will have to be developed to analyze expression of the antigen encoding genes in situ so as to control the efficiency and duration of expression. In addition, how long colonizing bacteria are present in the gut and what the effects are of continuous expression of a LAB-associated antigen remain to be determined.
Acknowledgements
The authors wish to thank Chul-Joogh Kim, Jong-Soo Lee and Guang-Li Wei for their excellent technical assistance. This work supported from the fund of the 10th Five-Year-Plan in Key Science and Technology Research in Heilongjiang Province of China (GB05B501-2). JE IL FEED COMPANY supported this research.
Contributor Information
Li-Yun Yu, Email: yuliyun@hlau.cn.
Jianzhu Liu, Email: liujz@sdau.edu.cn.
References
- 1.Shaw D.M., Gaerthé B., Leer R.J., Van Der Stap J.G., Smittenaar C., Heijne Den Bak-Glashouwer M.-J. Engineering the microflora to vaccinate the mucosa: serum immunoglobulin G responses and activated draining cervical lymph nodes following mucosal application of tetanus toxin fragment C-expressing lactobacilli. Immunology. 2000;100(4):510–518. doi: 10.1046/j.1365-2567.2000.00069.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Enjuanes L., Van der Zeijst B.A.M. Molecular basis of transmissible gastroenteritis coronavirus (TGEV) epidemiology. In: Siddell S.G., editor. Coronavirus. Plenum; New York, NY: 1995. pp. 337–376. [Google Scholar]
- 3.Krempl C., Herrler G. Sialic acid binding activity of transmissible gastroenteritis coronavirus affects sedimentation behavior of virions and solubilized glycoproteins. J Virol. 2001;75(2):844–849. doi: 10.1128/JVI.75.2.844-849.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ashiuchi M., Nawa C., Kamei T., Song J.J., Hong S.P., Sung M.H. Physiological and biochemical characteristics of poly gamma-glutamate synthetase complex of Bacillus subtilis. Eur J Biochem. 2001;268(20):5321–5328. doi: 10.1046/j.0014-2956.2001.02475.x. [DOI] [PubMed] [Google Scholar]
- 5.Lee J.S., Poo H., Han D.P., Hong S.P., Kim K., Cho M.W. Mucosal immunization with surface-displayed severe acute respiratory syndrome coronavirus spike protein on Lactobacillus casei induces neutralizing antibodies in mice. J Virol. 2006;80(8):4079–4087. doi: 10.1128/JVI.80.8.4079-4087.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Laitinen R., Malinen E., Palva A. PCR-ELISA I: application to simultaneous analysis of mixed bacterial samples composed of intestinal species. Syst Appl Microbiol. 2002;25(2):241–248. doi: 10.1078/0723-2020-00118. [DOI] [PubMed] [Google Scholar]
- 7.Burnette W.N. “Western blotting”: electrophoretic transfer of proteins from sodium dodecyl sulfate-polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981;112(2):195–203. doi: 10.1016/0003-2697(81)90281-5. [DOI] [PubMed] [Google Scholar]
- 8.Ogra P.L., Faden H., Welliver R.C. Vaccination strategies for mucosal immune responses. Clin Microbiol Rev. 2001;14(2):430–445. doi: 10.1128/CMR.14.2.430-445.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.de Waard R., Claassen E., Bokken G.C., Buiting B., Garssen J., Vos J.G. Enhanced immunological memory responses to Listeria monocytogenes in rodents, as measured by delayed-type hypersensitivity (DTH), adoptive transfer of DTH, and protective immunity, following Lactobacillus casei Shirota ingestion. Clin Diagn Lab Immunol. 2003;10(1):59–65. doi: 10.1128/CDLI.10.1.59-65.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hou X.L., Yu L.Y., Liu J. Development and evaluation of enzyme-linked immunosorbent assay based on recombinant nucleocapsid protein for detection of porcine epidemic diarrhea (PEDV) antibodies. Vet Microbiol. 2007;123(1–3):86–92. doi: 10.1016/j.vetmic.2007.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mannam P., jones K.F., Geller B.L. Mucosal vaccine made from live, recombinant Lactococcus lactis protects mice against pharyngeal infection with Streptococcus pyogenes. Infect Immun. 2004;72:3444–3450. doi: 10.1128/IAI.72.6.3444-3450.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Seegers J.F. Lactobacilli as live vaccine delivery vectors: progress and prospects. Trends Biotechnol. 2002;20(12):508–515. doi: 10.1016/s0167-7799(02)02075-9. [DOI] [PubMed] [Google Scholar]
- 13.Xin K.Q., Hoshino Y., Toda Y., Igimi S., Kojima Y., Jounai N. Immunogenicity and protective efficacy of orally administered recombinant Lactococcus lactis expressing surface-bound HIV Env. Blood. 2003;102(1):223–228. doi: 10.1182/blood-2003-01-0110. [DOI] [PubMed] [Google Scholar]
- 14.Leenhouts K., Buist G., Kok J. Anchoring of proteins to lactic acid bacteria. Antonie Van Leeuwenhoek. 1999;76(1–4):367–376. [PubMed] [Google Scholar]
- 15.Mutwiri G., Watts T., Lew L., Beskorwayne T., Papp Z., Baca-Estrada M.E. Ileal and jejunal Peyer's patches play distinct roles in mucosal immunity of sheep. Immunology. 1999;97(3):455–461. doi: 10.1046/j.1365-2567.1999.00791.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ogra P.L., Mestecky J., Lamm M.E., Strober W., Bienstock J., McGhee J.R., editors. Mucosal immunology. 2nd ed. Academic Press; New York, NY: 1999. [Google Scholar]
- 17.Zhang Y., Pacheco S., Acuna C.L., Switzer K.C., Wang Y., Gilmore X. Immunoglobulin A-deficient mice exhibit altered T helper 1-type immune responses but retain mucosal immunity to influenza virus. Immunology. 2002;105(3):286–294. doi: 10.1046/j.0019-2805.2001.01368.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Almeida A.J., Alpar H.O. Nasal delivery of vaccines. J Drug Target. 1996;3(6):455–467. doi: 10.3109/10611869609015965. [DOI] [PubMed] [Google Scholar]
- 19.Kleanthous H., Myers G.A., Georgakopoulos K.M., Tibbitts T.J., Ingrassia J.W., Gray H.L. Rectal and intranasal immunizations with recombinant urease induce distinct local and serum immune responses in mice and protect against Helicobacter pylori infection. Infect Immun. 1998;66(6):2879–2886. doi: 10.1128/iai.66.6.2879-2886.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rudin A., Johansson E.L., Bergquist C., Holmgren J. Differential kinetics and distribution of antibodies in serum and nasal and vaginal secretions after nasal and oral vaccination of humans. Infect Immun. 1998;66(7):3390–3396. doi: 10.1128/iai.66.7.3390-3396.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Russell M.W., Moldoveanu Z., White P.L., Sibert G.J., Mestecky J., Michalek S.M. Salivary, nasal, genital and systemic antibody responses in monkeys immunized intranasally with a bacterial protein antigen and the Cholera toxin B subunit. Infect Immun. 1996;64(4):1272–1283. doi: 10.1128/iai.64.4.1272-1283.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Staats H.F., Jackson R.J., Marinaro M., Takahashi I., Kiyono H., McGhee J.R. Mucosal immunity to infection with implications for vaccine development. Curr Opin Immunol. 1994;6(4):572–583. doi: 10.1016/0952-7915(94)90144-9. [DOI] [PubMed] [Google Scholar]
- 23.Staats H.F., Nichols W.G., Palker T.J. Mucosal immunity to HIV-1: systemic and vaginal antibody responses after intranasal immunization with the HIV-1 C4/V3 peptide T1SP10 MN(A) J Immunol. 1996;157(1):462–472. [PubMed] [Google Scholar]
- 24.Moon H.W., Bunn T.O. Vaccines for preventing enterotoxigenic Escherichia coli infections in farm animals. Vaccine. 1993;11(2):200–213. doi: 10.1016/0264-410X(93)90020-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Saif L.J. Mucosal immunity: an overview and studies of enteric and respiratory coronavirus infections in a swine model of enteric disease. Vet Immunol Immunopathol. 1996;54(1–4):163–169. doi: 10.1016/S0165-2427(96)05702-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Saif L.J., Jackwood D.J. Enteric virus vaccines: theoretical considerations. Current status and future approaches. In: Saif L.J., Theil K.W., editors. Viral diarrhea of man and animal. CRC Press; Boca Raton, FL: 1990. pp. 313–329. [Google Scholar]
- 27.Butler J.E. Immunoglobulins and immunocytes in animal milks. In: Orga P., Mestecky J., Lamm M., Strober W., Bienenstock J., McGhee J.R., editors. Mucosal immunology. 2nd ed. Academic Press Inc.; New York, NY: 1999. pp. 1531–1534. [Google Scholar]
- 28.Kweon C.H., Kwon B.J., Woo S.R., Kim J.M., Woo G.H., Son D.H. Immunoprophylactic effect of chicken egg yolk immunoglobulin (Ig Y) against porcine epidemic diarrhea virus (PEDV) in piglets. J Vet Med Sci. 2000;62(9):961–964. doi: 10.1292/jvms.62.961. [DOI] [PubMed] [Google Scholar]
- 29.Heritage P.L., Loomes L.M., Jianxiong J., Brook M.A., Underdown B.J., McDermott M.R. Novel polymer-grafted starch microparticles for mucosal delivery of vaccines. Immunology. 1996;88(1):162–168. doi: 10.1046/j.1365-2567.1996.d01-639.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Scinicariello F., Attanasio R. Intraspecies heterogeneity of immunoglobulin alpha-chain constant region genes in rhesus macaques. Immunology. 2001;103(4):441–448. doi: 10.1046/j.1365-2567.2001.01251.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gentschev I., Dietrich G., Spreng S., Kolb-Mäurer A., Brinkmann V., Grode L. Recombinant attenuated bacteria for the delivery of subunit vaccines. Vaccine. 2001;19(17–19):2621–2628. doi: 10.1016/s0264-410x(00)00502-8. [DOI] [PubMed] [Google Scholar]
- 32.Collins L.V., Schodel F. Live bacterial vector. In: Pastorel P.P., Blancou Vannier J., Verschueren P.C., editors. Veterinary vaccinology. Elsevier Science Press; New York, NY: 1997. pp. 293–308. [Google Scholar]
- 33.Ho P.S., Kwang J., Lee Y.K. Intragastric administration of Lactobacillus casei expressing transmissible gastroentritis coronavirus spike glycoprotein-induced specific antibody production. Vaccine. 2005;23(11):1335–1342. doi: 10.1016/j.vaccine.2004.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]


