Abstract
The recombinant canarypox virus ALVAC is being extensively studied as vaccine vector for the development of new vaccine strategies against chronic infectious diseases and cancer. However, the mechanisms by which ALVAC initiates the immune response have not been completely elucidated. In order to determine the type of innate immunity triggered by ALVAC, we characterized the gene expression profile of human monocyte derived dendritic cells (MDDCs) upon ALVAC infection. These cells are permissive to poxvirus infection and play a key role in the initiation of immune responses. The majority of the genes that were up-regulated by ALVAC belong to the type I interferon signaling pathway including IRF7, STAT1, RIG-1, and MDA-5. Genes involved in the NF-κB pathway were not up-regulated. The gene encoding for the chemokine CXCL10, a direct target of the transcription factor IRF3 was among those up-regulated and DC secretion of CXCL10 following exposure to ALVAC was confirmed by ELISA. Many downstream type I interferon activated genes with anti-viral activity (PKR, Mx, ISG15 and OAS among others) were also up-regulated in response to ALVAC. Among these, ISG15 expression in its unconjugated form by Western blot analysis was demonstrated. In view of these results we propose that ALVAC induces type I interferon anti-viral innate immunity via a cytosolic pattern-recognition-receptor (PRR) sensing double-stranded DNA, through activation of IRF3 and IRF7. These findings may aid in the design of more effective ALVAC-vectored vaccines.
Keywords: Innate immunity, Microarray, Dendritic cell, ALVAC, IRF
1. Introduction
Viral vectors are increasingly being used as a vaccine delivery system. Advantages of the use of viral vectors include their ability to carry genes encoding for multiple antigens and the ability to infect cells thereby stimulating both the humoral and cellular arm of the immune system [1].
Poxviruses comprise a large family of dsDNA viruses that replicate in the cytoplasm and are capable of infecting a broad range of animal species. Recombinant canarypox virus ALVAC replication is limited to avian hosts. ALVAC-vectored vaccines encoding for viral [1], [2] or tumor antigens [3], [4], [5] have been administered to humans and have been shown to be safe and immunogenic. ALVAC can accommodate a large number of exogenous genes because of its large dsDNA genome and specific humoral and cellular immune responses can be raised against their products [6], [7], [8], [9], [10], [11], [12], [13].
Besides its antigen delivery function, ALVAC also possesses potent adjuvant activities, however the mechanisms involved in immunopotentiation by ALVAC are not well understood [14]. We have previously shown that ALVAC enhanced both Th1 humoral and cellular immune responses to co-injected protein antigens in a mouse model. Precisely, ALVAC increased the levels of antigen-specific IgG2a antibodies and the number of IFNγ secreting cells in response to co-injected cytomegalovirus (CMV) gB or Human Immunodeficiency Virus (HIV) Tat protein antigens, compared to antigen alone. The adjuvant effect of ALVAC was dependent on the presence of IFNγ produced by natural killer (NK) cells [15]. Neutrophil infiltration at the ALVAC injection site was demonstrated by Boudet et al. also suggesting that ALVAC induces pro-inflammatory mechanisms [14].
Although full replication of canarypox virus is restricted to avian species, mammalian cells can be infected and viral as well as exogenous protein expression can be detected as early as one hour post-infection [16]. Both murine and human cells of the myeloid lineage, particularly immature MDDCs, are readily infected by ALVAC resulting in maturation of these cells [17], [18], [19]. In the present study we also observed DC partial maturation following ALVAC infection of immature MDDCs. Given the key role played by DCs in the orientation and initiation of acquired immune response, particularly priming of naïve T cells, the ability of ALVAC to potentially deliver foreign antigens directly to these potent antigen-presenting-cells (APC) increases its attractiveness as a vaccine vector.
Although immunogenic for some indications such as CMV pp65 [20], limitations in the immunogenicity of ALVAC have been reported in other models such as HIV, as defined by the number of cytotoxic T cells against the encoded antigens [21], [22], [23], [24]. Delivered intramuscularly, ALVAC induced inconsistent antigen-specific cytotoxic T-cell responses (HIV) in only about one third of the volunteers [6], [7], [9], [10].
A better understanding of the molecular mechanisms of action of ALVAC would provide insight into how to enhance its efficacy for future vaccine development. The current study aimed at identifying the pattern of gene expression in human immature dendritic cells (DC) upon ALVAC infection by microarray technology.
Comparing the gene signatures of ALVAC treated and non-treated MDDCs revealed a number of pertinent up-regulated genes belonging to families of genes such as cytokines, transcriptional regulators, RNA helicases, ubiquitination and ISGylation, anti-viral-, nuclease- and antigen-processing genes. The majority of the genes up-regulated following ALVAC infection was involved in the type I IFN pathway. In contrast, genes involved in the NF-κB signaling pathway (leading to the production of pro-inflammatory cytokines) were not up-regulated. The gene encoding for the chemokine CXCL10, a direct target of the transcription factor IRF3, was up-regulated in response to ALVAC. CXCL10 protein secretion in the supernatants of ALVAC infected DCs was confirmed by ELISA. The expression of a number of known interferon responsive genes such as IRF7, RIG-1 and MDA-5 as well as anti-viral genes like OAS, Mx, ISG15, and PKR were enhanced in human MDDCs cells following ALVAC infection. In this study, we focused on the ISG15 gene in view of increasing evidence suggesting its role in the regulation of immune responses [25], [26], [27], [28], [29]. The induction of the free form of ISG15 in human MDDCs was confirmed by Western blot analysis. These results support the hypothesis that ALVAC activates IFR3 and IFR7 mediated induction of the type I IFN signaling cascade through a cytosolic pattern-recognition receptor (PRR) sensing dsDNA viruses, such as the recently reported “DNA-dependent activator of IFN-regulatory molecule” (DAI) [30], [31].
2. Materials and methods
2.1. Antigens
Lyophilized recombinant canarypox vector ALVAC-HIV, vCP1521 (Sanofi-Pasteur, Marcy l’Etoile, France) containing the gene for HIV-1 envelope gp120 (strain TH023) linked to the trans-membrane portion of HIV-1 gp41 (strain LAI) and the gene HIV-1 LAI gag encoding for the entire gag protein was used.
ALVAC was re-constituted in sterile 0.9% saline or medium prior to usage. All vaccines used in this study were GMP clinical grade and were verified to be free of lipopolysaccharide (LPS) and other impurities.
2.2. Preparation of MDDCs
Heparinized human blood was obtained from healthy donors from the French blood bank E.F.S. Rhone-Alpes. Peripheral blood mononuclear cells (PBMCs) were isolated by centrifugation on a LymphoPrep gradient (Axis-Shield, Oslo, Norway). Monocytes were purified from total PBMCs by positive selection using CD14 magnetic beads (Miltenyi Biotec, Bergisch-Gladbach, Germany). To obtain immature DC, monocytes were cultured in RPMI supplemented with 10% FCS in presence of 50 ng/ml of GM-CSF and 10 ng/ml of IL-4 for 6 days. On day 6, the purity of the culture was analyzed by flow cytometry as percentage of CD11c positive cells and used for further experiments if it exceeded 80%.
2.3. Infection of DCs with ALVAC
Immature DCs prepared as described above were cultured at 106 cells/ml in RPMI supplemented with 10% FCS in the presence of ALVAC at 0.2 multiplicity of infection (MOI) or 10 μg/ml Poly (I:C) (Invivogen, San Diego, CA) and incubated at 37 °C for either 2, 6, 16, 24 or 48 h. ALVAC was left together with the DCs for the indicated times and was not washed out. When indicated 250 U of human Leukocyte Interferon (Hu-IFN-α and Hu-IFN-ω) (PBL Biomedical Laboratories), 5 μg/ml neutralizing interferon alpha/beta receptor chain 2 (CD118) human antibody (#RDI-PB21385, Fitzgerald, Concord, MA) or 5 μg/ml non-neutralizing interferon alpha/beta receptor chain 2 antibody (#RDI-PB31385 Fitzgerald) were added to the cultures during incubation time.
2.4. FACS analysis
Immature DCs were infected with ALVAC as described above or else incubated in the presence of 10 ng/ml LPS (Invivogen, Toulouse, France) as a positive control or left untreated as a negative control for 24 h. After the indicated times, DCs were washed once and resuspended in PBS with 2% FCS and 0.01% NaN3; then labeled with mAbs specific for CD11c (DAKO, Glostrup, Denmarkl), CD14, CD40, CD80, CD83, CD86, CD25, HLA-1, and HLA-DR (all purchased from BD Biosciences, Franklin Lakes, NJ). Cells were acquired and analyzed by flow cytometry using a FACSCalibur (BD Biosciences) and results analyzed using CellQuest Pro™ Software (BD Biosciences).
2.5. RNA isolation and oligonucleotide expression arrays
Total RNA was isolated from immature DCs prepared as described above, using the Nucleospin RNA II Kit (Macherey Nagel, Dueren, Germany) according to the manufacturer's protocol. Briefly, 5 × 106 MDDCs in 5 ml RPMI supplemented with 10% FCS, l-glutamine and penicillin/streptomycin were stimulated with 0.2 MOI ALVAC for 6 h. After 6 h cells were pelleted and washed twice with PBS. The cells were lysed in 350 μl RA1 lysis buffer from the Nucleospin II kit (Machery Nagel) supplemented with 3.5 μl of β-mercapto-ethanol. RNA was then eluted from the column using 60 μl RNAse-free H2O pre-warmed to 60 °C. The concentration and quality of the RNA was measured using a ND-1000 spectrophotometer (NanoDrop Technologies, Wilmington, DE) and Agilent 2100 Bioanalyzer (Agilent Technologies, Palo Alto, CA), respectively.
The Low RNA Input Fluorescent Linear amplification kit (Agilent Technologies) was used to generate cyanine 3- and cyanine 5-labeled complimentary RNA (cRNA). First, the total RNA was transcribed into cDNA using oligo dT-Promoter primer and MMLV-Reverse Transcriptase. Next, cRNA was synthesized from the double-stranded cDNA using T7 RNA polymerase, which simultaneously incorporates cyanine 3- or cyanine 5- labeled CTP (Perkin Elmer, Waltham, MA). Once labeling was complete, the labeled cRNA was purified with RNAeasy Mini Kit (Qiagen, Valencia, CA) following the manufacturer's protocol. Using the ND-1000 spectrophotometer (NanoDrop Technologies) the concentration and the amount of dye incorporated into the amplified cRNA were measured. After the purification of the labeled RNA, 1 μg of each of the differently labeled samples were combined, fragmented and subsequently hybridized to the whole human genome oligo-microarray slide (44 K, Agilent Technologies). Hybridization and washing of the slides were performed as described in the manufacturer's protocol (Agilent Technologies).
Slides were scanned using Scanner GenePix Personal 4100 (Axon Instrument, Union City, CA). Fluorescence intensity for each feature of the array was obtained using GenePix Pro v6.1 software. Rosetta Resolver (Rosetta Biosoftware, Seattle, WA) and Ingenuity Pathway Analysis (Ingenuity Systems, Redwood City, CA) software were used for further analysis.
Two dye-swapped hybridizations were performed for each donor. In one, the untreated sample was Cy3 labeled, and the ALVAC-infected sample was Cy5 labeled; in the second, labeling was reversed. Double labeling was used to abolish dye-specific labeling and hybridization differences.
2.6. RT-PCR
RNA was extracted from human MDDCs infected with ALVAC at MOI of 0.2 for 2, 6 or 16 h or stimulated with Poly I:C at 10 μg/ml, as described above. The reverse transcription (RT)-PCR analysis was carried out using the Super-Script II Reverse Transcriptase kit (Invitrogen). Briefly, first strand synthesis (cDNA) reaction mixture was incubated at 42 °C for 2 h. The following gene-specific primers were used for amplification: IFNβ sense 5′-GAT TCA TCT AGC ACT GGC TGG-3′, antisense 5′-CTT CAG GTA ATG CAG AAT CC-3′ (amplicon of 185 nt); b-actin sense 5′-ACA ATG AGC TGC GTG TGG CT-3′, antisense 5′-GAT GGG CAC AGT GTG GGT GA-3′ (amplicon of 223 nt). The RT-PCR reaction mixture contained: 1/10 of the cDNA, 100 nM of each primer, 1 U of Platinum Taq DNA Polymerase (Invitrogen), 200 mM dNTP mix, in 20 mM Tris–HCl (pH 8.4), 50 mM KCl, and 1.5 mM MgCl2 buffer in a final volume of 50 μl. The PCR was performed in an automated thermocycler (Perkin-Elmer Cetus, Norwalk, Conn.) under the following conditions: denaturing at 94 °C for 2 min, 35 amplification cycles, each consisting of three rounds at 94 °C for 30 s, 55 °C for 30 s, and 72 °C for 1 min and a final extension for 5 min at 72 °C. After the PCR amplification reaction, 20 μl of each reaction was loaded on 1.5% agarose gels in TAE buffer.
2.7. Immunoblotting
Monocyte derived dendritic cells (MDDC) were generated as described above and after 6 days in culture were resuspended in RPMI supplemented with 10% FCS at a concentration of 1 × 106 cells/ml. Cells were infected with 0.2 MOI ALVAC or 1000 U/ml human leukocyte interferon (hIFN-α and hIFNω) (PBL Biomedical Laboratories, New Brunswick, NJ) for 48 h at 37 °C. After incubation cells were lysed by addition of 1× lysis buffer (50 mM Tris–HCl, 150 mM NaCl, 1% NP40, 0.5% sodium deoxycholate, and protease inhibitors (source)). Cell debris was removed by centrifugation at 13,000 rpm for 5 min. SDS PAGE sample buffer (Bio-Rad Laboratories, Hercules, CA) was added to the protein extract which was then boiled for 5 min. Following separation of the whole cell extracts (2.5 × 106 MDDC per lane) by reducing SDS-PAGE electrophoresis, samples were transferred to Hybond-PVDF (GE Healthcare, UK) and blocked with TBS containing 0.05% Tween-20 (Sigma–Aldrich, St. Louis, MO) and 5% skim milk (BD, Franklin Lakes, NJ) at room temperature (RT) for one hour. Membranes were washed and incubated overnight with anti-ISG15 polyclonal antibody (Cell Signaling Technology, Danvers, MA) at 4 °C or for 3 h at RT. Membranes were washed three times and incubated with secondary anti-rabbit-HRP antibody (GE Healthcare, UK) for 1 h at RT. Blots were developed using ECL Plus (GE Healthcare, UK). In order to verify equal protein loading, membranes were washed, blocked as described above and incubated for 3 h with anti-GAPDH (Abcam, Cambridge, UK) directly coupled to HRP.
2.8. Cytokine detection
Cell supernatants were collected after 48 h in vitro stimulation and frozen at −20 °C until analysis. Cytokines in the supernatants were quantified by sandwich ELISA following the manufacturers’ protocols using the commercially available human CXCL10 DuoSet kit (R&D Systems, Minneapolis, MN), human IFN-α ELISA kit (PBL Biomedical Laboratories) or the human IFN-β ELISA Kit (Fujirebio Inc, Tokyo, Japan).
3. Results
3.1. Maturation of human immature MDDCs upon exposure to ALVAC
Human immature MDDCs were obtained by culturing CD14 positive monocytes from PBMCs from healthy volunteers in the presence of GM-CSF and IL-4 for 6 days. Flow cytometric analysis was then performed to assess their purity and phenotype (positive for CD11c and negative for CD14) (Fig. 1 A). To confirm that ALVAC induced DC maturation as previously described in human cells [32] and in mouse bone marrow derived dendritic cells [15] immature MDDCs were cultured for 24 h in the presence of ALVAC at a MOI of 0.2. Some prior dose response experiments using murine [15] and human (data not shown) DCs have indicated that 0.2 MOI is an optimal dose for DCs stimulation with minimal cell death. DCs treated with LPS at 10 ng/ml or left untreated were used as positive and negative controls, respectively.
Fig. 1.
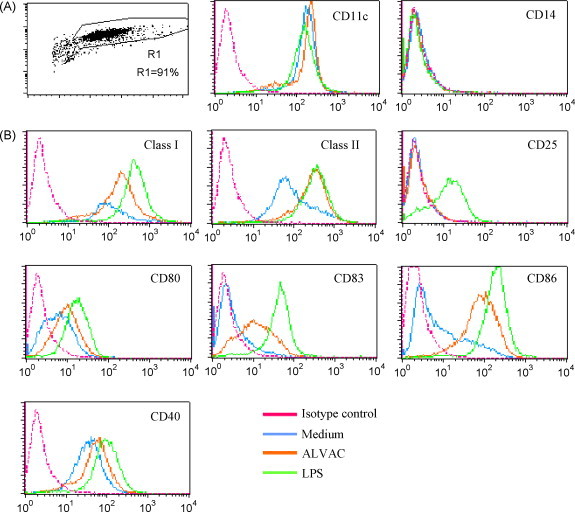
Maturation of human immature MDDCs upon exposure to ALVAC. (A) Human immature MDDCs (CD11c+ and CD14−), obtained from CD14 purified monocytes upon 6-day culture with GM-CSF and IL-4, were stimulated for 24 h with ALVAC (0.2 MOI), LPS (10 ng/ml) or left untreated (indicated by color code). (B) Plots show super-imposition of the surface expression of each maturation marker relative to the Ab isotype control under the three experimental conditions (ALVAC, LPS or medium). ALVAC infection increased the expression of DC maturation-associated markers CD40, CD80, CD86, CD83, HLA-1 (MHC-I), HLA-DR (MHC-II) on the surface of human MDDC. However, in contrast to LPS, ALVAC failed to up-regulate the CD25 marker.
As expected, ALVAC induced the up-regulation of maturation-associated surface markers: CD40, CD80, CD83, CD86, HLA-1 and HLA-DR although to a lesser extent than the positive control, LPS (Fig. 1B). Surprisingly, in contrast to LPS stimulation ALVAC failed to induce the expression of CD25, a molecule recently shown to be important for DC mediated T cell activation and IL-2 production [33].
3.2. Global gene expression in ALVAC treated versus untreated human MDDCs
A kinetic study was conducted in order to determine the optimal time point(s) after ALVAC infection for gene expression analysis. Immature MDDCs from a single donor were infected with ALVAC at 0.2 MOI for 2, 6 or 16 h and gene expression evaluated by microarray analysis (data not shown). Since the number of differentially regulated genes was highest at 6 h post-infection, this time point was chosen for further experiments in MDDCs. In addition, minimal viral cytopathic effects were expected to occur at this early time point as was reported by Guerra et al. for MVA and NYVAC poxvirus vectors [34].
RNA was isolated from ALVAC-treated or untreated MDDCs from four different healthy volunteers upon 6 h of infection, and then transcribed into labeled complimentary RNA as described in Section 2. Differentially labeled samples, to distinguish between cRNA from treated and untreated cells, were combined and hybridized to a 60-mer oligomer microarray slide, which represent 41,000 unique human genes and transcripts. A minimum of ±1.5-fold change in expression and a p-value of less than 0.05 were the criteria used to select for differentially regulated genes. Under these conditions, a total of 1433 differentially up-regulated and 1044 differentially down-regulated genes were identified. Extensive variation in the number of up- and down-regulated genes was observed among the four donors. Therefore, we focused our analysis on those genes differently regulated in all four donors, referred to as ‘core-regulated genes’. As shown in Fig. 2 , 123 core-up-regulated genes were identified (Fig. 2A), in contrast, no core-down-regulated genes were found (Fig. 2B). Therefore, we did not further investigate down-regulated genes in the present study.
Fig. 2.
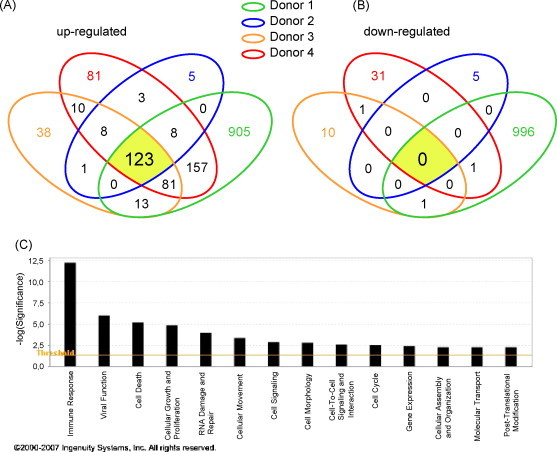
Venn diagram showing the overlap between 1433 differentially up-regulated (A) and 1044 differentially down-regulated genes (B) in ALVAC infected MDDCs from four independent healthy donors. Each of the four circles represents the set of genes differentially expressed in each donor (fold increase >±1.5, p < 0.05). Numbers depicted in the intersections between the circles represent numbers of genes differentially up-regulated in two, three or four donors. (C) Ontology analysis of the genes differently regulated in response to ALVAC relative to untreated cells. In the bar chart, the Y-axis represents the significance threshold of a given gene involved in a particular function.
Using Ingenuity Pathway Analyzer, 104 over the 123 core-up-regulated genes were recognized as known genes and classified according to their molecular function (selection shown in Table 1 ). Sixty-five (65) of them were incorporated into networks on the basis of the molecular interaction information found within the Ingenuity Pathways Knowledge Base. In decreasing order of magnitude, the biological functions they belonged to were: (1) immune response; (2) viral function; (3) cell death; (4) cellular growth and proliferation; (5) cellular motility; and (6) gene expression (Fig. 2C). The majority of the genes identified were associated with more than one biological function. Of these 65 genes, 25 (∼1/3) were involved in the immune response. As shown in Table 1, the list of immune-related genes include chemokines (e.g. CXCL10, CCL2, and CCL8), anti-viral proteins (e.g. Mx1, Mx2, OAS1), interferon responsive genes (IRF1, IRF7, IFI6, IFI44, ISG15, etc.), Myd88 (adaptor of TLR signaling), TAP1 (antigen processing,) and STAT1 (interferon signaling), among many others (see complete list in Supplementary Data).
Table 1.
Selection of core-up-regulated genes in human MDDCs in response to ALVAC
| GenBank ID | Common name | Description | Fold changea | Standard deviation |
|---|---|---|---|---|
| Cytokines | ||||
| M24545 | CCL2 | Chemokine (C–C motif) ligand 2 | 3.78 | ±2.87 |
| NM_005623 | CCL8 | Chemokine (C–C motif) ligand 8 | 17.17 | ±19.02 |
| NM_001565 | CXCL10 | Chemokine (C–X–C motif) ligand 10 | 8.06 | ±1.97 |
| NM_005746 | PBEF1 | Pre-B-cell colony enhancing factor 1 | 2.37 | ±1.14 |
| NM_005746.1 | PBEF1 | Pre-B-cell colony enhancing factor 1 | 2.41 | ±1.11 |
| U57059 | TNFSF10 | Tumor necrosis factor (ligand) superfamily, member 10 | 8.84 | ±5.21 |
| Transcriptional regulators | ||||
| NM_020740 | ANKFY1 | Ankyrin repeat and FYVE domain containing 1 | 2.05 | ±0.22 |
| BC006322 | ATF3 | Activating transcription factor 3 | 4.10 | ±1.61 |
| NM_138456.2 | BATF2 | Basic leucine zipper transcription factor, ATF-like 2 | 5.53 | ±2.30 |
| BC020597 | GTF2B | General transcription factor IIB | 1.78 | ±0.26 |
| U82811 | HESX1 | Homeobox, ES cell expressed 1 | 7.79 | ±4.81 |
| AY358248 | MAML2 | Mastermind-like 2 (Drosophila) | 2.15 | ±0.69 |
| NM_012385 | P8 | Nuclear protein 1 | 3.00 | ±2.08 |
| NM_002675 | PML | Promyelocytic leukemia | 2.77 | ±0.95 |
| NM_006074 | TRIM22 | Tripartite motif-containing 22 | 3.78 | ±2.27 |
| NM_021035 | ZNFX1 | Zinc finger, NFX1-type containing 1 | 2.12 | ±0.43 |
| NM_181782 | NCOA7 | Nuclear receptor coactivator 7 | 2.53 | ±0.79 |
| NM_080424 | SP110 | SP110 nuclear body protein | 4.71 | ±2.34 |
| ISGylation and ubiquitinylation | ||||
| NM_017414 | USP18 | Ubiquitin specific peptidase 18 | 10.67 | ±4.22 |
| AJ586979 | USP41 | Ubiquitin specific peptidase 41 | 3.26 | ±1.45 |
| BC007922 | ISG20 | Interferon stimulated exonuclease gene 20 kDa | 5.66 | ±3.44 |
| NM_016323 | HERC5 | Hect domain and RLD 5 | 16.79 | ±12.97 |
| NM_017912 | HERC6 | Hect domain and RLD 6 | 4.47 | ±2.21 |
| NM_005101 | G1P2 | ISG15 ubiquitin-like modifier | 10.07 | ±4.05 |
| X02492 | G1P3 | Interferon, alpha-inducible protein 6 | 3.62 | ±2.29 |
| Interferon inducible and anti-viral genes | ||||
| X67325 | IFI27 | Interferon, alpha-inducible protein 27 | 6.97 | ±6.63 |
| NM_005532 | IFI27 | Interferon, alpha-inducible protein 27 | 10.18 | ±12.10 |
| BC022870 | IFI44 | Interferon-induced protein 44 | 8.73 | ±2.45 |
| NM_006820 | IFI44L | Interferon-induced protein 44-like | 11.41 | ±7.01 |
| BC032839 | IFIT2 | Interferon-induced protein with tetratricopeptide repeats 2 | 77.00 | ±16.52 |
| NM_001547 | IFIT2 | Interferon-induced protein with tetratricopeptide repeats 2 | 21.30 | ±1.76 |
| NM_001549 | IFIT3 | Interferon-induced protein with tetratricopeptide repeats 3 | 26.33 | ±6.67 |
| U34605 | IFIT5 | Interferon-induced protein with tetratricopeptide repeats 5 | 3.50 | ±0.86 |
| NM_012420 | IFIT5 | Interferon-induced protein with tetratricopeptide repeats 5 | 3.40 | ±0.89 |
| J04164 | IFITM1 | Interferon-induced trans-membrane protein 1 (9–27) | 5.47 | ±4.83 |
| NM_021034 | IFITM3 | Interferon-induced trans-membrane protein 3 (1–8 U) | 3.27 | ±2.18 |
| BC009483 | IRF1 | Interferon regulatory factor 1 | 2.29 | ±0.31 |
| NM_004031 | IRF7 | Interferon regulatory factor 7 | 4.83 | ±0.52 |
| NM_139266 | STAT1 | Signal transducer and activator of transcription 1, 91 kDa | 3.37 | ±1.69 |
| BC038247 | TRIM25 | Tripartite motif-containing 25 | 3.17 | ±1.12 |
| X04371 | OAS1 | 2′,5′-Oligoadenylate synthetase 1, 40/46 kDa | 4.22 | ±1.67 |
| NM_016817 | OAS2 | 2′-5′-Oligoadenylate synthetase 2, 69/71 kDa | 3.70 | ±1.20 |
| NM_006187 | OAS3 | 2′-5′-Oligoadenylate synthetase 3, 100 kDa | 7.16 | ±2.86 |
| NM_003733 | OASL | 2′-5′-Oligoadenylate synthetase-like | 9.60 | ±2.89 |
| BC032602 | MX1 | Myxovirus (influenza virus) resistance 1, interferon-inducible protein p78 (mouse) | 8.64 | ±2.58 |
| M30818 | MX2 | Myxovirus (influenza virus) resistance 2 (mouse) | 9.35 | ±4.10 |
| NM_002463 | MX2 | Myxovirus (influenza virus) resistance 2 (mouse) | 4.21 | ±1.26 |
| BC002666 | GBP1 | Guanylate binding protein 1, interferon-inducible, 67 kDa | 5.52 | ±2.73 |
| NM_002053 | GBP1 | Guanylate binding protein 1, interferon-inducible, 67 kDa | 6.56 | ±5.15 |
| AL832576 | GBP4 | Guanylate binding protein 4 | 4.68 | ±3.51 |
| NM_080657 | RSAD2 | Radical S-adenosyl methionine domain containing 2 | 13.83 | ±10.25 |
| RNA helicases | ||||
| AF038963 | DDX58 | Dead (Asp-Glu-Ala-Asp) box polypeptide 58/RIG-1 | 12.81 | ±7.30 |
| AF095844 | IFIH1 | Interferon-induced with helicase C domain 1/MDA-5 | 5.09 | ±2.25 |
| BC040851 | EIF2AK2 | Eukaryotic translation initiation factor 2-alpha kinase 2/PKR | 4.50 | ±2.10 |
| AK021416 | LGP2 | Likely ortholog of mouse D11lgp2 | 3.43 | ±1.60 |
| Miscellaneous immune system | ||||
| NM_002468.2 | MYD88 | Myeloid differentiation primary response gene (88) | 2.14 | ±0.53 |
| BC014081 | TAP1 | Transporter 1, ATP-binding cassette, sub-family B (MDR/TAP) | 2.31 | ±0.67 |
| BC007037 | CD69 | CD69 molecule | 2.96 | ±0.71 |
| NM_002127 | HLA-G | HLA-G histocompatibility antigen, class I, G | 1.80 | ±0.18 |
| AK122586 | SIGLEC1 | Sialic acid binding Ig-like lectin 1, sialoadhesin | 3.94 | ±3.29 |
The fold change represents the mean of four different donors.
3.3. ALVAC induces a type I interferon pattern gene expression
Inducible interferon regulatory factor 7 (IRF7) was significantly increased (+4.8 mean fold change, Table 1) in human MDDCs from all four donors upon ALVAC infection. Inducible interferon regulatory factor 7, together with IRF3, the latter constitutively expressed in the majority of cell types, are key regulators of type I interferon (IFN) gene expression in response to pathogen associated molecular patterns (PAMPs) via either the trans-membrane Toll Like Receptor (TLR) or the cytosolic pattern-recognition systems [31]. Activation of IRF3 and IRF7 (the latter constitutively present only in small amounts) occurs through phosphorylation and formation of homo- and heterodimers. These dimers translocate into the nucleus where they promote IFN-α- and -β gene transcription (Fig. 3 ). Following the activation of IFNR1 and IFNR2 receptors by secreted IFNs, the expression of IRF7 and other molecules such as RIG-1 and MDA-5 as well as type I IFNs themselves are induced in a positive-feedback loop that amplifies this signaling pathway.
Fig. 3.
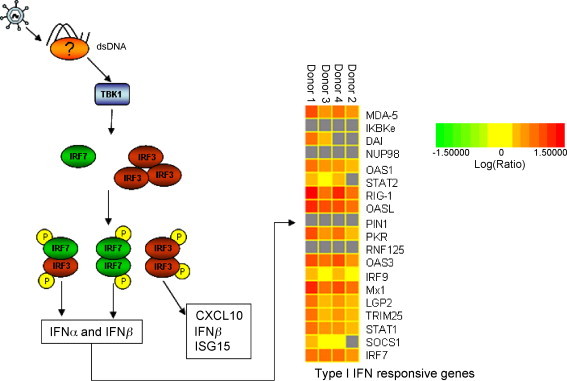
Proposed model of ALVAC induction of the type I IFN pathway in MDDCs (left side) and representation of type I IFN responsive gene expression levels from microarray analysis (right side). (Left side) We propose that ALVAC is detected by a cytoplasmic dsDNA sensor which leads to the phosphorylation of IRF3 via TBK1. Phosphorylated IRF3 homodimerizes or heterodimerizes with IRF7 and migrates to the nucleus where it directly induces the expression of IFN-α/β, CXCL10 and ISG15 among others. In a positive feedback loop type I IFNs induce gene expression of IRF7, MDA-5, RIG-1, etc. (Right side) Two-dimensional hierarchical clustering was performed using Rosetta Resolver System Software. Columns represent gene expression data for four distinct donors from an individual experiment. The intensity of the color red indicates the degree of up-regulation, genes which did not meet the required p-value of 0.05 are colored in grey.
Surprisingly, the up-regulation of IFN-β gene expression was observed only in two of the four donors while IFN-α expression was not up-regulated in the MDDCs of any donor included in the microarray analysis. When tested by reverse-transcripase PCR (RT-PCR) IFN-β was expressed at the highest level after 16 h of ALVAC infection in accordance with the positive-feedback loop (Fig. 4 ). Neither IFN-α nor IFN-β were detected in the supernatants of ALVAC infected MDDCs by ELISA (detection level ∼15 pg/ml). To explain this observation, it should be considered that IFN-α subtype expression is cell-type, donor and stimuli dependent and that not all the subtypes (13 in humans) were represented on the microarray chip (IFN-α subtypes present on the microarray chip: 2, 4, 5, 6, 8, 10, 14, and 21). Furthermore, MDDCs are known to be poor producers of type I IFNs, these are mainly secreted by plasmacytoid DCs and their precursors [35]. Therefore, the levels of type I IFNs in the supernatants might be below the detection limit of the assays used.
Fig. 4.
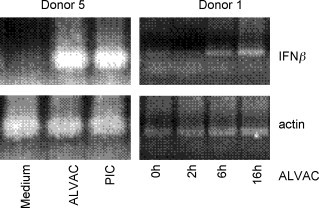
Enhanced IFNβ gene transcription in ALVAC infected human MDDCs. MDDCs were incubated as indicated for 2, 6 or 16 h (Donor 1) or 16 h (Donor 5) with either medium alone or ALVAC at a MOI of 0.2 or Poly I:C at 10 μg/ml as a positive control as indicated. Amplification of the IFNβ and β-actin encoding genes was performed by RT-PCR as described in Section 2. A band of the expected size of 185 nt was detected in MDDCs upon ALVAC infection and Poly I:C stimulation. Under the current experimental conditions IFNβ gene transcription in MDDCs was evident after 6 h exposure to ALVAC (right figure, Donor 1).
Our observation that signaling molecules downstream of the IFN receptor such as STAT1 and IRF9 (the latter in three donors) were up-regulated by ALVAC infection of MDDCs is consistent with the hypothesis that ALVAC triggers the type I IFN signaling pathway. Furthermore, IFN responsive genes such as positive regulators RIG-1, MDA-5 and IRF7 and anti-viral proteins OAS, Mx, ISG15 and PKR (indicated as red squares in Fig. 3) were among the genes up-regulated in response to ALVAC.
3.4. ALVAC induces production of CXCL10
Among the immune response-related genes up-regulated by MDDCs in response to ALVAC infection several encoded for chemokines such as CXCL10 (IP-10), CCL2 (MCP-1) and CCL8 (MCP-2). To determine if the strong up-regulation of CXCL10 transcription by ALVAC (+8.06-fold, Table 1) correlated to its expression at the protein level, the secretion of CXCL10 into the culture supernatants of ALVAC stimulated MDDCs was evaluated. Human MDDCs were incubated for 48 h with ALVAC at a MOI of 0.2, subsequently the supernatants were collected and the concentration of CXCL10 was measured by ELISA. In accordance with the enhanced gene transcription, MDDCs released high amounts of CXCL10 in response to ALVAC (Fig. 5 ). These results confirm our previous findings in the mouse model, in which high levels of CXCL10 secretion by bone marrow derived dendritic cells in response to ALVAC was observed [15]. In addition, this finding supports the hypothesis that ALVAC activates an IRF3-mediated signaling pathway, as the activated form of IRF3 directly targets CXCL10 gene transcription [31], [36].
Fig. 5.
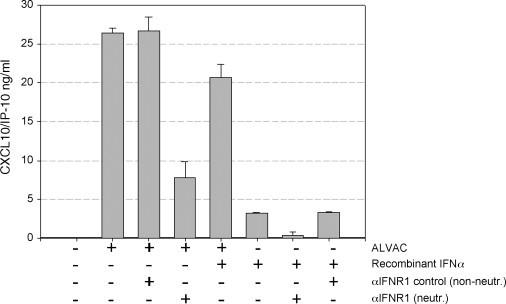
Secretion of CXCL10 by ALVAC infected human MDDCs. MDDCs were incubated for 48 h in the presence or absence of ALVAC, recombinant IFN-α, neutralizing IFN antibody or control antibody as indicated in the figure. Supernatants were collected and levels of CXCL10 were quantified by ELISA. Results are expressed as mean concentration ± standard error and are representative of three independent experiments. Infection of MDDCs with the viral vector ALVAC induced secretion of CXCL10 that was partially inhibited by anti-type I IFN receptor antibody. As expected the non-neutralizing anti-type I IFN receptor antibody used as negative control had no effect on CXCL10 release.
3.5. CXCL10 secretion is partially blocked by type I IFN receptor neutralizing antibody
Next, the role of type I IFNs on the responses elicited by MDDCs upon ALVAC infection, such as the CXCL10 release, was investigated. As shown in the Fig. 5 MDDCs infected with ALVAC in the presence of the anti-IFN type I receptor neutralizing antibody secreted significantly lower levels of CXCL10 than either MDDCs infected with ALVAC alone or ALVAC in the presence of the non-neutralizing anti-IFN type I receptor antibody. Interferon alpha induced release of CXCL10 by MDDCs at a lesser extent than ALVAC and this secretion was completely blocked by the neutralizing anti-IFN type I receptor antibody. These findings strongly argue in favor of the release of type I IFNs in this system which could not be detected in the ELISA experiments. Therefore, these results support a key role for type I IFN in the amplification of CXCL10 secreted by MDDCs in response to ALVAC infection, in accordance with the proposed mechanism of action (Fig. 3).
3.6. Genes encoding for ISG15 and its conjugating and deconjugating enzymes are up-regulated in ALVAC infected MDDCs
ISG15, which belongs to the ubiquitin-like protein modifier family together with its deconjugating (USP18/UBP43) and conjugating (Herc5/UBE1L) enzymes [37], [38] were found to be consistently highly up-regulated (ISG15, 10-fold on average) in ALVAC treated MDDCs (Table 1). Interferon via an IRF3 mediated pathway strongly activates the production of free ISG15 as well as protein ISGylation, which is the conjugation/deconjugation of ISG15 to target proteins via specific enzymes (UBP43 and UBE1L; whose expression is also regulated by type I IFN). Although, the functional consequences of ISGylation are not yet completely understood, there is increasing evidence to suggest a role in innate anti-viral responses [39], [40]. The free form of ISG15 has been described as an interferon-induced cytokine with immunoregulatory properties [27], [28]. Considering the potential role of ISG15 in the IFN mediated innate anti-viral response, we further analyzed the effect of ALVAC on this interferon responsive gene in MDDCs. We evaluated the induction of ISG15 in MDDCs following ALVAC infection at the protein level by Western blot analysis. Human MDDCs were cultured in the presence of 0.2 MOI ALVAC or human leukocyte interferon (IFN-α and IFN-ω), as positive control, for 48 h, then cells were harvested, lysed and subjected to SDS-PAGE and subsequently analyzed by Western blot. A band of 15 kDa, corresponding to the expected size of the free form of ISG15, was detected in the ALVAC as well as in the IFN treated MDDCs but not in the untreated MDDCs. Equal protein loading was verified by blotting the same membrane for the housekeeping gene GAPDH (Fig. 5). This data confirmed the induction of ISG15 in response to ALVAC at the protein level. Further analyses will establish if ISG15 is also released into the supernatant thereby influencing other cell types.
4. Discussion
In order to optimize the benefit of prophylactic or therapeutic immunization strategies, particularly those targeting cancer or viral infections, vaccines need to be able to induce and sustain cellular immune responses. ALVAC, a recombinant canarypox virus, is considered to be a good candidate vaccine vector with the potential to meet this need. ALVAC vaccine constructs can induce specific cellular and humoral immune responses against multiple pathogens and have a good safety profile [41]. Moreover, several canarypox virus recombinant vaccines are licensed for veterinary use [42]. ALVAC can be engineered to encode different foreign antigens and in contrast to vaccinia virus vectors, the problem of pre-existing immunity to the vector is circumvented. In addition to the antigen carrier function, it has also been reported that ALVAC itself exerts an adjuvant effect when administered with recombinant protein antigens in mice [14], [15]. This property is most likely linked to the stimulation of the innate immune system by ALVAC virus infection. Innate immunity is initiated by cells sensing foreign molecules known as “pathogen associated molecular patterns” (PAMP) through pattern-recognition receptors (PRR). Two classes of PRRs have been identified [31], [43], the trans-membrane PRRs, including the TLRs and some scavengers or lectin-like receptors and the cytosolic PRRs, including NOD and RIG-1 among others. Toll-like receptors sense bacterial or viral PAMPS and lead to secretion of pro-inflammatory cytokines via the NF-kB signaling pathway and for some TLRs (TLR3, TLR7/9) secretion of type I IFN via the IRF3/IRF7 signal transducers [31]. In the cytoplasm, the Nod-like receptors detect intracellular bacterial PAMPs, while RIG-1 and MDA-5 detect virus-derived RNA leading to activation of IFR3/IRF7 and NF-κB signaling cascades [43]. The existence of cytoplasmic dsDNA sensors has been long suspected since some DNA viruses, replicating in the cytoplasm, are able to induce type I interferon responses. Recently, Toneguchi and co-workers discovered the first cytosolic dsDNA sensor DAI (DLM-1/ZBP1), ligation of which leads to type I IFN gene induction through IRF3 and IRF7 and independently from TLR activation [30].
While, we have evidence that ALVAC triggers the innate immune response [14], [15], the specific pathways downstream of ALVAC infection have not yet been elucidated. Several recent papers have demonstrated that attenuated strains of vaccinia virus (MVA and NYVAC) trigger both the TLR-dependent and TLR-independent signaling pathways eliciting pro-inflammatory cytokines and type I IFNs [34], [44]. In this study, we analyzed the gene profile of human MDDC following ALVAC infection to determine the innate pathway preferentially induced by this virus vector. Dendritic cells were selected since they are permissive to ALVAC infection [32] and crucial in orchestrating immune responses [45].
The majority of genes up-regulated by ALVAC belong to the type I IFN pathway. Interaction between IFNs and their receptors (IFNR1 and IFNR2) promotes activation of the ISG3 signal transducer, a heterotrimer composed of STAT1, STAT2 and IRF9. Once ISG3 translocates to the nucleus, this complex activates the transcription of IRF7, MDA-5 and RIG-1 genes, all of which contribute to the positive feedback regulation of the IFN pathway. We found that the transcription of STAT1, IRF7, MDA-5 and RIG-1 was up-regulated in response to ALVAC. In addition, the expression of the interferon inducible and anti-viral genes OAS, PKR, Mx and ISG15 was enhanced following ALVAC infection of MDDCs. However, genes involved in the NF-κB signaling cascade were not found to be up-regulated in human MDDC upon ALVAC infection. In the light of these results, we propose that ALVAC, a dsDNA virus that resides in the cytoplasm of the cell, induces an interferon-mediated innate immune response in an IRF3/IRF7 dependent and TLR-independent way through recognition by a cytosolic dsDNA sensing PRR, such as the recently reported DAI molecule [30].
The up-regulation of CXCL10 and ISG15 transcription and protein expression, two molecules directly regulated by IRF3 (outlined in Fig. 3), following ALVAC infection of human MDDC is consistent with our hypothesis. Furthermore, the fact that ALVAC infected MDDCs secreted reduced levels of CXCL10 in the presence of neutralizing type I IFN-receptor antibody suggests a primary effect of ALVAC and a secondary effect of type I IFNs on MDDCs, thereby supporting the proposed model. CXCL10 (IP-10), is a chemotactic factor for T cells [46] and NK cells [47] expressing CXCR3 receptor. Recent studies with CXCL10 neutralizing antibodies or CXCL10 deficient mice demonstrated that the absence of functional CXCL10 resulted in increased mortality and reduced T cell infiltration in the brains of mice intracerebrally infected with a murine coronavirus, illustrating the importance of this chemokine in co-coordinating a protective immune response against a viral pathogen [48], [49], [50]. In addition, CXCL10 release might attract NK cells to DC rich areas bringing them in close contact and thereby triggering a downstream TH1 response by secreting IFNγ [51]. Here, we have demonstrated that human MDDCs secrete high levels of CXCL10 following incubation with ALVAC, thus confirming our microarray results, as well as data obtained in vivo in mice following ALVAC injections [15].
ISG15, is an ubiquitin-like protein modifier whose gene expression is induced by several stimuli, besides IRF3, type I interferon (ISG15 is one of the earliest ISGs induced), LPS, retinoic acid, campothecin and radiation [52]. The conjugation of ISG15 to intracellular protein targets is a reversible process controlled by a conjugating enzyme and a deconjugating enzyme, UBE1L and UBP43, respectively. Both enzymes are induced by type I interferon. Currently about 200 ISGylated proteins have been identified, among them IFN-α/β-induced anti-viral proteins, such as PKR, MxA, HuP56, and RIG-1 [40], [53]. The effect of ISGylation on protein function, distribution or stability is not yet fully understood. However, accumulating evidence, including the ability of many viruses to either inhibit the production of free ISG15 or the formation of ISG15 conjugates, indicates an anti-viral role for this protein [26], [26], [40], [53]. The free form of ISG15 has been described as an interferon-induced cytokine with immunoregulatory properties [27], [28]. Human lymphocytes and monocytes release ISG15 into culture medium following in vitro stimulation with IFN-β and free ISG15 has been detected in the serum of IFN-β-treated patients [28]. Furthermore, it has been shown that recombinant ISG15 stimulates IFN-γ production by CD3+ lymphocytes and increase proliferation of CD56+ NK cells [27]. We show here, for the first time, that ALVAC infection of human MDDCs induced gene expression of ISG15 and both its conjugating and deconjugating enzymes. Furthermore, we demonstrate that the protein expression of ISG15 is enhanced in ALVAC infected MDCCs by Western blot analysis (Fig. 6 ).
Fig. 6.
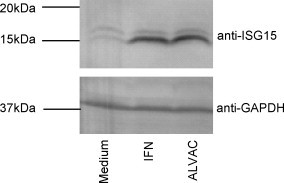
Protein expression of ISG15 in MDDCs upon ALVAC exposure. MDDCs were stimulated with 0.2 MOI ALVAC or 1000 U/ml human leukocyte interferon (IFN-α and IFN-ω) for 48 h, harvested, lysed and subjected to SDS-PAGE. Expression of ISG15 was analyzed using anti-ISG antibody. Equal loading was confirmed by re-probing blots with an antibody against GAPDH. One representative result of three experiments is shown.
While, our microarray data failed to show the up-regulation of IFN-α, we detected enhanced IFN-β expression in two of four donors. Recent data suggest that the expression of the different IFN-α subtypes (13 in human) is cell-type and stimulus specific [28]. The fact that not all IFN-α subtypes were represented on the microarray chip may explain the lack of IFN-α gene up-regulation observed in the microarray data. However, the secretion of type-I IFN by ALVAC infected DCs was indirectly demonstrated by the reduced capacity of these cells to secrete CXCL10 in the presence of a type I IFN-receptor neutralizing antibody (Fig. 5). Surprisingly, MyD88 gene transcription was also up-regulated by ALVAC in all four donors. The adaptor molecule MyD88 is essential for all TLR signaling leading to the production of pro-inflammatory cytokines (except TLR3) and is also required for TLR 7 and 9 signaling resulting in type I interferon secretion [43], [54]. Since we found no evidence for enhanced pro-inflammatory cytokine gene transcription following ALVAC infection upon analysis of our microarray results and type I interferon MyD88-mediated secretion via TLR7/9 does not involve IRF3 activation, the MyD88 induction might be attributed to other functions rather than the adaptor one [54]. This study confirms that ALVAC infection induces the maturation of MDDCs [15], [17], [55]. However, we made the additional observation that as opposed to LPS stimulation, ALVAC induced lower levels of co-stimulatory molecules and failed to up-regulate CD25 which has been demonstrated to be essential for DC mediated T cell activation and IL-2 secretion [33]. These results suggest that ALVAC does not induce full DC maturation. The consequences of this finding on the activation of the specific T cell response against ALVAC encoded antigens were not investigated in the present study. This is of great interest and will be subject of further research.
Overall our results highlight the ability of the recombinant canarypox viral vector ALVAC to induce a type I interferon-mediated innate anti-viral immune response in human MDDCs possibly through the cytosolic pattern-recognition system. Recently, Nociari et al. and Waibler et al. reported similar results with two other dsDNA vectors, Adenovirus vector [56] and modified vaccinia virus Ankara vector MVA [57], respectively. The stimulation of the innate immune response is essential to develop an effective acquired immune response against the foreign encoded antigens. Our findings offer new insight in the design of more effective ALVAC-vectored vaccines.
Acknowledgements
A.H. was funded by a Marie-Curie Industry Host Fellowship from the European Union. We thank Fred Vogel for critical reading of the manuscript and scientific discussions.
Footnotes
Supplementary data associated with this article can be found, in the online version, at doi:10.1016/j.vaccine.2008.07.050.
Contributor Information
Anke Harenberg, Email: anke.harenberg2@sanofipasteur.com.
Franca Spada, Email: Franca.Spada@sanofipasteur.com.
Appendix. Supplementary data
References
- 1.Marovich M.A. ALVAC-HIV vaccines: clinical trial experience focusing on progress in vaccine development. Expert Rev Vaccines. 2004;3(4 Suppl.):S99–S104. doi: 10.1586/14760584.3.4.s99. [DOI] [PubMed] [Google Scholar]
- 2.de Bruyn G., Rossini A.J., Chiu Y.L., Holman D., Elizaga M.L., Frey S.E. Safety profile of recombinant canarypox HIV vaccines. Vaccine. 2004;22(5–6):704–713. doi: 10.1016/j.vaccine.2003.08.023. [DOI] [PubMed] [Google Scholar]
- 3.Van der Burg S.H., Menon A.G., Redeker A., Bonnet M.C., Drijfhout J.W., Tollenaar R.A. Induction of p53-specific immune responses in colorectal cancer patients receiving a recombinant ALVAC-p53 candidate vaccine. Clin Cancer Res. 2002;8(5):1019–1027. [PubMed] [Google Scholar]
- 4.van Baren N., Bonnet M.C., Dreno B., Khammari A., Dorval T., Piperno-Neumann S. Tumoral and immunologic response after vaccination of melanoma patients with an ALVAC virus encoding MAGE antigens recognized by T cells. J Clin Oncol. 2005;23(35):9008–9021. doi: 10.1200/JCO.2005.08.375. [DOI] [PubMed] [Google Scholar]
- 5.Tartaglia J., Bonnet M.C., Berinstein N., Barber B., Klein M., Moingeon P. Therapeutic vaccines against melanoma and colorectal cancer. Vaccine. 2001;19(17–19):2571–2575. doi: 10.1016/s0264-410x(00)00491-6. [DOI] [PubMed] [Google Scholar]
- 6.Belshe R.B., Gorse G.J., Mulligan M.J., Evans T.G., Keefer M.C., Excler J.L. Induction of immune responses to HIV-1 by canarypox virus (ALVAC) HIV-1 and gp120 SF-2 recombinant vaccines in uninfected volunteers. NIAID AIDS Vaccine Evaluation Group. AIDS. 1998;12(18):2407–2415. doi: 10.1097/00002030-199818000-00009. [DOI] [PubMed] [Google Scholar]
- 7.Clements-Mann M.L., Weinhold K., Matthews T.J., Graham B.S., Gorse G.J., Keefer M.C. Immune responses to human immunodeficiency virus (HIV) type 1 induced by canarypox expressing HIV-1MN gp120, HIV-1SF2 recombinant gp120, or both vaccines in seronegative adults. NIAID AIDS Vaccine Evaluation Group. J Infect Dis. 1998;177(5):1230–1246. doi: 10.1086/515288. [DOI] [PubMed] [Google Scholar]
- 8.Fries L.F., Tartaglia J., Taylor J., Kauffman E.K., Meignier B., Paoletti E. Human safety and immunogenicity of a canarypox-rabies glycoprotein recombinant vaccine: an alternative poxvirus vector system. Vaccine. 1996;14(5):428–434. doi: 10.1016/0264-410x(95)00171-v. [DOI] [PubMed] [Google Scholar]
- 9.Salmon-Ceron D., Excler J.L., Sicard D., Blanche P., Finkielstzjen L., Gluckman J.C. Safety and immunogenicity of a recombinant HIV type 1 glycoprotein 160 boosted by a V3 synthetic peptide in HIV-negative volunteers. AIDS Res Hum Retroviruses. 1995;11(12):1479–1486. doi: 10.1089/aid.1995.11.1479. [DOI] [PubMed] [Google Scholar]
- 10.Gupta K., Hudgens M., Corey L., McElrath M.J., Weinhold K., Montefiori D.C. Safety and immunogenicity of a high-titered canarypox vaccine in combination with rgp120 in a diverse population of HIV-1-uninfected adults: AIDS Vaccine Evaluation Group Protocol 022A. J Acquir Immune Defic Syndr. 2002;29(3):254–261. doi: 10.1097/00126334-200203010-00005. [DOI] [PubMed] [Google Scholar]
- 11.Egan M.A., Pavlat W.A., Tartaglia J., Paoletti E., Weinhold K.J., Clements M.L. Induction of human immunodeficiency virus type 1 (HIV-1)-specific cytolytic T lymphocyte responses in seronegative adults by a nonreplicating, host-range-restricted canarypox vector (ALVAC) carrying the HIV-1MN env gene. J Infect Dis. 1995;171(6):1623–1627. doi: 10.1093/infdis/171.6.1623. [DOI] [PubMed] [Google Scholar]
- 12.Konishi E., Kurane I., Mason P.W., Shope R.E., Kanesa-thasan N., Smucny J.J. Induction of Japanese encephalitis virus-specific cytotoxic T lymphocytes in humans by poxvirus-based JE vaccine candidates. Vaccine. 1998;16(8):842–849. doi: 10.1016/s0264-410x(97)00265-x. [DOI] [PubMed] [Google Scholar]
- 13.Fischer L., Le Gros F.X., Mason P.W., Paoletti E. A recombinant canarypox virus protects rabbits against a lethal rabbit hemorrhagic disease virus (RHDV) challenge. Vaccine. 1997;15(1):90–96. doi: 10.1016/s0264-410x(96)00102-8. [DOI] [PubMed] [Google Scholar]
- 14.Boudet F., Chevalier M., Jourdier T.M., Tartaglia J., Moste C. Modulation of the antibody response to the HIV envelope subunit by co-administration of infectious or heat-inactivated canarypoxvirus (ALVAC) preparations. Vaccine. 2001;19(30):4267–4275. doi: 10.1016/s0264-410x(01)00150-5. [DOI] [PubMed] [Google Scholar]
- 15.Ryan E.J., Harenberg A., Burdin N. The Canarypox-virus vaccine vector ALVAC triggers the release of IFN-gamma by Natural Killer (NK) cells enhancing Th1 polarization. Vaccine. 2007;25(17):3380–3390. doi: 10.1016/j.vaccine.2006.12.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Taylor J., Meignier B., Tartaglia J., Languet B., VanderHoeven J., Franchini G. Biological and immunogenic properties of a canarypox-rabies recombinant, ALVAC-RG (vCP65) in non-avian species. Vaccine. 1995;13(6):539–549. doi: 10.1016/0264-410x(94)00028-l. [DOI] [PubMed] [Google Scholar]
- 17.Ignatius R., Marovich M., Mehlhop E., Villamide L., Mahnke K., Cox W.I. Canarypox virus-induced maturation of dendritic cells is mediated by apoptotic cell death and tumor necrosis factor alpha secretion. J Virol. 2000;74(23):11329–11338. doi: 10.1128/jvi.74.23.11329-11338.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Motta I., Andre F., Lim A., Tartaglia J., Cox W.I., Zitvogel L. Cross-presentation by dendritic cells of tumor antigen expressed in apoptotic recombinant canarypox virus-infected dendritic cells. J Immunol. 2001;167(3):1795–1802. doi: 10.4049/jimmunol.167.3.1795. [DOI] [PubMed] [Google Scholar]
- 19.Engelmayer J., Larsson M., Lee A., Lee M., Cox W.I., Steinman R.M. Mature dendritic cells infected with canarypox virus elicit strong anti-human immunodeficiency virus CD8+ and CD4+ T-cell responses from chronically infected individuals. J Virol. 2001;75(5):2142–2153. doi: 10.1128/JVI.75.5.2142-2153.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Berencsi K., Gyulai Z., Gonczol E., Pincus S., Cox W.I., Michelson S. A canarypox vector-expressing cytomegalovirus (CMV) phosphoprotein 65 induces long-lasting cytotoxic T cell responses in human CMV-seronegative subjects. J Infect Dis. 2001;183(8):1171–1179. doi: 10.1086/319680. [DOI] [PubMed] [Google Scholar]
- 21.Klein M. Current progress in the development of human immunodeficiency virus vaccines: research and clinical trials. Vaccine. 2001;19(17–19):2210–2215. doi: 10.1016/s0264-410x(00)00547-8. [DOI] [PubMed] [Google Scholar]
- 22.Dolin R. HIV vaccines for prevention of infection and disease in humans. Infect Dis Clin North Am. 2000;14(4):1001–1016. doi: 10.1016/s0891-5520(05)70144-6. [DOI] [PubMed] [Google Scholar]
- 23.Goepfert P.A., Horton H., McElrath M.J., Gurunathan S., Ferrari G., Tomaras G.D. High-dose recombinant Canarypox vaccine expressing HIV-1 protein, in seronegative human subjects. J Infect Dis. 2005;192(7):1249–1259. doi: 10.1086/432915. [DOI] [PubMed] [Google Scholar]
- 24.Bos R., van Duikeren S., van Hall T., Lauwen M.M., Parrington M., Berinstein N.L. Characterization of antigen-specific immune responses induced by canarypox virus vaccines. J Immunol. 2007;179(9):6115–6122. doi: 10.4049/jimmunol.179.9.6115. [DOI] [PubMed] [Google Scholar]
- 25.Lenschow D.J., Giannakopoulos N.V., Gunn L.J., Johnston C., O’Guin A.K., Schmidt R.E. Identification of interferon-stimulated gene 15 as an antiviral molecule during Sindbis virus infection in vivo. J Virol. 2005;79(22):13974–13983. doi: 10.1128/JVI.79.22.13974-13983.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lenschow D.J., Lai C., Frias-Staheli N., Giannakopoulos N.V., Lutz A., Wolff T. IFN-stimulated gene 15 functions as a critical antiviral molecule against influenza, herpes, and Sindbis viruses. Proc Natl Acad Sci USA. 2007;104(4):1371–1376. doi: 10.1073/pnas.0607038104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.D’Cunha J., Ramanujam S., Wagner R.J., Witt P.L., Knight E., Jr., Borden E.C. In vitro and in vivo secretion of human ISG15, an IFN-induced immunomodulatory cytokine. J Immunol. 1996;157(9):4100–4108. [PubMed] [Google Scholar]
- 28.D’Cunha J., Knight E., Jr., Haas A.L., Truitt R.L., Borden E.C. Immunoregulatory properties of ISG15, an interferon-induced cytokine. Proc Natl Acad Sci USA. 1996;93(1):211–215. doi: 10.1073/pnas.93.1.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Knight E., Jr., Cordova B. IFN-induced 15-kDa protein is released from human lymphocytes and monocytes. J Immunol. 1991;146(7):2280–2284. [PubMed] [Google Scholar]
- 30.Takaoka A., Wang Z., Choi M.K., Yanai H., Negishi H., Ban T. DAI (DLM-1/ZBP1) is a cytosolic DNA sensor and an activator of innate immune response. Nature. 2007;448(7152):501–505. doi: 10.1038/nature06013. [DOI] [PubMed] [Google Scholar]
- 31.Honda K., Taniguchi T. IRFs: master regulators of signalling by Toll-like receptors and cytosolic pattern-recognition receptors. Nat Rev Immunol. 2006;6(9):644–658. doi: 10.1038/nri1900. [DOI] [PubMed] [Google Scholar]
- 32.Yu Q., Jones B., Hu N., Chang H., Ahmad S., Liu J. Comparative analysis of tropism between canarypox (ALVAC) and vaccinia viruses reveals a more restricted and preferential tropism of ALVAC for human cells of the monocytic lineage. Vaccine. 2006;24(40–41):6376–6391. doi: 10.1016/j.vaccine.2006.06.011. [DOI] [PubMed] [Google Scholar]
- 33.Velten F.W., Rambow F., Metharom P., Goerdt S. Enhanced T-cell activation and T-cell-dependent IL-2 production by CD83+, CD25high, CD43high human monocyte-derived dendritic cells. Mol Immunol. 2007;44(7):1544–1550. doi: 10.1016/j.molimm.2006.08.020. [DOI] [PubMed] [Google Scholar]
- 34.Guerra S., Najera J.L., Gonzalez J.M., Lopez-Fernandez L.A., Climent N., Gatell J.M. Distinct gene expression profiling after infection of immature human monocyte-derived dendritic cells by the attenuated poxvirus vectors MVA and NYVAC. J Virol. 2007;81(16):8707–8721. doi: 10.1128/JVI.00444-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Asselin-Paturel C., Trinchieri G. Production of type I interferons: plasmacytoid dendritic cells and beyond. J Exp Med. 2005;202(4):461–465. doi: 10.1084/jem.20051395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Nakaya T., Sato M., Hata N., Asagiri M., Suemori H., Noguchi S. Gene induction pathways mediated by distinct IRFs during viral infection. Biochem Biophys Res Commun. 2001;283(5):1150–1156. doi: 10.1006/bbrc.2001.4913. [DOI] [PubMed] [Google Scholar]
- 37.Wong J.J., Pung Y.F., Sze N.S., Chin K.C. HERC5 is an IFN-induced HECT-type E3 protein ligase that mediates type I IFN-induced ISGylation of protein targets. Proc Natl Acad Sci USA. 2006;103(28):10735–10740. doi: 10.1073/pnas.0600397103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Dastur A., Beaudenon S., Kelley M., Krug R.M., Huibregtse J.M. Herc5, an interferon-induced HECT E3 enzyme, is required for conjugation of ISG15 in human cells. J Biol Chem. 2006;281(7):4334–4338. doi: 10.1074/jbc.M512830200. [DOI] [PubMed] [Google Scholar]
- 39.Yuan W., Krug R.M. Influenza B virus NS1 protein inhibits conjugation of the interferon (IFN)-induced ubiquitin-like ISG15 protein. EMBO J. 2001;20(3):362–371. doi: 10.1093/emboj/20.3.362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zhao C., Denison C., Huibregtse J.M., Gygi S., Krug R.M. Human ISG15 conjugation targets both IFN-induced and constitutively expressed proteins functioning in diverse cellular pathways. Proc Natl Acad Sci USA. 2005;102(29):10200–10205. doi: 10.1073/pnas.0504754102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Hutchings C.L., Gilbert S.C., Hill A.V., Moore A.C. Novel protein and poxvirus-based vaccine combinations for simultaneous induction of humoral and cell-mediated immunity. J Immunol. 2005;175(1):599–606. doi: 10.4049/jimmunol.175.1.599. [DOI] [PubMed] [Google Scholar]
- 42.Poulet H., Minke J., Pardo M.C., Juillard V., Nordgren B., Audonnet J.C. Development and registration of recombinant veterinary vaccines. The example of the canarypox vector platform. Vaccine. 2007;25(30):5606–5612. doi: 10.1016/j.vaccine.2006.11.066. [DOI] [PubMed] [Google Scholar]
- 43.Akira S., Uematsu S., Takeuchi O. Pathogen recognition and innate immunity. Cell. 2006;124(4):783–801. doi: 10.1016/j.cell.2006.02.015. [DOI] [PubMed] [Google Scholar]
- 44.Zhu J., Martinez J., Huang X., Yang Y. Innate immunity against vaccinia virus is mediated by TLR2 and requires TLR-independent production of IFN-beta. Blood. 2007;109(2):619–625. doi: 10.1182/blood-2006-06-027136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Banchereau J., Steinman R.M. Dendritic cells and the control of immunity. Nature. 1998;392(6673):245–252. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- 46.Liu M.T., Keirstead H.S., Lane T.E. Neutralization of the chemokine CXCL10 reduces inflammatory cell invasion and demyelination and improves neurological function in a viral model of multiple sclerosis. J Immunol. 2001;167(7):4091–4097. doi: 10.4049/jimmunol.167.7.4091. [DOI] [PubMed] [Google Scholar]
- 47.Trifilo M.J., Montalto-Morrison C., Stiles L.N., Hurst K.R., Hardison J.L., Manning J.E. CXC chemokine ligand 10 controls viral infection in the central nervous system: evidence for a role in innate immune response through recruitment and activation of natural killer cells. J Virol. 2004;78(2):585–594. doi: 10.1128/JVI.78.2.585-594.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Walsh K.B., Edwards R.A., Romero K.M., Kotlajich M.V., Stohlman S.A., Lane T.E. Expression of CXC chemokine ligand 10 from the mouse hepatitis virus genome results in protection from viral-induced neurological and liver disease. J Immunol. 2007;179(2):1155–1165. doi: 10.4049/jimmunol.179.2.1155. [DOI] [PubMed] [Google Scholar]
- 49.Liu M.T., Chen B.P., Oertel P., Buchmeier M.J., Armstrong D., Hamilton T.A. The T cell chemoattractant IFN-inducible protein 10 is essential in host defense against viral-induced neurologic disease. J Immunol. 2000;165(5):2327–2330. doi: 10.4049/jimmunol.165.5.2327. [DOI] [PubMed] [Google Scholar]
- 50.Dufour J.H., Dziejman M., Liu M.T., Leung J.H., Lane T.E., Luster A.D. IFN-gamma-inducible protein 10 (IP-10; CXCL10)-deficient mice reveal a role for IP-10 in effector T cell generation and trafficking. J Immunol. 2002;168(7):3195–3204. doi: 10.4049/jimmunol.168.7.3195. [DOI] [PubMed] [Google Scholar]
- 51.Megjugorac N.J., Young H.A., Amrute S.B., Olshalsky S.L., Fitzgerald-Bocarsly P. Virally stimulated plasmacytoid dendritic cells produce chemokines and induce migration of T and NK cells. J Leukoc Biol. 2004;75(3):504–514. doi: 10.1189/jlb.0603291. [DOI] [PubMed] [Google Scholar]
- 52.Pitha-Rowe I.F., Pitha P.M. Viral defense, carcinogenesis and ISG15: novel roles for an old ISG. Cytokine Growth Factor Rev. 2007;18(5–6):409–417. doi: 10.1016/j.cytogfr.2007.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Giannakopoulos N.V., Luo J.K., Papov V., Zou W., Lenschow D.J., Jacobs B.S. Proteomic identification of proteins conjugated to ISG15 in mouse and human cells. Biochem Biophys Res Commun. 2005;336(2):496–506. doi: 10.1016/j.bbrc.2005.08.132. [DOI] [PubMed] [Google Scholar]
- 54.O’Neill L.A., Bowie A.G. The family of five: TIR-domain-containing adaptors in Toll-like receptor signalling. Nat Rev Immunol. 2007;7(5):353–364. doi: 10.1038/nri2079. [DOI] [PubMed] [Google Scholar]
- 55.Marovich M.A., Mascola J.R., Eller M.A., Louder M.K., Caudrelier P.A., El Habib R. Preparation of clinical-grade recombinant canarypox-human immunodeficiency virus vaccine-loaded human dendritic cells. J Infect Dis. 2002;186(9):1242–1252. doi: 10.1086/344302. [DOI] [PubMed] [Google Scholar]
- 56.Nociari M., Ocheretina O., Schoggins J.W., Falck-Pedersen E. Sensing infection by adenovirus: toll-like receptor-independent viral DNA recognition signals activation of the interferon regulatory factor 3 master regulator. J Virol. 2007;81(8):4145–4157. doi: 10.1128/JVI.02685-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Waibler Z., Anzaghe M., Ludwig H., Akira S., Weiss S., Sutter G. Modified vaccinia virus Ankara induces toll-like receptor-independent type I interferon responses. J Virol. 2007;81(22):12102–12110. doi: 10.1128/JVI.01190-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


